Preamble
The International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO) has played an integral role in educating both the metabolic surgical and the medical community at large about the role of innovative and new surgical and/or endoscopic interventions in treating adiposity-based chronic diseases.
The mini gastric bypass is also known as the one anastomosis gastric bypass. The IFSO has agreed that the standard nomenclature should be the mini gastric bypass-one anastomosis gastric bypass (MGB-OAGB). The IFSO commissioned a task force (Appendix 1) to determine if MGB-OAGB is an effective and safe procedure and if it should be considered a surgical option for the treatment of obesity and metabolic diseases.
The following position statement is issued by the IFSO MGB-OAGB task force and approved by the IFSO Scientific Committee and Executive Board. This statement is based on current clinical knowledge, expert opinion, and published peer-reviewed scientific evidence. It will be reviewed in 2 years.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
In weight loss surgery, the concept of a “loop” gastric bypass consisting of one anastomosis was first introduced by Mason in 1967 [1]. In Mason’s configuration, the gastric pouch was wide and short, and had a horizontal shape, exposing the esophageal mucosa to caustic bile reflux coming from the jejunal loop. Because it was a reflux-inducing procedure, this bypass concept was quickly abandoned. In 1997, Rutledge introduced a different version of one anastomosis gastric bypass and named it “mini gastric bypass” (MGB) because the procedure initially was described through a “mini-laparotomy”, in analogy with “mini-laparotomy cholecystectomy”.
MGB consisted of a lesser curvature-based long-sleeved gastric pouch starting 2–3 cm below the level of the crow’s foot and extending proximally slightly to the left of the angle of His. An antecolic 3–5-cm-wide anastomosis was then constructed between the pouch and the jejunum, about 180–220 cm distal to Treitz’ ligament. In the super obese, the distance to Treitz’ ligament would be about 250 cm, in the elderly or vegetarians 180–200 cm and in type II diabetics without major obesity about 150 cm [2].
In 2002, Carbajo and Caballero (Spain) proposed a technical variation to prevent gastroesophageal (GE) bile reflux. They called their technique one anastomosis gastric bypass (OAGB) or in Spanish bypass gastrico de una anastomosis (BAGUA). According to this technique, OAGB had a latero-lateral anastomosis between the loop of jejunum and the pouch, and the distance to Treitz’ ligament averaged 250–350 cm [3].
Since then, other names such as “single anastomosis gastric bypass” (SAGB) or “omega loop gastric bypass” (OLGB) have been proposed to define the same technique [4, 5]. In 2013, the confusion created by the various names led a group of surgeons to use the name mini gastric bypass-one anastomosis gastric bypass (MGB-OAGB) to define this surgery [6].
Despite an increase in the utilisation of MGB-OAGB, particularly in Europe and the Asia Pacific regions [7], there remains concern that the MGB-OAGB could create bilio-enteric reflux, and may increase the risk of esophageal and gastric cancer.
The task force undertook a systematic review to summarise the current evidence on the efficacy and safety of these procedures with the aim of providing the most up-to-date information to guide practice.
Methods
Literature Search
We performed a comprehensive literature search to identify studies reporting any experience or outcomes with the MGB-OAGB. The search was done in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. We searched MEDLINE (1946 to November 2017), EMBASE (1974 to November 2017), PubMed (until November 2017) and the Cochrane Library (until November 2017). Search terms were broad, to encompass all mini gastric bypass procedures (MGB). These include terms specifying the bariatric procedure (gastric bypass, mini gastric bypass, one anastomosis gastric bypass, bariatric surgery), single anastomosis (single anastomosis, loop anastomosis, one anastomosis, omega loop, mini). A full list of search terms is presented in Appendix Table 4. Manual searching of reference lists from reviews, as well as references from selected primary studies, was performed to identify any additional studies.
Inclusion Criteria
Studies were selected that reported on outcomes after single anastomosis gastric bypass procedures. All study designs were accepted. We summarised data for studies with greater than 15 participants, and with greater than 1-year follow-up; however, studies of all sizes and follow-up time frames were collected. Only full text articles were included.
Data Extraction
Information extracted from eligible studies included basic study data (year, country, design, study size), demographic data, surgical technique, follow-up, weight loss, evolution of co-morbidities and complications.
Results
Literature Search
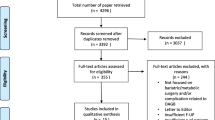
Using the search strategy described, we identified 3936 studies. After 877 duplicates were removed, we screened titles and abstracts for 3059 records. Full text articles for 255 eligible studies were screened, and 168 articles were subsequently excluded. Hence, 87 full length publications were identified for inclusion.
