Abstract
Vereda is a wetland ecosystem typical of the Cerrado biome characterized by diverse vegetation with dynamic and transitional areas of riparian forests, gallery forests, flooded forests, and humid grassland. In general, they are associated with controlling the flow of the water table, carbon storage, and high biodiversity and are essential to the maintenance of most rivers in the Cerrado biome. Besides that, Vereda is poorly studied, especially zooplanktonic groups. To lessen this knowledge gap, we evaluated the effects of seasonality and environmental predictors on the zooplanktonic community of Vereda. For that, we sampled zooplanktonic assemblages in the Veredas in the dry and wet seasons. We found environmental influence but not for the zooplankton community. The characteristic low conductivity seems to be an important environmental filter for zooplankton species occurrence in these systems, since the Veredas that registered major levels of richness and density were those with high electrical conductivity. Highlighting that some zooplankton species with a more restricted distribution were detected in this study: Acroperus tupinamba, which occurs in Brazil and Ecuador; Monospilus sp., for which only two species of this genus were registered in Brazil, both with restricted distribution, found thus far in only two protected areas of the Cerrado biome.
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wetlands are aquatic ecosystems that are essential worldwide because they are very productive and harbor high biodiversity. In parallel, these aquatic ecosystems provide various ecosystem services, such as water purification, nutrient cycling, and food resources (Mitsch et al. 2015). Recognizing their importance, the Ramsar Convention advocates for the protection and sustainable use of wetlands (https://www.ramsar.org/). Nonetheless, human activities are a significant threat to wetlands; according to Davidson (2014), more than 50% of the total area of wetlands was lost, making them the most vulnerable natural ecosystem (Wantzen and Junk 2000; Hu et al. 2017; Lynch et al. 2023). Despite their importance, wetlands are understudied (Junk 2013; Junk et al. 2014) worldwide. In Brazil, there has been emphasis on large wetlands, such as the Pantanal, Amazon, and Paraná, and little attention has been given to smaller wetlands, such as the Veredas of the Cerrado biome (Brazilian savannah) (Junk 2013; Junk et al. 2014).
Vereda is a typical ecosystem in the Cerrado, characterized by diverse vegetation, with transitional areas composed of riparian forests, gallery forests, flooded forests, and humid grassland (Da Cunha et al. 2015). The Veredas are formed adjacent to small watercourses (Lima and Silveira 1991; Ab’Sáber 2003), formed by hydromorphic soils and the presence of organic turfs associated with shallow water tables (Oliveira et al. 2009; Bijos et al. 2017). Wetlands of the Veredas type can be observed in flat terrain (i.e., lowlands) or steep areas (i.e., hills or plateau areas). In lowlands, the most common places to form, Veredas cover extensive areas and do not have well-defined watercourses. On the other hand, the Veredas found in valleys or steep areas are less extensive and have better-defined watercourses, in general, small streams (Silveira et al. 2022). In the Cerrado, Veredas play important roles in regulating the water table and maintaining rivers (Lima and Silveira 1991; Ab’Sáber 2003).
There have been few studies dedicated to characterizing the biodiversity in the Veredas (Gomes et al. 2020; Faquim et al. 2021), and even fewer focus on the zooplanktonic groups, resulting in knowledge gaps about local biodiversity. For example, among zooplanktonic groups, it is estimated that for rotifers, at least 30% of the species recorded in Brazil come from aquatic environments in the Cerrado, and approximately 4% are possibly endemic (Padovesi-Fonseca et al. 2015). Similar knowledge gaps are expected for other zooplanktonic groups, given their high endemism levels (Padovesi-Fonseca et al. 2015, 2021). Furthermore, only a small proportion of studies about zooplankton in the Cerrado addressed the different zooplankton groups together (Alarcão et al. 2014; Pinese et al. 2015; Gomes et al. 2020; Picapedra et al. 2022). Most studies have been limited to a single group of these organisms, as is the case for microcrustaceans (Sousa and Elmoor-Loureiro 2008, 2013; Sousa et al. 2013; Elmoor-Loureiro 2014; Fonseca et al. 2018).
For zooplankton communities, it is well established that different habitats and local environmental conditions play a crucial role in the survival and reproduction of different species in ecosystems (i.e., ecological niche theory—Hutchinson 1957). Studies on zooplankton community ecology have emphasized the influence of local factors such as morphometric characteristics (Paquette et al. 2022), seasonal and climate (Stephan et al. 2017), and water quality on the spatial distribution of these organisms in ecosystems (Padovesi-Fonseca and Rezende 2017; Wan Maznah et al. 2018). On the other hand, other studies have highlighted the influence of spatial factors and dispersal ability as important contributors to establishing and structuring the community (i.e., Unified Neutral Theory of Biodiversity and Biogeography—Hubbell 2001). Previous studies have shown that seasonal, environmental, and spatial factors contribute to clarify the structuring of zooplanktonic communities in Cerrado streams (Gomes et al. 2020; Padovesi-Fonseca et al. 2021; Pedroso et al. 2021).
