Abstract
Purpose
Bone is one of the main targets of hormones and endocrine diseases are frequent causes of secondary osteoporosis and fractures in real-world clinical practice. However, diagnosis of skeletal fragility and prediction of fractures in this setting could be a challenge, since the skeletal alterations induced by endocrine disorders are not generally captured by dual-energy X-ray absorptiometry (DXA) measurement of bone mineral density (BMD), that is the gold standard for diagnosis of osteoporosis in the general population. The aim of this paper is to review the existing evidence related to bone quality features in endocrine diseases, proposing assessment with new techniques in the future.
Methods
A comprehensive search within electronic databases was performed to collect reports of bone quality in primary hyperparathyroidism, hypoparathyroidism, hyperthyroidism, hypercortisolism, growth hormone deficiency, acromegaly, male hypogonadism and diabetes mellitus.
Results
Using invasive and non-invasive techniques, such as high-resolution peripheral quantitative computed tomography or DXA measurement of trabecular bone score (TBS), several studies consistently reported altered bone quality as predominant determinant of fragility fractures in subjects affected by chronic endocrine disorders.
Conclusions
Assessment of skeletal fragility in endocrine diseases might take advantage from the use of techniques to detect perturbation in bone architecture with the aim of best identifying patients at high risk of fractures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The silent pandemic of osteoporosis and its logical consequence, i.e. fragility fractures, are globally spreading, constituting a challenge for the majority of health systems [1]. Assessment and treatment of osteoporosis is mandatory in both sexes, in order to decrease the risk of fracture and the burden of osteoporotic fractures worldwide [2]. Up to 40% of osteoporotic postmenopausal women and 60% of osteoporotic men can harbor a secondary cause of bone loss, including rheumatological, gastrointestinal, hematological, neurological and endocrine diseases [3]. Thus, in the diagnostic work-up of a patient with osteoporosis, secondary causes of osteoporosis, most of all of endocrine nature, must always be ruled out [4]. Conversely, bone assessment and evaluation of fracture risk is mandatory for a comprehensive evaluation of patients with most endocrinopathies before and after treatment. Indeed, bone is a target organ of major hormones and bone remodeling is deeply influenced by pathological changes in their levels. The evaluation of bone mineral density (BMD) by means of dual-energy X-ray absorptiometry (DXA) with its incorporation into prediction algorithms, constitutes a mainstay in the evaluation of the risk of fragility fractures and has provided the practical definition of osteoporosis, as based on the T-score less than -2.5 standard deviation, originally established for postmenopausal women [5]. Nonetheless, the majority of patients with fragility fractures show T-scores above -2.5 standard deviation, especially in the setting of secondary osteoporosis and, in particular, endocrine diseases. Several reports in the last years have shown that endocrinopathies are associated with altered bone strength leading to increased fracture risk, at least in part independent of bone quantity as derived from DXA parameters, but mainly relying on hampering of bone quality as directly or indirectly assessed by different techniques [3].
To our knowledge, so far, no study has focused on available data on bone quality in major endocrinopathies affecting skeletal tissue in adults. The aim of this paper is to review the existing evidence related to bone quality features in diseases characterized by hyperfunction or hypofunction of major endocrine glands, namely parathyroid and thyroid disorders, hypercortisolism, growth hormone disorders, male hypogonadism and diabetes, starting from the definition and assessment of bone quality in clinical practice. For this purpose, a PubMed search up to December 2022 has been performed using the terms: “bone quality/qualities”, “trabecular bone score/TBS”, “bone strength index/BSI”, “central quantitative computed tomography/cQCT”, “peripheral quantitative computed tomography/pQCT”, “high resolution peripheral QCT/HR-pQCT”, “radiofrequency echographic multispectrometry/REMS”, “magnetic resonance imaging/MRI”, “histomorphometry”, “microindentation”, “fractures”, “meta-analysis” and major endocrinopathies (“hyperparathyroidism”, “hypoparathyroidism”, “hyperthyroidism”, “hypothyroidism”, “GH eccess/acromegaly”, “GH deficiency”, “hypercortisolism/Cushing syndrome”, “hypogonadism”, “diabetes”), with a main focus on in vivo results.
Bone quality or bone qualities: definition and clinical significance
In major endocrinopathies, bone is a target organ for altered bone quantity and quality, both leading to a decrease in its strength, as demonstrated by the increased rate of prevalent vertebral fractures, as assessed by vertebral morphometry, and previous/incident clinical fractures, regardless of BMD values [4, 6]. Indeed, although BMD as determined by DXA is one of the major determinants of fracture risk, reducing a 3-D structure to a 2-dimensional analysis poses great limit for the assessment of the complexity of bone structure.
Total bone mass, bone geometry and design, and material composition contributes to structural integrity of bones, shaped and constantly re-cycled by modeling and remodeling processes, respectively [7, 8].
In the last two decades, the concept of bone quality and its assessment has acquired a major role in the understanding and proper estimating bone fragility and explain the reduction of fracture risk as obtained by anti-osteoporotic treatments. Bone quality is a complex term and refers to the multifaceted properties of bone tissues, first of all microarchitecture, as determinants of bone competence to resist to fracture [9].
Bone quality, as distinct from bone quantity, stands for the intrinsic biomechanical characteristics, conferring strength and stiffness to the skeletal tissue, which are not accounted for bone mass (quantity) determination by DXA, which provide areal bone mineral density. Indeed, the knowledge of bone quality ensues from integrated competencies of a number of disciplines such as clinicians, biologists, physicists, and engineers and refers to the essential or distinctive characteristics, properties, or attributes of bone [10]. These features, whose integrity is needed for bone resistance, reflect the hierarchical macro-to-nanoscale structure of the bone tissue, which can be mainly assessed in research settings [11] (Fig. 1).
Bone quality measurement techniques depicted on a logarithmic scale of the hierarchical structure of bone. Each characterization technique is categorized as geometric, compositional, or mechanical and is depicted by a bar showing the approximate range of resolutions currently achievable. AFM atomic force microscopy; FTIR Fourier transform infrared; HPLC high-performance liquid chromatography; HR-MRI high resolution magnetic resonance imaging; HR-pQCT high-resolution peripheral quantitative computed tomography; micro-CT micro-computed tomography; NMR nuclear magnetic resonance imaging; qBEI quantitative back scattered electronic imaging; QCT quantitative computed tomography; RPI reference point indentation; SAXS small-angle scattering; SEM scanning electron microscopy; TGA thermogravimetric analysis; XRD x-ray diffraction (reproduced with permission from Ref. [11])
The balance between toughness and stiffness must be preserved in the skeleton to maintain structural strength. Collagen fibrils organized in triple helixes appropriately cross-linked together, confer strength in tightness, nevertheless allowing flexibility. The addition of hydroxyapatite crystals to the matrix confers stiffness. Nonetheless, since human skeletal tissue is about 60% mineralized, flexibility and lightness are preserved to deform to bending or compression forces without breaking and to allow movement, respectively. The complexity of the architecture of the skeletal tissue at a microscopic level, i.e., the number of osteons in cortical bone, contributes to limit the propagation of microcracks in the bone, allowing remodeling process to resorb and substitute the damaged tissue with new bone [12].
At a macroscopic level, trabecular and cortical bone are shaped to display a structure specifically suitable to the purpose of sustain, absorb forces and resist to fracture. In trabecular bone, the connectivity of the trabeculae as determined by their thickness and number ensure lightness and flexibility. Diameter and thickness of cortical bone, shaped by periosteal apposition and endosteal resorption, especially during modeling, contribute to the resistance of long bone to bending. Adaptation in size and shape during modeling occur in response to load [12].
Negative balance in the bone multicellular unit between bone formation and resorption observed during aging or diseases, somehow combined with a higher remodeling rate lead to bone loss, deterioration of bone qualities and increased fracture risk. In cancellous bone this is reflected by a decrease in the number and thickness, and an increase in trabecular perforation, all leading to loss of connectivity of trabecular plates. In cortical bone, the increased intracortical and endocortical remodeling causes the trabecularization of the cortex. Moreover, a decreased periosteal apposition contributes to the thinning of the bone cortex together with the increased endocortical reabsorption [9, 13].
These changes in the macroscopic structure of the bone are nowadays recognized as some of the main culprits of bone fragility during the aging process or diseases affecting bone and can even precede or occur in the absence of modifications of areal bone mineral density as identified by DXA [14]. Moreover, all the listed properties, which can be also affected during endocrine malfunction, cannot be merely reconducted just to the 2-dimensional DXA report, so that other tools have been developed to assess these features and provide better estimate of bone fragility.
Assessment of bone quality in clinical practice and research
Despite BMD derived by DXA is not able to fully evaluate the above-mentioned bone qualitative aspects that predispose to fragility fractures, this technique is still considered the standard of reference in clinical practice for bone densitometry. This is because of its several strengths over more advanced techniques, such as the extremely low radiation dose to patients, the evaluation of central skeletal sites, its low costs and wide availability [15]. Therefore, many DXA-based tools have been developed during time to explore skeletal features of bone quality, to refine fracture risk prediction.
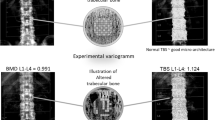
In the early 2010s, the Trabecular Bone Score (TBS) has been developed as a DXA-based software applied to lumbar spine images, and represents one of the most successful and well-known experiments in this field. The software can discriminate gray-level variations in the DXA image pixels; despite TBS does not provide a direct measurement of trabecular microarchitecture (due to technical constraints related to the pixel width of DXA image, which is about four time larger the mean size of trabeculae), ex-vivo and in-vivo studies showed how TBS can predict fracture risk independently of BMD [16]. It is now recognized that TBS provides indirect information about trabecular microarchitecture by means of a textural analysis: higher TBS values (> 1.310) are associated with normal microarchitecture, while lower TBS values (< 1.230) are associated with degraded microarchitecture [17] (Fig. 2). TBS report can be easily obtained on the same DXA region of interest. Other advantages of TBS are that, compared to BMD, is less affected by degenerative spinal changes, but also that it can be incorporated into the FRAX® to adjust a 10-year fracture probability [18, 19]. Adjustment of BMD T-score according to TBS values was recently proposed as a new approach for using TBS in clinical practice and enhancing fracture risk prediction independently of BMD [17]. TBS adjustment to the BMD T-score was based upon the change in TBS that would give the same predicted risk as a unit change in the BMD T-score. The Cox proportional hazards model used for TBS adjustment of BMD T-score paralleled the approach used for developing the TBS-adjustment to FRAX [19]. Interestingly, high agreement was found between probabilities of major osteoporotic fractures estimated by TBS-adjusted FRAX and those estimated by TBS-adjusted BMD [17].
Dual-energy X-ray absorptiometry (DXA) measurement of bone mineral density (BMD) and trabecular bone score (TBS) in a 49 years old post-menopausal woman with Cushing syndrome. This is a paradigmatic clinical case in which TBS was degraded notwithstanding BMD values were in the range of osteopenia. Noteworthy, the BMD T-score values moved in the range of osteoporosis after adjustment for TBS, according to the mathematical model proposed by Leslie et al. [17], which parallels the approach used for developing the TBS-adjustment to FRAX [19]
A more recent DXA-based software has been introduced in 2019 with the name of Bone Strain Index (BSI) and relies on the application of finite element analysis (FEA) mathematical model to DXA images [20]. The principle beyond FEA is that the analysis of a complex object can be made simple by dividing it into a model of many smaller elements (the so-called “finite elements”), and then simulate on this model any specific conditions. BSI analysis can be done both at lumbar spine and proximal femur, with the aim of identifying those regions of the bone at higher mechanical stress, hence easier to fracture [20]. BSI software divides DXA area of analysis into small triangles, to generate a specific FEA mathematical model both for lumbar spine and proximal femur. At the spine, the load is applied to the upper vertebral plate simulating the gravity, while at the femur the load is applied to the greater trochanter simulating a lateral fall. The supposed bone load is obtained from patient-specific simulated forces, as well as patient’s body mass index. Higher BSI values indicate higher strain and fracture risk, while lower BSI values indicate less strain to the bone and lower fracture risk. Proposed BSI threshold values for the Italian population indicate normal resistance to strain for BSI ≤ 1.7, and poor resistance for BSI ≥ 2.5 [21].
As for DXA, computed tomography (CT) based techniques relies on X-rays, but the latter offer a cross-sectional visualization of bony structures. Therefore, with CT is possible to get measurements of trabecular and cortical bone separately. Quantitative CT (QCT) and high-resolution peripheral quantitative CT (HR‑pQCT) are the two most used CT techniques for bone evaluation. They differ, among the others, for spatial resolution, and this is reflected in the type and size of bone they can measure. QCT, with a namely resolution of 250–300 μm, is typically applied to lumbar spine and proximal femur using whole body scanners [15, 22]. To obtain volumetric BMD (vBMD) values, a calibration of CT Hounsfield Unit (HU) is needed, and this is done with the use of phantoms containing a set of known density values of hydroxyapatite samples. In the case of synchronous calibration, such phantoms are placed under the patients inside the CT table. For asynchronous calibration, the phantom is scanned periodically allowing vBMD values to be obtained during routine CT exams in the absence of a phantom. The International Society for Clinical Densitometry (ISCD) indicates that T-score values obtained from QCT can be used for osteoporosis diagnosis only at femoral neck and total hip, as they are equivalent to DXA T-score [23]. At the spine, however, there is discordance between T-scores obtained by DXA and QCT [24], that could be more evident in presence of artifacts due to degenerative diseases or vertebral fractures (Fig. 3). To avoid diagnostic confusion that could result from different T-scores and facilitate the interpretation of QCT spine results, diagnostic cut points (120 mg/cm3 and 80 mg/cm3) were used to assign a diagnostic category based on QCT measurements [25].
Image showing the added value of QCT analysis in a female subject of 82 years old with severe osteoarthrosis at lumbar spine. The DXA scan on the left side has poor diagnostic value due to extensive arthrosis related to degenerative scoliosis, and normal T-score values. The corresponding QCT analysis on the right side allows for selective selection of trabecular region of interests, avoiding areas of bone sclerosis, with volumetric BMD values (mg/cm3) in the range of osteoporosis. QCT thresholds are expressed according to the American College of Radiology (ACR) classification [25]
HR-pQCT is currently the most powerful tool for the evaluation of bone quality. Thanks to its very high spatial resolution (82 μm, very close to the in-vivo range of trabecular scale) HR-pQCT can measure total, trabecular and cortical vBMD, also providing several parameters related to bone microarchitecture and quality. Among the various parameters, cortical vBMD, trabecular thickness (a measure of the mean thickness of trabeculae) and bone stiffness (a measure of bone strength derived by HR-pQCT) are reported to be the best predictor of fragility fracture, especially when measured at the distal radius [26]. Advantages of HR-pQCT are the short acquisition time (about 3 min) and the very low radiation exposure to patient. Drawbacks are the need for small field of views that limit HR-pQCT applicability to non-central sites (distal ulna and radius), as well the fact that it remains a rather expensive technique with limited clinical applicability.
