Abstract
The continuous depletion of fossil resources, energy-crisis, and environmental pollution has become the main drivers for the transition of linear petroleum-refinery to a sustainable biorefinery. Lignocellulosic biomass (LCB) remains the most abundant underutilized renewable biomaterial on our earth that offers a sustainable production of various value-added products like biofuels and other platform chemicals. However, presence of impenetrable lignin sheath along with highly crystalline structure of holocellulose barricades its commercial utilization. In this context, a suitable pretreatment method is mandatory to reduce the recalcitrance of the LCB material. Pretreatment capabilities derived from the extraordinary conditions for its effectiveness in laboratories and even at industrial scale for conversion of LCB are limited. In order to make these LCB bioconversion processes economically viable, more research is needed to focus on developing biorefinery approach. In current scenario, a pretreatment method that can fractionate the variety of LCB to produce lignin free carbohydrates with negligible waste biomass generation is of utmost need. Therefore, to promote the emerging technologies, the present review summarizes the recent advances in the most advanced pretreatment technologies being utilized for the LCB based biorefinery. Pros and cons associated with various pretreatment methods have been critically reviewed. Further, efforts have been made to create a better understanding of the already researched pretreatment technologies to facilitate the future research direction to realize the idea of biorefinery an actuality.
Graphical Abstract

Highlights
-
Pretreatment methods for valorization of LCB have been extensively reviewed
-
Applicability of pretreatment methods to LCB biorefinery has been discussed
-
Mechanisms of pretreatment methods have been discussed
-
Challenges associated with pretreatment methods have been highlighted
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Statement of Novelty
The novelty of this review lies in its comprehensive analysis of recent advances in pretreatment technologies especially for lignocellulosic biomass, with a focus on fractionating the biomass to produce lignin-free carbohydrates while minimizing waste biomass generation. By critically evaluating the pros and cons of various pretreatment methods, this review not only consolidates existing knowledge but also provides insights into the future research directions required to realize the concept of a LCB-based biorefinery. This comprehensive review serves as a valuable resource for researchers and industry professionals aiming to overcome the barriers associated with LCB utilization and facilitate the transition towards a sustainable and economically viable biorefinery approach.
Introduction
Energy is placed in the core of the most crucial sectors for development of human race. It acts as an essential factor for determining the socio-economic and high living standards of a country. Drastic increase in global human population and industrialization has exponentially increased the energy demand. Currently, fossil-based energy sources such as oil, coal, and natural gas are the major non-renewable resources are being used to accomplish the increasing energy demands. Concern over the exhausting fossil fuel resources and environmental pollution has extremely influenced the global scientific interest towards biofuels and other renewable energy sources. The transition from linear petroleum refinery to renewable biorefinery process is identified as a crucial step towards the progress of a viable industrialization, energy independence, and efficient management of greenhouse gas emissions. The US Department of Energy (US DOE) has identified 15 ideal target chemicals including biofuels and value-added bio-chemicals, as a result of conversion of biomass through a biorefinery platform [1]. Different types of biological materials such as agricultural residues, forest residues, and municipal solid waste; generally referred as lignocellulosic biomass (LCB) are being produced globally in massive capacities and remained underutilized. These biomasses are composed of several types of chemical entities, which may act as starting points for the production of large variety of value-added products, platform chemicals, or intermediates through different biochemical/chemical conversion routes. The value addition to the hugely abundant, cheaper, and renewable carbon source in the form of biomass may deliver substantial benefits in response to reducing fossil fuel resources and increasing environmental concerns [2].
Lignocellulosic biomass has diverse potential for value-added chemicals like biofuels, platform chemicals and materials production just because of dissimilarities in their compositions. The cellulose and hemicellulose are the two main carbohydrates present in LCB and are strongly bound with lignin making a complex and recalcitrant structure (Fig. 1). This complex structure is highly strong and resistant to depolymerization, therefore demands an effective pretreatment process to break the recalcitrance and avail the sugars for further processing steps [3,4,5,6]. The fractions of lignin, cellulose, and hemicellulose may differ with the type of plant and even at species level too. Plants of same species growing in different geographical locations and seasons may have different fractions of their chemical constituents’. Even the harvesting time may also contribute the difference in biochemical composition [7, 8]. This compositional variation in biomass suggests that a pretreatment method may suite or not for a particular biomass, and may require further optimization and standardization studies to scale up the process [9].
Pretreatment is the primary step posing crucial role in the economic viability of the overall bioconversion of LCB [10, 11]. A recent study has estimated the cost linked with the pretreatment process of LCB around 70–150 US dollars per ton of biomass [12]. The selection of pretreatment method and its conditions are highly dependent on the type and the chemical composition of the biomass. In the last few decades, a large number of pretreatment methods have been developed and validated for different types of LCB. These methods are broadly categorized under physical, chemical, physico-chemical, and biological methods [11]. For economic feasibility of any pretreatment method the energy and solvent requirements, depolymerization, conversion of lignocellulose, generation of inhibitory compounds, losses in terms of fermentable sugars should be taken into consideration. In addition to above requisites, the pretreatment method should be simple in operation and possesses minimum cost of operation and capital cost. Considering above points, the physical, chemical, and physico-chemical pretreatment methods are most likeable as compared to biological methods [13]. Since biological methods can incur higher costs compared to other pretreatment approaches. The higher cost and potentially lower productivity associated with biological methods and depends upon the type of biomass and specific process chosen for lignin fractionation. However, it is worth noting that biological pretreatment remains highly popular for anaerobic digestion [14, 15].
Biorefinery and Pretreatment: At a Glance
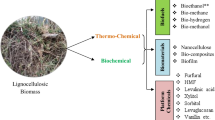
The biorefinery is a sustainable model that facilitates the usage of underutilized renewable natural biomass to replace the wide range of non-renewable fossil-based products and also focuses on minimizing the waste generation. The utilization of waste LCB especially agricultural residues as feedstock for biorefinery has great potential to bring down negative socio-economic and environmental impacts of linear petroleum economy. It is noteworthy that LCB do not demand additional crop land, thus have no adverse effect on the human or animal food economies [16]. However, a pretreatment method is an essential necessity to fractionate the biomass into its chemical constituents that can be further processed to liquid biofuels, biogas, biofertilizer, biopesticide, antimicrobial agent, cement composites, bioplastic, paints, coatings, resins, lubricants, single cell protein, animal feed, industrial enzymes, organic acids, carotenoids, xylose, xylitol etc. through biorefinery approach (Fig. 2). Pretreatment is recognised as the second most expensive processing step after downstream processing in bioconversion of LCB to biofuels and other value-added products generation. It has been estimated that about 20% of overall LCB processing cost is being incurred by pretreatment methods. A versatile pretreatment should fulfil the desired goals viz., (i) The possibility to use high solid (biomass) to liquid concentration, (ii) Should disintegrate the lignocellulosic structure by disrupting its structural bonds, (iii) Avoid the size reduction of biomass particles and inhibitors generation, (iv) Production of highly digestible solids to enhances enzymatic saccharification, (v) Fewer or no loss of carbohydrates (hemicellulose fraction must be preserved), (vi) Disrupt the lignin polymer and increase of surface area, (vii) Break the cellulose crystallinity and enhance the porosity of cellulose, (viii) Degradation of lignin polymers and partial depolymerization of hemicellulose, (ix) Allow the recovery of lignin fraction for reconstruction of valuable products, (x) Operation in reasonably sized and moderately priced reactors, and (xi) To be cost effective with low energy and power requirements. Previously, numerous pretreatment methods have been developed and employed for valorisation of various LCB (Fig. 3). They are encouragingly utilized to fractionate the LCB into intermediates for variety of industrial sectors like food, paper; they may substitute petroleum or may have new functionalities or better properties than the traditionally manufactured products such as: liquid biofuels [17]; chemicals like furfuraldehydes [18], 5-hydroxymethyl furfuraldehydes (HMF) [19], 2,5-furandicarboxylic acid [20], g-valerolactone (GVL) [21, 22], polymers [23, 24] and organic acids [25,26,27,28,29]; carbon fiber, adhesives, additives, dispersants [30,31,32,33,34,35]; and biogas [36, 37]. However, most of the existing pretreatment processes have several limitations such as sparse separation of holocellulosic and lignin complexes, elevated utilization of catalysts and/or energy, and liberation of by-products that inhibit successive conversion steps.
Physical Methods of Pretreatment: Decisive But Cost Intensive
Physical pretreatment avoids the addition of chemicals, enzymes or microorganisms during the pretreatment processes thus minimize the liberation of by-products and inhibitors for successive processes. Instead, physical pretreatment methods utilize the application of temperature, pressure, and mechanical force to obtain a smaller particle size and alter the lignocellulosic structure without modifying the chemical compositions of the component. Generally, physical treatments increase the porosity and the surface area of biomass. Physical pretreatment helps in reducing the crystallinity and degree of polymerization of cellulose present in biomass [38]. The physical methods include mechanical extrusion, milling, microwave assisted methods, ultrasound, pulsed electric field etc. (Table 1). Some common drawbacks of physical pretreatment methods include: inability to remove sufficient amount of lignin, high energy and capital-cost investment, and limited information about the mode of action prohibiting its large-scale implementation. Nevertheless, physical pretreatment methods are often essential step employed prior to subsequent chemical or biochemical processes.
Mechanical Extrusion
Methods like mechanical extrusion are well known to produce char and gaseous products. The LCB treated at 50–200 °C in combination to shearing and mixing to remove and shorten the biomass fibers [39]. Zheng et al. (2014) applied the mechanical extrusion using modified twin screw extruder to remove the xylose from steam exploded corncobs at 205 °C, which resulted in structural variation and enhanced glucose from 41 to 66% depending on different extrusion parameters. Optimization of extrusion method can be done by varying the operating temperature, screw speed and depend upon the cellulose content present in biomass (Table 1) [40]. A comparative study to elaborate the effect of mechanical extrusion on switch grass, blue stem, and prairie cord grass reported the glucose yield of about 28%, 66%, and 49% with screw speed of 200 rpm, 200 rpm, and 150 rpm and at 75 °C, 150 °C, and 100 °C temperature, respectively [41]. The mechanical extrusion method utilizes high amount of operational and energy cost, which makes it unsuitable for scale up to commercial setup [42].
Milling Pretreatment
The mechanical size reduction is generally recognized as a first type of physical pretreatment, where the biomass is subjected to various mechanical pretreatment processes to obtain smaller particles with a given size distribution. This size reduction of biomass triggers the mass and heat transfer for subsequent pretreatment steps. Coarse size reduction, hammering, shredding, chipping, grinding, and milling are several types of mechanical size reduction processes employed in biomass conversion route (Table 1). The selection of the milling process usually depends up on the moisture content of the biomass. Generally, two-roll mills and knife mills are used for dry biomass having moisture content to about 10–15% (wet basis), whereas colloid mills are suitable for wet biomass having moisture content more than 15–20% (wet basis). Comparatively, vibratory ball milling is a much advance method than an ordinary milling process in transforming cellulose crystallinity and magnifying biomass digestibility. Disk milling is recognized as a popular mechanical pretreatment which enhances the cellulose digestibility by producing fibres in comparison to hammer milling which produces finer bundles. On the other hand, the wet disk milling demands lower energy consumption over other traditional milling processes. Wet disk milling demands lower energy consumption and delivers efficient particle breakage, heat dissipation, reduced wear and tear of the milling equipment, improved process control (milling time, rotational speed, and concentration of the liquid medium), and increased throughput facilitated by the liquid medium [43]. According to an estimate, wet disk milling is 30% more energy efficient than dry milling methods [44].
The native complex structure of cellulose i.e. crystallinity and degree of polymerization should be modified by milling to make it more amenable to further processing. Cellulases are responsible for the hydrolysis of cellulose, but can only perform best when substrate is accessible for the enzymes. The accessibility of substrate to the enzymes can be enhanced by increasing surface area, porosity and decreasing crystallinity. Hence, milling of the biomass should be done prior to hydrolysis to increase the surface area and porosity of biomass and to decrease the crystallinity and degree of polymerization of cellulose. For the milling of wet biomass colloid mill, dissolver and fibrillator are mostly used, while for dry biomass hammer mill, extruder, cryogenic mill and roller are most suited [45] whereas ball milling can be performed for both the types of biomass. The reduction in particle size up to a certain limit is beneficial for the successive chemical and enzymatic pretreatment. The particle size in finishing biomass is dependent on the nature of physical pretreatment applied. Harvesting and pre-conditioning steps transform the wood stub and logs to coarse sizes of 10–45 mm, chipping can reduce the particles size to about 5–25 mm, and grinding/milling lowers the feedstock size to 0.4−2 mm [36]. It has been demonstrated that reduction in the particles size beyond 0.4 mm has no substantial impact on hydrolysis yields of lignocellulose, however it is mainly depends upon the type of biomass and subsequent processing techniques [46]. Biomass pretreatment with ball mill is cost expensive with respect to energy consumption, which is unfavorable for the industrial scale application. The ball milling method is also incapable of delignification, which makes it undesirable for enzymatic hydrolysis which requires higher accessibility/porosity to the substrate. The changes in crystallinity, degree of polymerization and surface area are highly dependent upon the type of biomass, milling method and on the duration of pretreatment [47,48,49].
Microwave Assisted Pretreatment (MWAP) Methods
During the last few years, microwave assisted methods for pretreatment of LCB have received more research attention. The fractionation of composite LCB during MWAP is a result of molecular collisions possessed by dielectric polarization. The MWAP methods are studied under two categories namely, atmospheric and high-pressure treatment. Comparatively, the high-pressure microwave treatments are performed at an elevated pressure and temperature (150−250 °C) in a closed reactor. Significant increase in bioethanol yield has been reported by several researchers using MWAP methods (Table 1). These methods are being employed progressively from lab to pilot scale [50]. The MWAP methods confer the advantages of lower activation energy of the reaction and short duration of reaction, which makes them suitable pretreatment method for LCB [51]. The recalcitrant structure of the biomass breaks down when heated under microwave as the irradiation penetrates inside the rigid structure of LCB [52, 53]. The recent scientific advancements in the area of MWAP were more focused on the design of novel microwave reactors for better biomass handling to enhance the scalability [50, 54].
The use of microwave irradiation in combination to other pretreatment method has been proven to a positive strategy for efficient degradation of LCB. It has been demonstrated that combination of mild alkali reagent (NaOH at different loadings 0.05–0.3 g/g biomass) with microwave pretreatment yielded a high concentration of reducing sugars (70–90%) [55]. Similarly, Nomanbhay et al. (2013) reported that microwave (180 W for 12 min) assisted alkali (NaOH 3% w/v) pretreatment increased the enzymatic hydrolysis to 5.8-fold from oil palm empty fruit bunch, in comparison to microwave pretreatment alone [56]. MWAP method using glycerol was found suitable to enhance the fermentable sugar yield from corncob along with eliminating the use of alkali metals [57]. Similarly, calcium chloride mediated microwave treatment enhanced the hemicellulose removal to 85.90% consequently specific surface area was increased to 168.93% as compared to untreated corn stalks [58]. In a recent study using maize distillery stillage, the high yield of enzymatic hydrolysis (75.8%) along with high sugar concentration (104.4 mg/g dry weight) were obtained with very low inhibitory compound formation [53].
Ultrasound Pretreatment
The effects of ultrasound pretreatment method on LCB have been explored for delignification and to reduce the recalcitrance of LCB to make it more accessible towards enzymatic hydrolysis (Table 1). The severity of the ultrasound pretreatment depends upon the frequency of the ultrasound waves, solvent used and on the type of reactor used [59].
