Abstract
The recent advance in hybrid imaging techniques enables offering simultaneous positron emission tomography (PET)/magnetic resonance imaging (MRI) in various clinical fields. 18F-fluorodeoxyglucose (FDG) PET has been widely used for diagnosis and evaluation of oncologic patients. The growing evidence from research and clinical experiences demonstrated that PET/MRI with FDG can provide comparable or superior diagnostic performance more than conventional radiological imaging such as computed tomography (CT), MRI or PET/CT in various cancers. Combined analysis using structural information and functional/molecular information of tumors can draw additional diagnostic information based on PET/MRI. Further studies including determination of the diagnostic efficacy, optimizing the examination protocol, and analysis of the hybrid imaging results is necessary for extending the FDG PET/MRI application in clinical oncology.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The new hybrid imaging modality, whole-body positron emission tomography (PET)/magnetic resonance imaging (MRI), has become more promising in oncologic and non-oncologic clinical fields. Comprehensive structural information from MRI and functional characteristics from PET can be obtained in a single session examination. PET scan using 18F-fluorodeoxyglucose (FDG) has been an established modality in initial diagnosis, planning treatments, and monitoring patients in various cancers.
Measurement of glucose metabolism by PET/MRI showed reproducible results and the PET image quality from PET/MRI is not deteriorated when comparing with that from PET/CT scans [1–4]. Furthermore, PET/MRI provide superior soft tissue contrast to PET/CT and its roles for accurate diagnosis and evaluating therapeutic response in oncologic patients has been expanded [5, 6].
In this review article, the current status of hybrid FDG PET/MR imaging in malignancies is discussed.
Materials and Methods
A systematic literature search was based on the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [7] (Fig. 1). The main research question was determined by the Eligible patients, Index test, Comparator/reference test, Target condition and Study design (PICTS) strategy [8, 9]. Literature searches using PubMed database were performed on October 10 and 29, 2015, by keyword “PET/MRI” and “PET/MR”, respectively. Exclusion criteria of original articles were followed: 1) review papers, letters to the editor, abstracts, small case series that includes less than five patients, 2) patients with non-malignant disease, 3) radiotracers other than FDG, 4) non-human studies, 5) articles that were written in languages other than English, and 6) duplicated publications [10]. Representative cases were originated from previous research approved by the Institutional Review Board of our hospital (1306-055-495).
The study-selection protocol in this review. PICTS strategy demonstrates the Eligible patients, Index test, Comparator/reference test, Target condition and Study design; FDG 18F-fluorodeoxyglucose, PET positron emission tomography, MRI magnetic resonance imaging, CT computed tomography, RCT randomized controlled trial
Retrospective Image Fusion of PET and MRI
Coregistration of PET and MRI from separated examinations has been studied for accurate detection of tumor lesions and additive diagnostic information provided by combination of anatomical and functional imaging. Earlier studies assessed spatial relationship between PET and MRI in brain imaging by development of spatial normalization technique. FDG PET enabled us to detect regional brain activation for pre-operative planning [11]. In this study, results of FDG PET scans after activation tasks of upper and lower extremities were compared with those of direct cortical stimulation by surgically placed electrodes. FDG PET could suggest a 95 % of diagnostic accuracy for detecting functional activation zone. FDG uptake of brain tumor is related with malignancy grade, and direct comparison of PET and MRI images provided additive diagnostic information such as tumor location, boundary, and heterogeneity [12–14].
Coregistration of PET and MRI in body part raises concern about quality of fused images in terms of global quality, alignment, and diagnostic confidence. Relatively good image quality can be achieved in head, thorax, and pelvic area [15]. In head and neck cancer, accurate T staging by fused PET/MRI was superior to that by PET/CT (87 % vs. 67 %) and N staging by PET/MRI was superior to that by MRI (77 % vs. 63 %) [16]. The level of diagnostic confidence was higher in fused PET/MRI than in PET/CT (85.9 % vs. 70.3 %) [17]. PET/MRI coregistration reading showed additional diagnostic information that altered the management plan in 46 % of the study population and no additional information in 18 % in patients with thyroid cancer [18]. The image fusion of PET and MRI improved anatomic correlation of PET and MRI finding, but little effect was observed in view of decision making in pancreas cancer (one of eight patients having additive information by image fusion) [19]. Another study reported that fused PET/MRI improved diagnostic accuracy for pancreas cancer compared with PET/CT (96.6 % vs. 86.6 %) and showed additional findings, especially for cystic lesions [20]. T1-weighted images as well as T2-weighted images on MRI (93.0 and 90.7 %) were more helpful for increasing diagnostic accuracy than PET/CT (88.4 %) [21]. In one study, the sensitivity of fused PET/MRI for detecting liver metastases of colorectal cancer was 98.3 %, which is higher than that of PET/CT (84.2 %) [22]. Improvement of sensitivity in PET/MRI was not more significant than MRI [23]. In uterine cervical cancer, fused PET/MRI and non-fused PET/MRI showed higher diagnostic accuracy of T-staging (83.3 %) than PET/CT (53.3 %). Difference of diagnostic accuracy for N-staging between fused PET/MRI (90.0 %), PET/CT (90.0 %), and MRI (86.7 %) showed no statistical difference [24]. In endometrial cancer, fused PET/MRI showed better sensitivity for detecting myometrial invasion than PET/CT (90.5 % vs. 76.2 %) and metastatic lymph nodes than MRI (100 % vs. 66.7 %) [25]. Fused PET/MRI improved lesion depiction, localization, and quality of diagnosis more than PET/CT in gynecologic malignancy [26]. MR signal intensity assessement on fused PET/MRI improved diagnostic accuracy for pre-operative staging more than PET/CT in lung cancer (97.1 % vs. 85.0 %, respectively) [27].
Image fusion of PET and MRI can enable us to localize metabolically active tumor focus exactly. One study reported that the histopathologic response to neoadjuvant chemotherapy was significantly correlated with high standardized uptake value (SUV) on residual tumor tissue in patients with osteosarcoma [28]. In addition to the maximum SUV, metabolic tumor volume on FDG PET aided by volume measure on MRI had a further predictive value of chemotherapy response in patients with osteosarcoma [29].
Diagnostic Performance of Hybrid PET/MR Imaging in Major Cancers
Head and Neck Cancer
Early PET/MRI study has been applied in diagnosis of Head and Neck (HN) cancer because of MRI information for accurate T staging and development of prototype hybrid PET/MRI scanner. PET scans acquired from PET/MRI scanner showed better image resolution in comparison to PET/CT scans, and results of FDG uptake in normal tissue and tumor were highly correlated between PET/MRI and PET/CT [30].
