Abstract
Purpose
Percutaneous cementoplasty (PC) is rarely applied to long bone tumours, since cement is not considered to be sufficiently resistant to torsional forces. We reviewed the literature to understand the effects of percutaneous long bone cementoplasty (PLBC) in terms of analgesia, limb function and complications.
Materials and Methods
This study followed the Cochrane’s guidelines for systematic reviews of interventions. Inclusion criteria were (1) prospective/retrospective studies concerning PC; (2) cohort including at least ten patients; (3) at least one patient in the cohort undergoing PLBC; (5) published in English; (6) results not published by the same author more than once.
Results
One thousand five hundred and ninety-eight articles were screened and 13 matched the inclusion criteria covering 196 PLBC patients. Pain improvement was high in 68.2 % patients (σ = 0.2) and mild in 27.4 % (σ = 0.2). Functional improvement was high in 71.9 % patients (σ = 0.1) and mild in 6 % (σ = 0.1). Use of PLBC correlated with pain reduction (P < 0.001). Secondary fractures occurred in 16 cases (8 %, σ = 2.5); other complications in 2 % cases. Percutaneous stabilisation (PS) was coupled with PLBC in 17 % of cases without any subsequent fracture. PS was not associated with absence of secondary fracture (P = 0.08).
Conclusion
PLBC is safe, offering good pain relief and recovery of impaired limb function. Secondary fractures are uncommon and PS may reduce their occurrence. However, no evidence is currently available to support PS plus PLBC as compared to PLBC alone.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The role of interventional radiologists (IR) in the curative treatment of bone malignancies is limited to a few patients presenting with oligometastatic bone disease and relatively good life expectancy [1]. IR are more frequently involved in palliative therapies aimed at preventing or treating skeletal-related events (i.e. pain, fractures and syndromes related to spinal cord or nervous compression). Among the available techniques, percutaneous cementoplasty (PC) is widely used due to its analgesic and consolidative properties. Injection of poly-methyl-methacrylate (PMMA) into malignant bone lesions allows micro- and macro-fracture stabilisation [2] along with temperature-mediated destruction of nociception nervous terminals [3].
PMMA is considered resistant to compressive forces, but less to torsional forces [4–6]. As a result, PC has mainly been applied to consolidate spinal and acetabular bone lesions [7–9] where pure compressive forces are involved. PC on long bones which are more subject to torsional forces is limited to only a few series.
The aim of the present study was to systematically review the current evidence for PC in long bones (PLBC) for the palliative treatment of malignant lesions in terms of pain relief and recovery of limb function.
Materials and Methods
Literature Searches
Informed consent was not required for this retrospective literature review. This study was realised following the guidelines of the Cochrane Collaboration for systematic reviews of interventions [10]. We searched MEDLINE, MEDLINE in-process, EMBASE and the Cochrane databases between 1994 and 2014, basing our search on condition and intervention using the keywords “percutaneous cementoplasty”.
Selection Criteria
Studies matching the following criteria were selected:
-
(a)
Prospective/retrospective cohort study applying PC to treat primary or secondary bone tumours;
-
(b)
Cohort ≥10 patients;
-
(c)
≥1 patient in the cohort undergoing PC in the humerus, radius, ulna, femur, tibia or fibula;
-
(d)
Published in English;
-
(e)
Results not already published by the same author in a previous paper.
Two reviewing authors (blinded for review) assessed the studies for inclusion. All conflicts were resolved in consensus.
Data Collection
The full text of all papers included was reviewed, collecting specific study information in an electronic spreadsheet (Excel 2011; Microsoft, Seattle, WA, USA). The following data were extracted: study design; cohort size; number of PLBC patients; mean age; average follow-up; histology of the treated lesion; affected bone, epiphyseal, metaphyseal or diaphyseal involvement; radiological guidance used during PLBC; number of procedures performed under general anaesthesia (GA); mean lesion size; radiological aspect of the treated lesion (lytic or mixed); number and type of treatments preceding or following PLBC (radiotherapy, surgery, ablation); percutaneous stabilisation (PS) performed along with PLBC; technical success; questionnaires applied to assess pain and limb function improvements following PLBC; and rates of occurrence of pain and limb function improvements as well as secondary fractures and complications.
