Abstract
The aim of this study was to assess the feasibility of and venous leakage reduction in percutaneous vertebroplasty (PV) using a new high-viscosity bone cement (PMMA). PV has been used effectively for pain relief in osteoporotic and malignant vertebral fractures. Cement extrusion is a common problem and can lead to complications. Sixty patients (52 female; mean age, 72.2 ± 7.2) suffering from osteoporosis (46), malignancy (12), and angiomas (2), divided into two groups (A and B), underwent PV on 190 vertebrae (86 dorsal, 104 lumbar). In Group A, PV with high-viscosity PMMA (Confidence, Disc-O-Tech, Israel) was used. This PMMA was injected by a proprietary delivery system, a hydraulic saline-filled screw injector. In Group B, a standard low-viscosity PMMA was used. Postprocedural CT was carried out to detect PMMA leakages and complications. Fisher’s exact test and Wilcoxon rank test were used to assess significant differences (p < 0.05) in leakages and to evaluate the clinical outcome. PV was feasible, achieving good clinical outcome (p < 0.0001) without major complications. In Group A, postprocedural CT showed an asymptomatic leak in the venous structures of 8 of 98 (8.2%) treated vertebrae; a discoidal leak occurred in 6 of 98 (6.1%). In Group B, a venous leak was seen in 38 of 92 (41.3%) and a discoidal leak in 12 of 92 (13.0%). Reduction of venous leak obtained by high-viscosity PMMA was highly significant (p < 0.0001), whereas this result was not significant (p = 0.14) related to the disc. The high-viscosity PMMA system is safe and effective for clinical use, allowing a significant reduction of extravasation rate and, thus, leakage-related complications.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Percutaneous vertebroplasty (PV) consists of the injection of polymethylmethacrylate (PMMA) into a collapsed vertebra in order to obtain pain relief and mechanical strengthening of the vertebral body. Galibert and Deramond proposed this procedure for the treatment of aggressive vertebral hemangiomas of C2 [1], and at present, it is used extensively worldwide in osteoporotic and malignant vertebral fractures when conventional therapies are not effective or not well tolerated. Although, clinically, PV is a relatively safe and effective procedure for back pain treatment, several studies have reported some major complications that can lead to paraplegia and death [2–11]. The most often described complications concerned pulmonary embolism, soft tissue damage, and nerve root compression related to leakage of bone cement [12–19]. Bone cement leakage is detected very frequently in vertebroplasty, as it is seen to occur in 38% [20] to 72.5% [21] of patients with vertebral metastases, in 59.5% [22] to 65% [23] of patients with osteoporotic vertebral collapse, and in 81% of treated patients [24] when computed tomography (CT) is carried out after PV. Even if this frequent minimal leakage is well tolerated or asymptomatic in the large majority of patients, cement extravasation is the main source of clinically relevant complications, depending on the site and volume of leakage.
PMMA symptomatic leakages are, in some astonishingly reported cases, related to poor technique, where initial extravasation is not detected and injection not suspended, thus allowing extensive endocanalar cement perfusion [25] or massive lung embolization [12]; when PV technique and radiological equipment are optimal, leakages are due to the low viscosity of PMMA [26, 27]. To reduce PMMA leakages, some authors have proposed technical optimization and implementation such as vertebral venography [28, 29], gel-foam embolization [30], and kyphoplasty [31–33], but the results were not conclusive. Even if minor complications related to venous leakages in the posterior epidural plexus, such as radicular pain, can be successfully treated [34], a technical improvement to reduce these leakages is necessary. Adequate patient selection and accurate imaging in the hands of skilled operators remain the major points for minimizing the risk of complications. Baroud and coworkers demonstrated the linkage between the viscosity of the injected cement and leakages [35]. In their experimental model, cement leakage ceased completely when its viscosity was very high. However, this study concluded that no delivery system in clinical use could inject such a highly viscous cement.
Even the above conditions regarding the venous leakage of PMMA cannot always be avoided or predicted by the sort of fracture in the treated vertebra [9, 36]. The high rate of minimal and asymptomatic venous leakage, which can expose patients to the risk of major complications, led us to verify whether PV is feasible and safer using a very high-viscosity PMMA. The studied high-viscosity bone cement is a special new formulation of PMMA designed for injection through a proprietary delivery system. This cement reaches a constant putty-like viscosity immediately after mixing, without waiting a few minutes as in other PMMA cements, and remains at a constant high viscosity, and consistently injectable for 8–10 min before it solidifies. This high-viscosity cement can be injected using 11-, 13-, and 15-G needles as well as a side-firing needle which enables directional cement injection. Cadaveric laboratory testing showed that in two excised vertebrae fractured with a chisel, injection of common PMMA resulted in leakage through the defect, while high-viscosity cement maintained a spherical filling configuration confined to the vertebral body, with no leakage.
