Abstract
Aim
To review outcomes and local evolution of treated lesions following percutaneous image-guided screw fixation (PIGSF) of pathological/insufficiency fractures (PF/InF) and impeding fractures (ImF) in cancer patients at two tertiary centres.
Materials and methods
Thirty-two consecutive patients (mean age 67.5 years; range 33–86 years) with a range of tumours and prognoses underwent PIGSF for non/minimally displaced PF/InF and ImF. Screws were placed under CT/fluoroscopy or cone-beam CT guidance, with or without cementoplasty. Clinical outcomes were assessed using a simple 4-point scale (1 = worse; 2 = stable; 3 = improved; 4 = significantly improved). Local evolution was reviewed on most recent follow-up imaging. Technical success, complications, and overall survival were evaluated.
Results
Thirty-six lesions were treated with 74 screws mainly in the pelvis and femoral neck (58.2 %); including 47.2 % PF, 13.9 % InF, and 38.9 % ImF. Cementoplasty was performed in 63.9 % of the cases. Technical success was 91.6 %. Hospital stay was ≤3 days; 87.1 % of lesions were improved at 1-month follow-up; three major complications (early screw-impingement radiculopathy; accelerated coxarthrosis; late coxofemoral septic arthritis) and one minor complication were observed. Unfavourable local evolution at imaging occurred in 3/24 lesions (12.5 %) at mean 8.7-month follow-up, including poor consolidation (one case) and screw loosening (two cases, at least 1 symptomatic). There were no cases of secondary fractures.
Conclusions
PIGSF is feasible for a wide range of oncologic patients, offering good short-term efficacy, acceptable complication rates, and rapid recovery. Unfavourable local evolution at imaging may be relatively frequent, and requires close clinico-radiological surveillance.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Bone metastatic disease affects up to 50 % of the cancer patients, and incidence increases as overall survival (OS) improves [1–4]. Symptomatic lesions may result from the following: (a) fractures secondary to bone resorption following chemotherapy/steroid treatment (insufficiency fracture; InF); (b) fracture secondary to bone replacement by tumoural tissue (pathological fracture; PF); or (c) Extensive tumoural involvement of a weight-bearing bone at high risk of fracture (impending fracture; ImF) [5–7]. Lesions are typically associated with pain, morbidity [2], mortality [8], and health resource utilisation [9]; and may significantly delay systemic therapy [6].
Therapeutic or prophylactic fixation is therefore essential to optimise outcome, and is typically performed via surgery [10–15]. However, invasive procedures may be unsuitable for high-risk patients with complex (especially pelvic) lesions, and may result in significant complications and prolonged post-operative recovery [15, 16].
Percutaneous image-guided screw fixation (PIGSF) is an emerging technique which utilises cross-sectional imaging guidance to precisely fix even complex fractures, avoiding significant soft tissue trauma and damage to adjacent structures [17, 18]. Several single-centre studies have reported good short-term outcomes and acceptable complication rates for treatment of oncologic bone lesions [16–20]. However, little is known regarding the local evolution of treated lesions on imaging follow-up and associated clinical consequences. This aspect is likely to become increasingly significant as OS improves and longer-term construct stability is necessitated.
The purpose of this study is to retrospectively review our double-centre experience of PIGSF for ImF, InF, and PF in cancer patients. We present short-term clinical outcomes; describe local evolution of treated lesions on serial imaging follow-up; and discuss the rationale and role of this novel technique in management of this complex patient cohort.
Materials and Methods
This double-centre single-arm retrospective observational study was conducted with Institutional Review Board approval and a waiver of informed consent at both Centres.
Study Population
Retrospective review of electronic records identified 32 consecutive oncologic patients (18 male, 14 female; mean age 67.5 years, range 33–86 years) who had undergone PIGSF for PF, InF, or ImF across two tertiary referral centres.
Disease spectrum ranged from solitary bone lesions to polymetastatic involvement. All patients had been treated with systemic chemotherapy, and over 60 % had undergone prior local treatment (Table 1).
