Abstract
Introduction
The so-called radiation-induced glioma (RIG, a secondary glioma after cranial irradiation), is a serious late effect after cranial radiation therapy. The clinical characteristics of and ideal treatment for these tumors are unclear. We analyzed our case series and conducted a comprehensive literature review to reveal the precise characteristics of RIGs.
Methods
We analyzed the cases of six patients with RIGs treated at our institution and 354 patients with RIGs from the literature. The latency period from irradiation to the development of each RIG and the median overall survival of the patients were subjected to Kaplan–Meier analyses. Spearman’s correlation test was used to determine the relationship between age at irradiation and the latency period.
Results
The mean age of the 360 patients at the development of RIG was 27.42 ± 17.87 years. The mean latency period was 11.35 ± 8.58 years. Multiple gliomas were observed in 28.4%. WHO grade 3 and 4 RIGs accounted for 93.3%. The latency periods were significant shorter in the higher WHO grade group (p = 0.0366) and the concomitant systemic chemotherapy group (p < 0.0001). Age at irradiation was negatively associated with the latency period (r =− 0.2287, p = 0.0219). The patients treated with radiotherapy achieved significantly longer survival compared to those treated without radiotherapy (p = 0.0011).
Conclusions
Development in younger age, multiplicity, and high incidence of grade 3 and 4 are the clinical characteristics of RIGs. Cranial irradiation at older ages and concomitant chemotherapy were associated with shorter latency for the development of RIG. Radiation therapy may be the feasible treatment option despite radiation-induced gliomas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gliomas are the most frequent parenchymal brain tumors of the human central nervous system (CNS) [1]. The etiology of gliomas remains unclear, but it is known that some inherited syndromes such as neurofibromatosis type 1 (NF1), Li-Fraumeni syndrome, Lynch syndrome, and constitutional mismatch repair deficiency increase the risk of the occurrence of a glioma, as does the use of ionized radiation [2]. A study of the lifespans of atomic bomb survivors revealed a risk of glioma development with increasing radiation exposure [3].
Cranial radiation therapy (RT) plays important roles in the treatment of diseases such as brain tumors, hematological malignancies, and head and neck disorders. Although the use of cranial RT has improved survival times, cranial RT is also known to increase the risks of leukoencephalopathy [4], cystic malacia [5], cognitive impairment [6], pituitary dysfunction [7], vascular arteritis including radiation-induced moyamoya syndrome [8], cavernous malformation [9], and secondary neoplasms [10].
Radiation-induced secondary intracranial neoplasms occur in the brain parenchyma or meninges; meningiomas, sarcomas, and gliomas are the most common secondary intracranial neoplasms. A secondary glioma that develops after the applications of irradiation, i.e., a radiation-induced glioma (RIG), is a more serious secondary neoplasm compared to meningiomas. Although sporadic diffuse gliomas are histologically subdivided into CNS World Health Organization (WHO) grades 2–4 and are also classified based on molecular features [11], RIGs have not been well characterized. In addition, ideal treatment protocols for RIGs have not been established. We conducted the present study to determine the precise characteristics of RIGs and investigate their optimal treatment. To do so, we performed a retrospective analysis of the series of patients with RIGs treated at our institution and the cases identified in a comprehensive literature review.
Methods
To reveal the clinical and pathological characteristics of RIGs and evaluate feasible treatment strategies for this tumor, we evaluated our case series of six patients with RIGs and performed a review of the relevant literature, which identified 354 cases.
Design of the study
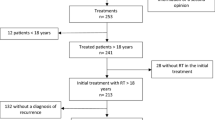
This retrospective study was approved by our institutional review board (approval no. E2022-0031). We first collected the medical records of all cases of surgically confirmed gliomas treated at our institution during the period from 2000 to 2021. We identified six patients who had a previous history of irradiation. We excluded the patients without pathological confirmation of a RIG.
Criteria for secondary glioma after irradiation
In this study, tumors that met the following criteria established by Cahan et al. [12] were considered RIGs: (1) the tumor arose within an irradiated field, (2) a sufficient latency period had passed between the time point of irradiation and the development of the second tumor (in this study, RIG cases with > 11-month latency periods were included), (3) the secondary tumor was histologically distinct from the primary tumor, and (4) the patient had no genetic history of cancer predisposition (e.g., Li-Fraumeni syndrome or neurofibromatosis).
