Abstract
Microbial fuel cells (MFC) have been foreseen as a sustainable renewable energy resource to meet future energy demand. In the past, several studies have been executed in both benchtop and pilot scale to produce electrical energy from wastewater. The key role players in this technology that leads to the operation are microbes, mainly bacteria. The dominant among them is termed as “exoelectrogens” that have the capability to produce and transport electron by utilizing waste source. The current review focuses on such electrogenic bacteria’s involvement for enhanced power generation of MFC. The pathway of electron transfer in their cell along and its conduction to the extracellular environment of the MFC system are critically discussed. The interaction of the microbes in various MFC operational conditions, including the role of substrate and solid electron acceptors, i.e., anode, external resistance, temperature, and pH, was also discussed in depth along with biotechnological advancement and future research perspective.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
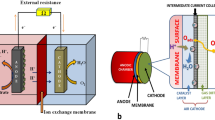
Microbial fuel cell (MFC) is a microbial-assisted process for the direct conversion of stored chemical energy in organics to electrical energy (Kang et al. 2015; Kang et al. 2017; Kim et al. 2017; Mukherjee and Saravanan 2019; Rabaey and Verstraete 2005). The demand for renewable power production pinned the interest in the MFC technology. Since the MFC functions in the presence of microbes (biocatalyst), it accomplishes wastewater treatment and organic waste deterioration too (Mukherjee et al. 2018). This technology is sub-classified into two types, namely single- and dual-chambered depending upon the reactor configuration (Mukherjee and Saravanan 2019). In general, an anoxic condition is maintained in the anode section of the dual-chambered, while the same is omnipresent in single-chambered (Logan 2009; Mukherjee and Saravanan 2019). The microbes present in the anode chamber catabolize the complex organic compounds into carbon dioxide and water releasing electrons and protons (Rabaey and Verstraete 2005). Thus, generated electrons are then transferred to the anode, which acts as an electron acceptor for the overall reaction to take place (Kang et al. 2015). The anode must possess the highest potential for the electron transfer to take place (Kang et al. 2015). Though a variety of mechanisms are preferred for this electron transfer, direct electron transfer (DET) and indirect electron transfer (IET) were considered as the two potential pathways (He et al. 2015; Mukherjee and Saravanan 2019). DET mechanism transfers the electrons via physical contact with the bacteria to the anode via pili or c-type cytochromes (Kumar et al. 2016). These types of microbes are also known as anodophiles or anode-respiring bacteria, and examples include Geobacter sulfurreducens and Shewanella oneidensis. On the other hand, the IET also known as mediated electron transfer (MET) utilizes mediators that act as electron conductor in the transfer process, and example include S. oneidensis (Kumar et al. 2016). The system may employ a single or mixed bacterial community. However, a common terminology of “exoelectrogens” is given to such electron-producing bacteria.
Although these exoelectrogens are a constant in the MFC system, their chemical and biological interactions have not been fully understood. Although the enhancement in power production in MFC was initiated by the crucial advancement in the biological side, most researchers focused their aim on improving the materials, cell architecture, electrolyte, etc. The review focuses on the exoelectrogen species, metabolism, interaction with various MFC components, techniques for isolation, identification and characterization, and finally the recent biotechnological advancements with futuristic
Microbiology of MFC
Varieties of microbes have been found in MFC, of which some are well known while others have recently been acknowledged. Methods like 16sRNA analysis for identifying bacterial pure cultures and denaturing gradient gel electrophoresis (DGGE) for identifying the microbial community composition and dynamics are commonly employed for bacterial identification (Borole et al. 2009). Scanning electron microscopy (SEM), transmission electron microscopy (TEM), and confocal laser scanning microscope (CLSM) are utilized to understand the morphological biofilm development on the electrode. The commonly found bacteria in MFC along with their electron transfer mechanism in the cell are discussed below.
Geobacter
Geobacter sp. is the Gram-negative bacteria belonging to the phylotype Proteobacteria that form thick biofilms in habitable condition (Shi et al. 2019). To a large extent, they are found in anaerobic soils and sediments actively involved in the reduction of insoluble Fe(III) oxides (Sun et al. 2019). The NCBI (National Center for Biotechnology Information) hosts 21 different species of Geobacter; however, G. sulfurreducens strain was much more emphasized. The reason is being one of the earliest discovered MFC exoelectrogen, having a rapid growth rate and easily cultivable in the laboratory (Coppi et al. 2001). However, the strain lacks in MFC performance as it is not effective in the reduction of Fe(III) oxides and resulting in lower power production as compared to other species of the genus (Rotaru et al. 2015; Sun et al. 2014; Zhou et al. 2014). It was also observed that different species of the genus show different extracellular electron transfer (EET) pathways, but all the genomes have multi-hem c-type chromosome and conductive pili (e-pili) (Butler et al. 2010; Holmes et al. 2016; Lovley 2017). For instance, G. sulfurreducens strain use extracellular c-type cytochrome PgcA for Fe(III) oxide reduction, and OmcS facilitates electron transfer to Fe(III) oxide in thin biofilm anodes, while OmcZ is involved for thick anode biofilms (Aklujkar et al. 2013; Holmes et al. 2006; Inoue et al. 2010; Mehta et al. 2005; Nevin et al. 2009; Zacharoff et al. 2017), where else G. metallireducens lacks OmcS cytochrome and uses e-pili and another c-type cytochrome (Gmet_2896) for the same (Shrestha et al. 2013; Tremblay et al. 2012). The outer membrane of multi-heme c-type cytochromes OmcP (GSU2913) and OmcO (GSU2912) are inessential for the reduction of Fe(III) oxide in G. sulfurreducens, but in the case of G. metallireducens, their homologs (Gmet_0557 and Gmet_0558) are mandatory for Fe(III) oxide respiration (Aklujkar et al. 2013; Smith et al. 2013). Some species like G. bemidjiensis, although having e-pili and OmcZ cytochrome, are incapable of electron generation (Nevin et al. 2005; Rotaru et al. 2015; Sun et al. 2019).
In a common mechanism for EET expressed by Geobacter, the extracellular substrate is used as a terminal electron acceptor. The pathways follow the conduction of electron from the quinone/quinol pool in the inner membrane to the periplasm and finally across the outer membrane to the extracellular environment (Lovley et al. 2011; Shi et al. 2007). Two inner membranes composed of c-type cytochromes and cytochrome c (ImcH) and a cytochrome protein with b- and c-type domains (CbcL) appear to be involved in the early steps of electron transfer to extracellular substrates in G. sulfurreducens (Levar et al. 2014; Levar et al. 2017; Zacharoff et al. 2016). A distinctive electron transfer (ET) component of the species is pilin monomers that enable the electrical conductivity in them. The subunits of e-pili were proteins built by aromatic amino acids in specific regions of its surface (Sun et al. 2019). This feature allows thick biofilm growth, and a study has also found that due to such thick attachment, the electricity production by G. sulfurreducens was 1047% more than that of Shewanella species (Engel et al. 2019). In contrast, the porin-cytochrome (Pcc) protein complex transfers the electron from the outer membrane to the electrolyte in the majority of Geobacter species (Liu et al. 2014; Shi et al. 2014). Diagram showing this is presented in Fig. 1a–e.
a ET mechanism of Geobacter sulfurreducens (Wang et al. 2019). b Comparative range of power density produced by various G. species showing highest for G. sulfurreducens (Rotaru et al. 2015). c Comparative study of Geobacter and Shewanella showing better current generation by G. species (Engel et al. 2019). d Confocal laser scanning microscope images of Geobacter on graphite anode (Engel et al. 2019) showing thick biofilm growth compared to e. of Shewanella showing thin biofilm growth (Engel et al. 2019).
Shewanella
It is a facultative anaerobe also belonging to the Gammaproteobacteria class found in both aquatic (fresh and marine) and soil environment (Fredrickson et al. 2008). The genus is widely popular owing to its respiratory versatility that enables the utilization of varieties of organic and inorganic substances, including anode (Kouzuma et al. 2015). However, the characteristic of a lack of thick biofilm formation reduces its versatility (Sun et al. 2019). The most widely used among the family is S. oneidensis as they can respire using dimethyl sulfoxide (DMSO), fumarate, nitrate, nitrite, N-oxide, and anthraquinone-2,6-disulfonate, oxygen, thiosulfate, trimethylamine sulfur, as well as both solid and soluble metals including chromium, cobalt, iron, manganese, technetium, uranium, and vanadium as electron acceptors (Fredrickson et al. 2008).
The major pathway followed by them for electron transfer is known as Mtr pathway consisting of mainly 5 protein components (CymA, MtrA, MtrB, MtrC, and OmcA) (Kouzuma et al. 2015). The Mtr protein is capable of ease converting flavin into soluble electron that shuttles for ET from an inner membrane cytochrome (CymA) to the MtrCAB porin-multi-heme c-type cytochrome complex (Shi et al. 2016; Shi et al. 2019). The EET is dominated by their outer membrane extensions with multi-heme c-type cytochromes (c-Cyts) via multistep hopping mechanism (El-Naggar et al. 2008; El-Naggar et al. 2010; Gorby et al. 2006; Gorby et al. 2008; Pirbadian and El-Naggar 2012; Pirbadian et al. 2014; Subramanian et al. 2018; Xu et al. 2018). On the bacterial surface, MtrC and probably OmcA transfer electron to the electrode surface either directly or indirectly via electron shuttle flavins (Kouzuma et al. 2015). The ET can also occur from the electrode surface to quinone in the cytoplasmic membrane through the same pathway. Furthermore, the electron transfer reactions to anodes are dominated by MtrC, and solid surface attachment is promoted by OmcA (Coursolle et al. 2010; Mitchell et al. 2012).
