Abstract
An important provision of the Minamata Convention on Mercury is to monitor and evaluate the effectiveness of the adopted measures and its implementation. Here, we describe for the first time currently available biotic mercury (Hg) data on a global scale to improve the understanding of global efforts to reduce the impact of Hg pollution on people and the environment. Data from the peer-reviewed literature were compiled in the Global Biotic Mercury Synthesis (GBMS) database (>550,000 data points). These data provide a foundation for establishing a biomonitoring framework needed to track Hg concentrations in biota globally. We describe Hg exposure in the taxa identified by the Minamata Convention: fish, sea turtles, birds, and marine mammals. Based on the GBMS database, Hg concentrations are presented at relevant geographic scales for continents and oceanic basins. We identify some effective regional templates for monitoring methylmercury (MeHg) availability in the environment, but overall illustrate that there is a general lack of regional biomonitoring initiatives around the world, especially in Africa, Australia, Indo-Pacific, Middle East, and South Atlantic and Pacific Oceans. Temporal trend data for Hg in biota are generally limited. Ecologically sensitive sites (where biota have above average MeHg tissue concentrations) have been identified throughout the world. Efforts to model and quantify ecosystem sensitivity locally, regionally, and globally could help establish effective and efficient biomonitoring programs. We present a framework for a global Hg biomonitoring network that includes a three-step continental and oceanic approach to integrate existing biomonitoring efforts and prioritize filling regional data gaps linked with key Hg sources. We describe a standardized approach that builds on an evidence-based evaluation to assess the Minamata Convention’s progress to reduce the impact of global Hg pollution on people and the environment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Global recognition of mercury (Hg) as an environmental contaminant with effects on humans and wildlife has resulted in the global establishment of a multilateral environmental agreement - the Minamata Convention on Mercury (Bank 2020). To date over 145 countries have ratified the Convention, agreeing to reduce anthropogenic sources of Hg to the environment and to evaluate if the established provisions are effective in meeting its goals. One important provision of the Convention is to monitor and evaluate the effectiveness of its implementation (Articles 1 and 22). This will require having standardized measurements of environmental concentrations of Hg in abiotic and biotic compartments over time (Evers et al. 2016). The establishment of “baseline” levels is critical for determining whether Hg levels have declined in response to source reductions mandated by the Convention, a monitoring principle that is required in other disciplines (e.g., Verra 2021). In order to track Hg concentrations over time, it will be necessary to select the environmental abiotic matrices (e.g., air, precipitation, water, soil, sediments) and various biotic tissue matrices (e.g., muscle, keratin materials such as feather or fur, blood, and eggs) from relevant bioindicators (e.g., fish, sea turtles, birds and marine mammals) that can provide reliable and repeatable measures both temporally and spatially. Ideally the tissue concentrations should also provide information on whether the indicator species are negatively affected by their Hg exposure by linking their Hg body burdens to effect levels determined for related species.
To date, there have been many spatial studies of Hg at the regional scale and temporal studies over timescales of decades in some locations; often these studies focus on locations or areas with known Hg contamination. However, to evaluate the effectiveness of the Convention, a global monitoring system that promotes standardized spatial and temporal coverage is needed. Given that establishing appropriate and effective monitoring frameworks has been a persistent challenge for wildlife conservation (e.g., Tear et al. 2005), increased efforts are needed to embrace more evidence-based approaches for assessing the effectiveness of actions (e.g., Salafsky et al. 2019). The overall goal of this paper is to describe the currently available repositories of peer-reviewed published biotic Hg data and existing monitoring programs to set the stage for developing a global Hg biomonitoring network that can provide accurate information for the assessment of the overall effectiveness of global efforts to reduce the adverse impacts of Hg pollution on people and the environment.
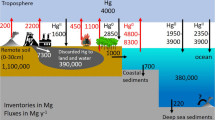
Globally, Hg enters ecosystems through the air (e.g., emissions from coal-fired power plants, incinerators, and volcanic activity), water (e.g., both inactive and active chlor-alkali facilities and artisanal small-scale gold mining), and land (e.g., natural geological formations, mine tailings, landfills, and other contaminated sites) (UNEP 2013; Pacyna et al. 2016; Kocman et al. 2017; Streets et al. 2017, Hsu-Kim et al. 2018; Obrist et al. 2018; Keane et al. 2023). Mercury emitted to the air and released into landscapes, where it can be transported across great distances, remains available for days to years, where its fate is complex as it moves through both terrestrial and aquatic ecosystems into biota (Driscoll et al. 2013; Gustin et al. 2016; Eagles-Smith et al. 2018).
Inorganic Hg emitted from natural or anthropogenic sources becomes more toxic and bioavailable in the environment when it is converted to methylmercury (MeHg), by a wide range of microbial communities (Fleming et al. 2006; Gilmour et al. 2013; Hsu-Kim et al. 2013; Yu et al. 2013). Certain ecosystem conditions (primarily those with an aquatic component, especially wetlands) can encourage the production and bioavailability of MeHg in the environment. Bacteria often produce more MeHg when moderate amounts of sulfate and low oxygen (hypoxic or anoxic) conditions are present to provide optimal conditions for the metabolic processes of the microorganisms (Hsu-Kim et al. 2013, 2018; Hu et al. 2020).
Environmental factors such as water pH, dissolved organic carbon, sulfur concentrations, and land use are important in influencing both inorganic Hg input and methylation potential (Gorski et al. 2008; Wyn et al. 2009; Gabriel et al. 2014; Schartup et al. 2015b; Chaves-Ulloa et al. 2016; Chételat et al. 2018; Rudd et al. 2018; Braaten et al. 2018, 2020). Ecological processes at the base of the food webs such as primary productivity and biomass dilution are also important in the trophic transfer of MeHg from algae to primary and secondary consumers (Pickhardt et al. 2002; Chen et al. 2005; Wu et al. 2019). The complex of redox and biological processes involved in Hg cycling make it particularly challenging to predict levels of potential concern in upper tropic level fish and wildlife from concentrations in air, water, and sediment (Gustin et al. 2016; Sunderland et al. 2016; Eagles-Smith et al. 2018). Ecological and biogeochemical factors play a large role in altering MeHg bioavailability to biota, although Hg concentrations in water and sediment are often not generally correlated with MeHg concentrations in biota (Tsui et al. 2023). Therefore, in sites where Hg deposition or Hg sources are low, levels and effects on biota may still be disproportionately high if the ecological conditions are conducive to MeHg production, bioaccumulation, and biomagnification. For example, MeHg concentrations in fish across freshwater ecosystems in western North America and in estuaries are poorly correlated with either total Hg or MeHg concentrations in aquatic sediments (Eagles-Smith et al. 2016a; Chen et al. 2014; Buckman et al. 2019). The difference between relatively high fish Hg levels in Sweden with low environmental levels of Hg (Braaten et al. 2020) and the low levels of Hg in many fish from waterbodies in China where levels of Hg in sediment, water and even the base of the food web are relatively high, is another example of the importance of understanding relationships between ecological conditions and the levels of biotic Hg. (Wu et al. 2023). The decoupling of inorganic Hg sources from MeHg production and bioavailability is evident at local (Evers et al. 2007) and landscape levels (Eagles-Smith et al. 2016b, 2018; Wang et al. 2023a, 2023b).
Mercury, in its methyl form, is a neurotoxicant and can impair physiological and neurological functions, behavior, reproduction, and survival in fish and wildlife (Scheuhammer et al. 2011; Ackerman et al. 2016; Evers 2018; Whitney and Cristol 2017), as well as humans (Tan et al. 2009; Karagas et al. 2012; Ha et al. 2017; Eagles-Smith et al. 2018; Basu et al. 2023). It readily biomagnifies through foodwebs, resulting in increasing MeHg concentrations as it moves from water and sediment to phytoplankton and plants, zooplankton, aquatic and terrestrial invertebrates, fish, wildlife, and humans. As MeHg moves through the base of the foodwebs, it can efficiently biomagnify in both aquatic and terrestrial organisms. As a result, top predators in foodwebs, including specific species of fish, amphibians, reptiles, birds, and mammals, may have MeHg concentrations in their tissues that are orders of magnitude higher than the concentrations found in water (often >106 to 107 higher). Generally, each trophic increase in the food web accounts for roughly an order of magnitude increase in MeHg concentrations, with the largest enrichment step occurring between water and phyto and zooplankton in aquatic systems (Lee and Fisher 2016; Wu et al. 2019).
Exposure to MeHg has been well documented in fish and wildlife around the world. Contamination can arise directly from inorganic Hg point sources, such as those along rivers (Jackson et al. 2011a; Kinghorn et al. 2007; Nguetseng et al. 2015; Santschi et al. 2017; Geyer and Ralston 2018), around lakes (Anderson et al. 2008; Suchanek et al. 2008; Kumari and Maiti 2019; Chen et al. 2021), and in estuaries (Eagles-Smith and Ackerman 2009; Chen et al. 2014; Buckman et al. 2015; Sullivan and Kopec 2018). Owing to atmospheric transport, inorganic Hg sources may not be local (i.e., <100 km) and subsequent impacts to biota are well described in most continents, including North America (Evers and Clair 2005; Evers et al. 2011a; Ackerman et al. 2016; Eagles-Smith et al. 2016a, b; Evers et al. 2020; AMAP 2021), South America (Sebastiano et al. 2016; May Junior et al. 2017; Manhães et al. 2022), Europe (Åkerblom et al. 2014; Nguetseng et al. 2015; Pacyna et al. 2017), Asia (Kim et al. 2012; Watanuki et al. 2016; Abeysinghe et al. 2017; Noh et al. 2017), Africa (Hanna et al. 2015; van Rooyen et al. 2023), and multiple ocean basins (Carravieri et al. 2014, 2016; Peterson et al. 2015; Drevnick et al. 2015; Lee et al. 2016; Bodin et al. 2017; Drevnick and Brooks 2017; Chastel et al. 2022).
Numerous studies document adverse impacts across many fish and wildlife species. In fish, adverse impacts of MeHg exposure include immunological, reproductive, and behavioral impairment (Hammerschmidt et al. 2002; Depew et al. 2012a; Carvan et al. 2017) as well as reduced capacity for predator avoidance (Webber and Haines 2003). In birds, numerous studies have documented reduced reproductive success, behavioral change (e.g., reduced time incubating), and neurological problems (e.g., ataxia) (Depew et al. 2012a, b; Basu 2015; Ackerman et al. 2016; Evers 2018; Whitney and Cristol 2017; Cristol and Evers 2020). However, many species vary in their sensitivity to MeHg toxicity - potentially based on foraging guilds and phylogeny as identified and discussed by Heinz et al. 2009). For example, embryo survival and hatching success in Passeriforms (i.e., songbirds), appears to be more sensitive to MeHg toxicity than in other orders of birds that have been more extensively studied, such as Anseriformes ducks. In mammals, elevated MeHg concentrations can result in biochemical changes in the brain, ataxia, and reduced reproductive output (Basu et al. 2007; Dietz et al. 2013, 2019, 2021, 2022; Evers 2018; Manhães et al. 2021). Based on these and other in situ studies collectively, the evidence is clear that biomagnification and bioaccumulation of MeHg is shown to adversely affect the reproductive success of many fish and wildlife populations. These biota represent multiple foraging guilds across many habitats and geographic areas of the world.
Understanding exposure pathways of MeHg in terrestrial and aquatic foodwebs and how MeHg adversely affects upper trophic level wildlife is important for developing meaningful assessments and monitoring efforts. Ultimately, identifying the proper fish and wildlife bioindicators for MeHg biomonitoring is complex, because their suitability differs according to geographic area, timescale of interest, conservation concern, and whether the overall goal is for ecological or human health or simply to track changes over time in a consistent and representative species. Herein, we describe some of the regional and global spatial and temporal patterns of MeHg exposure in fish and wildlife based on peer-reviewed literature with an emphasis on relevant bioindicators.
The objectives of this paper are to provide an overview of global, peer-reviewed biotic Hg data to: 1) spatially describe selected global human exposure and ecological bioindicators 2) assess existing biomonitoring data and programs in select regions around the world, and 3) develop strategies for establishing a global Hg biomonitoring framework that can respond to the global strategy defined by the Minamata Convention that calls for establishing an evidence-based monitoring approach to improve our understanding of linkages with Hg sources, spatial gradients and temporal trends.
Methods
Data within the primary or peer-reviewed literature represent numerous studies that include Hg concentrations in taxa identified in Article 19 of the Minamata Convention on Mercury. Between 2013 and 2023, the Center for Mercury Studies of Biodiversity Research Institute created and maintained a database of Hg concentrations in biotic tissue called the Global Biotic Mercury Synthesis (GBMS). The published data compiled in the GBMS database are summarized here with an emphasis on organisms identified to be of interest for monitoring within the Minamata Convention, which include fish (both teleosts and elasmobranchs), sea turtles, birds, and marine mammals (see Supplementary Information for a listing of the 1701 references used). In some cases, these peer-reviewed sources incorporate data from national monitoring studies, and in other cases are individual scientific studies conducted by governmental agencies, academic researchers, and others. Many Hg concentration measurements in biota, especially fish, have been generated by government agencies around the world. Many of these associated data are not represented here because they are not published in the peer-reviewed literature. However, these unpublished data are significant sources of information that should be collected in a standardized way for evaluating the effectiveness of the Minamata Convention.
Lastly, data for Hg concentrations in tissues that were collected from the published literature include individual (30.4%), composited (4.3%), and averaged (65.2%) data. Mercury concentrations in fish and wildlife are typically log-normally distributed, including the pooled GBMS dataset, and therefore using geometric means are preferable to using arithmetic means. However, the published data were often presented as arithmetic means. Therefore, the composited and averaged data were typically extracted as arithmetic means from the published literature. For consistency, all Hg concentration data presented here are arthimetic means with assoicated standard deviations unless otherwise noted.
For this initial assessment, we did not standardize Hg concentrations. For evaluating the effectiveness of the Minamata Convention, the primary goal will be to evaluate long-term trends in Hg concentrations in fish and wildlife. Because sampling effort and specifics vary among sites and years, it will be important in future trend analyses to account for ecological covariates that are known to influence Hg concentrations in animals. Ecological covariates can be accounted for either in the experimental design (by using a specified subset of the data and excluding any samples that do not meet strict criteria) or during statistical analysis (if sample sizes are sufficient, by including these covariates in the statistical model). For example, in fish, Hg concentrations generally increase substantially with length (Eikenberry et al. 2015), and therefore fish Hg concentrations are generally size-standardized for statistical comparisons (Eagles-Smith et al. 2016a; Drevnick and Brooks 2017). In birds, Hg concentrations generally are higher in males than females (Evers et al. 1998, 2005; Ackerman et al. 2008) and can change with age (Evers et al. 2005; Ackerman et al. 2011). In most wildlife, Hg concentrations vary substantially among sites and day of the year (Ackerman et al. 2019; Chételat et al. 2020). Thus, for effectiveness evaluation of the Minamata Convention, these additional metadata will be important for standardizing Hg concentrations. Several examples of large-scale statistical evaluations of wildlife Hg concentrations are available as a guide (Ackerman et al. 2016; Eagles-Smith et al. 2016a; Drevnick and Brooks 2017; Schoch et al. 2020).
All biotic samples were assigned a Taxonomic Serial Number (TSN) based on the identification provided in the published literature using the Integrated Taxonomic Information System (ITIS) to allow for standardization (ITIS 2023). Species level assignments were made where possible and the lowest taxonomic level that could be reliably assigned was used where species level data was not available.
Fish trophic levels were assigned by species using ‘Fishbase’ and the mean trophic level for the genus or family used, respectively, when presented (Boettiger et al. 2012). The widespread occurrence of ‘fishing down’ is the reason why, in 2004, the Convention on Biological Diversity (CBD) chose the mean trophic level of fisheries catches as an index of the biodiversity of large fishes (defined as fish with trophic levels >3.5), called the Marine Trophic Index or MTI (Pauly and Watson 2005). As a result, a threshold of trophic level 3.5 was used for visualization for some graphs.
Selection of bioindicators
A key initial step in bioindicator selection is to decide whether an organism is linked to a human exposure or ecological health endpoint – which can often be combined for both purposes if carefully considered. Biota that have been identified to best fit these two categories are well described and are categorized within their respective biomes and associated aquatic ecosystems (Table 1). Where applicable, utilization of Indigenous Knowledge is important to incorporate (Houde et al. 2022). Additionally, bioindicators should be reflective of changes in the availability of MeHg in the environment. One of the challenges of using multiple bioindicators is that their Hg concentrations are also affected by food web processes, physical movement, and physiology in addition to changes in the availability of MeHg in the environment (Chételat et al. 2020). Therefore, a key aspect of bioindicator selection for evaluating the effectiveness of reductions in environmental loads of Hg driven by the Minamata Convention is that their concentrations are less sensitive to variability caused by these other factors.
The extensive datasets of Hg concentrations in biota found in the published literature provide a basis for choices of species for potential monitoring (Fig. 1; Evers et al. 2016). Careful selection can ensure comparability at regional and global scales. A systematic literature search (range of years covered was 1972 to 2023) emphasized Hg data from: (1) biota identified in Article 19 of the Minamata Convention (fish, sea turtles, birds, and marine mammals), (2) species for human consumption, (3) taxonomic groups at high risk of MeHg exposure, (4) potential bioindicators for MeHg monitoring purposes, and (5) species from areas of concern due to current significant Hg sources (e.g., coal-fired power plants and artisanal small-scale gold mining [ASGM]). The taxonomic presentation structure follows phylogenetic order.
Distribution of five major taxa and their total Hg concentrations in three risk categories based on mean data derived from a survey of the available peer-reviewed English literature. Risk categories by major taxa and tissue type are: (1) cartilaginous fish (sharks and allies) and (2) bony fish muscle (µg/g, ww): <0.23 = low, 0.23–0.46 = moderate, >0.46 = high; (3) sea turtle muscle and egg (µg/g, ww): <0.22 = low, 0.22–0.46 = moderate, >0.46 = high; (4) bird body feathers (adult; µg/g, fw): <10.0 = low, 10.0–20.0 = moderate, >20.0 = high; bird blood (adult; µg/g, ww): <1.0 = low, 1.0–3.0 = moderate, >3.0 = high; eggs (µg/g, ww): <0.5 = low, 0.5–1.0 = moderate, >1.0 = high; (5) marine mammal muscle (µg/g, ww): <0.22 = low, 0.22–0.46 = moderate, >0.46 = high. Letters indicate additional available fish Hg samples that were not mapped: a >330,000 additional fish Hg concentrations within the Canadian Fish Mercury Database (Depew et al. 2013); b an estimated >500,000 additional fish Hg concentrations available within state databases in the United States, and; c >54,000 additional fish Hg concentrations within Fennoscandia (Braaten et al. 2019). Data for migratory species are plotted according to the reported location of sampling, which in some cases may not reflect the region where Hg exposure occurs
Because the selection of the taxa recommended for bioindicator species emphasizes the animal groups identified in the Minamata Convention’s Article 19, and their ability to represent MeHg exposure in a particular system, invertebrates are not considered. Therefore, organisms such as phyto and zooplankton are not included for biomonitoring given their high temporal and spatial variability within waterbodies and that they require maximizing within-date replication and higher frequency sampling during a season (Ward et al. 2012; Chen et al. 2012). In addition, the percent MeHg concentrations (generally less than 75%) are not as high as those in higher trophic level fish (>95%; Driscoll et al. 2007).
Lastly, published studies included here are those for which there is reasonable confidence about their validity, including those with: (1) sufficient description of the characteristics of the organism sampled (i.e., species, date, location, size/age, and tissue analyzed); (2) an appropriate method of sample collection; and (3) detailed information on sampling location (i.e., market-based fish Hg concentrations are excluded). For North America, extensive biotic Hg datasets published in response to three regional, one state, and one National Park synthesis efforts are included for the: (1) northeastern United States and eastern Canada (Evers and Clair 2005), (2) Great Lakes Region of the United States and Canada (Evers et al. 2011a), (3) western United States and Canada (Eagles-Smith et al. 2016b), (4) New York State (Evers et al. 2020), and (5) Acadia National Park, Maine, United States (Burton et al. 2024); although, all of the data are not mapped (Fig. 1 and see qualifiers “a” and “b”).
The data collected and incorporated into GBMS represent the arithmetic mean, or individual sample concentrations (when available), standard deviation (SD), minimum and maximum values, total number of individuals for each species, and tissue type that could be georeferenced within a peer-reviewed publication. These data were then joined by taxa and tissue type to generate a global average and variation. The raw data underlying the averaged statistics used herein were not always available and therefore, individual metadata of biota were not included (e.g., no adjustments or normalization for age, sex and size were conducted). Each of the published studies’ Hg concentrations was mapped by major taxonomic group (i.e., cartilaginous and bony fish, sea turtles, birds and marine mammals) and tissue type and were placed in three risk categories based on human exposure or ecological health thresholds (i.e., low, medium and high).
Risk categories
The health-related risk categories based on human exposure are developed from a combination of benchmarks pertaining to animal tissues consumed by humans and generated from standards used in the United States (USFDA 2022), for Arctic communities (AMAP 2015), and by the World Health Organization that generally relate to MeHg exposure levels of concern recognized for humans (Višnjevec et al. 2014; Basu et al. 2018). For human populations, those most at risk of MeHg exposure include: (1) sensitive individuals (e.g., women of childbearing age, pregnant women, and children); (2) communities dependent on a diet of aquatic organisms (e.g., Indigenous and subsistence fish consumers); and (3) diets regularly including high trophic-level fish (e.g., recreational anglers). The greatest risks to humans from dietary uptake of MeHg are observed with high consumption of upper trophic level species. For example, primary consumers (e.g., shellfish such as mussels) at trophic level 2 generally have relatively low MeHg concentrations and are usually considered safe for consumption (Chase et al. 2001). Secondary consumers (e.g., salmon, herring) are at trophic level 3, but are usually considered to be healthy choices.
For tertiary or higher consumers, which are carnivorous fish that generally consume vertebrate prey, MeHg concentrations can be elevated to levels that trigger human health concerns. The variability of concentrations in fish with a trophic level of 3.5 or more can be related to size, species, and location (Keppeler et al. 2020). Therefore, fish exceeding a trophic level of 3.5 that are commonly harvested are important bioindicators to assess potential exposure risk of Hg to humans. Large marine predatory fish such as tuna, swordfish and shark can have elevated MeHg concentrations, frequently exceeding the no consumption limits (or choices to avoid) identified by the USEPA and U.S. Food and Drug Administrations (USFDA [i.e., 0.46 µg/g, ww; Table 2]). While the European Union (EU) identifies the maximum level for total Hg in fish muscle for human consumption is 0.5 μg/g ww, they also include an exception for ‘large predators’ for which the maximum level is 1.0 μg/g ww (EU 2023). The USEPA - USFDA consumption limits are used herein.
The impacts of MeHg on fish health and reproductive welfare are not well established, but have been summarized previously (Depew et al. 2012a, b; Table 3a) and include threshold limits identified by Sandheinrich et al. (2011). While fish Hg concentrations are commonly examined for their impacts on humans (i.e., muscle tissue) or for wildlife exposure (e.g., whole body), the MeHg concentrations in fish tissues also can be assessed for their impact on behavior, reproductive abilities, and overall health.
Fish may exhibit impaired reproductive success at relatively low dietary MeHg concentrations as low as 0.04 µg/g, ww (Depew et al. 2012a) and may have adverse visible behavioral impacts at dietary MeHg concentrations of 0.50 µg/g, ww or higher (Depew et al. 2012a) (Table 3a). A recent synthesis of the effects of Hg on freshwater fish further summarizes adverse effects at physiologic, histologic, biochemical, enzymatic, and genetic levels; and that some fish species demonstrate greater sensitivity to MeHg than others (Morcillo et al. 2017). Ultimately, lower reproductive success reduces the size and sustainability of healthy fish populations, which could have adverse impacts on associated populations of piscivores and human recreational and commercial interests. Unlike freshwater fish, there have been few rigorous published studies evaluating toxicity of MeHg to marine fish (Scheuhammer et al. 2015; Morcillo et al. 2017).
For understanding risk to the health of birds, known risk categories for diet (Table 3a) and various tissue types (e.g., eggs, blood, and feathers) are well-established for some piscivores and invertivores (Table 3b). The science behind characterizing risk benchmarks that are based on both laboratory and wildlife populations has improved significantly from the initial study of lab-based lowest-observed adverse effect levels on a single species – the mallard (Anas platyrhynchos) (Heinz 1979) to more recent efforts. Current research documents effect concentrations related to different levels of breeding success in invertivore songbirds (Carolina wren, Thryothorus ludovicianus) and piscivorous waterbirds (common loons, Gavia immer) and is well supported by data collected in both laboratory and wild bird populations (Ackerman et al. 2016; Evers 2018; Whitney and Cristol 2017).
Adverse effect thresholds are not as well established for marine mammals, primarily because of field study and ethical challenges (Dietz et al. 2022). Brain Hg concentrations were found to have a significant positive correlation with liver concentrations, and brain Hg concentrations reported in cetaceans were one order of magnitude higher than pinnipeds and generally exceeded neurotoxicity thresholds (López-Berenguer et al. 2020).
Preferred tissue types and important metadata
Understanding the pharmacodynamics of Hg species concentrations in organisms is important because MeHg biomagnifies through foodwebs in polar (Ruus et al. 2015; Seco et al. 2021; Matias et al. 2022), temperate (Arcagni et al. 2018), and tropical (Bisi et al. 2012; Seixas et al. 2014) ecosystems, and bioaccumulates over time in individual fish (Drevnick and Brooks 2017), birds (Evers et al. 1998), and marine mammals (Lailson-Brito et al. 2002, 2012; Krey et al. 2015). The cycling, speciation, and toxicology of Hg can vary substantially among different tissues, which can have important implications for interpreting Hg concentrations (Manhães et al. 2021). Understanding how the selection of tissue types dictate interpretative power in the bioaccumulation and biomagnification of MeHg and subsequent potential health impacts is a critical aspect for developing monitoring designs (Eagles-Smith et al. 2016b; Chételat et al. 2020). Additionally, to establish relevant species, tissues, and timing of sampling of importance for human Hg exposure in Indigenous Populations, Indigenous Knowledge can provide crucial information (AMAP 2021; Houde et al. 2022).
This review focuses on tissues with well-established methods of measurement and interpretation and for which there is a large body of data and are regularly used for monitoring purposes (Table 4). There are many available matrices and tissue choices dependon monitoring objectives, interests, and outcomes. Often the most useful tissues can be non-lethally collected in the field. Samples that can be analyzed to assess total Hg or MeHg exposure are often from tissue types for targeted biotic groups (Table 4). Composite samples are sometimes used to estimate population Hg concentrations at a decreased cost (Gandhi et al. 2016) and are especially useful for cost-effective long-term trend assessments (Gandhi et al. 2016). Because most of the Hg in tissues that are commonly tested for biomonitoring purposes is in the MeHg form (i.e., generally >95%), analyses of total Hg (which is less expensive to analyze) is also more cost effective. The development of direct analyzers that couple thermal decomposition with Hg amalgamation and atomic absorption detection has simplified Hg determination and made analysis more accessible to those without advanced and costly laboratory facilities (Windmöller et al. 2017).
Other metadata that are important to improve interpretive power include physiological, demographic, and ecological factors (Chételat et al. 2020). For example, accounting for the health and fitness of indicator organisms is important for standardized comparisons, as is the identification of species, size, age, and sex. Covariation between Hg concentration and fish size (length and weight) and age requires a standardization to allow for investigation of temporal trends of Hg concentrations. However, for most of this data compilation, biotic Hg concentrations were not indexed or standardized according to size, age, or sex. This is a weakness of the dataset and is an important reason for designing a standardized sampling framework to strengthen the ability to interpret the data.
In general, larger and older individuals have higher MeHg concentrations than smaller and younger individuals, and males that are larger in body size than females tend to have higher concentrations in fish and birds (Evers et al. 2005; Robinson et al. 2012; Ackerman et al. 2008, 2015, 2016; Hartman et al. 2017), with a few exceptions related to foraging segregation between sexes like in albatrosses (Carravieri et al. 2014). An exception from the evaluation of fish Hg concentrations without data on age and/or size are fish Hg databases in Scandinavia (Braaten et al. 2019) and across North America (Kamman et al. 2005; Monson et al. 2011; Eagles-Smith et al. 2016a). Braaten et al. (2019) used the individual fish weight and Hg concentration in combination with fish species information and sampling year to find the modeled (i.e., expected) Hg concentration for fish at a standard weight; there are similar findings in tuna species as well (Médieu et al. 2021, 2022).
Changes in an animal’s physiology, health status, or ecological life history events can also have a substantial effect on MeHg concentrations, regardless of an animal’s actual environmental MeHg exposure. For example, the maternal transfer of MeHg to offspring during reproduction can reduce the female’s tissue concentrations of MeHg but increases risk to offspring, and the amount of MeHg transferred from females to their offspring differs among species (Ackerman et al. 2020). Weight change can also influence the interpretation of MeHg concentrations in animals. For instance, rapid growth of juvenile birds can cause mass dilution of contaminants and substantially reduce MeHg concentrations as juvenile birds age (Ackerman et al. 2011). Rapid growth in fish can also result in “growth dilution” and has been measured in freshwater and marine fish (Ward et al. 2010; Baumann et al. 2017). In contrast, annual life changes in adult body mass, such as fasting- and breeding-associated declines in body mass during periods of haul-out on land for marine mammals, can substantially increase MeHg concentrations (Peterson et al. 2018). In the same way, infections can cause MeHg remobilization and changes its body distribution (Manhães et al. 2021).
Seasonality can have large implications for biotic Hg monitoring programs (Eagles-Smith and Ackerman 2009; Braaten et al. 2014). Seasonal changes in MeHg exposure may be related to changing methylation rates and bioavailability in estuaries (e.g., saltmarsh sparrows, Ammodramus caudacutus, increase in blood Hg concentrations from early to late summer; Lane et al. 2011), molt strategies (Condon and Cristol 2009), migratory patterns for birds (Ackerman et al. 2019) and arrival to over-wintering areas (Eagles-Smith et al. 2009a), or lake-specific variation in Hg dynamics (e.g., Clark’s and western grebes, Aechmophorus clarkia and A. occidentalis, decrease in blood Hg from spring to autumn; Hartman et al. 2017). Lower food availability in winter can also result in losses in body condition factor and increases in Hg concentrations in fish tissue (Martyniuk et al. 2020; Piro et al. 2023).
Lastly, as outlined in AMAP (2021), and summarized by Houde et al. (2022), Indigenous Knowledge provides invaluable information for the interpretation of tissue Hg concentrations in the Arctic environment and should be appropriately utilized together with scientific evidence for a holistic and comprehensive analysis. Examples include explaining Hg concentrations in whitefish in Nunavut, Canada, that were lower than expected. Indigenous Knowledge explained that whitefish migrated out to sea to feed after the ice went out, which was not known to scientists, and explained the lower Hg levels that were found in these fish. Similarly, research in Nunavik found elevated levels of selenoneine in the blood of women. Selenoneine is a protective compound against negative impacts of Hg. Knowledge holders explained that this could be connected to only women eating the tail of the beluga, and analysis confirmed that selenoneine concentrations in the skin of the beluga tail are nearly twice as high compared to the skin from other areas of the whale (AMAP 2021; Houde et al. 2022). Other research on beluga whales in Nunavik investigated Indigenous Knowledge including on migration, body condition, foraging ecology, predation, breeding, calving and behavior of animals - all of which can help understand beluga exposure to Hg and other contaminants (Breton-Honeyman et al. 2016). Indigenous Peoples have lived on their lands for hundreds or even thousands of years, in an intricate relationship with their environment, with knowledge being passed on through many generations and ensuring their survival. Consequently, they have the most intimate understanding of their ecosystems and their complex connections, as well as any changes that occurred over time – be it in the Arctic, the Amazon, or other biomes.
Results
Biotic Hg concentrations for targeted taxa (based on Article 19 of the Minamata Convention) were collected from over 1700 peer-reviewed (See Supplementary Materials) scientific publications that represent >588,000 individuals at over 4100 unique locations in 139 countries (Fig. 1). The coverage of biotic Hg tissues concentrations in the GBMS data repository is global and comprises every continent and ocean basin (Table 5). When considering the geographical patterns in ‘risk levels’ it should be noted that the data shown in Fig. 1 represent samples collected over several decades and include many studies specifically focussing on areas with known Hg contamination, which can bias the resulting picture.
Furthermore, the density of datapoints in each region varies greatly and reveals the areas that are less studied than others. The GBMS dataset demonstrates the extent of the global dataset (Table 6) and shows the greatest availability of data at the continental level from North America, Europe, and South America whereas there is less availability from Africa, Antarctica, Asia, and Australia. For ocean basins, the greatest availability of data comes from the Arctic Ocean and Mediterranean Sea, with lesser availability from the Caribbean Sea, Indian Ocean, and North Atlantic and Pacific Oceans and minimal data from the South Atlantic and Pacific Oceans.
The most well-represented species group in the GBMS database are teleosts; bony, ray-finned fishes that are extremely diverse, as they contain over 95% of all fish species and are ubiquitous around the world in freshwater and marine ecosystems. In the GBMS database, marine teleosts were represented in 30 Orders by 92,426 individuals at 826 distinct locations, while freshwater teleosts included 26 Orders with 312,335 individuals at 973 distinct locations. By comparison, cartilaginous fish (elasmobranchs) comprised of sharks, skates, and rays, were represented in 13 Orders by 13,605 individuals at 212 distinct locations. Birds were the second most abundant species group, represented in 26 Orders by 134,427 individuals at 1296 distinct locations. Marine mammals were divided into four groups (i.e., toothed and baleen cetaceans, pinnipeds, and polar bears) and represented by 30,698 individuals at 601 locations. Finally, sea turtles were represented by 5007 individuals from a total of 82 distinct locations.
Many of the data collected reflect the various monitoring programs that exist at local (e.g., New York State, USA), national (e.g., Northern Contaminants Program in Canada), and regional scales (e.g., the Caribbean Regional Mercury Monitoring Network), and even multi-hemispheric scales (e.g., the Arctic Monitoring and Assessment Programme [AMAP]). A summary of these programs is provided based on a review by UNEP (UNEP 2016). In the interest of developing a Hg monitoring network that uses existing Hg data and biomonitoring programs, a framework has been developed for oceans and continents that can draw on the existing Hg data and potentially meet the biomonitoring interests of the Minamata Convention if key geographic and taxonomic data gaps can be filled. These three broad elements are herein covered: (1) biotic data Hg exposure profiles from GBMS, (2) existing Hg monitoring programs, and (3) a path forward for new Hg monitoring frameworks.
To provide sustainable and long-term biomonitoring capacity in key regions around the world where Hg inputs are likely having adverse impacts to human communities and ecological health (e.g., Arctic, tropical areas associated with artisanal small-scale gold mining, and oceanic islands), the focus should be placed on expanding and stabilizing existing national initiatives that use relevant sample sizes that can meet statistical power for confidence in understanding spatial gradients (e.g., ecosystem sensitivity spots; Evers et al. 2011b; Evers et al. 2023) and temporal trends (Bignert et al. 2004; Rigét et al. 2011; Braaten et al. 2019; Morris et al. 2022a). Moreover, it is crucial to foster international collaboration and coordination among national or local projects to create harmonized regional approaches, and to strive, where possible, to integrate biomonitoring activities into a standardized framework to properly assess regional and global spatiotemporal patterns of risk to human and environmental health.
The GBMS database and associated peer-reviewed publications provide a platform to assess broad spatial scales of Hg tissue concentrations in key food items related to human health for general (e.g., tuna, swordfish) and regional fish populations (e.g., sharks, freshwater fish), Indigenous Peoples (marine and freshwater fish, toothed whales, pinnipeds) and subsistence communities (which can include all the major taxa of concern). The health of ecosystems can also be viewed through bioindicators that are not necessarily key food items but are representative of taxa where the literature is robust (e.g., sea turtles, seabirds, loons, raptors, freshwater birds, landbirds, and marine mammals). We begin each section with a brief rationale for why each taxonomic group is important for Hg biomonitoring and then discuss associated caveats. We include data from the GBMS database, which summarizes Hg data from over 1700 peer-reviewed publications, to demonstrate the breadth of biotic tissue Hg data availability (spatially and temporally) and to better understand local, regional, and global patterns that can be used as a beginning point for identifying data and knowledge gaps for effectiveness evaluation purposes of the Minamata Convention.
Below, biotic data are organized by 1) Human exposure bioindicators - those organisms which are consumed by humans and may potentially pose a risk to human health; and 2) Ecological health bioindicators – those organisms that best represent Hg impacts to ecological health (Table 1).
Human exposure bioindicators
Many Indigenous Peoples in remote places depend on their local ecosystems for sustenance. For example, Arctic Indigenous Peoples rely on access to their traditional country foods for food security, for their general health and well-being, and as part of their spiritual and cultural identity, among many other things (AMAP 2021; Basu et al. 2022). However, due to exposure through the diet, Arctic Indigenous Peoples can experience some of the highest Hg levels globally (Basu et al. 2018, 2023). In such situations, good risk communication is essential to ensure that the proper messages are conveyed in balancing the risks associated with Hg exposure against the nutritional and cultural benefits of traditional diets.
In many other parts of the world, communities depend in part, and sometimes completely, on wild animals for subsistence. The following section describes known Hg concentrations for a broad range of biota and geographic areas. Specifically highlighted with data summaries are: (1) high trophic level marine fish that are widespread across the world’s oceans: tuna, billfish, and sharks; (2) the Caribbean and Mediterranean Seas; (3) freshwater fish within six continents; (4) seabirds and waterfowl in subarctic marine systems; and (5) marine mammals (e.g., toothed whales in the northern oceans). Due to the importance of dietary Hg exposure and the global impact on human health, patterns depicting the interaction of dietary MeHg uptake in humans are herein described for all the world’s major biomes from the Arctic and subarctic to temperate and tropical aquatic ecosystems. Ingesting elevated fish muscle Hg concentrations, such as in sharks, can exceed commonly suggested reference concentrations in less than two weeks (Baek et al. 2023). Often, biotic Hg concentrations can be linked to anthropogenic Hg point sources, such as ASGM activities, which have been connected to elevated Hg levels in nearby communities (Gibb and O’Leary 2014; Basu et al. 2023).
While other environmental (e.g., contaminant mixes; Alves et al. 2022), micronutrient (e.g., selenium; Lailson-Brito et al. 2012; Gochfeld and Burger 2021; Storelli et al. 2022; Sabino et al. 2022), and nutritional factors (e.g., omega-3s; Sardenne et al. 2020) clearly can confound assessments of Hg on human health those costs and benefits are not evaluated herein. The following biotic groups illustrate how biotic Hg exposure can be linked to human exposure concerns in several key ecosystems in the world using select bioindicators.
Marine fish - tuna
Rationale and caveats for Hg biomonitoring
Tuna species are one of the most important global sources of seafood and inhabit broad areas the Atlantic, Pacific, and Indian Oceans. Commercial harvests tracked by the Food and Agriculture Organization (FAO) for the seven most commercially available species totaled 5.2 million metric tons in 2018, worth an estimated value of $41 billion; this does not include substantial value associated with subsistence and artisanal fisheries and sport fisheries (McKinney et al. 2020). Projections indicate that the global market may reach over $50 billion by 2028. However, sustainably managing tuna fisheries to allow depleted stocks to recover has been challenging and generally does not account for the adverse impacts of MeHg tissue concentrations that may reduce reproductive output and growth rates. Excessive fishing pressure continues to threaten tuna stocks of eastern Pacific yellowfin, Pacific bluefin, Atlantic bigeye, Indian Ocean yellowfin, and southern bluefin. Mercury biomonitoring deliberations should consider tracking Hg concentrations in all nine of the tuna species that average or range above the 0.22 µg/g, ww threshold of “a two-meal limit/week” (see Table 2 for human meal frequency and Fig. 2 for the Hg profile) and biomonitoring considerations should account for species differences, size classes, changes in stock abundance from overfishing, differences in foodweb structure, and size of home range (Schartup et al. 2019).
The GBMS database includes 10,722 Hg concentrations of 9 species representing 120 publications. Muscle Hg concentrations and commercial harvest vary widely by species. The smallest tuna species (e.g., skipjack tuna, Katsuwonus pelamis) has average Hg concentrations under the USEPA-USFDA advisory level of 0.23 µg/g, ww while the largest (e.g., Pacific and Atlantic bluefin tunas, Thunnus orientalis and T. thynnus, respectively) have the highest average Hg concentrations and often exceed advisory levels (Fig. 2).
Mercury concentrations in nine species of tuna and their associated global harvest. Light gray bars represent the arithmetic mean ± SD of total Hg concentrations (µg/g, ww) in muscle tissue. Dark gray bars show FAO harvests estimates (in tonnes). Tuna with harvests of <15,000 tonnes are depicted with “**” while tuna with harvest of <5000 tonnes are depicted with “*”. Data are not normalized by size. Canned tuna Hg data are not included here. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines)
These patterns vary by size class within species and ocean basin origin. For example, whereas yellowfin tuna (Thunnus albacares) tends to have lower average muscle Hg concentrations than seven of the nine tuna species with data (Fig. 2) larger individuals (e.g., weighing over 70 kg) typically have Hg concentrations that are of human health concerns (Bosch et al. 2016a). Yellowfin, bigeye tuna (Thunnus obesus), and albacore tuna (Thunnus alalunga) Hg concentrations grouped by major ocean basin indicates that the eastern and northern areas of the Pacific Ocean have significantly higher Hg concentrations than other ocean basins (Ferriss and Essington 2011; Nicklisch et al. 2017; Houssard et al. 2019; Médieu et al. 2021). This area of the Pacific Ocean is where increasing tuna Hg concentrations have been recorded over the past decade (Drevnick et al. 2015; Drevnick and Brooks 2017) and modeled for several decades into the future (Sunderland et al. 2009). Tuna Hg concentrations in other ocean basins are known to be decreasing (North Atlantic Ocean; Lee et al. 2016) or remaining stable (southwestern Pacific Ocean; Médieu et al. 2021). As well as size and origin, other interpretive factors to consider include whether the tuna is canned or fresh (for the same species; canned tuna tend to have lower Hg concentrations; García et al. 2016) and farmed vs. wild. Although farmed tuna tend to have lower Hg concentrations (Balshaw et al. 2008; Annibaldi et al. 2019), the amount of Hg bioaccumulation in muscle tissue in wild-caught, pen-raised tuna depends on time spent in rearing pens (Srebocan et al. 2007).