Selection of articles and literature review
Two of the authors (SO and FY) conducted a comprehensive literature search for “radiation-induced glioma,” “radiation associated glioma,” “secondary glioma,” and “secondary neoplasms after radiation therapy” in the Pubmed and MEDLINE databases up to December 29, 2021. The terms “glioma,” “glioblastoma,” “gliosarcoma,” “brain tumor,” and “secondary neoplasm” were searched in combination with “radiotherapy-induced,” “radiation therapy,” and “after radiation.” We obtained all of the articles that were potentially eligible for inclusion in our review. The references listed in all potentially eligible articles were inspected in order to identify other eligible articles. Review articles that did not report original individual patients’ data were excluded, although their references were checked for other eligible articles. We identified published cases from 1960 to 2021 (Suppl. Table S1) and added the six cases from our institution.
Statistical analyses
After the review of all RIG cases in the literature, we evaluated the differences in the number of latency years for the development of RIGs by conducting Kaplan–Meier analyses between pairs of groups based on factors such as gender, WHO grade, irradiation dose, and chemotherapy. The overall survival (OS) of the patients with RIGs were also analyzed by a Kaplan–Meier analysis between different treatment groups. The log-rank tests were used to compare the latency years and OS between groups. Fisher’s exact test was performed to evaluate the relationship between the RIG lesions and the primary diseases. Spearman’s correlation test was used to determine the relationship between the patient's age at irradiation and the number of years to RIG diagnosis. Statistical significance was assigned as p < 0.05. The statistical analyses were performed using JMP pro 15.0 (SAS, Cary, NC, USA) and GraphPad Prism, ver. 7.00 for Mac (GraphPad Software, San Diego, CA).
Results
Our case series
Our institutional case series was six patients: three females and three males. None of these patients had undergone radiation exposure other than the radiation to their primary lesions. The median age at the development of their RIGs was 43.5 years (range 27–65 years). The latency period from irradiation to the development of RIG was from 5 to 38 years, with a median latency period of 21 years. Half of the six patients presented multiple lesions. Histopathological examinations had revealed three cases of WHO grade 4 (two cases of glioblastoma and a case of gliosarcoma), two cases of WHO grade 3 (one of anaplastic astrocytoma and one of anaplastic oligodendroglioma), and a WHO grade 2 diffuse astrocytoma. The histological diagnoses were based on the WHO guideline at the time the RIGs were diagnosed. In additional immunohistochemical analyses, all six cases were negative for IDH1-R132H immunohistochemical staining. A summary of this case series is provided in Table 1. Details of Case 5 were presented in an earlier report [13].
Literature review results
We identified 181 literatures that met the inclusion criteria. For this review, we collected the information of patients with RIGs including the six cases described above. Overall, we analyzed 360 cases of RIG. The details of these cases are summarized in Supplementary Table S1. The patient's age at the development of RIG was available in 356 cases. The mean age and standard deviation was 27.42 ± 17.87 years (median 20 years) with a peak in the 10 s decade; the numbers of RIG patients by decade of life were < 10 years (n = 31), 10 s (n = 141), 20 s (n = 54), 30 s (n = 41), 40 s (n = 38), 50 s (n = 28), 60 s (n = 12), 70 s (n = 10) and 80 s (n = 5). Table 2 presents a summary of the patients’ clinical information, including gender, primary disease, total irradiation dosage, WHO grade, isocitrate dehydrogenase (IDH) status, chemotherapy for the primary lesion, age at irradiation, number of years to the development (latency) of RIGs, and the patient's age at the development of RIGs. The age distribution of the patients at the development of RIG is depicted in Fig. 1a, and Fig. 1b shows the age distribution of the patients with malignant gliomas (WHO grades 3 and 4) based on records we obtained from the Brain Tumor Registry of Japan (BTRJ) 2005–2008.
