Abstract
Production of bioethanol from macroalgal feedstock involves pretreatment and hydrolysis of biomolecules for release of sugar followed by the fermentation of the sugar. The most commonly used pretreatment method for macroalgal biomass is dilute acid hydrolysis using mineral acids H2SO4 and HCl. The optimization of dilute acid hydrolysis of Ulva lactuca yielded 214.67 mg g−1 using 0.5 N H2SO4, 5% w/w substrate concentration at 121 °C for 45 min, and hydrolysis of Enteromorpha intestinalis yielded 239.94 mg g−1 of reducing sugar using 0.7 N H2SO4, 5% w/w substrate concentration at 120 °C for 45 min. Crude enzyme extracted from marine bacteria Vibrio parahaemolyticus and purified by the two-step purification produced 61.82% yield with 2.97-fold purification. Enzymatic hydrolysis of pretreated macroalgal biomass produced onefold higher reducing sugar than acid hydrolysis for Ulva lactuca (261.76 ± 0.9 mg g−1) and Enteromorpha intestinalis (289.89 ± 2.4 mg g−1).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The exploitation of finite fossil fuel resources has given rise to increased price fluctuations and elevated greenhouse gas emissions, contributing mainly to global warming. These drawbacks have escalated the need for alternative, renewable, sustainable, and economically viable energy resources such as carbohydrate-rich biomass to produce bioethanol. Bioethanol production is obtained from the carbohydrate fraction of the biomass, which is extracted and fermented [1,2,3,4]. Biomass conversion involves the separation of carbohydrate fraction to simple sugar through pretreatment methods, a vital step in biofuel production [5]. Several attempts have been made towards pretreatment of macroalgae or seaweeds [6,7,8,9,10,11], including chemical, physical, or biological or combination of these techniques. Pretreatment is carried out to enhance the surface area of the feedstock for the release of the constituent fermentable (reducing) sugars, which depends on the characteristics of chosen feedstock, catalysts, operation parameters, and strength [8, 12, 13]. Macroalgal biomasses are composed of a wide range of polysaccharides such as cellulose, ulvan, laminarin, Floridean starch, etc. These polysaccharides are broken down into monosaccharides which serve as raw materials for bioethanol production. Constituents of these monosaccharides vary in the macroalgal biomass, as summarized in Table 1.
Macroalgae with higher moisture (80–85%) content and devoid of lignin polymer is well suited for microbial conversion than combustion or thermochemical conversion [14, 33, 34]. Also, the absence of lignin avoids the necessity of employing harsh pretreatment processes required in lignocellulosic biomass [14, 33]. Pretreatment of biomass is carried out for (i) size reduction and (ii) alter or remove structural and compositional impediments before enzymatic hydrolysis. Pretreatments are required to be cost-effective with minimal inhibitor formation while releasing a higher quantum of sugar.
Physicochemical pretreatment involves liquid catalysts with higher process conditions to treat the feedstock. Pretreatment using chemicals such as acid, alkaline, and ammonia fiber expansion as well as soaking in aqueous ammonia and inorganic salts has been tried and is economical [8, 9, 35]. Ulva lactuca feedstock was subjected to four different pretreatments: ethanol organ solvent, alkaline, liquid hot water, and ionic liquid treatments. Organosolvent and liquid hot water treatment produced the highest sugar recovery of 808 mg g−1 dry weight (DW) and 629 mg g−1 DW, respectively [35]. In the hot water pretreatment, holes on algal feedstock surface (observed under scanning electron microscopy) indicated crystallinity (index of 97.5%) and cracks, which has enhanced enzyme digestibility of the feedstock. Gelidium amansii pretreated with 0.05–0.2 N Ca (OH)2 at 121 °C for 15 min resulted in gel formation. Hence, alkaline pretreatment is not opted for pre-processing of macroalgal feedstock, especially red and brown macroalgae containing hydrocolloids such as agar, carrageenan, and algin [36]. The most commonly used chemical pretreatment method employs mineral acids such as H2SO4 and HCl at milder concentrations of 0.3–0.9 N [9, 10]. Various reaction parameters such as reaction time, acid concentration, and substrate concentration are involved for efficient sugar release from the macroalgal feedstock. Pretreatment with dilute H2SO4 at different concentrations (~ 0.5–1%) and moderate temperature (~ 140–190 °C) [37] has been used widely for macroalgal cell wall depolymerization. Energy consumption in acid pretreatment is comparatively low compared to other pretreatments as it requires lower temperature as well as lesser incubation time (Table 2). Sulfuric acid reduces the production of inhibitors and improves the solubilization of seaweed polysaccharides [38]. The US National Renewable Energy Laboratory study reveals that the use of dilute acid (0.5–1%; 160–180 °C for 10 min) pretreatment aided in the release of different simple sugars (xylose, arabinose, galactose, glucose) [39, 40].
Reducing sugar (RS) released using H2SO4 from various macroalgal species such as Gracilaria verrucosa (430 mg g−1 RS, 1.5% H2SO4) [46]; Kappaphycus alvarezii (300 mg g−1 RS, 0.9 N H2SO4) [47]; Gracilaria verrucosa (7 g L−1 RS, 373 mM H2SO4) [48]; Laminaria japonica (29.09% RS, 0.06% H2SO4) [49]; Kappaphycus alvarezii (81.62 g L−1 RS, 1% v/v H2SO4) [15]; Gelidium amansii (33.7% RS, 3% H2SO4) [10]; Gracilaria verrucosa (7.47 g L−1 RS, 0.1 N H2SO4) [50]; and Kappaphycus alvarezii (30.5 g L−1 RS, 0.2 M H2SO4) has been reported [16]. However, drawback of dilute acid (higher concentration > 0.9 N) pretreatment is the generation of a higher concentration of 5-hydroxymethyl furfural (HMF) and levulinic acid (LA) (with the degradation of hexose sugars and furfurals from pentose sugar degradation), which acts as inhibitors for microorganisms during the fermentation process by reducing enzymatic and biological activities, breaking down the DNA and inhibiting protein and RNA synthesis [51]. In order to overcome this, enzyme saccharification or biological pretreatment using either cellulase enzyme (of commercial-grade) or enzymes isolated from fungi or bacteria has been tried.
The most common enzymes employed for seaweed hydrolysis in earlier studies are commercial enzymes such as Cellulase, Celluclast 1.5 L, Viscozyme L, Novozyme 188, Termamyl 120 L, β-glucosidase, Multifect, Meicelase, and Amyloglucosidase operated at pH 4.5–5.5 and temperature 35–55 °C; incubation time varies based on the algal feedstock [8, 15, 51,52,53,54,55,56,57,58,59]. The current study focuses on the evaluation of sugar release from Enteromorpha intestinalis and Ulva lactuca using dilute acid hydrolysis and enzyme (extracted from Vibrio parahaemolyticus) saccharification of the dilute acid pretreated biomass.