Marine fish - billfish
Rationale and caveats for biomonitoring
Large and relatively long-lived pelagic species such as billfishes can be used as bioindicators for understanding expansive spatial gradients of MeHg contamination in the world’s oceans using current commercial resources. Mercury concentrations in billfish, such as marlin (multiple genera; Drevnick and Brooks 2017, Vega-Sánchez et al. 2017, Bille et al. 2020, Rudershausen et al. 2023) and swordfish (Xiphias gladius, Mendez et al. 2001, Branco et al. 2007), are some of the highest known for marine teleost fish (Rodrigues and Amorim 2016) and adverse impacts to their physiology and body condition may be of concern for some populations (Biton-Porsmoguer et al. 2022). Swordfish are the most widespread of the billfish and northern hemisphere stocks are generally managed sustainability (western, central, and eastern North Pacific and North Atlantic stocks) (National Oceanic Atmospheric Administration, Department of Commerce unpubl. data) and provide a long-term opportunity for broad geographic and robust sampling options. Southern hemisphere swordfish stocks are less understood and in the case within the Indian Ocean are declining. Mercury biomonitoring deliberations should consider tracking Hg concentrations in swordfish, which average above the 0.46 µg/g, ww threshold of “choices to avoid” (see Table 2 for human meal frequency and Fig. 3a for the Hg profile) and biomonitoring considerations should account for differences among billfish species, size classes, changes in stock abundance from overfishing, differences in foodweb structure, and size of home range.
a Mercury concentration in seven species of billfish. Gray bars illustrate the arithmetic mean ± SD of total Hg concentrations (µg/g, ww) in dorsal muscle tissue. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines). b Mercury concentrations in swordfish. Gray bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in dorsal muscle tissue of swordfish from six ocean basins. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines)
The GBMS database includes over 3778 Hg concentrations of seven billfish species representing 54 publications. Of the billfish, the highest average Hg concentrations are in blue marlin (Makaira nigricans), nearly 4× global averages of the swordfish (Fig. 3a). In swordfish, Hg tissue concentrations vary according to major ocean basin with a tendency for a doubling of Hg concentrations in the Northern Hemisphere compared to the Southern Hemisphere (Fig. 3b; 0.79 ± 0.52 µg/g, ww and 0.54 ± 0.42 µg/g, ww, respectively). Elevated Hg levels in swordfish are to be expected because of their high trophic level and relatively long lifespan (>10 years). As these data illustrate, swordfish often exceed human health thresholds (see Fig. 3b vertical lines – only the South Atlantic population has mean levels below the “do not eat” threshold), making their consumption a human health concern. However, swordfish have important commercial value and are an important source of income for many oceanic island communities.
The Indian Ocean is a good case study where approximately 30,000 tonnes of swordfish are harvested annually (i.e., 25% of annual global catch during 2016–2018), half of it being caught by fleets of Indian Ocean coastal countries (FAO 2018; IOTC 2020). Sri Lanka, India, and Seychelles fisheries are the main contributors accounting for 24%, 10% and 8% of the annual total catch of swordfish in the Indian Ocean, respectively, contributing mostly to the global export market. Exports/imports of fish products are however strictly monitored when it comes to fish Hg content particularly for Europe, which is the top importing market for swordfish (FAO 2018). Indeed, the EU requires predatory pelagic fish (e.g., tuna and swordfish) imports to have <1.0 µg/g, ww of Hg for human consumption (EU Commission 2006). Total Hg concentrations in swordfish have been well investigated since the 2000s in the Indian Ocean, highlighting variable Hg concentrations depending on the swordfish size/age (largest and oldest swordfish having the highest Hg levels), and the area where it was caught. Overall, higher Hg concentrations were recorded in swordfish from the Western Indian Ocean compared to the Eastern Indian Ocean (Hg: 0.9 ± 0.1 and 0.6 ± 0.1 µg/g, ww, respectively) (Esposito et al. 2018), and from the southern Indian Ocean compared to the central-northern Indian Ocean (Hg: 2.0 ± 0.1 and 0.9 ± 0.1 µg/g, ww, respectively) (Sabino et al. 2022).
Approximately 13%, 13% and 43% of swordfish caught from the Sri Lanka, Seychelles and Reunion waters, respectively, were found to exceed the EU advisory level (Hollanda et al. 2017; Jinadasa and Fowler 2019; Kojadinovic et al. 2006). Swordfish with concentrations over this EU advisory level are not permitted for export to the EU. These large, high commercial value specimens therefore must either remain within island communities or are exported to other countries for less value. Thus, high Hg concentrations in fish can result in significant adverse economic, ecological and human health impacts, especially in the case of Small Island Developing States that rely highly on their fisheries such as the Seychelles (Bistoquet et al. 2018). Indeed, the last EU ban on Seychelles swordfish exports (2014) led the Seychelles longline fleet to favor exports of large tropical tunas over swordfish, as Hg concentrations in the tuna species within the central-western Indian Ocean are generally below 0.5 µg/g, ww (Bodin et al. 2017). However, this may not be a long-term solution due to the declining status of tuna populations in the Indian Ocean (e.g., yellowfin tuna: overfished and subject to overfishing) (IOTC 2018).
Marine fish – sharks
Rationale and caveats for biomonitoring
Sharks are a diverse and important group of marine species, as there are over 470 species of sharks that are defined within eight Orders in the subclass Elasmobrachii (i.e., elasmobranchs, which include sharks, skates, and rays), containing several top predators that are known to have cascading impacts on ecosystems they inhabit (Hammerschlag et al. 2019, 2022). Sharks are an important source of food in many cultures, and have been severely overexploited, with many species facing high extinction risk (Gallagher et al. 2012; Pacoureau et al. 2021; Sherman et al. 2023; Worm et al. 2024). Most shark species are known to contain elevated muscle Hg concentrations and their use as top trophic level bioindicators for marine ecosystems is well established. Mercury biomonitoring deliberations should consider tracking Hg concentrations in the 21 of 24 (88%) shark genera that average or range above the 0.46 µg/g, ww threshold of ”choices to avoid” (see Table 2 for human meal frequency and Fig. 4 for the Hg profile) and biomonitoring considerations should account for species differences, size classes, knowledge of prey availability, foraging depth (that can be measured with stable isotopes), and size of home range. Shark fins can be used as an indicator of Hg exposure (Kim et al. 2016; Vélez et al. 2021).
Mercury concentrations in sharks. Gray bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in muscle tissue of sharks by genus (24 genera) from the Orders of Mackerel and Ground Sharks from the GBMS database. USEPA-USFDA human health thresholds for mercury consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines)
The GBMS database shows that species within two of the eight Orders, the mackerel (Order Lamniformes) and ground (Order Carcharhiniformes) sharks generally have tissue Hg concentrations well above the human health advisory levels of no consumption set by the USEPA (0.46 µg/g, ww) and World Health Organization (1.0 µg/g, ww; Fig. 4). Implications of these elevated Hg tissue concentrations are also of concern for overall shark health, which adds to population stresses due to overfishing. Many species are on the IUCN Red List of Threatened Species and are overfished for their fins and meat.
The GBMS database includes 10,578 Hg concentrations of 24 genera of sharks. Many of the measurements are from blue (Prionace glauca), mako (Isurus spp.), great white (Carcharodon carcharias), hammerhead (Sphyrna spp.), silky (Carcharhinus falciformis), bull (Carcharhinus leucas), lemon (Negaprion brevirostris), and porbeagle (Lamna nasus) sharks, where large individuals well exceed human health advisory standards (Branco et al. 2004, 2007; Escobar-Sánchez et al. 2011; Maz-Courrau et al. 2012; de Carvalho et al. 2014; McKinney et al. 2016; Nicolaus et al. 2016; Matulik et al. 2017; Biton-Porsmoguer et al. 2018; Terrazas-López et al. 2019; Rodriguez-Gutiérrez et al. 2020; Maurice et al. 2021; Erasmus et al. 2022a, 2022b; Riesgo et al. 2023). Pelagic foraging piscivorous species tend to have higher Hg tissue concentrations compared to those foraging in benthic habitats and on invertebrates (de Pinho et al. 2002; Matulik et al. 2017). Further, the mesopelagic zone may be an important entry point for MeHg into the foodweb (Choy et al. 2009) – a zone that provides over 70% of the prey for larger species, such as the great white sharks (Carcharodon carcharias) in the northeastern Pacific Ocean (Le Croizier et al. 2020). Ultimately, trophic level of prey dictates muscle MeHg concentrations in sharks (Le Croizier et al. 2022b), but distribution of prey species in the ocean water column may also be an important factor (Choy et al. 2009; Furtado et al. 2021).
Of the 24 shark genera with published muscle Hg concentrations, the GBMS data shows that average levels exceed the USEPA human health standards of 0.46 µg/g, ww for 83% of genera and the WHO and EU standard of 1.0 µg/g, ww in 50% of genera (Fig. 4). Mercury concentrations are similar in all muscle tissue including fins (O’Bryhim et al. 2017; Kim et al. 2016), which are often also used as a basis for soup (Barcia et al. 2020). This is noteworthy given the practice of shark fin soup consumption in Asia (Worm et al. 2024).
Despite having among the highest levels of Hg recorded in any vertebrate, the physiological and behavioral effects of Hg concentrations on elasmobranchs remains largely unknown (Wosnick et al. 2023). Although chronic dietary MeHg uptake of 0.2 µg/g, ww in freshwater fish have been found to affect reproduction and other subclinical endpoints (Depew et al. 2012a), studies on the effects of MeHg in the shark brain indicate abilities to demethylate (Ehnert-Russo and Gelsleichter 2020) and potentially use detoxifying mechanisms through selenium-Hg liaisons (Branco et al. 2007; Dutton and Venuti 2019; Medina-Morales et al. 2020) or other physiological abilities (Le Croizier et al. 2020). For example, while Merly et al. (2019) found that blood concentrations of Hg in white shark (Carcharodon carcharias) exceeded levels that are known to be toxic to humans, no negative effects on shark health parameters were detected, including body condition, total leukocytes, or granulocyte to lymphocyte ratios. The authors speculated that sharks may have protective mechanisms that mitigate harmful effects of heavy metal exposure. However, only circulating blood Hg concentrations were measured, which may be more transient and less likely to impact shark health. Conversely, Wosnick et al. (2021) found that Hg concentrations in hepatic and gill tissues of sharks were associated with increased activity of alkaline phosphatase and deregulation of urea and lactate markers, respectively. The former relationship suggests possible alterations in liver-kidney functioning from Hg toxicity, while the later association suggests potential compromised gill functioning in osmoregulation. Clearly, there is a need to better understand the effects of Hg exposure on elasmobranch fitness and survival.
In addition to high Hg concentrations, as long-lived and high trophic level generalist species, sharks are prone to bioaccumulation and biomagnification of various heavy metals and other toxins (e.g., Hammerschlag et al. 2016; Shipley et al. 2021), which may additively or synergistically impact shark health and survival.
Marine fish – Mediterranean Sea
Rationale and caveats for biomonitoring
The Mediterranean Sea is a semi-enclosed area characterized by strong North-South and West-East gradients of environmental conditions with a residence time of waters of approximately a century (Millot and Taupier-Letage 2005). It covers an area of about 2,500,000 km2 (970,000 mi2) and has an average depth of 1500 m (4900 ft) with the deepest point at 5267 m (17,280 ft) in the Ionian Sea. Total captured fisheries production in the Mediterranean and Black Seas peaked in 1988 at approximately 1.8 million tonnes and although this has since fallen to around 1.2 million tonnes/yr for the period 2018–2022 (FAO 2020, 2022) fish stocks are still an important food source for local communities. Herrings, sardines, and anchovies accounted for 56% of the total landings with a mean annual amount over 665,000 tonnes, followed by miscellaneous coastal fishes (10%, 117,300 tonnes), and miscellaneous pelagic fish such as cods, hakes, and haddocks (10%, 123,500 tonnes). Catches of small pelagic species presented large fluctuations during this period linked to the variability of environmental factors, while decreasing landings were observed for some demersal species (European hake, whiting, turbot and sole) and increasing landings for a few other ones (red mullet, surmullet and blackspot seabream) (FAO 2022).
In the Mediterranean Sea, the Western Mediterranean continues to be the most productive area, accounting for 20% of the total landings, followed by the Eastern Mediterranean (15%), the Adriatic (14%) and the central Mediterranean (14%), while the Black Sea provided 38% of the total catch with 446,067 tonnes during the 2018–2020 period. Mercury biomonitoring deliberations should consider tracking fish Hg concentrations in the 24 of 36 (67%) fish families that average or range above the 0.46 µg/g, ww threshold of “choices to avoid” (see Table 2 for human meal frequency and Fig. 5 for the Hg profile) and account for differences in species distributions and abundance, as well as location within the Mediterranean Sea.
Mercury concentrations in Mediterranean Sea fish (including estuarine species). Gray bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in muscle tissue of 36 fish families from the GBMS database representing the Mediterranean Sea. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines). The double line is a break in the x-axis to better depict and view lower Hg concentrations
The GBMS dataset for the Mediterranean Sea includes 13,720 Hg concentrations for 111 fish species in 58 families – including 36 focal families– based on 59 publications (Fig. 5). These findings indicate several families of sharks – including Bonnethead/Hammerhead sharks (Sphyrnidae; x = 13.4 ± 3.5; n = 5), Gulper sharks (Centrophoridae; x = 9.66 µg/g; n = 25), Chimaeras (Chimaeridae; x = 3.14 µg/g; n = 160), Spiny dogfishes (Squalidae; x = 2.2 ± 2.4 µg/g; n = 123) - have the highest Hg concentrations for this region. Swordfish and tunas (Thunnus and Katsuwonus) have some of the highest Hg concentrations in bony fishes and average well above the USEPA safety threshold level for human exposure in sensitive populations (0.15 µg/g, ww). While, Indigenous Peoples from the Amazon and the Arctic have been found to have the highest Hg levels globally (Basu et al. 2018, 2023), non-indigenous people living in the Mediterranean region have the second highest Hg levels (Petrova et al. 2020). Most marine fish from the Mediterranean Sea have average Hg concentrations that are restrictive for safe human consumption (Cinnirella et al. 2019).
However, the lowest trophic level fish species (e.g., those that depend on zooplankton as primary prey) including herring, sardines, anchovies, and picarels, that accounted for more than half the total landings, have the lowest average Hg concentrations and are generally safe for human consumption (Fig. 5). Higher Hg concentrations of Mediterranean fish are generally recorded in larger/older individuals than in smaller/younger ones, in high trophic level predators than in low trophic level herbivores and zooplanktivores, in benthic than in pelagic species, in deeper than shallower environments, and in oligotrophic than mesotrophic waters (Cresson et al. 2014, 2015; Maulvault et al. 2016; Chouvelon et al. 2018; Sánchez-Muros et al. 2018). This explains why the short-lived pelagic zooplanktivores (Engraulidae and Clupeidae) exhibited Hg concentrations lower than the minimum USEPA threshold (<0.16 µg/g, ww), while deep demersal families (Scorpaenidae) and large, long-live pelagic predators (Xiphiidae and Scombridae) presented much higher Hg concentrations (>0.60 µg/g, ww) (Biton-Porsmoguer et al. 2022), that are restrictive for human health (Fig. 5). Demersal and deep sharks and rays also presented very high Hg concentrations in the Mediterranean Sea (Storelli et al. 2002).
Several studies have shown that Mediterranean fish species have higher concentrations of Hg in their tissues than the same species from the Atlantic Ocean (e.g., Renzoni et al. 1998; Cossa et al. 2012; Cransveld et al. 2017; Cammilleri et al. 2018; Chouvelon et al. 2018; Di Bella et al. 2018; Mauffret et al. 2023). More generally, high concentrations of Hg have been observed in Mediterranean predatory organisms, likely because the Mediterranean is one of the places in the World Ocean where Hg methylation potential is the highest (Cossa and Coquery 2005). The high Hg-enrichment in Mediterranean fish compared to other regions at the same latitudes results from the synergy of several factors: (1) the shallower location of the MeHg maximal concentration in the water column that induces a higher MeHg transfer into the biota, (2) the slower growth rates of fishes resulting in a higher age-at-length that induces a longer exposure to Hg at a given length, (3) higher concentrations in zooplankton, and (4) longer food webs linked to oligotrophic conditions and small sizes of phytoplankton cells (Buckman et al. 2018; Cossa et al. 2022).
The synergy of environmental and biological factors induces a high spatial variability in Hg concentrations of Mediterranean fishes (Cinnirella et al. 2019), exemplified here by the high standards deviations on Hg concentration means in Fig. 5. At the basin level, fishes from the Western Mediterranean appear more contaminated than those from the Eastern Mediterranean, in relation to higher MeHg concentrations in the Western basin waters (Cossa et al. 2022). At a regional scale, the areas of particular concern are the North of the Western basin (Cresson et al. 2014), the Adriatic Sea (Storelli et al. 2005; Grgec et al. 2020), the Tyrrhenian Sea (Buckman et al. 2018), some places in the Ionian Sea (Signa et al. 2017) and the Sea of Marmara (Keskin et al. 2007), while lower concentrations are reported from fishes from the Aegean Sea (Kucuksezgin et al. 2001), the Black Sea (Harmelin-Vivien et al. 2009) and the coast of Tunisia (Joiris et al. 1999).
Marine fish – Caribbean Sea
Rationale and caveats for biomonitoring
The Caribbean Sea includes numerous islands of the West Indies, and adjacent coasts of North and South America and has an area of about 2,754,000 km2 (1,063,000 mi2). The Sea’s deepest place is the Cayman Trough, between the Cayman Islands and Jamaica, at 7686 m (25,217 ft) below sea level. The Caribbean Sea has the world’s second largest barrier reef, the Mesoamerican Barrier Reef. It extends over 1000 km along the coasts of Mexico, Belize, Guatemala, and Honduras. The area generates a relatively robust fishing industry, accounting for 500,000 tonnes of fish a year (FAO 2018). Mercury biomonitoring deliberations should consider tracking fish Hg concentrations in 25 of 39 (64%) fish families that average or range above the 0.46 µg/g, ww threshold of “choices to avoid” (see Table 2 for human meal frequency and Fig. 6 for the Hg profile) and account for differences in species distributions and abundance, as well as location within the Caribbean Sea.
Mercury concentrations in Caribbean Sea fish (including estuarine species). Gray bars illustrate the arithmetic mean ± SD of total Hg concentrations (µg/g, ww) in muscle tissue of 39 fish families from the GBMS database that represent the Caribbean Sea. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines). The double line is a break in the x-axis to better depict and view lower Hg concentrations
A new monitoring effort, the Caribbean Region Mercury Monitoring Network, is now established with the laboratory hub in Antigua and Barbuda. The Network has selected key bioindicators of Hg for purposes that meet economic, human health safety, and logistical reasons for long-term Hg monitoring. The focal species include three that have average Hg concentrations below 0.22 µg/g, ww and have important local and commercial importance: yellowfin tuna (Thunnus albacares), red snapper (Lutjanus campechanus) and mahi-mahi (Coryphaena hippurus). Other species such as the great barracuda (Sphyraena barracuda) routinely have elevated Hg concentrations but are not as regularly consumed because of ciguatera fish poisoning concerns (Chinain et al. 2021). There are also multiple grouper species that are of local economic interests and should have regular monitoring of their Hg concentrations – species, size class and location are important factors for interpretation (Sinkus et al. 2021; Christian et al. 2024). For teleost fish, large pelagic species are generally of greater concern to human health than small pelagic and reef species (Shrestha et al. 1988; Ricketts et al. 2016). All shark species have mean Hg concentrations that are well above human health standards (Fig. 6) and especially for some areas such as in Trinidad and Tobago (Mohammed and Mohammed 2017).
The GBMS dataset for fish (elasmobranchs and teleosts) for the Caribbean Sea includes 8,480 Hg concentrations for 193 species in 67 families – including 39 families of greatest interest from 26 publications (Fig. 6). The findings indicate that mackerel sharks/Porbeagles/white sharks (Lamnidae; 4.3 ± 0.95 µg/g, ww; n = 4), thresher sharks (Alopiidae; x = 3.68 ± 1.29 µg/g, ww; n = 3), requiem sharks (Carcharhinidae; x = 1.81 ± 1.25 µg/g, ww; n = 866), billfish (x = 1.35 ± 2.42 µg/g, ww; n = 191) and swordfish (x = 1.14 ± 1.39 µg/g, ww; n = 3) have the highest Hg concentrations for this region. Species that have the lowest risk of Hg contamination to people include mahi-mahi, herring, sardines, lionfish (in the family Scorpaenidae), and mullets (Fig. 6: Adams 2009; Ahmed et al. 2020; Acosta-Coley et al. 2023). As part of the now established Caribbean Region Mercury Monitoring Network, a more recent analyses of over 1600 fish muscle samples for total Hg found a lower ratio of 26% of species exceeding the 0.46 µg /g, ww guideline (although few sharks and billfish were included) (Christian et al. 2024).
Areas of particular concern, which often times are related to Hg sources in the watersheds that flow into Caribbean Sea, include deltas, mangroves, and nearshore marine waters from ASGM activities in Suriname (Mol et al. 2001), from chlor-alkali facilities in Colombia (Alonso et al. 2000; Olivero-Verbel et al. 2008; Gallego Ríos et al. 2018) or other less defined sources such as long-distance transport (Guzmán and Garcı́a 2002). Based on Hg concentrations in barred grunt (Conodon nobilis) from Trinidad and Tobago, levels are generally highly elevated in the Gulf of Paria and the Colombus Channel and could be related to river runoff with Hg from ASGM activities in countries of northern South America and carried towards Trinidad by the Guiana Current (Christian et al. 2024).
Freshwater fish - Africa
Rationale and caveats for biomonitoring
The major river basins of Africa include the Nile (~6700 km or 4160 miles), the Congo (~4670 km or 2900 miles), the Niger (~4170 km or 2590 miles), and the Zambesi (~2740k or 1700 miles), while the largest lakes include Lakes Victoria, Tanganyika, and Malawi. These and other areas have nearly four million people engaged in fishing-related activities (Heck et al. 2007) and for some countries provide up to 70% of their animal protein (FAO 2012; Hanna et al. 2015). Whereas industrial Hg releases are relatively small in Africa (with the exception of coal-fired power plants in South Africa), approximately 70% of the estimated total Hg emissions and releases are associated with artisanal and small-scale gold mining (ASGM; UNEP 2019a).
Because of uncertainty of Hg in African fish, Hg biomonitoring deliberations should consider tracking fish Hg concentrations in seven of the 16 (44%) fish families that average or range above the 0.22 µg/g, ww threshold of (see Table 2 for human meal frequency and Fig. 7 for the Hg profile) and account for differences in species, size class, type of freshwater system (e.g., lake vs. river), association with small-scale gold mining activities and subsistence communities, as well as seasonality (wet versus dry season; Kouame et al. 2020).
Mercury concentrations in freshwater fish in Africa (including estuarine species). Gray bars show the arithmetic mean ± SD of total Hg concentrations (µg/g, ww) in muscle tissue of 16 teleost fish families selected from the GBMS database that represent Africa. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines). The double line is a break in the x-axis to better depict and view lower Hg concentrations
Studies documenting Hg concentrations in fish from lakes contaminated through atmospheric deposition (vs. releases of Hg into the water from ASGM activities) reveal relatively low concentrations for fish communities as illustrated in the Okavengo Delta in Botswana (Black et al. 2011), Lake Tanganyika in Tanzania (Campbell et al. 2008), Aiba Reservoir in Nigeria (Atobatele and Olutona 2015), and in rift valley lakes (Campbell et al. 2003a) including Lake Tana (Habiba et al. 2017), Lake Victoria (Campbell et al. 2003b; Drouillard et al. 2024), and Lake Malawi (Kidd et al. 2003); although only 4% of inland water bodies have been sampled for Hg concentrations in fish (Hanna et al. 2015). Importantly, Hg concentrations in Nile perch (Lates niloticus) and tilapia (representing multiple genera), the two most important commercial species, tend to be <0.5 µg/g, ww (Hanna et al. 2016; Drouillard et al. 2024). Conversely, snakeheads (Channidae) and African pike (Hepsetidae) are generally elevated and may be important for Hg biomonitoring for human health purposes.
However, local studies within ecosystems that are sensitive to Hg input indicate aquatic ecosystems in Africa can have elevated Hg levels of concern in fish and other aquatic food items used by humans, especially when associated with ASGM activities. Concentrations in high trophic level fish from lakes and rivers in Burkina Faso, Egypt, Ghana, Kenya, Senegal, South Africa, Tanzania, Zimbabwe as well as estuaries in Cote d’Ivoire have documented Hg levels of concern for human consumption (Ouédraogo and Amyot 2013; Hanna et al. 2015; Niane et al. 2015; Rajaee et al. 2015; Gbogbo et al. 2017; Walters et al. 2017; Elawady et al. 2019; Mason et al. 2019, 2022; Debrah et al. 2020; Makaure et al. 2023; van Rooyen et al. 2023).
A review of fish Hg concentrations in the GBMS database (6,126 individuals in 41 families from 183 species) from 171 locations in 21 African countries in 66 papers found mean Hg concentrations were relatively low (i.e., below the 0.22 µg/g, ww human health threshold commonly used). Sixteen families with ≥15 individuals are depicted (Fig. 7). Hanna et al. (2015) reviewed 30 studies in Africa that documented fish Hg concentrations and found that only locations near ASGM operations had mean Hg levels above recommended human health guidelines. A similar pattern was found in Ghana (n = 1305 measures in 65 species) where only sampling sites associated with ASGM had fish Hg levels that exceeded human health thresholds, especially for those species at high trophic levels (Rajaee et al. 2015; Kortei et al. 2020).
Piscivore fish species that have been identified in the GBMS database to have muscle tissue over 0.22 µg/g, ww include the saddled bichir (Polypterus endlicherii), African pike (Hepsetus spp.), African tigerfish (Hydrocynus vittatus), snakeheads, and multiple catfish species within the order Siluriformes, including species within the families of Bagridae, Clariidae, Claroteidae, Mochokidae, and Schilbeidae.
Freshwater fish - South America
Rationale and caveats for biomonitoring
The major river basins of South America, including the Magdalena, Orinoco, Amazon, and La Plata, support a large freshwater fishery, providing livelihoods for small-scale artisanal fishers as well as major commercial enterprises (Barletta et al. 2010). In the remote interior areas of South America, indigenous communities are highly dependent on freshwater resources for subsistence, and for communities with high fish consumption (FAO 2018), the risk of MeHg exposure can be high (Uryu et al. 2001; Passos et al. 2008; Oliveira et al. 2010; Olivero-Verbel et al. 2015; Hacon et al. 2020; Montaña et al. 2021). Research over several decades in the Amazon Basin has repeatedly identified a link between a diet high in fish, especially piscivorous and omnivorous species, and elevated Hg concentrations in human biomarkers such as hair (Bidone et al. 1997; Lebel et al. 1997; Castilhos et al. 1998; Boischio and Henshel 2000; Bastos et al. 2006; Faial et al. 2015; Ouboter et al. 2018; Feingold et al. 2020).
Mercury biomonitoring deliberations should consider tracking fish Hg concentrations in 17 of the 36 (47%) fish families that average or range above the 0.46 µg/g, ww threshold of ”choices to avoid” (see Table 2 for human meal frequency and Fig. 8 for the Hg profile) and accounting for differences in species, size class, type of freshwater system (e.g., lake vs. reservoir vs. river), and association with small-scale gold mining activities and Indigenous and subsistence communities. Seasonality of sample collection is also important as low water time periods result in higher fish Hg concentrations than otherwise (Nyholt et al. 2022).
Mercury concentrations in freshwater fish in South America (including estuarine species). Gray bars illustrate the arithmetic mean ± SD of total Hg concentrations (µg/g, ww) in muscle tissue of 36 teleost fish families selected from the GBMS database that represents South America. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines). The double line is a break in the x-axis to better depict and view lower Hg concentrations
Four hotspots in the Amazon Region have been identified as areas of particular concern because of the magnitude of ASGM activities since 2002 (Alvarez-Berríos and Mitchell Aide 2015): (1) the Madre de Dios region, Peru (Asner and Tupayachi 2017; Caballero Espejo et al. 2018; Diringer et al. 2020; Barocas et al. 2023); (2) the Guiana Shield region that includes French Guiana, Guyana, and Suriname; (3) the Tapajós–Xingú region (Malm et al. 1995; dos Santos et al. 2000; Nevado et al. 2010; Lino et al. 2019; Passos et al. 2008) that includes Central Amazon (Kasper et al. 2014) and the Madeira River (Bastos et al. 2006, 2015; Mussy et al. 2022; da Silva Montes et al. 2022) in Brazil; and (4) the Magdalena–Urabá region of Colombia (Ashe 2012; Hacon et al. 2014; Martinez et al. 2018; Gonzalez et al. 2019; Hacon et al. 2020). These and other Hg point sources (e.g., petroleum extraction; Webb et al. 2015) that are connected with river floodplain habitats, where daily and seasonal water level fluctuations can be extensive, appear to be sensitive to elevated methylation rates - during both droughts (Azevedo et al. 2018) and flood periods (da Silva et al. 2019).
The GBMS database for South America contains over 144 peer-reviewed publications on fish Hg concentrations from more than 319 different locations; 38,126 Hg concentrations from 350 species in 62 families are represented. The Hg dataset for fish in South America is taxonomically diverse with a description of Hg concentrations shown for 36 families with a sample size ≥15 (Fig. 8).
Of the 63 fish families with representative data, 49% have mean Hg concentrations over 0.22 µg/g, ww. Fifteen fish families exceed the USEPA human safety threshold (0.46 µg/g, ww on average) for avoiding consumption. The family Trichomycteridae (pencil or parasitic catfish) demonstrates the highest muscle Hg concentrations; this family is diverse with over 40 genera reflecting nearly 300 species. The most sampled taxa include high trophic-level species within the genus Hoplias (tigerfishes), Serrasalmus (piranhas), Pseudoplatystoma (sorubim catfishes), Cichla (neotropical cichlids), Salminus (dorado), and Hoplias (wolf fish). The GBMS dataset highlights areas of broad freshwater fish sampling on the continent, specifically in Brazil (Malm 1998; Ferreira da et al. 2019), and with some additional coverage in Colombia (Olivero et al. 1998; Salazar-Camacho et al. 2020), Ecuador (Webb et al. 2004), French Guiana (Richard et al. 2000; Gentès et al. 2019), Peru (Gammons et al. 2006; Diringer et al. 2015; Martinez et al. 2018), and Suriname (Ouboter et al. 2012; Vreedzaam et al. 2023); as well as in estuaries in Argentina (Marcovecchio et al. 2001) and Suriname (Mol et al. 2001).
From these data, it is also possible to identify ecologically sensitive hotspots of concern for ecological and human health (see Fig. 1). Much of the research on Hg to document ecological and human Hg exposure has been conducted in downstream areas potentially impacted by ASGM activities (Olivero-Verbel et al. 2015; Diringer et al. 2015; Moreno-Brush et al. 2016; Salazar-Camacho et al. 2017; Ouboter et al. 2018; Watson et al. 2020), especially when contaminated rivers flow into lakes (Lake Titicaca in Peru; Gammons et al. 2006) or reservoirs (Brokopondo Reservoir, Suriname; Ouboter et al. 2012; Tucurui Reservoir, Arrifano et al. 2018; and other reservoirs; Pestana et al. 2019). Watersheds that are downwind from ASGM activities also may have elevated biotic Hg concentrations (Gerson et al. 2022).
Freshwater fish – Asia
Rationale and caveats for biomonitoring
Mercury emissions in Asia represent some of the highest in the world, and reductions are now being attempted by countries, such as China and India, in part due to the provisions of the Minamata Convention (Sharma et al. 2019; Feng et al. 2022). While freshwater ecosystems in the vast Asian landscape are dominated by rivers in the south (e.g., Ganges, Indus, Mekong, Yangtze, and Yellow rivers) and the north (Lena and Ob Rivers) and numerous oligotrophic lakes in the north – relatively few studies have documented biotic Hg concentrations in this most water-stressed continent of the world. Mercury biomonitoring deliberations should consider tracking fish Hg concentrations in 12 of the 31 (39%) fish families that average or range above the 0.46 µg/g, ww threshold of “choices to avoid” (see Table 2 for human meal frequency and Fig. 9 for the Hg profile) and account for differences in species, size class, type of freshwater system (e.g., lake vs. reservoir vs. river), overfishing, and association with ASGM areas and rice fields.
Mercury concentrations in freshwater fish in Asia (including estuarine species). Gray bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in muscle tissue of 31 teleost fish families selected from the GBMS database that represents Asia. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines). The double line is a break in the x-axis to better depict and view lower Hg concentrations
The knowledge of Hg in freshwater fish for Asia is sparse for the size and diversity of the landscape. China is the largest nation for the consumption and export of fish and fish products; however reported Hg concentrations in fish are generally low (Wang and Wang 2019; Feng et al. 2022; Souza-Araujo et al. 2022), even in relatively new reservoirs, such as in the Guizhou Province (Yan et al. 2010a, 2010b) and in the Three Gorges Reservoir (Xu et al. 2018; Wang et al. 2019b), and when there are point sources such as abandoned Hg mines (Qiu et al. 2009) and others (Zhu et al. 2012). A comprehensive review over the past 10 years did document significant geographical differences from the north to the south: Fish in north China rivers had more Hg than those in south China (Zhang and Wong 2007); Additionally, the Tibetan Plateau exhibited the highest total Hg levels (up to 0.87 µg/g, ww) (Wang and Wang 2019). Fish Hg concentrations are generally low across China because of fast-growing farmed and stocked fish species dominance, coupling with declining wild fish populations in freshwaters where trophic level enrichment of MeHg is generally dampened by eutrophication, and water chemistry parameters (e.g., alkaline pH and low dissolved organic carbon) that are not conducive to high methylation rates (Cheng and Hu 2012; Liu et al. 2012). Minimal shoreline wetland area due to anthropogenic activities, lower trophic biomagnification factor in temperate freshwater food webs compared to boreal ones, and the general lack of trophic level 4 fish in eutrophic waters also contribute to patterns of lower fish Hg concentrations (Chen et al. 2008; Liu et al. 2012; Xu et al. 2018; Wang and Wang 2019; Wu et al. 2019; Jing et al. 2020).
Exceptions have been reported by Razavi et al. (2014) in the mesotrophic Qiandao Lake, where the food web in this remote 50-year-old reservoir of East China demonstrated a high degree of omnivory and a long food web with trophic level up to 4.9 including wild fish species. Consequently, wild fish in this reservoir had generally higher Hg concentrations (up to 1.78 µg/g, ww in the Mandarin fish (Siniperca chuatsi) than stocked fish (up to 0.58 µg/g, ww in herbivorous fish species, such as the goldfish [Carassius auratus]). Although average fish Hg concentrations from Qiandao Lake was well under 0.5 µg/g, ww, it was still significantly higher than fish from Taihu Lake and reservoirs in Guizhou. Nevertheless, a further meta-analysis of both freshwater and marine consumer fish in China over the last three decades from 1980 presented that fish Hg concentrations were decreasing despite increased Hg emission over time, a phenomenon likely due to overfishing and aquaculture (Zhang et al. 2022).
The GBMS database includes 14,093 fish Hg concentrations from 75 families representing 304 species based on 93 publications. Of those families, 31 are featured here that have sufficient sample sizes (≥12) or are otherwise of interest (e.g., high Hg concentrations) (Fig. 9). The highest Hg concentrations were in spiny eels (Mastacembelidae) with mean Hg concentrations over 2.0 µg/g, ww. Milkfish, needlefish, true minnows, and knifefish families had particularly elevated Hg concentrations, averaging over 0.5 µg/g, ww. Several families have wide variation in Hg concentration that indicate other factors such as species, size class, freshwater type, and location are important to understand prior to identifying best bioindicators.
Representation of Asian freshwater fish Hg concentrations outside of China are relatively sparse. In Russia, a summary of 21 fish species from the Oka, Moskva, Osetr, Volga, and Akhtuba Rivers found relatively low Hg concentrations with average concentrations at or under 0.26 µg/g, ww (Gorbunov et al. 2016). However, lakes sampled in Russia contained fish with relatively elevated Hg concentrations (Buck et al. 2019; Dudarev et al. 2019).
Investigation of selected Korean reservoirs from 2016 to 2020 showed low fish Hg concentrations well under 0.5 µg/g, ww in barbel steed (Hemibarbus labeo), largemouth bass, and bluegill (Jung et al. 2022). Whereas in Japan, Watanabe et al. (2021) presented 95% of salmon and trout samples contained MeHg at less than 0.05 µg/g, ww, extracted from the Japanese National Health and Nutrition Survey. Otherwise, there is no recent publicized freshwater fish Hg data in Japan since Matsunago (1975), who reported average fish Hg concentrations of 0.72 µg/g, ww in three different fish species (dace, crucian carp, and Zacco spp.) sampled in two rivers that received Hg mining waste at the time.
Freshwater fish Hg concentrations in certain South East Asia countries remain safe despite rapid urban development activities: Low fish Hg concentrations (0.051 ± 0.04 µg/g, ww) in Mekong River confirmed its pristine state of the ecosystem of Vientiane area, Laos (Guédron et al. 2014); Lobus and Komov (2016) validated that 76% of freshwater fish contained less than 0.10 µg/g, ww of total Hg in muscle tissue sampled from rivers, lakes, and reservoirs of Central and South Vietnam. Freshwater fish in Cambodia are generally low and do not exceed 0.12 µg/g, ww in striped snake-head fish (Channa striata), common climbing perch (Anabas testudineus) and peacock eel (Macrognathus siamensis), as demonstrated by Agusa et al. (2005a, 2005b). Fish Hg remain well below 0.46 µg/g, ww as seen in indicator species collected from West Bay area of Laguna Lake, in the Philippines (Cuvin-Aralar 1990).
High levels of Hg in fish stocks have been found mainly in coastal areas in Thailand, Indonesia, and India. With increasing inland industrial activities in these regions, increased freshwater fish Hg concentrations have been reported in industrial sites in Thailand: Sampled striped snakehead from Tha Tum industrial complex contained elevated fish Hg concentrations up to 0.52 µg/g, ww (Tremlová et al. 2017). Surprisingly, fish Hg was even higher up to 0.56 µg/g, ww in the same species sampled from Thap Lan National Park neighboring Prachinburi industrial park. In another scenario of adverse anthropogenic impact on fish Hg and human health, Castilhos et al. (2006) reported elevated fish Hg concentrations of 0.58 ± 0.44 µg/g, ww with more than 45% of fish having Hg levels above 0.46 µg/g, ww across 154 specimens of 10 freshwater species from gold mining areas in Tatelu, Indonesia. Fish from the Ganges River at West Bengal in India was investigated showing that wallago catfish (Wallago attu) possessed high Hg content at 0.93 ± 0.61 µg/g, ww, while small-sized fishes from the same sampling site showed low fish Hg concentrations below DL (Pal et al. 2011).
Freshwater, estuarine, and marine fish – Australia
Rationale and caveats for biomonitoring
Australia, including Tasmania and numerous islands, has rich and varied ecosystems, from desert to tropical rainforest and straddles the Indian and Pacific Oceans. Australia’s coastal seas include the world’s largest barrier reef, the Great Barrier Reef, encompassing almost 350,000 km2 off the northeast coast, and is a biodiversity hotspot. Mercury biomonitoring deliberations should consider tracking fish Hg concentrations in 7 of 18 (38%) fish families that average or range above the 0.46 µg/g, ww threshold of “choices to avoid” (see Table 2 for human meal frequency and Fig. 10 for the Hg profile) and account for differences in species distributions and abundance, as well as location across the continent.
Mercury concentrations in freshwater, estuarine and coastal fish sampled from the continental shelf in Australia. Gray bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in muscle tissue of 18 teleost and elasmobranchs families of interest selected from the GBMS database that represents Australia. Centropomidae (Snooks) includes barramundi. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines). The double line is a break in the x-axis to better depict and view lower Hg concentrations
The GBMS dataset for fish (elasmobranchs and teleosts) from Australia extending to the continental shelf (including estuarine and freshwater samples) includes 2646 Hg concentrations for 100 species in 48 families – including 18 families of greatest interest – from 16 publications (Fig. 10). The findings indicate that the five families with the highest Hg concentration are elasmobranchs, including mackerel sharks/Porbeagles/white sharks (Lamnidae; 2.82 ± 1.56 µg/g; n = 87), sleeper sharks (Somniosidae; x = 1.90 ± 1.15 µg/g; n = 106), Bonnethead and Hammerhead sharks (Sphyrnidae; 1.37 ± 0.23 µg/g; n = 212). The highest teleost families include swordfishes (Xiphiidae; 0.56 ± 0.02; n = 13), jacks (Carangidae; 0.28 ± 0.38; n = 16) and tunas, mackerels, bonitos (Scombridae; 0.24 ± 0.13; n = 146). Species that have the lowest risk of Hg contamination to people include gobies, herrings/sardines, and mojarras (Fig. 10).
Several independent research studies have been conducted to evaluate Hg concentrations in fish collected from Australian estuaries and nearshore coastal ecosystems with the focus on understanding health implications, environmental processes, and anthropogenic impacts (Gagnon et al. 2016; Maher et al. 2020; Butler et al. 2022). Mercury contamination of fish in nearshore Australian marine environments is not evident except at several locations with historical Hg contamination (Maher et al. 2020). In Australia, as in other regions like the Caribbean Sea, areas that of particular concern for elevated Hg concentrations in fish include wetland (particularly mangroves) and estuarine habitats. Butler et al. (2022) found that barramundi (Lates calcarifer) in floodplain wetlands concentrated Hg at almost twice the level of those that remained in saline habitats.
The Australian Government’s scientific research organization, the Commonwealth Scientific and Industrial Research Organization (CSIRO), has recently conducted an extensive review of published data on contaminants (including Hg) in sea turtles, birds, and marine mammals. Most samples were collected opportunistically in Australia between the 1970s and 2022 (Jarolimek et al. 2023). While long-term datasets are not available for the same species, the review provides baseline information on Hg levels in Australian marine fauna. Lastly, the Food Standards Australia and New Zealand (FSANZ) conducts the Australian Total Diet Study (ATDS) and evaluates Hg levels in a wide range of Australian foods (including seafood) with the aim to estimate the dietary exposure of the Australian population to Hg, identify risks and risk management options, and provides recommendations on the safe consumption of fish (note that Fig. 10 uses the standards set by the USEPA and USFDA (Table 2).
Freshwater fish – North America and Europe
Rationale and caveats for biomonitoring
The freshwaters of North America and Europe are extensive, and the lakes and rivers have a diverse fish community from temperate to Arctic waters. While the Great Lakes Basin in the U.S. and Canada provide a dominant recreational fishery for the region, riverine and lake fisheries across this extensive area are important contributors to local economies (Evers et al. 2011a; Wiener et al. 2012b) and for some areas, subsistence purposes such as in Arctic Inuit communities (AMAP 2021). Mercury biomonitoring deliberations should consider tracking fish Hg concentrations in 12 of the 25 (48%) fish families that average or range above the 0.46 µg/g, ww threshold of ”choices to avoid” (see Table 2 for human meal frequency and Fig. 11 for the Hg profile) and account for differences in species, size class, type of freshwater system (e.g., lake vs. reservoir vs. river), and association with Indigenous and subsistence communities.
