Abstract
In recent decades, emerging data have highlighted the critical role of extracellular vesicles (EVs), especially (exosomes) Exos, in the progression and development of several cancer types. These nano-sized vesicles are released by different cell lineages within the cancer niche and maintain a suitable platform for the interchange of various signaling molecules in a paracrine manner. Based on several studies, Exos can transfer oncogenic factors to other cells, and alter the activity of immune cells, and tumor microenvironment, leading to the expansion of tumor cells and metastasis to the remote sites. It has been indicated that the cell-to-cell crosstalk is so complicated and a wide array of factors are involved in this process. How and by which mechanisms Exos can regulate the behavior of tumor cells and non-cancer cells is at the center of debate. Here, we scrutinize the molecular mechanisms involved in the oncogenic behavior of Exos released by different cell lineages of tumor parenchyma. Besides, tumoricidal properties of Exos from various stem cell (SC) types are discussed in detail.
Similar content being viewed by others
Introduction
Cancer diseases have been debilitating conditions in human medicine in the last decades with high-rate morbidity and mortality [1]. In clinical settings, surgical approaches, chemotherapy, radiation, and neo-adjuvant therapies are still effective strategies for early cancer treatment [2]. Despite recent advances in cancer theranostics, tumor heterogeneity increases the probability of drug resistance, leading to treatment failure and cancer recurrence [3]. During the last decades, the advent and development of stem cell-related technologies have led to prominent progress in the treatment and alleviation of several pathological conditions [4]. Different stem cell types, including embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), and adult stem cells exhibit differentiation capacity to several lineages, making them as valid cell source for restoration of injured cells [5]. Among different stem cell types, adult mesenchymal stem cells (MSCs) have been extensively applied in various diseases with eminent regenerative outcomes. However, data confirmed that small fractions of transplanted MSCs are alive after direct introduction into the injured sites or a very low cell population can be recruited into the targeted sites after systemic administration [6, 7]. In light of these outcomes, it is believed that MSC therapeutic properties are mainly associated with the paracrine capacity and release of diverse signaling molecules such as cytokines, interleukins (ILs), growth factors, etc. via extracellular vesicles (EVs) into the extracellular matrix and biofluids [8]. Besides these facts, the direct injection of stem cells is associated with a short lifespan and survival rate, off-target delivery, infusion toxicity, activation of allogeneic immune cells, and various malignancies [5]. Meanwhile, the isolation and expansion of stem cells are laborious and expensive and the possibility of genetic and epigenetic instabilities, and loss of stemness are the main challenges in the clinical setting [9]. These features increase the application of stem cell secretome as an alternate to whole-cell-based therapies in clinics with at least biosafety concerns [10].
Exos with lipid bilayer membrane and nano-sized dimensions (30–150 nm) have the potential to carry several signaling molecules between the cells in a paracrine manner [11, 12]. Exos can easily be distributed in several biofluids such as blood, urine, saliva, and other biofluids, reflecting the metabolic status of parent cells [13]. A long with these comments, the origin and metabolic status of parent cells can pre-determine exosomal cargo under different conditions such as cancers [14, 15]. Emerging data have revealed the critical role of Exos in the dynamic growth of cancer cells. These magic bullets can orchestrate cell-to-cell crosstalk within the tumor microenvironment (TME) to regulate tumor mass expansion and cancer cell survival. Such functions can control the development of cancer stem cells (CSCs), TME remodeling, angiogenesis, and invasion of remote sites [16]. Unlike oncogenic properties, Exos can also exert tumoricidal effects on cancer cell lineages [17, 18]. These features make the Exos suitable alternates for tumoricidal therapies. Using smart loading techniques and surface modifications, specific therapeutics can be loaded onto the Exos with appropriate on-target effects [19]. To be specific, Exo-drug delivery can reduce side effects and off-target toxicity following direct administration of chemotherapeutics [20]. In this regard, engineered Exos can intelligently deliver the therapeutic cargo to the targeted sites and diminish the possibility of drug resistance issues (Table 1) [21]. The ability to cross several natural barriers such as blood–brain-barrier etc. makes the Exos superior to synthetic nanoparticles in terms of drug delivery purposes [22]. As above-mentioned, Exos can harbor several signaling molecules that are identical to the parent cells. The Exo molecular signature can be used as a platform for early-stage detection of anaplastic changes, progression, and follow-up of the therapeutic protocols (Fig. 1) [23]. For example, CSC-derived Exos exert pro-oncogenic effects on the non-CSC lineages and normal cells. Monitoring these Exos and their contents can give us invaluable data about the dynamic growth of tumor cells within the tumor mass [24]. Here, the tumorigenic and tumoricidal properties of Exos will be discussed in different cancer types focusing on the possible molecular mechanisms. Recent advances in the application of stem cells Exos in cancer therapy were also highlighted as cell-free therapeutic approaches in cancer therapy.
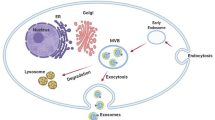
Biogenesis of exosomes (Exos). The endosomal system is actively involved in the generation of Exos. The internalized Exos are sorted into early endosomes. In the next steps, these endosomes mature into late endosomes and MVBs where new nano-sized vesicles, ILVs, are generated via the invagination of the endosomal membrane. These particles are named Exos upon their release into the ECM. MVBs can direct lysosomal degradation to directly fuse with the cell membrane to release their contents into the ECM. Abbreviations: MHC1: major histocompatibility complex 1, CD: Cluster of differentiation, EGFR: Epidermal Growth Factor Receptor, HSP: Heat shock proteins, HIF-1α: Hypoxia-inducible Factor 1α, GAPDH: Glyceraldehyde-3-phosphate dehydrogenase, TSG101: Tumor susceptibility gene 101, ARF-1: ADP-ribosylation factor 1, PI3K: Phosphoinositide 3-kinases, Rab: Ras-associated binding, SNARE: soluble N- ethylmaleimide- sensitive fusion attachment protein receptor, MVB: Multivesicular Body, ESCRT: Endosomal sorting complexes required for transport, STAM: Signaling transducing adaptor molecule, VPS4: Vacuolar protein sorting 4, ALIX: ALG-2-interacting protein X
Exo biogenesis
Exos are produced by the activity of the endosomal system via engaging several signaling molecules [54]. The phenomenon of Exo biogenesis is promoted by the engulfment of recently internalized Exos via endocytosis inside the early endosomes or fusion of trans-Golgi network vesicles with later endosomes or multivesicular bodies (MVBs) (Fig. 1) [55]. The endosomal system is promoted by the maturation of early endosomes into later endosomes and MVBs. Inside the later endosomes and especially MVBs, invagination of the vesicular membrane leads to the formation of numerous intraluminal vesicles (ILVs) [56]. This phenomenon is regulated by the participation of several proteins and factors that help to simultaneous sequestration of signaling molecules into the lumen of ILVs [57]. Molecular investigations have revealed the crucial role of endosomal sorting complex required for transport (ESCRT)-dependent and ESCRT-independent complexes in the formation of ILVs and cargo sorting [58]. The ESCRT complex is composed of four subunits, I, II, and III, with auxiliary factors including vacuolar protein sorting 4 (VPS4), vesicle trafficking 1 (VTA1), and ALG-2-interacting Protein X (ALIX). The close interaction of these factors leads to the sorting of ubiquitinated molecules into the ILVs [59, 60]. The sorting of non-ubiquitinated cargos is mediated via a non-conventional ESCRT-dependent complex which is composed of Syndecan-Syntenin-Alix-ESCRTIII [55, 61]. Besides these factors, tetraspanins (CD63, CD81, and CD9), and sphingomyelinase 2 enzymes (nSMase 2) are involved in the sorting of non-ubiquitinated molecules into the ILVs [62,63,64]. To be specific, tetraspanins are located in the endosomal membrane microdomains with key roles in the invagination of membrane and sorting of special proteins and intracellular factors into MVBs [64, 65]. Neutral sphingomyelinase 2 (nSMase 2)-enriched microdomains via conversion of endosomal membrane sphingomyelin to ceramide, induction of negative curvature of and formation of cone-shaped structure lead lateral separation vesicular membrane and formation of ILVs [66]. Inside the cytosol, the activity of different Ras-associated binding (Rab) GTPase types orchestrates the intracellular transport of endosomes [67]. Depending on the activation of specific Rabs, MVBs can be directed toward lysosomal degradation and release of cargo into the host cells. In alternative pathways, MVBs can be guided toward the Golgi apparatus or fuse with the cell membrane to release the content into extracellular matrix (ECM) [68]. The activation of Rab9 can contribute to endosomal trafficking to the Golgi apparatus while Rab7 increases the lysosomal degradation via inter-endosome-lysosome connection [69]. It should not be forgotten that the activation of similar Rab type in normal or cancer cells yields different outcomes in terms of MVBs destination. For instance, Rab7 activation in cancer cells enhances ILV secretion into the ECM [70]. Other GTPases such as Rab27a and Rab27b promote physical connection, tethering, and fusion of MVBs with cell membranes [71, 72]. Other Rabs such as Rab3, Rab11, and Rab35 are involved in endosomal recycling and ILV cargo secretion [73, 74]. Along with the activation of the Rabs, the soluble NSF Attachment Protein Receptor (SNARE) complex (SYX-5, YKT6, vesicle-associated membrane protein (VAMP)3/7, SNAP23) strengthens the fusion of MVBs with the plasma membrane [55] (Fig. 1). Upon the release of ILVs into ECM, these nanoparticles are hereafter Exos.
Oncogenic and anti-oncogenic properties of Exos
Oncogenic properties of Exos
Exos and TME
Some studies have indicated the transfer of different oncogenic products in the lumen of Exos and their influences on tumorigenesis via engaging several mechanisms [75]. For example, proteins related to the Ras superfamily of GTPases, and mRNAs of H-ras and K-ras, along with several oncomiRNAs were detected in prostate cancer cell Exos [76]. It is also possible that nucleus and mitochondria DNA are sorted into Exos inside the cancer cells in the levels of these elements were higher in cancer Exos than that of normal cells [77]. TME remodeling and stimulation of several anti-tumor activities such as polarization of macrophages toward M2 type are induced in the presence of miRNA-21A bearing cancer cell Exos. In lung cancer cells, this miRNA can directly target the programmed cell death protein 4 and inactivate myeloid-derived suppressor cells (MDSCs) [78]. Exos can play a certain role in TME for cell-to-cell intercommunication via a paracrine manner and regulation of tumor cell metastasis, angiogenesis, and immune cell function [16]. Of note, it should not be forgotten that the production and release of Exos from cancer cells is higher compared to normal cells [79]. Therefore, one can hypothesize that the role of paracrine interaction between the cells is more prominent compared to normal cell counterparts. As a common belief, tumor cell Exos are uptaken by neighboring tumor cells, CSCs, endothelial cells (ECs), and immune cells [80]. The existence of specific cargo inside the Exos can lead to stimulation of certain signaling pathways inside the tumor cells. For example, it was indicated that signaling cascades such as JAK/STAT3, KIT/ERK/BCL2, KIT/ERK/Akt/mTOR, KIT/PI3K/Akt/mTOR, HGF/MET/RAF1/MEK, HGF/MET/PI3K/Akt/mTOR and PDCD1/mTOR are the targeted molecular pathways with different cargo associated with tumorigenesis [81]. Based on molecular investigations, genomics (miRNAs, lncRNAs, etc.) and several factors can initiate the mechanisms associated with carcinogenesis inside the cancer cells (Table 2). Inside the tumor parenchyma, TME with specific physicochemical properties exists for the regulation of cancer cell dynamic growth [82]. TME is composed of heterogeneous cells (tumor cells, stromal cells, ECs, epithelial cells, MSCs, fibroblasts, and immune cells), ECM components, vascular units, and secretory ingredients [82, 83]. The orchestrated and mutual cross-talk between cancer cells with TME can lead to tumor development, expansion, and metastasis [83]. Commensurate with these comments, the organization, and alignment of TME components are critical to tumor cell function [84]. Whether and how Exos can affect the physicochemical properties of TME, non-cancer cells, and cancer cells is at the center of the debate. Emerging data have indicated that Exos can educate cells inside the tumor parenchyma and alter the physicochemical properties of TME. Tumor cell-derived Exos can change the function of TME cells and vice versa. The mutual interaction between the cancer cells and non-cancer stromal cells can dynamically alter the physicochemical properties of TME [79]. Noteworthy, inside the solid tumor parenchyma, the existence of hypoxic conditions increases the local levels of lactic acid, and ECM acidosis [85]. Under such conditions, cancer cells can exhibit rapid proliferation by engaging a mechanism that is so-called metabolic reprogramming [86]. Exos can increase the resistance of vulnerable cancer cells and non-cancer stromal cells to lower pH values via the transfer of mitochondrial particles to restore the production of ATP in these cells [80]. Unlike solid tumors, TME is different in hematologic cancers. Tumor cells can interact with the bone marrow microenvironment and prolonged interaction can lead to the acquisition of a cancerous niche [87]. The role of Exos in the progression of leukemia, invasion, angiogenesis, and inhibition of hematopoiesis has been addressed [88]. Under hypoxic conditions, Exos can foster tumorigenic properties via the regulation of EMT, invasion, survival rate, and maintenance of stemness features. Molecular analyses have confirmed that the density of hypoxia-Inducible factor-1 alpha (HIF-1α) is high in hypoxic cancer cell Exos [82]. Exos can alter the number of TME cells like T lymphocytes, NK cells, T regulatory lymphocytes, dendritic cells (DCs), MSCs, ECs, and MDSCs [89]. In a study conducted by Hou et al., they found that chondrosarcoma cell Exos promote the polarization of macrophages towards M2 type in response to hypoxia, ultimately leading to enhanced metastasis rate [90]. In a similar experiment, it was shown that hypoxic lung cancer Exos with luminal miRNA-21 affects IRF1 and increases M2 type macrophages [91]. It is thought that hypoxic conditions can alter the cargo type, biogenesis and secretion of Exos from cancer cells [92]. The levels of ceramides are increased by the activity of ceramide enzymes in response to hypoxia [93]. Of note, the type of molecules sequestrated into hypoxic ILVs is also changed compared to the normoxic conditions. Along with the expression of HIF-1α, miRNA-210, -21-3p, 125b-5p, 181d-5p levels are increased in released Exos in a HIF-1α-dependent manner [94,95,96]. Interestingly, the size of Exos is reduced under hypoxic conditions because the lack of coordination between the different parts of endosomal system [97]. Taken together, hypoxia is influencing factor in invasion, and metastasis of tumors toward remote site via the release of Exos with specific cargo from host cancer cells.
Fibroblasts and other tumor-associated cells
Cancer-associated fibroblasts (CAFs) are specific fibroblast types within the TME in several tumors [137]. CAFs do not solely originate from activated tumor fibroblasts. Different cells inside the TME such as MSCs, monocytes, adipocytes, smooth muscle cells, pericytes, and CSCs can commit into CAFs [138]. This biological activity is promoted by mechanisms called epithelial-mesenchymal transition (EMT) and endothelial-mesenchymal transition (EndMT) [139]. The process of transformation of normal fibroblasts to CAFs is stimulated via the modulation of several signaling pathways like transforming growth factor beta (TGFβ1)/suppressor of mothers against decapentaplegic (SMAD), stromal-derived factor 1 alpha (SDF-1α)/C-X-C chemokine receptor type 4 (CXCR4), IL-1β/NF-κB, IL-6/JAK/ ROCK/STAT3, Wnt, and HIF-1α. Depending on the malignancy rate and type of cancer, specific signaling pathways can be involved in the production of CAFs from normal fibroblasts [140, 141] (Fig. 2).
Underlying mechanisms associated with the generation of CAFs. These cells can regulate several cancer resistance mechanisms in a paracrine manner via the release of Exos with certain cargoes. Abbreviations: Notch: Neurogenic locus notch homolog protein, TGF-β: Transforming growth factor-β, IL-6: Interleukin 6 WNT: Wingless-related integration site, HIF-1α: Hypoxia-inducible Factor 1α, NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells, Snail: Zinc finger protein SNAI1, Slug: Zinc-Finger Protein Slug, Zeb: Zinc finger E-box-binding homeobox
It is postulated that the activity of non-cancer stromal cells is controlled by the CAFs. CAFs can regulate tumor cell proliferation, resistance to chemotherapeutics, metastasis, and apoptotic changes [140, 142,143,144]. Studies have shown that the activity of factors associated with Exo biogenesis in CAFs is higher as compared to normal fibroblasts [145]. CAF-derived Exos can regulate cancer cell proliferation, vascularization, and blood supply for tumor niches [146]. In ovarian cancers, CAFs produce Exos with low-levels of miR-29c-3p and metastatic behavior [137]. As such, CAFs can control the progression and expansion of colorectal cancer via the alteration of CDKN1A and SNX2 signaling pathways via exosomal miR-345-5p [103, 142]. The existence of miR-345-5p in CAF Exos can down-regulated LIMA1 leading to the activation of the Wnt/β-catenin pathway and hepatic carcinoma cell proliferation [104]. CAFs can also change the metabolism of cancer cells via the production of Exos with specific cargoes. For instance, exosomal lncRNA, namely LINC01614, stimulates the metabolism of glutamine, and thus cancer cell function is dependent on this amino acid [147]. In another work done by Yang and co-workers, CAF exosomal circular RNA, named circEIF3K, increased colorectal cancer progression in a hypoxia-dependent manner via the modulation of miR-214/PDL1 [148]. Like circEIF3K, CAFs can release Exos with other circular RNAs such as circZFR with the potential to alter Stat3/NF-κB molecular pathway and enhance hepatocellular carcinoma cancer growth and resistance to chemotherapy [149].
TME cells
MSCs are TME cellular components with self-renewal and multi-lineage differentiation capacity [150]. Although the immune-modulatory properties of MSCs have been previously addressed [81], MSCs participate in TME remodeling via the production of Exos [150]. For example, MSC Exos can induce angiogenesis, proliferation, apoptosis, metastasis, dormancy, drug resistance, and immune cell suppression via the alteration of certain effectors such as mTOR, AKT, PKC, MAPK, JNK, p53, NFE2L2 and ERK1/2 [150,151,152,153]. Of course, the function and tumorigenic behavior of MSCs within the TME can be regulated in a paracrine manner via cancer cell Exos. In a study conducted by Gyukity-Sebestyén et al., they claimed that melanoma cell Exos up-regulate PD-1 and phenotype acquisition of MSCs, leading to increased cell survival signals and tumor progression [81]. The active and mutual cross-talk between bone marrow MSCs and tumor cells can result in the progression of leukemia [154, 155]. Of course, it should not be forgotten that MSC Exos can also exert tumoricidal effects. How and by which mechanisms the tumorigenic and/or tumoricidal properties of MSC Exos are prominent needs further investigation.