Complications were defined as adverse events requiring further intervention during follow-up. The event “secondary PMMA stress fracture” was categorised separately. PS was defined as any technique applied in addition to PLBC to increase bone strength.
All papers except one [11] deemed the procedure as technically successful when a sufficient amount of PMMA was injected. Cazzato et al. [11] specified that if no PMMA leaked from the pathological site, the procedure could be defined as successful.
With regard to pain assessment following PLBC, an improvement of at least three out of five points or five out of ten was labelled as “high”, depending on the scale used. Any other improvement was classified as “mild”. A patient was considered to show high functional improvement when they could resume a previously precluded activity (e.g. leaving the wheelchair). Any other improvement was ranked as “mild”. All available anaesthesia data were used to calculate rate of occurrence in the PLBC population, but many papers reported only a percentage on the entire cohort and not on the PLBC group.
Methodological Quality Assessment
The quality of studies was assessed with a modified version of the Newcastle–Ottawa scale (NOS, Table 1) [12] previously reported [13]. This system evaluates the quality of non-randomised studies included in a systematic review using a “star system” to judge three aspects of the study group (1) selection; (2) comparability; (3) ascertainment of either the exposure or outcome of interest for case–control or cohort studies, respectively. The lower the risk of bias, the higher the number of stars awarded to the manuscript. The best possible article was awarded six stars.
Statistical Analysis
Reduction of subjective pain after PLBC was tested using a two-sided t test with independent samples and assuming equal variances. In this analysis, we included only those articles in which a 10-point visual analogue scale (VAS) was adopted. Furthermore, we tested the correlation between PS and incidence of secondary fractures using the Fisher’s exact test.
All continuous variables are described with their mean value and standard deviation (σ). A P value <0.05 was considered significant for all tests.
Results
The first search query produced 1598 papers. After applying the inclusion criteria, we selected 14 full manuscripts [11, 14–26]. One article was excluded because no PLBCs were performed [21], so the final review included 13 articles (4 prospective, 9 retrospective) with an overall population of 382 patients (Table 2). Among these, 196 were PLBC patients (103 females, 80 males, 13 not specified) with a mean age of 57.9 years (σ = 7.8). Average follow-up was 6 months (σ = 4.5).
An overall high methodological quality was observed with a high mean number of stars (5.85/6) after methodological quality assessment with a subsequent very low risk of bias. Inclusion and exclusion criteria reported by each single paper are summarised in Table 3.
Malignancies originated from: breast (52 patients), bone marrow (44 patients), not specified (35 patients), lung (23 patients), kidney (16 patients), gut (9 patients), melanoma (5 patients), liver (3 patients), prostate (3 patients), thyroid (2 patients), bladder (1 patient), bone (1 patient), unknown (1 patient) and ovary (1 patient).
Six papers combined computed tomography (CT) and fluoroscopy to guide bone trocar insertion, four used fluoroscopy, two cone-beam CT (CBCT) and one exclusively CT.
Technical success was 100 % in 8 articles out of 13. A low (59 %) success rate was observed in one paper [11]. However, the criteria used to judge success did not include any kind of PMMA leakage at all, even including minimal amounts. The rates of technical success [16, 17, 24] were not reported in three papers. GA was used in 44 % of all procedures.
Overall, 223 lesions were treated. They were localised in the femur (64 %), humerus (17 %), tibia (10 %), fibula (3 %) and radius (1 %). The remaining 5 % were in unspecified locations. No ulnar lesions were found. Four papers [11, 17, 18, 24] gave further details about the tumour location inside the affected bone: 50 % (σ = 0.39) of the lesions were epiphyseal, 44 % (σ = 0.42) metaphyseal and 6 % (σ = 0.12) diaphyseal.
