Abstract
In this paper, ferrous powder has been used as flame retardant and smoke suppression synergism with ammonium polyphosphate (APP) in flame retardant thermoplastic polyurethane (TPU) composites. The synergistic flame retardant and smoke suppression effects between ferrous powder and APP have been studied using cone calorimeter test (CCT), smoke density test (SDT), and limiting oxygen index (LOI). The CCT results showed that appropriate amount of ferrous powder can greatly decrease heat release rate, total heat release, mass loss, smoke production rate, total smoke release, and smoke factor. The SDT results indicated that ferrous powder can greatly improve the luminous flux of flame retardant TPU composites in the test with flame; however, the luminous flux decreases with the addition of ferrous powder in the test without flame. And the LOI results showed that the LOI value of the samples with ferrous powder is higher than that of the sample with only APP. The above results imply there are synergistic flame retardant and smoke suppression effects between ferrous powder and APP in TPU composites. Then, the thermo-gravimetric (TG) and scanning electron microscopy (SEM) were used to investigate the synergistic flame retardant and smoke suppression mechanism between ferrous powder and APP in TPU composites. The TG and DTG results showed that ferrous powder can decrease the initial decomposition temperature and improve the thermal stability at high temperature for flame retardant TPU composites. The SEM results showed that ferrous powder can improve the quality of char residues after CCT, resulting good flame retardant and smoke suppression properties for TPU composites containing both APP and ferrous powder. This is a very meaningful result in fire safety materials fields.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
As an engineering plastic, thermoplastic polyurethane (TPU) has been applied in many fields such as automotive parts, electrical and electronic industries, mechanical transmission parts, household and cosmetic parts, due to its excellent physical properties, chemical resistance, abrasion resistance, and good adhesion to chemicals and self-lubrication performance. However, the high flammability of TPU and its production of much smoke and toxic gas (i.e., benzene and other aromatic compounds) during burning has greatly limited its broad applications in various fields above mentioned [1–4]. So, the improvement of the flame retardancy and smoke suppression during combustion of TPU becomes increasingly vital to satisfy the increasing requirements in the practical use [5]. And developing the retardants with good flame retardancy and smoke suppression is the key to further explore the TPU applications. Many flame retardant additives used to reduce the flammability and smoke formation of polymer have been studied and reported [6–11].
Indeed, it has been reported that most fire deaths are due to toxic gases, oxygen deprivation, and other effects that have been widely referred to as smoke inhalation instead of burns in the USA [12]. In many cases, the visibility impairing and narcotic irritating effect of fire gases is regarded as the decisive factor preventing many fire victims from perceiving their possibilities of escape [13]. As a consequence, reducing the flammability and smoke formation of polymer of TPU becomes more important.
Many investigations demonstrated that halogen-containing flame retardants show effective flame retardant properties in TPU. However, the utilization of such retardants has been limited due to generation of toxic gas, causing life and environmental problems. Thus, a wide range of halogen-free flame retardants has been developed and used in TPU, which are mostly phosphorus-containing flame retardants and achieve good improvement [14]. The addition of flame retardants is an effective way to reduce flammability [15]. With advantages such as low release of smoke and toxic gas characteristics, eco-friendly intumescent flame retardants have been well developed as replacements for the halogen-containing flame retardants [16, 17]. In addition, the additives include compounds containing antimony, tin, zinc, copper, iron, and molybdenum, etc., while the most important commercial smoke suppressants are iron and molybdenum compounds. In our previous work, it has been found that ferrous powder can be used as intumescent flame retardant synergism in PP composites [18]. To the best of our knowledge, no work has been reported the synergistic effects between ferrous powder and APP on achievement of smoke suppression and flame retardant properties in TPU composites.
In this paper, ferrous powder was used as flame retardant and smoke suppression synergism to improve the flame retardant and smoke suppression efficiency in TPU composites based on APP. The synergistic flame retardant and smoke suppression properties of ferrous powder with APP were intensively investigated using CCT, SDT, and LOI. And the synergistic flame retardant and smoke suppression mechanism was further studied using SEM and TG.