Studies have also suggested that both DET and MET are adopted by the species of the family. The DET involves the direct electron transferring from c-Cyts to the solid electron acceptors (anodes) (Kouzuma et al. 2015). In the case of MET, the electrons are transferred via electron-shuttle compounds, such as flavins. It has been reported that purified OmcA and MtrC proteins in MR-1 conduct the electrons to crystalline Fe(III) oxides and graphite anodes in the DET process. In the case of MET, MR-1 can reduce Fe(III) oxides located away from the cells without direct contact. And the species also secrete riboflavin and flavin mononucleotide (FMN), which function as electron shuttles. However, regardless of the pathway followed, soluble flavin secretion is inevitable in the ET by the family (Kouzuma et al. 2015; Roy et al. 2012). The schematics of ET and power performance of S. oneidensis strains are shown in Fig. 2.
a ET mechanism of S. oneidensis (Shi et al. 2012). b Voltage output graph of S. oneidensis strain wild type in black and flavin modified strain of S. oneidensis in red (republished with permission from Yang et al. 2015) c CV graph of S. oneidensis strains (republished with permission from Yang et al. 2015). d Polarization curve S. oneidensis strains (republished with permission from Yang et al. 2015). e Power density curve of S. oneidensis strains (republished with permission from Yang et al. 2015).
Escherichia coli
Again it is a facultative anaerobe of class Gamaproteobacteria mainly found in the lower intestine of warm-blooded organisms. The capability of the E. coli to utilize a number of organic compounds as a substrate for electron production makes it a viable MFC biocatalyst (Ojima et al. 2020; Qiao et al. 2008; Zhang et al. 2008). However, E. coli cannot directly transfer the electron produced in the cell, and electron mediators are necessary for their operation (Qiao et al. 2008). The absence of a direct ET pathway makes it unfamiliar as compared to Shewanella oneidensis or Geobacter sulfurreducens (Ojima et al. 2020). This has led to gene modification that enables direct ET pathway thereby improving electron transfer rate to the anode (Ojima et al. 2020). MFC involving E. coli produced hydroquinone-type endogenous compound. This compound was responsible for the direct electrochemical redox behavior of E. coli cells. The study proposed a membrane-related mechanism for the quinone excretion as followed by mature E. coli cells (Qiao et al. 2008). The figure showing the mechanism and growth of the E. coli cells were reproduced in Fig. 3a–b.
a EET mechanism of E. coli (Qiao et al. 2008). b AFM topography and section analysis of original and matured E. coli cells showing rougher and thicker biofilm growth evolved in E. coli cells (Qiao et al. 2008). c and d The current density performance of pure E.coli cells with co-culture growth showing better performance in co-cultures (republished with permission from Wang et al. 2015).
The E. coli cells are also commonly found in mixed cultures in wastewater and are often used in co-culture studies aiming at performance enhancement. Figure 3c and d establish that the co-culturing activity of E. coli with other exoelectrogens improves the power performance of the MFC.
Pseudomonas
Pseudomonas species are among the well-known exoelectrogenic bacteria that utilizes redox-active metabolites for electron production and transferring (Qiao et al. 2015). Pseudomonas aeruginosa, for example, produces pyocyanin (PYO) and 1-hydroxyphenazine (OHPHZ) as secondary metabolites for facilitating ET (Bellin et al. 2014; Chen et al. 2015). It has also been found in a study by Yong et al. that the overexpression of methyltransferase encoding gene phzM, i.e., one of the dominant genes responsible for PYO biosynthesis, can enhance PYO production. The accelerated PYO production by such genetic mutation significantly improved electrocatalytic activity (Yong et al. 2014a, 2014b). Similarly, Shen et al. (2014) reported that sophorolipid addition promotes bacterial membrane permeability, simultaneously enhancing PYO secretion, thereby improving overall MFC performance (Shen et al. 2014). The increment in phenazines excretion is also studied to produce a positive role in P. aeruginosa performance in MFCs. However, the details of these in the cellular mechanism of P. aeruginosa are not well defined. The lack of real-time detection of the metabolites during cell operation leads to non-clarity of the mechanism by Pseudomonas. Moreover, the Pseudomonas species are mainly applied in co-culture either with other exoelectrogens or in mixed cultures. The metabolites supplied by Pseudomonas improve the ET in the mixed cultures contributing to MFC performance. The SEM image of P. aeruginosa in the initial stage and after acclimatization in the anode is shown in Fig. 4 a and b. The ET mechanism is shown in Fig. 4c, and the power performance and electrochemical activity of P. aeruginosa in MFC are represented in Fig. 4d and e, respectively.
a SEM image of P. aeruginosa biofilm at 72 h (Read et al. 2010). b SEM image of P. aeruginosa biofilm at 144h showing attachment and clustering at the anode (Read et al. 2010). c ET mechanism of P. aeruginosa (republished with permission from Qiao et al. 2015). d Current density performance of P. aeruginosa in anaerobic and integrated aerobic-anaerobic MFC (republished with permission from Yong et al. 2017). e Cyclic voltammogram of P. aeruginosa in anaerobic and integrated aerobic-anaerobic MFC (republished with permission from Yong et al. 2017).
Rhodoferax
Rhodoferax is an anoxic genus of Betaproteobacteria belonging to the Comamonadaceae family. It utilizes Fe(III) as an electron acceptor for the oxidation of glucose to CO2, releasing electrons and falls under metal-reducing bacteria possessing DET (Chaudhuri and Lovley 2003; Schröder 2007). Rhodoferax has been reported to produce electricity from different types of sugars like glucose, fructose, xylose, and sucrose. A major advantage of Rhodoferax over other bacteria was its conversion of 80% glucose for electricity (Chaudhuri and Lovley 2003). Other microbes, including Geobacter and Shewanella species, are incapable of utilization of glucose as sole substrate and depend on fermentation by other microbes for the conversion of glucose to low organic acids and alcohols for usage (Chaudhuri and Lovley 2003).
Others
Some other microbes are also highly found in MFC cells that work as exoelectrogens, but the mechanism is not thoroughly studied. Exoelectrogen like Comamonas of Comamonadaceae family is one of them having the same family as Rhodoferax and similar mechanisms like Pseudomonas (Mukherjee and Saravanan 2020). Desulfovibrio desulfuricans have also shown an electrogenic response in MFC using microbial nanowires as a DIET mechanism (Kumar et al. 2016). Klebsiella pneumoniae is another Gammaproteobacteria found in various MFC cells. Clostridium sps. of the phylum Firmicutes are also a common occurrence in many mixed cultures and active in bioelectricity generation (Chandrasekhar et al. 2020). Table 1 lists variously studied exoelectrogens with their advantages and limitations.
Interaction of exoelectrogens with MFC parameters
The activities of the exoelectrogens discussed above however is dominated by the MFC reactor parameters. The MFC system mainly comprises of anode, anolyte, membrane, cathode, and catholyte. The anode and anolyte in combination with microbes make the oxidation half-cell, and the cathode and catholyte makes the reduction half-cell, while both were separated by a membrane. These two half sections were connected by wire and external resistance to ensure the circuit for the flow of electrons from the anode to the cathode section. The exoelectrogens dominate the oxidation half-cell and hence are influenced by the anode and anolyte used in the MFC. The anolyte comprises of substrate used for microbial growth. Apart from the anode and substrate, reactor operating conditions like temperature, pH, external resistance used, or external forces like magnetic field also influences the exoelectrogenic bacteria growth and activity. The present section deals with the different reactor condition’s influence on exoelectrogenic activity.
Influence of substrate
The electrogenesis of complex organic matter in the electrolyte by the microbes is the mechanism that results in electron generation for the overall power production. The exoelectrogens can directly utilize organics like acetate as substrates for electron production whereas complex substrates like glucose and lactate may require fermentation before bacterial utilization. These substrate interactions also lead to substrate-specific anodic communities (Kiely et al. 2011a). A predominance of Geobacter is seen in acetate-fed cells, and it is also seen that acetic acid electron donor supports a high range of exoelectrogens. Also, acetate being one of the simplest organics supports a wide range of microbes and hence mostly preferred in MFCs operation (Jung and Regan 2007; Mukherjee and Saravanan 2020; Xing et al. 2009).
Lactate needs to be fermented first by the bacterial group before being utilized as a substrate. The fermentation of lactate produces acetate and propionate in a 1:2 molar ratio (Kiely et al. 2011a). Lactic acid has been used as a substrate for culturing by S. oneidensis in MFC operation (Logan et al. 2005). The propionate carbon source supports large communities of Gram-positive microbes, mainly Firmicutes (Chae et al. 2009). Butyrate-supplied MFCs favor Pseudomonas and Bacillus sp. (Freguia et al. 2010). Although the power production of butyrate is half of that using acetate, and its low degradability compared to that acetate or propionate makes it is unsuitable as an MFC substrate. The presence of acetate in low concentration in the end product of butyrate-fed MFCs suggests the possibility of fermentation of butyrate to acetate (Kiely et al. 2011a). The bacterial community dominant in the substrate was similar to that of propionate.