Materials and Methods
Study Population
Written informed consent was obtained before study inclusion from all patients in accordance with the national legislation and the Declaration of Helsinki. As the high-viscosity PMMA was CE approved for PV, institutional medical ethics committee approval was not required for this study.
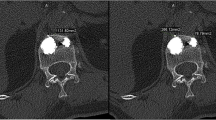
From February 2006, 60 patients underwent PV on 190 painful collapsed vertebrae not responding to conventional therapy (fractures aged from 3 to 24 months; mean, 6.9 ± 5.5 months). MRI and plain radiographs were evaluated before PV to assess correct indications to the procedure and to plan the levels to treat; up to seven vertebrae (1/30; 3.3%) were treated in the same sessions when MRI vertebral hyperintensity on T2-weighted sequences (Fig. 1) was concordant to focal pain evocated by clinical examination.
MR T2-stir-weighted sequences identified hyperintensity in multiple levels that were concordant with clinical pain in a patient with steroid–induced osteoporosis. Up to seven levels were treated in one session, with a good clinical outcome and without complication (a discoidal leak occurred in D11; white arrow)
Patients were randomized and divided into two homogeneous groups: in Group A, 30 patients (24 female and 6 male; age, from 56 to 84 years; mean, 71.3 ± 7.8 years) underwent PV on 98 vertebrae (39 dorsal and 59 lumbar; from D6 to L5), whereas in control Group B, 92 vertebrae (47 dorsal and 45 lumbar; from D4 to L5) were treated in 30 patients (28 female and 2 male; age, from 54 to 84 years; mean, 73.2 ± 6.4 years). Respectively, in the two groups 23 of 30 patients (76,6%) had osteoporotic vertebral fractures, 5 of 30 patients (16,6%) suffered from malignant fracture, 1 of 30 (3.3%) from multiple myeloma, and 1 of 30 (3.3%) from two symptomatic angiomas (Tables 1 and 2). In 12 of 46 osteoporotic patients (7 in Group A and 5 in Group B), multiple vertebral collapses were caused by high-grade osteoporosis induced by continued and long-lasting corticosteroid therapy.
Procedural Technique
All procedures were performed using a C-arm angiographic unit (Advantx Tilt-C; GE Medical Systems, Milwaukee, WI) with the patient in the prone position. Vertebroplasty was always performed under sterile conditions and IV antibiotics (1 g of vancomycin hydrochloride; Abbott SpA, Campoverde di Aprilia, LT, Italy) was administrated 3 days before and 5 days after the procedure. Patient pressure, heart rate, and oxygen saturation were monitored during the whole procedure. PV was always performed in local anesthesia by injecting 2 ml of 2% lidocaine hydrochloride (Lidosan; Industria Farmaceutica Galenica Senese, Monteroni d’Arbia, Siena, Italy) both at skin level and deep to include the periostium of the pedicle. An 11-G (125 vertebrae; 65.8%) or 13 G (65 vertebrae; 34.2%) beveled vertebroplasty needle was used in either a monolateral (188 vertebrae; 98.9%) or a bilateral (2 vertebrae; 1.1%) approach; the pathway was transpedicular in the lumbar spine and parapedicular intercostovertebral in the dorsal tract.
A core biopsy was always performed coaxially, using an 18-G through-cut needle (Magnum, Bard Inc., USA), to ensure the etiology of the fracture. Vertebral venography was not used following reports in the literature demonstrating no significant differences in frequency or amount of extravasation and no differences in clinical outcome between venography and no venography [28, 29].