Here, it is evident that there is a need to lessen gaps in biodiversity knowledge of the mechanisms that determine the distribution and structure of zooplanktonic communities in the Veredas. For this to happen, we want to contribute to current knowledge by evaluating the influence of seasonal, environmental, and spatial factors on the structuring of zooplanktonic communities in the Veredas. For that, we outline the following specific hypotheses and premises: (i) Due to the little knowledge available about Veredas, it is expected to register new occurrences or new records of zooplanktonic species. (ii) As a complex and dynamic environment (Da Cunha 2015), we expect the environmental variables to reveal dissimilarities between the sampling seasons. (iii) Knowing that the zooplankton community responds quickly to environmental changes (Fernández-Aláez et al. 2018), we expect that the zooplankton community will respond seasonally, and (iv) we expect that environmental factors will influence the composition of zooplankton more than spatial factors.
Methods
Study area
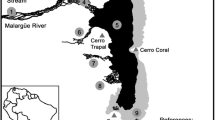
Veredas are localized between the cities of Barra do Garças and Nova Xavantina, state of Mato Grosso (Cerrado Biome), and distributed in headwaters of the microbasins of the Araguaia and Rio das Mortes (Fig. 1). The climate is classified as Aw according to the Köppen classification and has two defined seasons: dry winter and rainy summer (Kottek et al. 2006; Alvares et al. 2013). The annual mean temperature ranges from 22 to 25 °C, and the annual mean precipitation ranges from 1200 to 1800 mm (Alvares et al. 2013). The altitude above sea level ranges from 734 to 300 m. The Veredas streams sampled are waterways from first to third order according to the classification of Sthaller (Horton 1945; Strahler 1957). The streams associated with the Veredas are characterized by vegetation surrounded by grasses and herbs and in general with the presence of Mauritia flexuosa (Buriti) palms (Ribeiro and Walter 2008). The palms do not form a canopy, leaving vegetation coverage ranging from 5 to 10%.
Zooplanktonic community sampling
The zooplanktonic communities were sampled in the dry season (2016–2017) and rainy season (2020). We sampled 14 streams during the dry season, and out of those, 11 streams were resampled during the rainy season. To perform sampling, we chose a lentic stretch of stream and filtered 600 L of water through a mesh plankton net (68 μm) using a motor pump (Honda WX10T model). Then, we fixed the collected material in 4% formaldehyde buffered with sodium tetraborate (borax). The zooplankton was identified, and the density of each taxon was counted using a binocular optical microscope (Olympus CX23) and 1 mL Sedgewick-Rafter counting chambers. Then, the entire sample was analysed. The identification of the collected material was carried out using specialized bibliographic material (e.g., Koste 1978; Dussart and Defaye 1995; Segers 1995; Elmoor-Loureiro 1997; Suárez-Morales et al. 2020).
Environmental data
To measure environmental variables such as pH, turbidity, water conductivity, water temperature, dissolved oxygen, and total dissolved solids, we used a multiparameter limnological probe (Horiba, model U-50) at a single point of the stream. We measured depth and width at five points in the sample section. The surface water flow was measured by the time it takes a floating object (a rubber ball) to travel for one meter in the center of the stream channel.
Data analysis
To assess the environmental dissimilarity between the sampling seasons (hypothesis ii), we first performed a Principal Component Analysis (PCA) with the environmental variables. The aim PCA is to demonstrate the relationship of environmental variables with the sampled sites between seasons (Legendre and Legendre 1998). Additionally, we performed nonmetric multidimensional scaling (NMDS) using a Euclidean distance matrix to detect the environmental gradient in each sampling season. The environmental variables were standardized (scale x to zero mean and unit variance) and checked for high correlations between them, but no high correlation (r > 6.0) was found. Then, we performed an ANOSIM to test the environmental dissimilarity between the dry and rainy seasons.
To characterize the zooplankton community (hypothesis iii), we first performed a T test using log-transformed total density data and total richness data (response variables) and dry and rainy seasons (independent variables). Both data sets were previously tested for normality (Shapiro‒Wilk test). The zooplankton composition data were obtained by the Hellinger transformation of zooplankton density data. Later, we performed an ANOSIM accompanied by NMDS to compare the zooplankton composition in the dry and rainy seasons. For these analyses, we used the functions “anosim” and “metaMDS” in the vegan package (Oksanen et al. 2022).