Ethanol has also been utilized as a substrate as it can be fermented to generate acetic acid. Syntrophic interaction is used for the conversion of ethanol to electricity. Ethanol-fed MFC study has shown the dominant presence of the Geobacter community. The Betaproteobacteria also dominates the ethanol-fed MFCs (Kiely et al. 2011b; Kim et al. 2007). On the other hand, formic acid is less efficient in MFCs performance as compared to acetic acid, ethanol, and lactic acid due to its interaction with microbes (Kiely et al. 2011b). The community profiling data had suggested three possible formic acid conversion for electricity generation by exoelectrogens. The first is the direct oxidation of formic acid by Desulfitobacterium hafniense DCB2 in the presence of mediators like humic acids or anthraquinone-2,6-disulfonate (Milliken and May 2007). The second involves syntrophic interactions by microbes mainly homoacetogens for acetate formation from formic acid (Ha et al. 2008). The third archetype requires microbe Paracoccus denitrificans that oxidizes formate to hydrogen. This microbe utilizes formate dehydrogenase for the oxidation of formate to produce carbon dioxide and hydrogen. Thus, generated hydrogen molecules are then converted into an electron by Geobacteraceae to deliver electricity (Bond and Lovley 2003).
Simple carbohydrate (glucose)-fed MFCs also rely on the syntrophic process for electricity production employing the fermentation process. The fermentation leads to the production of several by-products like acetate, propionate, and hydrogen (Kiely et al. 2011a). In contrast complex carbohydrates like cellulose requires step like hydrolysis, fermentation, and electrolysis its MFC usage. It has been reported that Enterobacter cloacae are able of producing electrons from cellulose substrate (Rezaei et al. 2009). Further, a study has also demonstrated that the co-culturing of fermentative and exoelectrogenic bacteria together for cellulose usage resulted in the generation of high power density. The by-products formed by cellulose fermentation include acetate, ethanol, and hydrogen (Ren et al. 2007).
The substrate concentration also influences ammonia inhibition in the MFC system (Tice and Kim 2014). It is reported that a highly concentrated wastewater used in MFC inoculation contained a high amount of ammonia which negatively affected the exoelectrogens growth. Under low substrate concentration in such ammonia concentrated wastewater, the exoelectrogens activity is limited, leading to deprived power production. On the other hand, under high substrate conditions, the exoelectrogens resist the ammonia influence on their metabolism resulting in improved and stable power production. The higher substrate conditions resulted in the continuous production of H+ ion by the oxidation of the substrate. The presence of excess H+ ion drastically declines the pH of the anolyte, and under such lower pH conditions, ammonia (NH3) exists as ammonium (NH4+) ion that is less toxic than NH3 (Tice and Kim 2014).
The simpler the substrate, the easier is its oxidation by exoelectrogens releasing electron and proton. Utilizing pure cultures reduces the start-up time and aids thick biofilm growth in such case (Ullah and Zeshan 2020). However, in case of complex substrate, much more steps are involved in its oxidation influencing the start-up time and exoelectrogenic metabolism (Zhao et al. 2017). Hence, in such case, a mixed or co-culture species are more beneficial for high power generation (Li et al. 2018). The substrate utilization by exoelectrogens are calculated using Monod’s equation shown below.
where j is the current density obtained, jmax is the maximum current density of the biofilm, S is the concentration of substrate present, and Ks,app is the apparent half-saturation substrate concentration in a biofilm (Torres et al. 2010).
Influence of temperature
The temperature range of 4 °C to 45 °C is reported to be active for the anodic biofilms (Jadhav and Ghangrekar 2009; Patil et al. 2010). Both the biofilm establishment and its performance depend on the initially available temperature of the reactor. Temperature variation study showed that 35 °C was an ideal temperature for MFC operation. In a study on the development of microbial growth on anode surface, it was estimated that biofilm formation time decreased with an increase in temperature, 35 °C for 3.5 days as compared to 15 °C for 40 days (Patil et al. 2010). The spike in temperature from 30 to 45 °C found that the isolates Proteus sp. (N6) and Candida parapsilosis (S10) demonstrated the highest power densities in the range between 35 and 40°C (Nwagu et al. 2019).
In a psychrophilic MFC system, the electrogenic biofilm grows between −20 and 25°C (Lu et al. 2019). A maximum power density was delivered at 25°C; however, maximized chemical oxygen demand (COD) removal was obtained at 10°C. Moreover, it was also concluded that the pre-treated inoculum at varied temperatures determined the microbial communities on the anode surface. The bacterial population was dominated by Geobacter community having a relative abundance of 17–70% varying with the varied pre-acclimated temperature along with the presence of Arcobacter, Dechloromonas, Janthinobacterium, Limnohabitans, and Sejongia. It has also been found the Geobacter species were also majorly present at the lowest temperature (4°C) with a varying population of Accumulibacter, Caldilinea, Candidatus Desulfobulbus, Methylibium, and Nitrospira (Lu et al. 2019).
In some studies, thermophilic MFC operating at a temperature higher than 40 °C has gained interest. This system operates with microbes like Calditerrivibrio nitroreducens, Thermincola ferriacetica, and Thermincola potens (Fu et al. 2013; Marshall and May 2009; Wrighton et al. 2011). These bacteria utilizes exogenous mediators for the electron transfer for bioelectricity production. A recent study has demonstrated MFC operation as high as 95 °C (Fu et al. 2015).
It has been demonstrated by researchers that for macro and mesoscale MFCs, the Geobacter domination exhibits maximum power at a temperature between 25 and 30°C. However, in the case of micro, the optimal temperature range increases and ranged between 49 and 53°C (Ren et al. 2017). This was reasoned due to the limited electron transfer from cytochrome c of the Geobacter species when scaled down to micro. It was clarified that with an increase in temperature the cytochrome c contributing to electron transfer also increases leading to high-temperature activation in miniaturized MFCs (Ren et al. 2017).
The temperature variation showed a direct relationship with electrode potential where with a decrease in temperature the cathode potential decreased leading to lower power generation (Gadkari et al. 2020). It also leads to changes in ohmic losses. Thus, the operating temperature thus not only influences the microbial species present in the system but also influences the conductivity of the anolyte, activation energy, and diffusion coefficients further affecting the charge transfer rate (Oliveira et al. 2013; Nouri and Najafpour 2017). Therefore it is concluded that the temperature is a crucial parameter in determining the exoelectrogenic metabolism and start-up time and losses associated with influencing power performance in the MFC system
Influence of pH
pH certainly affects the microbial kinetics and hence is one of the deciding factors for the type of microbes present in the anodic chamber (Nwagu et al. 2019; Cheng et al. 2011). The pH of the system mostly depends upon the substrate used; a highly fermentable substrate leads to the formation of acidic products (Ren et al. 2007). Hence buffer is added to maintain neutral pH in most MFC operation. It has also been found that Shewanella can operate at pH as low as 5 (Biffinger et al. 2008). It has also been found in research that the growth and metabolism of Geobacter sulfurreducens are negatively affected by a change in pH from neutral to acidic (Franks et al. 2009). A biodegradation experiment employing Klebsiella sp. at various pH ranges (5,7, and 10) in MFC found that the neutral pH leads to the highest biodegradation (Holkar et al. 2018), while in the pH variation from 6.0 to 9.5, pH 8.5 produced the highest power generation in the MFC. Bacteria like Clavispora lusitaniae, Candida parapsilosis, and Clavispora lusitaniae dominated the chamber suggesting that alkaline pH is more preferred for MFC operation (Nwagu et al. 2019).
In a recent study, an alkaliphilic electrogenic bacteria (Bacillus alkalogaya BW2 ) was identified and utilized for MFC operation. It is a new culture used in MFC operation at pH as high as 10. The culture was able to utilize both acetate and lactate as substrate and produced higher voltage at pH 10. This extreme pH operation leads to future opportunities for utilizing high pH industrial wastewater as substrate (Dhundale et al. 2020).
In general it can be concluded that an acidic pH negatively impacts the voltage efficiency due to the negative impact on the exoelectrogens metabolism. Studies showed that under low pH conditions, the oxidation of substrate is incomplete releasing lower electron species and leads to concentration over potential (Ou et al. 2017). For instance a lower pH between 5 and 7 can be sustained by exoelectrogens, but power production was drastically affected (Babauta et al. 2011; Ou et al. 2017). Below this pH, concentration losses can occur in the anode chamber. It is also proved that pH influences the proton transport from anode to cathode. A low pH leading to increased acidity also decreases the resistance of the proton exchange membrane. The substrate oxidation is higher in neutral pH conditions indicating neutral pH is the best favored by exoelectrogens (Ou et al. 2017).
Influence of external resistance
It is established that the maximum performance of an MFC can be achieved only when the external resistance equates to its internal resistance. It also affects the anodic biofilm formation. A study on the effect of external resistance on anodic community found that lower external resistance of 100 Ω produced simpler DGGE patterns, whereas the band patterns were considerable in case of higher resistance (Katuri et al. 2012). The biofilm produced by S. oneidensis MR-1 was ca. 50 μm thick in case of high resistance (1MΩ) applied anode and was only ca. 5 μm in case of the anode with 100 Ω resistance (Mclean et al. 2010). In another study, external resistance of 20, 200, 470, and 1000 Ω was optimized for 10days, and its influence on exoelectrogens and methanogens distribution was seen (Cai et al. 2018). It was determined that the lower external resistance leads to enhanced dominance of exoelectrogens and limits methanogens growth (Cai et al. 2018).