Of these studies, 78 focused on outcomes of OAGB. Only 52 of these studies had reasonable follow-up and study numbers, with 26 having less than 1 year follow-up [8,9,10,11,12,13,14,15,16,17,18,19,20,21,22] or ≤ 15 OAGB patients [23,24,25,26,27,28,29,30,31,32,33]. A further nine studies focused on complications following OAGB (Fig. 1).
Overall Summary
A total of 52 reasonable quality (n > 15, follow-up > 1 year) studies were identified. Of these, there were 16,546 patients (excluding 2 studies with significant overlap in patient cohort), with a median of 94.5 (IQR 34.5–203, range 16–2678) patients per study. The average study body mass index (BMI) ranged from 25.3 to 67 kg/m2, with a mean study BMI of 44.6 ± 6.4 kg/m2.
Over a range of follow-up times, the average excess weight loss was 74.8 ± 12.0%. In studies that reported diabetes remission, there was observed resolution of diabetes in 2495 of 2855 diabetic patients (87.4%).
Outcomes from MGB-OAGB
There are currently 4 randomised controlled trials, 34 single-arm cohort studies and 14 multiple-arm comparison cohort studies on MGB-OAGB, which are summarised in Table 1. Sixteen studies reported on just primary MGB-OAGB procedures, 6 on revision operations, 15 analysed a mix of both and 15 did not state whether they were primary or revision operations.
MGB-OAGB as a Primary Procedure
In total, there are 191 patients who have been enrolled in 4 RCT and a further 8724 patients reported up in retrospective and prospective cohort studies. This number is likely to be an overestimation due to shared patients in multiple series.
Weight Loss
In the four randomised controlled trials, weight loss was reported at 12 months (EWL 66.9 ± 23.7% [35] and EWL 66.9 ± 10.9% [37]), 2 years (EWL 64.4 ± 8.8% [34]) and 5 years (TBWL 22.8 ± 5.9% [36]). Of note, in the Lee trial comparing MGB-OAGB to SG at 5 years, the mean starting BMI was substantially lower than the other trials at 30.2 ± 2.2 and the EWL achieved was over 100% with a mean finishing BMI of 23.3 ± 2.2 kg/m2. These results were not significantly different to comparator operations.
In the prospective cohort studies, weight loss at 12 months was EWL 80.5% (n = 2410; follow-up not reported) [38], EWL 70.1 ± 8.4% (n = 838; 94.8% follow-up) [51] and EWL 70 ± 20% (n = 89; 100% follow-up) [52]; 23 months 68.4% (n = 126; 45.2% follow-up) [20]; 3 years EWL 81.5 ± 5.0% (n = 570; 89.4% follow-up) [51]; 5 years EWL 72.9% (n = 1163; 56% follow-up) [44] and 77.0 ± 5.1 (n = 254; 79.1% follow-up). Two other prospective studies were not included as they failed to differentiate primary or secondary procedures and did not accurately report weight loss as the focus of these papers was health change rather than weight loss [53, 55]. Weight loss reported in the retrospective cohort studies was similar (Table 1).
Change in T2DM Management
Diabetes or metabolic syndrome was reported upon as a co-morbidity of interest in all 4 RCT. In the Lee trial comparing MGB-OAGB to RYGB, there was 100% resolution of the metabolic syndrome at 2 years [34]. No other treatment for T2DM was required at 12 months for 50% of participants with T2DM in-trial (n = 2) [35] and 84% (n = 49) [37]. At 5 years, 60% of participants with T2DM at baseline had a HbA1c < 6.5% without medications in the low BMI trial focusing on change in diabetes [36]. Again, these results were not significantly different to the comparator populations.
There are six prospective cohort studies that addressed change in diabetes status following MGB-OAGB. The change in T2DM management was reported in various ways, but all reported major improvement (Table 1).
Complications
There was one early death in-trial reported in the four RCT (3.3% death rate for that trial [36]; 0.05% for pooled data). A further 15 deaths were reported in the prospective and retrospective cohort studies giving a crude death rate overall of 0.17%. This is likely to be an underestimation due to shared patients between reports.
Early complications were experienced by 17/191 (8.9%) patients in the RCT cohort with 3 patients requiring a return to theatre (1.5%). This is likely to be an underestimation as minor complications were not reported by Darabi [35] and no complications other than the death were reported by Lee [36]. The early complication rate reported in the retrospective and prospective cohort studies was again similar (Table 1). Early complications included anastomotic leak, wound infection, haemorrhage, anastomotic stricture and organ perforation (Table 2).
