Abstract
Purpose
Positron emission tomography (PET)/magnetic resonance imaging (MRI) is a hybrid imaging modality that combines MRI and PET imaging into a comprehensive modality for oncologic evaluation. MRI contributes with excellent soft tissue contrast resolution along with multiparametric information and PET with exquisite high sensitivity. Together they facilitate lesion detection and characterization, TNM staging, and assessment of treatment response. This review aimed to survey the published PET/MRI research findings for body oncology and reflect upon them.
Methods
This narrative overview of the literature summarizes the findings of published research articles on PET/MRI for oncology (excluding neurologic applications) indexed in the online databases Google Scholar, PubMed, and Scopus, from its commercial introduction in 2011 to the present (2023).
Results
The theoretical advantages of PET/MRI have been demonstrated in practice with studies showing PET/MRI has comparable or superior sensitivity and specificity to PET/CT and MRI in most cancers, with the advantage of being acquired in a single session. Limitations include the comparatively lesser availability and the higher cost, both of which are predicted to be offset by increased adoption.
Conclusions
PET/MRI has the potential to become the standard test for staging and post-treatment evaluation of many primary tumors.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Purpose
Simultaneous positron emission tomography (PET)/magnetic resonance imaging (MRI) is a hybrid imaging technique commercially introduced in 2011 [1], after the development of avalanche photodiodes [2], and most recently silicon photomultipliers, breakthroughs in engineering that allowed incorporation of PET detectors into the MRI system without meaningful interference [3]. Previously used photomultiplier tubes suffered interference from magnetic fields making such achievement impossible. PET/MRI promised to address some of the limitations of the current oncologic standard of care imaging, namely, PET/CT. These include the poor soft tissue contrast of CT [4], which is exacerbated when performed with low radiation doses for attenuation correction purposes only. Another shortcoming of the current PET/CT technology is that it cannot acquire PET and CT data at once, but rather does it sequentially. Usually, the attenuation correction CT is acquired first, only subsequently the PET data are obtained [5]. This may lead to misregistration artifacts due to motion, such as breathing and peristalsis [6,7,8]. These artifacts might result in missed lesions in sites adjacent to motion, such as the liver capsule. Moreover, the quantitative measurements extracted from PET, like the ubiquitous maximum standardized uptake value (SUVmax), might be underestimated due to motion averaging [9]. PET/MRI introduces the simultaneous acquisition of PET and MRI and leverages MRI data to motion-correct PET events, resulting in more accurate measurements [10, 11]. MRI also allows protocol customization for specific primaries, adding sequences, such as diffusion-weighted imaging (DWI) and perfusion imaging to evaluate tumors beyond their anatomy. Given all the potential applications of PET/MRI in cancer staging and post-treatment evaluation, this review aims to consolidate the research findings on body oncologic applications of this diagnostic technology.
Methods
This narrative overview of the literature summarizes the findings of published research articles on PET/MRI for oncology (excluding neurologic applications) indexed in the online databases Google Scholar, PubMed, and Scopus, from its commercial introduction in 2011 to the present (2023). The same keyword combinations were used across these platforms. For each organ/system a combination of relevant terms on the topic, plus the term “PET/MRI,” was entered in the appropriate search field. Detailed search queries for each topic are included in Table 1. The returned articles were curated by an expert in PET/MRI, with 12 years of clinical and research experience, and included in the manuscript according to scientific soundness and relevance of the findings. When relevant, the references from returned articles were also reviewed. The review is structured with the following outlines: Head and Neck, Thorax, Breast, Cholangiocarcinoma, Pancreas, Colon and Rectum, Genitourinary Cancer, and Gynecological Cancer.
Results
Head and neck
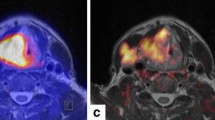
Head and neck cancers (HNC) comprise 3% of all malignancies, with 66,000 cases annually in the United States [12]. Histologically, most of them are squamous cell carcinomas. This anatomic region contains several critical structures. Hence, precise evaluation of the primary tumor size, as well as the involvement of regional lymph nodes, surrounding soft tissues, and osseous structures, is crucial for choosing the ideal treatment strategy. The superior soft tissue contrast of MRI makes it the preferred imaging modality for head and neck cancer evaluation, given its ability to delineate tumor invasion more accurately [13]. Metabolic imaging with 2-Deoxy-2-[18F]fluoroglucose ([18F]FDG) PET can enhance staging accuracy, identification of lymph node involvement, and differentiation of residual or recurrent disease [14]. Therefore, integrated PET/MRI, which combines the advantages of both methods, can meaningfully improve diagnostic accuracy (Fig. 1).
Previous studies show PET/MRI has a higher sensitivity and specificity in detecting HNC than stand-alone PET, CT, or MRI [15,16,17,18]. The high efficacy of PET/MRI in T staging of HNC has been demonstrated in several studies [19,20,21]. For example, PET/MRI showed high accuracy in the assessment of intracranial, retropharyngeal, skull, and prevertebral invasion, in addition to depicting perineural infiltration and muscular involvement [22, 23]. Another study reported a good imaging-pathological correlation between PET/MRI and surgical specimens in 67% of patients [24].
Before starting any therapy, assessment of nodal metastases is crucial since cervical lymph node involvement is common in HNC patients and is considered one of the most important predictors of prognosis. Nodal metastases are usually [18F]FDG avid. Thus, [18F]FDG-PET combined with DWI and dynamic contrast-enhanced MRI can be complementary to overcome the limitations of stand-alone MRI in detecting the small size and variable morphologic appearances of cervical lymph nodes [25].
Regarding M staging, the evaluation of lung and liver metastases is crucial, considering that those are the most common sites of spread in the 15% of patients who do develop distant metastases [26, 27]. While PET/MRI is superior in evaluating distant metastases from a range of primaries to a variety of organs, and especially to the bones, liver, lymph nodes, and peritoneum [28,29,30,31,32], it is limited in detecting lung metastases, especially if < 7 mm in maximal diameter [33]. Therefore, a dedicated chest CT needs to be obtained in those undergoing PET/MRI.
Thorax
Primary lung tumors
Tumors originating in the lung are highly lethal and are projected to account for 20% of cancer-related deaths in 2022 [12]. Non-small-cell lung cancer (NSCLC) comprises 85% of lung tumors and encompasses squamous cell carcinoma, adenocarcinoma, and large cell carcinoma [34]. For this group, [18F]FDG PET/MRI is comparable to [18F]FDG PET/CT in the initial staging [35]. Moreover, PET/MRI outperforms PET/CT in detecting brain and liver metastases [36]. The determination of tumor resectability is also adequately performed using [18F]FDG PET/MRI with a tailored protocol including breath-hold T1-weighted and respiratory-gated T2-weighted images [37]. Another prospective study with 50 patients showed a specificity of 92% and a sensitivity of 97% for [18F]FDG PET/MRI in determining primary tumor resectability [38] (Fig. 2).