All cases were discussed by a multidisciplinary tumour board including oncologists, orthopaedic surgeons, interventional radiologists, radiotherapists, and anaesthesiologists. Patients were referred for PIGSF if they had a symptomatic non-displaced or mildly displaced PF/InF; an ImF in long bone with Mirels’ score ≥8 [7] and less than 3 cm cortical involvement [5]; life expectancy >1 month; and were unsuitable for surgical treatment due to suboptimal fitness, refusal of consent, or unacceptable delay to systemic therapy. Exclusion criteria were severe and irreversible coagulation disorders and/or local/systemic sepsis.
Procedures
All procedures were performed in an interventional radiology suite under strict sterile conditions, following induction of general anaesthesia, by a senior Interventional Radiologist (minimum 6 years experience) and a resident/fellow (≤2 years experience).
Imaging guidance consisted of combined CT (Somatom Definition AS, Siemens, Erlangen, Germany) and Fluoroscopy (Arcadis Orbit; Siemens, Erlangen, Germany) in Centre 1 (Strasbourg); and Cone-Beam CT (CBCT) equipped with XPerCT and X-PerGuide tools (Philips Healthcare, Amsterdam, Netherlands) in Centre 2 (Bordeaux) .
Anticoagulants were stopped 5 days prior and blood clotting parameters were tested 24 h pre-procedure, ensuring minimum prothrombin time of 50 % and platelet count of 50,000/mm3, according to Society of Interventional Radiology (SIR) guidelines [21]. Broad-spectrum intravenous antibiotic cover (2 g cefazolin) was administered pre-procedurally.
Patients were positioned prone or supine (according to lesion location), and initial CT/CBCT was acquired to facilitate planning of screw trajectories and cutaneous entry-point marking. Multi-planar reconstructed images were used to measure screw length from cortical surface to intended intra-osseous anchor point.
Two different systems of cannulated self-tapping and self-drilling screws were used (6.5 mm Asnis™ III Cannulated Screw System, Kalamazoo, Michigan, USA; 7.2 mm Koenigsee-implantate, Königsee Implantate GmbH, Allendorf, Germany). Screws from the second system have multiple holes in the tip, thus allowing poly-methyl-methacrylate (PMMA, Osteopal V, Heraeus Medical GmbH, Wehrheim, Germany) injection inside the screw in order to fix its tip.
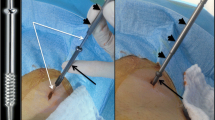
For each screw, following a small skin incision, a threaded guidewire was advanced along the planned trajectory using an electric driver (Stryker Universal Driver, Kalamazoo, Michigan, USA). Repeat CT/CBCT was performed to confirm adequate position. If inadequate, the wire was unscrewed using the driver and then repositioned.
The cannulated screw was advanced co-axially and manually over the wire using a driver that comes along with the screw kit, under fluoroscopic guidance, until the distal screw-tip was anchored in a satisfactory position. Successful placement was confirmed on final CT/CBCT.
For PF/InF, screws were placed perpendicularly across the fracture line, with the distal tip anchored in a healthy bone if possible. Iliac, sacral, and acetabular fractures were treated with single or multiple screws across each planar fracture component (Figs. 1, 2). Pubic ramus fractures were stabilised using a single retrograde screw (Fig. 3). An inverted triangle construct was used to treat femoral neck fractures, with one screw along the inferior neck and two screws anteriorly and posteriorly in the superior neck, to maximise biomechanical stability [22, 23]. Shoulder-girdle fractures were treated with 1–2 screws across the clavicle or coracoid, depending on the complexity as reported elsewhere [18]. ImF were similarly stabilised, with the screw traversing the lesion and ideally anchoring in normal bone.
