Abstract
The remediation of Cr(VI) is one of the hot issues in groundwater pollution remediation. The influences and effects of pH on Cr(VI) reduction by organic reducing substances in sugarcane molasses were studied under the sterilization and unsterilization conditions by means of batch laboratory-scale experiments. The results showed that pH of reaction solution has a great influence on the reduction of Cr(VI) by sugarcane molasses. When pH ≥ 3.0, the final removal efficiency of Cr(VI) on the condition of sterilization and unsterilization at pH values of 8.0, 5.8, 4.0 and 3.0 was 15.441%, 17.945%, 29.205%, 36.258% and 99.236%, and 98.577%, 99.235% and 99.368%, respectively. When pH < 3.0, the final removal efficiency of Cr(VI) on the condition of sterilization and unsterilization at pH values of 2.5 and 2.0 was 99.631% and 99.895%, and 99.631% and 99.895%, respectively. The main mechanism involves chemical reduction and biological reduction. And biological reduction in the process of Cr(VI) reduction by sugarcane molasses is of the first priority when pH ≥ 3.0; when pH < 3.0, the rate of chemical reduction was higher and it was not the basic existing biological reduction. The relationship between pH and pseudo-first-order rate constant (kobs) on both the sterilization and unsterilization conditions was satisfied with Boltzmann model. There is an inflection point at the pH of about 2.6, which also indicated the influence mechanism of pH on the reduction of Cr(VI) by sugarcane molasses.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Groundwater is an important resource in people’s life and production. However, in the process of industrial society, a large amount of hazardous waste such as heavy metals, organic matter and harmful radioactive substances enters into the groundwater, causing the pollution-induced water shortage problem (Barrera-Díaz et al. 2012). Chromium pollution is the most common type of heavy metal pollution in groundwater pollution site. In general, chromium exists in groundwater with two stable oxidation states: hexavalent C(VI) and trivalent Cr(III) (Yurik and Pikaev 1999). The most possibly existing Cr(VI) species in aqueous solution are Cr2O72−, CrO42−, H2CrO4 and HCrO4−, which are known to be toxic, carcinogenic and mobile, causing health problems such as liver damage, pulmonary congestion, vomiting and severe diarrhea, whereas Cr(III) is less toxic and can be precipitated out of solution in the form of Cr(OH)3 (Wu et al. 2017). In China, there were up to 7 million tons chromium wastes, and more than 400 million tons chromium wastes were released into the environment directly, which severely polluted surface water environment, groundwater environment and soil environment (Gu et al. 2005). The remediation of Cr(VI) has become an urgent problem in the world.
Nowadays, the reduction of Cr(VI) to Cr(III) in groundwater is considered an important remediation strategy targeted at contaminant immobilization. The reduction methods of Cr(VI) in groundwater are mainly based on the chemical, electrochemical and biological reduction (Kim et al. 2001; Cheung and Gu 2007), such as zero-valent iron (Fe0), ferrous ions (Fe2+), sulfur compounds, electrocoagulation, electrodissolution and organic reducing substances and so on (Wittbrodt and Palmer 1997; Agrawal et al. 2009; Carbonaro and Stone 2014; Liu et al. 2018). Many research results showed that the removal rate and effect of the reduction methods of chromium were influenced by the solution pH. When Fe0 and Fe2+ are used as the Cr(VI)-reducing agent, the reaction usually occurs under acidic conditions because Fe0 and Fe2+ appear as free ions in aqueous solution (park et al. 2004), and many research experiments showed that the reduction rates were achieved at low pH values (Chen et al. 2007; Gheju et al. 2008). At low pH values, the reduction of Cr(VI) to Cr(III) by Fe2+ ions is favored, but under these pH conditions, there is no precipitation of Fe(III)/Cr(III) hydroxides (Arroyo et al. 2009). Jardine et al. (1999) also thought believed that the reduction kinetics of Cr(VI) to Cr(III) seemed to be slow at pH 3.7, while the reaction rate was accelerated when pH was less than 3. Compared with inorganic reductants, organic compounds are inexpensive and environmentally harmless, and their reducing effects are also influenced by pH. Cr(VI) reduction by organic material is most rapid under acidic conditions (Zhilin et al. 2004; Deiana et al. 2007). The most favorable pH for Cr(VI) reduction by organic materials ranges from 1 to 4 (Wittbrodt and Palmer 1996). Deiana et al. (2007) emphasized that when pH > 4.2, the reduction of Cr(VI) by caffeic acid would occur slowly.