Zhang et al. showed the influences of the external resistance (10, 50, 250, and 1000 Ω) on the start-up of MFC and biofilm growth. Their reports substantiated that the lower resistance, the start-up time is higher (3days), and under higher external resistance, the start-up time is lower (0.6 days). Their study also confirmed that it would be difficult to start-up an MFC at very low resistance (10Ω) due to unstable anodic potential. Hence, they suggested to start-up the MFC at higher external resistance and then gradually reduce the resistance to obtain higher power performance. It was also revealed that at decreased external resistance, better energy output with thick biofilm formation can be achieved (Zhang et al. 2017).
Study by Cai and co-workers showed that the lower resistance was beneficial for a thick exoelectrogenic biofilm formation (Cai et al. 2018). A number of external resistance were used (20, 200, 470, and 1000 Ω), and its effect on microbial composition and metabolism was studied. It was seen that at higher external resistance, the exoelectrogens bacterial population decreased and the methanogens population increased in the reactor. However, at lower resistance, the exoelectrogens were dominant and leading to more power production (Cai et al. 2018). Their study also discuss on the anode potential where notable such potential was observed at lower external resistances. Such change in anode potential can also change the microbial community present (Cai et al. 2018). Anode potential being an electromotive driving force leads to the electron flow from a bacterial cell to the anode and further to cathode from anode. Lower the external resistance used, the higher is the current generation and substrate oxidation thus enhancing chemical oxygen demand removal rate. (Cai et al. 2018).
Influence of anode material
The pore structure, surface morphology, and properties like roughness and hydrophilicity are the main features affecting microbial acclimatization and stability in the MFC (Mukherjee and Saravanan 2019; Mukherjee and Saravanan 2020). The positively charged anode materials are generally more preferred by exoelectrogens (Kumar et al. 2016). Thus carbon-based anode is highly favorable as an anode in MFC (Mukherjee and Saravanan 2019). In a study, inoculated with Shewanella putrefaciens, the performance of an MFC was determined by the anode potential. The microbial growth and power performance increased with an incement in the positive potential of the system (Carmona Martinez et al. 2013), where else the surface functionalization of graphite anode materials with aryl-mannoside layers (provides high hydrophilicity) was also found to result in the accelerated start-up of MFC (Iannaci et al. 2020). Du et al. demonstrated that polydopamine utilization enhanced biofilm growth due to its super hydrophilic nature (Du et al. 2017). Further, their study substantiated that the inclusion of super hydrophilicity enhanced the growth of exoelectrogenic Proteobacteria and Firmicutes phyla leading to the superior performance of MFC (Du et al. 2017). It has also been suggested that the start-up time of MFCs can be minimized by the inclusion of such polymeric substances in the anode. Reports also showed that Geobacter colonization was favored by using polyaniline-modified graphene anode (Lin et al. 2019). The polymeric anode improves OmcZ expression level favoring advanced microbial colonization in the anode. The octaheme c-type cytochromes, OmcB, and OmcZ presence in number provided the pathway for electron transfer to the anodes (Lin et al. 2019).
It has also been studied that the exoelectrogens can convert non-conductive graphene to conductive form (Yoshida et al. 2016). The oxidized form of graphene, i.e., graphene oxide (GO), is found to enhance electron transfer to the reaction chamber in MFCs. The non-conductive GO can be made conductive by the microbial reduction of the material. The reduced form is simply stated as reduced GO (rGO), as the chemical identity is not detailed. It was revealed that the GO provided selective growth of the exoelectrogens on its surface. GO-respiring bacteria (GORBs) were hence obtained from the environment for the purpose. A composite with rGO and GORBs formed a conductive hydrogel showing preferable Geobacteraceae growth having 51–68% relative abundance. Secondly, Azospira, well-known acetate oxidizers, comprised 28–42% of the anodic species. Shewanella and E. coli along with mixed cultures were also capable of GO reduction (Akhavan and Ghaderi 2012; Salas et al. 2010). The redox protein and biomolecules like vitamin C of the ET were critically involved in the reduction (Fernandez-Merino et al. 2010). This insight shows that GO may probably serve as an electron acceptor from the microbial cell by the exoelectrogens favoring their acclimatization. It has also been identified that GO has antibacterial or bactericidal properties limiting their role in MFCs anode. Thus, further information on GO electrode is required for the possible mechanism followed by microbes. However, GO being more economic and hydrophilic provides better properties as MFC anode compared to graphene providing better bacterial attachment on its surface. The self-aggregation of GO to hydrogel on reduction in solution favors its stability and reusability as a terminal electron acceptor for exoelectrogens (Yoshida et al. 2016).
Recent trends focused on utilizing 3D anode material with high surface area and high surface to volume ratio for enhanced exoelectrogenic colonization and ease anolyte transfer in the anode for bacterial substrate (Mukherjee and Saravanan 2020). These 3D anodes are favored by anodophilic bacteria for colonization increasing exoelectrogens concentration in anode.
Influence of magnetic field
In general, magnets are a good conductor and are applied for various fields marking its entry into MFC operation as well (Li et al. 2018a; Zhou et al. 2019). The researches in MFCs have established that magnetic fields utilized in the system can promote power generation owing to oxidative stress and magneto-hydrodynamic effects produced by it (Tong et al. 2015). It has also been found that the magnetic fields affect biofilm growth and biodegradability (Łebkowska et al. 2011; Wang et al. 2012). A weaker magnetic field is capable of increasing microbial growth by more than 40% (Yavuz and Çelebi 2000). In some instances, it was shown that bacterial diversity has declined under magnetic exposed conditions (Liu et al. 2008). It has been evident through researches that the cytochrome c-mediated bioelectrochemical transformations are also facilitated by its application (Katz et al. 2004; Katz et al. 2005). This in turn facilitates enhanced performance of the biofuel cells. A pulse electromagnetic field enhanced ET and favored Geobacter establishment on the anode surface resulting in higher efficiency (Zhou et al. 2017). The intensity of the magnetic field decides the stimulation and inhibition of biofilm growth (Zhou et al. 2019). However, an appropriate or optimum range of intensity for the enrichment of exoelectrogens has not yet been clarified, providing a new research stream for its application in MFCs. The direction of the magnetic field and its respective intensity are constantly changing in the case of a pulsed system, which leads to changes in aggregation and stability of the microbes (Zhou et al. 2019). However, in the case of a static, the direction and intensity remain the same leading to stable biofilm growth in MFCs. Several studies have been focused on a low stable magnetic field intensity on the anode side of MFCs showing positive results on power performance (Li et al. 2018a; Zhou et al. 2019). The enhancement is known to be due to stimulating enzyme activity of the exoelectrogenic community on the anode surface. In a recent study exploring magnetic fields effects on MFC performance and exoelectrogenic growth, magnets were used as anodes. Stable magnetic field intensities were achieved by utilizing different thermal-demagnetizing temperatures. The result obtained showed that both voltage and power production increased in the case of the magnetic anode and the diffusion resistance of the system is decreased due to the presence of a magnetic field (Zhou et al. 2019). The study also demonstrated a higher abundance of Geobacter growth in the magnetic MFC compared to the non-magnetic one (Zhou et al. 2019). The effect of low intensity stable magnetic field on mixed culture on anode showed that a magnetic field of (105 and 150 mT ) on anodic biofilms significantly decreased the start-up time and hence enhanced the power generation of the single-chambered MFC (Li et al. 2018a). It also concluded that the application of the magnetic field enhanced the microbial conductivity likely due to the increment of the Geobacteraceae population (Li et al. 2018a). Table 2 presents the interaction of exoelectrogens for improved MFC performance.
Co-culturing and genetic modifications and new developments in exoelectrogens
The exoelectrogens can also form co-culture with other non-exoelectrogenic bacteria for better efficiency. For instance, G. sulfurreducens have been studied to form co-cultures with a number of other microbes for improving electron transport to the anode in the cell. Co-culture of G. sulfurreducens-E. coli showed improved power production compared to the pure culture of G. sulfurreducens (Qu et al. 2012). The improved performance of the co-culture is attributed to O2 reduction by E. coli in the cell and DET by the G. sulfurreducens to anodes (Qu et al. 2012). Similarly, co-culturing demonstrated a higher conversion of methane to electricity. A genetically modified methanogen Methanosarcina acetivorans was used for the conversion of methane to acetate. This acetate was then further oxidized to produce electron by G. sulfurreducens for anode electron transfer. The transport of electron is also facilitated by Paracoccus denitrificans that produces redox molecules along with other microbes in the reactor (McAnulty et al. 2017).
In a co-culture study for ET mechanism by Geobacter sulfurreducens and Pseudomonas aeruginosa, it was revealed that the co-culture evolved to utilize the pathway between DIET and hydrogen IET (HIT) (Semenec et al. 2018). The co-culture was also capable of growing using formate and fumarate as substrate. It was also revealed by sequential window acquisition of all theoretical spectra (SWATH) and mass spectrometry studies that upregulation in HybA was seen in the co-cultures. This gene is critical for HIT pathway by Geobacter species. The study had also seen an increment in OmcS, PgcA, OmcC, and MacA genes required for Geobacter electron transfer metabolism (Semenec et al. 2018). The schematics showing the mechanism of conversion is shown in Fig. 5.