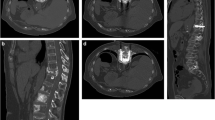
74-Year old male affected by non-Hodgkin’s lymphoma, presenting with a huge mass around the left iliac wing (dotted circle; A). This was complicated by a complex fracture with axial (arrows; B) and coronal (arrows; C) components. The patient underwent percutaneous cryoablation using 5 cryo-probes to treat the soft tissue mass (arrows; D). At the same time, 5 screws were applied to fix the complex fracture: 2 for the axial component (E) and 3 for the coronal component (F). Screws were stabilised with PMMA injection (F)
70-Year old female with breast cancer presenting with non-consolidated fracture of left iliac wing (arrow; A). The fracture was fixed with 4 screws, with satisfactory placement on coronal and axial images (B, C). However, at 8-month CT follow-up, one of the screws had backed out of the cortex, consistent with loosening (arrows; D, E). Unfortunately the patient was lost to clinical follow-up
81-Year old male with osteolytic metastases from prostate cancer in the acetabulum (arrow; A) and left ischio-pubic ramus (arrow; B). The patient underwent percutaneous screw fixation with cement augmentation of the left ilio-pubic ramus in order to treat the acetabular lesion (C); in the same session, a single screw was applied to treat the left ischio-pubic fracture (D). Plain X-ray obtained 1-month post-procedure demonstrated satisfactory appearances (E); however, repeat radiograph at 11 months demonstrated peri-prosthetic lucency consistent with screw loosening (arrows, F). The patient was mildly symptomatic on clinical review
Additional cementoplasty was performed at the discretion of the attending senior radiologist, on the basis of substantial osteolysis or osteopaenia around the screw (especially around its tip), which might compromise stability. In these cases, injection was performed through the Königsee Implantate screw system or through a dedicated 10G co-axial trocar (Gangi Special Vertebroplasty Needle, Optimed, Ettlingen, Germany) inserted adjacent to the screws. PMMA cement was injected under continuous fluoroscopy until adequate filling or leakage was adjudged.
Following the procedure, compressive dressings were applied to skin entry sites, and patients were transferred to a recovery ward for discharge once medically fit.
Follow-Up
All patients were clinically reviewed 1 month post-procedure by the treating interventional radiologist, with assessment of the treated lesion using a simple 4-point scale (1 = worse; 2 = unchanged; 3 = improved; 4 = significantly improved).
Systematic imaging follow-up was not performed; however, all patients underwent routine imaging follow-up every 3–6 months for disease monitoring, and additional imaging if local complications were suspected on the site of the treated lesion. Therefore, patients’ electronic records were reviewed to find out their last imaging follow-up available, including the area of PIGSF in the field of view, in order to investigate local evolution of the treated foci.
Data Collection and Statistical Analysis
Demographics, tumour type, lesion location, Mirels’ score for long bones [7], and prior treatments were tabulated. Number of screws, length of hospital stay, technical success, complications (according to SIR guidelines [21]), short-term clinical outcomes (change in status on 4-point scale at 1 month), local evolution of the treated focus on latest follow-up imaging, and overall survival (last record on institutional records in known deceased patients) were evaluated.
Technical success was defined as satisfactory screw placement perpendicular to the fracture line (PF and InF) or lytic lesion (ImF), with both ends securely anchored in good-quality bone; if cementoplasty was coupled to PIGSF and some leakage occurred, the procedure did not account for technical success.
Local evolution was assessed on the most recent imaging covering the treated lesion (identified using retrospective chart review). Screw loosening was defined as unwinding or >2 mm peri-screw lucency. If available, contemporaneous clinical data was referenced to evaluate symptoms.
Statistical analysis was performed using MATLAB® (Math Works, Inc.). Fisher’s test was used to compare outcomes between ImF and PF/InF groups. A Kaplan–Meier curve was plotted to outline population OS.
Results
Between December 2012 and December 2015, 36 lesions were treated in 33 sessions (mean 1.09 per session; range 1–2) under CT/Fluoroscopy (21/32; 65.6 % in Centre 1) or CBCT guidance (11/32; 34.4 % in Centre 2). The most commonly treated locations were the ilio-pubic/ischio-pubic rami (22.2 %), iliac crest (19.4 %), and femoral neck (16.6 %; mean Mirels’ score 10.2). Seventeen (47.2 %) lesions were treated due to PF, 5 due to InF (13.9 %), and 14 (38.9 %) due to ImF.
