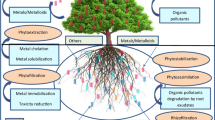
Abstract
Lead (Pb) is a heavy metal used in various industrial processes, so its levels are considerably increased in the soil, sediments, surface water, and groundwater. Pb is a non-biodegradable and persistent environmental pollutant that causes toxicity in humans, plants, animals, and microorganisms. Phytoremediation is a clean, eco-friendly, and cost-effective technology to remove Pb from aquatic and terrestrial environments. This technology uses plants to remove, immobilize, and contain Pb through phytoextraction, phytostabilization, and rhizofiltration. This chapter describes the characteristics of plants used in phytoremediation, focusing on the mechanisms employed by the plants to assist in the removal or immobilization of Pb. Moreover, it shows the state of the art on phytoremediation assisted by microorganisms for enhancing phytoremediation of Pb-polluted soils.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Lead (Pb) is a soft, malleable, bluish-gray metal located in group IV of the periodic table of elements (Al-Fartusie and Mohssan 2017). Pb occurs naturally in the soil at a concentration from 0.002 to 0.2 mg/kg, while in fresh waters, from 0.001 to 0.010 mg/L, worldwide (Carrillo-Chávez et al. 2006). The presence of Pb in the environment can be from natural or anthropogenic sources (Li et al. 2012). The primary natural sources of Pb are weathering and erosion of lead-rich rocks, forest fires, and volcanic eruptions (Zhang et al. 2019). While anthropogenic Pb results from mining, smelting, leaded gasoline combustion, coal burning, and industrial production of pigments, lead-acid batteries, cable sheathing, ammunition, alloys, solder, and pipes (Ballantyne et al. 2018; Eichler et al. 2015). Both natural and anthropogenic Pb sources have caused a significant increase in their levels in the environment (Zhang et al. 2019). For example, high Pb concentration has been detected in arable lands of Ireland (Nag and Cummins 2022), urban agricultural soils of Cameroon (Aboubakar et al. 2021), and in the arable soil in Southwest China (Wu et al. 2018).
Pb is a non-biodegradable and persistent pollutant that exerts toxic effects on plants, animals, and microorganisms at low concentrations (Wong and Li 2004; Abdelkrim et al. 2020; Rahman and Singh 2019). Pb ranks second on the list of hazardous substances according to the U.S. Agency for Toxic Substances and Disease Registry (ATSDR 2022). In humans, Pb causes haematological and cardiovascular disorders, kidney dysfunction, gastrointestinal disease, and central nervous system damage (Rahimpoor et al. 2020; Khanam et al. 2020; Rahman and Singh 2019). Lead also affects brain development in children, causing behavioural changes and lowering IQ score (Heidari et al. 2022).
Different physical and chemical methods have been developed to remove Pb from contaminated sites. These methods include soil washing, landfilling, vitrification, electrokinetic treatments, surface capping, encapsulation, and soil flushing (Liu et al. 2018; Song et al. 2017). Besides, bioremediation has been proposed as an eco-sustainable alternative for removing natural and anthropogenic Pb from soils, sediments, surface water, and groundwater (Bala et al. 2022). In this technology, living organisms such as bacteria, fungi, microalgae, or plants are used to degrade or transform environmental contaminants into non-toxic forms (Vidali 2001). According to the application site, bioremediation techniques have been classified as in situ or ex situ (He et al. 2021). In situ techniques are carried out directly at the contaminated site, while ex situ techniques are applied outside the contaminated site (Boopathy 2000).
2 Phytoremediation: Definition and Strategies
Phytoremediation is an in situ bioremediation technology that uses plants to degrade, immobilize, neutralize, and contain environmental contaminants (Wang et al. 2017). Phytoremediation has been successfully applied to clean up heavy metals, radionuclides, petroleum hydrocarbons, explosives, pesticides, pharmaceutical and personal care products (PPCPs) from aquatic and terrestrial environments (Jee 2016; Kurade et al. 2021). Plants perform phytoextraction, phytovolatilization, phytostabilization, rhizofiltration, phytodegradation, and phytostimulation to remove xenobiotics (Alsafran et al. 2022).
Phytoextraction or phytoaccumulation is used to remove heavy metals and other inorganic compounds from soil and sediment to the aerial parts of plants. In this process occurs the absorption of contaminants by the plant roots, translocation through stems, and their accumulation in shoots and leaves (Mahar et al. 2016; Etim 2012).
Phytostabilization or phytoimmobilization is a strategy in which a plant species reduces the mobility of pollutants and decreases its bioavailability for other plants or microorganisms (Alsafran et al. 2022). In this strategy, heavy metals and other inorganic compounds are adsorbed on root cell walls, absorbed within root tissues, or immobilized as non-toxic forms in the rhizosphere through mechanisms including sorption, precipitation, complexation, or metal valence reduction (Rai et al. 2021; Etim 2012; Yan et al. 2020).
Rhizofiltration is a hydroponic-based phytoremediation technique that uses plant roots to eliminate pollutants from the impacted aquatic environments (Srivastava et al. 2021). In this strategy, heavy metals and other inorganic compounds are adsorbed or precipitated on the root surface or absorbed in the roots (Kristanti et al. 2021).
Phytovolatilization is a strategy in which plants uptake environmental contaminants, transform them into volatile forms, and release them into the atmosphere through transpiration (Etim 2012). This process is applied for the treatment of some metals and metalloids such as arsenic (As), selenium (Se), and mercury (Hg) (Rai et al. 2021). This strategy is controversial because the pollutants are not destroyed but transferred to the atmosphere, where they can be redeposited (Mahar et al. 2016).
Phytodegradation is when plants uptake, store, metabolize or mineralize organic contaminants in their tissues (Rai et al. 2021). The phytodegradation process requires degrading enzymes involved in various metabolic processes and enzymes such as nitrilase, nitroreductase, peroxidase, dehalogenase, oxygenase, and laccases (Chatterjee et al. 2013).
Phytostimulation is a strategy in which plants and microorganisms localized in the rhizosphere degrade organic contaminants (Rai et al. 2021). In this strategy, plants secrete root exudates and metabolites that stimulate the growth of degrading microorganisms (Favas et al. 2014).
3 Phytoremediation of Lead
Rhizofiltration, phytostabilization, and phytoextraction are the main strategies for Pb removal from polluted environments. Phytostabilization and phytoextraction are applicable for the remediation of Pb in soils and sediments, while rhizofiltration is used for the remediation of surface water, groundwater, and wastewater (Yan et al. 2020; Otte and Jacob 2006).
3.1 Phytostabilization of Lead
Phytostabilization reduces the Pb migration from contaminated to non-contaminated soils (Alsafran et al. 2022; Bolan et al. 2011). In this process, the immobilization of Pb can occur by either adsorption on root cell walls or Pb precipitation in the rhizosphere (Ashraf et al. 2015; Arshad et al. 2016).
The bioavailability of Pb in the soil depends on its speciation, which is influenced by various factors such as pH, redox potential, organic matter, sulphur, and carbonate contents (Olaniran et al. 2013; John and Leventhal 1995). Besides, plant roots play an essential role in the metal and nutrient solubility in the soil (Wenzel et al. 1999). The metabolic activity of the plant roots can change the pH, the redox conditions, concentrations of Dissolved Organic Matter (DOM), and microbial activity in the rhizosphere, which enhance the uptake of nutrients such as iron (Fe), phosphorus (P), and zinc (Zn) or immobilize non-essential metals (Li et al. 2021; Seshadri et al. 2015; Rai et al. 2021). For example, Pelargonium × hortorum L.H. Bailey increased the DOM content and acidified the rhizosphere soil in response to Pb (Manzoor et al. 2020). Root exudation is one of the most critical factors affecting the physicochemical characteristics of the soil (Li et al. 2011b). The root exudates are a mixture of metabolites, including sugars, amino acids, and organic acids, produced by plants and secreted to the soil (Vives-Peris et al. 2020). These compounds can affect the bioavailability of Pb in the rhizosphere (Li et al. 2021; Seshadri et al. 2015). For example, the waterlogged (Oenanthe javanica DC.) and yellow melon (Cucumis melo L.) roots release citric acid and others organic acids to the rhizosphere which form soluble Pb–organic complexes (Liu and Luo 2019; Irias Zelaya et al. 2020). Similarly, organic acids secreted by pea plants (Pisum sativumpea L.) favour the formation of stable metal complexes in the root region (Austruy et al. 2014).