Proposed ET mechanism in the co-culture of G. sulfurreducens DL-1 strain (red cells) and P. aeruginosa PAO1, PA14, and PA14 phz strains (yellow cells) throughout evolution for electron production (republished with permission from Semenec et al. 2018).
In another co-culture study, S. oneidensis MR-1 along with Klebsiella pneumoniae was utilized for glycerol oxidization as substrate. A pure culture of S. oneidensis MR-1 is incapable for glycerol metabolization; however, the syntropic culture was capable of electron generation. The K. pneumoniae oxidized the glycerol to lactate that was then oxidized by S. oneidensis MR-1 for electron generation (Li et al. 2018b).
C. ljungdahlii was genetically modified by heterogeneous expression of the formate dehydrogenase gene. This gene modification in the microbe results in the regeneration of nicotinamide adenine dinucleotide (NADH). This higher NADH pool the facilitated ease electron transfer leading to higher power production of the system (Han et al. 2016).
Feng and coworkers genetically modified S. oneidensis species for utilizing xylose as a carbon source (Li et al. 2017). The utilization of this wood sugar as a substrate in MFC for electricity generation benefited sustainability as it is one of the primary ingredients of lignocellulosic hydrolysis. It is also the second most carbohydrate biomolecule present after glucose. However, as discussed above, xylose is not effectively utilized by many microbes and is limited by a slow consumption rate due to inefficient metabolism. The genetic modification within the study was enforced by collecting one in every of the wood sugar transporters from fungus intermedia (Candida intermedia )and true bacteria (Clostridium acetobutylicum) with one wood sugar metabolic pathways from the enzyme pathway from E. coli and also the enzyme pathway from Scheffersomyces stipites. It was found that the strain generated by modifying C. intermedia and S. stipites as xylose facilitator and xylose oxidoreductase respectively showed the highest result (Han et al. 2016).
A new species of Geobacter (Geobacter anodireducens) has been isolated for producing high power performance in excessive salt conditions (Sun et al. 2019). The G. anodireducens species has about 81 of the 87 c-type cytochromes homologous as compared to G. soli and G. sulfurreducens species. The species showed the presence of OmcS, OmcZ, and PgcA cytochromes responsible for ET. The presence of several membrane complexes and channels are responsible for proton and sodium ion transfer in their cell and thus protect them from osmotic shock. The presence of relatively additional polymer repair genes than most Geobacter species provides them protection in high salt and low pH scale conditions (Sun et al. 2019). Thus, power production was higher in high salt condition by G. anodireducens compared to metallireducens, sulfurreducens, and soli species of Geobacter.
Another novel exoelectrogen, namely Kluyvera georgiana MCC 3673, has been isolated from MFC culture grown in oilseed cake substrate. Genetic analysis by 16S rDNA sequencing disclosed that this organism is closely associated with Kluyvera georgiana. K. georgiana MCC 3673 is a facultative being, Gram-negative, non-spore-forming, non-motile, rod-shaped, measurement 0.3–0.6 μm × 1–2 μm being. Growth is fast in Luria-Bertani (LB) broth during a temperature variation between 25 and 37°C. Tiny yellowish smooth circular colonies with a diameter of 0.2–0.3mm were fashioned on agar within 12h of incubation. The power density produced by the culture was 379±8 mW/m2 (Thapa and Chandra 2019).
Future perspective and conclusion
This review dealt with the key role players, i.e., bacterial metabolism for electron transfer for the commonly occurring exoelectrogens of MFC. The factors affecting the exoelectrogenic electron production and transfer abilities are also discussed. The low power production and instability in the MFC system, in the long run, is a hindrance in its commercialization. The exoelectrogens are the crucial members for its interaction with various MFC parameters that influences the power production behavior. Hence the understanding of the EET by exoelectrogens in power production is of paramount importance. The knowledge of the proteins involved in EET electron production and transfer can lead to the development of new strategies for improving EET pathways of exoelectrogens. This provides scope in genetic engineering for manipulating existent exoelectrogens and discovery of new exoelectrogens having similar protein pathway for electron transfer. However, the MFC systems are still limited by the EET pathways of well-known exoelectrogens like G. sulfurreducens, S. oneidensis, E. coli only. A number of new exoelectrogens have been discovered along with genetic mutation of the widely known exoelectrogens in the recent studies. However real-time analysis of the changes in the cell activity during MFC operation is not yet thoroughly focused. The mechanism of electron transfer from the mentioned exoelectrogens in various growth media to obtain the stage-wise breakdown of substrate and number of electrons generated needs to be focused. The competition in case of mixed culture growth also presents future research opportunities. The isolation and transfer of EET genes present in Geobacter or Shewanella species to other non-exoelectrogens also presents biotechnological growth in future times. The MFC operation factors influencing exoelectrogenic growth and metabolism are also not studied completely, mostly only one factor influencing on the exoelectrogenic growth is discussed. However, the various factors like, temperature, pH, and substrate as a whole can affect other parameters providing future opportunities for establishing the interdependency of the factors on each other.
References
Akhavan O, Ghaderi E (2012) Escherichia coli bacteria reduce graphene oxide to bactericidal graphene in a self-limiting manner. Carbon 50:1853–1860
Aklujkar M, Krushkal J, DiBartolo G, Lapidus A, Land ML, Lovley DR (2009) The genome sequence of Geobacter metallireducens: features of metabolism, physiology and regulation common and dissimilar to Geobacter sulfurreducens. BMC Microbiol 9:109
Aklujkar M, Coppi M, Leang C et al (2013) Proteins involved in electron transfer to Fe (III) and Mn (IV) oxides by Geobacter sulfurreducens and Geobacter uraniireducens. Microbiology 159:515–535
Babauta JT, Nguyen HD, Beyenal H (2011) Redox and pH microenvironments within Shewanella oneidensis MR-1 biofilms reveal an electron transfer mechanism. Environ Sci Technol 45:6654–6660
Bellin DL, Sakhtah H, Rosenstein JK et al (2014) Integrated circuit-based electrochemical sensor for spatially resolved detection of redox-active metabolites in biofilms. Nat Commun 5:1–10
Biffinger JC, Pietron J, Bretschger O, Nadeau LJ, Johnson GR, Williams CC, Nealson KH, Ringeisen BR (2008) The influence of acidity on microbial fuel cells containing Shewanella oneidensis. Biosens Bioelectron 24:900–905
Bond DR, Lovley DR (2003) Electricity production by Geobacter sulfurreducens attached to electrodes. Appl Environ Microbiol 69:1548–1555
Borole AP, Hamilton CY, Vishnivetskaya TA, Leak D, Andras C, Morrell-Falvey J, Keller M, Davison B (2009) Integrating engineering design improvements with exoelectrogen enrichment process to increase power output from microbial fuel cells. J Power Sources 191:520–527
Butler JE, Young ND, Lovley DR (2010) Evolution of electron transfer out of the cell: comparative genomics of six Geobacter genomes. BMC Genomics 11:40
Cai WF, Geng JF, Pu KB, Ma Q, Jing DW, Wang YH, Chen QY, Liu H (2018) Investigation of a two-dimensional model on microbial fuel cell with different biofilm porosities and external resistances. Chem Eng J 333:572–582
Carmona Martinez AA, Harnisch F, Kuhlicke U et al (2013) Electron transfer and biofilm formation of Shewanella putrefaciens as function of anode potential. Bioelectrochemistry 93:23–29
Chae KJ, Choi MJ, Lee JW, Kim KY, Kim IS (2009) Effect of different substrates on the performance, bacterial diversity, and bacterial viability in microbial fuel cells. Bioresour Technol 100:3518–3525
Chandrasekhar K, Kumar G, Mohan SV, et al (2020) Microbial electro-remediation (MER) of hazardous waste in aid of sustainable energy generation and resource recovery. Environ Technol Innov p.100997.