Different mechanisms are involved in immunity against tumor cells along with the activity of natural killer (NK) cells [156]. Tumor-associated antigens are captured by antigen-presenting cells (APCs) like macrophages, T lymphocytes, etc. and further presentation of these antigens to effector immune cells results in tumor cell cytotoxicity [157, 158]. It has been elucidated that TME Exos can reduce the function of immune cells such as NK cells, DCs, and B and T lymphocytes via the regulation of TGF-β TGF I β-6, TNF-α, CTLA4, PD1 [158]. Under these conditions, Exos can increase the polarization of macrophages toward the M2 type [159, 160]. It has been indicated that macrophages have a dual function inside the TME. The M1 macrophages exhibit tumoricidal effects while M2 macrophages can help the tumor cells to proliferate and metastasize [83]. Within the TME, the largest fraction of macrophages is the M2 type while in the early stages of tumor formation, M1 macrophages are dominant and they commit to the M2 type over time [161]. This phenomenon is promoted in part via the production of IL-6-loaded Exos via cancer cells that dictate specific phenotypes for tumor-associated macrophages (TAMs) [162, 163]. Such mechanism has been indicated in pancreatic cancer cell Exos. These Exos harbor FGD5-AS1 and IL-6 with the potential to increase tumor cell metastasis and survival via the promotion of M2 TAMs via the STAT3/NF-κB pathway [162]. M2 TAM Exos with lncMMPA can increase the glucose metabolism within the TME of hepatocellular carcinoma [163]. M2 TAM Exosomal miR-221-3p can increase osteosarcoma cell metastasis via the modulation of the SOCS3/JAK2/STAT3 pathway [105]. In line with the induction of tumor cell metastasis and proliferation, M2 TAM Exos can increase vasculogenesis, known also vasculogenic mimicry (VM), within the tumor parenchyma by increasing vascular density and blood supply. M2 TAM Exos containing miR193a-5p can increase tumor progression in VM-dependent mechanisms via the TIMP2 pathway [106] (Fig. 3).
Cancer cells can use Exos for the regulation of various signaling factors associated with tumor metastatic behavior, chemoresistance, vascularization, and immune escape. Abbreviations: CAFs: Cancer-associated fibroblasts, TGF-β: Transforming growth factor-β, IL-1 β: Interleukin 1 β, FGF: Fibroblast Growth Factor, PDGF: Platelet-derived growth factor, TNF-α: Tumor Necrosis Factor-alpha, PI3K: Phosphoinositide 3-kinases, MAPK: Mitogen-activated protein kinase, RhoA: Ras Homolog Family Member A, Notch: Neurogenic locus notch homolog protein, IL: Interleukin, GM-CSF: Granulocyte–macrophage colony-stimulating factor, CXCL: CXC motif chemokine ligand, MMP: Matrix metallopeptidase, PTEN: Phosphatase and tensin homolog, AKT: Protein kinase B, WNT: Wingless-related integration site, Snail: Zinc finger protein SNAI1, SMAD2: SMAD family member 2, SMAD3: SMAD family member 3, ERK: Extracellular signal-regulated kinase, FAK: Focal adhesion kinase, YAP: Yes-associated protein 1, SDF1: Stromal cell-derived factor 1, HDGF: Hepatoma-derived growth factor, EMT: Epithelial-mesenchymal transition, EndMT: Endothelial-mesenchymal transition, ECM: Extracellular Matrix, MSC: Mesenchymal stem cells
Despite the tumoricidal properties of T lymphocytes, TME Exos can suppress the activity of these cells against tumor cells via the transfer of several signaling molecules such as miRNAs, circular RNAs, lncRNAs, TGF-β, PDL1, and PGE2. TGF-β can inhibit the commitment toward Th1 and Th17 phenotypes. Exosomal miRNA, PDL1, and TGF-β induce the activity of Treg lymphocytes. On the other hand, Exos can stimulate T cell apoptosis and exhaustion via FasL, TRAIL, TIM3, LAG3, and miRNA [13, 120]. Exosomal PGE2, CD39, and CD73 can alter the metabolic state of T cells, and the function of T lymphocytes is inhibited indirectly via PDL1, TGFβ, and PGE2 after suppression of DCs [164]. Hepatocellular carcinoma Exos with circCCAR1 can promote inactivation of CD8+ lymphocytes via the stimulation of PDL1 [120]. Along with these changes, the phosphorylation of hepatocyte growth factor receptor substrate (HRS) can limit the recruitment of CD8+ lymphocytes [165]. The increase of the Th17 subset within the tumor niche is related to tumor mass expansion. The release of Exos containing lncRNA CRNDE-h from colorectal cancer cells promotes the number of Th17 cells and thus cancer mass development [166]. Tumor cell Exo miR-208b and SNHG16 can affect the function of Treg lymphocytes and DCs within the TME. Along with these changes, the number of recruited CD4+ T lymphocytes and local IFN-γ is reduced [83]. These miRNAs can increase the number of CD73 + γδ1 Treg lymphocytes via the modulation of PDCD4 and TGFβ/SMAD pathways [108, 167] (Fig. 3).
Like T lymphocytes, the critical roles of NK cells should not be neglected in different malignancies. These cells and frontline cells promote tumoricidal effects via functional receptors [13]. The physical contact of NK cells with tumor cells leads to whole-cell lysis although the production of various cytokines can affect the anti-tumor activity of MK cells [158, 168]. Like other non-cancer stromal cells, tumor cell Exos can impair the function of NK cells via stimulation/inhibition of specific receptors within the cancerous niche, resulting in anti-tumor activity suppression [13]. In this scenario, hepatocellular carcinoma cells can decrease the local contents of IFN-γ and TNF-α via exosomal circUHRF1 and thus NK cell activity [169]. The stimulation of NK cell TGFβ/SMAD pathway by renal cell carcinoma Exos decreases the anti-tumor sensitivity following NKG2D suppression and induces tumor immune escape [83, 158, 170]. It was suggested that the attachment of certain exosomal factors such as ProNGF and Sortilin to surface receptor p75NTR increases the apoptotic changes in NK cells within the parenchyma of lung tissue cancers [170]. Likewise, colorectal cancer Exos with lncRNA SNHG10 can increase the NK cytotoxicity via up-regulation of INHBC from the TGF-β pathway [109]. The interaction of exosomal miRNA-221-5p and miRNA-186-5p with certain mRNAs (DAP10, and CD96), and perforin genes has been approved in bladder cancers [158]. Along with these comments, the TGF-β signaling pathway is one of the main targets for tumor cell Exos to control the activity of NK cells. In support of this notion, acute lymphocytic leukemia cell Exos can diminish the anti-tumor activity, proliferation, cytotoxicity, and inhibition of cytotoxic granules of NK cells via the TGF-β signaling pathway [168] (Fig. 3).
MDSCs are heterogeneous and immature bone marrow progenitor cells with morphologies similar to neutrophils and monocytes [89, 171, 172]. It is suggested that MDSCs can be committed into M1 and M2 macrophages [173]. The dynamic growth and differentiation of MDSCs in TME are regulated by several cytokines such as G-CSF, M-CSF, SCF, VEGF, and unsaturated fatty acids, IFN-γ, IL-1β, TNF-α, IL-4, -6, -13 by the modulation of NF-κB, STAT1, and STAT6 signaling pathways [174]. The activity of MDSCs can lead to suppression of CD8+ lymphocytes, stimulation of Treg cells, increase of Th17 lymphocytes, orientation of macrophages toward M2 type, and inhibition of B lymphocytes and NK cells [171, 172]. MDSC Exos harbor several factors (S100A8/A9, HSP72, CD47, TSP1, TGF-β, and PGE2), miRNAs (miRNA-21, -9, and -181a) to target certain signaling molecules such as STAT3, RORα, SOCS3 and PIAS3 inside the immune cells [83, 172, 175]. In response to exosomal miRNA-21, and miRNA-29a, MDSCs can promote the growth of tumor cells after the modulation of ROR-A/PTEN and Prkar1α signaling pathways [89]. Noteworthy, CAF exosomal miR-21 and IL-6 can increase the differentiation of MDSCS toward monocyte-macrophage lineage via the modulation of STAT3 [25, 172] (Fig. 3).
Effects of Exos on tumor cell invasion and metastasis
The metastasis is a complex biological phenomenon with sequential steps that help the tumor cells to separate from primary sites and migrate to close and remote sites. Upon reaching the new microenvironments, these cells can proliferate and produce ectopic foci [176]. To increase the possibility of metastasis, the suppression of immune cells and cancer cell resistance are critical features for the development of a pre-metastatic niche. As above-mentioned Treg lymphocytes are the main cell elements in the promotion of tumor cell metastasis to remote sites [177]. Emerging data confirmed the influence of tumor cell Exos in the formation of the pre-metastatic niches. In terms of dynamic trafficking, it should be noted that Exos can be easily distributed inside TME and separate from each other due to net negative charge at their surface [80]. These features mitigate in situ Exo agglutination inside the TME and increase the transfer into remote sites. On the other hand, certain cargo types potentiate Exos to alter the physicochemical behavior of TME, and metastatic behavior of tumor cells via the alteration of targeted signaling pathways, induction of angiogenesis, and immune cell suppression [178]. Some tumor cells are supposed to pass the tissue natural barrier, i.e. blood–brain barrier (BBB), and lung-blood barrier, in addition to blood–tumor barrier (BTB) [179, 180]. Due to specific physicochemical properties, and the existence of certain ligands (integrins) and internalization mechanisms, Exos can, in part, circumvent these obstacles and transfer the cytokines, and growth factors into the TME and remote sites. Exos can change the composition of ECM by the alteration of specific molecular pathways in favor of tumor cell survival and proliferation [180, 181]. Of note, the type and amount of exosomal integrins can pre-determine the on-target tissues and place of metastatic foci [84]. Tumor cell Exos exhibit the prominent capacity to cross the BBB interface. For instance, the transfer of lung cancer cell Exos from BBB increases the apoptotic astrocytes inside the brain parenchyma. Besides, because of specific immunosuppressive agents and inflammatory cytokines, these Exos can prepare the brain microenvironment for the development of metastatic sites [179] (Fig. 3).
Like tumor cells, CAFs actively participate in the formation of pre-metastatic via the release of several chemokines, growth factors, synthesis of certain ECM components, and matrix metalloproteinases (MMPs) [182]. Colorectal cancer cells produce HSPC111 (c-Myc target gene)-enriched Exos that facilitate the development of metastatic foci in other tissues by the change of lipid metabolism [178]. CAF Exos can reduce the activity of the mitochondrial electron transport chain and induce the glycolysis pathway in tumor cells, making these cells resistant to a lack of O2 and nutrients [183]. To increase the metastatic behavior of tumor cells, their migration capacity should be stimulated. Tumor cell Exos with specific cytokines TGF-β, HIF-1α, β-Catenin, and Caveolin-1 can increase the motility of neighboring cells within the TME [184]. Upon reaching the target sites, migrating tumor cells hide and undergo dormancy. Dormant tumor cells educate the resident immune cells to acquire tumor-supporting phenotype to mimic pre-metastatic TME [79]. It was well-established that Exos can affect the dormant tumor cells and their subsequent biological properties [185]. At the primary site, Exos can weaken intercellular communication via the disassociation of adhesion molecules. For example, colorectal tumor cell Exos with luminal ADAM-17 content disassociates E-cadherin in juxtaposed cells and loosens cell-to-cell attachment, leading to enhanced tumor cell migration and the possibility of metastasis to hepatic tissue [176]. The loss of vascular EC-to-EC connection is thought to be another mechanism for the metastasis of tumor cells to remote sites. Exosomal miRNAs such as miR-105 produced by breast tumor cells weaken the tight junction of vascular cells and diminish the integrity of basal membrane, leading to the permeability of blood and lymphatic vessel and increase of metastasis to remote sites [79, 83].
The modulation of EMT and balance between the epithelial and mesenchymal phenotypes is another mechanism in the development of the pre-metastatic niche. By the promotion of EMT, the levels of E-cadherin are reduced while the cellular content of vimentin, N-cadherin, and fibronectin is increased. Besides to induction of cell resistance to apoptotic changes, these features weaken the connection of tumor cells with the underlying basal membrane and increase the possibility of metastasis [186, 187]. CAF and tumor cell Exos with specific cargo types can stimulate the process of EMT via targeting certain effectors Snail, Slug, Zeb1/2, Twist, etc. [186, 188, 189]. Along with EMT, the stimulation of EndMT and differentiation of CSCs into ECs has been indicated by CAF Exos that lead to blood supply into the TME and metastasis [190] (Fig. 4).
Oncogenic properties of Exos. Exos can transfer specific signaling molecules with the potential to increase tumor cell metastasis and the formation of metastatic foci in remote sites. Abbreviations: MMPs: Matrix metallopeptidase, ERK: Extracellular signal-regulated kinase, MAPK: Mitogen-activated protein kinase, PTEN: Phosphatase and tensin homolog, AKT: Protein kinase B, TGF-β: Transforming growth factor-β, IL-1 β: Interleukin 1 β, TSP-1: Thrombospondin-1, Snail: Zinc finger protein SNAI1, Slug: Zinc-Finger Protein Slug, HIF-1α: Hypoxia-inducible Factor 1α, PDL1: Programmed death-ligand 1, FasL: Fas ligand, SMAD:, PI3K: Phosphoinositide 3-kinases, STAT: Signal transducers and activators of transcription, JAK: Janus kinases, NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells, HSP: Heat shock proteins, PGE2: Prostaglandin E2, IL: Interleukin, TNFα: Tumour Necrosis Factor alpha, VEGF: Vascular endothelial growth factor, TSG A10: Testis-specific gene antigen 10, ANGPT2: Angiopoietin-2, TGF-βR3: Transforming growth factor-β receptor 3, S100A4:, TP53INP1: Tumor Protein P53 inducible nuclear Protein 1, mTOR: Mammalian target of rapamycin, SOCS3: Suppressor of cytokine signaling 3, EMT: Epithelial-mesenchymal transition, ECM: Extracellular Matrix, CAF: Cancer Associated fibroblast, DCs: Dendritic cells, Treg: Regulatory T cell, Th1: Type 1 T helper, Th17: Type 17 T helper, CTLs: Cytotoxic T lymphocytes, MDSCs: Myeloid-derived suppressor cells, MSC: Mesenchymal stem cells, NK: Natural killer cells
Exos and tumor angiogenesis
Angiogenesis is the process of de novo blood vessels from parent vessels to support tumor cell survival, growth, and metastasis [85]. It has been shown that Exos can harbor pro-angiogenesis factors and stimulate TME vascularization [85, 110]. Exos can affect specific effectors associated with angiogenesis such as Akt, PTEN, β-Catenin, TSGA10, and ANGPT2 [85]. The uptake of colorectal tumor cell Exos containing B7-H3 molecule by human ECs led to tubulogenic behavior via the activation of Akt/mTOR and the VEGFA molecular pathways [128]. Likewise, lung cancer cell miR-3157-3p-enriched Exos up-regulates VEGF, MMP2, and 9 and Occludin [107]. It is believed that hypoxic tumor cells can produce Exos with angiogenic potential [107]. Prolonged hypoxic conditions increase the accumulation of HIF-1α and angiogenesis via the release of exosomal Wnt4a and activation of β-Catenin [83, 85]. In light of hypoxia, the Exos with higher levels of lncRNA SNHG1 and mir-216b-5p are released form breast cancer cells and the uptake of these Exos promotes angiogenesis in human ECs via Janus kinase 2 (JAK2) [110]. In a similar study, data confirmed that hypoxic pancreatic cancer cells produce Exos with high levels of miR-30b-5p. This factor can stimulate angiogenesis via the inhibition of Gap Junction Protein Alpha 1 (GJA1) [191]. Likewise, thyroid cancer cells can control the angiogenesis in a paracrine manner via the release of Exos enriched in lncRNA FGD5-AS1. This factor targets miR-6838-5p and VAV2 related to actin re-organization and cytoskeletal remodeling [192]. Along with the direct effect of tumor cell Exos and endothelial lineage, the uptake of these nanoparticles by M2-type macrophages can lead to the promotion of angiogenesis. It has been found that tumor cell Exos can recruit neutrophils and increase M2-type polarization of macrophages to support ECs [79] (Fig. 3).
Exos and tumor cell resistance
Chemo-resistance is one of the major challenges that reduce the efficiency of therapeutic protocols [23]. Exos with specific cargoes [P-gp, Survivin, DNMT1, Annexin A3, ATP7A, ATP7B, MRP1, p-STAT3) and miRNAs (miRNA-222-3p, -214, -100-5p, -567, -155-3p, -21, -433, -21-3p, -1246, -223, -365, -19b, -20a, -32-5p, -501, -447-5p, -99a-5p, -125b-5p, -210 & and -155] can increase tumor cell resistance via engaging different mechanisms such as DNA repair, apoptosis inhibition, alteration of drug targets, and efflux, up-regulation of MDR and oncogenes, down-regulating of tumor suppressor genes, EMT induction, autophagy stimulation [23, 193]. The transfer of Exos from resistant cells to sensitive cells is an effective way to treatment failure. In this regard, CAF Exos can educate the neighboring cells to resist chemotherapeutics [194]. Of note, in response to chemotherapy, tumor cells produce Exos containing ANXA6 that induces stemness phenotype in cancer cells via the regulation of ONECUT2. Along with these changes, exosomal levels of miR-378a-3p and miR-378d are increased in breast tumor cells after chemotherapy, resulting in cancer resistance via the EZH2/STAT3 pathway [26]. It seems that the levels of resistance factors are higher in Exos from resistant tumor cells compared to non-resistant counterparts. Tamoxifen-resistant breast tumor cells release Exos with higher luminal miRNA-205 which increases resistance to these drugs in other cells by targeting E2F1 [26]. In a similar work, it was indicated that doxorubicin-resistant neuroblastoma cells with prominent glycolysis activity produce Exos with higher circDLGAP4 contents that induce resistance in sensitive cells by targeting Hexokinase 2 [41].