Organic reducing substance as a microbial carbon source stimulated microbial metabolism and enhanced bioreduction to convert Cr(VI) into Cr(III). Michailides et al. (2014) first screened functional microorganisms from industrial wastewater to reduce Cr(VI) to Cr(III). Molasses were used as carbon source to reduce Cr(VI) pollution under suspension growth and adhesion growth of microorganisms. The results showed that molasses was a cheaper and more efficient microbial carbon source for bioremediation of Cr(VI) pollution than sucrose. Somasundaram et al. (2009) used molasses as carbon source to study the characteristics of the reduction of chromium (VI) by chromium-reducing bacteria (CBR), sulfate-reducing bacteria (SBR) and iron-reducing bacteria (IBR) and the reduce effect under different combinations of bacteria. Mathematical models based on the reduction of hexavalent chromium, COD consumption and biomass were established. The results showed that different combinations of reaction systems could remove hexavalent chromium very well. The mathematical model was also in good agreement with the simulation results. Sugiyama et al. (2012) evaluated the effect of transforming Cr(VI) into Cr(III) by strain Flexivirga alba ST13T. The results showed that when the concentration of Cr(VI) was 3.0 mM, the reaction was slightly inhibited, and the concentration of Cr(VI) dropped below the detection limit 13 days after the operation of the reaction system, and the treatment effect of molasses was significantly better than that of glucose.
Sugarcane molasses has been successfully used in the remediation of groundwater contaminated Cr(VI) as a kind of organic materials. It was demonstrated that sugarcane molasses could reduce Cr(VI) to Cr(III) by chemical reduction and microbial reduction (Chen et al. 2015, 2016). Chen et al. (2017) discussed the reaction pathways and dynamics of chemical reduction of Cr(VI), and the reducing reaction of Cr(VI) could be described by a pseudo-first-order kinetic model. The reducing effect and reaction mechanism are also influenced by pH, and it has been confirmed that biological reduction is of the first priority and chemical reduction plays a secondary role in the process of Cr(VI) reduction by sugarcane molasses when pH ≥ 3.0 of reaction solution; and chemical reduction is of the first priority and biological reduction plays a secondary role in the process of Cr(VI) reduction by sugarcane molasses when pH ≤ 2.5 of reaction solution. But the studies of Chen are qualitative description and there are no quantitative data to prove his results. The mechanism of chemical reduction and biological reduction is not clear. And the current studies of Cr(VI) reduction by sugarcane molasses are only limited to reaction mechanism and acidic pH’s influence. There is no influence mechanism literature of Cr(VI) reduction by sugarcane molasses, which severely restricts the theoretical development of Cr(VI) remediation in groundwater. The study aimed to explore the influence mechanism of pH on Cr(VI) reduction by organic reducing substances from sugarcane molasses and evaluate the kinetics of Cr(VI) reduction under different pH values. Besides, in order to find out biological reduction, the experiments under sterilization and unsterilization conditions were designed. The results of this article would provide useful basis for using sugarcane molasses to remediate groundwater contaminated by hexavalent chromium.
Materials and methods
Reagents
Sugarcane molasses was purchased from Changchun Yingtang Molasses CO., Ltd., which was produced in accordance with the Chinese National Standard (QB/T2684-2005). Sugarcane molasses contained rich plant polyphenols (38.4–48.8 mg/L) and a small amount of organic acid. Sugar brix and total sugar are ≥ 70% and 48–52%, respectively (Table 1).