Chaudhuri SK, Lovley DR (2003) Electricity generation by direct oxidation of glucose in mediatorless microbial fuel cells. Nat Biotechnol 21:1229–1232
Chen W, Liu XY, Qian C, Song XN, Li WW, Yu HQ (2015) An UV–vis spectroelectrochemical approach for rapid detection of phenazines and exploration of their redox characteristics. Biosens Bioelectron 64:25–29
Cheng S, Xing D, Logan BE (2011) Electricity generation of single chamber microbial fuel cells at low temperatures. Biosens Bioelectron 26:1913–1917
Coppi MV, Leang C, Sandler SJ, Lovley DR (2001) Development of a genetic system for Geobacter sulfurreducens. Appl Environ Microbiol 67:3180–3187
Coursolle D, Baron DB, Bond DR, Gralnick JA (2010) The Mtr respiratory pathway is essential for reducing flavins and electrodes in Shewanella oneidensis. J Bacteriol 192:467–474
Dhundale V, Hemke V, Desai D et al (2020) Evaluation of bioelectricity productivity using alkaliphilic Bacillus alkalogaya BW2 (1) as a possible exoelectrogens for improvement of microbial fuel cell performance. J Appl Biol Biotechnol 8:69–75
Du Q, An J, Li J et al (2017) Polydopamine as a new modification material to accelerate startup and promote anode performance in microbial fuel cells. J Power Sources 343:477–482
El-Naggar MY, Gorby YA, Xia W et al (2008) The molecular density of states in bacterial nanowires. Biophys J 95:L10–L12
El-Naggar MY, Wanger G, Leung KM et al (2010) Electrical transport along bacterial nanowires from Shewanella oneidensis MR-1. P Natl Acad Sci USA 107:18127–18131
Engel C, Schattenberg F, Dohnt K, Schröder U, Müller S, Krull R (2019) Long-term behavior of defined mixed cultures of Geobacter sulfurreducens and Shewanella oneidensis in bioelectrochemical systems. Front Bioeng Biotechnol 7:60
Fernandez-Merino MJ, Guardia L, Paredes JI et al (2010) Vitamin C is an ideal substitute for hydrazine in the reduction of graphene oxide suspensions. J Phys Chem C 114:6426–6432
Franks AE, Nevin KP, Jia H, Izallalen M, Woodard TL, Lovley DR (2009) Novel strategy for three-dimensional real-time imaging of microbial fuel cell communities: monitoring the inhibitory effects of proton accumulation within the anode biofilm. Energy Environ Sci 2:113–119
Fredrickson JK, Romine MF, Beliaev AS, Auchtung JM, Driscoll ME, Gardner TS, Nealson KH, Osterman AL, Pinchuk G, Reed JL, Rodionov DA, Rodrigues JLM, Saffarini DA, Serres MH, Spormann AM, Zhulin IB, Tiedje JM (2008) Towards environmental systems biology of Shewanella. Nat Rev Microbiol 6:592–603
Freguia S, Teh EH, Boon N, Leung KM, Keller J, Rabaey K (2010) Microbial fuel cells operating on mixed fatty acids. Bioresour Technol 101:1233–1238
Fu Q, Kobayashi H, Kawaguchi H, Wakayama T, Maeda H, Sato K (2013) A thermophilic Gram-negative nitrate-reducing bacterium, Calditerrivibrio nitroreducens, exhibiting electricity generation capability. Environ Sci Technol 47:12583–12590
Fu Q, Fukushima N, Maeda H, Sato K, Kobayashi H (2015) Bioelectrochemical analysis of a hyperthermophilic microbial fuel cell generating electricity at temperatures above 80 °C. Biosci Biotechnol Biochem 79:1200–1206
Gadkari S, Fontmorin JM, Yu E, Sadhukhan J (2020) Influence of temperature and other system parameters on microbial fuel cell performance: numerical and experimental investigation. Chem Eng J 388:124176
Gorby YA, Yanina S, McLean JS et al (2006) Electrically conductive bacterial nanowires produced by Shewanella oneidensis strain MR-1 and other microorganisms. Proc Natl Acad Sci U S A 103:11358–11363
Gorby Y, McLean J, Korenevsky A et al (2008) Redox-reactive membrane vesicles produced by Shewanella. Geobiology 6:232–241
Ha PT, Tae B, Chang IS (2008) Performance and bacterial consortium of microbial fuel cell fed with formate. Energ Fuels 22:164–168
Han S, Gao XY, Ying HJ, Zhou CC (2016) NADH gene manipulation for advancing bioelectricity in Clostridium ljungdahlii microbial fuel cells. Green Chem 18:2473–2478
He CS, Mu ZX, Yang HY, Wang YZ, Mu Y, Yu HQ (2015) Electron acceptors for energy generation in microbial fuel cells fed with wastewaters: a mini-review. Chemosphere 140:12–17
Holkar CR, Arora H, Halder D, Pinjari DV (2018) Biodegradation of reactive blue 19 with simultaneous electricity generation by the newly isolated electrogenic Klebsiella sp. C NCIM 5546 bacterium in a microbial fuel cell. Int Biodeterior Biodegradation 133:194–201
Holmes DE, Chaudhuri SK, Nevin KP, Mehta T, Methe BA, Liu A, Ward JE, Woodard TL, Webster J, Lovley DR (2006) Microarray and genetic analysis of electron transfer to electrodes in Geobacter sulfurreducens. Environ Microbiol 8:1805–1815
Holmes DE, Dang Y, Walker DJ et al (2016) The electrically conductive pili of Geobacter species are a recently evolved feature for extracellular electron transfer. Microb Genom 2:e000072
Iannaci A, Myles A, Flinois T, Behan JA, Barrière F, Scanlan EM, Colavita PE (2020) Tailored glycosylated anode surfaces: addressing the exoelectrogen bacterial community via functional layers for microbial fuel cell applications. Bioelectrochemistry 136:107621
Inoue K, Qian X, Morgado L, Kim BC, Mester T̈, Izallalen M, Salgueiro CA, Lovley DR (2010) Purification and characterization of OmcZ, an outer-surface, octaheme c-type cytochrome essential for optimal current production by Geobacter sulfurreducens. Appl Environ Microbiol 76:3999–4007
Jadhav GS, Ghangrekar MM (2009) Performance of microbial fuel cell subjected to variation in pH, temperature, external load and substrate concentration. Bioresour Technol 100:717–723
Jung S, Regan JM (2007) Comparison of anode bacterial communities and performance in microbial fuel cells with different electron donors. Appl Microbiol Biotechnol 77:393–402
Kang YL, Ibrahim S, Pichiah S (2015) Synergetic effect of conductive polymer poly (3, 4-ethylenedioxythiophene) with different structural configuration of anode for microbial fuel cell application. Bioresour Technol 189:364–369
Kang YL, Pichiah S, Ibrahim S (2017) Facile reconstruction of microbial fuel cell (MFC) anode with enhanced exoelectrogens selection for intensified electricity generation. Int J Hydrog Energy 42:1661–1671
Katuri KP, Enright AM, O'Flaherty V, Leech D (2012) Microbial analysis of anodic biofilm in a microbial fuel cell using slaughterhouse wastewater. Bioelectrochemistry 87:164–171
Katz E, Lioubashevski O, Willner I (2004) Magnetic field effects on cytochrome c-mediated bioelectrocatalytic transformations. J Am Chem Soc 126:11088–11092
Katz E, Lioubashevski O, Willner I (2005) Magnetic field effects on bioelectrocatalytic reactions of surface-confined enzyme systems: enhanced performance of biofuel cells. J Am Chem Soc 127:3979–3988
Kiely PD, Regan JM, Logan BE (2011a) The electric picnic: synergistic requirements for exoelectrogenic microbial communities. Curr Opin Biotechnol 22:378–385
Kiely PD, Rader G, Regan JM, Logan BE (2011b) Long-term cathode performance and the microbial communities that develop in microbial fuel cells fed different fermentation endproducts. Bioresour Technol 102:361–366
Kim JR, Jung SH, Regan JM et al (2007) Electricity generation and microbial community analysis of alcohol powered microbial fuel cells. Bioresour.Technol 98:2568–2577
Kim C, Lee CR, Song YE, Heo J, Choi SM, Lim DH, Cho J, Park C, Jang M, Kim JR (2017) Hexavalent chromium as a cathodic electron acceptor in a bipolar membrane microbial fuel cell with the simultaneous treatment of electroplating wastewater. Chem Eng J 328:703–707
Kouzuma A, Kasai T, Hirose A et al (2015) Catabolic and regulatory systems in Shewanella oneidensis MR-1 involved in electricity generation in microbial fuel cells. Front Microbiol 6:609
Kumar R, Singh L, Zularisam AW (2016) Exoelectrogens: recent advances in molecular drivers involved in extracellular electron transfer and strategies used to improve it for microbial fuel cell applications. Renew. Sust Energ Rev 56:1322–1336
Łebkowska M, Rutkowska-Narożniak A, Pajor E, Pochanke Z (2011) Effect of a static magnetic field on formaldehyde biodegradation in wastewater by activated sludge. Bioresour Technol 102:8777–8782
Levar CE, Chan CH, Mehta-Kolte MG et al (2014) An inner membrane cytochrome required only for reduction of high redox potential extracellular electron acceptors. MBio 5:e02034
Levar CE, Hoffman CL, Dunshee AJ, Toner BM, Bond DR (2017) Redox potential as a master variable controlling pathways of metal reduction by Geobacter sulfurreducens. ISME J 11:741–752
Li F, Li Y, Sun L, Li X, Yin C, An X, Chen X, Tian Y, Song H (2017) Engineering Shewanella oneidensis enables xylose-fed microbial fuel cell. Biotechnol Biofuels 10:196