There were no late deaths in the RCT cohort, and four in the cohort studies. Late complications included marginal ulcers, bowel obstruction, malnutrition and gastroesophageal reflux including biliary reflux.
MGB-OAGB as a Secondary Procedure
There is one prospective cohort comparator study, one retrospective cohort comparator study and four retrospective studies specifically addressing the use of MGB-OAGB as a revisional procedure with a total of 222 patients enrolled in these 6 studies.
Weight loss overall at each time-point appears to be lower than in the primary procedures, although one retrospective cohort study with a follow up rate of 91% (n = 30) did achieve EWL 66 ± 22% at 5 years [81].
Change in T2DM management was only reported in one retrospective cohort study (n = 30). There were seven patients with T2DM at baseline and at 5 years six patients required no treatment other than surgery for their T2DM giving an 85% resolution rate [81].
One early death has been reported with an in-trial death rate of 3.4% [79] and an overall death rate in these studies of 0.45%. No late deaths have been reported.
Early complication rates range from 2.6 to 21.6%. The wide variation probably reflects differences in reporting. Early complications include anastomotic leak, haemorrhage, anastomotic stricture and organ perforation.
Late complication rates are only reported in two studies (6.6% [81] and 8.1% [83]). They include gastroesophageal reflux disease (bile reflux not specifically reported) and bowel obstruction.
Operative Technique for MGB-OAGB
Operative technique (Table 3) varied among groups in various domains—pouch and bougie size, gastrojejunostomy anastomosis technique, limb length.
Pouch and Bougie Size
The description of the starting point for gastric stapling varied; however, most groups started at the level or just below the Crow’s foot on the lesser curve. The majority of studies used a 36 French bougie; however, the bougie size varied from a 1 cm diameter nasogastric tube to a 42 French bougie.
Gastrojejunostomy
A linear stapler was used in most cases, varying in length from 30 to 60 mm. Only a partial length of the stapler was used in some cases, creating an anastomosis as small as 1.5 cm. Handsewn anastomoses were not commonly used (described in one study).
Limb Length
The most common limb length used was 200 cm, reported by 27 studies. Nine studies reported forming limbs < 200 cm, five reported > 200 cm and five reported the “Rutledge” technique but no length. Ten studies tailored the limb length according to pre-operative body mass index (BMI).
Discussion
The current evidence suggests that MGB-OAGB provides effective weight loss that is durable to 5 years. Weight loss appears to be more effective in primary operations when compared to revisional procedures; however, small numbers limit our ability to completely assess this parameter.
MGB-OAGB appears to have a favourable effect on T2DM, although numbers in the reports are small, and durability of glycaemic effect has not been reported.
There is an acceptable early and late complication rate, and the rates of symptomatic bile reflux are lower than first feared. Complication rates appear to be higher in the revisional setting. There is a lack of long-term nutritional information and rates of bile reflux rely mainly on self-reporting. These are areas of concern and it is imperative that patients who undergo these procedures understand the need for on-going care from their bariatric team.
The ideal operative technique has not been defined. The most common description commences the pouch below the crows-foot with a stapled anastomosis and 200 cm common limb length; however, there is a great deal of variance in each of these elements. This may be an important issue to be addressed by an RCT in the future.
There is a paucity of RCT evidence, with the majority of evidence coming from retrospective cohort studies. There is a need for well-designed large prospective cohort studies as well as RCT in to the future to better define where MGB-OAGB should be placed in the current suite of bariatric procedures.
The term mini gastric bypass/one anastomosis gastric bypass (MGB-OAGB) has been used throughout this position statement as this has been the agreed nomenclature endorsed by the Executive Board of IFSO. Whilst the initial use of the word “mini” reflected the minimally invasive approach used for the procedure compared to a laparotomy, there is a risk that the term will be misinterpreted as meaning the surgery itself is a lesser procedure—both in terms of surgical risk and metabolic benefit. The current systematic review reinforces that the procedure is effective in terms of weight loss and metabolic benefit, but also carries surgical risk that is very similar to RYGB. Therefore, the taskforce recommends that in the future the procedure be referred to as “one anastomosis gastric bypass (OAGB)”.
The need for more RCT’s is paramount to our understanding of our interventions; however, the need for guidance for emerging procedures is the responsibility of organisations, such as IFSO. Professional societies must continue to extrapolate the existing data against the needs of the patients we serve and the availability of current technology on a micro and macro level. Though position statements are not without bias, they are meant to be temporal in nature. Continued re-analysis is necessary in order to remain relevant.