Precise diagnosis and staging of HN cancer is important for planning treatment modality such as radical surgery and/or combined chemoradiation therapy. PET/MR provided more accurate T staging of HN cancer than PET/CT and MRI (75, 59, 50 %, respectively), but could not achieve statistical significance. N staging results also showed no significant difference between these modalities [31]. One study reported that PET/MRI increased sensitivity for detecting cervical lymph node metastasis over MRI only (89.5 % vs. 65.8 %, respectively) but there was no significant difference with standalone PET (86.8 %) [32]. In another study, diagnostic performance of PET/MRI showed 80.5 % of sensitivity, 88.2 % of specificity, 75.6 % of positive predictive value (PPV), and 92.5 % of negative predictive value (NPV) and those of PET/CT showed 82.7, 87.3, 73.2, and 92.4 %, respectively [33].
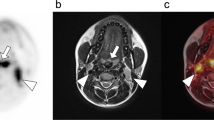
Detecting lymph nodes and/or distant metastases of HN cancer is comparable between PET/MRI and PET/CT (k value of interreader agreement of 0.85 and 0.70) [34]. Another study reported that combined PET/MRI using regional PET images increased sensitivity for a suspicious metastatic lymph node resulting in re-classification of stage in two of ten patients [35]. CT scans have limitation for evaluating oral cavity lesion when metallic and/or dental artifact exists. One study showed that most artifacts are noted in the suprahyoid region in CT and infrahyoid region in MRI [36]. PET/CT and PET/MRI would be considered as alternative diagnostic modality of HN cancer according to the location of primary tumor (Fig. 2).
(a) The maximal intensity projection (MIP) image of whole-body PET in patient with right tonsil cancer. The post-contrast computed tomography (CT) (b) and T1-weighted MR image (c) showed moderately enhanced cervical lymph nodes in bilateral neck. On the contrary, PET image (d) and fusion image (e) showed focal hypermetabolism in right cervical lymph node (red arrow). This lymph node was pathologically proven as a metastatic node
Advanced MRI techniques can provide multiple parameters of tumor such as perfusion index (K trans, k ep) and cell density (apparent diffusion coefficient (ADC), Ve). One study reported a significant correlation between the region of interest (ROI) area, SUV, and metabolic parameters such as metabolic tumor volume (MTV) measured from PET/CT and PET/MRI. ADC value showed significant negative correlation with both of K trans and SUV [37]. However, another study showed that adding diffusion weighted imaging (DWI) sequence in PET/MRI examination did not improve diagnostic accuracy more than PET/MRI without DWI (84.0 % vs. 86.7 %, respectively) [38].
FDG PET/CT has been a useful modality for evaluating histopathologic differentiation and monitoring of recurrence in patients with differentiated thyroid cancer (DTC) [39]. In DTC, PET/MRI showed equal performance of detecting local relapse, lymph node, and bone metastases. However, PET/CT was superior to PET/MRI to detect pulmonary metastatic lesions [40].
Breast Cancer
MRI has shown high diagnostic accuracy for primary breast cancer lesions. PET/CT has established modality for the detection of lymph node and/or distant metastasis of breast cancer. Hence, simultaneous PET/MRI has been evaluated for the improvement of diagnostic performance in breast cancer.
One study reported that PET/MRI and MRI provided identical performance for correct T-staging (82 %) and were superior to PET/CT (68 %). For the N-staging, PET/MRI showed similar results of diagnostic accuracy with PET/CT (86 % vs. 88 %) and tended to be higher than MRI (80 %) [41]. Another study showed that PET/MRI provided lower sensitivity for primary tumor than MRI (77 % vs. 100 %), but higher specificity (100 % vs. 67 %). For the N-staging, difference between PET/MRI and MRI was not statistically significant [42].
PET/MRI showed superior sensitivity to PET/CT for detecting bone metastasis of breast cancer (0.96 vs. 0.85) [43]. PET/MRI mammography using dedicated radiofrequency coil for breast improved detection sensitivity of breast cancer lesions more than PET alone or whole-body PET/MRI images (100, 79.2, 87.5 %, respectively) [44].
Cancer in Gastrointestinal Tract and Abdomen
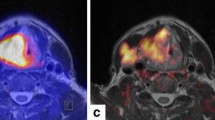
The recent technical advances of PET/MRI modality allow improved diagnostic accuracy of malignancies in intraabdominal organs. Advanced MRI protocols including dynamic contrast scans and diffusion weighted imaging provided could provide higher diagnostic confidence of liver metastasis, and additive information for FDG non-avid lesions [45]. Sensitivity and NPV of PET/MRI has been reported to be 92.2 and 95.1 %, higher than those of PET/CT (sensitivity 67.8 % and NPV 82.2 %, respectively). Non-enhanced fast T1 weighted MR imaging showed superiority to low-dose CT in terms of conspicuity rating in liver lesions [36]. Benign lesions of liver as well as metastatic lesions were better characterized by PET/MRI than PET/CT [46] (Fig. 3).
This patient with treated breast cancer underwent PET/MRI examination for evaluation of hepatic metastases (a MIP image of PET). (b) Delayed-phase (10 min) of post-contrast MR image revealed additional hepatic metastasis lesion (white arrow) without hypermetabolism (c PET). Furthermore, whole-body PET/MR imaging enabled us to detect asymptomatic brain metastasis (red arrow) (d apparent diffusion coefficient image; e PET)
PET/MRI showed detailed information for initial staging of colorectal cancer. Accurate identification of disease involvement in mesorectal fat and/or fascia allow an accurate plan for patient management. PET/MRI also provided comparable diagnostic accuracy for N and M staging with PET/CT (true positive findings in 86 % vs. 71 %, respectively) [47]. In initial diagnosis and clinical follow-up, metastasis of colorectal cancer occurs in lymph nodes, peritoneum, and distant organ. Comprehensive PET/MRI study improved diagnostic accuracy of liver metastasis more than PET/CT (0.74 vs. 0.56) and peritoneal seeding more than MRI (0.55 vs. 0.37) [48]. In this study, diagnostic accuracy of mucinous cancer decreased when comparing non-mucinous cancer even with PET/MRI (0.52 vs. 0.76). Further investigation will be needed to improve diagnostic accuracy of mucinous tumors.