The ultrasound based pretreatment methods work on principles of delignification and surface erosion. The benefits, it implies are: small pretreatment duration, lower heating requirement and use of lesser chemicals [60]. Increased biomethane production was reported using grape pomace when it was pretreated with ultrasound at 50 kHz and 25 °C for 40–70 min [61]. Chang et al. (2017) studied the effect of ultrasound in combination with ionic liquids along with SDS on the production of fermentable sugars. They observed that production of fermentable sugars, cellulose hydrolysis, and lignin removal were enhanced by 72.23%, 58.74%, and 21.01%, respectively as compared to ultrasound plus ionic liquid pretreatment. They further reported the increased delignification efficiency and decrease in cellulose crystallinity when SDS was added [62]. Saratale et al. (2020) developed an ultrasound assisted alkaline (NaOH) pretreatment method and were able to achieve 70% delignification of wheat straw and up to 84.5% hydrolysis yield along with 90% glucose and 65% xylose yield after enzymatic hydrolysis. The efficacy of the combined pretreatment method was significantly higher as compared to alkali (NaOH 2% w/v) and ultrasound (20 kHz for 30 min) treatments applied individually. The physicochemical analysis of biomass revealed that ultrasound assisted alkaline pretreatment method can break complex ultrastructure of LCB allowing enzymes to access the greater surface area of biomass [63]. Another study using ultrasound with 4% alkaline potassium permanganate for the pretreatment of spent coffee waste reported the maximum cellulose recovery of 98% with 46% delignification and an increase of 1.7-fold reducing sugar yield was observed after enzymatic hydrolysis as compared to spent coffee waste pretreated with KMnO4 alone [64].
Pulsed Electric Field (PEF) Pretreatment
Pulsed electric field is an emerging pretreatment technology that utilizes high voltage (0.1–100 kV/cm) exposure to the LCB for very short pulse duration of nanoseconds to milliseconds. This is carried out by a device carrying two electrodes which are inserted into the biomass/biomass-suspension. The exposure of the pulsed electric field to the LCB disrupts the structural components of biomass, causing an increase in permeability and hence facilitating the access of enzymes to the carbohydrate and lignin (Table 1) [65]. PEF is generally recognized as an energy efficient method to induce permeabilization of cellular components, while it has also been proposed as a novel stress promoter, capable of inducing the production of bioactive compounds. The electric pulses in the form of square or exponential waves are used for this pretreatment method. The setup of pulsed electric field consist of a pulse generator, a control system, data acquisition system and material handling vessel [45, 66]. This technology is at incipient stage, there are still significant room exist for the improvement in fractionation capability, for example, utilizing a combination of PEF method with organic solvent/salts may achieve higher yield.
Chemical Methods of Pretreatment: The Industrially Oriented Process
Chemical pretreatment methods utilize the application of special chemicals to destruct the rigid structure of LCB. In comparison to biological and physical pretreatment methods, chemical processes are employed more frequently because they are more effective to dissolve the complex lignocellulose. They are mostly operated at atmospheric pressure and across a range of ambient temperature employing oxidizing, acidic or alkali reagent. These chemicals are responsible to disrupt the intra- and inter polymer linkages of main organic components which in turn facilitate the hydrolysis of polymeric structure and degradation of lignin units [90]. Compared to other pretreatment methods, the chemical methods are considered to be very promising, since it can be quite effective in degrading more complex LCB and has incredible potential for upgrades in efficiency [91]. A range of chemical pretreatment methods were devised by different researchers till date for the fractionation of diverse LCB. These chemical pretreatment strategies have been proved to be effective on variety of LCB but in case of softwoods the yield of sugars is low. The chemical pretreatment processes are mostly performed utilizing alkali, acid, oxidizing agents, ionic liquids, and organic solvents to improve the biodegradability of LCB (Table 2).
Alkaline Pretreatment Methods
The alkaline pretreatment methods are more selective to delignify the biomass without disturbing carbohydrate content as much, though some amount of hemicellulose may also solubilize. Several strategies being followed by different researchers, focused on using different alkaline reagents like sodium hydroxide, calcium hydroxide, lime, ammonium hydroxide etc. (Table 2) [7]. Alkaline pretreatment of LCB can be carried out at ambient temperature for a residence period varying from few minutes to number of days and depending upon the type of alkali and lignin content [66]. Generally, alkaline pretreatment involves the alkali concentration between 1.0 and 8.0% and 90–220 °C temperature for a shorter residence time (5−100 min), or a dilute alkali concentration (0–2%) for several hours at 50–100 °C. The alkali processes affect the cellulosic residues by swelling mechanisms leading to a decrease in the crystallinity and degree of polymerization, subsequently improve the convenience of enzymes towards cellulosic fraction. It is effective in increasing the porosity and surface area accessible to enzymes of LCB, and hence improves the enzymatic hydrolysis of the biomass. These advantages make the alkaline methods suitable for the pretreatment of LCB [92, 93]. However, there are some major drawbacks associated with these methods such as longer reaction time which may vary from hours to days, high downstream processing cost required for recycling of salts, high post pretreatment cost involved for the neutralization of slurry, inability to fractionate the feedstocks with high lignin content, and sometimes adverse impacts on environment as well [94].
Alkaline pretreatment methods in combination to other chemical and physical pretreatment methods are getting much interest as the pretreatment efficiency and further hydrolysis can be significantly improved by doing so. A recent study conducted with NaOH/Urea with high biomass loading shown an increase of 31.89% in reducing sugar conversion as compared to untreated biomass. Further, FTIR and XRD analysis of cold NaOH/Urea pretreatment with 100% (w/v) biomass loading demonstrated effective disruption in lignin along with cellulose crystalline structure [95]. You et al. (2019) devised a cost effective pretreatment method (NaOH with CaO assisted by ultrasound), and were able to increase the lignin breakdown up to an extent of 60% and biogas production of more than 500mL per gram of total solid content of corn stover [83]. Another study optimized the combined effect of aqueous ammonia and ozone and found that biogas production from rice straw can be increased by 114.2−172.8% after 90 min ozonation and 9 h soaking in aqueous ammonia [96]. Chaudhary et al. (2012) observed 85% yield of reducing sugars from Saccharum spontaneum after aqueous ammonia pretreatment (soaked in 30% ammonia at 10% biomass loading for 40 days at 30 °C) followed by an acid hydrolysis (H2SO4 60% (v/v), 10% biomass loading at 30 °C for 4 h) process [8]. The advantage of ammonia based methods is that the ammonia can be most easily recovered and reused for further pretreatment [8]. Chundawat et al. (2020) reported a method using ammonia-salt based pretreatment at low temperatures, which decreased the cellulolytic enzyme requirements approximately by 50-fold [97].
Acidic Pretreatment Methods
Application of acids for the pretreatment of LCB is widely accepted especially in case of ethanol production (Table 2). During acid pretreatments, the hemicellulose present in biomass solubilizes in majority and a little extent of delignification takes place, this causes the increase in porosity of biomass [98]. Acid pretreatment methods are also capable of disrupting the lignocellulosic structure by cleaving interactions namely van der Waals forces, hydrogen and covalent bonds leading to the conversion of polymeric carbohydrates to oligomer sugar and monomer sugars [93, 99]. Phosphoric, acetic, sulfuric and hydrochloric acid are most commonly used acids for acid pretreatments [100]. The effectiveness of these methods depends upon the reaction time, temperature, concentration of acid, and biomass loading [99]. The major advantages of these methods include the use of highly commercial acids such as organic and inorganic acids, commercial level flexibility, greater hemicellulose removal and larger glucose yield. While the disadvantages associated are heavy cost of acid recovery, requirement of acid resistant vessels and equipment’s, and inhibitory/toxic compounds formation [93, 99]. However, it is important to know that in last two to three decades the economic and environmental aspects of the acid pretreatment methods have been greatly improved with time [93, 99].
Acidic pretreatment methods are studied under two categories (i) dilute acid pretreatment (ii) concentrated acid pretreatment. The dilute acid pretreatments (0–5% w/w) are generally performed at high temperature (120–210 °C) for a long residence time (5–30 min), whereas concentrated acid pretreatments (30–70% w/w) are carried out at low temperatures (< 100 °C) for long duration (30–120 min). The dilute acid pretreatment has high reaction rate and sugar conversion, but the energy required for the overall process is higher due to the higher temperature. Whereas, concentrated acid pretreatment requires relatively lower energy consumption, but the higher acidity leads to generation of fermentation inhibitors viz., furfurals, HMF, phenol derivatives, and aldehydes due to undesired degradation of carbohydrates. It has been estimated that the presence of these inhibitory compounds enhances the processing cost of the feedstock, ranging from 0.02 to 1.10 US dollars per kilogram of feedstock [12].
The dilute acid pretreatments are preferred, but harsh conditions of temperature and pressure are maintained for maximum recovery of fermentable sugars from LCB [101, 102]. Several variations have been made to search out the best operating parameters of acid pretreatment methods in terms of techno-economic and environmental aspects. These variations include the reactor configurations, operating conditions, severity factor etc. [99]. Among these parameters one of the most important parameter is combined severity factor (CSF) for low acid concentration, used to express the efficiency of the reaction parameters like temperature (T in oC), pH, reaction time (t in min), as given in the equation below [103, 104].
Where Tg is reference temperature (100oC) and pH is the initial pH value of reaction mixture.
Tang et al. 2021 explored the effect of dilute sulfuric acid in combination with humic acid (a natural surfactant) on pretreatment of waste wheat straw and were able to achieve 40.6% delignification and 96.2% hemicellulose removal, resulting in 92.9% enzymatic hydrolysis efficiency [105]. Pretreatment of Eucalyptus urophylla with GVL/H2O solution (4:1, v/v) containing 100 mM H2SO4 at 120 °C for 60 min, the highest glucose yield of 89.1% was achieved, furthermore 16.5% high purity lignin was also fractionated and could be used for its valorization applications [106].
Ionic Liquids Pretreatment Methods
Ionic liquids (ILs) have gained considerable attention recently for lignocellulosic biorefinery due to their recyclable capabilities. Their high solvation capabilities towards LCB leading to high yield of lignin free fermentable sugars make them promising candidate for future biorefinery platform [107, 108]. Ionic liquids are highly thermostable and possess low toxicity and work well at even low vapor pressure. They are highly selective in removing lignin and hemicellulose from LCB, yielding pure cellulose for subsequent hydrolysis process (Table 2) [4]. The main challenges with ionic liquid pretreatment are the complex processing, pH compatibility, toxicity, and higher costs involved [109]. Attempts have been made to synthesize low cost, environment friendly ILs using lignin and hemicellulose derived compounds such as [FurEt2NH][H2PO4], [VanEt2NH][H2PO4], and [p-AnisEt2NH][H2PO4] and were found effective compared to other ILs [110]. The ILs specifically recognized for the cellulose dissolution are imidazolium, pyridinium, ammonium, phosphonium or morpholinium based cations and anions. The capability of these ionic liquids to form strong hydrogen bonds with hydroxyl groups make them efficient for the cellulose dissolution [60].
Beside all the encouraging features of ILs, their commercial scale application is limited because of high viscosity, requirement of costly ionic liquids in large amounts, and negative effect of ILs on cellulases [111]. Hu et al. (2018) had overcome the problem of viscosity by adding 50% (w/w) water to the [Bmim]BF4 ionic liquid and were able to increase enzymatic hydrolysis efficiency up to 81.68% [112]. Pretreatment of Eucalyptus sawdust using ionic liquid in combination with microwave increased the delignification and cellulosic crystallinity deconstruction, which in turn increased the enzymatic hydrolysis [77]. Acetate based ionic liquid along with probe sonication pretreatment method was found capable of completely dissolving the bamboo biomass in merely 40 min, showing significant reduction in time for complete LCB dissolution and considerably change in thermo-physical characteristics of regenerated cellulose rich material [113]. Recently, Chuetor et al. (2022) optimized the pretreatment conditions of rice straw using 1-ethyl-3-methylimidazolium acetate and reported 89% more ethanol production than the untreated rice straw even after five recycles of IL [114]. Such findings have inspired the researchers to utilize the various IL pretreatment methods to make the idea of biorefinery an actuality.
Organosolv Pretreatment Methods
Organosolv pretreatment strategies have been established for industrial manufacturers of pulp & paper and bioethanol [11, 115]. Organosolv pretreatment method utilizes organic solvents like ethanol, acetone, ethylene glycol, formic acid, acetic acid, etc. for the delignification and hemicellulose solubilization (Table 2) [11]. The major advantage associated with organosolv pretreatment is that the solvent used can be recovered and reused. The organosolv methods of pretreatment are effective in fractionating LCB to its structural component: cellulose, hemicellulose, and lignin with high purity [93]. Organosolv pretreatment is in general carried out at an elevated temperature stretching from 100 oC to more than 200 oC. The acid catalysts like hydrochloric acid, sulfuric acid, oxalic acids etc. can also be used to increase the lignin extraction efficiency and in promoting hemicellulose breakdown [116, 117].
A recent study used methanol/dioxane binary solvent in addition to microwave irradiation to fractionate poplar wood into high quality cellulose, lignin, and monomeric sugar derivatives under mild condition of pretreatment i.e. 120 oC temperature for 10 min. Further, enzymatic hydrolysis of fractionated cellulose resulted in almost theoretical glucan conversion yield over 99% [78]. The high fractionation ability of organosolv pretreatment methods makes it the most encouraging biorefinery approach for meeting the objective of complete utilization of LCB without or minimal waste generation [118]. Kabakcı and Tanış (2020) compared different organosolv solvents (ethanol, alkaline glycerol, acetic acid, and formic acid/acetic acid/water) for the fractionation at atmospheric pressure and lower temperature and observed lignin precipitation yield ranging from 6.6% in ethanol organosolv and 42% in alkaline glycerol organosolv [119].
Ozonolysis Pretreatment
Ozonolysis is an oxidation reaction which utilizes ozone, a very strong oxidising agent for lignocellulose solubilization. The ozonolysis is much selective in breaking lignin component of the LCB leaving behind the cellulose and hemicellulose minimally affected (Table 2). Lignin is highly vulnerable towards the attack of ozone because of its electron rich properties and high contents of C = C bonds[120, 121]. The ozonolysis of lignin generates low molecular weight compounds majorly organic acids like formic and acetic acid, which results in lowering the pH of the solution up to 2. It poses several advantages over other pretreatment methods like selective delignification, milder reaction conditions, minimal inhibitory compound formation etc., while it also poses risks of fire and corrosion along with high cost of ozone generation, which limits its usage at industrial scale [122]. In a recent study, sugarcane straw was subjected to ozonolysis in different soaking media (acid, neutral and basic) and compared. The method preserved almost all the holocellulose while lignin content was reduced by 47% along with significant reduction in recalcitrance of the biomass, resulting in 60 and 71% yield of glucose and xylose respectively after enzymatic hydrolysis [123].
Deep Eutectic Solvent (DES) Pretreatment
In recent years, the development of mild and sustainable green process for industrial biorefining has got huge research attention (Table 2) [124]. Among different pretreatment methods, deep eutectic solvents are deliberately used as unique green solvents for pretreating LCB with added advantages like easier synthesis, extraordinary purity, lesser toxicity, biodegradable in nature, low melting points, high thermal stability, low volatility, non-flammability, and high air stability [125, 126], and most important they possess all the twelve principles of green chemistry[127]. Above all these DESs have outstanding competence of delignification from LCB [128,129,130,131,132,133,134,135]. The capability of delignification and hemicellulose removal from LCB during pretreatment can be determined by the ability of dissociation of protons by the DESs [136].
A recent study with DESs in addition with FeCl3, ZnCl2, CuCl2 and AlCl3 was used to treat hybrid Fountain grasses, the authors observed high level of cellulose retention (95.2%) and up to 99.5% cellulose enzymatic hydrolysis with DES when used with FeCl3 [137]. Whereas, in another study a two-step pretreatment strategy was followed, which includes liquid hot water extraction prior to mild acidic DES pretreatment. Out of three acidic DES (formic-, acetic-, and lactic acid-choline chloride) for selective delignification of poplar wood, the formic- and acetic acid DES contributed similar results with 76.4 and 76.5% delignification, while acetic acid deep eutectic acid was found most selective for the delignification with selectivity index of 7.9 [138].
In recent years, Natural deep eutectic solvents (NADES) have also gained more research interest for the delignification of LCB in a biofuels production route (Table 2). NADES are basically advanced liquid salts and are generated by mixing of cheap and readily available components such as quaternary ammonium salt (cholinium chloride) and naturally occurring hydrogen bond donor like amines, sugars, alcohols and carboxylic acids [139]. NADES offer several eco-friendly and economic advantages which include; cheaper synthesis, easy availability of components, easy synthesis, less toxicity, and high sustainability. They possess very low melting point even below 0oC, negligibly volatile, wide range of polarity, adjustable viscosity and great solubilization property for different molecules [139]. Few years back several stable NADES based on natural compounds (primary metabolites like organic acids, amino acids and sugars) were reported [140, 141]. Kumar et al. (2016) reported a method for high quality lignin and holocellulose separation in a single step from rice straw using NADESs. More than 60% delignification was achieved with greater than 90% purity of extracted lignin [142].