In Group A patients PV was performed using a high-viscosity PMMA with a specially designed delivery system (Confidence Type I; Disc-O-Tech, Israel). According to the manufacturer’s specifications, after 1 min of mixing of the liquid monomer into the powder polymer (containing 30% barium sulfate for optimal fluoroscopic visualization), the cement shows the consistency of Plasticine (Fig. 2). The delivery system consists of a hydraulic saline-filled screw injector with a long connection tube allowing the operator’s hands to be out of the X-ray beam during the injection (Fig. 3). The reservoir containing the cement is then distally Luer-lock connected to 11- and 13-G proprietary vertebroplasty needles and proximally to the delivery system (Fig. 4). In patients in control Group B, PV was carried out with standard low-viscosity PMMA (Mendec Spine; Tecres, Sommacampagna, Italy) CE approved for vertebroplasty with a screw injector (CementoSet; Optimed, Germany).
Considering our previous clinical experience and the data in the literature on the filling volume and clinical outcome ratio [37, 38], from 1 to 3.5 ml (mean, 2.5 ± 1.1 ml) of cement was injected in the anterior two-thirds of each treated vertebra.
After the procedure all patients remained supine in bed for 1 h and had 6 h of clinical observations, then was discharged from the hospital.
Study Design
In each treated patient, CT scans with two-dimensional reconstruction (LightSpeed16; GE Medical Systems, Milwaukee, WI, USA) was performed 1 h after PV to identify bone cement extravasation. Postprocedural CTs were examined by two experienced radiologists (not by the operator) from two different radiology centers, and allowed precise evaluation of vertebral cement perfusion, PMMA leakages, and needle pathways and possible complications (Fig. 5). When a venous leak was detected, CT scan of the lungs was carried out to evaluate the possibility of PMMA embolism. Blinded data about PMMA leakages on every treated vertebra were collected by another radiologist.
The 11-point (0, no pain, to 10, worst possible pain) Visual Analog Scale (VAS) [39] and the Oswestry Self-Evaluation Disability Questionnaire [40] were compiled by the anesthesiologist before PV, 7 days after the procedure, and after 6 months; a clinical interview follow-up was performed every month during a 6-month period.
Statistical Analysis
The Wilcoxon paired signed rank test was used to evaluate significant differences (p < 0.05) on clinical pain regression on the VAS and Oswestry disability questionnaire in each group and the Student paired t-test to investigate differences in clinical outcome between the two groups. Fisher’s exact test was conducted to evaluate differences in PMMA leakages between Group A and Group B and to compare the results of Group A to other published series.
Statistical studies were performed using Graphpad Instat software (GraphPad Instat version 3.0b for Mac; GraphPad Software, San Diego, CA, USA; http://www.graphpad.com) [41].
Results
Clinical Outcome
PV was feasible in all patients without any early or delayed complications, achieving a good clinical outcome; in Group A the mean VAS score before PV of 8.4 ± 1.4 improved significantly, to a mean of 0.5 ± 0.7 at the end point after the procedure (two-tailed p < 0.0001, Wilcoxon signed rank test). In Group B the mean VAS of 8.3 ± 1.5 preprocedure dropped to 0.9 ± 0.9 postprocedure (p < 0.0001). Patients’ quality of life, evaluated by the Oswestry Disability Self-Evaluation Questionnaire, also improved (p < 0.0001) significantly in the two groups, from a mean index of 59.7% ± 18.0 to 8.1% ± 5.6% (Group A) and from 67.7% ± 12.5% to 8.7% ± 7.6% (Group B). No differences in clinical outcome were noted between the two groups (p = 0.05, paired t-test).
Two patients in Group A (6.6%) and one patient in Group B (3.3%) suffering from osteoporosis (nos. 7 and 15, Table 1; no. 10, Table 2) reported a new fracture within 2 months of the first procedure on the contiguous above vertebra; they were all successfully retreated without any complication. No statistical difference in new fracture rate was demonstrated between the groups.
PMMA Leakages
No symptomatic cement leakages occurred in the two groups. In Group A, a minimal asymptomatic venous leak was detected on postprocedural CT in 6 of 30 patients (20%) and in 8 of 98 treated vertebrae (8.2%). In two of six patients fluoroscopy did not detect the venous leakage that CT showed after the procedure due to the high sensibility of CT as previously demonstrated [24, 42]. PMMA leakage into the disc occurred in 6 of 30 patients (20%) and on 6 of 98 treated levels (6.1%); they were all demonstrated on procedural fluoroscopy. Considering the different pathology, 4 of 23 osteoporotic patients (17.4%) reported a venous leak in 6 of 77 (7.8%) treated vertebrae, whereas a venous leak occurred in 2 of 6 (33.3%) malignant patients on 2 of 19 (10.5%) levels. Two symptomatic angiomas, treated in the same patient, did not show any leakage. A discoidal leak was observed in 5 of 23 osteoporotic patients (21.7%) in 5 of 77 vertebrae (6.5%) and in 1 of 6 patients (16.6%) with malignancy in 1 of 19 (5.3%) vertebrae. Cement vertebral perfusion assumed a spherical configuration in osteoporosis (Fig. 6), whereas deposition was more diffuse and irregular in metastases (Fig. 7).