As mentioned earlier, the reduction of therapeutic agents inside the tumor cells is another anti-tumoricidal property [23]. In this scenario, tumor cells can eliminate internalized chemotherapeutics via the activation of transport pumps. It is suggested that ABC transmembrane transporters (ABCB1, P-gp, MDR1, ABCCs, ABCG2, and MXR) can contribute to the efflux of various drugs from tumor cells [195]. Exos can regulate the expression and activity of cell membrane transporters. For instance, Exos containing P-gp promotes the transfer of drug resistance in recipient tumor cells. MSC Exos with miR-301-3p stimulates multidrug resistance of gastric tumor cells by inhibiting thioredoxin-interacting protein TXNIP [31]. Immune escape, angiogenesis, and the creation of CAFs are other mechanisms associated with tumor cell resistant [23]. As above-mentioned, MDSCs increase the chemo-resistance of tumor cells by different mechanisms, such as inhibition of macrophage polarization towards M1 type, promotion of angiogenesis, interaction with IL6, and increasing the secretion of S100A8/A9 [172]. The transfer of specific factors from CAFs to tumor cells makes them cells resistant to chemotherapeutics. Following gemcitabine treatment, pancreatic ductal adenocarcinoma CAFs can internalize the Exos with ACLS4, followed by induction of gemcitabine resistance in cancer cells via miR-3173-5p [29]. Besides, the transfer of MMP-14 via Exos from resistant pancreatic ductal adenocarcinoma cells to sensitive tumor cells increases their survival [47]. A recent study showed that acute myeloid leukemia cell Exos induce drug resistance by upregulating S100A4 (calcium-binding protein) in other cells [196]. The critical role of exosomal miR-21-5b and S100A6 has been documented in other tumor cell types [30] (Fig. 3).
Anti-oncogenic properties Exos
Besides their oncogenic roles, Exos can exert inhibitory effects on inhibiting tumor cell growth, progression, migration, and invasion via genetic cargo with tumor-suppressing capacities like miRNAs, pro-apoptotic factors, and anti-inflammatory cytokines [197]. In contrast to the resistant tumor cells and CSCs Exos, normal cells and non-resistant cancer cell Exos can expedite the immune system reactivity and anti-tumor properties [198]. The activation of DCs by hepatocellular carcinoma cell Exos increase the number of recruited T lymphocytes in TME with simultaneous elevation of IFN-γ. Under such conditions, leukocytosis and increased cytotoxic T lymphocytes (CTLs) are prominent [199]. Interestingly, brain microvascular ECs Exos with high levels of ECRG4 can suppress the inflammation and angiogenesis inside the glioma tumor parenchyma by inhibiting the P38-MAPK signaling pathway [200]. In an experiment conducted by Wang et al., they showed that exosomal miRNA-363-5p can target the PDGFB pathway and can inhibit breast cancer tumor cell proliferation and migration [201]. It was suggested that some tumor-specific antigens (such as Her2/Neo, Mart1, TRP, and gp100) can be transferred by Exos, leading to the promotion of the immune system against cancer cells [202]. Multiple myeloma Exos IL15/IL15R complex can initiate the proliferation and expansion of NK cells. In activated NK cells, the continuous production of IFN-γ occurs via the stimulation of the TLR2/HSP70/NF-κB pathway. To be specific, tumor cell Exos can frustrate NK cells and increase cytolytic and migration properties in an HSP70-dependent manner [203]. The inhibition of PD-1 in CD8+ lymphocytes was reported after exposure to miR-15a-5p containing hepatocellular carcinoma cell Exos. PD-1L-expressing tumor cells can easily escape from the immune system [204]. In pulmonary cancer, the release of GPC5 (belonging to heparan sulfate proteoglycan) containing Exos contributes to the reduction of angiogenic potential in lymphatic ECs via suppression of PTK2, and endothelial migration. These features are associated with the expression of the CTDSP1 gene and activation of the AhR-PRNT signaling pathway [205].
MSC and immune cell Exos
MSC Exos with various miRNAs and tumor suppressor profiles are suggested biological weapons against several cancer types [151]. In this regard, Xu et al. claimed that bone marrow MSC Exos containing miR-16-5p can inhibit the ITGA2, resulting in the reduction of colorectal cancer cell proliferation, migration, and invasion. Meanwhile, the number of apoptotic tumor cells also increased [206]. In another study, it was indicated that miRNA-let-7c and miRNA-34a containing MSC Exos can effectively reduce the dynamic growth and metastasis of resistant prostate and breast tumor cells, respectively [153, 207]. It seems that several tumorigenic mechanisms can be controlled via MSC Exos in different cell types. Signaling pathways such as LIMK1/Wnt/β-Catenin [208], EMT, TGF-β [209], ZNF367 [17], KLF7/AKT/HIF-1α [210], and Galectin-3 [211] can be modulated via exosomal miRNAs and cargo. These features indicate the anti-tumor activity of MSC Exos with a wide range of functions. Inside the TME, antigen-presenting properties of DCs can be stimulated after exposure to tumor cell Exos. Although DC Exo with notable levels of MHC-I, and -II, CD86, CD80, and HSP can promote T lymphocytes and CD8+ cells [14]. Molecular investigations have revealed that the levels of sphingomyelin and phosphatidyl inositol are high in DC Exos, resulting in enhanced stability and circulation time compared to Exo types [212]. The process of antigen presentation from DCs to immune cells is orchestrated via several mechanisms. Naïve DC Exos may be internalized by T lymphocytes or cross-dressed and coated with DC membrane components before uptake by T lymphocytes. Some authorities have documented the internalization of DC Exos by tumor cells and the addition of tumor-specific antigens with stronger immunological properties [213]. Decoration of DC Exos with specific integrin types αMβ2 and ICAM1 can increase the on-target potential effects [212].
Along with DCs, B, and T lymphocytes (CD4+ and CD8+ subsets) exhibit anti-tumor activities [203]. T cell Exos, especially CD8+ lymphocyte Exos, are potent destructive agents after activation by DCs. The Exos can directly attack tumor cells, eliminate TME MSCs, and activate other T lymphocytes. The inhibition of PDL-1 on the surface of tumor cells is also done via the release of PD-1+ Exos via specific miRNAs such as miR-16p [14, 214]. Likewise, NK cell Exos can exert tumoricidal effects via the stimulation of apoptosis-related factors such as certain Caspases [160, 215]. The existence of HSP70, and granzyme B in NK cell Exos increases the possibility of apoptosis in tumor cells [203]. NK cell and M1 macrophage Exos with specific cargo, miR-30-3p, and miR-16-5p respectively can reduce the proliferation and invasion of esophageal squamous carcinoma cells and gastric tumor cells via the modulation of PD-L1 [216, 217] (Fig. 5).
Different stem cell types produce Exos with tumoricidal properties. Abbreviations: MiR: MicroRNA, WNT: Wingless-related integration site, CTDP: Chemotherapy drugs preloaded, AKT: Protein kinase B, TRIM59: Tripartite motif-containing 59, CXCR: C-X-C Motif Chemokine Receptor, PEG: polyethylene glycol, RHPN2: Rhophilin-2, L-PGDS: Lipocalin-type prostaglandin D synthase. TRIL: TLR4 interactor with leucine-rich repeats, BCL-2: B-cell lymphoma 2, VEGF: Vascular Endothelial Growth Factor, BAX: Bcl-2-associated X protein, MMPs: Matrix metalloproteinases, MAPK: Mitogen-activated protein kinase, PBX3: Pre-B-cell leukemia transcription factor 3, CAFs: Cancer-associated fibroblasts, MSCs: Mesenchymal stem cells, CSCs: Cancer stem cells, EMT: Epithelial-mesenchymal transition
Application of stem cell Exos in cancer therapy
MSC Exos
Exos can be isolated from MSCs of different tissues [218]. Of note, there are controversies in the application of MSCs for cancer therapy purposes. On one hand, MSCs increase TME remodeling and can foster tumor cell dynamic growth, metastasis, and EMT via the suppression of immune system function [219]. On the other hand, various MSC anti-tumor properties have been shown in several in vitro and in vivo conditions [179, 220, 221]. Data confirmed that MSC Exos possess appropriate biocompatibility, healing capacity, and low-rate toxicity, making them valid tools for therapeutic purposes. The tumoricidal properties of MSC Exos are associated with immune system function, regulation of cell-to-cell interaction, induction of apoptotic changes, inhibition of angiogenesis and tumor cell proliferation, and modulation of drug resistance [222,223,224]. The anti-angiogenesis potential of MSC Exos in the context of tumor parenchyma leads to the reduction of VEGF, inhibition of NF-κB [225], and mTOR/HIF1A/VEGF axis [226]. The interaction of MSC Exos with CSCs promotes MET, loss of stemness features, and increase of non-CSC phenotype within the parenchyma, resulting in the reduction of tumor cell resistance [227]. In the presence of MSC Exos, NK cells and CD8+ T lymphocytes proliferate and these changes coincide with the inhibition of Treg cells and polarization of macrophage to M2 phenotype [228]. As above-mentioned MSC Exos increase the chemo-sensitivity of tumor cells by improving anti-drug resistance. It was found that adipose tissue MSC Exos sensitize breast cancer cells to cisplatin [229]. The combination of photobiomodulation with MSC Exos is suggested as an effective therapeutic protocol in cancer patients [229]. In line with several studies, data have confirmed the eligibility of MSC Exos as valid bioshuttles for delivery of anti-tumor factors, increasing drug sensitivity, and targeted delivery purposes [20]. Compared to transplant cells, the trap of administrated Exos is less in hepatic, splenic, and pulmonary tissues which increases the lifespan, circulation time, and affinity to tumor sites [230]. The tumoricidal properties of umbilical cord MSC Exos have been indicated in cancer of renal, endometrial, and breast tissues [231]. Bone marrow MSC Exos with miRNA-16 can suppress the VEGF factor and thus the angiogenesis and vascular density [232]. Besides, the existence of various anti-tumor factors has been indicated inside these Exos [233]. The anti-tumor potential of MSC Exos is closely associated with cargo type, tissue source, and dose and injection interval. In line with the claim, the anti-tumor properties of umbilical cord MSCs is more than bone marrow MSCs and their Exos [150, 234]. Of course, it should not be forgotten that cancer cell type, malignancy degree, and heterogeneity of TME can affect the function of MSC Exos [235, 236]. In general, the effects of different sources of MSC Exos on various cancers remain unclear (Table 3).
CSCs Exos
CSC Exos can be a suitable target for cancer treatment because of their active interaction with TME and control of several mechanisms associated with anaplastic conditions [251]. By sophisticated manipulation, CSC Exos can be used for the disruption of CSCs and non-CSC cancer cells, inhibition of resistance mechanisms, and transmission of stemness features to other cells [252]. The available protocols target certain factors or pathways such as the Notch axis that are eminent in CSCs [253]. Due to distinct physicochemical properties, chemotherapeutics, siRNAs, and immunomodulatory agents can be loaded onto CSC Exos to increase on-target delivery efficiency and reduce off-target side-effects [252, 254]. The conversion of EMT and compelling CSCs to commit to the non-CSC phenotype can lead to tumor cell sensitivity to conventional therapeutic protocols [227]. This approach can be achieved by using certain factors such as all-trans retinoic acid in leukemia cells [255]. The inhibition of paracrine activity, especially Exo biogenesis, in CSCs has been thought of as a promising therapeutic tool [231, 256]. For this purpose, specific endosomal factors such as ESCRT, sphingomyelinase, GTPase proteins, etc. can be regulated to reduce Exo biogenesis and abscission. For example, using sphingomyelinase inhibitor, GW4869, and Rab27a siRNA, Exo biogenesis and release were diminished respectively in CSCs [252, 257]. The exposure of cancer cells to dimethyl amiloride can block the acidification step inside the endosomes [258, 259]. The application of a genetic approach for the suppression or down-regulation of genes responsible for Exo biogenesis, i.e. ESCRT-III protein CHMP4B, is another anti-tumor medication [260]. The advent of nanoparticle technology can help to control Exo biogenesis, formation, and abscission. For instance, gold nanoparticles exhibit anti-Exo activity via the regulation of lipid metabolism [256]. CSC Exos can be manipulated to intensify the immune system response against tumor cells or suppress the immunosuppressive signals. Emerging evidence support the fact CSC Exos are eligible immunogenic tools for developing cancer vaccines to enhance anti-tumor immune-reactivity [261]. In this regard, the isolation of patient CSC Exos enables us to fabricate personalized vaccines for specific tumor types in the clinical setting. To select appropriate therapeutic strategies, a more profound knowledge related to CSC Exo bioactivities and challenges is imperative and warrants further research (Table 4).
Clinical application of SC Exos in cancers
SC Exos possess unique features that make them suitable for therapeutic purposes in cancer treatment.
SC Exos as the natural delivery platform
The unwanted impact of chemotherapy protocols on non-targeted tissues and organs is a challenging issue in cancer patients [278]. To achieve anti-tumor features, it is essential to use elevated doses of drugs despite the possibility of high toxicity for non-target cells [20]. The release of chemotherapeutics using Exos has been thought of as a more efficient approach to circumvent these side effects. SC Exos are valid delivery tools with suitable interaction between the homogenous and heterogeneous cell types [279, 280]. Compared to synthetic nanoparticles such as liposomes, Exos are non-immunogenic with a specific life span [279]. Due to the dynamic distribution of Exos and different uptake systems, these nanoparticles can be used in personalized medicine. These features make possible the load of several therapeutics onto the exosomal lumen and decoration with specific ligands (integrins) to increase on-target delivery and make them cross natural barriers such as BBB [22, 281]. The existence of a lipid bilayer around the therapeutic compounds keeps them away from degradation inside the TME [281]. Besides, these features, the load of chemotherapeutics inside Exos reduces the efficient dose and thus possible side effects [282, 283]. The target molecules can be loaded onto the Exos by using several strategies. In passive cargo loading, the compounds are trapped using a diffusion process like incubation, but the loading efficiency is low [284, 285]. Compared to passive methods, in active cargo loading the compounds are actively injected into the Exo lumen using techniques such as ultrasound and electroporation. These approaches can exert reversible injury to the exosomal membrane. However, the load of the drug, retainability, and stability are high in this method compared to passive drug loading [285, 286]. In an alternative approach, the parent SC is manipulated genetically before Exo isolation or co-cultured with the target molecules, leading to the sequestration of therapeutic compounds onto the Exos in the conditioned medium [284, 285]. Of course, the application of these methods depends on the type of cargo. In the case of drug loading using electroporation or ultrasound approaches, the aggregation of proteins and genetic materials is so high that can increase the possibility of Exo membrane injury and delivery efficiency [284]. Emerging data have indicated a load of small molecules, mRNAs, and proteins with tumoricidal properties onto Exos for therapeutic purposes [202]. Further studies are mandatory to find suitable loading techniques with minimum damage to the Exo structure. The identification of valid anti-tumor cargoes with possible translation capacity to clinical settings is at the center of the debate.
SC Exos for targeted cancer therapy
Recently, scientific society has concentrated on finding novel and sophisticated methods for the direction of SC Exos toward anaplastic sites to yield better therapeutic outcomes [287]. Tumor cells are at the center of targeted therapy by aiming certain factors required for dynamic growth, proliferation, and survival which are not overactive in normal healthy cells [288]. Compared to conventional therapies which target all dividing cells, targeted therapy compounds specifically aim for certain effectors in tumor cells. Using engineering tools, it is possible to develop specific Exo types with higher on-target delivery approaches. For instance, tumor-targeting proteins, peptides, or antibodies can increase the delivery efficiency in tumor cells compared to normal cells [221, 289]. Despite the superiority of Exo-based approaches compared to whole-cell-based therapies, cancer therapy resistance was reported in a study after the application of bone marrow MSC Exos [290]. Under such conditions, TME was remodeled and chemoresistance capacity was induced. However, the modulatory effects of bone marrow MSC Exos on CSCs have been approved by targeting specific intracellular signaling pathways or membrane-bound factors [184, 291].
Several documents have revealed the suitable tumor-homing capacity of MSC Exos [292]. It was suggested that MSC Exos can easily cross the BTB, and respond to gradient density of chemotactic factors [293]. This property can be intensified by the decoration of specific ligands against tumor cell receptors on the Exo surface [294]. MSC exosomal integrin α4β1 can easily interact with VCAM-1 on the tumor cells, leading to the increase of Exo uptake in TME [220]. In general, SC Exos facilitates a promising tumor-targeted therapy by offering more efficient and less harmful outcomes.
SC Exos as diagnostic tools
FDA has approved several Exo-based diagnostic kits for clinical settings [295]. Like several Exo types, CSCs Exos are potential diagnostic tools. As expected, these particles can harbor specific biomolecules associated with stemness, metastasis, tumor initiation, and resistance. The real-time changes in the metabolic profile of parent cells can be precisely monitored using Exos (Fig. 6) [252]. By monitoring specific biomarkers, it is possible to predict and evaluate the efficiency of therapeutic protocols [296]. Regarding the fact that Exos can easily distribute in different biofluids they are valid non-invasive tools for the detection of anaplastic changes with suitable sensitivity and specificity. It should not be neglected that Exos are stable in ECM with heterogeneous compounds. Therefore, serial and consequential sampling enables us for precise and in-time detection of tumorigenesis [136]. Compared to Exo examination, conventional tissue sampling gives information related to a single time point and makes it difficult to make accurate decisions [296]. Despite the promising roles of circulating tumor DNA in accurate clinical detection, these molecules are released into the circulation from cells with apoptotic or necrotic changes [297, 298]. While tumor cell Exos are continuously released into the blood at all phases of tumor cell development and growth with valid data about alive cancer cells [297, 299]. Circulating tumor cells and DNAs at certain numbers and concentrations can be used as prognostic and predictive markers. Any fluctuation in these features can weaken the tumor detection rate.
As described previously, due to a lack of high-quality isolation and purification protocols, and batch-to-batch variation the bulk application of Exos has been limited in cancer patients (Fig. 6) [269]. The lack of exclusive cancer biomarkers and discrimination of cancerous and normal Exos make precise detection challengeable [269, 300]. In line with these descriptions, further investigations are mandatory for the detection of suitable Exo sources for monitoring the dynamic growth of tumor cells, and propagation. The combination of tumor cell Exos with conventional approaches can increase the sensitivity and specificity of diagnostic tools [301]. Even though, CSC Exos can reflect real genetic signatures and are unparalleled biological tools for precise cancer detection and therapy.
Clinical trials and future perspectives
The safety and efficacy of SC Exos have been investigated for the treatment of various cancers in preclinical studies (Table 5). However, there are few clinical trials in this regard. For example, researchers at the MD Anderson Cancer Center (NCT03608631) are conducting a phase 1 study to assess the appropriate dosage and potential adverse effects of MSC Exo with KrasG12D siRNA in patients with pancreatic cancer [302]. By launching another phase 1 clinical trial (NCT04592484), Codiak Biosciences aims to explore the efficacy and safety of exoSTING8, engineered Exos, in treating multiple solid tumors. Data confirmed that manipulating SC Exos through engineering approaches holds promise for future therapeutic applications [303].