Relationship between primary disease and the irradiated field and the RIG locations
Whole-brain irradiation was performed in 106 cases: 85 cases (80.2%) of them were patients with hematologic malignancies, six cases (5.7%) were patients with medulloblastomas, and three patients had brain metastasis. Cranio-spinal irradiation (CSI) was performed in 70 cases: 48 cases (68.6%) were patients with medulloblastoma, 11 cases (15.7%) were a hematologic malignancy, and six cases (8.6%) were germ cell tumors. Stereotactic radiosurgery (SRS) was performed in 14 cases: five (35.7%) were patients with arteriovenous malformation, four (28.6%) were vestibular schwannomas, three (21.4%) were meningiomas, and two cases (14.2%) were brain metastasis. In the rest of the cases, the details of radiation therapy were not available.
The locations of the RIGs were obtained in 275 cases; among them 197 RIGs (71.6%) had developed in the supratentorial region. In 77 cases (28.0%), the RIGs had developed in the infratentorial region. A single patient (0.4%) presented RIGs at both the supratentorial and infratentorial regions.
The patients' primary diseases were subclassified into medulloblastoma, germ cell tumor, other primary brain tumor, brain metastasis, hematological malignancy, cerebrovascular disease, scalp disease, and other disease. In the medulloblastoma group, (66%) of the RIGs developed predominantly in the infratentorial region. In the brain metastasis group, the incidence of RIGs in the supratentorial and infratentorial regions were comparable. In the other groups, the RIGs developed predominantly in the supratentorial region. Fisher’s exact test showed that the medulloblastoma cases were significantly more likely to develop an infratentorial RIG compared to the other primary-disease groups (p < 0.01). The detailed results of this analysis are presented in Fig. 1c.
Pathological and molecular characteristics of RIGs
The histological WHO grade of the RIGs was available in 341 cases. The proportion of WHO glioma grades were as follows; grade 1: 0.3%, grade 2: 6.5%, grade 3: 29.6%, and grade 4: 63.6%. Multiple gliomas were observed in 50 of 178 RIG cases (28.1%).
The IDH status was obtained in 24 cases. IDH-wildtype gliomas accounted for 23 (95.8%) of the 24 cases. Only one case of an IDH-mutant RIG has been reported.
Clinical factors associated with the latency period for the development of RIG
Among all 360 cases, the mean and standard deviation of the latency period from irradiation to the development of the patient's RIGs were 11.35 ± 8.58 years (range 0.92–61 years). The latency periods did not show a significant difference between the female and male patients (p = 0.1601, log-rank test) (Fig. 2a). The median latency period of the high-grade RIGs (WHO grades 3 or 4) was 9 years, which is significantly shorter than the 13-year latency period of the low-grade RIGs (WHO grade 1 or 2) (p = 0.0366, log-rank test) (Fig. 2b).
Kaplan–Meier analysis for the number of years (latency) from irradiation to the development of the RIGs. The latency years were classified by gender (a), WHO grade of RIG (b), radiation dose (c), and the use vs. non-use of systemic chemotherapy for the primary lesion (d). In panel b, WHO grades 3/4 are presented as high-grade tumors and WHO grades 1/2 are presented as low-grade tumors. In panel c, the radiation doses were classified into the high-dose (≥ 40 Gy), intermediate (< 40 Gy, ≥ 20 Gy), and low-dose (< 20 Gy) groups. e: Spearman’s correlation test between age at irradiation and the number of years to the development of the RIG
We next subclassified the patients into three groups based on the irradiation doses they had received: the high-dose (≥ 40 Gy) group, the intermediate-dose (< 40 Gy, ≥ 20 Gy) group, and the low-dose (< 20 Gy) group. The median latency period was 9.5 years in the high-dose group, 9 years in the intermediate-dose group, and 8 years in the low-dose group. These latency periods for the development of RIG did not show a significant difference among the irradiation dose groups (p = 0.1035, log-rank test) (Fig. 2c).
To evaluate the effect of chemotherapy, we compared the latency periods between the patients with and without systemic chemotherapy for their primary disease. The latency period for the development of RIG was significantly shorter in the patients who had received systemic chemotherapy (p < 0.0001, log-rank test) (Fig. 2d). The examination of effect of the patient’s age at irradiation revealed that older age at irradiation was correlated with a significantly shorter latency period for the development of RIG (r =− 0.2287, p = 0.0219, Spearman’s correlation test) (Fig. 2e).