Experimental
Materials
Commercial TPU (9380A) was produced by Bayer, German. The basic properties of TPU are as follows, density: 1.110 g cm−3 (ISO1183); hardness: 82A (ISO868); tensile strength: 40 MPa (ISO527-1, -3); elongation at break: 500 % (ISO527-1, -3). APP with particle size of 2,500 mesh was purchased from new thin Metal and Chemical Co., Ltd., Guangzhou, China. Ferrous powder with the size of 0.5–10 μm was purchased from the Qingdao Zhicheng Trade Co., Ltd., Qingdao, China.
Sample preparation
Before processing experiment, TPU was dried in oven at 80 °C for 8 h. APP was dried in oven at 100 °C for 10 h. A certain amount of TPU was melted in the mixer at 175 °C. Then, a certain amount of ferrous powder and APP were added into the mixer, respectively. The blends were mixed for 10 min and pressed into sheets using tablet press machine. The formulations of flame retardant TPU composites are listed in Table 1.
Measurements
Cone calorimeter test (CCT)
The cone calorimeter (Stanton Redcroft, UK) tests were performed according to ISO 5660 standard procedures. Each specimen with dimensions of 100 × 100 × 3 mm3 was wrapped in aluminum foil and exposed horizontally to an external heat flux of 50 kW m−2.
Smoke density test (SDT)
A smoke density test machine (JQMY-2, Jianqiao Co, China) was used to measure the smoke characteristics according to ISO 5659-2 (2006). Each specimen with dimensions of 75 × 75 × 2.5 mm3 was wrapped in aluminum foil and exposed horizontally to an external heat flux of 25 kW m−2 with or without the application of a pilot flame.
Thermo-gravimetric (TG)
Thermo-gravimetric analysis (TG) of the sample was performed using a DT-50 (Setaram, France) instrument. About 10.0 mg of sample was put in an alumina crucible and heated from ambient temperature to 750 °C. The heating rates were set as 20 K min−1 (nitrogen atmosphere, flow rate of 20 mL min−1).
Scanning electron microscopy (SEM)
Scanning electron microscopy (SEM) studies were performed using a Hitachi X650 scanning electron microscope.
Limiting oxygen index (LOI)
LOI was measured according to ASTM D2863. The apparatus used was an HC-2 oxygen index meter (Jiangning Analysis Instrument Company, China). The specimens used for the test were of dimensions 10 × 6.5 × 3 mm3.
Results and discussion
Cone calorimeter test
Heat release rate (HRR)
Figure 1 shows a comparison between the heat release rate (HRR) curves for pure TPU and TPU blends with different amount of APP and ferrous powder. It can be observed from Fig. 1 that pure TPU (TPU-0) burns quickly with a high peak HRR (PHRR) value of 1,460.1 kW m−2. For TPU-1 with only APP, the value of PHRR was dramatically reduced to 152.4 kW m−2; and two peaks can be identified in the HRR curve, which can be considered characteristic feature for intumescent flame retardant polymer composites. The first peak followed immediately the ignition point. And the second peak corresponded to a huge increase in the HRR value over a short period of time. In this case, the first peak is identified as the development of the intumescent char, which could protect samples. After the first peak, the HRR curves tended to a relatively steady case, in which the increase in HRR was suppressed because of the presence of the efficient protective carbon layer. The second peak is due to the gradual degradation of the protective carbon layer as the sample is continuously exposed to the heat, and the formation of a new protective char because of the flame retardant, which then gives rise to the third and then the fourth HRR peak [19, 20]. Moreover, the PHRR values of the samples with both APP and ferrous powder decrease significantly compared with that of TPU-1 with only APP. And when ferrous powder is incorporated into TPU/APP system, the PHRR value further decreases. The PHRR value of TPU-2 with 0.625 mass% ferrous powder is 126.5 kW m−2, which is lower than that of TPU-1 (152.4 kW m−2). And TPU-5 with 3.750 mass% ferrous powder has the lowest PHRR value (98.6 kW m−2) among all samples. The dramatic decrease of PHRR in the samples with the addition of ferrous powder can be interpreted that ferrous powder can improve the compact, expansion degree and mass of char residue in the cone calorimeter test [21–24].