Coronal [18F]FDG-PET from PET/CT (a), coronal CT (b), fused PET/CT (c), coronal [18F]FDG-PET from PET/MR (d), coronal STIR (e), fused PET/MRI (f). There is an [18F]FDG-avid mass in the right upper lobe (arrowhead), corresponding to primary lung cancer, with associated [18F]FDG avid right hilar (narrow arrow) and subcarinal (broad arrow) lymphadenopathy. Infiltration of the visceral pleura can be appreciated on STIR
A marked advantage of PET/MRI over PET/CT is the possibility of integrating a brain MRI into the whole-body scan. This integration saves time, contrast media, and is more convenient for the patient. The nervous system is the most common site of lung cancer metastases [39]; therefore, brain evaluation is of utmost importance in lung cancer staging.
Lung metastases
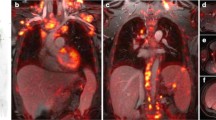
The lung is the third most common site of metastases for men and women [40]. Therefore, besides evaluating primary masses, it is essential to examine the lungs in the setting of any whole-body staging. In this regard, MRI is intrinsically limited due to low proton density and motion artifacts. Even though specialized MRI sequences, such as ultrashort echo time and zero-echo time, provide a better evaluation than standard Dixon images, MRI still falls short of CT performance for lung nodule detection [41,42,43,44]. PET/MRI inherits this deficiency, presenting subpar sensitivity for lung nodules ranging from 30 to 80% [33, 45,46,47,48]. This problem becomes more evident as nodule size decreases, with a sensitivity < 15% for ≤ 5 mm nodules [33]. However, in several types of malignancies, if metastatic disease is already detected elsewhere, diagnosing additional lung lesions may not change management. Thus, in most cases, [18F]FDG-PET/MRI is well positioned as a one-stop-shop modality for cancer staging (Fig. 3). Notwithstanding, in the specific cases where identifying a lung metastasis would result in management changes, PET/MRI should be complemented by chest CT if negative for extrathoracic metastases and lung nodules.
Coronal [18F]FDG-PET (a), coronal T1-weighted fat saturated MRI (b), fused PET/MRI (c). An [18F]FDG-avid nodule (arrow), corresponding to pulmonary metastasis, in the left upper lobe is easily detected on PET. Detection on MRI might be challenging, given small size, in the absence of corresponding marked [18F]FDG avidity
Breast
Breast cancer is the most common neoplasm worldwide, and its annual incidence continues to increase in the United States [12]. MRI is the most accurate cross imaging modality for breast cancer evaluation, with a sensitivity of 99% and specificity of 89%, according to pooled data [49]. Regarding molecular imaging’s role in breast cancer management, the National Comprehensive Cancer Network currently considers [18F]FDG PET/CT an optional test [50]. It is thought that PET/CT helps identify occult nodal or metastatic disease. By incorporating PET into the already best-in-class MRI, [18F]FDG PET/MRI might serve as a one-stop-shop modality in the staging of this disease. The proven diagnostic yield of MRI, combined with the incremental N and M staging performance delivered by PET, results in an even more robust test (Fig. 4). Case in point, [18F]FDG PET/MRI was superior to [18F]FDG PET/CT in whole-body staging [51, 52]. A retrospective study with 36 patients reported an increase in diagnostic confidence and changed the management in one-third of the cases when using [18F]FDG PET/MRI for the initial staging of invasive ductal carcinoma [53]. PET/MRI could detect additional nodal and distant metastases, which led to upstaging in 39% of the cases. A later meta-analysis found that this rate of change was lower when PET/CT was used for initial staging instead (25% for PET/CT vs. 39% for PET/MRI) [54]. In a prospective head-to-head comparison, [18F]FDG PET/MRI identified and correctly classified more lesions than [18F]FDG PET/CT [55]. In another direct comparison, PET/MRI was more effective than PET/CT in detecting bone metastases, presenting a sensitivity of 96% and a specificity of 99% [29]. [18F]FDG PET/MRI-derived biomarkers may also provide in-depth assessment of tumor biology and aggressiveness [56,57,58]. Ductal carcinoma has higher [18F]FDG uptake than lobular carcinoma. In the settings of lobular cancer, while detection of primary tumor, associated synchronous breast lesions, and overall local staging can be achieved by the MRI part of the study, [18F]FDG negative distant metastases in occult areas could be overlooked and may be better evaluated with other radiotracers discussed below [59]. In summary, in ductal breast cancer, [18F]FDG PET/MRI is an excellent tool to identify nodal and distant metastases, especially in younger patients and intermediate to high-grade tumors [53].
Axial [18F]FDG-PET (a), axial high-resolution post-contrast T1-weighted MRI (b), subtraction early arterial phase T1-weighted dynamic contrast-enhanced MRI (c), fused PET/MRI (d). A large, [18F]FDG avid, markedly enhancing mass, corresponding to known invasive ductal carcinoma, infiltrates the left breast (arrows). [18F]FDG avidity improves detection of lymphadenopathy, including the small left internal thoracic lymphadenopathy (arrowheads)
Fibroblast activation protein inhibitor (FAPI) has emerged as an alternative to [18F]FDG, targeting cancer-associated fibroblasts instead of increased glucose metabolism. A retrospective study reported [68Ga]Ga-FAPI uptake in 100% of the 19 primary breast tumors evaluated [60]. However, only 16% (3) of them had lobular carcinoma, which would be the histology in most need of a better radiotracer since [18F]FDG has already been proven effective for ductal carcinoma. For treatment response monitoring, the [68Ga]Ga-FAPI tumor-to-background ratio was associated with complete pathologic response [61]. Moreover, [68Ga]Ga-FAPI PET/MRI performed better than MRI alone for treatment response assessment in breast cancer [61]. PET/CT studies suggest that [68Ga]Ga-FAPI outperforms [18F]FDG in breast cancer, achieving a sensitivity of 100% and specificity of 96% versus 78% and 100%, respectively, for [18F]FDG. Further prospective PET/MRI studies are needed to determine if the same advantages are observed.