To investigate the primary predictor for the zooplankton community (hypothesis iv), we performed a multiple regression on distance matrices (Zapala and Schork 2006; Lichstein 2007; Haynes et al. 2013). The response variable was a zooplanktonic community matrix (represented by the Bray‒Curtis distance of Hellinger transformed density data) in the dry and rainy seasons separately. The independent variables were the spatial predictor (an Euclidean distance matrix of geographical coordinates) and the environmental predictor (the Euclidean distance of all standardized environmental variables). The Veredas that could not be sampled in the rainy season (V05, V06, V10) were also removed from the matrices of the dry season. Multiple regression on distance matrices (MRM) was carried out using the “MRM” function available on the ecodist package (Goslee and Urban 2007).
To achieve a multivariate response between environmental predictors and biological data, we performed a redundancy analysis (RDA). We then used forward variable selection to obtain an ordination constrained to the explanatory variable of interest (P < 0.05). For that, we used the “ordistep” function available on the vegan package (Oksanen et al. 2022). We checked the collinearity with the variance inflation factor (VIF) using the “vif” function available in the car package (Fox and Weisberg 2019). VIF values < 10 indicate variables that are independent of each other (Graham 2003; Borcard et al. 2018). For the RDA in the rainy season, the variables conductivity and dissolved oxygen were removed to control for multicollinearity. Prior to analysis, the environmental data matrix was standardized, and the zooplankton density matrix was transformed using log(x + 1). All analyses were performed in the R programming environment (R Core Team 2022).
Results
Environmental characterization
In both seasons, the Veredas streams sampled had very low electrical conductivity and total dissolved solids. The availability of dissolved oxygen varied widely in the dry season, while the mean value of dissolved oxygen was lower during the rainy season. The pH was always below seven, with the mean value being lower during the rainy season. On average, the depth of the Veredas streams increased considerably during the rainy season, along with the water temperature, while the water flow decreased slightly. The average stream width increased little during the rainy season, and the standard deviation of these values was lower at this time of the year, indicating that the Veredas had more similar widths in the rainy season (Table 1).
The PCA explained 60% of the environmental variability in both seasons in the first two axes (Axis 1 and 2, explained 36 and 24% of the variability, respectively). Furthermore, the ANOSIM (R = 0.32; P = 0.001) and the NMDS analysis (stress = 0.105) showed differences between seasons. These results showed that the environmental characteristics of the streams were different between the dry and rainy seasons (Fig. 2a, b).
a Principal component analysis (PCA) with all environmental variables of Veredas streams in the Araguaia River basin in the dry (D) and rainy (R) seasons; b nonmetric multidimensional scaling (NMDS) of Veredas streams in the Araguaia River basin in the dry and rainy seasons using the environmental variables
Zooplankton characterization
We recorded a total of 69 zooplankton taxa, including 41 rotifers, 16 cladocerans, and 12 copepods (9 adult copepods). Of these, 48 taxa were found in the dry season samples, and 43 were found in the rainy season (21 of which were not found in the dry season). The rotifers and cladocerans were mostly identified down to the species level. Whereas copepods, the adult forms were distributed into the family (for Harpacticoida) and genus (for Cyclopoida), and the other stages of development were counted as distinct taxa, due to their different ecological roles: nauplii (larval stage), Harpacticoida copepodites, and Cyclopoida copepodites (stage juvenile). We highlight the record of the genus Monospilus sp. (Cladocera) in Vereda V10 in the dry season as a new occurrence for this region (Table S1), and some zooplankton species with a more restricted distribution were detected in this study: Acroperus cf. tupinamba.
Veredas with higher richness also presented a higher density of organisms during both dry and rainy seasons. The zooplanktonic community was similar across all Veredas, with a greater proportion of rotifer group compared to other zooplanktonic groups as well as for the density of organisms. There were no significant differences in zooplankton richness (t = − 0.8, df = 10, P = 0.442), density (t = − 0.54, df = 10, P = 0.604) and composition (ANOSIM R = 0.06; P = 0.121) between seasons. The similarity in the zooplanktonic community between the two seasons was evident in the NMDS ordering analysis (stress = 0.195, Fig. 3).
By analysing the data for each climate season, we found that environmental conditions among Veredas were an important predictor for zooplanktonic community variability during the rainy season but not in the dry season (Table 2). The forward selection in RDA revealed two significant environmental variables (water temperature and total dissolved solids) as the most important predictors for rainy season data (R²adj = 0.44, F = 4.94, P = 0.001). For dry season data, dissolved oxygen was identified as the most important predictor of zooplankton density data (R²adj = 0.10, F = 2.46, P = 0.037). For the entire data set, we identified conductivity as the most important environmental variable for explaining zooplankton density data (R²adj = 0.12, F = 3.92, P = 0.008).