Pb precipitates have also been observed in the root of Pelargonium cultivars, Indian mustard (Brassica juncea), and two poplar species (Arshad et al. 2016; Yang et al. 2021; Shi et al. 2021). In Pelargonium cultivars, Pb precipitates on the root surface in the form of αPbO, PbOH+, carbonates, and ferrite derivatives (Arshad et al. 2016). On the other hand, in the Indian mustard root cells, Pb precipitates as lead phosphate Pb3(PO4)2, pyromorphite Pb5(PO4)3(OH, Cl), and other Pb phosphates (Yang et al. 2021). Similarly, Populus × canescens and P. nigra precipitate the Pb as phosphates and oxalates in their roots (Shi et al. 2021).
Pb immobilization in the root can be due to the interaction between the heavy metal ions and the components of the root cell wall (Krzesłowska 2011). The cell wall is the first structure of root cells to come in contact with heavy metals and is involved in ions metal binding (Parrotta et al. 2015). The cell wall’s main components are polysaccharide such as cellulose, hemicellulose (HC), and pectin which play an important role in Pb binding in cell walls (Zhang et al. 2021a; Sumranwanich et al. 2018). Pb adsorption by different cell wall components has been reported previously in tea (Camellia sinensis L.) and Athyrium wardii (Hook.) roots. In the cell wall of tea plant roots, the most significant amount of Pb is adsorbed mainly by cellulose and lignin (68.42%), followed by pectin (20%) and HC2 (5.26%) (Wang et al. 2015). On the other hand, pectin and HC are the primary binding sites for Pb in root cell walls of A. wardii (Zhan et al. 2020). The Pb-binding capacity of cell wall polysaccharides is attributed mainly to the presence of carboxyl (–COOH) and hydroxyls (–OH) groups (Sumranwanich et al. 2018; Wang et al. 2015; Zhan et al. 2020).
Previous studies have reported that Pb increases the biosynthesis of polysaccharides in some plant species’ root cell walls. For instance, Pb induced pectin and hemicellulose production in root cell walls of tall fescue (Festuca arundinacea Schreb) (Zhang et al. 2020), A. wardii (Zhan et al. 2020), Populus × canescens, and P. nigra (Shi et al. 2021), which may be a mechanism of tolerance to Pb stress of these plants.
In the phytostabilization process, the plant roots take up Pb ions or Pb-soluble complexes from the rhizosphere and accumulate them internally (Yan et al. 2020). The Pb within root tissue can be associated with the cell wall in apoplastic space or immobilized intracellularly in vacuoles, limiting Pb translocation from roots to shoots (Yan et al. 2020; Rahman et al. 2022; Wierzbicka 1998).
In signal grass (Brachiaria decumbens), Indian mustard (B. juncea), and Neyraudia reynaudiana it have been observed that most Pb precipitates in the cell wall as insoluble deposits inside the roots (Kopittke et al. 2008; Zhou et al. 2016; Yang et al. 2021). Pb mainly exists as lead phosphate precipitates [Pb5(PO4)3(OH, Cl), and Pb3(PO4)2] in the Indian mustard roots cells (Yang et al. 2021). Insoluble deposits of chloropyromorphite [Pb5(PO4)3Cl] in root cells have also been observed in signal grass (B. decumbens) roots (Kopittke et al. 2008).
Vacuolar sequestration of Pb in radicular cells limits its translocation within plants (Sharma and Dubey 2005). Vacuoles are the largest organelle of plant cells and play an essential function in the heavy metal detoxification (Sharma et al. 2016). In this organelle, intracellular Pb is stored by complexation with organic acids and sulfur-rich peptides known as phytochelatins (Zhang et al. 2018; Singh et al. 2017; Zhao et al. 2015). In addition to cell walls, the vacuoles are one of the main storage sites of Pb in Allium sativum and N. reynaudiana roots (Jiang and Liu 2010; Zhou et al. 2016). Approximately, 31.2–41.3% of total Pb is stored in the vacuoles of roots A. wardii (Hook.) (Zhao et al. 2015).
3.2 Phytoextraction of Lead
Phytoextraction is a method used to reduce Pb levels in soil and sediments. This method requires Pb uptake by plant roots, root-to-shoot translocation, and intracellular compartmentalization of Pb in aerial tissues (Yan et al. 2020). These processes are dependent on plant species, variety, genotype, environmental conditions, and Pb bioavailability in the soil (Asare et al. 2023).
The first step in Pb accumulation is the Pb uptake by the root (Gong et al. 2022). The Pb ions from the soil are absorbed by the root epidermal cells and can be transported inside the root by apoplastic or symplastic pathways (He et al. 2023; Zhou et al. 2018). In the apoplastic pathway, Pb in the extracellular fluid is transferred from one cell wall to another, whereas in the symplastic pathway, the Pb ions cross the plasma membrane and transfer cell to cell through channels called plasmodesmata (Pasricha et al. 2021). As Pb is not an essential element, plants do not have a specific channel for Pb uptake, so it has been suggested that Pb enter the plant cells via channels or transporters for other essential cations (Peralta-Videa et al. 2009; Gong et al. 2022). Different proteins have been associated with Pb transport across the membrane, such as AtCNGC1 (cyclic nucleotide-gated channel 1), NtCBP4 (Plasma membrane Calmodulin-Binding Protein 4), and OsNRAMP5 (Natural Resistance-Associated Macrophage Proteins 5) (Arazi et al. 1999; Sunkar et al. 2000; Chang et al. 2022). In tobacco (Nicotiana tabacum) and Arabidopsis thaliana, two cation channels for K+ and Ca2+ called NtCBP4 and AtCNGC1, respectively, have been associated with Pb uptake across the plant plasma membrane and Pb accumulation (Arazi et al. 1999; Sunkar et al. 2000). On the other hand, the OsNRAMP5, a divalent metal transporter, is associated with transporting intracellular Pb in rice (Oryza sativa) roots (Chang et al. 2022). Once inside the root cells, Pb is associated with amino acids and organic acids and can be translocated to shoots and leaves by the xylem (Gall and Rajakaruna 2013; Pourrut et al. 2013).
At the shoot level, intracellular Pb is detoxified by metal-binding ligands such as phytochelatins and metallothioneins (Mitra et al. 2014; Pourrut et al. 2011; Eapen and D’Souza 2005). Phytochelatins (PC) are oligopeptides that contain glutamic acid (Glu), cysteine (Cys), and glycine (Gly) [(γ–Glu–Cys)n–Gly (n = 2 − 11)], whose synthesis is catalyzed by phytochelatin synthase (PCS) from glutathione (Scarano and Morelli 2002; Gupta et al. 2013b). While metallothionein (MT) are low-molecular-weight proteins (7–10 kDa) with 9–16 cysteine residues that are encoded by a family of MT genes (Eapen and D’Souza 2005; Cobbett and Goldsbrough 2002). Previous studies have reported that the plants synthesize PC and MT in response to Pb stress. For example, it has been observed that Pb exposure induces the synthesis of PC in Salvinia minima Baker, Dwarf bamboo (Sasa argenteostriata), and coontail (Ceratophyllum demersum L.) (Jiang et al. 2020; Estrella-Gómez et al. 2009; Mishra et al. 2006), and increases the expression of MT genes in tomato (Lycopersicon esculentum), Bruguiera gymnorrhiza, and rice (O. sativa) plants (Kim and Kang 2018; Kisa et al. 2017; Huang and Wang 2009).