2 Materials and methods
2.1 Sample collection
E. intestinalis and U. lactuca seaweed samples were collected from Aghanashini estuary, Kumta taluk, Uttara Kannada district, Karnataka. Samples were thoroughly cleaned of epiphytes, then shade dried and powdered to obtain uniform size, and stored in polythene sachets for further analysis.
2.2 Dilute acid hydrolysis
Pretreatment of macroalgal feedstock was carried out at different acid concentrations, substrate concentrations, reaction time, and temperatures. Initially, acid hydrolysis was carried out using H2SO4 and HCl with concentrations ranging from 0.05, 0.1, 0.3, 0.5, 0.7, 0.9 to 1 N and keeping other parameters constant. The concentration of acid required for optimal sugar production was assessed, and further optimization was carried out. The reaction temperature for pretreatment was carried out with 2% w/v substrate at 30, 60, 90, and 120 °C for 45 min. Reaction time pretreatment with 2% w/v substrate concentration was carried out for 30, 60, 90,105, and 120 min. Pretreatment for algal biomass at different substrate concentrations of 1%, 2%, 3%, 5%, 7%, and 9% w/v was carried out at 120 °C for 45 min. After hydrolysis, residues were separated by filtration, and total sugar and reducing sugar were determined by phenol sulfuric acid [60] and dinitrosalicylic acid (DNS) [61] methods, respectively. DNS method is widely used to determine reducing sugar content in fields of food, bioprocess, medicine, etc. [62, 63]. Neutralization was carried out for acid hydrolysate using Na2CO3, NaOH, activated charcoal, and Ca (OH)2 [64, 65]. The significance of the above factors in influencing sugar release was determined using ANOVA. Thin-layer chromatography analysis of algal hydrolysate is obtained following optimized acid hydrolysis on silica gel plates using mobile phase butanol/ethanol/water (3:2:1 v/v/v). Later, these plates were dried at room temperature and dipped in AgNO3 solution for 1 min, and when dried, the plates were sprayed with ethanolic sodium hydroxide solution until dark brown spots appeared [66].
The efficiency of acid hydrolysis pretreatment (%) is calculated using Equation 1:
where:
- E p :
-
is the efficiency of acid hydrolysis pretreatment (%).
- ΔS:
-
is the disaccharide increase (mg) during acid hydrolysis pretreatment.
- TS:
-
is the total sugar (mg).
2.3 Response surface method
Response surface method (RSM) was used to evaluate the relationship between independent variables, reaction temperature (°C, X1), reaction time (min, X2), and substrate concentration (% w/v, X3), and dependent variable, reducing sugar (mg g−1, Y). The experimental data was analyzed and the probable relationship follows the second-order polynomial Eq. 2:
where Y is a response variable and X1, X2, and X3 are independent variables, β0 is the offset term; β1, β2, and β3 are the linear coefficients as per least squares method; and β11, β22, and β33 are the first, second, and third linear coefficients, respectively [48]. The Student’s t test was performed for the determination of the statistical significance of the regression coefficient [55, 67].
2.4 Crude enzyme production from Vibrio parahaemolyticus
Crude enzyme is extracted from Vibrio parahaemolyticus [68] using CMC as a sole source of carbon for screening cellulose-degrading bacteria. Endoglucanase was determined using the carboxymethyl cellulase method (CMCase) [69]; the endoglucanase enzyme cleaves the intermolecular β-1–4-glycosidic bonds present in cellulose. Cellulase was purified by centrifuging bacterial culture at 12,000 rpm for 15 min at 4 °C and supernatant was collected. Proteins were precipitated to 80% saturation with (NH4)2SO4 at 4 °C and pelletized through centrifugation. Pellet was dissolved in Tris–HCl (pH 7) and purified using ion exchange chromatography, wherein the sample was applied to Superdex 200 column equilibrated with Tris–HCl. Fractions were collected, and fractions with the highest enzyme activity were pooled and considered for other characterization. Enzymatic activity refers to the amount of enzyme that releases 1 µmol of reducing sugar per minute.
2.5 Enzyme characterization
The effect of pH on the enzyme activities was estimated using a buffer of different concentrations: 50 mM citrate buffer (pH 3–4), citrate phosphate buffer (pH 5–6), Tris–HCl (pH 7), and potassium phosphate buffer (pH 8). The effect of temperature on enzyme activity was determined by incubating the enzyme assay mixture of optimum pH at different temperatures ranging from 25 to 60 °C. The effect of salinity on enzyme assay mixture and salinity (NaCl) ranging from 4 to 20% was determined. Samples were incubated for 1 h with CMC as a sole source of carbon.
2.6 Efficient sugar release from macroalgal biomass through pretreatment: dilute acid hydrolysis and enzyme saccharification
Macroalgal feedstock Enteromorpha intestinalis (EI) and Ulva lactuca (UL) samples were subjected to dilute acid hydrolysis using an optimized acid concentration of 0.7 N and 0.5 N H2SO4 at optimal temperature and time of 121 °C and 45 min. Dilute acid pretreated macroalgal biomass EI and UL was subjected to enzymatic hydrolysis at 55 °C pH 6 for 36 h. The reducing sugar released from the above processes was recovered through a centrifuge and was estimated using DNS method [61].
2.7 SEM analysis
Macroalgal biomass surface morphology (untreated, acid-treated, and enzyme-treated biomass) was qualitatively analyzed using SEM (JEOL-IT 300). Macroalgal samples were placed on an aluminum specimen mount using conductive carbon tape. Sputter gold coating was performed to prevent charging. Samples were then examined in SEM under vacuum condition at accelerating voltage of 10 kV.
3 Results and discussion
Total sugar and reducing sugar (after dilute acid hydrolysis) were estimated for both E. intestinalis and U. lactuca. The optimization variables considered are acid concentration, reaction temperature, substrate concentration, and reaction time.
