Abstract
The study used scattered literature to summarize the effects of excess Cd, As, and Pb from contaminated soils on plant secondary metabolites/bioactive compounds (non-nutrient organic substances). Hence, we provided a systematic overview involving the sources and forms of Cd, As, and Pb in soils, plant uptake, mechanisms governing the interaction of these risk elements during the formation of secondary metabolites, and subsequent effects. The biogeochemical characteristics of soils are directly responsible for the mobility and bioavailability of risk elements, which include pH, redox potential, dissolved organic carbon, clay content, Fe/Mn/Al oxides, and microbial transformations. The radial risk element flow in plant systems is restricted by the apoplastic barrier (e.g., Casparian strip) and chelation (phytochelatins and vacuole sequestration) in roots. However, bioaccumulation is primarily a function of risk element concentration and plant genotype. The translocation of risk elements to the shoot via the xylem and phloem is well-mediated by transporter proteins. Besides the dysfunction of growth, photosynthesis, and respiration, excess Cd, As, and Pb in plants trigger the production of secondary metabolites with antioxidant properties to counteract the toxic effects. Eventually, this affects the quantity and quality of secondary metabolites (including phenolics, flavonoids, and terpenes) and adversely influences their antioxidant, antiinflammatory, antidiabetic, anticoagulant, and lipid-lowering properties. The mechanisms governing the translocation of Cd, As, and Pb are vital for regulating risk element accumulation in plants and subsequent effects on secondary metabolites.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plants possess unique strategies to remain cellularly and physiologically unaffected during harsh environmental effects, e.g., high/low temperatures and excess risk elements (Cd, As, and Pb). To achieve adaption to fluctuating environmental effects, plants exhibit a series of metabolic mechanisms that generate secondary metabolites (Arnold et al. 2019; Movahedi et al. 2021).
Plant secondary metabolites are non-nutrient bioactive substances manufactured via the metabolic pathway under biotic (e.g., fungi, viruses, and insects attack) and abiotic (e.g., UV radiation, salinity, light intensity, temperature changes, drought, and metals) stresses (Zheljazkov et al. 2006; Teoh 2015). Hence, climatic changes can influence the production of secondary metabolites in plants (Kroymann 2011; Arnold et al. 2019). In principle, secondary metabolites are manufactured by plants under environmental stress for structural and functional stability via signaling processes and pathways (Edreva et al. 2008). For instance, orchinol (a type of phenanthrene), primarily produced as an adaptive approach by orchids, is directly associated with pathogen attacks (Stoessl and Arditti 1984). Plant secondary metabolites offer enormous health benefits to consumers (Table 1). Different plants (e.g., poplar, orchids, and artemisia species) can produce a wide range of secondary metabolites, including alkaloids, anthocyanins, bibenzyls, flavonoids, phenols, polysaccharides, and stilbenes (Harper 1998; Teoh 2015). These compounds exhibit antioxidant, antimicrobial, antiinflammatory, antidiabetic, and anticoagulant properties. For example, some phenanthrene and stilbenoids possess cytotoxic properties, prevent angiogenesis (formation of new blood vessels) and tumor spread, and inhibit cancer cell replication, e.g., in human colon carcinoma cell lines (Kovács et al. 2008; De Luca et al. 2012; Vukmirovic et al. 2018). Recently, secondary metabolites from different plant species possessing antiviral properties have exhibited the potential for treating SARS-CoV-2 — severe acute respiratory syndrome coronavirus-2 (Bhuiyan et al. 2020). Elevated concentrations of Cd, As, and Pb from natural (geogenic-geochemical composition of source rock) and anthropogenic sources (e.g., industrial and mining processes) in soils can pose a threat to plants (Edreva et al. 2008). These elements are toxic to plants with no relevance to growth and development. The uptake, translocation, and accumulation of elements in plants depend on species and ecotype, plant organ, climatic conditions, and different seasons (Kabata-Pendias and Pendias 2001). Risk elements mostly remain in the plant’s root/rhizome system (belowground biomass). However, As and Cd are relatively easily transported to aboveground biomass, e.g., the leaf (Száková et al. 2018). The elements accumulated in edible organs of crops or spontaneous plants represent the main entry point in the food chain. Exposure to excess risk elements alters plant physiology, e.g., the rate of photosynthesis, respiration, enzymatic activities, and element distribution at the cellular level (Rout and Das 2003).
Abiotic stress, e.g., As, Cd, and Pb, can trigger the production of bioactive substances by changing aspects of secondary metabolism (Kováčik and Klejdus 2008; Nasim and Dhir 2010). Risk element stress on medicinal and other plants can also suppress secondary metabolite production (García-Calderón et al. 2015). High-risk elements in plant use for medicines can stimulate the production of bioactive compounds. Oxidative stress, induced by risk elements, initiates pathways that affect specific secondary metabolite production (Yadav 2010). Meanwhile, apoplastic barrier (e.g., Casparian strip), vacuolar sequestration, and phytochelatins in roots and other compartments of plants can maintain an adequate amount of essential metal ions (e.g., Zn) and reduce the damaging effects of non-essential ones (Yamaguchi et al. 2011). However, during an excess of risk elements, plants generate harmful oxygen species/singlets (e.g., superoxide (O2−) and hydrogen peroxide (H2O2)), which eventually lead to stress (Huang et al. 2019). Low molecular weight substances, enzymes, and many secondary metabolites (e.g., flavonoids, phenylpropanoids, and phenolic acid) serve as effective antioxidants (Kumar and Pandey 2013). Studies (e.g., Michalak 2006; Kumar et al. 2013) indicate that phenolics can form chelates with metals or scavenge ROS.
Several studies have described the effects of individual risk elements (Cd, As, and Pb) in soils and subsequent translocation in plants and metabolites. Meanwhile, most of these studies are site-specific. Therefore, we aim to compile and highlight the effects of elevated levels of Cd, As, and Pb in soils, uptake by plants, and their translocation to different aerial parts. Additionally, we collated vital literature to provide an in-depth overview and reliable inferences on the effects of As, Cd, and Pb after their uptake on plant secondary metabolites, as they show potency as herbal medication.
The next aspect of the study reviews Cd, As, and Pb uptake, translocation in plants, interactions with bioactive substances, and the potential effects on human health. Species of Cd, As, and Pb and their influence on secondary metabolites of plants are well-discussed. Commonalities and variabilities among different species of risk elements in plants may represent a prerequisite in determining the level of toxicity.
Soil-to-root pathway of risk elements
Natural processes (weathering of the parent material) and anthropogenic interferences (e.g., metallurgy, agricultural, and industrial activities) contribute primarily to the elevated Cd, As, and Pb levels in soils (Redondo-Gómez et al. 2010; Pourrut et al. 2011; Hu et al. 2018). However, anthropogenic accumulation produces the highest concentrations of risk elements in the soil than the former (Geiszinger et al. 2002).
The physicochemical forms of Cd, As, and Pb may exist in soils as inorganic species, organic complexes, adsorbed on solid phases, or constituents of solid phases, with varying solubility levels (Aydinalp and Marinova 2003). Plants absorb only the available fraction associated with the exchange complexes (organic and inorganic components) and soluble fractions in soil solution. Generally, the distribution and bioavailability of risk elements in the soil ecosystem are often well-governed by soil reaction (pH), redox condition, dissolved organic carbon (DOC), clay content, and oxides of Fe, Mn, and Al (Sungur et al. 2020).
Cadmium (Cd)
Cadmium toxicity in soils occurs via industrial (e.g., metallurgical activities and Zn refining) and agricultural practices, e.g., extensive herbicides and fertilizers (phosphate) applications (Redondo-Gómez et al. 2010). Although Cd-containing pesticides remained banned in Europe, Cd toxicity is possible by applying biosolids (e.g., sewage sludge) to arable fields (European Council Directive 1989).
Geochemically, the behavior of Cd in soils operates in a soluble and exchangeable portion, precipitated with carbonates, occluded in amorphous Fe/Mn oxides, complexed with organic matter (OM), and residual form (Wiggenhauser et al. 2021). The soluble and exchangeable fractions of Cd are more bioaccessible to plants. Non-residual Cd: (CO2−3-bound, Fe–Mn oxides, and OM fractions) becomes bioaccessible with changing pH and redox potential Eh (Rinklebe and Shaheen 2014). Under oxidizing conditions, the decrease in sulfide (S2-) produces protons, leading to increased solubility of Cd (Rinklebe and Shaheen 2014).
Cadmium can also form complexes with soil colloids. Soil acidity increases Cd desorption in colloids, allowing root uptake. The formation of Cd(OH)2 reduces the mobility of Cd at high alkaline pH, resulting in low plant accumulation (Gong et al. 2021). Soil Eh plays a vital role in Cd solubility. In flooded fields, Eh is again well-mediated by oxidized FeIII, MnIII/VI, NO3–, SO42−, and other soil components useful in microbial respiration (Cui et al. 2020). In a reduction reaction, the ions/ionic compounds receive electrons to generate reduced substances, which result in decreased Eh (Lin et al. 2021). The decrease in Eh can lead to the reductive dissolution of Fe and Mn oxides, which release bonded Cd into soil solution and increase bioavailability (Shaheen et al. 2016; Wu et al. 2019). Conversely, reduced Cd can precipitate under sulfide conditions, decreasing solubility and bioavailability (Rinklebe et al. 2016). Eh may increase, which can cause the dissolution of CdS and release of Cd2+ in well-drained soils (Rinklebe et al. 2016; Shaheen and Rinklebe 2017).
As Cd adsorbent in soils, OM can immobilize Cd, reduce phytoavailability, and decrease the uptake of Cd. For example, Ali et al. (2020) recorded a decreased Cd accumulation in the roots, straws, and grains of Oryza sativa in the presence of OM. Organic acids, as negative anions, react with Cd2+ and can immobilize Cd, thus reducing its phytoavailability (Li et al. 2019a, b). However, in soils managed for wet cultivation (e.g., Entisols and Inceptisols), OM as chelates can enhance the phytoavailability of Cd (Subašić et al. 2022). It indicates that remediation of Cd-contaminated soils with OM should be well-managed. The application of OM should be well-controlled to prevent further contamination by other risk elements.
Cadmium uptake by plants depends directly on soil properties, plant type, chemical properties of Cd, mobility, and immobilization by root exudates in the rhizosphere (Fig. 1). The chelation of Cd can affect pH and Eh in the rhizosphere and subsequently influence the mobility of Cd within the soil. For example, an arbuscular mycorrhizal association can reduce the toxicity of Cd2+ in the rhizosphere by inducing alkalinity, which can immobilize Cd2+ (Janousková and Pavlíková 2010).
Schematic diagram showing uptake and translocation of Cd by plants under different soil physicochemical properties. Here Cd uptake is mediated by many transporters, NRAMP families, P-type ATPases, and ABC transporters. The absorption of Cd from soil and its distribution between roots and shoots is a regulated process involving metal transporters of the root cell, plasma membrane, xylem, phloem loading/unloading, and leaf/shoot sequestration and detoxification. NRAMP natural resistance-associated macrophage protein; ABC ATP-binding cassette; Eh redox potential; OM organic matter (adapted and modified from Shahid et al. 2016)
Root exudates (e.g., oxalic, fumaric, malic, and acetic acids) have different detoxification mechanisms for plants to alleviate the toxicity of free Cd2+ through organic complexation (Lapie et al. 2019). Metal chelators and many other organic acids also play essential roles in reducing Cd toxicity in some organelles (e.g., via glutathione in the vacuole) and macromolecules (Shahid et al. 2016). Hence, chelation performs a vital function in the tolerance of plants to Cd toxicity.
Free Cd2+ uptake into the root epidermal layer undergoes a series of interactions. At the plasma membranes of root cells, H2CO3 dissociates to release H+ and HCO3 in the course plant’s respiration (Yamaguchi et al. 2011). Hydrogen ions swiftly shift for Cd2+ adsorption in root epidermal cells and subsequently to the epidermis layer via the apoplast pathway. Cadmium ion enters plant cells in Ca2+, Fe2+, and Zn2+ channels (Sadana et al. 2003). However, this is more restricted to specific areas of Cd concentration in soils, where the physiology of the root system remains unaffected. Cadmium combines with transporter proteins for Zn2+ and Ca2+ as it passes through the corresponding ion channels for these molecules and enters the layer of the root epidermis through the symplast pathway. Other viable absorption channels of Cd2+ include diffusion to the root surface (Shaaria et al. 2021).
Inside the root, phytochelatins form chelates with Cd2+ through phytochelatin synthases to resist transfer to the shoot (Bayçu 2002). However, many transporters regulate Cd uptakes, e.g., P-type ATPase, natural resistance-associated macrophage protein (NRAMP), Ca2+/cation antiporter (CaCA), and lysosomal cystine transporter (LCT) (Mao et al. 2021). Identifying Cd uptake mechanisms and transporters will enhance the fast and effective elimination of Cd contamination in soils.