In Group B, postprocedural CT demonstrated an asymptomatic venous leak in 24 of 30 patients (80%) and on 38 of 92 treated vertebrae (41.3%). PMMA into the disc was detected in 11 of 30 patients (36.6%) and 12 of 92 vertebrae (13%). Nineteen of 23 osteoporotic patients (82.6%) had a venous leak on 30 of 71 (42.3%) levels; a venous leak occurred in 5 of 6 (83.3%) malignant patients on 8 on 19 (42.1%) levels, whereas no leak was observed in the two symptomatic angiomas. A leak occurred into the disc in 8 of 23 osteoporotic patients (34.8%) in 9 of 71 vertebrae (12.7%) and in 3 of 6 cancer patients (50%) in 3 of 19 (15.8%) vertebrae. In one osteoporotic patient in this group, CT showed small asymptomatic PMMA emboli in the left lung.
Data Analysis
Comparison of data showed a statistically highly significant difference (p < 0.0001, Fisher’s exact test) of venous leakages between patients treated with high-viscosity cement (Group A) and patients in Group B. On the other hand, this difference was not significant concerning leakages into the disc (p = 0.1374) (Table 3).
Venous leakages detected in osteoporotic vertebrae in Group A were also compared to other published series (Table 4) where postprocedural CT was carried out to assess vertebral PMMA perfusion and leak-related complications [23, 24, 30, 42–44]. The Confidence bone cement and delivery system showed a statistically significant difference (p value from 0.0303 to <0.0001, Fisher’s exact test) compared to all previous studies concerning venous leakages; this difference is statistically significant (p = 0.0303) even if PV, performed with standard low-viscosity bone cement injection, is performed by preinjection gelfoam embolization [30]. If leakage into the disc is analyzed, the high-viscosity cement system showed a statistically significant reduction (p = 0.0069) from that reported by Jung and collaborators [44] but did not demonstrate any statistically significant differences from PV performed with standard bone cement by Bhatia et al [30] and Perez-Higueras and coworkers [43] (p = 0.2126 and p = 1.0000, respectively).
Discussion
This study investigated whether PV is feasible and whether the rate of cement leakage can be reduced by means of a high-viscosity, specifically designed, bone cement. In the majority of patients (23/30; 76.6%) treated with high-viscosity PMMA, the underlying cause was osteoporosis and the demographic characteristics report elderly patients (71.3 ± 7.8 years), with female prevalence (24/30; 80%); the same distribution is applicable to control Group B. The reason for testing the new high-viscosity cement was that such demographic characteristics are also found in most of the series reported in the literature, and to date, painful osteoporotic vertebral collapse not responding to conservative therapy is the main indication for PV [23, 26, 28, 45–47]. The good clinical outcome achieved by this minimally invasive procedure and the wide distribution of digital fluoroscopy and CT are increasing PV performance worldwide. These have led to PV’s being employed even at hospitals where the procedure is not performed daily. Although PV is generally safe, even well-trained physicians [9, 45–49] and high-quality imaging cannot prevent PMMA leakages, thus the patient is virtually exposed to a risk of serious complication. Furthermore, PV can be very dangerous, independent of the cement used, if proper technique is not applied [10, 12, 19, 25] or patient selection is inadequate [5]. As a consequence of these dramatic reports, the risk of potential leakage and their complications often prevents clinicians from offering the procedure to their patients.