Total heat release (THR)
The results of THR during combustion are shown in Fig. 2. The THR value of TPU-0 is 121.3 MJ m−2 at 400 s. When APP is introduced into TPU, the THR value of TPU-1 is reduced to 40.8 MJ m−2, which is much lower than that of TPU-0. In the case of sample TPU-2 with 0.625 mass% ferrous powder, the THR value is larger than that of TPU-0 before 200 s, which indicates that ferrous powder promotes pyrolysis process of TPU/APP composites. In this pyrolysis process, there is a compact char residue shell with high expansion degree formed on the surface of TPU-2, leading low THR between 200 and 650 s. When the combustion time exceeds 650 s, the THR of TPU-2 is higher than that of TPU-1. This is because the initial quality of TPU-2 is larger than that of TPU-1. As shown in Fig. 3, the mass loss of TPU-1 is larger than that of TPU-2. With the content of ferrous powder further increasing, the THR decreased greatly. And the TPU-5 with 3.750 mass% ferrous powder shows the lowest THR value of 34.4 MJ m−2 among all samples. It is reported that the gradient of THR curve can be assumed as representative of flame spread [25, 26]. The gradient of THR curve from TPU-1 with only APP is reduced more than that from TPU-0, which indicated that the flame spread speed slows down. In the case of the samples with ferrous powder, the flame spread speed further decreases, which is due to that there is a much expansive and compact char residue forming on the surface of the sample, restricting flames spread.
Mass loss
Figure 3 gives the mass loss curves versus time for all samples. From Fig. 3, it can be seen that the mass loss of TPU-0 is very large, just 10 % remained at 200 s. However, there is about 75.5 % char residue of TPU-1 at 200 s. This can be illustrated that an intumescent char, creating as a physical protective barrier for heat and mass transfer, may emerge on the surface of the flame retardant sample during combustion. The char would prevent oxygen from diffusing to the underlying substrate or give a low volatilization rate. When ferrous powder is incorporated into TPU/APP system, the mass loss is lower than that of TPU-1. This can be explained that ferrous powder can change the structure of char residue in the cone calorimeter test. It has been reported that ferrous powder can make the char residue much compact with high expansion degree [18]. The mass loss result is in agreement with the behavior of heat release and smoke suppression. The decrease in mass loss is attributed to char formation and its morphological structure on the surface of the composites [25].
Smoke production rate (SPR)
Smoke performance of flame retardant material during combustion plays an important factor concerning fire safety: Heavy smoke can hinder escape and toxic gases act as one killer during the fire hazard. The smoke production rate (SPR) curves of all samples are illustrated in Fig. 4. It can be clearly seen that a significant decrease of the SPR curves taken on as the flame retardants added. The peak SPR value of TPU-1 containing only APP is decreased to 0.057 m2 s−1 from 0.105 m2 s−1 of TPU-0 in the combustion process. The time to peak SPR of TPU-1 is 30 s, which is much shorter than that of TPU-0 (160 s). This can be attributed to the fact that APP decomposes at low temperature to form some smoke particulates. Moreover, the samples containing both APP and ferrous powder show lower peak SPR values than that of TPU-1 in the combustion process. The main reason is that ferrous powder would help to promote charring and formation of carbon layer. So, the SPR decreased in the process of formation of carbon layer. Because of the protection of the carbon layer, combustible gases and smoke-forming materials reduced rapidly in the gas phase during combustion [27]. As a result, the SPR values of the samples (TPU-2 to TPU-5) are all below 0.03 m2 s−1 after 50 s in the cone calorimeter test. Especially, the SPR value of TPU-5 is below 0.02 m2 s−1 after 50 s. It can be concluded that ferrous powder is an effective additive to suppress smoke production in the combustion process of TPU/APP composites.