In the cytoplasm of shoot cells, PC and MT binding to intracellular Pb and form stable complexes. The PC–Pb complex is finally transported into the vacuole, where it is stored (Andra et al. 2009; Inouhe et al. 2012).
3.3 Rhizofiltration of Lead
Rhizofiltration is a technique used to remove Pb from surface water, groundwater, and effluents with low levels of contaminants (Ekta and Modi 2018; Jadia and Fulekar 2009). Similarly, to the phytostabilization process, in the rhizofiltration, the Pb ions can be absorbed within root tissues (Kristanti et al. 2021; Rawat et al. 2012), adsorbed by root cell walls (Ho et al. 2021), or immobilized in the root surface (Delgado-González et al. 2021; Dushenkov et al. 1995).
In aquatic and wetland plants, iron plaque plays an essential role in the sequestration of heavy metals in the roots (Tripathi et al. 2014). The Fe plaques are deposits of different iron oxides and hydroxides on the root surface (Tripathi et al. 2014; Khan et al. 2016). The presence of ferrihydrite [Fe4–5(OH,O)12], lepidocrocite [γ–FeOOH], siderite [FeCO3], and goethite [FeO(OH)] has been observed in the Fe plaques of Oenanthe javanica, Phalaris arundinacea, and Vallisneria americana (Liu and Luo 2019; Hansel et al. 2001; St-Cyr et al. 1993). These Fe (hydr)oxides are result of oxidation of ferrous iron (Fe2+) in the rhizosphere by the oxygen loss from the roots, and the biological activity of microorganisms (Tripathi et al. 2014; Khan et al. 2016). Previous studies have reported that iron plaque can sequester Pb on the root surface (Zandi et al. 2022). In rice (O. sativa), the most significant amount of plant taken Pb (> 60%) is stored in the iron plaque of the root (Cheng et al. 2014; Ma et al. 2013). Similarly, most of the total Pb uptake (50–60%) performed by Phalaris arundinacea L. and Carex cinerascens Kukenth. plants was found in the iron plaque, and only small amounts was found in roots and shoots (Liu et al. 2015, 2016). The Pb–binding capacity of Fe plaques is attributed mainly to the Pb’s specific and high affinity for iron (hydr)oxides (Hansel et al. 2001).
3.4 Potential Plants for Phytoremediation of Pb
The concentration of heavy metals in plants determines the success of phytoremediation; therefore, selecting suitable plant species is crucial in phytoextraction, phytostabilization, and rhizofiltration efficiency (Yan et al. 2020; Gupta et al. 2013a).
Hyperaccumulator plants can potentially remove Pb from the soil through phytoextraction (Lone et al. 2008). Pb hyperaccumulators are plants able to grow in contaminated soils with heavy metals and accumulate more than 1000 mg/kg of Pb in aerial organs without show phytotoxicity signs (Sytar et al. 2021; Manara et al. 2020). However, Pb hyperaccumulation in plants is uncommon because Pb ions are easily precipitated in the rhizosphere, limiting their uptake by roots and the translocation to shoots (Baker and Brooks 1989). In the Global Hyperaccumulator Database (http://hyperaccumulators.smi.uq.edu.au/collection/), Alyssum wulfenianum, Noccaeae rotundifolium subsp. cepaeifolium, Polycarpaea synandra, Sesbania drummondii, Armeria maritima var. Halleri, Dactyloctenium aegyptium, Microstegium ciliatum, Polygala umbonata, and Spermacoce mauritiana plants, belonging to seven families, have been identified as Pb hyperaccumulators (Reeves et al. 2018). Although these plants accumulate high concentrations of Pb, hyperaccumulator plants are small and present slow growth, which limits their use in the phytoremediation process (Saifullah et al. 2009; Yan et al. 2020).
Different fast-growing crops with high biomass production, like sorghum (Sorghum bicolor L.), sunflower (Helianthus annuus L.), and corn (Zea mays) have been studied to remove Pb from lead contaminated soil under field conditions (Cheng et al. 2015; Zehra et al. 2020; Yuan et al. 2019). Despite the lower concentrations of Pb in their tissues, the total metal remotion exerted by these plants can be like levels reached by hyperaccumulator plants (Van Slycken et al. 2008).
Native plants grown on heavy metal contaminated sites are another option for Pb remediation. These plant species can survive, grow, and reproduce under metal stress better than plants introduced from other environments (Midhat et al. 2019; Yoon et al. 2006). Several studies have evaluated the phytoremediation potential of native plants growing in heavy metal-contaminated sites (Table 7.1). For example, Salazar and Pignata (2014) studied the vegetal community growing around a lead smelter plant in Argentina. On the other hand, Mahdavian et al. (2017) and Nouri et al. (2011) investigated plants colonizing a lead–zinc mining area in Iran. In Marocco, Midhat et al. (2019) and Hasnaoui et al. (2020) identified metal-tolerant native plant species from three abandoned mining sites and a contaminated site near a Pb/Zn mining area, respectively. In these sites, some plants belonging to the Asparagaceae, Asteraceae, Brassicaceae, Cucurbitaceae, Cyperaceae, Euphorbiaceae, Fabaceae, Gramineae, Lamiaceae, Liliaceae, Resedaceae, and Tamaricaceae families have been observed (Table 7.1).
The phytoremediation potential of plants can be estimated using Bioconcentration Factors (BCF) and Translocation Factors (TF) (Rolón-Cárdenas et al. 2022). The BCF is the ratio between the heavy metal content in the plant roots and the substrate (Zou et al. 2012; Lorestani et al. 2011). Various native plant species like Artemisia sieberi Besser, Fortuynia bungei Boiss., Astragalus durandianus Aitch. & Baker, Mentha longifolia L., and Allium umbilicatum Boiss. have showed BCF > 1 for Pb (Table 7.1), indicating the potential of these plants to be used in Pb phytostabilization (Lorestani et al. 2013).
On the other hand, TF is the ratio of heavy metal content in the shoots and the roots (Midhat et al. 2019; Lorestani et al. 2011). Values TF > 1 for Pb has been reported in different native plants such as Lactuca viminea (L.) J. Presl & C. Presl, Scariola orientalis (Boiss.) Sojak, Scolymus hispanicus, Cyperus iria, Juncellus serotinus, Euphorbia macroclada Boiss., Echinophora platyloba DC., Paspalum paspaloides, Phragmites australis, Reseda alba, and Tamarix ramosissima Ledeb. (Table 7.1). This TF value indicates high efficiency in Pb translocation from the roots to the shoots and, therefore, their potential to be used in phytoextraction (Midhat et al. 2019).
Rhizofiltration uses heavy metal tolerant plants with a fibrous root system and large surface areas to ‘filter’ Pb ions in solution (Chatterjee et al. 2013; Nedjimi 2021). Different aquatic plants have been studied to remove Pb from water through rhizofiltration (Kafle et al. 2022). For example, Alternanthera sessilis, Enhydra fluctuans, Pistia stratiotes, Salvinia cucullata, Typha latifolia, and Vetiveria zizanioides can remove between 84 and 99% of Pb from solution and accumulate into root and shoot (Das et al. 2021; Parven et al. 2022; Veselý et al. 2011; Alonso-Castro et al. 2009; Singh et al. 2015). Some terrestrial plants such as Indian mustard (B. juncea), Cosmos sulphureus Cav., sunflower (H. annuus L.), Iris lactea var. chinensis, and Talinum paniculatum are also suitable for rhizofiltration due to they remove high amount of Pb from the hydroponic medium and accumulate it in roots and shoots (Liu et al. 2000; Aftab et al. 2021; Seth et al. 2011; Han et al. 2008; dos Reis et al. 2022).
The plants used for rhizofiltration are first cultivated in hydroponic conditions to favour the root system development, later transferred to the contaminated water source, and finally harvested when the root of plants are saturated with contaminants (Mansoor et al. 2022; Yan et al. 2020).