Recommendation of the IFSO MGB-OAGB Taskforce
Based on the existing data, we recommend the following:
-
1.
OAGB should be the identifier for this procedure in future publications.
-
2.
Whilst early results are promising in terms of weight and T2DM management, there is a lack of long-term evidence for durability of effect as well as long-term nutritional complications. Bile reflux is either under reported or does not seem to be a major issue, but remains a theoretical risk. Patients should be encouraged to remain in long-term multidisciplinary care.
-
3.
Patients undergoing OAGB in the revisional setting have less weight loss and more complications than with primary procedures.
-
4.
Surgeons performing this, as well as any other bariatric/metabolic procedure, are encouraged to participate in a national or international registry so that long-term data may be more effectively identified.
-
5.
OAGB is a recognised bariatric/metabolic procedure and should not be considered investigational.
References
Mason EE, Ito C. Gastric bypass in obesity. Surg Clin North Am. 1967;47(6):1345–51.
Rutledge R. The mini-gastric bypass: experience with the first 1,274 cases. Obes Surg. 2001;11(3):276–80.
Carbajo M, García-Caballero M, Toledano M, et al. One-anastomosis gastric bypass by laparoscopy: results of the first 209 patients. Obes Surg. 2005;15(3):398–404.
Lee WJ, Lin YH. Single-anastomosis gastric bypass (SAGB): appraisal of clinical evidence. Obes Surg. 2014;24(10):1749–56.
Chevallier JM, Arman GA, Guenzi M, et al. One thousand single anastomosis (omega loop) gastric bypasses to treat morbid obesity in a 7-year period: outcomes show few complications and good efficacy. Obes Surg. 2015;25(6):951–8.
Musella M, Milone M. Still "controversies" about the mini gastric bypass? Obes Surg. 2014;24(4):643–4.
Angrisani, L., et al., Bariatric surgery and Endoluminal procedures: IFSO worldwide survey 2014. Obes Surg, 2017.
Blanc P, Lointier P, Breton C, et al. The hand-sewn anastomosis with an absorbable bidirectional monofilament barbed suture Stratafix during laparoscopic one anastomosis loop gastric bypass. Retrospective study in 50 patients. Obes Surg. 2015;25(12):2457–60.
Blanchet MC, Gignoux B, Matussière Y, et al. Experience with an enhanced recovery after surgery (ERAS) program for bariatric surgery: comparison of MGB and LSG in 374 patients. Obes Surg. 2017;27(7):1896–900.
Celik A, Pouwels S, Karaca FC, et al. Time to glycemic control—an observational study of 3 different operations. Obes Surg. 2017;27(3):694–702.
Dardzinska JA et al. Fasting and post-prandial peptide YY levels in obese patients before and after mini versus roux-en-Y gastric bypass. Minerva Chir. 2017;72(1):24–30.
Garcia-Caballero M et al. Resolution of diabetes mellitus and metabolic syndrome in normal weight 24-29 BMI patients with one anastomosis gastric bypass. Nutricion Hospitalaria. 2012;27(2):623–31.
Genser L, Carandina S, Tabbara M, et al. Presentation and surgical management of leaks after mini-gastric bypass for morbid obesity. Surgery for Obesity & Related Diseases. 2016;12(2):305–12.
Greco, F. and R. Tacchino, Ileal food diversion: a simple, powerful and easily revisable and reversible single-anastomosis gastric bypass. Obes Surg, 2014. 19.
Kaska L, Proczko M, Wiśniewski P, et al. A prospective evaluation of the influence of three bariatric procedures on insulin resistance improvement. Should the extent of undiluted bile transit be considered a key postoperative factor altering glucose metabolism? Wideochir Inne Tech Maloinwazyjne. 2015;10(2):213–28.
Kim MJ, Park HK, Byun DW, et al. Incretin levels 1 month after laparoscopic single anastomosis gastric bypass surgery in non-morbid obese type 2 diabetes patients. Asian Journal of Surgery. 2014;37(3):130–7.
Kim Z, Hur KY. Laparoscopic mini-gastric bypass for type 2 diabetes: the preliminary report. World J Surg. 2011;35(3):631–6.
Meydan C, Raziel A, Sakran N, et al. Single anastomosis gastric bypass-comparative short-term outcome study of conversional and primary procedures. Obes Surg. 2017;27(2):432–8.
Mokhber S et al. Anemia outcome after laparascopic mini bypass: analysis of 107 consecutive patients. Acta Gastroenterol Belg. 2016;79(2):201–5.