R²adj adjusted regression coefficient, P p value
Discussion
There is still limited knowledge about the biodiversity of the Veredas ecosystems, especially regarding microscopic groups such as zooplankton (Junk et al. 2006; Fonseca et al. 2018; Pedroso et al. 2021). The zooplankton taxa listed in this study (Table S1) are primarily cosmopolitan and neotropical (Smirnov 1996; Segers 2007). Among those, Acroperus cf. tupinamba has a more restricted distribution, with records in the Neotropical region registered thus far in Brazil and Ecuador (Sinev and Elmoor-Loureiro 2010). Notably, the genus Monospilus sp. was registered only in the dry season of the Veredas stream V10. In Brazil, only two protected areas of the Cerrado biome reported the occurrence of two species of this genus, Monospilus brachyspinus and Monospilus macroerosus (Sousa et al. 2017, 2018). As predicted in our hypothesis (i), our study could increase knowledge about the species that can be found in Veredas streams. Therefore, encouraging studies about biodiversity in understudied regions such as Veredas is necessary to lessen these knowledge gaps.
Veredas streams are mainly lotic water. In general, lotic ecosystems are unfavourable for the development of zooplanktonic organisms, due to rapid temperature fluctuations, water fast flows, and other factors (see more at Aggio et al. 2022). Zooplankton prefer backwater areas (Padovesi-Fonseca et al. 2021), but some taxa tolerate these conditions well (Matsumura-Tundisi et al. 2015), such as those adapted to living in the littoral zone of aquatic ecosystems. For example, Rotifera was the most abundant group in the sampled locations. This dominance pattern can be explained by the morphological and adaptive characteristics of this group, such as relatively small bodies, short life cycles, high reproductive rates, and predominantly parthenogenetic reproduction and resistance eggs (Allan 1976; Wallace et al. 2006). Among the organisms identified in the samples, the majority were representatives of littoral habit zooplankton, including rotifers of the genus Lecane (Segers 1996) and cladocerans of the family Chydoridae (Elmoor-Loureiro 1997). Copepods of the Cyclopoida and Harpacticoida orders also represent organisms with littoral and benthic habits (Esteves 1998).
The hydrological dynamics of drought and rain influenced the environmental conditions of the Veredas streams. Our study showed significant differences between the climatic seasons regarding the environmental gradient, in line with what was predicted by hypothesis (ii). Veredas are complex and heterogeneous systems with environmental characteristics that vary depending on location. The environmental structures of these locations can be determined by geological characteristics and historical factors associated with changes in relief (Gordon et al. 1997). For example, the type of bedrock in which the stream is located can influence the amount of solids dispersed in the water, while soil conditions can influence vegetation composition on the streambanks (Lewis 2008). The shape of the relief and its slope can also determine the flow of water, with steeper environments tending to have greater water velocity and narrower and deeper streams compared to less steep environments (Gordon et al. 1997; Lewis 2008).
Although it was not possible to find statistical differences in the zooplankton community in relation to seasonality, which refutes our hypothesis (iii), environmental variables are often important predictors of biological communities (e.g., Pinel-Alloul 1995; Bini et al. 2008; Declerck et al. 2011; Lopes et al. 2018; Pedroso et al. 2021). To Veredas streams was to recognize that conductivity, dissolved oxygen, water temperature, and total dissolved solids were the most important variable to organize the zooplanktonic community. These variables may act as an environmental filter for the development of the zooplanktonic community in the Veredas, as predicted by hypothesis (iv). Previous studies have already reported the importance of water temperature on zooplanktonic communities. In turn, the high temperature of water reduces the dissolved oxygen (Pinese et al. 2015), especially in shallow environments such as Veredas streams. Furthermore, Cerrado aquatic ecosystems are characterized by low electrical conductivity (Wantzen 2003, 2006; Gonçalves et al. Jr 2006), which may restrict the occurrence and/or establishment of certain species. Therefore, the Veredas streams with slightly higher conductivity values than expected may provide suitable conditions for more taxa to coexist, increasing local richness and abundance (Tundisi and Matsumura-Tundisi 2011).