4 Microbial-Assisted Pb Phytoremediation
Soil microorganisms fulfill essential ecosystem processes since they regulate biogeochemical cycles and decompose organic matter to maintain soil fertility (Basu et al. 2021). Plants establish associations with different types of soil microorganisms like bacteria and fungi which contribute to the host adaptation to environmental conditions (Gan et al. 2017; Narula et al. 2012). The rhizosphere is the zone of the soil around the plants’ roots where occurs intense biological activity during the plant-soil-microorganism interactions (More et al. 2019; Pathan et al. 2020).
Rhizobacteria and epiphytic bacteria are a broad group of soil bacteria that colonize the area around the roots, and the root surface, respectively (Taulé et al. 2021). While the endophytic bacteria colonize the internal plant tissues without causing adverse effects on their host plants (Ma et al. 2011).
Plant-associated microorganisms play an essential role in the metal phytoremediation process. These microorganisms can promote plant growth, reduce metal phytotoxicity, modify metal uptake and accumulation in the plant, and increase metal bioavailability in soil or water (Ma et al. 2016; Rajkumar et al. 2012). For example, two rhizospheric bacteria identified as Bacillus proteolyticus and B. licheniformis, increased the biomass of Solanum nigrum plants growing in heavy metal-contaminated soil, and the total Pb content in roots and shoots (He et al. 2020). Under axenic conditions, a rhizospheric arsenic-resistant bacteria also increased Pb concentration in the root of Pteris vittata (Manzoor et al. 2019). An endophytic microbial consortium isolated from three native plants increased Pb accumulation in roots and shoots of B. juncea, and Pb concentration in sunflower (H. annuus) roots (Pietrini et al. 2021).
In recent years, it has been demonstrated the potential of in situ plant-bacteria interaction for promote plant growth under Pb stress and Pb removal from water and contaminated sites. In an agricultural field contaminated with Pb and Cd, a consortium of four heavy metals resistant bacteria (Rhizobium leguminosarum, Bacillus simplex, Luteibacter sp. and, Variovorax sp.) increased plant length, dry biomass, nodule number of Lathyrus sativus plants, and enhance Pb accumulation in roots in comparison with uninoculated plants (Abdelkrim et al. 2020). Endophyte bacteria Pseudomonas putida RE02 reduced the mortality percentage of Trifolium repens seedlings under metal stress and improved Pb uptake by T. repens plants grown in heavy metal contaminated tailings (Liu et al. 2021).
In a constructed wetland, a consortium of five rhizobacteria (Bacillus cereus, B. pumilus, B. subtilis, Brevibacillus choshinensis, and Rhodococcus rhodochrous) increased Pb sorption by Scirpus grossus plants from contaminated water (Tangahu et al. 2022).
Bacterial communities can improve growth and tolerance to metal stress in host plants by producing phytohormones and enzyme 1–aminocyclopropane–1–carboxylate (ACC) deaminase which reduces ethylene production (Kong and Glick 2017; Sharma 2021). These bacteria also promote plant growth and favour nutritional status by improving the absorption of water and nutritive elements such as nitrogen (N), phosphorus (P), and iron (Fe) through mechanisms like nitrogen fixation, P-solubilization, and siderophores production (Ma et al. 2011; Etesami 2018; Manoj et al. 2020).
Like bacteria, fungi have also been evaluated to increase phytoremediation efficiency. Arbuscular Mycorrhizae (AM) are fungal endophytes that colonize the internal root tissues of higher plants (Deng and Cao 2017; Gaur and Adholeya 2004). AM fungi have also been shown to promote plant growth under Pb stress and increase Pb accumulation in plants. For example, Funneliformis mosseae, Claroideoglomus etunicatum, and Rhizophagus intraradices, promote the growth of the soybean (Glycine max L.) exposed to 100 and 300 mg/kg Pb, and increase Pb accumulation in the roots compared to non-inoculated plants (Adeyemi et al. 2021). F. mosseae inoculation also increased Pb accumulation in root and dry weights of Bidens parviflora under Pb stress (Yang et al. 2022). Similarly, Rhizophagus irregularis increases the shoot biomass of Medicago truncatula under Pb stress (800 mg/kg), and enhances the Pb concentration and content in its roots (Zhang et al. 2021b).
Arbuscular mycorrhizae inoculation may increase the Pb tolerance of the host plant and accumulation in the roots through immobilization of Pb ions by the root cell wall or by the fungal cells (Zhang et al. 2010, 2021c). R. irregularis inoculation induced pectin and hemicellulose production in root cell walls of M. truncatula, which increases the Pb immobilization (Zhang et al. 2021c). On the other hand, in maize (Z. mays) plants inoculated with AM fungi, the most significant amount of Pb in roots is localized in the hyphal wall and within fungal cells (Zhang et al. 2010).
5 Conclusion
Rhizofiltration, phytostabilization, and phytoextraction are the main phytoremediation strategies for Pb removal from polluted environments. In the phytostabilization and rhizofiltration process, the Pb ions can be precipitated in the rhizosphere, immobilized on root cell walls, or sequestered on the root surface. In contrast, in phytoextraction, the Pb is uptake by plant roots, translocated from roots to shoots, and accumulated in aerial tissues. The Pb hyperaccumulator plants, fast-growing crops with high biomass production, and the native plants growing on heavy metal contaminated sites have been used to remove Pb from the soil, while the aquatic and terrestrial plants with fibrous root systems are suitable for Pb removal of surface water, and groundwater. The plant-associated microorganisms like bacteria and fungi could be used as an alternative to improve the Pb phytoextraction efficiency.
References
Abdelkrim S, Jebara SH, Saadani O et al (2020) In situ effects of Lathyrus sativus- PGPR to remediate and restore quality and fertility of Pb and Cd polluted soils. Ecotoxicol Environ Saf 192:110260
Aboubakar A, Douaik A, Mewouo YCM et al (2021) Determination of background values and assessment of pollution and ecological risk of heavy metals in urban agricultural soils of Yaoundé, Cameroon. J Soils Sediments 21:1437–1454
Adeyemi NO, Atayese MO, Sakariyawo OS et al (2021) Arbuscular mycorrhizal fungi species differentially regulate plant growth, phosphorus uptake and stress tolerance of soybean in lead contaminated soil. J Plant Nutr 44:1633–1648
Aftab N, Saleem K, Khan AHA et al (2021) Cosmos sulphureus Cav. is more tolerant to lead than copper and chromium in hydroponics system. Int J Environ Sci Technol 18:2325–2334
Al-Fartusie FS, Mohssan SN (2017) Essential trace elements and their vital roles in human body. Indian J Adv Chem Sci 5:127–136
Alonso-Castro AJ, Carranza-Álvarez C, Alfaro-De La Torre MC et al (2009) Removal and accumulation of cadmium and lead by Typha latifolia exposed to single and mixed metal solutions. Arch Environ Contam Toxicol 57:688–696
Alsafran M, Usman K, Ahmed B et al (2022) Understanding the phytoremediation mechanisms of potentially toxic elements: a proteomic overview of recent advances. Front Plant Sci 13
Andra SS, Datta R, Sarkar D et al (2009) Induction of lead-binding phytochelatins in vetiver grass [Vetiveria zizanioides (L.)]. J Environ Qual 38:868–877
Arazi T, Sunkar R, Kaplan B, Fromm H (1999) A tobacco plasma membrane calmodulin-binding transporter confers Ni2+ tolerance and Pb2+ hypersensitivity in transgenic plants. Plant J 20:171–182
Arshad M, Merlina G, Uzu G et al (2016) Phytoavailability of lead altered by two Pelargonium cultivars grown on contrasting lead-spiked soils. J Soils Sediments 16:581–591