All chemicals were of analytical grade and used as received without further pretreatment. Potassium dichromate, diphenylcarbazide spectrophotometric, phenolphthalein, sulfuric acid (98%), phosphorus acid (85%), sodium hydroxide, glacial acetic acid and anhydrous sodium carbonate were supplied by Sinopharm Chemical Reagent Co., Ltd. or Beijing Chemical Plant (Beijing, China). Folin–Ciocalteu was purchased from Beijing Dingguo Changsheng Biotechnology Co., Ltd. Reverse osmosis Milli-Q water (18 M) was used to make all solutions and dilutions.
Experimental methods
Batch laboratory-scale experiments were conducted to investigate the effects of pH on Cr(VI) reduction with organic reducing substances in the form of sugarcane molasses at different pH conditions. The reaction environments of the experiments include two groups of reaction systems under sterilized and unsterilized conditions. Each group of reaction system has six brown reaction bottles with the volume of 500 mL. Diluted molasses solution was prepared by high temperature and sterilization. Approximately 0.435 mL of sugarcane molasses was dissolved into deionized water and then diluted to 1 L to obtain sugarcane molasses concentrations of 0.60 mg/L. The reaction dynamic process was also analyzed, and the mathematical relationship of reaction rate constant with [H+] was studied by weighted least squares linear regression method.
Six groups of experiments were conducted to determine the effects of pH on Cr(VI) reduction with organic reducing substances from sugarcane molasses. First, six copies of sterilization and unsterilization sand were weighed, each with the weight of 600 g, respectively, and put them into the brown reaction bottles under the condition of constant room temperature 20 °C ± 1.0 °C. Then, reactions mixtures were obtained by taking 100 mL Cr(VI) solutions, adjusting the concentration values of 20 mg/L, adding 100 mL of diluted molasses solutions with the Brix of 0.6% and adjusting the pH values of 2.0, 2.5, 3.0, 4.0, 5.8 and 8.0 ± 0.2. At regular time intervals, 10 mL of mixed solution was withdrawn to determine Cr(VI), total Cr concentration and organic reducing substances. Each experiment was repeated thrice under sterilization conditions.
Analytical methods
Total Cr concentration, Cr(VI) concentration, organic reducing substances, pH and temperature were measured in all the experiments. The Cr(VI) concentration was determined by the diphenylcarbazide spectrophotometric method according to the “Standard Methods for the Examination of Water and Wastewater” at 540 nm using a UNIC 7200 visible spectrophotometer (Macy China Instruments Inc., Beijing, China). Total chromium was determined by flame atomic absorption spectrometry (Shimadzu International Trading Ltd, Shimane, Japan) according to the “Standard Methods for the Examination of Water and Wastewater” using a Shimadzu AA-7000F/AAC atomic absorption spectrophotometer (USEPA 2015). The content of organic reducing substances was quantified by the Folin–Ciocalteu method at 765 nm using a WFZUV-2802H ultraviolet spectrophotometer (Shimadzu International Trading Ltd, Shimane, Japan) as described in the literature (Singleton et al. 1999). The pH and temperature were determined by HI99141 Portable pH/Temperature Measuring Instrument.
The steps of microbial community structure analysis are as follows: (1) genomic DNA extraction of samples, quantitative analysis of the extracted samples by gel electrophoresis and ultraviolet analyzer; (2) target region amplification of samples; (3) quantitative mixing of processed samples; (4) high-throughput sequencing of mixed samples. The sequencing work was entrusted to Bioengineering (Shanghai) Co., Ltd. The main contents of analysis were: data preprocessing (data quality control, data disassembly and primer region column removal), removal of chimera and target region sequences, operation taxonomic unit (OTU) classification (similarity 97% for OTU clustering), endowing species taxon, Alpha diversity analysis, etc.