Physico–Chemical Methods of Pretreatment
In general, physico-chemical pretreatment methods exploits the physical properties like high temperature or pressure along with some chemicals to pretreat the LCB. Most of these methods are conducted at high temperature and sometimes with high pressure. In physicochemical pretreatment, lignin and xylan fractions of the lignocellulose are broken-down by altering the working conditions (like moisture, biomass particles size, solid loading, temperature, and pressure) in the presence or absence of a catalyst. The physico-chemical pretreatment methods include steam explosion, liquid hot water, wet oxidation, oxidative treatment, ammonia fiber explosion, supercritical carbon dioxide, sulfite pretreatment etc. These methods primarily increase the surface area for enzyme accessibility, decreases cellulose crystallinity, extract hemicellulose, and delignify biomass [5, 100]. Some recent studies on chemical pretreatment methods and their effects on further processing are summarized in Table 3.
Steam Explosion Pretreatment
Lignocellulosic pretreatment with steam explosion is a method of choice at commercial scale. Steam explosion is a potential thermochemical pretreatment method for LCB as it increases the accessibility of biomass towards the action of hydrolyzing enzymes by disrupting its structural components. This method also alters the cellulose crystallinity by dissolving the amorphous region of lignin and xylan. It affects the biomass by three actions i.e. steam heating, shearing of biomass due to sudden pressure release, and auto hydrolysis of glycosidic bonds [189]. The method employs saturated steam at high temperature (generally more than 160 oC) and high pressure (more than 5 bar) for few seconds to many minutes, causing disruption of structural components of cell wall and solubilization of acid soluble lignin and hemicellulose [93, 190]. The particle size of biomass, temperature, moisture content, solid loading, residence time, and the cumulative effect of temperature and duration (severity factor) are the main parameters affecting the efficacy of steam explosion pretreatment (Table 3). The starting material and biomass particles size determines the correlation among processing temperature and residence period. Low temperature for longer residence time has been reported better than high temperature for lower residence time treatment due to less fermentation inhibition product formation. The severity factor for steam explosion can be expressed as [191]:
where, logRo: the severity factor as a function of treatment time; T: temperature (oC); t: residence time (min); 14.75: activation energy where the process obeys first order kinetics and the Arrhenius temperature dependence.
The major advantage of steam explosion is that no chemicals are required and hence it is pollution free method, and it requires low energy and cheaper in recycling waste stream whereas the disadvantages account lower carbohydrate saccharification efficiency and generation of inhibitory products (like furfurals and HMF) [189].
A recent study using steam explosion in a semi continuous pre-pilot reactor for the pretreatment of switchgrass reported up to 88.3% enzymatic saccharification, which was found similar to the enzymatic hydrolysis of commercial cellulose pulp [192]. Matsakas and his team fractionated the spruce biomass using lignin first strategy. They applied hybrid organosolv-steam explosion pretreatment technology, and obtained the solid residue comprised of up to 72% w/w cellulose component and achieved 79.4% delignification. Though, the saccharification efficiency could reach only up to 61%. Furthermore, the high purity lignin is an added advantage to this method, which can be further used for high value added materials [193].
Liquid Hot Water (Hydrothermal) Pretreatment
Hydrothermal or liquid hot water pretreatment method employs the liquid water at high temperatures and high pressure to maintain the liquidity of water for the LCB. Generally, 160−240 oC temperature is maintained [60, 93, 190]. Though this method does not employ any chemical and catalyst to carry out the pretreatment, but acidic pH water at high temperature classifies the method under physico-chemical pretreatments (Table 3). The pretreatment conditions solubilize most of the hemicellulosic portion and partially delignify the LCB and make the cellulose more accessible towards hydrolysis [194]. A pH between 4 and 7 is desirable to limit the production of inhibitors and preventing sugar degradation and loss [189]. Also as in case of steam explosion, the sudden pressure release is not required during hydrothermal pretreatment method, therefore make this process easier to operate [189].
The severity factor is one of the most important parameter for the effectiveness of hydrothermal pretreatment as well. A lower severity factor (3.39) was reported ineffective in deconstruction of sugarcane straw while severity factor greater than 4.70 causes the loss of fermentable sugars and generates inhibitors like furfural and formation of pseudo-lignin structures, although around 97% hemicellulosic removal was achieved [195]. Li et al. (2017) evaluated the effect of hydrothermal pretreatment on poplar wood and observed substantial xylan solubilization (50–77%) along with changes in lignin structure such as decrease of β-O-4′ ether linkages and removal of cinnamyl alcohol end group and acetyl group [196]. The upcoming research should be focused in such a way that the maximal sugars can be recovered and with reasonable amount of hemicellulose solubilization.
Wet Oxidation Pretreatment
Wet oxidation pretreatment of LCB employs water at a temperature above 120 oC and pressure ranging from 0.5 to 2 MPa for residence period of up to 30 min. The method is efficient for the LCB having higher amounts of lignin. The major parameters affecting wet oxidation are pretreatment duration, oxygen/air pressure and temperature of reaction (Table 3). Water at elevated temperatures behaves like an acid and hence hydrolysis of biomass favors with increase in hydrogen ions with enhancement in temperature leading to lowering in pH of the reaction mixture [45]. Oxygen or air is used in this pretreatment as a catalyst [189]. This method of pretreatment generally acts upon hemicellulose leading to its solubilization and further hydrolysis into constituting monomeric sugars and delignification through decomposition of lignin in CO2, H2O and carboxylic acids [197, 198]. A study using wet oxidation (6% oxygen in the form of H2O2) pretreatment for oil palm empty fruit bunch reported an increase of 43% in methane yield as compared to control during mesophilic conditions of biogas production [199].
Oxidative Pretreatment
Oxidative pretreatment is carried out using different oxidizing agents such as air, oxygen, hydrogen peroxide, and ozone. The mechanism of the lignocellulose breakdown depends on the oxidizing agent used and/or the pretreatment conditions [38, 65]. This method involves the oxidation which mainly results in breakdown of lignin, hemicellulose depolymerization into its constituting monomeric sugars and organic acids and the partial breakdown of cellulose increasing the cellulose accessibility towards enzymatic hydrolysis [38, 200]. It has been reported that oxidative pretreatment is not selective and generally simultaneous degradation of heteropolymeric lignin along with hemicellulose and cellulose can be observed from the lignocellulose. During oxidative pretreatment delignification occurs because of oxidation of aromatic rings to the carboxylic acid with the help of oxidant as catalyst [201]. Beside oxidation, electrophilic substitution, side chain displacement, and alkyl-aryl linkage breakdown also occurs during oxidative pretreatment [200]. Fernández–Delgado et al. (2019) compared mild alkaline and oxidative pretreatment methods for biobutanol production from brewer’s spent grain and observed 62.8% glucose yield and 28.1% xylose yield after enzymatic hydrolysis. Later on after fermentation of fermentable sugars a maximum of 11.0 g/L butanol concentration along with 13.7 g ABE/L was observed [202].
Ammonia Fiber Explosion (AFEX) Pretreatment
The ammonia fiber explosion method is quite similar to the steam explosion pretreatment method in terms of operation; however, the pretreatment conditions are quite different because of volatile nature of ammonia. The digestion temperatures range between 50 and 110 °C are significantly lower in comparison to steam-explosion, therefore it requires lower energy and total investment cost [38]. The duration of pretreatment may vary from few minutes to hours depending upon the reaction temperature. AFEX pretreatment causes the swelling of biomass, disruption of structural components of the LCB, reducing crystallinity of the cellulose, increasing surface area and lignin-carbohydrate linkages disruption [93, 203]. The modified lignin structure increases the water-retention capacity; thereby facilitate the digestibility of feedstocks. AFEX pretreatment may be less effective with LCB that have high lignin content because the presence of lignin hinders the penetration and reaction of ammonia during the pretreatment process. However, AFEX pretreatment offers the advantage of not requiring much size reduction compared to other methods, as the rapid fiber expansion facilitates effective ammonia penetration even with larger particle sizes [189]. The degree of biomass disruption can be improved by process parameters optimization viz., treatment time, blow down pressure, temperature, and ammonia and water loading (Table 3). The major advantage of using AFEX is that ammonia can be easily recovered and reused for further pretreatment purpose.
A recent study was conducted by Zhao and his team to see the synergistic effect of hydrogen peroxide and ammonia on lignin and formulated that the oxidation and ammonolysis was led by hydrogen peroxide and ammonia, respectively. The oxidation occurs at S unit of lignin whereas ammonolysis occurs at ferulate and p-coumarate esters. They observed that the combined effect of hydroxide peroxide and ammonia is highly effective in degrading lignin and reducing the polydisperity index generating homogeneous lignin fragments [204]. Wang and co-workers (2019) performed the AFEX pretreatment of corn stover to extract lignin and utilize it for the lipid production. They observed that Rhodococcus opacus can utilize the AFEX treated lignin without any treatment and produced lipid up to 32 mg/L in 72 h utilizing 20% of total lignin. The lipid accumulated has the same composition as of the vegetable oil [205]. Mokomele et al. (2019) compared steam explosion and AFEX pretreatment on biogas production from sugarcane bagasse and observed that AFEX pretreatment gave the highest specific methane yield along with high methane content [206]. Unlike other biomass pretreatment processes, AFEX process does not generate inhibitors; therefore, the subsequent downstream processes become more efficient. There is a wide literature available on the advantages of AFEX process which highlights the lower moisture requirement, fewer formations of inhibitory sugar by-products, 100% recovery of feedstocks material, and positive effect on enzymatic hydrolysis in comparison to other chemical methods. Besides, there are some disadvantages such as high costs due to chemicals recycling and appropriate reactors requirement still need to be overcome.
Supercritical Carbon Dioxide (Sc-CO2) Based Pretreatment
Supercritical fluids possess the characteristics of gases and liquids. Their diffusivity and viscosity are same as of gases and density is same as of liquids [207]. The fluids at a temperature and pressure above their critical point are termed as supercritical fluids. They possess high diffusivity and solvolytic efficiency and the density of the fluid can be adjusted by changing the temperature and pressure[207]. In comparison to several supercritical fluids like ammonia, methanol and water, the CO2 is most widely used and well recognized supercritical fluid as it has lower critical temperature (31 oC), pressure (73.8 bar), and solubility (7.118 (cal/cm3)0.5) (Table 3) [4, 208]. These properties make it an efficient solvent for different monomeric and non-polar compounds and inefficient for the polymeric and polar compounds. Hence, it is very much suitable to fractionation of LCB as compared to other solvents. CO2 can easily penetrate the surface of lignocelluloses and reduces recalcitrance and enhances the penetrability of the cellulose [4]. Further, supercritical fluids can be considered as potential option for the pretreatment of LCB because of relatively mild pretreatment conditions, higher fermentable sugar yield, lower inhibitory compound generation, and greater exposure for enzymatic hydrolysis with reduced use of chemicals [209].
A very recent study employing Sc-CO2 for date pulp pretreatment, reported the temperature and pressure as significant parameters and achieved a maximum sugar yield of 74.1 g/100 g for Sc-CO2 pretreated date pulp at 10 MPa and 46 °C for 59 min [210]. Zhao et al. (2019) conducted pretreatment of different agricultural residues (corn stover, corn cob, and sorghum stalk) having 75 wt % moisture content using supercritical CO2, at low temperature (50–80 °C) and a pressure of 17.5–25.0 MPa. After enzymatic hydrolysis of pretreated biomass, a 3 to 4-fold increase in sugar yield was observed as compared to control [211]. In a similar study on blue agave bagasse 40% increase in total reducing sugars was achieved with enzymatic hydrolysis of Sc-CO2 pretreated biomass [212]. Mitraka et al. (2022) examined the effect of Sc-CO2 pretreatment on sewage sludge biogas production and observed an increase of 8.7% increase in methane production [213].
Sulfite Pretreatment
The Sulfite Pretreatment to Overcome the Recalcitrance of Lignocellulosics (SPORL) is often abbreviated as SPORL. This method in its first step employs the treatment of biomass with calcium or magnesium sulfite which in turn removes the hemicellulosic and lignin fractions followed by the reduction of size using disk miller [45, 190, 214]. Further new sulfite pretreatment methods have been developed which can utilize various sulfite or bisulfite solutions with broad range of pH and temperatures to weaken the structural components of the LCB (Table 3). This pretreatment method is specially intended and established for the woody biomass, softwoods such as pines and other coniferous plants, hardwood like poplar, willow tree, eucalyptus etc. [215]. This method has high scale up potential for industrial application. The reduced energy requirements for the size reduction and enhanced enzymatic saccharification of pretreated biomass within shorter period of reaction make this method much suitable for the fermentative ethanol production [215]. However, there are still some prone and cons associated with sulfite pretreatment of LCB, need to be tackled before real time implementation of this method at a commercial scale.
Biological Pretreatment Methods
Biological pretreatments are considered to be nontoxic and eco-friendly in nature, require less energy, and generate no inhibitors; hence, minimize the cost of subsequent downstream processing (though overall cost of biological pretreatment is higher than other pretreatment method). Several bacterial and fungal strains possess the enzymes (ligninases), which can degrade the LCB are being utilized for the biological pretreatment. Among the various lignin degrading microbes, white-rot fungi, especially selective delignifiers, have received much research interest over non-selective delignifiers due to high lignin-free cellulosic biomass yield [1]. Likewise, ligninases possess a critical role in the modification and degradation of lignin biopolymers. The structural rigidity of lignin, mandate the lignolytic microorganisms to produce an array of extracellular oxidative enzymes to achieve efficient degradation. Broadly, these extracellular oxidative machineries include; laccase, manganese peroxidases, lignin peroxidases, and versatile peroxidases that catalyse a direct role in lignin degradation [234]. Additionally, some other ancillary enzymes, such as glyoxal oxidases, feruloyl esterase, alcohol oxidases, aryl-alcohol dehydrogenase, and quinone reductase, which enhance the peroxidase activities and facilitate the lignin fractionation, are being recognized as the sole component of lignin degradation system of microbes [235].
The biological method of pretreatment in general used for the breakdown of cellulose and depolymerization of lignin [11, 236]. The efficiency of the biological pretreatment method highly depends on the temperature, size of biomass, moisture content, incubation time, aeration, accessible surface area, pH, and composition of culture media like source of carbon and nitrogen, cellulose crystallinity, inorganic and organic compounds, roles of enzymes and hydrolysates, microbial strain etc. These factors considerably affect the rate of pretreatment and play vital role in modifying physio-chemical structure of the LCB [237]. Some recent studies on biological pretreatment methods and their effects on further processing are summarized in Table 4.
Zanellati et al. (2021) explored and evaluated the phenol and furan degrading fungi for biological pretreatment of LCB. The study analyzed 40 fungal strains having capability to grow on different concentrations of furfural, vanillin, 4-hydroxybenzaldehyde, and syringaldehyde. Byssochlamysnivea MUT 6321 was found as the only fungus that can grow on all the four molecules present simultaneously [238]. Arora et al. (2016) evaluated the effect of biological pretreatment method for paddy straw. The saccharification efficiency of white-rot fungus, Trametes hirsute treated biomass was evaluated in comparison to steam pretreated biomass. The cellulose content in steam and biological pretreated biomass was found 39.5 and 37.6%, respectively, whereas lignin content was observed 14.2 and 4.7%, respectively. The saccharification efficiency was also found similar for steam as well as biological pretreatment [239]. The conventional pretreatment methods using single microbial culture are economically non-viable as it usually takes longer incubation time. Therefore, to overcome these obstacles mixed microbial consortium or microbial co-cultures should be utilized to increase the lignin degradation efficiency and to reduce pretreatment time.
Conclusions
The lignocellulosic biorefinery is a more sustainable future for human’s low value resources. Lignocellulosic biomass and pretreatment strategy represent the crucial factors in attaining these concepts. Selection of cheaper pretreatment process can ensure economical and sustainable viability of a biorefinery process. Furthermore, it becomes mandatory to completely operate the biomass source with minimum loss of mass and waste generation. Physico-chemical and thermo-chemical conversion processes have the potential to meet the technical and economical objectives of a biorefinery framework.