Arsenic (As)
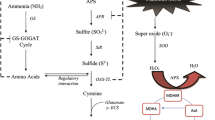
The various forms of As in soils include free ionic species, precipitated as solids, adsorbed on organic or inorganic constituents, and exchangeable and structural composition of primary and secondary minerals (Shahid et al. 2014; Joseph et al. 2015). Inorganic species of As (i.e., AsIII and AsV) are present in different forms, e.g., fully protonated As or arsenous acids (Sadiq 1997). The primary and thermodynamically stable forms of AsIII/V in soils include hydrogen arsenate (HAsO42−), dihydrogen arsenate (H2AsO4−), and arsenous acid (H3AsO3). The presence of As in an inorganic form can depend on pH and redox potential (Eh), and among these, H2AsO4 frequently occurs in aerobic soils (Warren and Alloway 2003). The arsenate form (AsO3−4) of As is more stable and adsorbed quickly to clay minerals and Fe/Mn (hydro)oxides (Khalid et al. 2017a). However, arsenite (As(OH)30) occurs primarily under reduced soil conditions and is more soluble, mobile, and toxic (Khalid et al. 2017a). For example, As mainly exists as arsenate (V) in oxic soil conditions as its main chemical species. Arsenic presents in an anionic form in combination with oxygen, unlike cationic elements (e.g., Cd and Zn) that exist in an independent anionic form (Masscheleyn et al. 1991). Thus, inorganic As species in soils are considered more toxic than their organic counterparts because of their higher solubility, mobility, and rapid absorption. Organic As species in the natural environment include the following: monomethylarsonite (MMAIII), monomethylarsonate (MMAV), dimethyl-arsenite (DMAIII), dimethylarsinate (DMAV), arsenocholine, arsenosugars, arsenobetaine, trimethylarsine oxide, and tetramethyl-arsonium ions (Joseph et al. 2015; Fig. 2).
Diagrammatic representation of uptake pathway, detoxification, and transport of arsenic in the root cell and vascular system of plants. Arsenite—AsIII, arsenate—AsV, monomethylarsonite—MMAIII/V, and dimethylarsenite—DMAIII/V. Cysteine is synthesized from sulfate and is transformed into glutathione by glutathione synthetase (GS). Phytochelatin synthase (PCS) produces phytochelatins from glutathione. Arsenite binds to phytochelatins (AsIII-PCn) and is transported into vacuoles or to the shoot-like arsenite or as a phytochelatin-arsenite complex. Efflux of arsenite from the cell can also be performed by Nodulin-26-like intrinsic protein (NIP) aquaporins. Aquaporin contributes to As uptake. Arsenate reductase (AS) reduces arsenate to arsenite by using glutathione as a reductant
The soil reaction (pH) represents a key variable governing the chemical distribution of metals in soils (Shahid et al. 2012; Khalid et al. 2017b). The biogeochemical behavior of As in the soil–plant system depends mainly on soil pH (Khalid et al. 2017a). Under acidic soil conditions, e.g., below a pH of 5.5, As is highly mobile and phytoavailable (Signes-Pastor et al. 2007). Admittedly, this is due to the transformation of As into a more soluble As fraction (AsIII) at low soil pH. A relative abundance of inorganic As species — AsIII and AsV — varies with soil pH, usually 8–9 and 3–5, respectively (Adra et al. 2016). Again, the pH dependence of AsIII/V adsorption differs in Fe and Al (oxy)hydroxides. The difference in sorption behavior is especially critical at acidic pH. Retention of AsV in ferrihydrite usually occurs at pH 3–5, and for AsIII is at pH 8–9 (Dixit and Hering 2003). Iron oxyhydroxides are generally considered superior As adsorbents than Al hydroxides, with maximum AsV adsorption on amorphous Al hydroxide at pH 4–4.5 (Anderson et al. 1976). Soil redox status has a significant effect on As speciation and geochemical behavior. Usually, the solubility of As increases at a decreasing Eh rate. For example, in an oxidation state, AsV predominates (Khalid et al. 2017a). However, AsV eventually is converted to AsIII at a low Eh value-reduction state (Gorny et al. 2015). Thus, the alteration of oxidation and reduction states of As affect the As solubility greatly. The solubility of As in soils is partly a function of Eh.
The mobility of As in soils increases with an increasing level of DOC, and vice versa in the case of adsorption (Mombo et al. 2016). Soil colloids and metal oxides are crucial adsorption sites for As and amorphous Fe hydroxides (Armendariz et al. 2016). In the process of oxidizing Fe, amorphous Fe oxides form. Arsenic is easily and quickly absorbed to amorphous Fe by ligand exchange of As for the OH and OH2 groups (Finnegan and Chen 2012). Hence, it is vital to consider the bound form of As and the total concentration to assess the status of soil contamination. Other competing ionic species, such as P and Si, affect the mobility of As and bioavailability in soils (Armendariz et al. 2016; Chandrakar et al. 2016).
The mobility, bioavailability, and toxicity of As in the soil–plant system greatly depend on As oxidation states (Shahid et al. 2013). Both AsV and AsIII are highly soluble in H2O and may change valency states depending on the pH (Parvin et al. 2022) and Eh (Marin et al. 1993). In reducing environments, As remains as AsIII, while H3AsO3 is the predominant species. Arsenic in aerated soils often occurs as H2AsO4− (acid soils) or HAsO42− (neutral species and base form) (Moreno-Jiménez et al. 2012). However, in anaerobic soil, HAs3O3 predominates where As availability is higher, and AsIII becomes more weakly retained than the AsV. Changes in environmental conditions can affect the occurrence of As at different oxidation states. The biogeochemical characteristics of As are not entirely dependent on the total concentration (a constituent of stable minerals) in soils (Rafiq et al. 2017) and are generally not bioavailable. The uptake of As by plant species depends on the concentration and its speciation in soils, depending on the exchangeable (bioavailable) level (Martínez-Sánchez et al. 2011; Rafiq et al. 2017).
Arsenic enters plants mainly in an inorganic form (AsIII or AsV) through transporter proteins likely governed by the concentration gradient of As between growth media and plant cells (Neidhardt et al. 2015; Fig. 2). Both AsV and phosphate use the same transporters to cross the plasma membrane of the root cell, e.g., in Brassica napus L. and Brassica juncea (L.) Czern. (Niazi et al. 2017). Arsenic transporters play crucial roles in As uptake, translocation, and detoxification. Several As transporters associated with its accumulation in grains; that is, Carey et al. (2010) reported phloem transport of 55 and 90% of AsIII and DMA, respectively, in Oryza sativa L. grains.
Microbes are vital in the transformation of As species in soils. Environmental microbes have the potential to produce highly toxic AsIII by the process of biomethylation of AsIII or reducing AsV. Microbes have evolved detoxification mechanisms, among which oxidation of AsIII to much less toxic AsV by the ArsH oxidase used in some bacteria (Chen et al. 2015). In As-contaminated soils, organoarsenicals (e.g., MMA, DMA, TMA, TMAO, and TETRA) usually represent a minor component or are not detected in soils (Geiszinger et al. 2002). Arsenic reduction, methylation, and demethylation can occur depending on the type, condition, and microbes of the soil. Under anaerobic conditions, organoarsenicals reduce to volatile arsine, including monomethylarsine (MMAIII), dimethylarsine (DMAIII), and trimethylarsine (TMAIII)) (Zhang et al. 2021). Microbial As methylation is an effective method for As detoxification in micro-organisms and an ideal bioremediation method. However, approaches to recycle volatile or collect methyl As should be developed.
Lead (Pb)
Soil contaminated with Pb oxide results from anthropogenic inputs of leaded fuels, old Pb plumbing pipes, and metallurgy (Retief and Cilliers 2006; Pourrut et al. 2011) and accumulates in the upper soil layers in decreasing concentration with depth (Cecchi et al. 2008). For example, in an abandoned Pb smelter in Příbram, the Czech Republic, the concentration of Pb in the arable layers was up to 2500 mg kg−1 and 400 mg kg−1 in the subsoils (Šichorová et al. 2004). Lead in soils exists as a free metal ion, mostly complexed with inorganic components (e.g., PbCO32–, PbHCO3–, PbSO42–, and PbCl2–), or occurs as organic ligands (e.g., humic and amino acids). Ionic Pb, PbII, Pb oxides and hydroxides, and Pb-metal oxyanion complexes are the general forms released into soils. The most stable forms of Pb are PbII and Pb-hydroxy complexes. The dominant insoluble Pb compounds are Pb-phosphates, Pb carbonates (at pH > 6), and Pb-(hydr)oxides (Yan et al. 2012).
Under anaerobic conditions, volatile organo-Pb (tetramethyl Pb) can form from microbial alkylation (Aten et al. 1980). In addition to the inorganic compounds of Pb, several organo-Pb compounds, such as tetraethyl Pb, are formed. Organo-Pb compounds of commercial and toxicological value are predominantly limited to the alkyl (methyl and ethyl) Pb compounds and their salts (e.g., diethyl-Pb-dichloride, dimethyl-diethyl-Pb, and trimethyl-Pb-chloride). In nature, Pb usually occurs as a mineral combined with other elements, such as S (e.g., PbS and PbSO4) or oxygen (PbCO3) (Wang and Mulligan 2006). Lead (Pb) in soils, in the context of species, solubility, mobility, and bioavailability undergo complex relationships engineered by some biogeochemical factors. The uptake of Pb by plants depends individually or collectively on soil pH, Eh, mineralogy, Pb concentration, organic and inorganic ligand, cation levels, biological and microbial conditions, and plant species (Kopittke et al. 2007; Rizwan et al. 2018). Due to the strong binding of Pb with organic and colloidal materials, only small amounts of Pb in the soil solution are phytoavailable (Punamiya et al. 2010). The bioavailability of Pb directly depends on its speciation and concentration of free Pb ions (Uzu et al. 2009). A significant plant uptake route for many cationic metals (especially free metal ions) is through the soil solution in dissolved form (Punamiya et al. 2010).
Concentrations of free Pb2+ in soils depend on both adsorption or desorption processes in which they participate (Vega et al. 2010; Campillo-Cora et al. 2020). By using surface complexation models, Gustafsson et al. (2011) determined that free Pb adsorption is dominated by OM at soil pH < 6, whereas adsorption in Fe oxides is more prevalent at pH > 6. Soil pH remains vital in determining the free Pb2+ activity in soil solution, as Pb adsorption and most precipitation reactions often are favored by high acidic pH. An increase in pH induces increases in the negative charge of OM that binds with the cationic charge of metals, e.g., Pb, Zn, and Cd. Dissolved OM also controls Pb solubility in soils, forming organo-Pb complexes that increase Pb solubility at high alkaline pH (Sauve et al. 1998). Water-soluble and potentially leachable Pb in non-acid soils often occurs as Pb-DOM complexes (Weng et al. 2002; Ashworth and Alloway 2008). In many contaminated soils, mineral components of Pb, for example, litharge (PbO), hydrocerussite (Pb3 (CO3)2 (OH)2), and cerussite (PbCO3), are stable to limit Pb solubility (Cabała et al. 2020) and pyromorphite (Pb5(PO4)3Cl), form under high phosphate concentration (Scheckel et al. 2005).
Lead is absorbed in the soil solution by plants through the roots and enters the food chain (Sharma and Dubey 2005; Uzu et al. 2009). Absorption of Pb by roots occurs through the apoplastic pathway or permeable channels of Ca2+, further transported by symplastic movement (Fig. 3). Part of Pb in soil solution is adsorbed on roots and bound to carboxyl groups of mucilage uronic acid or directly by polysaccharides on the surface of the cell of the rhizoderm by root exudation (Seregin and Ivanov 2001). There is also possible adsorption of Pb ions on the surfaces of the root, which eventually diffuse into root cells, such as in the cases of Lactuca sativa L. and Vigna unguiculata L. Walp. (Kopittke et al. 2007; Uzu et al. 2009). Additionally, Pb may enter roots in several ways, predominantly through ionic channels. The protective effect of Ca2+ on Pb2+ toxicity relates to its inhibition of the metal accumulation in the root tip, a potential target site of Pb2+ toxicity (Kim et al. 2002).
Schematic representation of the transfer of Pb2+ from the soils to the root and further translocation in plant parts. Translocation of Pb2+ metal ions occurs by apoplastic movement resulting in the deposition of metal ions in the endodermis and is further transported by symplastic movement. DMT1 divalent metal (ion) transporter 1, ROS reactive oxygen species
Although Pb uptake is a non-selective phenomenon, it nevertheless depends on the functioning of an H+/ATPase pump to maintain a negative membrane potential in rhizoderm cells (Wang et al. 2007). However, Ca2+-permeable channels represent one of the main pathways Pb enters roots (Demidchik et al. 2018). Studies on transgenic plants have shown the penetration of Pb in roots through different non-selective pathways, e.g., cyclic nucleotide-gated ion channels (Eapen and D'Souza 2005). The restrictive transport of Pb2+ into the root system indicates that remediation requires a critical selection of plants (hyperaccumulators — above-ground accumulators of metals), uptake high levels of Pb, and translocate to aerial plant parts with no or minimal toxicity. After Pb adsorption on the rhizoderm, root entry can occur passively and follow the translocation of the H2O stream (Kopittke et al. 2007). Non-uniform Pb absorption along the gradient of plant apical roots offers another option for root uptake (Seregin et al. 2004). High Pb concentrations in root apical meristem where root cells are young, with thin cell walls, facilitate Pb root uptake (Seregin et al. 2004). The low rhizodermic pH of the apical area increases Pb solubility in soil solution.