Total smoke release (TSR)
Figure 5 shows the total smoke release (TSR) curves of TPU composites in the cone calorimeter test at a flux 50 kW m−2. It can be seen that there is no difference between pure TPU and the flame retarded samples until an obviously distinction is apparent after 50 s. For example, at the end of the experiments, the TSR values of TPU-0 and TPU-1 are 980.0 m2 m−2 and 590.1 m2 m−2 at 400 s, respectively. But, the TSR value of TPU-4 is 407.2 m2 m−2. It can conclude that ferrous powder plays a significant role in the decrease in the TSR values. Some studies have shown that the smoke suppression effect of ferrous powder has two aspects. One can be by promoting the soot particle oxidation to CO and CO2; another can be by promoting the formation of compact carbon layer to prevent combustible gases and smoke precursor from diffusing into air [28, 29]. The TSR value of TPU-4 is lower than that of TPU-5 which can be illustrated by mass loss results from Fig. 3. It can be seen from Fig. 3, the mass of TPU-4 and TPU-5 is 56 and 50 mass%, respectively, at the end of the combustion test.
Smoke factor (SF)
SF is the product of PHRR and TSR [30]. Figure 6 gives the smoke factor (SF) as a function of time for all samples. It can be seen that the SF value of TPU-1 containing only APP is decreased to 115.8 MW m−2 from 1,609.9 MW m−2 of TPU-0. It is very clear that the addition of flame retardant significantly reduces the SF values of TPU composites. Furthermore, the samples containing both APP and ferrous powder show further decrease in SF values compared with TPU-1. It is apparent that the SF values for different samples decrease in turn as the amount of ferrous powder increased. And the TPU-5 is just 51.1 MW m−2.
Smoke density test
The smoke density test gives detailed information about the smoke production. Figure 7a presents the luminous flux curves of flame retardant TPU composites with flame in the smoke density test. In the case of TPU-0, the luminous flux rapidly decreases in the first 200 s and gets the lowest luminous flux value (7.5 %) at 480 s and increases gradually after 480 s. The luminous flux mainly decreases in the first 450 s, and it attains the lowest value (48.9 %) at 600 s in the smoke density test as APP is added into TPU. The luminous flux increases from 18.7 to 56.0 % at the end of the experiments. This indicates that smoke produced by TPU-1 is significantly less than that of TPU-0. Here, the addition of APP in TPU decomposes at low temperature to form polyphosphoric acid compounds which can catalyze the carbonization of TPU to generate condensed carbon shell on the surface of TPU-1 resulted the decrease of smoke production.
Compared with TPU-1 containing only APP and pure TPU, the luminous flux of the samples containing ferrous powder increased rapidly. In addition, the luminous flux increases along with the increase in ferrous powder content. For example, the luminous flux of TPU-5 is, respectively, up to 89.5 % at the end of experiments, which is much higher than that of TPU-1 (56.0 %). It is concluded that there may be an apparently synergistic effect of smoke suppression between ferrous powder and APP in TPU composites.
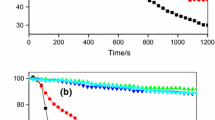
The luminous flux curves of TPU composites without flame in the smoke density test are presented in Fig. 7b. It can be seen from the Fig. 7b that there is a small grow of the luminous flux, and we can conclude that there is a small amount of smoke produced from TPU-0 at the first 300 s. But then, the luminous flux rapidly reduces and reaches 8.4 % at 1,200 s. Also, we can see that the luminous flux of the sample containing only APP (TPU-1) is much lower than that of the pure TPU (TPU-0) in the initial 720 s. The luminous flux of TPU-1 is higher than that of TPU-0 with the increase in time. The reason is that APP can decompose at low temperature to form some smoke precursors and char residue shell and that the smoke precursors lead to low luminous flux before 720 s. On the other hand, the luminous flux in TPU-1 is higher than that of TPU-0, which may be resulted from the generation rate of the smoke precursor decreases with the formation of char residue shell. It is interesting that the luminous flux is significantly improved compared with TPU-0 or TPU-1 when 0.625 mass% ferrous powder is incorporated into TPU/APP composite (TPU-2). And the maximum luminous flux value (61.2 %) is obtained when the content of ferrous powder is raised to 1.25 mass%. But then, the luminous flux decreases with the addition of ferrous powder. This implies that ferrous powder plays a negative role in the process of the formation of smoke precursors. And it is noted that a moderate amount of ferrous powder is favorable for improving the structure of char residue from TPU composites during the smoke density test without flame, so results low luminous flux.