Abdomen
Cholangiocarcinoma
Cholangiocarcinomas are tumors arising from the biliary tree whose treatment is often surgical. Thus, careful evaluation of its relationship to adjacent vasculature and lymph node involvement is essential [62]. The majority of cholangiocarcinomas are perihilar, with extrahepatic disease representing 40% of the cases and intrahepatic tumors the other 10% [63]. Morphologically, cholangiocarcinoma may be mass forming, periductal infiltrating, intraductal, superficial spreading, or undefined [62]. The staging of cholangiocarcinoma often involves a multimodal strategy, including contrast-enhanced CT, MRI with MRCP, and PET/CT imaging to completely evaluate the tumoral extension, nodal involvement, and distant metastases, even though only the first two are listed in the National Comprehensive Cancer Network guidelines [64,65,66,67]. The main limitations of such methods that can be overcome by PET/MRI are detecting small lesions, especially subcentimeter intrahepatic lesions, and peritoneal metastases [30].
[18F]FDG PET/MRI resulted in management changes in 30% of the cases with untreated, mass-forming intrahepatic cholangiocarcinoma when compared to the management that would have been proposed after conventional imaging [32]. Another study of [68Ga]Ga-FAPI PET/CT plus PET/MRI versus [18F]FDG in hepatic tumors, including 13 patients with cholangiocarcinoma, showed that [68Ga]Ga-FAPI was better than [18F]FDG for hepatic lesions, but was similar to MRI alone [68]. However, [68Ga]Ga-FAPI PET may contribute by identifying additional positive nodes and distant metastases. Therefore, the available data suggest that PET/MRI, especially when performed with [68Ga]Ga-FAPI, may perform better than CT, MRI, or PET alone in cholangiocarcinoma.
Pancreas
Pancreatic cancer is a lethal pathology with a rising incidence in the United States [69] and the 7th leading cause of cancer-related death worldwide [70]. Pancreatic cancer staging is usually performed with contrast-enhanced CT or MRI [71]. Notwithstanding, the use of [18F]FDG PET/MRI in the staging and post-treatment evaluation may be warranted since a retrospective study found that in 49% of cases [18F]FDG PET/MRI resulted in changes in management when compared to standard of care imaging [72]. Further, post-neoadjuvant therapy response is well correlated to [18F]FDG PET/MRI metrics, such as a change in SUVmax [73]. [68Ga]Ga-FAPI PET/MRI has also been used to successfully used to differentiate pancreatic cancer from IgG4-related pancreatitis in a case of ambiguous [18F]FDG PET imaging [74]. A prospective study with 33 patients comparing [68Ga]Ga-FAPI to [18F]FDG PET/CT showed a higher sensitivity for lymph node metastases but a lower detection rate for hepatic metastases for the former. While more studies are needed to provide more definitive conclusions, PET/MRI is positioned to become a staple in pancreatic cancer staging and restaging.
Colon and Rectum
Colorectal cancer is responsible for a major societal burden, being the third most prevalent cancer in males after prostate and lung and in females after breast and lung [12]. The standard of care for whole-body evaluation in colorectal cancer includes a pelvic MRI and a CT scan of the chest and abdomen. Whole-body [18F]FDG PET/MRI adds clinical value to this diagnostic process by detecting additional lesions and allowing better characterization of extracolonic lesions [75]. In the local staging of rectal tumors, PET/MRI improves T staging [76], the assessment of tumor size, and sphincteric infiltration [77], both of which may change surgical planning (Fig. 5). Moreover, PET/MRI presents better N staging versus MRI alone, thanks to the PET component, which can identify pathologic lymph nodes that do not meet cross sectional imaging size criteria for suspicion (Fig. 6). The M staging is also improved when using PET/MRI versus CT for liver metastases [28, 78, 79] (Fig. 7). This is especially important considering that the liver is the most common site of metastatic spread in colorectal cancer, and up to 25% of patients with colorectal cancer will develop liver metastases over the course of their disease [80, 81]. In oligometastatic colorectal cancer, [18F]FDG PET/MRI may change patient management in 19% of the cases (95% confidence interval 9–37%) [82]. Based on an initial study with [68Ga]Ga-FAPI PET/CT, [68Ga]Ga was superior to [18F]FDG in the detection of primary and metastatic lesions in gastric, duodenal, and colorectal cancers. Additionally, FAPI presented higher uptake in most lesions [83]. [68Ga]Ga-FAPI also led to upstaging when compared to the TNM-assigned stage by [18F]FDG PET/CT in 21% of the cases.
Axial [18F]FDG-PET (a), axial T2-weighted high-resolution FSE MRI (b), fused PET/MRI (c). Semi-circumferential rectal wall thickening, corresponding to known rectal cancer, demonstrates intermediate signal intensity in (b) and marked [18F]FDG uptake in (a). Extension beyond the tunica muscularis (arrowhead) for about 10 mm (T3c) would have been missed on PET images but is well depicted on MRI
Axial [18F]FDG-PET (a), T2-weighted SSFSE (b), fused PET/MRI (c), corresponding level DWI (d).Metastatic lymph node from colorectal cancer. On PET/MRI, metastatic lymph nodes (arrow) tend to concordantly demonstrate increased [18F]FDG avidity, heterogeneous internal signal and diffusion restriction as in this case. Note also exquisite delineation of lymph node from adjacent vasculature despite lack of IV contrast
Axial [18F]FDG-PET (a), axial fat saturated post-contrast T1-weighted MRI (b), fused PET/MRI (c). A subcentimeter, mildly [18F]FDG avid liver metastasis (arrow) might have been missed on stand-alone PET or stand-alone MRI; however, simultaneously acquired MRI and PET facilitate lesion detection and characterization
Musculoskeletal
Emerging studies on the role of PET/MRI in primary bone malignancies found it a promising modality that allows for precise local staging and detection of distant metastases with reduced patient exposure to radiation [84,85,86,87]. To evaluate musculoskeletal (MSK) malignancies, in addition to [18F]FDG, the most widely used radiopharmaceutical, one can use [18F]-sodium fluoride ([18F]-NaF) as a bone-seeking agent capable of detecting bone metastases, especially in tumors with low [18F]FDG affinity, like renal cell carcinoma and thyroid cancer [88]. Based on the published studies, PET/MRI is an excellent modality for the evaluation of soft tissue neoplasms. PET/MRI for T staging in sarcoma is equivalent to MRI, while the PET component improves N and M staging l [89]. MRI is an accurate tool for local staging in soft tissue sarcomas thanks to its ability for excellent delineation of tumor margins as well as detection of osseous and neurovascular invasion. Adding these potentialities of MRI to simultaneously acquire metabolic information from PET can yield a more accurate local staging [90]. The same applies regarding lymph node involvement since [18F]FDG-PET showed 96% sensitivity in the detection of involved lymph nodes compared with stand-alone CT and MRI [91]. Metastases detection by PET/MRI faces the aforementioned limitation of this modality for the assessment of pulmonary metastases, while it remains superior for detecting other distant metastases [4, 28, 33, 52, 92]. FAPI can overcome some of the limitations of [18F]FDG, such as the high [18F]FDG avidity of bone marrow and its low uptake in low-grade sarcoma. FAPI has an excellent tumor-to-background ratio in sarcomas. Therefore, FAPI-PET/MRI can be used as a single examination to provide all the necessary information regarding treatment and staging in affected patients [93, 94]. PET/MRI is also useful to guide treatment and identify biopsy targets in sarcomas [95]. The detailed anatomic (MRI) and functional (PET) information provided simultaneously on PET/MRI can yield more accurate and reliable TNM staging in both primary and metastatic MSK neoplasms. The diagnostic value of this modality may be optimized by the appropriate radiopharmaceutical and MRI protocol selection [96, 97].
