Abstract
Inhibitor of apoptosis (IAP) family comprises a group of endogenous proteins that function as main regulators of caspase activity and cell death. They are considered the main culprits in evasion of apoptosis, which is a fundamental hallmark of carcinogenesis. Overexpression of IAP proteins has been documented in various solid and hematological malignancies, rendering them resistant to standard chemotherapeutics and radiation therapy and conferring poor prognosis. This observation has urged their exploitation as therapeutic targets in cancer with promising pre-clinical outcomes. This review describes the structural and functional features of IAP proteins to elucidate the mechanism of their anti-apoptotic activity. We also provide an update on patterns of IAP expression in different tumors, their impact on treatment response and prognosis, as well as the emerging investigational drugs targeting them. This aims at shedding the light on the advances in IAP targeting achieved to date, and encourage further development of clinically applicable therapeutic approaches.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Apoptosis, or programmed cell death, is an essential cell process in homeostasis of multicellular organisms. Strict regulation of apoptosis has been involved in many human diseases, including cancer [1]. The inhibitor of apoptosis (IAP) proteins are a class of apoptosis regulators, that perform a critical role in the control of survival and cell death by regulating crucial factors in signaling events such as caspase activation and NF-κB signaling [1]. Targeting critical apoptosis regulators, like IAPs, is an attractive therapeutic way undertaken for the development of new classes of therapies for cancer. Overexpression of IAPs has been repeatedly encountered in various cancer cells, and is hypothesized to be associated with tumorigenesis, treatment resistance, and poor prognosis. These features render IAPs promising therapeutic targets in a wide range of human tumors by either direct induction of cell death or reduction of the threshold for cell death caused by anticancer drugs [2].
IAP family members
The IAP gene was first recognized in insect SF-21 cells infected by baculovirus, and was identified as a potent inhibitor of apoptosis in insect cells [3]. IAP homologs, sharing similar structure, were subsequently discovered in various species including nematode, yeasts, and mammals [4]. The first cellular IAP to be identified was the mammalian gene neuronal apoptosis inhibitory protein (NAIP) [5]. Since the discovery of NAIP, the human IAP gene family has expanded to include seven more members: cellular IAP1; cellular IAP2; X-linked inhibitor of apoptosis (XIAP) [6,7,8]; IAP-like protein 2 [9, 10]; BIR-containing ubiquitin conjugating enzyme (BRUCE/Apollon) [11]; Survivin [12]; and Livin (ML-IAP) [13, 14]. Among IAP family members, XIAP is the only one that can inhibit caspases through direct physical interaction. Cellular IAPs (cIAP1 and cIAP2) have similar roles as XIAP in regulating caspases activity, but are also involved in regulating NF-κB pathways [15]. The mechanism by which other IAP family members inhibit apoptosis is less understood. Several IAPs are capable of binding to caspases, yet lack the ability to directly inhibit the proteolytic activity of those enzymes [16].
Structural features of IAPs
The IAP family is characterized by the presence of Baculovirus IAP Repeat (BIR) at the N-terminal end of the protein, which constitutes of one to three tandem specific motifs of approximately 70 amino acids. The BIRs have a core component of cysteine-histidine (Cys-His) motif that coordinates a zinc ion [17]. The structure is organized in a series of short alpha-helices with intervening β-sheets, yielding a specific fold stabilized by Zinc tetrahedrally coordinated by three cysteine and one histidine residues [17]. BIRs are protein interacting modules with distinct binding properties, necessary for the anti-apoptotic activity of most IAPs [18]. Three subtypes of BIR domain, BIR1, BIR2, and BIR3, have been identified so far [19]. Most BIRs form a hydrophobic groove which binds conserved IAP binding motifs (IBMs), located in the extreme N-terminus of some caspases and IAP antagonists. The N-terminal exposure of IBM is essential for the recognition and binding by IAPs. Thus, only processed, activated caspases can bind to the BIR hydrophobic groove [17]. There are numerous proteins that bind to BIR in an IBM-dependent fashion, including caspases [16], the second mitochondrial activator of caspase (SMAC—also known as DIABLO) [20], HtrA2 (also known as Omi) [21], and the Drosophila proteins Hid, Grim and Reaper [22]. Different IAP family members possess specific intrinsic binding selectivity, which explains why subtle changes in the peptide-binding groove of BIR domains can markedly alter the target protein selectivity [18]. As opposed to BIR2, type I BIRs do not possess the binding properties with IBM, but can interact with a different set of proteins primarily involved in cell signaling pathways [23, 24].
At the C-terminal, the second conserved motif of IAP subsists, namely the really interesting new gene (RING) which displays a characteristic E3-ubiquitin ligase activity. It also enables homo- or hetero-dimerization of IAP proteins, which is crucial for their stability and possibly their activity [25]. For example, the RING domain is required for cIAP1 and 2 homodimerization, autoubiquitylation and subsequent proteasomal degradation [25, 26]. It is also noteworthy that cIAP1 exists in an inactive state due to the interaction between its BIR3 and RING domains which precludes intermolecular RING–RING dimerization [26, 27]. Other conserved protein domains are also found in some IAPs including caspase activation recruitment domain (CARD) which regulates E3-ubiquitin ligase activity [28]. The ubiquitin-associated domain (UBA) recognizes mono- and polyubiquitin chains allowing the recruitment of IAP in protein complexes [29, 30]. All IAPs are homologs sharing remarkably conserved sequences. The distinct association between baculoviral IAPs and insect IAPs proposes that the former might have been acquired by gene transfer from infected host insect cells. Some baculoviral IAPs can even represses apoptosis in mammals [31].
Regulatory mechanisms of IAP in apoptosis
Apoptosis is considered one of the main mechanisms of programmed cell death, which can be triggered in response to variable endogenous and exogenous stimuli. Key morphological changes occurring during apoptosis include nuclear condensation and fragmentation, as well as blebbing of plasma membranes leading to apoptotic body formation [32]. This process is strictly regulated by a series of signal cascades, under the influence of three critical factors: IAP, IAP antagonists, and caspases [33]. The regulation of these factors is crucial for cellular homeostasis, and their disruption is noted in many diseases, including cancer [34].
Apoptotic pathways
In mammals, apoptosis is mediated by a sequential activation cascade of cysteine proteases (caspases) that are responsible for distinct biochemical and morphological changes [35]. Caspases are inactive-zymogens constituting of one pro-domain and two active subunits. According to the length of the pro-domain and the activation mechanism, they are further sub-divided into initiator and effector caspases. The initiator caspases are characterized by the presence of a long pro-domain that allows their recruitment into caspase-activating complexes. In mammals, there are four apoptotic initiator caspases (caspase-2, -8, -9 and -10) [36, 37]. The effector caspases-3 and -7 are activated by proteolytic cleavage leading to the assembly of two large and two small subunits into a single active tetramer. They can cleave a wide spectrum of cellular proteins leading to loss of cellular integrity [17]. Caspases can also mediate other non-apoptotic processes, such as cellular proliferation and inflammatory response [35].
It has been documented that caspases are activated through several overlapping pathways for apoptosis initiation: (1) the mitochondrial pathway (intrinsic pathway) in which cytochrome c is released from the mitochondria and apoptosomes are generated activating caspase-9 and in turn caspase-3; (2) the death receptor pathway (extrinsic pathway), activated by the ligand binding of extracellular signals and death receptors on cell membrane [FasL (Fas ligand)/Fas, tumor necrosis factor (TNF)/TNF receptor] (Fig. 1); (3) the endoplasmic reticulum (ER) stress-induced apoptotic pathway that activates caspase-2 and caspase-9; and (4) the apoptosis-inducing protease, granzyme B, mediated activation of effector caspases, specifically in cytotoxic T lymphocytes and natural killer cells [19]. (5) A nuclear pathway that depends on specific nuclear organelles, named Pml oncogenic domains (PODs) or nuclear bodies (NBs) was proposed. The mechanism of caspase activation in this pathway is not clearly understood. Several apoptosis-promoting proteins have been localized to PODs, and their defects have been linked to tumorigenesis [38]. Given the dire consequences of caspase activation, strict regulation of these pathways at each step is of paramount importance.
Schematic diagram of the extrinsic (death receptor) and intrinsic (mitochondrial) apoptotic pathways. The first step in initiation of the extrinsic pathway is the binding of death ligands to their respective receptors on the plasma membrane: tumor necrosis factor (TNF) with TNF receptor (TNFR) and Fas ligand (FasL) with Fas receptor (FasR). This is followed by the binding of TNF receptor-associated death domain (TRADD) and/or Fas-associated death domain protein (FADD) to the intracellular domains of death receptors. These reactions result in the formation of death-inducing signaling complex (DISC) which promote the activation of pro-caspases 8 and 10. Once they become in the active state, they either activate the executioner caspases-3 and -7 resulting in apoptosis, or converge onto the intrinsic pathway via BID activation (mitochondrial amplification loop). On the other hand, the intrinsic pathway is initiated in response to apoptotic stimuli which activate the pro-apoptotic BCL2 family members: BH3-only proteins. Bax and/or BAK are consequently activated and induce mitochondrial outer membrane permeabilization (MOMP). Several proteins are released from the mitochondria, including second mitochondria-derived activator of caspases/direct IAP-binding protein with low pI (Smac/Diablo), Omi stress-regulated endoprotease/High temperature requirement protein A2 (Omi/HtrA2) and cytochrome c. The latter, together with apoptotic protease activating factor 1 (Apaf-1) and pro-caspase 9, form the apoptosome. The latter induces the activation of caspase 9 and subsequent activation of caspase-3 and 7, which eventually lead to apoptosis
The intrinsic pathway is largely regulated by BCL2 family, which comprises several anti- and pro-apoptotic proteins. Anti-apoptotic proteins (eg: BCL2 and BCLXL) share a structural homology in specific domains, namely BCL2 homology (BH) 1, 2, 3 and 4. On the other hand, some pro-apoptotic proteins share only BH3 domain homology and thus named BH3-only proteins, including PUMA, NOXA, BIM, BAD and BIK. The effector pro-apoptotic proteins, Bcl2- associated X protein (BAX) and Bcl2 homologous antagonist/killer (BAK) share multi-domain homology (BH1, 2 and 3). Apoptotic stimuli result in an imbalance between pro- and anti-apoptotic proteins, which consequently activates the effector BAX and BAK proteins [39].
Upon activation, BAX translocates from the cytosol to be integrated in the mitochondrial outer membrane. Together with BAK, a membrane-resident protein, they become fully inserted in the mitochondrial membrane leading to mitochondrial outer membrane permeabilization (MOMP) [40]. This eventually leads to supramolecular channels releasing several proteins from the mitochondrial inter-membrane space (IMS), the most important of which is cytochrome c [41, 42]. Other released proteins include the IAP antagonists Smac/Diablo (second mitochondria-derived activator of caspases/direct IAP-binding protein with low pI) [43] and Omi/HtrA2 (Omi stress-regulated endoprotease/High temperature requirement protein A2) [44]. Cytochrome c initiates apoptosome formation through binding the apoptotic protease activating factor 1 (Apaf-1), triggering its oligomerization into a wheel-like heptamer and exposing its caspase activation and recruitment domains (CARDs) [45]. The latter bind to procaspase-9 CARDs inducing autocatalysis, and active caspase 9 consequently activates caspases 3 and 7, executing cell death within minutes [46].
Notably, MOMP eventually results in energy depletion and cell death caused by progressive mitochondrial dysfunction, even in the presence of inactive caspases [40, 41]. Moreover, the mitochondrial pathway seems to be crucial for amplification of upstream signals of the extrinsic apoptotic pathway, highlighting the importance of MOMP in apoptosis execution [40]. Several other pro-apoptotic proteins, including p53-upregulated modulator of apoptosis (PUMA) and NOXA, can also be activated in response to DNA damage. The latter can alternatively lead to p53-dependent caspase 2 activation, which in turn induces MOMP. The mitochondrial proteins released, including endonuclease G (EndoG) and apoptosis-inducing factor (AIF), are capable of initiating caspase-independent apoptosis. This process can also be induced through lysosomal membrane permeabilization that releases MOMP-triggering cathepsins into the cytosol [32].
In the extrinsic pathway, caspases-8 and -10 are activated in response to death receptor signals from tumor necrosis factor (TNF) receptor superfamily. Death ligands are mainly produced by the immune system (eg: T-cells, macrophages, natural killer cells, and dendritic cells) and include TNF, Fas ligand and TNF-related apoptosis-inducing ligand (TRAIL) [47]. The TNF receptor superfamily is characterized by distinct protein motifs, namely death domains (DD) and death effector domains (DED), which are capable of monovalent interactions and pivotal for apoptotic signal transduction [46]. Stimulation of Fas (DR2, CD95) or TRAIL Receptor I or II (DR4 and DR5) induces the recruitment of DD-containing molecules, FADD (Fas-associated death domain protein) and/or TNF receptor-associated death domain (TRADD). FADD triggers pro-apoptotic pathways by activating pro-caspases-8 and -10 in a receptor-associated platform called DISC (death-inducing signalling complex) [17, 37]. Once those caspases are activated, they amplify death signaling, by either direct activation of effector caspases-3, -6 and -7, or engaging the intrinsic apoptotic pathway [46]. This convergence is mediated by caspase-8 that triggers cleavage of the pro-apoptotic BH3 family member BID. This results in its activation and subsequent involvement of BAX and BAX which eventually induce MOMP, in a mitochondrial amplification loop [47]. In contrast, TRADD initiates anti-apoptotic signals via forming complex I with receptor interacting protein-1 (RIP1), TNF receptor-associated factors 2 and 5 (TRAF2 and TRAF5), and the inhibitor of apoptosis protein-1 and - 2 (cIAP1/2). This complex promotes survival signaling, such as those regulated by NF-κB [46, 48, 49].