The difference of testing condition in the smoke density tests with and without flame is whether to ignite. The tests with flame are carried out using flame of methane gas to ignite the sample under the radiant heat flux of 25 kW m−2. And the tests without flame are not ignited by any flame and just under the radiant heat flux of 25 kW m−2. Comparing Fig. 7b with 7a, it can be seen that there is much difference between the smoke density tests with flame and without flame. For example, the luminous flux of TPU-5 is 89.5 % in the test with flame, but it is only 32.5 % in the test without flame. This phenomenon illustrates that the smoke precursors formed in the thermal decomposition process and burn out in the smoke density test with flame.
Thermo-gravimetric (TG)
The key factor of improving the flame retardancy of TPU is to enhance the char-forming ability on the TPU surface. The APP/ferrous powder flame retardant system used in this paper has been, accordingly, designed for flame retarding TPU by considering the aspect mentioned above.
It is well known that thermo-gravimetry (TG) was developed by Professor Kotaro Honda (Tohoku Imperial University) in 1916 and that it is a high-precision method for the study of pyrolysis under well-defined conditions [31–34]. The TG also provides the identification of different surfactant environments and structure arrangements, and it can be also used for revealing of thermal and structural stabilities [35, 36]. Figure 8a, b shows the thermo-gravimetric (TG) and derivative thermo-gravimetric (DTG) curves of various flame retardant TPU composites in the temperature range of 25–700 °C under air atmosphere. For sample TPU-0, it can be seen that the base TPU begins to decompose rapidly at about 439.7 °C and this decomposition ends at about 700 °C with almost no char residue. Compared with TPU-0, the initial decomposition temperature of TPU-1 with only APP is 309.2 °C, which is lower than TPU-0. This can be attributed to the decomposition of APP at low temperature [37]. As usual, thermal degradation of TPU has two steps: The first stage is responsible for the rupture of the TPU main chains, whereas the second stage is attributed to the further destruction of the C–C and C–O bonds on the main chain [38]. But for the TPU-1 with only APP, the thermal degradation has three steps according to Fig. 8b. The three thermal degradation steps are assigned to ammonia release, catalysis of the charring process, and decomposition of the polyphosphoric acid chain, respectively. It can be clearly seen that the addition of APP reduces the formation temperature of carbon layer so that prevents TPU composites from further thermal degradation [39, 40].
For TPU-2 sample, the initial decomposition temperature is 287.5 °C, which is lower than that of TPU-1. The decrease in the decomposition temperature of the samples containing ferrous powder owe to that ferrous powder as a kind of active metal, which can catalyze APP decomposing. Furthermore, ferrous powder decreases the mass of flame retardant TPU composites at high temperature. It figured out that the samples with both APP and ferrous powder have four decomposition processes, but TPU-1 with only APP shows three. From Fig. 8b, it can obviously be seen that the decomposition rate of the samples with both APP and ferrous powder is larger than that of TPU-1 at the first stage. The phenomena may attribute to the catalyzing deamination by ferrous powder. For the samples TPU-2 to TPU-5 containing ferrous powder, the second decomposition stage is the reaction between polyphosphoric acid and ferrous powder. The third stage is responsible for the rupture of the TPU main chains. And the fourth stage is attributed to the further destruction of the C–C and C–O bonds on the main chain [38].
Photographs and scanning electron microscopy (SEM) of char residue
Figure 9 is the photographs and SEM of char residues for the TPU composites after cone calorimeter test. The heat transfer between flame zone and burning substrate can be prevented by the formation of efficient carbon layer, and thus protecting the underlying materials from further burning and pyrolysis of polymer composites. It can be seen that TPU-0 has the lightest and loosest char residue that is responding to the highest HRR and largest mass loss among all samples. Compared with TPU-0; the char residue of TPU-1 containing only APP is relatively high. What is more, a coherent and dense char can be formed only at a suitable ratio of ferrous powder into TPU/APP composites. It is apparent that the samples containing both APP and ferrous powder have high and regular char residues. For the TPU/APP/ferrous powder series, TPU-4 has the densest and most compact char residue surface among all samples, which can explain the related data tested by cone calorimeter.