Representative Hg concentrations in selected game fish species from North America and Europe. Gray bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in muscle tissue of 25 teleost game fish species selected from the GBMS database that represent North America and Europe. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines)
Both continents have or have had well established Hg monitoring programs that generally revolve around game fish, though there has been an absence of harmonized, national monitoring programs that span decades of time. However, monitoring efforts have generated hundreds of thousands of fish analyzed for Hg over the past 4–5 decades. In the U.S., such programs have focused on the Great Lakes, interior lakes, rivers, and streams including in the Great Lakes Fish Monitoring and Surveillance Program (Monson et al. 2011; Zhou et al. 2017) and the National Rivers and Streams Assessment (Wathen et al. 2015a, 2015b). Mercury data have also been summarized for lakes and rivers for the Northeast (Kamman et al. 2005; Millard et al. 2020), Midwest (Monson et al. 2011), and the West (Eagles-Smith et al. 2016a). In Canada, massive datasets (>300,000 Hg concentrations in the Canadian Fish Mercury database; Depew et al. 2013) describing spatial and temporal game fish Hg trends have been summarized as well (Gandhi et al. 2014; Eagles-Smith et al. 2016a). Similar efforts in Europe, especially in Scandinavia – spanning 55°–70°N, also provide definitive baseline information that can be used for temporal comparison purposes (Braaten et al. 2019).
To evaluate spatial and temporal variation of Hg in fish, the game fish Hg data are generally normalized with regard to size using well-established standard units (e.g., 55 cm and 1 kg for northern pike; Sorensen et al. 1990). Such standards have been applied to robust Hg datasets (i.e., >50,000 data points) in the U.S., Canada, and northern Europe for pike, bass, and walleye (Johnels et al. 1967; Kamman et al. 2005; Monson 2009; Monson et al. 2011; Gandhi et al. 2015; Eagles-Smith et al. 2016a) for human health assessments and using perch (Perca spp.) (Scheuhammer et al. 2016) for evaluating concerns for ecological health purposes. Other normalizing techniques include using individual fish weight and Hg concentration in combination with fish species information and sampling year to find expected Hg concentration for fish at a standard weight. In Braaten et al. (2019), multiple linear regression models were applied to describe Hg concentrations, where potential explanatory variables included fish weight, fish species, sampling year, and the interaction terms year × species and weight × species, to evaluate changes in fish Hg concentrations with weight and species over time.
Records of Hg in freshwater fish across Fennoscandia (Norway, Sweden, Finland, Kola Peninsula in Russian Federation) have been collected for over 50 years (since 1965) in almost 3000 lakes and rivers and collated into a single database by the International Cooperative Programme for assessment and monitoring of the effects of air pollution on rivers and lakes (or ICP Waters), under the UNECE Air Convention (Braaten et al. 2017) – only the peer-reviewed published data are in the GBMS database. Fish Hg concentrations vary widely among lakes in Fennoscandia owing partly to differences in local and regional Hg pollution in the lakes, but particularly factors controlling net methylation, trophic structures, and subsequent biomagnification (Braaten et al. 2019).
Measured Hg concentrations in the south (55°N–60°N) of Fennoscandia are generally higher than in the north (60°N–70°N), with over 40% of all lakes containing fish muscle Hg concentrations exceeding the WHO/FAO limit of 0.5 µg/g, ww widely used as a trigger for human consumption safety in Europe. The dataset includes important species for recreational fishing such as northern pike (Esox lucius) (South: 0.63 ± 0.01 µg/g,ww [n = 24,849], North: 0.60 ± 0.01 µg/g,ww [n = 3360]), Arctic char (Salvelinus alpinus) (South: 0.45 ± 0.18 µg/g,ww [n = 284], North: 0.11 ± 0.10 µg/g,ww [n = 514]), European perch (Perca fluviatilis) (South: 0.26 ± 0.02 µg/g,ww [n = 20,276], North: 0.20 ± 0.01 µg/g,ww [n = 2326]), and brown trout (Salmo trutta) (South: 0.14 ± 0.03 µg/g,ww [n = 1816], North: 0.16 ± 0.40 µg/g,ww [n = 230]). Half a century of fish Hg concentrations in Fennoscandian lakes have shown a clear decline (Åkerblom et al. 2014; Braaten et al. 2017, 2019). However, there is no consistent decline in lakes for which Hg originates from atmospheric sources only (e.g., Rask et al. 2024). Closing of local industrial pollution sources over the past 50 years is likely to have led to a reduction in fish Hg concentrations.
The GBMS database for North America and Europe contains over 170 peer-reviewed publications on fish Hg concentrations from more than 240 sites across 100 different waterbodies (e.g., lakes, rivers, estuaries, and bays); more than 253,667 individual fish from more than 240 genera are represented. The Hg dataset for fish is robust, especially for game fish, of which 25 species of 8 families are featured here as key bioindicators for human health purposes, including walleye (Sander vitreus), largemouth bass (Micropterus salmoides), and northern pike (Fig. 11). Of these species, 48% have a mean Hg concentration over the USEPA human health benchmark of 0.46 µg/g, ww.
Seabirds – human consumption assessment
Rationale and caveats of biomonitoring
Although hunting of seabirds is not as common globally as it once was, in some areas of the world, marine birds (waterfowl, shorebirds, and seabirds) and their eggs can still be a regular and necessary food source for remote subsistence communities. This is especially true across the circumpolar Arctic and Subarctic (e.g., Naves 2018; Otsuki et al. 2024), and in a scattering of small island nations, like Grenada (Smart et al. 2020), where alternative sources of protein may be limited at times, driving both legal and illegal consumptive harvests of seabirds.
In North America and Europe, there are several examples of both indigenous and non-indigenous harvests of seabirds and/or their eggs, some with significant cultural relevance. For example, Baffin Island in the eastern Canadian Arctic is an ecologically significant area, supporting many species of seabirds and marine mammals. Inuit communities across the Canadian Arctic, such as those on Baffin Island, rely heavily on a variety of marine resources, including seabirds (Chan et al. 1995; Mallory et al. 2004). The presence of elevated Hg in seabirds throughout the Canadian Arctic is well established (Muir et al. 1999; Mallory et al. 2004; Campbell et al. 2005; Mallory and Braune 2012; Burnham et al. 2021), with either stable or increasing Hg trends observed in seabirds in recent decades (Braune et al. 2015, 2016).
Indigenous People and subsistence hunters in Alaska also have a long history of harvesting and consuming marine resources, including seabirds and their eggs (Burger et al. 2007; Naves 2009). Mercury accumulation in seabirds from the Aleutian Islands has also been well documented (Burger et al. 2007, 2008, 2009; Burger and Gochfeld 2009; Savoy et al. 2017). According to Burger et al. (2007), 90% of households from an Aleutian village consumed birds to some degree each year. Previous studies have shown that some seabird species from the Aleutian Islands contain edible parts (e.g., breast meat, eggs) with Hg levels that approach or exceed human consumption advisory levels (0.22 µg/g, ww; Burger et al. 2007, 2009). Although estimates of subsistence harvests of seabirds may have declined somewhat across the state in recent decades, birds and their eggs remain a necessary source of nutrition in some particularly isolated Alaskan communities (e.g., the St. Lawrence–Diomede Islands; Naves 2018), as well as remaining culturally important across the region. Abundance and ease of collection mean the eggs of the glaucous-winged gull (Larus glaucescens) are highly sought after, and the number collected each year can exceed 6000 (Naves 2009). Larger gull species regularly exhibit elevated Hg concentrations within a seabird colony. The eggs of murres and gulls represent the majority of egg harvests, but the eggs of many smaller seabird species, such as auklets and terns, are also widely harvested in Alaska (Naves 2018).
The GBMS database includes 3943 individual eggs from 20 species of marine bird of interest in the Arctic and subarctic were included from 32 publications. Generally, Hg concentrations in seabirds in the Canadian Arctic are below levels associated with health effects in wildlife. Whereas edible parts for human consumption (breast muscle, eggs) (Fig. 12) may approach or exceed the action level (0.22 µg/g, ww) (Table 2). For example, a recent review of Hg concentrations in Arctic seabirds found that 50% of individuals sampled (n > 5000) showed tissue Hg concentrations exceeding 0.20 µg/g, ww (Chastel et al. 2022).
Mercury concentrations from Arctic and subarctic birds. Gray bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in eggs of 20 species selected from the GBMS database. USEPA-USFDA human health thresholds for Hg consumption (µg/g, ww) are shown as dotted (0.15) dashed (0.23) and solid (0.46) lines (see Table 2 for consumption guidelines)
Using kittiwakes (Rissa spp.), fulmars (Fulmarus spp.) and murres (Urias spp.) as bioindicators, Hg concentrations in seabird eggs from the Canadian Arctic have increased significantly over recent decades (Braune 2007; Braune et al. 2016; Burnham et al. 2021). In the Aleutian Islands of Alaska, there has been an effort to quantify MeHg uptake by local North Pacific fisheries and wildlife due to potential cumulative inputs of Hg from historic military activity (Burger and Gochfeld 2006; Anthony et al. 2007; Ricca et al. 2008), emissions from local volcanic activity (Ricca et al. 2008), and atmospheric and oceanic transport of Hg from Asia (Rocque and Winker 2004; Anthony et al. 2007; Driscoll et al. 2013) and Russia (Fisher et al. 2012).
Mercury biomonitoring deliberations could consider tracking bird egg Hg concentrations in the six seabird species that average or range above the 0.46 µg/g, ww threshold of restricted consumption (see Table 2 for human meal frequency and Fig. 12 for the Hg profile) and incorporate differences in species, interpretation of tissue type that incorporates species’ ecology, and association with Indigenous and subsistence communities.
Marine mammals – toothed whales
Rationale and caveats for biomonitoring
Toothed whales are the marine mammal taxa of greatest concern for human and ecological health because their Hg concentrations regularly exceed levels of concern for many species (Fig. 13). Although the effect levels in marine mammals are not well defined (Desforges et al. 2016), brain tissue levels are associated with neurotoxic effects (Dietz et al. 2013; Krey et al. 2015) and a study on bottlenose dolphins found lesions in the liver at 61 µg/g, ww and is being used by scientists as a benchmark for assessing ecological concern in marine mammals (Dietz et al. 2013). However, more recently groupings of marine mammals including toothed whales has been divided into groups such as “No risk, Low risk, Moderate risk, High risk and Severe risk” based on controlled experiments of harp seals (Pagophilus groenlandicus) (Dietz et al. 2022). Because liver tissue has limitations for assessing risk (e.g., levels of MeHg vary and are generally a small percentage of total Hg) and is not often consumed as a major part of the human diet, a more useful tissue to use for assessing the potential exposure of MeHg to humans is muscle tissue (AMAP 2021). Mercury biomonitoring deliberations should consider tracking muscle Hg concentrations in four of the toothed whale species that are regularly consumed and average above the 0.46 µg/g, ww threshold of ”choices to avoid” (see Table 2 for human meal frequency and Fig. 13 for the Hg profile and target species) and incorporate differences in species, home range, interpretation of tissue type that incorporates species’ ecology, and association with Indigenous and subsistence communities.
Muscle Hg concentrations in toothed whales. Bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in muscle tissue of 38 toothed whale species. Dark gray bars are for species most regularly consumed by humans. All species exceed the USEPA-USFDA human health threshold for Hg consumption of 0.46 µg/g, ww level of “choices to avoid” (see Table 2 for consumption guidelines)
Many Indigenous Peoples and subsistence communities, mostly in the Arctic, depend on the harvest of marine mammals such as beluga (Delphinapterus leucas), narwhal (Monodon monoceros), and pilot whale (Globicephala spp.). Elevated tissue concentrations of Hg in these species are of high human health concern. Pilot whale harvesting by some subsistence-oriented countries, such as the Faroe Islands (Dam and Bloch 2000; Weihe and Debes Joensen 2012) have now ended (Krümmel and Gilman 2016; AMAP 2021), while in other countries whaling remains a concern such as in St. Vincent and the Grenadines (Fielding and Evans 2014; McCormack et al. 2020).Various species of porpoises and dolphins (Aubail et al. 2013; Correa et al. 2013), as well as beaked whales (which specialize in foraging on deep water cephalopods) also generally have elevated Hg tissue concentrations (Fig. 13; Bustamante et al. 2003; Garrigue et al. 2016, 2024). Other marine mammals, such as elephant seals (Mirounga angustirostris) foraging in the mesopelagic zone, also have elevated Hg concentrations (Peterson et al. 2015) and are especially vulnerable when concentrations can increase during haul-out periods when body mass declines as they fast and breed on land (Peterson et al. 2018).
The GBMS database includes 29,816 individuals of 79 species representing 199 publications. Based on the GBMS database, over 25 species of toothed whales have average muscle tissue Hg concentrations above 1.0 µg/g, ww. Therefore, toothed whales appear to be one of the more Hg contaminated groups of marine mammals. Toothed whales have global mean Hg concentrations in muscle tissue (3.2 µg/g, ww) that are well above general recognized consumption advisory levels recognized by most national standards (most relevant for beluga and pilot whales) because of the dependence of certain Arctic human communities on them) (Fig. 13).
Canadian Inuit regions published health advice outlining the importance of traditional country foods for Inuit health and well-being (AMAP 2015). However, Hg exposure through parts of the traditional diet has been found to be of concern in two Inuit regions. For example, health officials in Nunavik found that the main source of Hg exposure in their region is beluga meat and recommend that pregnant women and those of childbearing age should decrease their consumption of beluga meat. In Nunavut, Inuit women who are or may become pregnant are advised to avoid ringed seal (Pusa hispida) liver, while ringed seal meat is recommended as a healthy alternative.
Other marine mammals are also important bioindicators for human or ecological health and should be monitored. Ringed seals are a good candidate as they are common, widely distributed, and regularly harvested (Braune et al. 2015; see marine mammal section under “Ecological Health Bioindicators” for further information about ringed seals and other pinnipeds).
Ecological health bioindicators
There are many species of fish and wildlife that are impacted by the adverse effects of elevated Hg on their physiology, behavior, and reproductive success (see summaries: Crump and Trudeau 2009; Dietz et al. 2013; Scheuhammer et al. 2015; Ackerman et al. 2016; Evers 2018; Whitney and Cristol 2017; AMAP 2021). Some are considered high profile species and are included by the IUCN on their Red List of Threatened Species, or formally listed by the United States Endangered Species Act of 1972.
The selection of the organism or suite of bioindicators depends on the objective. Taxa suitability may vary according to ecosystem interests (e.g., at habitat or biome levels of relevance), spatial gradient resolution (e.g., local, regional or global), temporal trends (e.g., short- or long-term), human or ecological health interests, endpoints of importance (e.g., reproductive impairment), known adverse toxicity thresholds (e.g., by tissue and taxa using endpoints of interest), sample availability (e.g., simple or challenging), and sampling outcome (e.g., non-lethal or lethal). A provisional list of some potential bioindicators for evaluating and monitoring environmental Hg loads for ecological health purposes can be grouped into four target biomes and their associated waterbodies and by major taxa of interest (Table 2; Evers et al. 2016). Some of these major taxa of interest for use as bioindicators are summarized below for sea turtles, birds, and marine mammals (the use of fish as indicators for Hg biomonitoring is covered under the “Human Health Bioindicators” section).
Sea turtles
Rationale and caveats for biomonitoring
Sea turtles have a wide distribution in tropical and subtropical regions, their dietary habits vary according to the species (e.g., herbivores, omnivores, and carnivores), and their lifespan is compatible with the residence time of Hg in the surface layer of the oceans (approximately 30 years) (Aguirre and Lutz 2004; Barbieri 2009; UNEP 2013; Evers 2018). These characteristics enable a spectrum of Hg concentrations through different trophic levels and facilitate the comparison among regions (Anan et al. 2001; Rodriguez et al. 2022). For instance, when examining the Hg levels in liver samples from juvenile green turtles (Chelonia mydas) from two distinct environments – one highly influenced by human activities (Bahia, Brazil) and another with less anthropogenic impact (Ceará, Brazil), the comparison revealed elevated concentrations of Hg in both the green sea turtles and their food items (algae and mollusks) from the highly affected location (Bezerra et al. 2015). Although comparisons were made with other tissues (e.g., muscle, kidney, and scutes), the liver was the only one that showed a significant relationship with the environmental concentrations, which can be explained by the role it plays in the storage and redistribution of recently ingested Hg (Schneider et al. 2013). Thus, the characteristics and function of each tissue are essential to understanding the metabolism of Hg and other metals in sea turtles.
Most Hg monitoring studies using sea turtles involved the utilization of internal organs (e.g., liver, kidney, and muscle), proving effective as estimators of environmental Hg concentrations (Bezerra et al. 2015; Rodriguez et al. 2022). However, due to their status as endangered species, this type of sampling is not viable for monitoring programs, while the use of non-invasive methods such as blood and keratinized matrices (e.g., scutes and nails) allows periodic monitoring (Sakai et al. 2000; Day et al. 2005; Bezerra et al. 2012; Rodríguez-Gutiérrez et al. 2020). Both tissue types can show more recent (e.g., blood) and longer term (e.g., scutes and nails) exposure (Day et al. 2005; Benjamin et al. 2018), characterizing bioaccumulation, patterns of temporal exposure to Hg and other trace metals (Bezerra et al. 2012; Schneider et al. 2015; Barraza et al. 2019; Villa et al. 2019). The study conducted by Day et al. (2005), found in the loggerhead sea turtle (Caretta caretta) along the southeastern coast of the United States a relationship between Hg concentrations in blood and scute samples and the foraging areas with greater contamination. Proximity to sources of contamination allows us to understand the differences between Hg concentrations in species of sea turtles, not only at the regional level but also at the global level.
According to Rodriguez et al. (2022), the high concentrations of Hg found for loggerhead sea turtles in the Mediterranean Sea compared to the Atlantic and Pacific Oceans, can be explained by the high density of submarine volcanoes and regional anthropogenic contamination (Selin 2009; Cinnirella et al. 2019; Tseng et al. 2021). Robust findings for Hg concentrations in fish and marine mammals supports the trend of elevated levels in sea turtles and the tendency to be higher than other areas within the Atlantic Ocean (Gworek et al. 2016; Kershaw and Hall 2019; Rodriguez et al. 2022).
Regions used as foraging areas by different species of sea turtles are especially important in Hg monitoring (Rodríguez-Gutiérrez et al. 2020) since diet is considered the main route of MeHg exposure (Gray 2002). Comprehending how the diet influences Hg concentrations in sea turtles allows for an understanding of the possible risks of consuming their meat and eggs (Green et al. 2010; Ross et al. 2016); generalized dietary groupings include vegetation (green sea turtles), jellyfish (leatherback sea turtles, Dermochelys coriacea), sponges (hawksbill sea turtles, Eretmochelys imbricata), and crustaceans (loggerhead sea turtle).
The GBMS database revealed that of the six species of sea turtles with tissue Hg concentrations (egg or blood), loggerhead sea turtles have the highest average egg Hg concentrations (Fig. 14). This finding holds significant implications for local communities that rely on this species’ eggs as supplemental protein (Ross et al. 2016; Guzmán et al. 2020; Tapilatu et al. 2020). Regions, where sea turtles may need to be monitored more intensively for elevated levels of Hg, include the Caribbean Sea, the Arabian Sea, and especially the Mediterranean Sea (Rodriguez et al. 2022). Areas with knowledge gaps in Hg exposure information are across the South Pacific (Rodriguez et al. 2022).
The lack of a standardized sampling methodology poses challenges in utilizing these data for global-scale environmental monitoring programs. The establishment of a standardized sampling methodology is imperative for future studies. Furthermore, using the carapace, as documented by Barrios-Rodríguez et al. (2023), is essential to standardize the collection point of scute samples due to differences among the vertebral, costal, and marginal scutes. The application of these guidelines together with a thorough assessment of characteristics such as diet, distribution area, contamination sources near foraging areas, and the age range of the individuals under study would allow a distinct comprehension of the origin of Hg in sea turtles. This approach would also aid in delineating species’ disparities and investigating worldwide Hg contamination.
Seabirds – ecological health assessment
Rationale and caveats for biomonitoring
Seabirds occupy a broad range of trophic levels, but most seabirds occur high in the food web and therefore biomagnify elevated concentrations of Hg and are therefore important bioindicators (Monteiro and Furness 1995; Mallory and Braune 2012; Elliott and Elliott 2013; Provencher et al. 2014; Gilmour et al. 2019a, b; Albert et al. 2021). Seabirds permit Hg monitoring across large geographical scales and variations within the same species or family over longitudinal (e.g., brown noddy, Anous stolidus, for the intertropical zone, and Adélie penguin, Pygoscelis adeliae, for the circumantarctic area (Cusset et al. 2023) or latitudinal scales (e.g., skuas and jaegers for both southern and northern hemispheres; Albert et al. 2022; Carravieri et al. 2017; Fleishman et al. 2019). The use of different tissues with different integration time (e.g., generally blood reflects short-term exposure and adult feathers reflect long-term exposure) constitute relevant approaches to provide integrated values of Hg contamination over different time scales. The variation in Hg contamination in seabird tissues can thus reveal differences in the degree of contamination between major ocean basins, as well as latitudinal gradients of contamination within basins, and trends at a series of spatial and temporal scales.
Mercury concentrations in adult body feathers generally reflect the bird’s exposure since the last molt. The significant remobilization of Hg stored in internal tissues during molting leads to depuration of up to 90% of the Hg stored since the last molt (Agusa et al. 2005a, 2005b; Braune 1987; Honda et al. 1986). Interpretation of feather Hg concentrations can be challenging as levels can relate to MeHg dietary uptake from different foraging sites from where the bird has been (breeding, migratory, or wintering) and prey items, level of stress, and age. Mercury biomonitoring deliberations could consider tracking adult feather, blood and egg Hg concentrations in Procellariforms or other seabirds (Fig. 15) and incorporate differences in ocean basins, proper interpretation of tissue types as associated with species’ ecology, movements, and diet. Tracking selenium body burdens is also important when interpreting toxicity of MeHg to seabirds (Cruz-Flores et al. 2024).
Global Hg concentrations in seabirds. Stacked bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in three tissues: light gray (fw in feathers; n = 5314), gray (ww in blood; n = 2872) and dark gray (ww in eggs; n = 1051) of four seabird families within the Order Procellariformes
Use of feather Hg concentrations from chicks is difficult as they are challenging to interpret without accounting for age because rapid chick growth rates disassociate Hg in feathers with internal tissues (Peterson et al. 2019) as well as carryover from adult body burdens (Carravieri et al. 2023). However, it provides the advantage to reflect local contamination from the food brought by the adults to feed their chicks (Blévin et al. 2013). Mercury from multiple tissue types can be converted into a standard unit such as blood equivalencies to assess changes over time in seabirds (Pollet et al. 2022).
Due to their foraging strategies, behavioral ecologies, and life-history traits (e.g., breeding sequence, molting strategies, foraging ranges, migration patterns), seabirds generally have elevated body burdens of Hg that can ultimately impact their fitness, reduce their reproductive capacity and affect their population sizes over time (Braune et al. 2006; Tartu et al. 2013; Goutte et al. 2014a, b; Bond et al. 2015; Bauch et al. 2022; Chastel et al. 2022). For instance, particular concerns arise for adverse Hg effects on the increasingly rare ivory gull (Pagophila eburnea, Braune et al. 2006; Bond et al. 2015) and there is evidence of physiological harm to the near-threatened, black-vented shearwater (Puffinus opisthomelas; Soldatini et al. 2020) and sublethal effects on immunity, liver function and breeding parameters in an Antarctic seabird – the brown skua (Stercorarius antarcticus) (Ibañez et al. 2024). Many studies have focused on seabird Hg tissue concentrations from tropical to polar regions and from coastal to oceanic zones, covering most of the world’s oceans. Nevertheless, the South Pacific and other areas of the Southern Hemisphere appear to be less documented while the Arctic has received special attention, partly because seabirds are a food resource for human populations (AMAP 2011; Albert et al. 2021; Schneider et al. 2023).
The GBMS database shows that seabirds exhibit a wide range of Hg concentrations across tissue types (e.g., feathers, blood, eggs), driven by spatial differences, trophic ecology as well as phylogeny. For example, penguins generally have the lowest Hg concentrations in feathers, blood, and eggs, whereas Procellariiforms (e.g., petrels, shearwaters, storm-petrels, and albatrosses) generally have the highest Hg body burdens (Fig. 15). Because of their large diversity, the Procellariiforms are a well-studied group and display a wide range of tissue Hg concentrations that reflect phylogenetic and physiological differences with albatrosses exhibiting the highest Hg concentrations (Muirhead and Furness 1988; Stewart et al. 1999; Anderson et al. 2010; Tavares et al. 2013; Bustamante et al. 2016; Cherel et al. 2018; Mills et al. 2020).
The most important factor for predicting seabird Hg exposure is their foraging ecology (e.g., Carravieri et al. 2014). Because seabirds use a wide range of habitats, from the coastal margins to the open ocean, species or individuals with differing foraging behaviors can reflect Hg contamination from different parts of the ecosystems both horizontally (e.g., coastal and oceanic food webs) and vertically (i.e., benthic, epipelagic, and mesopelagic food webs). Therefore, the study of a group of seabirds with contrasting ecology from the same region allows determination of MeHg availability for multiple marine zones and thus provides a more holistic view (Ochoa-Acuña et al. 2002; Bond and Diamond 2009; Stenhouse et al. 2018; Pollet et al. 2023). For example, crustacean-feeding seabirds have lower Hg exposure than cephalopod- and fish-feeders (Carravieri et al. 2014) and epipelagic seabirds have lower Hg exposure than those relying on mesopelagic prey (Ochoa-Acuña et al. 2002; Furtado et al. 2021). Seabirds of the highest trophic levels (e.g., albatrosses or skuas) are therefore at risk to the effects of MeHg toxicity that are associated with potential long-term population declines and potentially can be impacted by co-occurring contaminants such as persistent organic pollutants (Goutte et al. 2014a, b) or by deficiency in selenium, which protects against Hg toxic effects (Manceau et al. 2021b).
Based on the GBMS database, storm-petrels breeding in the Northern Hemisphere have feather Hg concentrations that are ten-fold higher (13.8 ± 3.7 µg/g) than populations breeding in the Southern Hemisphere (2.1 ± 1.8 µg/g). Such a difference is not found for the Procellariidae (8.4 ± 7.4 vs 11.1 ± 12.5 µg/g, respectively). Differences between hemispheres could be explored further using seabirds with similar trophic ecology as well as close phylogeny.
Loons/Divers
Rationale and caveats for biomonitoring
Species within the Order Gaviiformes (loons or divers) are piscivores that breed on freshwater ponds and lakes in temperate and Arctic ecosystems of the Northern Hemisphere. In the winter, all loon species migrate to marine ecosystems (with parts of some populations overwintering on freshwater lakes). The two largest loon species (common loon and yellow-billed loon, Gavia adamsii) are obligate piscivores and accordingly have some of the highest average Hg body burdens of birds in the world. Mercury biomonitoring deliberations could consider tracking adult and egg Hg concentrations in all loon/diver species, with an emphasis on the two largest species (Fig. 16) and account for differences in body size, age, sex, prey availability and waterbody type (e.g., lake or reservoir).
Mercury concentrations in loons (n = 4797). Gray bars illustrate the arithmetic mean ± SD of global total Hg concentrations (µg/g, ww) in common and yellow-billed loon indexed blood across parts of the northern hemisphere (from 10–180° W) from the GBMS database. Environmental concentrations that have a 10% or more reduction in reproductive success (e.g., EC10) are based on Table 3b
Loons have been used as bioindicators of MeHg availability in both their breeding and wintering areas for several decades for the common loon (Meyer et al. 1998; Scheuhammer et al. 1998; Evers et al. 1998, 2003, 2008, 2011a; Burgess et al. 2005; Jackson et al. 2016; Schoch et al. 2020) and more recently for the yellow-billed loon (Evers et al. 2014) and red-throated loon (Gavia stellata; Eriksson et al. 1992; Schmutz et al. 2009). The effects of Hg on loon reproductive success are well established (Burgess and Meyer 2008; Evers et al. 2008, 2011b; Depew et al. 2012b) and are used as benchmarks for evaluating ecological concern in piscivorous birds.
In Canada, the common loon and its prey are being monitored to evaluate the success of national regulatory standards to reduce Hg emissions (Scheuhammer et al. 2016); and recent findings indicate continued adverse reproductive impacts from Hg across Canada (Tozer et al. 2013), including an annual loss of 0.01 fledged chicks per territorial pair over the past 40 years in Ontario due to MeHg burden in prey (Bianchini et al. 2020). Loons are being used as a standard bioindicator across the United States as well (Evers et al. 1998, 2003). Based on original research (Evers et al. 1998), and supported by other datasets over the past two decades, the GBMS data was used to demonstrate a west to east increasing gradient of MeHg availability in lakes within temperate and boreal forest ecosystems, with Alaskan breeding populations having the lowest Hg concentrations and eastern North American populations the highest (Fig. 16). The smaller loon/diver species, while less piscivorous and having lower Hg concentrations (Jackson et al. 2016), remain potential bioindicators for MeHg availability across their ranges, especially in Scandinavia (Eriksson et al. 1992; Eriksson 2015).
Raptors
Rationale and caveats for biomonitoring
Birds of prey, or raptors, comprise a large and varied group of birds generally characterized as predators. Several raptors at the species (e.g., osprey, Pandion haliaetus) or genus (Haliaeetus eagles) levels have near global distribution and so are commonly used in spatial and temporal contaminant monitoring efforts (Bowerman et al. 2002; Hollamby et al. 2004; Weech et al. 2006; Grove et al. 2009; Henny et al. 2009; DeSorbo et al. 2018; Sun et al. 2019; Bjedov et al. 2023). In breeding areas, developing nestlings of many raptor species are often more efficiently captured for tissue sampling than resident adults – even though chick feather Hg concentrations are challenging to interpret. Nestlings can be effective for spatial and temporal Hg monitoring since they reflect exposure over a well-defined period of nesting development (e.g., six weeks), while adult exposure is more easily linked to risk (Ackerman et al. 2016; Evers 2018).
Adult raptors consistently exhibit higher Hg concentrations than nestlings, largely due to nestlings’ ability to depurate MeHg into growing feathers (Ackerman et al. 2011) and the bioaccumulation that outpaces depuration and demethylation, especially for older individuals. In both age groups, individuals sampled in association with nesting territories generally reflect MeHg exposure in the food web from that territory (Bowerman et al. 1994; DeSorbo et al. 2018). Greater MeHg availability associated with increasing elevational gradients (DeSorbo et al. 2020) or varying across subpopulations (Sun et al. 2019) have also been shown for Haliaeetus eagles. Mercury biomonitoring deliberations could consider tracking adult, nestling, and egg Hg concentrations in piscivorous raptors (e.g., Pandion and Haliaeetus species) and some terrestrial raptors (e.g., Accipiter and Falco species) (Fig. 17) as long as an accounting of differences in body size, dietary habits, and habitat type use among species is considered.
Piscivorous raptors, namely osprey and Haliaeetus eagles, are well-suited for Hg biomonitoring within and across multiple habitat types (marine, estuarine, river, lake) (Jackson et al. 2016; Rumbold et al. 2017; DeSorbo et al. 2018; Sun et al. 2019). Haliaeetus eagles are key sentinels in environmental programs used to monitor spatial and temporal Hg exposure patterns in North America, particularly in the U.S. Great Lakes ecosystem (Bowerman et al. 2002) and in Fennoscandia (Sun et al. 2019; Gómez-Ramírez et al. 2023). A study of bald eagles (Haliaeetus leucocephalus) in the Great Lakes of the United States found evidence that Hg adversely affects a proportion of this population (Rutkiewicz et al. 2011); in that study, 14–27% of individuals sampled were exposed to Hg at concentrations associated with subclinical neurological damage.
The GBMS database reveals that piscivorous raptors such as osprey and Haliaeetus eagles tend to exhibit the highest adult blood Hg concentrations among raptors (Fig. 17). Raptor species specializing in bird prey (e.g., many Accipiter and Falco spp.) generally have higher average Hg concentrations (Keyel et al. 2020) than those predominantly targeting small mammals (e.g., Buteo and Circus spp.) (Bourbour et al. 2019), while obligate scavengers are generally exposed to low levels of Hg (Herring et al. 2018).
While piscivorous raptors were predominantly emphasized in past Hg biomonitoring, recent studies show that MeHg is also prevalent in terrestrial-based food webs, and that invertivorous birds (Passeriformes) can have elevated MeHg concentrations (Jackson et al. 2011a, 2015; Evers 2018) that can result in levels of concern in raptors such as Accipiters and falcons that feed on those birds (Newton et al. 1999; Barnes and Gerstenberger 2015, 2019; Bourbour et al. 2019; Keyel et al. 2020). Studies that documented sublethal dietary MeHg exposure in captive American kestrels (Falco sparverius) demonstrated neurotoxic impacts (Bennett et al. 2009) and reproductive harm (Albers et al. 2007) and provide threshold benchmarks for wild populations. Other foodwebs of raptors should also be considered and mindfully assessed – for example, the trophic transfer in a novel foodweb of some striated caracara populations (Phalcoboenus australis), which focused on southern rockhopper penguins (Eudyptes chrysocome) for parts of the year, resulted in highly elevated Hg concentrations (Balza et al. 2021).
Freshwater birds
Rationale and caveats for biomonitoring
As conferred, freshwater habitats are often conducive to MeHg production and bioaccumulation, and freshwater birds are among the most numerous non-marine birds exposed to naturally elevated levels of MeHg contamination. There are numerous freshwater bird species that are appropriate for long-term sampling for environmental Hg biomonitoring (Fig. 18). Among these, Forster’s terns (Sterna forsteri), Caspian terns (Hydroprogne caspia), and Clark’s grebes (Aechmophorus clarkii) are the most Hg contaminated bird species in western North America (Ackerman et al. 2016). Shorebirds can also be highly exposed to Hg contamination in breeding (Ackerman et al. 2007; Eagles-Smith et al. 2009a; Hargreaves et al. 2010; Perkins et al. 2016; Chastel et al. 2022; Perkins et al. 2023) and wintering areas (Lucia et al. 2014; Burger et al. 2018; Correia et al. 2023), and small differences in foraging strategies can result in large differences in Hg concentrations among species (Ackerman et al. 2007).
Other freshwater and estuarine birds of interest for Hg biomonitoring include kingfishers (Evers et al. 2005; Zamani-Ahmadmahmoodi et al. 2009; Hurtado et al. 2023; Oliveira et al. 2023; Pisconte et al. 2024), rails (Cumbee et al. 2008; Tsao et al. 2009; Ackerman et al. 2012; Casazza et al. 2014), and wading birds such as herons (Goutner et al. 2001), night-herons (Henny et al. 2002, 2007), ibis (Mullié et al. 1992; Klekowski et al. 1999; Heath and Frederick 2005), and egrets (Frederick et al. 1999, 2002; Sepúlveda et al. 1999a, 1999b; Rumbold et al. 2001; Zamani-Ahmadmahmoodi et al. 2010).
Documented adverse effects of Hg are known for shorebirds (Ackerman et al. 2008; Eagles-Smith et al. 2009a; Lucia et al. 2012), terns (Hoffman et al. 2011; Herring et al. 2010; Braune et al. 2012), rails (Heinz et al. 2009; Ackerman et al. 2012), and wading birds (Heinz et al. 2009; Sepúlveda et al. 1999a, 1999b; Frederick and Jayasena 2011) and therefore provide baseline information that can support the interpretation of meaningful Hg trends. As with other bird species, monitoring programs for freshwater birds could include tracking adult Hg concentrations in blood and eggs and should account for differences in sexes, sampling dates, locations, and habitats.
Landbirds
Rationale and caveats for biomonitoring
Many species of invertebrate-eating birds (herein called landbirds) are also at elevated risk to Hg exposure. Remarkably, landbirds can exhibit higher tissue Hg concentrations than fish-eating birds within the same ecosystem (Evers et al. 2005; Kopec et al. 2018; Sayers et al. 2023). They may also be more sensitive to MeHg, resulting in a higher likelihood of adverse impacts on reproductive success (Heinz et al. 2009; Jackson et al. 2011b; Whitney and Cristol 2017). An increasing number of studies characterizing Hg exposure in songbirds (Passeriformes) are demonstrating that certain clades are at higher risk than others, based largely on foraging behavior, and breeding habitats (Cristol and Evers 2020). Generally, gleaning, flycatching, and “predatory” songbirds that breed in wetland habitats (Edmonds et al. 2010; Jackson et al. 2011a, 2015, 2020; Hartman et al. 2013; Ackerman et al. 2016; Pacyna et al. 2017; Ackerman et al. 2019), including estuaries (Lane et al. 2011; Kopec et al. 2018; Sayers et al. 2021), rice fields (Abeysinghe et al. 2017; Xu et al. 2024) and tropical evergreen forest floodplains (Sayers et al. 2023) are at highest risk of Hg exposure, especially species that forage on predaceous arthropods such as spiders (Cristol et al. 2008; Janssen et al. 2023). The availability of MeHg to tropical resident songbirds (and other landbird groups) is increasingly becoming more evident and are more elevated for certain foraging guilds and habitat types (Lane et al. 2013; Townsend et al. 2013). Sayers et al. (2023) analyzed over 1800 individual neotropical Passeriformes and found warblers, woodcreepers, antbirds, and wrens to have the most elevated body burdens of Hg.
Mercury biomonitoring deliberations could consider tracking adult Hg concentrations in blood or feathers of landbirds, with an emphasis on families known to exceed thresholds of interest (0.7 µg/g blood Hg), including 8 of 21 (38%) passerine families (see Table 3b for thresholds and Fig. 19 for Hg profiles) and incorporate differences in body size, prey availability and habitat type. A similar analyses of passerine blood Hg data collected within the new initiative called Tropical Research for Avian Conservation and Ecotoxicology (TRACE) Initiative found 23 of 51 (45%) Neotropical families with individuals exceeding thresholds of interest (Sayers et al. 2023).
Mercury concentrations in in 21 families of songbirds in the United States (n = 11,606). Gray bars illustrate the arithmetic mean ± SD of total Hg concentrations in blood (µg/g, ww) from the GBMS database. For EC level definitions see Table 3b
Songbird species that spend most of their annual life cycle within wetland-oriented ecosystems and that migrate long-distances (e.g., neotropical migrants or Palearctic migrants) may also be at great risk of chronic Hg exposure adversely impacting migratory and reproductive success (Jackson et al. 2011b; Varian-Ramos et al. 2014; Ma et al. 2018a). New findings on elevated Hg exposure and migration physiology/behavior indicate significant adverse impacts are possible, especially for long-distance migrants that may experience decreased flight endurance (Seewagen et al. 2016, Seewagen 2020; Ma et al. 2018b; Branco et al. 2022) that could also be related to increasing flight feather asymmetry in high Hg individuals (Herring et al. 2017). Models including ones predicting Hg exposure to neotropical migrants now demonstrate that warblers (Paruilidae) are a particularly vulnerable group (Sayers et al. 2023). The GBMS data can be used for quantifying broad trends in different taxa – such as the higher Hg burdens in some songbird families (e.g., Troglodytidae, Emberizidae, and Icteridae)) versus those that likely forage more on seeds and berries from upland habitats (e.g., Cardinalidae, Calcariidae and Fringillidae).
Marine mammals – toothed and baleen whales, pinnipeds, and polar bears
Rationale and caveats for biomonitoring
Recent studies suggest that Hg concentrations in marine mammals have increased approximately 20 times relative to pre-industrial concentrations (Dietz et al. 2009; AMAP 2011). Of the four broad groups of marine mammals (toothed and baleen whales, seals and other pinnipeds, and polar bears, Ursus maritimus), toothed whales (Odontoceti) generally have the highest Hg body burdens (Fig. 20). Toothed whales include about 88 species of whales, dolphins, and porpoises and prey on higher trophic level organisms than baleen whales. Seals and other pinnipeds, such as walruses (Odobenus rosmarus) and Stellar’s sea lions (Eumetopias jubatus), are distributed around the world’s oceans and can also serve as biomonitoring options. Mercury biomonitoring deliberations should consider tracking muscle Hg concentrations in toothed whale and various pinniped species (Fig. 20) and account for differences in species, home range, ecology, and interpretation of tissue types. Lastly, Hg biomonitoring in polar bears has been used to provide insight into temporal changes in MeHg availability in Arctic ecosystems and should also be included in Arctic biomonitoring programs.
Compared to baleen whales, toothed whales generally forage higher on the food web and as a result average 47 times higher Hg concentrations (Fig. 20). Average Hg concentrations in many species of toothed whales are highly elevated — and potentially high enough to cause physiological, behavioral, or reproductive harm (Wagemann et al. 1998; Wagemann and Kozlowska 2005; Dietz et al. 2013, 2022).
The species of greatest concern from Hg contamination, based on existing GBMS data, include the false killer whale, killer whale, short-finned pilot whale, pygmy killer whale, striped dolphin, Risso’s dolphin, bottlenose dolphin, and various species of beaked whales (all with average Hg concentrations greater than 5.0 µg/g, ww in the muscle; Fig. 13). One reason for these elevated concentrations, in addition to their high trophic position, is that cetaceans cannot depurate MeHg through hair formation such as in polar bears and seals or through feathers by birds (Dietz et al. 2013). In general, Hg concentrations observed in small toothed whales (e.g., the Franciscana and Guiana dolphins) are considered low, and less relevant, when compared to those found in larger odontoceti, especially delphinids. However, it is important to note that these small dolphins weigh almost half, or less, than the weight of most delphinids (e.g., bottlenose dolphin) and therefore, because of increased metabolism in smaller species, the doses of Hg can be similar when accounting for individual size. Elevated concentrations of muscular Hg have been observed in endangered species of odontoceti, representing yet another risk factor impacting their populations (Manhães et al. 2022).
Some pinnipeds, such as the ringed seal are considered medium-trophic level predators and rely on large zooplankton, epibenthic and under-ice crustaceans, and pelagic and demersal fishes including Arctic cod and polar cod (Boreogadus saida and Arctogadus glacialis, respectively) for their diet (e.g., Lowry et al. 1980; Weslawski et al. 1994; Wathne et al. 2000). Other species, including bearded seals (Erignathus barbatus) and walrus, are more omnivorous, with diets that include benthic invertebrates (Born et al. 1981). This variation in diet influences the level of Hg exposure. Spatial variation in Hg concentrations in ringed seals suggests that the central and western Canadian Arctic is higher in Hg than other Arctic regions including Alaska, Greenland, Norway, and Russia (Rigét et al. 2005; Brown et al. 2016). Mercury in seals has also been shown to vary seasonally and is linked to variations in sea ice cover. Periods of greater sea ice are related to higher Hg concentrations, in part because seals are more reliant on fish. During warmer seasons when sea ice is reduced, seals can forage on a broader range of prey items, effectively reducing their exposure to Hg (Houde et al. 2020).