Mean lesion size was 45 mm [σ = 11, number of papers (n p) = 8]; 87 % lesions (σ = 0.27, n p = 6) were lytic, 2 % (σ = 0.04, n p = 6) mixed, and the rest were not specified. In 51 % (n p = 4) of the lesions there was evidence of cortical bone disruption.
Radiation therapy was performed before PLBC in 39 % cases (n p = 9, σ = 0.4); 7 % cases received the same treatment after PLBC (n p = 2, σ = 0.4). Percutaneous thermal ablation was combined with PLBC in 6 % of cases (n p = 4).
PS was performed in three papers, in 17 % procedures (n p = 3, σ = 0.5). Deschamps et al. [18] performed stabilisation of impending pathological fractures of the proximal femur by means of cannulated screws inserted percutaneously; Kim et al. [20] combined cementoplasty with flexible nails to stabilise bone lesions of the lower limbs; Sun et al. [25] used a biliary cement-filled catheter left inside the bone marrow cavity. The correlation test between PS and absence of secondary fractures did not provide statistical significance (P = 0.08).
Eleven papers (85 %) used the 10-point VAS to assess pain before and after PLBC and ten of them provided data suitable for statistical analysis: 68.2 % patients showed high improvement in the considered time intervals (n p = 10, σ = 0.2); 27.4 % reported mild improvement (σ = 0.2) and only 4.4 % experienced no change at all (σ = 0.1). The correlation test between PLBC and reduction of pain was extremely significant in this subgroup of 115 patients (P < 0.001, n p = 10, Fig. 1).
Seven papers (54 %) also evaluated functional improvement following PLBC. Three of them measured the return to ambulation, which was impossible before PLBC (n p = 3), one used the functional independence measure (FIM) scale, one used the activities of daily living (ADL) index, one a custom 4-point scale and the final paper used the Western Ontario and McMaster Universities Osteoarthritis Index. On average, 71.9 % patients (n p = 7, σ = 0.1) reported high improvement after PLBC, 6 % (σ = 0.1) mild and 22.1 % (σ = 0.1) presented negligible modifications. Rates of pain and functional improvement after PLBC are shown in Fig. 2.
Twelve papers studied the incidence of “secondary fractures” as an event (16 cases, 8 %, σ = 2.5, Table 4).
Five complications were reported across four papers (2 %): three heamatomas at the puncture site needing blood transfusions; one PMMA leakage into the elbow joint leading to a permanent reduction of the range of motion; and one abscess. Two PLBC cases obtained from the present authors’ personal experience are shown in Figs. 3 and 4.
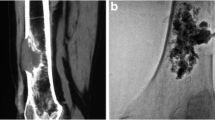
Case obtained from present authors’ personal experience: 42-year old female affected by breast cancer. A Axial CT scans of the pelvis shows a lytic lesion of the right femoral head (black arrow) thinning the anterior cortical bone and thus carrying a risk of pathological fracture. B, C, D The patient underwent CBCT-guided cementoplasty (white arrow) through one 11G × 10 cm bone trocar (Osteo-Site Bone Biopsy Needle, Cook Medical, Bloomington, IN, USA)
Case obtained from present authors’ personal experience: 84-year old female affected by cervical cancer. A Sagittal and B coronal CT scans of the lower limb shows a lytic lesion of the proximal tibia disrupting the posterior cortical bone (black arrow). C The patient underwent CBCT-guided cementoplasty through two 11G × 10 cm bone trocar (Osteo-Site Bone Biopsy Needle, Cook Medical, Bloomington, IN, USA) in order to obtain a D, E progressive, homogeneous and complete filling of the bone defect (white arrow). F Sagittal and G coronal CBCT scans obtained at the end of the procedure confirm the optimal filling of the bone defect
Discussion
Across 196 patients and 223 treated lesions, we found an overall 95.6 % rate of pain control within a mean follow-up of 6.3 months, confirming the efficacy of PMMA analgesia for the treatment of lytic or mixed bone tumours. Such an improvement was associated with the use of PLBC (P < 0.001). Although the exact mechanism by which PMMA assures fast and effective analgesia is not completely understood, it seems likely that micro- and macro-fracture stabilisation [2] as well as temperature-mediated destruction of nociception nervous terminals [3] are the phenomena most likely to be involved. Both mechanisms could also be responsible for the improvement in functional performance (overall 77.9 %). This result is relevant considering the high incidence of lesions we found in lower limbs (77 %) carrying a non-negligible risk of disability due to the risk of secondary fracture. Sixteen cases (8 %) presented a secondary fracture and 14 needed surgical treatment.