Wetlands play an important role in water purification, nutrient cycling, and other ecosystem services (Convention on Wetlands 2021; Lynch et al. 2023). In particular, the Veredas are important for the maintenance of water resources once they are in the headwaters of the drainage basins. However, habitat fragmentation, land use conversion to agriculture, and siltation pose significant threats to the conservation of these Cerrado environments (Carvalho et al. 2009; De Marco et al. 2014), including the Veredas (Gonçalves et al. 2022). In recent decades, the rapid loss of wetland integrity has been reported worldwide (Hu et al. 2017). As underscored by a recent overview, “Biodiversity conservation is especially critical for freshwater biodiversity” (Lynch et al. 2023). Thus, the in-depth understanding gained from our study regarding the Veredas can lead to better conservation efforts for these small wetlands. Specifically, to maintain the water depth required for local zooplanktonic communities, it is crucial to prevent the loss of zooplankton communities and a whole resulting food web.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ab’Sáber AN (2003) Os domínios de natureza no Brasil: potencialidades paisagísticas. Ateliê editorial, São Paulo
Aggio CEG, Oliveira FR, Progênio M, Bello JR, Lansac-Toha FM, Velho LFM (2022) The zooplankton of tropical streams: is it determinism or stochasticity that drives the spatial and temporal patterns in community structure? Commun Ecol 23(2):219–229
Alarcão AG, Sena-Souza JP, Maciel BLO et al (2014) A relevância de múltiplos grupos zooplanctônicos para o monitoramento ambiental no cerrado: estudo da estação ecológica Águas. Emendadas Rev Espaço e Geogr 17:1–21
Allan JD (1976) Life history patterns in zooplankton. Am Nat 110:165–180
Alvares CA, Stape JL, Sentelhas PC et al (2013) Köppen’s climate classification map for Brazil. Meteorol Z 22:711–728. https://doi.org/10.1127/0941-2948/2013/0507
Bijos NR, Ulysses Orlando Eugênio C, de Roure Bandeira Mello T et al (2017) Plant species composition, richness, and diversity in the palm swamps (Veredas) of Central Brazil. Flora 236–237:94–99. https://doi.org/10.1016/j.flora.2017.10.002
Bini LM, Silva LCF, Velho LFM et al (2008) Zooplankton assemblage concordance patterns in brazilian reservoirs. Hydrobiologia 598:247–255. https://doi.org/10.1007/s10750-007-9157-3
Borcard D, Gillet F, Legendre P (2018) Numerical ecology with R, 2nd edn. Springer, New York
Carvalho FMV, De Marco P, Ferreira LG (2009) The Cerrado into-pieces: habitat fragmentation as a function of landscape use in the savannas of central Brazil. Biol Conserv 142:1392–1403. https://doi.org/10.1016/j.biocon.2009.01.031
Convention on Wetlands (2021) Global wetland outlook: special edition 2021. https://www.global-wetland-outlook.ramsar.org. Accessed 26 Sept 2022
Da Cunha CN, Piedade MTF, Junk WJ (2015) Classificação e delineamento das áreas úmidas brasileiras e de seus macrohabitats. EdUFMT, Cuiabá
Davidson NC (2014) How much wetland has the world lost? long-term and recent trends in global wetland area. Mar Freshw Res 65:934. https://doi.org/10.1071/MF14173
Esteves F (1998) Fundamentos de limnologia, 2nd edn. Interciência, Rio de Janeiro
De Marco P, Nogueira DS, Correa CC et al (2014) Patterns in the organization of Cerrado pond biodiversity in Brazilian pasture landscapes. Hydrobiologia 723:87–101. https://doi.org/10.1007/s10750-013-1695-2
Declerck SAJ, Coronel JS, Legendre P, Brendonck L (2011) Scale dependency of processes structuring metacommunities of Cladocerans in temporary pools of High-Andes wetlands. Ecography (Cop) 34:296–305. https://doi.org/10.1111/j.1600-0587.2010.06462.x
Dussart BH, Defaye D (1995) Copepoda: introduction to the Copepoda. In: Dumont HJ (ed) Guides to the identification of the microinvertebrates of the continental waters of the world. SPB Academic Publishing, The Hague, p 277
Elmoor-Loureiro LMA (1997) Manual de identificação de cladóceros límnicos do Brasil. Universa, Brasília
Elmoor-Loureiro LMA (2014) Ephemeroporus quasimodo sp. nov. (Crustacea: Cladocera: Chydoridae), a new species from the Brazilian Cerrado. Zootaxa 3821:88. https://doi.org/10.11646/zootaxa.3821.1.6
Faquim RCP, Machado KB, Teresa FB et al (2021) Shortcuts for biomonitoring programs of stream ecosystems: evaluating the taxonomic, numeric, and cross-taxa congruence in phytoplankton, periphyton, zooplankton, and fish assemblages. PLoS ONE 16:e0258342. https://doi.org/10.1371/journal.pone.0258342