This review article concludes that the eco-friendly pretreatment processes which utilize the green solvents and low chemical loads receiving much interest as compared to the conventional chemical-based pretreatment methods and such methods are being developed and made feasible for industrial application. In recent past deep eutectic solvents have extensively been explored for their feasibility to fractionate variety of LCB. Further selection of pretreatment method highly depends upon type of the biomass to be treated and the purpose or end use for which biomass needs to be pretreated. Following points need to be taken care of while choosing a pretreatment method:
-
The pretreatment method employed should considerably be able to alter the complex three dimensional structure of the lignocellulose by depolymerizing its different components and decrystallization of cellulose.
-
The method should be able to increase the porosity and surface area of lignocellulose leading to increased enzyme accessibility for hydrolysis of biomass.
-
After hydrolysis of pretreated pulp, the total reducing sugar recovery should be close to the theoretical sugar yield.
-
The generation of inhibitory compounds and sugar degradation products should be at such minimum level that detoxification step prior to hydrolysis or fermentation should not be required.
-
Pretreated pulp or liquid processing like washing, separation, neutralization, etc. should be inexpensive and easy to process.
-
Equipments and vessels used for the pretreatment should be simple and capable of processing large quantities of solids with easy operation in terms of commissioning and decommissioning.
-
Pretreatment methods with mild process conditions should be favored.
-
The methods with lesser requirements of chemicals and water should be favored.
-
Pretreatment Methods with green and easy to recover chemicals should be favored to avoid further waste processing.
-
Pretreatment methods favoring higher extent of fractionation having capability of generating lignin and cellulose rich fraction of high purity should be favored. The purity lignin can further be valorized to variety of high value products.
Data Availability
Not Applicable.
Abbreviations
- AFEX:
-
Ammonia Fiber Explosion
- COD:
-
Chemical oxygen demand
- CRI:
-
Cellulose crystallinity index
- CSF:
-
Combined severity factor
- DES:
-
Deep Eutectic Solvent
- GVL:
-
g-valerolactone
- HMF:
-
5-hydroxymethyl furfuraldehydes
- IL:
-
Ionic liquid
- MW:
-
Microwave
- MWAP:
-
Microwave Assisted Pretreatment
- NADES:
-
Natural Deep Eutectic Solvent
- LCB:
-
Lignocellulosic biomass
- PEF:
-
Pulsed Electric Field
- S/L:
-
Solid/Liquid
- Sc-CO2 :
-
Supercritical Carbon Dioxide
- SDS:
-
Sodium dodecyl sulfate
- Temp:
-
Temperature
- TRS:
-
Total reducing sugars
- TS:
-
Total solids
- US:
-
Ultrasound
- US DOE:
-
US Department of Energy
References
Shankar, A., Saini, S., Sharma, K.K.: Fungal-integrated second-generation lignocellulosic biorefinery: utilization of agricultural biomass for co-production of lignocellulolytic enzymes, mushroom, fungal polysaccharides, and bioethanol. Biomass Convers. Biorefin. 1–15 (2022). https://doi.org/10.1007/S13399-022-02969-1
Cho, E.J., Trinh, L.T.P., Song, Y., Lee, Y.G., Bae, H.J.: Bioconversion of biomass waste into high value chemicals. Bioresour. Technol. 298, 122386 (2020). https://doi.org/10.1016/j.biortech.2019.122386
Lorenci Woiciechowski, A., Dalmas Neto, C.J., Porto de Souza Vandenberghe, L., de Carvalho Neto, D.P., Novak Sydney, A.C., Letti, L.A.J., Karp, S.G., Zevallos Torres, L.A., Soccol, C.R.: Lignocellulosic biomass: acid and alkaline pretreatments and their effects on biomass recalcitrance – conventional processing and recent advances. Bioresour. Technol. 304, 122848 (2020). https://doi.org/10.1016/j.biortech.2020.122848
Bhatia, S.K., Jagtap, S.S., Bedekar, A.A., Bhatia, R.K., Patel, A.K., Pant, D., Rajesh Banu, J., Rao, C.V., Kim, Y.G., Yang, Y.H.: Recent developments in pretreatment technologies on lignocellulosic biomass: effect of key parameters, technological improvements, and challenges. Bioresour. Technol. 300, 122724 (2020). https://doi.org/10.1016/j.biortech.2019.122724
Zabed, H., Sahu, J.N., Boyce, A.N., Faruq, G.: Fuel ethanol production from lignocellulosic biomass: an overview on feedstocks and technological approaches. Renew. Sustain. Energy Rev. 66, 751–774 (2016). https://doi.org/10.1016/j.rser.2016.08.038
Seidl, P.R., Goulart, A.K.: Pretreatment processes for lignocellulosic biomass conversion to biofuels and bioproducts. Curr. Opin. Green Sustain. Chem. 2, 48–53 (2016). https://doi.org/10.1016/j.cogsc.2016.09.003
Allison, G.G., Morris, C., Clifton-Brown, J., Lister, S.J., Donnison, I.S.: Genotypic variation in cell wall composition in a diverse set of 244 accessions of miscanthus. Biomass Bioenergy. 35(11), 4740–4747 (2011). https://doi.org/10.1016/j.biombioe.2011.10.008
Chaudhary, G., Singh, L.K., Ghosh, S.: Alkaline pretreatment methods followed by acid hydrolysis of Saccharum spontaneum for bioethanol production. Bioresour. Technol. 124, 111 (2012). https://doi.org/10.1016/j.biortech.2012.08.067
Boerjan, W., Ralph, J., Baucher, M.: Lignin biosynthesis. Annu. Rev. Plant. Biol. 54, 519–546 (2003). https://doi.org/10.1146/annurev.arplant.54.031902.134938
Amin, F.R., Khalid, H., Zhang, H., Rahman, S., Zhang, R., Liu, G., Chen, C.: Pretreatment methods of lignocellulosic biomass for anaerobic digestion. AMB Express. 7, 1–12 (2017). https://doi.org/10.1186/s13568-017-0375-4
Tu, W.C., Hallett, J.P.: Recent advances in the pretreatment of lignocellulosic biomass. Curr. Opin. Green Sustain. Chem. 20, 11–17 (2019). https://doi.org/10.1016/j.cogsc.2019.07.004
Preethi, Gunasekaran, M.: Indexing energy and cost of the pretreatment for economically efficient bioenergy generation. Front. Energy Res. 10, 1060599 (2023). https://doi.org/10.3389/fenrg.2022.1060599
Baral, N.R., Shah, A.: Comparative techno-economic analysis of steam explosion, dilute sulfuric acid, ammonia fiber explosion and biological pretreatments of corn stover. Bioresour. Technol. 232, 331–343 (2017). https://doi.org/10.1016/j.biortech.2017.02.068
Poddar, B.J., Nakhate, S.P., Gupta, R.K., Chavan, A.R., Singh, A.K., Khardenavis, A.A., Purohit, H.J.: A comprehensive review on the pretreatment of lignocellulosic wastes for improved biogas production by anaerobic digestion. Int. J. Environ. Sci. Technol. 194, 193429–193456 (2021). https://doi.org/10.1007/S13762-021-03248-8
Yadav, M., Balan, V., Varjani, S., Tyagi, V.K., Chaudhary, G., Pareek, N., Vivekanand, V.: Multidisciplinary pretreatment approaches to improve the bio-methane production from lignocellulosic biomass. BioEnergy Res. 16, 228–247 (2022). https://doi.org/10.1007/S12155-022-10489-Z
Saini, S., Chandel, A.K., Sharma, K.K.: Past practices and current trends in the recovery and purification of first generation ethanol: A learning curve for lignocellulosic ethanol. J. Clean. Prod. 268, 122357 (2020). https://doi.org/10.1016/J.JCLEPRO.2020.122357
De Op, B., Dusselier, M., Geboers, J., Holsbeek, J., Morré, E., Oswald, S., Giebeler, L., Sels, B.F.: Direct catalytic conversion of cellulose to liquid straight-chain alkanes. Energy Environ. Sci. 8, 230–240 (2015). https://doi.org/10.1039/c4ee01523a
Cai, C.M., Zhang, T., Kumar, R., Wyman, C.E.: Integrated furfural production as a renewable fuel and chemical platform from lignocellulosic biomass. J. Chem. Technol. Biotechnol. 89, 2–10 (2014). https://doi.org/10.1002/jctb.4168
Wang, T., Nolte, M.W., Shanks, B.H.: Catalytic dehydration of C6 carbohydrates for the production of hydroxymethylfurfural (HMF) as a versatile platform chemical. Green. Chem. 16, 548–572 (2014). https://doi.org/10.1039/c3gc41365a
Zhang, J., Li, J., Tang, Y., Lin, L., Long, M.: Advances in catalytic production of bio-based polyester monomer 2,5-furandicarboxylic acid derived from lignocellulosic biomass. Carbohydr. Polym. 130, 420–428 (2015). https://doi.org/10.1016/j.carbpol.2015.05.028
Tang, X., Zeng, X., Li, Z., Hu, L., Sun, Y., Liu, S., Lei, T., Lin, L.: Production of γ-valerolactone from lignocellulosic biomass for sustainable fuels and chemicals supply. Renew. Sustain. Energy Rev. 40, 608–620 (2014). https://doi.org/10.1016/j.rser.2014.07.209
Shuai, L., Questell-Santiago, Y.M., Luterbacher, J.S.: A mild biomass pretreatment using γ-valerolactone for concentrated sugar production. Green. Chem. 18, 937–943 (2016). https://doi.org/10.1039/c5gc02489g
Pang, J., Zheng, M., Sun, R., Wang, A., Wang, X., Zhang, T.: Synthesis of ethylene glycol and terephthalic acid from biomass for producing PET. Green. Chem. 18, 342–359 (2016). https://doi.org/10.1039/c5gc01771h
Hassanin, A.H., Hamouda, T., Candan, Z., Kilic, A., Akbulut, T.: Developing high-performance hybrid green composites. Compos. Part. B Eng. 92, 384–394 (2016). https://doi.org/10.1016/j.compositesb.2016.02.051
Pileidis, F.D., Titirici, M.M.: Levulinic acid biorefineries: new challenges for efficient utilization of Biomass. ChemSusChem 9, 562–582 (2016). https://doi.org/10.1002/cssc.201501405
Choi, S., Song, C.W., Shin, J.H., Lee, S.Y.: Biorefineries for the production of top building block chemicals and their derivatives. Metab. Eng. 28, 223–239 (2015). https://doi.org/10.1016/j.ymben.2014.12.007
Jansen, M.L.A., van Gulik, W.M.: Towards large scale fermentative production of succinic acid. Curr. Opin. Biotechnol. 30, 190–197 (2014). https://doi.org/10.1016/j.copbio.2014.07.003
Deng, W., Zhang, Q., Wang, Y.: Catalytic transformations of cellulose and cellulose-derived carbohydrates into organic acids. Catal. Today 234, 31–41 (2014). https://doi.org/10.1016/j.cattod.2013.12.041
Koutinas, A.A., Vlysidis, A., Pleissner, D., Kopsahelis, N., Lopez Garcia, I., Kookos, I.K., Papanikolaou, S., Kwan, T.H., Lin, C.S.K.: Valorization of industrial waste and by-product streams via fermentation for the production of chemicals and biopolymers. Chem. Soc. Rev. 43, 2587–2627 (2014). https://doi.org/10.1039/c3cs60293a
Strassberger, Z., Tanase, S., Rothenberg, G.: The pros and cons of lignin valorisation in an integrated biorefinery. RSC Adv. 4, 25310–25318 (2014). https://doi.org/10.1039/c4ra04747h
Li, S.H., Liu, S., Colmenares, J.C., Xu, Y.J.: A sustainable approach for lignin valorization by heterogeneous photocatalysis. Green. Chem. 18, 594–607 (2016). https://doi.org/10.1039/c5gc02109j
Duval, A., Lawoko, M.: A review on lignin-based polymeric, micro- and nano-structured materials. React. Funct. Polym. 85, 78–96 (2014). https://doi.org/10.1016/j.reactfunctpolym.2014.09.017
Laurichesse, S., Avérous, L.: Chemical modification of lignins: towards biobased polymers. Prog Polym. Sci. 39, 1266–1290 (2014). https://doi.org/10.1016/j.progpolymsci.2013.11.004
Lievonen, M., Valle-Delgado, J.J., Mattinen, M.L., Hult, E.L., Lintinen, K., Kostiainen, M.A., Paananen, A., Szilvay, G.R., Setälä, H., Österberg, M.: A simple process for lignin nanoparticle preparation. Green. Chem. 18, 1416–1422 (2016). https://doi.org/10.1039/c5gc01436k
Fache, M., Boutevin, B., Caillol, S.: Epoxy thermosets from model mixtures of the lignin-to-vanillin process. Green. Chem. 18, 712–725 (2016). https://doi.org/10.1039/c5gc01070e
Zheng, Y., Zhao, J., Xu, F., Li, Y.: Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog. Energy Combust. Sci. 42, 35–53 (2014). https://doi.org/10.1016/j.pecs.2014.01.001
Joelsson, E., Dienes, D., Kovacs, K., Galbe, M., Wallberg, O.: Combined production of biogas and ethanol at high solids loading from wheat straw impregnated with acetic acid: experimental study and techno-economic evaluation. Sustain. Chem. Process. 4, 1–19 (2016). https://doi.org/10.1186/s40508-016-0058-5
Jȩdrzejczyk, M., Soszka, E., Czapnik, M., Ruppert, A.M., Grams, J.: Physical and chemical pretreatment of lignocellulosic biomass. Second and Third Generation of Feedstocks. 143–196 (2019). https://doi.org/10.1016/B978-0-12-815162-4.00006-9
Maurya, D.P., Singla, A., Negi, S.: An overview of key pretreatment processes for biological conversion of lignocellulosic biomass to bioethanol. 3 Biotech 5, 597–609 (2015). https://doi.org/10.1007/s13205-015-0279-4
Zheng, J., Choo, K., Bradt, C., Lehoux, R., Rehmann, L.: Enzymatic hydrolysis of steam exploded corncob residues after pretreatment in a twin-screw extruder. Biotechnol. Rep. 3, 99–107 (2014). https://doi.org/10.1016/j.btre.2014.06.008
Karunanithy, C., Muthukumarappan, K., Gibbons, W.R.: Effect of extruder screw speed, temperature, and enzyme levels on sugar recovery from different biomasses. ISRN Biotechnol. (2013). https://doi.org/10.5402/2013/942810
Menon, V., Rao, M.: Trends in bioconversion of lignocellulose: biofuels, platform chemicals and biorefinery concept. Prog. Energy Combust. Sci. 38(4), 522–550 (2012). https://doi.org/10.1016/j.pecs.2012.02.002
Konde, K.S., Nagarajan, S., Kumar, V., Patil, S.V., Ranade, V.V.: Sugarcane bagasse based biorefineries in India: potential and challenges. Sustain. Energy Fuels 5, 52–78 (2021). https://doi.org/10.1039/D0SE01332C
Chelgani, S. C., Parian, M., Parapari, P. S., Ghorbani, Y., Rosenkranz, J.: A comparative study on the effects of dry and wet grinding on mineral flotation separation–a review. J. Mater. Res. Technol. 8(5), 5004–5011 (2019). https://doi.org/10.1016/j.jmrt.2019.07.053
Nauman Aftab, M., Iqbal, I., Riaz, F., Karadag, A., Tabatabaei, M.: Different pretreatment methods of lignocellulosic biomass for use in biofuel production. Biomass Bioenergy—Recent Trends Futur. Challenges. 1–24 (2019). https://doi.org/10.5772/intechopen.84995