The Mirels’ score [27] is the most applied method for estimating the risk of fracture in long bones. According to this system, a score >9 categorises patients at high risk. Deschamps et al. [17] suggested another practical way to ascertain risky femoral lesions: cortical involvement >30 mm or a previous fracture of the lesser trochanter, both favour fractures. Accordingly, patients presenting with the aforementioned features should receive some form of bone stabilization. Ideally, these patients should be referred for surgical treatment, provided they have a good performance status and a relatively long life expectancy. Although surgical techniques are safe and effective [28], very few patients are suitable candidates due to the frequently advanced stage of disease [29]. Moreover, surgery carries substantial drawbacks in terms of hospitalisation duration (up to 19.3 days) and complications (8 %) including death (13.9 % at 3 months) [30]. Given these conditions, minimally invasive consolidative strategies are desirable to replace surgical approaches. On the whole, 32 patients received some kind of minimally invasive endomedullary stabilisation coupled with PLBC in the same session. Kim et al. applied flexible nails in 15 patients under spinal anaesthesia [20]. Sun et al. [25] used a 8-Fr PMMA-filled biliary drainage catheter in five patients under conscious sedation and Deschamps et al. [18] systematically performed percutaneous osteosynthesis by means of cannulated screws (8 mm) plus PC to prevent impending pathological fractures of the proximal femur in 12 patients, under different types of anaesthesia (GA or conscious sedation, local anaesthesia and continuous nerve block). Additionally, Deschamps et al. reported the mean procedural time (110 ± 43 min; range 60–180) and the mean duration of in-hospital stay (4 days) [18].
Despite the heterogeneity of PS techniques applied and the low proportion of patients receiving PS in addition to PLBC, no secondary fractures occurred in this sub-population (mean follow-up 9.9 months). These results were not statistically significant, with a borderline P value (P = 0.08), probably because the subgroup of patients analysed was very small. Compared to PLBC alone, PS plus PLBC is a more complex and longer procedure requiring experienced operators, presence of an anaesthesiologist and, if not performed under GA, good patient compliance. Furthermore, at the moment it is currently not possible to identify the best PS technique among those reported.
PLBC showed a low rate of complications and the possibility to be combined with other interventional and non-interventional (e.g. percutaneous ablation, radiotherapy) treatments. A large proportion of patients received radiation therapy at a certain point before PLBC. This was expected since radiation therapy is widely accepted as a first-line palliative treatment for bone metastases [31], even though up to one-third of patients do not experience a real benefit in terms of pain relief [32].
PLBC requires a 3D intra-procedural control, due to the anatomical configuration of extra-spinal lesions. Probably, for this reason CBCT or CT plus fluoroscopy were the most frequently applied techniques of guidance.
Few papers reported the exact tumour location within the affected bone. As a result, we were not able to evaluate any associations between lesion site and eventual complications. However, severe adverse effects (i.e. chondrolysis, permanent articular impairment) have been reported after intra-articular PMMA leakage [33].
Conclusion
PLBC is safe, offering good pain relief and recovery of impaired limb function. Secondary fractures are uncommon and they may be prevented by adding minimally invasive PS to PLBC. The combined strategy of PS + PLBC may be best proposed to active patients presenting with high risk lesions (e.g. Mirels’ score >9, cortical involvement >30 mm and/or previous fractures of the lesser trochanter). However, there is a substantial lack of evidence supporting the superiority of PS plus PLCB in comparison to PLBC alone. Therefore, further studies comparing both strategies are desirable.