In order to further evaluate the influence of ferrous powder on the structure of intumescent char determining flame retardant properties, SEM micrographs of intumescent chars of TPU-0 (TPU-0S), TPU-1 (TPU-1S), and TPU-5 (TPU-5S) magnified 200× are shown in Fig. 9, respectively. For TPU-0S sample, there are many holes and crevices, and the carbon layer presents an irregular appearance. But for TPU-1S composites with APP (Fig. 9), a very smooth fracture surface is observed. Compared with TPU-0S and TPU-1S, the char residue of TPU-4S shows a compact appearance and smooth surface clearly as well as clear interface. This can be explained that ferrous powder can help to promote charring and forming compact carbon layer, restraining the heat release and smoke generation.
Limiting oxygen index (LOI)
The LOI values testing results of the flame retardant TPU composites are presented in Fig. 10. It can be seen that the LOI value of the composite containing 20.00 mass% APP is as high as 31.25 compared with that of the pure TPU. Moreover, the values of the composites containing ferrous powder at the corresponding additive level are higher than the TPU/APP composite. And TPU-3 with 1.25 mass% ferrous powder shows the highest LOI value (32.75) among all samples. With ferrous powder further increasing, the LOI values of TPU/APP/ferrous powder composites decrease. For example, the LOI value of TPU-4 with 2.5 mass% ferrous powders is 31.75, but the LOI value of TPU-5 with 3.75 mass% ferrous powder increases slightly. The above results imply ferrous powder can improve the structure of char residue from TPU composites. Indeed, ferrous powder not only can react with polyphosphoric acid from the decomposition of APP to form iron pyrophosphate compounds which increases the melt viscosity of the composites, resulting high LOI value, but also can change the expansion degree and density of char residue which plays an important role in LOI. Moderate expansion degree and density may correspond to high LOI. The sample TPU-3 containing 1.25 mass% ferrous powders plays a vivid example. But it can be seen that the LOI values of TPU-4 and TPU-5 are lower than that of TPU-3 when the ferrous powder content is raised. This phenomenon contributed by that the density of char residue increases but the expansion degree of char residue decreases as the ferrous powder content is raised. As the LOI values of TPU-4 are lower than that of TPU-5, which may be due to that ferrous powder particles aggregated severely in the APP/TPU blend, suggesting poor dispersibility, it can be concluded that there is synergistic effect between APP and ferrous powder in improving LOI value of TPU composites.
Conclusions
As to all the results of TPU/APP/Fe composites tested by CCT, SDT, TG, SEM and LOI, the following conclusion was deduced. Firstly, the combination of the ferrous powder with APP intumescent system has the best synergistic effect in flameretardant of TPU, greatly enhancing the quality of the formed condensed phase charred layer and hence improving the flame retardancy. Secondly, ferrous powder can help to change the structure of char residue layer that restrain the heat release and smoke generation. Thirdly, ferrous powder represents dramatically excellent smoke suppression properties inflame retardant TPU composites based on APP.
References
Nie SB, Peng C, Yuan SJ, Zhang MX. Thermal and flame retardant properties of novel intumescent flame retardant polypropylene composites. J Therm Anal Calorim. 2013;113:865–71.
Hatsuhiko H, Yoshihisa T, Takahito I. Synergistic effect of red phosphorus, novella and melamine ternary combination on flame retardancy of poly (oxymethylene). Polym Degrad Stab. 2006;91:1996–2002.
Kongkhlang T, Kousaka Y, Umemura T, Nakaya D, Thuamthong W, Pattamamongkolchai Y, Chirachanchai S. Role of primary amine in polyoxymethylene (POM)/bentonite nanocomposite formation. Polymer. 2008;49:1676–84.