3.1 Effect of dilute acid concentration
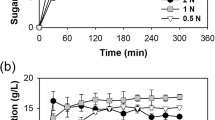
The effect of dilute acid concentration on E. intestinalis and U. lactuca was determined using acid concentrations of 0.01, 0.05, 0.3, 0.5, 0.7, and 1 N, for 1% substrate concentration at 121 °C for 1 h. Higher total sugar for E. intestinalis and U. lactuca biomass was obtained for pretreatment using H2SO4 (Fig. 1.). The sugar content gradually decreased with increase in acid concentrations as sugar decomposition varies based on concentrations and different acid catalytic activities [70]. Acid-catalyzed glucose decomposition is more dependent on the concentration of hydrogen ions at a particular temperature than on hydrogen ion sources [71].
Reducing sugar estimation was carried out for E. intestinalis and U. lactuca using HCl and H2SO4 (Fig. 2). Reducing sugar increased gradually with the increase in H2SO4 concentration for E. intestinalis, whereas for U. lactuca, reducing sugar increased up to 0.5 N H2SO4 and then decreased drastically. Acid hydrolysis efficiency was calculated for both the acid, and it was found that 0.7 N H2SO4 with the conversion efficiency of 80.18% was suitable for E. intestinalis and 0.5 N H2SO4 achieved a conversion efficiency of 60.07% for U. lactuca. These concentrations were kept constant for further optimization study. H2SO4 hydrolysis exhibited better reducing sugars compared to HCl and was considered for optimization. Hydrolysis using different acid concentrations released different concentrations of reducing sugars from the macroalgal biomass depending on their structure and biochemical composition, demonstrating that a customized approach is needed for hydrolysis.
3.2 Effect of reaction temperature on acid hydrolysis
Reducing sugar at different temperatures of 30, 60, 90, and 120 °C was recorded (Fig. 3). The highest reducing sugar of 549.45 mg g−1 and 528.46 mg g−1 was obtained for E. intestinalis and U. lactuca at 120 °C with 90.9% and 97.7% sugar conversion, respectively. Pretreatment of terrestrial biomass involves higher temperature (165–210 °C); this is attributed to their rigid structures [59], whereas macroalgal biomass requires milder temperatures. Studies involving red algae Kappaphycus alvarezii pretreated using 1% H2SO4 at 120 °C for 60 min obtained 81 g L−1 of reducing sugar [15], whereas earlier study [16] of similar pretreatment conditions at 130 °C obtained 22.4 g L−1 of reducing sugar. Reducing sugar of 65 mg g−1 was obtained after pretreatment of U. pinnatifida at 120 °C for 24 h [17]. Pretreatment of Saccharina japonica using 40 mM H2SO4 at 121 °C for 60 min yielded 20.6 g L−1 of reducing sugar [57]. An earlier study [59] of similar pretreatment conditions using 1 mM H2SO4 for 120 min achieved 34 g L−1 reducing sugar, indicating that the concentration of acid in hydrolysis plays a critical role in incubation temperature. Higher pretreatment conditions lead to degradation of sugars to hydroxymethyl furfural, which inhibits yeast growth by reducing the biological enzymatic activities, causing DNA and cell wall damage, inhibition of RNA, and protein synthesis [72].
3.3 Effect of reaction time on hydrolysis
To investigate the release of total sugar and reducing sugar, E. intestinalis and U. lactuca were hydrolyzed by 0.7 N and 0.5 N H2SO4, respectively, at 121 °C and different reaction times varying from 15, 30, 60, 90 to 120 min (Fig. 4). Maximum total sugar of 399 mg g−1was obtained at 105 min for E. intestinalis; maximum reducing sugar (121 mg g−1) was recorded at 45 min with a conversion efficiency of 42.1%. Maximum total and reducing sugars were produced for U. lactuca at 45 min and were seen decreasing with the increase in incubation time. G. verrucosa was subjected to pretreatment using 0.1 N H2SO4, and maximum total sugar (12.06 g L−1) and reducing sugar (6.99 g L−1) were obtained at 15 min incubation time. A shorter incubation time is required for red algae as a major fraction of sugar (i.e., Floridean starch) is composed in the cytoplasm of the red algae [73], which gets released easily. Hence, the longer incubation time was not considered as it leads to increase energy and cost, as well as accelerates the degradation of sugars to 5-HMF, levulinic acid, and formic acid [8, 10, 52, 65]. Therefore, 45 min was considered as the optimum incubation time for further studies.
3.4 Effect of substrate concentration on acid hydrolysis
The effect of varying substrate concentrations (1–9% w/v) on acid hydrolysis of E. intestinalis and U. lactuca was investigated at 121 °C for 45 min (Fig. 5). Total and reducing sugar concentration decreased with an increase in substrate concentrations. Similar results were observed for E. intestinalis, and the total reducing sugar decreased with an increase in solid to liquid ratio [8]. In a conventional simultaneous saccharification and fermentation process, substrate concentration of 10% (w/v) is considered optimal due to high viscosity and difficulty in handling the slurry [70]. Hydrolysis of Kappaphycus alvarezii required a 10% substrate concentration [16]. The highest sugar conversion rate was achieved at 2% (w/v) of G. verrucosa [50]. Higher efficiency of 85.43% and 62.97% was obtained for 5% (w/v) of E. intestinalis and U. lactuca substrate. Therefore, 5% (w/v) was considered as optimum substrate concentration.
3.5 Optimized sugar from E. intestinalis and U. lactuca
Acid hydrolysis of E. intestinalis and U. lactuca feedstock was carried out at an optimum acid concentration of 0.7 N and 0.5 N H2SO4, respectively at 121 °C for 45 min 5% (w/v) substrate concentration (Table 3). The highest reducing sugar of 206.82 ± 14.96 mg g−1was recorded from U. fasciata using sodium acetate (pH 4.8) buffer pretreatment process at 120 °C for 60 min. Undaria pinnatifida was pretreated at a higher concentration of acid, 5% H2SO4 at 120 °C for 24 h to obtain 65 mg glucose/g biomass [17]. U. pertusa was subjected to a high thermal liquefaction process (HTLP), with a process condition of 400 °C at 40 mPa and obtained 352 mg g−1of reducing sugar. HTLP pretreatment loosens the complex structure and increases the porosity of the cell membranes allowing the entry of the solvent for further degradation [74]. Reducing sugar concentration of 145 ± 2.1 mg g−1was obtained from pretreatment of Saccharina japonica (10% w/v) at 121 °C for 60 min using 40 mM H2SO4 [57]. Pretreatment of red seaweed Gracilaria sp. was carried out using 0.1 N H2SO4 at 121 °C for 1 h at 20% w/v biomass loading and obtained 277 mg g−1of reducing sugar [75]. Inhibitors from acid hydrolysate hydroxymethyl furfural (HMF) and levulinic acid (LA) were detoxified using activated charcoal [65], which removed 70.37% HMF and 38.8% LA; similarly, Na2CO3 detoxified the 56.1% from U. lactuca and 23.3% from E. intestinalis [64] indicating that hydrolysis using dilute acid concentration resulted in a lower concentration of inhibitors.