De la Rubia, M.A., Fernández-Cegrí, V., Raposo, F., Borja, R.: Influence of particle size and chemical composition on the performance and kinetics of anaerobic digestion process of sunflower oil cake in batch mode. Biochem. Eng. J. 58–59, 162 (2011). https://doi.org/10.1016/J.BEJ.2011.09.010
Muller, C.D., Abu-Orf, M., Novak, J.T.: Application of mechanical shear in an internal-recycle for the enhancement of Mesophilic anaerobic digestion. Water Environ. Res 79, 297–304 (2007). https://doi.org/10.2175/106143006x101935
Jin, S., Chen, H.: Superfine grinding of steam-exploded rice straw and its enzymatic hydrolysis. Biochem. Eng. J. 30, 225–230 (2006). https://doi.org/10.1016/j.bej.2006.05.002
Mais, U., Esteghlalian, A.R., Saddler, J.N., Mansfield, S.D.: Enhancing the enzymatic hydrolysis of cellulosic materials using simultaneous ball milling. Appl. Biochem. Biotechnol. A 98–100, 815–832 (2002). https://doi.org/10.1385/ABAB:98-100:1-9:815
Li, H., Qu, Y., Yang, Y., Chang, S., Xu, J.: Microwave irradiation - a green and efficient way to pretreat biomass. Bioresour. Technol. 199, 34–41 (2016). https://doi.org/10.1016/j.biortech.2015.08.099
Kang, K., Nanda, S., Sun, G., Qiu, L., Gu, Y., Zhang, T., Zhu, M., Sun, R.: Microwave-assisted hydrothermal carbonization of corn stalk for solid biofuel production: optimization of process parameters and characterization of hydrochar. Energy 186, 115795 (2019). https://doi.org/10.1016/j.energy.2019.07.125
Chen, W.H., Tu, Y.J., Sheen, H.K.: Disruption of sugarcane bagasse lignocellulosic structure by means of dilute sulfuric acid pretreatment with microwave-assisted heating. Appl. Energy 88, 2726–2734 (2011). https://doi.org/10.1016/j.apenergy.2011.02.027
Mikulski, D., Kłosowski, G., Menka, A., Koim-Puchowska, B.: Microwave-assisted pretreatment of maize distillery stillage with the use of dilute sulfuric acid in the production of cellulosic ethanol. Bioresour. Technol. 278, 318–328 (2019). https://doi.org/10.1016/j.biortech.2019.01.068
Ooshima, H., Aso, K., Harano, Y., Yamamoto, T.: Microwave treatment of cellulosic materials for their enzymatic hydrolysis. Biotechnol. Lett. 6, 289–294 (1984). https://doi.org/10.1007/BF00129056
Hu, Z., Wen, Z.: Enhancing enzymatic digestibility of switchgrass by microwave-assisted alkali pretreatment. Biochem. Eng. J. 38, 369–378 (2008). https://doi.org/10.1016/j.bej.2007.08.001
Nomanbhay, S.M., Hussain, R., Palanisamy, K., Nomanbhay, S.M., Hussain, R., Palanisamy, K.: Microwave-assisted alkaline pretreatment and microwave assisted enzymatic saccharification of oil palm empty fruit bunch fiber for enhanced fermentable sugar yield. J. Sustain. Bioenergy Syst. 3, 7–17 (2013). https://doi.org/10.4236/JSBS.2013.31002
Zheng, A., Zhao, Z., Huang, Z., Zhao, K., Wei, G., Jiang, L., Wang, X., He, F., Li, H.: Overcoming biomass recalcitrance for enhancing sugar production from fast pyrolysis of biomass by microwave pretreatment in glycerol. Green. Chem. 17, 1167–1175 (2015). https://doi.org/10.1039/C4GC01724B
Li, H., Xu, J.: Optimization of microwave-assisted calcium chloride pretreatment of corn stover. Bioresour Technol. 127, 112–118 (2013). https://doi.org/10.1016/j.biortech.2012.09.114
Bussemaker, M.J., Zhang, D.: Effect of ultrasound on lignocellulosic biomass as a pretreatment for biorefinery and biofuel applications. Ind. Eng. Chem. Res. 52, 3563–3580 (2013). https://doi.org/10.1021/ie3022785
Cheah, W.Y., Sankaran, R., Show, P.L., Ibrahim, T., Baizura, T.N., Chew, K.W.: Pretreatment methods for lignocellulosic biofuels production: current advances, challenges and future prospects. Biofuel Res. J. 7, 1115–1127 (2020). https://doi.org/10.18331/BRJ2020.7.1.4
El Achkar, J.H., Lendormi, T., Salameh, D., Louka, N., Maroun, R.G., Lanoisellé, J.-L., Hobaika, Z.: Influence of pretreatment conditions on lignocellulosic fractions and methane production from grape pomace. Bioresour. Technol. 247, 881–889 (2018). https://doi.org/10.1016/j.biortech.2017.09.182
Chang, K.L., Han, Y.J., Wang, X.Q., Chen, X.M., Leu, S.Y., Liu, J., Peng, Y.P., Liao, Y.L., Potprommanee, L.: The effect of surfactant-assisted ultrasound-ionic liquid pretreatment on the structure and fermentable sugar production of a water hyacinth. Bioresour. Technol. 237, 27–30 (2017). https://doi.org/10.1016/j.biortech.2017.02.044
Saratale, G.D., Saratale, R.G., Varjani, S., Cho, S.K., Ghodake, G.S., Kadam, A., Mulla, S.I., Bharagava, R.N., Kim, D.S., Shin, H.S.: Development of ultrasound aided chemical pretreatment methods to enrich saccharification of wheat waste biomass for polyhydroxybutyrate production and its characterization. Ind. Crops Prod. 150, 112425 (2020). https://doi.org/10.1016/j.indcrop.2020.112425
Ravindran, R., Jaiswal, S., Abu-Ghannam, N., Jaiswal, A.K.: Evaluation of ultrasound assisted potassium permanganate pre-treatment of spent coffee waste. Bioresour. Technol 224, 680–687 (2017). https://doi.org/10.1016/j.biortech.2016.11.034
Kumar, B., Bhardwaj, N., Agrawal, K., Chaturvedi, V., Verma, P.: Current perspective on pretreatment technologies using lignocellulosic biomass: an emerging biorefinery concept. Fuel Process. Technol. 199, 106244 (2020). https://doi.org/10.1016/j.fuproc.2019.106244
Kumar, P., Barrett, D.M., Delwiche, M.J., Stroeve, P.: Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind. Eng. Chem. Res 48, 3713–3729 (2009). https://doi.org/10.1021/ie801542g
Kozłowski, K., Lewicki, A., Czekała, W., Wójtowicz, A., Kupryaniuk, K., Drózdz, D.: Extrusion pretreatment of maize straw-case study for a polish biogas plants. Int. Agrophys. 33, 527–535 (2019). https://doi.org/10.31545/intagr/113548
Wang, Z., He, X., Yan, L., Wang, J., Hu, X., Sun, Q., Zhang, H.: Enhancing enzymatic hydrolysis of corn stover by twin-screw extrusion pretreatment. Ind. Crops Prod. 143, 111960 (2020). https://doi.org/10.1016/j.indcrop.2019.111960
Pérez-Rodríguez, N., García-Bernet, D., Domínguez, J.M.: Faster methane production after sequential extrusion and enzymatic hydrolysis of vine trimming shoots. Environ. Chem. Lett. 16, 295–299 (2018). https://doi.org/10.1007/s10311-017-0668-5
Gu, B.J., Wang, J., Wolcott, M.P., Ganjyal, G.M.: Increased sugar yield from pre-milled Douglas-fir forest residuals with lower energy consumption by using planetary ball milling. Bioresour. Technol. 251, 93–98 (2018). https://doi.org/10.1016/j.biortech.2017.11.103
Zhang, H., Zhang, P., Ye, J., Wu, Y., Liu, J., Fang, W., Xu, D., Wang, B., Yan, L., Zeng, G.: Comparison of various pretreatments for ethanol production enhancement from solid residue after rumen fluid digestion of rice straw. Bioresour. Technol. 247, 147–156 (2018). https://doi.org/10.1016/j.biortech.2017.09.065
Bychkov, A.L., Podgorbunskikh, E.M., Ryabchikova, E.I., Lomovsky, O.I.: The role of mechanical action in the process of the thermomechanical isolation of lignin. Cellulose 25, 1–5 (2018). https://doi.org/10.1007/s10570-017-1536-y
Liu, H., Zhang, Y.X., Hou, T., Chen, X., Gao, C., Han, L., Xiao, W.: Mechanical deconstruction of corn stover as an entry process to facilitate the microwave-assisted production of ethyl levulinate. Fuel Process. Technol. 174, 53–60 (2018). https://doi.org/10.1016/j.fuproc.2018.02.011
Bai, X., Wang, G., Yu, Y., Wang, D., Wang, Z.: Changes in the physicochemical structure and pyrolysis characteristics of wheat straw after rod-milling pretreatment. Bioresour Technol. 250, 770–776 (2018). https://doi.org/10.1016/j.biortech.2017.11.085
López-Linares, J.C., García-Cubero, M.T., Lucas, S., González-Benito, G., Coca, M.: Microwave assisted hydrothermal as greener pretreatment of brewer’s spent grains for biobutanol production. Chem. Eng. J. 368, 1045–1055 (2019). https://doi.org/10.1016/j.cej.2019.03.032
Yunqing, Z., Han, X., Hui, J., Weihua, Q.: Enhance the ethanol fermentation performance of kitchen waste using microwave pretreatment. Biomass Convers. Biorefin. 1–10 (2022). https://doi.org/10.1007/S13399-022-02330-6
Hou, X., Wang, Z., Sun, J., Li, M., Wang, S., Chen, K., Gao, Z.: A microwave-assisted aqueous ionic liquid pretreatment to enhance enzymatic hydrolysis of eucalyptus and its mechanism. Bioresour. Technol. 272, 99–104 (2019). https://doi.org/10.1016/j.biortech.2018.10.003
Zhang, Y., Wang, Z., Feng, J., Pan, H.: Maximizing utilization of poplar wood by microwave-assisted pretreatment with methanol/dioxane binary solvent. Bioresour. Technol. 300, 122657 (2020). https://doi.org/10.1016/j.biortech.2019.122657
Kumar, B., Verma, P.: Optimization of microwave-assisted pretreatment of Rice Straw with FeCl3 in combination with H3PO4 for improving enzymatic hydrolysis. Adv. Plant. Microb. Biotechnol. 41–48 (2019). https://doi.org/10.1007/978-981-13-6321-4_7.
Bhardwaj, N., Kumar, B., Verma, P.: Microwave-assisted pretreatment using alkali metal salt in combination with orthophosphoric acid for generation of enhanced sugar and bioethanol. Biomass Convers. Biorefinery. (2020). https://doi.org/10.1007/s13399-020-00640-1
Dawid, M., Grzegorz, K.: Microwave-assisted hydrotropic pretreatment as a new and highly efficient way to cellulosic ethanol production from maize distillery stillage. Appl. Microbiol. Biotechnol. 105, 3381–3392 (2021). https://doi.org/10.1007/S00253-021-11258-2
Dhandayuthapani, K., Sarumathi, V., Selvakumar, P., Temesgen, T., Asaithambi, P., Sivashanmugam, P.: Study on the ethanol production from hydrolysate derived by ultrasonic pretreated defatted biomass of chlorella sorokiniana NITTS3. Chem. Data Collect 31, 100641 (2021). https://doi.org/10.1016/J.CDC.2020.100641
You, Z., Pan, S.-Y., Sun, N., Kim, H., Chiang, P.-C.: Enhanced corn-stover fermentation for biogas production by NaOH pretreatment with CaO additive and ultrasound. J. Clean. Prod. 238, 117813 (2019). https://doi.org/10.1016/j.jclepro.2019.117813
Chen, S.J., Chen, X., Zhu, M.J.: Xylose recovery and bioethanol production from sugarcane bagasse pretreated by mild two-stage ultrasonic assisted dilute acid. Bioresour. Technol 345, 126463 (2022). https://doi.org/10.1016/J.BIORTECH.2021.126463
Szwarc, D., Szwarc, K.: Use of a pulsed electric field to improve the biogas potential of maize silage. Energies 14, 119 (2020). https://doi.org/10.3390/EN14010119
Safavi, S.M., Unnthorsson, R.: Enhanced methane production from pig slurry with pulsed electric field pre-treatment. Environ. Technol. 39, 479–489 (2018). https://doi.org/10.1080/09593330.2017.1304455
Szwarc, D., Głowacka, K.: Increasing the biogas potential of rapeseed straw using pulsed electricfield pre-treatment. Energies 14, 8307 (2021). https://doi.org/10.3390/EN14248307
Putranto, A.W., Abida, S.H., Adrebi, K.: Lignocellulosic analysis of corncob biomass by using non thermal pulsed electric field NaOH pretreatment. Proc. Int. Conf. Sustain. Biomass. 202, 273–280 (2021). https://doi.org/10.2991/AER.K.210603.049
Wang, Y.Z., Zhang, L., Xu, T., Ding, K.: Improving lignocellulose enzymatic saccharification in a bioreactor with an applied electric field. Ind. Crops Prod. 109, 404–409 (2017). https://doi.org/10.1016/J.INDCROP.2017.08.061
Norrrahim, M.N.F., Ilyas, R.A., Nurazzi, N.M., Rani, M.S.A., Atikah, M.S.N., Shazleen, S.S.: Chemical pretreatment of lignocellulosic biomass for the production of bioproducts: an overview. Appl. Sci. Eng. Prog. 14, 588–605 (2021). https://doi.org/10.14416/J.ASEP.2021.07.004
Jose, D., Kitiborwornkul, N., Sriariyanun, M., Keerthi, K.: A review on chemical pretreatment methods of lignocellulosic biomass: recent advances and progress. Appl. Sci. Eng. Prog. 15, 6210–6210 (2022). https://doi.org/10.14416/J.ASEP.2022.08.001
Kim, J.S., Lee, Y.Y., Kim, T.H.: A review on alkaline pretreatment technology for bioconversion of lignocellulosic biomass. Bioresour. Technol. 199, 42–48 (2016). https://doi.org/10.1016/j.biortech.2015.08.085
Rezania, S., Oryani, B., Cho, J., Talaiekhozani, A., Sabbagh, F., Hashemi, B., Rupani, P.F., Mohammadi, A.A.: Different pretreatment technologies of lignocellulosic biomass for bioethanol production: an overview. Energy 199, 117457 (2020). https://doi.org/10.1016/j.energy.2020.117457
Bali, G., Meng, X., Deneff, J.I., Sun, Q., Ragauskas, A.J.: The effect of alkaline pretreatment methods on cellulose structure and accessibility. ChemSusChem 8, 275–279 (2015). https://doi.org/10.1002/cssc.201402752
Dong, L., Cao, G., Wu, J., Liu, B., Xing, D., Zhao, L., Zhou, C., Feng, L., Ren, N.: High-solid pretreatment of rice straw at cold temperature using NaOH/urea for enhanced enzymatic conversion and hydrogen production. Bioresour. Technol. 287, 121399 (2019). https://doi.org/10.1016/j.biortech.2019.121399
Ai, P., Zhang, X., Dinamarca, C., Elsayed, M., Yu, L., Xi, J., Mei, Z.: Different effects of ozone and aqueous ammonia in a combined pretreatment method on rice straw and dairy manure fiber for enhancing biomethane production. Bioresour. Technol. 282, 275–284 (2019). https://doi.org/10.1016/j.biortech.2019.03.021
Chundawat, S.P.S., Sousa, L.C., Roy, S., Yang, Z., Gupta, S., Pal, R., Zhao, C., Liu, S.-H., Petridis, L., O’Neill, H., Pingali, S.V.: Ammonia-salt solvent promotes cellulosic biomass deconstruction under ambient pretreatment conditions to enable rapid soluble sugar production at ultra-low enzyme loadings. Green. Chem. 22, 204–218 (2020). https://doi.org/10.1039/C9GC03524A
Chaudhary, G., Ghosh, S.: Two-reactor, continuous culture fermentation for fuel ethanol production from lignocellulosic acid hydrolysate using Zymomonas mobilis and Scheffersomyces stipitis. RSC Adv. 4, 36412 (2014). https://doi.org/10.1039/c4ra02377c