Polar bears live most of their lives on sea ice, hunting pinnipeds and other marine mammals. Seasonal variation in polar bear Hg exposure is related to sea ice fluctuations and availability of prey (Morris et al. 2022b). Higher Hg concentrations have been observed in spring and autumn, whereas low Hg concentrations have been observed during winter months when prey are more difficult to locate (AMAP 2011). Research has shown sex-specific differences in Hg levels but also Hg sensitivity (Bechshoft et al. 2016). While adult female polar bears have been found to have higher Hg concentrations in hair than adult males, adult males appear to be more sensitive with regards to Hg-related health responses. Also, in polar bear offspring, males were found to have higher Hg levels (Bechshoft et al. 2016). The different Hg concentrations in adult polar bears may be due to sex-specific variability in the diet. It has been suggested that female bears generally target smaller prey items, such as ringed seals (mostly fish-eaters) that are higher in Hg while males prey upon larger pinnipeds, such as bearded seals and walruses, that primarily forage on mollusks (Bechshoft et al. 2016). Males may also have reduced Hg exposure because of greater consumption of blubber, which is low in MeHg. Elevated Hg concentrations in female polar bears and the transfer of MeHg to the polar bear fetus represent a potential long-term conservation concern that could affect future populations of polar bears (Bechshoft et al. 2016). Like persistent organic pollutants, MeHg is transferred from mother to fetus in polar bears and the potential impacts require attention and further research (Cardona-Marek et al. 2009; Knott et al. 2012; Bechshoft et al. 2016).
As observed in ringed seals, spatial variation in Hg concentrations in polar bears indicates hotspot areas in the Canadian Arctic Archipelago and northwestern Greenland (e.g., AMAP 1998; Brown et al. 2016; Dietz et al. 1998, 2000, 2013; Routti et al. 2011, 2012; AMAP 2021, Dietz et al. 2022). These hotspots have recently been linked to the presence of elevated MeHg in the upper 400 m of the water column (AMAP 2021, Dietz et al. 2022).
Discussion
The GBMS database compiles relevant data at multiple geographic levels that can be useful platforms for understanding the breadth of existing biotic Hg data around the world. As illustrated above, the existence of a standardized, queryable database allows for data to be analyzed according to a variety of relevant factors. Geographically, the world’s ocean basins and continents are suitable regional scales, although further reduction in area would likely be more manageable and insightful. Taxonomically, bioindicators of human exposure and/or ecological health that are important for policy decisions within the Minamata Convention are now identified, although their selection depends on many factors identified in this paper.
The relationships among past and ongoing biomonitoring programs and databases (that exist at multiple spatial and jurisdictional levels), and how they can support the flow of data that are both comparable and sufficient to meet specified requirements are the core of international policy-related assessments. In this synthesis, the analyses have focused on assembling information currently available in the public domain into relevant categories that help inform the overarching monitoring-related interests within the Minamata Convention. This analysis provides insight into gaps in spatial coverage of data around the world, which in turn can facilitate the prioritization of cost-efficient and strategic global biomonitoring frameworks. An example would be a global monitoring framework distributed across continents and ocean basins that ultimately can reflect changes in environmental Hg loads directly related to human health and the environment (Fig. 21).
Conceptual diagram of the relationships and process for integrating existing data from biomonitoring programs (e.g., Arctic Monitoring and Assessment Program [AMAP], Asia Pacific Mercury Monitoring Network [APMMN], and the Caribbean Region Mercury Monitoring Network [CRMMN]) to design appropriate biomonitoring frameworks to inform global policy objectives of the Minamata Convention
Existing biomonitoring programs
Biomonitoring programs exist worldwide, particularly in developed countries in the northern hemisphere (e.g., U.S., Canada, across several European countries, South Korea, and Japan). However, only a few programs track long-term patterns of both environmental inorganic Hg (e.g., air) and the bioavailability of MeHg (e.g., fish). Existing biomonitoring programs were identified in a United Nations Environment Programme review (UNEP 2016). For the UNEP compilation, Hg biomonitoring programs were identified following a formal global request. Responses were compiled and provide the most up-to-date record of existing local, regional, and global abiotic and biotic Hg monitoring programs. These include programs underway within many national networks, including initiatives in Brazil, Canada, Colombia, Japan, Norway, Poland, South Korea, Spain, Sweden, United Kingdom, and the United States as well as a few regional and global networks (UNEP 2016). Data from these monitoring programs are in the GBMS database if they were published in peer-reviewed journals.
Some parts of the world are covered by long-established regional monitoring programmes. Examples include the Arctic, through the Arctic Monitoring and Assessment Programme (AMAP 2021) which is largely based on national programs such as Canada’s Northern Contaminants Program (NCP) (Chételat et al. 2015) and comparable programmes in other Arctic countries. The AMAP assessments also utilize data from other relevant monitoring initiatives such as the ARCTOX program for tracking Hg in seabirds (Albert et al. 2019). Other established international monitoring programmes exist for the European regional seas: the Northeast Atlantic (OSPAR Convention), Baltic Sea (HELCOM Convention) and Mediterranean Sea (Barcelona Convention).
There are several programs in the temperate regions of the United States (e.g., the U.S. Environmental Protection Agency’s seafood Hg monitoring program), European national programs (e.g., ROCCH, Réseau d’Observation de la Contamination Chimique), and Japan (e.g., Japanese National Institute for Environmental Studies). However, in developing countries and countries in economic transition there are fewer national or regional long-term initiatives. There are very few long-term Hg biomonitoring efforts in tropical biomes that include global development priorities such as Small Island Developing States or Least Developed Countries.
A review of the geographical coverage of Hg biomonitoring networks reveals a general lack of regional initiatives around the world, especially in Africa and Australia (UNEP 2016), which is notably similar to coverage of Hg concentrations in humans as well (Basu et al. 2018, 2023). Most Asian countries are minimally involved with national initiatives to monitor Hg levels in biota, notable exceptions being Japan and South Korea where more extensive programs exist and may be expanded by the Asia Pacific Mercury Monitoring Network (APMMN; www.apmmn.org). Conversely, Hg biomonitoring is ongoing in many countries within Europe, Oceana and across the Western Hemisphere. Also, Environmental Specimen Banks can be used as monitoring tools to provide long-term trends for contaminants in the environment, including Hg (Day et al. 2014; Paulus et al. 2015; Qiu et al. 2015; García-Seoane et al. 2017). Data that were generated from these monitoring programs and were published in peer-reviewed journals are now housed in the GBMS database.
To provide sustainable and long-term biomonitoring capacity in key regions (e.g., Arctic, tropical areas associated with ASGM, and Small Island Developing States), it could be valuable to focus on stabilizing and expanding existing local and national initiatives that use sample sizes with sufficient statistical power for understanding spatial gradients (such as biological MeHg hotspots; Evers et al. 2011b) and temporal trends (Bignert et al. 2004; Rigét et al. 2011; Morris et al. 2022a). Moreover, international collaboration and coordination among national programs could help create harmonized regional approaches and integrate biomonitoring activities into an interdisciplinary framework to assess environmental and human health risk. This creates an efficient, hierarchical framework whereby regional efforts can then be amalgamated to represent global spatio-temporal patterns. Mercury scientists from around the world, comprising the Open-Ended Science Group (OESG) and Technical Experts, are currently collaborating to compile global data to support the first effectiveness evaluation required by the Minamata Convention (UNEP 2019b, 2022).
A recent example of robust Hg monitoring efforts happening at a local level that are linked to local and regional policy regulations to lower Hg emissions and releases from anthropogenic sources is within New York State in the United States. Here, over 47,000 Hg concentrations have been collected in biota over nearly a 50-year period alongside detailed measurements in atmospheric deposition (both wet and dry) across the state (Evers et al. 2020). However, it was not until this information was collected into a standardized database designed to answer specific, policy-related questions that it was possible to adequately assess the status and trends of Hg contamination and risk, and to design more cost-effective monitoring programs in the future. By assembling long-term tracking using standard tissues and species of fish and birds, it was possible to identify important declines in environmental Hg loads (Adams et al. 2023). Several similar efforts have assembled vast amounts of Hg data into U.S. regional databases and serve to demonstrate the value of such synthesis for the northeastern United States and eastern Canada (Evers and Clair 2005), Great Lakes Region of the United States and Canada (Evers et al. 2011a), and western United States and Canada (Eagles-Smith et al. 2016b).
Although not included in the original Article 19 list of bioindicators, some invertebrate taxa are emerging as effective tools for tracing temporal and spatial trends in Hg risk to ecosystems. In particular, the interagency (US Geological Survey, National Park Service, US Fish and Wildlife Service) Dragonfly Mercury Project (Eagles-Smith et al. 2020) is the largest ongoing (2011-present) Hg monitoring network in the United States. Mercury in dragonfly larvae have been shown to be strongly correlated with those in paired fish and amphibians from the same water body (Eagles-Smith et al. 2020), as well as biogeochemical factors associated with Hg cycling (Willacker et al. 2023). As such, these bioindicators can also be used to better understand the factors and mechanisms influencing MeHg bioavilability to food webs. Additionally, an Integrated Impairment Index has been developed for dragonfly larvae that equates their Hg concentrations to various ecosystem health risk benchmarks (Eagles-Smith et al. 2020).
Linking existing biota Hg data and biomonitoring programs with objectives of the Minamata Convention
Biomonitoring programs and summarized biotic Hg data can provide information to respond to three of the four identified policy questions under the Effectiveness Evaluation process established in Article 22 of the Minamata Convention (UNEP 2019b):
i. Have the actions taken by Parties of the Minamata Convention resulted in changes in mercury supply, use, emissions, and releases into the environment?
ii. Have those changes resulted in changes in levels of mercury in the environment, biotic media and vulnerable populations that can be attributed to the Minamata Convention?
iii. To what extent are existing measures under the Minamata Convention meeting the objective of protecting human health and the environment from mercury?
To answer these questions, biomonitoring will help with: (1) establishing linkages between Hg source types and biota, (2) describing spatial gradients, and (3) tracking temporal trends.
Establishing linkages between Hg source types and biota
A promising new method for linking major Hg source types and Hg found in biota is the use of variations in stable Hg isotope ratios (e.g., Blum et al. 2014; Kwon et al. 2014, 2020; Li et al. 2016, 2022; Renedo et al. 2020; Manceau et al. 2021c). Mercury has seven stable isotopes and undergoes mass fractionation following many different patterns of isotope ratio variation during chemical reactions. The most widely used isotopic ‘signals’ of sources and chemical processes are: mass dependent fractionation (MDF), odd isotope mass independent fractionation (odd-MIF), and even isotope mass independent fractionation (even-MIF). The magnitude of the three ‘signals’ as well as the ratios between them can be combined to distinguish Hg sources and chemical processes in the environment (Blum and Johnson 2017).
By measuring the isotopic ratios of Hg in environmental samples, certain linkages can be established, and others can be eliminated in investigations of Hg sources (Le Croizier et al. 2020). Conclusive source receptor-relationships are challenging for Hg, even when using isotopes, in part due to the extensive re-emission from the earth surface back to the atmosphere after initial deposition (Outridge et al. 2018). So, while the method is often not definitive by itself, by combining isotopic data with other information based on Hg concentrations and chemical speciation, the evidence considered together can be conclusive. There are many examples in the literature where Hg isotopes have been used to separate the origin of Hg from global gaseous Hg background, global precipitation, coal burning facilities, chlor-alkali facilities, gold mining, and other industrial sources – particularly at local scales (Obrist et al. 2018).
Several studies have shown that local atmospheric sources of Hg from industrial output can be identified in precipitation and in gaseous Hg because they contrast in isotopic composition with globally well-mixed atmospheric reservoirs (Sherman et al. 2012). Similarly, industrial inputs of Hg to rivers, lakes, and marine coastal areas can often be distinguished from natural background Hg and atmospherically deposited Hg based on isotopic composition (Donovan et al. 2014). Mercury isotopes have also been used as an indicator of Hg methylation and demethylation rates and hotspots within ecosystems, and more broadly as a tool for understanding Hg biogeochemistry (Donovan et al. 2016). In situations where at least two isotopically distinct sources of Hg are present, Hg isotopes have also been used to trace the source of Hg in biota and humans (Sherman and Blum 2013).
To link Hg sources with changing Hg concentrations in biota is complicated due to the complex post-emission processing of Hg (i.e., between emission/release and uptake in the food chain; Li et al. 2022). However, efforts have been made to make the link evaluating Hg burdens in different biotic media. For example, in the Fennoscandian fish Hg database more than 3000 lakes were classified per dominant Hg pollution source based on expert judgment, including (1) lakes with no local Hg pollution sources, implying that atmospheric deposition of Hg is the dominating pollution source, and (2) lakes with known local industry point source(s) (Braaten et al. 2019). The data indicated that for the point-source lakes (2), the temporal trends showed a significant long-term decreasing trend between 1965 and 2015. However, since 1995, the temporal trends were not decreasing, indicating that most of the change in concentrations happened earlier. The authors argue that, in Fennoscandia, a peak in industry emissions and releases occurred during the 1950s and 60 s and since the 1980s local emissions and releases in Fennoscandia have been reduced significantly. This is more recently confirmed in an evaluation of the effectiveness of Norwegian Hg regulations and policies (i.e., the National Mercury Assessment for Norway; Braaten et al. 2022) where the official governmental total emissions of Hg to the atmosphere and releases to soil and water are documented. The reasons for the decline in discharge and emissions in Scandinavia are, in addition to regional and national control legislation, improved technology and reduction of polluting industrial production.
Describing spatial gradients
The availability of MeHg to high trophic level organisms can vary widely in relation to environmental conditions. Some ecosystems are more sensitive to inorganic Hg input than others (Driscoll et al. 2007; Eagles-Smith et al. 2016a, 2018; Branfireun et al. 2020) and it is these areas where biological MeHg hotspots (or ecosystem sensitive areas) can form and are especially pronounced in higher trophic-level organisms (Evers et al. 2007). For terrestrial ecosystems, such areas are generally associated with wetlands and other temporally wetted habitats and can be particularly pronounced in ecosystems with water chemistry variables such as low pH, moderate to high dissolved organic carbon concentrations, low to moderate primary productivity, or availability of sulfur (Bishop et al. 2020). In particular, fluctuating water levels can have an important contribution in generating higher methylation rates and increases in MeHg bioavailability (Evers et al. 2007; Willacker et al. 2016); and may happen at daily (e.g., tidal), monthly (e.g., artificial reservoirs and pools), or seasonal (river floodplains and dry tropical areas flooded during the wet season) timeframes, as well as in areas where water levels are managed (e.g., rice agriculture).
Therefore, determining which areas may have elevated MeHg availability requires consideration of other environmental factors in addition to the deposition or release of inorganic Hg into the environment. Globally, models can now identify the sensitivity of ecosystems and therefore areas of greatest concern (Evers et al. 2023). Because of an understanding of environmental factors that drive methylation rates in temperate ecosystems in North America and Europe, as well as a history of abiotic and biotic Hg data collection there are many good examples that quantitatively assess the dynamics between atmospheric deposition of Hg and the ecological response in lakes.
For example, Kejimkujik National Park (Nova Scotia, Canada) experiences relatively low precipitation-weighted mean concentrations and deposition of total Hg (<5 ng/L and <7.5 µg/m2/y, respectively; Dastoor and Larocque 2004; Dastoor et al. 2015), yet biotic MeHg accumulation is some of the highest in North America (e.g., 0.30 and 3.0 µg/g, ww in fish muscle and bird blood, respectively; Evers et al. 1998; Burgess and Hobson 2006; Burgess and Meyer 2008; Wyn et al. 2009, 2010). Fennoscandia is another example where the contemporary global “background” levels of atmospheric Hg deposition support Hg concentrations in fish that often exceed healthy advisory levels, even after half a century of decline in the measured fish Hg levels of the region (Braaten et al. 2017, 2019). Most lakes and catchments in the area are sensitive to inorganic Hg input and have high methylation potential and MeHg bioavailability because of important habitat characteristics including a combination of low pH, high dissolved organic carbon, high percentage of hydrologically-connected wetlands, low primary productivity, and in some cases catchment disturbance such as forest management or beaver activity (Bishop et al. 2009; de Wit et al. 2014; Eklöf et al. 2018; Negrazis et al. 2022). Ultimately, the identification of biological MeHg hotspots for freshwater and terrestrial eocsystems can be improved via better standardization of existing biotic data (Evers et al. 2011b; Ackerman et al. 2016; Eagles-Smith et al. 2016a) to inform modeling of ecosystem sensitivity at multiple spatial scales (Evers et al. 2023).
In marine regions, spatial patterns in biological MeHg concentrations are less resolved but will be facilitated by the development of a global biotic database of Hg concentrations in marine species and supporting modeling efforts to help explain observed spatial patterns. Differences in MeHg concentrations across ocean basins are clear from the literature. For example, Médieu et al. (2021) reported a five-fold spatial gradient in total Hg concentrations in albacore tuna across the North Pacific Ocean. This trend is driven by local anthropogenic Hg release along the Asia coast – where total Hg concentrations in albacore tuna are highest. Globally, the highest concentrations of MeHg in seawater have been reported in some regions of the Southern Ocean, which also have elevated concentrations of MeHg in some food webs (Cossa et al. 2011). Considerable spatial variability in seawater MeHg concentrations has been reported among other ocean basins, with highest levels in subsurface waters of the most biologically productive areas (Cossa et al. 2009; Sunderland et al. 2009; Bowman et al. 2014, 2016; Munson et al. 2015; Kim et al. 2017). The Arctic appears to have higher concentrations of MeHg in near-surface seawater, which may reflect unique microbial activity resulting from the combination of stratification, freshwater discharges, and ice cover (Lehnherr et al. 2011; Heimbürger et al. 2015; Schartup et al. 2015a). Much work remains to gather more data from data-poor basins (see Tables 5 and 6), and link MeHg production areas in the ocean to tissue concentrations in marine biota.
Tracking temporal trends
Models simulating the deposition of Hg from anthropogenic emissions at global scales (using several anthropogenic emissions scenarios) indicate a best scenario of a decrease of up to 50% in the Northern Hemisphere and up to 35% in the Southern Hemisphere by 2035 relative to 2010 (Pacyna et al. 2016). Although tracking Hg emissions, deposition, and releases are important tools for understanding patterns of environmental Hg loads (Sundseth et al. 2017), the relationship between modeled (or measured) deposition and MeHg concentrations in biota is poorly understood in both freshwaters and oceans, and usually when measured divergence in Hg trends in air and biota is likely (Wang et al. 2019a). Reasons for this divergence are often unexplained but can sometimes be linked to shifts in trophic structure and dietary preferences initiated by invasive species (Lepak et al. 2019). Observations of long-term trends are critical for improving the understanding of the linkages and can be viewed through fish, birds, and marine mammal studies using data within the GBMS database.
Trends in inorganic Hg concentrations are thought to differ among ocean basins because anthropogenic emissions have strongly declined in North America and Europe, leading to large declines in atmospheric concentrations, especially in the Atlantic Ocean (Zhang et al. 2016). Lee and Fisher (2016) postulated that this may also explain observed declines in Atlantic bluefin tuna MeHg concentrations between 2004 and 2012 in the North Atlantic Ocean – which are supported in measured Hg concentrations in blue marlin (Makaira nigricans) (Rudershausen et al. 2023). In a Norwegian study, Braaten et al. (2020), argued for a link between declines in Hg in fish in pristine lakes since the 1970s and reduced sulfate deposition in northern Europe. Mercury trends in biota (downward) and sediment (upward) indicated a disconnect between lake Hg loading and food web Hg bioaccumulation. The authors suggested that reduced sulfate deposition constrains substrate availability for sulfate-reducing methylating bacteria (causing reduced food web MeHg exposure despite increased Hg loading to the lake).
The relationship of changing fish MeHg concentrations in different ocean basins is germane to a better understanding of the geographic origins of Hg in seafood by country or region. For example, in the U.S., 45% of population-wide MeHg exposure originates from open oceans (particularly the Pacific Ocean), 37% from domestic coastal ecosystems, and 18% from aquaculture and freshwater fisheries (Sunderland et al. 2018). While, in the North Pacific Ocean, both atmospheric emissions and freshwater discharges of Hg have been growing on the Asian continent leading to increased Hg levels (Amos et al. 2013, 2014; Streets et al. 2009; Sunderland et al. 2009; Zhang et al. 2015). Most recent data indicate the rate of growth in Hg emissions has been slowed by widespread implementation of emissions controls on new coal-fired utilities (Giang et al. 2015; Streets et al. 2017, 2019). While temporal data on fisheries in the North Pacific are more limited some researchers have suggested that there is evidence for increases in tuna MeHg concentrations over recent decades (Drevnick et al. 2015), which is further supported by additional analysis of bigeye tuna for the same area (Drevnick and Brooks 2017).
In freshwaters, the regional fish observations across the northeastern United States (e.g., Millard et al. 2020; Richter and Skinner 2020) and Fennoscandia (Braaten et al. 2017) stand out as examples of both the potential and challenges of long-term monitoring of biota. In the northeastern United States, records are decades long for fish. The patterns here are complex, despite documented decreases in atmospheric deposition and regional Hg emissions to the atmosphere (Evers et al. 2020). While fish of some species have shown long-term declines, gamefish (e.g., largemouth bass) have had stable Hg concentrations (Richter and Skinner 2020). In the Adirondack, New York region within the United States, comparative surveys of lakes sampled a decade apart in the 2000s showed Hg increases in fish (Millard et al. 2020). The wide variation of responses seen in the biota of the northeastern United States reveal the role of environmental factors on Hg biomagnification, including climate and dissolved organic carbon concentrations in surface waters.
In Fennoscandia, the half century of records dating back to the mid-1960s is composed of fish collected from different lakes, different species, and different sampling methods. Nonetheless, the large number of fish and the decades of Hg data make it possible to reasonably associate both regional declines in atmospheric deposition and reductions in local pollution sources have led to reduced levels of Hg in fish (Braaten et al. 2019). Such large, long-term data and associated analyses are a resource for both designing new, long-term programs and the interpretation of existing datasets compiled from different sources to achieve better spatial and temporal coverage.
While freshwater fish Hg data regularly exist for the North American and European continents because of intensive field sampling efforts linked to human exposure concerns since the 1960s (Johnels et al. 1967), birds are also useful bioindicators of those and other continents and biomes. Multi-decadal Hg biomonitoring in birds is well-established in North American lakes using blood, feathers and eggs in breeding common loons (Evers et al. 1998, 2003; Meyer et al. 2011; Schoch et al. 2020), in eggs for herring gulls for the Great Lakes (Blukacz-Richards et al. 2017), and in eggs for seabirds in the Arctic (Braune et al. 2016; Bianchini et al. 2022) and Atlantic Canada for the Leach’s storm petrel (Calvert et al. 2024).
Through the use of feathers from museum specimens, birds can also provide a temporal profile that commonly exceeds one century. Recent studies that have combined retrospective Hg analyses of museum feathers with their contemporary counterparts from field samples are particularly important. For example, from the tundra of Alaska’s North Slope feather Hg concentrations from 1845 to 2012 indicated a doubling of Hg body burdens for the yellow-billed loon with projections of a four-fold increase by 2050 (Evers et al. 2014). Another retrospective study across Canada and northeastern United States found feather Hg concentrations from 1871 to 2015 from the rusty blackbird (Euphagus carolinus) to have substantially increased by 10-fold or more (Perkins et al. 2020).
To investigate changes in Hg over the past century in the Arctic landscape, Dietz et al. (2011) analyzed Hg in polar bear hair from northwest Greenland during 1892–2008. Mercury concentrations showed yearly significant increases of nearly 2% over that nearly 120-year time period. No change in trophic levels over this period was detected from stable isotopes, so changes in feeding patterns do not explain the change in Hg exposure. These trends were in accordance with an earlier review (Dietz et al. 2009). The latter study examined the literature concerning the long-term changes of Hg in humans and selected Arctic marine mammals and birds of prey since preindustrial times (i.e., before 1800 A.D.), to determine the anthropogenic contribution to present-day Hg concentrations and the historical timing of any changes. The authors calculated historical trends in Hg concentration in hard tissues of various Arctic biota. They found that “on average, 92% of the present-day Hg in Arctic wildlife is likely to be of anthropogenic origin” (AMAP 2011), while studies across tissues and species from the circumpolar Arctic generally did not demonstrate a consistent trend during the last 30 years or so (Rigét et al. 2011; Morris et al. 2022a, b). This is related to varied responses in different tissues (e.g., muscle vs. liver tissues) and that biota Hg concentrations are influenced by many factors, including changes in the food web (Morris et al. 2022a).
Framework for global mercury biomonitoring
To develop a sustainable and long-term global biomonitoring framework that could link existing biotic Hg data and biomonitoring programs with objectives of the Minamata Convention, several criteria could be considered, including: (1) stabilizing and expanding existing monitoring programs, (2) identifying areas that have regional data gaps so new programs can be purposely launched that define relevant sample sizes necessary for understanding spatial gradients that incorporate ecosystem sensitivity (Evers et al. 2011b) and temporal trends (Rigét et al. 2011; Morris et al. 2022a, b), and (3) identifying an existing queryable global environmental database platform to serve as a standardized and interpretive source of biota Hg information (e.g., United Nations Environment Program’s World Environment Situation Room; www.wesr.unep.org).
Moreover, international collaboration and coordination among national projects (emphasizing ratified countries) could create harmonized regional approaches and would integrate biomonitoring activities into an interdisciplinary framework to assess ecological and human health risks. Using the example of successful programs such as the Northern Contaminants Program (NCP) in Canada for the development of other national programs may also aid in building capacity of Indigenous Peoples in monitoring and research activities, which can lead to more robust monitoring and a more comprehensive understanding on Hg levels in the ecosystem and their possible drivers in addition to helping guide policy development (Houde et al. 2022). By developing a hierarchical framework, data compilation would be easier and regional and temporal trends could be assessed. Based on an information document for the Conference of Parties for their fourth meeting of the Minamata Convention on Mercury (UNEP 2022), guidance on monitoring of Hg and Hg compounds to support evaluation of the effectiveness can be grouped to achieve six objectives: (1) Estimation of contemporary Hg concentrations for areas without (i.e., background sites) or with (i.e., affected sites) local anthropogenic sources; (2) Identification of temporal trends; (3) Characterization of spatial patterns; (4) Estimation of source attribution; (5) Estimation of exposure and adverse impacts, and; (6) Quantification of key environmental processes to improve our understanding of cause-effect relationships.
Based on the knowledge of existing biotic Hg exposure, data availability is generally sufficient for tracking temporal trends and spatial gradients for all major taxa as bioindicators for both human health and the environment in the Arctic (AMAP 2021), as well as for fish in Canada and Europe (covering parts of the boreal and temperate mixed forests). There are some long-term Hg monitoring programs that include birds in North America (e.g., loons [Gavia spp] in temperate lakes of the United States and Canada; Scheuhammer et al. 2016; Evers et al. 1998, 2005, 2011a, 2020) and in the Arctic and subarctic (Fort et al. 2017). Retrospective MeHg analyses of museum bird feathers (Frederick et al. 2004; Head et al. 2011; Evers et al. 2014; Perkins et al. 2020) and mammal fur, whiskers, and baleen (Dietz et al. 2011) are a promising approach for expanding options for examining temporal trends in many regions of the world. Therefore, in the interest of using comparable data for relevant terrestrial biomes and associated aquatic areas, based on existing data (See Tables 5 and 6), we suggest that a matrix of available data and museum specimens can respond sufficiently to initial overarching questions related to temporal trends and spatial gradients.
A generalized assessment of global Hg data availability describes areas where existing data gaps are most notable - such as within the tropical rainforest biome and associated marine areas (Table 7). These areas are most problematic when coupled with Hg releases from artisanal small-scale mining activities and other major Hg source types. Information for marine mammals is generally missing as well, except for the Arctic Ocean. The preferred choice of trophic level 4 or higher bioindicators by biome and general ecosystem type (i.e., land, freshwater, marine) is influenced by objective (e.g., linking human Hg exposure, source types, understanding spatial gradients and tracking temporal trends) and several other factors (e.g., practicality, sustainability, comparability, and cost effectiveness are all factors to consider for Hg monitoring in biota).
One way to make a relatively standard comparison of global Hg exposure in high trophic level fish species is to assess the percentage that may exceed human health standards. Based on the GBMS database, the percentage of fish, seabirds (based on eggs), and marine mammals that average over or include individuals that exceed 0.46 µg/g, ww of total Hg in edible tissue can be determined for taxonomic groups of concern to people (Table 8). Notably, over half (or nearly half) of the species, genera or families include individuals that exceed 0.46 µg/g, ww in tuna, billfish, sharks, marine fish in the Mediterranean and Caribbean Seas, and freshwater fish in North and South America, and Europe, as well as marine mammals. Africa has the greatest number of fish families that are considered safe to consume by humans (for at least one meal per week). A global analysis of freshwater fish for muscle Hg concentrations within the GBMS database (n = 312,335) indicates that 45% of the 131 families include individuals that exceed 0.46 µg/g, ww.
A further breakdown of global Hg exposure as indicated by biota can be viewed by biome. In the Arctic, standard bioindicators have been selected by AMAP to monitor Hg for human health and the environment and represent a long-term existing dataset and confidence for future coverage (AMAP 2021). In the taiga and boreal areas of the Northern Hemisphere comparable Hg data are available (because of relatively similar game fish species) in Canada, the United States and Fennoscandia. The practicality and sustainability of Canada’s NCP and those directed by the other country’s respective governments makes the operation of standardized Hg monitoring programs cost-effective (Depew et al. 2013; Gandhi et al. 2014). The major exception for these northern biomes is Russia (Morris et al. 2022a).
For temperate biomes in the western hemisphere, existing (or recent) efforts are primarily in place in parts of the United States (e.g., state efforts such as in New York; Millard et al. 2020) and parts of Europe (Braaten et al. 2019) for freshwater ecosystems and some marine areas – although they rarely reflect long-term datasets and are generally not standardized across states, provinces, and countries. However, regional efforts in the Great Lakes and national efforts in United States rivers have provided standardized abiotic data over time (Wathen et al. 2015a, 2015b; Grieb et al. 2020). Southern hemisphere Hg biomonitoring efforts in temperate biomes are not as strong as the northern hemisphere and if added could contribute to the knowledge of hemispheric Hg cycling (Chen and Evers 2023).
In tropical and subtropical areas, few Hg monitoring efforts and datasets are in place. Environmental Hg-related research has been significant in some countries, such as Brazil and China, but are not as robust for Hg biomonitoring as in temperate areas. The practicality, sustainability and comparability are also all challenging because of limited infrastructure and history of monitoring activities. In addition, ecosystems and habitats that are susceptible to creating elevated levels of MeHg availability that may or may not be associated with ASGM contamination include reservoirs (Ouédraogo and Amyot 2013), estuaries (Diop and Amara 2016), and large wetlands (Daso et al. 2015). River deltas and estuaries are especially of interest as they have high methylating abilities and fisheries resources that are important to local communities. There are very few data from these ecosystems along the African coast, which are among many examples where more investigations could help to fill the extensive data gaps on the African continent (see Table 6). In particular, human and ecological exposure to MeHg is thought to be generally lower in African versus North American and European temperate lakes (i.e., the “tropical African mercury anomaly”; Black et al. 2011). Since MeHg biomagnification rates appear to be similar in African lakes to temperate and Arctic lakes (Kidd et al. 2003), the selection of bioindicators needs to be cautiously made in geographic areas considered to have high sensitivity to Hg input (Evers et al. 2023). Ultimately, tropical ecosystems are especially limited with Hg biomonitoring programs.
Across ocean basins (outside of the Arctic and Antarctic Oceans), commercial fisheries for tuna and billfish provide a platform for long-term, sustainable, and cost-effective monitoring of Hg based on existing and regular capture opportunities that can be coupled with sampling (Esposito et al. 2018; Médieu et al. 2023). Nearshore fish monitoring signifies Hg concentrations that may differ from those in offshore more pelagic fish, especially when considering the complexity and variable processes related to offshore Hg deposition, methylation, bioavailability, and biomagnification (Médieu et al. 2021). In response, high trophic level species such as barracuda, snapper and grouper are important bioindicators for evaluating nearshore MeHg availability to fish (Christian et al. 2024).
Mercury biomonitoring will need to incorporate potential confounding impacts from global climate change (Pinkney et al. 2015; Sundseth et al. 2017; Schartup et al. 2019; Bishop et al. 2020; Sonke et al. 2023), which is supported by findings in marine ecosystems (McKinney et al. 2015; Sundseth et al. 2015; Wang et al. 2023a, 2023b; Bargagli and Rota 2024), Arctic ecosystems (McKinney et al. 2015; Sundseth et al. 2015; Chételat et al. 2022; McKinney et al. 2022; Grunst et al. 2023), subarctic and temperate lakes (Chen et al. 2018), temperate estuaries (Jonsson et al. 2017; Willacker et al. 2017), and terrestrial temperate (Eagles-Smith et al. 2018) and tropical ecosystems (Yang et al. 2023). Specific effects of global climate change that impact MeHg availability include enhanced air-seawater exchange, melting of polar ice caps and glaciers, increased thawing of permafrost, and changes in estuarine sulfur biogeochemistry. However, how these landscape processes relate to changes in biotic Hg exposure is relatively unknown. Sunderland et al. (2018) demonstrated that global climate change is altering fish harvest MeHg exposures in species such as cod and pollock that are sensitive to climate-driven warming of seawater.
Iterative efforts to link realistic and applied biomonitoring efforts at local levels with regional science-policy groups aimed at assisting the Conference of Parties of the Minamata Convention may ultimately help keep pace with the many emerging scientific findings that may fill existing information gaps. As the overall understanding of source types and their ecosystem linkages, spatial gradients, and temporal trends, and the interest of using bioindicators for human exposure and ecological health that reasonably reflect terrestrial, freshwater, and marine environments, two overarching global biotic Hg monitoring approaches have been identified for continents and oceans; these approaches are partly described below and are more fully detailed in Evers and Sunderland (2019).
Continental framework for integrated mercury monitoring
To identify the best locations for global Hg monitoring requires multiple defined steps (Fig. 22). An initial step is to understand the complexities of a landscape and its ability to methylate Hg and make it available in the foodweb. Net mercury methylation rates are generally high in wetlands – particularly in estuarine wetlands such as mangroves, peatlands, and lake or rivers with shoreline wetlands especially those associated with fluctuating water levels. Forested areas are also an important factor for increasing dry deposition rates of atmospheric Hg in temperate (Driscoll et al. 2007; Obrist et al. 2018) and tropical (Gerson et al. 2022) ecosystems, while agricultural areas that introduce large amounts of phosphorus and nitrogen to freshwater systems tend to dampen methylation levels by promoting biodilution (Chen et al. 2008; Lavoie et al. 2013). As many of the most important wetland areas in the world are identified and protected through the Ramsar Convention (https://www.ramsar.org/), their 2,341 locations covering 252,489,973 ha, along with ASGM activities (e.g., Steckling et al. 2017), FAO data, and Red List species as identified through the International Union for Conservation of Nature (https://www.iucnredlist.org/), can be feasibly combined with the GBMS database to further advance analytical assessments. For example, the identification and potential overlap of ecosystem sensitivity spots with priority lakes, rivers and wetlands may help prioritize areas of greatest concern for protecting human health and the environment. Similarly, summarizing information by watershed has proven to be an important base area for mapping, providing additional hierarchical structure whereby the choice of the most appropriate scale for analysis (i.e., choice of hydrologic unit codes [HUC], and land-use impacts) will depend on the specific objectives being assessed (Evers et al. 2023).
Stepwise components for developing a conceptual continental approach using biota for Hg monitoring (see Evers and Sunderland 2019)
Oceanic framework for integrated mercury monitoring
The approach for monitoring Hg in oceanic areas greatly differs from the continental approach (Fig. 23). The cycling and movement of Hg in the world’s oceans varies by hemisphere, basin, ocean depth, and juxtaposition with the continental land masses and associated river deltas. Therefore, Hg concentrations in fish, birds, and marine mammals vary significantly. For example, bluefin tuna (representing three sibling species – the Atlantic, Pacific, and Southern) have average Hg concentrations in their muscle tissue across six ocean regions that may vary three-fold (Fig. 2). Reasons for this variation differ and need to be accounted for when globally monitoring Hg in oceanic areas.
Stepwise components for developing a conceptual oceanic approach using biota for Hg monitoring (see Evers and Sunderland 2019)
Based on the GBMS database, the species of highest Hg concern with the greatest interest for human consumption are the larger tuna species and swordfish. The Hg concentrations in tuna vary greatly by species (Fig. 2) because of their growth rates, ultimate size, age, trophic level, and ocean basin and therefore species selection is important. Médieu et al. (2023) provide a template for monitoring Hg in tuna, to standardize data collection and reporting, and also suggest other environmental variables that could be integrated into monitoring to aid in interpretation of spatial and temporal trends. Coastal fish biomonitoring would generally include different species – for the Caribbean a new Hg biomonitoring network has identified four key species (barracuda, mahi-mahi, and various snapper and grouper species (Christian et al. 2024). While commercial harvest of some of the highest-trophic level fish is important for global Hg biomonitoring, perhaps some of the most vulnerable populations are Indigenous Peoples and subsistence communities who depend on a broad variety of biota for their local, traditional diet – which includes many species with established highly elevated MeHg body burdens in the Arctic and subarctic (Basu et al. 2018, 2023), within tropical systems (Salazar-Camacho et al. 2020), and across oceanic islands (Sabino et al. 2022).
Conclusions
Efforts to assemble disparate but related biotic Hg data into standardized and comparable databases are essential for understanding the patterns and trends of Hg exposure, and for informing critical policies designed to lower Hg risks and impacts to ecosystems and people. Building from a history of successful regional efforts in North America, we describe the use of existing monitoring programs and a first effort to assemble a standardized global database of published, peer-reviewed Hg concentrations in biotic tissue - called the Global Biotic Mercury Synthesis (GBMS). We use this database to identify critical knowledge gaps and describe adoption of specific quantitative and replicable approaches to create harmonized biomonitoring efforts that can be developed and made available to countries. We provide examples of how to standardize efforts to document where, when, how, and what to monitor for tracking environmental Hg loads, their changes over time, and potential impacts on human and ecological health.
To illustrate the value of GBMS data to environmental policies, we present new syntheses of global Hg data in relation to Minamata Convention objectives. Our findings demonstrate that while there are a few large biological Hg datasets, they generally do not provide the ability to determine changes in biotic Hg exposure at regional or global scales over decadal periods (with the notable exceptions of AMAP, the Northern Contaminants Program in Canada, and the Fennoscandian fish database) in response to the obligations of the Minamata Convention (Evers et al. 2016; Potera 2019; Rosendal et al. 2020). Robust statistical approaches are critical for confidently tracking biotic Hg concentrations in the many different biomes around the world, and controlling for the effects of other factors, such as global climate change, altered foraging habitat, changes in primary productivity, and changing growth rates that can drive shifts in biotic MeHg concentrations that are not due to altered anthropogenic loading of Hg to the ecosystem. One factor in particular, global climate change, will alter future MeHg concentrations in biota in all biomes and ocean basins. Specific effects of global climate change include enhanced air-seawater exchange, melting of polar ice caps and glacier ice sheets, increased thawing of permafrost and changes in estuarine sulfur biogeochemistry – but how these landscape processes relate to changes in biotic Hg exposure is relatively unknown (Wang et al. 2019a).
Iterative efforts to link realistic and applied biomonitoring efforts at local levels with science-policy groups aimed at assisting the Conference of Parties of the Minamata Convention will ultimately help keep pace with the many emerging scientific findings that may fill existing information gaps that are key for local landscape management as well as global policymaking. Ultimately, the careful selection and use of bioindicators that closely match provisions of the Minamata Convention (e.g., linkages to Hg sources, spatial gradients, and temporal trends) can be a cost-effective and time-efficient way to track human and ecological health of anthropogenic loading of Hg into the air and onto the water and landscape at a global level (Evers et al. 2016; Evers and Sunderland 2019). As described, the methods for biomonitoring and the interpretation of the tissues sampled are generally well-established for many target taxa. The extensive knowledge of Hg exposure in a wide range of fish and wildlife that are available in existing monitoring programs and research efforts are described in the peer-reviewed literature, and now in the GBMS database. This therefore provides a platform for informed selection of the appropriate taxa within specific biomes or waterbodies. For example, a synthesis of the compiled global Hg datasets that represent Hg concentrations of biota ingested by people (i.e., freshwater and marine fish, seabirds, and marine mammals) found 45% of the 131 families of representative organisms include individuals that exceed the 0.46 µg/g, ww newly updated benchmark identified by the United States government as food “choices to avoid” (n = 312,335 individuals; Table 8).
Biomonitoring should build from existing programs, which are generally found within developed countries at local, national, and sometimes regional levels. Global pilot projects based on existing networks with local organizations and governmental agencies have been tested for fish (Buck et al. 2019) and humans (Trasande et al. 2016), and regional biomonitoring approaches in temperate and tropical marine ecosystems are described (Evers et al. 2008; Christian et al. 2024). Generating a more coordinated global approach that provides best practice examples, can connect existing biomonitoring programs and identify the ecosystem, taxa, or geographic gaps that are both needed and feasible. Research and monitoring efforts that work in an equitable and ethical partnership with Indigenous Peoples and utilize Indigenous Knowledge have been found to be particularly successful and can also be used as examples (Houde et al. 2022).
In order to better understand and reduce the impact of Hg on people and the environment, additional effort is needed to bridge information and knowledge gaps more effectively. There are many landscape, ecological, and demographic factors that influence MeHg generation and bioavailability – many of which are known and can be used for scaling models. Other factors that affect spatial gradients of biotic MeHg exposure still need further investigations (e.g., ASGM and climate change). Once global needs and interests of the Minamata Convention are determined by the Conference of Parties, we suggest that it is feasible to generate cost-efficient and reliable biomonitoring approaches at geographic scales of interest that can be integrated with existing local and regional Hg biomonitoring networks. Invariably, a commitment to long-term standardized regional biomonitoring approaches is needed - as proven by a 50+ year global tuna Hg assessment that identified multiple limitations generated by high inter-annual variability among species and geographical scale (Médieu et al. 2024).
Lastly, there is an urgency to monitor and assess the influence of MeHg on biota because of the potential adverse impacts to biological diversity (e.g., at ASGM sites: Palacios-Torres et al. 2018; Dossou et al. 2024) during a time when global stressors are causing long-term and significant declines (Leclère et al. 2020; Eddy et al. 2021). Recent evidence demonstrates that the multifaceted effects of anthropogenic chemicals and other pollutants such as Hg in the environment are posing a growing threat to biodiversity (Sigmund et al. 2023) and that there is justification in targeting a wider scope of environmental contaminants within strategies and actions associated with the post-2020 global biodiversity framework of the Convention on Biological Diversity (Sigmund et al. 2022).
Data availability
All data are available from Biodiversity Research Institute (www.briwildlife.org).