Asare MO, Száková J, Tlustoš P (2023) The fate of secondary metabolites in plants growing on Cd-, As-, and Pb-contaminated soils—a comprehensive review. Environ Sci Pollut Res 30:11378–11398
Ashraf U, Kanu AS, Mo Z et al (2015) Lead toxicity in rice: effects, mechanisms, and mitigation strategies—a mini review. Environ Sci Pollut Res 22:18318–18332
ATSDR (2022) Agency for toxic substances and disease registry substance priority list. [Online]. Available: https://www.atsdr.cdc.gov/spl/index.html#2022spl. Accessed 05 Apr 2023
Austruy A, Shahid M, Xiong T et al (2014) Mechanisms of metal-phosphates formation in the rhizosphere soils of pea and tomato: environmental and sanitary consequences. J Soils Sediments 14:666–678
Baker AJM, Brooks RR (1989) Terrestrial higher plants which hyperaccumulate metallic elements—a review of their distribution, ecology and phytochemistry. Biorecovery 1:81–126
Bala S, Garg D, Thirumalesh BV et al (2022) Recent strategies for bioremediation of emerging pollutants: a review for a green and sustainable environment. Toxics 10
Ballantyne AD, Hallett JP, Riley DJ et al (2018) Lead acid battery recycling for the twenty-first century. R Soc Open Sci 5:171368
Basu S, Kumar G, Chhabra S, Prasad R (2021) Role of soil microbes in biogeochemical cycle for enhancing soil fertility. In: Verma JP, Macdonald CA, Gupta VK et al (eds) New and future developments in microbial biotechnology and bioengineering. Elsevier
Bolan NS, Park JH, Robinson B et al (2011) Phytostabilization: a green approach to contaminant containment. In: Sparks DL (ed) Advances in agronomy. Academic Press
Boopathy R (2000) Factors limiting bioremediation technologies. Bioresour Technol 74:63–67
Carrillo-Chávez A, González-Partida E, Morton-Bermea O et al (2006) Heavy metal distribution in rocks, sediments, mine tailings, leaching experiments, and groundwater from the Mineral de Pozos historical mining site, North-Central Mexico. Int Geol Rev 48:466–478
Chang J-D, Gao W, Wang P, Zhao F-J (2022) OsNRAMP5 is a major transporter for lead uptake in rice. Environ Sci Technol 56:17481–17490
Chatterjee S, Mitra A, Datta S, Veer V (2013) Phytoremediation protocols: an overview. In: Gupta DK (ed) Plant-based remediation processes. Springer Berlin Heidelberg, Berlin, Heidelberg
Cheng H, Wang M, Wong MH, Ye Z (2014) Does radial oxygen loss and iron plaque formation on roots alter Cd and Pb uptake and distribution in rice plant tissues? Plant Soil 375:137–148
Cheng S-F, Huang C-Y, Lin Y-C et al (2015) Phytoremediation of lead using corn in contaminated agricultural land—an in situ study and benefit assessment. Ecotoxicol Environ Saf 111:72–77
Cobbett C, Goldsbrough P (2002) Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu Rev Plant Biol 53:159–182
Das S, Das A, Mazumder PET et al (2021) Lead phytoremediation potentials of four aquatic macrophytes under hydroponic cultivation. Int J Phytoremediation 23:1279–1288
Delgado-González CR, Madariaga-Navarrete A, Fernández-Cortés JM et al (2021) Advances and applications of water phytoremediation: a potential biotechnological approach for the treatment of heavy metals from contaminated water. Int J Environ Res Public Health [Online] 18
Deng Z, Cao L (2017) Fungal endophytes and their interactions with plants in phytoremediation: a review. Chemosphere 168:1100–1106
dos Reis PE, De Souza KRD, Romão GF et al (2022) Potential of Talinum paniculatum cuttings in lead and manganese rhizofiltration. Water Air Soil Pollut 233:243
Dushenkov V, Kumar PBAN, Motto H, Raskin I (1995) Rhizofiltration: the use of plants to remove heavy metals from aqueous streams. Environ Sci Technol 29:1239–1245
Eapen S, D’Souza SF (2005) Prospects of genetic engineering of plants for phytoremediation of toxic metals. Biotechnol Adv 23:97–114
Eichler A, Gramlich G, Kellerhals T et al (2015) Pb pollution from leaded gasoline in South America in the context of a 2000-year metallurgical history. Sci Adv 1:e1400196
Ekta P, Modi NR (2018) A review of phytoremediation. J Pharmacogn Phytochem 7:1485–1489
Estrella-Gómez N, Mendoza-Cózatl D, Moreno-Sánchez R et al (2009) The Pb-hyperaccumulator aquatic fern Salvinia minima Baker, responds to Pb2+ by increasing phytochelatins via changes in SmPCS expression and in phytochelatin synthase activity. Aquat Toxicol 91:320–328
Etesami H (2018) Bacterial mediated alleviation of heavy metal stress and decreased accumulation of metals in plant tissues: mechanisms and future prospects. Ecotoxicol Environ Saf 147:175–191
Etim E (2012) Phytoremediation and its mechanisms: a review. Int J Environ Bioenergy 2:120–136
Favas P, Pratas J, Varun M et al (2014) Phytoremediation of soils contaminated with metals and metalloids at mining areas: potential of native flora. In: Hernández-Soriano (ed) Environmental risk assessment of soil contamination. InTech
Gall JE, Rajakaruna N (2013) The physiology, functional genomics, and applied ecology of heavy metal-tolerant Brassicaceae. In: Lang M (ed) Brassicaceae: characterization, functional genomics and health benefits. Nova Science Pub Inc.
Gan H, Churchill ACL, Wickings K (2017) Invisible but consequential: root endophytic fungi have variable effects on belowground plant–insect interactions. Ecosphere 8:e01710
Gaur A, Adholeya A (2004) Prospects of arbuscular mycorrhizal fungi in phytoremediation of heavy metal contaminated soils. Curr Sci 86
Gong X, Yang F, Pan X, Shao JF (2022) Accumulation of silicon in shoots is required for reducing lead uptake in rice. Crop J
Gupta DK, Huang HG, Corpas FJ (2013a) Lead tolerance in plants: strategies for phytoremediation. Environ Sci Pollut Res 20:2150–2161
Gupta DK, Vandenhove H, Inouhe M (2013b) Role of phytochelatins in heavy metal stress and detoxification mechanisms in plants. In: Gupta DK, Corpas FJ, Palma JM (eds) Heavy metal stress in plants. Springer Berlin Heidelberg, Berlin, Heidelberg
Han Y-L, Huang S-Z, Gu J-G et al (2008) Tolerance and accumulation of lead by species of Iris L. Ecotoxicology 17:853–859
Hansel CM, Fendorf S, Sutton S, Newville M (2001) Characterization of Fe plaque and associated metals on the roots of mine-waste impacted aquatic plants. Environ Sci Technol 35:3863–3868
Hasnaoui SE, Fahr M, Keller C et al (2020) Screening of native plants growing on a Pb/Zn mining area in eastern Morocco: perspectives for phytoremediation. Plants [Online] 9
He X, Xu M, Wei Q et al (2020) Promotion of growth and phytoextraction of cadmium and lead in Solanum nigrum L. mediated by plant-growth-promoting rhizobacteria. Ecotoxicol Environ Saf 205:111333
He J, Chen X, Achal V (2021) Bioremediation: concepts and application. In: Sharma N, Sodhi AS, Batra N (eds) Basic concepts in environmental biotechnology. CRC Press, Boca Raton
He W, Wang S, Wang Y et al (2023) Pathways and characteristics of lead uptake and transportation in Rhus chinensis Mill. Forests 14:90
Heidari S, Mostafaei S, Razazian N et al (2022) The effect of lead exposure on IQ test scores in children under 12 years: a systematic review and meta-analysis of case-control studies. Syst Rev 11:106
Ho LB, Huynh BV, Bui TC (2021) Accumulation and distribution of lead (Pb) in different tissues of Lucky bamboo plants (Dracaena sanderiana). JDAE 20:50–60
Huang G-Y, Wang Y-S (2009) Expression analysis of type 2 metallothionein gene in mangrove species (Bruguiera gymnorrhiza) under heavy metal stress. Chemosphere 77:1026–1029
Inouhe M, Huang H, Chaudhary SK, Gupta DK (2012) Heavy metal bindings and their interactions with thiol peptides and other biological ligands in plant cells. In: Gupta DK, Sandalio LM (eds) Metal toxicity in plants: perception, signaling and remediation. Springer Berlin Heidelberg, Berlin, Heidelberg