Incidentalomas from cysts to malignancy in abdominal organs are frequently detected in whole-body and/or abdominal imaging. PET/MRI can detect a greater number of incidental findings than PET/CT (635 vs. 407), and reduce the indeterminate diagnostic finding [49].
Gynecologic Cancer and Pelvic Malignancy
High soft tissue contrast information from MRI provides greater improvement of primary tumor delineation by PET/MRI than PET/CT in terms of tissue invasion, relation to surrounding structures, and characterization [50]. In patients with suspected recurrence of cervical or ovarian cancer, detection rate of malignant lesion was higher in simultaneous PET/MR imaging than MRI alone (98.9 % vs. 88.8 %) and diagnostic confidence increased in PET/MRI more than MRI or PET/CT [51, 52]. MR DWI and ADC imaging show increases in tissue cellularity and improve the diagnostic confidence in differentiating malignancy from benign [53]. However, recent study showed that DWI had no additional information for diagnosis of pelvic malignancy [54]. Optimized PET/MRI study for whole-body MRI can be applied to oncologic patients for accurate diagnostic information and reduced examination time [55, 56].
FDG PET/CT has limitation for evaluation of bladder cancer because of urinary FDG activity and possible misregistration of PET and CT images due to time-delay between image acquisitions. Simultaneous PET/MRI acquisition reduced misregistration of bladder wall, mass, and pelvic lymph nodes resulting in improvement of diagnostic confidence [57].
Lung Cancer
Accurate diagnosis and staging of non-small cell lung cancer (NSCLC) is needed to decide treatment modality such as surgery and/or concurrent chemoradiation therapy. FDG PET/CT showed high diagnostic accuracy in NSCLC although limited specificity in false-positive lymph node. PET/MRI showed high diagnostic accuracy for lymph node metastasis of NSCLC when comparing with PET/CT (concordance rate of 91 %) [58]. Another study reported that diagnostic performance of PET/CT for N staging is a sensitivity of 0.75, specificity of 0.79, PPV of 0.75, NPV of 0.79, and 0.70, 0.75, 0.71, 0.73 for PET/MRI, respectively [59]. Diagnostic accuracy for intial staging was not significantly different in PET/MR and PET/CT [60–62].
The size of the pulmonary nodule is a significant factor for evaluating the diagnostic performance of MRI [63]. The detection rates for small nodules less than 1 cm in diameter on MR images were only 27.3 and 45.5 % of diagnostic CT images when using Dixon volume interpolated breath hold examination (VIBE) sequence, respectively [64]. Non-FDG avid small lung nodules less than 1 cm are usually only detected on CT scan of the PET/CT. Low signal-to-noise ratio for detecting small nodules in aerated lung is a limitation of PET/MR application in lung cancer. However, one study reported that the 96.4 % (81/84) of small non-FDG avid lung nodules missed on PET/MRI showed benign clinical courses such as resolved or stable status [65]. To overcome the size effect, a free-breathing ultrashort echo time (UTE) sequence has been evaluated in patients having small metastatic lung nodules. UTE sequence showed a higher detection rate for lung nodules with small size, non-FDG avid, not-subpleural location (p < 0.001) [66]. On the other hand, one study reported that post-contrast VIBE sequence images of MRI could not improve detection rate of pulmonary nodules more than non-contrast VIBE images (jackknife alternative free-response receiver-operating-characteristics value of 0.848 vs. 0.837, respectively) [67].
Others
Diagnosis of bone metastasis in oncologic patients has been crucial for treatment planning and predicting prognosis. Nevertheless, systemic development of bone metastasis is hard to detect by regional anatomic imaging. Whole-body PET/MR showed better detection rate of malignant bone lesions than PET/CT (100 % vs. 94 %), whereas diagnostic confidence of benign bone lesions was higher in PET/CT [68]. MR T1-weighted images in PET/MRI provided more conspicuous results for delineating and differentiating bone lesions than CT [69, 70]. Chemotherapy-related change of metabolic activity and fat density in proximal bone marrow can be evaluated by PET and MRI [71].
PET/MR imaging has been regarded as a promising alternative modality in pediatric oncologic patients due to reduced radiation exposure. The effective dose from PET/MR examination was about 20∼50 % that of PET/CT examinations [72–74]. Despite the SUV decrease in bone marrow in PET with MR-based attenuation correction method, lesion detection performance of PET/MR showed comparable to that of PET/CT (61 focal uptakes among 62 focal uptakes on PET/CT) [73]. Single PET/MR examination could result in a potential change of management plan in five of nine patients based on superior soft tissue contrast of MRI than that of CT [74].
The clinical usefulness of MR or PET/MR has not been clear for diagnosis and staging of lymphoma. One study reported that the sensitivity and specificity of PET/MR were 93.8 and 99.4 % based on the standard of reference [75]. Simultaneous PET/MR imaging identified nodal involvement of lymphoma with higher sensitivity than that of DWI (100 % vs. 62.7 %) and correctly diagnosed bone marrow involvement which was missed by PET/CT scan [76].
Cardiac MR imaging has been essential in the diagnosis of cardiac disease including tumors. Hybrid PET/MR imaging provides the diagnostic cut-off value by FDG PET and morphologic characterization of tissue by MRI, which yields 100 % of sensitivity and 100 % of specificity for differentiating malignancy from benign lesions [77]. PET/MR can provide acceptable diagnostic accuracy for T staging (66.7 %) and better accuracy for N staging (83.3 %) than endoscopic ultrasonography (EUS) or PET/CT in esophageal cancer [78].
Consideration for Study Protocol
In recent technical advances of MRI acquisition, PET/MRI examination protocol will be optimized to the patient’s indication with reducing the examination time (Tables 1 and 2). Earlier study reported that simultaneous acquisition of PET and MRI could be adopted with high image quality and short acquisition time of less than 20 min including whole-body coronal 2-point Dixon 3-dimensional T1 VIBE sequence which provided T1 in-phase, out-phase images automatically [79].
The advantage of MRI over CT is the easy capability of acquiring specific image sequences for each purpose. Simple PET/MRI examination based on only whole-body T1 weighted images for achieving attenuation correction map could not improve diagnostic accuracy more than PET/CT [80]. MRI sequences for evaluating tumor characteristics have been developed. DWI and ADC measurement has been widely used for diagnosis of malignant lesion. T1 and T2 weighted imaged can be acquired with short TR and TE using advanced techniques such as VIBE sequences and half Fourier acquired single shot turbo spin echo (HASTE) [81]. High resolution contrast-enhanced T1 is also available based on this protocol. Turbo inversion recovery magnitude (TIRM) sequence is used for detecting bone metastasis. Composition of these sequences could omit the non-enhanced T1 images without detrimental effect of diagnostic performance [82].