Fernández-Aláez C, Trigal C, García-Girón J, Fernández-Aláez M (2018) Zooplankton is more strongly controlled by nutrients than predation in a vegetation-free Mediterranean shallow lake: a mesocosm experiment. Inl Waters 8:474–487. https://doi.org/10.1080/20442041.2018.1474686
Fonseca BM, Mendonça-Galvão L, Sousa FDR et al (2018) Biodiversity in pristine wetlands of Central Brazil: a multi-taxonomic approach. Wetlands 38:145–156. https://doi.org/10.1007/s13157-017-0964-7
Fox J, Weisberg S (2019) An R companion to applied regression, 3rd edn. Sage, Thousand Oaks
Gomes LF, Barbosa JC, Oliveira Barbosa H et al (2020) Environmental and spatial influences on stream zooplankton communities of the Brazilian Cerrado. Commun Ecol 21:25–31. https://doi.org/10.1007/s42974-020-00008-5
Gonçalves JF Jr, Graça MAS, Callisto M (2006) Leaf-litter breakdown in 3 streams in temperate, Mediterranean, and tropical Cerrado climates. J North Am Benthol Soc 25:344–355. https://doi.org/10.1899/0887-3593(2006
Gonçalves RVS, Cardoso JCF, Oliveira PE et al (2022) The role of topography, climate, soil and the surrounding matrix in the distribution of Veredas wetlands in Central Brazil. Wetl Ecol Manag 30:1261–1279. https://doi.org/10.1007/s11273-022-09895-z
Gordon ND, McMahon TA, Finlayson BL et al (1997) Stream hydrology: an introduction for ecologists, 1st edn. Wiley, Hoboken
Goslee SC, Urban DL (2007) The ecodist package for dissimilarity-based analysis of ecological data. J Stat Softw 22:1–19. https://doi.org/10.18637/jss.v022.i07
Graham MH (2003) Confronting multicollinearity ecological multiple regression. Ecology 84:2809–2815. https://doi.org/10.1890/02-3114
Haynes KJ, Bjørnstad ON, Allstadt AJ, Liebhold AM (2013) Geographical variation in the spatial synchrony of a forest-defoliating insect: isolation of environmental and spatial drivers. Proc R Soc B Biol Sci 280:20122373. https://doi.org/10.1098/rspb.2012.2373
Horton RE (1945) Erosional development of streams and their drainage basins; hydrophysical approach to quantitative morphology. Geol Soc Am Bull 56:275–370
Hu S, Niu Z, Chen Y et al (2017) Global wetlands: potential distribution, wetland loss, and status. Sci Total Environ 586:319–327. https://doi.org/10.1016/j.scitotenv.2017.02.001
Hubbell SP (2001) The unified neutral theory of biodiversity and biogeography. Princeton University Press, Princeton
Hutchinson G (1957) Concluding remarks. Cold Spring Harb Symp Quant Biol 22:415–427
Junk WJ (2013) Current state of knowledge regarding South America wetlands and their future under global climate change. Aquat Sci 75:113–131. https://doi.org/10.1007/s00027-012-0253-8
Junk WJ, Brown M, Campbell IC et al (2006) The comparative biodiversity of seven globally important wetlands: a synthesis. Aquat Sci 68:400–414. https://doi.org/10.1007/s00027-006-0856-z
Junk WJ, Piedade MTF, Lourival R et al (2014) Brazilian wetlands: their definition, delineation, and classification for research, sustainable management, and protection. Aquat Conserv Mar Freshw Ecosyst 24:5–22. https://doi.org/10.1002/aqc.2386
Koste W (1978) Rotatoria: die Rädertiere Mitteleuropas, I tafelband and II tafelband. Gebrüder Borntraeger, Stuttgart
Kottek M, Grieser J, Beck C et al (2006) World map of the Köppen-Geiger climate classification updated. Meteorol Z 15:259–263. https://doi.org/10.1127/0941-2948/2006/0130
Legendre P, Legendre L (1998) Numerical ecology, 2nd edn. Elsevier, Amsterdam
Lewis WM (2008) Physical and chemical features of tropical flowing waters. In: Dudgeon D (ed) Tropical stream ecology, 5th edn. Elsevier, Londres, pp 1–21
Lichstein JW (2007) Multiple regression on distance matrices: a multivariate spatial analysis tool. Plant Ecol 188:117–131. https://doi.org/10.1007/s11258-006-9126-3
Lima SC, Silveira FP (1991) A preservação das veredas para manutenção do equilíbrio hidrológico dos cursos d’água. In: Encontro Nacional de Estudos Sobre o Meio Ambiente. pp 204–218
Lopes VG, Castelo Branco CW, Kozlowsky-Suzuki B et al (2018) Environmental distances are more important than geographic distances when predicting spatial synchrony of zooplankton populations in a tropical reservoir. Freshw Biol 63:1592–1601. https://doi.org/10.1111/fwb.13188