Bhatia and coworkers [30] recently demonstrated that significant PMMA leakage reduction can be achieved by embolization with gelfoam preinjection; this is an easy and well-known method for interventional radiologists but it can be unfamiliar to orthopedics, neurosurgeons, and anesthesiologists who also perform PV; on the other hand, the high-viscosity PMMA system does not substantially change the technique of PV. The link between the viscosity of the bone cement and leakage was recently demonstrated by Baroud and coworkers, however, they concluded that delivery of high-viscosity cement may approach or exceed the human physical limit of injection forces [35]. However, the hydraulic cement delivery system enables the introduction of constant high-viscosity cement immediately after mixing the cement components during a 8- to 10-min injection. Use of a system with high-viscosity PMMA demonstrates, in our experience, a highly significant reduction of extravasation into the vein and, consequently, into the systemic venous circulation. This difference was demonstrated to occur in comparison to standard PV with low-viscosity vertebroplasty-designed bone cement (p < 0.001) but also when gelfoam embolization preceded standard PV (p = 0.0303). Although all detected leakages were asymptomatic and high-viscosity PMMA did not change patients’ clinical condition at all compared to low-viscosity PMMA, in our opinion, a reduced PMMA leakage rate makes PV a safer procedure.
Finally, our data show that a relative (statistically not significant) reduction in the rate of leakages into the disc was achieved by use of high-viscosity PMMA. In an osteoporotic vertebral fracture the leak into the disc more frequently occurs through an intravertebral vacuum cleft or through a perforation of the endplate created by the needle tip [50]. Although this event is not related to serious complications and does not affect the clinical outcome [50], it could increase the risk of a new fracture in the contiguous vertebra [51]; in our series two patients in Group A and one patient in Group B who reported a new vertebral fracture did not show any leak into the disc during the first treatment. Our results did not show a statistically significant difference between the two groups (p = 0.1374) or from results reported by Bhatia and coworkers [30] (p = 0.2126) and Perez-Higueras et al [43] (p = 1.000). Jung et al [44] reported a rate of 22.3% discoidal leak in 85 osteoporotic treated levels that are more similar to the 77 vertebrae treated in our series with the high-viscosity system, where the rate of 6.5% shows a significant difference (p = 0.0069).
As application of the high-viscosity PMMA system does not substantially change the technique of PV as it is routinely performed by different physicians, in our opinion, it should be considered to reduce the rate of asymptomatic leakages and, consequently, the risk of complications related to cement extravasation. If PV is performed at centers where it is not part of the daily routine, the Confidence system could avoid further complications, usually due to limited experience of the operator in correct evaluation of the consistency of standard PMMA before injection and in manual syringe PMMA injection.
A limitation of this study could be that the same experienced operator used to avoid complications even with low-viscosity cement tested the high-viscosity PMMA; only a multicenter randomized study can assess whether physicians with different degrees of experience can also achieve this reduction in leakages.
Conclusion
This study has demonstrated that utilization of high-viscosity PMMA (the Confidence system) during routine PV is safe and feasible and can significantly reduce venous cement leakage without any substantial changes in the vertebroplasty technique. Serious complications reported in the literature are related to PMMA leakage in most cases; we think that the application of high-viscosity bone cement can reduce the complication rate, improving the safety of the PV technique, and consequently, even more referring physicians will propose this procedure to patients with vertebral collapses not responding to conservative therapy whose quality of life is poor.
References
Galibert P, Deramond H, Rosat P et al (1987) Preliminary report on the percutaneous vertebroplasty with acrylic cement as a treatment of vertebral angioma. Neurochirurgie 33:166–168