The future perspectives of SC Exos in cancer treatment have garnered significant interest in the scientific and medical communities. Exos can successfully combat drug resistance and ameliorate the frequently encountered side effects associated with conventional treatments [304]. Recent advancements in engineered Exo technologies provide exciting opportunities for targeted therapies by modifying surface receptors and loading specific molecules. Despite these features, more investigations are required to overcome the challenges of standardizing isolation techniques and unraveling the intricate mechanisms behind the anti-tumor effects exerted by Exos. To be specific, SC Exos are essential elements in personalized medicine strategies for cancer patients, offering improved effectiveness alongside limited toxicity. Nonetheless, we have just started along this path, and to continue, meticulously planned prospective randomized clinical trials are necessary.
Availability of data and materials
No datasets were generated or analysed during the current study.
Abbreviations
- ACLS4:
-
Acyl-CoA synthetase long chain family member 4
- ADAM-17:
-
A disintegrin and metalloprotease 17
- AKT:
-
Protein kinase B
- ALIX:
-
ALG-2-interacting protein X
- ANGPT2:
-
Angiopoietin-2
- ANXA6:
-
Annexin A6
- APCs:
-
Antigen-presenting cells
- BBB:
-
Blood–brain barrier
- BTB:
-
Blood–tumor barrier
- CAFs:
-
Cancer-associated fibroblasts
- CSCs:
-
Cancer stem cells
- CTDSP1:
-
C-terminal domain small phosphatase 1
- CTLA4:
-
Cytotoxic T-lymphocyte associated protein 4
- CTLs:
-
Cytotoxic T lymphocytes
- CXCR:
-
C-X-C Motif Chemokine Receptor
- DCs:
-
Dendritic cells
- E2F1:
-
E2F transcription factor 1
- ECM:
-
Extracellular Matrix
- ECs:
-
Endothelial cells
- EMT:
-
Epithelial-mesenchymal transition
- EndMT:
-
Endothelial-mesenchymal transition
- ERK:
-
Extracellular signal-regulated kinase
- ESCRT:
-
Endosomal sorting complexes required for transport
- ESCs:
-
Embryonic stem cells
- EVs:
-
Extracellular vesicles
- Exos:
-
Exosomes
- ECM:
-
Extracellular matrix
- EZH2:
-
Enhancer of zeste homolog 2
- FasL:
-
Fas ligand
- G-CSF:
-
Granulocyte colony-stimulating factor
- GJA1:
-
Gap Junction Protein Alpha 1
- gp100:
-
Glycoprotein Gp 100
- Her2/Neo (ERBB2):
-
Erb-B2 receptor tyrosine kinase 2
- HGF:
-
Hepatocyte growth factor
- HIF-1α:
-
Hypoxia-inducible Factor 1α
- HRS:
-
Hepatocyte growth factor receptor substrate
- HSP:
-
Heat shock protein
- IFN-γ:
-
Interferon γ
- ILs:
-
Interleukins
- ILVs:
-
Intraluminal vesicles
- iPSCs:
-
Induced pluripotent stem cells
- JAK:
-
Janus kinases
- JNK:
-
C-Jun N-terminal kinases
- KIT:
-
Receptor tyrosine kinase
- KLF7:
-
Kruppel-like factor 7
- LIMA1:
-
LIM domain and actin-binding protein 1
- LIMK1:
-
LIM domain kinase 1
- MAPK:
-
Mitogen-activated protein kinase
- Mart1:
-
Melanoma-associated antigen recognized by T cells
- M-CSF:
-
Macrophage colony-stimulating factor
- MDR:
-
Multiple drug resistance;
- MDSCs:
-
Myeloid-derived suppressor cells
- MEK:
-
Mitogen-activated protein kinase kinase
- MET (HGF receptor):
-
Hepatocyte growth factor receptor
- miR:
-
MicroRNA
- MMPs:
-
Matrix metalloproteinases
- MSCs:
-
Mesenchymal stem cells
- mTOR:
-
Mammalian target of rapamycin
- MVBs:
-
Multivesicular bodies
- NFE2L2:
-
Nuclear factor erythroid 2-related factor 2 (NRF2)
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- NK:
-
Natural killer cells
- NKG2D:
-
Natural killer group 2D
- nSMase 2:
-
Neural sphingomyelinase 2 enzymes
- PD-1:
-
Programmed death-1
- PDCD1:
-
Programmed cell death protein 1
- PDGFB:
-
Platelet-derived growth factor subunit B
- PGE2:
-
Prostaglandin E2
- PI3K:
-
Phosphoinositide 3-kinases
- PIAS3:
-
E3 SUMO-protein ligase
- Prkar1α:
-
Protein kinase A regulatory subunit Iα
- PTEN:
-
Phosphatase and tensin homolog
- PTK2:
-
Protein tyrosine kinase 2
- Rab:
-
Ras-associated binding
- RAF1:
-
Rapidly Accelerated Fibrosarcoma
- ROCK:
-
Rho-associated protein kinase
- RORα:
-
RAR-related orphan receptor alpha
- S100A8/A9:
-
S100 calcium-binding proteins A8 and A9
- SMAD:
-
Suppressor of mothers against decapentaplegic
- Stem cell:
-
SCs
- SCF:
-
Stem cell factor
- SDF-1α:
-
Stromal-derived factor 1 alpha
- Snail:
-
Zinc finger protein SNAI1
- SNAP23:
-
Synaptosomal-associated protein 23
- SNARE:
-
Soluble N- ethylmaleimide- sensitive fusion attachment protein receptor
- SOCS3:
-
Suppressor of cytokine signaling 3
- STAT:
-
Signal transducers and activators of transcription
- SYX-5:
-
Syntaxin 5
- TAMs:
-
Tissue associated macrophages
- TGF-β:
-
Transforming growth factor-β
- Th1:
-
Type 1 T helper
- Th17:
-
Type 17 T helper
- TIM3:
-
T cell immunoglobulin and mucin domain-containing protein 3
- TIMP2:
-
Tissue inhibitor of metalloproteinases 2
- TLR2:
-
Toll-like receptor 2
- TME:
-
Tumor microenvironment
- TNF-α:
-
Tumor necrosis factor alpha
- TRAIL:
-
TNF-related apoptosis inducing ligand
- Treg :
-
Regulatory T cell
- TRP:
-
Transient receptor potential channel
- TSGA10:
-
Testis-specific gene antigen 10
- TSP1:
-
Thrombospondin 1
- TXNIP:
-
Thioredoxin-interacting protein
- VAMP3/7:
-
Vesicle-associated membrane protein 3
- VEGF:
-
Vascular Endothelial Growth Factor
- VM:
-
Vasculogenic mimicry
- VPS4:
-
Vacuolar protein sorting 4
- VTA1:
-
Vacuolar protein sorting-associated protein
- VAMP:
-
Vesicle-associated membrane protein
- WNT:
-
Wingless-related integration site
- YKT6:
-
N-ethylmaleimide-sensitive factor attachment protein receptor
- Zeb:
-
Zinc finger E-box-binding homeobox
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209–49. https://doi.org/10.3322/caac.21660.
Köpke MM, Aktas B, Ditsch N. Recommendations for the diagnosis and treatment of patients with early breast cancer: update 2023. Curr Opin Obstet Gynecol. 2023;35(1):67–72. https://doi.org/10.1097/gco.0000000000000835.
Fang H, Cavaliere A, Li Z, Huang Y, Marquez-Nostra B. Preclinical advances in theranostics for the different molecular subtypes of breast cancer. Front Pharmacol. 2021;12:627693. https://doi.org/10.3389/fphar.2021.627693.
Wang Y, Ma D, Wu Z, Yang B, Li R, Zhao X, Yang H, Zhang L. Clinical application of mesenchymal stem cells in rheumatic diseases. Stem Cell Res Ther. 2021;12(1):567. https://doi.org/10.1186/s13287-021-02635-9.
Zhang K, Cheng K. Stem cell-derived exosome versus stem cell therapy. Nature Reviews Bioengineering. 2023:1-2.
Fan XL, Zhang Y, Li X, Fu QL. Mechanisms underlying the protective effects of mesenchymal stem cell-based therapy. Cell Mol Life Sci. 2020;77(14):2771–94. https://doi.org/10.1007/s00018-020-03454-6.
Sanchez-Diaz M, Quiñones-Vico MI, Sanabria de la Torre R, Montero-Vílchez T, Sierra-Sánchez A, Molina-Leyva A, Arias-Santiago S.Biodistribution of Mesenchymal Stromal Cells after Administration in Animal Models and Humans: A Systematic Review. J Clin Med. 2021;10(13). https://doi.org/10.3390/jcm10132925.
L PK, Kandoi S, Misra R, S V, K R, Verma RS. The mesenchymal stem cell secretome: A new paradigm towards cell-free therapeutic mode in regenerative medicine. Cytokine Growth Factor Rev. 2019;46:1–9. https://doi.org/10.1016/j.cytogfr.2019.04.002.
Zhou T, Yuan Z, Weng J, Pei D, Du X, He C, Lai P. Challenges and advances in clinical applications of mesenchymal stromal cells. J Hematol Oncol. 2021;14(1):24. https://doi.org/10.1186/s13045-021-01037-x.
Lotfy A, AboQuella NM, Wang H. Mesenchymal stromal/stem cell (MSC)-derived exosomes in clinical trials. Stem Cell Res Ther. 2023;14(1):66. https://doi.org/10.1186/s13287-023-03287-7.
Salimi L, Seyedaghamiri F, Karimipour M, Mobarak H, Mardi N, Taghavi M, Rahbarghazi R. Physiological and pathological consequences of exosomes at the blood-brain-barrier interface. Cell Commun Signal. 2023;21(1):118. https://doi.org/10.1186/s12964-023-01142-z.
Kumar DN, Chaudhuri A, Dehari D, Shekher A, Gupta SC, Majumdar S, Krishnamurthy S, Singh S, Kumar D, Agrawal AK. Combination Therapy Comprising Paclitaxel and 5-Fluorouracil by Using Folic Acid Functionalized Bovine Milk Exosomes Improves the Therapeutic Efficacy against Breast Cancer. Life. 2022;12(8):1143.
Zhou Y, Zhang Y, Gong H, Luo S, Cui Y. The role of exosomes and their applications in cancer. Int J Mol Sci. 2021;22(22):12204.
Kim SB. Function and therapeutic development of exosomes for cancer therapy. Arch Pharmacal Res. 2022;45(5):295–308.
Panigrahi AR, Srinivas L, Panda J. Exosomes: Insights and therapeutic applications in cancer. Translational Oncology. 2022;21: 101439.
Li X, Li X, Zhang B, He B. The Role of Cancer Stem Cell-Derived Exosomes in Cancer Progression. Stem Cells Int. 2022;2022:9133658. https://doi.org/10.1155/2022/9133658.
Du L, Tao X, Shen X. Human umbilical cord mesenchymal stem cell-derived exosomes inhibit migration and invasion of breast cancer cells via miR-21-5p/ZNF367 pathway. Breast Cancer. 2021;28(4):829–37. https://doi.org/10.1007/s12282-021-01218-z.
Yang D, Zhu L, Lin X, Zhu J, Qian Y, Liu W, Chen J, Zhou C, He J. Therapeutic Effects of Synthetic Triblock Amphiphilic Short Antimicrobial Peptides on Human Lung Adenocarcinoma. Pharmaceutics. 2022;14(5). https://doi.org/10.3390/pharmaceutics14050929.
Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977.
Moon B, Chang S. Exosome as a delivery vehicle for cancer therapy. Cells. 2022;11(3):316.
Fathi-Karkan S, Heidarzadeh M, Narmi MT, Mardi N, Amini H, Saghati S, Abrbekoh FN, Saghebasl S, Rahbarghazi R, Khoshfetrat AB. Exosome-loaded microneedle patches: promising factor delivery route. Int J Biol Macromol. 2023;243:125232. https://doi.org/10.1016/j.ijbiomac.2023.125232.
Roy A, As SG, Ganesh PS, Saravanan M, Sunny B. Exosome Mediated Cancer Therapeutic Approach: Present Status and Future Prospectives. Asian Pacific Journal of Cancer Prevention: APJCP. 2023;24(2):363.
Li J, Gao N, Gao Z, Liu W, Pang B, Dong X, Li Y, Fan T. The emerging role of exosomes in cancer chemoresistance. Front Cell Dev Biol. 2021;9:737962.
Rahbarghazi R, Jabbari N, Sani NA, Asghari R, Salimi L, Kalashani SA, Feghhi M, Etemadi T, Akbariazar E, Mahmoudi M, Rezaie J. Tumor-derived extracellular vesicles: reliable tools for Cancer diagnosis and clinical applications. Cell Commun Signal. 2019;17(1):73. https://doi.org/10.1186/s12964-019-0390-y.
Zhao Q, Huang L, Qin G, Qiao Y, Ren F, Shen C, Wang S, Liu S, Lian J, Wang D. Cancer-associated fibroblasts induce monocytic myeloid-derived suppressor cell generation via IL-6/exosomal miR-21-activated STAT3 signaling to promote cisplatin resistance in esophageal squamous cell carcinoma. Cancer Lett. 2021;518:35–48.
Yang Q, Zhao S, Shi Z, Cao L, Liu J, Pan T, Zhou D, Zhang J. Chemotherapy-elicited exosomal miR-378a-3p and miR-378d promote breast cancer stemness and chemoresistance via the activation of EZH2/STAT3 signaling. J Exp Clin Cancer Res. 2021;40(1):1–18.
Zhao Y, Jin L-J, Zhang X-Y. Exosomal miRNA-205 promotes breast cancer chemoresistance and tumorigenesis through E2F1. Aging (Albany NY). 2021;13(14):18498.
Song Z, Jia G, Ma P, Cang S. Exosomal miR-4443 promotes cisplatin resistance in non-small cell lung carcinoma by regulating FSP1 m6A modification-mediated ferroptosis. Life Sci. 2021;276:119399. https://doi.org/10.1016/j.lfs.2021.119399.
Qi R, Bai Y, Li K, Liu N, Xu Y, Dal E, Wang Y, Lin R, Wang H, Liu Z. Cancer-associated fibroblasts suppress ferroptosis and induce gemcitabine resistance in pancreatic cancer cells by secreting exosome-derived ACSL4-targeting miRNAs. Drug Resist Updates. 2023;68:100960.
Luo T, Liu Q, Tan A, Duan L, Jia Y, Nong L, Tang J, Zhou W, Xie W, Lu Y. Mesenchymal stem cell-secreted exosome promotes chemoresistance in breast cancer via enhancing miR-21-5p-mediated S100A6 expression. Molecular Therapy-Oncolytics. 2020;19:283–93.
Zhu T, Hu Z, Wang Z, Ding H, Li R, Wang J, Wang G. microRNA-301b-3p from mesenchymal stem cells-derived extracellular vesicles inhibits TXNIP to promote multidrug resistance of gastric cancer cells. Cell Biol Toxicol. 2022:1–15.
Zhang K, Chen J, Li C, Yuan Y, Fang S, Liu W, Qian Y, Ma J, Chang L, Chen F, Yang Z, Gu W. Exosome-mediated transfer of SNHG7 enhances docetaxel resistance in lung adenocarcinoma. Cancer Lett. 2022;526:142–54. https://doi.org/10.1016/j.canlet.2021.10.029.
Deng X, Ruan H, Zhang X, Xu X, Zhu Y, Peng H, Zhang X, Kong F, Guan M. Long noncoding RNA CCAL transferred from fibroblasts by exosomes promotes chemoresistance of colorectal cancer cells. Int J Cancer. 2020;146(6):1700–16. https://doi.org/10.1002/ijc.32608.
Pan R, Zhou H. Exosomal Transfer of lncRNA H19 Promotes Erlotinib Resistance in Non-Small Cell Lung Cancer via miR-615-3p/ATG7 Axis. Cancer Manag Res. 2020;12:4283–97. https://doi.org/10.2147/cmar.s241095.
Liu C, Lu C, Yixi L, Hong J, Dong F, Ruan S, Hu T, Zhao X. Exosomal Linc00969 induces trastuzumab resistance in breast cancer by increasing HER-2 protein expression and mRNA stability by binding to HUR. Breast Cancer Res. 2023;25(1):124. https://doi.org/10.1186/s13058-023-01720-6.
Chen X, Wang Z, Tong F, Dong X, Wu G, Zhang R. lncRNA UCA1 Promotes Gefitinib Resistance as a ceRNA to Target FOSL2 by Sponging miR-143 in Non-small Cell Lung Cancer. Mol Ther Nucleic Acids. 2020;19:643–53. https://doi.org/10.1016/j.omtn.2019.10.047.
Mao G, Mu Z, Wu DA. Exosomal lncRNA FOXD3-AS1 upregulates ELAVL1 expression and activates PI3K/Akt pathway to enhance lung cancer cell proliferation, invasion, and 5-fluorouracil resistance. 2021;53(11):1484–1494. https://doi.org/10.1093/abbs/gmab129.
Yuan Z, Yang Z, Li W, Wu A, Su Z, Jiang B. Exosome-Mediated Transfer of Long Noncoding RNA HOTAIR Regulates Temozolomide Resistance by miR-519a-3p/RRM1 Axis in Glioblastoma. Cancer Biother Radiopharm. 2020. https://doi.org/10.1089/cbr.2019.3499.
Wang D, Zhou X, Yin J, Zhou Y. Lnc-PICSAR contributes to cisplatin resistance by miR-485-5p/REV3L axis in cutaneous squamous cell carcinoma. Open life sciences. 2020;15(1):488–500. https://doi.org/10.1515/biol-2020-0049.
Geng X, Zhang Y, Lin X, Zeng Z, Hu J, Hao L, Xu J, Wang X, Wang H, Li Q. Exosomal circWDR62 promotes temozolomide resistance and malignant progression through regulation of the miR-370–3p/MGMT axis in glioma. 2022;13(7):596. https://doi.org/10.1038/s41419-022-05056-5.
Tan W-Q, Yuan L, Wu X-Y, He C-G, Zhu S-C, Ye M. Exosome-delivered circular RNA DLGAP4 induces chemoresistance via miR-143-HK2 axis in neuroblastoma. Cancer Biomark. 2022;34(3):375–84.
Xu X, Tao R, Sun L, Ji X. Exosome-transferred hsa_circ_0014235 promotes DDP chemoresistance and deteriorates the development of non-small cell lung cancer by mediating the miR-520a-5p/CDK4 pathway. 2020;20(1):552. https://doi.org/10.1186/s12935-020-01642-9.
Xie H, Yao J, Wang Y, Ni B. Exosome-transmitted circVMP1 facilitates the progression and cisplatin resistance of non-small cell lung cancer by targeting miR-524-5p-METTL3/SOX2 axis. Drug Delivery. 2022;29(1):1257–71. https://doi.org/10.1080/10717544.2022.2057617.