Translocation and speciation of Cd, As, and Pb in plant tissues
The epidermal cell layer in plant species is a well-elaborated system considered an absorbing tissue for ion uptake and membrane transporters. After passing through all blockages on the root surface, metals end up in the root cortex, including the apoplast barrier (in the endodermis). Metal ions enter the symplast, the stele, and the xylem (Broadley et al. 2007; Akhter et al. 2014). Eventually, after a series of passages, metals are loaded into stems.
Transport of Cd in plant tissues
After passing through all barriers from root surfaces over the root cortex, including apoplastic space, e.g., the apical parts where Casparian bands are less developed, Cd2+ enters the symplast and transported to the stele and xylem elements (Redjala et al. 2009; Akhter et al. 2014).
The loading of Cd2+ in the xylem of roots represents one of the vital processes in Cd transport involved in differential Cd accumulation in hyperaccumulating plants (Verbruggen et al. 2009a; Yamaguchi et al. 2011). Translocation of Cd2+ primarily is a function of the retention in roots and loading activity in the xylem (forms part of apoplasmic space) (Verbruggen et al. 2009b). Retention occurs through Cd-chelating molecules, such as apoplastic barriers, phytochelatins, and vacuole sequestration. Competition between sequestration in root cells, e.g., transport from the symplast to the stele and xylem loading, enables most higher plants to accumulate many Cd2+ in roots compared to shoots (Yamaguchi et al. 2011). However, plant hyperaccumulators mostly accumulate a higher concentration of metals in the aboveground parts than in their roots (Verbruggen et al. 2009a). In plant xylem sap, Cd2+ has the potential to form complexes with ligands (Ana et al. 2014). These complexes, with free Cd2+, are transported to the aerial parts of the plant with the flow of xylem sap driven by transpiration. Hyperaccumulation of As defined as a concentration above (in mg kg−1) 1000 was in ferns growing on industrially polluted soils (Reeves 2006), 8350 for Pityrogramma calomelanos L. (Visoottiviseth et al. 2002), and 22,630 also for Pteris vittata L. (Ma et al. 2001). Several species of the Brassicaceae family, such as Thlaspi caerulescens J., accumulated 3000 mg kg−1 (Reeves et al. 2001). Free Cd2+ also interacts with the cell walls of xylem vessels and partly adsorbs on them or can migrate through the cell walls of the xylem to stem and leaf cells, mainly under the influence of transpiration flow (Sterckeman and Thomine 2020).
Additionally, Cd2+ can re-enter the apoplast before loading into xylem vessels upon reaching the stele (Hu et al. 2009; Sterckeman and Thomine 2020). For instance, Akhter et al. (2014) recorded high Cd concentration in the walls of xylem vessels in Hordeum vulgare L. Furthermore, Cd2+ in stele can coordinate with S and acetate ligands. The process suggests the possibility of Cd2+ complexation with phytochelatins (heavy-metal-binding peptide) and carboxylic acids in vacuoles and subsequent binding to cell walls. Chelation and sequestration of metals by particular ligands are approaches plants use to cope with stress. Higher plants respond to heavy metals by synthesizing phytochelatins and related cysteine. For example, phytochelatin synthases and γ-glutamylcysteine (γ-Glu-Cys) catalyze the synthesis of heavy metal binding (Yadav 2010).
The tendency to move Cd2+ from the xylem parenchyma to xylem vessels depends on the activities of transporters (Mori et al. 2009). A difference in the ability of transporters to load Cd2+ into the xylem is vital in determining the differences between Cd concentrations in the xylem sap (Mori et al. 2009). Yamaguchi et al. (2011) detected the differences in Cd2+ uptake into stem xylem in cultivars of Solanum torvum Sw reflected differences in membrane transporters. Mendoza-Cózatl et al. (2008) identified high concentrations of phytochelatins, glutathione, and Cd2+ in the phloem sap of B. napus. Thus, the phloem is also a conduit for long-distance source-to-sink transport of Cd2+. Identified transporters/protein-regulated Cd include the P-type ATPase family and HMA subfamily (heavy metal ATPases), involved in the transmembrane transfer of ions belonging to the symplastic pathway (Maccaferri et al. 2019).
Cadmium forms chelate with organic ligands in the leaf and migrate within cells (Yamaguchi et al. 2011). Free ions of the complexes enter the cell vacuole for storage as chelates. In the shoot, Cd2+ distributes in the various units of the cell and tissues (Sterckeman and Thomine 2020). Additionally, part of this Cd2+ redistributes to other organs (e.g., the reproductive parts) and through the phloem into the root system. Excess Cd can also preferentially excrete via the leaf surface (e.g., through hydathodes) to avoid toxicity (Dong et al. 2019).
Transport of As species in plant tissues
Transports of different As species from roots have been well-studied among plants (Zhao et al. 2009; Huang et al. 2011). Xylem tissues are directly involved in the root translocation of As and subsequent distribution among various plant tissues (Kumar et al. 2015). Arsenate is reduced to AsIII in the plant cell with the assistance of As reductase (Yoshinaga et al. 2011). For example, As hyperaccumulating plant species, P. vittata can accumulate AsV and transport more AsIII through the xylem with less tendency for AsIII-thiol complex formation (Zhou et al. 2000).
Arsenite efflux mainly occurs via aquaporin channels through the epidermal cells (Fig. 2). Typical protein-coding in the transport of AsIII and As-related substances includes Nodulin 26-like intrinsic protein, OsNIP2;1- Si-influx transporters mostly in O. sativa As-transport (Yan et al. 2021), and other transporters in the aquaporin family (Bienert et al. 2008). Additionally, the ability to load As directly into the xylem affects the shoot accumulation in many plants. The uptake rate of methylated organic As species is slower than those of inorganic As species (Agre and Kozono 2003). Meanwhile, the translocation of methylated As species from root to shoot via the xylem is fast (Raab et al. 2007a; Shakoor et al. 2015). Moreover, in xylem and phloem tissues, methylated As species are often more mobile than their inorganic counterparts (Carey et al. 2010).
Some organic As species get transported from soil to the xylem stream through the Si influx and efflux transporters (Li et al. 2009; Mitani-Ueno et al. 2011). Influx Si (e.g., Lsi1) transporter allows the bidirectional (inclusion and exclusion) flux of Si and AsIII between soil and plant roots cells, while Lsi2, an efflux, involves in the exclusion of Si/AsIII into the stele and xylem tissues (Khalid et al. 2017a; Yibin et al. 2019). A hydroponic experiment conducted by Raab et al. (2007b), using over 40 species of Zea mays L., showed that the transport of DMAV from root to shoot was about 10 and 3 times greater than MMAV and AsV, respectively. Thus, As species in plants reflect differential transport mechanisms.
Trivalent As is considered more toxic than Asv as it reacts like a soft metal with thiols (Ramírez-Solis 2004; Fig. 2). Arsenite may form relatively weak bonds with monothiols, and high intracellular concentrations of AsIII can deplete glutathione cells. It forms strong bonds with dithiols in small molecules, e.g., lipoic acid cofactor, and with vicinal thiols in proteins, leading to the inactivation of various enzymes and receptors (Ramírez-Solis 2004). In contrast to AsV, a negatively charged oxyanion in solution, AsIII, is the neutral undissociated acid As(OH)3 at physiological pH.
Meanwhile, the transports of these substrates across the aquaporins operate mainly by two pores, the conserved asparagine-proline-alanine boxes and aromatic/arginine (Maurel et al. 2008). Nodulin-26-like intrinsic protein (NIP) aquaporins can also perform the efflux of As from plant cells. All NIPs have different pore structures at arginine selective filters. NIPs are into subgroups: (1) archetype nodulin-26 (permeable to C3H8O3, C3H6O3, and H2O), (2) a pore size larger than archetype nodulin-26 (permeable to large solutes, e.g., CH3NO, H3BO3, and CH4N2O but less permeable to H2O) (Wallace et al. 2006), and (3) finally for the transport of silicic acid (Si(OH)4). The AsIII species is permeable to all NIP subgroups and transported by Si influx and efflux transporters (Panda et al. 2010).
Arsenic accumulation also depends on plant species. For example, Tremlová et al. (2017) observed a higher accumulation of As by Equisetum fluviatile L. than by Juncus filiformis L. living under similar conditions. The difference in As accumulation probably depends on different regulations of the phosphate transporters or the aquaglyceroporins involved in the cellular uptake of both As species. According to Tlustoš et al. (2002), radish has AsIII dominated in roots, while AsV translocates to the leaves. Arsenic speciation in the phloem of many plants and its role in As accumulation remain largely uncharacterized.
The translocation efficiency of As from roots to shoots affects the tolerance of plants and determines the ability of hyperaccumulators to accumulate As (Verbruggen et al. 2009a). The reduction of AsV to AsIII efficiently in hyperaccumulator plants is often reported. Thus, AsIII predominates in root-to-shoot transport, whether there is AsV or AsIII supply to the plants (Raab et al. 2007a, b). It is, therefore, convenient to know the predominant As species to select the most suitable plant accumulator for effective phytoremediation.
Transport of Pb in plant tissues
Lead accumulates primarily in root cells because of the blockage by Casparian strips (Dogan et al. 2018). Unlike As, Pb shows a more translocation restriction phenomenon. Thus, a higher fraction of accumulated Pb remains in roots compared to the aerial plant parts (Kiran and Prasad 2017). And this is in support of negative charges that exist on root cell walls that trap Pb ions (Pourrut et al. 2011; Tariq 2018). Thus, hyperaccumulation of Pb is often restricted in many plants and generally dominates in the root (Probst et al. 2009; Fahr et al. 2013). A comparative analysis of different fruiting parts of plants indicated that root edibles accumulate the highest Pb concentration in aboveground organs (Fahr et al. 2013).
Deposition of Pb in various organelles of plants and vascular tissues in the root system of plants confirms the retention of Pb in this section (Yuan et al. 2015; Li et al. 2016). However, the translocation of Pb to aboveground biomass is concentration and duration dependent, e.g., in B. juncea and Neyraudia reynaudiana (Kunth) (Zhou et al. 2016; Kohli et al. 2018). Root retention of Pb partly involves the binding of Pb to ion exchange on the cell wall and extracellular precipitation in the form of Pb CO2−3. Meanwhile, the un-precipitated Pb ions intermittently are transported to other parts of cells via pleiotropic drug resistance, mitochondrial inner membrane protein, and ATP-binding cassette transporters (Lee et al. 2005; Fig. 3). The synthesis of phytochelatins, peptides, and organic acids forms chelates with Pb upon entry into root cells to tolerate and toxify Pb (Hall 2002; Patra et al. 2004). Active efflux of Pb across the plasma membrane is known to be mediated by specific transporters (Kaur et al. 2013; Jiang et al. 2017). Other transporters contribute to Pb resistance by regulating the transport of Pb outside cells, e.g., leucine-rich repeat protein (Zhu et al. 2013).
In leaf tissues, Pb is well-reflected near the xylem, phloem, sclerenchyma, and mesophyll cells; indication of transport to the aerial parts requires translocation via xylem vessels (Verbruggen et al. 2009b). Again, Pb can form complexes with minerals (e.g., P, Ca, and S) (Zheng et al. 2012) and organic or amino acids while passing through the xylem tissues (Maestri et al. 2010). Lead translocation could occur through the phloem (Rascio and Navari-Izzo 2011), confirmed through an X-ray mapping. In this case, a high fraction of Pb deposition occurred near the xylem and phloem cells of Pb-treated Prosopis sp. (Arias et al. 2010).
Interaction of risk elements with plant physiological and metabolic processes
Excess As, Cd, and Pb can change the expression of vital enzymes in the respiratory process and eventually lead to physiological changes in plants. In addition, stress from risk elements can also result in reduced shoot length and biomass, decreased respiratory rate, and reduced crop quality. Plant physiological disorders under risk element stresses can increase ROS. Examples of ROS include free oxygen radicals — superoxide (O2−), hydroxyl (OH), and hydrogen peroxide (H2O2); peroxyl radical (ROO); and non-radicals — H2O2, 1O2, and ozone (O3) (Riley 1994; Okem et al. 2015). Reactive oxygen species are dangerous for plant metabolism and cause detrimental effects on vital macromolecules, e.g., proteins, carbohydrates, lipids, and DNA (Singh et al. 2017; Talukdar 2017).
Meanwhile, plants undergo a series of internal modifications that include metabolic secretion to protect them from the toxicity of risk elements (Michalak 2006; Okem et al. 2015). These mechanisms, e.g., production and accumulation of antioxidant enzymes, free-proline, glutathione, and phenolics, protect plants from free radicals and prevent biomolecule damage (Michalak 2006; Bhattacharya et al. 2010). Phenolic compounds have antioxidative and metal-chelating properties and, thus, play a vital role in protecting plants against metal-induced stresses (Tatipamula and Kukavic 2021). The concentration of metals is a function of the level of phenolics (Melato et al. 2012). Hence, high-risk element concentrations enhance the production of phenolic compounds. Meanwhile, an increased amount of cell wall-bound phenolics signifies a mechanism for detoxification.