The endoplasmic reticulum (ER) majorly contributes to both mitochondrial and ER stress-induced apoptotic pathways [50,51,52]. It has been proven that ER stress induces down-regulation of the anti-apoptotic Bcl2, up-regulation of the pro-apoptotic BIM and PUMA, as well as BAX activation with subsequent apoptosis execution [53]. Some studies also showed a correlation between ER stress and regulation of the tumor suppressor p53. The latter is stabilized in response to ER stress, promoting transcriptional activation of pro-apoptotic PUMA and NOXA [54]. This pathway is mainly mediated by caspases-2 and -9 which in turn activate the executioner caspases-3 and -7 leading to cell death [19, 55].
Mechanism of action of IAP
IAPs are considered to be the only known endogenous proteins that are capable of suppressing both initiator and effector caspases, the key executioners of apoptosis [56]. They impose negative regulation on apoptotic pathways by direct inhibition of caspases through several mechanisms. First, their conserved BIR domains bind the active site of caspases inhibiting their proteolytic function, as do XIAP, cAP1/2 and Survivin to caspases-3 and -7 [57]. This results in stimulating the breakdown of active caspases, or their isolation away from their substrates [57]. Also, IAPs repress caspases-2 and -9 in the ER stress-induced apoptotic pathway through BIR domain binding [55]. Second, direct inhibition of pro-caspase 9 activation by XIAP. Third, some IAP family members are capable of targeting effector caspases for ubiquitination and proteasomal degradation. Additionally, cIAPs may play some role in the activation of anti-apoptotic signals, such as NF-κB, which explains their pivotal role in regulating NF-κB during TNF signaling [58].
While different IAPs can suppress caspases-2, -3, -7 and -9, other caspases, such as -1, -6, -8 and -10, are thought to be resistant to IAP inhibition. IAPs do not bind caspase-8 but rather inhibit its substrate, namely caspase-3 [19]. Caspase-8 can also be negatively regulated through the induction of survival signaling pathways that in turn inhibit its activation. The BIR domain, CARD and RING E3 ligases in cIAP1/2 act to recruit TRAF1 and 2 and inhibit TNFα-apoptotic signaling. Thus, cIAP1/2 have the potential of inhibiting caspase-8 by inducing pro-survival signals, mainly NF-κB pathway [46, 59, 60]. Moreover, cIAPs are capable of interacting with caspases-9 and -7 in an IBM-dependent fashion, and with the pro-domain of caspase-3 independently of IBM [61]. It has been observed that neither BIR2 nor BIR3 domains of cellular IAPs can directly inhibit caspases; so they execute their anti-apoptotic function through caspase binding, with a lower affinity than XIAP [19]. Of note, cellular IAP 2 is the only IAP family member that is capable of binding and inhibiting caspase-2, through its BIR2 domain [55]. cIAPs can also control the stability of activated caspases through a UPS (ubiquitin/proteasome system)-dependent mechanism [61]. Furthermore, cellular IAPs, as well as Melanoma IAP (ML-IAP/Livin) and Apollon, have the ability of binding to SMAC, to prevent XIAP neutralization [62]. In addition to SMAC binding and degradation, Livin can also exert its anti-apoptotic activity by inhibiting caspases-3, -7 and -9 [63]. On the other hand, neuronal apoptosis inhibitory protein (NAIP) can distinctly interact with pro-caspase 9 inhibiting its cleavage when present in the apoptosome complex. This mechanism is ATP-dependent and IBM-independent, resulting in early inhibition of the intrinsic pathway [64]. The anti-apoptotic functions of NAIP have been demonstrated both in vivo and in vitro, and extend to include inhibitory action on caspases-3 and -7 as well [19].
XIAP, the best identified IAP so far, is regarded as the most powerful caspase inhibitor. It possesses three BIR domains (BIR1, BIR2, and BIR3) of high affinity and varying functions to caspases [16]. BIR2 domain binds the executioner caspases-3 and -7, while BIR3 binds initiator caspase 9 [16]. The interaction with effector caspases is believed to be via steric hindrance, where BIR2 domain blocks the substrate entry site [65]. On the other hand, the interaction between BIR3 and the Apaf-1/caspase 9 complex occurs via sequestration of the N-terminus of caspase-9 small subunit [19]. The latter distinctly resembles the N-terminus of mitochondrial SMAC/Diablo, raising the suspicion that both compete for XIAP-BIR3 binding [19]. XIAP BIR3 domain, together with caspase 9, form a heterodimer which results in stabilization of inactive caspase 9 by preventing its homodimerization and subsequent autocatalytic activity [66]. It has been shown that the capacity of XIAP to control capase-9 activity is directly correlated to the level of APAF-1 and apoptosome activity [67]. Thus, in cells harbouring low Apaf-1 levels, such as neuronal cells and cardiac myocytes, XIAP is an effective regulator of response to apoptotic stimuli [68]. Furthermore, the E3 ligase activity of the XIAP RING domain plays an important role in caspase inhibition. Surprisingly, neither BIR2, BIR3 nor RING domains alone is capable of caspase inhibition [19]. Of note, XIAP is cleaved in response to Fas-induced apoptosis into two separate fragments; one contains both BIR1 and 2 domains while the other consists of BIR3 and RING domains [69]. How these fragments contribute to the apoptotic process is still unclear [65].
Several studies have documented the ability of Survivin to inhibit both intrinsic and extrinsic apoptotic pathways [12, 70]. The exact mechanism of Survivin-mediated caspase inhibition is not yet fully understood, as its capability of binding caspases-3 and -9 is still controversial [70]. The inhibitory action of Survivin on apoptosis is mainly mediated by co-operative interactions with other apoptotic regulators in vivo. One example is the interaction between Survivin and XIAP BIR 1 and three domains, expanding its functional repertoire [71]. Survivin also exerts an indirect action through hepatitis B X-interacting protein (HBXIP) that binds pro-caspase 9 [70], and through triggering XIAP inhibitory effect on caspases-3 and -9 [72]. Another mechanism is Survivin binding with the pro-apoptotic Smac/Diablo, hindering caspase activation [70]. Notably, it also plays a pivotal role in regulating chromosomal segregation during cell division [73]. These two vital cellular functions are mediated by the characteristic structure of Survivin, where the C-terminal is involved in cell division and the N-terminal is responsible for regulating apoptosis [74].
Another mechanism of caspase inhibition by IAP proteins is through the ubiquitination process. This regulated process entails post-translational protein modification, where ubiquitin is covalently bonded to lysine on a substrate protein [75]. Through the interplay of ubiquitin activating enzyme (E1), ubiquitin conjugating enzyme (E2), and ubiquitin protein ligase (E3), ubiquitins attach to target proteins and undergo proteasome-mediated degradation [19, 76], as illustrated in Fig. 2. A fine balance between ubiquitination and auto-ubiquitination is one way of IAP regulation. The loss of one IAP protein has been noted to cause an increase in the levels of other IAP family members [77]. The RING domain of cIAP1 has been proven to play an important role in the degradation of RING-containing IAPs, as well as being involved in XIAP binding and degradation [78]. Moreover, the E3 ligase activity of cIAP2 mediates both auto-ubiquitination and mono-ubiquitination of caspases-3 and -7 [79]. These different regulatory mechanisms act to control the endogenous IAP levels and activities, promoting apoptosis.
Illustrative diagram of ubiquitination enzymatic reactions. The first step in this cascade of events is the ATP-dependent activation of ubiquitin by E1 via formation of a thioester bond. Ubiquitin then binds to E2 conjugating enzyme, followed by subsequent isopeptide bond formation between the carboxyl-terminal glycine in ubiquitin and lysine residue in a substrate protein, via the action of E3 ligase. This process eventually results in proteasomal-mediated substrate degradation
All BIR-containing proteins, with the exception of NAIP, are associated with ubiquitin. In addition, the RING-containing IAPs, including XIAP, cellular IAPs 1/2, and ML-IAP act as ubiquitin E3 ligases [80]. Huang et al. demonstrated that both full-length cellular IAP 2, and its RING domain alone, possess the capacity of acting as an E3 ligase in vitro [79]. It has also been demonstrated that in-vivo removal of the RING domain results in XIAP protein stabilization, increase in caspase 3 activity and TNF sensitivity [81]. Similar to cIAP1/2, the XIAP-RING domain can conjugate various ubiquitin chain types to target proteins [82, 83]. XIAP also has the ability to induce caspase 3 ubiquitination by itself [80]. This demonstrates the role of ubiquitination of XIAP-bound caspases in the inhibition of apoptosis. BRUCE/Apollon is a distinct IAP with dual function owing to the possession of two specific domains [11]. At the N-terminal, the BIR domain mediates anti-apoptotic functions by antagonizing Smac/Diablo and multiple caspases. On the other hand, the C-terminal ubiquitin conjugating (UBC) domain mediates E2 ubiquitination activities. BRUCE has been discovered to be a hybrid E2/E3 enzyme owing to the dual function of its UBC domain as both conjugating enzyme (E2) and protein ligase (E3) [84].
IAP antagonists
IAP activities are strictly regulated through several feedback mechanisms that involve pro-apoptotic proteins. Numerous mammalian IAP antagonists have been identified, including second mitochondrial activator of caspases/direct IAP binding protein with low pI (Smac/Diablo) [85], high temperature-regulated A2/Omi (HtrA2/Omi) [86], X-linked IAP associated factor 1 (XAF1) [87], the endoplasmic reticulum protein, GSPT1/eRF3 (G1 to S phase transition protein/eukaryotic Release Factor 3) [88], the septin-like mitochondrial protein ARTS [89, 90], glutamate dehydrogenase, Nipsnap 3 and 4, and 3-hydroxyisobutyrate dehydrogenase [91]. The functional execution of IAP binding proteins entails their physical interaction with IAPs through a conserved IAP Binding Motif (IBM). The latter specifically binds IAP BIR domain promoting apoptosis partly by replacing the bound caspases [20]. The whole spectrum of actions of IAP antagonists and their role in IAP regulation are not yet fully understood. For example, various apoptotic triggers, such as DNA damaging agents, seem to induce IAP degradation and possibly RIPoptosome assembly without a clear role for IAP antagonists [17, 92].
Smac/Diablo and HtrA2 are perhaps the most studied mammalian IAP antagonists [21, 43, 44, 85, 86]. During apoptosis, they are released into the cytosol and activated into arc-shaped dimmers and pyramid-shaped homotrimers, respectively [19, 41]. Smac/Diablo has a characteristic ability of binding both BIR2 and BIR3 domains halting XIAP-mediated caspase inhibition, namely caspases-9 and -3 [85]. It can also inhibit cellular IAPs 1/2, and induce their degradation through auto-ubiquitination [30, 93]. Similar to Smac, its isoform Smac3 can also inhibit XIAP by binding with BIR2 and BIR3 domains. Smac3, produced as a result of exon 4 splicing, stimulates XIAP ubiquitination and destruction [94]. HtrA2/Omi exerts its pro-apoptotic function via binding XIAP, thus releasing its inhibitory effect on caspases, as well as irreversible proteolytic cleavage of XIAP and cellular IAPs [21]. However, it has a weaker affinity than Smac as regards XIAP BIR3 binding [19]. Notably, the tumor suppressor p53 upregulates and activates HtrA2, which is one of the mechanisms by which p53 promotes apoptosis and supresses carcinogenesis [95].
XIAP-associated factor (XAF1) is a tumor suppressor gene that acts as an IAP antagonist, sequestrating XIAP in the nucleus and counteracting its anti-caspase activity [87]. Arora et al. demonstrated the ability of XAF1 to directly bind all IAP members, with the exception of Survivin which is indirectly inhibited through XIAP-Survivin complex [96]. Also, the destruction of Survivin is regulated by XAF1 through activating the E3 activity of XIAP RING domain [96]. Another proposed mechanism of XAF1 action is through promoting IFN-mediated sensitization to TRAIL in tumors [97]. Several studies have shown that XAF1 is expressed in normal tissues, but is nearly undetectable in cancer cell lines and its suppression in several tumors has been confirmed [98,99,100,101]. Moreover, the ratio of expression levels of both XIAP and XAF1 seems to be crucial for determination of cell fate. Carcinogenesis is favoured when XIAP is overexpressed with respect to XAF1, evading apoptotic pathways [102]. Interestingly, a regulatory relationship has been established between the tumor suppressor genes XAF1 and p53 [103]. Wild type p53 exerts a negative feedback and transcriptional repression of XAF1, which is probably a mechanism to avoid duplication of function. On the other hand, XAF1 induces p53 phosphorylation in response to DNA damage, leading to its nuclear accumulation and enhanced transcriptional activity. These findings illustrate the function of XAF1 as a promoter of p53-mediated apoptosis in cancer [103]. Hence, induction of XAF1 expression could be exploited in cancer therapy, especially in cancers having low expression levels of wild type p53 [99, 103, 104].