Radiomics analysis on opportunistic CT scan has been proposed as promising technique for analyzing the textures and spatial heterogeneity in subjects with skeletal fragility at risk of vertebral fractures [27, 28]. Radiomics refers to comprehensive, automated high-throughput mining of quantitative medical image features to capture disease characteristics that are difficult to identify by the human eye; in turn, this supports clinical decision making with improved diagnostic and/or predictive performance [29, 30].
The last in-vivo technique that can provide information about cortical and trabecular parameters is high-resolution MRI (HR-MRI), which of course has the advantages over CT of being a non-ionizing technique that can be used both at central and peripheral sites. MRI image relies on the fact that cortical and trabecular bone do not produce signal, compared to the fatty replaced marrow spaces. Despite HR-MRI sequences can reach very high spatial resolution, its use is still mainly limited to research practice because it is technically difficult to perform and requires long post-processing time from experienced operators, resulting in limited availability and consistency of measurements between different scanners [22].
Another non-ionizing technique recently introduced is the Radiofrequency Echographic Multi-Spectrometry (REMS), which uses an ultrasound device to obtain spectral analysis of the raw unfiltered ultrasound signals reflected by bones. REMS is applied to lumbar spine and femur and is able to obtain -similarly to DXA- ultrasound BMD value expressed as g/cm2. A significant correlation between BMD measured by REMS and DXA has been reported, together with very good precision values [31]. A recent REMS-derived “fragility score” has been developed as indicator of bone quality that is independent of BMD: low values of fragility score are associated with higher quality of the evaluated bone [32].
Techniques to be exploited in the future to directly assess bone resistance to applied forces include impact microindentation, a method to directly assess tissue-level material properties of cortical bone in vivo in humans, also referred to as Reference Point Indentation (RPI). This method employs a single hand-held RPI device, i.e. a stainless-steel probe with spheroconical tip (Osteoprobe®) which imparts a single impact load to the bone surface and receiving a feedback from the bone proportional to its resistance, defined as bone material strength index (BMSi) [33]. To date, no reference range has been published and no data are available in patients with endocrine diseases.
Ex vivo techniques such as back scattering electron imaging (BSEI) to assess mineralization, both static and dynamic histomorphometry on bone specimens, and vibrational spectroscopic techniques are additional systems to assess skeletal microstructure. Nonetheless, they have been left out of this paper since they are referred mainly to research settings and not routinely performed in clinical practice.
Bone quality in parathyroid disorders
Besides the effects on calcium homeostasis, parathyroid hormone (PTH) directly influences bone remodeling with significant effect on BMD and bone quality exerted by both chronic hormone excess and deficiency.
Primary hyperparathyroidism
Primary hyperparathyroidism (PHPT) is defined as hypercalcemia and concomitant hyperparathyroidism or inappropriately elevated PTH secretion. It is the third most common endocrine disorder, after diabetes and thyroid diseases, in postmenopausal women. It is sustained by parathyroid cells proliferations, mainly as benign adenomas or multiglandular hyperplasia, rarely as carcinomas. PTH is deeply involved in bone metabolism by promoting both bone resorption and formation. Therefore, bone is a main target of PHPT. In the rare classical PHPT phenotype, osteitis fibrosa cystica (OFC), was the hallmark of the disease [34]. It was characterized by brown tumors on long bones, evidence of subperiosteal bone resorption, "salt and pepper" radiographic erosions of the skull bones, and tapering of the distal portions of the finger bones and clavicles [34]. However, it has been reported that prevalence of milder signs of OFC, such as subperiosteal bone resorption in the fingers and skull mottling, are not uncommon in current PHPT clinical presentation [35].
The current most common clinical presentation of PHPT is characterized by mild disease with an increased risk of fragility fractures. PHPT predominantly occurs in post-menopausal women, who have an increased risk of osteoporosis and fractures due to ageing and loss of the protective effects of estrogen, and PHPT further increases this risk.
Early studies assessing BMD by DXA in PHPT patients demonstrated preferential reduction of BMD at sites with predominantly cortical bone such as the radius, with relative sparing of sites with predominantly trabecular bone such as the lumbar spine [36, 37].
Histomorphometry from iliac crest biopsies of patients with PHPT demonstrated increased bone turnover, thinning of cortical bone, and increased cancellous bone volume, suggesting preservation and even an increase in trabecular bone volume in PHPT patients [38].
Despite the preserved BMD at the lumbar spine by DXA, individuals with PHPT have an increased incidence of both vertebral and non-vertebral fractures [39,40,41], suggesting an impairment of bone microarchitecture in PHPT patients. Consequently, non-invasive techniques assessing trabecular and cortical microstructure, such as peripheral QCT, HRpQCT and TBS have been considered of interest. By pQCT and HRpQCT, abnormalities were observed at both trabecular and cortical sites in postmenopausal women with PHPT [42,43,44]. In adults with PHPT, a good correlation between HRpQCT and TBS has been found [45]. TBS has been shown to be reduced in more than a half of PHPT patients [46] and significantly associated with vertebral fractures in postmenopausal women and in older men with PHPT, independently of lumbar spine BMD, with less evidence of correlation with non-vertebral or total fractures [47, 48]. Recently, in a small cohort of adult Australian PHPT patients follow up for 10 years, the addition of TBS to BMD showed a trend to improve fracture prediction without reaching statistical significance [46].
Besides low TBS, there is evidence that PHPT might be associated also with high BSI values that resulted to have moderate accuracy in identifying subjects with vertebral fractures [49].
A clue of the outcome of surgical resolution of PHPT by parathyroidectomy is related to the reduction of the risk of fractures [50]. However, studies examining changes in TBS after parathyroidectomy are conflicting, with some showing improvement in TBS [48, 51] and others showing no change [46, 52, 53], which may be related to differences in the population studied, disease severity, or sample size. Using HRpQCT, 1- and 2-year studies showed improvements in cortical and trabecular compartments after surgical cure of primary PHPT [54, 55]. It is still unknown whether cinacalcet might induce effects on bone quality in subjects with PHPT [56].
In conclusion, bone quality assessment by HRpQCT or TBS may improve the fracture risk estimation in PHPT patients and can be useful in the indication of parathyroidectomy; however, further robust studies are advisable.
Hypoparathyroidism
Hypoparathyroidism (HypoPT) is characterized by absence or deficient PTH secretion causing hypocalcemia. It is a rare disorder, whose clinical expression depends on the etiology [57]; genetic, idiopathic and autoimmune disorders are by definition chronic conditions, while chronic postsurgical HypoPT is defined when the disorder persists 6 months after neck surgery [58]. As in PHPT, complications of persistently deficient or absent PTH secretion involve the skeleton.
Bone remodeling is persistently low in HypoPT, as demonstrated by histomorphometric studies [59, 60]. The assessment of bone formation rate by tetracycline labeling showed lower values in HypoPT compared to control subjects on the cancellous, endocortical and intracortical surfaces [59, 60]. Mineral apposition rate and mineralizing surface, as well as osteoid width and surface are reduced, concomitantly with a significant lower bone resorption rate [59, 60]. As a consequence, higher, unmineralized, bone tissue is made [59, 60]. Trabecular bone volume is increased in HypoPT, mostly in association with higher trabecular width, and there is a trend towards an increase in cortical width and decreased cortical porosity [60, 61]. The low bone remodeling and the increased bone volume described in histomorphometric studies are concordant with the assessment of biochemical markers of bone turnover, that are typically in the low range of normal, and the high BMD by DXA observed in HypoPT [60, 62].
Assessment of trabecular microarchitecture by TBS was performed in five clinical studies with a total of 317 patients [52, 63,64,65,66]. Overall, they were concordant in illustrating that the degraded trabecular microarchitecture is evident in HypoPT postmenopausal women with postsurgical HypoPT [64, 66]. Sakane et al. studied a cohort of patients with postsurgical HypoPT (85.4% female, 68.6% postmenopausal) and found that more than 30% (mostly postmenopausal women) had TBS values below the normal cutoff value (1.31) [64]. In an Italian cohort of 50 postmenopausal women with postsurgical hypoparathyroidism, mean TBS values were well below the normal range and were not different compared to healthy age- and sex-matched controls [66]. Conversely, mean TBS values are above the normal range in HypoPT cohorts including non-surgical patients [52, 63, 65]. As far as the effect of menopause on TBS in this group, results are not concordant [63, 65]. Similar values in pre- and post-menopausal HypoPT women are described in one study [63], while lower TBS values and higher rate of values within the “degraded microarchitecture” pattern in postmenopausal compared to the pre-menopausal counterpart are reported in another cohort [65].
Results on the role of TBS as determinant of vertebral fractures risk are not concordant among studies; two of them excluded [65, 66], while one confirmed [64] that TBS could be considered as predictor of vertebral fractures in HypoPT subjects. Non definitive results were observed in terms of TBS changes during recombinant human (rh) PTH(1–84) therapy; a trend towards an increase in TBS values, although not significant in some cases, was observed [52, 63]
Noninvasive assessment of bone quality by pQCT showed higher trabecular vBMD values in HypoPT compared to healthy subjects [67]. By HRpQCT, trabecular vBMD and trabecular number are higher in younger HypoPT compare to age and sex-matched controls [68, 69]. Trabecular thickness and separation was lower at the tibia in post-surgical and nonsurgical HypoPT of different age classes [68,69,70]. As far as the cortical compartment, different results are reported by clinical studies [68,69,70]. Cortical volumetric density was reported as higher, comparable and lower in HypoPT compared to controls, according to different population studied and methodologies used [62, 68,69,70]. Cortical porosity was lower at the radius and tibia in HypoPT [68,69,70]. During rhPTH(1–84) therapy, trabecular vBMD declines after 5 years and returned to baseline values afterwards; conversely, there is a continuous decline in cortical vBMD up to 12 years [71].
The overall evidence shows that bone quality may be impaired in HypoPT, particularly at the trabecular sites, with potential higher risk of vertebral fractures in postmenopausal women. More data are definitely needed to assess eventual differences in bone quality between different age groups and etiologies of the disease, as well as the association with fracture risk.
Bone quality in thyroid disorders
Thyroid hormones have physiological stimulatory effects on bone remodeling and bone mineralization, and normal euthyroid status during childhood and adolescence is required for acquisition of peak bone mass [72]. Interestingly, also thyrotropin (TSH) can act directly on bone cells possibly modulating the skeletal effects of thyroid hormones. Specifically, in pre-clinical and clinical studies, TSH was shown to inhibit osteoclastogenesis and bone resorption [73]. Indeed, low TSH values might contribute to bone loss in subjects with hyperthyroidism [74].
Both excess and defect of thyroid hormones have been linked to bone fragility with various degree of evidence [75].
Prolonged, overt endogenous or exogenous thyrotoxicosis is a known cause risk of secondary osteoporosis and fragility fractures [76]. For this reason, long-standing hyperthyroidism is included in fracture-risk assessment of major algorithms such as FRAX®.
The treatment of hyperthyroidism with the achievement of a euthyroid status is well documented to significantly increase BMD in the medium-long term along with a reversion to normal of fracture risk which is increased at baseline [76]. Also TBS has been shown to improve in a group of young adults of both sexes with Graves’ disease under anti-thyroid treatment even in the short-medium term in a retrospective study [77]. The effects of hyperthyroidism on bone quality have been assessed in a recent paper comparing in a case–control study 61 women with a similar number of euthyroid women matched for age and menopause status by means of HR-pQCT [78]. While no DXA changes at the lumbar spine and the hip were detected, lower vBMD and estimated bone strength, and compromised cortical microarchitecture at the radius were demonstrated at baseline [78]. These results confirmed in vivo for the first time old histomorphometric analyses on bone biopsies, which showed reduction in cortical thickness and increased cortical porosity as effects of increased bone turnover [79]. In the longitudinal study, a significant improvement in vBMD and cortical microstructural parameters (cortical thickness and cortical area, as assessed at the radius and tibia) were observed 1 year after restoration of euthyroidism and independently of bone specific treatments, underlining the need to treat hyperthyroidism for bone quality improvement [78].
Studies assessing the effect on BMD of Levo-thyroxine (L-T4)-mediated TSH suppression, also referred to as subclinical thyrotoxicosis, have shown bone detrimental outcomes with increased risk of major osteoporotic fractures especially in postmenopausal women and older males [80], with contradictory results in premenopausal women or younger men [75]. Nonetheless, a meta-analysis including 13 prospective international cohorts has shown that in age- and sex-adjusted analyses subclinical thyrotoxicosis is associated with increased risk of hip fracture and other major osteoporotic fractures, particularly if TSH levels are less than 0,1 mIU/ml [81].
The effects of subclinical thyrotoxicosis (i.e. suppressed TSH and normal free thyroid hormones levels) on bone quality have been mainly analyzed in patients with differentiated thyroid cancer (DTC) receiving L-T4 therapy at a dosage able to suppress TSH levels, generally assessing TBS [80]. The finding that lower TSH is associated with a deterioration of bone quality in postmenopausal women, as assessed by TBS has been highlighted by various studies [80, 82, 83].
In particular, Chung et al. have shown in a retrospective study that in a large group of thyroidectomized postmenopausal women with DTC treated with suppressive doses of L-T4, TBS was markedly decreased, while no significant difference was evidenced as compared to the ones receiving L-T4 non-suppressing TSH [82]. In patients receiving anti-osteoporotic medications, positive significant changes in TBS were observed just in the group receiving non-suppressive L-T4 therapy [82].
The duration of TSH suppression was shown to be linked to higher risk of fragility fractures and deterioration of TBS in postmenopausal women with DTC, while no changes in microarchitecture was detected in premenopausal women in the long-term, as demonstrated in several retrospective studies [84,85,86,87]. These findings were recently confirmed in a following study group mainly composed of postmenopausal women with DTC with TSH constantly below 0.1 mIU/ml [88].
Better detection of osteopenia or osteoporosis was obtained by means of cQCT versus DXA in patients with DTC under TSH suppressive therapy, despite no control group was included [89]. vBMD and bone geometry (i.e. trabecular bone mineral content and cortical thickness) only at a non-bearing site (distal radius) were found to be altered in postmenopausal women with DTC under TSH suppression as compared to premenopausal patients, although pQCT scans were obtained just once in a cross-sectional design study [90].
The influence of either TSH levels or FT4 levels on bone quality as assessed by TBS has been tried to be dissected in studies in cohorts composed of euthyroid subjects.