Kinetic model
Previous studies have demonstrated that the overall reaction of Cr(VI) could be characterized by the pseudo-first-order kinetic model (Nakayasu et al. 1999; Park et al. 2017; Chen et al. 2016), and the rate model can be expressed as follows:
where v is the reaction rate, c is the hexavalent chromium concentration, and kobs is the pseudo-first-order rate constant. Equation (1) can be integrated to calculate Eq. (2) as follows:
Thus, half-time (t1/2) is calculated by Eq. (3):
where c0 is the initial hexavalent chromium concentration and t is the reaction time. v initial is the initial reaction rate of Cr(VI) reduction with sugarcane molasses:
Based on the kobs obtained at different pH values on the conditions of sterilization and unsterilization environment, the relationship between pH and reaction rate (kobs) was determined by regression analysis method. The relationship between pH and reaction rate (kobs) on the conditions of sterilization and unsterilization was described by Boltzmann reaction kinetic model (Yan et al. 2017) as Eq. (5).
where kobs is the pseudo-first-order rate constant, and pH is the reaction solution pH. A0, A1, A2 and A3 are the constants.
Results and discussion
Effects of pH on reduction of Cr(VI)
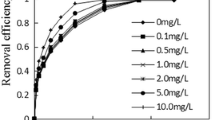
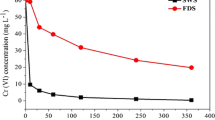
Figure 1 shows the relationship between reaction solution pH and removal efficiency of Cr(VI) reduction. The curves characteristics of Cr(VI) removal efficiency were different with different pH values. The curves characteristics of Cr(VI) removal efficiency were approximately identical, when pH is larger than 3.0. The final removal efficiency of Cr(VI) on the condition of sterilization and unsterilization at pH values of 8.0, 5.8, 4.0 and 3.0 was 15.441%, 17.945%, 29.205% and 36.258%, and 99.236%, 98.577%, 99.235% and 99.368%, respectively. When pH < 3.0, the curves of Cr(VI) removal efficiency had the same characteristics. The final removal efficiency of Cr(VI) on the condition of sterilization and unsterilization at pH values of 2.5 and 2.0 were 99.631% and 99.895%, and 99.631% and 99.895%, respectively. From the results above, the final removal efficiency of Cr(VI) on the sterilization environment was far less than that of unsterilization environment when pH was higher than 3.0. The final removal efficiency values on the same pH (8.0, 5.8, 4.0 and 3.0) were 83.795%, 80.632%, 70.03% and 63.11%, respectively. And the greater pH, the more the added value of the unsterilization condition than sterilization condition. When pH < 3.0, the final removal efficiency of Cr(VI) on the condition of sterilization and unsterilization was equal. Compared to the sterilization and unsterilization experiments, the final removal efficiency of Cr(VI) by biological reduction was 83.795%, 80.612%, 70.03%, 63.11%, 0 and 0 on the pH values of 8.0, 5.8, 4.0, 3.0, 2.5 and 2.0; and those of by chemical reduction were 15.441%, 17.945%, 29.205%, 36.258%, 99.631% and 99.895% on the same pH values. When pH values were 2.5 and 2.0, the removal efficiency value of Cr(VI) by biological reduction was both 0, indicating that the biological reduction is very weak when pH < 3.0. This phenomenon is mainly influenced by the remediation mechanism of the Cr(VI). Biological reduction in the process of Cr(VI) reduction by sugarcane molasses is of the first priority when pH ≥ 3.0 of reaction solution; and chemical reduction in the process of Cr(VI) reduction by sugarcane molasses is of the first priority when pH < 3.0 of reaction solution. And the rate of chemical reduction was higher, and it was not the basic existing biological reduction when pH was lower than 3.0.