Irias Zelaya CR, Gadelha JC, Hernandez FFF, Ortiz Escobar ME (2020) Heavy metals behavior in the presence of organic acids and proteins in the rhizosphere and plant tissues of yellow melon var. natal (Cucumis melo L.) cultivated in a Quartzarenic neosol. Water Air Soil Pollut 231:572
Jadia CD, Fulekar M (2009) Phytoremediation of heavy metals: recent techniques. Afr J Biotechnol 8
Jee C (2016) Advances in phytoremediation and rhizoremediation. Octa J Environ Res 4:18–32
Jiang W, Liu D (2010) Pb-induced cellular defense system in the root meristematic cells of Allium sativum L. BMC Plant Biol 10:40
Jiang M, Cai X, Liao J et al (2020) Different strategies for lead detoxification in dwarf bamboo tissues. Ecotoxicol Environ Saf 193:110329
John DA, Leventhal JS (1995) Bioavailability of metals. In: Bray EAD (ed) Preliminary compilation of descriptive geoenvironmental mineral deposit models. U.S. Geological Survey, Denver, Colorado
Kafle A, Timilsina A, Gautam A et al (2022) Phytoremediation: mechanisms, plant selection and enhancement by natural and synthetic agents. Environ Adv 8:100203
Khan N, Seshadri B, Bolan N et al (2016) Root iron plaque on wetland plants as a dynamic pool of nutrients and contaminants. In: Sparks DL (ed) Advances in agronomy. Academic Press
Khanam R, Kumar A, Nayak AK et al (2020) Metal(loid)s (As, Hg, Se, Pb and Cd) in paddy soil: bioavailability and potential risk to human health. Sci Total Environ 699:134330
Kim Y-O, Kang H (2018) Comparative expression analysis of genes encoding metallothioneins in response to heavy metals and abiotic stresses in rice (Oryza sativa) and Arabidopsis thaliana. Biosci Biotechnol Biochem 82:1656–1665
Kisa D, Öztürk L, Doker S, Gökçe İ (2017) Expression analysis of metallothioneins and mineral contents in tomato (Lycopersicon esculentum) under heavy metal stress. J Sci Food Agric 97:1916–1923
Kong Z, Glick BR (2017) The role of plant growth-promoting bacteria in metal phytoremediation. In: Poole RK (ed) Advances in microbial physiology. Academic Press
Kopittke PM, Asher CJ, Blamey FPC et al (2008) Localization and chemical speciation of Pb in roots of signal grass (Brachiaria decumbens) and rhodes grass (Chloris gayana). Environ Sci Technol 42:4595–4599
Kristanti RA, Ngu WJ, Yuniarto A, Hadibarata T (2021) Rhizofiltration for removal of inorganic and organic pollutants in groundwater: a review. Biointerface Res Appl Chem 4:12326–12347
Krzesłowska M (2011) The cell wall in plant cell response to trace metals: polysaccharide remodeling and its role in defense strategy. Acta Physiol Plant 33:35–51
Kurade MB, Ha Y-H, Xiong J-Q et al (2021) Phytoremediation as a green biotechnology tool for emerging environmental pollution: a step forward towards sustainable rehabilitation of the environment. Chem Eng J 415:129040
Li G-Y, Hu N, Ding D-X et al (2011a) Screening of plant species for phytoremediation of uranium, thorium, barium, nickel, strontium and lead contaminated soils from a uranium mill tailings repository in South China. Bull Environ Contam Toxicol 86:646–652
Li T, Di Z, Islam E et al (2011b) Rhizosphere characteristics of zinc hyperaccumulator Sedum alfredii involved in zinc accumulation. J Hazard Mater 185:818–823
Li F-L, Liu C-Q, Yang Y-G et al (2012) Natural and anthropogenic lead in soils and vegetables around Guiyang city, southwest China: a Pb isotopic approach. Sci Total Environ 431:339–347
Li X, Huang S, Mcbride MB (2021) Rhizosphere effect on Pb solubility and phytoavailability in Pb-contaminated soils. Environ Pollut 268:115840
Liu J, Luo L (2019) Uptake and transport of Pb across the iron plaque of waterlogged dropwort (Oenanthe javanica DC.) based on micro-XRF and XANES. Plant Soil 441:191–205
Liu D, Jiang W, Liu C et al (2000) Uptake and accumulation of lead by roots, hypocotyls and shoots of Indian mustard [Brassica juncea (L.)]. Bioresour Technol 71:273–277
Liu CY, Gong XF, Tang YP, Chen CL (2015) Lead sequestration in iron plaques developed on Phalaris arundinacea Linn. and Carex cinerascens Kukenth. from Poyang Lake (China). Aquat Bot 122:54–59
Liu C, Gong X, Chen C et al (2016) The effect of iron plaque on lead translocation in soil-Carex cinerascens Kukenth. system. Int J Phytoremediation 18:1–9
Liu L, Li W, Song W, Guo M (2018) Remediation techniques for heavy metal-contaminated soils: principles and applicability. Sci Total Environ 633:206–219
Liu C, Lin H, Li B et al (2021) Endophyte Pseudomonas putida enhanced Trifolium repens L. growth and heavy metal uptake: a promising in-situ non-soil cover phytoremediation method of nonferrous metallic tailing. Chemosphere 272:129816
Lone MI, He Z-L, Stoffella PJ, Yang X-E (2008) Phytoremediation of heavy metal polluted soils and water: progresses and perspectives. J Zhejiang Univ Sci B 9:210–220
Lorestani B, Cheraghi M, Yousefi N (2011) Phytoremediation potential of native plants growing on a heavy metals contaminated soil of copper mine in Iran. Int J Geol 5:299–304
Lorestani B, Yousefi N, Cheraghi M, Farmany A (2013) Phytoextraction and phytostabilization potential of plants grown in the vicinity of heavy metal-contaminated soils: a case study at an industrial town site. Environ Monit Assess 185:10217–10223
Ma Y, Prasad MNV, Rajkumar M, Freitas H (2011) Plant growth promoting rhizobacteria and endophytes accelerate phytoremediation of metalliferous soils. Biotechnol Adv 29:248–258
Ma X, Liu J, Wang M (2013) Differences between rice cultivars in iron plaque formation on roots and plant lead tolerance. Adv J Food Sci Technol 5:160–163
Ma Y, Rajkumar M, Zhang C, Freitas H (2016) Beneficial role of bacterial endophytes in heavy metal phytoremediation. J Environ Manage 174:14–25
Mahar A, Wang P, Ali A et al (2016) Challenges and opportunities in the phytoremediation of heavy metals contaminated soils: a review. Ecotoxicol Environ Saf 126:111–121
Mahdavian K, Ghaderian SM, Torkzadeh-Mahani M (2017) Accumulation and phytoremediation of Pb, Zn, and Ag by plants growing on Koshk lead–zinc mining area, Iran. J Soils Sediments 17:1310–1320
Manara A, Fasani E, Furini A, Dalcorso G (2020) Evolution of the metal hyperaccumulation and hypertolerance traits. Plant Cell Environ 43:2969–2986
Manoj SR, Karthik C, Kadirvelu K et al (2020) Understanding the molecular mechanisms for the enhanced phytoremediation of heavy metals through plant growth promoting rhizobacteria: a review. J Environ Manage 254:109779
Mansoor S, Khan NF, Farooq I et al (2022) Phytoremediation at molecular level. In: Bhat RA, Tonelli FMP, Dar GH et al (eds) Phytoremediation. Academic Press
Manzoor M, Abid R, Rathinasabapathi B et al (2019) Metal tolerance of arsenic-resistant bacteria and their ability to promote plant growth of Pteris vittata in Pb-contaminated soil. Sci Total Environ 660:18–24
Manzoor M, Gul I, Manzoor A et al (2020) Lead availability and phytoextraction in the rhizosphere of Pelargonium species. Environ Sci Pollut Res 27:39753–39762
Midhat L, Ouazzani N, Hejjaj A et al (2019) Accumulation of heavy metals in metallophytes from three mining sites (Southern Centre Morocco) and evaluation of their phytoremediation potential. Ecotoxicol Environ Saf 169:150–160
Mishra S, Srivastava S, Tripathi RD et al (2006) Lead detoxification by coontail (Ceratophyllum demersum L.) involves induction of phytochelatins and antioxidant system in response to its accumulation. Chemosphere 65:1027–1039
Mitra A, Chatterjee S, Datta S et al (2014) Mechanism of metal transporters in plants. In: Kumar Gupta D, Chatterjee S (eds) Heavy metal remediation: transport accumulation in plants. Nova Science Publishers, Inc.