TLC analysis showed glucose and xylose in the both acid hydrolysate of E. intestinalis and U. lactuca (Fig. 6).
3.6 Assessing the optimal pretreatment conditions through RSM (Response Surface Method)
RSM involved assessing the optimal pretreatment conditions (Table 4) for maximum reducing sugar yield from E. intestinalis and U. lactuca. The possible combinations of independent variables were chosen through stepwise regression, and the probable relationship with the yield of sugar (Y) is expressed in Eqs. 3 for E. intestinalis and 4 for U. lactuca, respectively (p < 0.05). Response surface curves were generated using 14 data points of each variable as depicted in Fig. 7 for E. intestinalis and U. lactuca reducing sugar yield at different reaction temperatures, time, and substrate concentrations, which aided in arriving at the optimum level of each variable for maximum response. An increase in substrate concentrations led to a decline in reducing sugar release, which could be due to sorption loss.
Response surface plots of reducing sugar yield for E. intestinalis (1a–b) and U. lactuca (2a–b) after dilute acid pretreatment at different reaction temperature, time, and substrate concentrations. 1a and 2a Reducing sugar yield at substrate concentration = 5% w/v; 1b and 2b reducing sugar yield at temperature = 75 °C; 1c and 2c reducing sugar yield at reaction time = 75 min (p < 0.05)
The effect of reaction temperature and incubation time on hydrolysis of E. intestinalis and U. lactuca when substrate concentration was kept constant as shown in Fig. 7 and reducing sugar yield decreased with an increase in incubation temperature and incubation time. Higher reducing sugar yield was recorded at lower temperatures (30–60 °C) and incubation time (30–90 min). In order to obtain reducing sugar yield between 400 abd 600 mg/g, the optimum reaction temperature of 75 °C, reaction time 75 min, and substrate concentration of 5% w/v were recorded from the RSM 3D plot. However, in this study, dilute acid hydrolysis of algal biomass for efficient reducing sugar yield between 200 and 240 mg/g was achieved at temperature 121 °C and time 45 min at 5% w/v substrate concentration. Higher glucose yield for Sargassum spp. is achieved at optimized acid concentration of 3.75 and 4.5% (w/v) substrate concentration and optimum temperature 115 °C for 86–90 min [55]. It is seen that the pretreatment temperature and incubation time obtained in this study to treat E. intestinalis and U. lactuca were milder than the terrestrial biomass. The presence of cellulose, hemicellulose, and lignin imparts the rigidity to the terrestrial biomass. It hence requires a temperature between 165 and 210 °C at a high concentration of acids for a longer incubation time (4 weeks) [55, 57].
It is seen that the pretreatment temperature and incubation time obtained to treat E. intestinalis and U. lactuca were milder than the terrestrial biomass requiring temperature between 165 and 210 °C at a high concentration of acids for longer incubation time (4 weeks). A harsh pretreatment condition was required due to the rigidity of the biomass with cellulose, hemicellulose, and lignin [55, 59]. Estimated effects, standard errors (SE), Student’s t test, and significance value for the model representing reducing sugar yield from U. lactuca and E. intestinalis are represented in Table 5.
3.7 Enzyme purification and characterization
The purification of cellulase enzyme is summarized in Table 6, which is a two-step purification and includes ammonium sulfate precipitation and size exclusion chromatography. The purified enzyme exhibited 7.24 U mg−1 of specific activity, and 61.82% yield was obtained with 2.97-fold purification. Purification was further confirmed by observing a single protein band on SDS-PAGE (Fig. 8) with an estimated molecular mass of 29 kDa. Similarly, molecular mass was obtained for cellulase extracted from Salinivibrio sp. NTU-05 exhibiting 32.4 U mg−1 specific activity and 18.9% recovery with 29.5-fold purification [76]. Extraction of Endo-β-1, 4-glucanase Cel5A from Vibrio sp. exhibited a molecular mass of 50 kDa, indicating functional cellulase gene in Vibrio genus (Gao et al., 2010).
The enzyme exhibited the highest activity at pH 6, and the activity profile showed that the enzyme was active over a wide range of pH, retaining 90% of its activity (Fig. 9). Similar characteristic pH tolerance over a wide range has been studied earlier for Paenibacillus sp. pH 7 [77]; Marinobacter sp. MS1032 [78]; Vibrio sp. G21 pH 6.5–7.5 [79]; Bacillus sp. H1666 pH 3–9 [80]; Bacillus sp. [81]; Stachybotrys atra BP-A [82], Bacillus flexus pH 8–12 [83]; and Salinivibrio sp. pH 6.5–8.5 [76].
The highest activity of the enzyme was recorded at 50 °C with higher stability between 40 and 55 °C (Fig. 10). Enzyme activity decreased due to the fluidity of protein conformation with an increase in temperature above 55 °C. It was seen that around 60% of the activity remained at 60 °C. The optimum temperature for cellulase-producing bacteria was 40–60 °C [76,77,78, 80,81,82,83].
Enzyme activity declined with an increase in salt concentration; at NaCl concentration > 10%, the enzyme retained 5% of its activity after 24 h (Fig. 11). Enzyme activity was above 20% up to 10% NaCl concentration. It is seen that NaCl concentration induces the activity of endo-β-1, 4-glucanase Cel5A from Vibrio sp. G21 and EgI-AG from alkaliphilic Bacillus agaradhaerens [79, 84]. Enzyme displayed activity in a broad range of 0–10% NaCl concentration with optimum NaCl concentration of 3%. Enzyme activity was stable only up to 10% NaCl concentration despite the enzyme being extracted from marine bacteria V. parahaemolyticus.
3.8 Dilute acid pretreatment and enzymatic hydrolysis
Acid hydrolysis of E. intestinalis and U. lactuca biomass was carried out at an optimum acid concentration of 0.7 N and 0.5 N H2SO4, respectively, at a temperature of 121 °C for 45-min incubation time and 5% (w/v) substrate concentration (Table7).