Results of kinetic mechanism
The reduction of Cr(VI) with sugarcane molasses could be described by the pseudo-first-order kinetic model with respect to the Cr(VI) concentration at different pH values. As shown in Table 2, the rate constants, half-time of Cr(VI) reduction and initial reaction rate (vinitial) were different with the pH of reaction solution. On the condition of sterilization environment, the rate constants (kobs) at the pH values of 8.0, 5.8, 4.0, 3.0, 2.5 and 2.0 were 0.0036, 0.0043, 0.0072, 0.0094, 0.2436 and 0.2981, respectively. And on the condition of unsterilization environment, the rate constants (kobs) at the pH values of 8.0, 5.8, 4.0, 3.0, 2.5 and 2.0 were 0.1059, 0.0924, 0.1015, 0.1055, 0.2436 and 0.2981, respectively. The rate constant (kobs) of pH (2.0) on the sterilization environment was 1.224, 31.713, 41.403, 69.326 and 82.805 times those of pH values of 8.0, 5.8, 4.0, 3.0 and 2.5, respectively. There was an obvious rule; that is, the rate constants (kobs) of Cr(VI) reduction reaction were decreased gradually with the increase in the initial solution pH. This may be because the rates of electrons transfer between reducing organic matters and Cr(VI) could be accelerated with the decrease in pH. And in the unsterilization environment, the rate constant (kobs) of pH (2.0) was 1.224, 2.826, 2.937, 3.226 and 2.815 times than those of pH values of 8.0, 5.8, 4.0, 3.0 and 2.5, respectively. Relative to the unsterilization environment, it has a bigger impact on rate constants (kobs) of pH on the sterilization environment. And the initial reaction rate (vinitial) and half-time (t1/2) have the same rule with rate constants (kobs). These results indicated that pH is one of the decisive factors of chemical reduction of Cr(VI) reduction by sugarcane molasses.
Figures 2 and 3 show the relationship between pH and pseudo-first-order rate constant (kobs). The curve patterns of pH and kobs on both the sterilization and unsterilization conditions were satisfied with Boltzmann model and with the kinetic equations which are (6) and (7) as follows:
From the Boltzmann model, the kobs was decreased with the increase in pH. It was an inflection point at the pH of about 2.6, and it was accurately predicted and analyzed reaction rate of sugarcane molasses and Cr(VI) and the change of Cr(VI) concentration at different pH conditions (2.0–8.0) by the Boltzmann model Eqs. (6) and (7). Biological reduction was of the first priority and chemical reduction played a secondary role in the process of Cr(VI) reduction by sugarcane molasses when pH ≥ 3.0 of reaction solution; and chemical reduction was of the first priority and biological reduction played a secondary role in the process of Cr(VI) reduction by sugarcane molasses when pH ≤ 3.0 of reaction solution. The chemical reduction by sugarcane molasses was gradually decreased with the increase in pH value.
Microbial community structure
There were 28 groups microbial flora at gate level, which were detected in unsterilization reaction system by analyzing microbial flora test (Fig. 4). The main microbial flora were Proteobacteria, Actinobacteria, Firmicutes, Bacteroidetes, and their proportion were 39.93%, 33.37%, 21.05% and 3.09%, respectively. It also showed that the diversity of microbial flora was high and the dominant microbial flora was obvious in the unsterilization reaction system, which may be filtrated due to the toxicity of Cr(VI).
There were 491 microbial species at genus level in unsterilization reaction system (Fig. 5). The main microbial species were Arthrobacter, Lactococcus, Pseudomonas, Bacillus, Massilia, Unclassified Comamonadaceae, and their proportion were 31.42%, 10.59%, 4.29%, 4.17%, 3.38% and 6.67%, respectively. The results showed that microbial species diversity was higher and microbial abundance distribution was more uniform. Arthrobacter is the most common microbial genus in groundwater environment, which belonged to gram-positive specific aerobic bacteria. At present, many scholars had reported the reduction of Cr(VI) to Cr(III) by Arthrobacter (Megharaj et al. 2003; Camargo et al. 2004; Elangovan and Philip 2009). Pseudomonas was a special aerobic gram-negative, sporeless, capsular bacterium with rod-shaped or slightly curved shape. Pseudomonas also has good reduction effect of Cr(VI) to Cr(III) (Ishibashi et al. 1990; Dmitrenko et al. 2003).