References
Abeysinghe KS, Qiu G, Goodale E et al. (2017) Mercury flow through an Asian rice-based food web. Environ Pollut 229:219–228
Abma RA, Paterson G, McLeod A, Haffner GD (2015) Cross-basin comparison of mercury bioaccumulation in Lake Huron lake trout emphasizes ecological characteristics. Environ Toxicol Chem 34:355–359
Ackerman JT, Eagles-Smith CA, Takekawa JY et al. (2007) Mercury concentrations and space use of pre-breeding American avocets and black-necked stilts in San Francisco Bay. Sci Total Environ 384:452–466
Ackerman JT, Eagles-Smith CA, Takekawa JY et al. (2008) Mercury concentrations in blood and feathers of pre-breeding Forster’s terns in relation to space use of San Francisco Bay habitats. Environ Toxicol Chem 27:897–908
Ackerman JT, Eagles-Smith, Herzog MP (2011) Bird mercury concentrations change rapidly as chicks age: toxicological risk is highest at hatching and fledging. Environ Sci Technol 45:5418–5425
Ackerman JT, Overton CT, Casazza ML et al. (2012) Does mercury contamination reduce body condition of endangered California clapper rails? Environ Pollut 162:439–448
Ackerman JT, Herzog MP, Schwarzbach SE (2013) Methylmercury is the predominant form of mercury in bird eggs: a synthesis. Environ Sci Technol 47:2052–2060
Ackerman JT, Hartman CA, Eagles-Smith CA (2015) Estimating mercury exposure of piscivorous birds and sport fish using prey fish monitoring. Environ Sci Technol 49:13596–13604
Ackerman JT, Eagles-Smith CA, Herzog MP (2016) Avian mercury exposure and toxicological risk across western North America: A synthesis. Sci Total Environ 568:749–769
Ackerman JT, Hartman CA, Herzog MP (2019) Mercury contamination in resident and migrant songbirds and potential effects on body condition. Environ Pollut 246:797–810
Ackerman JT, Herzog MP, Evers DC et al. (2020) Synthesis of maternal transfer of mercury in birds: implications for altered toxicity risk. Environ Sci Technol 54:2878–2891
Acosta-Coley I, Cabarcas-Montalvo M, Hernandez-Lambraño RE et al. (2023) Mercury assessment in invasive Lionfish Pterois (Oken, 1817) from marine protected areas in the Colombian Caribbean. Mar Pollut Bull 189:114753
Adams DH, McMichael RH (2007) Mercury in king mackerel, Scomberomorus cavalla, and Spanish mackerel, S. maculatus, from waters of the south-eastern USA: regional and historical trends. Mar Fresh Res 58:187–193
Adams DH (2009) Consistently low mercury concentrations in dolphinfish Coryphaena hippurus an oceanic pelagic predator. Environ Res 109:697–701
Adams E, Guilka JE, Yang Y, Burton MEH, Burns DA, Buxton V, Cleckner L, DeSorbo CR, Driscoll CT, Fisher N, Lane O, Mao H, Riva-Murray K, Millard G, Razavi NR, Richter W, Sauer AK, Schoch N (2023) Distribution and trends of mercury in aquatic and terrestrial biota of New York, USA: a synthesis of 50 years of research and monitoring. Ecotoxicology 32:959–976
Aguirre AA, Lutz PL (2004) Marine Turtles as Sentinels of Ecosystem Health: Is Fibropapillomatosis an Indicator? EcoHealth 1:275–283. 101007/s10393-004-0097-3
Agusa T, Kunito T, Iwata H et al. (2005a) Mercury contamination in human hair and fish from Cambodia: levels specific accumulation and risk assessment. Environ Pollut 134:79–86
Agusa T, Matsumoto T, Ikemoto T et al. (2005b) Body distribution of trace elements in Black-tailed gulls from Rishiri Island, Japan: age-dependent accumulation and transfer to feathers and eggs. Environ Toxicol Chem 24:2107
Ahmed S, Wajid SA, Donohue S et al. (2020) Mercury concentration testing in local fish populations in Anguilla with correlations of potential complications related to long term mercury ingestion in Caribbean populations. J Evolut Med Dent Sci 9:3117–3122
Åkerblom S, Bignert A, Meili M et al. (2014) Half a century of changing mercury levels in Swedish freshwater fish. Ambio 43:91–103
Åkerblom S, de Jong J (2017) Mercury in fur of Daubenton’s bat (Myotis daubentonii) in Southern Sweden and Comparison to Ecotoxicological Thresholds. Bull Environ Contam Toxicol 99:561–566
Albers PH, Koterba MT, Rossmann R et al. (2007) Effects of methylmercury on reproduction in American kestrels. Environ Toxicol Chem Intern J 26:1856–1866
Albert C, Renedo M, Bustamante P, Fort J (2019) Using blood and feathers to investigate large-scale Hg contamination in Arctic seabirds: a review. Environ Res 177:108588
Albert C, Helgason HH, Brault-Favrou M et al. (2021) Seasonal variation of mercury contamination in Arctic seabirds: A pan-arctic assessment. Sci Total Environ 750:142201
Albert C, Strøm H, Helgason HH et al. (2022) Spatial variations in winter Hg contamination affect egg volume in an Arctic seabird the great skua (Stercorarius skua). Environ Pollut 314C:120322
Alonso D, Pineda P, Olivero J et al. (2000) Mercury levels in muscle of two fish species and sediments from the Cartagena Bay and the Ciénaga Grande de Santa Marta, Colombia. Environ Pollut 109:157–163
Alvarez-Berríos NoraL, Mitchell Aide T (2015) Global demand for gold is another threat for tropical forests. Environ Res Lett 10:1
Alves LMF, Lemos MFL, Cabral H, Novais SC (2022) Elasmobranchs as bioindicators of pollution in the marine environment. Mar Pollut Bull 176:113418
Alves LM, Lemos MF, Moutinho AB et al. (2023) Assessment of contaminants in blue sharks from the Northeast Atlantic: profiles accumulation dynamics and risks for human consumers. Environ Pollut 316:120467
AMAP (1998) Arctic Pollution Issues. Arctic Monitoring and Assessment Programme (AMAP). AMAP, Oslo Norway p 859
AMAP (2011) AMAP Assessment 2011: Mercury in the Arctic. Arctic Monitoring and Assessment Programme (AMAP). AMAP, Oslo Norway p 193
AMAP (2015) AMAP Assessment 2015: Human health in the arctic. Arctic Monitoring and Assessment Programme (AMAP). AMAP, Oslo Norway p 65
AMAP (2021) AMAP Assessment 2021: Mercury in the Arctic Arctic. Monitoring and Assessment Programme (AMAP). AMAP, Tromsø Norway p 324
Amorim-Lopes C, Willmer IQ, Araujo NL et al. (2020) Mercury screening in highly consumed sharpnose sharks (Rhizoprionodon lalandii and R. porosus) caught artisanally in southeastern Brazil. Elem Sci Anth 8:022
Amos HM, Jacob DJ, Streets DG, Sunderland EM (2013) Legacy impacts of all-time anthropogenic emissions on the global mercury cycle. Global Biogeochem Cycle 27:410–421
Amos HM, Jacob DJ, Kocman D et al. (2014) Global biogeochemical implications of mercury discharges from rivers and sediment burial. Environ Sci Technol 48:9514–9522
Anan Y, Kunito T, Watanabe I et al. (2001) Trace element accumulation in hawksbill turtles (Eretmochelys imbricata) and green turtles (Chelonia mydas) from Yaeyama Islands, Japan. Environ Toxicol Chem Intern J 20:2802–2814
Anderson DW, Suchanek TH, Eagles-Smith CA, Cahill TM (2008) Mercury residues and productivity in osprey and grebes from a mine-dominated ecosystem. Ecol Appl 18:A227–A238
Anderson ORJ, Phillips RA, Shore RF et al. (2010) Element patterns in albatrosses and petrels: influence of trophic position foraging range and prey type. Environ Pollut 158:98–107
Annibaldi A, Truzzi C, Carnevali O et al. (2019) Determination of Hg in farmed and wild Atlantic bluefin tuna (Thunnus thynnus L) muscle. Molecules 24:1273
Anthony RG, Miles AK, Ricca MA, Estes JA (2007) Environmental contaminants in bald eagle eggs from the Aleutian archipelago. Environ Toxicol Chem 26:1843–1855
Arcagni M, Juncos R, Rizzo A et al. (2018) Species-and habitat-specific bioaccumulation of total mercury and methylmercury in the food web of a deep oligotrophic lake. Sci Total Environ 612:1311–1319
Arrifano GP, Martín-Doimeadios RCR, Jiménez-Moreno M, Ramírez-Mateos V, da Silva NF, Souza-Monteiro JR, Augusto-Oliveira M, Paraense RS, Macchi BM, doNascimento JLM, Crespo-Lopez ME (2018) Large-scale projects in the amazon and human exposure to mercury: The case-study of the Tucuruí Dam. Ecotoxicol Environ Saf 147:299–305
Ashe K (2012) Elevated Mercury Concentrations in Humans of Madre de Dios Peru. PLoS ONE 7(3):e33305. https://doi.org/10.1371/journalpone0033305
Asner GP, Tupayachi R (2017) Accelerated losses of protected forests from gold mining in the Peruvian Amazon. Environmental Res Lett 12:094004
Atobatele OE, Olutona GO (2015) Distribution of three non-essential trace metals (Cadmium Mercury and Lead) in the organs of fish from Aiba Reservoir Iwo Nigeria. Toxicol Rep 2:896–903
Aubail A, Méndez-Fernandez P, Bustamante P et al. (2013) Use of skin and blubber tissues of small cetaceans to assess the trace element content of internal organs. Mar Pollut Bull 76:158–169
Azevedo JS, Sarkis JES, Hortellani MA, Ladle RJ (2012) Are catfish (Ariidae) effective bioindicators for Pb Cd Hg Cu and Zn? Water Air Soil Pollut 223:3911–3922
Azevedo LS, Pestana IA, Rocha ARM et al. (2018) Drought promotes increases in total mercury and methylmercury concentrations in fish from the lower Paraíba do Sul River, southeastern Brazil. Chemosphere 202:483–490
Baek K, Park C, Sakong J (2023) Increase of blood mercury level with shark meat consumption: A repeated-measures study before and after Chuseok, Korean holiday. Chemosphere 344:140317
Balshaw S, Edwards JW, Ross KE, Daughtry BJ (2008) Mercury distribution in the muscular tissue of farmed southern bluefin tuna (Thunnus maccoyii) is inversely related to the lipid content of tissues. Food Chem 111:616–621
Balza U, Brasso R, Lois NA et al. (2021) The highest mercury concentrations ever reported in a South American bird, the Striated Caracara (Phalcoboenus australis). Polar Biol 44:2189–2193
Bank MS, Chesney E, Shine JP et al. (2007) Mercury bioaccumulation and trophic transfer in sympatric snapper species from the Gulf of Mexico. Ecolog Appl 17:2100–2110
Bank MS (2020) The mercury science-policy interface: History evolution and progress of the Minamata Convention. Sci Total Environ 722:137832
Barbieri E (2009) Concentration of heavy metals in tissues of green turtles (Chelonia mydas) sampled in the Cananéia estuary Brazil Brazilian. J Oceanogr 57(3):243–248
Barcia LG, Argiro J, Babcock EA et al. (2020) Mercury and arsenic in processed fins from nine of the most traded shark species in the Hong Kong and China dried seafood markets: The potential health risks of shark fin soup. Mar Pollut Bull 157:111281
Bargagli R, Rota E (2024) Mercury Biogeochemistry and Biomagnification in the Mediterranean Sea: Current Knowledge and Future Prospects in the Context of Climate Change. Coasts 4:89–107
Barletta M, Jaureguizar AJ, Baigun C (2010) Fish and aquatic conservation in South America: a continental overview with emphasis on neotropical systems. J Fish Biol 76:2118–2176
Barnes JG, Gerstenberger L (2015) Using feathers to determine mercury contamination in Peregrine Falcons and their prey. J Raptor Res 49:43–58
Barnes JG, Doney GE, Yates YA (2019) A broadscale assessment of mercury contamination in peregrine falcons across the northern latitudes of North America. J Raptor Res 53:1–13
Barocas A, Claudia V, Alejandro A et al. (2023) Local intensity of artisanal gold mining drives mercury accumulation in neotropical oxbow lake fishes. Sci Total Environ 886:164024
Barraza AD, Komoroske LM, Allen C et al. (2019) Trace metals in green sea turtles (Chelonia mydas) inhabiting two southern California coastal estuaries. Chemosphere 223:342–350. https://doi.org/10.1016/j.chemosphere.2019.01.107
Barrios-Rodríguez CA, de Lacerda LD, Fernandes-Bezerra MA(2023) Pilot Study of Mercury Distribution in the Carapace of Four Species of Sea Turtles from Northeastern Brazil Bull. Environ Contam Toxicol 110:99. https://doi.org/10.1007/s00128-023-03745-1
Bastos WR, Gomes JPO, Oliveira RC et al. (2006) Mercury in the environment and riverside population in the Madeira River basin Amazon Brazil. Sci Total Environ 368:344–351
Bastos WR, Dórea JG, Bernardi JV et al. (2015) Mercury in fish of the Madeira River (temporal and spatial assessment) Brazilian Amazon. Environ Res 140:191–197
Basu N (2015) Applications and implications of neurochemical biomarkers in environmental toxicology. Environ Toxicol Chem 34:22–29
Basu N, Scheuhammer AM, Rouvinen-Watt K et al. (2007) Decreased N-methyl-D-aspartic acid (NMDA) receptor levels are associated with mercury exposure in wild and captive mink. Neurotoxicol 28:587–593
Basu N, Horvat M, Evers DC et al. (2018) A state-of-the-science review of mercury biomarkers in human populations worldwide between 2000 and 2018. Environ Health Perspec 126:106001-14
Basu N, Abass K, Dietz R (2022) The impact of mercury contamination on human health in the Arctic: A state of the science review. Sci Total Environ 831:154793
Basu N, Bastiansz A, Dórea JG et al. (2023) Our evolved understanding of the human health risks of mercury. Ambio 52:877–896
Bauch C, Gatt MC, Verhulst S et al. (2022) Higher mercury contamination is associated with shorter telomeres in a long-lived seabird-A direct effect or a consequence of among-individual variation in phenotypic quality? Sci Total Environ 839:156359
Baumann Z, Mason RP, Conover DO et al. (2017) Mercury bioaccumulation increases with latitude in a coastal marine fish (Atlantic silverside, Menidia menidia). Can J Fish Aquat Sci 74:1009–1015
Bechard MJ, Perkins DN, Kaltenecker GS, Alsup S (2009) Mercury contamination in Idaho bald eagles Haliaeetus leucocephalus. Bull Environ Contam Toxicol 83:698
Bechshoft T, Derocher AE, Richardson E et al. (2016) Hair mercury concentrations in western Hudson Bay polar bear family groups. Environ Sci Technol 50:5313–5319
Benjamin T, Brasso R, Midway S et al. (2018) Using non-destructive techniques to measure mercury (Hg) concentrations in gravid Blanding’s turtles (Emydoidea blandingii) in northeastern Illinois. Bull Environ Contam Toxicol 101:295–299
Bennett RS, French JB, Rossmann R, Haebler R (2009) Dietary toxicity and tissue accumulation of methylmercury in American kestrels. Arch Environ Contam Toxicol 56:149–156
Bentzen R, Castellini JM, Gerlach R et al. (2016) Mercury concentrations in Alaska Pacific halibut muscle relative to stable isotopes of C and N and other biological variables. Mar Pollut Bull 113:110–116
Bezerra MF, Lacerda LD, Costa BGB, Lima EHSM(2012) Mercury in the sea turtle Chelonia mydas (Linnaeus 1958) from Ceará coast NE Brazil Anais Da Academia Brasileira De Ciências 84:123–128. https://doi.org/10.1590/S0001-37652012000100012
Bezerra M, Lacerda L, Rezende C et al. (2015) Food preferences and Hg distribution in Chelonia mydas assessed by stable isotopes. Environ Pollut 206:236–246. https://doi.org/10.1016/j.envpol.2015.07.011
Bianchini K, Tozer DC, Alvo R et al. (2020) Drivers of declines in common loon (Gavia immer) productivity in Ontario Canada. Sci Total Environ 738:139724
Bianchini K, Mallory ML, Braune BM et al. (2022) Why do we monitor? Using seabird eggs to track trends in Arctic environmental contamination. Environ Reviews 30:245–267
Bidone ED, Casilhos ZC, Cid TM et al. (1997) Fish contamination and human exposure to mercury in the Tapajoas River Basin Para State Amazon Brazil: a screening approach. Bull Environ Contam Toxicol 59:194–201
Bighetti GP, Padilha JA, Cunha LST et al. (2021) Bioaccumulation of mercury is equal between sexes but different by age in seabird (Sula leucogaster) population from southeast coast of Brazil. Environ Pollut 285:117222
Bignert A, Riget F, Braune B et al. (2004) Recent temporal trend monitoring of mercury in Arctic biota - how powerful are the existing data sets? J Environ Monit 6:351–355
Bille L, Crovato S, Manfrin A et al. (2020) Mercury contents in commercial Billfish species of the Western Central Atlantic: Assessing the potential risks to human health of Billfish consumption. Food Control 110:107002
Bishop K, Allan C, Bringmark L et al. (2009) The effects of forestry on Hg bioaccumulation in nemoral/boreal waters and recommendations for good silvicultural practice. Ambio A J Human Environ 38:373–380
Bishop K, Shanley JB, Riscassi A et al. (2020) Recent advances in understanding and measurement of mercury in the environment: Terrestrial Hg cycling. Sci Total Environ 721:137647
Bisi TL, Lepoint G, de Freitas Azevedo A et al. (2012) Trophic relationships and mercury biomagnification in Brazilian tropical coastal food webs. Ecol Indicators 18:291–302
Bistoquet K, Marguerite M, Lucas T, et al. (2018) Development of the Fishery Satellite Account in the Seychelles. In: Fourteenth Session of the IOTC Working Party on Data Collection and Statistics (WPDCS), Mahé, Seychelles, p IOTC-2018-WPDCS14-29_Rev2 7p
Biton-Porsmoguer S, Bǎnaru D, Boudouresque CF et al. (2018) Mercury in blue shark (Prionace glauca) and shortfin mako (Isurus oxyrinchus) from north-eastern Atlantic: Implication for fishery management. Mar Pollut Bull 127:131–138
Biton-Porsmoguer S, Bӑnaru D, Harmelin-Vivien M et al. (2022) A study of trophic structure physiological condition and mercury biomagnification in swordfish (Xiphias gladius): evidence of unfavorable conditions for the swordfish population in the Western Mediterranean. Mar Pollut Bull 176:113411
Bjedov D, Mikuska A, Begović L, et al. (2023) Effects of white-tailed eagle (Haliaeetus albicilla) nestling diet on mercury exposure dynamics in Kopački rit Nature Park, Croatia. Environ Pollut 336:122377
Black FJ, Bokhutlo T, Somoxa A et al. (2011) The tropical African mercury anomaly: lower than expected mercury concentrations in fish and human hair. Sci Total Environ 409:1967–1975
Blévin P, Carravieri A, Jaeger A et al. (2013) Wide range of mercury contamination in chicks of Southern Ocean seabirds. Plos One 8(1):e54508
Bloom NS (1992) On the chemical form of mercury in edible fish and marine invertebrate tissue. Can J Fish Aquat Sci 49:1010–1017
Blukacz-Richards EA, Visha A, Graham ML et al. (2017) Mercury levels in herring gulls and fish: 42 years of spatio-temporal trends in the Great Lakes. Chemosphere 172:476–487
Blum JD, Johnson MW (2017) Recent developments in mercury stable isotope analysis. Rev Mineral Geochem 82:733–757
Blum JD, Sherman LS, Johnson MW (2014) Mercury isotopes in earth and environmental sciences. Ann Rev Earth Planet Sci 42:249–269
Bodin N, Lesperance D, Albert R et al. (2017) Trace elements in oceanic pelagic communities in the western Indian Ocean. Chemosphere 174:354–362
Boettiger C, Lang DT, Wainwright PC (2012) rfishbase: exploring, manipulating and visualizing FishBase data from R. J Fish Biol 81(6):2030–2039
Boischio AAP, Henshel D (2000) Fish consumption fish lore and mercury pollution - risk communication for the Madeira River people. Environ Res 84A:108–126
Bond AL, Diamond AW (2009) Mercury concentrations in seabird tissues from Machias Seal Island New Brunswick Canada. Sci Total Environ 407:4340–4347
Bond AL, Hobson KA, Branfireun BA (2015) Rapidly increasing methyl mercury in endangered ivory gull (Pagophila eburnea) feathers over a 130-year record. Proc R Soc London B Biological Sci 282:20150032
Born EW, Kraul I, Kristensen T (1981) Mercury DDT and PCB in the Atlantic walrus (Odobenus rosmarus rosmarus) from the Thule District North Greenland. Arctic 34:255–260
Bosch AC, O’Neill B, Sigge GO et al. (2016a) Mercury accumulation in Yellowfin tuna (Thunnus albacares) with regards to muscle type muscle position and fish size. Food Chem 190:351–356
Bosch AC, O’Neill B, Sigge GO et al. (2016b) Heavy metals in marine fish meat and consumer health. Rev J Sci Food Agric 96:32–48
Bourbour RP, Martinico BL, Ackerman JT et al. (2019) Feather mercury concentrations in North American raptors sampled at migration monitoring stations. Ecotoxicology 28:379–391
Bowerman IVWW, Evans ED, Giesy JP, Postupalsky S (1994) Using feathers to assess risk of mercury and selenium to bald eagle reproduction in the Great Lakes region. Arch Environ Contam Toxicol 27:294–298
Bowerman WW, Roe AS, Gilbertson MJ et al. (2002) Using bald eagles to indicate the health of the Great Lakes’ environment. Lakes Reservoirs Res Manag 7:183–187
Bowman KL, Hammerschmidt CR, Lamborg CH, Swarr G (2014) Mercury in the North Atlantic Ocean: The US GEOTRACES zonal and meridional sections. Deep Sea Res Part II Topical Stud Oceanogr 116:251–261
Bowman KL, Hammerschmidt CR, Lamborg CH et al. (2016) Distribution of mercury species across a zonal section of the eastern tropical South Pacific Ocean (US GEOTRACES GP16). Mar Chem 186:156–166
Braaten HFV, Fjeld E, Rognerud S et al. (2014) Seasonal and year-to-year variation of mercury concentration in perch (Perca fluviatilis) in boreal lakes. Environ Toxicol Chem 33:2661–2670
Braaten HFV, Åkerblom S, de Wit HA, et al. (2017) Spatial patterns and temporal trends of mercury in freshwater fish in Fennoscandia. In NIVA-Report 7179/2017 ICP Waters Report 132/2017, Norwegian Institute for Water Research, Oslo, Norway
Braaten HFV, de Wit HA, Larssen T, Poste AE (2018) Mercury in fish from Norwegian lakes: the complex influence of aqueous organic carbon. Sci Total Environ 627:341–348
Braaten HFV, Åkerblom S, Kahilainen KK et al. (2019) Improved environmental status: 50 years of declining fish mercury levels in boreal and subarctic Fennoscandia. Environ Sci Technol 53:1834–1843
Braaten HFV, Lindholm M, de Wit HA (2020) Five decades of declining methylmercury concentrations in boreal foodwebs suggest pivotal role for sulphate deposition. Sci Total Environ 714:136774
Braaten HFV, Pfaffhuber KA, Routti H, et al. (2022) National mercury assessment - an evaluation of the effectiveness of Norwegian mercury regulations and policies. The Norwegian environment agency. Available at https://www.miljodirektoratet.no/publikasjoner/2022/september/national-mercury-assessment-an-evaluation-of-the-effectiveness-of-norwegian-mercury-regulations-and-policies/
Branco V, Canario J, Vale C et al. (2004) Total and organic mercury concentrations in muscle tissue of the blue shark (Prionace glauca L. 1758) from the Northeast Atlantic. Mar Pollut Bull 49:871–874
Branco V, Vale C, Canário J, dos Santos MN (2007) Mercury and selenium in blue shark (Prionace glauca L 1758) and swordfish (Xiphias gladius L 1758) from two areas of the Atlantic Ocean. Environ Pollut 150:373–380
Branfireun BA, Cosio C, Poulain AJ et al. (2020) Mercury cycling in freshwater systems-An updated conceptual model. Sci Total Environ 745:140906
Braune BM (1987) Comparison of total mercury levels in relation to diet and molt for nine species of marine birds. Arch Environ Contam Toxicol 16:217–224
Braune BM (2007) Temporal trends of organochlorines and mercury in seabird eggs from the Canadian Arctic 1975-2003. Environ Pollut 148:599–613
Braune BM, Mallory ML, Gilchrist HG (2006) Elevated mercury levels in a declining population of ivory gulls in the Canadian Arctic. Mar Pollut Bull 52:978–982
Braune BM, Mallory ML, Butt CM et al. (2010) Persistent halogenated organic contaminants and mercury in northern fulmars (Fulmarus glacialis) from the Canadian Arctic. Environ Pollut 158:3513–3519
Braune BM, Scheuhammer AM, Crump D et al. (2012) Toxicity of methylmercury injected into eggs of thick-billed murres and arctic terns. Ecotoxicology 21:2143–2152
Braune BM, Chételat M, Amyot T et al. (2015) Mercury in the marine environment of the Canadian Arctic: Review of recent findings. Sci Total Environ 509:67–90
Braune BM, Gaston AJ, Mallory ML (2016) Temporal trends of mercury in eggs of five sympatrically breeding seabird species in the Canadian Arctic. Environ Pollut 214:124–131
Breton-Honeyman K, Hammill MO, Furgal CM, Hickie B (2016) Inuit knowledge of beluga whale (Delphinapterus leucas) foraging ecology in Nunavik (Arctic Quebec), Canada. Can J Zool 94:713–726
Branco JM, Hingst-Zaher E, Jordan-Ward R et al. (2022) Interrelationships among feather mercury content, body condition and feather corticosterone in a Neotropical migratory bird, the Purple Martin (Progne subis subis). Environ Pollut 314:20284
Brookens TJ, O’Hara TM, Taylor RJ et al. (2008) Total mercury body burden in Pacific harbor seal Phoca vitulina richardii pups from central California. Mar Pollut Bull 56:27–41
Brown TM, Fisk AT, Wang X et al. (2016) Mercury and cadmium in ringed seals in the Canadian Arctic: Influence of location and diet. Sci Total Environ 545:503–511
Brunborg LA, Graff IE, Frøyland L, Julshamn K (2006) Levels of non-essential elements in muscle from harp seal (Phagophilus groenlandicus) and hooded seal (Cystophora cristata) caught in the Greenland Sea area. The Sci Total Environ 366:784–798
Buck DG, Evers DC, Adams E (2019) A global-scale assessment of fish mercury concentrations and the identification of biological hotspots. Sci Total Environ 687:956–966
Buckman KL, Marvin-DiPasquale M, Taylor VF et al. (2015) Influence of a chlor-alkali superfund site on mercury bioaccumulation in periphyton and low-trophic level fauna. Environ Toxicol Chem 34:1649–1658
Buckman KL, Lane O, Kotnik J et al. (2018) Spatial and taxonomic variation of mercury concentration in low trophic level fauna from the Mediterranean Sea. Ecotoxicology 27:1341–1352
Buckman KL, Seelen EA, Mason RP et al. (2019) Sediment Organic Carbon and Temperature Effects on Methylmercury Concentration: A Mesocosm Experiment. Sci Total Environ. 666:1316–1326
Burger J (1993) Metals in avian feathers: bioindicators of Environ Pollut. Rev Environ Toxicol 5:203–311
Burger J, Gochfeld M (2000) Metal levels in feathers of 12 species of seabirds from Midway Atoll in the northern Pacific Ocean. Sci Total Environ 257:37–52
Burger J, Gochfeld M (2006) Locational differences in heavy metals and metalloids in Pacific blue mussels Mytilus [edulis] trossulus from Adak Island in the Aleutian Chain Alaska. Sci Total Environ 368:937–950
Burger J, Gochfeld M (2009) Comparison of arsenic cadmium chromium lead manganese mercury and selenium in feathers in bald eagle (Haliaeetus leucocephalus) and comparison with common eider (Somateria mollissima) glaucous-winged gull (Larus glaucescens) pigeon guillemot (Cepphus columba) and tufted puffin (Fratercula cirrhata) from the Aleutian Chain of Alaska. Environ Monit Assess 152:357–367
Burger J, Gochfeld M, Jeitner S et al. (2007) Mercury levels and potential risk from subsistence foods from the Aleutians. Sci Total Environ 384:93–105
Burger J, Gochfeld M, Jeitner D et al. (2008) Assessment of metals in down feathers of female common eiders and their eggs from the Aleutians: arsenic cadmium chromium lead manganese mercury and selenium. Environ Monit Assess 143:247–256
Burger J, Gochfeld M, Jeitner S et al. (2009) Mercury and other metals in eggs and feathers of glaucous-winged gulls (Larus glaucescens) in the Aleutians. Environ Monit Assess 152:179–194
Burger J, Mizrahi D, Tsipoura N et al. (2018) Mercury lead cadmium cobalt arsenic and selenium in the blood of semipalmated sandpipers (Calidris pusilla) from Suriname South America: age-related differences in wintering site and comparisons with a stopover site in New Jersey USA. Toxics 6:27
Burgess NM, Hobson KA (2006) Bioaccumulation of mercury in yellow perch (Perca flavescens) and common loons (Gavia immer) in relation to lake chemistry in Atlantic Canada. Hydrobiologia 567:275–282
Burgess NM, Evers DC, Kaplan JD et al. (2005) Mercury and other contaminants in common loons breeding in Atlantic Canada. Ecotoxicology 14:241–252
Burgess NM, Meyer MW (2008) Methylmercury exposure associated with reduced productivity in common loons. Ecotoxicology 17:83–91
Burgess NM, Bond AL, Hebert CE et al. (2013) Mercury trends in herring gull (Larus argentatus) eggs from Atlantic Canada 1972-2008: temporal change or dietary shift? Environ Pollut 172:216–222
Burnham KK, Meyer FK, Burnham JL et al. (2021) Mercury contamination of seabird and sea duck eggs from high Arctic Greenland. Polar Biol 44:1195–1202
Burton MEH, Chen C, Buck D et al. (2024) Monitoring Mercury in Times of Environmental Transformation: A Case Study of Acadia National Park. Ecotoxicology
Bustamante P, Garrigue C, Breau L et al. (2003) Trace elements in two odontocetes species (Kogia breviceps and Globicephala macrorhynchus) stranded in New Caledonia (South Pacific). Environ Pollut 124:263–271
Bustamante P, Carravieri A, Goutte A et al. (2016) High feather mercury concentrations in the wandering albatross are related to sex breeding status and trophic ecology with no demographic consequences. Environ Res 144:1–10
Bustamante P, Le Verge T, Bost CA et al. (2023) Mercury contamination in the tropical seabird community from Clipperton Island, eastern Pacific Ocean. Ecotoxicol 32:1–12
Butler EC, Harries SJ, McAllister KA et al. (2022) Influence of life history variation and habitat on mercury bioaccumulation in a high-order predatory fish in tropical Australia. Environ Res 212:113152
Caballero Espejo J, Messinger M, Román-Dañobeytia F et al. (2018) Deforestation and Forest Degradation Due to Gold Mining in the Peruvian Amazon: A 34-Year Perspective. Remote Sens 10:1903. https://doi.org/10.3390/rs10121903
Cai Y, Rooker JR, Gill GA, Turner JP (2007) Bioaccumulation of mercury in pelagic fishes from the northern Gulf of Mexico. Can J Fish Aquat Sci 64:458–469
Calvert AM, Gutowsky SE, Fifield DA (2024) Inter-colony variation in predation, mercury burden and adult survival in a declining seabird. Sci Total Environ 911:168549
Cammilleri G, Vazzana M, Arizza V et al. (2018) Mercury in fish products: what’s the best for consumers between bluefin tuna and yellowfin tuna? Nat Prod Res 32:457–462
Campbell LM, Hecky RE, Nyaundi J et al. (2003a) Distribution and food-web transfer of mercury in Napoleon and Winam Gulfs, Lake Victoria, East Africa. J Great Lakes Res 29:267–282
Campbell L, Dixon DG, Hecky RE (2003b) A review of mercury in Lake Victoria, East Africa: implications for human and ecosystem health. J Toxicol Environ Health Part B 6:325–356
Campbell LM, Norstrom RJ, Hobson KA et al. (2005) Mercury and other trace elements in a pelagic Arctic marine food web (Northwater Polynya Baffin Bay). Sci Total Environ 351–352:247–263
Campbell L, Verburg P, Dixon DG, Hecky RE (2008) Mercury biomagnification in the food web of Lake Tanganyika (Tanzania, East Africa). Sci Total Environ 402:184–191
Cardona-Marek T, Knott KK, Meyer BE, O’Hara TM (2009) Mercury concentrations in southern Beaufort Sea polar bears: variation based on stable isotopes of carbon and nitrogen. Environ Toxicol Chem Internat J 28:1416–1424
Carravieri A, Cherel Y, Blévin P et al. (2014) Mercury exposure in a large subantarctic avian community. Environ Pollut 190C:51–57
Carravieri A, Cherel Y, Jaeger A et al. (2016) Penguins as bioindicators of mercury contamination in the southern Indian Ocean: geographical and temporal trends. Environ Pollut 213:195–205
Carravieri A, Cherel Y, Brault-Favrou M et al. (2017) From Antarctica to the subtropics: Contrasted geographical concentrations of selenium mercury and persistent organic pollutants in skua chicks (Catharacta spp). Environ Pollut 228:464–473
Carravieri A, Lorioux S, Angelier F et al. (2023) Carryover effects of winter mercury contamination on summer concentrations and reproductive performance in little auks. Environ Pollut 318:120774
Carvan MJ, Kalluvila TA, Klingler RH et al. (2017) Mercury-induced epigenetic transgenerational inheritance of abnormal neurobehavior is correlated with sperm epimutations in zebrafish. PLoS One 12:e0176155
Casazza ML, Ricca MA, Overton CT et al. (2014) Dietary mercury exposure to endangered California clapper rails in San Francisco Bay. Mar Pollut Bull 86:254–260
Castellini JM, Rea LD, Avery JP et al. (2022) Total mercury total selenium and monomethylmercury relationships in multiple age cohorts and tissues of Steller sea lions (Eumetopias jubatus). Environ Toxicol Chem 41:1477–1489
Castilhos ZC, Bidone ED, Lacerda LD (1998) Increase of the background human exposure to mercury through fish consumption due to gold mining at the Tapajos River region Para State Amazon. Bull Environ Contam Toxicol 61:202–209
Castilhos ZC, Rodrigues-Filho S, Rodrigues APC et al. (2006) Mercury contamination in fish from gold mining areas in Indonesia and human health risk assessment. Sci Total Environ 368:320–325
Cerveny D, Roje S, Turek J, Randak T (2016) Fish fin-clips as a non-lethal approach for biomonitoring of mercury contamination in aquatic environments and human health risk assessment. Chemosphere 163:290–295
Chalabis-Mazurek A, Rechulicz J, Pyz-Lukasik R (2021) A food-safety risk assessment of mercury lead and cadmium in fish recreationally caught from three lakes in Poland. Animals 11:3507
Chan HM, Kim C, Khoday K et al. (1995) Assessment of dietary exposure to trace-metals in Baffin Inuit food. Environ Health Perspec 103:740–746
Charapata P, Clark CT, Miller N et al. (2023) Whiskers provide time-series of toxic and essential trace elements, Se: Hg molar ratios, and stable isotope values of an apex Antarctic predator, the leopard seal. Sci Total Environ 854:158651
Chase ME, Jones SH, Hennigar P et al. (2001) Gulfwatch: Monitoring spatial and temporal patterns of trace metal and organic contaminants in the Gulf of Maine (1991-1997) with the blue mussel (Mytilus edulis L). Mar Pollut Bull 42:490–504
Chastel O, Fort J, Ackerman J et al. (2022) Mercury contamination and potential health risks to Arctic seabirds and shorebirds. Sci Total Environ 844C:156944
Chaves-Ulloa R, Taylor BW, Broadley HJ et al. (2016) Dissolved organic carbon modulates mercury concentrations in insect subsidies from streams to terrestrial consumers. Ecol Appl 26:1771–1784
Chen CY, Stemberger RS, Kamman NC et al. (2005) Patterns of Hg bioaccumulation and transfer in aquatic food webs across multi-lake studies in the Northeast US. Ecotoxicol 14:135–147
Chen CY, Evers DC (2023) Global mercury impact synthesis: Processes in the Southern Hemisphere. Ambio 52:827–832
Chen CY, Pickhardt PC, Xu MQ, Folt CL (2008) Mercury and arsenic bioaccumulation and eutrophication in Baiyangdian Lake China. Water Air Soil Pollut 190:115–127
Chen C, Kamman N, Williams J et al. (2012) Spatial and temporal variation in mercury bioaccumulation by zooplankton in Lake Champlain. Environ Pollut 161:343–349
Chen CY, Borsuk ME, Bugge DM et al. (2014) Benthic and pelagic pathways of methylmercury bioaccumulation in estuarine food webs of the northeast United States. PloS One 9:89305
Chen L, Zhang X, Cao M et al. (2021) Release of legacy mercury and effect of aquaculture on mercury biogeochemical cycling in highly polluted Ya-Er Lake, China. Chemo 275:130011
Chen MM, Lopez L, Bhavsar SP, Sharma S (2018) What’s hot about mercury? Examining the influence of climate on mercury levels in Ontario top predator fishes. Environ Res 162:63–73
Cheng H, Hu Y (2012) Understanding the Paradox of Mercury Pollution in China: High Concentrations in Environmental Matrix yet Low Levels in Fish on the Market. Environ Sci Technol 46:4695–4696
Cherel Y, Barbraud C, Lahournat M et al. (2018) Accumulate or eliminate? Seasonal mercury dynamics in albatrosses the most contaminated family of birds. Environ Pollut 241:124–135
Chételat J, Braune B, Stow J, Tomlinson S (2015) Special issue on mercury in Canada’s North: Summary and recommendations for future research. Sci Total Environ 509:260–262
Chételat J, Richardson MC, MacMillan GA et al. (2018) Ratio of methylmercury to dissolved organic carbon in water explains methylmercury bioaccumulation across a latitudinal gradient from North-Temperate to Arctic Lakes. Environ Sci Technol 52:79–88
Chételat J, Ackerman KT, Eagles-Smith CA, Hebert CE (2020) Methylmercury exposure in wildlife: a review of the ecological and physiological processes affecting contaminant concentrations and their interpretation. Sci Total Environ 711:135117
Chételat J, McKinney MA, Amyot M et al. (2022) Climate change and mercury in the Arctic: Abiotic interactions. Sci Total Environ 824:153715
Chinain M, Gatti CMI, Darius HT et al. (2021) Ciguatera poisonings: A global review of occurrences and trends. Harmful Algae 102:101873
Chouvelon T, Brach-Papa C, Auger D et al. (2017) Chemical contaminants (trace metals persistent organic pollutants) in albacore tuna from western Indian and south-eastern Atlantic Oceans: Trophic influence and potential as tracers of populations. Sci Total Environ 596:481–495
Chouvelon T, Cresson P, Bouchoucha M et al. (2018) Oligotrophy as a major driver of mercury bioaccumulation in medium-to high-trophic level consumers: A marine ecosystem-comparative study. Environ Pollut 233C:844–854
Choy CA, Popp BN, Kaneko JJ, Drazen JC (2009) The influence of depth on mercury levels in pelagic fishes and their prey. Proc Nat Acad Sci 106:13865–13869
Christian L, Burton MEH, Mohammed A et al. (2024) An evaluation of fish and invertebrate mercury concentrations in the Caribbean Region. Ecotoxicology https://doi.org/10.1007/s10646-024-02754-y
Cinnirella S, Bruno DE, Pirrone N et al. (2019) Mercury concentrations in biota in the Mediterranean Sea a compilation of 40 years of surveys. Sci Data 6:205
Cizdziel JV, Hinners TA, Heithmar EM (2002) Determination of total mercury in fish tissues using combustion atomic absorption spectrometry with gold amalgamation. Water Air Soil Pollut 135:355–370
Cladis DP, Kleiner AC, Santerre CR (2014) Mercury content in commercially available finfish in the United States. J Food Protection 77:1361–1366
Condini MV, Malinowski CR, Hoeinghaus DJ et al. (2023) Spatial analysis of mercury and stable isotopes in the vulnerable Dusky Grouper Epinephelus marginatus along the Brazilian coast. Mar Pollut Bull 187:114526
Condon AM, Cristol DA (2009) Feather growth influences blood mercury level of young songbirds. Environ Toxicol Chem 28:395–401
Correa L, Castellini JM, Wells RS, O’Hara T (2013) Distribution of mercury and selenium in blood compartments of bottlenose dolphins (Tursiops truncatus) from Sarasota Bay Florida. Environ Toxicol Chem 32:2441–2448
Correia E, Granadeiro JP, Vale C, Catry T (2023) Trace elements in relation to trophic ecology of long-distance migratory shorebirds and seabirds in West Africa. Environ Pollut 316:120674
Cossa D, Coquery M (2005) The Mediterranean mercury anomaly a geochemical or a biological issue in the Mediterranean Sea. Handbook of Environmental Chemistry, vol 5 Part K 177–208. Springer-Verlag, Berlin Heidelberg, p 414
Cossa D, Averty B, Pirrone N (2009) The origin of methylmercury in open Mediterranean waters. Limnol Oceanogr 54:837–844
Cossa D, Heimbürger LE, Lannuzel D et al. (2011) Mercury in the Southern Ocean. Geochim Cosmochim Acta 75:4037–4052
Cossa D, Harmelin-Vivien M, Mellon-Duval M et al. (2012) Influences of Bioavailability Trophic Position and Growth on Methylmercury in Hakes (Merluccius merluccius) from Northwestern Mediterranean and Northeastern Atlantic. Environ Sci Technol 46:4885–4893
Cossa D, Knoery J, Banaru D et al. (2022) Mediterranean mercury assessment 2022: an updated budget health consequences and research perspectives. Environ Sci Technol 56:3840–3862
Costa F, Coelho JP, Baptista J et al. (2021) Lifelong mercury bioaccumulation in Atlantic horse mackerel (Trachurus trachurus) and the potential risks to human consumption. Mar Pollut Bull 173:113015
Cransveld A, Amouroux D, Tessier E et al. (2017) Mercury stable isotopes discriminate different populations of European seabass and trace potential Hg sources around Europe. Environ Sci Technol 51:12219–12228
Cresson P, Fabri M-C, Bouchoucha M et al. (2014) Mercury in organisms from the Northwestern Mediterranean slope: importance of food sources. Sci Total Environ 497-498:229–238
Cresson P, Bouchoucha M, Morat F et al. (2015) A multitracer approach to assess the spatial contamination pattern of hake (Merluccius merluccius) in the French Mediterranean. Sci Total Environ 532:184–194
Cristol DA, Brasso RL, Condon AM et al. (2008) The movement of aquatic mercury through terrestrial food webs. Science 320:335–335
Cristol DA, Evers DC (2020) The impact of mercury on North American songbirds: effects trends and predictive factors. Ecotoxicology 29:1107–1116