An analysis in a large group of 1475 women, a subgroup belonging to an ongoing prospective study carried out in Switzerland looking at the relationship between cardiovascular diseases and osteoporosis (CoLaus/OsteoLaus cohort) has been performed. While no association was found between TSH and DXA parameters, a positive association was described between TSH and TBS at baseline (cross-sectional study) and a negative association between TSH and incident fractures in the 5-year follow-up period (longitudinal study) [91]. Nonetheless, in a previous study in euthyroid patients of both sexes (n. 1648 patients, 648 postmenopausal women and 728 men), an independent association between high-normal FT4 levels and TBS, irrespective of TSH levels, was found in community-dwelling, postmenopausal women [92].
Subclinical hypothyroidism does not seem to be associated with alteration on bone quantity and quality (as assessed by TBS), nor the proper replacement with L-T4 leads to any effect on bone health in a randomized nested trial [93]. Nonetheless, a small study including women with either overt or subclinical hypothyroidism has shown that this disease is associated with increased trabecular bone area and a lower mineral density of cortical bone in the radius, as examined by HR-pQCT [94]. In this study, restoration of euthyroid state was accompanied by an increase in cortical porosity, without variations in estimated bone strength [94].
In conclusion, the results of these studies suggest that TBS should always be included in the baseline skeletal evaluation of patients receiving L-T4 therapy at TSH suppressive dosage, in particular in postmenopausal women, but even in premenopausal women lately transitioning to menopause. Therefore, TBS-adjusted FRAX should be calculated to assess fracture risk in these patients, even more so considering the long survival of these patients. Studies employing HR-pQCT as well as longitudinal/prospective studies are still needed in overt hyperthyroidism and hypothyroidism and subclinical thyrotoxicosis, with a particular focus in bone quality issues in older men.
Bone quality in hypercortisolism
Glucocorticoids in excess induce direct and indirect effects on bone remodeling and metabolism, these comprising suppression of osteoblastogenesis and bone formation, relative increase in bone resorption, inhibition of osteocyte function and survival, and alterations in calcium homeostasis by inducing negative calcium balance [73]. Moreover, cortisol hypersecretion can cause functional growth (GH) deficiency (GHD) and central hypogonadism, both contributing to skeletal fragility in this clinical setting [73].
Consistent with the aforementioned actions of glucocorticoids on bone cells [73], uncoupled bone remodeling with low bone formation and slight increase in bone resorption has been described in subjects with endogenous hypercortisolism [95]. These alterations in bone remodeling are cause of deterioration in bone structure and quality, predominantly in trabecular bone, as evidenced by HR-pQcT [96], DXA evaluation of BMD at the lumbar spine and femoral neck [97], DXA measurement of TBS [98], assessment of bone marrow fat by MRI [99, 100], and volumetric bone evaluation by forearm QCT [95]. However, also cortical bone structure can be altered in individuals with endogenous hypercortisolism [101].
A number of studies consistently documented increased incidence and prevalence of fragility fractures in endogenous hypercortisolism, with vertebrae and ribs being the skeletal sites most frequently involved [73, 102]. The relationship between decreased BMD and fractures in hypercortisolism is only partly maintained. Subjects with densitometric diagnosis of osteoporosis have the highest likelihood of developing fractures [103, 104], but a remarkable number of fragility fractures can occur even in the context of normal or low-normal BMD [103,104,105,106]. It is still uncertain whether evaluation of bone microstructure in endogenous can improve prediction of fractures. In a cohort of 182 young subjects with hypercortisolism, fragility fractures did not result to be significantly associated with TBS [106]. In another study performed in 102 elderly subjects with “subclinical” hypercortisolism associated with adrenal incidentaloma, measurement of TBS improved prediction of incident vertebral fractures [107]. In line with this finding, a more recent study reported a significant association between vertebral fractures and increase in bone marrow fat measured by MRI in subjects with endogenous hypercortisolism and low-normal BMD [99], consistent with the pathophysiological concept that enhanced adipogenesis in bone marrow reflects the impairment of osteoblastogenesis induced by glucocorticoid excess [73].
After treatment of hypercortisolism, bone quality as assessed by TBS measurement seems to improve more rapidly compared to BMD, since more than two-thirds of 110 individuals with alterations in bone microstructure at diagnosis of Cushing syndrome were shown to have a clinically relevant improvement of TBS by 24 months after remission of hypercortisolism [98]. The impact of this improvement in bone quality on outcome of fracture risk is still uncertain, since fragility fractures can occur even in subjects with cured hypercortisolism, especially if they had experienced previous fractures [108, 109].
Bone quality in growth-hormone disorders
GH and insulin-like growth factor 1 (IGF1) are important regulators of bone remodeling and metabolism and have an essential role in the achievement and maintenance of bone mass throughout life. In experimental models and in humans diseases, both GHD and GH excess cause alterations in bone structure with high risk of fragility fractures [110].
Growth hormone deficiency
GHD is the main determinant of skeletal fragility in subjects with hypopituitarism [111]. Moreover, a deregulation of GH-IGF1 axis with functional defect in GH and/or IGF1 can contribute to bone loss in several clinical conditions as ageing, anorexia nervosa, diabetes mellitus and chronic exposure to glucocorticoid excess [110, 112, 113]. Individuals with deficiency in GH and IGF1 have suppressed bone turnover due to impaired osteoblastogenesis [114]. Consequently, bone quality is altered in GHD and risk of fractures is 2–5 higher as compared to general population, regardless of the existence of other pituitary hormone deficiencies [73]. Importantly, symptomatic vertebral fractures occur in more than 30% of adult men and women with acquired untreated GHD, even in the context of a normal BMD [115]. Data on bone structure in GHD are scanty and no studies evaluated the value of bone quality assessment in predicting the risk of fractures in this clinical setting [73]. Bone biopsies from male adult patients with GHD reveal decreased osteoid and mineralizing surfaces and decreased bone formation rate [116]. Studies of 34 individuals with either childhood-onset or long-standing GHD that used a 3D DXA algorithm at the hip site and HR-pQCT of the distal radius and tibia demonstrated decreased cortical area and cortical thickness, as well as decreased trabecular thickness in cancellous bone [117, 118]. However, bone microarchitecture might not be altered in adults with GHD with onset after the achievement of peak bone mass [119]. Indeed, some of these patients had previously received GH replacement more than 12 months before the study and this may have counteracted any abnormalities in bone structure and strength at the time of evaluation, consistently with the concept that beneficial effects of replacement therapy may be persistent after recombinant human GH withdrawal [120].
Acromegaly
In the context of GH and IGF1 excess, bone resorption is increased to a greater extent than bone formation and as a result bone loss ensues [121]. Consistently, a large number of individuals with acromegaly have abnormalities in bone structure characterized by decreased cancellous bone volume and increased cortical porosity [119, 122, 123]. The first evidence of impaired bone quality in acromegaly was provided by Ueland et al. who described loss of trabecular connections and low trabecular bone biochemical competence in iliac crest biopsies of 13 acromegalic patients with active disease [124]. Following this evidence, many other studies confirmed that trabecular bone structure is profoundly deteriorated in acromegaly regardless of gonadal status of subjects [119, 122, 125]. Indeed, TBS results to be frequently degraded in subjects with acromegaly [126,127,128,129]. In a recent meta-analysis including 336 acromegalic patients and 490 non-acromegalic subjects, TBS resulted to be significantly lower in acromegaly as compared to controls irrespective of disease activity and gonadal status [129]. Besides the abnormalities in trabecular microstructure, acromegaly was shown to affect also cortical bone in terms of increased porosity [119, 123, 130] and impaired cortical properties [131]. As a direct consequence of alterations in bone microstructure and quality, vertebral fractures develop in a large number of subjects with acromegaly [121]. Indeed, more than one-third of individuals exposed to GH hypersecretion develop vertebral fractures, with a fracture risk which is 3- to 8-fold greater compared to that in control subjects [121]. Vertebral fractures occur even in subjects with normal BMD [132]. In this setting, vertebral fractures were associated with deterioration in bone microstructure as assessed by histomorphometry, tridimensional DXA of the proximal femur, and peripheral CT of distal radius [123, 126, 130]. Moreover, in a meta-analysis of two studies including 132 subjects with acromegaly who were evaluated for vertebral fractures [126, 133], TBS values were found to be significantly lower in fractured patients as compared to those who did not fracture, with a pooled mean difference of − 0.099 [129]. Biochemical control of acromegaly induces a rapid decrease in bone turnover, however, bone microstructure remains deteriorated in a number of individuals following disease control [73]. Indeed, treatment of acromegaly can cause a worsening in TBS values, an increase in cortical porosity and a reduction in cortical bone thickness and structural properties [73, 123, 133, 134]. The mechanisms responsible of deteriorated bone quality in individuals with controlled acromegaly are still largely unknown, but a persistent impairment of osteoblastogenesis and bone formation could be hypothesized [123, 125]. Coexistent hypopituitarism, as well as medications used for the treatment of acromegaly, might contribute to the persistent alterations in bone remodeling and structure in subjects with treated disease [135,136,137]. As a direct consequence of persistent alterations in bone structure, a high number of individuals with acromegaly are at substantial risk of vertebral fractures after biochemical control of the disease [136, 138].
Only one study including 33 acromegalic subjects has so far evaluated performance of REMS in acromegaly [139]. This technique did not seem to provide any significant advantage in evaluating bone quality as compared to DXA.
Bone quality in male hypogonadism
Hypogonadism is a well-known risk factor for secondary osteoporosis in males [140,141,142,143]. Animal and clinical evidence have clarified that testosterone (T) plays a crucial role in regulating bone homeostasis during all ages. Androgen, along with estrogen receptors, are widely expressed on osteoblasts, osteoclasts, osteocytes and marrow stromal cells [143]. Both T and estradiol are involved in periosteal apposition and trabecular bone growth, during pubertal period, contributing to pubertal growth spurts and peak bone mass [142, 144]. However, due to the higher circulating levels observed in males, T contributes to develop higher peak bone mass characterized by wider bones with a thicker cortex when compared to what observed in women [142, 143, 145]. After puberty, androgens still contribute to bone homeostasis by reducing the bone remodeling rate and by regulating a balance between bone resorption and formation [143, 145].
In line to what observed in the pre-clinical models, data derived from the general population have confirmed an inverse association between aBMD and circulating T levels [146,147,148]. However, the European Male Aging Study, including more than 3400 subjects aged 40–80 years old, documented a reduced aBMD only when severe hypogonadism (total T < 8 nmol/L) was observed [146]. Similar data were derived from a randomized controlled trial including 198 healthy men, receiving goserelin acetate to suppresses endogenous gonadal steroid production [149]. In line with these observations, available guidelines indicate to routinely assess BMD, through DXA, in patients with organic hypogonadism, which is usually associated with more severe reduction of T levels when compared to what observed in late onset hypogonadism (LOH) [140, 150,151,152,153]. However, several male subjects with hypogonadism can have skeletal fragility with high risk of fractures even in the context of a normal BMD [154, 155]. In this setting, evaluation of bone quality is mandatory. Using HR-MRI technique at distal tibia, two studies including 20 hypogonadal men provided evidence that bone loss and skeletal fragility in male hypogonadism are caused by a conversion of plate-like to rod-like trabeculae characterized by low surface-to-curve ratio, high erosion index and low bone properties and mechanical competence, as defined by FEA [156, 157]. The alterations in trabecular bone architecture associated with hypogonadism were also captured by CT techniques [158,159,160] and DXA-measurement of TBS [161, 162] in 146 subjects with congenital hypogonadism. Noteworthy, alterations in bone microstructure were detected even in subjects with central hypogonadism in whom serum follicle-stimulating hormone (FSH) are very low [160, 163], questioning the relevance in humans of experimental data suggesting a role of high FSH in pathogenesis of hypogonadal bone loss [73]. HR-pQCT technique revealed also reduced cortical area and thickness that could contribute to compromised bone strength in males with hypogonadism [158, 159, 163]. Interestingly, deterioration in bone quality has been associated with increase in visceral fat induced by hypogonadism [162], consistently with the concept that visceral-fat derived proinflammatory cytokines can influence bone remodeling and structure [164]. Moreover, alterations in cortical and trabecular bone microstructure resulted to be more severe in hypogonadal subjects with lower appendicular lean mass index [159].
The positive effects of T replacement therapy (TRT) in males with hypogonadism are well-documented [141, 165, 166]. Data derived from the largest meta-analysis published so far, including all available placebo-controlled Randomized Control Trials and observational studies, showed that TRT resulted in more evident effects on aBMD, at lumbar level, only when studies including hypogonadal subjects (total T < 12 nmol/L) at enrolment were considered. The results were inversely related to baseline T levels and confirmed when subjects without LOH were excluded from the analysis [141]. The magnitude of the effects of TRT on aBMD at lumbar site in LOH, as derived from Corona et al. [141] study, was similar to that observed by the same group analyzing the results derived from subjects with Klinefelter’s Syndrome [165]. Furthermore, the same meta-analysis [141] showed better outcomes in subjects with diabetes mellitus (DM), which is frequently co-morbid in patients with LOH [167].
The effects of TRT on bone quality have been investigated in several clinical contexts. In 211 older hypogonadal subjects, 1-year TRT increased vBMD as determined by QCT, especially in trabecular bone than cortical-rich peripheral bone, and increased estimated bone strength, as determined by FEA [168], whereas TBS did not change significantly [169]. The reasons of such a methodological variability are unknown but one could argue that TBS might not be clinically useful in monitoring the short-term effects of TRT on bone quality. Interestingly, the favorable effects of TRT on bone structure resulted to be more evident in hypogonadal subjects with type 2 diabetes (T2D) than those without T2D [170]. Based on the changes of biochemical markers of bone turnover, it is reasonable to hypothesize a predominant anabolic effect of T on the skeleton of men with T2D, while the antiresorptive effect seems to prevail among those without T2D. In 51 subjects with congenital hypogonadism, the effects of TRT on trabecular bone resulted to be larger when treatment was started early during adolescence, whereas when diagnosis was late and treatment was started after closure of epiphysis only cortical bone can improve as effect of replacement therapy [163].