Seventy-four screws were implanted (mean number of screws per session 2.2; range 1–5; mean number screws per lesion 2.0; range 1–5; mean screws length 82.5 ± 21.7 mm; and range 50–120). Adjunctive cementoplasty was performed in 23/36 (63.9 %) lesions, including 12 PF/InF and 11 ImF.
Technical success was 91.6 % (33/36 lesions), since PMMA leakage occurred in 3 lesions (13.0 %, 1 ImF and 2 PF/InF; p = 0.59).
In-patient hospital stay was ≤3 days in all cases.
Three major complications (3/33 sessions, 9.1 %) were observed, including a painful L5 radiculopathy secondary to screw impingement 10 days following an uncemented sacral fracture fixation (Grade D; necessitated image-guided screw removal to relieve symptoms following failed local steroid injection); a septic arthritis of the coxofemoral joint requiring surgical revision and arthrodesis 12 months following PIGFS (grade D); and an accelerated coxarthrosis 2 months following treatment of a plasmacytoma-related acetabular fracture (Grade E).
There was 1 (3 %) minor complication consistent with an asymptomatic intra-articular cement leakage following acetabular PIGSF (Grade A).
One month clinical follow-up was available for 29/32 patients (90.6 %) and 31/36 lesions (86.1 %) due to early mortality of 3 patients. On a per-lesion basis, 87.1 % were improved from baseline (Table 2). There was no significant difference in distribution of “significantly improved” and “improved” cases vs. “unchanged” and “worse” cases between ImF and PF/InF groups (p = 0.6), and between cemented and uncemented PIGSF (p = 0.5).
Imaging follow-up (predominantly with CT) was available in 22/32 patients (68.7 %) and 24/36 lesions (66.6 %), with mean duration of 8.7 months (range 0.3–25 months; Table 2).
Ten patients were lost to follow-up. Unfavourable local evolution was observed in 3/24 lesions (12.5 %): absent bone consolidation was seen in 1 case of post-cryoablation (thyroid metastasis) acromion InF treated with uncemented PIGSF after 5 months. However, the patient remained asymptomatic with good arm mobility despite the persistent cavity—probably due to good-quality distal fixation in the healthy bone; screw loosening occurred in 2 cases, with unwinding of a screw-head 8 months post-PIGSF of an iliac PF (Fig. 2; lost to clinical follow-up); and peri-screw lucency on plain radiograph 11 months post-fixation of an ischio-pubic ramus PF (Fig. 3; symptomatic with 3/10 pain score on walking at contemporaneous clinical follow-up). All unfavourable local evolutions revealed at imaging follow-up were observed in uncemented cases; there was a significant difference in occurrence between Uncemented and Cemented groups (p = 0.001); on the other hand, there was no significant difference in occurrence between ImF and PF/InF groups (p = 0.22). No secondary fractures were detected on imaging follow-up.
OS at 3, 12, and 24 months were 81.2, 75.0, and 68.8 %, respectively (Fig. 4).
Discussion
PIGSF is a novel technique for fracture fixation, which has recently been applied for the treatment of long bone, pelvic, and shoulder-girdle lesions in oncologic patients [16–20]. It offers advantages over conventional surgery including excellent visualisation of surrounding structures to avoid iatrogenic injury, and a rapid post-procedural recovery to minimise onco-therapeutic delay [16–19]. The technique involves precise placement of co-axial cannulated screws over a guidewire placed perpendicular to the fracture/lesion under imaging guidance, securely anchoring the distal screw in good-quality bone [16–18]. Prior studies have demonstrated technical feasibility, safety, and good short/mid-term outcomes [16, 17, 19], which appear comparable to surgery [11–13].
Our study similarly illustrates good technical success (91.6 %), good short-term symptomatic improvement (87.1 % of lesions improved at 1 month), and an acceptable major complication rate (9.1 %). Although we utilised a simple 4-point scale (due to the retrospective nature of the study) rather than a formal Visual Analogue Scale (VAS), our outcomes are comparable to those of Deschamps et al. and Hartung et al. [16, 17, 19].