Chen SW, Zhu SQ, Pei X, Qiu BQ, Xiong D, Long X, Lin K, Lu F, Xu JJ, Wu YB. Cancer cell-derived exosomal circUSP7 induces CD8(+) T cell dysfunction and anti-PD1 resistance by regulating the miR-934/SHP2 axis in NSCLC. Mol Cancer. 2021;20(1):144. https://doi.org/10.1186/s12943-021-01448-x.
Chen Z, Xu Z, Wang Q, Wang L, Zhang H, Wang W, Zhao H, Guo Y, Cui J. Exosome-delivered circRNA circSYT15 contributes to cisplatin resistance in cervical cancer cells through the miR-503-5p/RSF1 axis. Cell cycle (Georgetown, Tex). 2023;22(20):2211–28. https://doi.org/10.1080/15384101.2023.2281768.
Yao W, Guo P, Mu Q, Wang Y. Exosome-Derived Circ-PVT1 Contributes to Cisplatin Resistance by Regulating Autophagy, Invasion, and Apoptosis Via miR-30a-5p/YAP1 Axis in Gastric Cancer Cells. Cancer Biother Radiopharm. 2021;36(4):347–59. https://doi.org/10.1089/cbr.2020.3578.
Li X, Li K, Li M, Lin X, Mei Y, Huang X, Yang H. Chemoresistance transmission via exosome-transferred MMP14 in pancreatic cancer. Front Oncol. 2022;12:844648.
Tian T, Han J, Huang J, Li S, Pang H. Hypoxia-Induced Intracellular and Extracellular Heat Shock Protein gp96 Increases Paclitaxel-Resistance and Facilitates Immune Evasion in Breast Cancer. Front Oncol. 2021;11. https://doi.org/10.3389/fonc.2021.784777.
Wu S, Luo M, To KKW, Zhang J, Su C, Zhang H, An S, Wang F, Chen D, Fu L. Intercellular transfer of exosomal wild type EGFR triggers osimertinib resistance in non-small cell lung cancer. 2021;20(1):17. https://doi.org/10.1186/s12943-021-01307-9.
Lin S, Zhu B. Exosome-transmitted FOSL1 from cancer-associated fibroblasts drives colorectal cancer stemness and chemo-resistance through transcriptionally activating ITGB4. 2023. https://doi.org/10.1007/s11010-023-04737-9.
Wei QT, Liu BY, Ji HY, Lan YF, Tang WH, Zhou J, Zhong XY, Lian CL, Huang QZ, Wang CY, Xu YM, Guo HB. Exosome-mediated transfer of MIF confers temozolomide resistance by regulating TIMP3/PI3K/AKT axis in gliomas. Molecular Therapy Oncolytics. 2021;22:114–28. https://doi.org/10.1016/j.omto.2021.08.004.
Hu J, He Q, Tian T, Chang N, Qian L. Transmission of Exosomal TPX2 Promotes Metastasis and Resistance of NSCLC Cells to Docetaxel. 2023;16:197–210. https://doi.org/10.2147/ott.s401454
Wang X, Cheng K, Zhang G, Jia Z, Yu Y, Guo J, Hua Y, Guo F, Li X, Zou W, Sun H, Dong J, Yang Z. Enrichment of CD44 in Exosomes From Breast Cancer Cells Treated With Doxorubicin Promotes Chemoresistance. Front Oncol. 2020;10:960. https://doi.org/10.3389/fonc.2020.00960.
Gao W, Liang T, He R, Ren J, Yao H, Wang K, Zhu L, Xu Y. Exosomes from 3D culture of marrow stem cells enhances endothelial cell proliferation, migration, and angiogenesis via activation of the HMGB1/AKT pathway. Stem Cell Res. 2020;50:102122. https://doi.org/10.1016/j.scr.2020.102122.
Mardi N, Haiaty S, Rahbarghazi R, Mobarak H, Milani M, Zarebkohan A, Nouri M. Exosomal transmission of viruses, a two-edged biological sword. Cell Commun Signal. 2023;21(1):1–29.
Peng X, Li X, Yang S, Huang M, Wei S, Ma Y, Li Y, Wu B, Jin H, Li BJJoE, Research CC. LINC00511 drives invasive behavior in hepatocellular carcinoma by regulating exosome secretion and invadopodia formation. 2021;40 (1):1–17.
Wei H, Chen Q, Lin L, Sha C, Li T, Liu Y, Yin X, Xu Y, Chen L, Gao W. Regulation of exosome production and cargo sorting. Int J Biol Sci. 2021;17(1):163.
Larios J, Mercier V, Roux A, Gruenberg J. ALIX- and ESCRT-III-dependent sorting of tetraspanins to exosomes. J Cell Biol. 2020;219 (3). https://doi.org/10.1083/jcb.201904113.
Ju Y, Bai H, Ren L, Zhang L. The Role of Exosome and the ESCRT Pathway on Enveloped Virus Infection. Int J Mol Sci. 2021;22 (16). https://doi.org/10.3390/ijms22169060.
Lee YJ, Shin KJ, Jang H-J, Ryu J-S, Lee CY, Yoon JH, Seo JK, Park S, Lee S, Je AR. GPR143 controls ESCRT-dependent exosome biogenesis and promotes cancer metastasis. Dev Cell. 2023;58:320–34.
Leblanc R, Kashyap R, Barral K, Egea-Jimenez A, Kovalskyy D, Feracci M, Garcia M, Derviaux C, Betzi S, Ghossoub R. Pharmacological inhibition of syntenin PDZ2 domain impairs breast cancer cell activities and exosome loading with syndecan and EpCAM cargo. J Extracell Vesicles. 2020;10(2):e12039.
Xu J, Sims EKJD (2022) 370-OR: Proinflammatory Stress Activates Neutral Sphingomyelinase 2 Based Generation of Ceramide-Enriched ß-Cell Exosomes. 71 (Supplement_1)
Šála M, Hollinger KR, Thomas AG, Dash RP, Tallon C, Veeravalli V, Lovell L, Kögler M, Hřebabecký H, Procházková E. Novel human neutral sphingomyelinase 2 inhibitors as potential therapeutics for Alzheimer’s disease. J Med Chem. 2020;63(11):6028–56.
Mathieu M, Névo N, Jouve M, Valenzuela JI, Maurin M, Verweij FJ, Palmulli R, Lankar D, Dingli F, Loew D, Rubinstein E, Boncompain G, Perez F, Théry C. Specificities of exosome versus small ectosome secretion revealed by live intracellular tracking of CD63 and CD9. Nat Commun. 2021;12(1):4389–4389. https://doi.org/10.1038/s41467-021-24384-2.
Li Y, Yu S, Li L, Chen J, Quan M, Li Q, Gao Y. KLF4-mediated upregulation of CD9 and CD81 suppresses hepatocellular carcinoma development via JNK signaling. Cell Death Dis. 2020;11(4):299.
Arya SB, Chen S, Jordan-Javed F, Parent CA. Ceramide-rich microdomains facilitate nuclear envelope budding for non-conventional exosome formation. Nat Cell Biol. 2022;24(7):1019–28.
Rinaldi C, Waters CS, Li Z, Kumbier K, Rao L, Nichols RJ, Jacobson MP, Wu LF, Altschuler SJ. Dissecting the effects of GTPase and kinase domain mutations on LRRK2 endosomal localization and activity. Cell Rep. 2023;42(5):112447. https://doi.org/10.1016/j.celrep.2023.112447.
Bala S, Babuta M, Catalano D, Saiju A, Szabo G. Alcohol Promotes Exosome Biogenesis and Release via Modulating Rabs and miR-192 Expression in Human Hepatocytes. Front Cell Dev Biol. 2022;9:787356–787356. https://doi.org/10.3389/fcell.2021.787356.
Yap CC, Digilio L, McMahon LP, Wang T, Winckler B. Dynein Is Required for Rab7-Dependent Endosome Maturation, Retrograde Dendritic Transport, and Degradation. J Neurosci. 2022;42(22):4415–34. https://doi.org/10.1523/JNEUROSCI.2530-21.2022.
Hwang J, Choi D, Han S, Jung SY, Choi J, Hong J. Potential toxicity of polystyrene microplastic particles. Sci Rep. 2020;10(1):7391. https://doi.org/10.1038/s41598-020-64464-9.
Wu J, Zhao Y, Chen Q, Chen Y, Gu J, Mao L. Enterovirus A71 Promotes Exosome Secretion by the Nonstructural Protein 3A Interacting with Rab27a. Microbiol Spectr. 2023;11(2):e0344622–e0344622. https://doi.org/10.1128/spectrum.03446-22.
Yu F, Wu W, Liang M, Huang Y, Chen C. Prognostic Significance of Rab27A and Rab27B Expression in Esophageal Squamous Cell Cancer. Cancer Manag Res. 2020;12:6353–61. https://doi.org/10.2147/cmar.S258940.
Diaz-Rohrer B, Castello-Serrano I, Chan SH, Wang H-Y, Shurer CR, Levental KR, Levental I. Rab3 mediates a pathway for endocytic sorting and plasma membrane recycling of ordered microdomains. Proc Natl Acad Sci U S A. 2023;120(10):e2207461120. https://doi.org/10.1073/pnas.2207461120.
Solinger JA, Rashid HO, Prescianotto-Baschong C, Spang A. FERARI is required for Rab11-dependent endocytic recycling. Nat Cell Biol. 2020;22(2):213–24. https://doi.org/10.1038/s41556-019-0456-5.
Heidarzadeh M, Zarebkohan A, Rahbarghazi R, Sokullu E. Protein corona and exosomes: new challenges and prospects. Cell Commun Signal. 2023;21(1):1–15.
Jakhar R, Crasta K. Exosomes as emerging pro-tumorigenic mediators of the senescence-associated secretory phenotype. Int J Mol Sci. 2019;20(10):2547.
Wortzel I, Dror S, Kenific CM, Lyden D. Exosome-mediated metastasis: communication from a distance. Dev Cell. 2019;49(3):347–60. https://doi.org/10.1016/j.devcel.2019.04.011.
Zhang X, Li F, Tang Y, Ren Q, Xiao B, Wan Y, Jiang S. miR-21a in exosomes from Lewis lung carcinoma cells accelerates tumor growth through targeting PDCD4 to enhance expansion of myeloid-derived suppressor cells. Oncogene. 2020;39(40):6354–69. https://doi.org/10.1038/s41388-020-01406-9.
Liu T, Hooda J, Atkinson JM, Whiteside TL, Oesterreich S, Lee AV. Exosomes in breast cancer – mechanisms of action and clinical potential. Mol Cancer Res. 2021;19(6):935–45. https://doi.org/10.1158/1541-7786.mcr-20-0952.
Thakur A, Johnson A, Jacobs E, Zhang K, Chen J, Wei Z, Lian Q, Chen HJ. Energy sources for exosome communication in a cancer microenvironment. Cancers. 2022;14(7):1698.
Gyukity-Sebestyén E, Harmati M, Dobra G, Németh IB, Mihály J, Zvara Á, Hunyadi-Gulyás É, Katona R, Nagy I, Horváth P, Bálind Á, Szkalisity Á, Kovács M, Pankotai T, Borsos B, Erdélyi M, Szegletes Z, Veréb ZJ, Buzás EI, Kemény L, Bíró T, Buzás K (2019) Melanoma-Derived Exosomes Induce PD-1 Overexpression and Tumor Progression via Mesenchymal Stem Cell Oncogenic Reprogramming. Front Immunol. 10. https://doi.org/10.3389/fimmu.2019.02459.
Meng W, Hao Y, He C, Li L, Zhu G. Exosome-orchestrated hypoxic tumor microenvironment. Mol Cancer. 2019;18(1):57. https://doi.org/10.1186/s12943-019-0982-6.
Qi R, Zhao Y, Guo Q, Mi X, Cheng M, Hou W, Zheng H, Hua B. Exosomes in the lung cancer microenvironment: biological functions and potential use as clinical biomarkers. Cancer Cell Int. 2021;21(1):333. https://doi.org/10.1186/s12935-021-01990-0.
Yilmaz G, Tavsan Z, Cagatay E, Kursunluoglu G, Kayali HA. Exosomes released from cisplatin-resistant ovarian cancer cells modulate the reprogramming of cells in tumor microenvironments toward the cancerous cells. Biomed Pharmacother. 2023;157:113973. https://doi.org/10.1016/j.biopha.2022.113973.
Paskeh MDA, Entezari M, Mirzaei S, Zabolian A, Saleki H, Naghdi MJ, Sabet S, Khoshbakht MA, Hashemi M, Hushmandi K, Sethi G, Zarrabi A, Kumar AP, Tan SC, Papadakis M, Alexiou A, Islam MA, Mostafavi E, Ashrafizadeh M. Emerging role of exosomes in cancer progression and tumor microenvironment remodeling. J Hematol Oncol. 2022;15(1):83. https://doi.org/10.1186/s13045-022-01305-4.
Yuan Y, Li H, Pu W, Chen L, Guo D, Jiang H, He B, Qin S, Wang K, Li N, Feng J, Wen J, Cheng S, Zhang Y, Yang W, Ye D, Lu Z, Huang C, Mei J, Zhang H-F, Gao P, Jiang P, Su S, Sun B, Zhao S-M. Cancer metabolism and tumor microenvironment: fostering each other? Science China Life Sciences. 2022;65(2):236–79. https://doi.org/10.1007/s11427-021-1999-2.
Ghiaur G, Valkenburg KC, Esteb C, Ambinder A, Imus PH, Pienta KJ, Jones RJ. Bone marrow niche chemoprotection of metastatic solid tumors mediated by CYP3A4. Cancer. 2023.
Yamaguchi T, Kawamoto E, Gaowa A, Park EJ, Shimaoka M. Remodeling of bone marrow niches and roles of exosomes in leukemia. Int J Mol Sci. 2021;22(4):1881.
Shao X, Hua S, Feng T, Ocansey DKW, Yin L. Hypoxia-regulated tumor-derived exosomes and tumor progression: a focus on immune evasion. Int J Mol Sci. 2022;23(19):11789.
Hou SM, Lin CY, Fong YC, Tang CH. Hypoxia-regulated exosomes mediate M2 macrophage polarization and promote metastasis in chondrosarcoma. Aging (Albany NY). 2023;15(22):13163–75. https://doi.org/10.18632/aging.205230.
Jin J, Yu G. Hypoxic lung cancer cell-derived exosomal miR-21 mediates macrophage M2 polarization and promotes cancer cell proliferation through targeting IRF1. World J Surg Oncol. 2022;20(1):241. https://doi.org/10.1186/s12957-022-02706-y.
Mortezaee K, Majidpoor J. The impact of hypoxia on extracellular vesicle secretome profile of cancer. Med Oncol. 2023;40(5):128. https://doi.org/10.1007/s12032-023-01995-x.
Novgorodov SA, Gudz TI. Ceramide and mitochondria in ischemia/reperfusion. J Cardiovasc Pharmacol. 2009;53(3):198.
Chen X, Zhou J, Li X, Wang X, Lin Y, Wang X. Exosomes derived from hypoxic epithelial ovarian cancer cells deliver microRNAs to macrophages and elicit a tumor-promoted phenotype. Cancer Lett. 2018;435:80–91.
Huang X, Ding L, Bennewith KL, Tong RT, Welford SM, Ang KK, Story M, Le Q-T, Giaccia AJ. Hypoxia-inducible mir-210 regulates normoxic gene expression involved in tumor initiation. Mol Cell. 2009;35(6):856–67.
Umezu T, Tadokoro H, Azuma K, Yoshizawa S, Ohyashiki K, Ohyashiki JH. Exosomal miR-135b shed from hypoxic multiple myeloma cells enhances angiogenesis by targeting factor-inhibiting HIF-1. Blood J Am Soc Hematol. 2014;124(25):3748–57.
To KKW, Cho WCS. Exosome secretion from hypoxic cancer cells reshapes the tumor microenvironment and mediates drug resistance. Cancer Drug Resistance (Alhambra, Calif). 2022;5(3):577–94. https://doi.org/10.20517/cdr.2022.38.
Qiu S, Xie L, Lu C, Gu C, Xia Y, Lv J, Xuan Z, Fang L, Yang J, Zhang L, Li Z, Wang W, Xu H, Li B, Xu Z. Gastric cancer-derived exosomal miR-519a-3p promotes liver metastasis by inducing intrahepatic M2-like macrophage-mediated angiogenesis. J Exp Clin Cancer Res. 2022;41(1):296. https://doi.org/10.1186/s13046-022-02499-8.
Zhao S, Mi Y, Guan B, Zheng B, Wei P, Gu Y, Zhang Z, Cai S, Xu Y, Li X, He X, Zhong X, Li G, Chen Z, Li D. Correction to: Tumor-derived exosomal miR-934 induces macrophage M2 polarization to promote liver metastasis of colorectal cancer. J Hematol Oncol. 2021;14(1):33. https://doi.org/10.1186/s13045-021-01042-0.
Yang S-S, Ma S, Dou H, Liu F, Zhang S-Y, Jiang C, Xiao M, Huang Y-X. Breast cancer-derived exosomes regulate cell invasion and metastasis in breast cancer via miR-146a to activate cancer associated fibroblasts in tumor microenvironment. Exp Cell Res. 2020;391(2):111983. https://doi.org/10.1016/j.yexcr.2020.111983.
Bai J, Zhang X, Shi D, Xiang Z, Wang S, Yang C, Liu Q, Huang S, Fang Y, Zhang W, Song J, Xiong B Exosomal miR-128–3p Promotes Epithelial-to-Mesenchymal Transition in C olorectal Cancer Cells by Targeting FOXO4 via TGF-β/SMAD and JAK/STAT3 Signaling. Front Cell Dev Biol. 9:568738. https://doi.org/10.3389/fcell.2021.568738
Du C, Duan X, Yao X, Wan J, Cheng Y, Wang Y, Yan Y, Zhang L, Zhu L, Ni C, Wang M, Qin Z. Tumour-derived exosomal miR-3473b promotes lung tumour cell intrapulmonary colonization by activating the nuclear factor-κB of local fibroblasts. J Cell Mol Med. 2020;24(14):7802–13. https://doi.org/10.1111/jcmm.15411.
Shi W, Liu Y, Qiu X, Yang L, Lin G. Cancer-associated fibroblasts-derived exosome-mediated transfer of miR-345-5p promotes the progression of colorectal cancer by targeting CDKN1A. Carcinogenesis. 2023;44(4):317–27. https://doi.org/10.1093/carcin/bgad014.
Qi Y, Wang H, Zhang Q, Liu Z, Wang T, Wu Z, Wu W. CAF-Released Exosomal miR-20a-5p Facilitates HCC Progression via the LIMA1-Mediated β-Catenin Pathway. Cells. 2022;11(23):3857.