Vasconcelos C, Gailloud Ph, Martin JB et al (2001) Transient arterial hypotension induced by polymethylmethacrylate injection during percutaneous vertebroplasty [Letter). J Vasc Interv Radiol 12:1001–1002
Padovani B, Kasriel O, Brunner P et al (1999) Pulmonary embolism caused by acrylic cement: a rare complication of percutaneous vertebroplasty. Am J Neuroradiol 20:375–377
Scroop R, Eskridge J, Britz GW (2002) Paradoxical cerebral arterial embolization of cement during intraoperative vertebroplasty: case report. Am J Neuroradiol 23:868–870
Syed MI, Jan S, Patel NA et al (2006) Fatal fat embolism after vertebroplasty: identification of the high-risk patient. Am J Neuroradiol 27(2):343–345
Freitag M, Gottschalk A, Schuster M et al (2006) Pulmonary embolism caused by polymethylmethacrylate during percutaneous vertebroplasty in orthopaedic surgery. Acta Anaesthesiol Scand 50(2):248–251
MacTaggart JN, Pipinos II, Johanning JM et al (2006) Acrylic cement pulmonary embolus masquerading as an embolized central venous catheter fragment. J Vasc Surg 43(1):180–183
Baumann A, Tauss J, Baumann G et al (2006) Cement embolization into the vena cava and pulmonal arteries after vertebroplasty: interdisciplinary management. Eur J Vasc Endovasc Surg 31(5):558–561
Barragan-Campos HM, Vallee JN, Lo D et al (2006) Percutaneous vertebroplasty for spinal metastases: complications. Radiology 238(1):354–362
Chung SE, Lee SH, Kim TH et al (2005) Renal cement embolism during percutaneous vertebroplasty. Eur Spine J 14:1–5
Laredo JD, Hamze B (2005) Complications of percutaneous vertebroplasty and their prevention. Semin Ultrasound CT MR 26(2):65–80
Monticelli F, Meyer HJ, Tutsch-Bauer E (2005) Fatal pulmonary cement embolism following percutaneous vertebroplasty (PVP). Forensic Sci Int 20;149(1):35–38
Choe DH, Marom EM, Ahrar K et al (2004) Pulmonary embolism of polymethyl methacrylate during percutaneous vertebroplasty and kyphoplasty. AJR 183(4):1097–1102
Yoo KY, Jeong SW, Yoon W et al (2004) Acute respiratory distress syndrome associated with pulmonary cement embolism following percutaneous vertebroplasty with polymethylmethacrylate. Spine 29(14):E294–E297
Francois K, Taeymans Y, Poffyn B et al (2003) Successful management of a large pulmonary cement embolus after percutaneous vertebroplasty: a case report. Spine 28(20):E424–E425
Koessler MJ, Aebli N, Pitto RP (2003) Fat and bone marrow embolism during percutaneous vertebroplasty. Anesth Analg 97(1):293–294
Bernhard J, Heini PF, Villiger PM (2003) Asymptomatic diffuse pulmonary embolism caused by acrylic cement: an unusual complication of percutaneous vertebroplasty. Ann Rheum Dis 62(1):85–86
Jang JS, Lee SH, Jung SK (2002) Pulmonary embolism of polymethylmethacrylate after percutaneous vertebroplasty: a report of three cases. Spine 27(19):E416–E418
Chen HL, Wong CS, Ho ST et al (2002) A lethal pulmonary embolism during percutaneous vertebroplasty. Anesth Analg 95(4):1060–1062
Weill A, Chiras J, Simon JM et al (1996) Spinal metastases: indications for and results of percutaneous injection of acrylic surgical cement. Radiology 199:241–247
Cotten A, Dewatre F, Cortet B et al (1996) Percutaneous vertebroplasty for osteolytic metastases and myeloma: effects of the percentage of lesion filling and the leakage of methyl methacrylate at clinical follow-up. Radiology 200:525–530
Gaughen J, Jensen ME, Schweickert PA et al (2002) Relevance of antecedent venography in percutaneous vertebroplasty for the treatment of osteoporotic compression fractures. Am J Neuroradiol 23:594–600
Cortet B, Cotten A, Boutry N et al (1999) Percutaneous vertebroplasty in the treatment of osteoporotic vertebral compression fractures: an open prospective study. J Rheumatol 26:2222–2228
Schmidt R, Cakir B, Mattes T et al (2005) Cement leakage during vertebroplasty: an underestimated problem? Eur Spine J 14(5):466–473
Chen YJ, Tan TS, Chen WH et al (2006) Intradural cement leakage: a devastatingly rare complication of vertebroplasty. Spine 31(12):E379–E382
Mathis JM (2003) Percutaneous vertebroplasty: complications avoidance and tTechnique optimization. Am J Neuroradiol 24:1697–1706
Moreland DB, Landi MK, Grand W (2001) Vertebroplasty: techniques to avoid complications. Spine J 1(1):66–71
Vasconcelos C, Gailloud P, Beauchamp NJ et al (2002) Is percutaneous vertebroplasty without pretreatment venography safe? Evaluation of 205 consecutives procedures. Am J Neuroradiol 23(6):913–917