Another identified IAP antagonist is the endoplasmic reticulum protein, GSPT1/eRF3 (G1 to S phase transition protein/eukaryotic Release Factor 3). During apoptosis, its IBM is exposed and selectively mediates cellular IAP1 auto-ubiquitination and degradation [88]. On the other hand, the septin-like mitochondrial protein, ARTS, is a peculiar IAP antagonist that lacks the characteristic IAP binding motif [89]. In spite of that, it can bind XIAP and induce its ubiquitination [105, 106]. An increased incidence of lymphomas and leukemias has been observed with ARTS inactivation in mice, which can be reversed by XIAP inactivation. This phenomenon highlights the key role of ARTS-mediated XIAP inhibition in maintaining normal hematopoiesis and tumor suppression [17, 107]. Edison et al. have concluded that ARTS is capable of activating caspases upstream of MOMP. It is localized in the outer mitochondrial membrane and is rapidly translocated to the cytoplasm upon induction of apoptosis, in a caspase-independent manner. This leads to XIAP binding and inhibition prior to the release of cytochrome c and Smac [90]. Furthermore, it has been shown that ARTS knockdown precludes the release of cytochrome c from the mitochondria, suggesting that ARTS is pivotal for the regulation of mitochondrial proteins release in response to MOMP [90]. In addition to well characterized IAP binding proteins, various other proteins have the capacity of antagonizing IAP actions. For example, interleukins-3 and -5 as well as granulocyte-macrophage colony stimulating factor (GM-CSF) have been shown to regulate cIAP2 and Survivin [108]. Some mitochondrial proteins, including glutamate dehydrogenase, can also inhibit XIAP via BIR2 binding [91]. However, their detailed mechanism of action and regulatory process in cancer are yet to be fully understood.
Clinical applications of IAP
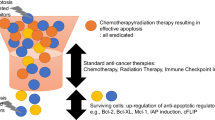
Evasion of apoptosis is one of the fundamental hallmarks of carcinogenesis. Cancer cells are known to enhance survival and proliferation by overexpressing anti-apoptotic and inactivating pro-apoptotic proteins [46]. Members of IAP and tumor necrosis factor families are known to promote cancer cell survival synergistically. For instance, TNFα can increase the expression levels of XIAP and cellular IAPs in cancer cell lines [109]. On the contrary, IAP antagonists induce degradation of various IAPs, and stimulate NF-κB that in turn activates TNFα-mediated apoptosis [93, 110]. Besides apoptosis, IAPs are capable of regulating various other processes that are known to be culprits in carcinogenesis. These include cell cycle regulation, cancer-mediated inflammation, tumor invasion and metastasis [59, 111].
IAP overexpression has been documented in various malignancies (Table 1), possibly rendering them resistant to standard chemotherapeutics and radiation therapy. In prostate cancer, adverse clinic-pathological features seem to be correlated with IAPs, including cellular IAPs 1/2 and tumor stage, cIAP2 and positive surgical margins, as well as survivin and perineural invasion [112].
Survivin, normally limited to embryonic tissues, is correlated with treatment resistance and increased incidence of relapse when overexpressed in tumor tissues [12, 202]. It is of particular importance in diagnosis and prognostication of gastric and colorectal cancers, a finding that has been consistent in several studies [203, 204]. In addition to evasion of apoptosis and induction of tumor proliferation [202], Survivin also promotes angiogenesis. It is known to upregulate vascular endothelial growth factor (VEGF) and enhance proliferation of vascular endothelial cells [205]. Under normal physiological condition, Survivin plays an important role in regulation of stem cell homeostasis, in intestinal, hematopoietic and nervous systems [206,207,208]. This drives the speculation that Survivin may be involved in cancer stem cell regulation as well [70].
XIAP overexpression has been identified as a well characterized prognostic factor in various malignancies. In pediatric acute myeloid leukemia (AML), XIAP confers poor response to induction therapy, short relapse-free survival as well as intermediate and poor cytogenetics [209, 210]. In adult AML, as well, XIAP is associated with poor cytogenetics, monocytic differentiation and short overall survival [211]. Moreover, XIAP expression in pediatric acute lymphoid leukemia (ALL) heralds resistance to glucocorticoid-mediated apoptosis, an established poor prognostic factor [187]. As for solid malignancies, XIAP is identified as a biomarker of poor survival, chemo-resistance and metastatic potential in ovarian and hepatocellular carcinomas, respectively [142, 212]. In breast cancer, nuclear overexpression of XIAP was identified as an independent prognostic factor, harbouring threefold increased risk of disease-specific death [122].
Cellular IAPs 1 and 2 have been characterized in pancreatic neoplasms conferring short overall survival [174]. Endo et al. revealed a preferential overexpression of cIAP2 and Survivin in elderly colon cancer patients, compared with younger ones [213]. This observation suggests dysregulation of apoptosis in the elderly population contributing to increased incidence of tumors. It is hypothesized that cellular IAPs adversely impact prognosis of head and neck cancer patients. Tanimoto et al. stated that nuclear overexpression of cIAP1 was associated with advanced disease stage, lymph node involvement and poor prognosis in head and neck cancers [145]. In a more recent study in oral squamous cell carcinomas, cIAP2 overexpression was linked to advanced disease stage, but had no impact on survival [214]. Bladder carcinogenesis is also affected by expression levels of cIAP1, where its nuclear overexpression correlates with muscle invasive disease, tumor grade, short recurrence-free and overall survival [115].
Promising therapeutic targets
In cancer, any defect along the apoptotic pathways may offer an interesting therapeutic target. The pivotal role of IAPs in development and progression of cancers compelled their targeting as a promising strategy of cancer treatment. Drugs that can restore the apoptotic signaling pathways towards normality have the potential to eliminate cancer cells which depend on these defects for survival. Many recent and important discoveries have opened new doors into potential new classes of anticancer drugs. To date, several studies investigated different IAP inhibiting agents, achieving a breakthrough in cancer treatment:
IAPs
Novel therapy targeting inhibitor of apoptosis proteins include antisense strategies that are capable of reducing IAP mRNA, short interfering RNA (siRNA) molecules, and Smac mimetics [65, 215]. The latter are synthetic small molecules that mimic the action of endogenous Smac, antagonize IAP actions and induce apoptosis [215]. Using the antisense approach, inhibition of XIAP has been reported to improve tumor control by radiotherapy and chemotherapy [216]. Moreover, when used together with anticancer drugs, XIAP antisense oligonucleotides have been recognized to exhibit enhanced chemotherapeutic activity [217]. On the other hand, some researchers reported that siRNA targeting of XIAP increased radiation sensitivity of human cancer cells, especially in the presence of p53 mutation [218]. Others reported that targeting XIAP or Survivin by siRNAs sensitize hepatoma cells to death receptor- and chemotherapeutic agent-induced cell death [219]. However, when AEG 35156 -a second generation antisense oligonucleotide- was tested in several clinical trials, it yielded contradictory results as shown in Table 2.
Another approach of targeting IAP is the concomitant use of Smac mimetics with chemotherapeutics, which has been proven to induce cancer cell apoptosis in various tumor types [118, 224,225,226,227,228,229]. In pancreatic tumors, apoptosis can be initiated by the combined effect of Smac mimetic with gemcitabine chemotherapy. This process is mediated by NF-κB resulting in caspase activation and subsequent cell death [226]. Reversal of TRAIL resistance is another mechanism by which Smac mimetics exert their pro-apoptotic function [225, 230]. This results in cleavage and activation of procaspases -3 and -7 which mediate apoptotic cell death [225]. Servida et al. have proven that Smac mimetics sensitize leukemic cells to cytotoxicity of chemotherapy and biological agents augmenting TRAIL [227]. In a preclinical animal model, concomitant administration of Smac mimetic with combination chemotherapy, resembling ALL induction, was tested. Significant reduction in tumor load and prolonged survival were observed with combination treatment [231]. The synergy between Smac mimetics and chemotherapy is regulated by RIP1, the inhibition of which by Necrostatin-1 results in inhibition of caspases [228]. In addition, Smac mimetics have been proven to sensitize tumor cells to radiotherapy as well [118]. This approach is of specific interest in malignant gliomas that are usually resistant to standard treatment. The addition of Smac mimetics to radiotherapy and temozolomide can offer a favourable therapeutic ratio in brain gliomas [118, 229]. Synergy between Smac mimetics and oncolytic viruses has also been described [232]. When the Smac mimetic LCL161 was combined with oncolytic rhabdovirus vesicular stomatitis virus, they exerted a synergistic bystander cell death in tumor cells. Moreover, when tested in vivo, they also induced significant tumor regression and durable response [232]. Notably, combination of Smac mimetics with standard cancer therapy seems crucial for tumor cytotoxicity, as the former is ineffective when used as single agent [230]. Several Smac mimetics have been tested in clinical trials, with promising outcomes (Table 3).
A natural small molecule XIAP inhibitor, namely Embelin, was discovered to block the binding of caspase 9, but not caspase 3, to XIAP BIR3 domain [246]. Its anti-cancerous activity is also partially mediated by PTEN-dependent suppression of the oncogenic STAT3 pathway [247]. Embelin has been proven to be of therapeutic value in non-small cell lung cancer (NSCLC), as it reverses XIAP-mediated cisplatin resistance [162]. It can also potentiate fluorouracil cytotoxicity in gastric carcinoma, leading to reduced tumor viability [139]. Moreover, Embelin has been shown to sensitize prostate cancer cells to radiation therapy both in vitro and in vivo [248]. In vitro, it induced cell cycle arrest in the S-phase, inhibiting tumor proliferation and inducing apoptosis in a caspase-independent manner. In vivo, it suppressed angiogenesis, delayed tumor progression and improved survival in combination with radiotherapy [248].
To date, several other XIAP inhibitors have been identified to exert anti-tumor activity. For example, XIAP antagonist compound (XAC) 1396-11, has shown considerable activity in NSCLC in combination with vinerolbine chemotherapy [224]. An adamantane thiadiazole derivative, ATD-4, was recently characterized for its binding affinity to XIAP-BIR3 domain. It has been shown to stimulate mitochondrial apoptotic pathway in lung carcinoma cell lines, exerting anti-tumor activity [249]. Two cyclopeptidic Smac mimetics were found to bind to XIAP and cIAP-1/2, restoring the activities of caspases-9 and -3/-7 [250]. On the other hand, SM-164, a non-peptidic IAP inhibitor was reported to strongly enhance TRAIL activity by concurrently targeting XIAP and cIAP1 [251]. Interestingly, Mitsuuchi et al. have recently proven bivalent IAP antagonists to be superior to monovalent compounds in inhibiting TNF-mediated NF-κB [252]. They demonstrated high levels of residual TRAF2-associated cIAP1 following monovalent compound treatment. This reflects the lack of formation of cIAP1 E3-ubiquitin ligase complex which is responsible for TRAF2-associated cIAP1 degradation [252]. These findings warrant tailoring the choice of specific IAP antagonist classes according to their biochemical properties, and emphasize that bivalent compounds are the drug of choice for targeting TNF-dependent signaling in cancer.
Survivin is another IAP family member that has been extensively investigated as a therapeutic target in cancer. One example of its targeting is the use of antisense oligonucleotides that were described in human melanoma cells [253]. It was shown that transfection of antisense Survivin into YUSAC-2 and LOX malignant melanoma cells resulted in their spontaneous apoptosis. This approach has also been applied in head and neck squamous cell carcinoma and has been reported to induce apoptosis and sensitize these cells to chemotherapy [254]. In colorectal cancer cells, Survivin antisense oligonucleotides enhanced tumor response to radiotherapy as well [255]. Moreover, it has been found to inhibit growth and proliferation of medullary thyroid carcinoma cells [256]. However, these findings didn’t translate into clinical benefit when antisense oligonucleotide, LY2181308, was investigated in combination with docetaxel in a phase II study involving NSCLC patients [257].
Another approach of Survivin therapeutic targeting entails its downregulation by the use of siRNAs. This mechanism can be exploited to overcome radio-resistance in pancreatic carcinoma and NSCLC [258, 259], as well as enhancing chemotherapy effects in AML [260], HCC [261, 262], bladder [263], ovarian [264], and breast cancers [265]. In addition, several small molecule antagonists of Survivin have shown promising anti-tumor activity [266]. For example, YM-155, which directly binds to Survivin promoter inhibiting its activation, has shown activity in osteosarcoma [267], rhabdomyosarcoma [268], prostate [269], and pancreatic cancers [270]. Another small molecule inhibitor, namely FL 118, has the potential of inhibiting Survivin, XIAP and cIAP2. It has been proven to be more effective than standard chemotherapy in human tumor xenograft models [271]. Clinical trials evaluating these drugs are demonstrated in Table 4. More recently, Survivin gene therapy is being investigated as an alternative method of its targeting. A substitution mutation in Survivin, for example Thr34 to Ala, prevents its phosphorylation and the mutant form acts as a competitive antagonist of the wild form [272].