In similar studies, the highest reducing sugar of 206.82 ± 14.96 mg g−1 was recorded from U. fasciata using sodium acetate (pH 4.8) buffer pretreatment at 120 °C for 60 min. Undaria pinnatifida was pretreated at a higher concentration of acid, 5% H2SO4 at 120 °C for 24 h to obtain 65 mg glucose g−1 biomass [17]. U. pertusa was subjected to a high thermal liquefaction process (HTLP), with a process condition of 400 °C at 40 mPa, and obtained 352 mg g−1 of reducing sugar. HTLP pretreatment loosens the complex structure and increases the porosity of the cell membranes allowing the entry of the solvent for further degradation [74]. Reducing sugar concentration of 145 ± 2.1 mg g−1was obtained from pretreatment of Saccharina japonica (10% w/v) at 121 °C for 60 min using 40 mM H2SO4 [57]. Pretreatment of red seaweed Gracilaria sp. using 0.1 N H2SO4 at 121 °C for 1 h at 20% w/v biomass loading yielded 277 mg g−1 of reducing sugar [75].
Enzyme hydrolysis is affected by various factors, such as temperature, pH, and concentration (enzyme/substrate). Increasing enzyme concentration will speed up the reaction, as long as there is substrate availability; however, if once all of the substrates are bound, the reaction will cease to speed up. On the other hand, increasing substrate concentration also increases the rate of reaction to a certain extent. But once all enzymes are bound, any increase in substrate will have no effect on the reaction rate due to saturation of available enzymes. Trivedi et al. (2015) isolated cellulase enzyme from Cladosporium sphaerospermum and subjected Ulva lactuca, green seaweed to enzymatic hydrolysis and obtained 112 mg/g of reducing sugar. However, in this study, the highest reducing sugar of 107.6 mg/g was obtained from U. lactuca, whereas 135.9 mg/g reduced sugar from E. intestinalis indicating enzyme ability to hydrolyze the macroalgal polysaccharide.
Acid pretreated macroalgal biomass (E. intestinalis and U. lactuca) was subjected to enzymatic hydrolysis using purified enzyme and incubated for 24 h and observed twofold increase in reducing sugar in both biomass and 1.2-fold increase from dilute acid pretreatment, compared to crude enzymatic hydrolysis. Enzymatic hydrolysis of U. lactuca using purified enzyme extracted from Bacillus sp. H1666 yielded 450 mg g−1 of reducing sugar, indicating the potential applicability of the enzyme for algal biomass saccharification [80]. Enzymes secreted from the cell are generally found along with other proteins, lipids, polysaccharides, and nucleic acids. The measurement of enzyme purity is defined as the relation of the activity of the enzyme to the total protein present (i.e., the specific activity). Enzyme purification is carried out in order to remove the contaminants and increase the specific activity [85]. In this study, purified enzyme yielded higher reducing sugar due to increased specific activity (Table 6).
3.9 Scanning electron microscopy
Scanning electron microscopic (SEM) analysis of macroalgal biomass revealed ultrastructural changes in the biomass during dilute acid pretreatment. Figures 12a and 12b depict the untreated surface of E. intestinalis and U. lactuca; raw or untreated biomass had continuous, even, and smooth surfaces, whereas biomass after dilute acid pretreatment had loosened the rugged surface, which increased the surface area, exposing more internal cellulose for enzymatic hydrolysis. The roughness of the seaweed surface after dilute acid hydrolysis makes it more liable for enzymatic hydrolysis. The presence of strong hydrogen bonding of cellulose and Van der Waal forces of glucose molecules imparts the crystalline structure to biomass [52, 86, 87]. Scanning electron microscope (SEM) images revealed cracks and holes on the pretreated algal surface. Gelidium amansii treated at 121 °C were observed under SEM; electron micrographs revealed fibers exposed in autoclaved samples allowing enzymes to easily degrade the cells [52].
a Scanning electron micrograph of E. intestinalis depicting ultrastructural changes in the feedstock — untreated sample compared with the acid and enzyme treated. b Ultrastructural changes evident in the scanning electron micrograph of U. lactuca macroalgal biomass — untreated, acid, and enzyme treated
4 Conclusions
The present study demonstrates the potential of E. intestinalis and U. lactuca as marine source for production of sugar for biofuel production. Feedstock E. intestinalis and U. lactuca were subjected to dilute acid pretreatment and yielded 239.94 ± 1.3 mg g−1 and 214.67 ± 0.9 mg g−1of reducing sugar. An enzyme extracted from marine bacteria Vibrio parahaemolyticus hydrolyzed the algal biomass efficiently, releasing onefold higher reducing sugar than dilute acid pretreatment. Enzymatic hydrolysis of pretreated macroalgal biomass produced onefold higher reducing sugar than the dilute acid pretreatment. Dilute acid pretreatment prior to enzymatic hydrolysis improves algal biomass saccharification and releases higher reducing sugar, increasing bioethanol yield. However, tailor-made approaches need to be employed depending on algal species, as some sugars are easily hydrolyzed by acid and few by a direct enzyme.