Treatment and prognosis of radiation-induced gliomas
Several treatment strategies for RIGs have been reported: surgery, chemotherapy, and radiation therapy. In our present review of the literature, the details of adjuvant therapy for RIGs and the survival time were described in 123 of 362 cases (34.0%). Adjuvant chemotherapy was performed in 82.1% using various chemotherapeutic agents; temozolomide, bevacizumab, CCNU (lomustine), and others. Radiotherapy for a radiation-induced glioma was performed in 69.1% of the RIG patients.
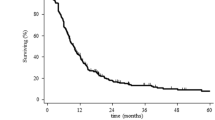
The median overall survival (OS) among all of the RIG patients studied herein was 11 months. The median survival benefit of radiotherapy for RIG was 6 months; the median survival was 16 months with radiotherapy and 10 months without radiotherapy (hazard ratio [HR] for death, 0.5226; 95%CI: 0.3254–0.8398, p = 0.0011, log-rank test) (Fig. 3a). The comparison of the prognosis of the patients treated with or without systemic chemotherapy for RIG revealed that the median OS did not differ significantly between these groups (HR for death, 0.81225; 95%CI: 0.4637–1.424, p = 0.4201, log-rank test) (Fig. 3b). The median OS also showed no significant difference between the two groups of high and low WHO grades (p = 0.9231, log-rank test) (Fig. 3c).
a Kaplan–Meier analyses of the patients' median overall survival (OS). The median OS was 16 months in the patients treated with radiotherapy and 10 months for the patients treated without radiotherapy (p = 0.0011, log-rank test). b Comparison of the patients treated with or without systemic chemotherapy for RIG: the median OS did not differ significantly between these groups (p = 0.4201, log-rank test). c The median OS was not significantly different between the WHO grade groups (p = 0.9231, log-rank test)
Discussion
This study assessed the characteristics of RIGs and considered the ideal treatment strategies for these tumors. Our analyses revealed that higher patient age at cranial irradiation and concomitant chemotherapy were associated with shorter latency to the development of RIGs. The results also suggest that radiation therapy may be the feasible treatment option for RIGs despite radiation-induced gliomas.
Gliomas are the one of the common intraparenchymal brain tumors. In a general population examined during the period 2012–2016 and described in the Central Brain Tumor Registry of the United States (CBTRUS) Statistical Report, the median patient ages at the diagnoses of diffuse astrocytoma, anaplastic astrocytoma and glioblastoma were 47, 53, and 65 years, respectively [1]. Gliomas are also one of the most common subsequent CNS neoplasms after cranial irradiation [14]. In contrast to the gliomas in general populations, in our present investigation the median age of RIG patients was 20 years with a peak in the 10 s decade. The age distribution of the development of gliomas (Fig. 2) indicates that RIGs develop at younger ages compared to sporadic malignant gliomas. Our analyses also revealed that RIGs were more likely to present multiple lesions compared to sporadic gliomas. In a 2019 meta-analysis, the prevalence rate of multiple high-grade glioma was 19% [15]. In the present study, multiple gliomas were observed in 50 of 178 (28.1%) RIG cases.
We also observed that the location of the development of RIGs varied depending on the type of primary disease, and thus on the irradiation field. For example, in the present patients with a medulloblastoma in the posterior fossa, which is commonly treated with local radiation with craniospinal irradiation, the RIGs developed predominantly in the infratentorial region. In the patients with germ cell tumors that were mainly at a suprasellar or pineal region (which is treated with whole ventricular radiotherapy followed by a boost to the tumor bed), the RIGs developed predominantly in the supratentorial region. These results imply that a concentrated irradiated field may be associated with the development of a RIG.
Regarding the pathological and molecular characteristics of RIGs, we found only one IDH-mutant RIG case. In this patient, concurrent mutations were also observed; a missense mutation of TP53 and a short in-frame deletion of MLH1; although an analysis of germline variants was not performed, the authors of that case report did not rule out the possibility of Lynch syndrome [16]. In another report, RIGs were genetically distinct from sporadic gliomas [17]. Different diagnostic and clinical approaches to RIGs are thus necessary.