Caspases
Several drugs have been developed to synthetically activate caspases. For example, Apoptin is a caspase-inducing agent which was initially derived from chicken anaemia virus [283]. It is characterized by selective induction of apoptosis in malignant, but not normal cells, owing to its differential cellular localization. The nuclear aggregation of Apoptin, or its truncated variant, in tumor cells promotes its pro-apoptotic function and tumor cytotoxicity, whereas its cytoplasmic localization in normal cells leads to its degradation [284]. Small molecule caspase activators are peptides which contain the arginin-glycine-aspartate motif. They possess a pro-apoptotic activity by directly inducing auto-activation of procaspase 3. They have also been demonstrated to decrease the activation threshold of caspases, contributing to an increase in drug sensitivity of cancer cells [285]. In addition, caspase gene therapy has been investigated in several studies. For instance, human caspase-3 gene therapy was used with etoposide in an AH130 liver tumor model and was observed to induce extensive apoptosis and tumor regression [286]. Gene transfer of constitutively active caspse-3 into HuH7 human hepatoma cells selectively induced apoptosis in these cells [286]. Also, a recombinant adenovirus carrying immunocaspase-3 has been indicated to exert anticancer effects in hepatocellular carcinoma, both in vitro and in vivo [287].
Conclusion
Inhibitors of apoptosis and their antagonists, along with caspases, are complex key players in apoptosis regulation. Over expression of various IAP family members has been repeatedly documented in solid and hematological malignancies. They are culprits in mediating hallmarks of carcinogenesis, as evasion of apoptosis and sustained proliferation. This heralds their use as therapeutic targets in cancer treatment through different approaches. The promising preclinical data existing to date support the notion that IAPs can be effectively used in combination with standard anti-cancer therapy yielding favourable outcome. However, this mandates further extensive research to validate these data on clinical grounds.
References
Fulda S (2014) Molecular pathways: targeting inhibitor of apoptosis proteins in cancer—from molecular mechanism to therapeutic application. Clin Cancer Res 20(2):289–295
Fulda S, Vucic D (2012) Targeting IAP proteins for therapeutic intervention in cancer. Nat Rev Drug Discov 11(2):109
Crook NE, Clem RJ, Miller LK (1993) An apoptosis-inhibiting baculovirus gene with a zinc finger-like motif. J Virol 67(4):2168–2174
Orme M, Meier P (2009) Inhibitor of apoptosis proteins in drosophila: gatekeepers of death. Apoptosis 14(8):950–960
Roy N, Mahadevan MS, McLean M et al (1995) The gene for neuronal apoptosis inhibitory protein is partially deleted in individuals with spinal muscular atrophy. Cell 80(1):167–178
Duckett CS, Nava VE, Gedrich RW et al (1996) A conserved family of cellular genes related to the baculovirus iap gene and encoding apoptosis inhibitors. EMBO J 15(11):2685
Liston P, Roy N, Tamai K, Lefebvre C (1996) Suppression of apoptosis in mammalian cells by NAIP and a related family of IAP genes. Nature 379(6563):349
Rothe M, Pan MG, Henzel WJ, Ayres TM, Goeddel DV (1995) The TNFR2-TRAF signaling complex contains two novel proteins related to baculoviral inhibitor of apoptosis proteins. Cell 83(7):1243–1252
Lagacé M, Xuan JY, Young SS, McRoberts C, Maier J, Rajcan-Separovic E, Korneluk RG (2001) Genomic organization of the X-linked inhibitor of apoptosis and identification of a novel testis-specific transcript. Genomics 77(3):181–188
Richter BW, Mir SS, Eiben LJ et al (2001) Molecular cloning of ILP-2, a novel member of the inhibitor of apoptosis protein family. Mol Cell Biol 21(13):4292–4301
Chen Z, Naito M, Hori S, Mashima T, Yamori T, Tsuruo T (1999) A human IAP-family gene, apollon, expressed in human brain cancer cells. Biochem Biophys Res Commun 264(3):847–854
Ambrosini G, Adida C, Altieri DC (1997) A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma. Nat Med 3(8):917–921
Kasof GM, Gomes BC (2001) Livin, a novel inhibitor of apoptosis protein family member. J Biol Chem 276(5):3238–3246
Vucic D, Stennicke HR, Pisabarro MT, Salvesen GS, Dixit VM (2000) ML-IAP, a novel inhibitor of apoptosis that is preferentially expressed in human melanomas. Curr Biol 10(21):1359–1366
Vucic D, Dixit VM, Wertz IE (2011) Ubiquitylation in apoptosis: a post-translational modification at the edge of life and death. Nat Rev Mol Cell Biol 12(7):43
Pop C, Salvesen GS (2009) Human caspases: activation, specificity, and regulation. J Biol Chem 284(33):21777–21781
Berthelet J, Dubrez L (2013) Regulation of apoptosis by inhibitors of apoptosis (IAPs). Cells 2(1):163–187
Eckelman BP, Drag M, Snipas SJ, Salvesen GS (2008) The mechanism of peptide-binding specificity of IAP BIR domains. Cell Death Differ 15(5):920
Wei Y, Fan T, Yu M (2008) Inhibitor of apoptosis proteins and apoptosis. Acta Biochim Biophys Sin 40(4):278–288
Cossu F, Milani M, Mastrangelo E et al (2009) Structural basis for bivalent Smac-mimetics recognition in the IAP protein family. J Mol Biol 392(3):630–644
Singh N, D’souza A, Cholleti A, Sastry GM, Bose K (2014) Dual regulatory switch confers tighter control on HtrA2 proteolytic activity. FEBS J 281(10):2456–2470
Bergmann A (2010) The role of ubiquitylation for the control of cell death in Drosophila. Cell Death Differ 17(1):61
Lu M, Lin SC, Huang Y et al (2007) XIAP induces NF-κB activation via the BIR1/TAB1 interaction and BIR1 dimerization. Mol Cell 26(5):689–702
Mace PD, Smits C, Vaux DL, Silke J, Day CL (2010) Asymmetric recruitment of cIAPs by TRAF2. J Mol Biol 400(1):8–15
Mace PD, Linke K, Feltham R et al (2008) Structures of the cIAP2 RING domain reveal conformational changes associated with ubiquitin-conjugating enzyme (E2) recruitment. J Biol Chem 283(46):31633–31640
Feltham R, Bettjeman B, Budhidarmo R et al (2011) Smac mimetics activate the E3 ligase activity of cIAP1 protein by promoting RING domain dimerization. J Biol Chem 286(19):17015–17028
Dueber EC, Schoeffler AJ, Lingel A et al (2011) Antagonists induce a conformational change in cIAP1 that promotes autoubiquitination. Science 334(6054):376–380
Lopez J, John SW, Tenev T et al (2011) CARD-mediated autoinhibition of cIAP1’s E3 ligase activity suppresses cell proliferation and migration. Mol Cell 42(5):569–583
Gyrd-Hansen M, Darding M, Miasari M et al (2008) IAPs contain an evolutionarily conserved ubiquitin-binding domain that regulates NF-κB as well as cell survival and oncogenesis. Nat Cell Biol 10(11):1309–1317
Blankenship JW, Varfolomeev E, Goncharov T et al (2009) Ubiquitin binding modulates IAP antagonist-stimulated proteasomal degradation of c-IAP1 and c-IAP2 1. Biochem J 417(1):149–165
Huang Q, Deveraux QL, Maeda S, Salvesen GS, Stennicke HR, Hammock BD, Reed JC (2000) Evolutionary conservation of apoptosis mechanisms: lepidopteran and baculoviral inhibitor of apoptosis proteins are inhibitors of mammalian caspase-9. Proc Natl Acad Sci 97(4):1427–1432
Marino G, Niso-Santano M, Baehrecke EH, Kroemer G (2014) Self-consumption: the interplay of autophagy and apoptosis. Nat Rev Mol Cell Biol 15(2):81
Wong RS (2011) Apoptosis in cancer: from pathogenesis to treatment. J Exp Clin Cancer Res 30(1):87
Giansanti V, Torriglia A, Scovassi AI (2011) Conversation between apoptosis and autophagy: “Is it your turn or mine?” Apoptosis 16(4):321–333
Chowdhury I, Tharakan B, Bhat GK (2008). Caspases—an update. Comp Biochem Physiol B Biochem Mol Biol 151(1):10–27
Würstle ML, Laussmann MA, Rehm M (2012) The central role of initiator caspase-9 in apoptosis signal transduction and the regulation of its activation and activity on the apoptosome. Exp Cell Res 318(11):1213–1220
Mace PD, Riedl SJ (2010) Molecular cell death platforms and assemblies. Curr Opin Cell Biol 22(6):828–836
Pinton P, Giorgi C, Pandolfi PP (2011) The role of PML in the control of apoptotic cell fate: a new key player at ER–mitochondria sites. Cell Death Differ 18:1450–1456
Hata AN, Engelman JA, Faber AC (2015) The BCL2 family: key mediators of the apoptotic response to targeted anticancer therapeutics. Cancer Discov 5(5):475–487
Volkmann N, Marassi FM, Newmeyer DD, Hanein D (2014) The rheostat in the membrane: BCL-2 family proteins and apoptosis. Cell Death Differ 21(2):206
Tait SW, Green DR (2010) Mitochondria and cell death: outer membrane permeabilization and beyond. Nat Rev Mol Cell Biol 11(9):621
Dewson G, Kratina T, Czabotar P, Day CL, Adams JM, Kluck RM (2009) Bak activation for apoptosis involves oligomerization of dimers via their α6 helices. Mol Cell 36(4):696–703
Flanagan L, Sebastia J, Tuffy LP et al (2010) XIAP impairs Smac release from the mitochondria during apoptosis. Cell Death Dis 1(6), e49
Walle LV, Lamkanfi M, Vandenabeele P (2008) The mitochondrial serine protease HtrA2/Omi: an overview. Cell Death Differ 15(3):453–460
Bratton SB, Salvesen GS (2010) Regulation of the Apaf-1–caspase-9 apoptosome. J Cell Sci 123(19):3209–3214
Koff JL, Ramachandiran S, Bernal-Mizrachi L (2015) A time to kill: targeting apoptosis in cancer. Int J Mol Sci 16(2):2942–2955
Sayers TJ (2011) Targeting the extrinsic apoptosis signaling pathway for cancer therapy. Cancer Immunol Immunother 60(8):1173–1180
Zhang DW, Shao J, Lin J et al (2009) RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 325(5938):332–336
Long JS, Ryan KM (2012) New frontiers in promoting tumour cell death: targeting apoptosis, necroptosis and autophagy. Oncogene 31(49):5045
Puthalakath H, O’Reilly LA, Gunn P et al (2007) ER stress triggers apoptosis by activating BH3-only protein Bim. Cell 129(7):1337–1349
Deniaud A, El Dein OS, Maillier E, Poncet D, Kroemer G, Lemaire C, Brenner C (2008) Endoplasmic reticulum stress induces calcium-dependent permeability transition, mitochondrial outer membrane permeabilization and apoptosis. Oncogene 27(3):285
Hamanaka RB, Bobrovnikova-Marjon E, Ji X, Liebhaber SA, Diehl JA (2009) PERK-dependent regulation of IAP translation during ER stress. Oncogene 28(6):910
Tabas I, Ron D (2011) Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat Cell Biol 13(3):184–190
Moenner M, Pluquet O, Bouchecareilh M, Chevet E (2007) Integrated endoplasmic reticulum stress responses in cancer. Cancer Res 67(22):10631–10634
Cheung HH, Kelly NL, Liston P, Korneluk RG (2006) Involvement of caspase-2 and caspase-9 in endoplasmic reticulum stress-induced apoptosis: a role for the IAPs. Exp Cell Res 312(12):2347–2357
Baig S, Seevasant I, Mohamad J, Mukheem A, Huri HZ, Kamarul T (2016). Potential of apoptotic pathway-targeted cancer therapeutic research: where do we stand?. Cell Death Dis 7(1):e2058
Silke J, Meier P (2013) Inhibitor of apoptosis (IAP) proteins-modulators of cell death and inflammation. Cold Spring Harb Perspect Biol 5(2):a008730
Estornes Y, Bertrand MJ (2015) IAPs, regulators of innate immunity and inflammation. Semin Cell Dev Biol 39:106–114
Gyrd-Hansen M, Meier P (2010) IAPs: from caspase inhibitors to modulators of NF-[kappa] B, inflammation and cancer. Nat Rev Cancer 10(8):561