References
Gangi A, Tsoumakidou G, Buy X, Quoix E (2010) Quality improvement guidelines for bone tumour management. Cardiovasc Intervent Radiol 33(4):706–713
Dean JR, Ison KT, Gishen P (2000) The strengthening effect of percutaneous vertebroplasty. Clin Radiol 55(6):471–476
Deramond H, Wright NT, Belkoff SM (1999) Temperature elevation caused by bone cement polymerization during vertebroplasty. Bone 25(Suppl 2):17S–21S
Gangi A, Buy X (2010) Percutaneous bone tumor management. Semin Intervent Radiol 27(2):124–136
Heini PF, Franz T, Fankhauser C, Gasser B, Ganz R (2004) Femoroplasty-augmentation of mechanical properties in the osteoporotic proximal femur: a biomechanical investigation of PMMA reinforcement in cadaver bones. Clin Biomech 19(5):506–512
Sutter EG, Mears SC, Belkoff SM (2010) A biomechanical evaluation of femoroplasty under simulated fall conditions. J Orthop Trauma 24(2):95–99
Weill A, Chiras J, Simon JM, Rose M, Sola-Martinez T, Enkaoua E (1996) Spinal metastases: indications for and results of percutaneous injection of acrylic surgical cement. Radiology 199(1):241–247
Weill A, Kobaiter H, Chiras J (1998) Acetabulum malignancies: technique and impact on pain of percutaneous injection of acrylic surgical cement. Eur Radiol 8(1):123–129
Gupta AC, Hirsch JA, Chaudhry ZA, Chandra RV, Pulli B, Galinsky JG et al (2012) Evaluating the safety and effectiveness of percutaneous acetabuloplasty. J Neurointerv Surg 4(2):134–138
Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated 2011). In: Cochrane Collab. www.cochrane-handbook.org. Accessed Jan 2015
Cazzato RL, Buy X, Eker O, Fabre T, Palussiere J (2014) Percutaneous long bone cementoplasty of the limbs : experience with fifty-one non-surgical patients. Eur Radiol 24(12):3059–3068
Wells G, Shea B, O’connell D, Peterson J. The Newcastle–Ottawa Scale (NOS) for assessing the quality of non-randomised studies in meta-analyses. http://www.medicine.mcgill.ca/rtamblyn/Readings/The Newcastle-scale for assessing the quality of nonrandomised studies in meta-analyses.pdf. Accessed 8 May 2014
Lanza E, Thouvenin Y, Viala P, Sconfienza LM, Poretti D, Cornalba G et al (2014) Osteoid osteoma treated by percutaneous thermal ablation: when do we fail? A systematic review and guidelines for future reporting. Cardiovasc Intervent Radiol 37(6):1530–1539
Anselmetti GC, Manca A, Ortega C, Grignani G, Debernardi F, Regge D (2008) Treatment of extraspinal painful bone metastases with percutaneous cementoplasty: a prospective study of 50 patients. Cardiovasc Intervent Radiol 31(6):1165–1173
Basile A, Giuliano G, Scuderi V, Motta S, Crisafi R, Coppolino F et al (2008) Cementoplasty in the management of painful extraspinal bone metastases: our experience. Radiol Med 113(7):1018–1028
Botton E, Edeline J, Rolland Y, Vauléon E, Le Roux C, Mesbah H et al (2012) Cementoplasty for painful bone metastases: a series of 42 cases. Med Oncol 29(2):1378–1383
Deschamps F, Farouil G, Hakime A, Barah A, Guiu B, Teriitehau C et al (2012) Cementoplasty of metastases of the proximal femur: is it a safe palliative option? J Vasc Interv Radiol 23(10):1311–1316
Deschamps F, Farouil G, Hakime A, Teriitehau C, Barah A, de Baere T (2012) Percutaneous stabilization of impending pathological fracture of the proximal femur. Cardiovasc Intervent Radiol 35(6):1428–1432
Iannessi A, Amoretti N, Marcy P-Y, Sedat J (2012) Percutaneous cementoplasty for the treatment of extraspinal painful bone lesion, a prospective study. Diagn Interv Imaging 93(11):859–870