PIGSF may be combined with thermal ablation or cementoplasty to optimise palliation and stabilisation. We performed thermal ablation prior to PIGSF in three cases to control local disease and/or treat pain related to extra-osseous tumour extension, without any particular technical difficulty.
In 64 % of cases (11 ImF and 12 PF/InF), we performed cementoplasty following screw placement, intending to improve screw-tip anchorage within regions of osteolysis and osteopaenia; no significant benefit in terms of pain management was noted between cemented and uncemented cases. This approach is similar to Hartung et al. [19], but differs from Deschamps et al. [17], who did not perform PMMA injection for PF/InFs due to risk of leakage. Although theoretically more likely for InF/PF than ImF, we did not encounter any clinically relevant leakage in either group similar to other series [30]. Additionally, several biomechanical and clinical studies suggest that cementoplasty may enhance screw fixation and optimise biomechanics [25–29]; and all our unfavourable local cement augmentation evolution rated at imaging follow-up occurred in non-cemented cases. Although definitive proof is lacking [24], it is probably an effective and safe option to optimise screw fixation with PMMA injection when it is performed under continuous fluoroscopic guidance. Moreover, the development of perforated cannulated screws offers a novel method of implementation with an unlikely event of PMMA leakage [31].
Improved life expectancy of oncologic patients necessitates longer-term construct stability. In our series (approximately 2/3 survival at 24 months), 12.5 % of treated lesions with available follow-up imaging (mean 8.7 months) demonstrated poor osseous consolidation or screw loosening; and at least 1 patient was symptomatic. Despite limited sample size and data quality, local unfavourable evolution at imaging follow-up may be relatively frequent similar to surgical series [28, 32]. Our non-consolidated case can be explained by poor osseous substrate in a post-ablation cavity. On the other hand, the two cases of screw loosening occurred in uncemented cases with mildly displaced or distracted fractures. Other contributory factors may include altered biomechanics and fluted design of self-tapping screws [33]. At the moment, our data are insufficient to quantify the real incidence of poor bone consolidation around the screw or screw loosening. However, longer-term local evolution of treated lesions may have significant implications for patient selection and optimal technique; therefore, prospective studies are required to investigate this increasingly significant issue.
The role of PIGSF in management of PF, InF, and ImF in cancer patients is rapidly evolving. Given the lack of long-term follow-up data, we believe that PIGSF is best suited for fracture consolidation in high-risk, non-surgical candidates with short life expectancy. Conversely, surgery is favoured for low-risk patients with isolated solitary bone metastasis and good prognosis, since it offers potentially curative resection and optimal biomechanical reconstruction [10, 13, 15, 34]. Patients between these extremes should ideally be treated with cytoreduction followed by stabilisation—either via limited surgical resection and reconstruction; or using percutaneous thermal ablation with PIGSF/cementoplasty [10, 35]. PIGSF with close clinical and radiological monitoring is therefore a viable option for a wide range of patients.
Several alternative techniques have been described for percutaneous treatment of oncologic bone lesions, including internal fixation with modified trocars, bone marrow nails, Kirschner wires, and cement-filled catheters [29, 36–39]; and augmentation using micro-needles mesh and cement [40]. Although supported by some biomechanical evidence [41], these techniques have not widespread and clinical evidence is lacking. Conversely, screw fixation is commonly used by orthopaedic surgeons in patients unsuitable for invasive surgery in a variety of locations and settings [14, 22, 23, 32, 42]. In particular, several clinical and biomechanical studies support the use of an “inverted triangle” screw-configuration with/without cement augmentation to provide intramedullary reinforcement in proximal femoral neck fractures [22, 23, 25, 43]. The PIGSF technique described herein and elsewhere [16] is comparable to existing surgical techniques, and probably represents the most appropriate procedure for most lesions. Cementoplasty alone appears structurally inadequate for larger or complex lesions, and is best utilised in combination with PIGSF [16, 18, 24, 30].
The principal limitation of this study relies on its retrospective design, which precluded utilisation of validated scales, systematic clinico-radiological follow-up, and comparison of symptoms in patients with/without radiological loosening. Our small, heterogeneous sample limits generalisability, but reflects clinical practice in two different tertiary centres. The high attrition rate (1/3 of patients lost to follow-up) is unfortunately common to all studies in this population [13].