Phenolics and other plant secondary metabolites have pharmaceutical significance due to their properties (Dai and Mumper 2010; Melato et al. 2012). Thus, a high accumulation of risk elements can affect the quality, quantity, or composition of phytochemicals, questioning the effectiveness of medicinal plant/herb products. For example, excess treatments with Cd, Pb, and Cu altered the chemical composition of essential oils of Ocimum basilicum L. (Prasad et al. 2011) and dill and reduced the menthol content in peppermint oil (Zheljazkov et al. 2006). Meanwhile, many plants have different levels of toxicity to stress from metals. According to Stancheva et al. (2005), different levels of (in mg kg−1) Cd — 3, Pb — 80, and Zn — 340 only resulted in sage biomass inhibition, but essential oil yield and quality remained unaffected.
Formation of secondary metabolites under risk element stress
Plant secondary metabolite formations are plant/stress-specific. Meanwhile, secondary metabolites are known to develop from primary metabolites (precursors) in plants (Aharoni and Galili 2011). At molecular, anatomical, and morphological levels in plants, varieties of tolerance or adaptation mechanisms develop, including interrelated gene networking (Goharrizi et al. 2022). Different plants exhibit varying biosynthesis pathways during secondary metabolite formation. For instance, putative transcription factors in the formation of glandular secretory trichomes and the biosynthesis of artemisinin include ENHANCER OF GLABRA3 (Aa-EGL3) and TRANSPARENT TESTA GLABRA1 (Aa-TTG1) (Guerriero et al. 2018).
Risk element uptake by plants affects photosynthetic pigments, sugars, proteins, and non-protein production. Reactive oxygen species in plants are part of the metabolism of chloroplasts, mitochondria, and peroxisomes. However, high exposure to risk elements can overwhelm the systems protecting the plants and result in oxidative stress. Oxidative stress then results from an imbalance between the production and accumulation of reactive oxygen species.
The main effects of risk element-induced oxidative stress in cells ignite the enzymatic response and metabolite production (Fig. 4). For example, Cd, a non-redox metal, can indirectly increase the production of reactive oxygen species (ROS) in plants, which elevates free oxygen radicals. In a proteomic analysis of alfalfa stem exposed to Cd, Gutsch et al. (2018) observed that an abundance of peroxidases and their corresponding genes remained upregulated after stress treatment. Additionally, soybean and lupin exposed to 15–25 mg Cd kg−1 and 150–350 mg Pb kg−1 induced phenylalanine ammonia-lyase activities. However, the increase did not correlate with the induction of gene expression and lignin accumulation in lupin (Pawlak-Sprada et al. 2011). Thus, this indicates the presence of transcriptional and posttranscriptional regulation and involvement of activated phenylpropanoid pathway in the synthesis of secondary metabolites than lignin building blocks.
Processes involved in the formation of plant secondary metabolite under heavy metal stress. The stress triggers a ROS burst, which exerts an induction on phenylalanine ammonia-lyase (PAL) (stress-responsive enzymes). The phenylpropanoid pathway is boosted with the subsequent production of, e.g., phenolic acids and flavonoids. These secondary metabolites, together with the enzymatic antioxidant system, contribute to counterbalancing ROS. At the cell, an increased lignification is observed, as well as a modification of the pectins (increase in acidic pectins) which can sequester risk elements through the “box” structure. PME: pectin methylesterase; SOD: superoxide dismutase; GST: glutathione-S-transferase; CAT: catalase (adapted and modified from Berni et al. 2018)
Impact of excess Cd, As, and Pb on secondary plant metabolites
Plants exposed to excess Cd, As, and Pb exhibit a marked effect on metabolic activities. Excess Pb destroys chloroplast ultrastructure, prevents electron and nutrient transport, and seizes CO2 entry by stomatal closure (Srivastava et al. 2014; Kumar et al. 2015).
Cadmium
Cadmium phytotoxicity has inhibitory effects on the metabolism of some micro-nutrients, photosynthesis, disturbs transpiration, and fixation of CO2 (Prasad 2005). Under an increasing concentration of Cd, different plant species exhibit differential accumulations of specific metabolites. For example, a high Cd load of 83 mg kg−1 caused an increase in proline levels in Drimia elata Jacq. (Okem et al. 2015). In D. elata bulbs, Okem et al. (2015) reported a reduction in chlorophyll, phenolic, and flavonoid concentrations under increasing Cd loads. Reduced chlorophyll content under Cd stress affects plant physiology, as photosynthetic electron transport processes are altered, suppressing energy transformation (Paunov et al. 2018) and microbial activities.
The low phenolic and flavonoid levels can partly result from the sudden halt of electron transport. Meanwhile, decreased secondary metabolite levels under the risk element stress of plants result in poor antimicrobial activity (Ibrahim et al. 2017).
Under the same loads of excess risk elements, plants exert differential accumulation and production of specific metabolites. In the analysis of metabolic profiles of two Cynodon dactylon L. genotypes exposed to the same level of Cd stress, Xie et al. (2014) recorded different levels of amino acids (glutamic acid, gulonic acid, glycine, norvaline, proline, serine, and threonine), organic acids (citric, glyceric, malic, and oxoglutaric acids), and sugars (galactose, talose, and xylulose). The differential accumulations indicate different tolerance to Cd by the two genotypes, especially in the case of proline production. Notably, the reduction of some acids during Cd stress, e.g., aspartic acid, regardless of plant species and genotype, partly relates to the buffering effects through modulations of other amino acids (Chaffei et al. 2004; Xu et al. 2012).
In plants, proline production represents the most common stress marker, vital in alleviating Cd toxicity (Kishor et al. 2005; Liang et al. 2013). During risk element detoxification, proline scavenges ROS and forms chelates, which increase glutathione and protect antioxidant enzyme activities (Perveen et al. 2012). Proline synthesis often occurs in plants’ leaves as organs for the photoactivation of vital enzymes (Arora and Saradhi 1995). Additionally, proline can act as an osmoprotectant when plants are stressed. In this case, proline offsets water deficit during metal stress in many plants by stomatal closure. Notable, this inactivates processes: metal translocation and transpiration (Liang et al. 2013). Hence, what then becomes the fate of plants and other metabolites on increasing loads of risk elements? However, increased or decreased metabolites under stress from risk elements are a symptom of damage. Thus, metabolites need critical elemental analysis before use. Consumable metabolites can contain < 0.3 mg kg−1 Cd, according to WHO (2007), to avoid detrimental health effects on humans.
Stress induced by, e.g., drought and risk elements in plants can regulate the biosynthesis of amino acids (Curtis et al. 2018). The biosynthesis and accumulation of asparagine, an amino acid derivative, represent an example of a Cd stress plant (Okem et al. 2015). Asparagine provides a high N:C used by plants as an N storage compound to ensure future recovery (Chaffei et al. 2004). Meanwhile, high asparagine can participate in detoxification processes or the biosynthetic way of chelating peptides (Costa and Spitz 1997). Some secondary metabolites are involved in stress defense, e.g., isoprenoids, phenylpropanoids, alkaloids, or polypeptide pathways (Dixon 2001). Xu et al. (2012) found an accumulation of branched-chain amino acids (e.g., isoleucine and valine) in Solanum nigrum L. roots under high Cd accumulation and tolerance. However, under Cd stress, isoleucine and valine contents can increase for Cd-sensitive plants but unchanged for Cd-tolerant (Okem et al. 2015), suggesting that two different stress defense pathways exist in different genotypes. According to Mahajan et al. (2020), Cd increases diosgenin content in Trigonella foenum-graecum L. The accumulation of sesquiterpenoids (lubimin and 3-hydroxylubimin) is high in Datura stramonium L. treated with Cd salts (Furze et al., 1991).
Arsenic
The most dangerous biochemical effect of As at the subcellular level is the production of ROS. Under As stress, many plants show changes in metabolome and reduce trends within the chloroplast, e.g., fronds of the hyperaccumulator plant Pteris cretica L. (Zemanová et al. 2020). Melato et al. (2012) reported a decrease of > 35% in the length of Chrysopogon zizanioides L. plants treated with As compared to an identical plant without As stress. Total soluble phenolic content in C. zizanioides increased with an increasing concentrations of As, Cr, Cu, Ni, Pb, and Zn in the growth medium. During As uptake by plants, some of the AsV taken up by cells eventually convert to AsIII employing arsenate reductase (Kalita et al. 2018). The AsIII forms a complex with thiol groups in proteins, inhibiting arsenate reductase activities (Hu et al. 2020). Thus, excess AsIII may probably be more deleterious to the quality of secondary metabolite. However, this is so far not well-studied.
Plants produce secondary metabolites as a response to adaptation. The accumulation of metabolites can represent a function of increasing environmental (abiotic) stress conditions. For example, Withania somnifera L. plants tolerated high concentrations of trivalent/tetravalent As species until 5 and 10 days by increasing antioxidant enzymes and synthesis of thiols, especially in the roots (Siddiqui et al. 2015). Phenolics and flavonoids can scavenge active oxygen species due to their ability to donate H atoms or electrons (Okem et al. 2015). In a combined application of methyl jasmonate to As-stressed, B. napus increased leaf chlorophyll fluorescence, biomass production, and reduced malondialdehyde content, compared to the same plant under only As stress (Farooq Muhammad et al. 2016). Methyl jasmonate regulates plant defense against abiotic stress and produces antioxidants and secondary metabolites (Ahmed et al. 2015). Under induced methyl jasmonate, As-stressed B. napus cultivars observed enhanced phenylalanine ammonia-lyase, polyphenol peroxidase, and antioxidants (Farooq Muhammad et al. 2016). These compounds stimulate the phenylpropanoid pathway and produce derivatives such as phenols and flavonoids, which play a vital role in minimizing oxidative stress and As concentration in active reproductive parts, e.g., root and leaf (Wang et al. 2011).
Lead
Lead toxicity inhibits ATP production, lipid peroxidation, and DNA damage by ROS over-production (Hou et al. 2018; Melato et al. 2012).
Plants exhibit defense mechanisms to cope under Pb stress, e.g., reduced uptake into cells, sequestration into vacuoles by forming complexes, binding by phytochelatins, glutathione, and amino acids, including the synthesis of osmolytes (Yuan et al. 2015). Notwithstanding, Pb affects photosynthetic mechanisms by inhibiting enzyme activities such as δ-aminolaevulinic acid dehydratase (Gupta et al. 2009; Cenkci et al. 2010) involved in enzymatic pathways of photosynthetic pigments. Inhibition of chlorophyll biosynthesis leads to reductions in pigment contents and alteration in chlorophyll fluorescence (Hou et al. 2018). An increase in phenolic content in Phaseolus vulgaris (Wall.) with increasing Pb concentrations indicated the prevention of oxidative damage by scavenging active oxygen species and breaking the radical chain reactions during lipid peroxidation.
Metabolomics often is dedicated to a comprehensive analysis of organic metabolites within cells and tissues. Due to the beneficial properties of metabolites, the interest in characterization and quantification is pertinent, resulting in the identification and usage of different instrumental methods. Different physicochemical methods are applicable in analyzing plant metabolites (Table 2). Moreover, the application of elicitors in plant cultures triggers the large-scale production of secondary metabolites and is vital in the pharmaceutical and therapeutic industries. The selection of sites for unpolluted soils needs close monitoring to ensure the safety and efficacy of cultivated plants.
Conclusions
The study revealed that high levels of risk elements above permissible limits in soils affect the metabolic activities of plants, including the production of secondary metabolites after the uptake of Cd, As, and Pb. The accumulation of risk elements is associated with different plant tissues according to their functions: (i) absorbing tissues responsible for the entry of Cd, As, and Pb from soils through root rhizoderm; (ii) without competing ions, risk elements flow through soils into roots in all root zones. However, tissues functioning as barriers (e.g., Casparian strips) restrict the radial transport in roots. Meanwhile, these appear in mature cells, with modifications of cell walls for some metals; (iii) and finally, to the metal-accumulating and metal-containing tissues, plants uptake only accessible Cd, As, and Pb in exchange complexes (organic and inorganic components) and soluble fractions in the soil solution. However, the mobility and bioavailability of the risk elements to plants often get eased by factors such as pH, redox condition, DOC, clay content, and metal oxides.
Plants counteract the toxic effects of Cd, As, and Pb by engaging antioxidant enzymes and proline, glutathione, and phenolic compound production to protect them against free radicals and biomolecule damage. Phytotoxicity by risk elements has inhibitory effects on the metabolism of nutrient photosynthesis, disturbs transpiration, and fixation of CO2. Excess risk elements cause chlorophyll reduction, increased free proline, and secondary metabolites (e.g., phenolics, flavonoids, alkaloids, and terpenes).