Noun R, Riachi E, Zeidan S, et al. Mini-gastric bypass by mini-laparotomy: a cost-effective alternative in the laparoscopic era. Obes Surg. 2007;17(11):1482–6.
Piazza L, di Stefano C, Ferrara F, et al. Revision of failed primary adjustable gastric banding to mini-gastric bypass: results in 48 consecutive patients. Updat Surg. 2015;67(4):433–7.
Shenouda, M.M., et al., Bile gastritis following laparoscopic single anastomosis gastric bypass: pilot study to assess significance of bilirubin level in gastric aspirate. Obes Surg, 2017.
Ahmetasevic E et al. Bariatric surgery in university clinic center Tuzla—results after 30 operations. Acta Inform Med. 2016;24(2):139–42.
Betry C et al. Need for intensive nutrition care after bariatric surgery. JPEN J Parenter Enteral Nutr. 2017;41(2):258–62.
Garciacaballero M et al. Improvement of C peptide zero BMI 24-34 diabetic patients after tailored one anastomosis gastric bypass (BAGUA). Nutricion Hospitalaria. 2013;28(Suppl 2):35–46.
Greco F. Conversion of vertical sleeve gastrectomy to a functional single-anastomosis gastric bypass: technique and preliminary results using a non-adjustable ring instead of stapled division. Obes Surg. 2017;27(4):896–901.
Himpens JM, Vilallonga R, Cadière GB, et al. Metabolic consequences of the incorporation of a roux limb in an omega loop (mini) gastric bypass: evaluation by a glucose tolerance test at mid-term follow-up. Surg Endosc. 2016;30(7):2935–45.
Milone M, di Minno MN, Leongito M, et al. Bariatric surgery and diabetes remission: sleeve gastrectomy or mini-gastric bypass? World J Gastroenterol. 2013;19(39):6590–7.
Tolone S, Cristiano S, Savarino E, et al. Effects of omega-loop bypass on esophagogastric junction function. Surgery for Obesity & Related Diseases. 2016;12(1):62–9.
Yeh C, Huang HH, Chen SC, et al. Comparison of consumption behavior and appetite sensations among patients with type 2 diabetes mellitus after bariatric surgery. PeerJ. 2017;5:e3090.
Guo, X., et al., [Impacts of laparoscopic bariatric surgery on GLP-1 and Ghrelin level in patients with type 2 diabetes mellitus]. Chung-Hua Wai Ko Tsa Chih [Chinese Journal of Surgery], 2013. 51(4): p. 323–7.
Guo X, Yin K, Zhuo GZ, et al. Efficacy comparison between 2 methods of laparoscopic gastric bypass surgery in the treatment of type 2 diabetes mellitus. Zhonghua Weichang Waike Zazhi. 2012;15(11):1125–8.
Ding D, Chen DL, Hu XG, et al. Outcomes after laparoscopic surgery for 219 patients with obesity. Zhonghua Weichang Waike Zazhi. 2011;14(2):128–31.
Lee WJ, Yu PJ, Wang W, et al. Laparoscopic roux-en-Y versus mini-gastric bypass for the treatment of morbid obesity: a prospective randomized controlled clinical trial. Ann Surg. 2005;242(1):20–8.
Darabi S, Talebpour M, Zeinoddini A, et al. Laparoscopic gastric plication versus mini-gastric bypass surgery in the treatment of morbid obesity: a randomized clinical trial. Surgery for Obesity & Related Diseases. 2013;9(6):914–9.
Lee WJ, Chong K, Lin YH, et al. Laparoscopic sleeve gastrectomy versus single anastomosis (mini-) gastric bypass for the treatment of type 2 diabetes mellitus: 5-year results of a randomized trial and study of incretin effect. Obes Surg. 2014;24(9):1552–62.
Seetharamaiah S, Tantia O, Goyal G, et al. LSG vs OAGB-1 year follow-up data-a randomized control trial. Obes Surg. 2017;27(4):948–54.
Rutledge R, Walsh TR. Continued excellent results with the mini-gastric bypass: six-year study in 2,410 patients. Obes Surg. 2005;15(9):1304–8.
Noun R, Zeidan S. Laparoscopic mini-gastric bypass: an effective option for the treatment of morbid obesity. J Chir. 2007;144(4):301–4.
Peraglie C. Laparoscopic mini-gastric bypass (LMGB) in the super-super obese: outcomes in 16 patients. Obes Surg. 2008;18(9):1126–9.
Lee WJ, Wang W, Lee YC, et al. Laparoscopic mini-gastric bypass: experience with tailored bypass limb according to body weight. Obes Surg. 2008;18(3):294–9.