Crump KL, Trudeau VL (2009) Mercury‐induced reproductive impairment in fish. Environ Toxicol Chem An InternatJ 28:895–907
Cruz-Flores M, Lemaire J, Brault-Favrou M et al. (2024) Spatial distribution of selenium-mercury in Arctic seabirds. Environ Pollut 343:123110
Cumbee JC, Gaines KF, Mills GL et al. (2008) Clapper rails as indicators of mercury and PCB bioavailability in a Georgia saltmarsh system. Ecotoxicol 17:485–494
Cusset F, Reynolds SJ, Carravieri A et al. (2023) A century of mercury: Ecosystem-wide changes drive increasing contamination of a tropical seabird species in the South Atlantic Ocean. Environ Pollut 323:121187
Cuvin-Aralar MLA (1990) Mercury levels in the sediment water and selected finfishes of Laguna Lakes The Philippines. Aquaculture 84:277–288
da Silva SF, Oliveira DC, Pereira JPG et al. (2019) Seasonal variation of mercury in commercial fishes of the Amazon Triple Frontier, Western Amazon Basin. Ecological Indicators 106:105549
da Silva Montes C, Ferreira MAP, Giarrizzo T et al. (2022) The legacy of artisanal gold mining and its impact on fish health from Tapajós Amazonian region: A multi-biomarker approach. Chemosphere 287:132263
Dam M, Bloch D (2000) Screening of mercury and persistent organochlorine pollutants in long-finned pilot whale (Globicephala melas) in the Faroe Islands. Mar Pollut Bull 40:1090–1099
Damseaux F, Siebert U, Pomeroy P et al. (2021) Habitat and resource segregation of two sympatric seals in the North Sea. Sci Total Environ 764:142842
Daso AP, Okonkwo JO, Jansen R et al. (2015) Mercury concentrations in eggshells of the southern ground-hornbill (Bucorvus leadbeateri) and wattled crane (Bugeranus carunculatus) in South Africa. Ecotoxicol Environ Saf 114:61–66
Dastoor AP, Larocque Y (2004) Global circulation of atmospheric mercury: A modelling study. Atmospheric Environ 38:147–161
Dastoor A, Ryzhkov A, Durnford D et al. (2015) Atmospheric mercury in the Canadian Arctic Part II: Insight from modeling. Sci Total Environ 509:16–27
Day RD, Christopher SJ, Becker PR, Whitaker DW (2005) Monitoring mercury in the loggerhead sea turtle Caretta caretta. Environ Sci Technol 39:437–446
Day RD, Becker PR, Donard OF et al. (2014) Environmental specimen banks as a resource for mercury and mercury isotope research in marine ecosystems. Environ Sci Processes Impacts 16:10–27
de Carvalho GGA, Degaspari IAM, Branco V et al. (2014) Assessment of total and organic mercury levels in blue sharks (Prionace glauca) from the south and southeastern Brazilian coast. Biol Trace Element Res 159:128–134
de Matos LS, Correa ASAS, da Silva SAA et al. (2021) Mercury concentrations in fish and human health assessment in preflood phase of a hydro dam in Teles Pires River Southern Brazilian Amazon. Elem Sci Anth 9:020
de Medeiros Costa G, Lázaro WL et al. (2021) Rhamphotheca as a useful indicator of mercury in seabirds. Sci Total Environ 754:141730
de Pinho AP, Guimarães JRD, Martins AS et al. (2002) Total mercury in muscle tissue of five shark species from Brazilian offshore waters: effects of feeding habit sex and length. Environ Res 89:250–258
de Wit HA, Granhus A, Lindholm M et al. (2014) Forest harvest effects on mercury in streams and biota in Norwegian boreal catchments. Forest Ecol Manag 324:52–63
Debrah CA, Ribeiro PF, Baah D (2020) Total levels of mercury concentrations in marine fish-Kumasi Ghana European. J Nutr Food Saf 12:81–86
Depew DC, Basu N, Burgess NM et al. (2012a) Toxicity of dietary methylmercury to fish: derivation of ecologically meaningful threshold concentrations. Environ Toxicol Chem 31:1536–1547
Depew DC, Basu N, Burgess NM et al. (2012b) Derivation of screening benchmarks for dietary methylmercury exposure for the common loon (Gavia immer): rationale for use in ecological risk assessment. Environ Toxicol Chem 31:2399–2407
Depew DC, Burgess NM, Anderson MR et al. (2013) An overview of mercury concentrations in freshwater fish species: a national fish mercury dataset for Canada Canadian. J Fisheries Aquatic Sci 70:436–451
Desforges JPW, Sonne C, Levin M et al. (2016) Immunotoxic effects of environmental pollutants in marine mammals. Environ Int 86:126–139
DesGranges JL, Rodrigue J, Tardif B, Laperle M (1998) Mercury accumulation and biomagnification in ospreys (Pandion haliaetus) in the James Bay and Hudson Bay regions of Quebec. Arch Environ Contam Toxicol 35:330–341
DeSorbo CR, Burgess NM, Todd CS et al. (2018) Mercury concentrations in bald eagles across an impacted watershed in Maine USA. Sci Total Environ 627:1515–1527
DeSorbo CR, Burgess NM, Nye PE et al. (2020) Bald eagle mercury exposure varies with region and site elevation in New York USA. Ecotoxicology 29:1862–1876
Di Bella G, Tardugno R, Cicero N (2018) Investigation of Hg content by a rapid analytical technique in Mediterranean pelagic fishes. Separations 5:51
Dibbern M, Elmeros M, Dietz R et al. (2021) Mercury exposure and risk assessment for Eurasian otters (Lutra lutra) in Denmark. Chemosphere 272:129608
Dietz RC, Pacyna J, Thomas DJ, et al. (1998) Chapter 7: Heavy metals AMAP Assessment Report: Arctic Pollution Issues Arctic Monitoring and Assessment Programme. AMAP, Oslo Norway, p 373–524
Dietz RC, Riget F, Born EW (2000) Geographical differences of zinc cadmium mercury and selenium in polar bears (Ursus maritimus) from Greenland. Sci Total Environ 245:25–48
Dietz R, Outridge PM, Hobson KA (2009) Anthropogenic contributions to mercury levels in present-day Arctic animals—a review. Sci Total Environ 407:6120–6131
Dietz R, Born EW, Riget F et al. (2011) Temporal trends and future predictions of mercury concentrations in Northwest Greenland polar bear (Ursus maritimus) hair. Environ Sci Technol 45:1458–1465
Dietz RC, Basu N, Braune B et al. (2013) What are the toxicological effects of mercury in Arctic biota? Sci Total Environ 443:775–790
Dietz RC, Letcher RJ, Desforges JP, et al. (2019) Current state of knowledge on biological effects from contaminants on arctic wildlife and fish. Sci Total Environ 696:133792
Dietz RC, Sonne C, Albert JO et al. (2021) A risk assessment of the effects of mercury on Baltic Sea Greater North Sea and North Atlantic wildlife fish and bivalves. Environ Int 146:106178
Dietz RC, Letcher RJ, Aars J et al. (2022) A risk assessment review of mercury exposure in Arctic marine and terrestrial mammals. Sci Total Environ 845:154445
Diop M, Amara R (2016) Mercury concentrations in the coastal marine food web along the Senegalese coast. Environ Sci Pollut Res 23:11975–11984
Diringer SE, Feingold BJ, Ortiz EJ et al. (2015) River transport of mercury from artisanal and small-scale gold mining and risks for dietary mercu ry exposure in Madre de Dios Peru. Environ Sci 17:478–487. https://doi.org/10.1039/C4EM00567H
Dodino S, Riccialdelli L, Polito MJ et al. (2022) Mercury exposure driven by geographic and trophic factors in Magellanic penguins from Tierra del Fuego. Mar Pollut Bull 174:113184
Dolgova S, Crump D, Porter E et al. (2018) Stage of development affects dry weight mercury concentrations in bird eggs: Laboratory evidence and adjustment method. Environ Toxicol Chem 37:1168–1174
Dominguez L, Montevecchi W, Burgess NM et al. (2003) Reproductive success, environmental contaminants, and trophic status of nesting Bald Eagles in eastern Newfoundland, Canada. J Raptor Res 37:209–218
Donovan PM, Blum JD, Demers JD (2014) Identification of multiple mercury sources to stream sediments near Oak Ridge TN USA. Environ Sci Technol 48:3666–3674
Donovan PM, Blum JD, Singer MB, Marvin-DiPasquale M, Tsui MT (2016) Isotopic composition of inorganic mercury and methylmercury downstream of a historical gold mining region. Environ Sci Technol 50(4):1691–1702
dos Santos LDS, Müller RC, de S Sarkis JE et al. (2000) Evaluation of total mercury concentrations in fish consumed in the municipality of Itaituba Tapajos River Basin Para Brazil. Sci Total Environ 261:1–8
Dossou EI, Stylo M, Davis K, Evers DC, Slaveykova V (2024) Artisanal and Small-scale Gold Mining and Biodiversity: A Global Literature Review. In Assessing Environmental Mercury Loads in Biota and Impacts on Biodiversity to Meet the Needs of the Minamata Convention. Ecotoxicology https://doi.org/10.1007/s10646-024-02748-w
Drevnick PE, Lamborg CH, Horgan MJ (2015) Increase in mercury in Pacific yellowfin tuna. Environ Toxicol Chem 34:931–934
Drevnick PE, Brooks BA (2017) Mercury in tunas and blue marlin in the North Pacific Ocean. Environ Toxicol Chem 36:1365–1374
Diringer SE, Axel J, Berky Marco Marani Ernesto J et al. (2020) Deforestation Due to Artisanal and Small-Scale Gold Mining Exacerbates Soil and Mercury Mobilization in Madre de Dios Peru. Environ Sci Technol 54:286–296
Driscoll CT, Han YJ, Chen CY et al. (2007) Mercury contamination in remote forest and aquatic ecosystems in the northeastern US: Sources transformations and management options. Bioscience 57:17–28
Driscoll CT, Mason RP, Chan HM et al. (2013) Mercury as a global pollutant: sources pathways and effects. Environ Sci Technol 47:4967–4983
Drouillard KG, Campbell L, Otieno D et al. (2024) Increasing mercury bioaccumulation and biomagnification rates of Nile perch (Lates niloticus L.) in Winam Gulf, Lake Victoria, Kenya. Sci Total Environ 916:170059
Dudarev AA, Chupakhin VS, Vlasov SV, Yamin-Pasternak S (2019) Traditional diet and environmental contaminants in coastal Chukotka III: metals. Int J Environ Res Public Health 16:699
Durante CA, Reis BMM, Azevedo A et al. (2020) Trace elements in trophic webs from South Atlantic: The use of cetaceans as sentinels. Mar Pollut Bull 150:110674
Dutton J, Venuti VM (2019) Comparison of maternal and embryonic trace element concentrations in common thresher shark (Alopias vulpinus) muscle tissue. Bull Environ Contam Toxicol 103:380–384
Eagles-Smith CA, Ackerman JT (2009) Rapid changes in small fish mercury concentrations in estuarine wetlands: Implications for wildlife risk and monitoring programs. Environ Sci Technol 43:8658–8664
Eagles-Smith CA, Ackerman JT (2014) Mercury bioaccumulation in estuarine wetland fishes: evaluating habitats and risk to coastal wildlife. Environ Pollut 193:147–155
Eagles-Smith CA, Ackerman JT, De La Cruz SEW, Takekawa JY (2009a) Mercury bioaccumulation and risk to three waterbird foraging guilds is influenced by foraging ecology and breeding stage. Environ Pollut 157:1993–2002
Eagles-Smith CA, Ackerman JT, Willacker JJ et al. (2016a) Spatial and temporal patterns of mercury concentrations in freshwater fish across the Western United States and Canada. Sci Total Environ 568:1171–1184
Eagles-Smith CA, Wiener JG, Eckley CS et al. (2016b) Mercury in western North America: A synthesis of environmental contamination fluxes bioaccumulation and risk to fish and wildlife. Sci Total Environ 568:1213–1226
Eagles-Smith CA, Silbergeld EK, Basu N et al. (2018) Modulators of mercury risk to wildlife and humans in the context of rapid global change. Ambio 47:170–197
Eagles-Smith CA, Willacker JJ, Nelson SJ et al. (2020) A national-scale assessment of mercury bioaccumulation in United States national parks using dragonfly larvae as biosentinels through a citizen-science framework. Environ Sci Technol 54(14):8779–8790
Eddy TD, Lam VW, Reygondeau G et al. (2021) Global decline in capacity of coral reefs to provide ecosystem services. One Earth 4:1278–1285
Edmonds ST, Evers DC, O’Driscoll NJ et al. (2010) Geographic and seasonal variation in mercury exposure of the declining Rusty Blackbird. Condor 112:789–799
Ehnert-Russo SL, Gelsleichter J (2020) Mercury accumulation and effects in the brain of the Atlantic sharpnose shark (Rhizoprionodon terraenovae). Arch Environ Contam Toxicol 78:267–283
Eikenberry BCS, Riva-Murray K, Knightes CD (2015) Optimizing fish sampling for fish–mercury bioaccumulation factors. Chemosphere 135:467–473
Elawady E, El Bayomi R, Darwish W, El-Atabany A (2019) Risk assessment of some heavy metals from Claris gariepinus (African catfish) consumed in Sharkia Governorate Egypt Zagazig. Veterinary Journal 47:193–202
Erasmus JH, Smit NJ, Gerber R et al. (2022a) Total mercury concentrations in sharks skates and rays along the South African coast. Mar Pollut Bull 184:114142
European Environmental Bureau (EEB) (2021) Tackling Mercury Pollution of EU Waters. European Environmental Bureau, Brussels, Belgium
Eklöf K, Bishop K, Bertilsson S et al. (2018) Formation of mercury methylation hotspots as a consequence of forestry operations. Sci Total Environ 613:1069–1078
Elliott JE, Elliott KH (2013) Tracking marine pollution. Science 340:556–558
Erasmus JH, Smit NJ, Gerber R et al. (2022b) Total mercury concentrations in sharks, skates and rays along the South African coast. Mar Pollut Bull 184:114142
Eriksson MO, Johansson I, Ahlgren CG (1992) Levels of mercury in eggs of Red-throated Diver Gavia stellata and Black-throated Diver G arctica in Southwest Sweden. Ornis Svecica 2:29–36
Eriksson MO (2015) Reduced survival of Black-throated Diver Gavia arctica chicks-an effect of changes in the abundance of fish light conditions or exposure to mercury in the breeding lakes. Ornis Svecica 25:131–152
Escobar-Sánchez O, Galván-Magaña F, Rosíles-Martínez R (2011) Biomagnification of mercury and selenium in blue shark Prionace glauca from the Pacific Ocean off Mexico. Biologic Trace Element Res 144:550–559
Esposito M, De Roma A, La Nucara R et al. (2018) Total mercury content in commercial swordfish (Xiphias gladius) from different FAO fishing areas. Chemosphere 197:14–19
European Union Commission (EU Commission) (2006) No 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs L 364/5. European Union Commission, Brussels, Belgium
European Union (EU) (2023) The EU Fish Market - 2023 Edition. Publications office of the European Union. Luxembourg
Evans MS, Muir DC, Keating J, Wang X (2015) Anadromous char as an alternate food choice to marine animals: A synthesis of Hg concentrations population features and other influencing factors. Sci Total Environ 509:175–194
Evans RD, Addison EM, Villeneuve JY et al. (2000) Distribution of inorganic and methylmercury among tissues in mink (Mustela vison) and otter (Lutra canadensis). Environ Res 84:133–139
Evers DC (2018) The Effects of Methylmercury on Wildlife: A Comprehensive Review and Approach for Interpretation. Encyclopedia Anthropocene 5:181–194. 10.1016
Evers DC, Clair TA (2005) Mercury in northeastern North America: A synthesis of existing databases. Ecotoxicology 14:7–14
Evers DC, Sunderland E (2019) Technical information report on mercury monitoring in biota. United Nations Environment Programme, Geneva, Switzerland, p 44
Evers DC, Kaplan JD, Meyer MW et al. (1998) Bioavailability of environmental mercury measured in Common Loon feathers and blood across North American. Environ Toxicol Chem 17:173–183
Evers DC, Taylor KM, Major A et al. (2003) Common loon eggs as indicators of methylmercury availability in North America. Ecotoxicology 12:69–81
Evers DC, Tear T, Burton M (2023) Mercury in the global environment: Impacts to biodiversity? BRI Science Communications Series 2023-12. Biodiversity Research Institute, Portland, Maine, USA, p 21
Evers DC, Burgess N, Champoux L et al. (2005) Patterns and interpretation of mercury exposure in freshwater avian communities in northeastern North America. Ecotoxicology 14:193–222
Evers DC, Han YJ, Driscoll CT et al. (2007) Identification and Evaluation of Biological Hotspots of Mercury in the Northeastern US and Eastern Canada. Bioscience 57:29–43
Evers DC, Mason RP, Kamman NC et al. (2008) An integrated mercury monitoring program for temperate estuarine and marine ecosystems on the North American Atlantic Coast. EcoHealth 5:426–441
Evers DC, Graham RT, Perkins P et al. (2009) Mercury concentrations in the goliath grouper of Belize: an anthropogenic stressor of concern. Endangered Species Res 7:249–256
Evers DC, Wiener JG, Basu N et al. (2011a) Mercury in the Great Lakes region - Bioaccumulation spatial and temporal patterns ecological risks and policy. Ecotoxicology 20:1487–1499
Evers DC, Williams KA, Meyer MW et al. (2011b) Spatial gradients of methylmercury for breeding common loons in the Laurentian Great Lakes region. Ecotoxicology 20:1609–1625
Evers DC, Schmutz JA, Basu N et al. (2014) Mercury exposure and risk in Yellow-billed Loons breeding in Alaska and Canada. Waterbirds 37:147–159
Evers DC, Keane SE, Basu N, Buck D (2016) Evaluating the effectiveness of the Minamata Convention on Mercury: Principles and recommendations for next steps. Sci Total Environ 569:888–903
Evers DC, Sauer AK, Burns DA et al. (2020) A synthesis of patterns of environmental mercury inputs exposure and effects in New York State. Ecotoxicology 29:1565–1589
Ewald JD, Kirk JL, Li M, Sunderland EM (2019) Organ-specific differences in mercury speciation and accumulation across ringed seal (Phoca hispida) life stages. Sci Total Environ 650:2013–2020
Faial KR, Deus S, Deus R et al. (2015) Mercury levels assessment in hair of riverside inhabitants of the Tapajos River Para State Amazon Brazil: fish consumption as a possible route of exposure. J Trace Elements Med Biol 30:66–76
Feingold BA, Berky H, Hsu-Kim E et al. (2020) Population-based dietary exposure to mercury through fish consumption in the Southern Peruvian Amazon. Environ Res 183:108720. https://doi.org/10.1016/j.envres.2019.108720
Feng X, Li P, Fu X et al. (2022) Mercury pollution in China: implications on the implementation of the Minamata Convention. Environ Sci 24:634–648
Ferreira da SS, Diomar C, Oliveira J et al. (2019) Seasonal variation of mercury in commercial fishes of the Amazon Triple Frontier Western Amazon Basin. Ecol Indicators 106:105549. https://doi.org/10.1016/j.ecolind.2019.105549
Ferriss BE, Essington TE (2011) Regional patterns in mercury and selenium concentrations of yellowfin tuna (Thunnus albacares) and bigeye tuna (Thunnus obesus) in the Pacific Ocean Canadian. J Fisheries Aquatic Sci 68:2046–2056
Fielding R, Evans DW (2014) Mercury in Caribbean dolphins (Stenella longirostris and Stenella frontalis) caught for human consumption off St Vincent West Indies. Mar Pollut Bull 89(1-2):30–34
Finkelstein M, Keitt BS, Croll DA (2006) Albatross species demonstrate regional differences in North Pacific marine contamination. Ecol Appl 16:678–686
Fisher JA, Jacob DJ, Soerensen AL (2012) Riverine source of Arctic Ocean mercury inferred from atmospheric observations. Natural Geosci 5:499–504
Fleishman AB, Orben RA, Kokubun N et al. (2019) Wintering in the western subarctic Pacific increases mercury contamination of red-legged kittiwakes. Environ Sci Technol 53:13398–13407
Fleming EJ, Mack EE, Green PG, Nelson DC (2006) Mercury methylation from unexpected sources: molybdate-inhibited freshwater sediments and an iron-reducing bacterium. Appl Environ Microbiol 72:457–464
Fonseca FRD, Malm O, Waldemarin HF (2005) Mercury levels in tissues of Giant otters (Pteronura brasiliensis) from the Rio Negro Pantanal Brazil. Environ Res 98:368–371
Food and Agriculture Organization (FAO) (2012) The State of World Fisheries and Aquaculture 2012. FAO, Rome Italy
Food and Agriculture Organization (FAO) (2018) The state of world fisheries and aquaculture 2018. Meeting the sustainable development goal. FAO fisheries and Aquaculture Department, Rome Italy
Food and Agriculture Organization (FAO) (2020) The state of Mediterranean and Black Sea fisheries 2020. FAO fisheries and Aquaculture Department, Rome Italy
Food and Agriculture Organization (FAO) (2022) The State of Mediterranean and Black Sea Fisheries 2022. General Fisheries Commission for the Mediterranean. FOA, Rome Italy, p 188
Fort J, Helgason H, Amelineau F, et al. (2017) ARCTOX: a pan-Arctic sampling network to track mercury contamination across Arctic marine food webs. In: 13th International Conference on Mercury as a Global Pollutant. ICMGP, Providence, Rhode Island, USA
Fournier F, Karasov WH, Kenow KP et al. (2002a) The oral bioavailability and toxicokinetics of methylmercury in common loon (Gavia immer) chicks. Comparative Biochem Physiol Part A 133:703–714
Fournier F, Karasov WH, Meyer MW, Kenow KP (2002b) Daily energy expenditures of free-ranging Common Loon (Gavia immer) chicks. Auk 119:1121–1126
Frederick PC, Spalding MG, Sepälveda MS et al. (1999) Exposure of great egret (Ardea albus) nestlings to mercury through diet in the Everglades ecosystem. Environ Toxicol Chem 18:1940–1947
Frederick PC, Spalding MG, Dusek R (2002) Wading birds as bioindicators of mercury contamination in Florida USA: Annual and geographic variation. Environ Toxicol Chem 21:163–167
Frederick PC, Hylton B, Heath JA, Spalding MG (2004) A historical record of mercury contamination in southern florida (USA) as inferred from avian feather tissue: Contribution R‐09888 of the Journal Series, Florida Agricultural Experiment Station. Environ Toxicol Chem 23:1474–1478
Frederick P, Jayasena N (2011) Altered pairing behaviour and reproductive success in white ibises exposed to environmentally relevant concentrations of methylmercury. Proc Royal Soc B 278:1851–1857
Furtado R, Pereira ME, Granadeiro JP, Catry P (2019) Body feather mercury and arsenic concentrations in five species of seabirds from the Falkland Islands. Mar Pollut Bull 149:110574
Furtado R, Granadeiro JP, Gatt MC et al. (2021) Monitoring of mercury in the mesopelagic domain of the Pacific and Atlantic oceans using body feathers of Bulwer’s petrel as a bioindicator. Sci Total Environ 775:145796
Gabriel MC, Howard N, Osborne TZ (2014) Fish mercury and surface water sulfate relationships in the Everglades Protection Area. Environ Manag 53:583–593
Gagnon MM, Baker JK, Long et al. (2016) Contaminant (PAHs, OCs, PCBs and trace metals) concentrations are declining in axial tissue of sand flathead (Platycephalus bassensis) collected from an urbanised catchment (Port Phillip Bay, Australia). Marine Pollut Bull 109:661–666
Gajdosechova Z, Brownlow A, Cottin NT et al. (2016) Possible link between Hg and Cd accumulation in the brain of long-finned pilot whales (Globicephala melas). Sci Total Environ 545:407–413
Gallagher AJ, Kyne PM, Hammerschlag N (2012) Ecological risk assessment and its application to elasmobranch conservation and management. J Fish Biol 85:1727–1748
Gallego Ríos, Ramírez SE, López CM et al. (2018) Evaluation of mercury, lead, arsenic, and cadmium in some species of fish in the Atrato River Delta, Gulf of Urabá, Colombian Caribbean. Water Air Soil Pollut 229:1–13
Gammons CH, Slotton DG, Gerbrandt B et al. (2006) Mercury concentrations of fish, river water, and sediment in the Río Ramis-Lake Titicaca watershed, Peru. Sci Total Environ 368:637–648
Gandhi N, Tang RW, Bhavsar SP, Arhonditsis GB (2014) Fish mercury levels appear to be increasing lately: a report from 40 years of monitoring in the province of Ontario Canada. Environ Sci Technol 48:5404–5414
Gandhi N, Bhavsar SP, Tang RW, Arhonditsis GB (2015) Projecting Fish Mercury Levels in the Province of Ontario Canada and the Implications for Fish and Human Health. Environ Sci Technol 49:14494–14502
Gandhi N, Bhavsar SP, Gewurtz SB et al. (2016) Is it appropriate to composite fish samples for mercury trend monitoring and consumption advisories? Environ Int 88:80–85
Gantner NM, Power D, Iqaluk M et al. (2010) Mercury concentrations in landlocked Arctic char (Salvelinus alpinus) from the Canadian Arctic Part I: insights from trophic relationships in 18 lakes. Environ Toxicol Chem 29:621–632
García MÁ, Núñez R, Alonso J, Melgar MJ (2016) Total mercury in fresh and processed tuna marketed in Galicia (NW Spain) in relation to dietary exposure. Environ Sci Pollut Res 23:24960–24969
García-Seoane R, Varela Z, Carballeira A et al. (2017) Temporal trends in mercury concentrations in raptor flight feathers stored in an environmental specimen bank in Galicia (NW Spain) between 2000 and 2013. Ecotoxicology 26:196–201
Garrigue C, Oremus M, Dodémont R et al. (2016) A mass stranding of seven Longman’s beaked whales (Indopacetus pacificus) in New Caledonia South Pacific. Mar Mamm Sci 32:884–910
Garrigue C, Derville S, Bonneville C et al. (2024) Marine mammal strandings recorded in New Caledonia, South West Pacific Ocean, 1877 to present. Pac Cons Biol 30:PC23016
Gbogbo F, Otoo SD, Huago RQ, Asomaning O (2017) High levels of mercury in wetland resources from three river basins in Ghana: a concern for public health. Environ Sci Pollut Res 24:5619–5627
Gentès S, Coquery M, Vigouroux R et al. (2019) Application of the European Water Framework Directive: Identification of reference sites and bioindicator fish species for mercury in tropical freshwater ecosystems (French Guiana). Ecol Indicators 106:105468
Gerson JR, Szponar N, Zambrano AA et al. (2022) Amazon forests capture high levels of atmospheric mercury pollution from artisanal gold mining. Nat Comm 13:559
Geyer WR, Ralston DK (2018) A mobile pool of contaminated sediment in the Penobscot Estuary Maine USA. Sci Total Environ 612:694–707
Giang A, Stokes LC, Streets DG (2015) Impacts of the Minamata Convention on mercury emissions and global deposition from coal-fired power generation in Asia. Environ Sci Technol 49:5326–5335
Gibb H, O’Leary KG (2014) Mercury exposure and health impacts among individuals in the artisanal and small-scale gold mining community: a comprehensive review. Environ Health Perspec 122:667–672
Gilmour CC, Podar M, Bullock AL (2013) Mercury methylation by novel microorganisms from new environments. Environ Sci Technol 47:11810–11820
Gilmour ME, Hudson ST, Lamborg C et al. (2019a) Tropical seabird sample broadscale patterns of marine contaminants. Sci Total Environ 691:631–643
Gilmour ME, Lavers JL, Lamborg C et al. (2019b) Mercury as an indicator of foraging ecology but not the breeding hormone prolactin in seabirds. Ecol Indicators 103:248–259
Gilmour ME, Holmes ND, Fleishman AB, Kriwoken LK (2019c) Temporal and interspecific variation in feather mercury in four penguin species from Macquarie Island, Australia. Marine Pollut Bull 142:282–289
Gimeno M, Giménez J, Chiaradia A, Davis LS, Seddon PJ, Ropert‐Coudert Y, Reisinger RR, Coll M, Ramírez F (2024) Climate and human stressors on global penguin hotspots: Current assessments for future conservation. Glob Change Biol 30(1):e17143
Gochfeld M, Burger J (2021) Mercury interactions with selenium and sulfur and the relevance of the Se: Hg molar ratio to fish consumption advice. Environ Sci Pollut Res 28:18407–18420
Gómez-Ramírez P, Bustnes JO, Eulaers I (2023) Mercury Exposure in Birds of Prey from Norway: Relation to Stable Carbon and Nitrogen Isotope Signatures in Body Feathers. Bull Environ Contam Toxicol 110:1–7
Gonzalez JDX, Aubrey A, Fernandez LE (2019) Mercury exposure risk factors and perceptions among women of childbearing age in an artisanal gold mining region of the Peruvian Amazon. Environ Res 108786. https://doi.org/10.1016/j.envres.2019.108786
Goodale MW, Evers DC, Mierzykowski SE et al. (2008) Marine foraging birds as bioindicators of mercury in the Gulf of Maine. EcoHealth 5:409–425
Gopakumar A, Giebichenstein J, Raskhozheva E, Borgå K (2021) Mercury in Barents Sea fish in the Arctic polar night: Species and spatial comparison. Mar Pollut Bull 169:112501
Gorbunov AV, Ermolaev BV, Lyapunov SM et al. (2016) Estimation of mercury intake from consumption of fish and seafood in Russia. Food Nutri Sci 7:516–523
Gorski PR, Armstrong DE, Hurley JP, Krabbenhoft DP (2008) Influence of natural dissolved organic carbon on the bioavailability of mercury to a freshwater alga. Environ Pollut 154:116–123
Goutner V, Furness RW (1997) Mercury in feathers of little egret Egretta garzetta and night heron Nycticorax nycticorax chicks and in their prey in the Axios Delta, Greece. Arch Environ Contam Toxicol 32:211–216
Goutner V, Furness RW, Papakostas G (2001) Mercury in feathers of Squacco Heron (Ardeola ralloides) chicks in relation to age, hatching order, growth, and sampling dates. Environ Pollut 111:107–115
Goutte A, Barbraud C, Meillère A et al. (2014a) Demographic consequences of heavy metals and persistent organic pollutants in a vulnerable long-lived bird the wandering albatross. Proc R Soc B 281:20133313
Goutte A, Bustamante P, Barbraud C et al. (2014b) Demographic responses to mercury exposure in two closely-related Antarctic top predators. Ecology 95:1075–1086
Grajewska A, Falkowska L, Reindl A (2019) Evaluation of claws as an alternative route of mercury elimination from the herring gull (Larus argentatus). Oceanological Hydrobiological Stud 48:165–173
Gray JS (2002) Biomagnification in marine systems: The perspective of an ecologist. Mar Pollut Bull 45(1-12):46–52. https://doi.org/10.1016/S0025-326X(01)00323-X
Green AD, Buhlmann KA, Hagen C et al. (2010) Mercury contamination in turtles and implications for human health. J Environ Health 72(10):14–22
Grgec AS, Kljakovic-Gašpic Z, Orct T et al. (2020) Mercury and selenium in fish from the eastern part of the Adriatic Sea: A risk-benefit assessment in vulnerable population groups. Chemosphere 261:127742
Grieb TM, Fisher NS, Karimi R, Levin L (2020) An assessment of temporal trends in mercury concentrations in fish. Ecotoxicol 29:1739–1749
Grove RA, Henny CJ, Kaiser JL (2009) Osprey: worldwide sentinel species for assessing and monitoring environmental contamination in rivers lakes reservoirs and estuaries. J Toxicol Environ Health B 12:25–44
Grunst AS, Grunst ML, Grémillet D et al. (2023) Mercury contamination challenges the behavioral response of a keystone species to Arctic climate change. Environ Sci Technol 57:2054–2063
Guédron S, Tisserand D, Garambois S et al. (2014) Baseline investigation of (methyl) mercury in waters soils sediments and key foodstuffs in the Lower Mekong Basin: The rapidly developing city of Vientiane (Lao PDR). J Geochemical Exploration 143:96–102
Gustin M, Evers DC, Bank M et al. (2016) Importance of integration and implementation of emerging and future mercury research into the Minamata Convention. Environ Sci Technol 50:2767–2770
Guzmán HM, Garcı́a EM (2002) Mercury levels in coral reefs along the Caribbean coast of Central America. Marine Pollut Bull 44:1415–1420
Guzmán HM, Kaiser S, van Hinsberg VJ (2020) Accumulation of trace elements in leatherback turtle (Dermochelys coriacea) eggs from the south-western Caribbean indicates potential health risks to consumers. Chemosphere 24:125424. https://doi.org/10.1016/j.chemosphere.2019.125424
Gworek B, Bemowska-Kalabun O, Kijenska M et al. (2016) Mercury in Marine and Oceanic Waters-a Review. Water Air Soil Pollut 227:371. https://doi.org/10.1007/s11270-016-3060-3
Ha E, Basu N, Bose-O’Reilly S et al. (2017) Current progress on understanding the impact of mercury on human health. Environ Res 152:419–433
Habiba G, Abebe G, Bravo AG et al. (2017) Mercury human exposure in populations living around Lake Tana (Ethiopia). Biol Trace Element Res 175:237–243
Hacon SS, Dórea JG, Fonseca MDF et al. (2014) The influence of changes in lifestyle and mercury exposure in riverine populations of the Madeira River (Amazon Basin) near a hydroelectric project. Int J Environ Res Public Health 11:2437–2455
Hacon SDS, Oliveira-da-Costa M, Gama CDS et al. (2020) Mercury exposure through fish consumption in traditional communities in the Brazilian Northern Amazon. Int J Environ Res Public Health 17:5269
Hammerschlag N, Davis DA, Mondo K et al. (2016) Cyanobacterial neurotoxin BMAA and mercury in sharks. Toxins 8:238
Hammerschlag N, Schmitz OJ, Flecker AJ et al. (2019) Ecosystem Function and Services of Aquatic Predators in the Anthropocene. Trends Ecol Evol 34:369–383
Hammerschlag N, Fallows C, Meÿer M et al. (2022) Loss of an apex predator in the wild induces physiological and behavioural changes in prey. Biol Lett 18:20210476
Hammerschmidt CR, Sandheinrich MB, Wiener JG, Rada RG (2002) Effects of dietary methylmercury on reproduction of fathead minnows. Environ Sci Technol 36:877–883
Hanna DE, Solomon CT, Poste AE et al. (2015) A review of mercury concentrations in freshwater fishes of Africa: Patterns and predictors. Environ Toxicol Chem 34:215–223
Hanna DEL, Buck DG, Chapman LJ (2016) Effects of habitat on mercury concentrations in fish: a case study of Nile perch (Lates niloticus) in Lake Nabugabo Uganda. Ecotoxicology 25:178–191
Hargreaves AL, Whiteside DP, Gilchrist G (2010) Concentrations of 17 elements including mercury and their relationship to fitness measures in arctic shorebirds and their eggs. Sci Total Environ 408:3153–3161
Harmelin-Vivien M, Cossa D, Crochet S (2009) Difference of mercury bioaccumulation in red mullets from the north-western Mediterranean and Black Seas. Mar Pollut Bull 58:679–685
Hartman CA, Ackerman JT, Herring G et al. (2013) Marsh wrens as bioindicators of mercury in wetlands of Great Salt Lake: Do blood and feathers reflect site-specific exposure risk to bird reproduction? Environ Sci Technol 47:6597–6605
Hartman CA, Ackerman JT, Herzog MP, Eagles-Smith CA (2017) Season molt and body size influence mercury concentrations in grebes. Environ Pollut 229:29–39
Head JA, DeBofsky A, Hinshaw J, Basu N (2011) Retrospective analysis of mercury content in feathers of birds collected from the state of Michigan (1895–2007). Ecotoxicol 20:1636–1643
Heath JA, Frederick PC (2005) Relationships among mercury concentrations, hormones, and nesting effort of White Ibises (Eudocimus albus) in the Florida Everglades. Auk 122:255–267
Heck S, Bene C, Reyes-Gaskin R (2007) Investing in African fisheries: Building links to the Millennium Development Goals. Fish and Fisheries. 8:211–226
Heimbürger LE, Sonke JE, Cossa D et al. (2015) Shallow methylmercury production in the marginal sea ice zone of the central Arctic Ocean. Sci Rep 5:10318
Heinz GH (1979) Methylmercury: reproductive and behavioral effects on three generations of mallard ducks. J Wildlife Manag 43:394–401
Heinz GH, Hoffman DJ, Klimstra JD et al. (2009) Species differences in the sensitivity of avian embryos to methylmercury. Arch Environ Contam Toxicol 56:129–138
Henny CJ, Kaiser JL, Grove RA (2009) PCDDs PCDFs PCBs OC pesticides and mercury in fish and osprey eggs from Willamette River Oregon (1993 2001 and 2006) with calculated biomagnification factors. Ecotoxicology 18:151–173
Henny CJ, Hill EF, Hoffman DJ et al. (2002) Nineteenth century mercury: hazard to wading birds and cormorants of the Carson River, Nevada. Ecotoxicol 11:213–231
Henny CJ, Hill EF, Grove RA, Kaiser JL (2007) Mercury and drought along the lower Carson River, Nevada: I. Snowy egret and black-crowned night-heron annual exposure to mercury, 1997–2006. Arch Environ Contam Toxicol 53:269–280
Herring G, Ackerman JT, Eagles‐Smith CA (2010) Embryo malposition as a potential mechanism for mercury‐induced hatching failure in bird eggs. Environ Toxicol Chem 29:1788–1794
Herring G, Eagles-Smith CA, Ackerman JT (2017) Mercury exposure may influence fluctuating asymmetry in waterbirds. Environ Toxicol Chem 36:1599–1605
Herring G, Eagles-Smith CA, Varland DE (2018) Mercury and lead exposure in avian scavengers from the Pacific Northwest suggest risks to California condors: Implications for reintroduction and recovery. Environ Pollut 243:610–619
Hill EF, Henny CJ, Grove RA (2008) Mercury and drought along the lower Carson River, Nevada: II. Snowy egret and black-crowned night-heron reproduction on Lahontan Reservoir, 1997–2006. Ecotoxicol 17:117–131
Hoffman DJ, Eagles‐Smith CA, Ackerman JT et al. (2011) Oxidative stress response of Forster’s terns (Sterna forsteri) and Caspian terns (Hydroprogne caspia) to mercury and selenium bioaccumulation in liver, kidney, and brain. Environ Toxicol Chem 30:920–929
Hollamby S, Afema-Azikuru J, Sikarskie JG et al. (2004) Mercury and persistent organic pollutant concentrations in African fish eagles, marabou storks, and Nile tilapia in Uganda. J Wildlife Dis 40(3):501–514
Hollanda S, Bodin N, Churlaud C, Bustamante P (2017) Mercury and Selenium Levels in Swordfish (Xiphias gladius) Fished in the Exclusive Economic Zone of the Republic of Seychelles. Int J Environ Ecol Eng 11:23–26
Honda K, Nasu T, Tatsukawa R (1986) Seasonal changes in mercury accumulation in the black-eared kite, Milvus migrans lineatus. Environ Pollut Ecol Biol 42:325–334
Hopkins WA, Hopkins LB, Unrine JM et al. (2007) Mercury concentrations in tissues of osprey from the Carolinas USA. J Wildlife Manag 71:1819–1829
Houde M, Taranu ZE, Wang X et al. (2020) Mercury in ringed seals (Pusa hispida) from the Canadian Arctic in relation to time and climate parameters. Environ Toxicol Chem 39:2462–2474
Houde M, Krümmel EM, Mustonen T et al. (2022) Contributions and perspectives of Indigenous Peoples to the study of mercury in the Arctic. Sci Total Environ 841:156566
Houssard P, Point D, Tremblay-Boyer L et al. (2019) A model of mercury distribution in tuna from the western and central Pacific Ocean: influence of physiology, ecology and environmental factors. Environ Sci Tech 53:1422–1431
Hsu-Kim H, Kucharzyk KH, Zhang T, Deshusses MA (2013) Mechanisms regulating mercury bioavailability for methylating microorganisms in the aquatic environment: a critical review. Environ Sci Technol 47:2441–2456
Hsu-Kim H, Eckley CS, Achá D et al. (2018) Challenges and opportunities for managing aquatic mercury pollution in altered landscapes. Ambio 47:141–169
Hu H, Wang B, Bravo AG et al. (2020) Shifts in mercury methylation across a peatland chronosequence: From sulfate reduction to methanogenesis and syntrophy. J Hazardous Mater 387:121967
Hughes KD, Ewins PJ, Clark KE (1997) A comparison of mercury levels in feathers and eggs of osprey (Pandion haliaetus) in the North American Great Lakes. Arch Environ Contam Toxicol 33:441–452
Hurtado TC, de Medeiros Costa G, de Carvalho GS, et al. (2023) Mercury and methylmercury concentration in the feathers of two species of Kingfishers Megaceryle torquata and Chloroceryle amazona in the Upper Paraguay Basin and Amazon Basin. Ecotoxicology 32:1084–1095
Ibañez AE, Mills WF, Bustamante P et al. (2024) Deleterious effects of mercury contamination on immunocompetence, liver function and egg volume in an antarctic seabird. Chemosphere 346:140630
IOTC (2018) Implementation of IOTC Conservation and Management Measures - Part A: Understanding IOTC and the international fisheries management framework Indian Ocean Tuna. Commission Food and Agric Organization of the United Nations Seychelles
IOTC (2020) Review of fishery trends for billfish. In: IOTC Proc Presented at the IOTC Working Party on Billfish. IOTC Online/Virtual p 57 Integrated Taxonomic Information System (ITIS) (2023) www.itis.gov, CC0. https://doi.org/10.5066/F7KH0KBK
Jackson AK, Evers DC, Folsom SB et al. (2011a) Mercury exposure in terrestrial birds far downstream of an historical point source. Environ Pollut 159:3302–3308
Jackson AK, Evers DC, Etterson MA et al. (2011b) Mercury exposure affects the reproductive success of free-living terrestrial songbird the Carolina wren (Thryothorus ludovicianus). Auk 128:759–769
Jackson A, Evers DC, Adams E et al. (2015) Mercury exposure in songbirds of eastern North America across habitats and guilds. Ecotoxicology 24:453–467
Jackson A, Evers DC, Eagles-Smith CA et al. (2016) Mercury risk to avian piscivores across the western United States and Canada. Sci Total Environ 568:685–696
Jackson AK, Eagles-Smith CA, Emery C (2020) Spatial variation in aquatic invertebrate and riparian songbird mercury exposure across a river-reservoir system with a legacy of mercury contamination. Ecotoxicol 29:1195–1204
Jagoe CH, Bryan AL, Brant HA et al. (2002) Mercury in bald eagle nestlings from South Carolina, USA. J Wildl Dis 38:706–712
Janssen SE, Kotalik CJ, Eagles-Smith CA et al. (2023) Mercury Isotope Values in Shoreline Spiders Reveal the Transfer of Aquatic Mercury Sources to Terrestrial Food Webs. Environ Sci Tech Lett 10:891–896