Lynch AJ, Cooke SJ, Arthington AH et al (2023) People need freshwater biodiversity. WIREs Water. https://doi.org/10.1002/wat2.1633
Matsumura-Tundisi T, Tundisi J, Souza-Soares F, Tundisi J (2015) Zooplankton community structure of the lower Xingu river (PA) related to the hydrological cycle. Braz J Biol 75:47–54. https://doi.org/10.1590/1519-6984.03814BM
Mitsch WJ, Bernal B, Hernandez ME (2015) Ecosystem services of wetlands. Int J Biodivers Sci Ecosyst Serv Manag 11:1–4. https://doi.org/10.1080/21513732.2015.1006250
Oksanen J, Simpson G, Blanchet F et al (2022) Vegan: community ecology package (R Package Version 2.6-2)
Oliveira GC, Araújo GM, Barbosa AAA (2009) Florística e zonação de espécies vegetais em Veredas no Triângulo Mineiro. Brasil Rodriguésia 60:1077–1085. https://doi.org/10.1590/2175-7860200960417
Padovesi-Fonseca C, Rezende R (2017) Factors that drive zooplankton diversity in Neo-Tropical savannah shallow lakes. Acta Limnol Bras 29:e15. https://doi.org/10.1590/s2179-975x1817
Padovesi-Fonseca C, Martins-Silva MJ, Puppin-Gonçalves CT (2015) Others Cerrado’s areas as a reference analysis for aquatic conservation in Brazil. Biodivers J 6:805–816
Padovesi-Fonseca C, Souza Rezende R, Costa DF, Martins-Silva MJ (2021) Spatial scales drive zooplankton diversity in savanna cerrado streams. Commun Ecol 22:249–259. https://doi.org/10.1007/s42974-021-00052-9
Paquette C, Gregory-Eaves I, Beisner BE (2022) Environmental drivers of taxonomic and functional variation in zooplankton diversity and composition in freshwater lakes across canadian continental watersheds. Limnol Oceanogr 67:1081–1097. https://doi.org/10.1002/lno.12058
Pedroso C, Lansac-Tôha F, Mantovano T, Lansac‐Tôha F (2021) Mechanistic approach and elements of metacommunity structure of the zooplankton of palm swamps in the neotropical region. Int Rev Hydrobiol 106:217–225. https://doi.org/10.1002/iroh.202102092
Picapedra PHDS, Fernandes C, Baumgartner G, Sanches PV (2022) Drivers of zooplankton spatial dynamics in a small neotropical river. Acta Limnol Bras 34:e13
Pinel-Alloul P (1995) Spatial heterogeneity as a multiscale characteristic of zooplankton community. Hydrobiologia 300–301:17–42. https://doi.org/10.1007/BF00024445
Pinese OP, Pinese JF, Del Claro K (2015) Structure and biodiversity of zooplankton communities in freshwater habitats of a Vereda Wetland Region, Minas Gerais, Brazil. Acta Limnol Bras 27:275–288. https://doi.org/10.1590/S2179-975X0415
R Core Team (2022) R: a language and environment for statistical computing
Ribeiro JF, Walter BMT (2008) As principais fitofisionomias do bioma Cerrado. Cerrado: ecologia e flora. Embrapa Informação Tecnológica Brasília, pp 151–212
Segers H (1995) Rotifera: the Lecanidae (Monogononta). In: Dumont H, Nogrady T (eds) Guides to the identification of the microinvertebrates of the continental waters of the world. SPB Academic Publishing, The Hague, p 226
Segers H (1996) The biogeography of littoral Lecane Rotifera. Hydrobiologia 323:169–197. https://doi.org/10.1007/BF00007845
Segers H (2007) Annotated checklist of the rotifers (Phylum Rotifera), with notes on nomenclature, taxonomy and distribution. Zootaxa 1564:1–104. https://doi.org/10.11646/zootaxa.1564.1.1
Silveira LJ, Barbosa RA, Spletozer AC, Dias HCT (2022) Soil characteristics in Veredas (Brazilian palm swamps) of Alto São Francisco, Minas Gerais, Brazil. In: Open science research. Editora Científica Digital 216–228
Sinev A, Elmoor-Loureiro LMA (2010) Three new species of chydorid cladocerans of subfamily Aloninae (Branchipoda: Anomopoda: Chydoridae) from Brazil. Zootaxa 2390:1–25. https://doi.org/10.11646/zootaxa.2390.1.1
Smirnov NN (1996) Cladocera: the Chydorinae and Sayciinae (Chydoridae) of the world. SPB Academic Publishing, Amsterdam
Sousa FDR, Elmoor-Loureiro LMA (2008) Cladóceros fitófilos (Crustacea, Branchiopoda) do Parque Nacional das Emas, estado de Goiás. Biota Neotrop 8:159–166. https://doi.org/10.1590/S1676-06032008000100019
Sousa F, Elmoor-Loureiro LM (2013) Cladocerans (Crustacea: Anomopoda and Ctenopoda) of the Sempre Vivas national park, Espinhaço range, Minas Gerais, Brazil. Check List 9:4–8
Sousa FDR, Elmoor-Loureiro LMA, Mendonça-Galvão L (2013) Cladocerans (Crustacea, Anomopoda and Ctenopoda) from Cerrado of Central Brazil: inventory of phytophilous community in natural wetlands. Biota Neotrop 13:222–229. https://doi.org/10.1590/S1676-06032013000300025