Liu W, Long Q, Zhang W, Zeng D, Hu B, Liu S, Chen L. miRNA-221-3p derived from M2-polarized tumor-associated macrophage exosomes aggravates the growth and metastasis of osteosarcoma through SOCS3/JAK2/STAT3 axis. Aging (Albany NY). 2021;13(15):19760.
Liu Q, Zhao E, Geng B, Gao S, Yu H, He X, Li X, Dong G, You B. Tumor-associated macrophage-derived exosomes transmitting miR-193a-5p promote the progression of renal cell carcinoma via TIMP2-dependent vasculogenic mimicry. Cell Death Dis. 2022;13(4):382.
Ma Z, Wei K, Yang F, Guo Z, Pan C, He Y, Wang J, Li Z, Chen L, Chen Y. Tumor-derived exosomal miR-3157-3p promotes angiogenesis, vascular permeability and metastasis by targeting TIMP/KLF2 in non-small cell lung cancer. Cell Death Dis. 2021;12(9):840.
Ni C, Fang Q-Q, Chen W-Z, Jiang J-X, Jiang Z, Ye J, Zhang T, Yang L, Meng F-B, Xia W-J. Breast cancer-derived exosomes transmit lncRNA SNHG16 to induce CD73+ γδ1 Treg cells. Signal Transduct Target Ther. 2020;5(1):41.
Huang Y, Luo Y, Ou W, Wang Y, Dong D, Peng X, Luo Y. Exosomal lncRNA SNHG10 derived from colorectal cancer cells suppresses natural killer cell cytotoxicity by upregulating INHBC. Cancer Cell Int. 2021;21:1–16.
Dai G, Yang Y, Liu S, Liu H. Hypoxic breast cancer cell-derived exosomal SNHG1 promotes breast cancer growth and angiogenesis via regulating miR-216b-5p/JAK2 axis. Cancer Manage Res. 2022:123–133
Ding N, Song X, Yu H, Wang J, Huang L, Zhou Y, He X. Mechanism of Exosomal LncRNA PART1 in Esophageal Cancer Angiogenesis by Targeting miR-302a-3p/CDC25A Axis. Technol Cancer Res Treat. 2023;22:15330338231184328. https://doi.org/10.1177/15330338231184327.
Wu Q, He Y, Liu X, Luo F, Jiang Y, Xiang M, Zhao R. Cancer stem cell-like cells-derived exosomal lncRNA CDKN2B-AS1 promotes biological characteristics in thyroid cancer via miR-122-5p/P4HA1 axis. Regenerative Therapy. 2023;22:19–29.
Wang Y, Jiang R, Zhao H, Li F, Li Y, Zhu M. TTN-AS1 delivered by gastric cancer cell–derived exosome induces gastric cancer progression through in vivo and in vitro studies. Cell Biol Toxicol. 2023;39(2):557–71.
Shen T, Miao S, Zhou Y, Yi X, Xue S, Du B, Tang C, Qu L, Fu D, Jia R. Exosomal AP000439. 2 from clear cell renal cell carcinoma induces M2 macrophage polarization to promote tumor progression through activation of STAT3. Cell Commun Signal. 2022;20(1):152.
Fang X, Xu Y, Li K, Liu P, Zhang H, Jiang Y, Tang J, Li Y. Exosomal lncRNA PCAT1 promotes tumor circulating cell-mediated colorectal cancer liver metastasis by regulating the activity of the miR-329–3p/Netrin-1-CD146 complex. J Immunol Res. 2022;2022.
Kong W, Zhang L, Chen Y, Yu Z, Zhao Z. Cancer cell-derived exosomal LINC00313 induces M2 macrophage differentiation in non-small cell lung cancer. Clin Transl Oncol. 2022;24(12):2395–408.
Hu Y, Zai H, Jiang W, Ou Z, Yao Y, Zhu Q. Hepatoblastoma: Derived exosomal lncrna neat1 induces bmscs differentiation into tumor-supporting myofibroblasts via modulating the mir-132/mmp9 axis. J Oncol. 2022;2022.
Yu Q, Zhang Y, Tian Y, Peng A, Cui X, Ding B, Yang L, Liu Y, Ju Y, Gao C. Exosomal Circ_FMN2 Derived from the Serum of Colorectal Cancer Patients Promotes Cancer Progression by miR-338–3p/MSI1 Axis. Appl Biochem Biotechnol. 2023:1–16.
Shang A, Gu C, Wang W, Wang X, Sun J, Zeng B, Chen C, Chang W, Ping Y, Ji P. Exosomal circPACRGL promotes progression of colorectal cancer via the miR-142-3p/miR-506-3p-TGF-β1 axis. Mol Cancer. 2020;19(1):1–15.
Hu Z, Chen G, Zhao Y, Gao H, Li L, Yin Y, Jiang J, Wang L, Mang Y, Gao Y. Exosome-derived circCCAR1 promotes CD8+ T-cell dysfunction and anti-PD1 resistance in hepatocellular carcinoma. Mol Cancer. 2023;22(1):1–21.
Huang X-Y, Huang Z-L, Huang J, Xu B, Huang X-Y, Xu Y-H, Zhou J, Tang Z-Y. Exosomal circRNA-100338 promotes hepatocellular carcinoma metastasis via enhancing invasiveness and angiogenesis. J Exp Clin Cancer Res. 2020;39:1–16.
Wang X, Dong F, Wang Y, Wei H, Li T, Li J. Exosomal circTGFBR2 promotes hepatocellular carcinoma progression via enhancing ATG5 mediated protective autophagy. 2023.
Jiang Q, Lei Z, Wang Z, Wang Q, Zhang Z, Liu X, Xing B, Li S, Guo X, Liu Y. Tumor-Associated Fibroblast-Derived Exosomal circDennd1b Promotes Pituitary Adenoma Progression by Modulating the miR-145-5p/ONECUT2 Axis and Activating the MAPK Pathway. Cancers. 2023;15(13):3375.
Zhu S, Liao B. Mechanism of hypoxia-induced exosome circ_0051799 regulating the progression of lung adenocarcinoma. Biol Chem. 2023(0).
Yu L, Zhang F, Wang Y. Circ_0005615 Regulates the Progression of Colorectal Cancer Through the miR-873–5p/FOSL2 Signaling Pathway. Biochem Genet. 2023:1–22
Chen X, Ren X, E J, Zhou Y, Bian R. Exosome-transmitted circ IFNGR2 Modulates Ovarian Cancer Metastasis via miR-378/ST5 Axis. Mol Cell Biol. 2023;43(1):22-42.
Liu P, Zu F, Chen H, Yin X, Tan X. Exosomal DNAJB11 promotes the development of pancreatic cancer by modulating the EGFR/MAPK pathway. Cell Mol Biol Lett. 2022;27(1):1–20.
Wu R, Zhang Y, Xu X, You Q, Yu C, Wang W, Mao Y. Exosomal B7-H3 facilitates colorectal cancer angiogenesis and metastasis through AKT1/mTOR/VEGFA pathway. Cellular Signalling. 2023:110737
Zheng X, Liu J, Li X, Tian R, Shang K, Dong X, Cao B. Angiogenesis is promoted by exosomal DPP4 derived from 5-fluorouracil-resistant colon cancer cells. Cancer Lett. 2021;497:190–201.
Jiang K, Dong C, Yin Z, Li R, Mao J, Wang C, Zhang J, Gao Z, Liang R, Wang Q. Exosome-derived ENO1 regulates integrin α6β4 expression and promotes hepatocellular carcinoma growth and metastasis. Cell Death Dis. 2020;11(11):972.
Wang M, Yu W, Cao X, Gu H, Huang J, Wu C, Wang L, Sha X, Shen B, Wang T, Yao Y, Zhu W, Huang F. Exosomal CD44 Transmits Lymph Node Metastatic Capacity Between Gastric Cancer Cells via YAP-CPT1A-Mediated FAO Reprogramming. Front Oncol. 2022;12. https://doi.org/10.3389/fonc.2022.860175.
Zhang W, Sun W, Gu X, Miao C, Feng L, Shen Q, Liu X, Zhang X. GDF-15 in tumor-derived exosomes promotes muscle atrophy via Bcl-2/caspase-3 pathway. Cell Death Discov. 2022;8(1):162.
Gao Q, An K, Gao Z, Wang Y, Ding C, Niu P, Lei F. Rectal cancer-derived exosomes activate the nuclear factor kappa B pathway and lung fibroblasts by delivering integrin beta-1. Korean J Physiol Pharmacol. 2023;27(4):375–81.
Tang Z, Yang Y, Chen W, Liang T. Epigenetic deregulation of MLF1 drives intrahepatic cholangiocarcinoma progression through EGFR/AKT and Wnt/β-catenin signaling. Hepatol Commun. 2023;7(8).
Chen G, Han Y, Liu S, Zhou G, Xu X, Zhang H, Li Z, Wu C, Liu Y, Fang K (2021) MUC13 promotes the development of esophageal cancer by upregulating the expression of O-glycan process-related molecules.
Yu C, Xue B, Li J, Zhang Q. Tumor cell-derived exosome RNF126 affects the immune microenvironment and promotes nasopharyngeal carcinoma progression by regulating PTEN ubiquitination. Apoptosis. 2022;27(7–8):590–605.
Han Q, Tan S, Gong L, Li G, Wu Q, Chen L, Du S, Li W, Liu X, Cai J, Wang Z. Omental cancer-associated fibroblast-derived exosomes with low microRNA-29c-3p promote ovarian cancer peritoneal metastasis. Cancer Sci. 2023;114(5):1929–42. https://doi.org/10.1111/cas.15726.
Lee P, Ho C, Ho H, Chen W, Lin C, Lai Y, Juan Y, Chu W, Lee J, Su S. Tumor microenvironment-based screening repurposes drugs targeting cancer stem cells and cancer-associated fibroblasts. Theranostics. 2021;11:9667–86. https://doi.org/10.7150/thno.62676.
Jiao K, Zhen J, Wu M, Teng M, Yang K, Zhou Q, Hu C, Zhou M, Li Y, Li Z. 27-Hydroxycholesterol-induced EndMT acts via STAT3 signaling to promote breast cancer cell migration by altering the tumor microenvironment. Cancer Biol Med. 2020;17(1):88.
Ye B, Duan Y, Zhou M, Wang Y, Lai Q, Yue K, Cao J, Wu Y, Wang X, Jing C. Hypoxic tumor-derived exosomal miR-21 induces cancer-associated fibroblast activation to promote head and neck squamous cell carcinoma metastasis. Cell Signal. 2023;108:110725. https://doi.org/10.1016/j.cellsig.2023.110725.
Mao X, Xu J, Wang W, Liang C, Hua J, Liu J, Zhang B, Meng Q, Yu X, Shi S. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Mol Cancer. 2021;20(1):131. https://doi.org/10.1186/s12943-021-01428-1.
Jiang Y, Qiu Q, Jing X, Song Z, Zhang Y, Wang C, Liu K, Ye F, Ji X, Luo F, Zhao R. Cancer-associated fibroblast-derived exosome miR-181b-3p promotes the occurrence and development of colorectal cancer by regulating SNX2 expression. Biochem Biophys Res Commun. 2023;641:177–85. https://doi.org/10.1016/j.bbrc.2022.12.026.
Shi L, Zhu W, Huang Y, Zhuo L, Wang S, Chen S, Zhang B, Ke B. Cancer-associated fibroblast-derived exosomal microRNA-20a suppresses the PTEN/PI3K-AKT pathway to promote the progression and chemoresistance of non-small cell lung cancer. 2022;12(7):e989.https://doi.org/10.1002/ctm2.989.
Lavie D, Ben-Shmuel A, Erez N, Scherz-Shouval R. Cancer-associated fibroblasts in the single-cell era. Nature Cancer. 2022;3(7):793–807. https://doi.org/10.1038/s43018-022-00411-z.
Fu W, Zhao H, Liu Y, Nie H, Gao B, Yin F, Wang B, Li T, Zhang T, Wang L, Wu X, Zhu M, Xia L. Exosomes derived from cancer-associated fibroblasts regulate cell progression in clear-cell renal-cell carcinoma. Nephron. 2021;146(4):383–92. https://doi.org/10.1159/000520304.
Strnadová K, Pfeiferová L, Přikryl P, Dvořánková B, Vlčák E, Frýdlová J, Vokurka M, Novotný J, Šáchová J, Hradilová M, Brábek J, Šmigová J, Rösel D, Smetana K, Kolář M, Lacina L. Exosomes produced by melanoma cells significantly influence the biological properties of normal and cancer-associated fibroblasts. Histochem Cell Biol. 2022;157(2):153–72. https://doi.org/10.1007/s00418-021-02052-2.
Liu T, Han C, Fang P, Ma Z, Wang X, Chen H, Wang S, Meng F, Wang C, Zhang E, Dong G, Zhu H, Yin W, Wang J, Zuo X, Qiu M, Wang J, Qian X, Shen H, Xu L, Hu Z, Yin R. Cancer-associated fibroblast-specific lncRNA LINC01614 enhances glutamine uptake in lung adenocarcinoma. J Hematol Oncol. 2022;15(1):141. https://doi.org/10.1186/s13045-022-01359-4.
Yang K, Zhang J, Bao C. Exosomal circEIF3K from cancer-associated fibroblast promotes colorectal cancer (CRC) progression via miR-214/PD-L1 axis. BMC Cancer. 2021;21(1):933. https://doi.org/10.1186/s12885-021-08669-9.
Zhou Y, Tang W, Zhuo H, Zhu D, Rong D, Sun J, Song J. Cancer-associated fibroblast exosomes promote chemoresistance to cisplatin in hepatocellular carcinoma through circZFR targeting signal transducers and activators of transcription (STAT3)/ nuclear factor -kappa B (NF-κB) pathway. Bioengineered. 2022;13(3):4786–97. https://doi.org/10.1080/21655979.2022.2032972.
Al-Awsi GRL, Alsaikhan F, Margiana R, Ahmad I, Patra I, Najm MAA, Yasin G, Rasulova I, Hammid AT, Kzar HH, Al-Gazally ME, Siahmansouri H. Shining the light on mesenchymal stem cell-derived exosomes in breast cancer. Stem Cell Res Ther. 2023;14(1):21. https://doi.org/10.1186/s13287-023-03245-3.
Weng Z, Zhang B, Wu C, Yu F, Han B, Li B, Li L. Therapeutic roles of mesenchymal stem cell-derived extracellular vesicles in cancer. J Hematol Oncol. 2021;14(1):136. https://doi.org/10.1186/s13045-021-01141-y.
Yuan Y-G, Wang J-L, Zhang Y-X, Li L, Reza AMMT, Gurunathan S. Biogenesis, Composition and Potential Therapeutic Applications of Mesenchymal Stem Cells Derived Exosomes in Various Diseases. Int J Nanomedicine. 2023:3177–3210.
Vakhshiteh F, Rahmani S, Ostad SN, Madjd Z, Dinarvand R, Atyabi F. Exosomes derived from miR-34a-overexpressing mesenchymal stem cells inhibit in vitro tumor growth: a new approach for drug delivery. Life Sci. 2021;266:118871.
Phetfong J, Tawonsawatruk T, Kamprom W, Ontong P, Tanyong D, Borwornpinyo S, Supokawej A. Bone marrow-mesenchymal stem cell-derived extracellular vesicles affect proliferation and apoptosis of leukemia cells in vitro. FEBS Open Bio. 2022;12(2):470–9.
Li Q, Wang M, Liu L. The role of exosomes in the stemness maintenance and progression of acute myeloid leukemia. Biochem Pharmacol. 2023:115539
Arianfar E, Shahgordi S, Memarian A. Natural Killer Cell Defects in Breast Cancer: A Key Pathway for Tumor Evasion. Int Rev Immunol. 2021;40(3):197–216. https://doi.org/10.1080/08830185.2020.1845670.
Xie Q-H, Zheng J-Q, Ding J-Y, Wu Y-F, Liu L, Yu Z-L, Chen G. Exosome-mediated immunosuppression in tumor microenvironments. Cells. 2022;11(12):1946.
Huyan T, Gao L, Gao N, Wang C, Guo W, Zhou X, Li Q. miR-221-5p and miR-186-5p are the critical bladder cancer derived exosomal miRNAs in natural killer cell dysfunction. Int J Mol Sci. 2022;23(23):15177.
Zhao Y, Liu L, Sun R, Cui G, Guo S, Han S, Li Z, Bai T, Teng L. Exosomes in cancer immunoediting and immunotherapy. Asian J Pharm Sci. 2022;17(2):193–205.
Li X, Liu Y, Zheng S, Zhang T, Wu J, Sun Y, Zhang J, Liu G. Role of exosomes in the immune microenvironment of ovarian cancer. Oncol Lett. 2021;21(5):1–17.
Xu Z, Chen Y, Ma L, Chen Y, Liu J, Guo Y, Yu T, Zhang L, Zhu L, Shu Y (2022) Role of exosomal non-coding RNAs from tumor cells and tumor-associated macrophages in the tumor microenvironment. Molecular Therapy.
He Z, Wang J, Zhu C, Xu J, Chen P, Jiang X, Chen Y, Jiang J, Sun C. Exosome-derived FGD5-AS1 promotes tumor-associated macrophage M2 polarization-mediated pancreatic cancer cell proliferation and metastasis. Cancer Lett. 2022;548:215751.
Xu M, Zhou C, Weng J, Chen Z, Zhou Q, Gao J, Shi G, Ke A, Ren N, Sun H. Tumor associated macrophages-derived exosomes facilitate hepatocellular carcinoma malignance by transferring lncMMPA to tumor cells and activating glycolysis pathway. J Exp Clin Cancer Res. 2022;41(1):253.
Ma F, Vayalil J, Lee G, Wang Y, Peng G. Emerging role of tumor-derived extracellular vesicles in T cell suppression and dysfunction in the tumor microenvironment. J Immunotherapy Cancer. 2021;9(10).
Guan L, Wu B, Li T, Beer LA, Sharma G, Li M, Lee CN, Liu S, Yang C, Huang L. HRS phosphorylation drives immunosuppressive exosome secretion and restricts CD8+ T-cell infiltration into tumors. Nat Commun. 2022;13(1):4078.
Sun J, Jia H, Bao X, Wu Y, Zhu T, Li R, Zhao H. Tumor exosome promotes Th17 cell differentiation by transmitting the lncRNA CRNDE-h in colorectal cancer. Cell Death Dis. 2021;12(1):123.
Ning T, Li J, He Y, Zhang H, Wang X, Deng T, Liu R, Li H, Bai M, Fan Q. Exosomal miR-208b related with oxaliplatin resistance promotes Treg expansion in colorectal cancer. Mol Ther. 2021;29(9):2723–36.