Decreased plant secondary metabolite contents result in poor antimicrobial activities of plants grown on risk element-contaminated sites. Risk element contamination of soils alters the physicochemical state of plants as they adapt to stress conditions. Eventually, excess risk elements affect the synthesis of secondary metabolites, consequently influencing the quality and efficacy of medicinal products by decreasing antimicrobial properties.
Knowledge gap
Limited information exists on specific physiological responses of medicinal plants to heavy metals in soils and the resulting changes in the efficacy of plant metabolites. With the increasing rate of metalliferous activities, many plants, including medicinal herbs, can accumulate a high concentration of metals. However, there are many existing gaps on the effects of other metal-(loid)s, e.g., Au, Ag, Ni, and Hg, with little or no detailed proteomic analysis of secondary metabolites and their derivatives developed by plants in affected areas. Finally, the optimization of both AsIII and Asv species determine the intensity of their deleterious effects on secondary metabolites, especially medicinal herbaceous plant species.
Data availability
The authors declare that data supporting the findings of this study are available within the article.
References
Adra A, Morin G, Ona-Nguema G, Brest J (2015) Arsenate and arsenite adsorption onto Al-containing ferrihydrites. Implications for arsenic immobilization after neutralization of acid mine drainage. Appl Geochem 64:2–9. https://doi.org/10.1016/j.apgeochem.2015.09.015
Agre P, Kozono D (2003) Aquaporin water channels: molecular mechanisms for human diseases. FEBS Lett 555:72–78. https://doi.org/10.1016/S0014-5793(03)01083-4
Aharoni A, Galili G (2011) Metabolic engineering of the plant primary–secondary metabolism interface. Curr Opin Biotechnol 22:239–244. https://doi.org/10.1016/j.copbio.2010.11.004
Ahmed IM, Nadira UA, Bibi N, Cao F, He X, Zhang G, Wu F (2015) Secondary metabolism and antioxidants are involved in the tolerance to drought and salinity, separately and combined, in Tibetan wild barley. Environ Exp Bot 111:1–12. https://doi.org/10.1016/j.envexpbot.2014.10.003
Akhter MF, Omelon CR, Gordon RA, Moser D (2014) Localization and chemical speciation of cadmium in the roots of barley and lettuce. Environ Exp Bot 100:10–19. https://doi.org/10.1016/j.envexpbot.2013.12.005
Ali W, Mao K, Zhang H, Junaid M, Xu N, Rasool A, Feng X, Yang Z (2020) Comprehensive review of the basic chemical behaviors, sources, processes, and endpoints of trace element contamination in paddy soil-rice systems in rice-growing countries. J Hazard Mater 397:122720. https://doi.org/10.1016/j.jhazmat.2020.122720
Aliferis KA, Chamoun R, Jabaji S (2015) Metabolic responses of willow (Salix purpurea L.) leaves to mycorrhization as revealed by mass spectrometry and 1H NMR spectroscopy metabolite profiling Front Plant Sci 6. https://doi.org/10.3389/fpls.2015.00344
Ana Á-F, Pablo D-B, Anunciación A, Ana-Flor L-M, Javier A (2014) Metal species involved in long-distance metal transport in plants. Front Plant Sci 397:122720. https://doi.org/10.3389/fpls.2014.00105
Anderson MA, Ferguson JF, Jerome Gavis J (1976) Arsenate adsorption on amorphous aluminum hydroxide. J Coll Inter Sci 54:391–399. https://doi.org/10.1016/0021-9797(76)90318-0
Arias JA, Peralta-Videa JR, Ellzey JT, Ren M, Viveros MN, Gardea-Torresdey JL (2010) Effects of Glomus deserticola inoculation on Prosopis: enhancing chromium and lead uptake and translocation as confirmed by X-ray mapping, ICP-OES, and TEM techniques. Environ Exp Bot 68:139–148. https://doi.org/10.1016/j.envexpbot.2009.08.009
Armendariz AL, Talano MA, Villasuso AL, Travaglia C, Racagni GE, Reinoso H, Agostini E (2016) Arsenicstress induces changes in lipid signaling and evokes the stomata closure in soybean. Plant Physiol Biochem 103:45–52. https://doi.org/10.1016/j.plaphy.2016.02.041
Arnold PA, Kruuk LE, Nicotra AB (2019) How to analyze plant phenotypic plasticity in response to a changing climate. New Phytol 222:1235–1241. https://doi.org/10.1111/nph.15656
Arora S, Saradhi PP (1995) Light-induced enhancement in proline levels in Vigna radiata exposed to environmental stresses. Aust J Plant Physiol 22:383–386
Ashworth DJ, Alloway BJ (2008) Influence of dissolved organic matter on the solubility of heavy metals in sewage-sludge-amended soils. Commun Soil Sci Plant Anal 39:538–550
Aten GF, Bourke JB, Martini JH, Walton JC (1980) Arsenic and lead in an orchard environment. Bull Environ Contam Toxicol 24:108–115
Aydinalp C, Marinova S (2003) Distribution and forms of heavy metals in some agricultural soils. Polish J Environ Stud 12:629–633
Bayçu G (2002) Phytochelatin biosynthesis and cadmium detoxification. J Cell Mol Biol 1:45–55
Berni R, Luyckx M, Xu X, Legay S, Sergeant K, Hausman J-F, Lutts S, Cai G, Guerrierod G (2018) Reactive oxygen species and heavy metal stress in plants: impact on the cell wall and secondary metabolism. Environ Exp Bot. https://doi.org/10.1016/j.envexpbot.2018.10.017
Bhattacharya A, Sood P, Citovsky V (2010) The roles of plant phenolics in defense and communication during Agrobacterium and Rhizobium infection. Mol Plant Pathol 11:705–719
Bhuiyan FR, Howlader S, Raihan T, Hasan M (2020) Plants metabolites: possibility of natural therapeutics against the COVID-19 pandemic. Front Med 7:444. https://doi.org/10.3389/fmed.2020.00444
Bienert GP, Thorsen M, Schüssler MD, Nilsson HR, Wagner A, Tamás MJ, Jahn TP (2008) A subgroup of plant aquaporins facilitate the bi-directional diffusion of As(OH)3 and Sb(OH)3 across membranes. BMC Biol 6:26. https://doi.org/10.1186/1741-7007-6-26
Broadley MR, White PJ, Hammond JP, Zelko I, Lux A (2007) Zinc in plants. New Phytol 173:677–702
Cabała J, Warchulski R, Rozmus D, Środek D, Szełęg E (2020) Pb-rich slags, minerals, and pollution resulted from a medieval Ag-Pb smelting and mining operation in the Silesian-Cracovian region (Southern Poland). Minerals 10:28. https://doi.org/10.3390/min10010028
Campillo-Cora C, Conde-Cid M, Arias-Estévez M, Fernández-Calviño D, Alonso-Vega F (2020) Specific adsorption of heavy metals in soils: individual and competitive experiments. J Agron 10:1113. https://doi.org/10.3390/agronomy10081113
Carey A-M, Scheckel KG, Lombi E, Newville M, Choi Y, Norton GJ, Charnock JM, Feldmann J, Price AH, Meharg AA (2010) Grain unloading of arsenic species in rice. Plant Physiol 152:309–319. https://doi.org/10.1104/pp.109.146126
Cecchi M, Dumat C, Alric A, Felix-Faure B, Pradere P, Guiresse M (2008) Multi-metal contamination of a calcic cambisol by fallout from a lead-recycling plant. Geoderma 144:287–298
Cenkci S, Cigerci IH, Yildiz M, Özay C, Bozdag A, Terzi H (2010) Lead contamination reduces chlorophyll biosynthesis and genomic template stability in Brassica rapa L. Environ Exp Bot 67:467–473
Chaffei C, Pageau K, Suzuki A, Gouia H, Ghorbel MH, Masclaux-Daubresse C (2004) Cadmium toxicity induced changes in nitrogen management in Lycopersicon esculentum leading to a metabolic safeguard throughan amino acid storage strategy. Plant Cell Physiol 45:1681–1693
Chandrakar V, Dubey A, Keshavkant S (2016) Modulation of antioxidant enzymes by salicylic acid in arsenic exposed Glycine max L. J Soil Sci Plant Nutr 16:662–676. https://doi.org/10.4067/S0718-95162016005000048
Chen P-H, Peng C-Y, Pai H-C, Teng C-M, Chen C-C, Yang R (2010) Denbinobin suppresses breast cancer metastasis through the inhibition of Src-mediated signaling pathways. J Nutr Biochem 22:732–740. https://doi.org/10.1016/j.jnutbio.2010.06.004
Chen J, Bhattacharjee H, Rosen BP (2015) ArsH is an organoarsenical oxidase that confers resistance to trivalent forms of the herbicide monosodium methyl arsenate and the poultry growth promoter rox-arsone. Mol Microbiol 96:1042–1052
Costa G, Spitz E (1997) Influence of cadmium on soluble carbohydrates, free amino acids, protein content of in vitro cultured Lupinus albus. Plant Sci 128:131–140
Cui H, Zhang X, Wu Q, Zhang S, Xu L, Zhou J, Zheng X, Zhou J (2020) Hematite enhances the immobilization of copper, cadmium, and phosphorus in soil amended with hydroxyapatite under flooded conditions. Sci Total Environ 708:134590
Curtis TY, Bo V, Tucker A, Halford NG (2018) Construction of a network describing asparagine metabolism in plants and its application to the identification of genes affecting asparagine metabolism in wheat under drought and nutritional stress. Food Energy Sec 7:e00126
Dai J, Mumper RJ (2010) Plant phenolics: extraction, analysis and their antioxidant, and anticancer properties. Molecules 15:7313–7352. https://doi.org/10.3390/molecules15107313
De Luca V, Salim V, Atsumi SM, Yu F (2012) Mining the biodiversity of plants: a revolution in the making. Science 336:1658–1661. https://doi.org/10.1126/science.1217410
Demidchik V, Shabala S, Isayenkov S, Cuin TA, Pottosin I (2018) Calcium transport across plant membranes: mechanisms and functions. New Phytol 220:49–69. https://doi.org/10.1111/nph.15266
Dixit S, Herring JG (2003) Comparison of arsenic(V) and arsenic(III) sorption onto iron oxide minerals:implications for arsenic mobility. Environ Sci Technol 37:4182–4189. https://doi.org/10.1021/es030309t
Dixon RA (2001) Natural products and plant disease resistance. Nature 411:843–847
Dogan M, Karatas M, Aasim M (2018) Cadmium and lead bioaccumulation potentials of an aquatic macrophyte Ceratophyllum demersum L.: a laboratory study. Ecotoxicol Environ Saf 148:431–440
Dong Q, Fei L, Wang C, Hu S, Wang Z (2019) Cadmium excretion via leaf hydathodes in tall fescue and its phytoremediation potential, Environmental Pollution 252. Part B 2019:1406–1411. https://doi.org/10.1016/j.envpol.2019.06.079
Eapen S, D’Souza SF (2005) Prospects of genetic engineering of plants for phytoremediation of toxic metals. Biotechnol Adv 23:97–114. https://doi.org/10.1016/j.biotechadv.2004.10.001
Edreva A, Velikova V, Tsonev T, Dagnon S, Gürel A, Aktaş L, Gesheva E (2008) Stress-protective role of secondary metabolites: diversity of functions and mechanisms. Gen Appl Plant Physiol 34:67–78
European Council Directive (1986) 86/278/EEC on the protection of the environment, and in particular of the soil when sewage sludge is used in agriculture. Retrieved from https://op.europa.eu/en/publicationdetail/-/publication/f76faa39-2b27-42f2-be1e-9332f795e324/language-en. Accessed on 20 Oct 2022
Fahr M, Laplaze L, Bendaou N, Hocher V, El Mzibri M, Bogusz D, Smouni A (2013) Effect of lead on root growth. Front Plant Sci 4:175. https://doi.org/10.3389/fpls.2013.00175
Farooq Muhammad A, Gill Rafaqat A, Faisal I, Basharat A, Hongbo L, Jianxiang X, Huiping H, Weijun Z (2016) Methyl Jasmonate regulates antioxidant defense and suppresses arsenic uptake in Brassica napus L. Front Plant Sci 7. https://doi.org/10.3389/fpls.2016.00468
Finnegan PM, Chen W (2012) Arsenic toxicity: the effects on plant metabolism. Front Physiol 3:182. https://doi.org/10.3389/fphys.2012.00182
Finster ME, Gray KA, Binns HJ (2004) Lead levels of edibles grown in contaminated residential soils: a field survey. Sci Total Environ 320:245–257
Furze JM, Rhodes MJC, Parr AJ et al (1991) Abiotic factors elicit sesquiterpenoid phytoalexin production but not alkaloid production in transformed root cultures of Datura stramonium. Plant Cell Rep 10:111–114. https://doi.org/10.1007/BF00232039
García-Calderón M, Pons-Ferrer T, Mrazova A, Pal'Ove-Balang P, Vilkova M, Pérez-Delgado C, Vega J, Eliašová A, Repčák M, Márquez A, Betti M (2015) Modulation of phenolic metabolism under stress conditions in a Lotus japonicus mutant lacking plastidic glutamine synthetase. Front Plant Sci 6. https://doi.org/10.3389/fpls.2015.00760
Geiszinger A, Goessler W, Kosmus W (2002) Organoarsenic compounds in plants and soil on top of an ore vein. Appl Organomet Chem 16:245–249