Lee WJ, Wang W, Lee YC, et al. Effect of laparoscopic mini-gastric bypass for type 2 diabetes mellitus: comparison of BMI>35 and <35 kg/m2. J Gastrointest Surg. 2008;12(5):945–52.
Piazza L, Ferrara F, Leanza S, et al. Laparoscopic mini-gastric bypass: short-term single-institute experience. Updat Surg. 2011;63(4):239–42.
Lee WJ, Ser KH, Lee YC, et al. Laparoscopic roux-en-Y vs. mini-gastric bypass for the treatment of morbid obesity: a 10-year experience. Obes Surg. 2012;22(12):1827–34.
Lee YC, Lee WJ, Liew PL. Predictors of remission of type 2 diabetes mellitus in obese patients after gastrointestinal surgery. Obesity Research & Clinical Practice. 2013;7(6):e494–500.
Lee YC, Liew PL, Lee WJ, et al. Gastrointestinal quality of life following bariatric surgery in Asian patients. Hepato-Gastroenterology. 2013;60(124):759–61.
Carbajo MA, Jiménez JM, Castro MJ, et al. Outcomes in weight loss, fasting blood glucose and glycosylated hemoglobin in a sample of 415 obese patients, included in the database of the European accreditation council for excellence centers for bariatric surgery with laparoscopic one anastomosis gastric bypass. Nutricion Hospitalaria. 2014;30(5):1032–8.
Kim MJ, Hur KY. Short-term outcomes of laparoscopic single anastomosis gastric bypass (LSAGB) for the treatment of type 2 diabetes in lower BMI (<30 kg/m(2)) patients. Obes Surg. 2014;24(7):1044–51.
Kular KS, Manchanda N, Rutledge R. A 6-year experience with 1,054 mini-gastric bypasses-first study from Indian subcontinent. Obes Surg. 2014;24(9):1430–5.
Musella, M., et al., A decade of bariatric surgery. What have we learned? Outcome in 520 patients from a single institution. International Journal Of Surgery, 2014. 12 Suppl 1: p. S183–8.
Musella M, Susa A, Greco F, et al. The laparoscopic mini-gastric bypass: the Italian experience: outcomes from 974 consecutive cases in a multicenter review. Surg Endosc. 2014;28(1):156–63.
Yang PJ, Lee WJ, Tseng PH, et al. Bariatric surgery decreased the serum level of an endotoxin-associated marker: lipopolysaccharide-binding protein. Surgery for Obesity & Related Diseases. 2014;10(6):1182–7.
Garcia-Caballero M et al. Super obese behave different from simple and morbid obese patients in the changes of body composition after tailored one anastomosis gastric bypass (BAGUA). Nutricion Hospitalaria. 2014;29(5):1013–9.
Luger M, Kruschitz R, Langer F, et al. Effects of omega-loop gastric bypass on vitamin D and bone metabolism in morbidly obese bariatric patients. Obes Surg. 2015;25(6):1056–62.
Milone M, Lupoli R, Maietta P, et al. Lipid profile changes in patients undergoing bariatric surgery: a comparative study between sleeve gastrectomy and mini-gastric bypass. Int J Surg. 2015;14:28–32.
Kular KS, Manchanda N, Cheema GK. Seven years of mini-gastric bypass in type II diabetes patients with a body mass index <35 kg/m(2). Obes Surg. 2016;26(7):1457–62.
Peraglie C. Laparoscopic mini-gastric bypass in patients age 60 and older. Surg Endosc. 2016;30(1):38–43.
Al-Shurafa H et al. Primary experience of bariatric surgery in a newly established private obesity center. Saudi Medical Journal. 2016;37(10):1089–95.
Jammu GS, Sharma R. A 7-year clinical audit of 1107 cases comparing sleeve gastrectomy, roux-en-Y gastric bypass, and mini-gastric bypass, to determine an effective and safe bariatric and metabolic procedure. Obes Surg. 2016;26(5):926–32.
Kansou, G., et al., Laparoscopic sleeve gastrectomy versus laparoscopic mini gastric bypass: One year outcomes. International Journal Of Surgery, 2016. 33 Pt A: p. 18–22.
Kruschitz R, Luger M, Kienbacher C, et al. The effect of roux-en-Y vs. Omega-loop gastric bypass on liver, metabolic parameters, and weight loss. Obes Surg. 2016;26(9):2204–12.
Musella M, Apers J, Rheinwalt K, et al. Efficacy of bariatric surgery in type 2 diabetes mellitus remission: the role of mini gastric bypass/one anastomosis gastric bypass and sleeve Gastrectomy at 1 year of follow-up. A European survey Obesity Surgery. 2016;26(5):933–40.