Genitourinary
Prostate cancer
Prostate cancer is the most common cancer type in males and is projected to account for 27% of the newly diagnosed cancers in this patient population [12]. Prostate-specific membrane antigen (PSMA) is a transmembrane enzyme that is overexpressed in prostate adenocarcinoma; its degree of expression is usually correlated with tumor grade [98]. PSMA analogs have been developed to target prostatic cancers. Their use is particularly useful in patients with low serum PSA, whose lesions can be more subtle and missed by anatomic imaging alone or non-specific tracers, such as [18F]FDG. Examples of FDA-approved agents to date include [18F]DCFPyL and [68Ga]PSMA-11. [68Ga]PSMA PET/MRI was superior to PET alone and multiparametric MRI alone for prostate cancer localization (AUROCs of 0.88, 0.83, and 0.73, respectively) [99]. Compared to multiparametric pelvic MRI, the current standard of care, PSMA PET/MRI, increases the sensitivity (46% vs. 69%) of extracapsular extension evaluation at the expense of specificity (94% vs. 90%) [100]. PSMA PET/MRI may also improve nodal staging and distant metastasis evaluation, especially in the bones, where MRI is superior to CT [101,102,103] (Fig. 8). Besides initial staging, PSMA PET/MRI performs well in the setting of restaging/biochemical recurrence, with a sensitivity of 99% [104]. PSMA PET/MRI detection rate in biochemical recurrence is related to the PSA levels, with values < 0.5 ng/mL presenting significantly less lesions vs. PSA value ≥ 2 ng/mL [105]. However, even at low PSA levels, PSMA positivity is still relevant at 54.5%, and most of these patients had a distant recurrence in the lymph nodes or bones [106]. Notably, besides stellar performance for whole-body metastases detection, PSMA PET/MRI performs better than PSMA PET/CT to assess recurrent disease in the prostatectomy bed.
Kidney cancer
Renal tumors accounted for 4% of new cancer diagnoses in the United States in 2022 [12]. PSMA is a expressed in several tissues, including the small intestine, proximal renal tubules, and salivary glands [107]. Unfortunately, this expression, coupled with the urinary excretion of most PSMA ligands, makes imaging primary kidney tumors challenging with the radiopharmaceutical [108, 109]. However, in the characterization of primary renal cancers, [68Ga]PSMA PET/CT SUVmax has been shown to differentiate histologic tumor grade [110] and also differentiate benign and malignant tumors [111]. Finally, [18F]-PSMA PET/CT has been used to evaluate treatment response in patients with metastatic renal cell carcinoma undergoing immune checkpoint inhibitor therapy, detecting changes that were missed by RECIST [112]. [18F]DCFPyL has been successfully used in PET/CT imaging of metastatic renal cell carcinoma, detecting more lesions than CT or MRI [113,114,115]. PET/MRI is yet to be applied in the context of renal cancers, but we can expect similar or even better results than with PET/CT.
Gynecological cancer
Cervical, ovarian, and uterine cancer are among the ten most common cancers in females, and gynecologic cancers accounted for about 672,00 deaths in 2020 worldwide [116]. They are classified based on the anatomic origin, including cancers of the ovaries and fallopian tubes, uterine corpus, uterine cervix, endometrium, vagina, and vulva [117]. The risk of gynecologic cancer increases with age, and early diagnosis is paramount for the most effective treatment. PET/MRI showed similar efficacy in the evaluation of local tumor extent with that of contrast-enhanced pelvic MRI, which is a common modality for the evaluation of tumor size and local extension in gynecologic settings [118,119,120,121]. In two studies of cervical cancer, PET/MRI showed high accuracy for T staging (83.3%, 85%) and N staging (90%, 87%) [119, 120, 122] (Fig. 9). Another study using PET/MRI for ovarian cancer reported accuracies of 96.4%, 93.9%, and 100% for T, N, and M staging, respectively (Fig. 10). Additionally, PET/MRI allows better treatment planning due to its higher sensitivity for distant metastasis [122]. In comparison with PET/CT, PET/MRI was reported to be equivalent or more accurate for detecting lymph node metastases in cervical cancer [120, 123]. Moreover, as reported in a study of 30 patients with endometrial cancer, PET/MRI and PET/CT showed the same sensitivity, specificity, and accuracy (100%, 96.3%, and 96.7%, respectively) for detecting pelvic nodal metastases [124]. Another study specifically focused on the detection of deep myometrial invasion and lymph node involvement of Endometrial Cancer using PET/MRI reported good performance in preoperative staging (sensitivity of 0.8571, specificity of 0.9286, accuracy of 0.9143) [125]. Moreover, this study showed that MRI parameters, such as tumor volume, volume index, and tumor volume ratio, together with PET parameters, like total lesion glycolysis, may predict lymphovascular space invasion, with MRI parameters also being able to classify patients into low or high risk. Regarding distant metastases, PET/MRI showed a high sensitivity for the detection of metastatic liver lesions (95%) as well as peritoneal carcinomatosis (97%) [28, 30]. However, more studies are needed to confirm the role of PET/MRI in gynecological malignancies.