More SS, Shinde SE, Kasture MC (2019) Root exudates a key factor for soil and plant: an overview. Pharm Innov J 8:449–459
Nag R, Cummins E (2022) Human health risk assessment of lead (Pb) through the environmental-food pathway. Sci Total Environ 810:151168
Narula N, Kothe E, Behl RK (2012) Role of root exudates in plant-microbe interactions. J Appl Bot Food Qual 82:122–130
Nedjimi B (2021) Phytoremediation: a sustainable environmental technology for heavy metals decontamination. SN Appl Sci 3:286
Nouri J, Lorestani B, Yousefi N et al (2011) Phytoremediation potential of native plants grown in the vicinity of Ahangaran lead–zinc mine (Hamedan, Iran). Environ Earth Sci 62:639–644
Olaniran AO, Balgobind A, Pillay B (2013) Bioavailability of heavy metals in soil: impact on microbial biodegradation of organic compounds and possible improvement strategies. Int J Mol Sci 14:10197–10228
Otte ML, Jacob DL (2006) Constructed wetlands for phytoremediation: rhizofiltration, phytostabilisation and phytoextraction. In: Mackova M, Dowling D, Macek T (eds) Phytoremediation rhizoremediation. Springer
Parrotta L, Guerriero G, Sergeant K et al (2015) Target or barrier? The cell wall of early- and later-diverging plants vs cadmium toxicity: differences in the response mechanisms. Front Plant Sci 6
Parven S, De A, Gupta A (2022) Cu and Pb accumulation and removal from aqueous medium by Enydra fluctuans Lour. (Asteraceae)—a medicinal plant with potential for phytoremediation. Environ Sci Pollut Res 29:66902–66912
Pasricha S, Mathur V, Garg A et al (2021) Molecular mechanisms underlying heavy metal uptake, translocation and tolerance in hyperaccumulators-an analysis: heavy metal tolerance in hyperaccumulators. Environ Chall 4:100197
Pathan SI, Ceccherini MT, Sunseri F, Lupini A (2020) Rhizosphere as hotspot for plant-soil-microbe interaction. In: Datta R, Meena RS, Pathan SI et al (eds) Carbon and nitrogen cycling in soil. Springer Singapore, Singapore
Peralta-Videa JR, Lopez ML, Narayan M et al (2009) The biochemistry of environmental heavy metal uptake by plants: implications for the food chain. Int J Biochem Cell Biol 41:1665–1677
Pietrini I, Grifoni M, Franchi E et al (2021) Enhanced lead phytoextraction by endophytes from indigenous plants. Soil Syst 5:55
Pourrut B, Shahid M, Dumat C et al (2011) Lead uptake, toxicity, and detoxification in plants. In: Whitacre DM (ed) Reviews of environmental contamination and toxicology, vol 213. Springer New York, New York, NY
Pourrut B, Shahid M, Douay F et al (2013) Molecular mechanisms involved in lead uptake, toxicity and detoxification in higher plants. In: Gupta DK, Corpas FJ, Palma JM (eds) Heavy metal stress in plants. Springer Berlin Heidelberg, Berlin, Heidelberg
Rahimpoor R, Rostami M, Assari MJ et al (2020) Evaluation of blood lead levels and their effects on hematological parameters and renal function in Iranian lead mine workers. Health Scope 9:e95917
Rahman Z, Singh VP (2019) The relative impact of toxic heavy metals (THMs) (arsenic (As), cadmium (Cd), chromium (Cr)(VI), mercury (Hg), and lead (Pb)) on the total environment: an overview. Environ Monit Assess 191:419
Rahman MM, Swaraz AM, El-Shehawi AM et al (2022) The mechanistic basis of sulfur-mediated alleviation of Pb toxicity in wheat. Gesunde Pflanz 74:571–581
Rai GK, Bhat BA, Mushtaq M et al (2021) Insights into decontamination of soils by phytoremediation: a detailed account on heavy metal toxicity and mitigation strategies. Physiol Plant 173:287–304
Rajkumar M, Sandhya S, Prasad MNV, Freitas H (2012) Perspectives of plant-associated microbes in heavy metal phytoremediation. Biotechnol Adv 30:1562–1574
Rawat K, Fulekar M, Pathak B (2012) Rhizofiltration: a green technology for remediation of heavy metals. Int J Innov Bio-Sci 2:193–199
Reeves RD, Baker AJM, Jaffré T et al (2018) A global database for plants that hyperaccumulate metal and metalloid trace elements. New Phytol 218:407–411
Rolón-Cárdenas GA, Martínez-Martínez JG, Arvizu-Gómez JL et al (2022) Enhanced Cd-accumulation in Typha latifolia by interaction with Pseudomonas rhodesiae GRC140 under axenic hydroponic conditions. Plants 11:1447
Saifullah, Meers E, Qadir M et al (2009) EDTA-assisted Pb phytoextraction. Chemosphere 74:1279–1291
Salazar MJ, Pignata ML (2014) Lead accumulation in plants grown in polluted soils. Screening of native species for phytoremediation. J Geochem Explor 137:29–36
Scarano G, Morelli E (2002) Characterization of cadmium- and lead-phytochelatin complexes formed in a marine microalga in response to metal exposure. BioMetals 15:145–151
Seshadri B, Bolan NS, Naidu R (2015) Rhizosphere-induced heavy metal(loid) transformation in relation to bioavailability and remediation. J Soil Sci Plant Nutr 15:524–548
Seth CS, Misra V, Singh RR, Zolla L (2011) EDTA-enhanced lead phytoremediation in sunflower (Helianthus annuus L.) hydroponic culture. Plant Soil 347:231–242
Sharma P (2021) Efficiency of bacteria and bacterial assisted phytoremediation of heavy metals: an update. Bioresour Technol 328:124835
Sharma P, Dubey R (2005) Lead toxicity in plants. Braz J Plant Physiol 17:35–52
Sharma SS, Dietz K-J, Mimura T (2016) Vacuolar compartmentalization as indispensable component of heavy metal detoxification in plants. Plant Cell Environ 39:1112–1126
Shi W, Zhou J, Li J et al (2021) Lead exposure-induced defense responses result in low lead translocation from the roots to aerial tissues of two contrasting poplar species. Environ Pollut 271:116346
Singh V, Thakur L, Mondal P (2015) Removal of lead and chromium from synthetic wastewater using Vetiveria zizanioides. Clean (Weinh) 43:538–543
Singh H, Verma A, Kumar M et al (2017) Phytoremediation: a green technology to clean up the sites with low and moderate level of heavy metals. Austin Biochem 2:1012
Song B, Zeng G, Gong J et al (2017) Evaluation methods for assessing effectiveness of in situ remediation of soil and sediment contaminated with organic pollutants and heavy metals. Environ Int 105:43–55
Srivastava S, Anand V, Singh P et al (2021) Microbial systems as a source of novel genes for enhanced phytoremediation of contaminated soils. In: Kumar A, Singh VK, Singh P et al (eds) Microbe mediated remediation of environmental contaminants. Woodhead Publishing
St-Cyr L, Fortin D, Campbell PGC (1993) Microscopic observations of the iron plaque of a submerged aquatic plant (Vallisneria americana Michx). Aquat Bot 46:155–167
Sumranwanich T, Boonthaworn K, Singh A (2018) The roles of plant cell wall as the first-line protection against lead (Pb) toxicity. KMUTNB IJAST 11:239–245
Sunkar R, Kaplan B, Bouché N et al (2000) Expression of a truncated tobacco NtCBP4 channel in transgenic plants and disruption of the homologous Arabidopsis CNGC1 gene confer Pb2+ tolerance. Plant J 24:533–542