Jarolimek CV, King JJ, Apte SC et al. (2023) A review of inorganic contaminants in Australian marine mammals, birds and turtles. Environ Chem 20:147–170
Jewett SC, Duffy LK (2007) Mercury in fishes of Alaska, with emphasis on subsistence species. Sci Total Environ 387:3–27
Jinadasa BKKK, Fowler SW (2019) Critical review of mercury contamination in Sri Lankan fish and aquatic products. Mar Pollut Bull 149:110526
Jinadasa BKKK, Edirisinghe EMRKB, Wickramasinghe I (2013) Total mercury content, weight and length relationship in swordfish (Xiphias gladius) in Sri Lanka. Food Addit Contam Part B 6:244–248
Jinadasa BKKK, Chathurika GS, Jayaweera CD, Jayasinghe GDTM (2018) Mercury and cadmium in swordfish and yellowfin tuna and health risk assessment for Sri Lankan consumers. Food Addit Contam Part B12 2:75–80
Jing M, Lin D, Wu P et al. (2020) Effect of aquaculture on mercury and polyunsaturated fatty acids in fishes from reservoirs in Southwest China. Environ Pollut 257:113543
Johnels AG, Westermark T, Berg W et al. (1967) Pike (Esox lucius L) and some other aquatic organisms in Sweden as indicators of mercury contamination in the environment. Oikos 1967:323–333
Jonsson S, Andersson A, Nilsson MB et al. (2017) Terrestrial discharges mediate trophic shifts and enhance methylmercury accumulation in estuarine biota. Sci Adv 3:1601239
Joiris CR, Holsbeek L, Laroussi Moatemri N (1999) Total and methylmercury in sardines Sardinella aurita and Sardina pilchardus from Tunisia. Mar Pollut Bull 38:188–192
Jung E, Kim H, Yun D et al. (2022) Importance of hydraulic residence time for methylmercury accumulation in sediment and fish from artificial reservoirs. Chemosphere 293:133545
Kalisinska E, Gorecki J, Lanocha N et al. (2014) Total and methylmercury in soft tissues of white-tailed eagle (Haliaeetus albicilla) and osprey (Pandion haliaetus) collected in Poland. Ambio 43:858–870
Kamman NC, Burgess NM, Driscoll CT et al. (2005) Mercury in freshwater fish of northeast North America-a geographic perspective based on fish tissue monitoring databases. Ecotoxicology 14:163–180
Karagas MR, Choi AL, Oken W et al. (2012) Evidence on the human health effects of low-level methylmercury exposure. Environ Health Perspect 120:799–806
Kasper D, Forsberg BR, Amaral Joao HF et al. (2014) Reservoir stratification affects methylmercury levels in river water plankton and fish downstream from Balbina hydroelectric dam Amazonas Brazil. Environ Sci Technol 48:1032–1040
Keane, Bernaudat S, Davis L et al. (2023) Mercury and artisanal and small-scale gold mining: Review of global use estimates and considerations for promoting mercury-free alternatives. Ambio 52:833–852
Kenney LA, Eagles-Smith CA, Ackerman JT, von Hippel FA (2014) Temporal variation in fish mercury concentrations within lakes from the western Aleutian archipelago Alaska. PloS One 9(7):102244
Keppeler FW, Montaña CG, Winemiller KO (2020) The relationship between trophic level and body size in fishes depends on functional traits. Ecol Monographs 90:e01415
Kershaw JL, Hall AJ (2019) Merc ury in cetaceans: Exposure bioaccumulation and toxicity. Sci Total Environ 694:133683. https://doi.org/10.1016/j.scitotenv.2019.133683
Keskin Y, Baskaya R, Özyaral O et al. (2007) Cadmium lead mercury and copper in fish from the Marmara Sea Turkey. Bull Environ Contam Toxicol 78:258–261
Keyel ER, Etterson MA, Niemi GJ, et al. (2020) Feather mercury increases with feeding at higher trophic levels in two species of migrant raptors Merlin (Falco columbarius) and Sharp-shinned Hawk (Accipiter striatus). Condor 122. https://doi.org/10.1093/condor/duz069
Kidd KA, Bootsma HA, Hesslein RH et al. (2003) Mercury concentrations in the food web of Lake Malawi, East Africa. J Great Lakes Res 29:258–266
Klekowski EJ, Temple SA, Siung-Chang AM, Kumarsingh K (1999) An association of mangrove mutation, scarlet ibis, and mercury contamination in Trinidad, West Indies. Environ Pollut 105:185–189
Kim E, Kim H, Shin KH et al. (2012) Biomagnification of mercury through the benthic food webs of a temperate estuary: Masan Bay Korea. Environ Toxicol Chem 31:1254–1263
Kim H, Soerensen AL, Hur J et al. (2017) Methylmercury Mass Budgets and Distribution Characteristics in the Western Pacific Ocean. Environ Sci Technol 51:1186–1194
Kim SJ, Lee HK, Badejo AC et al. (2016) Species-specific accumulation of methyl and total mercury in sharks from offshore and coastal waters of Korea. Mar Pollut Bull 102:210–215
Kinghorn A, Solomon P, Chan HM (2007) Temporal and spatial trends of mercury in fish collected in the English-Wabigoon River system in Ontario Canada. Sci Total Environ 372:615–623
Kiszka JJ, Aubail A, Hussey NE et al. (2015) Plasticity of trophic interactions among sharks from the oceanic south-western Indian Ocean revealed by stable isotope and mercury analyses. Deep Sea Res Part I 96:49–58
Klenavic KL, Champoux PY, Daoust RD et al. (2008) Mercury concentrations in wild mink (Mustela vison) and river otters (Lontra canadensis) collected from eastern and Atlantic Canada: relationship to age and parasitism. Environ Pollut 156:359–366
Knott KK, Boyd D, Ylitalo GM, O’Hara TM (2012) Lactational transfer of mercury and polychlorinated biphenyls in polar bears. Chemosphere 88:395–402
Kocman D, Wilson SJ, Amos HM et al. (2017) Toward an Assessment of the Global Inventory of Present-Day Mercury Releases to Freshwater. Environments. Int J Environ Res Public Health 14:138
Kojadinovic JM, Potier M, Le Corre RP et al. (2006) Mercury content in commercial pelagic fish and its risk assessment in the Western Indian Ocean. Sci Total Environ 366:688–700
Kojadinovic J, Bustamante P, Churlaud C et al. (2007) Mercury in seabird feathers: Insight on dietary habits and evidence for exposure levels in the western Indian Ocean. Sci Total Environ 384:194–204
Kopec AD, Bodaly RA, Lane OP et al. (2018) Elevated mercury in blood and feathers of breeding marsh birds along the contaminated lower Penobscot River, Maine, USA. Sci Total Environ 634:1563–1579
Kortei NK, Heymann ME, Essuman EK et al. (2020) Health risk assessment and levels of toxic metals in fishes (Oreochromis noliticus and Clarias anguillaris) from Ankobrah and Pra basins: Impact of illegal mining activities on food safety. Toxicol Rep 7:360–369
Korstian JM, Chumchal MM, Bennett VJ, Hale AM (2018) Mercury contamination in bats from the central United States. Environ Toxicol Chem 37:160–165
Kouame, Bolou Bi LBC, Aka EB et al. (2020) Seasonality of Hg dynamics in the Ebrié Lagoon (Côte d’Ivoire) ecosystem: influence of biogeochemical factors. Environ Sci Pollut Res 27:19810–19825
Krey A, Ostertag SK, Chan HM (2015) Assessment of neurotoxic effects of mercury in beluga whales (Delphinapterus leucas) ringed seals (Pusa hispida) and polar bears (Ursus maritimus) from the Canadian Arctic. Sci Total Environ 509:237–247
Krümmel EM, Gilman A (2016) An update on risk communication in the Arctic. Int J Circumpolar Health 75:33822
Kucuksezgin F, Altay O, Uluturhan E, Kontas A (2001) Trace metal and organochlorine residue levels in red mullet (Mullus barbatus) from the Eastern Aegean Turkey. Wat Res 35:2327–2332
Kumari P, Maiti SK (2019) Health risk assessment of lead, mercury, and other metalloids: A potential threat to the population consuming fish inhabiting, a lentic ecosystem in Steel City (Jamshedpur), India. Human Ecol Risk Assess Inter 25:2174–2192
Kwon SY, Blum JD, Chen CY et al. (2014) Mercury isotope study of sources and exposure pathways of methylmercury in estuarine food webs in the Northeastern US. Environ Sci Technol 48:10089–10097
Kwon SY, Blum JD, Yin R et al. (2020) Mercury stable isotopes for monitoring the effectiveness of the Minamata Convention on Mercury. Earth Sci Rev 203:103111
Lailson-Brito J, Azeredo MA, Malm O et al. (2002) Trace metals in liver and kidney of the (franciscana Pontoporia blainvillei) from the northern coast of Rio de Janeiro state Brazil. Lat Am J Aquatic Mamm 1:107-114
Lailson-Brito J, Cruz R, Dorneles PR et al. (2012) Mercury-selenium relationships in liver of Guiana dolphin: the possible role of Kupffer cells in the detoxification process by tiemannite formation. PLoS One 7:42162
Lane OP, O’Brien KM, Evers DC et al. (2011) Mercury in breeding saltmarsh sparrows (Ammodramus caudacutus). Ecotoxicology 20:1984–1991
Lane OP, Arendt WJ, Tórrez MA, Castellón JCG (2013) Pilot assessment of mercury exposure in selected biota from the lowlands of Nicaragua. Mesoamericana 17:19–28
Lavoie RA, Jardine TD, Chumchal MM et al. (2013) Biomagnification of mercury in aquatic food webs: a worldwide meta-analysis. Environ Sci Technol 47:13385–13394
Le Croizier G, Lorrain A, Sonke JE et al. (2020) The twilight zone as a major foraging habitat and mercury source for the great white shark. Environ Sci Technol 54:15872–15882
Le Croizier G, Point D, Renedo M et al. (2022a) Mercury concentrations biomagnification and isotopic discrimination factors in two seabird species from the Humboldt Current ecosystem. Mar Pollut Bull 177:113481
Le Croizier G, Sonke JE, Lorrain A et al. (2022b) Foraging plasticity diversifies mercury exposure sources and bioaccumulation patterns in the world’s largest predatory fish. J Hazardous Mater 425:127956
Leah RT, Evans SJ, Johnson MS (1992) Mercury in flounder (Platichthys flesus L.) from estuaries and coastal waters of the north-east Irish Sea. Environ Pollut 75:317–322
Lebel JM, Roulet D, Mergler M et al. (1997) Fish diet and mercury exposure in a riparian Amazonian population. Water Air Soil Pollut 97:31–44
Leclère D, Obersteiner M, Barrett M et al. (2020) Bending the curve of terrestrial biodiversity needs an integrated strategy. Nature 585:551–556
Lee CS, Fisher NS (2016) Methylmercury uptake by diverse marine phytoplankton. Limnol Oceanogr 61:1626–1639
Lee CS, Lutcavage ME, Chandler E et al. (2016) Declining mercury concentrations in bluefin tuna reflect reduced emissions to the North Atlantic Ocean. Environ Sci Technol 50:12825–12830
Lehnherr I, St Louis VL, Hintelmann H, Kirk JL (2011) Methylation of inorganic mercury in polar marine waters. Nature Geosci 4:298–302
Lepak RF, Hoffman JC, Janssen SE, Krabbenhoft DP, Ogorek JM, DeWild JF, Tate MT, Babiarz CL, Yin R, Murphy EW, Engstrom DR (2019) Mercury source changes and food web shifts alter contamination signatures of predatory fish from Lake Michigan. Proc Natl Acad Sci 116(47):23600–23608
Lescord GL, Johnston TA, Branfireun BA, Gunn JM (2018) Percentage of methylmercury in the muscle tissue of freshwater fish varies with body size and age and among species. Environ Toxicol Chem 37:2682–2691
Li M, Schartup AT, Valberg AP et al. (2016) Environmental origins of methylmercury accumulated in subarctic estuarine fish indicated by mercury stable isotopes. Environ Sci Technol 50:11559–11568
Li ML, Kwon SY, Poulin BA et al. (2022) Internal dynamics and metabolism of mercury in biota: A review of insights from mercury stable isotopes. Environ Sci Technol 56:9182–9195
Lian M, Field CL, van Wijngaarden E et al. (2021) Assessment of clinical outcomes associated with mercury concentrations in harbor seal pups (Phoca vitulina richardii) in central California. Sci Total Environ 758:143686
Lino AS, Kasper D, Guida YS et al. (2019) Total and methyl mercury distribution in water sediment plankton and fish along the Tapajós River basin in the Brazilian Amazon. Chemosphere 235:690–700
Lippold A, Aars J, Andersen M et al. (2021) Two decades of mercury concentrations in Barents Sea polar bears (Ursus maritimus) in relation to dietary carbon sulfur and nitrogen. Environ Sci Technol 54:7388–7397
Lippold A, Boltunov A, Aars J et al. (2022) Spatial variation in mercury concentrations in polar bear (Ursus maritimus) hair from the Norwegian and Russian. Arctic. Sci Total Environ 822:153572
Little ME, Burgess NM, Broders HG, Campbell LM (2015) Distribution of mercury in archived fur from little brown bats across Atlantic Canada. Environ Pollut 207:52–58
Liu B, Yan H, Wang C et al. (2012) Insights into low fish mercury bioaccumulation in a mercury-contaminated reservoir Guizhou China. Environ Pollut 160:109–117
Lobus NV, Komov VT, Thanh NTH (2011) Mercury concentration in ecosystem components in water bodies and streams in Khanh Hoa province (Central Vietnam). Water Resour 38:799–805
Lobus NV, Komov VT (2016) Mercury in the muscle tissue of fish in the Central and South Vietnam. Inland Water Biol 9:319–328
López-Berenguer G, Peñalver J, Martínez-López E (2020) A critical review about neurotoxic effects in marine mammals of mercury and other trace elements. Chemosphere 246:125688
Lowry LF, Frost JK, Burns JJ (1980) Variability in the diet of ringed seals Phoca hispida in Alaska. Can J Fish Aquat Sci 37:2254–2261
Lucia M, Bocher P, Chambosse M, Delaporte P, Bustamante P (2014) Trace element accumulation in relation to trophic niches of shorebirds using intertidal mudflats. J Sea Res 92:134–143
Lucia M, Bocher P, Cosson RP, Churlaud C, Bustamante P (2012) Evidence of species-specific detoxification processes for trace elements in shorebirds. Ecotoxicol 21:2349–2362
Lucia M, Verboven N, Strøm H et al. (2015) Circumpolar contamination in eggs of the high-Arctic ivory gull Pagophila eburnea. Environ Toxicol Chem 34:1552–1561
Lucia M, Strøm H, Bustamante P, Gabrielsen GW (2016) Trace element concentrations in relation to the trophic behaviour of endangered Ivory Gulls (Pagophila eburnea) during their stay at a breeding site in Svalbard. Arch Environ Contam Toxicol 71:518–529
Ma Y, Branfireun BA, Hobson KA, Guglielmo CG (2018a) Evidence of negative seasonal carry‐over effects of breeding ground mercury exposure on survival of migratory songbirds. J Avian Biol 49:01656
Ma Y, Perez CR, Branfireun BA, Guglielmo CG (2018b) Dietary exposure to methylmercury affects flight endurance in a migratory songbird. Environ Pollut 234:894–901
Ma Y, Zheng W, An Y et al. (2021) Mercury contamination in terrestrial predatory birds from Northeast China: Implications for species and feather type selection for biomonitoring. Ecologic Indicat 130:108108
Machovsky-Capuska GE, von Haeften G, Romero MA et al. (2020) Linking cadmium and mercury accumulation to nutritional intake in common dolphins (Delphinus delphis) from Patagonia Argentina. Environ Pollut 263:114480
MacMillan GA, Amyot M, Daoust PY, Lemire M (2022) Age-specific trace element bioaccumulation in grey seals from the Gulf of St Lawrence. Chemosphere 294:133640
Maffucci F, Caurant F, Bustamante P et al. (2005) Trace element (Cd Cu Hg Se Zn) accumulation and tissue distribution in loggerhead turtles (Caretta caretta) from the Western Mediterranean Sea (southern Italy). Chemosphere 58:535–542
Maher W, Krikowa F, Ellwood M (2020) Mercury cycling in Australian estuaries and near shore coastal ecosystems: Triggers for management. Elem Sci Anth 8:29
Makaure J, Dube T, Stewart D, Razavi NR (2023) Mercury Exposure in Two Fish Trophic Guilds from Protected and ASGM-Impacted Reservoirs in Zimbabwe and Possible Risks to Human Health. Arch Environ Contam Toxicol 84:199–213
Mallory ML, Wayland M, Braune BM, Drouillard KG (2004) Trace elements in marine birds, arctic hare and ringed seals breeding near Qikiqtarjuaq Nunavut Canada. Mar Pollut Bull 49:136–141
Mallory ML, Braune BM (2012) Tracking contaminants in seabirds of Arctic Canada: Temporal and spatial insights. Mar Pollut Bull 64:1475–1484
Mallory ML, Provencher JF, Robertson GJ et al. (2018) Mercury concentrations in blood brain and muscle tissues of coastal and pelagic birds from northeastern Canada. Ecotoxicol Environ Saf 157:424–430
Malm O (1998) Gold mining as a source of mercury exposure in the Brazilian Amazon. Environ Res 77:73–78
Malm O, Branches FJP, Akagi H et al. (1995) Mercury and methylmercury in fish and human hair from the Tapajós river basin Brazil. Sci Total Environ 175:141–150
Manceau A, Azemard S, Hédouin L et al. (2021a) Chemical forms of mercury in blue marlin billfish: Implications for human exposure. Environ Sci Technol Lett 8:405–411
Manceau A, Gaillot AC, Glatzel P et al. (2021b) In vivo formation of HgSe nanoparticles and Hg-tetraselenolate complex from methylmercury in seabird – Implications for the Hg-Se antagonism. Environ Sci Technol 55:1515–1526
Manceau A, Brossier R, Janssen SE et al. (2021c) Mercury isotope fractionation by internal demethylation and biomineralization reactions in seabirds: Implications for environmental mercury science. Environ Sci Technol 55:13942–13952
Manhães BMR, Santos-Neto EB, Tovar LR et al. (2021) Changes in mercury distribution and its body burden in delphinids affected by a morbillivirus infection: Evidences of methylmercury intoxication in Guiana dolphin. Chemosphere 263:128286
Manhães BMR, Vannuci-Silva M, Briao JA et al. (2022) Temporal trends of trace elements bioaccumulation by a vulnerable cetacean (Pontoporia blainvillei) before and after one of the largest mining disasters worldwide. Sci Total Environ 804:150196
Marcovecchio JE, Gerpe MS, Bastida RO (1994) Environmental contamination and marine mammals in coastal waters from Argentina: an overview. Sci Total Environ 154:141–151
Marcovecchio JE, Andrade S, Ferrer LD et al. (2001) Mercury distribution in estuarine environments from Argentina: the detoxification and recovery of salt marshes after 15 years. Wetlands Ecol Manage 9:317–322
Martyniuk MAC, Couture P, Tran L et al. (2020) Seasonal variation of total mercury and condition indices of Arctic charr (Salvelinus alpinus) in Northern Québec, Canada. Sci Total Environ 738:139450
Mason RP, Baumann Z, Hansen G et al. (2019) An assessment of the impact of artisanal and commercial gold mining on mercury and methylmercury levels in the environment and fish in Cote d’Ivoire. Sci Total Environ 665:1158–1167
Mason RP, Coulibaly M, Hansen G et al. (2022) An examination of mercury levels in the coastal environment and fish of Cote d’Ivoire. Chemosphere 300:134609
Matias RS, Guímaro HR, Bustamante P et al. (2022) Mercury biomagnification in an Antarctic food web of Antarctic Peninsula. Environ Pollut 304:119199
Martinez G, McCord SA, Driscoll CT et al. (2018) Mercury contamination in riverine sediments and fish associated with artisanal and small-scale gold mining in Madre de Dios Peru. Int J Environ Res Public Health 15:1584
Matulik AG, Kerstetter DW, Hammerschlag N et al. (2017) Bioaccumulation and biomagnification of mercury and methylmercury in four sympatric coastal sharks in a protected subtropical lagoon. Mar Pollut Bull 116:357–364
Mauffret A, Chouvelon T, Wessel N et al. (2023) Trace elements Dioxins and PCBs in different fish species and marine regions: importance of the taxon and regional features. Environ Res 216:114624
Maulvault AL, Custódio A, Anacleto P et al. (2016) Bioaccumulation and elimination of mercury in juvenile seabass (Dicentrarchus labrax) in a warmer environment. Environ Res 149:77–85
Maurice L, Le Croizier G, Morales G et al. (2021) Concentrations and stable isotopes of mercury in sharks of the Galapagos Marine Reserve: Human health concerns and feeding patterns. Ecotoxicol Environ Saf 215:112122
May Junior JA, Quigley H, Hoogesteijn R et al. (2017) Mercury content in the fur of jaguars (Panthera onca) from two areas under different levels of gold mining impact in the Brazilian Pantanal. Anais da Academia Brasileira de Ciências. https://doi.org/10.1590/0001-3765201720170190
Maz-Courrau A, López-Vera C, Galvan-Magaña F et al. (2012) Bioaccumulation and biomagnification of total mercury in four exploited shark species in the Baja California Peninsula Mexico. Bull Environ Contam Toxicol 88:129–134
McCormack MA, Fielding R, Kiszka JJ et al. (2020) Mercury and selenium concentrations and selenium: mercury molar ratios in small cetaceans taken off St Vincent West Indies. Environ Res 181:108908
McKinney MA, Pedro S, Dietz R et al. (2015) A review of ecological impacts of global climate change on persistent organic pollutant and mercury pathways and exposures in arctic marine ecosystems. Current Zool 61:617–628
McKinney MA, Dean K, Hussey NE et al. (2016) Global versus local causes and health implications of high mercury concentrations in sharks from the east coast of South Africa. Sci Total Environ 541:176–183
McKinney MA, Chételat J, Burke SM et al. (2022) Climate change and mercury in the Arctic: biotic interactions. Sci Total Environ 834:155221
McKinney R, Gibbon J, Wozniak E, Galland G (2020) Netting Billions 2020: A Global Tuna Valuation. The Pew Charitable Trusts, Washington DC USA, p 31
Médieu A, Point D, Receveur A et al. (2021) Stable mercury concentrations of tropical tuna in the southwestern Pacific Ocean: An 18-year monitoring study. Chemosphere 263:128024
Médieu A, Point D, Itai T et al. (2022) Evidence that Pacific tuna mercury levels are driven by marine methylmercury production and anthropogenic inputs. Proc Natl Acad Sci USA 119:e2113032119
Médieu A, Lorrain A, Point D (2023) Are tunas relevant bioindicators of mercury concentrations in the global ocean? Ecotoxicology 32:994–1009
Médieu A, Point D, Sonke JE et al. (2024) Stable Tuna Mercury Concentrations since 1971 Illustrate Marine Inertia and the Need for Strong Emission Reductions under the Minamata Convention. Environ Sci Technol Lett. https://doi.org/10.1021/acs.estlett.3c00949
Medina-Morales SA, Corro-Espinosa D, Escobar-Sánchez O et al. (2020) Mercury (Hg) and selenium (Se) content in the shark Mustelus henlei (Triakidae) in the northern Mexican Pacific. Environ Sci Pollut Res 27:16774–16783
Mendez E, Giudice H, Pereira A et al. (2001) Total mercury content-fish weight relationship in swordfish (Xiphias gladius) caught in the southwest Atlantic Ocean. J Food Compos Anal 14:453–460
Merly L, Lange L, Meÿer M et al. (2019) Blood plasma levels of heavy metals and trace elements in white sharks (Carcharodon carcharias) and potential health consequences. Mar Pollut Bull 142:85–92
Meyer MW, Evers DC, Hartigan JJ, Rasmussen PS (1998) Patterns of common loon (Gavia immer) mercury exposure, reproduction, and survival in Wisconsin, USA. Environ Toxicol Chem 17:184–190
Meyer MW, Rasmussen PW, Watras CJ et al. (2011) Bi-phasic trends in mercury concentrations in blood of Wisconsin common loons during 1992–2010. Ecotoxicol 20:1659–1668
Miljeteig C, Strom H, Gavrilo MV et al. (2009) High levels of contaminants in Ivory Gull Pagophila eburnea eggs from the Russian and Norwegian Arctic. Environ Sci Technol 43:5521–5528
Millard G, Driscoll C, Montesdeoca M et al. (2020) Patterns and trends of fish mercury in New York State. Ecotoxicology 29:1709–1720
Millot C, Taupier-Letage I (2005) Circulation in the Mediterranean Sea: Updated description and schemas of the circulation of the water masses in the whole Mediterranean Sea. In: Saliot A (ed) The Mediterranean Sea (5-K) Handbook of Environmental Chemistry. Springer, New York, USA, p 29–66
Mills WF, Bustamante P, McGill RA, Anderson OR et al. (2020) Mercury exposure in an endangered seabird: long-term changes and relationships with trophic ecology and breeding success. Proc Royal Soc B 287:20202683
Mills WF, Ibañez AE, Bustamante P et al. (2022) Spatial and sex differences in mercury contamination of skuas in the Southern Ocean. Environ Pollut 297:118841
Mohammed A, Mohammed T (2017) Mercury, arsenic, cadmium and lead in two commercial shark species (Sphyrna lewini and Caraharinus porosus) in Trinidad and Tobago. Mar Pollut Bull 119:214–218
Mol JH, Ramlal JS, Lietar C, Verloo M (2001) Mercury contamination in freshwater estuarine and marine fishes in relation to small-scale gold mining in Suriname South America. Environ Res 86:183–197
Mondragón ME, Luzardo OP, Henríquez-Hernández LA et al. (2023) Trophic behavior of inorganic elements in nesting sea turtles (Chelonia mydas, Eretmochelys imbricata, and Caretta caretta) in Quintana Roo: Biomagnification and biodilution effect in blood and scute tissues. Mar Pollut Bull 187:114582
Monson BA (2009) Trend reversal of mercury concentrations in piscivorous fish from Minnesota Lakes: 1982- 2006. Environ Sci Technol 43:1750–1755
Monson BA, Staples DF, Bhavsar SP et al. (2011) Spatiotemporal trends of mercury in walleye and largemouth bass from the Laurentian Great Lakes region. Ecotoxicology 20:1555–1567
Montaña CG, Liverpool E, Taphorn DC, Schalk CM (2021) The cost of gold: Mercury contamination of fishes in a neotropical river food web. Neotropical Ichthyology 19:3
Monteiro LR, Furness RW (1995) Seabirds as monitors of mercury in the marine environment. Water Air Soil Pollut 80:851–870
Monteiro LR, Furness RW (2001) Kinetics dose- response and excretion of methylmercury in free-living adult Cory’s shearwaters. Environ Sci Technol 35:739–746
Morcillo P, Esteban MA, Cuesta A (2017) Mercury and its toxic effects on fish. AIMS Environ Sci 4:386–402
Moreno-Brush M, Rydberg J, Gamboa N et al. (2016) Is mercury from small-scale gold mining prevalent in the southeastern Peruvian Amazon? Environ Pollut 218:150–159
Morris AD, Wilson SJ, Fryer RJ et al. (2022a) Temporal trends of mercury in Arctic biota: 10 more years of progress in Arctic monitoring. Sci Total Environ 839:155803
Morris AD, Braune BM, Gamberg M et al. (2022b) Temporal change and the influence of climate and weather factors on mercury concentrations in Hudson Bay polar bears, caribou, and seabird eggs. Environ Res 207:112169
Motas M, Jerez S, Esteban M et al. (2021) Mercury levels in feathers of penguins from the Antarctic Peninsula area: geographical and inter-specific differences International. J Environ Res Public Health 18:9918
Mott R, Herrod A, Clarke RH (2017) Post-breeding dispersal of frigatebirds increases their exposure to mercury. Mar Pollut Bull 119:204–210
Muir D, Braune B, DeMarch B et al. (1999) Spatial and temporal trends and effects of contaminants in the Canadian Arctic marine ecosystem: A review. Sci Total Environ 230:83–144
Muirhead SJ, Furness RW (1988) Heavy metal concentrations in the tissues of seabirds from Gough Island South Atlantic Ocean. Mar Pollut Bull 19:278–283
Mullié WC, Massi A, Focardi S, Renzoni A (1992) Residue levels of organochlorines and mercury in cattle egret, Bubulcus ibis, eggs from the Faiyum Oasis, Egypt. Bull Environ Contamin Toxicol 48:739–746
Munson KM, Lamborg CH, Swarr GJ, Saito MA (2015) Mercury species concentrations and fluxes in the Central Tropical Pacific Ocean. Global Biogeochem Cycles 29:656–676
Mussy MH, de Ronaldo A, de Carvalho DP et al. (2022) Evaluating total mercury and methylmercury biomagnification using stable isotopes of carbon and nitrogen in fish from the Madeira River basin Brazilian Amazon. Environ Sci Pollut Res 30:1–12
Naves LC (2009) Alaska Migratory Bird Subsistence Harvest Estimates 2004-2007. Alaska Migratory Bird Co-Management Council, Anchorage
Naves LC (2018) Geographic and seasonal patterns of seabird subsistence harvest in Alaska. Polar Biol 41:1217–1236
Negrazis L, Kidd KA, Erdozain M et al. (2022) Effects of forest management on mercury bioaccumulation and biomagnification along the river continuum. Environ Pollut 310:119810
Nevado JB, Martín-Doimeadios RR, Bernardo FG et al. (2010) Mercury in the Tapajós River basin Brazilian Amazon: a review. Environ Int 36:593–608
Newtoff KN, Emslie SD (2017) Mercury Exposure and Diet in Brown Pelicans (Pelecanus occidentalis) in North Carolina USA. Waterbirds 40:50–57
Newton I, Dale L, Little B (1999) Trends in organochlorine and mercurial compounds in the eggs of British merlins (Falco columbarius). Bird Study 46:356–362
Nguetseng R, Fliedner A, Knopf B et al. (2015) Retrospective monitoring of mercury in fish from selected European freshwater and estuary sites. Chemosphere 134:427–434
Niane B, Guédron S, Moritz R et al. (2015) Human exposure to mercury in artisanal small-scale gold mining areas of Kedougou region Senegal as a function of occupational activity and fish consumption. Environ Sci Pollut Res 22:7101–7111
Nicklisch SC, Bonito LT, Sandin S, Hamdoun A (2017) Mercury levels of yellowfin tuna (Thunnus albacares) are associated with capture location. Environ Pollut 229:87–93
Nicolaus EM, Bendall VA, Bolam TP et al. (2016) Concentrations of mercury and other trace elements in porbeagle shark Lamna nasus. Mar Pollut Bull 112:406–410
Noh S, Kim CK, Kim Y et al. (2017) Assessing correlations between monomethylmercury accumulation in fish and trophic states of artificial temperate reservoirs. Sci Total Environ 580:912–919
Nyholt K, Jardine TD, Villamarín F et al. (2022) High rates of mercury biomagnification in fish from Amazonian floodplain-lake food webs. Sci Total Environ 833:155161
O’Bryhim JR, Adams DH, Spaet JL et al. (2017) Relationships of mercury concentrations across tissue types, muscle regions and fins for two shark species. Environ Pollut 223:323–333
Obrist D, Kirk JL, Zhang J et al. (2018) A review of global environmental mercury processes in response to human and natural perturbations: Changes of emissions climate and land use. Ambio 47:116–140
Ochoa-Acuña H, Sepulveda MS, Gross TS (2002) Mercury in feathers from Chilean birds: Influence of location feeding strategy and taxonomic affiliation. Mar Pollut Bull 44:340–349
Odsjö T, Roos A, Johnels AG (2004) The Tail Feathers of Osprey Nestlings (Pandion haliaetus L) as Indicators of Change in Mercury Load in the Environment of Southern Sweden (1969–1998): Case Study with a Note on the Simultaneous Intake of Selenium. Ambio: A J Human Environ 33:133–137
Oliveira E, Ignácio ARA, Lázaro WL, et al. (2023) Green kingfishers as sentinel species for mercury contamination in Amazon. Arch Environ Contam Toxicol 85:34–45
Oliveira RC, Dorea JG, Bernardi JVE et al. (2010) Fish consumption by traditional subsistence villagers of the Rio Madeira (Amazon): impact on hair mercury. Ann Hum Biol 37:629–642
Olivero J, Solano B, Acosta I (1998) Total mercury in muscle of fish from two marshes in goldfields, Colombia. Bull Environ Contam Toxicol 61:182–187
Olivero-Verbel J, Johnson-Restrepo B, Baldiris-Avila R et al. (2008) Human and crab exposure to mercury in the Caribbean coastal shoreline of Colombia: impact from an abandoned chlor-alkali plant. Environ Internat 34:476–482
Olivero-Verbel J, Caballero-Gallardo K, Turizo-Tapia A (2015) Mercury in the gold mining district of San Martin de Loba South of Bolivar (Colombia). Environ Sci Pollut Res 22:5895–5907
Ono K, Knutsen H, Olsen EM et al. (2019) Possible adverse impact of contaminants on Atlantic cod population dynamics in coastal ecosystems. Proc R Soc B 286:20191167
Otsuki M, Ogawa M, Yutaka W (2024) Brood patch size as a field indicator for feather mercury concentration, but not plastic ingestion, in a harvested seabird of the high Arctic: The little auk Alle alle. Polar Sci 101053 https://doi.org/10.1016/j.polar.2024.101053
Ouboter PE, Landburg GA, Quik JH et al. (2012) Mercury levels in pristine and gold mining impacted aquatic ecosystems of Suriname, South America. Ambio 41:873–882
Ouboter PE, Landburg GA, Satnarain GU et al. (2018) Mercury levels in women and children from interior villages in Suriname, South America. Int J Environ Res Public Health 15:1007
Ouédraogo O, Amyot M (2013) Mercury arsenic and selenium concentrations in water and fish from sub-Saharan semi-arid freshwater reservoirs (Burkina Faso). Sci Total Environ 444:243–254
Outridge PM, Mason RP, Wang F et al. (2018) Updated global and oceanic mercury budgets for the United Nations Global Mercury Assessment 2018. Environ Sci Technol 52:11466–11477
Packull-McCormick S, Cowan A, Stark KD et al. (2023) Mercury bioaccessiblity in freshwater fish species from northern Canada. Sci Total Environ 899:165624
Pacoureau N, Rigby CL, Kyne PM et al. (2021) Half a century of global decline in oceanic sharks and rays. Nature 589:567–571
Pacyna JM, Travnikov O, De Simone F et al. (2016) Current and future levels of mercury atmospheric pollution on a global scale. Atmos Chem Phys 16(19):12495
Pacyna AD, Martínez CZ, Miguélez D et al. (2017) Mercury contamination a potential threat to the globally endangered aquatic warbler (Acrocephalus paludicola). Environ Sci Pollut Res 24:26478–26484
Pacyna-Kuchta AD, Jakubas D, Frankowski M et al. (2020) Exposure of a small Arctic seabird the little auk (Alle alle) breeding in Svalbard to selected elements throughout the course of a year. Sci Total Environ 732:139103
Pal M, Ghosh S, Mukhopadhyay M, Ghosh M (2011) Methyl mercury in fish-a case study on various samples collected from Ganges River at West Bengal. Environ Monit. Assessment 184:3407–3414
Palacios-Torres Y, Caballero-Gallardo K, Olivero-Verbel J (2018) Mercury pollution by gold mining in a global biodiversity hotspot the Choco biogeographic region Colombia. Chemosphere 193:421–430
Pandiyan J, Jagadheesan R, Karthikeyan G et al. (2020) Probing of heavy metals in the feathers of shorebirds of Central Asian Flyway wintering grounds. Scient Rep 10:22118
Passos CJ, Da Silva DS, Lemire M et al. (2008) Daily mercury intake in fish-eating populations in the Brazilian Amazon. J Expo Sci Environ Epidemiol 18:76–87
Paulus M, Teubner D, Rüdel H, Klein R (2015) Bioaccumulation and Long-Term Monitoring in Freshwater Ecosystems-Knowledge Gained from 20 Years of Zebra Mussel Analysis by the German Environmental Specimen Bank in Environmental Indicators. Springer, Dordrecht, p 781–803
Pauly D, Watson R (2005) Background andinterpretation of the ‘Marine Trophic Index’as a measure of biodiversity. Philosophical Transactions of the Royal Society B: Biological Sciences 360(1454):415–423
Pelletier AR, Castello L, Zhulidov AV et al. (2017) Temporal and longitudinal mercury trends in burbot (Lota lota) in the Russian Arctic. Environ Sci Technol 51:13436–13442
Perkins M, Basu N (2018) Dried blood spots for estimating mercury exposure in birds. Environ Pollut 236:236–246
Perkins M, Ferguson L, Lanctot RB et al. (2016) Mercury exposure and risk in breeding and staging Alaskan shorebirds. Condor 118:571–582
Perkins M, Lane OP, Evers DC et al. (2020) Historical patterns in mercury exposure for North American songbirds. Ecotoxicol 29:1161–1173
Pestana IA, Almeida MG, Bastos WR, Souza CM (2019) Total Hg and methylmercury dynamics in a river-floodplain system in the Western Amazon: Influence of seasonality, organic matter and physical and chemical parameters. Sci Total Environ 656:388–399
Perkins M, Stenhouse IJ, Lanctot RB, Brown S, Bêty J, Boldenow M, Cunningham J, English W, Gates R, Gilchrist HG, Giroux MA (2023) Factors influencing mercury exposure in Arctic-breeding shorebirds. Ecotoxicol 32:1062–1083
Peterson SA, Van Sickle J, Hughes RM et al. (2004) A biopsy procedure for determining filet and predicting whole-fish mercury concentration. Arch Environ Contam Toxicol 48:99–107
Peterson SH, Ackerman JT, Costa DP (2015) Marine foraging ecology influences mercury bioaccumulation in deep-diving northern elephant seals. Proc R Soc B 282:20150710
Peterson SH, Ackerman JT, Costa DP (2016a) Mercury correlations among blood muscle and hair of northern elephant seals during the breeding and molting fasts. Environ Toxicol Chem 35:2103–2110
Peterson SH, McHuron EA, Kennedy SN et al. (2016b) Evaluating hair as a predictor of blood mercury: the influence of ontogenetic phase and life history in pinnipeds. Arch Environ Contam Toxicol 70:28–45
Peterson SH, Ackerman JT, Eagles-Smith CA et al. (2017) A critical evaluation of the utility of eggshells for estimating mercury concentrations in avian eggs. Environ Toxicol Chem 36:2417–2427
Peterson SH, Ackerman JT, Crocker DE, Costa DP (2018) Foraging and fasting can influence contaminant concentrations in animals: an example with mercury contamination in a free-ranging marine mammal. Proc R Soc B 285:20172782
Peterson SP, Ackerman JT, Toney M, Herzog MP (2019) Mercury concentrations vary within and among individual bird feathers: A critical evaluation and guidelines for feather use in mercury monitoring programs. Environ Toxicol Chem 38:1164–1187
Petrova MV, Ourgaud M, Boavida JRH et al. (2020) Human mercury exposure levels and fish consumption at the French Riviera. Chemosphere 258:127232
Pickhardt PC, Folt CL, Chen CY et al. (2002) Algal blooms reduce the uptake of methylmercury in freshwater food webs. Proc Nat Acad Sci 99:4419–4423
Pilcher N, Gaw S, Eisert R et al. (2020) Latitudinal sex and inter-specific differences in mercury and other trace metal concentrations in Adélie and Emperor penguins in the Ross Sea Antarctica. Mar Pollut Bull 154:111047
Pinkney AE, Driscoll CT, Evers DC et al. (2015) Interactive effects of climate change with nutrients mercury and freshwater acidification on key taxa in the North Atlantic Landscape Conservation Cooperative region. Integ Environ Assess Manag 11:355–369
Piro AJ, Taipale SJ, Laiho HM (2023) Fish muscle mercury concentration and bioaccumulation fluctuate year-round-Insights from cyprinid and percid fishes in a humic boreal lake. Environ Res 231:116187
Pisconte JN, Vega CM, Sayers CJ et al. (2024) Elevated mercury exposure in bird communities inhabiting Artisanal and Small-Scale Gold Mining landscapes of the southeastern Peruvian Amazon. Ecotoxicology https://doi.org/10.1007/s10646-024-02740-4
Pollet IL, Provencher JF, Tranquilla LM et al. (2022) Mercury levels in North Atlantic seabirds: A synthesis. Mar Pollut Bull 181:113884
Pollet IL, McFarlane-Tranquilla L, Burgess NM et al. (2023) Factors influencing mercury levels in Leach’s storm-petrels at northwest Atlantic colonies. Sci Total Environ 860:160464
Potera C (2019) A Global Look at Mercury Exposures: Supporting the Goals of the Minamata Convention. Environ Health Perspec 127(8):084001
Pratte I, Noble DG, Mallory ML et al. (2020) The influence of migration patterns on exposure to contaminants in Nearctic shorebirds: A historical study. Environ Monit Assess 192:256
Provencher JF, Mallory ML, Braune BM et al. (2014) Mercury and marine birds in Arctic Canada: effects current trends and why we should be paying closer attention. Environ Rev 22:244–255
Qiu F, Meng XZ, Qiu YL et al. (2015) Historical development and future perspectives of Environmental Specimen Bank in China: a mini review. Environ Sci Pollut Res 22:1562–1567
Qiu G, Feng X, Wang S et al. (2009) Mercury distribution and speciation in water and fish from abandoned Hg mines in Wanshan Guizhou province China. Sci Total Environ 407:5162–5168
Quadri-Adrogué A, Gómez-Ramírez P, García-Fernández AJ et al. (2022) Feather mercury levels in beached Magellanic penguin (Spheniscus magellanicus) in northern Argentina during the non-breeding season. Environ Sci Pollut Res 29:24793–24801
Quillfeldt P, Bedolla-Guzmán Y, Libertelli MM et al. (2023) Mercury in Ten Storm-Petrel Populations from the Antarctic to the Subtropics. Arch Environ Contam Toxicol 85:55–72
Rajaee M, Obiri S, Green A et al. (2015) Integrated assessment of artisanal and small-scale gold mining in Ghana-Part 2: Natural sciences review. Int J Environ Res Public Health 12:8971–9011
Rask M, Malinen T, Nyberg K et al. (2024) Pike Mercury Concentration in Small Boreal Headwater Lakes During Four Decades of Regional and Local Changes. Water Air Soil Pollut 235:85
Razavi NR, Qu M, Jin B et al. (2014) Mercury biomagnification in subtropical reservoir fishes of eastern China. Ecotoxicology 23:133–146
Rea LD, Castellini JM, Avery JP et al. (2020) Regional variations and drivers of mercury and selenium concentrations in Steller sea lions. Sci Total Environ 744:140787
Reif JS, Schaefer AM, Bossart GD (2015) Atlantic bottlenose dolphins (Tursiops truncatus) as a sentinel for exposure to mercury in humans: closing the loop. Vet Sci 2:407–422
Renedo M, Bustamante P, Tessier E et al. (2017) Assessment of mercury speciation in feathers using species-specific isotope dilution analysis. Talanta 174:100–110