Previously used MRI sequences have limitations for whole-body imaging which required fast scan time and high image quality. For example, DWI based on a single-shot echo planar imaging (EPI) sequence often presented image distortion, which caused reduced quality of fusion images. Development of fusion-compatible MRI sequences will be helpful for increasing diagnostic accuracy [83–85].
Although results were highly correlated within PET/CT and PET/MR, SUV from PET/MR can be different from PET/CT. Careful interpretation is needed for direct comparison of quantitative PET/MR and PET/CT results [86, 87].
MRI techniques have been continuously developed and indications of MRI have been growing in the clinical oncology field, but CT also has its own advantages over MRI in several aspects such as easy accessibility, high-resolution image, and lung parenchyma evaluation. The role of PET/CT and PET/MRI might also be rather complementary than competitive for tumor diagnosis. The exact positioning of PET/MRI to reinforce the metabolic characterization of tumors where PET/CT is less useful has to be further investigated.
Complementary Role of PET/MR Imaging
PET/MRI enables high-quality fusion imaging and easy assessment of whole-body disease status, and the complementary role for PET/MR has been evaluated in major cancers (Table 3). Simultaneous acquisition of PET and MRI can provide more accurate spatial registration between two scans than sequential PET/CT acquisition. Improvement of spatial coregistration could be helpful for diagnosis and delineating treatment target tissue [88]. FDG uptake in a suspicious lesion can be further characterized by adding morphologic and functional information from MRI [89]. One study reported that additional findings revealed on PET/MRI affected clinical management in 17.9 % of patients due to re-evaluation of disease status and detecting metastases [90].
Hybrid PET/MRI enable to measure glucose metabolism and cellular density in tumor by simultaneous FDG, PET, and DWI acquisition. In malignant lesions, inverse correlation was found between SUV and ADC value [91, 92]. Volumetric measurement of tumor is highly correlated when comparing the results from PET images and MRI images (both of T2 weighted images and DWI) [93]. Diagnostic confidence of tumor can be increased by using the complementary information in PET/MRI. Addition of DWI information to PET/MRI increased lesion-based diagnostic accuracy more than PET/CT or PET/MR (0.69 vs. 0.66 and 0.57, respectively) [48]. In peritoneal calcinomatosis, combined information for ADC and SUV could enable us to differentiate histopathology or primary malignancy. Metastatic lesions from colorectal cancer showed relative higher ADC values and lower FDG uptakes than those from ovarian cancer [94].
Volumetric parameters such as MTV or total lesion glycolysis (TLG) driven by PET has been known to be useful for measuring tumor burden [95]. Changes of PET volumetric parameters can predict a more accurate response to chemotherapy than MRI volumetric measurements [96].
PET/MRI enabled us to provide functional parameters by combining tumor metabolism/volumetric parameters from PET and cellularity information from MRI. Glucose metabolism of tumor and/or ADC-corrected glucose metabolism showed linear correlation with tumor staging [97]. Further studies using new functional parameters from PET/MRI will be necessary for detailed characterization of tumor biology.
The role of PET and MRI scans has been increased in management of oncologic patients and performing both examinations could be cost-effective. Hybrid PET/MR imaging provides both the functional and structural information in a single examination, which could be convenient for the patient [98].
Conclusion
Hybrid FDG PET/MRI has become more widely used to overcome the limitation of conventional PET/CT in the oncologic field. PET/MRI showed comparable diagnostic performance and superiority to PET/CT in various malignancies in terms of excellent soft tissue contrast and flexible application of MRI sequences. Replacement of CT by MRI reduced the patient’s radiation dose, which becomes a more important issue in pediatric imaging and expansion of medical diagnostic imaging. With developing new hybrid imaging technique, the exact diagnostic indication and optimizing protocol of FDG PET/MRI in patient management has to be further investigated.
References
Rasmussen JH, Fischer BM, Aznar MC, Hansen AE, Vogelius IR, Lofgren J, et al. Reproducibility of (18)F-FDG PET uptake measurements in head and neck squamous cell carcinoma on both PET/CT and PET/MR. Br J Radiol. 2015;88:20140655.
Wiesmuller M, Quick HH, Navalpakkam B, Lell MM, Uder M, Ritt P, et al. Comparison of lesion detection and quantitation of tracer uptake between PET from a simultaneously acquiring whole-body PET/MR hybrid scanner and PET from PET/CT. Eur J Nucl Med Mol Imaging. 2013;40:12–21.
Quick HH, von Gall C, Zeilinger M, Wiesmuller M, Braun H, Ziegler S, et al. Integrated whole-body PET/MR hybrid imaging: clinical experience. Invest Radiol. 2013;48:280–9.
Pace L, Nicolai E, Luongo A, Aiello M, Catalano OA, Soricelli A, et al. Comparison of whole-body PET/CT and PET/MRI in breast cancer patients: lesion detection and quantitation of 18F-deoxyglucose uptake in lesions and in normal organ tissues. Eur J Radiol. 2014;83:289–96.
Schuler MK, Platzek I, Beuthien-Baumann B, Fenchel M, Ehninger G, van den Hoff J. (18)F-FDG PET/MRI for therapy response assessment in sarcoma: comparison of PET and MR imaging results. Clin Imaging. 2015;39:866–70.
Tian J, Fu L, Yin D, Zhang J, Chen Y, An N, et al. Does the novel integrated PET/MRI offer the same diagnostic performance as PET/CT for oncological indications? PLoS One. 2014;9:e90844.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151:264–9.
Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM, et al. Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD Initiative. Ann Intern Med. 2003;138:40–4.
Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM, et al. The STARD statement for reporting studies of diagnostic accuracy: explanation and elaboration. Ann Intern Med. 2003;138:W1–12.
von Elm E, Poglia G, Walder B, Tramer MR. Different patterns of duplicate publication: an analysis of articles used in systematic reviews. JAMA. 2004;291:974–80.
Schreckenberger M, Spetzger U, Sabri O, Meyer PT, Zeggel T, Zimny M, et al. Localisation of motor areas in brain tumour patients: a comparison of preoperative [18F]FDG-PET and intraoperative cortical electrostimulation. Eur J Nucl Med. 2001;28:1394–403.