References
Haghighi Mood S, Hossein Golfeshan A, Tabatabaei M et al (2013) Lignocellulosic biomass to bioethanol, a comprehensive review with a focus on pretreatment. Renew Sustain Energy Rev 27:77–93. https://doi.org/10.1016/j.rser.2013.06.033
Yin Y, Wang J (2018) Pretreatment of macroalgal Laminaria japonica by combined microwave-acid method for biohydrogen production. Bioresour Technol 268:52–59. https://doi.org/10.1016/j.biortech.2018.07.126
Montingelli ME, Benyounis K, Stokes J, Olabi AG (2016) Pretreatment of macroalgal biomass for biogas production. Energy Convers Manag 108:202–209. https://doi.org/10.1016/j.enconman.2015.11.008
Yun EJ, Kim HT, Cho KM et al (2016) Pretreatment and saccharification of red macroalgae to produce fermentable sugars. Bioresour Technol 199:311–318. https://doi.org/10.1016/j.biortech.2015.08.001
Agbor VB, Cicek N, Sparling R et al (2011) Biomass pretreatment: fundamentals toward application. Biotechnol Adv 29:675–685. https://doi.org/10.1016/j.biotechadv.2011.05.005
Feng D, Liu H, Li F et al (2011) Optimization of dilute acid hydrolysis of Enteromorpha. Chinese J Oceanol Limnol 29:1243–1248. https://doi.org/10.1007/s00343-011-0298-x
Jeong GT, Kim SK, Park DH (2013) Detoxification of hydrolysate by reactive-extraction for generating biofuels. Biotechnol Bioprocess Eng 18:88–93. https://doi.org/10.1007/s12257-012-0417-3
Kim D-H, Lee S-B, Jeong G-T (2014) Production of reducing sugar from Enteromorpha intestinalis by hydrothermal and enzymatic hydrolysis. Bioresour Technol 161:348–353. https://doi.org/10.1016/j.biortech.2014.03.078
Meinita MDN, Hong YK, Jeong GT (2012) Comparison of sulfuric and hydrochloric acids as catalysts in hydrolysis of Kappaphycus alvarezii (cottonii). Bioprocess Biosyst Eng 35:123–128. https://doi.org/10.1007/s00449-011-0609-9
Park JH, Hong JY, Jang HC et al (2012) Use of Gelidium amansii as a promising resource for bioethanol: a practical approach for continuous dilute-acid hydrolysis and fermentation. Bioresour Technol 108:83–88. https://doi.org/10.1016/j.biortech.2011.12.065
Yoon JJ, Kim YJ, Kim SH et al (2010) Production of polysaccharides and corresponding sugars from red seaweed. Adv Mater Res 93–94:463–466. https://doi.org/10.4028/www.scientific.net/AMR.93-94.463
Kang KE, Park DH, Jeong GT (2013) Effects of inorganic salts on pretreatment of Miscanthus straw. Bioresour Technol 132:160–165. https://doi.org/10.1016/j.biortech.2013.01.012
Nitsos CK, Matis KA, Triantafyllidis KS (2013) Optimization of hydrothermal pretreatment of lignocellulosic biomass in the bioethanol production process. Chemsuschem 6:110–122. https://doi.org/10.1002/cssc.201200546
Horn SJ, Aasen IM, Emptyvstgaard K (2000) Ethanol production from seaweed extract. J Ind Microbiol Biotechnol 25:249–254. https://doi.org/10.1038/sj.jim.7000065
Hargreaves PI, Barcelos CA, da Costa ACA, Pereira N (2013) Production of ethanol 3G from Kappaphycus alvarezii: evaluation of different process strategies. Bioresour Technol 134:257–263. https://doi.org/10.1016/j.biortech.2013.02.002
Meinita MDN, Kang JY, Jeong GT et al (2012) Bioethanol production from the acid hydrolysate of the carrageenophyte Kappaphycus alvarezii (cottonii). J Appl Phycol 24:857–862. https://doi.org/10.1007/s10811-011-9705-0
Lee S, Oh Y, Kim D et al (2011) Converting carbohydrates extracted from marine algae into ethanol using various ethanolic Escherichia coli strains. Appl Biochem Biotechnol 164:878–888. https://doi.org/10.1007/s12010-011-9181-7
Adams JMM, Toop TA, Donnison IS, Gallagher JA (2011) Seasonal variation in Laminaria digitata and its impact on biochemical conversion routes to biofuels. Bioresour Technol 102:9976–9984. https://doi.org/10.1016/J.BIORTECH.2011.08.032
Cho Y, Kim H, Kim SK (2013) Bioethanol production from brown seaweed, Undaria pinnatifida, using NaCl acclimated yeast. Bioprocess Biosyst Eng 36:713–719. https://doi.org/10.1007/s00449-013-0895-5
Choi D, Sim HS, Piao YL et al (2009) Sugar production from raw seaweed using the enzyme method. J Ind Eng Chem 15:12–15. https://doi.org/10.1016/j.jiec.2008.08.004
Dawczynski C, Schubert R, Jahreis G (2007) Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem 103(3):891–899. https://doi.org/10.1016/J.FOODCHEM.2006.09.041
Deniaud E, Quemener B, Fleurence J, Lahaye M (2003) Structural studies of the mix-linked β-(1 → 3)/β-(1 → 4)-D-xylans from the cell wall of Palmaria palmata (Rhodophyta). Int J Biol Macromol 33:9–18. https://doi.org/10.1016/S0141-8130(03)00058-8
Hou X, Yan X (1998) Study on the concentration and seasonal variation of inorganic elements in 35 species of marine algae. Sci Total Environ 222:141–156. https://doi.org/10.1016/S0048-9697(98)00299-X
Mutripah S, Meinita MDN, Kang JY et al (2014) Bioethanol production from the hydrolysate of Palmaria palmata using sulfuric acid and fermentation with brewer’s yeast. J Appl Phycol 26:687–693. https://doi.org/10.1007/s10811-013-0068-6
Robin A, Chavel P, Chemodanov A et al (2017) Diversity of monosaccharides in marine macroalgae from the Eastern Mediterranean Sea. Algal Res 28:118–127. https://doi.org/10.1016/j.algal.2017.10.005
Rupérez P (2002) Mineral content of edible marine seaweeds. Food Chem 79:23–26. https://doi.org/10.1016/S0308-8146(02)00171-1
Tan IS, Lam MK, Lee KT (2013) Hydrolysis of macroalgae using heterogeneous catalyst for bioethanol production. Carbohydr Polym 94:561–566. https://doi.org/10.1016/j.carbpol.2013.01.042
Trivedi N, Reddy CRK, Radulovich R, Jha B (2015) Solid state fermentation (SSF)-derived cellulase for saccharification of the green seaweed Ulva for bioethanol production. Algal Res 9:48–54. https://doi.org/10.1016/j.algal.2015.02.025
van der Wal H, Sperber BLHM, Houweling-Tan B et al (2013) Production of acetone, butanol, and ethanol from biomass of the green seaweed Ulva lactuca. Bioresour Technol 128:431–437. https://doi.org/10.1016/j.biortech.2012.10.094
Yaich H, Garna H, Besbes S et al (2011) Chemical composition and functional properties of Ulva lactuca seaweed collected in Tunisia. Food Chem 128:895–901. https://doi.org/10.1016/j.foodchem.2011.03.114