Archodoulaki VM, Lüftl S, Seidler S. Degradation behavior of polyoxymethylene: Influence of different stabilizer packages. J Appl Polym Sci. 2007;105:3679–88.
Zhang Q, Chen YH. Synergistic effects of ammonium polyphosphate/melamine intumescent system with macromolecular char former in flame-retarding polyoxymethylene. J Polym Res. 2011;18:293–303.
Carty P, White S. Relationship between char, flammability and smoke production in blends of chlorinated polyvinyl chloride, CPVC, and acrylonitrile–butadiene–styrene, ABS, containing a smoke suppressing iron compound. Polym Netw Blends. 1997;7:121–4.
Carty P, Whilte S. Anomalous flammability behavior of CPVC (chlorinated polyvinyl chloride) in blends with ABS (acrylonitrile–butadiene–styrene) containing flame-retarding/smoke-suppressing compounds. Polymer. 1997;38:1111–9.
Carty P, Metcalfe E, Annison WN. The optimization of the smoke suppressant and flame retardant properties of flexible PVC. J Appl Polym Sci. 1990;41:901–6.
Skinner GA, Haines P, Evans SJ. The effects of structure on the thermal degradation of polyester resins. Thermochim Acta. 1996;278:77–89.
Morgan AB, Gilman JW. An overview of flame retardancy of polymeric materials: application, technology, and future directions. Fire Mater. 2013;37:259–79.
Green DW, Dallavia AJ. Alumina trihydrate in flexible PVC-effects of alumina particle morphology. J Vinyl Addit Technol. 1988;10:178–82.
Gann RG, Babrauskas V, Peacock RD, Hall JR. Fire conditions for smoke toxicity measurement. Fire Mater. 1994;18:193–9.
Prager FH, Cabos HP. Fire–gas hazards in rail traffic. Fire Mater. 1994;18:131–49.
Chen XL, Jiang YF, Jiao CM. Smoke suppression properties of ferrite yellow on flame retardant thermoplastic polyurethane based on ammonium polyphosphate. J Hazard Mater. 2014;266:114–21.
Fang Y, Wang Q, Guo C, Song Y, Cooper PA. Effect of zinc borate and wood flour on thermal degradation and fire retardancy of polyvinyl chloride (PVC) composites. J Therm Anal Calorim. 2013;100:230–6.
Wang X, Hu Y, Song L, Xuan SY, Xing WY, Bai ZM, Lu HD. Flame retardancy and thermal degradation of intumescent flame retardant poly(lactic acid)/starch biocomposites. Ind Eng Chem Res. 2010;50:713–20.
Wu K, Hu Y, Song L, Lu HD, Wang ZZ. Flame retardancy and thermal degradation of intumescent flame retardant starch-based biodegradable composites. Ind Eng Chem Res. 2009;48:3150–7.
Chen XL, Jiao CM, Wang Y. Synergistic effects of iron powder on intumescent flame retardant polypropylene system. Express Polym Lett. 2009;3:359–65.
Tsai KC. Orientation effect on cone calorimeter test results to assess fire hazard of materials. J Hazard Mater. 2009;172:763–72.
Schartel B, Hull TR. Development of fire-retarded materials—interpretation of cone calorimeter data. Fire Mater. 2007;31:327–54.
Wang XY, Li Y, Liao WW, Gu J, Li D. A new intumescent flame-retardant: preparation, surface modification, and its application in polypropylene. Polym Adv Technol. 2008;19:1055–61.
Qian Y, Wei P, Zhao XM, Jiang PK, Yu HZ. Flame retardancy and thermal stability of polyhedral oligomeric silsesquioxane nanocomposites. Fire Mater. 2012;22:22037–43.
Chen XL, Jiao CM. Flammability and thermal degradation of epoxy acrylate modified with phosphorus-containing compounds. Polym Adv Technol. 2010;21:490–5.
Babrauskas V, Peacock RD. Heat release rate: the single most important variable in fire hazard. Fire Saf J. 1992;18:255–72.