Axial [18F]FDG-PET (a), axial T2-weighted MRI (b), fused PET/MRI (c). Large cervical cancer (asterisk) infiltrates the uterus, bilateral parametria, peritoneum (arrow) and metastasizes to pelvic lymph nodes (arrowhead). Note high-quality anatomic layout provided by MRI and perfect coregistration with PET, ensured by simultaneous PET/MR acquisition
Neuroendocrine tumors
Neuroendocrine tumors can arise in different organs and share neuromarker expression, such as chromogranin A and synaptophysin; they also variably express different classes of somatostatin receptors [126]. The evaluation of neuroendocrine tumors is thus facilitated on PET/MRI due to the superb soft tissue contrast resolution of MRI paired with the ability to customize the PET scan by choosing different tracers according to the underlying tumor biology. In a pilot study with [68Ga]DOTATOC including 8 patients, PET/MRI was able to detect all malignant neuroendocrine lesions, although 4 benign and indeterminate lung lesions detected on PET/CT were not seen on PET/MRI [127]. A larger study assessed the performance of the same radiotracer in PET/CT and PET/MRI in 28 patients with neuroendocrine tumors, where PET/MRI outperformed PET/CT in liver lesion detection thanks to the possibility of performing hepatobiliary phase contrast-enhanced imaging and DWI that increased the sensibility beyond PET-positive lesions. In a separate study with similar design, review of 197 lesions in 30 patients showed a higher proportion of correctly classified neuroendocrine lesions on PET/MRI than on PET/CT (90.8% vs. 86.7%, p = 0.031), with PET/MRI also presenting better lesion conspicuity than PET/CT, with the caveat of only ~ 10% of the lesions having had pathological confirmation [128]. A third study with 12 patients comparing [68Ga] DOTATOC PET/MRI and PET/CT showed PET/MRI superiority in lesion detection (72.5% vs. 62.7%, respectively, p = 0.01) [129]. Overall, these results suggest PET/MRI is equivalent to or better than PET/CT for neuroendocrine tumor evaluation with DOTATOC. Another study using a different tracer, [68Ga]DOTANOC, showed a slight advantage of PET/MRI over PET/CT for neuroendocrine tumor imaging, with an overall accuracy for PET/MRI of 97% (95% CI, 94.4%–99.6%), versus 94.6% (95% CI, 91.2%–98.1%) for PET/CT [130]. Of note, as mentioned beforehand the evaluation of lung metastases can be compromised on PET/MRI due to MRI limitations for chest imaging, and this study replicated such deficit – three pulmonary lesions and one pleural metastasis were missed by PET/MRI, resulting in 0% sensitivity (95% CI, 0%–70.8%) for this segment. In a retrospective study, PET/MRI with [68Ga]DOTATATE PET/MRI detected additional lesions when compared to [68Ga]PET/CT, with better characterization of lesions in the liver, pancreas, and pelvis [131]. Even without using gadolinium contrast, PET/MRI can achieve a performance similar or superior to contrast-enhanced PET/CT, which is important in patients with risk factors for nephrogenic systemic fibrosis [132]. In summary, a variety of tumor-specific radiotracers have been developed for neuroendocrine tumor imaging, and PET/MRI explores these tracers with remarkable synergy thanks to the intrinsic soft tissue contrast of MRI coupled with protocol flexibility that allows for inclusion of sequences, such as DWI and hepatobiliary phase imaging.
Limitations and Pitfalls
Although a promising technology with several paradigm-shifting results as presented above, PET/MRI presents some drawbacks. Patients with ferromagnetic implants or other devices that are not compatible with MRI might be better suited for PET/CT evaluation. Even in MRI-compatible materials, the associated field inhomogeneity leads to significant image distortion and compromise attenuation correction [133, 134]. Moreover, a comprehensive lung evaluation as part of the whole-body PET/MRI is still suboptimal, especially in lesions measuring 6 mm or smaller. A dedicated chest CT should be considered in cases where sufficient suspicion of lung lesions is warranted. Finally, cost, accessibility, and personnel training remain significant barriers to PET/MRI adoption, despite some studies already showing a favorable cost-effectiveness profile for PET/RMI given appropriate indications [135].
Conclusions
PET/MRI is in a unique position to become the standard whole-body staging for body oncology by combining precise anatomic localization, functional and metabolic data in a single and simultaneously acquired study.
References
Delso G, Fürst S, Jakoby B et al (2011) Performance measurements of the Siemens mMR integrated whole-body PET/MR scanner. J Nucl Med 52:1914–1922
Judenhofer MS, Wehrl HF, Newport DF et al (2008) Simultaneous PET-MRI: a new approach for functional and morphological imaging. Nat Med 14:459–465
Düppenbecker PM, Weissler B, Gebhardt P et al (2016) Development of an MRI-compatible digital SiPM detector stack for simultaneous PET/MRI. Biomed Phys Eng Express 2:015010
Buchbender C, Heusner TA, Lauenstein TC et al (2012) Oncologic PET/MRI, part 2: bone tumors, soft-tissue tumors, melanoma, and lymphoma. J Nucl Med 53:1244–1252
Torres Espallardo I (2017) PET/TAC: bases físicas, instrumentación y avances. Radiologia 59:431–445
Sureshbabu W, Mawlawi O (2005) PET/CT imaging artifacts. J Nucl Med Technol 33:156–161
Blake MA, Singh A, Setty BN et al (2006) Pearls and pitfalls in interpretation of abdominal and pelvic PET-CT. Radiographics 26:1335–1353
Shah SN, Huang SS (2015) Hybrid PET/MR imaging: physics and technical considerations. Abdom Imaging 40:1358–1365
Manber R, Thielemans K, Hutton BF et al (2015) Practical PET respiratory motion correction in clinical PET/MR. J Nucl Med 56:890–896
Fuin N, Catalano OA, Scipioni M et al (2018) Concurrent Respiratory Motion Correction of Abdominal PET and Dynamic Contrast-Enhanced-MRI Using a Compressed Sensing Approach. J Nucl Med 59:1474–1479