Renedo M, Bustamante P, Cherel Y et al. (2020) A seabird-eye on mercury stable isotopes and cycling in the Southern Ocean. Sci Total Environ 742:140499
Renedo M, Pedrero Z, Amouroux D et al. (2021) Mercury isotopes of key tissues document mercury metabolic processes in seabirds. Chemosphere 263:127777
Renzoni A, Zino F, Franchi E (1998) Mercury levels along the foodchain and risk for exposed populations. Environ Res 77:68–72
Ricca MA, Keith MA, Anthony RG (2008) Sources of organochlorine contaminants and mercury in seabirds from the Aleutian archipelago of Alaska: Inferences from spatial and trophic variation. Sci Total Environ 406:308–323
Richard S, Arnoux A, Cerdan P et al. (2000) Mercury levels of soils, sediments and fish in French Guiana, South America. Water Air Soil Pollut 124:221–244
Richter W, Skinner LC (2020) Mercury in the fish of New York’s Great Lakes: A quarter century of near stability. Ecotoxicology 29:1721–1738
Ricketts P, Basu N, Fletcher H et al. (2016) Assessment of fish consumption and mercury exposure among pregnant women in Jamaica and Trinidad & Tobago. Chemosphere 164:462–468
Riesgo L, Sanpera C, García-Barcelona S et al. (2023) Understanding the role of ecological factors affecting mercury concentrations in the blue shark (Prionace glauca). Chemosphere 313:37642
Rigét F, Muir D, Kwan M et al. (2005) Circumpolar pattern of mercury and cadmium in ringed seals. Sci Total Environ 351:312–322
Rigét F, Dietz R, Born EW (2007) Temporal trends of mercury in marine biota of west and northwest Greenland. Mar Pollut Bull 54:72–80
Rigét F, Braune B, Bignert A et al. (2011) Temporal trends of Hg in Arctic biota an update. Sci Total Environ 409:3520–3526
Rimmer CC, McFarland KP, Evers DC et al. (2005) Mercury concentrations in Bicknell’s thrush and other insectivorous passerines in montane forests of northeastern North America. Ecotoxicology 14:223–240
Ritonga IR, Bureekul S, Luadnakrob P, Sompongchaiyakul P (2023) Status Level of Total Mercury (T-Hg) in Barracuda (Sphyraena putnamae) from the Gulf of Thailand. Trends Sci 20:5353–5353
Robinson SA, Lajeunesse MJ, Forbes MR (2012) Sex differences in mercury contamination of birds: testing multiple hypotheses with meta-analysis. Environ Sci Technol 46:7094–7101
Rocque DA, Winker K (2004) Biomonitoring of contaminants in birds from two trophic levels in the North Pacific. Environ Toxicol Chem 23:759–766
Rodrigues T, Amorim AF (2016) Review and analysis of mercury levels in blue marlin (Makaira nigricans Lacepède 1802) and swordfish (Xiphias gladius Linnaeus 1758). bioRxiv https://www.biorxiv.org/content/10.1101/043893v1
Rodriguez CAB, de Lacerda LD, Bezerra MF (2022) Pan-oceanic distribution of mercury (Hg) in sea turtles: a review. Endangered Species Res 49:175–185
Rodriguez CAB, Bezerra MF, Rezende CED et al. (2019) Mercury and methylmercury in carapace of the marine turtle Caretta caretta in northeastern Brazil and its potential for Environ Monit. Anais da Acad Brasileira de Ciências 91:2
Rodriguez CAB, de Lacerda LD, Bezerra MF (2022) Pan-oceanic distribution of mercury (Hg) in sea turtles: a review. Endang Species Res 49:175–185
Rodríguez-Gutiérrez J, Galván-Magaña F, Jacobo-Estrada T et al. (2020) Mercury–selenium concentrations in silky sharks (Carcharhinus falciformis) and their toxicological concerns in the southern Mexican Pacific. Mar Pollut Bull 153:111011
Rosendal GK, Andresen S, Heggelund GM, Steindal EH (2020) The Minamata Convention and Mercury Policy in China: The Role of Science. Asian Perspect 44:435–460
Ross DA, Guzmán HM, Van Hinsberg VJ, Potvin C (2016) Metal contents of marine turtle eggs (Chelonia mydas, Lepidochelys olivacea) from the tropical eastern pacific and the implications for human health. J Environ Sci Health B 51(10):675–687
Routti H, Letcher RJ, Born EW et al. (2011) Spatial and temporal trends of selected trace elements in liver tissue from polar bears (Ursus maritimus) from Alaska Canada and Greenland. J Environ Monitor 13:2260–2267
Routti H, Letcher RJ, Born EW et al. (2012) Influence of carbon and lipid sources on variation of mercury and other trace elements in polar bears (Ursus maritimus). Environ Toxicol Chem 31(12):2739–2747
Rudd JW, Bodaly RA, Fisher NS et al. (2018) Fifty years after its discharge methylation of legacy mercury trapped in the Penobscot Estuary sustains high mercury in biota. Sci Total Environ 642:1340–1352
Rudershausen PJ, Cross FA, Runde BJ et al. (2023) Total mercury methylmercury and selenium concentrations in blue marlin Makaira nigricans from a long-term dataset in the western north Atlantic. Sci Total Environ 858:159947
Rumbold DG, Niemczyk SL, Fink LE et al. (2001) Mercury in eggs and feathers of great egrets (Ardea albus) from the Florida Everglades. Arch Environ Contam Toxicol 41:501–507
Rumbold DG, Miller KE, Dellinger TA, Haas N (2017) Mercury Concentrations in Feathers of Adult and Nestling Osprey (Pandion haliaetus) from Coastal and Freshwater Environments of Florida. Arch Environ Contam Toxicol 72:31–38
Rumbold DG, Lienhardt CT, Parsons ML (2018) Mercury biomagnification through a coral reef ecosystem. Arch Environ Contam Toxicol 75:121–133
Rutkiewicz J, Nam DH, Cooley T et al. (2011) Mercury exposure and neurochemical impacts in bald eagles across several Great Lakes states. Ecotoxicology 20:1669–1676
Ruus A, Øverjordet IB, Braaten HFV et al. (2015) Methylmercury biomagnification in an Arctic pelagic food web. Environ Toxicol Chem 34:2636–2643
Ryazanov SD, Fomin SV, Kalinchuk VV (2023) Mercury content in the fur of sea otters (Enhydra lutris) from the Commander Islands. Mar Pollut Bull 188:114638
Sabino MA, Bodin N, Govinden R et al. (2022) The role of tropical small-scale fisheries in trace element delivery for a Small Island Developing State community the Seychelles. Mar Pollut Bull 181:113870
Sakai H, Saeki K, Ichihashi H et al. (2000) Species-Specific Distribution of Heavy Metals in Tissues and Organs of Loggerhead Turtle (Caretta caretta) and Green Turtle (Chelonia mydas) from Japanese Coa stal Waters. Mar Pollut Bull 40:701–709. https://doi.org/10.1016/S0025-326X(00)00008-4
Salafsky N, Boshoven J, Burivalova Z et al. (2019) Defining and using evidence in conservation practice. Conserv Sci Pract 1(5):e27
Salazar-Camacho C, Manuel Salas-Moreno S, Marrugo-Madrid J et al. (2017) Dietary human exposure to mercury in two artisanal small-scale gold mining communities of northwestern Colombia. Environ Int 107:47–54
Salazar-Camacho C, Salas-Moreno M, Paternina-Uribe R et al. (2020) Dataset of concentrations of mercury and methylmercury in fish from a tropical river impacted by gold mining in the Colombian Pacific. Data Brief 33:106513
Sánchez-Fortún M, Ouled-Cheikh J, Jover C et al. (2020) Following up mercury pollution in the Ebro Delta (NE Spain): Audouin’s gull fledglings as model organisms to elucidate anthropogenic impacts on the environment. Environ Pollut 266:115232
Sánchez-Muros MJ, Morote E, Gil C et al. (2018) Mercury contents in relation to biometrics and proximal composition and nutritional levels of fish eaten from the Western Mediterranean Sea (Almería bay). Mar Pollut Bull 135:783–789
Sandheinrich MB, Bhavsar SP, Bodaly RA et al. (2011) Ecological risk of methylmercury to piscivorous fish of the Great Lakes region. Ecotoxicology 20:1577–1587
Santschi PH, Yeager KM, Schwehr KA, Schindler KJ (2017) Estimates of recovery of the Penobscot River and estuarine system from mercury contamination in the 1960s. Sci Total Environ 596:351–359
Sardenne F, Bodin N, Médieu A et al. (2020) Benefit-risk associated with the consumption of fish bycatch from tropical tuna fisheries. Environ Pollut 267:115614
Savoy LP, Flint D, Zwiefelhofer H et al. (2017) Geographic and temporal patterns of variation in total mercury concentrations in blood of harlequin ducks and blue mussels from Alaska. Mar Pollut Bull 117:178–183
Sayers CJ, Roeder MR, Forrette LM et al. (2021) Geographic variation of mercury in breeding tidal marsh sparrows of the northeastern United States. Ecotoxicol 30:1929–1940
Sayers C, Evers DC, Ruiz-Gutierrez V et al. (2023) Mercury in Neotropical birds: a synthesis and prospectus on 15 years of exposure data. Ecotoxicol 32:1–28
Schartup AT, Balcom PH, Soerensen AL et al. (2015a) Freshwater discharges drive high levels of methylmercury in Arctic marine biota. Proc Natl Acad Scie USA 112:11789–11794
Schartup AT, Ndu U, Balcom PH et al. (2015b) Contrasting effects of marine and terrestrially derived dissolved organic matter on mercury speciation and bioavailability in seawater. Environ Sci Technol 49:5965–5972
Schartup AT, Thackray CP, Qureshi A et al. (2019) Climate change and overfishing increase neurotoxicant in marine predators. Nature 572:648–650
Scheuhammer AM, Wong AH, Bond D (1998) Mercury and selenium accumulation in common loons (Gavia immer) and common mergansers (Mergus merganser) from eastern Canada. Environ Toxicol Chem 17:197–201
Scheuhammer AM, Basu N, Evers DC, et al. (2011) Ecotoxicologyogy of mercury in fish and wildlife: Recent advances. In: Bank M (ed) Mercury in the Environment: Pattern and Process. University of California Press, Berkeley CA, p 223–238
Scheuhammer AM, Braune B, Chan HM et al. (2015) Recent progress on our understanding of the biological effects of mercury in fish and wildlife in the Canadian Arctic. Sci Total Environ 509:91–103
Scheuhammer AM, Lord SI, Wayland M et al. (2016) Major correlates of mercury in small fish and common loons (Gavia immer) across four large study areas in Canada. Environ Pollut 210:361–370
Schmutz JA, Trust KA, Matz AC (2009) Red-throated loons (Gavia stellata) breeding in Alaska USA are exposed to PCBs while on their Asian wintering grounds. Environ Pollut 157:2386–2393
Schneider L, Maher W, Green A, Vogt RC (2013) Mercury contamination in reptiles: an emerging problem with consequences for wildlife and human health Mercury: Sources Applications and Health Impacts. Nova Science Publishers Inc, Hauppauge New York USA, p 173-232
Schneider L, Eggins S, Maher W et al. (2015) An evaluation of the use of reptile dermal scutes as a non-invasive method to monitor mercury concentrations in the environment. Chemosphere 119:163–170
Schneider L, Fisher JA, Diéguez MC et al. (2023) A synthesis of mercury research in the Southern Hemisphere, part 1: Natural processes. Ambio 52:897–917
Schoch N, Yang Y, Yanai RD et al. (2020) Spatial patterns and temporal trends in mercury concentrations in common loons (Gavia immer) from 1998 to 2016 in New York’s Adirondack Park: Has this top predator benefitted from mercury emission controls? Ecotoxicology 29:1774–1785
Sebastiano M, Bustamante P, Costantini D et al. (2016) High levels of mercury and low levels of persistent organic pollutants in a tropical seabird in French Guiana the Magnificent frigatebird Fregata magnificens. Environ Pollut 214:384–393
Sebastiano M, Bustamante P, Eulaers I et al. (2017) Trophic ecology drives contaminant concentrations within a tropical seabird community. Environ Pollut 227:183–193
Seco J, Aparício S, Brierley AS et al. (2021) Mercury biomagnification in a Southern Ocean food web. Environ Pollut 275:116620
Sedak M, Bilandžić N, Đokić M et al. (2022) Body burdens and distribution of mercury and selenium in bottlenose, striped and Risso’s dolphins along the Adriatic coast: A 20-year retrospective. Mar Pollut Bull 185:114298
Seewagen CL, Cristol DA, Gerson AR (2016) Mobilization of mercury from lean tissues during simulated migratory fasting in a model songbird. Scient Rep 6:1–5
Seewagen CL (2020) The threat of global mercury pollution to bird migration: potential mechanisms and current evidence. Ecotoxicology 29:1254–1267
Seixas TG, Moreira I, Siciliano S et al. (2014) Differences in methylmercury and inorganic mercury biomagnification in a tropical marine food web. Bull Environ Contam Toxicol 92:274–278
Selin NE (2009) Global Biogeochemical Cycling of Mercury: A Review. Annual Rev Environ Resour 34:43–63
Sharma BM, Bharat GK, Šebková K, Scheringer M (2019) Implementation of the Minamata Convention to manage mercury pollution in India: challenges and opportunities. Environ Sci Europe 31:1–12
Sherman CS, Simpfendorfer CA, Pacoureau N et al. (2023) Half a century of rising extinction risk of coral reef sharks and rays. Nat Comm 14:15
Sherman LS, Blum JD, Keeler GJ et al. (2012) Investigation of local mercury deposition from a coal-fired power plant using mercury isotopes. Environ Sci Technol 46:382–390
Sherman LS, Blum JD (2013) Mercury stable isotopes in sediments and largemouth bass from Florida lakes, USA. Sci Total Environ 448:163–175
Shipley ON, Lee CS, Fisher NS et al. (2021) Metal concentrations in coastal sharks from The Bahamas with a focus on the Caribbean Reef shark. Scient Rep 11:218
Shrestha KP, Noguera R, Chopite J, Sosa P (1988) Mercury content of some marine fish from the southern Caribbean Sea. Sci Total Environ 73:181–187
Signa G, Mazzola A, Tramati CD, Vizzini S (2017) Diet and habitat use influence Hg and Cd transfer to fish and consequent biomagnification in a highly contaminated area: Augusta Bay (Mediterranean Sea). Environ Pollut 230:394–404
Sigmund G, Ågerstrand M, Brodin T, Diamond ML, Erdelen WR, Evers DC, Lai A, Rillig MC, Schäffer A, Soehl A, Torres JPM (2022) Broaden chemicals scope in biodiversity targets. Science 376:1280
Sigmund G, Ågerstrand M, Antonelli A, Backhaus T, Brodin T, Diamond ML, Erdelen WR, Evers DC, Hofmann T, Hueffer T, Lai A (2023) Addressing chemical pollution in biodiversity research. Global Change Biol 29:3240–3255
Sinkus W, White B, Reed L, Shervette V (2021) Mercury accumulation in reef fishes: a comparison among red grouper, scamp, and gag of the Atlantic southeastern US and evaluation of “grouper” consumption guidelines. Environ Monitor Assess 193:574
Smart WA, Collier N, Rolland V (2020) A survey of Grenadians on seabird harvest in the Grenada Grenadines. J Caribbean Ornithol 33:67–77
Sonke JE, Angot H, Zhang Y et al. (2023) Global change effects on biogeochemical mercury cycling. Ambio 52:853–876
Sorensen JA, Glass GE, Schmidt KW et al. (1990) Airborne mercury deposition and watershed characteristics in relation to mercury concentrations in water sediments plankton and fish of eighty northern Minnesota lakes. Environ Sci Technol 24:1716–1727
Soldatini C, Sebastiano M, Albores-Barajas YV et al. (2020) Mercury exposure in relation to foraging ecology and its impact on the oxidative status of an endangered seabird. Sci Total Environ 724:138131
Soresini G, da Silva FA, Leuchtenberger C, Mourão G (2021) Total mercury concentration in the fur of free-ranging giant otters in a large Neotropical floodplain. Environ Res 198:110483
Souza-Araujo J, Andrades R, Hauser-Davis RA et al. (2022) Before the dam: a fish-mercury contamination baseline survey at the Xingu river Amazon basin before the Belo Monte dam. Bull Environ Contam Toxicol 108:861–866
Sepúlveda MS, Frederick PC, Spalding MG, Williams Jr GE (1999a) Mercury contamination in free‐ranging great egret nestlings (Ardea albus) from Southern Florida, USA. Environ Toxicol Chem 18:985–992
Sepúlveda MS, Williams Jr GE, Frederick PC, Spalding MG (1999b) Effects of mercury on health and first-year survival of free-ranging great egrets (Ardea albus) from southern Florida. Arch Environ Contam Toxicol 37:369–376
Squadrone S, Benedetto A, Brizio P et al. (2015) Mercury and selenium in European catfish (Silurus glanis) from Northern Italian Rivers: can molar ratio be a predictive factor for mercury toxicity in a top predator? Chemosphere 119:24–30
Srebocan E, Pompe-Gotal J, Prevendar-Crnic A, Ofner E (2007) Mercury concentrations of captive Atlantic bluefin tuna (Thunnus thynnus) farmed in the Adriatic Sea. Veterinarni Med 52:175–177
Steckling N, Tobollik M, Plass D et al. (2017) Global burden of disease of mercury used in artisanal small-scale gold mining. An Global Health 83:234–247
Stenhouse IJ, Adams EM, Goyette JL et al. (2018) Changes in mercury exposure of marine birds breeding in the Gulf of Maine 2008-2013. Mar Pollut Bull 128:156–161
Stern AH (2005) A revised probabilistic estimate of the maternal methyl mercury intake dose corresponding to a measured cord blood mercury concentration. Environ Health Perspect 113:155–163
Stewart FM, Phillips RA, Bartle JA et al. (1999) Influence of phylogeny diet moult schedule and sex on heavy metal concentrations in New Zealand Procellariiformes. Mar Ecol Progr Ser 1999:295–305
Stickel LF, Wiemeyer SN, Blus LJ (1973) Pesticide residues in eggs of wild birds: adjustment for loss of moisture and lipid. Bull Environ Contam Tox 9:193–196
Storelli A, Barone G, Garofalo R et al. (2022) Determination of mercury methylmercury and selenium concentrations in elasmobranch meat: Fish consumption safety. Int J Environ Res Public Health 19:788
Storelli MM, Marcotrigiano GO (2001) Total mercury levels in muscle tissue of swordfish (Xiphias gladius) and bluefin tuna (Thunnus thynnus) from the Mediterranean Sea (Italy). J Food Protection 64:1058–1061
Storelli MM, Giacominelli Stuffler R, Marcotrigiano GO (2002) Mercury accumulation and speciation in muscle tissue of different species of sharks from Mediterranean Sea Italy. Bull Environ Contam Toxicol 68:201–210
Storelli MM, Storelli A, Giacominelli-Stuffler R, Marcotrigiano GO (2005) Mercury speciation in the muscle of two commercially important fish hake (Merluccius merluccius) and striped mullet (Mullus barbatus) from the Mediterranean Sea: estimated weekly intake. Food Chem 89:295–300
Strandberg U, Palviainen M, Eronen A et al. (2016) Spatial variability of mercury and polyunsaturated fatty acids in the European perch (Perca fluviatilis)-Implications for risk-benefit analyses of fish consumption. Environ Pollut 219:305–314
Streets DG, Zhang Q, Wu Y (2009) Projections of global mercury emissions in 2050. Environ Sci Technol 43:2983–2988
Streets DG, Horowitz HM, Jacob DJ et al. (2017) Total Mercury Released to the Environment by Human Activities. Environ Sci Technol 51:5969–5977
Streets DG, Horowitz HM, Lu Z et al. (2019) Global and regional trends in mercury emissions and concentrations, 2010–2015. Atmos Environ 201:417–427
Suchanek TH, Eagles-Smith CA, Slotton DG et al. (2008) Spatiotemporal trends in fish mercury from a mine-dominated ecosystem: Clear Lake California. Ecol Appl 18:A177–95
Sullivan KM, Kopec AD (2018) Mercury in wintering American black ducks (Anas rubripes) downstream from a point-source on the lower Penobscot River Maine USA. Sci Total Environ 612:1187–1199
Sun J, Bustnes JO, Helander B et al. (2019) Temporal trends of mercury differ across three northern white-tailed eagle (Haliaeetus albicilla) subpopulations. Sci Total Environ 687:77–86
Sunderland EM, Krabbenhoft DP, Moreau JW et al. (2009) Mercury sources distribution and bioavailability in the North Pacific Ocean: Insights from data and models. Global Biogeochem Cycles 23:2
Sunderland EM, Driscoll CT, Hammitt Jr JK et al. (2016) Benefits of regulating hazardous air pollutants from coal and oil-fired utilities in the United States. Environ Sci Technol 50:2117–2120
Sunderland EM, Li M, Bullard K (2018) Decadal Changes in the Edible Supply of Seafood and Methylmercury Exposure in the United States. Environ Health Perspec 126:017006
Sundseth K, Pacyna JM, Banel A et al. (2015) Climate change impacts on environmental and human exposure to mercury in the Arctic. Int J Environ Res Public Health 12:3579–3599
Sundseth K, Pacyna JM, Pacyna EG et al. (2017) Global sources and pathways of mercury in the context of human health. Int J Environ Res Public Health 14:105. https://doi.org/10.3390/ijerph14010105
Symon TE, Murillo-Cisneros DA, Hernández-Camacho CJ et al. (2023) Mercury and selenium concentrations in lanugo of free-ranging California sea lions in the southern Gulf of California, Mexico. Mar Pollut Bull 197:115712
Tan SW, Meiller JC, Mahaffey KR (2009) The endocrine effects of mercury in humans and wildlife. Crit Rev Toxicol 39:228–269
Tapilatu RF, Wona H, Siburian RHS (2020) Data on environmental contaminants in sea turtle eggs at Venu Island Kaimana - West Papua Indonesia. Data Brief 31:105778. https://doi.org/10.1016/j.dib.2020.105778
Tartu S, Goutte A, Bustamante P et al. (2013) To breed or not to breed: endocrine response to mercury contamination by an arctic seabird. Biol Lett 9:20130317
Tavares S, Xavier JC, Phillips RA et al. (2013) Influence of age sex and breeding status on mercury accumulation patterns in the wandering albatross Diomedea exulans. Environ Pollut 181:315–320
Tear TH, Kareiva P, Angermeier P et al. (2005) How much is enough? The recurrent problem of setting measurable objectives in conservation. BioScience 55:835–849
Terrazas-López R, Arreola-Mendoza L, Galván-Magaña F et al. (2019) Understanding the antagonism of Hg and Se in two shark species from Baja California South, México. Sci Total Environ 650:202–209
Titcomb EM, Reif JS, Fair PA et al. (2017) Blood mercury concentrations in common bottlenose dolphins from the Indian River Lagoon Florida: Patterns of social distribution. Mar Mamm Sci 33:771–784
Townsend JM, Rimmer CC, Driscoll CT et al. (2013) Mercury concentrations in tropical resident and migrant songbirds on Hispaniola. Ecotoxicology 22:86–93
Tozer DC, Falconer CM, Badzinski DS (2013) Common Loon reproductive success in Canada: the west is best but not for long. Avian Conservation & Ecology 8(1)
Trasande LJ, DiGangi DC, Evers P et al. (2016) Economic implications of mercury exposure in the context of the global mercury treaty: hair mercury levels and estimated lost economic productivity in selected developing countries. J Environ Manag 183:229–235
Tremlová J, Petrilik J, Teebthaisong A, Ritthichat A (2017) Mercury in fish from industrial sites in Thailand: Report based on chemical analyses of fish and sediment samples collected by Arnika and EARTH in Thailand in 2016-2017, www.earththailand.org/th
Tsao DC, Miles AK, Takekawa JY, Woo I (2009) Potential effects of mercury on threatened California black rails. Arch Environ Contam Toxicol 56:292–301
Tseng C, Ang S, Chen Y et al. (2021) Bluefin tuna reveal global patterns of mercury pollution and bioavailability in the world’s oceans. Proc Natl Acad Sci 118(38):e2111205118
Tsui MTK, Kwon SY, Li ML, Bishop K (2023) Comments on “Decreasing mercury levels in consumer fish over the three decades of increasing mercury emissions in China”. Eco Environ Health 2:1–2
UNEP (2013) Sources emissions releases and environmental transport. United Nations Environment Programme, Chemicals Branch, Geneva, Switzerland, p 42
UNEP (2016) UNEP Global Review of Mercury Monitoring Networks. United Nations Environment Programme, Chemicals Branch, Geneva Switzerland
UNEP (2019a) Global Mercury Assessment 2018. United Nations Environment Programme, Chemicals Branch, Geneva, Switzerland pp 58
UNEP (2019b) Report of the ad hoc technical expert group for effectiveness evaluation: proposed framework for the effectiveness evaluation of the Minamata Convention on Mercury. United Nations Environment Programme, Chemicals Branch, Geneva, Switzerland, UNEP/MC/COP3/14
UNEP (2022) Guidance on monitoring of mercury and mercury compounds to support the evaluation of the effectiveness of the Minamata Convention. United Nations Environment Programme, Chemicals Branch, Geneva, Switzerland, UNEP/MC/COP4/18/
Uryu Y, Malm O, Thornton I et al. (2001) Mercury contamination of fish and its implications for other wildlife of the Tapajós Basin Brazilian Amazon. Conserv Biol 15:438–446
Utete B, Fregene BT (2020) Assessing the spatial and temporal variability and related environmental risks of toxic metals in Lake Asejire south-western Nigeria. Sci African 7:e00259
van Rooyen D, Erasmus JH, Gerber R et al. (2023) Bioaccumulation and trophic transfer of total mercury through the aquatic food webs of an African sub-tropical wetland system. Sci Total Environ 889:164210
Varian-Ramos CW, Swaddle JP, Cristol DA (2014) Mercury reduces avian reproductive success and imposes selection: an experimental study with adult-or lifetime-exposure in zebra finch. PLoS One 9:95674
Vega-Sánchez B, Ortega-García S, Ruelas-Inzunza J et al. (2017) Mercury in the Blue Marlin (Makaira nigricans) from the Southern Gulf of California: Tissue Distribution and Inter-Annual Variation (2005-2012). Bull Environ Contam Toxicol 98:156–161
Vélez N, Bessudo S, Barragán-Barrera DC et al. (2021) Mercury concentrations and trophic relations in sharks of the Pacific Ocean of Colombia. Mar Pollut Bull 173:113109
Verra (2021) Verra: Standards for a Sustainable Future. https://verra.org Accessed 26 March 2021
Villa C, Bell I, Madden Hof C et al. (2019) Elucidating temporal trends in trace element exposure of green turtles (Chelonia mydas) using the toxicokinetic differences of blood and scute samples. Sci Total Environ 651:2450–2459. https://doi.org/10.1016/j.scitotenv.2018.10.092
Višnjevec AM, Kocman D, Horvat M (2014) Human mercury exposure and effects in Europe. Environ Toxicol Chem 33:1259–1270
Vreedzaam A, Ouboter P, Hindori-Mohangoo AD et al. (2023) Contrasting mercury contamination scenarios and site susceptibilities confound fish mercury burdens in Suriname, South America. Environ Pollut 336:122447
Wagemann R, Kozlowska H (2005) Mercury distribution in the skin of beluga (Delphinapterus leucas) and narwhal (Monodon monoceros) from the Canadian Arctic and mercury burdens and excretion by moulting. Sci Total Environ 351:333–343
Wagemann R, Trebacz E, Boila G, Lockhart WL (1998) Methylmercury and total mercury in tissues of arctic marine mammals. Sci Total Environ 218:19–31
Walters C, Couto M, McClurg N et al. (2017) Baseline monitoring of mercury levels in environmental matrices in the Limpopo Province. Water Air Soil Pollut 228:57
Wang X, Wang WX (2019) The three ‘B’ of fish mercury in China: Bioaccumulation biodynamics and biotransformation. Environ Pollut 250:216–232
Wang B, Hu H, Bishop K et al. (2023a) Microbial communities mediating net methylmercury formation along a trophic gradient in a peatland chronosequence. J Hazard Mater 442:130057
Wang F, Outridge PM, Feng X et al. (2019a) How closely do mercury trends in fish and other aquatic wildlife track those in the atmosphere? - Implications for evaluating the effectiveness of the Minamata Convention. Sci Total Environ 674:58–70
Wang HS, Wei-Feng X, Zhuo-Jia C et al. (2013) In vitro estimation of exposure of Hong Kong residents to mercury and methylmercury via consumption of market fishes. J Hazard Mater 248:387–393
Wang Y, Wu P, Zhang Y (2023b) Climate-driven changes of global marine mercury cycles in 2100. Proc Nat Acad Sci 120:e2202488120
Wang Y, Yin D, Xiang Y et al. (2019b) A review of studies on the biogeochemical behaviors of mercury in the Three Gorges Reservoir China. Bull Environ Contam Toxicol 102:686–694
Ward DM, Nislow KH, Chen CY, Folt CL (2010) Rapid, efficient growth reduces mercury concentrations in stream-dwelling Atlantic salmon. Trans Am Fish Soc 139:1–10
Ward DM, Mayes B, Sturup S et al. (2012) Assessing element-specific patterns of bioaccumulation across New England lakes. Sci Total Environ 421-422:230–237
Watanabe T, Matsuda R, Uneyama C (2021) Probabilistic estimation of dietary intake of methylmercury from fish in Japan. Food Saf 9:1–9
Watanuki Y, Yamashita A, Ishizuka M et al. (2016) Feather mercury concentration in streaked shearwaters wintering in separate areas of southeast Asia. Marine Ecol Progr Ser 546:263–269
Wathen JB, Lazorchak JM, Olsen AR, Batt A (2015a) A national statistical survey assessment of mercury concentrations in fillets of fish collected in the US EPA national rivers and streams assessment of the continental USA. Chemosphere 122:52–61
Wathen JB, Lazorchak JM, Olsen AR, Batt A (2015b) A national statistical survey assessment of mercury concentrations in fillets of fish collected in the US EPA national rivers and streams assessment of the continental USA. Chemo 122:52–61
Wathne JA, Haug T, Lydersen C (2000) Prey preference and niche overlap of ringed seals (Phoca hispida) and harp seals (Phoca groenlandica) in the Barents Sea. Marine Ecol Progr Ser 194:233–239
Watson LC, Hurtado-Gonzales JL, Chin CJ, Persaud J (2020) Survey of methylmercury exposures and risk factors among indigenous communities in Guyana, South America. J Health Pollut 10(26):200604
Webb J, Mainville N, Mergler D et al. (2004) Mercury in fish-eating communities of the Andean Amazon, Napo River valley, Ecuador. EcoHealth 1:SU59–SU71
Webb J, Coomes OT, Mainville N, Mergler D (2015) Mercury contamination in an indicator fish species from Andean Amazonian rivers affected by petroleum extraction. Bull Environ Contam Toxicol 95:279–285
Webber HM, Haines TA (2003) Mercury effects on predator avoidance behavior of a forage fish golden shiner (Notemigonus crysoleucas). Environ Toxicol Chem 22:1556–1561
Weech SA, Scheuhammer AM, Elliott JE (2006) Mercury exposure and reproduction in fish-eating birds breeding in the Pinchi Lake region British Columbia Canada. Environ Toxicol Chem 25:1433–1440
Weihe P, Debes Joensen H (2012) Dietary recommendations regarding pilot whale meat and blubber in the Faroe Islands. Int J Circumpolar Health 71(1):p18594
Weis JS, Khan AA (1990) Effects of mercury on the feeding behavior of the mummichog (Fundulus heteroclitus) from a polluted habitat. Marine Environ Res 30:243–249
Weseloh DC, Moore DJ, Hebert CE et al. (2011) Current concentrations and spatial and temporal trends in mercury in Great Lakes Herring Gull eggs 1974-2009. Ecotoxicology 20:1644–1658
Weslawski MJ, Ryg M, Smith TG, Øritsland NA (1994) Diet of ringed seals (Phoca hispida) in a fjord of West Svalbard. Arctic 47:109-114
White AE, Cristol DA (2014) Plumage coloration in belted kingfishers (Megaceryle alcyon) at a mercury-contaminated river. Waterbirds 37:144–152
Whitney MC, Cristol DA (2017) Impacts of Sublethal Mercury Exposure on Birds: A Detailed Review. Rev Environ Contam Toxicol, https://doi.org/10.1007/398_2017_4
Wiemeyer SN, Bunck CM, Krynitsky AJ (1988) Organochlorine pesticides polychlorinated biphenyls and mercury in osprey eggs-1970-79-and their relationships to shell thinning and productivity. Arch Environ Contam Toxicol 17:767–787
Wiener JG, Sandheinrich MB, Bhavsar SP et al. (2012a) Toxicological significance of mercury in yellow perch in the Laurentian Great Lakes region. Environ Pollut 161:350–357
Wiener JG, Evers DC, Gay DA et al. (2012b) Mercury contamination in the Laurentian Great Lakes region: Introduction and overview. Environ Pollut 161:243–251
Willacker JJ, Eagles-Smith CA, Lutz MA et al. (2016) Reservoirs and water management influence fish mercury concentrations in the western United States and Canada. Sci Total Environ 568:739–748
Willacker JJ, Eagles-Smith CA, Ackerman JT (2017) Mercury bioaccumulation in estuarine fishes: Novel insights from sulfur stable isotopes. Environ Sci Technol 51:2131–2139
Willacker JJ, Eagles-Smith CA, Nelson SJ et al. (2023) The influence of short-term temporal variability on the efficacy of dragonfly larvae as mercury biosentinels. Sci Total Environ 867:161469. https://doi.org/10.1016/j.scitotenv.2023.161469
Windmöller CC, Silva NC, Andrade PHM et al. (2017) Use of a direct mercury analyzer(r) for mercury speciation in different matrices without sample preparation. Anal Methods 9:2159–2167
Worm B, Orofino S, Bradley D et al. (2024) Global shark fishing mortality still rising despite widespread regulatory change. Science 383:225–230
Wosnick N, Chaves AP, Leite RD et al. (2021) Nurse sharks, space rockets and cargo ships: Metals and oxidative stress in a benthic, resident and large-sized mesopredator, Ginglymostoma cirratum. Environ Pollut 288:117784
Wosnick N, Prado AC, Martins M, et al. (2023) Reviewing Cd Hg and Pb. Assessments and Effects in Elasmobranchs in Lead Mercury and Cadmium in the Aquatic Environment. CRC Press, Boca Raton, Florida, USA, p 46–75
Wu P, Bishop K, von Brömssen C et al. (2018) Does forest harvest increase the mercury concentrations in fish? Evidence from Swedish lakes. Sci Total Environ 622:1353–1362
Wu P, Kainz MJ, Bravo AG et al. (2019) The importance of bioconcentration into the pelagic food web base for methylmercury biomagnification: A meta-analysis. Sci Total Environ 646:357–367
Wu P, Yan H, Kainz M et al. (2023) Investigating the diet source influence on freshwater fish mercury bioaccumulation and fatty acids—Experiences from Swedish lakes and Chinese reservoirs. Ecotoxicology 32, https://doi.org/10.1007/s10646-023-02712-0
Wyn B, Kidd KA, Burgess NM, Curry RA (2009) Mercury biomagnification in the food webs of acidic lakes in Kejimkujik National Park and National Historic Site Nova Scotia. Can J Fish Aquat Sci 66:1532–1545
Wyn B, Kidd KA, Burgess NM et al. (2010) Increasing mercury in yellow perch at a hotspot in Atlantic Canada Kejimkujik National Park. Environ Sci Technol 44:9176–9181
Xu Q, Zhao L, Wang Y et al. (2018) Bioaccumulation characteristics of mercury in fish in the Three Gorges Reservoir China. Environ Pollut 243:115–126
Xu Z, Lu Q, Jia D et al. (2024) Significant biomagnification of methylmercury in songbird nestlings through a rice-based food web: Insights from stable mercury isotopes. J Hazard Mater 468:133783
Yan HY, Rustadbakken A, Yao H et al. (2010a) Total mercury in wild fish in Guizhou reservoirs China. J Environ Sci 22:1129–1136
Yang H, Macario-González L, Cohuo S et al. (2023) Mercury Pollution History in Tropical and Subtropical American Lakes: Multiple Impacts and the Possible Relationship with Climate Change. Environ Sci Technol 57:3680–3690
Yates DE, Mayack DT, Munney K et al. (2005) Mercury levels in mink (Mustela vison) and river otter (Lontra canadensis) from northeastern North America. Ecotoxicology 14:263–274
Yates DE, Adams EM, Angelo SE et al. (2014) Mercury in bats from the northeastern United States. Ecotoxicology 23:45–55
Yan HY, Rustadbakken A, Yao H et al. (2010b) Total mercury in wild fish in Guizhou reservoirs China. J Environ Sci 22:1129–1136
Yu RQ, Reinfelder JR, Hines ME, Barkay T (2013) Mercury methylation by the methanogen Methanospirillum hungatei. Appl Environ Microbiol 79:6325–6330
Yurkowski DJ, McCulloch E, Ogloff WR et al. (2023) Mercury accumulation, biomagnification, and relationships to δ13C, δ15N and δ34S of fishes and marine mammals in a coastal Arctic marine food web. Mar Pollut Bull 193:115233
Zamani-Ahmadmahmoodi R, Esmaili-Sari A, Ghasempouri SM, Savabieasfahani M (2009) Mercury levels in selected tissues of three kingfisher species, Ceryle rudis, Alcedo atthis, and Halcyon smyrnensi from Shadegan Marshes of Iran. Ecotoxicology 18:319–324
Zamani-Ahmadmahmoodi R, Esmaili-Sari A, Savabieasfahani M, Bahramifar N (2010) Cattle egret (Bubulcus ibis) and little egret (Egretta garzetta) as monitors of mercury contamination in Shadegan Wetlands of south-western Iran. Environ Mon Assess 166:371–377
Zarn AM, Valle CA, Brasso R et al. (2020) Stable isotope and mercury analyses of the Galápagos Islands seabird community. Marine Ornithol 48:71–80
Zhang H, Wang WX, Lin CJ et al. (2022) Decreasing mercury levels in consumer fish over the three decades of increasing mercury emissions in China. Eco Environ Health 1:46–52
Zhang L, Wong MH (2007) Environmental mercury contamination in China: Sources and impacts. Environ Int 33:108–121
Zhang Q, Pan K, Kang S et al. (2014) Mercury in wild fish from high-altitude aquatic ecosystems in the Tibetan Plateau. Environ Sci Technol 48:5220–5228
Zhang Y, Jacob DJ, Dutkiewicz S et al. (2015) Biogeochemical drivers of the fate of riverine mercury discharged to the global and Arctic oceans. Global Biogeochem Cycles 29:854–864
Zhang Y, Jacob DJ, Horowitz HM (2016) Observed decrease in atmospheric mercury explained by global decline in anthropogenic emissions. Proc Natl Acad Sci 113:526–531
Zhou C, Cohen MD, Crimmins BA et al. (2017) Mercury temporal trends in top predator fish of the Laurentian Great Lakes from 2004 to 2015: are concentrations still decreasing? Environ Sci Technol 51:7386–7394
Zhu L, Yan B, Wang L, Pan X (2012) Mercury concentration in the muscle of seven fish species from Chagan Lake Northeast China. Environ Monit Assess 184:1299–1310
Acknowledgements
We would like to thank the Global Environmental Facility – Scientific and Technical Advisory Panel (GEF-STAP) for providing funding to generate the early parts of the GBMS database. Further support was from the UNEP-Chemicals office in Geneva, Switzerland that included a workshop in Monaco to develop global frameworks for monitoring mercury. Olga Furman provided literature and guidance for the section on fish Hg concentrations in Australia. Further assistance with section reviews from staff at Biodiversity Research Institute, Portland, Maine, United States was from Chris DeSorbo, Dustin Meattey, Lucas Savoy, and Chris Sayers; Kate Taylor and Sarah Dodgin assisted with reference standardization, including the Supplemental Information section. The content and views expressed in this publication are those of the authors and do not necessarily reflect the views or carry the endorsement of the Secretariats of the Basel, Rotterdam, and Stockholm Conventions (BRS), the United Nations Environment Programme (UNEP), the United Nations (UN) or of contributory organizations. Designations employed and the presentation of the materials in this publication do not imply the expression of any opinion whatsoever on the part of BRS, UNEP or the UN, on the geopolitical situations or the legal status of any country, territory, city or area, or their authorities, or concerning the delimitation of their frontiers or boundaries. The use of trade, product, or firm names in the publication is for descriptive purposes and does not imply endorsement by the U.S. Government. Lastly, we thank three anonymous reviewers, the reviewer for the U.S. Geological Survey review, the Associate Editor, and Editor-in-Chief for providing us the opportunity to improve this manuscript via their constructive feedback.
Funding
Funding was from the Global Environmental Facility – Scientific and Technical Advisory Panel (GEF-STAP) and the United Nations Environment Programme for generating the early parts of the mercury database.
Author information
Authors and Affiliations
Contributions
Conceptualization: DCE; formal analysis: DCE, MB; resources: DCE; data curation: DCE, MB; writing—original draft: DCE, ES; writing—review & editing: DCE, PB, TT. All coauthors with specific additional contributions from the following for specific sections: Methods – MB, DCE, Marine fish-tuna – PD, Marine fish-billfish – NB, Marine fish-sharks – NH, Other Marine fish-Mediterranean Sea – MHV, PB, Other Marine fish-Caribbean Sea – LC, Freshwater fish-Africa – DB, Freshwater fish-South America – LF, JLB, GM, CV, Freshwater fish-Asia – WP, AT, Freshwater fish-North America and Europe – SA, KB, CE, HFVB, JC, SW, Seabirds-Human Consumption Assessment – IS, Marine Mammals-Toothed Whales – RD, EK, SW, Sea Turtles – CABR, Seabirds-Ecological Health Assessment – PB, IS, Loons/Divers – DCE, Raptors – DCE, Freshwater Birds – JA, Landbirds – DCE, CS, Marine Mammals-Toothed and Baleen Whales, Pinnipeds, and Polar Bears – RD, SW, Linking existing biota Hg data and biomonitoring programs with objectives of the Minamata Convention – NB, CC, AH, ES, Framework for global mercury monitoring – DCE, ES, TT; visualization: DCE, MB; supervision: DCE, ES, PB; project administration: DCE, MB; funding acquisition: DCE.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Evers, D.C., Ackerman, J.T., Åkerblom, S. et al. Global mercury concentrations in biota: their use as a basis for a global biomonitoring framework. Ecotoxicology 33, 325–396 (2024). https://doi.org/10.1007/s10646-024-02747-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-024-02747-x