Solarte-Toro, J.C., Romero-García, J.M., Martínez-Patiño, J.C., Ruiz-Ramos, E., Castro-Galiano, E., Cardona-Alzate, C.A.: Acid pretreatment of lignocellulosic biomass for energy vectors production: a review focused on operational conditions and techno-economic assessment for bioethanol production. Renew. Sustain. Energy Rev. 107, 587–601 (2019). https://doi.org/10.1016/j.rser.2019.02.024
Mosier, N., Wyman, C., Dale, B., Elander, R., Lee, Y.Y., Holtzapple, M., Ladisch, M.: Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour. Technol. 96, 673–686 (2005). https://doi.org/10.1016/j.biortech.2004.06.025
Loow, Y.-L., Wu, T.Y., Md. Jahim, J., Mohammad, A.W., Teoh, W.H.: Typical conversion of lignocellulosic biomass into reducing sugars using dilute acid hydrolysis and alkaline pretreatment. Cellulose 23, 1491–1520 (2016). https://doi.org/10.1007/s10570-016-0936-8
Rabemanolontsoa, H., Saka, S.: Various pretreatments of lignocellulosics. Bioresour. Technol. 199, 83–91 (2016). https://doi.org/10.1016/j.biortech.2015.08.029
Agbor, V.B., Cicek, N., Sparling, R., Berlin, A., Levin, D.B.: Biomass pretreatment: fundamentals toward application. Biotechnol. Adv. 29, 675–685 (2011). https://doi.org/10.1016/j.biotechadv.2011.05.005
Hsu, T.-C., Guo, G.-L., Chen, W.-H., Hwang, W.-S.: Effect of dilute acid pretreatment of rice straw on structural properties and enzymatic hydrolysis. Bioresour. Technol. 101, 4907–4913 (2010). https://doi.org/10.1016/j.biortech.2009.10.009
Tang, W., Wu, X., Huang, C., Ling, Z., Lai, C., Yong, Q.: Natural surfactant-aided dilute sulfuric acid pretreatment of waste wheat straw to enhance enzymatic hydrolysis efficiency. Bioresour. Technol. 324, 124651 (2021). https://doi.org/10.1016/J.BIORTECH.2020.124651
Sun, S.N., Chen, X., Tao, Y.H., Cao, X.F., Li, M.F., Wen, J.L., Nie, S.X., Sun, R.C.: Pretreatment of Eucalyptus urophylla in γ-valerolactone/dilute acid system for removal of non-cellulosic components and acceleration of enzymatic hydrolysis. Ind. Crops Prod. 132, 21–28 (2019). https://doi.org/10.1016/J.INDCROP.2019.02.004
Brandt, A., Gräsvik, J., Hallett, J.P., Welton, T.: Deconstruction of lignocellulosic biomass with ionic liquids. Green. Chem. 15, 550–583 (2013). https://doi.org/10.1039/C2GC36364J
Moniruzzaman, M., Goto, M.: Ionic liquid pretreatment of lignocellulosic biomass for enhanced enzymatic delignification. Appl. Ionic Liquids. Biotechnol. Springer International Publishing, 168, 61–77, (2019). https://doi.org/10.1007/10_2018_64
Singh, S.: Designing tailored microbial and enzymatic response in ionic liquids for lignocellulosic biorefineries. Biophys. Rev. 10, 911–913 (2018). https://doi.org/10.1007/s12551-018-0418-3
Socha, A.M., Parthasarathi, R., Shi, J., Pattathil, S., Whyte, D., Bergeron, M., George, A., Tran, K., Stavila, V., Venkatachalam, S., Hahn, M.G., Simmons, B.A., Singh, S.: Efficient biomass pretreatment using ionic liquids derived from lignin and hemicellulose. Proc. Natl. Acad. Sci. 111, E3587 LP-E3595 (2014). https://doi.org/10.1073/pnas.1405685111
Sriariyanun, M., Kitiborwornkul, N., Tantayotai, P., Rattanaporn, K., Show, P.L.: One-pot ionic liquid-mediated bioprocess for pretreatment and enzymatic hydrolysis of rice straw with ionic liquid-tolerance bacterial cellulase. Bioengineering 9, 17 (2022). https://doi.org/10.3390/BIOENGINEERING9010017
Hu, X., Cheng, L., Gu, Z., Hong, Y., Li, Z., Li, C.: Effects of ionic liquid/water mixture pretreatment on the composition, the structure and the enzymatic hydrolysis of corn stalk. Ind. Crops Prod. 122, 142–147 (2018). https://doi.org/10.1016/j.indcrop.2018.05.056
Rahim, A.H.A., Man, Z., Sarwono, A., Muhammad, N., Khan, A.S., Hamzah, W.S.W., Yunus, N.M., Elsheikh, Y.A.: Probe sonication assisted ionic liquid treatment for rapid dissolution of lignocellulosic biomass. Cellulose 27, 2135–2148 (2020). https://doi.org/10.1007/s10570-019-02914-y
Chuetor, S., Panakkal, E.J., Ruensodsai, T., Cheenkachorn, K., Kirdponpattara, S., Cheng, Y.S., Sriariyanun, M.: Improvement of enzymatic saccharification and ethanol production from rice straw using recycled ionic liquid: the effect of anti-solvent mixture. Bioengineering 9, 115 (2022). https://doi.org/10.3390/BIOENGINEERING9030115
Johansson, A., Aaltonen, O., Ylinen, P.: Organosolv pulping—methods and pulp properties. Biomass 13, 45–65 (1987). https://doi.org/10.1016/0144-4565(87)90071-0
Park, N., Kim, H.-Y., Koo, B.-W., Yeo, H., Choi, I.-G.: Organosolv pretreatment with various catalysts for enhancing enzymatic hydrolysis of pitch pine (Pinus rigida). Bioresour Technol. 101, 7046–7053 (2010). https://doi.org/10.1016/j.biortech.2010.04.020
Zhao, X., Cheng, K., Liu, D.: Organosolv pretreatment of lignocellulosic biomass for enzymatic hydrolysis. Appl. Microbiol. Biotechnol. 82, 815–827 (2009)
Shuai, L., Luterbacher, J.: Organic solvent effects in biomass conversion reactions. ChemSusChem 9, 133–155 (2016). https://doi.org/10.1002/cssc.201501148
Başakçılardan Kabakcı, S., Tanış, M.H.: Pretreatment of lignocellulosic biomass at atmospheric conditions by using different organosolv liquors: a comparison of lignins. Biomass Convers. Biorefin. 11, 2869–2880 (2020). https://doi.org/10.1007/s13399-020-00677-2
Ab Rasid, N.S., Zainol, M.M., Amin, N.A.S.: Pretreatment of agroindustry waste by ozonolysis for synthesis of biorefinery products. Refining Biomass Residues for Sustainable Energy and Bioproducts. Elsvier, 303–336 (2020). https://doi.org/10.1016/B978-0-12-818996-2.00014-4
Travaini, R., Marangon-Jardim, C., Colodette, J.L., Morales-Otero, M.: Bolado-Rodríguez, S.: Ozonolysis in Pretreatment of Biomass. Elsevier. 105–135 (2015). https://doi.org/10.1016/B978-0-12-800080-9.00007-4
Travaini, R., Martín-Juárez, J., Lorenzo-Hernando, A., Bolado-Rodríguez, S.: Ozonolysis: an advantageous pretreatment for lignocellulosic biomass revisited. Bioresour. Technol. 199, 2–12 (2016). https://doi.org/10.1016/j.biortech.2015.08.143
Orduña Ortega, J., Mora Vargas, J.A., Perrone, O.M., Metzker, G., Gomes, E., da Silva, R., Boscolo, M.: Soaking and ozonolysis pretreatment of sugarcane straw for the production of fermentable sugars. Ind. Crops Prod. 145, 111959 (2020). https://doi.org/10.1016/J.INDCROP.2019.111959
Jose, D., Tawai, A., Divakaran, D., Bhattacharyya, D., Venkatachalam, P., Tantayotai, P., Sriariyanun, M.: Integration of deep eutectic solvent in biorefining process of lignocellulosic biomass valorization. Bioresour. Technol. Rep. 21, 101365 (2023). https://doi.org/10.1016/J.BITEB.2023.101365
del Contreras-Gámez, M., Galán-Martín, M., Seixas, Ã., da Costa Lopes, N., Silvestre, A.M., Castro, A.: Deep eutectic solvents for improved biomass pretreatment: current status and future prospective towards sustainable processes. Bioresour. Technol. 369, 128396 (2023). https://doi.org/10.1016/J.BIORTECH.2022.128396
Bhagwat, P., Amobonye, A., Singh, S., Pillai, S.: Deep eutectic solvents in the pretreatment of feedstock for efficient fractionation of polysaccharides: current status and future prospects. Biomass Convers. Biorefin. 121, 171–195 (2021). https://doi.org/10.1007/S13399-021-01745-X
Xu, H., Peng, J., Kong, Y., Liu, Y., Su, Z., Li, B., Song, X., Liu, S., Tian, W.: Key process parameters for deep eutectic solvents pretreatment of lignocellulosic biomass materials: a review. Bioresour. Technol. 310, 123416 (2020). https://doi.org/10.1016/j.biortech.2020.123416
Alvarez-Vasco, C., Ma, R., Quintero, M., Guo, M., Geleynse, S., Ramasamy, K.K., Wolcott, M., Zhang, X.: Unique low-molecular-weight lignin with high purity extracted from wood by deep eutectic solvents (DES): a source of lignin for valorization. Green. Chem. 18, 5133–5141 (2016). https://doi.org/10.1039/C6GC01007E
Chen, Z., Reznicek, W.D., Wan, C.: Deep eutectic solvent pretreatment enabling full utilization of switchgrass. Bioresour. Technol. 263, 40–48 (2018). https://doi.org/10.1016/j.biortech.2018.04.058
Chen, Z., Wan, C.: Ultrafast fractionation of lignocellulosic biomass by microwave-assisted deep eutectic solvent pretreatment. Bioresour. Technol. 250, 532–537 (2018). https://doi.org/10.1016/j.biortech.2017.11.066
Kim, K.H., Dutta, T., Sun, J., Simmons, B., Singh, S.: Biomass pretreatment using deep eutectic solvents from lignin derived phenols. Green Chem. 20(4), 809–815 (2018). https://doi.org/10.1039/C7GC03029K
Procentese, A., Johnson, E., Orr, V., Garruto Campanile, A., Wood, J.A., Marzocchella, A., Rehmann, L.: Deep eutectic solvent pretreatment and subsequent saccharification of corncob. Bioresour. Technol. 192, 31–36 (2015). https://doi.org/10.1016/j.biortech.2015.05.053
Shen, X.-J., Wen, J.-L., Mei, Q.-Q., Chen, X., Sun, D., Yuan, T.-Q., Sun, R.-C.: Facile fractionation of lignocelluloses by biomass-derived deep eutectic solvent (DES) pretreatment for cellulose enzymatic hydrolysis and lignin valorization. Green. Chem. 21, 275–283 (2019). https://doi.org/10.1039/C8GC03064B
Xia, Q., Liu, Y., Meng, J., Cheng, W., Chen, W., Liu, S., Liu, Y., Li, J., Yu, H.: Multiple hydrogen bond coordination in three-constituent deep eutectic solvents enhances lignin fractionation from biomass. Green Chem. 20, 2711–2721 (2018). https://doi.org/10.1039/C8GC00900G
Chen, Z., Ragauskas, A., Wan, C.: Lignin extraction and upgrading using deep eutectic solvents. Ind. Crops Prod. 147, 112241 (2020). https://doi.org/10.1016/j.indcrop.2020.112241
Tang, X., Zuo, M., Li, Z., Liu, H., Xiong, C., Zeng, X., Sun, Y., Hu, L., Liu, S., Lei, T., Lin, L.: Green processing of lignocellulosic biomass and its derivatives in deep eutectic solvents. ChemSusChem 10, 2696–2706 (2017). https://doi.org/10.1002/cssc.201700457
Wang, Z.K., Li, H., Lin, X.C., Tang, L., Chen, J.J., Mo, J.W., Yu, R.S., Shen, X.J.: Novel recyclable deep eutectic solvent boost biomass pretreatment for enzymatic hydrolysis. Bioresour. Technol. 307, 123237 (2020). https://doi.org/10.1016/j.biortech.2020.123237
Tian, D., Guo, Y., Hu, J., Yang, G., Zhang, J., Luo, L., Xiao, Y., Deng, S., Deng, O., Zhou, W., Shen, F.: Acidic deep eutectic solvents pretreatment for selective lignocellulosic biomass fractionation with enhanced cellulose reactivity. Int. J. Biol. Macromol. 142, 288–297 (2020). https://doi.org/10.1016/j.ijbiomac.2019.09.100
Radošević, K., Ćurko, N., Srček, G., Cvjetko Bubalo, V., Tomašević, M., Kovačević Ganić, M., Radojčić Redovniković, K.: Natural deep eutectic solvents as beneficial extractants for enhancement of plant extracts bioactivity. LWT - Food Sci. Technol. 73, 45–51 (2016). https://doi.org/10.1016/j.lwt.2016.05.037
Choi, Y.H., van Spronsen, J., Dai, Y., Verberne, M., Hollmann, F., Arends, I.W.C.E., Witkamp, G.-J., Verpoorte, R.: Are natural deep eutectic solvents the missing link in understanding cellular metabolism and physiology? Plant Physiol. 156, 1701–1705 (2011). https://doi.org/10.1104/pp.111.178426
Dai, Y., van Spronsen, J., Witkamp, G.-J., Verpoorte, R., Choi, Y.H.: Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 766, 61–68 (2013). https://doi.org/10.1016/j.aca.2012.12.019
Kumar, A.K., Parikh, B.S., Pravakar, M.: Natural deep eutectic solvent mediated pretreatment of rice straw: bioanalytical characterization of lignin extract and enzymatic hydrolysis of pretreated biomass residue. Environ. Sci. Pollut. Res. 23, 9265–9275 (2016). https://doi.org/10.1007/s11356-015-4780-4
Łukajtis, R., Rybarczyk, P., Kucharska, K., Konopacka-Łyskawa, D., Słupek, E., Wychodnik, K., Kamiński, M.: Optimization of saccharification conditions of lignocellulosic biomass under alkaline pre-treatment and enzymatic hydrolysis. Energies 11, 886 (2018). https://doi.org/10.3390/EN11040886
Wang, W., Wang, X., Zhang, Y., Yu, Q., Tan, X., Zhuang, X., Yuan, Z.: Effect of sodium hydroxide pretreatment on physicochemical changes and enzymatic hydrolysis of herbaceous and woody lignocelluloses. Ind. Crops Prod. 145, 112145 (2020). https://doi.org/10.1016/J.INDCROP.2020.112145
Shimizu, F.L., Monteiro, P.Q., Ghiraldi, P.H.C., Melati, R.B., Pagnocca, F.C., de Souza, W., Sant’Anna, C., Brienzo, M.: Acid, alkali and peroxide pretreatments increase the cellulose accessibility and glucose yield of banana pseudostem. Ind. Crops Prod. 115, 62–68 (2018). https://doi.org/10.1016/J.INDCROP.2018.02.024
Az-Zahraa, B., Zakaria, S., Daud, M.F.B., Jaafar, S.N.S.: Removal of oil palm trunk lignin in ammonium hydroxide pretreatment. AIP Conference Proceeding. 020017 (1940). (2018). https://doi.org/10.1063/1.5027932
Liu, E., Das, L., Zhao, B., Crocker, M., Shi, J.: Impact of dilute sulfuric acid, ammonium hydroxide, and ionic liquid pretreatments on the fractionation and characterization of engineered switchgrass. BioEnergy Res. 104(10), 1079–1093 (2017). https://doi.org/10.1007/S12155-017-9868-X
Mejica, G.F.C., Unpaprom, Y., Whangchai, K., Ramaraj, R.: Cellulosic-derived bioethanol from Limnocharisflava utilizing alkaline pretreatment. Biomass Convers. Biorefin. 125(12), 1737–1743 (2021). https://doi.org/10.1007/S13399-020-01218-7
Chi, X., Liu, C., Bi, Y.H., Yu, G., Zhang, Y., Wang, Z., Li, B., Cui, Q.: A clean and effective potassium hydroxide pretreatment of corncob residue for the enhancement of enzymatic hydrolysis at high solids loading. RSC Adv. 9, 11558–11566 (2019). https://doi.org/10.1039/C9RA01555H