Yu H, Huang T, Wang D, Chen L, Lan X, Liu X, Chen K, He H, Li S, Zhou Y. Acute lymphoblastic leukemia-derived exosome inhibits cytotoxicity of natural killer cells by TGF-β signaling pathway. 3 Biotech. 2021;11(7):313.
Zhang P-F, Gao C, Huang X-Y, Lu J-C, Guo X-J, Shi G-M, Cai J-B, Ke A-W. Cancer cell-derived exosomal circUHRF1 induces natural killer cell exhaustion and may cause resistance to anti-PD1 therapy in hepatocellular carcinoma. Mol Cancer. 2020;19(1):1–15.
Kim H, Kim DW, Cho J-Y. Exploring the key communicator role of exosomes in cancer microenvironment through proteomics. Proteome Science. 2019;17(1):5. https://doi.org/10.1186/s12953-019-0154-z.
Giannotta C, Autino F, Massaia M. The immune suppressive tumor microenvironment in multiple myeloma: the contribution of myeloid-derived suppressor cells. Front Immunol. 2023;13:1102471.
Chen Z, Yuan R, Hu S, Yuan W, Sun Z. Roles of the exosomes derived from myeloid-derived suppressor cells in tumor immunity and cancer progression. Front Immunol. 2022;13:817942.
Su M-T, Kumata S, Endo S, Okada Y, Takai T. LILRB4 promotes tumor metastasis by regulating MDSCs and inhibiting miR-1 family miRNAs. Oncoimmunology. 2022;11(1):2060907.
Chen P, Liu Z, Xiao H, Yang X, Li T, Huang W, Zhou H. Effect of tumor exosome-derived Lnc RNA HOTAIR on the growth and metastasis of gastric cancer. Clin Transl Oncol. 2023. https://doi.org/10.1007/s12094-023-03208-3.
Jiang M, Zhang W, Zhang R, Liu P, Ye Y, Yu W, Guo X, Yu J. Cancer exosome-derived miR-9 and miR-181a promote the development of early-stage MDSCs via interfering with SOCS3 and PIAS3 respectively in breast cancer. Oncogene. 2020;39(24):4681–94.
Sun J, Lu Z, Fu W, Lu K, Gu X, Xu F, Dai J, Yang Y, Jiang J. Exosome-derived ADAM17 promotes liver metastasis in colorectal cancer. Front Pharmacol. 2021;12:734351.
Wang M, Qin Z, Wan J, Yan Y, Duan X, Yao X, Jiang Z, Li W, Qin Z. Tumor-derived exosomes drive pre-metastatic niche formation in lung via modulating CCL1+ fibroblast and CCR8+ Treg cell interactions. Cancer Immunol Immunother. 2022;71(11):2717–30.
Zhang C, Wang X-Y, Zhang P, He T-C, Han J-H, Zhang R, Lin J, Fan J, Lu L, Zhu W-W, Jia H-L, Zhang J-B, Chen J-H. Cancer-derived exosomal HSPC111 promotes colorectal cancer liver metastasis by reprogramming lipid metabolism in cancer-associated fibroblasts. Cell Death Dis. 2022;13(1):57. https://doi.org/10.1038/s41419-022-04506-4.
Aldoghachi AF, Chong ZX, Yeap SK, Cheong SK, Ho WY, Ong AHK. Stem cells for cancer therapy: translating the uncertainties and possibilities of stem cell properties into opportunities for effective cancer therapy. Int J Mol Sci. 2023;24(2):1012.
Gu P, Sun M, Li L, Yang Y, Jiang Z, Ge Y, Wang W, Mu W, Wang H. Breast tumor-derived exosomal microRNA-200b-3p promotes specific organ metastasis through regulating CCL2 expression in lung epithelial cells. Front Cell Dev Biol. 2021;9:657158.
Zhou Y, Chen F, Xie X, Nie H, Lian S, Zhong C, Fu C, Shen W, Li B, Ye Y. Tumor-derived exosome promotes metastasis via altering its phenotype and inclusions. J Cancer. 2021;12(14):4240.
Gonzalez-Avila G, Sommer B, Mendoza-Posada DA, Ramos C, Garcia-Hernandez AA, Falfan-Valencia R. Matrix metalloproteinases participation in the metastatic process and their diagnostic and therapeutic applications in cancer. Crit Rev Oncol Hematol. 2019;137:57–83. https://doi.org/10.1016/j.critrevonc.2019.02.010.
Gao Z, Pang B, Li J, Gao N, Fan T, Li Y. Emerging Role of Exosomes in Liquid Biopsy for Monitoring Prostate Cancer Invasion and Metastasis. Front Cell Dev Biol. 2021;9. https://doi.org/10.3389/fcell.2021.679527.
Dai J, Su Y, Zhong S, Cong L, Liu B, Yang J, Tao Y, He Z, Chen C, Jiang Y. Exosomes: key players in cancer and potential therapeutic strategy. Signal Transduct Target Ther. 2020;5(1):145. https://doi.org/10.1038/s41392-020-00261-0.
Hernández-Barranco A, Nogués L, Peinado H. Could extracellular vesicles contribute to generation or awakening of “sleepy” metastatic niches? Front Cell Dev Biol. 2021;9:625221.
Jiang J, Li J, Zhou X, Zhao X, Huang B, Qin Y. Exosomes regulate the epithelial-mesenchymal transition in cancer. Front Oncol. 2022;12:864980.
Kirave P, Gondaliya P, Kulkarni B, Rawal R, Garg R, Jain A, Kalia K. Exosome mediated miR-155 delivery confers cisplatin chemoresistance in oral cancer cells via epithelial-mesenchymal transition. Oncotarget. 2020;11(13):1157.
Lin Q, Zhou C-R, Bai M-J, Zhu D, Chen J-W, Wang H-F, Li M-A, Wu C, Li Z-R, Huang M-S. Exosome-mediated miRNA delivery promotes liver cancer EMT and metastasis. Am J Transl Res. 2020;12(3):1080.
Shi Z, Jiang T, Cao B, Sun X, Liu J. CAF-derived exosomes deliver LINC01410 to promote epithelial-mesenchymal transition of esophageal squamous cell carcinoma. Exp Cell Res. 2022;412(2):113033.
Kim K, Sohn YJ, Lee R, Yoo HJ, Kang JY, Choi N, Na D, Yeon JH. Cancer-associated fibroblasts differentiated by exosomes isolated from cancer cells promote cancer cell invasion. Int J Mol Sci. 2020;21(21):8153.
Chen K, Wang Q, Liu X, Wang F, Yang Y, Tian X. Hypoxic pancreatic cancer derived exosomal miR-30b-5p promotes tumor angiogenesis by inhibiting GJA1 expression. Int J Biol Sci. 2022;18(3):1220.
Liu B, Chen J, Shang F, Lian M, Shen X, Fang J. Tumor-derived exosome FGD5-AS1 promotes angiogenesis, vascular permeability, and metastasis in thyroid cancer by targeting the miR-6838–5p/VAV2 Axis. J Oncol. 2022;2022.
Mostafazadeh M, Samadi N, Kahroba H, Baradaran B, Haiaty S, Nouri M. Potential roles and prognostic significance of exosomes in cancer drug resistance. Cell Biosci. 2021;11(1):1–15.
Yang K, Zhang F, Luo B, Qu Z. CAFs-derived small extracellular vesicles circN4BP2L2 promotes proliferation and metastasis of colorectal cancer via miR-664b-3p/HMGB3 pathway. Cancer Biol Ther. 2022;23(1):404–16.
Wang Y, Wang Y, Qin Z, Cai S, Yu L, Hu H, Zeng S. The role of non-coding RNAs in ABC transporters regulation and their clinical implications of multidrug resistance in cancer. Expert Opin Drug Metab Toxicol. 2021;17(3):291–306.
Lyu T, Wang Y, Li D, Yang H, Qin B, Zhang W, Li Z, Cheng C, Zhang B, Guo R. Exosomes from BM-MSCs promote acute myeloid leukemia cell proliferation, invasion and chemoresistance via upregulation of S100A4. Exp Hematol Oncol. 2021;10(1):1–13.
Guo G, Tan Z, Liu Y, Shi F, She J. The therapeutic potential of stem cell-derived exosomes in the ulcerative colitis and colorectal cancer. Stem Cell Res Ther. 2022;13(1):1–18.
Singh D, Khan MA, Siddique HR. Specific targeting of cancer stem cells by immunotherapy: a possible stratagem to restrain cancer recurrence and metastasis. Biochem Pharmacol. 2022;198:114955. https://doi.org/10.1016/j.bcp.2022.114955.
Gunassekaran GR, Vadevoo SMP, Baek M-C, Lee B. M1 macrophage exosomes engineered to foster M1 polarization and target the IL-4 receptor inhibit tumor growth by reprogramming tumor-associated macrophages into M1-like macrophages. Biomaterials. 2021;278:121137.
Huo H, Yang S, Wu H, Sun Y, Zhao R, Ye R, Yan D, Shi X, Yang J. Brain endothelial cells-derived extracellular vesicles overexpressing ECRG4 inhibit glioma proliferation through suppressing inflammation and angiogenesis. J Tissue Eng Regen Med. 2021;15(12):1162–71.
Wang X, Qian T, Bao S, Zhao H, Chen H, Xing Z, Li Y, Zhang M, Meng X, Wang C. Circulating exosomal miR-363-5p inhibits lymph node metastasis by downregulating PDGFB and serves as a potential noninvasive biomarker for breast cancer. Mol Oncol. 2021;15(9):2466–79.
Nicolini A, Ferrari P, Biava PM. Exosomes and cell communication: from tumour-derived exosomes and their role in tumour progression to the use of exosomal cargo for cancer treatment. Cancers. 2021;13(4):822.
Kugeratski FG, Kalluri R. Exosomes as mediators of immune regulation and immunotherapy in cancer. FEBS J. 2021;288(1):10–35.
Zhang H-Y, Liang H-X, Wu S-H, Jiang H-Q, Wang Q, Yu Z-J. Overexpressed tumor suppressor exosomal miR-15a-5p in cancer cells inhibits PD1 expression in CD8+ T cells and suppresses the hepatocellular carcinoma progression. Front Oncol. 2021;11:622263.
Humphries B, Wang Z, Yang C. MicroRNA Regulation of Breast Cancer Stemness. Int J Mol Sci. 2021;22(7). https://doi.org/10.3390/ijms22073756.
Xu Y, Shen L, Li F, Yang J, Wan X, Ouyang M. microRNA-16-5p-containing exosomes derived from bone marrow-derived mesenchymal stem cells inhibit proliferation, migration, and invasion, while promoting apoptosis of colorectal cancer cells by downregulating ITGA2. J Cell Physiol. 2019;234(11):21380–94.
Kurniawati I, Liu M-C, Hsieh C-L, Do AD, Sung S-Y. Targeting castration-resistant prostate cancer using mesenchymal stem cell exosomes for therapeutic MicroRNA-let-7c delivery. Front Bioscience-Landmark. 2022;27(9):256.
Ding B, Lou W, Fan W, Pan J. Exosomal miR-374c-5p derived from mesenchymal stem cells suppresses epithelial-mesenchymal transition of hepatocellular carcinoma via the LIMK1-Wnt/β-catenin axis. Environ Toxicol. 2023;38(5):1038–52.
Li T, Wan Y, Su Z, Li J, Han M, Zhou C. Mesenchymal stem cell-derived exosomal microRNA-3940-5p inhibits colorectal cancer metastasis by targeting integrin α6. Dig Dis Sci. 2021;66:1916–27.
Xiao-Ni L, Zhang C-B, Hai L, Xiao-Yuan T, Rong Z, Hui-Lan W, Jie L. microRNA-204 shuttled by mesenchymal stem cell-derived exosomes inhibits the migration and invasion of non-small-cell lung cancer cells via the KLF7/AKT/HIF-1α axis. Neoplasma. 202168(4).
Xie X, Ji J, Chen X, Xu W, Chen H, Zhu S, Wu J, Wu Y, Sun Y, Sai W. Human umbilical cord mesenchymal stem cell-derived exosomes carrying hsa-miRNA-128-3p suppress pancreatic ductal cell carcinoma by inhibiting Galectin-3. Clin Transl Oncol. 2022;24(3):517–31.
Xia J, Miao Y, Wang X, Huang X, Dai J. Recent progress of dendritic cell-derived exosomes (Dex) as an anti-cancer nanovaccine. Biomed Pharmacother. 2022;152:113250.
Wang Y, Xiang Y, Xin VW, Wang X-W, Peng X-C, Liu X-Q, Wang D, Li N, Cheng J-T, Lyv Y-N. Dendritic cell biology and its role in tumor immunotherapy. J Hematol Oncol. 2020;13(1):1–18.
Chen HL, Luo YP, Lin MW, Peng XX, Liu ML, Wang YC, Li SJ, Yang DH, Yang ZX. Serum exosomal miR-16-5p functions as a tumor inhibitor and a new biomarker for PD-L1 inhibitor-dependent immunotherapy in lung adenocarcinoma by regulating PD-L1 expression. Cancer Med. 2022;11(13):2627–43.
Kim H, Kim DW, Cho J-Y. Exploring the key communicator role of exosomes in cancer microenvironment through proteomics. Proteome science. 2019;17(1):1–14.
Sun M, Li H, Feng B. miR-30e-3p in natural killer cell-derived exosomes inhibits the proliferation and invasion of human esophageal squamous carcinoma cells. Xi bao yu fen zi Mian yi xue za zhi= Chin J Cell Mol Immunol. 2023;39(4):295–302.
Li Z, Suo B, Long G, Gao Y, Song J, Zhang M, Feng B, Shang C, Wang D. Exosomal miRNA-16-5p derived from M1 macrophages enhances T cell-dependent immune response by regulating PD-L1 in gastric cancer. Front Cell Dev Biol. 2020;8:572689.
Wang S, Lei B, Zhang E, Gong P, Gu J, He L, Han L, Yuan Z. Targeted therapy for inflammatory diseases with mesenchymal stem cells and their derived exosomes: from basic to clinics. Int J Nanomedicine. 2022;17:1757–81. https://doi.org/10.2147/ijn.S355366.
Atiya H, Frisbie L, Pressimone C, Coffman L. Mesenchymal stem cells in the tumor microenvironment. Adv Exp Med Biol. 2020;1234:31–42. https://doi.org/10.1007/978-3-030-37184-5_3.
Zhang C, Wang X-Y, Zhang P, He T-C, Han J-H, Zhang R, Lin J, Fan J, Lu L, Zhu W-W. Cancer-derived exosomal HSPC111 promotes colorectal cancer liver metastasis by reprogramming lipid metabolism in cancer-associated fibroblasts. Cell Death Dis. 2022;13(1):57.
Lin Z, Wu Y, Xu Y, Li G, Li Z, Liu T. Mesenchymal stem cell-derived exosomes in cancer therapy resistance: recent advances and therapeutic potential. Mol Cancer. 2022;21(1):179.
Chen L-Y, Kao T-W, Chen C-C, Niaz N, Lee H-L, Chen Y-H, Kuo C-C, Shen Y-A. Frontier review of the molecular mechanisms and current approaches of stem cell-derived exosomes. Cells. 2023;12(7):1018.
Wang Q, Li T, Wu W, Ding G. Interplay between mesenchymal stem cell and tumor and potential application. Hum Cell. 2020;33:444–58.
Sun Z, Zhang J, Li J, Li M, Ge J, Wu P, You B, Qian H. Roles of mesenchymal stem cell-derived exosomes in cancer development and targeted therapy. Stem Cells Int. 2021;2021.
Rosenberger L, Ezquer M, Lillo-Vera F, Pedraza PL, Ortúzar MI, González PL, Figueroa-Valdés AI, Cuenca J, Ezquer F, Khoury M. Stem cell exosomes inhibit angiogenesis and tumor growth of oral squamous cell carcinoma. Sci Rep. 2019;9(1):663.
Lou G, Chen L, Xia C, Wang W, Qi J, Li A, Zhao L, Chen Z, Zheng M, Liu Y. MiR-199a-modified exosomes from adipose tissue-derived mesenchymal stem cells improve hepatocellular carcinoma chemosensitivity through mTOR pathway. J Exp Clin Cancer Res. 2020;39(1):4. https://doi.org/10.1186/s13046-019-1512-5.
Lee KS, Choi JS, Cho YW. Reprogramming of cancer stem cells into non-tumorigenic cells using stem cell exosomes for cancer therapy. Biochem Biophys Res Commun. 2019;512(3):511–6. https://doi.org/10.1016/j.bbrc.2019.03.072.
Shen D, He Z. Mesenchymal stem cell-derived exosomes regulate the polarization and inflammatory response of macrophages via miR-21-5p to promote repair after myocardial reperfusion injury. Ann Transl Med. 2021;9(16):1323. https://doi.org/10.21037/atm-21-3557.
Jia Z, Zhu H, Sun H, Hua Y, Zhang G, Jiang J, Wang X. Adipose mesenchymal stem cell-derived exosomal microRNA-1236 reduces resistance of breast cancer cells to cisplatin by suppressing SLC9A1 and the Wnt/β-catenin signaling. Cancer Manage Res. 2020:8733–8744.
Zargar MJ, Kaviani S, Vasei M, Soufi Zomorrod M, Heidari Keshel S, Soleimani M. Therapeutic role of mesenchymal stem cell-derived exosomes in respiratory disease. Stem Cell Res Ther. 2022;13(1):194.
Cao JY, Wang B, Tang TT, Wen Y, Li ZL, Feng ST, Wu M, Liu D, Yin D, Ma KL, Tang RN, Wu QL, Lan HY, Lv LL, Liu BC. Exosomal miR-125b-5p deriving from mesenchymal stem cells promotes tubular repair by suppression of p53 in ischemic acute kidney injury. Theranostics. 2021;11(11):5248–66. https://doi.org/10.7150/thno.54550.
Parfejevs V, Sagini K, Buss A, Sobolevska K, Llorente A, Riekstina U, Abols A. Adult stem cell-derived extracellular vesicles in cancer treatment: opportunities and challenges. Cells. 2020;9(5):1171.
Ren W, Hou J, Yang C, Wang H, Wu S, Wu Y, Zhao X, Lu C. Extracellular vesicles secreted by hypoxia pre-challenged mesenchymal stem cells promote non-small cell lung cancer cell growth and mobility as well as macrophage M2 polarization via miR-21-5p delivery. J Exp Clin Cancer Res. 2019;38:1–14.
Christodoulou I, Goulielmaki M, Devetzi M, Panagiotidis M, Koliakos G, Zoumpourlis V. Mesenchymal stem cells in preclinical cancer cytotherapy: a systematic review. Stem Cell Res Ther. 2018;9(1):1–38.
Weng Z, Zhang B, Wu C, Yu F, Han B, Li B, Li L. Therapeutic roles of mesenchymal stem cell-derived extracellular vesicles in cancer. J Hematol Oncol. 2021;14:1–22.