Goharrizi KJ, Karami S, Basaki T, Dehnavi MM, Momeni Nejat MA, MM, Meru G, (2022) Transcriptomic and proteomic mechanisms underlying cold tolerance in plants. Biologia Plantarum 66:240–254. https://doi.org/10.32615/bp.2022.030
Gong L, Wang J, Abbas T, Zhang Q, Cai M, Tahir M, Wu D, Di H (2021) Immobilization of exchangeable Cd in soil using mixed amendment and its effect on soil microbial communities under paddy upland rotation system. Chemosphere 262:127828
Gorny J, Billon G, Lesven L, Dumoulin D, Madé B, Noiriel C (2015) Arsenic behavior in river sediments under redox gradient: a review. Sci Total Environ 505:423–434
Guerriero G, Berni IDR, Muñoz-Sanchez JA, Apone F, Abdel-Salam EM, Qahtan AA, Alatar AA, Cantini C, Cai G, Hausman J-F, Siddiqui IDKS et al (2018) Production of plant secondary metabolites: examples, tips, and suggestions for biotechnologists. Genes 9:309. https://doi.org/10.3390/genes9060309
Gupta DK, Nicoloso FT, Schetinger MRC, Rossato LV, Pereira LB, Castro GY, Srivastava S, Tripathi RD (2009) Antioxidant defense mechanism in hydroponically grown Zea mays seedlings under moderate lead stress. J Hazard Mater 172:479–484
Gustafsson JP, Tiberg C, Edkymish A, Berggren Kleja D (2011) Modelling lead (II) sorption to ferrihydrite and soil organic matter. Environ Chem 8:485–492
Hall JL (2002) Cellular mechanisms for heavy metal detoxification and tolerance. J Exp Bot 53:1–11. https://doi.org/10.1093/jexbot/53.366.1
Harper DJ (1998) Early Chinese medical literature. The Mawangdui medical manuscripts. Kegan Paul Intern, London
Hou X, Han H, Cai L, Liu A, Ma X, Zhou C, Wang G, Meng F (2018) Pb stress effects on leaf chlorophyll fluorescence, antioxidative enzyme activities, and organic acid contents of Pogonatherum crinitum seedlings. Flora 240:82–88
Hu PJ, Qiu RL, Senthilkumar P, Jiang D, Chen ZW, Tang YT, Liu FJ (2009) Tolerance accumulation and distribution of zinc and cadmium in hyperaccumulator Potentilla griffithii. Environ Exp Bot 66:317–325
Hu W, Wang H, Dong L, Huang B, Borggaard OK, Bruun HC, Hansen Y, He PE (2018) Holm source identification of heavy metals in peri-urban agricultural soils of southeast China: an integrated approach. Environ Pollut 237:650–661. https://doi.org/10.1016/j.envpol.2018.02.070
Hu Y, Li J, Lou B, Wu R, Wang G, Lu C, Wang H, Pi J, Xu Y (2020) The role of reactive oxygen species in arsenic toxicity. Biomolecules 10:1–30
Huang Y, Hatayama M, Inoue C (2011) Characterization of As efflux from the roots of As hyperaccumulator Pteris vittata L. Planta 234:1275–1284
Huang H, Farhan U, Dao-Xiu Z, Ming Y, Yu Z. (2019) Mechanisms of ROS regulation of plant development and stress responses. Front Plant Sci 10. https://doi.org/10.3389/fpls.2019.00800
Ibrahim MH, Kong YC, Zain NAM (2017) Effect of cadmium and copper exposure on growth, secondary metabolites and antioxidant activity in the medicinal plant Sambung Nyawa (Gynura procumbens (Lour.) Merr). Molecules 22:1623. https://doi.org/10.3390/molecules22101623
Jan R, Asaf S, Numan M, Kim KM (2021) Plant secondary metabolite biosynthesis and transcriptional regulation in response to biotic and abiotic stress conditions. Agron J 11:968. https://doi.org/10.3390/agronomy11050968
Janousková M, Pavlíková D (2010) Cadmium immobilization in the rhizosphere of arbuscular mycorrhizal plants by the fungal extraradical mycelium. Plant Soil 332:511–520. https://doi.org/10.1007/s11104-010-0317-2
Jiang L, Wang W, Chen Z, Gao Q, Xu Q, Cao H (2017) A role for APX1 gene in lead tolerance in Arabidopsis thaliana. Plant Sci 256:94–102
Joseph T, Dubey B, McBean EA (2015) Human health risk assessment from arsenic exposures in Bangladesh. Sci Total Environ 527:552–560. https://doi.org/10.1016/j.scitotenv.2015.05.053
Kabata-Pendias A, Pendias H (2001) Trace elements in soils and plants, 3rd edn. CRC Press, Boca Raton
Kalita J, Pradhan AK, Shandilya ZM, Tanti B (2018) Arsenic stress responses and tolerance in rice: physiological, cellular and molecular approaches. Rice Sci 2:235–249. https://doi.org/10.1016/j.rsci.2018.06.007
Kaur G, Singh HP, Batish DR, Kohli RK (2012) A time course assessment of changes in reactive oxygen species generation and antioxidant defense in hydroponically grown wheat in response to lead ions (Pb2+). Protoplasma 249:1091–1100
Khalid S, Shahid M, Dumat C, Niazi NK, Bibi I, Gul Bakhat HFS, Abbas G, Murtaza B, Javeed HMR (2017b) Influence of groundwater and wastewater irrigation on lead accumulation in soil and vegetables: implications for health risk assessment and phytoremediation. Int J Phytoremed 19:1037–1046. https://doi.org/10.1080/15226514.2017.1319330
Khalid S, Shahid M, Niazi NK, Rafiq M, Bakhat HF, Imran M, Abbas T, Bibi I, Dumat C (2017a) Arsenic behavior in soil-plant system: biogeochemical reactions and chemical speciation influences. In: Anjum NA, Gill SS, Tuteja N (eds) Enhancing Cleanup of Environmental Pollutants, Vol 2: Non- Biological Approaches, Springer International Publishing, Switzerland, pp 97–140
Kim Y-Y, Yang Y-Y, Lee Y (2002) Pb and Cd uptake in rice roots. Physiol Plant 116(3):368–372. https://doi.org/10.1034/j.1399-3054.2002.1160312.x
Kiran BR, Prasad MNV (2017) Responses of Ricinus communis L. (castor bean, phytoremediation crop) seedlings to lead (Pb) toxicity in hydroponics. Selcuk J Agri Food Sci 31:73–80
Kishor PK, Sangam S, Amrutha R, Laxmi PS, Naidu KS, Rao KRSS, Rao S, Reddy KG, Theriappan P, Sreenivasulu N (2005) Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: its implications in plant growth and abiotic stress tolerance. Curr Sci 88:424–438
Kohli SK, Handa N, Sharma A, Gautam V, Arora S, Bhardwaj R, Alyemeni MN, Wijaya L, Ahmad P (2018) Combined effect of 24-epibrassinolide and salicylic acid mitigates lead (Pb) toxicity by modulating various metabolites in Brassica juncea L. seedlings. Protoplasma 255:11–24. https://doi.org/10.1007/s00709-017-1124-x
Kopittke PM, Asher CJ, Kopittke RA, Menzies NW (2007) Toxic effects of Pb2+ on growth of cowpea (Vigna unguiculata). Environ Pollut 150:280–287
Kováčik J, Klejdus B (2008) Dynamics of phenolic acids and lignin accumulation in metal-treated Matricaria chamomilla roots. Plant Cell Rep 27:605–615. https://doi.org/10.1007/s00299-007-0490-9
Kovács A, Vasas A, Hohmann J (2008) Natural phenanthrenes and their biological activity. Phytochemistry 69:1084–1110
Kroymann J (2011) Natural diversity and adaptation in plant secondary metabolism. Current Opin Plant Biol 14:246–251. https://doi.org/10.1016/j.pbi.2011.03.021
Kumar S, Pandey AK (2013) Chemistry and biological activities of flavonoids: an overview. Sci World J 2013:162750. https://doi.org/10.1155/2013/162750
Kumar A, Prasad MNV, Mohan V, Achary M, Panda BB (2013) Elucidation of lead-induced oxidative stress in Talinum triangulare roots by analysis of antioxidant responses and DNA damage at cellular level. Environ Sci Pollut Res 20:4551–4561
Kumar S, Dubey RS, Tripathi RD, Chakrabarty D, Trivedi PK (2015) Omics and biotechnology of arsenic stress and detoxification in plants: current updates and prospective. Environ Int 74:221–230
Lapie C, Leglize P, Paris C (2019) Profiling of main metabolites in root exudates and mucilage collected frommaize submitted to cadmium stress. Environ Sci Pollut Res 26:17520–17534. https://doi.org/10.1007/s11356-019-05168-0
Lee M, Lee K, Lee J, Noh EW, Lee Y (2005) AtPDR12 contributes to lead resistance in Arabidopsis. Plant Physiol 138:827–836
Li R-Y, Ago Y, Liu W-J, Mitani N, Feldmann J, McGrath SP, Ma JF, Zhao F-J (2009) The rice aquaporin Lsi1 mediates the uptake of methylated arsenic species. Plant Physiol 150:2071–2080
Li H-B, Li J, Zhu Y-G, Juhasz AL, Ma LQ (2015) Comparison of arsenic bioaccessibility in house dust and contaminated soils based on four in vitro assays. Sci Total Environ 532:803–811. https://doi.org/10.1016/j.scitotenv.2015.06.060
Li Y, Zhou C, Huang M, Luo J, Hou X, Wu P, Ma X (2016) Lead tolerance mechanism in Conyza canadensis: subcellular distribution, ultrastructure, antioxidative defense system, and phytochelatins. J Plant Res 129:251–262
Li Q, Guo J, Zhang X, Yu H, Huang F, Zhang L, Zhang M, Li T (2019a) Changes of non-protein thiols in root and organic acids in xylem sap involved in cadmium translocation of cadmium-safe rice line (Oryza sativa L.). Plant Soil 439:475–486
Li CF, Zhou KH, Qin WQ, Tian CJ, Qi M, Yan XM, Han WB (2019b) A review on heavy metals contamination in soil: effects, sources, and remediation techniques. Soil Sedim Contamin 28:380–394
Liang X, Zhang L, Kumar SN, Becker DF (2013) Antioxidants & redox signaling. Dickman M (ed), Proline Mechanisms of stress survival. Plant Redox. https://doi.org/10.1089/ars.2012.5074
Lin J, He F, Owens G, Chen Z (2021) How do phytogenic iron oxide nanoparticles drive redox reactions to reduce cadmium availability in a flooded paddy soil? J Hazard Mater 403:12
Ma LQ, Komar KM, Tu C, Zhang W, Cai Y, Kennelley ED (2001) A fern that hyperaccumulates arsenic. Nature 409:579
Maccaferri M, Harris NS, Twardziok SO, Pasam RK, Gundlach H, Spannagl M, Cattivelli L (2019) Durum wheat genome highlights past domestication signatures and future improvement targets. Nat Genet 51:885–895
Maestri E, Marmiroli M, Visioli G, Marmiroli N (2010) Metal tolerance and hyperaccumulation: costs and trade-offs between traits and environment. Environ Exp Bot 68:1–13
Mahajan M, Kuiry R, Pal PK (2020) Understanding the consequence of environmental stress for accumulation of secondary metabolites in medicinal and aromatic plants. J Appl Res Med Aromatic Plants 18:100255. https://doi.org/10.1016/j.jarmap.2020.100255
Mao K, Yang J, Wang M, Liu H, Guo X, Zhao X, Dong Q, Ma F (2021) Genome-wide analysis of the apple CaCA superfamily reveals that MdCAX proteins are involved in the abiotic stress response as calcium transporters. BMC Plant Biol 21:81. https://doi.org/10.1186/s12870-021-02866-1
Marin AR, Masscheleyn PH, Patrick WH (1993) Soil redox-pH stability of arsenic species and its influence on arsenic uptake by rice. Plant Soil 52:245–325
Martínez-Sánchez M, Martínez-López S, García-Lorenzo M, Martínez-Martínez L, Pérez-Sirvent C (2011) Evaluation of arsenic in soils and plant uptake using various chemical extraction methods in soils affected by old mining activities. Geoderma 160:535–541. https://doi.org/10.1016/j.geoderma.2010.11.001
Masscheleyn PH, Delaune RD, Patrick WH (1991) Effect of redox potential and pH on arsenic speciation and solubility in contaminated soil. Environ Sci Technol 25:1414–1419
Maurel C, Verdoucq L, Luu D-T, Santoni V (2008) Plant aquaporins: membrane channels with multiple integrated functions. Annu Rev Plant Biol 59:595–624
Melato FA, Regnier T, McCrindle RI, Mokgalaka NS (2012) Impact of metals on secondary metabolites production and plant morphology in Vetiver grass (Chrysopogon zizanioides). S Afr J Chem 65:178–183
Mendoza-Cózatl DG, Butko E, Springer F, Torpey JW, Komives EA, Kehr J, Schroeder JI (2008) Identification of high levels of phytochelatins, glutathione, and cadmium in the phloem sap of Brassica napus. A role for thiol-peptides in the long-distance transport of cadmium and the effect of cadmium on iron translocation. Plant J 54:249–259. https://doi.org/10.1111/j.1365-313X.2008.03410.x
Michalak A (2006) Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress. Pol J Environ Stud 15:523–530
Mitani-Ueno N, Yamaji N, Zhao F-J, Ma JF (2011) The aromatic/arginine selectivity filter of NIP aquaporins plays a critical role in substrate selectivity for silicon, boron, and arsenic. J Ex Bot 62:4391–4398
Mombo S, Foucault Y, Deola F, Gaillard I, Goix S, Shahid M, Schreck E, Pierart A, Dumat C (2016) Management of human health risk in the context of kitchen gardens polluted by lead and cadmium near a lead recycling company. J Soils Sedim 16:1214–1224. https://doi.org/10.1007/s11368-015-1069-7