Yanagisawa M, Kawai S, Murata K (2013) Strategies for the production of high concentrations of bioethanol from seaweeds. Bioengineered 4:224–235. https://doi.org/10.4161/bioe.23396
Lee YJ, Choi YR, Lee SY et al (2011) Screening wild yeast strains for alcohol fermentation from various fruits. Mycobiology 39:33–39. https://doi.org/10.4489/MYCO.2011.39.1.033
Kraan S (2013) Mass-cultivation of carbohydrate rich macroalgae, a possible solution for sustainable biofuel production. Mitig Adapt Strateg Glob Chang 18:27–46. https://doi.org/10.1007/s11027-010-9275-5
Ross AB, Jones JM, Kubacki ML, Bridgeman T (2008) Classification of macroalgae as fuel and its thermochemical behaviour. Bioresour Technol 99:6494–6504. https://doi.org/10.1016/j.biortech.2007.11.036
Jmel MA, Anders N, Ben YN et al (2018) Variations in physicochemical properties and bioconversion efficiency of Ulva lactuca polysaccharides after different biomass pretreatment techniques. Appl Biochem Biotechnol 184:777–793. https://doi.org/10.1007/s12010-017-2588-z
Jeong TS, Kim YS, Oh KK (2011) Two-stage acid saccharification of fractionated Gelidium amansii minimizing the sugar decomposition. Bioresour Technol 102:10529–10534. https://doi.org/10.1016/j.biortech.2011.09.017
Wyman CE, Dale BE, Elander RT et al (2005) Coordinated development of leading biomass pretreatment technologies. Bioresour Technol 96:1959–1966. https://doi.org/10.1016/j.biortech.2005.01.010
Singh LK, Chaudhary G, Majumder CB, Ghosh S (2011) Utilization of hemicellulosic fraction of lignocellulosic biomaterial for bioethanol production. Adv Appl Sci Res 2:508–521
Aden A, Ruth M, Ibsen K et al (2002) Lignocellulosic biomass to ethanol process design and economics utilizing co-current dilute acid prehydrolysis and enzymatic hydrolysis for corn stover
Hendriks ATWM, Zeeman G (2009) Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour Technol 100:10–18. https://doi.org/10.1016/j.biortech.2008.05.027
Jmel MA, Ben Messaoud G, Marzouki MN et al (2016) Physico-chemical characterization and enzymatic functionalization of Enteromorpha sp. cellulose. Carbohydr Polym 135:274–279. https://doi.org/10.1016/j.carbpol.2015.08.048
Mithra MG, Jeeva ML, Sajeev MS, Padmaja G (2018) Comparison of ethanol yield from pretreated lignocellulo-starch biomass under fed-batch SHF or SSF modes. Heliyon 4:e00885. https://doi.org/10.1016/j.heliyon.2018.e00885
Chundawat SPS, Pal RK, Zhao C et al (2020) Ammonia fiber expansion (AFEX) pretreatment of lignocellulosic biomass. J Vis Exp 2020:1–8. https://doi.org/10.3791/57488
El-Dalatony MM, Salama ES, Kurade MB et al (2017) Utilization of microalgal biofractions for bioethanol, higher alcohols, and biodiesel production: a review. Energies 10:1–19. https://doi.org/10.3390/en10122110
Pezoa-Conte R, Leyton A, Anugwom I et al (2015) Deconstruction of the green alga Ulva rigida in ionic liquids: closing the mass balance. Algal Res 12:262–273. https://doi.org/10.1016/j.algal.2015.09.011
Kumar S, Gupta R, Kumar G et al (2013) Bioethanol production from Gracilaria verrucosa, a red alga, in a biorefinery approach. Bioresour Technol 135:150–156. https://doi.org/10.1016/j.biortech.2012.10.120
Khambhaty Y, Mody K, Gandhi MR et al (2012) Kappaphycus alvarezii as a source of bioethanol. Bioresour Technol 103:180–185. https://doi.org/10.1016/j.biortech.2011.10.015
Nguyen TH, Ra CH, Sunwoo IY et al (2017) Bioethanol production from Gracilaria verrucosa using Saccharomyces cerevisiae adapted to NaCl or galactose. Bioprocess Biosyst Eng 40:529–536. https://doi.org/10.1007/s00449-016-1718-2
Lee JY, Kim YS, Um BH, Oh KK (2013) Pretreatment of Laminaria japonica for bioethanol production with extremely low acid concentration. Renew Energy 54:196–200. https://doi.org/10.1016/j.renene.2012.08.025
Kim SW, Hong CH, Jeon SW, Shin HJ (2015) High-yield production of biosugars from Gracilaria verrucosa by acid and enzymatic hydrolysis processes. Bioresour Technol 196:634–641. https://doi.org/10.1016/j.biortech.2015.08.016
Ra CH, Jeong GT, Shin MK, Kim SK (2013) Biotransformation of 5-hydroxymethylfurfural (HMF) by Scheffersomyces stipitis during ethanol fermentation of hydrolysate of the seaweed Gelidium amansii. Bioresour Technol 140:421–425. https://doi.org/10.1016/j.biortech.2013.04.122
Kim HM, Wi SG, Jung S et al (2015) Efficient approach for bioethanol production from red seaweed Gelidium amansii. Bioresour Technol 175:128–134. https://doi.org/10.1016/j.biortech.2014.10.050
Tan IS, Lee KT (2014) Enzymatic hydrolysis and fermentation of seaweed solid wastes for bioethanol production: an optimization study. Energy 78:53–62. https://doi.org/10.1016/j.energy.2014.04.080
Wei N, Quarterman J, Jin YS (2013) Marine macroalgae: an untapped resource for producing fuels and chemicals. Trends Biotechnol 31:70–77. https://doi.org/10.1016/j.tibtech.2012.10.009
Borines MG, de Leon RL, Cuello JL (2013) Bioethanol production from the macroalgae Sargassum spp. Bioresour Technol 138:22–29. https://doi.org/10.1016/j.biortech.2013.03.108
Lee SM, Lee JH (2011) The isolation and characterization of simultaneous saccharification and fermentation microorganisms for Laminaria japonica utilization. Bioresour Technol 102:5962–5967. https://doi.org/10.1016/j.biortech.2011.02.006
Jang JS, Cho YK, Jeong GT, Kim SK (2012) Optimization of saccharification and ethanol production by simultaneous saccharification and fermentation (SSF) from seaweed, Saccharina japonica. Bioprocess Biosyst Eng 35:11–18. https://doi.org/10.1007/s00449-011-0611-2
Yanagisawa M, Ojima T, Nakasaki K (2011) Bioethanol from sea lettuce with the use of crude enzymes derived from waste. J Mater Cycles Waste Manag 13:321–326. https://doi.org/10.1007/s10163-011-0026-9
Ge L, Wang P, Mou H (2011) Study on saccharification techniques of seaweed wastes for the transformation of ethanol. Renew Energy 36:84–89. https://doi.org/10.1016/j.renene.2010.06.001