Catalano OA, Umutlu L, Fuin N et al (2018) Comparison of the clinical performance of upper abdominal PET/DCE-MRI with and without concurrent respiratory motion correction (MoCo). Eur J Nucl Med Mol Imaging 45:2147–2154
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72:7–33
Ichimiya Y, Alluri K, Marcus C et al (2015) Imaging modality utilization trends in patients with stage III-IV oropharyngeal squamous cell carcinoma. Am J Nucl Med Mol Imaging 5:154–161
Boss A, Stegger L, Bisdas S et al (2011) Feasibility of simultaneous PET/MR imaging in the head and upper neck area. Eur Radiol 21:1439–1446
Patel LD, Bridgham K, Ciriello J et al (2022) PET/MR imaging in evaluating treatment failure of head and neck malignancies: a neck imaging reporting and data system-based study. AJNR Am J Neuroradiol 43:435–441
Park J, Pak K, Yun TJ et al (2020) Diagnostic Accuracy and Confidence of [18F] FDG PET/MRI in comparison with PET or MRI alone in Head and Neck Cancer. Sci Rep 10:9490
Loeffelbein DJ, Souvatzoglou M, Wankerl V et al (2014) Diagnostic value of retrospective PET-MRI fusion in head-and-neck cancer. BMC Cancer 14:846
Huang S-H, Chien C-Y, Lin W-C et al (2011) A comparative study of fused FDG PET/MRI, PET/CT, MRI, and CT imaging for assessing surrounding tissue invasion of advanced buccal squamous cell carcinoma. Clin Nucl Med 36:518–525
Sekine T (2017) PET+ MR versus PET/CT in the initial staging of head and neck cancer, using a trimodality PET/CT+ MR system. Clin Imaging 42:232–239
Sheng-Chieh C-H, Yeh T-C, Yen S-H et al (2018) Clinical utility of simultaneous whole-body 18F-FDG PET/MRI as a single-step imaging modality in the staging of primary nasopharyngeal carcinoma. Eur J Nucl Med Mol Imaging 45:1297–1308
Schaarschmidt BM, Heusch P, Buchbender C et al (2016) Locoregional tumour evaluation of squamous cell carcinoma in the head and neck area: a comparison between MRI, PET/CT and integrated PET/MRI. Eur J Nucl Med Mol Imaging 43:92–102
Chan S-C, Yeh C-H, Yen T-C et al (2018) Clinical utility of simultaneous whole-body 18F-FDG PET/MRI as a single-step imaging modality in the staging of primary nasopharyngeal carcinoma. Eur J Nucl Med Mol Imaging 45:1297–1308
Hayashi K, Kikuchi M, Imai Y et al (2020) Clinical value of fused PET/MRI for surgical planning in patients with oral/oropharyngeal carcinoma. Laryngoscope 130:367–374
Samolyk-Kogaczewska N, Sierko E, Dziemianczyk-Pakiela D et al (2020) Usefulness of hybrid PET/MRI in clinical evaluation of head and neck cancer patients. Cancers (Basel) 12:511
Chen J, Hagiwara M, Givi B et al (2020) Assessment of metastatic lymph nodes in head and neck squamous cell carcinomas using simultaneous 18F-FDG-PET and MRI. Sci Rep 10:20764
Queiroz MA, Huellner MW (2015) PET/MR in cancers of the head and neck. Semin Nucl Med 45:248–265
Ferlito A, Shaha AR, Silver CE et al (2001) Incidence and sites of distant metastases from head and neck cancer. ORL J Otorhinolaryngol Relat Spec 63:202–207
Zhang C, O’Shea A, Parente CA et al (2021) Evaluation of the diagnostic performance of positron emission tomography/magnetic resonance for the diagnosis of liver metastases. Invest Radiol. https://doi.org/10.1097/RLI.0000000000000782
Catalano OA, Nicolai E, Rosen BR et al (2015) Comparison of CE-FDG-PET/CT with CE-FDG-PET/MR in the evaluation of osseous metastases in breast cancer patients. Br J Cancer 112:1452–1460
Furtado FS, Wu MZ, Esfahani SA et al (2022) Positron emission tomography/magnetic resonance imaging versus the standard of care imaging in the diagnosis of peritoneal carcinomatosis. Ann Surg. https://doi.org/10.1097/SLA.0000000000005418
Amorim BJ, Hong TS, Blaszkowsky LS et al (2019) Clinical impact of PET/MR in treated colorectal cancer patients. Eur J Nucl Med Mol Imaging 46:2260–2269
Ferrone C, Goyal L, Qadan M et al (2020) Management implications of fluorodeoxyglucose positron emission tomography/magnetic resonance in untreated intrahepatic cholangiocarcinoma. Eur J Nucl Med Mol Imaging 47:1871–1884
Biondetti P, Vangel MG, Lahoud RM et al (2021) PET/MRI assessment of lung nodules in primary abdominal malignancies: sensitivity and outcome analysis. Eur J Nucl Med Mol Imaging 48:1976–1986
Ettinger DS, Akerley W, Bepler G et al (2010) Non–small cell lung cancer. J Natl Compr Canc Netw 8:740–801
Lee SM, Goo JM, Park CM et al (2016) Preoperative staging of non-small cell lung cancer: prospective comparison of PET/MR and PET/CT. Eur Radiol 26:3850–3857
Mayerhoefer ME, Prosch H, Beer L et al (2020) PET/MRI versus PET/CT in oncology: a prospective single-center study of 330 examinations focusing on implications for patient management and cost considerations. Eur J Nucl Med Mol Imaging 47:51–60
Messerli M (2019) Value of PET/MRI for assessing tumor resectability in NSCLCdIntra-individual comparison with PET/CT. The Br J Radiol. https://doi.org/10.1259/bjr.20180379
Fraioli F, Screaton NJ, Janes SM et al (2015) Non-small-cell lung cancer resectability: diagnostic value of PET/MR. Eur J Nucl Med Mol Imaging 42:49–55
Riihimäki M, Hemminki A, Fallah M et al (2014) Metastatic sites and survival in lung cancer. Lung Cancer 86:78–84
Riihimäki M, Thomsen H, Sundquist K et al (2018) Clinical landscape of cancer metastases. Cancer Med 7:5534–5542
Zeng F, Nogami M, Ueno YR et al (2020) Diagnostic performance of zero-TE lung MR imaging in FDG PET/MRI for pulmonary malignancies. Eur Radiol 30:4995–5003
Rausch I, Quick HH, Cal-Gonzalez J et al (2017) Technical and instrumentational foundations of PET/MRI. Eur J Radiol 94:A3–A13
Regier M, Schwarz D, Henes FO et al (2011) Diffusion-weighted MR-imaging for the detection of pulmonary nodules at 1.5 Tesla: Intraindividual comparison with multidetector computed tomography. J Med Imaging Radiat Oncol 55:266–274
Spick C, Herrmann K, Czernin J (2016) 18F-FDG PET/CT and PET/MRI perform equally well in cancer: Evidence from studies on more than 2,300 patients. J Nucl Med 57:420–430