Bone quality in diabetes mellitus
DM is a common disease, linked to several complications, including osteoporosis and fractures. Fracture risk is highly increased in type 1 diabetes (T1D), mostly in patients with longer duration of the disease, worse glucose control or family history for fractures [171]. In T2D, fracture risk is mostly related to cortical rich bone sites like proximal and distal femur and it is strongly associated to insulin or sulphonylureas use, falls, prolonged poor glycemic control [172, 173]. Contrary to what commonly believed, fracture risk is increased also in subjects with insulin resistance without DM, suggesting that pathophysiology of bone fragility in subjects with DM may begin even before the onset or diagnosis of the disease [174]. Underlying mechanisms of bone fragility in DM are still poorly understood with several factors like gluco-toxicity, inflammation, WNT impairment, oxidative stress, micro-angiopathy playing a main role [175]. Advanced glycation end products (AGEs), linking collagen, reduce bone flexibility and energy dissipation, impair strength and biomechanical properties of cortical and trabecular bone. In the meantime, AGE may interfere with immune cells, increasing cytokines and reactive oxygen species (ROS), impacting on bone resorption [176]. AGEs are also responsible of microvascular damage that independently affects bone health as well [177]. Low T frequently observed in T2D can represent another risk factor [178]. Serum sclerostin levels are consistently reported as increased in both T2D and Latent Autoimmune Diabetes in Adults (LADA) subjects, underlying the impairment of WNT signaling in DM [179, 180]. Subjects with T2D present also increased SOST and lower RUNX2 gene expression at the bone level, compared to healthy controls, further confirming that impaired WNT signaling plays a key role in determining lower bone formation in DM [181]. These data are in agreement with histological findings that have shown lower tetracycline labeling in bone biopsies in T2D subjects and with reports of lower serum bone formation markers. As a matter of fact, it is now clear that DM is a low bone turnover condition, characterized by both lower bone formation and resorption. This unique feature may cause accumulation of micro-damage, more homogeneous bone structure and, in turn, more propagation of microcracks.
Although fracture risk is increased, BMD in T2D is often normal or even higher than healthy subjects, indicating that bone quality rather than density is likely impaired [182]. On the contrary, T1D patients show low BMD although fracture risk is usually higher than that suggested for a given T or Z-score. On the attempt to investigate this apparent discrepancy between BMD and fracture risk, several studies have investigated bone micro-architecture, used TBS or have combined different available fracture algorithms. Using HRpQCT, cortical porosity has been shown to be increased in T2D in most studies, mostly in subjects with fractures, or cardio-vascular complications [183, 184]. Moreover, creep indentation distance and indentation distance increase were greater (by 18% and 20%, respectively) in cortical bone from T2D than in subjects without DM [185]. However, trabecular bone indices are either comparable or show even increased density compared to controls [186, 187]. Altogether, this evidence proves that subjects with DM may present a unique micro-architecture feature with normal trabecular bone and high cortical porosity that may impair failure load and bone strength. Two large studies including 3051 subjects with DM provided convincing evidence that TBS is more reliable than BMD in capturing the DM-associated fracture risk [188, 189]. Similar results were reported more recently using REMS in 90 post-menopausal women with T2D [190]. In this setting, the percentage of T2D women classified as "osteoporotic", on the basis of BMD by REMS was markedly higher with respect to those classified by DXA and T2D women with fragility fractures presented significantly lower values of BMD-LS by REMS with respect to those without fractures [190].
Treatment of diabetes can influence skeletal health and fracture risk. Fracture risk in DM is increased also because of higher risk of hypoglycemic events, falls, use of some medications like insulin or sulphonylureas [173]. Therefore, metformin and new medications with a safe bone profile like glucagon like peptide 1-receptor agonist (GLP1-RA) analogues or sodium-glucose co-transporters 2 (SGLT2) inhibitors should be preferred. Optimization of glucose control will not only prevent cardio-vascular complications but will determine also lower oxidative stress, glucose toxicity, bone microangiopathy, AGEs accumulation and inflammation, which will result in better bone quality. All subjects with DM with poor glycemic control or on insulin treatment should be screened for osteoporosis and should start anti-osteoporosis treatment when needed. According to the IOF algorithm, occurrence of fragility fractures (vertebral or proximal femur) or a T score of -2 SD should be considered as a threshold for treatment [191]. According to a recent metanalysis [192], all anti-resorptive medications have a positive effect on both BMD and fracture risk in subjects without DM. In this context of low-bone turnover osteoporosis, teriparatide is expected to expand the “anabolic window” with secondary improvement of bone quality. As a matter of fact, teriparatide has been shown to significantly prevent fracture risk in subjects with DM [193].
Conclusions
Osteoporosis is a frequent complication of several endocrine diseases, including primary hyperparathyroidism, hyperthyroidism, Cushing’s syndrome, GHD, acromegaly, male hypogonadism and DM. Prediction of fragility fractures in these clinical conditions, such as in other forms of secondary osteoporosis, is a challenge since BMD allows to identify only a minority of subjects at risk of fractures. Since bone quality is predominantly altered in endocrine-related osteoporosis, assessment of skeletal fragility in endocrine diseases may take advantage from the use of non-invasive techniques to detect perturbation in the trabecular and cortical bone architecture with the aim of best identify high risk patients (Table 1). In this context, HRpQCT represents the gold standard technology for the non-invasive evaluation of bone quality in several skeletal disorders. Notwithstanding, owing to the unavailability of HRpQCT in clinical practice, clinicians may be supported by the use of modern DXA machines and related DXA-derived parameters in the diagnostic work-up of endocrine disease-related osteoporosis. These technologies provide several additional information on bone structure, properties and geometry not captured by the isolated measurement of BMD and essential to identify subjects with skeletal fragility. For instance, evaluation of bone quality by using DXA measurement of TBS might be proposed for all individuals with endocrine disorders potentially causing skeletal fragility, which do not have BMD values in the range of osteoporosis. The finding of degraded TBS in these cases might help the clinicians to perform a careful monitoring of skeletal fragility and to put in place the prevention of fractures. In Fig. 4, a screening procedure for bone fragility assessment in endocrinopathies is proposed. Indeed, in this context, more data from longitudinal studies assessing changes in these parameters over time associated with the cure of the underlying endocrine disorder, as well as the effectiveness of bone active agents, will significantly impact the clinical management of these patients.
Proposed clinical assessment of bone fragility in secondary endocrine osteoporosis (treatment of underlying endocrine disease is taken for granted and mandatory); VF vertebral facture; DXA dual-energy X-ray absorptiometry; FRAX® international algorithm to estimate the risk of major osteoporotic fractures and hip fractures in a 10-year time; *, T-score is used in post-menopausal women and in men older than 50 years; **, Z-score is used in pre-menopausal women and in men younger than 50 years; ***, follow-up is advised if there are coexistent risk factors of fractures or the endocrine disorder is not resolved; ****, consider treatment with bone-active drugs if there are coexistent risk factors of fractures or the endocrine disorder is not or cannot be resolved. The proposed therapeutic flow-chart doesn’t consider the country-specific criteria of drug reimbursement for osteoporosis
Data availability
Not applied.
References
Borgstrom F, Karlsson L, Ortsater G, Norton N, Halbout P, Cooper C, Lorentzon M, McCloskey EV, Harvey NC, Javaid MK, Kanis JA, International Osteoporosis F (2020) Fragility fractures in Europe: burden, management and opportunities. Arch Osteoporos 15(1):59. https://doi.org/10.1007/s11657-020-0706-y
Curtis EM, Dennison EM, Cooper C, Harvey NC (2022) Osteoporosis in 2022: care gaps to screening and personalised medicine. Best Pract Res Clin Rheumatol 36(3):101754. https://doi.org/10.1016/j.berh.2022.101754
Eller-Vainicher C, Falchetti A, Gennari L, Cairoli E, Bertoldo F, Vescini F, Scillitani A, Chiodini I (2019) Diagnosis of endocrine disease: EVALUATION of bone fragility in endocrine disorders. Eur J Endocrinol. https://doi.org/10.1530/EJE-18-0991
Lewiecki EM (2022) Evaluating patients for secondary causes of osteoporosis. Curr Osteoporos Rep 20(1):1–12. https://doi.org/10.1007/s11914-022-00717-y
Anderson PA, Freedman BA, Brox WT, Shaffer WO (2021) Osteoporosis: recent recommendations and positions of the American Society for Bone and Mineral Research and the International Society for Clinical Densitometry. J Bone Jt Surg Am 103(8):741–747. https://doi.org/10.2106/JBJS.20.01248
Unnanuntana A, Rebolledo BJ, Khair MM, DiCarlo EF, Lane JM (2011) Diseases affecting bone quality: beyond osteoporosis. Clin Orthop Relat Res 469(8):2194–2206. https://doi.org/10.1007/s11999-010-1694-9
Seeman E (2003) Bone quality. Osteoporos Int 14(Suppl 5):S3-7. https://doi.org/10.1007/s00198-003-1465-5
Brandi ML (2009) Microarchitecture, the key to bone quality. Rheumatology (Oxford) 48(Suppl 4):3–8. https://doi.org/10.1093/rheumatology/kep273
Seeman E, Delmas PD (2006) Bone quality–the material and structural basis of bone strength and fragility. N Engl J Med 354(21):2250–2261. https://doi.org/10.1056/NEJMra053077
Bouxsein ML (2003) Bone quality: where do we go from here? Osteoporos Int 14(Suppl 5):S118-127. https://doi.org/10.1007/s00198-003-1489-x
Hunt HB, Donnelly E (2016) Bone quality assessment techniques: geometric, compositional, and mechanical characterization from macroscale to nanoscale. Clin Rev Bone Miner Metab 14(3):133–149. https://doi.org/10.1007/s12018-016-9222-4
Seeman E (2008) Bone quality: the material and structural basis of bone strength. J Bone Miner Metab 26(1):1–8. https://doi.org/10.1007/s00774-007-0793-5
Boskey AL, Imbert L (2017) Bone quality changes associated with aging and disease: a review. Ann N Y Acad Sci 1410(1):93–106. https://doi.org/10.1111/nyas.13572
Osterhoff G, Morgan EF, Shefelbine SJ, Karim L, McNamara LM, Augat P (2016) Bone mechanical properties and changes with osteoporosis. Injury 47(Suppl 2):S11-20. https://doi.org/10.1016/S0020-1383(16)47003-8
Adams JE (2013) Advances in bone imaging for osteoporosis. Nat Rev Endocrinol 9(1):28–42. https://doi.org/10.1038/nrendo.2012.217
Krohn K, Schwartz EN, Chung YS, Lewiecki EM (2019) Dual-energy X-ray absorptiometry monitoring with Trabecular Bone Score: 2019 ISCD official position. J Clin Densitom 22(4):501–505. https://doi.org/10.1016/j.jocd.2019.07.006
Leslie WD, Shevroja E, Johansson H, McCloskey EV, Harvey NC, Kanis JA, Hans D (2018) Risk-equivalent T-score adjustment for using lumbar spine trabecular bone score (TBS): the Manitoba BMD registry. Osteoporos Int 29(3):751–758. https://doi.org/10.1007/s00198-018-4405-0
Silva BC, Broy SB, Boutroy S, Schousboe JT, Shepherd JA, Leslie WD (2015) Fracture risk prediction by non-BMD DXA measures: the 2015 ISCD official positions part 2: Trabecular Bone Score. J Clin Densitom 18(3):309–330. https://doi.org/10.1016/j.jocd.2015.06.008
McCloskey EV, Odén A, Harvey NC, Leslie WD, Hans D, Johansson H, Kanis JA (2015) Adjusting fracture probability by trabecular bone score. Calcif Tissue Int 96(6):500–509. https://doi.org/10.1007/s00223-015-9980-x
Ulivieri FM, Rinaudo L (2020) Beyond bone mineral density: a new dual X-ray absorptiometry index of bone strength to predict fragility fractures, the bone strain index. Front Med 7:590139. https://doi.org/10.3389/fmed.2020.590139