Almeras X, Bras ML, Hornsby P, Bourbigot S, Marosi G, Keszei S, Poutch F. Effect of fillers on the fire retardancy of intumescent polypropylene compounds. Polym Degrad Stab. 2003;82:325–31.
Jiao CM, Chen XL. Flammability and thermal degradation of intumescent flame-retardant polypropylene composites. Polym Eng Sci. 2010;10:767–72.
Dong YY, Gui Z, Hu Y, Wu Y, Jiang SH. The influence of titanate nanotube on the improved thermal properties and the smoke suppression in poly(methyl methacrylate). J Hazard Mater. 2012;209:34–9.
Carty P, White S, Creghton JR. TG and flammability studies on polymer blends containing acrylonitrile–butadiene–styrene and chlorinated poly(vinyl chloride). J Therm Anal Calorim. 2001;63:679–87.
Carty P, White S. The effect of temperature on char formation in polymer blends: an explanation of the role of the smoke suppressant FeOOH acting in ABS/CPVC polymer blends. Polym Degrad Stab. 2002;75:173–84.
Ricciardi MR, Antonucci V, Zarrelli M, Giordano M. Fire behavior and smoke emission of phosphate-based inorganic fire-retarded polyester resin. Fire Mater. 2012;36:203–15.
Chen XL, Jiao CM, Zhang J. Thermal and combustion behavior of ethylene–vinyl acetate/aluminum trihydroxide/Fe-montmorillonite composites. Polym Eng Sci. 2012;10:414–9.
Chen XL, Hu Y, Jiao CM, Song L. Preparation and thermal properties of a novel flame-retardant coating. Polym Degrad Stab. 2007;92:1141–50.
Kimura T. Advanced topics of 15th international congress of thermal analysis and calorimetry. J Therm Anal Calorim. 2013;113:999–1002.
Li LL, Wang G, Wang SY, Qin S. Thermogravimetric and kinetic analysis of energy crop Jerusalem artichoke using the distributed activation energy model. J Therm Anal Calorim. 2013;114:1183–9.
Kubranová M, Jóna E, Rudinská E, Nemčeková K, Ondrušová D, Pajtášová M. Thermal properties of Co-, Ni- and Cu-exchanged montmorillonite with 3 hydroxypyridine. J Therm Anal Calorim. 2003;74:251–7.
Jona E, Sapietova M, Nircova S, Pajtasova M, Ondrusova D, Pavlık V, Lajdova L, Mojumdar SC. Characterization and thermal properties of Ni-exchanged montmorillonite with benzimidazole. J Therm Anal Calorim. 2008;94:69–73.
Zhao K, Xu W, Song L, Wang B, Feng H, Hu Y. Synergistic effects between boron phosphate and microencapsulated ammonium polyphosphate in flame-retardant thermoplastic polyurethane composites. Polym Adv Technol. 2012;23:894–900.
Lin M, Li B, Li Q, Li S, Zhang SQ. Synergistic effect of metal oxides on the flame retardancy and thermal degradation of novel intumescent flame-retardant thermoplastic polyurethanes. J Appl Polym Sci. 2011;121:1951–60.
Zhang Y, Chen X, Fang Z. Synergistic effects of expandable graphite and ammonium polyphosphate with a new carbon source derived from biomass in flame retardant ABS. J Appl Polym Sci. 2013;128:2424–32.
Fang G, Li H, Chen Z, Liu X. Preparation and characterization of flame retardant n-hexadecane/silicon dioxide composites as thermal energy storage materials. J Hazard Mater. 2010;181:1004–9.
Acknowledgements
The authors gratefully acknowledge the National Natural Science Foundation of China (Nos. 51106078, 51206084), the Out-standing Young Scientist Research Award Fund from Shandong Province (BS2011CL018), and the University Research and Development Projects Shandong Province (J14LA13).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiao, C., Zhao, X., Song, W. et al. Synergistic flame retardant and smoke suppression effects of ferrous powder with ammonium polyphosphate in thermoplastic polyurethane composites. J Therm Anal Calorim 120, 1173–1181 (2015). https://doi.org/10.1007/s10973-014-4377-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-014-4377-z