Karimi, M., et al., Trend of changes in serum albumin and its relation with sex, age, and BMI following laparoscopic mini-gastric bypass surgery in morbid obese cases. Obes Surg, 2017.
Carbajo M, García-Caballero M, Toledano M, et al. One-anastomosis gastric bypass by laparoscopy: results of the first 209 patients. Obes Surg. 2005;15(3):398–404.
Wang W, Wei PL, Lee YC, et al. Short-term results of laparoscopic mini-gastric bypass. Obes Surg. 2005;15(5):648–54.
Chakhtoura G, Zinzindohoué F, Ghanem Y, et al. Primary results of laparoscopic mini-gastric bypass in a French obesity-surgery specialized university hospital. Obes Surg. 2008;18(9):1130–3.
Noun R, Skaff J, Riachi E, et al. One thousand consecutive mini-gastric bypass: short- and long-term outcome. Obes Surg. 2012;22(5):697–703.
Disse E, Pasquer A, Espalieu P, et al. Greater weight loss with the omega loop bypass compared to the roux-en-Y gastric bypass: a comparative study. Obes Surg. 2014;24(6):841–6.
Bruzzi M, Rau C, Voron T, et al. Single anastomosis or mini-gastric bypass: long-term results and quality of life after a 5-year follow-up. Surgery for Obesity & Related Diseases. 2015;11(2):321–6.
Chevallier JM, Arman GA, Guenzi M, et al. One thousand single anastomosis (omega loop) gastric bypasses to treat morbid obesity in a 7-year period: outcomes show few complications and good efficacy. Obes Surg. 2015;25(6):951–8.
Guenzi M, Arman G, Rau C, et al. Remission of type 2 diabetes after omega loop gastric bypass for morbid obesity. Surg Endosc. 2015;29(9):2669–74.
Parmar CD, Mahawar KK, Boyle M, et al. Mini gastric bypass: first report of 125 consecutive cases from United Kingdom. Clinical Obesity. 2016;6(1):61–7.
Madhok B, Mahawar KK, Boyle M, et al. Management of super-super obese patients: comparison between mini (one anastomosis) gastric bypass and sleeve gastrectomy. Obes Surg. 2016;26(7):1646–9.
Carbajo MA, Luque-de-León E, Jiménez JM, et al. Laparoscopic one-anastomosis gastric bypass: technique, results, and long-term follow-up in 1200 patients. Obes Surg. 2017;27(5):1153–67.
Carbajo MA, Fong-Hirales A, Luque-de-León E, et al. Weight loss and improvement of lipid profiles in morbidly obese patients after laparoscopic one-anastomosis gastric bypass: 2-year follow-up. Surg Endosc. 2017;31(1):416–21.
Lessing Y, Pencovich N, Khatib M, et al. One-anastomosis gastric bypass: first 407 patients in 1 year. Obes Surg. 2017;27:2583–9.
Musella M, Susa A, Manno E, et al. Complications following the mini/one anastomosis gastric bypass (MGB/OAGB): a multi-institutional survey on 2678 patients with a mid-term (5 years) follow-up. Obes Surg. 2017;27:2956–67.
Taha O, Abdelaal M, Abozeid M, et al. Outcomes of omega loop gastric bypass, 6-years experience of 1520 cases. Obes Surg. 2017;27(8):1952–60.
Wang W, Huang MT, Wei PL, et al. Laparoscopic mini-gastric bypass for failed vertical banded gastroplasty. Obes Surg. 2004;14(6):777–82.
Moszkowicz D, Rau C, Guenzi M, et al. Laparoscopic omega-loop gastric bypass for the conversion of failed sleeve gastrectomy: early experience. Journal of visceral surgery. 2013;150(6):373–8.
Bruzzi M, Voron T, Zinzindohoue F, et al. Revisional single-anastomosis gastric bypass for a failed restrictive procedure: 5-year results. Surgery for Obesity & Related Diseases. 2016;12(2):240–5.
Salama TM, Sabry K. Redo surgery after failed open VBG: laparoscopic Minigastric bypass versus laparoscopic roux en Y gastric bypass-which is better? Minim Invasive Surg. 2016;2016:8737519.
Ghosh S, Bui TL, Skinner CE, et al. A 12-month review of Revisional single anastomosis gastric bypass for complicated laparoscopic adjustable gastric banding for body mass index over 35. Obes Surg. 2017;27:3048–54.