Ulivieri FM, Rinaudo L, Messina C, Aliprandi A, Sconfienza LM, Sardanelli F, Cesana BM (2022) Bone Strain Index: preliminary distributional characteristics in a population of women with normal bone mass, osteopenia and osteoporosis. Radiol Med (Torino) 127(10):1151–1158. https://doi.org/10.1007/s11547-022-01543-z
Geusens P, Chapurlat R, Schett G, Ghasem-Zadeh A, Seeman E, de Jong J, van den Bergh J (2014) High-resolution in vivo imaging of bone and joints: a window to microarchitecture. Nat Rev Rheumatol 10(5):304–313. https://doi.org/10.1038/nrrheum.2014.23
Engelke K, Lang T, Khosla S, Qin L, Zysset P, Leslie WD, Shepherd JA, Shousboe JT (2015) Clinical use of quantitative computed tomography-based advanced techniques in the management of osteoporosis in adults: the 2015 ISCD official positions-part III. J Clin Densitom 18(3):393–407. https://doi.org/10.1016/j.jocd.2015.06.010
Lin W, He C, Xie F, Chen T, Zheng G, Yin H, Chen H, Wang Z (2022) Discordance in lumbar bone mineral density measurements by quantitative computed tomography and dual-energy X-ray absorptiometry in postmenopausal women: a prospective comparative study. Spine J. https://doi.org/10.1016/j.spinee.2022.10.014
Radiology ACO (2018) ACR–SPR–SSR practice parameter for the performance of musculoskeletal quantitative computed tomography (QCT). American College of Radiology, Reston
Cheung WH, Hung VW, Cheuk KY, Chau WW, Tsoi KK, Wong RM, Chow SK, Lam TP, Yung PS, Law SW, Qin L (2021) Best performance parameters of HR-pQCT to predict fragility fracture: systematic review and meta-analysis. J Bone Miner Res 36(12):2381–2398. https://doi.org/10.1002/jbmr.4449
Mookiah MRK, Subburaj K, Mei K, Kopp FK, Kaesmacher J, Jungmann PM, Foehr P, Noel PB, Kirschke JS, Baum T (2018) Multidetector computed tomography imaging: effect of sparse sampling and iterative reconstruction on trabecular bone microstructure. J Comput Assist Tomogr 42(3):441–447. https://doi.org/10.1097/rct.0000000000000710
Biamonte E, Levi R, Carrone F, Vena W, Brunetti A, Battaglia M, Garoli F, Savini G, Riva M, Ortolina A, Tomei M, Angelotti G, Laino ME, Savevski V, Mollura M, Fornari M, Barbieri R, Lania AG, Grimaldi M, Politi LS, Mazziotti G (2022) Artificial intelligence-based radiomics on computed tomography of lumbar spine in subjects with fragility vertebral fractures. J Endocrinol Invest 45(10):2007–2017. https://doi.org/10.1007/s40618-022-01837-z
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures, they are data. Radiology 278(2):563–577. https://doi.org/10.1148/radiol.2015151169
Lambin P, Leijenaar RTH, Deist TM, Peerlings J, de Jong EEC, van Timmeren J, Sanduleanu S, Larue R, Even AJG, Jochems A, van Wijk Y, Woodruff H, van Soest J, Lustberg T, Roelofs E, van Elmpt W, Dekker A, Mottaghy FM, Wildberger JE, Walsh S (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14(12):749–762. https://doi.org/10.1038/nrclinonc.2017.141
Di Paola M, Gatti D, Viapiana O, Cianferotti L, Cavalli L, Caffarelli C, Conversano F, Quarta E, Pisani P, Girasole G, Giusti A, Manfredini M, Arioli G, Matucci-Cerinic M, Bianchi G, Nuti R, Gonnelli S, Brandi ML, Muratore M, Rossini M (2019) Radiofrequency echographic multispectrometry compared with dual X-ray absorptiometry for osteoporosis diagnosis on lumbar spine and femoral neck. Osteoporos Int 30(2):391–402. https://doi.org/10.1007/s00198-018-4686-3
Ciardo D, Pisani P, Conversano F, Casciaro S (2022) Pulse-echo measurements of bone tissues. Techniques and clinical results at the spine and femur. Adv Exp Med Biol 1364:145–162. https://doi.org/10.1007/978-3-030-91979-5_7
Diez-Perez A, Bouxsein ML, Eriksen EF, Khosla S, Nyman JS, Papapoulos S, Tang SY (2016) Technical note: recommendations for a standard procedure to assess cortical bone at the tissue-level in vivo using impact microindentation. Bone Rep 5:181–185. https://doi.org/10.1016/j.bonr.2016.07.004
Fuleihan GE-HCM, Cipriani C, Eastell R, Karonova T, Liu J-M, Minisola S, Mithal A, Moreira CA, Peacock M, Schini M, Silva B, Walker M, Zein OE, Marcocci C (2022) Classical and non-classical manifestations of primary hyperparathyroidism. J Bone Miner Res. https://doi.org/10.1002/jbmr.4679
Castellano EAR, Boriano A, Borretta V, Gennaro M, Latina A, Borretta G (2020) Radiologic manifestation of bone involvement in primary hyperparathyroidism: prevalence and clinical significance in a southern European series. End Pract 26(9):983–989. https://doi.org/10.4158/EP-2020-0095
Silverberg SJ, Shane E, de la Cruz L, Dempster DW, Feldman F, Seldin D, Jacobs TP, Siris ES, Cafferty M, Parisien MV et al (1989) Skeletal disease in primary hyperparathyroidism. J Bone Miner Res 4(3):283–291. https://doi.org/10.1002/jbmr.5650040302
Wishart JHM, Need A, Nordin BE (1990) Relationship between forearm and vertebral mineral density in postmenopausal women with primary hyperparathyroidism. Arch Intern Med 150(6):1329–1331
Parisien M, Silverberg SJ, Shane E, de la Cruz L, Lindsay R, Bilezikian JP, Dempster DW (1990) The histomorphometry of bone in primary hyperparathyroidism: preservation of cancellous bone structure. J Clin Endocrinol Metab 70(4):930–938. https://doi.org/10.1210/jcem-70-4-930
Vignali E, Viccica G, Diacinti D, Cetani F, Cianferotti L, Ambrogini E, Banti C, Del Fiacco R, Bilezikian JP, Pinchera A, Marcocci C (2009) Morphometric vertebral fractures in postmenopausal women with primary hyperparathyroidism. J Clin Endocrinol Metab 94(7):2306–2312. https://doi.org/10.1210/jc.2008-2006
Khosla S, Melton LJ 3rd, Wermers RA, Crowson CS, O’Fallon W, Riggs B (1999) Primary hyperparathyroidism and the risk of fracture: a population-based study. J Bone Miner Res 14(10):1700–1707. https://doi.org/10.1359/jbmr.1999.14.10.1700
Ejlsmark-Svensson H, Bislev LS, Lajlev S, Harslof T, Rolighed L, Sikjaer T, Rejnmark L (2018) Prevalence and risk of vertebral fractures in primary hyperparathyroidism: a nested case-control study. J Bone Miner Res 33(9):1657–1664. https://doi.org/10.1002/jbmr.3461
Charopoulos ITS, Trovas G, Raptou P, Kaldrymides P, Skarandavos G, Katsalira K, Lyritis GP (2006) Effect of primary hyperparathyroidism on volumetric bone mineral density and bone geometry assessed by peripheral quantitative computed tomography in postmenopausal women. J Clin Endocrinol Metab 91(5):1748–1753. https://doi.org/10.1210/jc.2005-2102
Stein EMSB, Boutroy S, Zhou B, Wang J, Udesky J, Zhang C, McMahon DJ, Romano M, Dworakowski E, Costa AG, Cusano N, Irani D, Cremers S, Shane E, Guo XE, Bilezikian JP (2013) Primary hyperparathyroidism is associated with abnormal cortical and trabecular microstructure and reduced bone stiffness in postmenopausal women. J Bone Miner Res 28(5):1029–1040
Vu TDTWX, Wang Q, Cusano NE, Irani D, Silva BC, Ghasem-Zadeh A, Udesky J, Romano ME, Zebaze R, Jerums G, Boutroy S, Bilezikian JP, Seeman E (2013) New insights into the effects of primary hyperparathyroidism on the cortical and trabecular compartments of bone. Bone 55(1):57–63. https://doi.org/10.1016/j.bone.2013.03.009
Silva BC, Boutroy S, Zhang C, McMahon DJ, Zhou B, Wang J, Udesky J, Cremers S, Sarquis M, Guo XD, Hans D, Bilezikian JP (2013) Trabecular bone score (TBS)–a novel method to evaluate bone microarchitectural texture in patients with primary hyperparathyroidism. J Clin Endocrinol Metab 98(5):1963–1970. https://doi.org/10.1210/jc.2012-4255
Jones ARSK, Harvey S, Grill V (2022) Bone mineral density compared to trabecular bone score in primary hyperparathyroidism. J Clin Med 11(2):330. https://doi.org/10.3390/jcm11020330
Romagnoli E, Cipriani C, Nofroni I, Castro C, Angelozzi M, Scarpiello A, Pepe J, Diacinti D, Piemonte S, Carnevale V, Minisola S (2013) “Trabecular Bone Score” (TBS): an indirect measure of bone micro-architecture in postmenopausal patients with primary hyperparathyroidism. Bone 53(1):154–159. https://doi.org/10.1016/j.bone.2012.11.041
Eller-Vainicher C, Battista C, Guarnieri V, Muscarella S, Palmieri S, Salcuni AS, Guglielmi G, Corbetta S, Minisola S, Spada A, Hendy GN, Cole DE, Chiodini I, Scillitani A (2014) Factors associated with vertebral fracture risk in patients with primary hyperparathyroidism. Eur J Endocrinol 171(3):399–406. https://doi.org/10.1530/EJE-14-0343
Tabacco G, Naciu AM, Messina C, Sanson G, Rinaudo L, Cesareo R, Falcone S, Manfrini S, Napoli N, Bilezikian JP, Ulivieri FM, Palermo A (2021) DXA-based bone strain index: a new tool to evaluate bone quality in primary hyperparathyroidism. J Clin Endocrinol Metab 106(8):2304–2312. https://doi.org/10.1210/clinem/dgab317
Anagnostis P, Vaitsi K, Veneti S, Potoupni V, Kenanidis E, Tsiridis E, Papavramidis TS, Goulis DG (2021) Efficacy of parathyroidectomy compared with active surveillance in patients with mild asymptomatic primary hyperparathyroidism: a systematic review and meta-analysis of randomized-controlled studies. J Endocrinol Invest 44(6):1127–1137. https://doi.org/10.1007/s40618-020-01447-7
Rolighed LRL, Sikjaer T, Heickendorff L, Vestergaard P, Mosekilde L, Christiansen P (2014) Vitamin D treatment in primary hyperparathyroidism: a randomized placebo controlled trial. J Clin Endocrinol Metab 99(3):1072–1080. https://doi.org/10.1210/jc.2013-3978
Cipriani C, Abraham A, Silva BC, Cusano NE, Rubin MR, McMahon DJ, Zhang C, Hans D, Silverberg SJ, Bilezikian JP (2017) Skeletal changes after restoration of the euparathyroid state in patients with hypoparathyroidism and primary hyperparathyroidism. Endocrine 55(2):591–598. https://doi.org/10.1007/s12020-016-1101-8
Miguel GACF, Rodríguez JCR, Ramos MA, Pablos DL, Herrero EF, Díaz-Guerra GM (2019) Trabecular Bone Score, bone mineral density and bone markers in patients with primary hyperparathyroidism 2 years after parathyroidectomy. Horm Metab Res 51(3):186–190. https://doi.org/10.1055/a-0850-8679
Cusano NE, Rubin MR, Silva BC, Tay YD, Williams JM, Agarwal S, Omeragic B, Guo XE, Bilezikian JP (2018) Skeletal microstructure and estimated bone strength improve following parathyroidectomy in primary hyperparathyroidism. J Clin Endocrinol Metab 103(1):196–205. https://doi.org/10.1210/jc.2017-01932
Hansen S, Hauge EM, Rasmussen L, Jensen JE, Brixen K (2012) Parathyroidectomy improves bone geometry and microarchitecture in female patients with primary hyperparathyroidism: a one-year prospective controlled study using high-resolution peripheral quantitative computed tomography. J Bone Miner Res 27(5):1150–1158. https://doi.org/10.1002/jbmr.1540
Ye Z, Silverberg SJ, Sreekanta A, Tong K, Wang Y, Chang Y, Zhang M, Guyatt G, Tangamornsuksun W, Zhang Y, Manja V, Bakaa L, Couban RJ, Brandi ML, Clarke B, Khan AA, Mannstadt M, Bilezikian JP (2022) The efficacy and safety of medical and surgical therapy in patients with primary hyperparathyroidism: a systematic review and meta-analysis of randomized controlled trials. J Bone Miner Res 37(11):2351–2372. https://doi.org/10.1002/jbmr.4685
Hamdy NAT, Decallonne B, Evenepoel P, Gruson D, van Vlokhoven-Verhaegh L (2021) Burden of illness in patients with chronic hypoparathyroidism not adequately controlled with conventional therapy: a Belgium and the Netherlands survey. J Endocrinol Invest 44(7):1437–1446. https://doi.org/10.1007/s40618-020-01442-y
Bilezikian JP (2020) Hypoparathyroidism. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgaa113
Rubin MR, Dempster DW, Zhou H, Shane E, Nickolas T, Sliney J Jr, Silverberg SJ, Bilezikian JP (2008) Dynamic and structural properties of the skeleton in hypoparathyroidism. J Bone Miner Res 23(12):2018–2024. https://doi.org/10.1359/jbmr.080803
Rubin MR, Dempster DW, Sliney J Jr, Zhou H, Nickolas TL, Stein EM, Dworakowski E, Dellabadia M, Ives R, McMahon DJ, Zhang C, Silverberg SJ, Shane E, Cremers S, Bilezikian JP (2011) PTH(1–84) administration reverses abnormal bone-remodeling dynamics and structure in hypoparathyroidism. J Bone Miner Res 26(11):2727–2736. https://doi.org/10.1002/jbmr.452
Rubin MR (2019) Skeletal manifestations of hypoparathyroidism. Bone 120:548–555. https://doi.org/10.1016/j.bone.2018.11.012
Silva BCBJ (2020) Skeletal abnormalities in hypoparathyroidism and in primary hyperparathyroidism. Rev Endocr Metab Disord. https://doi.org/10.1007/s11154-020-09614-0
Cipriani C, Pepe J, Silva BC, Rubin MR, Cusano NE, McMahon DJ, Nieddu L, Angelozzi M, Biamonte F, Diacinti D, Hans D, Minisola S, Bilezikian JP (2018) Comparative effect of rhPTH(1–84) on bone mineral density and Trabecular Bone Score in hypoparathyroidism and postmenopausal osteoporosis. J Bone Miner Res 33(12):2132–2139. https://doi.org/10.1002/jbmr.3554
Sakane ENVM, Lazaretti-Castro M, Maeda SS (2019) Predictors of poor bone microarchitecture assessed by Trabecular Bone Score in postsurgical hypoparathyroidism. J Clin Endocrinol Metab 104:5795–5803. https://doi.org/10.1210/jc.2019-00698