In conclusion, PIGSF is a technically feasible procedure for palliation and stabilisation of ImF, PF, and InF in cancer patients with a range of prognoses. It offers rapid post-operative recovery, acceptable complication rates, and good short-term efficacy. Unfavourable local evolution of treated lesions as assessed with imaging may be relatively frequent and potentially symptomatic. Therefore, close clinico-radiological monitoring may be advisable following PIGSF. Further prospective studies are required to evaluate long-term sequelae of this novel technique in this complex and vulnerable patient cohort.
References
Parker C, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, Fossa SD, Chodacki A, Wiechno P, Logue J, Seke M, Widmark A, Johannessen DC, Hoskin P, Bottomley D, James ND, Solberg A, Syndikus I, Kliment J, Wedel S, Boehmer S, Dall’Oglio M, Franzen L, Coleman R, Vogel-zang NJ, O’Bryan-Tear CG, Staudacher K, Garcia-Vargas J, Shan M, Bruland OS, Sartor O, Investigators A. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369(3):213–23.
Coleman RE. Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res. 2006;12:6243s–9s.
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2007. Cancer CA Cancer J Clin. 2007;57:43–66.
Perrin RG, Laxton AW. Metastatic spine disease: epidemiology, pathophysiology, and evaluation of patients. Neurosurg Clin N Am. 2004;15:365–73.
Deschamps F, Farouil G, Hakime A, Barah A, Guiu B, Teriitehau C, Auperin A, deBaere T. Cementoplasty of metastases of the proximal femur: is it a safe palliative option? J Vasc Interv Radiol. 2012;23(10):1311–6. doi:10.1016/j.jvir.2012.06.027.
Piccioli A, Spinelli MS, Maccauro G. Impending fracture: a difficult diagnosis. Injury. 2014;45(Suppl 6):S138–41.
Mirels H. Metastatic disease in long bones. A proposed scoring system for diagnosing impending pathologic fractures. Clin Orthop Relat Res. 1989;249:256–64.
Manglani HH, Marco RA, Picciolo A, Healey JH. Orthopedic emergencies in cancer patients. Semin Oncol. 2000;27:299–310.
Body JJ, Pereira J, Sleeboom H, Maniadakis N, Terpos E, Acklin YP, Finek J, Gunther O, Hechmati G, Mossman T, Costa L, Rogowski W, Nahi H, von Moos R. Health resource utilization associated with skeletal-related events: results from a retrospective European study. Eur J Health Econ. 2015.
Müller DA, Capanna R. The surgical treatment of pelvic bone metastases. Adv Orthop. 2015;2015:525363. doi:10.1155/2015/525363.
Choy WS, Kim KJ, Lee SK, Yang DS, Jeung SW, Choi HG, Park HJ. Surgical treatment of pathological fractures occurring at the proximal femur. Yonsei Med J. 2015;56(2):460–5.
Arvinius C, Parra JL, Mateo LS, Maroto RG, Borrego AF, Stern LL. Benefits of early intramedullary nailing in femoral metastases. Int Orthop. 2014;38(1):129–32.
Talbot M, Turcotte RE, Isler M, Normandin D, Iannuzzi D, Downer P. Function and health status in surgically treated bone metastases. Clin Orthop Relat Res. 2005;438:215–20.
Liu G, Hasan MY, Wong HK. Minimally invasive iliac screw fixation in treating painful metastatic lumbosacral deformity: a technique description and clinical results. Eur Spine J. 2016.
Wedin R. Surgical treatment for pathologic fracture. Acta Orthop Scand Suppl. 2001;72(302):1–29.
Deschamps F, Farouil G, Hakime A, Teriitehau C, Barah A, de Baere T. Percutaneous stabilization of impending pathological fracture of the proximal femur. Cardiovasc Intervent Radiol. 2012;35(6):1428–32.