Kao WP, Yang CY, Su TW, Wang YT, Lo YC, Lin SC (2015) The versatile roles of CARDs in regulating apoptosis, inflammation, and NF-κB signaling. Apoptosis 20(2):174–195
Choi YE, Butterworth M, Malladi S, Duckett CS, Cohen GM, Bratton SB (2009) The E3 ubiquitin ligase cIAP1 binds and ubiquitinates caspase-3 and-7 via unique mechanisms at distinct steps in their processing. J Biol Chem 284(19):12772–12782
Marivin A, Berthelet J, Plenchette S, Dubrez L (2012) The inhibitor of apoptosis (IAPs) in adaptive response to cellular stress. Cells 1(4):711–737
Chang H, Schimmer AD (2007) Livin/melanoma inhibitor of apoptosis protein as a potential therapeutic target for the treatment of malignancy. Mol Cancer Ther 6(1):24–30
Davoodi J, Ghahremani MH, Es-haghi A, Mohammad-gholi A, MacKenzie A (2010) Neuronal apoptosis inhibitory protein, NAIP, is an inhibitor of procaspase-9. Int J Biochem Cell Biol 42(6):958–964
Hunter AM, LaCasse EC, Korneluk RG (2007) The inhibitors of apoptosis (IAPs) as cancer targets. Apoptosis 12(9):1543–1568
Shiozaki EN, Chai J, Rigotti DJ et al (2003) Mechanism of XIAP-mediated inhibition of caspase-9. Mol Cell 11(2):519–527
Ho AT, Li QH, Okada H, Mak TW, Zacksenhaus E (2007) XIAP activity dictates Apaf-1 dependency for caspase 9 activation. Mol Cell Biol 27(16):5673–5685
Tait SW, Green DR (2008) Caspase independent cell death: leaving the set without the final cut. Oncogene 27(50):6452–6461
Deveraux QL, Leo E, Stennicke HR, Welsh K, Salvesen GS, Reed JC (1999) Cleavage of human inhibitor of apoptosis protein XIAP results in fragments with distinct specificities for caspases. EMBO J 18(19):5242–5251
Garg H, Suri P, Gupta JC, Talwar GP, Dubey S (2016) Survivin: a unique target for tumor therapy. Cancer Cell Int 16(1):49
Altieri DC (2010) Survivin and IAP proteins in cell-death mechanisms. Biochem J 430(2):199–205
Dohi T, Xia F, Altieri DC (2007) Compartmentalized phosphorylation of IAP by protein kinase A regulates cytoprotection. Mol Cell 27(1):17–28
van der Horst A, Lens SM (2014) Cell division: control of the chromosomal passenger complex in time and space. Chromosoma 123(1–2):25–42
Wheatley SP (2015) The functional repertoire of survivin’s tails. Cell Cycle 14(2):261–268
Komander D, Rape M (2012) The ubiquitin code. Annu Rev Biochem 81:203–229
Huang X, Dixit VM (2016) Drugging the undruggables: exploring the ubiquitin system for drug development. Cell Res 26(4):484–498
Conze DB, Albert L, Ferrick DA, Goeddel DV, Yeh WC, Mak T, Ashwell JD (2005) Posttranscriptional downregulation of c-IAP2 by the ubiquitin protein ligase c-IAP1 in vivo. Mol Cell Biol 25(8):3348–3356
Cheung HH, Plenchette S, Kern CJ, Mahoney DJ, Korneluk RG (2008) The RING domain of cIAP1 mediates the degradation of RING-bearing inhibitor of apoptosis proteins by distinct pathways. Mol Biol Cell 19(7):2729–2740
Huang HK, Joazeiro CA, Bonfoco E, Kamada S, Leverson JD, Hunter T (2000) The inhibitor of apoptosis, cIAP2, functions as a ubiquitin-protein ligase and promotes in vitro monoubiquitination of caspases 3 and 7. J Biol Chem 275(35):26661–26664
Galbán S, Duckett CS (2010) XIAP as a ubiquitin ligase in cellular signaling. Cell Death Differ 17(1):54–60
Schile AJ, García-Fernández M, Steller H (2008) Regulation of apoptosis by XIAP ubiquitin-ligase activity. Genes Dev 22(16):2256–2266
Damgaard RB, Nachbur U, Yabal M et al (2012) The ubiquitin ligase XIAP recruits LUBAC for NOD2 signaling in inflammation and innate immunity. Mol Cell 46(6):746–758
Oberoi TK, Dogan T, Hocking JC et al (2012) IAPs regulate the plasticity of cell migration by directly targeting Rac1 for degradation. EMBO J 31(1):14–28
Pohl C, Jentsch S (2008) Final stages of cytokinesis and midbody ring formation are controlled by BRUCE. Cell 132(5):832–845
Martinez-Ruiz G, Maldonado V, Ceballos-Cancino G, Grajeda JPR, Melendez-Zajgla J (2008) Role of Smac/DIABLO in cancer progression. J Exp Clin Cancer Res 27(1):48
Suzuki Y, Imai Y, Nakayama H, Takahashi K, Takio K, Takahashi R (2001) A serine protease, HtrA2, is released from the mitochondria and interacts with XIAP, inducing cell death. Mol Cell 8(3):613–621
Liston P, Fong WG, Kelly NL et al (2001) Identification of XAF1 as an antagonist of XIAP anti-Caspase activity. Nat Cell Biol 3(2):128
Hegde R, Srinivasula SM, Datta P et al (2003) The polypeptide chain-releasing factor GSPT1/eRF3 is proteolytically processed into an IAP-binding protein. J Biol Chem 278(40):38699–38706
Gottfried Y, Rotem A, Lotan R, Steller H, Larisch S (2004) The mitochondrial ARTS protein promotes apoptosis through targeting XIAP. EMBO J 23(7):1627–1635
Edison N, Zuri D, Maniv I et al (2012) The IAP-antagonist ARTS initiates caspase activation upstream of cytochrome C and SMAC/Diablo. Cell Death Differ 19(2):356–368
Verhagen AM, Kratina TK, Hawkins CJ, Silke J, Ekert PG, Vaux DL (2007) Identification of mammalian mitochondrial proteins that interact with IAPs via N-terminal IAP binding motifs. Cell Death Differ 14(2):348
Tenev T, Bianchi K, Darding M et al (2011) The Ripoptosome, a signaling platform that assembles in response to genotoxic stress and loss of IAPs. Mol Cell 43(3):432–448
Varfolomeev E, Blankenship JW, Wayson SM et al (2007) IAP antagonists induce autoubiquitination of c-IAPs, NF-κB activation, and TNFα-dependent apoptosis. Cell 131(4):669–681
Fu J, Jin Y, Arend LJ (2003) Smac3, a novel Smac/DIABLO splicing variant, attenuates the stability and apoptosis-inhibiting activity of X-linked inhibitor of apoptosis protein. J Biol Chem 278(52):52660–52672
Yamauchi S, Hou YY, Guo AK, Hirata H, Nakajima W, Yip AK, Tanaka N (2014) p53-mediated activation of the mitochondrial protease HtrA2/Omi prevents cell invasion. J Cell Biol 204(7):1191–1207
Arora V, Cheung HH, Plenchette S, Micali OC, Liston P, Korneluk RG (2007) Degradation of survivin by the X-linked inhibitor of apoptosis (XIAP)-XAF1 complex. J Biol Chem 282(36):26202–26209
Micali OC, Cheung HH, Plenchette S, Hurley SL, Liston P, LaCasse EC, Korneluk RG (2007) Silencing of the XAF1 gene by promoter hypermethylation in cancer cells and reactivation to TRAIL-sensitization by IFN-β. BMC Cancer 7(1):52
Huang J, Yao WY, Zhu Q et al (2010) XAF1 as a prognostic biomarker and therapeutic target in pancreatic cancer. Cancer Sci 101(2):559–567
Choo ZE, Koh RYL, Wallis K et al (2016) XAF1 promotes neuroblastoma tumor suppression and is required for KIF1Bβ-mediated apoptosis. Oncotarget 7(23):34229
Wang Y, Mao H, Hao Q, Wang Y, Yang Y, Shen L, Liu P (2012) Association of expression of XIAP-associated factor 1 (XAF1) with clinicopathologic factors, overall survival, microvessel density and cisplatin-resistance in ovarian cancer. Regul Pept 178(1):36–42
Zhu LM, Shi DM, Dai Q et al (2014) Tumor suppressor XAF1 induces apoptosis, inhibits angiogenesis and inhibits tumor growth in hepatocellular carcinoma. Oncotarget 5(14):5403–5415
Shibata T, Mahotka C, Wethkamp N, Heikaus S, Gabbert HE, Ramp U (2007) Disturbed expression of the apoptosis regulators XIAP, XAF1, and Smac/DIABLO in gastric adenocarcinomas. Diagn Mol Pathol 16(1):1–8
Zou B, Chim CS, Pang R et al (2012) XIAP-associated factor 1 (XAF1), a novel target of p53, enhances p53-mediated apoptosis via post-translational modification. Mol Carcinog 51(5):422–432
Plenchette S, Cheung HH, Fong WG, LaCasse EC, Korneluk RG (2007) The role of XAF1 in cancer. Curr Opin Investig Drugs 8(6):469
Bornstein B, Gottfried Y, Edison N, Shekhtman A, Lev T, Glaser F, Larisch S (2011) ARTS binds to a distinct domain in XIAP-BIR3 and promotes apoptosis by a mechanism that is different from other IAP-antagonists. Apoptosis 16(9):869
Garrison JB, Correa RG, Gerlic M et al (2011) ARTS and Siah collaborate in a pathway for XIAP degradation. Mol Cell 41(1):107–116
García-Fernández M, Kissel H, Brown S et al (2010) Sept4/ARTS is required for stem cell apoptosis and tumor suppression. Genes Dev 24(20):2282–2293
Vassina EM, Yousefi S, Simon D, Zwicky C, Conus S, Simon HU (2006) cIAP-2 and survivin contribute to cytokine-mediated delayed eosinophil apoptosis. Eur J Immunol 36(7):1975–1984
Gordon GJ, Mani M, Mukhopadhyay L, Dong L, Yeap BY, Sugarbaker DJ, Bueno R (2007) Inhibitor of apoptosis proteins are regulated by tumour necrosis factor-α in malignant pleural mesothelioma. J Pathol 211(4):439–446
Vince JE, Wong WWL, Khan N et al (2007) IAP antagonists target cIAP1 to induce TNFα-dependent apoptosis. Cell 131(4):682–693
Church DN, Talbot DC (2012) Survivin in solid tumors: rationale for development of inhibitors. Curr Oncol Rep 14(2):120–128
Rodríguez-Berriguete G, Torrealba N, Ortega MA et al (2015) Prognostic value of inhibitors of apoptosis proteins (IAPs) and caspases in prostate cancer: caspase-3 forms and XIAP predict biochemical progression after radical prostatectomy. BMC Cancer 15(1):809
Altieri B, Sbiera S, Della Casa S et al (2017) Livin/BIRC7 expression as malignancy marker in adrenocortical tumors. Oncotarget 8(6):9323
Sbiera S, Kroiss M, Thamm T et al (2013) Survivin in adrenocortical tumors–pathophysiological implications and therapeutic potential. Horm Metab Res 45(02):137–146
Che X, Yang D, Zong H et al (2012) Nuclear cIAP1 overexpression is a tumor stage-and grade-independent predictor of poor prognosis in human bladder cancer patients. Urol Oncol 30(4):450–456
Yang D, Song X, Zhang J et al (2010) Therapeutic potential of siRNA-mediated combined knockdown of the IAP genes (Livin, XIAP, and Survivin) on human bladder cancer T24 cells. Acta Biochim Biophys Sin 42(2):137–144
Poli G, Brancorsini S, Cochetti G, Barillaro F, Egidi MG, Mearini E (2015) Expression of inflammasome-related genes in bladder cancer and their association with cytokeratin 20 messenger RNA. Urol Oncol 33(12):505-e1
Ziegler DS, Keating J, Kesari S et al (2011) A small-molecule IAP inhibitor overcomes resistance to cytotoxic therapies in malignant gliomas in vitro and in vivo. Neuro Oncol 13(8):820–829
Zhang H, Xu F, Xie T, Jin H, Shi L (2012) β-elemene induces glioma cell apoptosis by downregulating survivin and its interaction with hepatitis B X-interacting protein. Oncol Rep 28(6):2083–2090
Li G, Zhang H, Liu Y, Kong L, Guo Q, Jin F (2015) Effect of temozolomide on livin and caspase-3 in U251 glioma stem cells. Exp Ther Med 9(3):744–750
Peng XH, Karna P, O’Regan RM et al (2007) Down-regulation of inhibitor of apoptosis proteins by deguelin selectively induces apoptosis in breast cancer cells. Mol Pharmacol 71(1):101–111
Zhang Y, Zhu J, Tang Y et al (2011) X-linked inhibitor of apoptosis positive nuclear labeling: a new independent prognostic biomarker of breast invasive ductal carcinoma. Diagn Pathol 6(1):49
Choi J, Hwang YK, Choi YJ et al (2007) Neuronal apoptosis inhibitory protein is overexpressed in patients with unfavorable prognostic factors in breast cancer. J Korean Med Sci 22(Suppl):S17–S23
Li F, Yin X, Luo X et al (2013) Livin promotes progression of breast cancer through induction of epithelial–mesenchymal transition and activation of AKT signaling. Cell Signal 25(6):1413–1422