Saha SMV, Kandasamy D, Sreenivas V, Goswami R (2022) Vertebral fractures, trabecular bone score and their determinants in chronic hypoparathyroidism. J Endocrinol Invest. https://doi.org/10.1007/s40618-022-01818-2
Cipriani CMS, Bilezikian JP, Diacinti D, Colangelo L, Piazzolla V, Angelozzi M, Nieddu L, Pepe J, Diacinti D (2021) Vertebral fracture assessment in postmenopausal women with postsurgical hypoparathyroidism. J Clin Endocrinol Metab 106:1303–1311. https://doi.org/10.1210/clinem/dgab076
Chen QKH, Iu M-F, Nomura R, Sowa H, Yamauchi M, Tsukamoto T, Sugimoto T, Chihara K (2003) Effects of an excess and a deficiency of endogenous parathyroid hormone on volumetric bone mineral density and bone geometry determined by peripheral quantitative computed tomography in female subjects. J Clin Endocrinol Metab 88:4655–4658. https://doi.org/10.1210/jc.2003-030470
Liu JCS, Quan T, Wang Y, Wang O, Nie M, Jiang Y, Li M, Xing X, Xia W (2020) Bone microstructure of adult patients with non-surgical hypoparathyroidism assessed by high-resolution peripheral quantitative computed tomography. Osteop Int 31:2219–2230. https://doi.org/10.1007/s00198-020-05506-w
Cusano NE, Nishiyama KK, Zhang C, Rubin MR, Boutroy S, McMahon DJ, Guo XE, Bilezikian JP (2016) Noninvasive assessment of skeletal microstructure and estimated bone strength in hypoparathyroidism. J Bone Miner Res 31(2):308–316. https://doi.org/10.1002/jbmr.2609
Cusano NE, Rubin MR, Williams JM, Agarwal S, Tabacco G, Tay D, Majeed R, Omeragic B, Bilezikian JP (2020) Changes in skeletal microstructure through four continuous years of rhPTH(1–84) therapy in hypoparathyroidism. J Bone Miner Res. https://doi.org/10.1002/jbmr.4005
Agarwal S, McMahon DJ, Chen J, Brossfield A, Fernando J, Bilezikian JP, Cusano NE, Rubin MR (2023) The clinical and skeletal effects of long-term therapy of hypoparathyroidism with rhPTH(1–84). J Bone Miner Res. https://doi.org/10.1002/jbmr.4780
Williams GR, Bassett JHD (2018) Thyroid diseases and bone health. J Endocrinol Invest 41(1):99–109. https://doi.org/10.1007/s40618-017-0753-4
Mazziotti G, Frara S, Giustina A (2018) Pituitary diseases and bone. Endocr Rev 39(4):440–488. https://doi.org/10.1210/er.2018-00005
Kim SM, Ryu V, Miyashita S, Korkmaz F, Lizneva D, Gera S, Latif R, Davies TF, Iqbal J, Yuen T, Zaidi M (2021) Thyrotropin, hyperthyroidism, and bone mass. J Clin Endocrinol Metab 106(12):e4809–e4821. https://doi.org/10.1210/clinem/dgab548
Apostu D, Lucaciu O, Oltean-Dan D, Muresan AD, Moisescu-Pop C, Maxim A, Benea H (2020) the influence of thyroid pathology on osteoporosis and fracture risk: a review. Diagnostics (Basel). https://doi.org/10.3390/diagnostics10030149
Vestergaard P, Mosekilde L (2003) Hyperthyroidism, bone mineral, and fracture risk–a meta-analysis. Thyroid 13(6):585–593. https://doi.org/10.1089/105072503322238854
Ock SY, Chung YS, Choi YJ (2016) Changes in bone mineral density and trabecular bone score in Graves’ disease patients after anti-thyroid therapy. Osteoporos Sarcopenia 2(3):175–179. https://doi.org/10.1016/j.afos.2016.05.004
Nicolaisen P, Obling ML, Winther KH, Hansen S, Hermann AP, Hegedus L, Bonnema SJ, Brix TH (2021) Consequences of hyperthyroidism and its treatment for bone microarchitecture assessed by high-resolution peripheral quantitative computed tomography. Thyroid 31(2):208–216. https://doi.org/10.1089/thy.2020.0084
Eriksen EF, Charles P, Melsen F, Mosekilde L, Risteli L, Risteli J (1993) Serum markers of type I collagen formation and degradation in metabolic bone disease: correlation with bone histomorphometry. J Bone Miner Res 8(2):127–132. https://doi.org/10.1002/jbmr.5650080202
Cellini M, Rotondi M, Tanda ML, Piantanida E, Chiovato L, Beck-Peccoz P, Lania A, Mazziotti G (2021) Skeletal health in patients with differentiated thyroid carcinoma. J Endocrinol Invest 44(3):431–442. https://doi.org/10.1007/s40618-020-01359-6
Blum MR, Bauer DC, Collet TH, Fink HA, Cappola AR, da Costa BR, Wirth CD, Peeters RP, Åsvold BO, den Elzen WP, Luben RN, Imaizumi M, Bremner AP, Gogakos A, Eastell R, Kearney PM, Strotmeyer ES, Wallace ER, Hoff M, Ceresini G, Rivadeneira F, Uitterlinden AG, Stott DJ, Westendorp RG, Khaw KT, Langhammer A, Ferrucci L, Gussekloo J, Williams GR, Walsh JP, Jüni P, Aujesky D, Rodondi N, Thyroid Studies Collaboration (2015) Subclinical thyroid dysfunction and fracture risk: a meta-analysis. JAMA 313(20):2055–2065. https://doi.org/10.1001/jama.2015.5161
Chung CW, Choi HS, Kong SH, Park YJ, Park DJ, Ahn HY, Cho SW (2021) Measurements of bone health after thyroid-stimulating suppression therapy in postmenopausal women with differentiated thyroid carcinoma: bone mineral density versus the trabecular bone score. J Clin Med. https://doi.org/10.3390/jcm10091964
Sousa B, Silva BC, de Oliveira GT, Pires MC, Soares MMS, Kakehasi AM (2021) Trabecular bone score in women with differentiated thyroid cancer on long-term TSH-suppressive therapy. J Endocrinol Invest 44(10):2295–2305. https://doi.org/10.1007/s40618-021-01537-0
Mendonca Monteiro de Barros G, Madeira M, Vieira-Neto L, de Paula Paranhos Neto F, Carvalho Mendonca LM, Correa Barbosa Lima I, Corbo R, Fleiuss Farias ML (2016) Bone mineral density and bone microarchitecture after long-term suppressive levothyroxine treatment of differentiated thyroid carcinoma in young adult patients. J Bone Miner Metab 34(4):417–421. https://doi.org/10.1007/s00774-015-0680-4
Moon JH, Kim KM, Oh TJ, Choi SH, Lim S, Park YJ, Park DJ, Jang HC (2017) The effect of TSH suppression on vertebral trabecular bone scores in patients with differentiated thyroid carcinoma. J Clin Endocrinol Metab 102(1):78–85. https://doi.org/10.1210/jc.2016-2740
De Mingo Dominguez ML, Guadalix Iglesias S, Martin-Arriscado Arroba C, Lopez Alvarez B, Martinez Diaz-Guerra G, Martinez-Pueyo JI, Ferrero Herrero E, Hawkins Carranza F (2018) Low trabecular bone score in postmenopausal women with differentiated thyroid carcinoma after long-term TSH suppressive therapy. Endocrine 62(1):166–173. https://doi.org/10.1007/s12020-018-1671-8
Mazziotti G, Formenti AM, Frara S, Olivetti R, Banfi G, Memo M, Maroldi R, Giubbini R, Giustina A (2018) High prevalence of radiological vertebral fractures in women on thyroid-stimulating hormone-suppressive therapy for thyroid carcinoma. J Clin Endocrinol Metab 103(3):956–964. https://doi.org/10.1210/jc.2017-01986
Hawkins Carranza F, Guadalix Iglesias S, De Mingo L, Dominguez M, Martin-Arriscado Arroba C, Lopez Alvarez B, Allo Miguel G, Martinez Diaz-Guerra G (2020) Trabecular bone deterioration in differentiated thyroid cancer: impact of long-term TSH suppressive therapy. Cancer Med 9(16):5746–5755. https://doi.org/10.1002/cam4.3200
Kim K, Kim IJ, Pak K, Kim SJ, Shin S, Kim BH, Kim SS, Lee BJ, Jeon YK (2018) Evaluation of bone mineral density using DXA and cQCT in postmenopausal patients under thyrotropin suppressive therapy. J Clin Endocrinol Metab 103(11):4232–4240. https://doi.org/10.1210/jc.2017-02704
Tournis S, Antoniou JD, Liakou CG, Christodoulou J, Papakitsou E, Galanos A, Makris K, Marketos H, Nikopoulou S, Tzavara I, Triantafyllopoulos IK, Dontas I, Papaioannou N, Lyritis GP, Alevizaki M (2015) Volumetric bone mineral density and bone geometry assessed by peripheral quantitative computed tomography in women with differentiated thyroid cancer under TSH suppression. Clin Endocrinol (Oxf) 82(2):197–204. https://doi.org/10.1111/cen.12560
Vendrami C, Marques-Vidal P, Gonzalez Rodriguez E, Hans D, Waeber G, Lamy O (2022) Thyroid-stimulating hormone is associated with trabecular bone score and 5-year incident fracture risk in euthyroid postmenopausal women: the OsteoLaus cohort. Osteoporos Int 33(1):195–204. https://doi.org/10.1007/s00198-021-06081-4
Hwangbo Y, Kim JH, Kim SW, Park YJ, Park DJ, Kim SY, Shin CS, Cho NH (2016) High-normal free thyroxine levels are associated with low trabecular bone scores in euthyroid postmenopausal women. Osteoporos Int 27(2):457–462. https://doi.org/10.1007/s00198-015-3270-3
Gonzalez Rodriguez E, Stuber M, Del Giovane C, Feller M, Collet TH, Lowe AL, Blum MR, van Vliet NA, van Heemst D, Kearney PM, Gussekloo J, Mooijaart S, Westendorp RGJ, Stott DJ, Aeberli D, Bauer DC, Hans D, Rodondi N (2020) Skeletal effects of levothyroxine for subclinical hypothyroidism in older adults: a TRUST randomized trial nested study. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgz058
Obling ML, Nicolaisen P, Brix TH, Winther KH, Hansen S, Hegedus L, Hermann AP, Bonnema SJ (2021) Restoration of euthyroidism in women with Hashimoto’s thyroiditis changes bone microarchitecture but not estimated bone strength. Endocrine 71(2):397–406. https://doi.org/10.1007/s12020-020-02398-y
Chiodini I, Carnevale V, Torlontano M, Fusilli S, Guglielmi G, Pileri M, Modoni S, Di Giorgio A, Liuzzi A, Minisola S, Cammisa M, Trischitta V, Scillitani A (1998) Alterations of bone turnover and bone mass at different skeletal sites due to pure glucocorticoid excess: study in eumenorrheic patients with Cushing’s syndrome. J Clin Endocrinol Metab 83(6):1863–1867. https://doi.org/10.1210/jcem.83.6.4880
Moraes AB, de Paula MP, de Paula Paranhos-Neto F, Cavalari EMR, de Morais FFC, Curi DSC, Lima LFC, de Mendonça LMC, Farias MLF, Madeira M, Vieira Neto L (2020) Bone evaluation by high-resolution peripheral quantitative computed tomography in patients with adrenal incidentaloma. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgaa263
Di Somma C, Pivonello R, Loche S, Faggiano A, Klain M, Salvatore M, Lombardi G, Colao A (2003) Effect of 2 years of cortisol normalization on the impaired bone mass and turnover in adolescent and adult patients with Cushing’s disease: a prospective study. Clin Endocrinol (Oxf) 58(3):302–308
Vinolas H, Grouthier V, Mehsen-Cetre N, Boisson A, Winzenrieth R, Schaeverbeke T, Mesguich C, Bordenave L, Tabarin A (2018) Assessment of vertebral microarchitecture in overt and mild Cushing’s syndrome using trabecular bone score. Clin Endocrinol (Oxf) 89(2):148–154. https://doi.org/10.1111/cen.13743
Ferraù F, Giovinazzo S, Messina E, Tessitore A, Vinci S, Mazziotti G, Lania A, Granata F, Cannavò S (2020) High bone marrow fat in patients with Cushing’s syndrome and vertebral fractures. Endocrine 67(1):172–179. https://doi.org/10.1007/s12020-019-02034-4
Maurice F, Dutour A, Vincentelli C, Abdesselam I, Bernard M, Dufour H, Lefur Y, Graillon T, Kober F, Cristofari P, Jouve E, Pini L, Fernandez R, Chagnaud C, Brue T, Castinetti F, Gaborit B (2018) Active Cushing syndrome patients have increased ectopic fat deposition and bone marrow fat content compared to cured patients and healthy subjects: a pilot 1H-MRS study. Eur J Endocrinol 179(5):307–317. https://doi.org/10.1530/eje-18-0318
dos Santos CV, Vieira Neto L, Madeira M, Alves Coelho MC, de Mendonca LM, Paranhos-Neto Fde P, Lima IC, Gadelha MR, Farias ML (2015) Bone density and microarchitecture in endogenous hypercortisolism. Clin Endocrinol (Oxf) 83(4):468–474. https://doi.org/10.1111/cen.12812
Apaydın T, Yavuz DG (2021) Assessment of non-traumatic vertebral fractures in Cushing’s syndrome patients. J Endocrinol Invest 44(8):1767–1773. https://doi.org/10.1007/s40618-020-01496-y
Valassi E, Santos A, Yaneva M, Toth M, Strasburger CJ, Chanson P, Wass JA, Chabre O, Pfeifer M, Feelders RA, Tsagarakis S, Trainer PJ, Franz H, Zopf K, Zacharieva S, Lamberts SW, Tabarin A, Webb SM (2011) The European Registry on Cushing’s syndrome: 2-year experience. Baseline demographic and clinical characteristics. Eur J Endocrinol 165(3):383–392. https://doi.org/10.1530/eje-11-0272
Tauchmanova L, Pivonello R, Di Somma C, Rossi R, De Martino MC, Camera L, Klain M, Salvatore M, Lombardi G, Colao A (2006) Bone demineralization and vertebral fractures in endogenous cortisol excess: role of disease etiology and gonadal status. J Clin Endocrinol Metab 91(5):1779–1784. https://doi.org/10.1210/jc.2005-0582
Trementino L, Appolloni G, Ceccoli L, Marcelli G, Concettoni C, Boscaro M, Arnaldi G (2014) Bone complications in patients with Cushing’s syndrome: looking for clinical, biochemical, and genetic determinants. Osteoporos Int 25(3):913–921. https://doi.org/10.1007/s00198-013-2520-5
Belaya ZE, Hans D, Rozhinskaya LY, Dragunova NV, Sasonova NI, Solodovnikov AG, Tsoriev TT, Dzeranova LK, Melnichenko GA, Dedov II (2015) The risk factors for fractures and trabecular bone-score value in patients with endogenous Cushing’s syndrome. Arch Osteoporos 10:44. https://doi.org/10.1007/s11657-015-0244-1
Eller-Vainicher C, Morelli V, Ulivieri FM, Palmieri S, Zhukouskaya VV, Cairoli E, Pino R, Naccarato A, Scillitani A, Beck-Peccoz P, Chiodini I (2012) Bone quality, as measured by trabecular bone score in patients with adrenal incidentalomas with and without subclinical hypercortisolism. J Bone Miner Res 27(10):2223–2230. https://doi.org/10.1002/jbmr.1648