Deschamps F, de Baere T, Hakime A, Pearson E, Farouil G, Teriitehau C, Tselikas L. Percutaneous osteosynthesis in the pelvis in cancer patients. Eur Radiol. 2015.
Garnon J, Koch G, Ramamurthy N, Caudrelier J, Rao P, Tsoumakidou G, Cazzato RL, Gangi A. Percutaneous CT and fluoroscopy-guided screw fixation of pathological fractures in the shoulder girdle: technical report of 3 cases. Cardiovasc Intervent Radiol. 2016.
Hartung MP, Tutton SM, Hohenwalter EJ, King DM, Neilson JC. Safety and efficacy of minimally invasive acetabular stabilization for periacetabular metastatic disease with thermal ablation and augmented screw fixation. J Vasc Interv Radiol. 2016.
Trumm CG, Rubenbauer B, Piltz S, Reiser MF, Hoffmann RT. Screw placement and osteoplasty under computed tomographic-fluoroscopic guidance in a case of advanced metastatic destruction of the iliosacral joint. Cardiovasc Intervent Radiol. 2011;34(Suppl 2):S288–93.
Sacks D, McClenny TE, Cardella JF, Lewis CA. Society of Interventional Radiology clinical practice guidelines. J Vasc Interv Radiol. 2003;14:S199–202.
Selvan VT, Oakley MJ, Rangan A, Al-Lami MK. Optimum configuration of cannulated hip screws for the fixation of intracapsular hip fractures: a biomechanical study. Injury. 2004;35(2):136–41.
Ye Y, Hao J, Mauffrey C, Hammerberg EM, Stahel PF, Hak DJ. Optimizing stability in femoral neck fracture fixation. Orthopedics. 2015;38(10):625–30.
Cazzato RL, Palussière J, Buy X, Denaro V, Santini D, Tonini G, Grasso RF, Zobel BB, Poretti D, Pedicini V, Balzarini L, Lanza E. Percutaneous long bone cementoplasty for palliation of malignant lesions of the limbs: a systematic review. Cardiovasc Intervent Radiol. 2015;38(6):1563–72. doi:10.1007/s00270-015-1082-7 Epub 2015 Mar 24.
Palumbo BT, Nalley C, Gaskins RB 3rd, Gutierrez S, Alexander GE III, Anijar L, Nayak A, Cheong D, Santoni BG. Biomechanical analysis of impending femoral neck fractures: the role of percutaneous cement augmentation for osteolytic lesions. Clin Biomech (Bristol, Avon). 2014;29(3):289–95.
Rüger M, Sellei RM, Stoffel M, von Rüden C. The effect of polymethyl methacrylate augmentation on the primary stability of cannulated bone screws in an anterolateral plate in osteoporotic vertebrae: a human cadaver study. Glob Spine J. 2016;6(1):46–52.
He D, Wu L, Sheng X, Xiao Q, Zhu Y, Yu W, Liu F, Zhu K. Internal fixation with percutaneous kyphoplasty compared with simple percutaneous kyphoplasty for thoracolumbar burst fractures in elderly patients: a prospective randomized controlled trial. Eur Spine J. 2013;22(10):2256–63.
Kim J-B, Park S-W, Lee Y-S, Nam T-K, Park Y-S, Kim Y-B. The effects of spinopelvic parameters and paraspinal muscle degeneration on S1 screw loosening. J Korean Neurosurg Soc. 2015;58(4):357–62.
Tian QH, He CJ, Wu CG, Li YD, Gu YF, Wang T, Xiao QP, Li MH. Comparison of percutaneous cementoplasty with and without interventional internal fixation for impending malignant pathological fracture of the proximal femur. Cardiovasc Intervent Radiol. 2016;39(1):81–9.
Cazzato RL, Buy X, Eker O, Fabre T, Palussiere J. Percutaneous long bone cementoplasty of the limbs: experience with fifty-one non-surgical patients. Eur Radiol. 2014;24(12):3059–68. doi:10.1007/s00330-014-3357-9.