Kim YI, Kang HG, Kim TS, Kim SK, Kim JH, Kim HS (2014) Palliative percutaneous stabilization of lower extremity for bone metastasis using flexible nails and bone cement. Surg Oncol 23(4):192–198
Kodama H, Aikata H, Uka K, Takaki S, Mori N, Waki K et al (2007) Efficacy of percutaneous cementoplasty for bone metastasis from hepatocellular carcinoma. Oncology 72(5–6):285–292
Lane MD, Le HBQ, Lee S, Young C, Heran MKS, Badii M et al (2011) Combination radiofrequency ablation and cementoplasty for palliative treatment of painful neoplastic bone metastasis: experience with 53 treated lesions in 36 patients. Skeletal Radiol 40(1):25–32
Masala S, Volpi T, Fucci FPM, Cantonetti M, Postorino M, Simonetti G (2011) Percutaneus osteoplasty in the treatment of extraspinal painful multiple myeloma lesions. Support Care Cancer 19(7):957–962
Plancarte-Sanchez R, Guajardo-Rosas J, Cerezo-Camacho O, Chejne-Gomez F, Gomez-Garcia F, Meneses-Garcia A et al (2013) Femoroplasty: a new option for femur metastasis. Pain Pract 13(5):409–415
Sun G, Jin P, Liu XW, Li M, Li L (2014) Cementoplasty for managing painful bone metastases outside the spine. Eur Radiol 24(3):731–737
Toyota N, Naito A, Kakizawa H, Hieda M, Hirai N, Tachikake T et al (2005) Radiofrequency ablation therapy combined with cementoplasty for painful bone metastases: initial experience. Cardiovasc Intervent Radiol 28(5):578–583
Mirels H (1989) Metastatic disease in long bones. A proposed scoring system for diagnosing impending pathologic fractures. Clin Orthop Relat Res 249:256–264
Dijstra S, Wiggers T, van Geel BN, Boxma H (1994) Impending and actual pathological fractures in patients with bone metastases of the long bones. A retrospective study of 233 surgically treated fractures. Eur J Surg 160(10):535–542
Wedin R (2001) Surgical treatment for pathologic fracture. Acta Orthop Scand Suppl 72(302):1–29
Ristevski B, Jenkinson RJ, Stephen DJG, Finkelstein J, Schemitsch EH, McKee MD et al (2009) Mortality and complications following stabilization of femoral metastatic lesions: a population-based study of regional variation and outcome. Can J Surg 52(4):302–308
Steenland E, Leer JW, van Houwelingen H, Post WJ, van den Hout WB, Kievit J et al (1999) The effect of a single fraction compared to multiple fractions on painful bone metastases: a global analysis of the Dutch Bone Metastasis Study. Radiother Oncol 52(2):101–109
Chow E, Harris K, Fan G, Tsao M, Sze WM (2007) Palliative radiotherapy trials for bone metastases: a systematic review. J Clin Oncol 25(11):1423–1436
Leclair A, Gangi A, Lacaze F, Javier RM, Bonidan O, Kempf JF et al (2000) Rapid chondrolysis after an intra-articular leak of bone cement in treatment of a benign acetabular subchondral cyst: an unusual complication of percutaneous injection of acrylic cement. Skeletal Radiol 29(5):275–278
Acknowledgments
All the authors would like to thank Pippa McKelvie-Sebileau from Institut Bergonie, Bordeaux, France, for medical editorial advice in English.
Conflict of interest
Roberto Luigi Cazzato, Jean Palussière, Xavier Buy, Vincenzo Denaro, Daniele Santini, GiuseppeTonini, Rosario Francesco Grasso, Bruno Beomonte Zobel, Dario Poretti, Vittorio Pedicini, Luca Balzarini and Ezio Lanza declare they have no conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cazzato, R.L., Palussière, J., Buy, X. et al. Percutaneous Long Bone Cementoplasty for Palliation of Malignant Lesions of the Limbs: A Systematic Review. Cardiovasc Intervent Radiol 38, 1563–1572 (2015). https://doi.org/10.1007/s00270-015-1082-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-015-1082-7