Shen, J., Zheng, Q., Zhang, R., Chen, C., Liu, G.: Co-pretreatment of wheat straw by potassium hydroxide and calcium hydroxide: Methane production, economics, and energy potential analysis. J. Environ. Manage. 236, 720–726 (2019). https://doi.org/10.1016/J.JENVMAN.2019.01.046
Xie, X., Feng, X., Chi, S., Zhang, Y., Yu, G., Liu, C., Li, Z., Li, B., Peng, H.: A sustainable and effective potassium hydroxide pretreatment of wheat straw for the production of fermentable sugars. Bioresour. Technol. Rep. 3, 169–176 (2018). https://doi.org/10.1016/J.BITEB.2018.07.014
Rani, P., Pathak, V.V., Bansal, M.: Co-digestion of wheat straw and animal manure pretreated with calcium hydroxide for biomethane production: kinetic study. Curr. Res. Green. Sustain. Chem. 4, 100145 (2021). https://doi.org/10.1016/J.CRGSC.2021.100145
Chang, M., Li, D., Wang, W., Chen, D., Zhang, Y., Hu, H., Ye, X.: Comparison of sodium hydroxide and calcium hydroxide pretreatments on the enzymatic hydrolysis and lignin recovery of sugarcane bagasse. Bioresour. Technol. 244, 1055–1058 (2017). https://doi.org/10.1016/J.BIORTECH.2017.08.101
Yu, Z., Du, Y., Shang, X., Zheng, Y., Zhou, J.: Enhancing fermentable sugar yield from cassava residue using a two-step dilute ultra-low acid pretreatment process. Ind. Crops Prod. 124, 555–562 (2018). https://doi.org/10.1016/J.INDCROP.2018.08.029
Kuglarz, M., Alvarado-Morales, M., Dąbkowska, K., Angelidaki, I.: Integrated production of cellulosic bioethanol and succinic acid from rapeseed straw after dilute-acid pretreatment. Bioresour. Technol. 265, 191–199 (2018). https://doi.org/10.1016/J.BIORTECH.2018.05.099
Santos, C.C., de Souza, W., Sant Anna, C., Brienzo, M.: Elephant grass leaves have lower recalcitrance to acid pretreatment than stems, with higher potential for ethanol production. Ind. Crops Prod. 111, 193–200 (2018). https://doi.org/10.1016/J.INDCROP.2017.10.013
Yildirim, O., Ozkaya, B., Altinbas, M., Demir, A.: Statistical optimization of dilute acid pretreatment of lignocellulosic biomass by response surface methodology to obtain fermentable sugars for bioethanol production. Int. J. Energy Res. 45, 8882–8899 (2021). https://doi.org/10.1002/ER.6423
Avila-Gaxiola, J., Velarde-Escobar, O.J., Millan-Almaraz, J.R., Ramos-Brito, F., Atondo-Rubio, G., Yee-Rendon, C., Avila-Gaxiola, E.: Treatments to improve obtention of reducing sugars from agave leaves powder. Ind. Crops Prod. 112, 577–583 (2018). https://doi.org/10.1016/J.INDCROP.2017.12.039
Zhang, J., Shen, W., Collings, C., Meulen, V., Fox, K.A., Vázquez, B.G., Ramos, L.M., Dumesic, J.A., Ding, S.Y.: Visualizing plant cell wall changes proves the superiority of hydrochloric acid over sulfuric acid catalyzed γ-valerolactone pretreatment. Chem. Eng. J. 412, 128660 (2021). https://doi.org/10.1016/J.CEJ.2021.128660
Dziekońska-Kubczak, U., Berłowska, J., Dziugan, P., Patelski, P., Pielech-Przybylska, K., Balcerek, M.: Nitric Acid Pretreatment of Jerusalem Artichoke Stalks for Enzymatic Saccharification and Bioethanol Production. Energies. 11, 2153 (2018). https://doi.org/10.3390/EN11082153
Morais, W.G., Pacheco, T.F., Corrêa, P.S., Martins, A.A., Mata, T.M., Caetano, N.S.: Acid pretreatment of sugarcane biomass to obtain hemicellulosic hydrolisate rich in fermentable sugar. Energy Rep. 6, 18–23 (2020). https://doi.org/10.1016/J.EGYR.2020.10.015
Liu, Z., Wan, X., Wang, Q., Tian, D., Hu, J., Huang, M., Shen, F., Zeng, Y.: Performances of a multi-product strategy for bioethanol, lignin, and ultra-high surface area carbon from lignocellulose by PHP (phosphoric acid plus hydrogen peroxide) pretreatment platform. Renew. Sustain. Energy Rev. 150, 111503 (2021). https://doi.org/10.1016/J.RSER.2021.111503
Sahu, S., Pramanik, K.: Evaluation and optimization of organic acid pretreatment of cotton gin waste for enzymatic hydrolysis and bioethanol production. Appl. Biochem. Biotechnol. 186, 1047–1060 (2018). https://doi.org/10.1007/S12010-018-2790-7/FIGURES/3
Wang, P., Chen, Y.M., Wang, Y., Lee, Y.Y., Zong, W., Taylor, S., McDonald, T., Wang, Y.: Towards comprehensive lignocellulosic biomass utilization for bioenergy production: efficient biobutanol production from acetic acid pretreated switchgrass with Clostridium saccharoperbutylacetonicum N1-4. Appl. Energy 236, 551–559 (2019). https://doi.org/10.1016/J.APENERGY.2018.12.011
Rattanaporn, K., Roddecha, S., Sriariyanun, M., Cheenkachorn, K.: Improving saccharification of oil palm shell by acetic acid pretreatment for biofuel production. Energy Procedia 141, 146–149 (2017). https://doi.org/10.1016/J.EGYPRO.2017.11.027
Papa, G., Feldman, T., Sale, K.L., Adani, F., Singh, S., Simmons, B.A.: Parametric study for the optimization of ionic liquid pretreatment of corn stover. Bioresour. Technol. 241, 627–637 (2017). https://doi.org/10.1016/J.BIORTECH.2017.05.167
Sorn, V., Chang, K.L., Phitsuwan, P., Ratanakhanokchai, K., Dong, C., Di: Effect of microwave-assisted ionic liquid/acidic ionic liquid pretreatment on the morphology, structure, and enhanced delignification of rice straw. Bioresour. Techno.l 293, 121929 (2019). https://doi.org/10.1016/J.BIORTECH.2019.121929
Sharma, V., Nargotra, P., Bajaj, B.K.: Ultrasound and surfactant assisted ionic liquid pretreatment of sugarcane bagasse for enhancing saccharification using enzymes from an ionic liquid tolerant aspergillus assiutensis VS34. Bioresour. Technol. 285, 121319 (2019). https://doi.org/10.1016/J.BIORTECH.2019.121319
Zhang, W., Liu, J., Wang, Y., Sun, J., Huang, P., Chang, K.: Effect of ultrasound on ionic liquid-hydrochloric acid pretreatment with rice straw. Biomass Convers. Biorefinery 2020. 115, 11, 1749–1757 (2020). https://doi.org/10.1007/S13399-019-00595-Y
Chang, K.L., Chen, X.M., Wang, X.Q., Han, Y.J., Potprommanee, L., Liu, J., Liao, Y.L., Ning, X., Sun, S., Huang, Q.: Impact of surfactant type for ionic liquid pretreatment on enhancing delignification of rice straw. Bioresour. Technol. 227, 388–392 (2017). https://doi.org/10.1016/J.BIORTECH.2016.11.085
Trinh, L.T.P., Lee, Y.J., Park, C.S., Bae, H.J.: Aqueous acidified ionic liquid pretreatment for bioethanol production and concentration of produced ethanol by pervaporation. J. Ind. Eng. Chem. 69, 57–65 (2019). https://doi.org/10.1016/J.JIEC.2018.09.008
Ramezani, N., Sain, M.: Thermal and physiochemical characterization of Lignin extracted from wheat straw by organosolv process. J. Polym. Environ. 267(26), 3109–3116 (2018). https://doi.org/10.1007/S10924-018-1199-2
Choi, J.H., Jang, S.K., Kim, J.H., Park, S.Y., Kim, J.C., Jeong, H., Kim, H.Y., Choi, I.G.: Simultaneous production of glucose, furfural, and ethanol organosolv lignin for total utilization of high recalcitrant biomass by organosolv pretreatment. Renew. Energy 130, 952–960 (2019). https://doi.org/10.1016/J.RENENE.2018.05.052
Tang, C., Shan, J., Chen, Y., Zhong, L., Shen, T., Zhu, C., Ying, H.: Organic amine catalytic organosolv pretreatment of corn stover for enzymatic saccharification and high-quality lignin. Bioresour. Technol. 232, 222–228 (2017). https://doi.org/10.1016/J.BIORTECH.2017.02.041
Joy, S.P., Krishnan, C.: Modified organosolv pretreatment for improved cellulosic ethanol production from sorghum biomass. Ind. Crops Prod. 177, 114409 (2022). https://doi.org/10.1016/J.INDCROP.2021.114409
Pascal, K., Ren, H., Sun, F.F., Guo, S., Hu, J., He, J.: Mild acid-catalyzed atmospheric glycerol organosolv pretreatment effectively improves enzymatic hydrolyzability of lignocellulosic biomass. ACS Omega 4, 20015–20023 (2019). https://doi.org/10.1021/ACSOMEGA.9B02993/ASSET
Singh, S., Sinha, R., Kundu, S.: Role of organosolv pretreatment on enzymatic hydrolysis of mustard biomass for increased saccharification. Biomass Convers. Biorefinery 125(12), 1657–1668 (2021). https://doi.org/10.1007/S13399-020-01251-6
Tan, X., Zhang, Q., Wang, W., Zhuang, X., Deng, Y., Yuan, Z.: Comparison study of organosolv pretreatment on hybrid pennisetum for enzymatic saccharification and lignin isolation. Fuel 249, 334–340 (2019). https://doi.org/10.1016/J.FUEL.2019.03.117
Andersen, S.L.F., Castoldi, R., Garcia, J.A.A., Bracht, A., Peralta, R.A., de Lima, E.A., Helm, C.V., Moreira, R., de Peralta, F.P.M.: Improving enzymatic saccharification of Eucalyptus grandis branches by ozone pretreatment. Wood Sci. Technol. 2018(531), 5349–5369 (2018). https://doi.org/10.1007/S00226-018-1061-7
Domański, J., Marchut-Mikołajczyk, O., Polewczyk, A., Januszewicz, B.: Ozonolysis of straw from Secale cereale L. for anaerobic digestion. Bioresour. Technol. 245, 394–400 (2017). https://doi.org/10.1016/J.BIORTECH.2017.08.090
Patil, R., Cimon, C., Eskicioglu, C., Goud, V.: Effect of ozonolysis and thermal pre-treatment on rice straw hydrolysis for the enhancement of biomethane production. Renew. Energy 179, 467–474 (2021). https://doi.org/10.1016/J.RENENE.2021.07.048
Perrone, O.M., Moretti, M.M., de Bordignon, S., Pereira, S.E., da Silva, J.C., Gomes, R., Boscolo, E.: Improving cellulosic ethanol production using ozonolysis and acid as a sugarcane biomass pretreatment in mild conditions. Bioresour Technol. Rep. 13, 100628 (2021). https://doi.org/10.1016/J.BITEB.2021.100628
Li, C., Wang, L., Chen, Z., Li, Y., Luo, X., Zhao, F.: Ozonolysis of wheat bran in subcritical water for enzymatic saccharification and polysaccharide recovery. J. Supercrit. Fluids 168, 105092 (2021). https://doi.org/10.1016/J.SUPFLU.2020.105092
Hossain, M.A., Rahaman, M.S., Yelle, D., Shang, H., Sun, Z., Renneckar, S., Dong, J., Tulaphol, S., Sathitsuksanoh, N.: Effects of polyol-based deep eutectic solvents on the efficiency of rice straw enzymatic hydrolysis. Ind. Crops Prod. 167, 113480 (2021). https://doi.org/10.1016/J.INDCROP.2021.113480
Zhao, Z., Chen, X., Ali, M.F., Abdeltawab, A.A., Yakout, S.M., Yu, G.: Pretreatment of wheat straw using basic ethanolamine-based deep eutectic solvents for improving enzymatic hydrolysis. Bioresour. Technol. 263, 325–333 (2018). https://doi.org/10.1016/J.BIORTECH.2018.05.016
Sharma, V., Nargotra, P., Sharma, S., Bajaj, B.K.: Efficacy and functional mechanisms of a novel combinatorial pretreatment approach based on deep eutectic solvent and ultrasonic waves for bioconversion of sugarcane bagasse. Renew. Energy 163, 1910–1922 (2021). https://doi.org/10.1016/J.RENENE.2020.10.101
Liang, Y., Duan, W., An, X., Qiao, Y., Tian, Y., Zhou, H.: Novel betaine-amino acid based natural deep eutectic solvents for enhancing the enzymatic hydrolysis of corncob. Bioresour. Technol. 310, 123389 (2020). https://doi.org/10.1016/j.biortech.2020.123389
Lu, Q., Yan, D., Wu, P., Chen, L., Yagoub, A.E.G.A., Ji, Q., Yu, X., Zhou, C.: Ultrasound-NATDES/DMSO system for corn straw biomass conversion into platform compounds. Renew. Energy 190, 675–683 (2022). https://doi.org/10.1016/J.RENENE.2022.03.154
Kumari, D., Singh, R.: Pretreatment of lignocellulosic wastes for biofuel production: A critical review. Renew. Sustain. Energy Rev. 90, 877–891 (2018). https://doi.org/10.1016/j.rser.2018.03.111
Moreno, A.D., Tomás-Pejó, E., Ballesteros, M., Negro, M.J.: Pretreatment technologies for lignocellulosic biomass deconstruction within a biorefinery perspective. Biofuels Altern. Feed. Convers. Process. Prod. Liq. Gaseous Biofuels (2019). https://doi.org/10.1016/b978-0-12-816856-1.00016-6
Overend, R.P., Chornet, E.: Fractionation of lignocellulosics by steam-aqueous pretreatments. Philos. Trans. R. Soc. London Series A. 321(1561), 523–536 (1987). https://doi.org/10.1098/rsta.1987.0029
Bonfiglio, F., Cagno, M., Rey, F., Torres, M., Böthig, S., Menéndez, P., Mussatto, S.I.: Pretreatment of switchgrass by steam explosion in a semi-continuous pre-pilot reactor. Biomass Bioenergy 121, 41–47 (2019). https://doi.org/10.1016/j.biombioe.2018.12.013
Matsakas, L., Raghavendran, V., Yakimenko, O., Persson, G., Olsson, E., Rova, U., Olsson, L., Christakopoulos, P.: Lignin-first biomass fractionation using a hybrid organosolv – steam explosion pretreatment technology improves the saccharification and fermentability of spruce biomass. Bioresour. Technol. 273, 521–528 (2019). https://doi.org/10.1016/j.biortech.2018.11.055
Cara, C., Romero, I., Oliva, J.M., Sáez, F., Castro, E.: Liquid hot water pretreatment of olive tree pruning residues. Appl. Biochem. Biotechnol. 137-140 (1-12), 379–394 (2007). https://doi.org/10.1007/s12010-007-9066-y
Batista, G., Souza, R.B.A., Pratto, B., dos Santos-Rocha, M.S.R., Cruz, A.J.G.: Effect of severity factor on the hydrothermal pretreatment of sugarcane straw. Bioresour. Technol. 275, 321–327 (2019). https://doi.org/10.1016/j.biortech.2018.12.073
Li, M., Cao, S., Meng, X., Studer, M., Wyman, C.E., Ragauskas, A.J., Pu, Y.: The effect of liquid hot water pretreatment on the chemical-structural alteration and the reduced recalcitrance in poplar. Biotechnol. Biofuels 10, 1–13 (2017). https://doi.org/10.1186/S13068-017-0926-6/SCHEMES/1
Banerjee, S., Sen, R., Pandey, R.A., Chakrabarti, T., Satpute, D., Giri, B.S., Mudliar, S.: Evaluation of wet air oxidation as a pretreatment strategy for bioethanol production from rice husk and process optimization. Biomass Bioenerg. 33, 1680–1686 (2009). https://doi.org/10.1016/j.biombioe.2009.09.001
Bjerre, A.B., Olesen, A.B., Fernqvist, T., Plöger, A., Schmidt, A.S.: Pretreatment of wheat straw using combined wet oxidation and alkaline hydrolysis resulting in convertible cellulose and hemicellulose. Biotechnol. Bioeng. 49, 568–577 (1996). https://doi.org/10.1002/(SICI)1097-0290(19960305)49:5%3c568::AID-BIT10%3e3.0.CO;2-6