Borgwardt L, Hojgaard L, Carstensen H, Laursen H, Nowak M, Thomsen C, et al. Increased fluorine-18 2-fluoro-2-deoxy-D-glucose (FDG) uptake in childhood CNS tumors is correlated with malignancy grade: a study with FDG positron emission tomography/magnetic resonance imaging coregistration and image fusion. J Clin Oncol. 2005;23:3030–7.
Estrada G, Gonzalez-Maya L, Celis-Lopez MA, Gavito J, Larraga-Gutierrez JM, Salgado P, et al. Diagnostic approach in suspected recurrent primary brain tumors using (18)FDG-PET/MRI, perfusion MRI, visual and quantitative analysis, and three dimensional stereotactic surface projections. First experience in Mexico. Rev Esp Med Nucl. 2008;27:329–39.
Kim JS, Cheon GJ, Lim SM. Presurgical mapping of brain tumors using statistical probabilistic anatomical maps. J Biomed Sci Eng. 2015;08:653–8.
Antonica F, Asabella AN, Ferrari C, Rubini D, Notaristefano A, Nicoletti A, et al. Useful diagnostic biometabolic data obtained by PET/CT and MR fusion imaging using open source software. Hell J Nucl Med. 2014;17 Suppl 1:50–5.
Kanda T, Kitajima K, Suenaga Y, Konishi J, Sasaki R, Morimoto K, et al. Value of retrospective image fusion of (1)(8)F-FDG PET and MRI for preoperative staging of head and neck cancer: comparison with PET/CT and contrast-enhanced neck MRI. Eur J Radiol. 2013;82:2005–10.
Huang SH, Chien CY, Lin WC, Fang FM, Wang PW, Lui CC, et al. A comparative study of fused FDG PET/MRI, PET/CT, MRI, and CT imaging for assessing surrounding tissue invasion of advanced buccal squamous cell carcinoma. Clin Nucl Med. 2011;36:518–25.
Seiboth L, Van Nostrand D, Wartofsky L, Ousman Y, Jonklaas J, Butler C, et al. Utility of PET/neck MRI digital fusion images in the management of recurrent or persistent thyroid cancer. Thyroid. 2008;18:103–11.
Ruf J, Lopez Hanninen E, Bohmig M, Koch I, Denecke T, Plotkin M, et al. Impact of FDG-PET/MRI image fusion on the detection of pancreatic cancer. Pancreatology. 2006;6:512–9.
Nagamachi S, Nishii R, Wakamatsu H, Mizutani Y, Kiyohara S, Fujita S, et al. The usefulness of (18)F-FDG PET/MRI fusion image in diagnosing pancreatic tumor: comparison with (18)F-FDG PET/CT. Ann Nucl Med. 2013;27:554–63.
Tatsumi M, Isohashi K, Onishi H, Hori M, Kim T, Higuchi I, et al. 18F-FDG PET/MRI fusion in characterizing pancreatic tumors: comparison to PET/CT. Int J Clin Oncol. 2011;16:408–15.
Yong TW, Yuan ZZ, Jun Z, Lin Z, He WZ, Juanqi Z. Sensitivity of PET/MR images in liver metastases from colorectal carcinoma. Hell J Nucl Med. 2011;14:264–8.
Donati OF, Hany TF, Reiner CS, von Schulthess GK, Marincek B, Seifert B, et al. Value of retrospective fusion of PET and MR images in detection of hepatic metastases: comparison with 18F-FDG PET/CT and Gd-EOB-DTPA-enhanced MRI. J Nucl Med. 2010;51:692–9.
Kitajima K, Suenaga Y, Ueno Y, Kanda T, Maeda T, Deguchi M, et al. Fusion of PET and MRI for staging of uterine cervical cancer: comparison with contrast-enhanced (18)F-FDG PET/CT and pelvic MRI. Clin Imaging. 2014;38:464–9.
Kitajima K, Suenaga Y, Ueno Y, Kanda T, Maeda T, Takahashi S, et al. Value of fusion of PET and MRI for staging of endometrial cancer: comparison with (18)F-FDG contrast-enhanced PET/CT and dynamic contrast-enhanced pelvic MRI. Eur J Radiol. 2013;82:1672–6.
Nakajo K, Tatsumi M, Inoue A, Isohashi K, Higuchi I, Kato H, et al. Diagnostic performance of fluorodeoxyglucose positron emission tomography/magnetic resonance imaging fusion images of gynecological malignant tumors: comparison with positron emission tomography/computed tomography. Jpn J Radiol. 2010;28:95–100.
Ohno Y, Koyama H, Yoshikawa T, Takenaka D, Seki S, Yui M, et al. Three-way comparison of whole-body MR, coregistered whole-body FDG PET/MR, and integrated whole-body FDG PET/CT imaging: TNM and stage assessment capability for non-small cell lung cancer patients. Radiology. 2015;275:849–61.
Kong CB, Byun BH, Lim I, Choi CW, Lim SM, Song WS, et al. (1)(8)F-FDG PET SUVmax as an indicator of histopathologic response after neoadjuvant chemotherapy in extremity osteosarcoma. Eur J Nucl Med Mol Imaging. 2013;40:728–36.
Cheon GJ, Kim MS, Lee JA, Lee SY, Cho WH, Song WS, et al. Prediction model of chemotherapy response in osteosarcoma by 18F-FDG PET and MRI. J Nucl Med. 2009;50:1435–40.
Boss A, Stegger L, Bisdas S, Kolb A, Schwenzer N, Pfister M, et al. Feasibility of simultaneous PET/MR imaging in the head and upper neck area. Eur Radiol. 2011;21:1439–46.
Schaarschmidt BM, Heusch P, Buchbender C, Ruhlmann M, Bergmann C, Ruhlmann V, et al. Locoregional tumour evaluation of squamous cell carcinoma in the head and neck area: a comparison between MRI, PET/CT and integrated PET/MRI. Eur J Nucl Med Mol Imaging. 2016;43:92–102.
Platzek I, Beuthien-Baumann B, Schneider M, Gudziol V, Kitzler HH, Maus J, et al. FDG PET/MR for lymph node staging in head and neck cancer. Eur J Radiol. 2014;83:1163–8.
Kubiessa K, Purz S, Gawlitza M, Kuhn A, Fuchs J, Steinhoff KG, et al. Initial clinical results of simultaneous 18F-FDG PET/MRI in comparison to 18F-FDG PET/CT in patients with head and neck cancer. Eur J Nucl Med Mol Imaging. 2014;41:639–48.