Yaqin M, Runhua L, Fuxi Z (2007) Analyses of Bcl-2, Survivin, and CD44v6 expressions and human papillomavirus infection in cervical carcinomas. Scand J Infect Dis 39(5):441–448
Imoto I, Tsuda H, Hirasawa A, Miura M, Sakamoto M, Hirohashi S, Inazawa J (2002) Expression of cIAP1, a target for 11q22 amplification, correlates with resistance of cervical cancers to radiotherapy. Cancer Res 62(17):4860–4866
Burstein DE, Idrees MT, Li G, Wu M, Kalir T (2008) Immunohistochemical detection of the X-linked inhibitor of apoptosis protein (XIAP) in cervical squamous intraepithelial neoplasia and squamous carcinoma. Ann Diagn Pathol 12(2):85–89
Huerta S, Heinzerling JH, Anguiano-Hernandez YM et al (2007) Modification of gene products involved in resistance to apoptosis in metastatic colon cancer cells: Roles of Fas, Apaf-1, NFκB, IAPs, Smac/DIABLO, and AIF. J Surg Res 142(1):184–194
Wang X, Xu J, Ju S, Ni H, Zhu J, Wang H (2010) Livin gene plays a role in drug resistance of colon cancer cells. Clin Biochem 43(7):655–660
Krajewska M, Kim H, Kim C et al (2005) Analysis of apoptosis protein expression in early-stage colorectal cancer suggests opportunities for new prognostic biomarkers. Clin Cancer Res 11(15):5451–5461
Xu X, Wu X, Jiang Q et al (2015) Downregulation of microRNA-1 and microRNA-145 contributes synergistically to the development of colon cancer. Int J Mol Med 36(6):1630–1638
Lambropoulou M, Papadopoulos N, Tripsianis G et al (2010) Co-expression of survivin, c-erbB2, and cyclooxygenase-2 (COX-2): prognostic value and survival of endometrial cancer patients. J Cancer Res Clin Oncol 136(3):427–435
Gagnon V, Van Themsche C, Turner S, Leblanc V, Asselin E (2008) Akt and XIAP regulate the sensitivity of human uterine cancer cells to cisplatin, doxorubicin and taxol. Apoptosis 13(2):259–271
Gagnon V, St-Germain ME, Parent S, Asselin E (2003) Akt activity in endometrial cancer cells: regulation of cell survival through cIAP-1. Int J Oncol 23(3):803–810
Zhang S, Ding F, Luo A et al (2007) XIAP is highly expressed in esophageal cancer and its downregulation by RNAi sensitizes esophageal carcinoma cell lines to chemotherapeutics. Cancer Biol Ther 6(6):974–979
Upadhyay R, Khurana R, Kumar S, Ghoshal UC, Mittal B (2011) Role of survivin gene promoter polymorphism (– 31G > C) in susceptibility and survival of esophageal cancer in northern India. Ann Surg Oncol 18(3):880–887
Zhang S, Tang W, Weng S et al (2014) Apollon modulates chemosensitivity in human esophageal squamous cell carcinoma. Oncotarget 5(16):7183
Wang TT, Qian XP, Liu BR (2007) Survivin: potential role in diagnosis, prognosis and targeted therapy of gastric cancer. World J Gastroenterol 13(20):2784
Wang DG, Sun YB, Ye F et al (2014) Anti-tumor activity of the X-linked inhibitor of apoptosis (XIAP) inhibitor embelin in gastric cancer cells. Mol Cell Biochem 386(1–2):143–152
Chung CY, Park YL, Kim N et al (2013) Expression and prognostic significance of Livin in gastric cancer. Oncol Rep 30(5):2520–2528
Falkenhorst J, Grunewald S, Mühlenberg T et al (2016) Inhibitor of apoptosis proteins (IAPs) are commonly dysregulated in GIST and can be pharmacologically targeted to enhance the pro-apoptotic activity of imatinib. Oncotarget 7(27):41390–41403
Shi YH, Ding WX, Zhou J et al (2008) Expression of X-linked inhibitor-of-apoptosis protein in hepatocellular carcinoma promotes metastasis and tumor recurrence. Hepatology 48(2):497–507
Fan L, Sun G, Ma T, Zhong F, Wei W (2013) Melatonin overcomes apoptosis resistance in human hepatocellular carcinoma by targeting survivin and XIAP. J Pineal Res 55(2):174–183
Guo H, Gao YT, Zhang Q et al (2013) Expression and clinical significance of livin protein in hepatocellular carcinoma. Dis Markers 35(5):489–496
Tanimoto T, Tsuda H, Imazeki N, Ohno Y, Imoto I, Inazawa J, Matsubara O (2005) Nuclear expression of cIAP-1, an apoptosis inhibiting protein, predicts lymph node metastasis and poor patient prognosis in head and neck squamous cell carcinomas. Cancer Lett 224(1):141–151
Scheper MA, Nikitakis NG, Sauk JJ (2007) Survivin is a downstream target and effector of sulindac-sensitive oncogenic Stat3 signalling in head and neck cancer. Int J Oral Maxillofac Surg 36(7):632–639
Qi G, Kudo Y, Ando T, Tsunematsu T, Shimizu N, Siriwardena SB, Takata T (2010) Nuclear survivin expression is correlated with malignant behaviors of head and neck cancer together with Aurora-B. Oral Oncol 46(4):263–270
Yang XH, Feng ZE, Yan M et al (2012) XIAP is a predictor of cisplatin-based chemotherapy response and prognosis for patients with advanced head and neck cancer. PloS ONE, 7(3):e31601
Lévy P, Vidaud D, Leroy K et al (2004) Molecular profiling of malignant peripheral nerve sheath tumors associated with neurofibromatosis type 1, based on large-scale real-time RT-PCR. Mol Cancer 3(1):20
Chen SM, Li YY, Tu CH et al (2016) Blockade of inhibitors of apoptosis proteins in combination with conventional chemotherapy leads to synergistic antitumor activity in medulloblastoma and cancer stem-like cells. PloS ONE, 11(8):e0161299
Abdel-Aziz A, Mohamed MAA, Akl FMF, Taha ANM (2013) Survivin expression in medulloblastoma: a possible marker for survival. Pathol Oncol Res 19(3):413–419
Kluger HM, McCarthy MM, Alvero AB et al (2007) The X-linked inhibitor of apoptosis protein (XIAP) is up-regulated in metastatic melanoma, and XIAP cleavage by Phenoxodiol is associated with Carboplatin sensitization. J Transl Med 5(1):6
McKenzie JA, Liu T, Goodson AG, Grossman D (2010) Survivin enhances motility of melanoma cells by supporting Akt activation and α5 integrin upregulation. Cancer Res 70(20):7927–7937
Mckenzie JA, Grossman D (2012) Role of the apoptotic and mitotic regulator survivin in melanoma. Anticancer Res 32(2):397–404
Lazar I, Perlman R, Lotem M, Peretz T, Ben-Yehuda D, Kadouri L (2012) The clinical effect of the inhibitor of apopotosis protein livin in melanoma. Oncology 82(4):197–204
Tassi E, Zanon M, Vegetti C et al (2012) Role of apollon in human melanoma resistance to antitumor agents that activate the intrinsic or the extrinsic apoptosis pathways. Clin Cancer Res 18(12):3316–3327
Cregan IL, Dharmarajan AM, Fox SA (2013) Mechanisms of cisplatin-induced cell death in malignant mesothelioma cells: Role of inhibitor of apoptosis proteins (IAPs) and caspases. Int J Oncol 42(2):444–452
Dasgupta A, Alvarado CS, Xu Z, Findley HW (2010) Expression and functional role of inhibitor-of-apoptosis protein livin (BIRC7) in neuroblastoma. Biochem Biophys Res Commun 400(1):53–59
Eschenburg G, Eggert A, Schramm A, Lode HN, Hundsdoerfer P (2012) Smac mimetic LBW242 sensitizes XIAP-overexpressing neuroblastoma cells for TNF-α–independent apoptosis. Cancer Res 72(10):2645–2656
Najem S, Langemann D, Appl B, Trochimiuk M, Hundsdoerfer P, Reinshagen K, Eschenburg G (2016) Smac mimetic LCL161 supports neuroblastoma chemotherapy in a drug class-dependent manner and synergistically interacts with ALK inhibitor TAE684 in cells with ALK mutation F1174L. Oncotarget 7(45):72634
Lamers F, van der Ploeg I, Schild L et al (2011) Knockdown of survivin (BIRC5) causes apoptosis in neuroblastoma via mitotic catastrophe. Endocr Relat Cancer 18(6):657–668
Cheng YJ, Jiang HS, Hsu SL, Lin LC, Wu CL, Ghanta VK, Hsueh CM (2010) XIAP-mediated protection of H460 lung cancer cells against cisplatin. Eur J Pharmacol 627(1):75–84
Fan J, Wang L, Jiang GN, He WX, Ding JA (2008) The role of survivin on overall survival of non-small cell lung cancer, a meta-analysis of published literatures. Lung Cancer 61(1):91–96
Sun JG, Liao RX, Zhang SX et al (2011) Role of inhibitor of apoptosis protein Livin in radiation resistance in nonsmall cell lung cancer. Cancer Biother Radiopharm 26(5):585–592
Dong X, Lin D, Low C et al (2013) Elevated expression of BIRC6 protein in non–small-cell lung cancers is associated with cancer recurrence and chemoresistance. J Thorac Oncol 8(2):161–170
Osaka E, Suzuki T, Osaka S et al (2007) Survivin expression levels as independent predictors of survival for osteosarcoma patients. J Orthop Res 25(1):116–121
Nedelcu T, Kubista B, Koller A et al (2008) Livin and Bcl-2 expression in high-grade osteosarcoma. J Cancer Res Clin Oncol 134(2):237–244
Qu Y, Xia P, Zhang S, Pan S, Zhao J (2015) Silencing XIAP suppresses osteosarcoma cell growth, and enhances the sensitivity of osteosarcoma cells to doxorubicin and cisplatin. Oncol Rep 33(3):1177–1184
Shaw TJ, Lacasse EC, Durkin JP, Vanderhyden BC (2008) Downregulation of XIAP expression in ovarian cancer cells induces cell death in vitro and in vivo. Int J Cancer 122(6):1430–1434
Chen L, Liang L, Yan X et al (2013) Survivin status affects prognosis and chemosensitivity in epithelial ovarian cancer. Int J Gynecol Cancer 23(2):256–263
Liu X, Wang A, Gao H, Yuan Z, Jiao Y (2012) Expression and role of the inhibitor of apoptosis protein livin in chemotherapy sensitivity of ovarian carcinoma. Int J Oncol 41(3):1021–1028
Cohen S, Bruchim I, Graiver D et al (2013) Platinum-resistance in ovarian cancer cells is mediated by IL-6 secretion via the increased expression of its target cIAP-2. J Mol Med 91(3):357–368
Wang L, Chen YJ, Hou J, Wang YY, Tang WQ, Shen XZ, Tu RQ (2014) Expression and clinical significance of BIRC6 in human epithelial ovarian cancer. Tumor Biol 35(5):4891–4896
Esposito I, Kleeff J, Abiatari I et al (2007) Overexpression of cellular inhibitor of apoptosis protein 2 is an early event in the progression of pancreatic cancer. J Clin Pathol 60(8):885–895
Lopes RB, Gangeswaran R, McNeish IA, Wang Y, Lemoine NR (2007) Expression of the IAP protein family is dysregulated in pancreatic cancer cells and is important for resistance to chemotherapy. Int J Cancer 120(11):2344–2352
Han Z, Lee S, Je S, Eom CY, Choi HJ, Song JJ, Kim JH (2016) Survivin silencing and TRAIL expression using oncolytic adenovirus increase anti-tumorigenic activity in gemcitabine-resistant pancreatic cancer cells. Apoptosis 21(3):351–364
Seligson DB, Hongo F, Huerta-Yepez S et al (2007) Expression of X-linked inhibitor of apoptosis protein is a strong predictor of human prostate cancer recurrence. Clin Cancer Res 13(20):6056–6063
Rodríguez-Berriguete G, Fraile B, de Bethencourt FR et al (2010) Role of IAPs in prostate cancer progression: immunohistochemical study in normal and pathological (benign hyperplastic, prostatic intraepithelial neoplasia and cancer) human prostate. BMC Cancer 10(1):18
Mizutani Y, Nakanishi H, Li YN et al (2007) Overexpression of XIAP expression in renal cell carcinoma predicts a worse prognosis. Int J Oncol 30(4):919–925
Lei Y, Geng Z, Guo-Jun W, He W, Jian-Lin Y (2010) Prognostic significance of survivin expression in renal cell cancer and its correlation with radioresistance. Mol Cell Biochem 344(1–2):23–31
Kempkensteffen C, Hinz S, Christoph F et al (2007) Expression of the apoptosis inhibitor livin in renal cell carcinomas: correlations with pathology and outcome. Tumor Biol 28(3):132–138
Simon-Keller K, Paschen A, Hombach AA et al (2013) Survivin blockade sensitizes rhabdomyosarcoma cells for lysis by fetal acetylcholine receptor–redirected T cells. Am J Pathol 182(6):2121–2131