Bolandi Z, Mokhberian N, Eftekhary M, Sharifi K, Soudi S, Ghanbarian H, Hashemi SM. Adipose derived mesenchymal stem cell exosomes loaded with miR-10a promote the differentiation of Th17 and Treg from naive CD4+ T cell. Life Sci. 2020;259:118218.
Shimbo K, Miyaki S, Ishitobi H, Kato Y, Kubo T, Shimose S, Ochi M. Exosome-formed synthetic microRNA-143 is transferred to osteosarcoma cells and inhibits their migration. Biochem Biophys Res Commun. 2014;445(2):381–7.
Kalimuthu S, Gangadaran P, Rajendran RL, Zhu L, Oh JM, Lee HW, Gopal A, Baek SH, Jeong SY, Lee S-W. A new approach for loading anticancer drugs into mesenchymal stem cell-derived exosome mimetics for cancer therapy. Front Pharmacol. 2018;9:1116.
Bagheri E, Abnous K, Farzad SA, Taghdisi SM, Ramezani M, Alibolandi M. Targeted doxorubicin-loaded mesenchymal stem cells-derived exosomes as a versatile platform for fighting against colorectal cancer. Life Sci. 2020;261:118369. https://doi.org/10.1016/j.lfs.2020.118369.
Li X, Wang K, Ai H. Human umbilical cord mesenchymal stem cell-derived extracellular vesicles inhibit endometrial cancer cell proliferation and migration through delivery of exogenous miR-302a. Stem Cells int. 2019;2019.
Zhang F, Lu Y, Wang M, Zhu J, Li J, Zhang P, Yuan Y, Zhu F. Exosomes derived from human bone marrow mesenchymal stem cells transfer miR-222-3p to suppress acute myeloid leukemia cell proliferation by targeting IRF2/INPP4B. Mol Cell Probes. 2020;51:101513.
Cheng H, Ding J, Tang G, Huang A, Gao L, Yang J, Chen L. Human mesenchymal stem cells derived exosomes inhibit the growth of acute myeloid leukemia cells via regulating miR-23b-5p/TRIM14 pathway. Mol Med. 2021;27(1):128. https://doi.org/10.1186/s10020-021-00393-1.
Zhou W, Zhou Y, Chen X, Ning T, Chen H, Guo Q, Zhang Y, Liu P, Zhang Y, Li C. Pancreatic cancer-targeting exosomes for enhancing immunotherapy and reprogramming tumor microenvironment. Biomaterials. 2021;268:120546.
Li D, Lin F, Li G, Zeng F. Exosomes derived from mesenchymal stem cells curbs the progression of clear cell renal cell carcinoma through T-cell immune response. Cytotechnology. 2021;73(4):593–604.
Qiu Y, Sun J, Qiu J, Chen G, Wang X, Mu Y, Li K, Wang W. Antitumor activity of cabazitaxel and MSC-TRAIL derived extracellular vesicles in drug-resistant oral squamous cell carcinoma. Cancer Manage Res. 2020;10809–10820.
Yu L, Gui S, Liu Y, Qiu X, Zhang G, Xa Z, Pan J, Fan J, Qi S, Qiu B. Exosomes derived from microRNA-199a-overexpressing mesenchymal stem cells inhibit glioma progression by down-regulating AGAP2. Aging (Albany NY). 2019;11(15):5300.
Yao X, Mao Y, Wu D, Zhu Y, Lu J, Huang Y, Guo Y, Wang Z, Zhu S, Li X, Lu Y. Exosomal circ_0030167 derived from BM-MSCs inhibits the invasion, migration, proliferation and stemness of pancreatic cancer cells by sponging miR-338-5p and targeting the Wif1/Wnt8/β-catenin axis. Cancer Lett. 2021;512:38–50. https://doi.org/10.1016/j.canlet.2021.04.030.
Xu Y, Lai Y, Cao L, Li Y, Chen G, Chen L, Weng H, Chen T, Wang L, Ye Y. Human umbilical cord mesenchymal stem cells-derived exosomal microRNA-451a represses epithelial–mesenchymal transition of hepatocellular carcinoma cells by inhibiting ADAM10. RNA Biol. 2021;18(10):1408–23.
Cai H, Yang X, Gao Y, Xu Z, Yu B, Xu T, Li X, Xu W, Wang X, Hua L. Exosomal microRNA-9-3p secreted from BMSCs downregulates ESM1 to suppress the development of bladder cancer. Molecular Therapy-Nucleic Acids. 2019;18:787–800.
Li P, Xin H, Lu L. Extracellular vesicle-encapsulated microRNA-424 exerts inhibitory function in ovarian cancer by targeting MYB. J Transl Med. 2021;19(1):1–17.
Barbato L, Bocchetti M, Di Biase A, Regad T. Cancer stem cells and targeting strategies. Cells. 2019;8(8):926.
Yang J, Teng Y. Harnessing cancer stem cell-derived exosomes to improve cancer therapy. J Exp Clin Cancer Res. 2023;42(1):1–15.
Venkatesh V, Nataraj R, Thangaraj GS, Karthikeyan M, Gnanasekaran A, Kaginelli SB, Kuppanna G, Kallappa CG, Basalingappa KM. Targeting Notch signalling pathway of cancer stem cells. Stem Cell Investig. 2018;5.
Landeros N, Castillo I, Pérez-Castro R. Preclinical and clinical trials of new treatment strategies targeting cancer stem cells in subtypes of breast cancer. Cells. 2023;12(5):720.
Orfali N, O’Donovan TR, Cahill MR, Benjamin D, Nanus DM, McKenna SL, Gudas LJ, Mongan NP. All-trans retinoic acid (ATRA)-induced TFEB expression is required for myeloid differentiation in acute promyelocytic leukemia (APL). Eur J Haematol. 2020;104(3):236–50. https://doi.org/10.1111/ejh.13367.
Catalano M, O’Driscoll L. Inhibiting extracellular vesicles formation and release: a review of EV inhibitors. J Extracell Vesicles. 2020;9(1):1703244. https://doi.org/10.1080/20013078.2019.1703244.
Song L, Tang S, Han X, Jiang Z, Dong L, Liu C, Liang X, Dong J, Qiu C, Wang Y. KIBRA controls exosome secretion via inhibiting the proteasomal degradation of Rab27a. Nat Commun. 2019;10(1):1639.
Kim JH, Lee C-H, Baek M-C. Dissecting exosome inhibitors: therapeutic insights into small-molecule chemicals against cancer. Exp Mol Med. 2022;54(11):1833–43. https://doi.org/10.1038/s12276-022-00898-7.
Peak TC, Panigrahi GK, Praharaj PP, Su Y, Shi L, Chyr J, Rivera-Chávez J, Flores-Bocanegra L, Singh R, Vander Griend DJ, Oberlies NH, Kerr BA, Hemal A, Bitting RL, Deep G. Syntaxin 6-mediated exosome secretion regulates enzalutamide resistance in prostate cancer. Mol Carcinog. 2020;59(1):62–72. https://doi.org/10.1002/mc.23129.
Marie PP, Fan SJ, Mason J, Wells A, Mendes CC, Wainwright SM, Scott S, Fischer R, Harris AL, Wilson C, Goberdhan DCI. Accessory ESCRT-III proteins are conserved and selective regulators of Rab11a-exosome formation. J Extracell Vesicles. 2023;12(3):e12311. https://doi.org/10.1002/jev2.12311.
Dhar R, Bhattacharya B, Mandal D, Devi A, Thorat ND. Exosome-based cancer vaccine: a cutting-edge approach – correspondence. Int J Surg. 2022;108:106993. https://doi.org/10.1016/j.ijsu.2022.106993.
Kim H, Kim EH, Kwak G, Chi S-G, Kim SH, Yang Y. Exosomes: cell-derived nanoplatforms for the delivery of cancer therapeutics. Int J Mol Sci. 2020;22(1):14.
Chen J, Li P, Zhang T, Xu Z, Huang X, Wang R, Du L. Review on strategies and technologies for exosome isolation and purification. Front Bioengineering Biotechnol. 2022;9:811971.
Zhang Y, Li J, Gao W, Xie N. Exosomes as anticancer drug delivery vehicles: prospects and challenges. Front Bioscience-Landmark. 2022;27(10):293.
Zhang R, Bu T, Cao R, Li Z, Wang C, Huang B, Wei M, Yuan L, Yang G. An optimized exosome production strategy for enhanced yield while without sacrificing cargo loading efficiency. J Nanobiotechnol. 2022;20(1):1–15.
Zhang Y, Liu Q, Zhang X, Huang H, Tang S, Chai Y, Xu Z, Li M, Chen X, Liu J. Recent advances in exosome-mediated nucleic acid delivery for cancer therapy. J Nanobiotechnol. 2022;20(1):279.
Jafari D, Malih S, Eini M, Jafari R, Gholipourmalekabadi M, Sadeghizadeh M, Samadikuchaksaraei A. Improvement, scaling-up, and downstream analysis of exosome production. Critical Rev Biotechnol. 2020:1–15. doi:https://doi.org/10.1080/07388551.2020.1805406.
Tacheny A. Scaling-up the production of stem cell-derived extracellular vesicles in stirred-tank bioreactors. Cell Gene Therapy Insights. 2021;7:1077–83.
Li X, Corbett AL, Taatizadeh E, Tasnim N, Little JP, Garnis C, Daugaard M, Guns E, Hoorfar M, Li IT. Challenges and opportunities in exosome research—Perspectives from biology, engineering, and cancer therapy. APL Bioengineering. 2019;3(1).
Whitford W, Guterstam P. Exosome manufacturing status. Future Med Chem. 2019;11(10):1225–36.
Han L, Zhao Z, He C, Li J, Li X, Lu M. Removing the stumbling block of exosome applications in clinical and translational medicine: expand production and improve accuracy. Stem Cell Res Ther. 2023;14(1):1–13.
Hussen BM, Faraj GSH, Rasul MF, Hidayat HJ, Salihi A, Baniahmad A, Taheri M, Ghafouri-Frad S. Strategies to overcome the main challenges of the use of exosomes as drug carrier for cancer therapy. Cancer Cell Int. 2022;22(1):1–23.
Jeyaraman M, Muthu S, Jeyaraman N. Challenges in the clinical translation of exosomal therapy in regenerative medicine. Future Medicine. 2022;17.
Makler A, Asghar W. Exosomal biomarkers for cancer diagnosis and patient monitoring. Expert Rev Mol Diagn. 2020;20(4):387–400. https://doi.org/10.1080/14737159.2020.1731308.
Gupta D, Zickler AM, El Andaloussi S. Dosing extracellular vesicles. Adv Drug Deliv Rev. 2021;178:113961.
Tzng E, Bayardo N, Yang PC. Current challenges surrounding exosome treatments. Extracellular Vesicle. 2023;2:100023. https://doi.org/10.1016/j.vesic.2023.100023.
Driscoll J, Yan IK, Patel T. Development of a lyophilized off-the-shelf mesenchymal stem cell-derived acellular therapeutic. Pharmaceutics. 2022;14(4):849.
Zeien J, Qiu W, Triay M, Dhaibar HA, Cruz-Topete D, Cornett EM, Urits I, Viswanath O, Kaye AD. Clinical implications of chemotherapeutic agent organ toxicity on perioperative care. Biomed Pharmacother. 2022;146:112503. https://doi.org/10.1016/j.biopha.2021.112503.
Crommelin DJ, van Hoogevest P, Storm G. The role of liposomes in clinical nanomedicine development. What now? Now what? J Control Release. 2020;318:256–63.
Logozzi M, Di Raimo R, Mizzoni D, Fais S What we know on the potential use of exosomes for nanodelivery. In: Seminars in Cancer Biology, 2022. Elsevier, pp 13-25.
Palazzolo S, Memeo L, Hadla M, Duzagac F, Steffan A, Perin T, Canzonieri V, Tuccinardi T, Caligiuri I, Rizzolio F. Cancer extracellular vesicles: next-generation diagnostic and drug delivery nanotools. Cancers. 2020;12(11):3165.
Aqil F, Gupta RC (2022) Exosomes in cancer therapy. vol 14. MDPI.
Chen L, Wang L, Zhu L, Xu Z, Liu Y, Li Z, Zhou J, Luo F. Exosomes as drug carriers in anti-cancer therapy. Front Cell Dev Biol. 2022;10:728616.
Ahmadi M, Mahmoodi M, Shoaran M, Nazari-Khanamiri F, Rezaie J. Harnessing normal and engineered mesenchymal stem cells derived exosomes for cancer therapy: opportunity and challenges. Int J Mol Sci. 2022;23(22):13974.
Akbari A, Nazari-Khanamiri F, Ahmadi M, Shoaran M, Rezaie J. Engineered exosomes for tumor-targeted drug delivery: a focus on genetic and chemical functionalization. Pharmaceutics. 2022;15(1):66.
Chen C, Sun M, Wang J, Su L, Lin J, Yan X. Active cargo loading into extracellular vesicles: highlights the heterogeneous encapsulation behaviour. J Extracell Vesicles. 2021;10(13):e12163. https://doi.org/10.1002/jev2.12163.
Kim JY, Rhim W-K, Yoo Y-I, Kim D-S, Ko K-W, Heo Y, Park CG, Han DK. Defined MSC exosome with high yield and purity to improve regenerative activity. J Tissue Eng. 2021;12:20417314211008624.
Piawah S, Venook AP. Targeted therapy for colorectal cancer metastases: a review of current methods of molecularly targeted therapy and the use of tumor biomarkers in the treatment of metastatic colorectal cancer. Cancer. 2019;125(23):4139–47. https://doi.org/10.1002/cncr.32163.
Bahr MM, Amer MS, Abo-El-Sooud K, Abdallah AN, El-Tookhy OS. Preservation techniques of stem cells extracellular vesicles: a gate for manufacturing of clinical grade therapeutic extracellular vesicles and long-term clinical trials. Int J Veterinary Sci Med. 2020;8(1):1–8.
Wang N, Pei B, Yuan X, Yi C, Wiredu Ocansey DK, Qian H, Mao F. Emerging roles of mesenchymal stem cell-derived exosomes in gastrointestinal cancers. Front Bioengineering Biotechnol. 2022;10:1019459.
Von Schulze A, Deng F. A review on exosome-based cancer therapy. J Cancer Metastasis Treatment. 2020;6:42.
Sun Y, Liu G, Zhang K, Cao Q, Liu T, Li J. Mesenchymal stem cells-derived exosomes for drug delivery. Stem Cell Res Ther. 2021;12(1):561. https://doi.org/10.1186/s13287-021-02629-7.
Wu Q, Zhou L, Lv D, Zhu X, Tang H. Exosome-mediated communication in the tumor microenvironment contributes to hepatocellular carcinoma development and progression. J Hematol Oncol. 2019;12(1):53. https://doi.org/10.1186/s13045-019-0739-0.
Salunkhe S, Basak M, Chitkara D, Mittal A. Surface functionalization of exosomes for target-specific delivery and in vivo imaging & tracking: Strategies and significance. J Control Release. 2020;326:599–614.
Xiao Y, Zhong J, Zhong B, Huang J, Jiang L, Jiang Y, Yuan J, Sun J, Dai L, Yang C. Exosomes as potential sources of biomarkers in colorectal cancer. Cancer Lett. 2020;476:13–22.
Li X, Li X, Zhang B, He B.The role of cancer stem cell-derived exosomes in cancer progression. Stem Cells Int. 2022;2022.
Yu W, Hurley J, Roberts D, Chakrabortty S, Enderle D, Noerholm M, Breakefield X, Skog J. Exosome-based liquid biopsies in cancer: opportunities and challenges. Ann Oncol. 2021;32(4):466–77.
Huerta M, Roselló S, Sabater L, Ferrer A, Tarazona N, Roda D, Gambardella V, Alfaro-Cervelló C, Garcés-Albir M, Cervantes A, Ibarrola-Villava M. Circulating Tumor DNA Detection by Digital-Droplet PCR in Pancreatic Ductal Adenocarcinoma: A Systematic Review. Cancers (Basel). 2021;13(5). https://doi.org/10.3390/cancers13050994.
Yamamoto H, Watanabe Y, Itoh F. Cell-Free DNA. Biomarkers in Cancer Therapy: Liquid Biopsy Comes of Age. 2019:11–24.
Wong C-H, Chen Y-C. Clinical significance of exosomes as potential biomarkers in cancer. World J Clin Cases. 2019;7(2):171.
Tao SC, Huang JY, Wei ZY, Li ZX, Guo SC. EWSAT1 Acts in Concert with Exosomes in Osteosarcoma Progression and Tumor-Induced Angiogenesis: The “Double Stacking Effect.” Adv Biosyst. 2020;4(9):e2000152. https://doi.org/10.1002/adbi.202000152.
Surana R, LeBleu VS, Lee JJ, Smaglo BG, Zhao D, Lee MS, Wolff RA, Overman MJ, Mendt MC, McAndrews KM, Yang S, Rezvani K, Kalluri R, Maitra A, Shpall EJ, Pant S. Phase I study of mesenchymal stem cell (MSC)-derived exosomes with KRASG12D siRNA in patients with metastatic pancreatic cancer harboring a KRASG12D mutation. J Clin Oncol. 2022;40 (4_suppl):TPS633-TPS633. https://doi.org/10.1200/JCO.2022.40.4_suppl.TPS633.
BioSciences C (2020) A first-in-human study of CDK-002 (exoSTING) in subjects with advanced/metastatic, recurrent, injectable solid tumors. ClinicalTrials. Gov.
Wu H, Mu X, Liu L, Wu H, Hu X, Chen L, Liu J, Mu Y, Yuan F, Liu W. Bone marrow mesenchymal stem cells-derived exosomal microRNA-193a reduces cisplatin resistance of non-small cell lung cancer cells via targeting LRRC1. Cell Death Dis. 2020;11(9):801.
Acknowledgements
We thank the personnel of the Faculty of Advanced Medical Sciences for their help and guidance.
Funding
This study is supported by a grant (No: 71399) from Tabriz University of Medical Sciences under the ethical code of IR.TBZMED.VCR.REC.1401.392.
Author information
Authors and Affiliations
Contributions
"Z.A-M., S.G.A., Z.A.S., N.M., and Z.A. collected data, and prepared manuscript. Z.S. and R.R. conceptualized the study. S.Z.M. drew the illustrations. R.R. supervised the study".
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Abbasi-Malati, Z., Azizi, S.G., Milani, S.Z. et al. Tumorigenic and tumoricidal properties of exosomes in cancers; a forward look. Cell Commun Signal 22, 130 (2024). https://doi.org/10.1186/s12964-024-01510-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12964-024-01510-3