Wong W, Mathis J (2002) Is intraosseous venography a significant safety measure in performance of vertebroplasty? J Vasc Interv Radiol 13:137–138
Bhatia C, Barzilay Y, Krishna M et al (2006) Cement leakage in percutaneous vertebroplasty: effect of preinjection gelfoam embolization. Spine 31(8):915–919
Bouza C, Lopez T, Magro A et al (2006) Efficacy and safety of balloon kyphoplasty in the treatment of vertebral compression fractures: a systematic review. Eur Spine J 15(7):1050–1067
Fourney DR, Schomer DF, Nader R et al (2003) Percutaneous vertebroplasty and kyphoplasty for painful vertebral body fractures in cancer patients. J Neurosurg 98:21–30
Phillips FM, Todd Wetzel F, Lieberman I et al (2002) An in vivo comparison of the potential for extravertebral cement leak after vertebroplasty and kyphoplasty. Spine 27:2173–2178
Kelekis AD, Martin JB, Somon T et al (2003) Radicular pain after vertebroplasty: compression or irritation of the nerve root? Initial experience with the “cooling system.” Spine 28(14):E265–E269
Baroud G, Crookshank M, Bohner M (2006) High-viscosity cement significantly enhances uniformity of cement filling in vertebroplasty: an experimental model and study on cement leakage. Spine 31(22):2562–2568
Baumann C, Fuchs H, Kiwit J et al (2007) Complications in percutaneous vertebroplasty associated with puncture or cement leakage. CardioVasc Interv Radiol 2007 30(2):161–168
Molloy S, Mathis JM, Belkoff SM (2003) The effect of vertebral body percentage fill on mechanical behavior during percutaneous vertebroplasty. Spine 28(14):1549–1554
Molloy S, Riley LH III, Belkoff SM (2005) Effect of cement volume and placement on mechanical-property restoration resulting from vertebroplasty. Am J Neuroradiol 26(2):401–404
Farrar JT, Young JP, LaMoreaux L et al (2001) Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain 94:149–158
Fairbank JCF, Couper JF, Davies JBF et al (1980) The Oswestry low back pain disability questionnaire. Physiotherapy 66:271–273
Motulsky H (1995) Intuitive biostatistics. Oxford University Press, New York
Yeom JS, Kim WJ, Choy WS et al (2003) Leakage of cement in percutaneous transpedicular vertebroplasty for painful osteoporotic compression fractures. J Bone Joint Surg Br 85(1):83–89
Perez-Higueras A, Alvarez A, Rossi RE et al (2002) Percutaneous vertebroplasty: long-term clinical and radiological outcome. Neuroradiology 44:950–954
Jung JY, Lee MH, Ahn JM (2006) Leakage of polymethylmethacrylate in percutaneous vertebroplasty: comparison of osteoporotic vertebral compression fractures with and without an intravertebral vacuum cleft. J Comput Assist Tomogr 30(3):501–506
Peh WCG, Gilula LA, Peck DD (2002) Percutaneous vertebroplasty for severe osteoporotic vertebral body compression fractures. Radiology 223:121–126
Gangi A, Guth S, Imbert JP et al (2003) Percutaneous vertebroplasty: indications, technique, and results. Radiographics 23(2):e10
Zoarski GH, Snow P, Olan WJ et al (2002) Percutaneous vertebroplasty for osteoporotic compression fractures: quantitative prospective evaluation of long-term outcomes. J Vasc Interv Radiol 13:139–148
Hochmuth K, Proschek D, Schwarz W et al (2006) Percutaneous vertebroplasty in the therapy of osteoporotic vertebral compression fractures: a critical review. Eur Radiol 16:998–1004
Anselmetti GC, Corrao G, Monica PD et al (2007) Pain relief following percutaneous vertebroplasty: results of a series of 283 consecutive patients treated in a single institution. CardioVasc Interv Radiol 30(3):441–447
Mirovsky Y, Anekstein Y, Shalmon E et al (2006) Intradiscal cement leak following percutaneous vertebroplasty. Spine 31(10):1120–1124
Lin EP, Ekholm S, Hiwatashi A et al (2004) Vertebroplasty: cement leakage into the disc Increase the risk of new fracture of adjacent vertebral body. Am J Neuroradiol 25:175–180
Acknowledgment
The authors acknowledge Mrs. Olga Bruno for her kind and lovely collaboration.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anselmetti, G.C., Zoarski, G., Manca, A. et al. Percutaneous Vertebroplasty and Bone Cement Leakage: Clinical Experience with a New High-Viscosity Bone Cement and Delivery System for Vertebral Augmentation in Benign and Malignant Compression Fractures. Cardiovasc Intervent Radiol 31, 937–947 (2008). https://doi.org/10.1007/s00270-008-9324-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-008-9324-6