Tirrò E, Consoli ML, Massimino M et al (2006) Altered expression of c-IAP1, survivin, and Smac contributes to chemotherapy resistance in thyroid cancer cells. Cancer Res 66(8):4263–4272
Antonaci A, Consorti F, Mardente S, Natalizi S, Giovannone G, Della Rocca C (2008) Survivin and cyclin D1 are jointly expressed in thyroid papillary carcinoma and microcarcinoma. Oncol Rep 20(1):63–67
Yim JH, Kim WG, Jeon MJ et al (2014) Association between expression of X-linked inhibitor of apoptosis protein and the clinical outcome in a BRAFV600E-prevalent papillary thyroid cancer population. Thyroid 24(4):689–694
El-Mesallamy HO, Hegab HM, Kamal AM (2011) Expression of inhibitor of apoptosis protein (IAP) livin/BIRC7 in acute leukemia in adults: correlation with prognostic factors and outcome. Leuk Res 35(12):1616–1622
Hundsdoerfer P, Dietrich I, Schmelz K, Eckert C, Henze G (2010) XIAP expression is post-transcriptionally upregulated in childhood ALL and is associated with glucocorticoid response in T-cell ALL. Pediatr Blood Cancer 55(2):260–266
Park E, Gang EJ, Hsieh YT et al (2011) Targeting survivin overcomes drug resistance in acute lymphoblastic leukemia. Blood 118(8):2191–2199
Ismail EAR, Mahmoud HM, Tawfik LM et al (2012) BIRC6/Apollon gene expression in childhood acute leukemia: impact on therapeutic response and prognosis. Eur J Haematol 88(2):118–127
Moreno-Martínez D, Nomdedeu M, Lara-Castillo MC et al (2014) XIAP inhibitors induce differentiation and impair clonogenic capacity of acute myeloid leukemia stem cells. Oncotarget 5(12):4337–4346
Carter BZ, Qiu Y, Huang X et al (2012) Survivin is highly expressed in CD34 + 38 – leukemic stem/progenitor cells and predicts poor clinical outcomes in AML. Blood 120(1):173–180
Loeder S, Zenz T, Schnaiter A et al (2009) A novel paradigm to trigger apoptosis in chronic lymphocytic leukemia. Cancer Res 69(23):8977–8986
Silva KL, de Souza PS, de Moraes GN et al (2013) XIAP and P-glycoprotein co-expression is related to imatinib resistance in chronic myeloid leukemia cells. Leuk Res 37(10):1350–1358
Stella S, Tirro E, Conte E et al (2013) Suppression of survivin induced by a BCR-ABL/JAK2/STAT3 pathway sensitizes imatinib-resistant CML cells to different cytotoxic drugs. Mol Cancer Ther 12(6):1085–1098
Okumu DO, East MP, Levine M et al (2017) BIRC6 mediates imatinib resistance independently of Mcl-1. PloS ONE 12(5):e0177871
Akyurek N, Ren Y, Rassidakis GZ, Schlette EJ, Medeiros LJ (2006) Expression of inhibitor of apoptosis proteins in B-cell non-Hodgkin and Hodgkin lymphomas. Cancer 107(8):1844–1851
Garcıa JF, Camacho FI, Morente M et al (2003) Hodgkin and Reed-Sternberg cells harbor alterations in the major tumor suppressor pathways and cell-cycle checkpoints: analyses using tissue microarrays. Blood 101(2):681–689
Ramakrishnan V, Painuly U, Kimlinger T, Haug J, Rajkumar SV, Kumar S (2014) Inhibitor of apoptosis proteins (IAPs) as therapeutic targets in multiple myeloma (MM). Leukemia 28(7):1519
Tsubaki M, Takeda T, Ogawa N et al (2015) Overexpression of survivin via activation of ERK1/2, Akt, and NF-κB plays a central role in vincristine resistance in multiple myeloma cells. Leuk Res 39(4):445–452
Cillessen SA, Reed JC, Welsh K et al (2008) Small-molecule XIAP antagonist restores caspase-9–mediated apoptosis in XIAP-positive diffuse large B-cell lymphoma cells. Blood 111(1):369–375
Jacquemin G, Granci V, Gallouet AS et al (2012) Quercetin-mediated Mcl-1 and survivin downregulation restores TRAIL-induced apoptosis in non-Hodgkin’s lymphoma B cells. Haematologica 97(1):38–46
Mobahat M, Narendran A, Riabowol K (2014) Survivin as a preferential target for cancer therapy. Int J Mol Sci 15(2):2494–2516
Krieg A, Werner TA, Verde PE, Stoecklein NH, Knoefel WT (2013). Prognostic and clinicopathological significance of survivin in colorectal cancer: a meta-analysis. PloS ONE 8(6):e65338
Chen J, Li T, Liu Q, Jiao H, Yang W, Liu X, Huo Z (2014). Clinical and prognostic significance of HIF-1α, PTEN, CD44v6, and survivin for gastric cancer: a meta-analysis. PloS ONE 9(3):e91842
Fernández JG, Rodríguez DA, Valenzuela M et al (2014) Survivin expression promotes VEGF-induced tumor angiogenesis via PI3K/Akt enhanced β-catenin/Tcf-Lef dependent transcription. Mol Cancer 13(1):209
Martini E, Schneider E, Neufert C, Neurath MF, Becker C (2016) Survivin is a guardian of the intestinal stem cell niche and its expression is regulated by TGF-β. Cell Cycle 15(21):2875–2881
Fukuda S, Hoggatt J, Singh P et al (2015) Survivin modulates genes with divergent molecular functions and regulates proliferation of hematopoietic stem cells through Evi-1. Leukemia 29(2):433–441
Feng R, Zhou S, Liu Y et al (2013) Sox2 protects neural stem cells from apoptosis via up-regulating survivin expression. Biochem J 450(3):459–468
Tamm I, Richter S, Oltersdorf D et al (2004) High expression levels of x-linked inhibitor of apoptosis protein and survivin correlate with poor overall survival in childhood de novo acute myeloid leukemia. Clin Cancer Res 10(11):3737–3744
Sung KW, Choi J, Hwang YK et al (2009) Overexpression of X-linked inhibitor of apoptosis protein (XIAP) is an independent unfavorable prognostic factor in childhood de novo acute myeloid leukemia. J Korean Med Sci 24(4):605–613
Tamm I, Richter S, Scholz F et al (2004) XIAP expression correlates with monocytic differentiation in adult de novo AML: impact on prognosis. Hematol J 5(6):489–495
Ma JJ, Chen BL, Xin XY (2009) XIAP gene downregulation by small interfering RNA inhibits proliferation, induces apoptosis, and reverses the cisplatin resistance of ovarian carcinoma. Eur J Obstet Gynecol Reprod Biol 146(2):222–226
Endo T, Abe S, Seidlar HBK et al (2004) Expression of IAP family proteins in colon cancers from patients with different age groups. Cancer Immunol Immunother 53(9):770–776
Cha JD, Kim HK, Cha IH (2014) Cytoplasmic HuR expression: Correlation with cellular inhibitors of apoptosis protein-2 expression and clinicopathologic factors in oral squamous cell carcinoma cells. Head Neck 36(8):1168–1175
Mannhold R, Fulda S, Carosati E (2010) IAP antagonists: promising candidates for cancer therapy. Drug Discov Today 15(5):210–219
Connolly K, Mitter R, Muir M, Jodrell D, Guichard S (2009) Stable XIAP knockdown clones of HCT116 colon cancer cells are more sensitive to TRAIL, taxanes and irradiation in vitro. Cancer Chemother Pharmacol 64(2):307–316
Holt SV, Brookes KE, Dive C, Makin GW (2011) Down-regulation of XIAP by AEG35156 in paediatric tumour cells induces apoptosis and sensitises cells to cytotoxic agents. Oncol Rep 25(4):1177–1181
Ohnishi K, Nagata Y, Takahashi A, Taniguchi S, Ohnishi T (2008) Effective enhancement of X-ray-induced apoptosis in human cancer cells with mutated p53 by siRNA targeting XIAP. Oncol Rep 20(1):57–61
Yamaguchi Y, Shiraki K, Fuke H et al (2005) Targeting of X-linked inhibitor of apoptosis protein or survivin by short interfering RNAs sensitize hepatoma cells to TNF-related apoptosis-inducing ligand-and chemotherapeutic agent-induced cell death. Oncol Rep 14(5):1311–1316
Schimmer AD, Estey EH, Borthakur G et al (2009) Phase I/II trial of AEG35156 X-linked inhibitor of apoptosis protein antisense oligonucleotide combined with idarubicin and cytarabine in patients with relapsed or primary refractory acute myeloid leukemia. J Clin Oncol 27(28):4741–4746
Mahadevan D, Chalasani P, Rensvold D et al (2013) Phase I trial of AEG35156 an antisense oligonucleotide to XIAP plus gemcitabine in patients with metastatic pancreatic ductal adenocarcinoma. Am J Clin Oncol 36(3):239–243
Lee FA, Zee BCY, Cheung FY et al (2016) Randomized phase II study of the X-linked inhibitor of apoptosis (XIAP) antisense AEG35156 in combination with sorafenib in patients with advanced hepatocellular carcinoma (HCC). Am J Clin Oncol 39(6):609–613
Schimmer AD, Herr W, Hänel M et al (2011) Addition of AEG35156 XIAP antisense oligonucleotide in reinduction chemotherapy does not improve remission rates in patients with primary refractory acute myeloid leukemia in a randomized phase II study. Clin Lymphoma Myeloma Leuk 11(5):433–438
Dean EJ, Ward T, Pinilla C et al (2010) A small molecule inhibitor of XIAP induces apoptosis and synergises with vinorelbine and cisplatin in NSCLC. Br J Cancer 102(1):97
Metwalli AR, Khanbolooki S, Jinesh G et al (2010) Smac mimetic reverses resistance to TRAIL and chemotherapy in human urothelial cancer cells. Cancer Biol Ther 10(9):885–892
Stadel D, Cristofanon S, Abhari BA et al (2011) Requirement of nuclear factor κB for Smac mimetic–mediated sensitization of pancreatic carcinoma cells for gemcitabine-induced apoptosis. Neoplasia 13(12):1162–1170
Servida F, Lecis D, Scavullo C et al (2011) Novel second mitochondria-derived activator of caspases (Smac) mimetic compounds sensitize human leukemic cell lines to conventional chemotherapeutic drug-induced and death receptor-mediated apoptosis. Invest New Drugs 29(6):1264–1275
Loeder S, Fakler M, Schoeneberger H et al (2012) RIP1 is required for IAP inhibitor-mediated sensitization of childhood acute leukemia cells to chemotherapy-induced apoptosis. Leukemia 26(5):1020–1029
Wagner L, Marschall V, Karl S et al (2013) Smac mimetic sensitizes glioblastoma cells to Temozolomide-induced apoptosis in a RIP1-and NF-[kappa] B-dependent manner. Oncogene 32(8):988
Finlay D, Vamos M, González-López M et al (2014) Small-molecule IAP antagonists sensitize cancer cells to TRAIL-induced apoptosis: roles of XIAP and cIAPs. Mol Cancer Ther 13(1):5–15
Schirmer M, Trentin L, Queudeville M et al (2016) Intrinsic and chemo-sensitizing activity of SMAC-mimetics on high-risk childhood acute lymphoblastic leukemia. Cell Death Dis 7(1):e2052
Beug ST, Tang VA, LaCasse EC et al (2014) Smac mimetics and innate immune stimuli synergize to promote tumor death. Nat Biotechnol 32(2):182–190
Infante JR, Dees EC, Olszanski AJ, Dhuria SV, Sen S, Cameron S, Cohen RB (2014) Phase I dose-escalation study of LCL161, an oral inhibitor of apoptosis proteins inhibitor, in patients with advanced solid tumors. J Clin Oncol 32(28):3103–3110
Dienstmann R, Adamo B, Vidal L, Dees EC, Chia S, Mayer EL et al (2012) Phase Ib study of LCL161, an oral antagonist of inhibitor of apoptosis proteins, in combination with weekly paclitaxel in patients with advanced solid tumors. Cancer Res. doi:10.1158/0008-5472.SABCS12-P6-11-06
Parton M, Bardia A, Kummel S et al (2015) A phase II, open-label, neoadjuvant, randomized study of LCL161 with paclitaxel in patients with triple-negative breast cancer (TNBC). J Clin Oncol 33(15):1014–1014
Hurwitz HI, Smith DC, Pitot HC et al (2015) Safety, pharmacokinetics, and pharmacodynamic properties of oral DEBIO1143 (AT-406) in patients with advanced cancer: results of a first-in-man study. Cancer Chemother Pharmacol 75(4):851–859
Tolcher AW, Papadopoulos KP, Patnaik A et al (2013) Phase I study of safety and pharmacokinetics (PK) of GDC-0917, an antagonist of inhibitor of apoptosis (IAP) proteins in patients (Pts) with refractory solid tumors or lymphoma. J Clin Oncol 31(15):2503–2503