Lee, J.T.E., Khan, M.U., Tian, H., Ee, A.W.L., Lim, E.Y., Dai, Y., Tong, Y.W., Ahring, B.K.: Improving methane yield of oil palm empty fruit bunches by wet oxidation pretreatment: mesophilic and thermophilic anaerobic digestion conditions and the associated global warming potential effects. Energy Convers. Manag. 225, 113438 (2020). https://doi.org/10.1016/J.ENCONMAN.2020.113438
Hendriks, A.T.W.M., Zeeman, G.: Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 100, 10–18 (2009). https://doi.org/10.1016/j.biortech.2008.05.027
Harmsen, P., Huijgen, W., Bermudez, L., Bakker, R.: Literature review of physical and chemical pretreatment processes for lignocellulosic biomass. Wageningen UR Food & Biobased Research 1184, (2010). https://edepot.wur.nl/150289
Fernández-Delgado, M., Plaza, P.E., Coca, M., García-Cubero, M.T., González-Benito, G., Lucas, S.: Comparison of mild alkaline and oxidative pretreatment methods for biobutanol production from brewer’s spent grains. Ind. Crops Prod. 130, 409–419 (2019). https://doi.org/10.1016/J.INDCROP.2018.12.087
Behera, S., Arora, R., Nandhagopal, N., Kumar, S.: Importance of chemical pretreatment for bioconversion of lignocellulosic biomass. Renew. Sustain. Energy Rev. 36, 91–106 (2014). https://doi.org/10.1016/j.rser.2014.04.047
Zhao, C., Qiao, X., Shao, Q., Hassan, M., Ma, Z., Yao, L.: Synergistic effect of hydrogen peroxide and ammonia on lignin. Ind. Crops Prod. 146, 112177 (2020). https://doi.org/10.1016/j.indcrop.2020.112177
Wang, Z., Li, N., Pan, X.: Transformation of ammonia fiber expansion (AFEX) corn stover lignin into microbial lipids by Rhodococcus opacus. Fuel 240, 119–125 (2019). https://doi.org/10.1016/j.fuel.2018.11.081
Mokomele, T., da Costa Sousa, L., Balan, V., van Rensburg, E., Dale, B.E., Görgens, J.F.: Incorporating anaerobic co-digestion of steam exploded or ammonia fiber expansion pretreated sugarcane residues with manure into a sugarcane-based bioenergy-livestock nexus. Bioresour. Technol. 272, 326–336 (2019). https://doi.org/10.1016/J.BIORTECH.2018.10.049
Mani Rathnam, V., Madras, G.: Conversion of Shizochitrium limacinum microalgae to biodiesel by non-catalytic transesterification using various supercritical fluids. Bioresour. Technol. 288, 121538 (2019). https://doi.org/10.1016/j.biortech.2019.121538
Morais, A.R.C., da Costa Lopes, A.M., Bogel-Łukasik, R.: Carbon dioxide in biomass processing: contributions to the green biorefinery concept. Chem. Rev. 115, 3–27 (2015). https://doi.org/10.1021/cr500330z
Escobar, E.L.N., da Silva, T.A., Pirich, C.L., Corazza, M.L., Pereira Ramos, L.: Supercritical fluids: a promising technique for biomass pretreatment and fractionation. Front. Bioeng. Biotechnol. 8, 1–18 (2020). https://doi.org/10.3389/fbioe.2020.00252
Arumugham, T., AlYammahi, J., Rambabu, K., Hassan, S.W., Banat, F.: Supercritical CO2 pretreatment of date fruit biomass for enhanced recovery of fruit sugars. Sustain. Energy Technol. Assess. 52, 102231 (2022). https://doi.org/10.1016/J.SETA.2022.102231
Zhao, M.J., Xu, Q.Q., Li, G.M., Zhang, Q.Z., Zhou, D., Yin, J.Z., Zhan, H.S.: Pretreatment of agricultural residues by supercritical CO2 at 50–80° C to enhance enzymatic hydrolysis. J. Energy Chem. 31, 39–45 (2019). https://doi.org/10.1016/J.JECHEM.2018.05.003
Navarro, A., Montiel, C., Gracia-Fadrique, J., Tecante, A., Bárzana, E.: Supercritical carbon dioxide “explosion” on blue agave bagasse to enhance enzymatic digestibility. Biomass Convers. Biorefinery (2021). https://doi.org/10.1007/S13399-021-01557-Z
Mitraka, G.C., Kontogiannopoulos, K.N., Tsivintzelis, I., Zouboulis, A.I., Kougias, P.G.: Optimization of supercritical carbon dioxide explosion for sewage sludge pre-treatment using response surface methodology. Chemosphere 297, 133989 (2022). https://doi.org/10.1016/J.CHEMOSPHERE.2022.133989
Kumar, A.K., Sharma, S.: Recent updates on different methods of pretreatment of lignocellulosic feedstocks: a review. Bioresour. Bioprocess. 4, 7 (2017). https://doi.org/10.1186/s40643-017-0137-9
Bajpai, P.: Pretreatment of lignocellulosic biomass for biofuel production. Green. Chem. Sustain. 34, 86 (2016). https://doi.org/10.1007/978-981-10-0687-6
Balan, R., Antczak, A., Brethauer, S., Zielenkiewicz, T., Studer, M.H.: Steam explosion pretreatment of Beechwood. Part 1: Comparison of the enzymatic hydrolysis of washed solids and whole pretreatment slurry at different solid loadings. Energies 2020. 13(13), 3653 (2020). Page 3653https://doi.org/10.3390/EN13143653
Eom, T., Chaiprapat, S., Charnnok, B.: Enhanced enzymatic hydrolysis and methane production from rubber wood waste using steam explosion. J. Environ. Manage 235, 231–239 (2019). https://doi.org/10.1016/j.jenvman.2019.01.041
Silva, T.A.L., Zamora, H.D.Z., Varão, L.H.R., Prado, N.S., Baffi, M.A., Pasquini, D.: Effect of steam explosion pretreatment catalysed by organic acid and alkali on chemical and structural properties and enzymatic hydrolysis of sugarcane bagasse. Waste Biomass Valoriz. 9(11), 2191–2201 (2017). https://doi.org/10.1007/S12649-017-9989-7
He, L., Wang, C., Shi, H., Zhou, W., Zhang, Q., Chen, X.: Combination of steam explosion pretreatment and anaerobic alkalization treatment to improve enzymatic hydrolysis of Hippophae rhamnoides. Bioresour. Technol. 289, 121693 (2019). https://doi.org/10.1016/j.biortech.2019.121693
Imman, S., Laosiripojana, N., Champreda, V.: Effects of liquid hot water pretreatment on enzymatic hydrolysis and physicochemical changes of corncobs. Appl. Biochem. Biotechnol. 184, 432–443 (2018). https://doi.org/10.1007/S12010-017-2541-1
Larnaudie, V., Ferrari, M.D., Lareo, C.: Enzymatic hydrolysis of liquid hot water-pretreated switchgrass at high solid content. Energy Fuels 33, 4361–4368 (2019). https://doi.org/10.1021/ACS.ENERGYFUELS.9B00513
Yang, H., Shi, Z., Xu, G., Qin, Y., Deng, J., Yang, J.: Bioethanol production from bamboo with alkali-catalyzed liquid hot water pretreatment. Bioresour. Technol. 274, 261–266 (2019). https://doi.org/10.1016/J.BIORTECH.2018.11.088
Xia, F., Gong, J., Lu, J., Cheng, Y., Zhai, S., An, Q., Wang, H.: Combined liquid hot water with sodium carbonate-oxygen pretreatment to improve enzymatic saccharification of reed. Bioresour. Technol. 297, 122498 (2020). https://doi.org/10.1016/J.BIORTECH.2019.122498
Sakuragi, K., Igarashi, K., Samejima, M.: Application of ammonia pretreatment to enable enzymatic hydrolysis of hardwood biomass. Polym. Degrad. Stab. 148, 19–25 (2018). https://doi.org/10.1016/J.POLYMDEGRADSTAB.2017.12.008
Iram, A., Cekmecelioglu, D., Demirci, A.: Optimization of dilute sulfuric acid, aqueous ammonia, and steam explosion as the pretreatments steps for distillers’ dried grains with solubles as a potential fermentation feedstock. Bioresour Technol. 282, 475–481 (2019). https://doi.org/10.1016/J.BIORTECH.2019.03.009
Sohni, S., Hashim, R., Nidaullah, H., Sulaiman, O., Leh, C.P., Lamaming, J., Arai, T., Kosugi, A.: Enhancing the enzymatic digestibility of oil palm biomass using supercritical carbon dioxide-based pretreatment towards biorefinery application. Ind. Crops Prod. 157, 112923 (2020). https://doi.org/10.1016/J.INDCROP.2020.112923
Rosero-Henao, J.C., Bueno, B.E., de Souza, R., Ribeiro, R., Lopes de Oliveira, A., Gomide, C.A., Gomes, T.M., Tommaso, G.: Potential benefits of near critical and supercritical pre-treatment of lignocellulosic biomass towards anaerobic digestion. Waste Manag. Res. 37, 74–82 (2019). https://doi.org/10.1177/0734242X18806998
Al Afif, R., Wendland, M., Amon, T., Pfeifer, C.: Supercritical carbon dioxide enhanced pre-treatment of cotton stalks for methane production. Energy 194, 116903 (2020). https://doi.org/10.1016/j.energy.2020.116903
An, S., Li, W., Liu, Q., Xia, Y., Zhang, T., Huang, F., Lin, Q., Chen, L.: Combined dilute hydrochloric acid and alkaline wet oxidation pretreatment to improve sugar recovery of corn stover. Bioresour. Technol. 271, 283–288 (2019). https://doi.org/10.1016/J.BIORTECH.2018.09.126
Morone, A., Chakrabarti, T., Pandey, R.A.: Assessment of alkaline peroxide-assisted wet air oxidation pretreatment for rice straw and its effect on enzymatic hydrolysis. Cellul. 2017 2411. 24, 4885–4898 (2017). https://doi.org/10.1007/S10570-017-1451-2
Noparat, P., Prasertsan, P., O-Thong, S., Pan, X.: Sulfite pretreatment to overcome recalcitrance of lignocellulose for enzymatic hydrolysis of oil palm trunk. Energy Procedia 138, 1122–1127 (2017). https://doi.org/10.1016/J.EGYPRO.2017.10.209
Kim, J.R., Karthikeyan, K.G.: Effects of severe pretreatment conditions and lignocellulose-derived furan byproducts on anaerobic digestion of dairy manure. Bioresour. Technol. 340, 125632 (2021). https://doi.org/10.1016/J.BIORTECH.2021.125632
Liu, Z.J., Lan, T.Q., Li, H., Gao, X., Zhang, H.: Effect of bisulfite treatment on composition, structure, enzymatic hydrolysis and cellulase adsorption profiles of sugarcane bagasse. Bioresour. Technol. 223, 27–33 (2017). https://doi.org/10.1016/J.BIORTECH.2016.10.029
Saini, S., Sharma, K.K.: Fungal lignocellulolytic enzymes and lignocellulose: a critical review on their contribution to multiproduct biorefinery and global biofuel research. Int. J. Biol. Macromol. 193, 2304–2319 (2021). https://doi.org/10.1016/J.IJBIOMAC.2021.11.063
Saini, S., Sharma, K.K.: Ligninolytic Fungi from the Indian subcontinent and their contribution to enzyme biotechnology. Prog Mycol. 139–184 (2021). https://doi.org/10.1007/978-981-16-3307-2_6/COVER
Chen, H., Liu, J., Chang, X., Chen, D., Xue, Y., Liu, P., Lin, H., Han, S.: A review on the pretreatment of lignocellulose for high-value chemicals. Fuel Process Technol. 160, 196–206 (2017)
Sharma, H.K., Xu, C., Qin, W.: Biological pretreatment of lignocellulosic biomass for biofuels and bioproducts: an overview. Waste Biomass Valoriz. 10, 235–251 (2019). https://doi.org/10.1007/s12649-017-0059-y
Zanellati, A., Spina, F., Bonaterra, M., Dinuccio, E., Varese, G.C., Scarpeci, T.E.: Screening and evaluation of phenols and furans degrading fungi for the biological pretreatment of lignocellulosic biomass. Int. Biodeterior. Biodegrad. 161, 105246 (2021). https://doi.org/10.1016/j.ibiod.2021.105246
Arora, A., Priya, S., Sharma, P., Sharma, S., Nain, L.: Evaluating biological pretreatment as a feasible methodology for ethanol production from paddy straw. Biocatal. Agric. Biotechnol. 8, 66–72 (2016). https://doi.org/10.1016/j.bcab.2016.08.006
Akyol, Ã., Ince, O., Bozan, M., Ozbayram, E.G., Ince, B.: Biological pretreatment with Trametes versicolor to enhance methane production from lignocellulosic biomass: a metagenomic approach. Ind. Crops Prod. (2019). https://doi.org/10.1016/j.indcrop.2019.111659
Machado, A.S., Ferraz, A.: Biological pretreatment of sugarcane bagasse with basidiomycetes producing varied patterns of biodegradation. Bioresour. Technol. 225, 17–22 (2017). https://doi.org/10.1016/J.BIORTECH.2016.11.053
Saha, B.C., Kennedy, G.J., Qureshi, N., Cotta, M.A.: Biological pretreatment of corn stover with Phlebia brevispora NRRL-13108 for enhanced enzymatic hydrolysis and efficient ethanol production. Biotechnol. Prog. 33, 365–374 (2017). https://doi.org/10.1002/BTPR.2420
Martínez-Patiño, J.C., Lu-Chau, T.A., Gullón, B., Ruiz, E., Romero, I., Castro, E., Lema, J.M.: Application of a combined fungal and diluted acid pretreatment on olive tree biomass. Ind. Crops Prod. 121, 10–17 (2018). https://doi.org/10.1016/J.INDCROP.2018.04.078
Dai, Y., Zhang, H.S., Huan, B., He, Y.: Enhancing the enzymatic saccharification of bamboo shoot shell by sequential biological pretreatment with galactomyces sp. CCZU11-1 and deep eutectic solvent extraction. Bioprocess. Biosyst. Eng. 40, 1427–1436 (2017). https://doi.org/10.1007/S00449-017-1800-4
Shah, T.A., Lee, C.C., Orts, W.J., Tabassum, R.: Biological pretreatment of rice straw by ligninolytic Bacillus sp. strains for enhancing biogas production. Environ. Prog Sustain. Energy 38, e13036 (2019). https://doi.org/10.1002/EP.13036
Li, P., He, C., Li, G., Ding, P., Lan, M., Gao, Z., Jiao, Y.: Biological pretreatment of corn straw for enhancing degradation efficiency and biogas production. Bioengineered 11, 251–260 (2020). https://doi.org/10.1080/21655979.2020.1733733
Zou, L., Ouyang, S., Hu, Y., Zheng, Z., Ouyang, J.: Efficient lactic acid production from dilute acid-pretreated lignocellulosic biomass by a synthetic consortium of engineered Pseudomonas putida and Bacillus coagulans. Biotechnol. Biofuels. 14 (2021). doi: 10.1186/S13068-021-02078-7.
Yan, X., Wang, Z., Zhang, K., Si, M., Liu, M., Chai, L., Liu, X., Shi, Y.: Bacteria-enhanced dilute acid pretreatment of lignocellulosic biomass. Bioresour. Technol. 245, 419–425 (2017). https://doi.org/10.1016/J.BIORTECH.2017.08.037
Acknowledgements
The authors are thankful to Chaudhary Charan Singh Haryana Agricultural University Hisar, Haryana, India for providing infrastructural facility to conduct the research.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
GC: Conceptualization, Methodology, Resources, Writing—Original Draft, Writing—Reviewing and Editing and Supervision. NC: Writing—Original Draft. SS: Writing—Reviewing and Editing. YG: Writing—Reviewing and Editing. VV: Writing—Reviewing and Editing. AP: Writing—Reviewing & Editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chaudhary, G., Chaudhary, N., Saini, S. et al. Assessment of Pretreatment Strategies for Valorization of Lignocellulosic Biomass: Path Forwarding Towards Lignocellulosic Biorefinery. Waste Biomass Valor 15, 1–36 (2024). https://doi.org/10.1007/s12649-023-02219-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-023-02219-z