Moreno-Jiménez E, Esteban E, Peñalosa JM (2012) The fate of arsenic in soil-plant systems. Rev Environ Contam Toxicol 215:1–37. https://doi.org/10.1007/978-1-4614-1463-6_1
Mori S, Uraguchi S, Ishikawa S, Arao T (2009) Xylem loading process is a critical factor in determining Cd accumulation in the shoots of Solanum melongena and Solanum torvum. Environ Exp Bot 67:127–132. https://doi.org/10.1016/j.envexpbot.2009.05.006
MovahediYaghutiWei AAAIZHP, Rutland W, SunMousaviLiZhuge MDQ (2021) Plant secondary metabolites with an overview of Populus. Int J Mol Sci 22:6890. https://doi.org/10.3390/ijms22136890
Müsel G, Schindler T, Bergfeld R, Ruel K, Jacquet G, Lapierre C, Speth V, Schopfer P (1997) Structure and distribution of lignin in primary and secondary cell walls of maize coleoptiles analyzed by chemical and immunological probes. Planta 201:146–159
Nasim SA, Dhir B (2010) Heavy metals alter the potency of medicinal plants. In: Whitacre D (Eds) Reviews of environmental contamination and toxicology. Reviews of environmental contamination and toxicology Continuation of residue reviews, p. 203. Springer, New York. https://doi.org/10.1007/978-1-4419-1352-4_5
Neidhardt H, Kramar U, Tang X, Guo H, Norra S (2015) Arsenic accumulation in the roots of Helianthus annuus and Zea mays by irrigation with arsenic-rich groundwater: Insights from synchrotron X-ray fluorescence imaging. Geochem 75:261–270. https://doi.org/10.1016/j.chemer.2015.04.001
Niazi NK, Bibi I, Fatimah A, Shahid M, Javed MT, Wang H, Ok YS, Bashir S, Murtaza B, Saqib ZA (2017) Phosphate-assisted phytoremediation of arsenic by Brassica napus and Brassica juncea: morphological and physiological response. Int J Phytoremed 19:670–678. https://doi.org/10.1080/15226514.2016.1278427
Okem A, Southway C, Stirk WA, Street RA, Finnie JF, Staden J (2015) Effect of cadmium and aluminum on growth, metabolite content and biological activity in Drimia elata (Jacq.) Hyacinthaceae. South African J Bot 98:142–147. https://doi.org/10.1016/j.sajb.2015.02.013
Panda S, Upadhyay R, Nath S (2010) Arsenic stress in plants. J Agron Crop Sci 196:161–174
Parvin S, Md Ehsanul Haque, Fahmida Akhter, Mubarak Ali (2022) Determination of arsenic in minor cereals (Barley, Foxtail Millet, Proso-Millet, Finger- Millet, Pearl -Millet, Buckwheat. Oat, Quinoa and Sorghum) in Gazipur, Bangladesh. Journal of Agricultural Research Pesticides and Biofertilizers, 3(1).
Patra M, Bhowmik N, Bandopadhyay B, Sharma A (2004) Comparison of mercury, lead, and arsenic with respect to genotoxic effects on plant systems and the development of genetic tolerance. Environ Exp Bot 52:199–223. https://doi.org/10.1016/j.envexpbot.2004.02.009
Paunov M, Koleva L, Vassilev A, Vangronsveld J, Goltsev V (2018) Effects of different metals on photosynthesis: cadmium and zinc affect chlorophyll fluorescence in durum wheat. Int J Mol Sci 19:787. https://doi.org/10.3390/ijms19030787
Pawlak-Sprada S, Arasimowicz-Jelonek M, Podgórska M, Deckert J (2011) Activation of phenylpropanoid pathway in legume plants exposed to heavy metals. Part I. Effects of cadmium and lead on phenylalanine ammonia-lyase gene expression, enzyme activity and lignin content. Acta Biochim Pol 58:211–216
Perveen S, Anis M, Aref IM (2012) In vitro morphogenic response and metal accumulation in Albizia lebbeck (L.) cultures grown under metal stress. Eur J for Res 131:669–681
Pourrut B, Shahid M, Dumat C, Winterton P, Pinelli E (2011) Lead uptake, toxicity, and detoxification in plants. Rev Environ Contam Toxicol 213:113–136. https://doi.org/10.1007/978-1-4419-9860-6_4
Prasad MNV (2005) Cadmium toxicity and tolerance in vascular plants. Environ Exp Bot 35:525–545
Prasad A, Kumar S, Khaliq A, Pandey A (2011) Heavy metals and arbuscular mycorrhizal (AM) fungi can alterthe yield and chemical composition of volatile oil of sweet basil (Ocimum basilicum L.). Biol Fertil Soils 47:853–861. https://doi.org/10.1007/s00374-011-0590-0
Probst A, Liu H, Fanjul M, Liao B, Hollande E (2009) Response of Vicia faba L. to metal toxicity on mine tailing substrate: geochemical and morphological changes in leaf and root. Environ Exp Bot 66:297–308
Punamiya P, Datta R, Sarkar D, Barber S, Patel M, Das P (2010) Symbiotic role of glomus mosseae in phytoextraction of lead in vetiver grass [Chrysopogon zizanioides (L.)]. J Hazard Mater 177:465–474
Raab A, Wright SH, Jaspars M, Meharg AA (2007a) Feldmann J Pentavalent arsenic can bind to biomolecules. Angew Chem Int Ed 46:2594–2597
Raab A, Williams PN, Meharg A, Feldmann J (2007b) Uptake and translocation of inorganic and methylated arsenic species by plants. Environ Chem 4:197–203
Rafiq M, Shahid M, Shamshad S, Khalid S, Niazi NK, Abbas G, Saeed MF, Ali M, Murtaza B (2017) A comparative study to evaluate efficiency of EDTA and calcium in alleviating arsenic toxicity to germinating and young Vicia faba L. seedlings. J Soils Sedim 1:11. https://doi.org/10.1007/s11368-017-1693-5
Ramírez-Solis A, Mukopadhyay R, Rosen BP, Stemmler TL (2004) Experimental and theoretical characterization of arsenite in water: insights into the coordination environment of As-O. Inorg 43:2954–2959
Rascio N, Navari-Izzo F (2011) Heavy metal hyperaccumulating plants: how and why do they do it? And what makes them so interesting? Plant Sci 180:169–181
Redjala T, Zelko I, Sterckeman T, Legué V, Lux A (2011) Relationship between root structure and root cadmium uptake in maize. Environ Exp Bot 71:241–248
Redondo-Gómez S, Mateos-Naranjo E, Andrades-Moreno L (2010) Accumulation and tolerance characteristics of cadmium in a halophytic Cd-hyperaccumulator Arthrocnemum macrostachyum. J Hazard Mater 184:299–307
Reeves R (2006) Hyperaccumulation of trace elements by plants. In: Morel JL, Echevarria G, Goncharova N (eds) Phytoremediation of Metal Contaminated Soils. NATO Science, Springer, Dordrecht
Reeves RD, Schwartz C, Morel JL, Edmondson J (2001) Distribution and metal accumulating behavior of Thlaspi caerulescens and associated metallophytes in France. Int J Phytoremediation 3:145–172
Retief F, Cilliers L (2006) Lead poisoning in ancient Rome. Acta Theologica 26:2. https://doi.org/10.4314/actat.v26i2.52570
Riley PA (1994) Free radicals in biology: oxidative stress and the effects of ionizing radiation. Int J Radiat Biol 65(27–33):10. https://doi.org/10.1080/09553009414550041
Rinklebe J, Shaheen SM (2014) Assessing the mobilization of cadmium, lead, and nickel using a seven-step sequential extraction technique in contaminated floodplain soil profiles along the central Elbe River, Germany. Water Air Soil Pollut 225:2039
Rinklebe J, Shaheen SM, Yu K (2016) Release of As, Ba, Cd, Cu, Pb, and Sr under pre-definite redox conditions in different rice paddy soils originating from the USA and Asia. Geoderma 270:21–32
Rizwan M, Ali S, Rehman MZu, Javed MR, Bashir A (2018) Lead toxicity in cereals and its management strategies: a critical review. Water Air Soil Pollut 229:211. https://doi.org/10.1007/s11270-018-3865-3
Rout GR, Das P (2003) Effect of metal toxicity on plant growth and metabolism. Agronomie 23:3–11. https://doi.org/10.1051/agro:2002073
Sadana US, Samal D, Claassen N (2003) Differences in manganese efficiency of wheat (Triticum aestivum L.) and Raya (Brassica juncea L.) as related to root-shoot relations and manganese influx. J Plant Nutr Soil Sci 166:385–389
Sadiq M (1997) Arsenic chemistry in soils: an overview of thermodynamic predictions and field observations. Water Air Soil Pollut 93:117–136
Sauve S, McBride M, Hendershot W (1998) Soil solution speciation of lead(II): effects of organic matter and pH. Soil Sci Soc Am J 62:618–621
Scheckel KG, Ryan JA, Allen D, Lescano NV (2005) Determining speciation of Pb in phosphate-amended soils: method limitations. Sci Total Environ 350:261–272
Seregin IV, Ivanov VB (2001) Physiological aspects of cadmium and lead toxic effects on higher plants. Russ J Plant Physiol 48:523–525
Seregin IV, Shpigun LK, Ivanov VB (2004) Distribution and toxic effects of cadmium and lead on maize roots. Russ J Plant Physiol 51:525–533
Shaaria NEM, Tajudina MTFM, Khandakera MM, Majrashib A, Alenazic MM, Abdullahia UA, Mohda KS (2022) Cadmium toxicity symptoms and uptake mechanism in plants: a review. Braz J Biol 7(84):e252143. https://doi.org/10.1590/1519-6984.252143
Shaheen SM, Rinklebe J (2017) Sugar beet factory lime affects the mobilization of Cd Co, Cr, Cu, Mo, Ni, Pb, and Zn under dynamic redox conditions in a contaminated floodplain soil. J Environ Manage 186:253–260
Shaheen SM, Rinklebe J, Frohne T, White JR, DeLaune RD (2016) Redox effects on release kinetics of arsenic, cadmium, cobalt, and vanadium in Wax Lake Deltaic freshwater marsh soils. Chemosphere 150:740–748
Shaheen SM, Shams MS, Khalifa MR, El-Dali MA, Rinklebe J (2017) Various soil amendments and environmental wastes affect the (im)mobilization and phytoavailability of potentially toxic elements in a sewage effluent irrigated sandy soil. Ecotoxicol Environ Saf 142:375–387
Shahid M, Pinelli E, Dumat C (2012) Review of Pb availability and toxicity to plants in relation with metal speciation; role of synthetic and natural organic ligands. J Hazard Mater 219:1–12. https://doi.org/10.1016/j.jhazmat.2012.01.060
Shahid M, Xiong T, Castrec-Rouelle M, Leveque T, Dumat C (2013) Water extraction kinetics of metals, arsenic and dissolved organic carbon from industrial contaminated poplar leaves. J Environ Sci 25:2451–2459. https://doi.org/10.1016/S1001-0742(12)60197-1
Shahid M, Dumat C, Pourrut B, Silvestre J, Laplanche C, Pinelli E (2014) Influence of EDTA and citric acid on lead-induced oxidative stress to Vicia faba roots. J Soils Sedim 14:835–843. https://doi.org/10.1007/s11368-013-0724-0
Shahid M, Dumat C, Khalid S, Niazi NK, Antunes PMC (2016) Cadmium bioavailability, uptake, toxicity and detoxification in soil-plant system. In: de Voogt, P. (eds) Reviews of Environmental Contamination and Toxicology. Reviews of Environmental Contamination and Toxicology, 241. Springer, Cham. https://doi.org/10.1007/398_2016_8
Shakoor MB, Niazi NK, Bibi I, Rahman MM, Naidu R, Dong Z, Shahid M, Arshad M (2015) Unraveling health risk and speciation of arsenic from groundwater in rural areas of Punjab, Pakistan. Int J Environ Res Public Health 12:12371–12390
Sharma P, Dubey RS (2005) Lead toxicity in plants. Braz J Plant Physiol 17:35–52
Šichorová K, Tlustoš P, Száková J, Kořínek K, Balík JK (2004) Horizontal and vertical variability of heavy metals in the soil of a polluted area. Plant Soil Environ 50:525–534
Siddiqui F, Tandon PK, Srivastava S (2015) Analysis of arsenic-induced physiological and biochemical responses in a medicinal plant, Withania somnifera. Physiol Mol Biol Plants 21:61–69. https://doi.org/10.1007/s12298-014-0278-7
Signes-Pastor A, Burló F, Mitra K, Carbonell-Barrachina A (2007) Arsenic biogeochemistry as affected by phosphorus fertilizer addition, redox potential, and pH in a west Bengal (India) soil. Geoderma 137:504–510. https://doi.org/10.1016/j.geoderma.2006.10.012
Singh PK, Indoliya Y, Chauhan AS, Singh SP, Singh AP, Dwivedi S, Tripathi RD, Chakrabarty D (2017) Nitric oxide mediated transcriptional modulation enhances plant adaptive responses to arsenic stress. Sci Rep 7:3592
Srivastava RK, Pandey P, Rajpoot R, Rani A, Dubey RS (2014) Cadmium and lead interactive effects on oxidative stress and antioxidative responses in rice seedlings. Protoplasma 251:1047–1065
Stancheva I, Genevaa M, Hristozkova M, Markovska Y, Salamon I (2010) Antioxidant capacity of sage grown on heavy metal-polluted soil. Russ J Plant Physiol 57:799–805
Sterckeman T, Thomine S (2020) Mechanisms of cadmium accumulation in plants. Crit Rev Plant Sci 39:322–359. https://doi.org/10.1080/07352689.2020.1792179
Stoessl A, Arditti J (1984) Orchid phytoalexins. In: Arditti J (ed) Orchid biology, reviews, and perspectives, III. Cornell University Press, Ithaca, New York.