Lin PP, Kang HG, Kim YI, Kim JH, Kim HS. Minimally invasive surgery for femoral neck fractures using bone cement infusible hollow-perforated screw in high-risk patients with advanced cancer. Surg Oncol. 2015;24(3):226–31.
Galbusera F, Volkheimer D, Reitmaier S, Berger-Roscher N, Kienle A, Wilke HJ. Pedicle screw loosening: a clinically relevant complication? Eur Spine J. 2015;24(5):1005–16.
Muller ME, Allgöwer M, Schneider R, et al. Manual of internal fixation: techniques recommended by the AO/ASIF group. New York: Springer; 1995.
Cazzato RL, Bonichon F, Buy X, Godbert Y, de Figuereido BH, Pointillart V, Palussière J. Over ten years of single-institution experience in percutaneous image-guided treatment of bone metastases from differentiated thyroid cancer. Eur J Surg Oncol. 2015;41(9):1247–55.
Cazzato RL, Buy X, Grasso RF, Luppi G, Faiella E, Quattrocchi CC, Pantano F, BeomonteZobel B, Tonini G, Santini D, Palussiere J. Interventional Radiologist’s perspective on the management of bone metastatic disease. Eur J Surg Oncol. 2015;41(8):967–74.
He C, Tian Q, Wu CG, Gu Y, Wang T, Li M. Feasibility of percutaneous cementoplasty combined with interventional internal fixation for impending pathologic fracture of the proximal femur. J Vasc Interv Radiol. 2014;25(7):1112–7.
Abdel-Aal AK, Underwood ES, Saddekni S. Use of cryoablation and osteoplasty reinforced with Kirschner wires in the treatment of femoral metastasis. Cardiovasc Intervent Radiol. 2012;35(5):1211–5.
Anselmetti GC, Manca A, Chiara G, Tutton S, Iussich G, Gino G, Grignani G, Ortega C, Moselli N, Regge D. Painful pathologic fracture of the humerus: percutaneous osteoplasty with bone marrow nails under hybrid computed tomography and fluoroscopic guidance. J Vasc Interv Radiol. 2011;22(7):1031–4.
Kawai N, Sato M, Iwamoto T, Tanihata H, Minamiguti H, Nakata K. Percutaneous osteoplasty with use of a cement-filled catheter for a pathologic fracture of the humerus. J Vasc Interv Radiol. 2007;18(6):805–9.
Kelekis A, Filippiadis D, Anselmetti G, Brountzos E, Mavrogenis A, Papagelopoulos P, Kelekis N, Martin JB. Percutaneous augmented peripheral osteoplasty in long bones of oncologic patients for pain reduction and prevention of impeding pathologic fracture: the rebar concept. Cardiovasc Intervent Radiol. 2016;39(1):90–6.
Kelekis A, Martin JB, Anselmetti G, Filipiadis D. Regarding, “Percutaneous augmented peripheral osteoplasty in long bones of oncologic patients for pain reduction and prevention of impeding pathologic fracture: the rebar concept”: reply. Cardiovasc Intervent Radiol. 2016;39(3):479–80.
Steensma M, Healey JH. Trends in the surgical treatment of pathologic proximal femur fractures among Musculoskeletal Tumor Society members. Clin Orthop Relat Res. 2013;471(6):2000–6.
Nawathe S, Nguyen BP, Barzanian N, Akhlaghpour H, Bouxsein ML, Keaveny TM. Cortical and trabecular load sharing in the human femoral neck. J Biomech. 2015;48(5):816–22.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Roberto Luigi Cazzato, Guillaume Koch, Xavier Buy, Nitin Ramamurthy, Georgia Tsoumakidou, Jean Caudrelier, Vittorio Catena, Julien Garnon, Jean Palussiere and Afshin Gangi have no conflict of interest to disclose.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Cazzato, R.L., Koch, G., Buy, X. et al. Percutaneous Image-Guided Screw Fixation of Bone Lesions in Cancer Patients: Double-Centre Analysis of Outcomes including Local Evolution of the Treated Focus. Cardiovasc Intervent Radiol 39, 1455–1463 (2016). https://doi.org/10.1007/s00270-016-1389-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-016-1389-z