Vestergaard P, Lindholm J, Jorgensen JO, Hagen C, Hoeck HC, Laurberg P, Rejnmark L, Brixen K, Kristensen LO, Feldt-Rasmussen U, Mosekilde L (2002) Increased risk of osteoporotic fractures in patients with Cushing’s syndrome. Eur J Endocrinol 146(1):51–56
Scillitani A, Mazziotti G, Di Somma C, Moretti S, Stigliano A, Pivonello R, Giustina A, Colao A (2014) Treatment of skeletal impairment in patients with endogenous hypercortisolism: when and how? Osteoporos Int 25(2):441–446. https://doi.org/10.1007/s00198-013-2588-y
Mazziotti G, Lania AG, Canalis E (2022) Skeletal disorders associated with the growth hormone–insulin- like growth factor 1 axis. Nat Rev Endocrinol 18(6):353–365. https://doi.org/10.1038/s41574-022-00649-8
Wuster C, Abs R, Bengtsson BA, Bennmarker H, Feldt-Rasmussen U, Hernberg-Stahl E, Monson JP, Westberg B, Wilton P (2001) The influence of growth hormone deficiency, growth hormone replacement therapy, and other aspects of hypopituitarism on fracture rate and bone mineral density. J Bone Miner Res 16(2):398–405. https://doi.org/10.1359/jbmr.2001.16.2.398
Canalis E, Mazziotti G, Giustina A, Bilezikian JP (2007) Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int 18(10):1319–1328. https://doi.org/10.1007/s00198-007-0394-0
Ardawi MS, Akhbar DH, Alshaikh A, Ahmed MM, Qari MH, Rouzi AA, Ali AY, Abdulrafee AA, Saeda MY (2013) Increased serum sclerostin and decreased serum IGF-1 are associated with vertebral fractures among postmenopausal women with type-2 diabetes. Bone 56(2):355–362. https://doi.org/10.1016/j.bone.2013.06.029
Colao A, Di Somma C, Pivonello R, Loche S, Aimaretti G, Cerbone G, Faggiano A, Corneli G, Ghigo E, Lombardi G (1999) Bone loss is correlated to the severity of growth hormone deficiency in adult patients with hypopituitarism. J Clin Endocrinol Metab 84(6):1919–1924. https://doi.org/10.1210/jcem.84.6.5742
Mazziotti G, Bianchi A, Bonadonna S, Nuzzo M, Cimino V, Fusco A, De Marinis L, Giustina A (2006) Increased prevalence of radiological spinal deformities in adult patients with GH deficiency: influence of GH replacement therapy. J Bone Miner Res 21(4):520–528. https://doi.org/10.1359/jbmr.060112
Bravenboer N, Holzmann P, de Boer H, Blok GJ, Lips P (1996) Histomorphometric analysis of bone mass and bone metabolism in growth hormone deficient adult men. Bone 18(6):551–557
Yang H, Yan K, Yuping X, Zhang Q, Wang L, Gong F, Zhu H, Xia W, Pan H (2019) Bone microarchitecture and volumetric bone density impairment in young male adults with childhood-onset growth hormone deficiency. Eur J Endocrinol 180(2):145–153. https://doi.org/10.1530/eje-18-0711
Gracia-Marco L, Gonzalez-Salvatierra S, Garcia-Martin A, Ubago-Guisado E, Garcia-Fontana B, Gil-Cosano JJ, Muñoz-Torres M (2021) 3D DXA hip differences in patients with acromegaly or adult growth hormone deficiency. J Clin Med. https://doi.org/10.3390/jcm10040657
Silva PPB, Amlashi FG, Yu EW, Pulaski-Liebert KJ, Gerweck AV, Fazeli PK, Lawson E, Nachtigall LB, Biller BMK, Miller KK, Klibanski A, Bouxsein M, Tritos NA (2017) Bone microarchitecture and estimated bone strength in men with active acromegaly. Eur J Endocrinol 177(5):409–420. https://doi.org/10.1530/eje-17-0468
Biller BM, Sesmilo G, Baum HB, Hayden D, Schoenfeld D, Klibanski A (2000) Withdrawal of long-term physiological growth hormone (GH) administration: differential effects on bone density and body composition in men with adult-onset GH deficiency. J Clin Endocrinol Metab 85(3):970–976. https://doi.org/10.1210/jcem.85.3.6474
Mazziotti G, Biagioli E, Maffezzoni F, Spinello M, Serra V, Maroldi R, Floriani I, Giustina A (2015) Bone turnover, bone mineral density, and fracture risk in acromegaly: a meta-analysis. J Clin Endocrinol Metab 100(2):384–394. https://doi.org/10.1210/jc.2014-2937
Madeira M, Neto LV, de Paula-Paranhos-Neto F, Barbosa-Lima IC, Carvalho-de-Mendonca LM, Gadelha MR, Fleiuss-de-Farias ML (2013) Acromegaly has a negative influence on trabecular bone, but not on cortical bone, as assessed by high-resolution peripheral quantitative computed tomography. J Clin Endocrinol Metab 98(4):1734–1741. https://doi.org/10.1210/jc.2012-4073
Dalle Carbonare L, Micheletti V, Cosaro E, Valenti MT, Mottes M, Francia G, Davì MV (2018) Bone histomorphometry in acromegaly patients with fragility vertebral fractures. Pituitary 21(1):56–64. https://doi.org/10.1007/s11102-017-0847-1
Ueland T, Ebbesen EN, Thomsen JS, Mosekilde L, Brixen K, Flyvbjerg A, Bollerslev J (2002) Decreased trabecular bone biomechanical competence, apparent density, IGF-II and IGFBP-5 content in acromegaly. Eur J Clin Invest 32(2):122–128
Silva PPB, Pereira RMR, Takayama L, Borba CG, Duarte FH, Trarbach EB, Martin RM, Bronstein MD, Tritos NA, Jallad RS (2021) Impaired bone microarchitecture in premenopausal women with acromegaly: the possible role of Wnt signaling. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgab260
Kuzma M, Vanuga P, Sagova I, Pavai D, Jackuliak P, Killinger Z, Binkley NC, Winzenrieth R, Genant HK, Payer J (2019) Non-invasive DXA-derived bone structure assessment of acromegaly patients: a cross-sectional study. Eur J Endocrinol 180(3):201–211. https://doi.org/10.1530/eje-18-0881
Sala E, Malchiodi E, Carosi G, Verrua E, Cairoli E, Ferrante E, Filopanti M, Eller-Vainicher C, Ulivieri FM, Spada A, Arosio M, Chiodini I, Mantovani G (2021) Spine bone texture assessed by trabecular bone score in active and controlled acromegaly: a prospective study. J Endocr Soc 5(8):bvab090. https://doi.org/10.1210/jendso/bvab090
Sorohan MC, Dusceac R, Sorohan BM, Caragheorgheopol A, Poiana C (2021) Trabecular bone score and bone mineral density in acromegalic osteopathy assessment: a cross-sectional study. Arch Osteoporos 16(1):134. https://doi.org/10.1007/s11657-021-00986-7
Bioletto F, Barale M, Prencipe N, Berton AM, Parasiliti-Caprino M, Gasco V, Ghigo E, Procopio M, Grottoli S (2022) Trabecular bone score as an index of bone fragility in patients with acromegaly: a systematic review and meta-analysis. Neuroendocrinology. https://doi.org/10.1159/000528199
Maffezzoni F, Maddalo M, Frara S, Mezzone M, Zorza I, Baruffaldi F, Doglietto F, Mazziotti G, Maroldi R, Giustina A (2016) High-resolution-cone beam tomography analysis of bone microarchitecture in patients with acromegaly and radiological vertebral fractures. Endocrine 54(2):532–542. https://doi.org/10.1007/s12020-016-1078-3
Malgo F, Hamdy NA, Rabelink TJ, Kroon HM, Claessen KM, Pereira AM, Biermasz NR, Appelman-Dijkstra NM (2017) Bone material strength index as measured by impact microindentation is altered in patients with acromegaly. Eur J Endocrinol 176(3):339–347. https://doi.org/10.1530/eje-16-0808
Wassenaar MJ, Biermasz NR, Hamdy NA, Zillikens MC, van Meurs JB, Rivadeneira F, Hofman A, Uitterlinden AG, Stokkel MP, Roelfsema F, Kloppenburg M, Kroon HM, Romijn JA, Pereira AM (2011) High prevalence of vertebral fractures despite normal bone mineral density in patients with long-term controlled acromegaly. Eur J Endocrinol 164(4):475–483. https://doi.org/10.1530/eje-10-1005
Calatayud M, Pérez-Olivares Martín L, Librizzi MS, Lora Pablos D, González Méndez V, Aramendi Ramos M, Martínez Diaz-Guerra G, Hawkins F (2021) Trabecular bone score and bone mineral density in patients with long-term controlled acromegaly. Clin Endocrinol (Oxf). https://doi.org/10.1111/cen.14439
Godang K, Lekva T, Normann KR, Olarescu NC, Oystese KAB, Kolnes A, Ueland T, Bollerslev J, Heck A (2019) Hip structure analyses in acromegaly: decrease of cortical bone thickness after treatment: a longitudinal cohort study. JBMR Plus 3(12):e10240. https://doi.org/10.1002/jbm4.10240
Chiloiro S, Giampietro A, Frara S, Bima C, Donfrancesco F, Fleseriu CM, Pontecorvi A, Giustina A, Fleseriu M, De Marinis L, Bianchi A (2020) Effects of pegvisomant and pasireotide LAR on vertebral fractures in acromegaly resistant to first-generation SRLs. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgz054
Mazziotti G, Battista C, Maffezzoni F, Chiloiro S, Ferrante E, Prencipe N, Grasso L, Gatto F, Olivetti R, Arosio M, Barale M, Bianchi A, Cellini M, Chiodini I, De Marinis L, Del Sindaco G, Di Somma C, Ferlin A, Ghigo E, Giampietro A, Grottoli S, Lavezzi E, Mantovani G, Morenghi E, Pivonello R, Porcelli T, Procopio M, Pugliese F, Scillitani A, Lania AG (2020) Treatment of acromegalic osteopathy in real-life clinical practice: the BAAC (Bone Active Drugs in Acromegaly) study. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgaa363
Vitali E, Palagano E, Schiavone ML, Mantovani G, Sobacchi C, Mazziotti G, Lania A (2022) Direct effects of octreotide on osteoblast cell proliferation and function. J Endocrinol Invest 45(5):1045–1057. https://doi.org/10.1007/s40618-022-01740-7
Pelsma ICM, Biermasz NR, Pereira AM, van Furth WR, Appelman-Dijkstra NM, Kloppenburg M, Kroon HM, Claessen K (2020) Progression of vertebral fractures in long-term controlled acromegaly: a 9-year follow-up study. Eur J Endocrinol 183(4):427–437. https://doi.org/10.1530/eje-20-0415
Rolla M, Halupczok-Żyła J, Jawiarczyk-Przybyłowska A, Bolanowski M (2020) Bone densitometry by radiofrequency echographic multi-spectrometry (REMS) in acromegaly patients. Endokrynol Pol 71(6):524–531. https://doi.org/10.5603/EP.a2020.0056
Rochira V, Antonio L, Vanderschueren D (2018) EAA clinical guideline on management of bone health in the andrological outpatient clinic. Andrology 6(2):272–285. https://doi.org/10.1111/andr.12470
Corona G, Vena W, Pizzocaro A, Giagulli VA, Francomano D, Rastrelli G, Mazziotti G, Aversa A, Isidori AM, Pivonello R, Vignozzi L, Mannucci E, Maggi M, Ferlin A (2022) Testosterone supplementation and bone parameters: a systematic review and meta-analysis study. J Endocrinol Invest 45(5):911–926. https://doi.org/10.1007/s40618-021-01702-5
Ferlin A, Selice R, Carraro U, Foresta C (2013) Testicular function and bone metabolism–beyond testosterone. Nat Rev Endocrinol 9(9):548–554. https://doi.org/10.1038/nrendo.2013.135
Almeida M, Laurent MR, Dubois V, Claessens F, O’Brien CA, Bouillon R, Vanderschueren D, Manolagas SC (2017) Estrogens and androgens in skeletal physiology and pathophysiology. Physiol Rev 97(1):135–187. https://doi.org/10.1152/physrev.00033.2015
Rochira V, Kara E, Carani C (2015) The endocrine role of estrogens on human male skeleton. Int J Endocrinol 2015:165215. https://doi.org/10.1155/2015/165215
Porcelli T, Maffezzoni F, Pezzaioli LC, Delbarba A, Cappelli C, Ferlin A (2020) Management of endocrine disease: male osteoporosis: diagnosis and management - should the treatment and the target be the same as for female osteoporosis? Eur J Endocrinol 183(3):R75-r93. https://doi.org/10.1530/eje-20-0034
Vanderschueren D, Pye SR, Venken K, Borghs H, Gaytant J, Huhtaniemi IT, Adams JE, Ward KA, Bartfai G, Casanueva FF, Finn JD, Forti G, Giwercman A, Han TS, Kula K, Labrie F, Lean ME, Pendleton N, Punab M, Silman AJ, Wu FC, O’Neill TW, Boonen S (2010) Gonadal sex steroid status and bone health in middle-aged and elderly European men. Osteoporos Int 21(8):1331–1339. https://doi.org/10.1007/s00198-009-1144-2
Greendale GA, Edelstein S, Barrett-Connor E (1997) Endogenous sex steroids and bone mineral density in older women and men: the Rancho Bernardo Study. J Bone Miner Res 12(11):1833–1843. https://doi.org/10.1359/jbmr.1997.12.11.1833
Bjørnerem A, Ahmed LA, Joakimsen RM, Berntsen GK, Fønnebø V, Jørgensen L, Øian P, Seeman E, Straume B (2007) A prospective study of sex steroids, sex hormone-binding globulin, and non-vertebral fractures in women and men: the Tromso Study. Eur J Endocrinol 157(1):119–125. https://doi.org/10.1530/eje-07-0032
Finkelstein JS, Lee H, Leder BZ, Burnett-Bowie SA, Goldstein DW, Hahn CW, Hirsch SC, Linker A, Perros N, Servais AB, Taylor AP, Webb ML, Youngner JM, Yu EW (2016) Gonadal steroid-dependent effects on bone turnover and bone mineral density in men. J Clin Invest 126(3):1114–1125. https://doi.org/10.1172/jci84137
Isidori AM, Aversa A, Calogero A, Ferlin A, Francavilla S, Lanfranco F, Pivonello R, Rochira V, Corona G, Maggi M (2022) Adult- and late-onset male hypogonadism: the clinical practice guidelines of the Italian Society of Andrology and Sexual Medicine (SIAMS) and the Italian Society of Endocrinology (SIE). J Endocrinol Invest. https://doi.org/10.1007/s40618-022-01859-7
Bhasin S, Brito JP, Cunningham GR, Hayes FJ, Hodis HN, Matsumoto AM, Snyder PJ, Swerdloff RS, Wu FC, Yialamas MA (2018) Testosterone therapy in men with hypogonadism: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 103(5):1715–1744. https://doi.org/10.1210/jc.2018-00229