Partovi S, Kohan A, Vercher-Conejero JL, Rubbert C, Margevicius S, Schluchter MD, et al. Qualitative and quantitative performance of (18)F-FDG-PET/MRI versus (18)F-FDG-PET/CT in patients with head and neck cancer. AJNR Am J Neuroradiol. 2014;35:1970–5.
Lee SJ, Seo HJ, Cheon GJ, Kim JH, Kim EE, Kang KW, et al. Usefulness of integrated PET/MRI in head and neck cancer: a preliminary study. Nucl Med Mol Imaging. 2014;48:98–105.
Kuhn FP, Hullner M, Mader CE, Kastrinidis N, Huber GF, von Schulthess GK, et al. Contrast-enhanced PET/MR imaging versus contrast-enhanced PET/CT in head and neck cancer: how much MR information is needed? J Nucl Med. 2014;55:551–8.
Covello M, Cavaliere C, Aiello M, Cianelli MS, Mesolella M, Iorio B, et al. Simultaneous PET/MR head-neck cancer imaging: preliminary clinical experience and multiparametric evaluation. Eur J Radiol. 2015;84:1269–76.
Queiroz MA, Hullner M, Kuhn F, Huber G, Meerwein C, Kollias S, et al. Use of diffusion-weighted imaging (DWI) in PET/MRI for head and neck cancer evaluation. Eur J Nucl Med Mol Imaging. 2014;41:2212–21.
Haugen BRM, Alexander EK, Bible KC, Doherty G, Mandel SJ, Nikiforov YE, et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26:1–133.
Vrachimis A, Burg MC, Wenning C, Allkemper T, Weckesser M, Schafers M, et al. [(18)F]FDG PET/CT outperforms [(18)F]FDG PET/MRI in differentiated thyroid cancer. Eur J Nucl Med Mol Imaging. 2016;43:212–20.
Grueneisen J, Nagarajah J, Buchbender C, Hoffmann O, Schaarschmidt BM, Poeppel T, et al. Positron emission tomography/magnetic resonance imaging for local tumor staging in patients with primary breast cancer: a comparison with positron emission tomography/computed tomography and magnetic resonance imaging. Invest Radiol. 2015;50:505–13.
Botsikas D, Kalovidouri A, Becker M, Copercini M, Djema DA, Bodmer A, et al. Clinical utility of 18F-FDG-PET/MR for preoperative breast cancer staging. Eur Radiol. 2015. doi:10.1007/s00330-015-4054-z.
Catalano OA, Nicolai E, Rosen BR, Luongo A, Catalano M, Iannace C, et al. Comparison of CE-FDG-PET/CT with CE-FDG-PET/MR in the evaluation of osseous metastases in breast cancer patients. Br J Cancer. 2015;112:1452–60.
Kong EJ, Chun KA, Bom HS, Lee J, Lee SJ, Cho IH. Initial experience of integrated PET/MR mammography in patients with invasive ductal carcinoma. Hell J Nucl Med. 2014;17:171–6.
Beiderwellen K, Geraldo L, Ruhlmann V, Heusch P, Gomez B, Nensa F, et al. Accuracy of [18F]FDG PET/MRI for the detection of liver metastases. PLoS One. 2015;10:e0137285.
Beiderwellen K, Gomez B, Buchbender C, Hartung V, Poeppel TD, Nensa F, et al. Depiction and characterization of liver lesions in whole-body [(18)F]-FDG PET/MRI. Eur J Radiol. 2013;82:e669–75.
Paspulati RM, Partovi S, Herrmann KA, Krishnamurthi S, Delaney CP, Nguyen NC. Comparison of hybrid FDG PET/MRI compared with PET/CT in colorectal cancer staging and restaging: a pilot study. Abdom Imaging. 2015;40:1415–25.
Brendle C, Schwenzer NF, Rempp H, Schmidt H, Pfannenberg C, la Fougere C, et al. Assessment of metastatic colorectal cancer with hybrid imaging: comparison of reading performance using different combinations of anatomical and functional imaging techniques in PET/MRI and PET/CT in a short case series. Eur J Nucl Med Mol Imaging. 2016;43:123–32.
Schaarschmidt BM, Grueneisen J, Heusch P, Gomez B, Umutlu L, Ruhlmann V, et al. Does 18F-FDG PET/MRI reduce the number of indeterminate abdominal incidentalomas compared with 18F-FDG PET/CT? Nucl Med Commun. 2015;36:588–95.
Queiroz MA, Kubik-Huch RA, Hauser N, Freiwald-Chilla B, von Schulthess G, Froehlich JM, et al. PET/MRI and PET/CT in advanced gynaecological tumours: initial experience and comparison. Eur Radiol. 2015;25:2222–30.
Grueneisen J, Beiderwellen K, Heusch P, Gratz M, Schulze-Hagen A, Heubner M, et al. Simultaneous positron emission tomography/magnetic resonance imaging for whole-body staging in patients with recurrent gynecological malignancies of the pelvis: a comparison to whole-body magnetic resonance imaging alone. Invest Radiol. 2014;49:808–15.
Beiderwellen K, Grueneisen J, Ruhlmann V, Buderath P, Aktas B, Heusch P, et al. [(18)F]FDG PET/MRI vs. PET/CT for whole-body staging in patients with recurrent malignancies of the female pelvis: initial results. Eur J Nucl Med Mol Imaging. 2015;42:56–65.
Kuang F, Ren J, Zhong Q, Liyuan F, Huan Y, Chen Z. The value of apparent diffusion coefficient in the assessment of cervical cancer. Eur Radiol. 2013;23:1050–8.
Grueneisen J, Schaarschmidt BM, Beiderwellen K, Schulze-Hagen A, Heubner M, Kinner S, et al. Diagnostic value of diffusion-weighted imaging in simultaneous 18F-FDG PET/MR imaging for whole-body staging of women with pelvic malignancies. J Nucl Med. 2014;55:1930–5.
Grueneisen J, Schaarschmidt BM, Heubner M, Suntharalingam S, Milk I, Kinner S, et al. Implementation of FAST-PET/MRI for whole-body staging of female patients with recurrent pelvic malignancies: a comparison to PET/CT. Eur J Radiol. 2015;84:2097–102.
Buchbender C, Hartung-Knemeyer V, Beiderwellen K, Heusch P, Kuhl H, Lauenstein TC, et al. Diffusion-weighted imaging as part of hybrid PET/MRI protocols for whole-body cancer staging: does it benefit lesion detection? Eur J Radiol. 2013;82:877–82.