Li F, Wang L, Liu C, Wu D, Song H (2018) Engineering exoelectrogens by synthetic biology strategies. Curr Opin Electrochem 10:37–45
Li C, Wang L, Liu H (2018a) Enhanced redox conductivity and enriched Geobacteraceae of exoelectrogenic biofilms in response to static magnetic field. Appl Microbiol Biotechnol 102:7611–7621
Li F, Yin C, Sun L, Li Y, Guo X, Song H (2018b) Synthetic Klebsiella pneumoniae-Shewanella oneidensis consortium enables glycerol-fed high-performance microbial fuel cells. Biotechnol J 13:1700491
Lin XQ, Li ZL, Liang B, Nan J, Wang AJ (2019) Identification of biofilm formation and exoelectrogenic population structure and function with graphene/polyanliline modified anode in microbial fuel cell. Chemosphere 219:358–364
Liu S, Yang F, Meng F, Chen H, Gong Z (2008) Enhanced anammox consortium activity for nitrogen removal: impacts of static magnetic field. J Biotechnol 138:96–102
Liu Y, Wang Z, Liu J, Levar C, Edwards MJ, Babauta JT, Kennedy DW, Shi Z, Beyenal H, Bond DR, Clarke TA, Butt JN, Richardson DJ, Rosso KM, Zachara JM, Fredrickson JK, Shi L (2014) A trans-outer membrane porin-cytochrome protein complex for extracellular electron transfer by Geobacter sulfurreducens PCA. Environ Microbiol Rep 6:776–785
Liu J, Guo T, Wang D, Ying H (2015) Clostridium beijerinckii mutant obtained atmospheric pressure glow discharge generates enhanced electricity in a microbial fuel cell. Biotechnol Lett 37:95–100
Liu X, Wang S, Xu A, Zhang L, Liu H, Ma LZ (2019) Biological synthesis of high-conductive pili in aerobic bacterium Pseudomonas aeruginosa. Appl Microbiol Biotechnol 103:1535–1544
Logan BE (2009) Exoelectrogenic bacteria that power microbial fuel cells. Nat Rev Microbiol 7:375–381
Logan BE, Murano C, Scott K, Gray ND, Head IM (2005) Electricity generation from cysteine in a microbial fuel cell. Water Res 39:942–952
Lovley DR (2017) Electrically conductive pili: biological function and potential applications in electronics. Curr Opin Electrochem 4:190–198
Lovley DR, Ueki T, Zhang T et al (2011) Geobacter: the microbe electric’s physiology, ecology, and practical applications, in: Poole, R.K. (Ed). Adv Microb Physiol 59:1–100
Lu S, Xie B, Liu B, Lu B, Xing D (2019) Neglected effects of inoculum preservation on the start-up of psychrophilic bioelectrochemical systems and shaping bacterial communities at low temperature. Front Microbiol 10:935
Marshall CW, May HD (2009) Electrochemical evidence of direct electrode reduction by a thermophilic Gram-positive bacterium, Thermincola ferriacetica. Energy Environ Sci 2:699–705
McAnulty MJ, Poosarla GV, Kim KY et al (2017) Electricity from methane by reversing methanogenesis. Nat Commun 8:1–8
Mclean SJ, Wanger G, Gorby YA et al (2010) Quantification of electron transfer rates to a solid phase electron acceptor through the stages of biofilm formation from single cells to multicellular communities. Environ Sci Technol 44:2721–2727
Mehta T, Coppi MV, Childers SE, Lovley DR (2005) Outer membrane c-type cytochromes required for Fe (III) and Mn (IV) oxide reduction in Geobacter sulfurreducens. Appl Environ Microbiol 71:8634–8641
Milliken CE, May HD (2007) Sustained generation of electricity by the spore-forming, Gram-positive, Desulfitobacterium hafniense strain DCB2. Appl Microbiol Biotechnol 73:1180–1189
Mitchell AC, Peterson L, Reardon CL et al (2012) Role of outer membrane c-type cytochromes MtrC and OmcA in Shewanella oneidensis MR-1 cell production, accumulation, and detachment during respiration on hematite. Geobiology 10:355–370
Mukherjee P, Saravanan P (2019) Perspective view on materialistic, mechanistic and operating challenges of microbial fuel cell on commercialisation and their way ahead. ChemistrySelect 4:1601–1612
Mukherjee P, Saravanan P (2020) Graphite nanopowder functionalized 3-D acrylamide polymeric anode for enhanced performance of microbial fuel cell. Int J Hydrog Energy 45:23411–23421
Mukherjee P, Mishra P, Saravanan P (2018) Microbial fuel cell: a prospective sustainable solution for energy and environmental crisis. Int J Biosen Bioelectron 4:191–193
Nevin KP, Holmes DE, Woodard TL, Hinlein ES, Ostendorf DW, Lovley DR (2005) Geobacter bemidjiensis sp. nov. and Geobacter psychrophilus sp. nov., two novel Fe (III)-reducing subsurface isolates. Int J Syst Evol Microbiol 55:1667–1674
Nevin KP, Kim BC, Glaven RH, Johnson JP, Woodard TL, Methé BA, DiDonato RJ, Covalla SF, Franks AE, Liu A, Lovley DR (2009) Anode biofilm transcriptomics reveals outer surface components essential for high density current production in Geobacter sulfurreducens fuel cells. PLoS One 4:e5628
Niessen J, Schröder U, Scholz F (2004) Exploiting complex carbohydrates for microbial electricity generation: a bacterial fuel cell operating on starch. Electrochem Commun 6:955–958
Nouri P, Najafpour DG (2017) Impacts of process parameters optimization on the performance of the annular single chamber microbial fuel cell in wastewater treatment. Eng Life Sci 17:545–551
Nwagu KE, Ekpo IA, Ekaluo BU, Ubi G, Elemba M, Uzoh V (2019) Optimization and molecular characterization of exoelectrogenic isolates for enhanced microbial fuel cell performance. Microbiol Biotechnol Lett 47:621–629
Ojima Y, Kawaguchi T, Fukui S, Kikuchi R, Terao K, Koma D, Ohmoto T, Azuma M (2020) Promoted performance of microbial fuel cells using Escherichia coli cells with multiple-knockout of central metabolism genes. Bioprocess Biosyst Eng 43:323–332
Oliveira VB, Simões M, Melo LF, Pinto AMFR (2013) Overview on the developments of microbial fuel cells. Biochem Eng J 73:53–64
Ou S, Kashima H, Aaron DS, Regan JM, Mench MM (2017) Full cell simulation and the evaluation of the buffer system on air-cathode microbial fuel cell. J Power Sources 347:159–169
Patil SA, Harnisch F, Kapadnis B, Schröder U (2010) Electroactive mixed culture biofilms in microbial bioelectrochemical systems: the role of temperature for biofilm formation and performance. Biosens Bioelectron 26:803–808
Pirbadian S, El-Naggar MY (2012) Multistep hopping and extracellular charge transfer in microbial redox chains. Phys Chem Chem Phys 14:13802–13808
Pirbadian S, Barchinger SE, Leung KM, Byun HS, Jangir Y, Bouhenni RA, Reed SB, Romine MF, Saffarini DA, Shi L, Gorby YA, Golbeck JH, el-Naggar MY (2014) Shewanella oneidensis MR-nanowires are outer membrane and periplasmic extensions of the extracellular electron transport components. Proc Natl Acad Sci U S A 111:12883–12888
Qiao Y, Li CM, Bao SJ et al (2008) Direct electrochemistry and electrocatalytic mechanism of evolved Escherichia coli cells in microbial fuel cells. ChemComm 11:1290–1292
Qiao Y, Qiao YJ, Zou L, Ma CX, Liu JH (2015) Real-time monitoring of phenazines excretion in Pseudomonas aeruginosa microbial fuel cell anode using cavity microelectrodes. Bioresour Technol 198:1–6
Qu Y, Feng Y, Wang X, Logan BE (2012) Use of a coculture to enable current production by geobacter sulfurreducens. Appl Environ Microbiol 78:3484–3487
Rabaey K, Verstraete W (2005) Microbial fuel cells: novel biotechnology for energy generation. Trends Biotechnol 23:291–298
Read ST, Dutta P, Bond PL, Keller J, Rabaey K (2010) Initial development and structure of biofilms on microbial fuel cell anodes. BMC Microbial 10:98
Ren Z, Ward TE, Regan JM (2007) Electricity production from cellulose in a microbial fuel cell using a defined binary culture. Environ Sci Technol 41:4781–4786
Ren H, Jiang C, Chae J (2017) Effect of temperature on a miniaturized microbial fuel cell (MFC). Micro and Nano Systems Letters 5:1–7
Rezaei F, Xing D, Wagner R, Regan JM, Richard TL, Logan BE (2009) Simultaneous cellulose degradation and electricity production by Enterobacter cloacae in a microbial fuel cell. Appl Environ Microbiol 75:3673–3678
Rotaru AE, Woodard TL, Nevin KP et al (2015) Link between capacity for current production and syntrophic growth in Geobacter species. Front Microbiol 6:744
Roy JN, Luckarift HR, Lau C, Falase A, Garcia KE, Ista LK, Chellamuthu P, Ramasamy RP, Gadhamshetty V, Wanger G, Gorby YA, Nealson KH, Bretschger O, Johnson GR, Atanassov P (2012) A study of the flavin response by Shewanella cultures in carbon-limited environments. RSC Adv 2:10020–10027
Salas EC, Sun Z, Luttge A et al (2010) Reduction of graphene oxide via bacterial respiration. ACS Nano 4:4852–4856
Schröder U (2007) Anodic electron transfer mechanisms in microbial fuel cells and their energy efficiency. Phys Chem Chem Phys 9:2619–2629