Sytar O, Ghosh S, Malinska H et al (2021) Physiological and molecular mechanisms of metal accumulation in hyperaccumulator plants. Physiol Plant 173:148–166
Tangahu BV, Sheikh Abdullah SR, Basri H et al (2022) Lead (Pb) removal from contaminated water using constructed wetland planted with Scirpus grossus: optimization using response surface methodology (RSM) and assessment of rhizobacterial addition. Chemosphere 291:132952
Taulé C, Vaz-Jauri P, Battistoni F (2021) Insights into the early stages of plant–endophytic bacteria interaction. World J Microbiol Biotechnol 37:13
Tripathi RD, Tripathi P, Dwivedi S et al (2014) Roles for root iron plaque in sequestration and uptake of heavy metals and metalloids in aquatic and wetland plants. Metallomics 6:1789–1800
Van Slycken S, Meers E, Meiresonne L et al (2008) The use of bio-energy crops for phytoremediation of metal enriched soils in the Campine region. Commun Agric Appl Biol Sci 73:19–22
Veselý T, Tlustoš P, Száková J (2011) The use of water lettuce (Pistia Stratiotes L.) for rhizofiltration of a highly polluted solution by cadmium and lead. Int J Phytoremediation 13:859–872
Vidali M (2001) Bioremediation. An overview. Pure Appl Chem 73:1163–1172
Vives-Peris V, De Ollas C, Gómez-Cadenas A, Pérez-Clemente RM (2020) Root exudates: from plant to rhizosphere and beyond. Plant Cell Rep 39:3–17
Wang M, Duan D, Xu C et al (2015) Adsorption ability of cell wall (CW) components in roots of tea plant (Camellia sinensis L.) to Pb and FTIR spectra of their functional groups. Acta Ecol Sin 35:1743–1751
Wang L, Ji B, Hu Y et al (2017) A review on in situ phytoremediation of mine tailings. Chemosphere 184:594–600
Wenzel WW, Lombi E, Adriano DC (1999) Biogeochemical processes in the rhizosphere: role in phytoremediation of metal-polluted soils. In: Prasad MNV, Hagemeyer J (eds) Heavy metal stress in plants: from molecules to ecosystems. Springer Berlin Heidelberg, Berlin, Heidelberg
Wierzbicka M (1998) Lead in the apoplast of Allium cepa L. root tips—ultrastructural studies. Plant Sci 133:105–119
Wong CSC, Li XD (2004) Pb contamination and isotopic composition of urban soils in Hong Kong. Sci Total Environ 319:185–195
Wu J, Long J, Liu L et al (2018) Risk assessment and source identification of toxic metals in the agricultural soil around a Pb/Zn mining and smelting area in southwest China. Int J Environ Res Public Health 15:1838
Yan A, Wang Y, Tan SN et al (2020) Phytoremediation: a promising approach for revegetation of heavy metal-polluted land. Front Plant Sci 11
Yang W, Luo L, Bostick BC et al (2021) Effect of combined arsenic and lead exposure on their uptake and translocation in Indian mustard. Environ Pollut 274:116549
Yang Y, Huang B, Xu J et al (2022) Heavy metal domestication enhances beneficial effects of arbuscular mycorrhizal fungi on lead (Pb) phytoremediation efficiency of Bidens parviflora through improving plant growth and root Pb accumulation. Environ Sci Pollut Res 29:32988–33001
Yoon J, Cao X, Zhou Q, Ma LQ (2006) Accumulation of Pb, Cu, and Zn in native plants growing on a contaminated Florida site. Sci Total Environ 368:456–464
Yuan X, Xiong T, Yao S et al (2019) A real filed phytoremediation of multi-metals contaminated soils by selected hybrid sweet sorghum with high biomass and high accumulation ability. Chemosphere 237:124536
Zandi P, Yang J, Darma A et al (2022) Iron plaque formation, characteristics, and its role as a barrier and/or facilitator to heavy metal uptake in hydrophyte rice (Oryza sativa L.). Environ Geochem Health
Zehra A, Sahito ZA, Tong W et al (2020) Assessment of sunflower germplasm for phytoremediation of lead-polluted soil and production of seed oil and seed meal for human and animal consumption. J Environ Sci 87:24–38
Zhan J, Huang H, Yu H et al (2020) The combined effects of Cd and Pb enhanced metal binding by root cell walls of the phytostabilizer Athyrium wardii (Hook.). Environ Pollut 258:113663
Zhang H-H, Tang M, Chen H et al (2010) Effect of inoculation with AM fungi on lead uptake, translocation and stress alleviation of Zea mays L. seedlings planting in soil with increasing lead concentrations. Eur J Soil Biol 46:306–311
Zhang J, Martinoia E, Lee Y (2018) Vacuolar transporters for cadmium and arsenic in plants and their applications in phytoremediation and crop development. Plant Cell Physiol 59:1317–1325
Zhang Y, Hou D, O’Connor D et al (2019) Lead contamination in Chinese surface soils: source identification, spatial-temporal distribution and associated health risks. Crit Rev Environ Sci Technol 49:1386–1423
Zhang J, Qian Y, Chen Z et al (2020) Lead-induced oxidative stress triggers root cell wall remodeling and increases lead absorption through esterification of cell wall polysaccharide. J Hazard Mater 385:121524
Zhang B, Gao Y, Zhang L, Zhou Y (2021a) The plant cell wall: biosynthesis, construction, and functions. J Integr Plant Biol 63:251–272
Zhang H, Ren W, Zheng Y et al (2021b) Arbuscular mycorrhizal fungi increase Pb uptake of colonized and non-colonized Medicago truncatula root and deliver extra Pb to colonized root segment. Microorganisms 9:1203
Zhang X, Hu W, Xie X et al (2021c) Arbuscular mycorrhizal fungi promote lead immobilization by increasing the polysaccharide content within pectin and inducing cell wall peroxidase activity. Chemosphere 267:128924
Zhao L, Li T, Yu H et al (2015) Changes in chemical forms, subcellular distribution, and thiol compounds involved in Pb accumulation and detoxification in Athyrium wardii (Hook.). Environ Sci Pollut Res 22:12676–12688
Zhou C, Huang M, Li Y et al (2016) Changes in subcellular distribution and antioxidant compounds involved in Pb accumulation and detoxification in Neyraudia reynaudiana. Environ Sci Pollut Res 23:21794–21804
Zhou J, Zhang Z, Zhang Y et al (2018) Effects of lead stress on the growth, physiology, and cellular structure of privet seedlings. PLoS ONE 13:e0191139
Zou T, Li T, Zhang X et al (2012) Lead accumulation and phytostabilization potential of dominant plant species growing in a lead–zinc mine tailing. Environ Earth Sci 65:621–630
Acknowledgements
This work was funded by grants from CONAHCYT, Programa Presupuestario F003. Fondo Sectorial de Investigación para la Educación, CB2017-2018 A1-S-40454, to Alejandro Hernández-Morales.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Rolón-Cárdenas, G.A., Hernández-Morales, A. (2024). Phytoremediation of Lead Present in Environment: A Review. In: Kumar, N., Jha, A.K. (eds) Lead Toxicity Mitigation: Sustainable Nexus Approaches. Environmental Contamination Remediation and Management. Springer, Cham. https://doi.org/10.1007/978-3-031-46146-0_7
Download citation
DOI: https://doi.org/10.1007/978-3-031-46146-0_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-46145-3
Online ISBN: 978-3-031-46146-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)