Chansaenroj P, Aung L, Lee WJ, et al. Revision procedures after failed adjustable gastric banding: comparison of efficacy and safety. Obes Surg. 2017;27:2861–7.
Chen CY, Lee WJ, Lee HM, et al. Laparoscopic conversion of gastric bypass complication to sleeve gastrectomy: technique and early results. Obes Surg. 2016;26(9):2014–21.
Chen MC, Lee YC, Lee WJ, et al. Diet behavior and low hemoglobin level after laparoscopic mini-gastric bypass surgery. Hepato-Gastroenterology. 2012;59(120):2530–2.
Chiu CC, Lee WJ, Wang W, et al. Prevention of trocar-wound hernia in laparoscopic bariatric operations. Obes Surg. 2006;16(7):913–8.
Saarinen T, Räsänen J, Salo J, et al. Bile reflux scintigraphy after mini-gastric bypass. Obes Surg. 2017;27(8):2083–9.
Lee WJ, Lee YC, Ser KH, et al. Revisional surgery for laparoscopic minigastric bypass. Surgery for Obesity & Related Diseases. 2011;7(4):486–91.
Mahawar KK, Reed AN, Graham YNH. Marginal ulcers after one anastomosis (mini) gastric bypass: a survey of surgeons. Clin Obes. 2017;7(3):151–6.
Mishra T, Lakshmi KK, Peddi KK. Prevalence of cholelithiasis and choledocholithiasis in morbidly obese south Indian patients and the further development of biliary calculus disease after sleeve gastrectomy, gastric bypass and mini gastric bypass. Obes Surg. 2016;26(10):2411–7.
Rutledge R. Hospitalization before and after mini-gastric bypass surgery. Int J Surg. 2007;5(1):35–40.
Salama TMS, Hassan MI. Incidence of biliary reflux esophagitis after laparoscopic omega loop gastric bypass in morbidly obese patients. J Laparoendosc Adv Surg Tech A. 2017;27(6):618–22.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict Statement
Dr. De Luca has nothing to disclose. Ms. Tie reports personal fees from Centre for Obesity Research and Education (CORE), grants from Apollo Endosurgery, grants from Novo Nordisc, outside the submitted work. Dr. Ooi reports personal fees from National Health and Medical Research Council, personal fees from Royal Australasian College of Surgeon, outside the submitted work. Dr. Himpens reports personal fees from Ethicon, personal fees from Medtronic, outside the submitted work. Dr. Higa has nothing to disclose. Dr. Carbajo reports he is the current President of the MGB-OAGB International Club. Dr. Mahawar reports he has been paid honoraria by Medtronic Inc. for mentoring consultant bariatric surgeons in the United Kingdom to help them start them start their One Anastomosis Gastric Bypass programme. Dr Shikora has nothing to disclose. Dr. Brown reports grants from Johnson and Johnson, grants from Medtronic, grants from GORE, personal fees from GORE, grants from Applied Medical, grants from Apollo Endosurgery, grants and personal fees from Novo Nordisc, personal fees from Merck Sharpe and Dohme, outside the submitted work.
Ethics Statement
Ethical approval is not required for this type of study.
Informed Consent
Informed consent is not required for this study.
Additional information
On behalf of the IFSO appointed task force reviewing the literature on MGB-OAGB
Appendix 1—Members of the IFSO Appointed Task Force Reviewing the Literature on MGB-OAGB
Appendix 1—Members of the IFSO Appointed Task Force Reviewing the Literature on MGB-OAGB
Maurizio De Luca—Italy
Kelvin Higa—USA
Tiffany Tie—Australia
Geraldine Ooi—Australia
Wendy Brown—Australia
Jacques Himpens—Belgium
Scott Shikora—USA
Rudolf Weiner—Germany
Miguel-A Carbajo—Spain
Kamal Mahawar—UK
Jean Marc Chevallier—France
Luigi Angrisani—Italy
Luque-de-Leon—Spain
Aparna G Bhasker—India
Alberto Sartori—Italy
Mario Musella—Italy
KS Kular—India
Emanuele Soricelli—Italy
Ramon VilallongaVilallonga—Spain
Muffazal Lakdawala—India
Enrico Facchiano—Italy
Alessio Corradi—Germany
Appendix 2
Rights and permissions
About this article
Cite this article
De Luca, M., Tie, T., Ooi, G. et al. Mini Gastric Bypass-One Anastomosis Gastric Bypass (MGB-OAGB)-IFSO Position Statement. OBES SURG 28, 1188–1206 (2018). https://doi.org/10.1007/s11695-018-3182-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-018-3182-3