Semenec L, Laloo AE, Schulz BL, Vergara IA, Bond PL, Franks AE (2018) Deciphering the electric code of Geobacter sulfurreducens in cocultures with Pseudomonas aeruginosa via SWATH-MS proteomics. Bioelectrochemistry 119:150–160
Shen HB, Yong XY, Chen YL, Liao ZH, Si RW, Zhou J, Wang SY, Yong YC, OuYang PK, Zheng T (2014) Enhanced bioelectricity generation by improving pyocyanin production and membrane permeability through sophorolipid addition in Pseudomonas aeruginosa-inoculated microbial fuel cells. Bioresour Technol 167:490–494
Shi L, Squier TC, Zachara JM, Fredrickson JK (2007) Respiration of metal (hydr) oxides by Shewanella and Geobacter: a key role for multihaem c-type cytochromes. Mol Microbiol 65:12–20
Shi L, Rosso KM, Clarke TA et al (2012) Molecular underpinnings of Fe (III) oxide reduction by Shewanella oneidensis MR-1. Front Microbiol 3:50
Shi L, Fredrickson JK, Zachara JM (2014) Genomic analyses of bacterial porin-cytochrome gene clusters. Front Microbiol 5:657
Shi L, Dong H, Reguera G, Beyenal H, Lu A, Liu J, Yu HQ, Fredrickson JK (2016) Extracellular electron transfer mechanisms between microorganisms and minerals. Nat Rev Microbiol 14:651–662
Shi M, Jiang Y, Shi L (2019) Electromicrobiology and biotechnological applications of the exoelectrogens Geobacter and Shewanella spp. Sci China Technol Sci 62:1670–1678
Shrestha PM, Rotaru AE, Summers ZM, Shrestha M, Liu F, Lovley DR (2013) Transcriptomic and genetic analysis of direct interspecies electron transfer. Appl Environ Microbiol 79:2397–2404
Smith JA, Lovley DR, Tremblay PL (2013) Outer cell surface components essential for Fe (III) oxide reduction by Geobacter metallireducens. Appl Environ Microbiol 79:901–907
Subramanian P, Pirbadian S, El-Naggar MY et al (2018) Ultrastructure of Shewanella oneidensis MR-1 nanowires revealed by electron cryotomography. Proc Natl Acad Sci U S A 115:E3246–E3255
Sun D, Wang A, Cheng S, Yates M, Logan BE (2014) Geobacter anodireducens sp. nov., an exoelectrogenic microbe in bioelectrochemical systems. Int J Syst Evol Microbiol 64:3485–3491
Sun D, Wan X, Liu W, Xia X, Huang F, Wang A, Smith JA, Dang Y, Holmes DE (2019) Characterization of the genome from Geobacter anodireducens, a strain with enhanced current production in bioelectrochemical systems. RSC Adv 9:25890–25899
Thapa BS, Chandra TS (2019) Kluyvera georgiana MCC 3673: a novel electrogen enriched in microbial fuel cell fed with oilseed cake. Curr Microbiol 76:650–657
Tice RC, Kim Y (2014) Influence of substrate concentration and feed frequency on ammonia inhibition in microbial fuel cells. J. Power Sources 271:360–365
Tong ZH, Yu HQ, Li WW, Wang YK, Sun M, Liu XW, Sheng GP (2015) Application of a weak magnetic field to improve microbial fuel cell performance. Ecotoxicology 24:2175–2180
Torres CI, Marcus AK, Lee H-S, Parameswaran P, Krajmalnik-Brown R, Rittmann BE (2010) A kinetic perspective on extracellular electron transfer by anode-respiring bacteria. FEMS Microbiol Rev 34:3–17
Tremblay PL, Aklujkar M, Leang C, Nevin KP, Lovley D (2012) A genetic system for Geobacter metallireducens: role of the flagellin and pilin in the reduction of Fe (III) oxide. Environ Microbiol Rep 4:82–88
Ullah Z, Zeshan S (2020) Effect of substrate type and concentration on the performance of a double chamber microbial fuel cell. Water Sci Technol 81:1336–1344
Wang XH, Diao MH, Yang Y, Shi YJ, Gao MM, Wang SG (2012) Enhanced aerobic nitrifying granulation by static magnetic field. Bioresour Technol 110:105–110
Wang VB, Sivakumar K, Yang L, Zhang Q, Kjelleberg S, Loo SCJ, Cao B (2015) Metabolite-enabled mutualistic interaction between Shewanella oneidensis and Escherichia coli in a co-culture using an electrode as electron acceptor. Sci Rep 5:11222
Wang Q, Jones AAD, Gralnick JA et al (2019) Microfluidic dielectrophoresis illuminates the relationship between microbial cell envelope polarizability and electrochemical activity. Sci Adv 5:eaat5664
Wrighton KC, Thrash JC, Melnyk RA, Bigi JP, Byrne-Bailey KG, Remis JP, Schichnes D, Auer M, Chang CJ, Coates JD (2011) Evidence for direct electron transfer by a Gram-positive bacterium isolated from a microbial fuel cell. Appl Environ Microbiol 77:7633–7639
Xing D, Cheng S, Regan JM, Logan BE (2009) Change in microbial communities in acetate-and glucose-fed microbial fuel cells in the presence of light. Biosens Bioelectron 25:105–111
Xu S, Barrozo A, Tender LM, Krylov AI, el-Naggar MY (2018) Multiheme cytochrome mediated redox conduction through Shewanella oneidensis MR-1 Cells. J Am Chem Soc 140:10085–10089
Yang Y, Ding Y, Hu Y, Cao B, Rice SA, Kjelleberg S, Song H (2015) Enhancing bidirectional electron transfer of Shewanella oneidensis by a synthetic flavin pathway. ACS Synth Biol 4:815–823
Yavuz H, Çelebi SS (2000) Effects of magnetic field on activity of activated sludge in wastewater treatment. Enzym Microb Technol 26:22–27
Yong XY, Shi DY, Chen YL, Jiao F, Lin X, Zhou J, Wang SY, Yong YC, Sun YM, OuYang PK, Zheng T (2014a) Enhancement of bioelectricity generation by manipulation of the electron shuttles synthesis pathway in microbial fuel cells. Bioresour Technol 152:220–224
Yong XY, Feng J, Chen YL, Shi DY, Xu YS, Zhou J, Wang SY, Xu L, Yong YC, Sun YM, Shi CL, OuYang PK, Zheng T (2014b) Enhancement of bioelectricity generation by cofactor manipulation in microbial fuel cell. Biosens Bioelectron 56:19–25
Yong XY, Yan ZY, Shen HB, Zhou J, Wu XY, Zhang LJ, Zheng T, Jiang M, Wei P, Jia HH, Yong YC (2017) An integrated aerobic-anaerobic strategy for performance enhancement of Pseudomonas aeruginosa-inoculated microbial fuel cell. Bioresour Technol 241:1191–1196
Yoshida N, Miyata Y, Doi K, Goto Y, Nagao Y, Tero R, Hiraishi A (2016) Graphene oxide-dependent growth and self-aggregation into a hydrogel complex of exoelectrogenic bacteria. Sci Rep 6:21867
Zacharoff L, Chan CH, Bond DR (2016) Reduction of low potential electron acceptors requires the CbcL inner membrane cytochrome of Geobacter sulfurreducens. Bioelectrochemistry 107:7–13
Zacharoff LA, Morrone DJ, Bond DR (2017) Geobacter sulfurreducens extracellular multiheme cytochrome PgcA facilitates respiration to Fe (III) oxides but not electrodes. Front Microbiol 8:2481
Zhang T, Cui C, Chen S, Yang H, Shen P (2008) The direct electrocatalysis of Escherichia coli through electroactivated excretion in microbial fuel cell. Electrochem Commun 10:293–297
Zhang L, Li J, Zhu X, Ye D, Fu Q, Liao Q (2017) Startup performance and anodic biofilm distribution in continuous-flow microbial fuel cells with serpentine flow fields: effects of external resistance. Ind Eng Chem Res 56:3767–3774
Zhao YG, Zhang Y, She Z, Shi Y, Wang M, Gao M, Guo L (2017) Effect of substrate conversion on performance of microbial fuel cells and anodic microbial communities. Environ Eng Sci 34:666–674
Zhou S, Yang G, Lu Q, Wu M (2014) Geobactersoli sp. nov., a dissimilatory Fe (III)-reducing bacterium isolated from forest soil. Int J Syst Evol Microbiol 64:3786–3791
Zhou H, Liu B, Wang Q, Sun J, Xie G, Ren N, Ren ZJ, Xing D (2017) Pulse electromagnetic fields enhance extracellular electron transfer in magnetic bioelectrochemical systems. Biotechnol Biofuels 10:238–249
Zhou H, Mei X, Liu B, Xie G, Xing D (2019) Magnet anode enhances extracellular electron transfer and enrichment of exoelectrogenic bacteria in bioelectrochemical systems. Biotechnol Biofuels 12:133
Acknowledgements
The authors are grateful to Science and Engineering Research Board, Department of Science and Technology (DST-SERB) for the financial support received under IMPRINT with grant code IMP/2019/000286.
Availability of data and materials
Not applicable
Author information
Authors and Affiliations
Contributions
PS and PM conceived idea for the article. PM executed literature search and paper drafting. PS, GP, and MJ critically revised the work. All authors read and approved the final manuscript
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent of publication
Figs 1a–e and 2a are open access articles with copyright by citation and are cited in the manuscript as per instruction. Citation is provided for Figs 3a and b and 4a and b. Copyright has been taken for reuse of Figs 2b, c, d, and e; 3 c and d; 4 c, d, and e; and 5.
Consent to publish
All co-authors agreed to publish the manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Weiming Zhang
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mukherjee, P., Pichiah, S., Packirisamy, G. et al. Biocatalyst physiology and interplay: a protagonist of MFC operation. Environ Sci Pollut Res 28, 43217–43233 (2021). https://doi.org/10.1007/s11356-021-15015-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-15015-w