Subašić M, Šamec D, Selović A, Karalija E (2022) Phytoremediation of cadmium polluted soils: current status and approaches for enhancing soil. System 6:3. https://doi.org/10.3390/soilsystems6010003
Sungur A, Kavdir Y, Özcan H, İlay R, Soylak M (2020) Geochemical fractions of trace metals in surface and core sections of aggregates in agricultural soils. Catena 197:2021
Száková J, Dziaková M, Kozáková A, Tlustoš P (2018) The risk element uptake by chamomile (Matricaria recutita (L.) Rauschert) growing in four different soils. Arch Environ Prot 44:12–21
Talukdar D (2017) Reactive oxygen species in plants. Balancing roles of reactive oxygen species in plants response to metalloid exposure: boon or bane—revisiting the role of ROS. Wiley, Hobokem
Tariq H (2018) Effect of lead on plants and animals. Retrieved (11/02/2022). https://www.slideshare.net/hazz12/effect-of-lead-on-plants-and-animals-86577938. Accessed on 22 Oct 2022
Tatipamula VB, Kukavica B (2021) Phenolic compounds as antidiabetic, anti-inflammatory, and anticancer agents and improvement of their bioavailability by liposomes. Cell Biochem Funct 39:926–944. https://doi.org/10.1002/cbf.3667
Teoh ES (2015) Secondary Metabolites of plants. Med Orchids Asia 2015:59–73. https://doi.org/10.1007/978-3-319-24274-3_5
Tlustoš P, Száková J, Daniela P, Balik A (2002) The accumulation of arsenic and cadmium by different species of vegetables. Ac 571:217–224. https://doi.org/10.17660/ActaHortic.2002.571.26
Tremlová J, Sehnal M, Száková J, Goessler W, Steiner O, Najmanová J, Horáková T, Tlustoš P (2017) A profile of arsenic species in different vegetables growing in arsenic-contaminated soils. Arch Agron Soil Sci 63:918–927
Uzu G, Sobanska S, Aliouane Y, Pradere P, Dumat C (2009) Study of lead phytoavailability for atmosphericindustrial micronic and sub-micronic particles in relation with lead speciation. Environ Pollut 157:1178–1185
Vega F, Andrade M, Covelo E (2010) Influence of soil properties on the sorption and retention of cadmium, copper, and lead, separately and together, by 20 soil horizons: comparison of linear regression and tree regression analyses. J Hazard Mater 174:522–533
Verbruggen N, Hermans C, Schat H (2009a) Mechanisms to cope with arsenic or cadmium excess in plants. Curr Opin Plant Biol 12:364–372
Verbruggen N, Hermans C, Schat H (2009b) Molecular mechanisms of metal hyperaccumulation in plants. New Phytol 181:759–776
Visoottiviseth P, Francesconi K, Sridokchan W (2002) The potential of Thai indigenous plant species for the phytoremediation of arsenic-contaminated land. EnvironPollut 118:453461
Vukmirovic D, Rollo D, Seymour C, Mothersill C (2018) Cytotoxic profiling of plant secondary metabolites on P53 variant human colon carcinoma cell lines. Dose-Response 15(4):1559325817748121. https://doi.org/10.1177/1559325817748121. (2017 Dec 21)
Wallace IS, Choi W-G, Roberts DM (2006) The structure, function, and regulation of the nodulin 26-like intrinsic protein family of plant aquaglyceroporins. Biochim Biophys Acta 1758:1165–1175
Wang S, Mulligan CN (2006) Effect of natural organic matter on arsenic release from soils and sediments into groundwater. Environ Geochem Health 28:197–214
Wang H, Shan X, Wen B, Owens G, Fang J, Zhang S (2007) Effect of indole-3-acetic acid on lead accumulation in maize (Zea mays L.) seedlings and the relevant antioxidant response. Environ Exp Bot 61:246–253
Wang C, Lu J, Zhang SH, Wang PF, Hou J, Qian J (2011) Effects of Pb stress on nutrient uptake and secondary metabolism in submerged macrophyte Vallisneria natans. Ecotoxicol Environ Saf 74:1297–1303. https://doi.org/10.1016/j.ecoenv.2011.03.005
Warren G, Alloway B (2003) Reduction of arsenic uptake by lettuce with ferrous sulfate applied to contaminated soil. J Environ Q 32:767–772. https://doi.org/10.2134/jeq2003.7670
Weng L, Temminghoff EJ, Lofts S, Tipping E, Van Riemsdijk WH (2002) Complexation with dissolved organic matter and solubility control of heavy metals in a sandy soil. Environ Sci Technol 36:4804–4810
WHO (2007) Guidelines for assessing quality of herbal medicines with reference to contaminants and residue. WHO Press, Geneva
Wiggenhauser M, Aucour AM, Bureau S, Campillo S, Telouk P, Romani M, Ma JF, Landrot G, Sarret G (2021) Cadmium transfer in contaminated soil-rice systems: insights from solid-state speciation analysis and stable isotope fractionation. Environ Pollut 269:115934
Wu C, Shi L, Xue S, Li W, Jiang X, Rajendran M, Qian Z (2019) Effect of sulfur-iron modified biochar on the available cadmium and bacterial community structure in contaminated soils. Sci Total Environ 647:1158–1168
Xie Y, Hu L, Du Z, Sun X, Amombo E, Fan J, Fu J (2014) Effects of cadmium exposure on growth and metabolic profile of bermudagrass [Cynodon dactylon (L.) Pers. PLoS One 29(9):115279. https://doi.org/10.1371/journal.pone.0115279
Xu J, Sun J, Du L, Liu X (2012) Comparative transcriptome analysis of cadmium responses in Solanum nigrum and Solanum torvum. New Phytol 196:110–124
Yadav SK (2010) Heavy metals toxicity in plants: An overview on the role of glutathione and phytochelatins in heavy metal stress tolerance of plants. S Afr J Bot 76:167–179
Yamaguchi N, Mori S, Baba K, Kaburagi-Yada S, Arao T, Kitajima N, Terada Y (2011) Cadmium distribution in the root tissues of solanaceous plants with contrasting root-to-shoot Cd translocation efficiencies. Environ Exp Bot 71:198–206
Yan X, Zhang M, Liao X, Tu S (2012) Influence of amendments on soil arsenic fractionation and phytoavailability by Pteris vittata L. Chemosphere 88:240–244
Yan G, Fan X, Tan L, Yin C, Li T, Liang Y (2021) Root silicon deposition and its resultant reduction of sodium bypass flow is modulated by OsLsi1 and OsLsi2 in rice. Plant Physiol Biochem 158:219–227. https://doi.org/10.1016/j.plaphy.2020.11.015
Yibin L, Zhongxiang S, Zhenfang L, Rongrong X, Weikang C, Shaozhi S, Tingting L, Rensen Z, Yuanyuan S (2019) Deficiency in silicon transporter lsi1 compromises inducibility of anti-herbivore defense in rice plants. Front Plant Sci 10. https://doi.org/10.3389/fpls.2019.00652
Yoshinaga M, Cai Y, Rosen BP (2011) Demethylation of methylarsonic acid by a microbial community. Environ Microbiol 13:1205–1215
Yu R, Jiang Q, Xv C, Li L, Bu S, Shi G (2019) Comparative proteomics analysis of peanut roots revealsdifferential mechanisms of cadmium detoxification and translocation between two cultivars differing in cadmium accumulation. BMC Plant Biol 19:137
Yuan H, Zhang Y, Huang Yang SY, Gu C (2015) Effects of exogenous glutathione and cysteine on growth, lead accumulation, and tolerance of Iris lactea var. Chinensis Environ Sci Pollut Res 22:2808–2816
Zemanová V, Popov M, Pavlíková D et al (2020) Effect of arsenic stress on 5-methylcytosine, photosynthetic parameters and nutrient content in arsenic hyperaccumulator Pteris cretica L. var. Albo-lineata. BMC Plant Biol 20:130. https://doi.org/10.1186/s12870-020-2325-6
Zhang J, Chen J, Wu Y-F, Wang Z-P, Qiu J-G, Li X-L, Cai F, Xiao K-Q, Sun X-X, Barry P, Zhao F-J (2021) Oxidation of organoarsenicals and antimonite by a novel flavin monooxygenase widely present in soil bacteria. Environ Microbiol 00:00-00. https://doi.org/10.1111/1462-2920.15488
Zhao FJ, Ma JF, Meharg AA, McGrath SP (2009) Arsenic uptake and metabolism in plants. New Phytol 181:777–794. https://doi.org/10.1111/j.1469-8137.2008.02716.x
Zheljazkov VD, Craker LE, Xing B (2006) Effects of Cd, Pb, and Cu on growth and essential oil contents in dill, peppermint, and basil. Environ Exp Bot 58:9–16. https://doi.org/10.1016/j.envexpbot.2005.06.008
Zheng L, Peer T, Seybold V, Lütz-Meindl U (2012) Pb-induced ultrastructural alterations and subcellular localization of Pb in two species of Lespedeza by TEM-coupled electron energy loss spectroscopy. Environ Exp Bot 77:196–206
Zhou T, Radaev S, Rosen BP, Gatti DL (2000) Structure of the ArsA ATPase: the catalytic subunit of a heavy metal resistance pump. EMBO J 19:1–8
Zhou C, Huang M, Li Y, Luo J, Cai L (2016) Changes in subcellular distribution and antioxidant compounds involved in Pb accumulation and detoxification in Neyraudia reynaudiana. Environ Sci Pollut Res 23:21794–21804
Zhu FY, Li L, Lam PY, Chen MX, Chye ML, Lo C (2013) Sorghum extracellular leucine-rich repeat protein SbLRR2 mediates lead tolerance in transgenic Arabidopsis. Plant Cell Physiol 54:1549–1559. https://doi.org/10.1093/pcp/pct101
Funding
This research was supported by the Nutrisk project (European Regional Development Fund—Project No. CZ.02.1.01/0.0/0.0/ 16_019/0000845).
Author information
Authors and Affiliations
Contributions
Conceptualization: Michael O. Asare, Jiřina Száková, and Pavel Tlustoš; data curation: Michael O. Asare and Jiřina Száková; formal Analysis: Michael O. Asare, Jiřina Száková, and Pavel Tlustoš; funding acquisition: Jiřina Száková and Pavel Tlustoš; methodology: Michael O. Asare, Jiřina Száková, and Pavel Tlustoš; project administration: Jiřina Száková and Pavel Tlustoš; resources: Jiřina Száková and Pavel Tlustoš; supervision: Prof. Jiřina Száková and Prof. Pavel Tlustoš; validation: Jiřina Száková and Pavel Tlustoš; writing — original draft: Michael O. Asare and Jiřina Száková; and writing — review and editing: Michael O. Asare, Jiřina Száková, and Pavel Tlustoš.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All authors have approved the final version of the manuscript and have given their consent for publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Elena Maestri
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Asare, M.O., Száková, J. & Tlustoš, P. The fate of secondary metabolites in plants growing on Cd-, As-, and Pb-contaminated soils—a comprehensive review. Environ Sci Pollut Res 30, 11378–11398 (2023). https://doi.org/10.1007/s11356-022-24776-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-24776-x