Sousa FDR, Elmoor-Loureiro LMA, Panarelli EA (2017) The amazing diversity of the genus Monospilus Sars, 1862 (Crustacea: Branchiopoda: Aloninae) in South America. Zootaxa 4242:467. https://doi.org/10.11646/zootaxa.4242.3.3
Sousa FDR, Elmoor-Loureiro LMA, Mendonça-Galvão L et al (2018) Cladoceran (Crustacea: Branchiopoda) biodiversity of protected areas in a Brazilian hotspot. Invertebr Zool 15:309–322. https://doi.org/10.15298/invertzool.15.3.09
Stephan LR, Castilho-Noll MS, Henry R (2017) Comparison among zooplankton communities in hydrologically different lentic ecosystems. Limnetica 36:99–112. https://doi.org/10.23818/limn.36.08
Strahler AN (1957) Quantitative analysis of watershed geomorphology. Trans Am Geophys Union 38:913. https://doi.org/10.1029/TR038i006p00913
Suárez-Morales E, Gutiérrez-Aguirre MA, Gómez S et al (2020) Class copepoda. In: Thorp and covich’s freshwater invertebrates. Academic Press, pp 663–796
Tundisi JG, Matsumura-Tundisi T (2011) Limnology. CRC Press, Leiden
Wallace RL, Snell TW, Ricci C, Thomas N (2006) Rotifera: biology, ecology and systematics. In: Dumont HJF (ed) Guides to the identification of the microinvertebrates of the continental waters of the world, 2nd edn. Kenobi Productions, Leiden, p 299
Wan Maznah WO, Intan S, Sharifah R, Lim CC (2018) Lentic and lotic assemblages of zooplankton in a tropical reservoir, and their association with water quality conditions. Int J Environ Sci Technol 15:533–542. https://doi.org/10.1007/s13762-017-1412-1
Wantzen KM (2003) Cerrado streams-characteristics of a threatened freshwater ecosystem type on the tertiary shields of Central South America. Amazoniana 17:481–502
Wantzen KM (2006) Physical pollution: effects of gully erosion on benthic invertebrates in a tropical clear-water stream. Aquat Conserv Mar Freshw Ecosyst 16:733–749. https://doi.org/10.1002/aqc.813
Wantzen KM, Junk WJ (2000) The importance of stream-wetland-systems for biodiversity: a tropical perspective. Biodivers Wetl Assess Funct Conserv 1:11–34
Zapala MA, Schork NJ (2006) Multivariate regression analysis of distance matrices for testing associations between gene expression patterns and related variables. Proc Natl Acad Sci 103:19430–19435. https://doi.org/10.1073/pnas.0609333103
Acknowledgements
We are grateful to the UNEMAT Graduate Program in Ecology and Conservation (UNEMAT/PPGEC) and Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT; “Projeto Veredas” 227925/2015) for financial support. We are also grateful to colleagues at the Laboratório de Ecologia e Conservação de Ecossistemas Aquáticos (LECEA/UFMT) for help in the field. We would like to thank all the landowners for permission to collect samples on their properties. R. G Strutz, L. B. Lima, and F. J. M. Oliveira were partially financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) Finance Code – 001; L. B. Lima was supported by Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT/CNPq Notice 001/2022, Process FAPEMAT-PRO.000073/2023),V.G. Lopes was supported by Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT; Edital DCR 003/2016, FAPEMAT.0582219/2017), and D. P. Lima-Junior was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Grants: 305923/2020-0). We also thank Sueide Vilela and Fernando Borges Vieira for assistance during collections.
Funding
R.G.S., L.B.L., F.J.M.O., and C.C., were partially financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) Finance Code – 001; L.B.L was supported by Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT/CNPq Notice 001/2022, Process FAPEMAT-PRO.000073/2023); V.G.L., was supported by Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT; Edital DCR 003/2016, FAPEMAT.0582219/2017); C.C., L.B.L., and D.P.L.J. were partially funded by Agência Nacional de Águas e Saneamento Básico – ANA (88881.178666/2018-01), and D.P.L.J. was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, 305923/2020-0).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by RGS, VGL, LBL, FJO, CC and DPL-J. The first draft of the manuscript was written by RGS and VGL and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lopes, V.G., Strutz, R.G., Lima, L.B. et al. Characterization and predictors of the zooplankton community in the Veredas wetlands in Brazilian savanna. Wetlands Ecol Manage 31, 733–743 (2023). https://doi.org/10.1007/s11273-023-09944-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-023-09944-1