Burris NS, Johnson KM, Larson PEZ et al (2016) Detection of small pulmonary nodules with ultrashort echo time sequences in oncology patients by using a PET/MR system. Radiology 278:239–246
Chandarana H, Heacock L, Rakheja R et al (2013) Pulmonary nodules in patients with primary malignancy: comparison of hybrid PET/MR and PET/CT imaging. Radiology 268:874–881
Rauscher I, Eiber M, Fürst S et al (2014) PET/MR imaging in the detection and characterization of pulmonary lesions: Technical and diagnostic evaluation in comparison to PET/CT. J Nucl Med 55:724–729
Sawicki LM, Grueneisen J, Buchbender C et al (2016) Comparative performance of 18F-FDG PET/MRI and 18F-FDG PET/CT in detection and characterization of pulmonary lesions in 121 oncologic patients. J Nucl Med 57:582–586
Bennani-Baiti B, Bennani-Baiti N, Baltzer PA (2016) Diagnostic performance of breast magnetic resonance imaging in non-calcified equivocal breast findings: Results from a systematic review and meta-analysis. PLoS ONE 11:e0160346
Groheux D, Hindié E, Espié M, Ulaner GA (2021) Letter to the editor: PET/CT in locally advanced breast cancer: Time for a guideline change? J Natl Compr Canc Netw. https://doi.org/10.6004/jnccn.2021.7050
Botsikas D, Bagetakos I, Picarra M et al (2019) What is the diagnostic performance of 18-FDG-PET/MR compared to PET/CT for the N- and M- staging of breast cancer? Eur Radiol 29:1787–1798
Catalano OA, Daye D, Signore A et al (2017) Staging performance of whole-body DWI, PET/CT and PET/MRI in invasive ductal carcinoma of the breast. Int J Oncol 51:281–288
Taneja S, Jena A, Goel R et al (2014) Simultaneous whole-body 18F-FDG PET-MRI in primary staging of breast cancer: a pilot study. Eur J Radiol 83:2231–2239
Han S, Choi JY (2021) Impact of 18F-FDG PET, PET/CT, and PET/MRI on staging and management as an initial staging modality in breast cancer: a systematic review and meta-analysis. Clin Nucl Med 46:271–282
Sawicki LM, Grueneisen J, Schaarschmidt BM et al (2016) Evaluation of 18F-FDG PET/MRI, 18F-FDG PET/CT, MRI, and CT in whole-body staging of recurrent breast cancer. Eur J Radiol 85:459–465
Incoronato M, Grimaldi AM, Mirabelli P et al (2019) Circulating miRNAs in untreated breast cancer: an exploratory multimodality Morpho-functional study. Cancers (Basel) 11:876
Incoronato M, Mirabelli P, Catalano O et al (2014) CA15-3 is a useful serum tumor marker for diagnostic integration of hybrid positron emission tomography with integrated computed tomography during follow-up of breast cancer patients. BMC Cancer 14:356
Incoronato M, Grimaldi AM, Cavaliere C et al (2018) Relationship between functional imaging and immunohistochemical markers and prediction of breast cancer subtype: a PET/MRI study. Eur J Nucl Med Mol Imaging 45:1680–1693
Groheux D, Cochet A, Humbert O et al (2016) 18F-FDG PET/CT for staging and restaging of breast cancer. J Nucl Med 57(Suppl 1):17S-26S
Backhaus P, Burg MC, Roll W et al (2022) Simultaneous FAPI PET/MRI targeting the fibroblast-activation protein for breast cancer. Radiology 302:39–47
Backhaus P, Burg MC, Asmus I, et al (2022) Initial results of FAPI-PET/MRI to assess response to neoadjuvant chemotherapy in breast cancer. J Nucl Med
Razumilava N, Gores GJ (2014) Cholangiocarcinoma. Lancet 383:2168–2179
DeOliveira ML, Cunningham SC, Cameron JL et al (2007) Cholangiocarcinoma: thirty-one-year experience with 564 patients at a single institution. Ann Surg 245:755–762
Hennedige TP, Neo WT, Venkatesh SK (2014) Imaging of malignancies of the biliary tract- an update. Cancer Imaging 14:14
Kim JY, Kim M-H, Lee TY et al (2008) Clinical role of 18F-FDG PET-CT in suspected and potentially operable cholangiocarcinoma: a prospective study compared with conventional imaging. Am J Gastroenterol 103:1145–1151
Seo S, Hatano E, Higashi T et al (2008) Fluorine-18 fluorodeoxyglucose positron emission tomography predicts lymph node metastasis, P-glycoprotein expression, and recurrence after resection in mass-forming intrahepatic cholangiocarcinoma. Surgery 143:769–777
Benson AB, D’Angelica MI, Abbott DE et al (2021) Hepatobiliary cancers, version 2.2021, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 19:541–565
Siripongsatian D, Promteangtrong C, Kunawudhi A et al (2022) Comparisons of quantitative parameters of Ga-68-labelled fibroblast activating protein inhibitor (FAPI) PET/CT and [18F]F-FDG PET/CT in patients with liver malignancies. Mol Imaging Biol. https://doi.org/10.1007/s11307-022-01732-2
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70:7–30
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424
Tempero MA, Malafa MP, Al-Hawary M, et al (2020) NCCN Guidelines Version 1.2021 Pancreatic Adenocarcinoma. In: National Comprehensive Cancer Network. https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf. Accessed 30 Jan 2021
Furtado FS, Ferrone CR, Lee SI et al (2021) Impact of PET/MRI in the treatment of pancreatic adenocarcinoma: a retrospective cohort study. Mol Imaging Biol 23:456–466
Panda A, Garg I, Truty MJ et al (2021) Borderline resectable and locally advanced pancreatic cancer: FDG PET/MRI and CT tumor metrics for assessment of pathologic response to neoadjuvant therapy and prediction of survival. AJR Am J Roentgenol 217:730–740
Shou Y, Xue Q, Yuan J, Zhao J (2021) 68Ga-FAPI-04 PET/MR is helpful in differential diagnosis of pancreatitis from pancreatic malignancy compared to 18F-FDG PET/CT: a case report. Eur J Hybrid Imaging 5:12
Kang B, Lee JM, Song YS et al (2016) Added value of integrated whole-body PET/MRI for evaluation of colorectal cancer: comparison with contrast-enhanced MDCT. AJR Am J Roentgenol 206:W10-20
Paspulati RM, Partovi S, Herrmann KA et al (2015) Comparison of hybrid FDG PET/MRI compared with PET/CT in colorectal cancer staging and restaging: a pilot study. Abdom Imaging 40:1415–1425
Catalano OA, Lee SI, Parente C et al (2021) Improving staging of rectal cancer in the pelvis: the role of PET/MRI. Eur J Nucl Med Mol Imaging 48:1235–1245