Our study also revealed clinical factors that ae significantly associated with shorter latency for the development of RIG: concomitant chemotherapy and higher age at cranial irradiation. An earlier investigation of RIGs including spinal gliomas and ependymomas described a shorter latency period in patients treated with concomitant systemic chemotherapy [18]. In our larger present review of intracranial RIGs, the patients who underwent systemic chemotherapy for their primary diseases showed significant shorter latency periods from simple radiotherapy to the development of the RIGs. Cytotoxic effects of chemotherapy (e.g., that with alkylating agents) lead to DNA damage and tumorigenesis [19, 20], which would cause a shorter latency to the development of RIGs.
Our present statistical analyses showed that older patient age at irradiation was associated with a shorter latency period for the development of a RIG (r =− 0.2286, p = 0.0219, Spearman’s correlation test). In the general population, the peak incidence of glioblastoma is in the 75–84-year age range. The age-adjusted incidence of glioblastoma increases with aging, except for a decrease in patients > 85 years old [1]. Aging is an independent risk factor for the development with gliomas, and we suspect that irradiation could have been the final trigger of tumorigenesis.
The indications for radiation therapy for radiation-induced tumors are controversial. In our review, nearly 70% of the RIG patients received radiation therapy. The patients who were treated with radiation therapy achieved longer survivals compared to the patients without radiation therapy. The prognosis of patients with a RIG is poor with the median OS of 11 months, and thus radiation therapy for a RIG could be considered as an acceptable treatment option even if it exceeds the total tolerable radiation exposure. In a recent analysis concerning reirradiation for glioblastoma, the risk of radiation necrosis was approx. 0–3% after < 101 Gy with a conventional fraction at a cumulative equivalent total dose normalized to 2 Gy/fraction [21]. However, for the prevention of further complications, radiation therapy for patients with a RIG requires careful consideration of the extent of the irradiation field and the irradiation methods. Regarding chemotherapy for RIGs, several cases of marked responses to nimustine hydrochloride (ACNU) or temozolomide were reported, but the RIGs eventually relapsed in these cases [22, 23]. In the present review, chemotherapy for RIG did not show a survival benefit.
Our study has several limitations. It was a retrospective analysis that included only patients who developed gliomas after cranial radiation therapy, rather than all patients who had received cranial radiation therapy. The clinical information is limited due to the lack of data from previous publications. Larger prospective studies are needed to determine the actual cumulative incidence and risk of RIGs.
Conclusions
Development at a younger age, multiplicity, and high incidence of grade 3 and 4 tumors are the clinical characteristics of RIGs. Cranial irradiation at older ages and concomitant chemotherapy were associated with a shorter latency period for the development of RIG. Radiation therapy is the most feasible treatment option despite the risk of radiation-induced gliomas.
References
Ostrom QT, Cioffi G, Gittleman H et al (2019) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2012–2016. Neuro Oncol 21:V1–V100. https://doi.org/10.1093/neuonc/noz150
Ostrom QT, Bauchet L, Davis FG et al (2014) The epidemiology of glioma in adults: a state of the science review. Neuro Oncol 16:896–913. https://doi.org/10.1093/neuonc/nou087
Brenner AV, Sugiyama H, Preston DL et al (2020) Radiation risk of central nervous system tumors in the life span study of atomic bomb survivors, 1958–2009. Eur J Epidemiol 35:591–600. https://doi.org/10.1007/s10654-019-00599-y
Terziev R, Psimaras D, Marie Y et al (2021) Cumulative incidence and risk factors for radiation induced leukoencephalopathy in high grade glioma long term survivors. Sci Rep 11:1–9. https://doi.org/10.1038/s41598-021-89216-1
Yamasaki F, Takayasu T, Nosaka R et al (2017) Development of cystic malacia after high-dose cranial irradiation of pediatric CNS tumors in long-term follow-up. Child’s Nerv Syst 33:957–964. https://doi.org/10.1007/s00381-017-3400-7