Conclusions
The pH of reaction solution has a great influence on the reduction of Cr(VI) by sugarcane molasses. When pH ≥ 3.0, the final removal efficiency of Cr(VI) on the condition of sterilization and unsterilization at pH values of 8.0, 5.8, 4.0 and 3.0 was 15.441%, 17.945%, 29.205% and 36.258%, and 99.236%, 98.577%, 99.235% and 99.368%, respectively. When pH < 3.0, the final removal efficiency of Cr(VI) on the condition of sterilization and unsterilization at pH values of 2.5 and 2.0 was 99.631% and 99.895%, and 99.631% and 99.895%, respectively. The main mechanism involves chemical reduction and biological reduction. And biological reduction in the process of Cr(VI) reduction by sugarcane molasses was of the first priority when pH ≥ 3.0 of reaction solution; and chemical reduction in the process of sugarcane molasses reducing Cr(VI) was of the first priority when pH < 3.0 of reaction solution. The rate of chemical reduction was higher, and it was not the basic existing biological reduction when pH is lower than 3.0. The relationship between pH and pseudo-first-order rate constant (kobs) on both the sterilization and unsterilization conditions was satisfied with Boltzmann model and with the kinetic equations. There is an inflection point at the pH of about 2.6, which also indicated the influence mechanism of pH on the reduction of Cr(VI) by sugarcane molasses.
References
Agrawal SG, Fimmen RL, Chin YP (2009) Reduction of Cr(VI) to Cr(III) by Fe(II) in the presence of fulvic acids and in lacustrine pore water. Chem Geol 262:328–335
Arroyo MG, Pérez- Herranz V, Montanés MT, García-Antón J, Guinón JL (2009) Effect of pH and chloride concentration on the removal of hexavalent chromium in a batch electrocoagulation reactor. J Hazard Mater 169:1127–1133
Barrera-Díaz CE, Lugo-Lugo V, Bilyeu B (2012) A review of chemical, electrochemical and biological methods for aqueous Cr(VI) reduction. J Hazard Mater 223–224:1–12
Camargo FAO, Bento FM, Okeke BC, Frankenberger WT (2004) Hexavalent chromium reduction by an actinomycete, Arthrobacter crystallopoietes ES32. Biol Trace Elem Res 97:183–194
Carbonaro RF, Stone AT (2014) Oxidation of Cr-III aminocarboxylate complexes by hydrous manganese oxide: products and time course behaviour. Environ Chem 12(1):33–51
Chen SS, Cheng CY, Li CW, Chai PH, Chang YM (2007) Reduction of chromate from electroplating wastewater from pH 1 to 2 using fluidized zero valent iron process. J Hazard Mater 142:362–367
Chen ZF, Zhao YS, Zhang JW, Bai J (2015) Mechanism and kinetics of hexavalent chromium chemical reduction with sugarcane molasses. Water Air Soil Pollut 226(11):363–372
Chen ZF, Zhao YS, Li Q (2016) Influence of Fe(II) on Cr(VI) reduction by organic reducing substances from sugarcane molasses. Water Air Soil Pollut 227(1):19–26
Chen ZF, Zhao YS, Li Q, Hong M (2017) Effect of hardness ions on Cr(VI) reduction with organic reducing substances. Environ Eng Sci 34(3):221–226
Cheung KH, Gu JD (2007) Mechanism of hexavalent Chromium detoxification by microorganisms and bioremediation application potential: a review. Int Biodeterior Biodegrad 59(1):8–15
Deiana S, Premoli A, Senette C (2007) Reduction of Cr(VI) by caffeic acid. Chemosphere 67(10):1919–1926
Dmitrenko GN, Konovalova VV, Shum OA (2003) The reduction of Cr(VI) by bacteria of the genus Pseudomonas. Microbiology 72:327–330
Elangovan R, Philip L (2009) Performance evaluation of various bioreactors for the removal of Cr(VI) and organic matter from industrial effluent. Biochem Eng J 44:174–186
Gheju M, Iovi A, Balcu I (2008) Hexavalent chromium reduction with scrap iron in continuous—flow system: part 1: effect of feed solution pH. J Hazard Mater 153(1–2):655–662
Gu CH, Shan ZX, Wang RQ (2005) Study of the soil pollution from chromium slag. Min Saf Environ Prot 32(6):18–20