Dubois M, Gilles KA, Hamilton JK et al (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Marsden W, Gray PP, Nippard GJ, Quinlan MR (1982) Evaluation of the Dns method for analysing lignocellulosic hydrolysates. J Chem Technol Biotechnol 32:1016–1022. https://doi.org/10.1002/jctb.5030320744
Hu R, Lin L, Liu T et al (2008) Reducing sugar content in hemicellulose hydrolysate by DNS method: a revisit. J Biobased Mater Bioenergy 2:156–161. https://doi.org/10.1166/jbmb.2008.306
Ramachandra TV, Hebbale D (2020) Bioethanol from macroalgae: prospects and challenges. Renew Sustain Energy Rev 117:109479. https://doi.org/10.1016/j.rser.2019.109479
Meinita MDN, Hong YK, Jeong GT (2012) Detoxification of acidic catalyzed hydrolysate of Kappaphycus alvarezii (cottonii). Bioprocess Biosyst Eng 35:93–98. https://doi.org/10.1007/s00449-011-0608-x
Bosch-Reig F, Marcote MJ, Minana MD, Cabello ML (1992) Separation and identification of sugars and maltodextrines by thin layer chromatography: application to biological fluids and human milk. Talanta 39:1493–1498. https://doi.org/10.1016/0039-9140(92)80131-V
Rahman SHA, Choudhury JP, Ahmad AL, Kamaruddin AH (2007) Optimization studies on acid hydrolysis of oil palm empty fruit bunch fiber for production of xylose. Bioresour Technol 98:554–559. https://doi.org/10.1016/j.biortech.2006.02.016
Hebbale D, Bhargavi R, Ramachandra TV (2019) Saccharification of macroalgal polysaccharides through prioritized cellulase producing bacteria. Heliyon 5:e01372. https://doi.org/10.1016/j.heliyon.2019.e01372
Percival Zhang YH, Himmel ME, Mielenz JR (2006) Outlook for cellulase improvement: screening and selection strategies. Biotechnol Adv 24:452–481. https://doi.org/10.1016/j.biotechadv.2006.03.003
Jang S-S (2012) Production of mono sugar from acid hydrolysis of seaweed. African J Biotechnol 11:1953–1963. https://doi.org/10.5897/AJB10.1681
Kupiainen L, Ahola J, Tanskanen J (2010) Comparison of formic and sulfuric acids as a glucose decomposition catalyst. Ind Eng Chem Res 49:8444–8449. https://doi.org/10.1021/ie1008822
Gonçalves DB, Batista AF, Rodrigues MQRB et al (2013) Ethanol production from macaúba (Acrocomia aculeata) presscake hemicellulosic hydrolysate by Candida boidinii UFMG14. Bioresour Technol 146:261–266. https://doi.org/10.1016/j.biortech.2013.07.075
Smith GM (1938) Cryptogamic botany. Volume I, Algae and fungi. Cryptogam Bot Vol I, Algae fungi
Choi WY, Han JG, Lee CG et al (2012) Bioethanol production from Ulva pertusa Kjellman by high-temperature liquefaction. Chem Biochem Eng Q 26:15–21. https://doi.org/10.2147/NSS.S6844
Wu FC, Wu JY, Liao YJ et al (2014) Sequential acid and enzymatic hydrolysis in situ and bioethanol production from Gracilaria biomass. Bioresour Technol 156:123–131. https://doi.org/10.1016/j.biortech.2014.01.024
Wang CY, Hsieh YR, Ng CC et al (2009) Purification and characterization of a novel halostable cellulase from Salinivibrio sp. strain NTU-05. Enzyme Microb Technol 44:373–379. https://doi.org/10.1016/j.enzmictec.2009.02.006
Islam F, Roy N (2018) Screening, purification and characterization of cellulase from cellulase producing bacteria in molasses. BMC Res Notes 11:1–6. https://doi.org/10.1186/s13104-018-3558-4
Shanmughapriya S, Kiran GS, Selvin J et al (2010) Optimization, purification, and characterization of extracellular mesophilic alkaline cellulase from sponge-associated Marinobacter sp. MSI032. Appl Biochem Biotechnol 162:625–640. https://doi.org/10.1007/s12010-009-8747-0
Gao Z, Ruan L, Chen X et al (2010) A novel salt-tolerant endo-β-1,4-glucanase Cel5A in Vibrio sp. G21 isolated from mangrove soil. Appl Microbiol Biotechnol 87:1373–1382. https://doi.org/10.1007/s00253-010-2554-y
Harshvardhan K, Mishra A, Jha B (2013) Purification and characterization of cellulase from a marine Bacillus sp. H1666: a potential agent for single step saccharification of seaweed biomass. J Mol Catal B Enzym 93:51–56. https://doi.org/10.1016/j.molcatb.2013.04.009
Kim JY, Hur SH, Hong JH (2005) Purification and characterization of an alkaline cellulase from a newly isolated alkalophilic Bacillus sp. HSH-810. Biotechnol Lett 27:313–316. https://doi.org/10.1007/s10529-005-0685-5
Picart P, Diaz P, Pastor FIJ (2008) Stachybotrys atra BP-A produces alkali-resistant and thermostable cellulases. Antonie van Leeuwenhoek. Int J Gen Mol Microbiol 94:307–316. https://doi.org/10.1007/s10482-008-9248-9
Trivedi N, Gupta V, Kumar M et al (2011) An alkali-halotolerant cellulase from Bacillus flexus isolated from green seaweed Ulva lactuca. Carbohydr Polym 83:891–897. https://doi.org/10.1016/j.carbpol.2010.08.069
Hirasawa K, Uchimura K, Kashiwa M et al (2006) Salt-activated endoglucanase of a strain of alkaliphilic Bacillus agaradhaerens Antonie van Leeuwenhoek. Int J Gen Mol Microbiol 89:211–219. https://doi.org/10.1007/s10482-005-9023-0
Robinson PK (2015) Enzymes: principles and biotechnological applications. Essays Biochem 59:1–41. https://doi.org/10.1042/BSE0590001
Timung R, Naik Deshavath N, Goud VV, Dasu VV (2016) Effect of subsequent dilute acid and enzymatic hydrolysis on reducing sugar production from sugarcane bagasse and spent citronella biomass. J Energy 2016:1–12. https://doi.org/10.1155/2016/8506214
Pilavtepe M, Celiktas MS, Sargin S, Yesil-Celiktas O (2013) Transformation of Posidonia oceanica residues to bioethanol. Ind Crops Prod 51:348–354. https://doi.org/10.1016/j.indcrop.2013.09.020
Acknowledgements
We are grateful to (i) ISRO-IISc Space Technology Cell [ISTC/BES/TVR/389], (ii) Gajanana and Madhavi Hegde [CES/TVR/GJH001] endowment for biofuel research for the financial support, and (iii) Indian Institute of Science [R1011] for the infrastructure support and colleagues at EWRG, IISc field station, Kumta, for assistance during the field sampling.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hebbale, D., Ramachandra, T.V. Optimal sugar release from macroalgal feedstock with dilute acid pretreatment and enzymatic hydrolysis. Biomass Conv. Bioref. 13, 8287–8300 (2023). https://doi.org/10.1007/s13399-021-01845-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-021-01845-8