Tolcher A, Bendell JC, Papadopoulos KP et al (2016) A phase I dose escalation study evaluating the safety tolerability and pharmacokinetics of cudc-427, a potent, oral, monovalent IAP antagonist, in patients with refractory solid tumors. Clin Cancer Res 22(18):4567–4573
Amaravadi RK, Schilder RJ, Martin LP et al (2015) A phase I study of the SMAC-mimetic birinapant in adults with refractory solid tumors or lymphoma. Mol Cancer Ther 14(11):2569–2575
Amaravadi RK, Senzer NN, Martin LP et al (2013) A phase I study of birinapant (TL32711) combined with multiple chemotherapies evaluating tolerability and clinical activity for solid tumor patients. J Clin Oncol 31(15):2504–2504
Senzer NN, LoRusso P, Martin LP et al (2013) Phase II clinical activity and tolerability of the SMAC-mimetic birinapant (TL32711) plus irinotecan in irinotecan-relapsed/refractory metastatic colorectal cancer. J Clin Oncol 31(15):3621–3621
Noonan AM, Bunch KP, Chen JQ et al (2016) Pharmacodynamic markers and clinical results from the phase 2 study of the SMAC mimetic birinapant in women with relapsed platinum-resistant or-refractory epithelial ovarian cancer. Cancer 122(4):588–597
Hamilton EP, Birrer MJ, DiCarlo BA et al (2015) A phase 1b, open-label, non-randomized multicenter study of birinapant in combination with conatumumab in subjects with relapsed epithelial ovarian cancer, primary peritoneal cancer, or fallopian tube cancer. J Clin Oncol 33(15):5571–5571
Donnellan WB, Diez-Campelo M, Heuser M et al (2016) A phase 2 study of azacitidine (5-AZA) with or without birinapant in subjects with higher risk myelodysplastic syndrome (MDS) or chronic myelomonocytic leukemia (CMML). J Clin Oncol 34(15):7060–7060
Sikic BI, Eckhardt SG, Gallant G et al (2011) Safety, pharmacokinetics (PK), and pharmacodynamics (PD) of HGS1029, an inhibitor of apoptosis protein (IAP) inhibitor, in patients (Pts) with advanced solid tumors: Results of a phase I study. J Clin Oncol 29(15):3008–3008
Nikolovska-Coleska Z, Xu L, Hu Z et al (2004) Discovery of embelin as a cell-permeable, small-molecular weight inhibitor of XIAP through structure-based computational screening of a traditional herbal medicine three-dimensional structure database. J Med Chem 47(10):2430–2440
Heo JY, Kim HJ, Kim SM et al (2011) Embelin suppresses STAT3 signaling, proliferation, and survival of multiple myeloma via the protein tyrosine phosphatase PTEN. Cancer Lett 308(1):71–80
Dai Y, DeSano J, Qu Y, Tang W, Meng Y, Lawrence TS, Xu L (2011) Natural IAP inhibitor Embelin enhances therapeutic efficacy of ionizing radiation in prostate cancer. Am J Cancer Res 1(2):128–143
Ali AG, Mohamed MF, Abdelhamid AO, Mohamed MS (2017) A novel adamantane thiadiazole derivative induces mitochondria-mediated apoptosis in lung carcinoma cell line. Bioorg Med Chem 25(1):241–253
Sun H, Liu L, Lu J, Qiu S, Yang CY, Yi H, Wang S (2010) Cyclopeptide Smac mimetics as antagonists of IAP proteins. Bioorg Med Chem Lett 20(10):3043–3046
Lu J, McEachern D, Sun H et al (2011) Therapeutic potential and molecular mechanism of a novel, potent, nonpeptide, Smac mimetic SM-164 in combination with TRAIL for cancer treatment. Mol Cancer Ther 10(5):902–914
Mitsuuchi Y, Benetatos CA, Deng Y et al (2017) Bivalent IAP antagonists, but not monovalent IAP antagonists, inhibit TNF-mediated NF-κB signaling by degrading TRAF2-associated cIAP1 in cancer cells. Cell Death Discov 3:16046
Grossman D, McNiff JM, Li F, Altieri DC (1999) Expression and targeting of the apoptosis inhibitor, survivin, in human melanoma. J Invest Dermatol 113(6):1076–1081
Sharma H, Sen S, Lo Muzio L, Mariggiò MA, Singh N (2005) Antisense-mediated downregulation of anti-apoptotic proteins induces apoptosis and sensitizes head and neck squamous cell carcinoma cells to chemotherapy. Cancer Biol Ther 4(7):720–727
Rödel F, Frey B, Leitmann W, Capalbo G, Weiss C, Rödel C (2008) Survivin antisense oligonucleotides effectively radiosensitize colorectal cancer cells in both tissue culture and murine xenograft models. Int J Radiat Oncol Biol Phys 71(1):247–255
Du ZX, Zhang HY, Gao DX, Wang HQ, Li YJ, Liu GL (2006) Antisurvivin oligonucleotides inhibit growth and induce apoptosis in human medullary thyroid carcinoma cells. Exp Mol Med 38(3):230
Natale R, Blackhall F, Kowalski D et al (2014) Evaluation of antitumor activity using change in tumor size of the survivin antisense oligonucleotide LY2181308 in combination with docetaxel for second-line treatment of patients with non–small-cell lung cancer: a randomized open-label phase II study. J Thorac Oncol 9(11):1704–1708
Kami K, Doi R, Koizumi M et al (2005) Downregulation of survivin by siRNA diminishes radioresistance of pancreatic cancer cells. Surgery 138(2):299–305
Yang CT, Li JM, Weng HH, Li YC, Chen HC, Chen MF (2010) Adenovirus-mediated transfer of siRNA against survivin enhances the radiosensitivity of human non-small cell lung cancer cells. Cancer Gene Ther 17(2):120
Karami H, Baradaran B, Esfahani A et al (2013) siRNA-mediated silencing of survivin inhibits proliferation and enhances etoposide chemosensitivity in acute myeloid leukemia cells. Asian Pac J Cancer Prev 14(12):7719–7724
Liu W, Zhu F, Jiang Y, Sun D, Yang B, Yan H (2013) siRNA targeting survivin inhibits the growth and enhances the chemosensitivity of hepatocellular carcinoma cells. Oncol Rep 29(3):1183–1188
Vogl TJ, Oppermann E, Qian J et al (2016) Transarterial chemoembolization of hepatocellular carcinoma in a rat model: the effect of additional injection of survivin siRNA to the treatment protocol. BMC Cancer 16(1):325
Cui M, Au JLS, Wientjes MG, O’donnell MA, Loughlin KR, Lu Z (2015) Intravenous siRNA silencing of survivin enhances activity of mitomycin C in human bladder RT4 xenografts. J Urol 194(1):230–237
Salzano G, Navarro G, Trivedi MS, De Rosa G, Torchilin VP (2015) Multifunctional polymeric micelles co-loaded with anti-survivin siRNA and paclitaxel overcome drug resistance in an animal model of ovarian cancer. Mol Cancer Ther 14(4):1075–1084
Wang T, Gantier MP, Xiang D et al (2015) EpCAM aptamer-mediated survivin silencing sensitized cancer stem cells to doxorubicin in a breast cancer model. Theranostics 5(12):1456
Pennati M, Folini M, Zaffaroni N (2007) Targeting survivin in cancer therapy: fulfilled promises and open questions. Carcinogenesis 28(6):1133–1139
Zhang S, Wang X, Gu Z, Wang L (2016) Small molecule survivin inhibitor YM155 displays potent activity against human osteosarcoma cells. Cancer Investig 34(8):401–407
Ueno T, Uehara S, Nakahata K, Okuyama H (2016) Survivin selective inhibitor YM155 promotes cisplatin induced apoptosis in embryonal rhabdomyosarcoma. Int J Oncol 48(5):1847–1854
Nakahara T, Takeuchi M, Kinoyama I et al (2007) YM155, a novel small-molecule survivin suppressant, induces regression of established human hormone-refractory prostate tumor xenografts. Cancer Res 67(17):8014–8021
Na YS, Yang SJ, Kim SM et al (2012) YM155 induces EGFR suppression in pancreatic cancer cells. PLoS ONE 7(6):e38625
Ling X, Cao S, Cheng Q, Keefe JT, Rustum YM, Li F (2012) A novel small molecule FL118 that selectively inhibits survivin, Mcl-1, XIAP and cIAP2 in a p53-independent manner, shows superior antitumor activity. PloS ONE 7(9):e45571
Coumar MS, Tsai FY, Kanwar JR, Sarvagalla S, Cheung CHA (2013) Treat cancers by targeting survivin: just a dream or future reality? Cancer Treat Rev 39(7):802–811
Grossman SA, Ye X, Peereboom D et al (2012) Phase I study of terameprocol in patients with recurrent high-grade glioma. Neuro-Oncol 14(4):511–517
Giaccone G, Zatloukal P, Roubec J et al (2009) Multicenter phase II trial of YM155, a small-molecule suppressor of survivin, in patients with advanced, refractory, non–small-cell lung cancer. J Clin Oncol 27(27):4481–4486
Cheson BD, Bartlett NL, Vose JM et al (2012) A phase II study of the survivin suppressant YM155 in patients with refractory diffuse large B-cell lymphoma. Cancer 118(12):3128–3134
Papadopoulos KP, Lopez-Jimenez J, Smith SE et al (2016) A multicenter phase II study of sepantronium bromide (YM155) plus rituximab in patients with relapsed aggressive B-cell Non-Hodgkin lymphoma. Leuk Lymphoma 57(8):1848–1855
Kudchadkar R, Ernst S, Chmielowski B et al (2015) A phase 2, multicenter, open-label study of sepantronium bromide (YM155) plus docetaxel in patients with stage III (unresectable) or stage IV melanoma. Cancer Med 4(5):643–650
Clemens MR, Gladkov OA, Gartner E et al (2015) Phase II, multicenter, open-label, randomized study of YM155 plus docetaxel as first-line treatment in patients with HER2-negative metastatic breast cancer. Breast Cancer Res Treat 149(1):171–179
Kelly RJ, Thomas A, Rajan A et al (2013) A phase I/II study of sepantronium bromide (YM155, survivin suppressor) with paclitaxel and carboplatin in patients with advanced non-small-cell lung cancer. Ann Oncol 24(10):2601–2606
Raetz EA, Morrison D, Romanos-Sirakis E et al (2014) A phase I study of EZN-3042, a novel survivin messenger ribonucleic acid (mRNA) antagonist, administered in combination with chemotherapy in children with relapsed acute lymphoblastic leukemia (ALL): a report from the therapeutic advances in childhood leukemia and lymphoma (TACL) consortium. J Pediatr Hematol Oncol 36(6):458
Erba HP, Sayar H, Juckett M et al (2013) Safety and pharmacokinetics of the antisense oligonucleotide (ASO) LY2181308 as a single-agent or in combination with idarubicin and cytarabine in patients with refractory or relapsed acute myeloid leukemia (AML). Investig New Drugs 31(4):1023–1034
Wiechno PJ, Chlosta P, Pikiel J et al (2013) Randomized phase II study with window-design to evaluate anti-tumor activity of the survivin antisense oligonucleotide (ASO) ly2181308 in combination with docetaxel for first-line treatment of castrate-resistant prostate cancer (CRPC). J Clin Oncol 31(15):5019–5019
Rohn JL, Noteborn MHM (2004) The viral death effector Apoptin reveals tumor-specific processes. Apoptosis 9(3):315–322
Ruiz-Martínez S, Castro J, Vilanova M et al (2017) A truncated apoptin protein variant selectively kills cancer cells. Investig New Drugs 35(3):260–268
Philchenkov A, Zavelevich M, Kroczak TJ, Los MJ (2004) Caspases and cancer: mechanisms of inactivation and new treatment modalities. Exp Oncol 26(2):82–97
Yamabe K, Shimizu S, Ito T et al (1999) Cancer gene therapy using a pro-apoptotic gene, caspase-3. Gene Ther 6(12):1952–1959
Li X, Fan R, Zou X et al (2007) Inhibitory effect of recombinant adenovirus carrying immunocaspase-3 on hepatocellular carcinoma. Biochem Biophys Res Commun 358(2):489–494
Acknowledgements
This work is supported by a research Grant S—1438-0013 from University of Tabuk, Kingdom of Saudi Arabia to Mervat S. Mohamed.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Mervat S. Mohamed has received a research Grant S – 1438-0013 from University of Tabuk, Kingdom of Saudi Arabia. Other authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mohamed, M.S., Bishr, M.K., Almutairi, F.M. et al. Inhibitors of apoptosis: clinical implications in cancer. Apoptosis 22, 1487–1509 (2017). https://doi.org/10.1007/s10495-017-1429-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-017-1429-4