Rosenkrantz AB, Balar AV, Huang WC, Jackson K, Friedman KP. Comparison of coregistration accuracy of pelvic structures between sequential and simultaneous imaging during hybrid PET/MRI in patients with bladder cancer. Clin Nucl Med. 2015;40:637–41.
Heusch P, Buchbender C, Kohler J, Nensa F, Gauler T, Gomez B, et al. Thoracic staging in lung cancer: prospective comparison of 18F-FDG PET/MR imaging and 18F-FDG PET/CT. J Nucl Med. 2014;55:373–8.
Kohan AA, Kolthammer JA, Vercher-Conejero JL, Rubbert C, Partovi S, Jones R, et al. N staging of lung cancer patients with PET/MRI using a three-segment model attenuation correction algorithm: initial experience. Eur Radiol. 2013;23:3161–9.
Huellner MW, Barbosa FG, Husmann L, Pietsch CM, Mader CE, Burger IA, et al. TNM staging of NSCLC: comparison of PET/MR and PET/CT. J Nucl Med. 2016;57:21–6.
Stolzmann P, Veit-Haibach P, Chuck N, Rossi C, Frauenfelder T, Alkadhi H, et al. Detection rate, location, and size of pulmonary nodules in trimodality PET/CT-MR: comparison of low-dose CT and Dixon-based MR imaging. Investig Radiol. 2013;48:241–6.
Fraioli F, Screaton NJ, Janes SM, Win T, Menezes L, Kayani I, et al. Non-small-cell lung cancer resectability: diagnostic value of PET/MR. Eur J Nucl Med Mol Imaging. 2015;42:49–55.
Yoon SH, Goo JM, Lee SM, Park CM, Cheon GJ. PET/MR imaging for chest diseases: review of initial studies on pulmonary nodules and lung cancers. Magn Reson Imaging Clin N Am. 2015;23:245–59.
Rauscher I, Eiber M, Furst S, Souvatzoglou M, Nekolla SG, Ziegler SI, et al. PET/MR imaging in the detection and characterization of pulmonary lesions: technical and diagnostic evaluation in comparison to PET/CT. J Nucl Med. 2014;55:724–9.
Raad RA, Friedman KP, Heacock L, Ponzo F, Melsaether A, Chandarana H. Outcome of small lung nodules missed on hybrid PET/MRI in patients with primary malignancy. J Magn Reson Imaging. 2015. doi:10.1002/jmri.25005.
Burris NS, Johnson KM, Larson PE, Hope MD, Nagle SK, Behr SC, et al. Detection of small pulmonary nodules with ultrashort echo time sequences in oncology patients by using a PET/MR system. Radiology. 2016;278:239–46.
Lee KH, Park CM, Lee SM, Lee JM, Cho JY, Paeng JC, et al. Pulmonary nodule detection in patients with a primary malignancy using hybrid PET/MRI: is there value in adding contrast-enhanced mr imaging? PLoS One. 2015;10:e0129660.
Beiderwellen K, Huebner M, Heusch P, Grueneisen J, Ruhlmann V, Nensa F, et al. Whole-body [(18)F]FDG PET/MRI vs. PET/CT in the assessment of bone lesions in oncological patients: initial results. Eur Radiol. 2014;24:2023–30.
Eiber M, Takei T, Souvatzoglou M, Mayerhoefer ME, Furst S, Gaertner FC, et al. Performance of whole-body integrated 18F-FDG PET/MR in comparison to PET/CT for evaluation of malignant bone lesions. J Nucl Med. 2014;55:191–7.
Samarin A, Hullner M, Queiroz MA, Stolzmann P, Burger IA, von Schulthess G, et al. 18F-FDG-PET/MR increases diagnostic confidence in detection of bone metastases compared with 18F-FDG-PET/CT. Nucl Med Commun. 2015;36:1165–73.
Schraml C, Schmid M, Gatidis S, Schmidt H, la Fougere C, Nikolaou K, et al. Multiparametric analysis of bone marrow in cancer patients using simultaneous PET/MR imaging: correlation of fat fraction, diffusivity, metabolic activity, and anthropometric data. J Magn Reson Imaging. 2015;42:1048–56.
Hirsch FW, Sattler B, Sorge I, Kurch L, Viehweger A, Ritter L, et al. PET/MR in children. Initial clinical experience in paediatric oncology using an integrated PET/MR scanner. Pediatr Radiol. 2013;43:860–75.
Schafer JF, Gatidis S, Schmidt H, Guckel B, Bezrukov I, Pfannenberg CA, et al. Simultaneous whole-body PET/MR imaging in comparison to PET/CT in pediatric oncology: initial results. Radiology. 2014;273:220–31.
Gatidis S, Schmidt H, Gucke B, Bezrukov I, Seitz G, Ebinger M, et al. Comprehensive oncologic imaging in infants and preschool children with substantially reduced radiation exposure using combined simultaneous 18F-fluorodeoxyglucose positron emission tomography/magnetic resonance imaging: a direct comparison to 18F-fluorodeoxyglucose positron emission tomography/computed tomography. Invest Radiol. 2016;51:7–14.
Platzek I, Beuthien-Baumann B, Ordemann R, Maus J, Schramm G, Kitzler HH, et al. FDG PET/MR for the assessment of lymph node involvement in lymphoma: initial results and role of diffusion-weighted MR. Acad Radiol. 2014;21:1314–9.
Heacock L, Weissbrot J, Raad R, Campbell N, Friedman KP, Ponzo F, et al. PET/MRI for the evaluation of patients with lymphoma: initial observations. AJR Am J Roentgenol. 2015;204:842–8.
Nensa F, Tezgah E, Poeppel TD, Jensen CJ, Schelhorn J, Kohler J, et al. Integrated 18F-FDG PET/MR imaging in the assessment of cardiac masses: a pilot study. J Nucl Med. 2015;56:255–60.
Lee G, H I, Kim SJ, Jeong YJ, Kim IJ, Pak K, et al. Clinical implication of PET/MR imaging in preoperative esophageal cancer staging: comparison with PET/CT, endoscopic ultrasonography, and CT. J Nucl Med. 2014;55:1242–7.
Drzezga A, Souvatzoglou M, Eiber M, Beer AJ, Furst S, Martinez-Moller A, et al. First clinical experience with integrated whole-body PET/MR: comparison to PET/CT in patients with oncologic diagnoses. J Nucl Med. 2012;53:845–55.