Salonia A, Bettocchi C, Boeri L, Capogrosso P, Carvalho J, Cilesiz NC, Cocci A, Corona G, Dimitropoulos K, Gül M, Hatzichristodoulou G, Jones TH, Kadioglu A, Martínez Salamanca JI, Milenkovic U, Modgil V, Russo GI, Serefoglu EC, Tharakan T, Verze P, Minhas S (2021) European Association of Urology guidelines on sexual and reproductive health-2021 update: male sexual dysfunction. Eur Urol 80(3):333–357. https://doi.org/10.1016/j.eururo.2021.06.007
Corona G, Filippi S, Bianchi N, Dicuio M, Rastrelli G, Concetti S, Sforza A, Maggi M (2021) Cardiovascular risks of androgen deprivation therapy for prostate cancer. World J Mens Health 39(3):429–443. https://doi.org/10.5534/wjmh.200109
Vena W, Pizzocaro A, Indirli R, Amer M, Maffezzoni F, Delbarba A, Leonardi L, Balzarini L, Ulivieri FM, Ferlin A, Mantovani G, Lania AG, Ferrante E, Mazziotti G (2020) Prevalence and determinants of radiological vertebral fractures in patients with Klinefelter syndrome. Andrology 8(6):1699–1704. https://doi.org/10.1111/andr.12841
Mazziotti G, Vena W, Pedersini R, Piccini S, Morenghi E, Cosentini D, Zucali P, Torrisi R, Sporeni S, Simoncini EL, Maroldi R, Balzarini L, Lania AG, Berruti A (2022) Prediction of vertebral fractures in cancer patients undergoing hormone deprivation therapies: reliability of who fracture risk assessment tool (frax) and bone mineral density in real-life clinical practice. J Bone Oncol 33:1021. https://doi.org/10.1016/j.jbo.2022.100421
Benito M, Gomberg B, Wehrli FW, Weening RH, Zemel B, Wright AC, Song HK, Cucchiara A, Snyder PJ (2003) Deterioration of trabecular architecture in hypogonadal men. J Clin Endocrinol Metab 88(4):1497–1502. https://doi.org/10.1210/jc.2002-021429
Zhang XH, Liu XS, Vasilic B, Wehrli FW, Benito M, Rajapakse CS, Snyder PJ, Guo XE (2008) In vivo microMRI-based finite element and morphological analyses of tibial trabecular bone in eugonadal and hypogonadal men before and after testosterone treatment. J Bone Miner Res 23(9):1426–1434. https://doi.org/10.1359/jbmr.080405
Shanbhogue VV, Hansen S, Jørgensen NR, Brixen K, Gravholt CH (2014) Bone geometry, volumetric density, microarchitecture, and estimated bone strength assessed by HR-pQCT in Klinefelter syndrome. J Bone Miner Res 29(11):2474–2482. https://doi.org/10.1002/jbmr.2272
Piot A, Plotton I, Boutroy S, Bacchetta J, Ailloud S, Lejeune H, Chapurlat RD, Szulc P, Confavreux CB (2022) Klinefelter bone microarchitecture evolution with testosterone replacement therapy. Calcif Tissue Int 111(1):35–46. https://doi.org/10.1007/s00223-022-00956-2
Varimo T, Miettinen PJ, Laine T, Salonen P, Tenhola S, Voutilainen R, Huopio H, Hero M, Raivio T (2021) Bone structure assessed with pQCT in prepubertal males with delayed puberty or congenital hypogonadotropic hypogonadism. Clin Endocrinol (Oxf) 95(1):107–116. https://doi.org/10.1111/cen.14466
Tahani N, Nieddu L, Prossomariti G, Spaziani M, Granato S, Carlomagno F, Anzuini A, Lenzi A, Radicioni AF, Romagnoli E (2018) Long-term effect of testosterone replacement therapy on bone in hypogonadal men with Klinefelter Syndrome. Endocrine 61(2):327–335. https://doi.org/10.1007/s12020-018-1604-6
Vena W, Carrone F, Delbarba A, Akpojiyovbi O, Pezzaioli LC, Facondo P, Cappelli C, Leonardi L, Balzarini L, Farina D, Pizzocaro A, Lania AG, Mazziotti G, Ferlin A (2023) Body composition, trabecular bone score and vertebral fractures in subjects with Klinefelter syndrome. J Endocrinol Invest 46(2):297–304. https://doi.org/10.1007/s40618-022-01901-8
Ostertag A, Papadakis GE, Collet C, Trabado S, Maione L, Pitteloud N, Bouligand J, De Vernejoul MC, Cohen-Solal M, Young J (2021) Compromised volumetric bone density and microarchitecture in men with congenital hypogonadotropic hypogonadism. J Clin Endocrinol Metab 106(9):e3312–e3326. https://doi.org/10.1210/clinem/dgab169
Blüher M (2009) Adipose tissue dysfunction in obesity. Exp Clin Endocrinol Diabetes 117(6):241–250. https://doi.org/10.1055/s-0029-1192044
Pizzocaro A, Vena W, Condorelli R, Radicioni A, Rastrelli G, Pasquali D, Selice R, Ferlin A, Foresta C, Jannini EA, Maggi M, Lenzi A, Pivonello R, Isidori AM, Garolla A, Francavilla S, Corona G (2020) Testosterone treatment in male patients with Klinefelter syndrome: a systematic review and meta-analysis. J Endocrinol Invest 43(12):1675–1687. https://doi.org/10.1007/s40618-020-01299-1
Zitzmann M, Aksglaede L, Corona G, Isidori AM, Juul A, T’Sjoen G, Kliesch S, D’Hauwers K, Toppari J, Słowikowska-Hilczer J, Tüttelmann F, Ferlin A (2021) European academy of andrology guidelines on Klinefelter Syndrome Endorsing Organization: European Society of Endocrinology. Andrology 9(1):145–167. https://doi.org/10.1111/andr.12909
Corona G, Rastrelli G, Vignozzi L, Barbonetti A, Sforza A, Mannucci E, Maggi M (2021) The Role of testosterone treatment in patients with metabolic disorders. Expert Rev Clin Pharmacol 14(9):1091–1103. https://doi.org/10.1080/17512433.2021.1938548
Snyder PJ, Kopperdahl DL, Stephens-Shields AJ, Ellenberg SS, Cauley JA, Ensrud KE, Lewis CE, Barrett-Connor E, Schwartz AV, Lee DC, Bhasin S, Cunningham GR, Gill TM, Matsumoto AM, Swerdloff RS, Basaria S, Diem SJ, Wang C, Hou X, Cifelli D, Dougar D, Zeldow B, Bauer DC, Keaveny TM (2017) Effect of testosterone treatment on volumetric bone density and strength in older men with low testosterone: a controlled clinical trial. JAMA Intern Med 177(4):471–479. https://doi.org/10.1001/jamainternmed.2016.9539
Cauley JA, Ellenberg SS, Schwartz AV, Ensrud KE, Keaveny TM, Snyder PJ (2021) Effect of testosterone treatment on the trabecular bone score in older men with low serum testosterone. Osteoporos Int 32(11):2371–2375. https://doi.org/10.1007/s00198-021-06022-1
Colleluori G, Aguirre L, Napoli N, Qualls C, Villareal DT, Armamento-Villareal R (2021) Testosterone therapy effects on bone mass and turnover in hypogonadal men with type 2 diabetes. J Clin Endocrinol Metab 106(8):e3058–e3068. https://doi.org/10.1210/clinem/dgab181
Leanza G, Maddaloni E, Pitocco D, Conte C, Palermo A, Maurizi AR, Pantano AL, Suraci C, Altomare M, Strollo R, Manfrini S, Pozzilli P, Schwartz AV, Napoli N (2019) Risk factors for fragility fractures in type 1 diabetes. Bone 125:194–199. https://doi.org/10.1016/j.bone.2019.04.017
Napoli N, Schwartz AV, Palermo L, Jin JJ, Wustrack R, Cauley JA, Ensrud KE, Kelly M, Black DM (2013) Risk factors for subtrochanteric and diaphyseal fractures: the study of osteoporotic fractures. J Clin Endocrinol Metab 98(2):659–667. https://doi.org/10.1210/jc.2012-1896
Napoli N, Strotmeyer ES, Ensrud KE, Sellmeyer DE, Bauer DC, Hoffman AR, Dam TT, Barrett-Connor E, Palermo L, Orwoll ES, Cummings SR, Black DM, Schwartz AV (2014) Fracture risk in diabetic elderly men: the MrOS study. Diabetologia 57(10):2057–2065. https://doi.org/10.1007/s00125-014-3289-6
Napoli N, Conte C, Pedone C, Strotmeyer ES, Barbour KE, Black DM, Samelson EJ, Schwartz AV (2019) Effect of insulin resistance on BMD and fracture risk in older adults. J Clin Endocrinol Metab 104(8):3303–3310. https://doi.org/10.1210/jc.2018-02539
Hofbauer LC, Busse B, Eastell R, Ferrari S, Frost M, Müller R, Burden AM, Rivadeneira F, Napoli N, Rauner M (2022) Bone fragility in diabetes: novel concepts and clinical implications. Lancet Diabetes Endocrinol 10(3):207–220. https://doi.org/10.1016/s2213-8587(21)00347-8
Manigrasso MB, Juranek J, Ramasamy R, Schmidt AM (2014) Unlocking the biology of RAGE in diabetic microvascular complications. Trends Endocrinol Metab 25(1):15–22. https://doi.org/10.1016/j.tem.2013.08.002
Shanbhogue VV, Hansen S, Frost M, Jørgensen NR, Hermann AP, Henriksen JE, Brixen K (2016) Compromised cortical bone compartment in type 2 diabetes mellitus patients with microvascular disease. Eur J Endocrinol 174(2):115–124. https://doi.org/10.1530/eje-15-0860
Corona G, Monami M, Rastrelli G, Aversa A, Sforza A, Lenzi A, Forti G, Mannucci E, Maggi M (2011) Type 2 diabetes mellitus and testosterone: a meta-analysis study. Int J Androl 34(6 Pt 1):528–540. https://doi.org/10.1111/j.1365-2605.2010.01117.x
Napoli N, Strollo R, Defeudis G, Leto G, Moretti C, Zampetti S, D’Onofrio L, Campagna G, Palermo A, Greto V, Manfrini S, Hawa MI, Leslie RD, Pozzilli P, Buzzetti R (2018) Serum sclerostin and bone turnover in latent autoimmune diabetes in adults. J Clin Endocrinol Metab 103(5):1921–1928. https://doi.org/10.1210/jc.2017-02274
Piccoli A, Cannata F, Strollo R, Pedone C, Leanza G, Russo F, Greto V, Isgrò C, Quattrocchi CC, Massaroni C, Silvestri S, Vadalà G, Bisogno T, Denaro V, Pozzilli P, Tang SY, Silva MJ, Conte C, Papalia R, Maccarrone M, Napoli N (2020) Sclerostin regulation, microarchitecture, and advanced glycation end-products in the bone of elderly women with type 2 diabetes. J Bone Miner Res 35(12):2415–2422. https://doi.org/10.1002/jbmr.4153
Hygum K, Starup-Linde J, Harsløf T, Vestergaard P, Langdahl BL (2017) Mechanisms in endocrinology: diabetes mellitus, a state of low bone turnover - a systematic review and meta-analysis. Eur J Endocrinol 176(3):R137-r157. https://doi.org/10.1530/eje-16-0652
Epstein S, Defeudis G, Manfrini S, Napoli N, Pozzilli P (2016) Diabetes and disordered bone metabolism (diabetic osteodystrophy): time for recognition. Osteoporos Int 27(6):1931–1951. https://doi.org/10.1007/s00198-015-3454-x
Yu EW, Putman MS, Derrico N, Abrishamanian-Garcia G, Finkelstein JS, Bouxsein ML (2015) Defects in cortical microarchitecture among African-American women with type 2 diabetes. Osteoporos Int 26(2):673–679. https://doi.org/10.1007/s00198-014-2927-7
Patsch JM, Burghardt AJ, Yap SP, Baum T, Schwartz AV, Joseph GB, Link TM (2013) Increased cortical porosity in type 2 diabetic postmenopausal women with fragility fractures. J Bone Miner Res 28(2):313–324. https://doi.org/10.1002/jbmr.1763
Karim L, Moulton J, Van Vliet M, Velie K, Robbins A, Malekipour F, Abdeen A, Ayres D, Bouxsein ML (2018) Bone microarchitecture, biomechanical properties, and advanced glycation end-products in the proximal femur of adults with type 2 diabetes. Bone 114:32–39. https://doi.org/10.1016/j.bone.2018.05.030
Hunt HB, Torres AM, Palomino PM, Marty E, Saiyed R, Cohn M, Jo J, Warner S, Sroga GE, King KB, Lane JM, Vashishth D, Hernandez CJ, Donnelly E (2019) Altered tissue composition, microarchitecture, and mechanical performance in cancellous bone from men with type 2 diabetes mellitus. J Bone Miner Res 34(7):1191–1206. https://doi.org/10.1002/jbmr.3711
Napoli N, Schwartz AV, Schafer AL, Vittinghoff E, Cawthon PM, Parimi N, Orwoll E, Strotmeyer ES, Hoffman AR, Barrett-Connor E, Black DM (2018) Vertebral fracture risk in diabetic elderly men: the MrOS study. J Bone Miner Res 33(1):63–69. https://doi.org/10.1002/jbmr.3287
Leslie WD, Aubry-Rozier B, Lamy O, Hans D (2013) TBS (trabecular bone score) and diabetes-related fracture risk. J Clin Endocrinol Metab 98(2):602–609. https://doi.org/10.1210/jc.2012-3118
Kim JH, Choi HJ, Ku EJ, Kim KM, Kim SW, Cho NH, Shin CS (2015) Trabecular bone score as an indicator for skeletal deterioration in diabetes. J Clin Endocrinol Metab 100(2):475–482. https://doi.org/10.1210/jc.2014-2047
Caffarelli C, TomaiPitinca MD, Al Refaie A, Ceccarelli E, Gonnelli S (2022) Ability of radiofrequency echographic multispectrometry to identify osteoporosis status in elderly women with type 2 diabetes. Aging Clin Exp Res 34(1):121–127. https://doi.org/10.1007/s40520-021-01889-w
Ferrari SL, Abrahamsen B, Napoli N, Akesson K, Chandran M, Eastell R, El-Hajj Fuleihan G, Josse R, Kendler DL, Kraenzlin M, Suzuki A, Pierroz DD, Schwartz AV, Leslie WD (2018) Diagnosis and management of bone fragility in diabetes: an emerging challenge. Osteoporos Int 29(12):2585–2596. https://doi.org/10.1007/s00198-018-4650-2
Eastell R, Vittinghoff E, Lui LY, Ewing SK, Schwartz AV, Bauer DC, Black DM, Bouxsein ML (2022) Diabetes mellitus and the benefit of antiresorptive therapy on fracture risk. J Bone Miner Res. https://doi.org/10.1002/jbmr.4697
Schwartz AV, Pavo I, Alam J, Disch DP, Schuster D, Harris JM, Krege JH (2016) Teriparatide in patients with osteoporosis and type 2 diabetes. Bone 91:152–158. https://doi.org/10.1016/j.bone.2016.06.017
Funding
The authors declare that they have no financial interests.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All the authors declare that they have no conflicts of interest.
Informed consent
For this type of study, formal consent is not required.
Research involving human participants and/or animals
The authors received the permission by two patients followed-up in the outpatient bone clinic for using anonymously for research purposes the images of their DXA and CT exams.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cianferotti, L., Cipriani, C., Corbetta, S. et al. Bone quality in endocrine diseases: determinants and clinical relevance. J Endocrinol Invest 46, 1283–1304 (2023). https://doi.org/10.1007/s40618-023-02056-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-023-02056-w