Greene-Schloesser D, Robbins ME (2012) Radiation-induced cognitive impairment-from bench to bedside. Neuro Oncol. https://doi.org/10.1093/neuonc/nos196
Vatner RE, Niemierko A, Misra M et al (2018) Endocrine deficiency as a function of radiation dose to the hypothalamus and pituitary in pediatric and young adult patients with brain tumors. I J Clin Oncol 36:2854–2862. https://doi.org/10.1200/JCO.2018.78.1492
Desai SS, Paulino AC, Mai WY, Teh BS (2006) Radiation-induced moyamoya syndrome. Int J Radiat Oncol Biol Phys 65:1222–1227. https://doi.org/10.1016/j.ijrobp.2006.01.038
Lew SM, Morgan JN, Psaty E et al (2006) Cumulative incidence of radiation-induced cavernomas in long-term survivors of medulloblastoma. J Neurosurg. https://doi.org/10.3171/ped.2006.104.2.103
Tsui K, Gajjar A, Li C et al (2015) Subsequent neoplasms in survivors of childhood central nervous system tumors: risk after modern multimodal therapy. Neuro Oncol 17:448–456. https://doi.org/10.1093/neuonc/nou279
Louis DN, Perry A, Wesseling P et al (2021) The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro Oncol 23:1231–1251. https://doi.org/10.1093/neuonc/noab106
Cahan WG, Woodard HQ (1948) Sarcoma arising in irradiated bone; report of 11 cases. Cancer 1:3–29. https://doi.org/10.1002/1097-0142(194805)1:1%3c3::aid-cncr2820010103%3e3.0.co;2-7
Doskaliyev A, Yamasaki F, Kenjo M et al (2008) Secondary anaplastic oligodendroglioma after cranial irradiation: a case report. J Neurooncol 88:299–303. https://doi.org/10.1007/s11060-008-9564-y
Neglia JP, Robison LL, Stovall M et al (2006) New primary neoplasms of the central nervous system in survivors of childhood cancer: a report from the childhood cancer survivor study. J Natl Cancer Inst 98:1528–1537. https://doi.org/10.1093/jnci/djj411
Di Carlo DT, Cagnazzo F, Benedetto N et al (2019) Multiple high-grade gliomas: epidemiology, management, and outcome. a systematic review and meta-analysis. Neurosurg Rev 42:263–275. https://doi.org/10.1007/s10143-017-0928-7
Mascelli S, Nozza P, Sak K et al (2016) Distinctive genetic profile with IDH1, TP53, and MLH1 mutations in a radiation-induced anaplastic astrocytoma. Pediatr Blood Cancer 63:179–179. https://doi.org/10.1002/pbc.25671
Whitehouse JP, Howlett M, Federico A et al (2021) Defining the molecular features of radiation-induced glioma: a systematic review and meta-analysis. Neuro-Oncology Adv 3:1–16. https://doi.org/10.1093/noajnl/vdab109
Yamanaka R, Hayano A, Kanayama T (2018) Radiation-induced gliomas: a comprehensive review and meta-analysis. Neurosurg Rev 41:719–731. https://doi.org/10.1007/s10143-016-0786-8
Blagosklonny MV (2005) Carcinogenesis, cancer therapy and chemoprevention. Cell Death Differ 12:592–602. https://doi.org/10.1038/sj.cdd.4401610
Borgmann A, Zinn C, Hartmann R et al (2008) Secondary malignant neoplasms after intensive treatment of relapsed acute lymphoblastic leukaemia in childhood. Eur J Cancer 44:257–268. https://doi.org/10.1016/j.ejca.2007.09.019
Minniti G, Niyazi M, Alongi F et al (2021) Current status and recent advances in reirradiation of glioblastoma. Radiat Oncol. https://doi.org/10.1186/s13014-021-01767-9
Monje ML, Ramakrishna NR, Young G et al (2007) Durable response of a radiation-induced, high-grade cerebellar glioma to temozolomide. J Neurooncol 84:179–183. https://doi.org/10.1007/s11060-007-9354-y
Kon T (2013) Radiation-induced glioblastoma following radiotherapy for pituitary adenomas: marked response to chemotherapy. J Neurol Neurophysiol 04:3–5. https://doi.org/10.4172/2155-9562.1000155
Funding
The authors declare that no funds, grants, or other support were received for the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The material preparation and the data collection and analyses were performed by SO and FY. The first draft of the manuscript was written by SO, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Onishi, S., Yamasaki, F., Amatya, V.J. et al. Characteristics and therapeutic strategies of radiation-induced glioma: case series and comprehensive literature review. J Neurooncol 159, 531–538 (2022). https://doi.org/10.1007/s11060-022-04090-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-04090-9