Ishibashi Y, Cervantes C, Silver S (1990) Chromium reduction in Pseudomonas putida. Appl Environ Microbiol 56:2268–2270
Jardine PM, Fendorf SE, Mayes MA, Larsen IL, Brooks SC, Bailey WB (1999) Fate and transport of hexavalent chromium in undisturbed heterogeneous soil. Environ Sci Technol 33:2939–2944
Kim C, Zhou Q, Deng B, Thornton EC, Xu H (2001) Chromium (VI) reduction by hydrogen sulfide in aqueous media: stoichiometry and kinetics. Environ Sci Technol 35:2219–2225
Liu XL, Dong HL, Yang XW, Kovarik L, Chen Y, Zeng Q (2018) Effects of citrate on hexavalent chromium reduction by structural Fe(II) in nontronite. J Hazard Mater 343:245–254
Megharaj M, Avudainayagam S, Naidu R (2003) Toxicity of hexavalent chromium and its reduction by bacteria isolated from soil contaminated with tannery waste. Curr Microbiol 47:51–54
Michailides MK, Tekerlekopoulou AG, Akratos CS et al (2014) Molasses as an efficient low-cost carbon source for biological Cr(VI) removal. J Hazard Mater 281:95–105
Nakayasu K, Fukushima M, Sasaki K, Tanaka S, Nakamura H (1999) Comparative studies of the reduction behavior of chromium(VI) by humic substances and their precursors. Environ Toxicol Chem 18(6):1085–1090
Park D, Yun YS, Park JM (2004) Reduction of hexavalent chromium with the brown seaweed Ecklonia biomass. Environ Sci Technol 38:4860–4864
Park D, Yun YS, Jo JH et al (2017) Mechanism of hexavalent chromium removal by dead fungal biomass of Aspergillus niger. Water Res 39:533–540
Singleton VL, Othofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. J Wiley Sons 229(2):152–178
Somasundaram V, Philip L, Bhallamudi SM (2009) Experimental and mathematical modeling studies on Cr(VI) reduction by CRB, SRB and IRB, individually and in combination. J Hazard Mater 172:606–617
Sugiyama T, Sugito H, Mamiya K et al (2012) Hexavalent chromium reduction by an actinobacterium Flexivirga alba ST13(T) in the family Dermacoccaceae. J Biosci Bioeng 113:367–371
USEPA (2015) EPA superfund record of decision: AvcoLycoming, Pennsylvania
Wittbrodt PR, Palmer CD (1996) Effect of temperature, ionic strength, background electrolytes, and Fe(III) on the reduction of hexavalent chromium by soil humic substances. Environ Sci Technol 30(8):2470–2477
Wittbrodt PR, Palmer CD (1997) Reduction of Cr(VI) by soil humic acids. Eur J Soil Sci 48(1):151–162
Wu M, Li GX, Jiang XL, Xiao QQ, Niu MX, Wang ZY, Wang YY (2017) Non- biological reduction of Cr(VI) by reacting with humic acids composted from cattle manure. RSC Adv 7(43):26903–26911
Yan BZ, Xiao CL, Liang XJ, Wu SL (2017) Influences of pH and CO2 on the formation of Metasilicate mineral water in Changbai Mountain, Northeast China. Appl Water Sci 7(4):1657–1667
Yurik TK, Pikaev AK (1999) Radiolysis of weakly acidic and neutral aqueous solutions of hexavalent chromium ions. High Energy Chem 33(4):208–212
Zhilin DM, Schmitt-Kopplin P, Perminova IV (2004) Reduction of Cr(VI) by peat and coal humic substances. Environ Chem Lett 2(3):141–145
Acknowledgements
This work was financially supported by the Scientific Research Initiation funds for Ph.D. scholars (BQ2017011); China’s Post-doctoral Science Fund (2018M631874); Scientific Research Projects of the Higher University in Hebei (ZD2014023); Hebei province water conservancy science and technology plan projects (2017-59), Youth Foundation of Hebei Province Department (QN2015061, QN2017026); Natural science fund project in Hebei province (D2018403040); and Institutions of higher learning scientific research project, Hebei province.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animals rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Yan, Bz., Chen, Zf. Influence of pH on Cr(VI) reduction by organic reducing substances from sugarcane molasses. Appl Water Sci 9, 61 (2019). https://doi.org/10.1007/s13201-019-0940-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-019-0940-x