Appenzeller P, Mader C, Huellner MW, Schmidt D, Schmid D, Boss A, et al. PET/CT versus body coil PET/MRI: how low can you go? Insights Imaging. 2013;4:481–90.
Schaarschmidt B, Buchbender C, Gomez B, Rubbert C, Hild F, Kohler J, et al. Thoracic staging of non-small-cell lung cancer using integrated (18)F-FDG PET/MR imaging: diagnostic value of different MR sequences. Eur J Nucl Med Mol Imaging. 2015;42:1257–67.
Schaarschmidt BM, Grueneisen J, Heusch P, Gomez B, Beiderwellen K, Ruhlmann V, et al. Oncological whole-body staging in integrated (18)F-FDG PET/MR: value of different MR sequences for simultaneous PET and MR reading. Eur J Radiol. 2015;84:1285–92.
Sagiyama K, Watanabe Y, Kamei R, Baba S, Honda H. Comparison of positron emission tomography diffusion-weighted imaging (PET/DWI) registration quality in a PET/MR scanner: Zoomed DWI vs. Conventional DWI. J Magn Reson Imaging. 2015. doi:10.1002/jmri.25059.
Huellner MW, Appenzeller P, Kuhn FP, Husmann L, Pietsch CM, Burger IA, et al. Whole-body nonenhanced PET/MR versus PET/CT in the staging and restaging of cancers: preliminary observations. Radiology. 2014;273:859–69.
Reiner CS, Stolzmann P, Husmann L, Burger IA, Hullner MW, Schaefer NG, et al. Protocol requirements and diagnostic value of PET/MR imaging for liver metastasis detection. Eur J Nucl Med Mol Imaging. 2014;41:649–58.
Varoquaux A, Rager O, Poncet A, Delattre BM, Ratib O, Becker CD, et al. Detection and quantification of focal uptake in head and neck tumours: (18)F-FDG PET/MR versus PET/CT. Eur J Nucl Med Mol Imaging. 2014;41:462–75.
Al-Nabhani KZ, Syed R, Michopoulou S, Alkalbani J, Afaq A, Panagiotidis E, et al. Qualitative and quantitative comparison of PET/CT and PET/MR imaging in clinical practice. J Nucl Med. 2014;55:88–94.
Rakheja R, DeMello L, Chandarana H, Glielmi C, Geppert C, Faul D, et al. Comparison of the accuracy of PET/CT and PET/MRI spatial registration of multiple metastatic lesions. AJR Am J Roentgenol. 2013;201:1120–3.
Jena A, Taneja S, Jha A. Simultaneous PET/MRI: impact on cancer management-A comprehensive review of cases. Indian J Radiol Imaging. 2014;24:107–16.
Catalano OA, Rosen BR, Sahani DV, Hahn PF, Guimaraes AR, Vangel MG, et al. Clinical impact of PET/MR imaging in patients with cancer undergoing same-day PET/CT: initial experience in 134 patients—a hypothesis-generating exploratory study. Radiology. 2013;269:857–69.
Schaarschmidt BM, Buchbender C, Nensa F, Grueneisen J, Gomez B, Kohler J, et al. Correlation of the apparent diffusion coefficient (ADC) with the standardized uptake value (SUV) in lymph node metastases of non-small cell lung cancer (NSCLC) patients using hybrid 18F-FDG PET/MRI. PLoS One. 2015;10:e0116277.
Shih IL, Yen RF, Chen CA, Chen BB, Wei SY, Chang WC, et al. Standardized uptake value and apparent diffusion coefficient of endometrial cancer evaluated with integrated whole-body PET/MR: correlation with pathological prognostic factors. J Magn Reson Imaging. 2015;42:1723–32.
Sun H, Xin J, Zhang S, Guo Q, Lu Y, Zhai W, et al. Anatomical and functional volume concordance between FDG PET, and T2 and diffusion-weighted MRI for cervical cancer: a hybrid PET/MR study. Eur J Nucl Med Mol Imaging. 2014;41:898–905.
Schwenzer NF, Schmidt H, Gatidis S, Brendle C, Muller M, Konigsrainer I, et al. Measurement of apparent diffusion coefficient with simultaneous MR/positron emission tomography in patients with peritoneal carcinomatosis: comparison with 18F-FDG-PET. J Magn Reson Imaging. 2014;40:1121–8.
Rahim MK, Kim SE, So H, Kim HJ, Cheon GJ, Lee ES, et al. Recent trends in PET image interpretations using volumetric and texture-based quantification methods in nuclear oncology. Nucl Med Mol Imaging. 2014;48:1–15.
Byun BH, Kong CB, Lim I, Kim BI, Choi CW, Song WS, et al. Early response monitoring to neoadjuvant chemotherapy in osteosarcoma using sequential (18)F-FDG PET/CT and MRI. Eur J Nucl Med Mol Imaging. 2014;41:1553–62.
Lee G, H I, Kim SJ, Pak K, Cho JS, Jeong YJ, et al. Initial experience of 18F-FDG PET/MRI in thymic epithelial tumors: morphologic, functional, and metabolic biomarkers. Clin Nucl Med. 2016;41:8–14.
Yen RF, Yen MF, Hong RL, Tzen KY, Chien CR, Chen TH. The cost-utility analysis of 18-fluoro-2-deoxyglucose positron emission tomography in the diagnosis of recurrent nasopharyngeal carcinoma. Acad Radiol. 2009;16:54–60.
Pinker K, Bogner W, Baltzer P, Karanikas G, Magometschnigg H, Brader P, et al. Improved differentiation of benign and malignant breast tumors with multiparametric 18fluorodeoxyglucose positron emission tomography magnetic resonance imaging: a feasibility study. Clin Cancer Res. 2014;20:3540–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Hyun Woo Kwon, Ann-Katharina Becker, Jin Mo Goo, and Gi Jeong Cheon declare that they have no conflict of interest.
Ethical Statement
This paper dose not contains any patient information and only includes a representative case originated from previous research approved by the Institutional Review Board of our hospital (1306-055-495). The manuscript has not been published before, or is not under consideration for publication anywhere else. This manuscript has been approved by all co-authors.
Rights and permissions
About this article
Cite this article
Kwon, H.W., Becker, AK., Goo, J.M. et al. FDG Whole-Body PET/MRI in Oncology: a Systematic Review. Nucl Med Mol Imaging 51, 22–31 (2017). https://doi.org/10.1007/s13139-016-0411-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-016-0411-3