Lee DH, Lee JM, Hur BY et al (2016) Colorectal cancer liver metastases: diagnostic performance and prognostic value of PET/MR imaging. Radiology 280:782–792
Yoon JH, Lee JM, Chang W et al (2020) Initial M staging of rectal cancer: FDG PET/MRI with a hepatocyte-specific contrast agent versus contrast-enhanced CT. Radiology 294:310–319
Hugen N, van de Velde CJH, de Wilt JHW, Nagtegaal ID (2014) Metastatic pattern in colorectal cancer is strongly influenced by histological subtype. Ann Oncol 25:651–657
Disibio G, French SW (2008) Metastatic patterns of cancers: results from a large autopsy study. Arch Pathol Lab Med 132:931–939
Furtado FS, Suarez-Weiss KE, Vangel M et al (2021) Clinical impact of PET/MRI in oligometastatic colorectal cancer. Br J Cancer. https://doi.org/10.1038/s41416-021-01494-8
Pang Y, Zhao L, Luo Z et al (2021) Comparison of 68Ga-FAPI and 18F-FDG uptake in gastric, duodenal, and colorectal cancers. Radiology 298:393–402
Shah SN, Oldan JD (2017) PET/MR imaging of multiple myeloma. Magn Reson Imaging Clin N Am 25:351–365
Purohit BS, Dulguerov P, Burkhardt K, Becker M (2014) Dedifferentiated laryngeal chondrosarcoma: combined morphologic and functional imaging with positron-emission tomography/magnetic resonance imaging. Laryngoscope 124:E274–E277
Christian A, Heusner TC, Lauenstein A, Bockisch G (2012) Oncologic PET/MRI, part 2: bone tumors, soft-tissue tumors, melanoma, and lymphoma. J Nucl Med 53:1244–1252
Eiber M, Takei T, Souvatzoglou M et al (2014) Performance of whole-body integrated 18F-FDG PET/MR in comparison to PET/CT for evaluation of malignant bone lesions. J Nucl Med 55:191–197
Mick CG, James T, Hill JD et al (2014) Molecular imaging in oncology: (18)F-sodium fluoride PET imaging of osseous metastatic disease. AJR Am J Roentgenol 203:263–271
Partovi S, Kohan AA, Zipp L et al (2014) Hybrid PET/MR imaging in two sarcoma patients - clinical benefits and implications for future trials. Int J Clin Exp Med 7:640–648
Holzapfel K, Regler J, Baum T et al (2015) Local staging of soft-tissue sarcoma: Emphasis on assessment of neurovascular encasement-value of MR imaging in 174 confirmed cases. Radiology 275:501–509
Elmanzalawy A, Vali R, Chavhan GB et al (2020) The impact of 18F-FDG PET on initial staging and therapy planning of pediatric soft-tissue sarcoma patients. Pediatr Radiol 50:252–260
Pace L, Nicolai E, Luongo A et al (2014) Comparison of whole-body PET/CT and PET/MRI in breast cancer patients: lesion detection and quantitation of 18F-deoxyglucose uptake in lesions and in normal organ tissues. Eur J Radiol 83:289–296
Kratochwil C, Flechsig P, Lindner T (2019) 68Ga-FAPI PET/CT: Tracer Uptake in 28 Different Kinds of Cancer. J Nucl Med 60:801–805
Fukuda T, Huang M, Janardhanan A et al (2019) Correlation of bone marrow cellularity and metabolic activity in healthy volunteers with simultaneous PET/MR imaging. Skeletal Radiol 48:527–534
Schuler MK, Richter S, Beuthien-Baumann B et al (2013) PET/MRI imaging in high-risk sarcoma: first findings and solving clinical problems. Case Rep Oncol Med 2013:793927
Chaudhry AA, Gul M, Gould E et al (2016) Utility of positron emission tomography-magnetic resonance imaging in musculoskeletal imaging. World J Radiol 8:268–274
Husseini JS, Balza R, Evangelista L et al (2022) PET/MR for evaluation of musculoskeletal malignancies. Clin Transl Imaging 10:71–83
Ross JS, Sheehan CE, Fisher HAG et al (2003) Correlation of primary tumor prostate-specific membrane antigen expression with disease recurrence in prostate cancer. Clin Cancer Res 9:6357–6362
Eiber M, Weirich G, Holzapfel K et al (2016) Simultaneous 68Ga-PSMA HBED-CC PET/MRI improves the localization of primary prostate cancer. Eur Urol 70:829–836
Muehlematter UJ, Burger IA, Becker AS et al (2019) Diagnostic accuracy of multiparametric MRI versus 68Ga-PSMA-11 PET/MRI for extracapsular extension and seminal vesicle invasion in patients with prostate cancer. Radiology 293:350–358
Donswijk ML, van Leeuwen PJ, Vegt E et al (2020) Clinical impact of PSMA PET/CT in primary prostate cancer compared to conventional nodal and distant staging: a retrospective single center study. BMC Cancer 20:723
Hofman MS, Lawrentschuk N, Francis RJ et al (2020) Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet 395:1208–1216
Zhao R, Li Y, Nie L et al (2021) The meta-analysis of the effect of 68Ga-PSMA-PET/CT diagnosis of prostatic cancer compared with bone scan. Medicine (Baltimore) 100:e25417
Guberina N, Hetkamp P, Ruebben H et al (2020) Whole-body integrated [68Ga]PSMA-11-PET/MR imaging in patients with recurrent prostate cancer: comparison with whole-body PET/CT as the standard of reference. Mol Imaging Biol 22:788–796
Mapelli P, Ghezzo S, Samanes Gajate AM et al (2022) 68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in recurrent prostate cancer: diagnostic performance and association with clinical and histopathological data. Cancers (Basel) 14:334
Kranzbühler B, Müller J, Becker AS et al (2020) Detection rate and localization of prostate cancer recurrence using 68Ga-PSMA-11 PET/MRI in patients with low PSA values ≤ 0.5 ng/mL. J Nucl Med 61:194–201
Hoffmann MA, Wieler HJ, Baues C et al (2019) The impact of 68Ga-PSMA PET/CT and PET/MRI on the management of prostate cancer. Urology 130:1–12
Siva S, Udovicich C, Tran B et al (2020) Expanding the role of small-molecule PSMA ligands beyond PET staging of prostate cancer. Nat Rev Urol 17:107–118
Urso L, Castello A, Rocca GC et al (2022) Role of PSMA-ligands imaging in Renal Cell Carcinoma management: current status and future perspectives. J Cancer Res Clin Oncol 148:1299–1311
Gao J, Xu Q, Fu Y et al (2021) Comprehensive evaluation of 68Ga-PSMA-11 PET/CT parameters for discriminating pathological characteristics in primary clear-cell renal cell carcinoma. Eur J Nucl Med Mol Imaging 48:561–569
Golan S, Aviv T, Groshar D et al (2021) Dynamic 68Ga-PSMA-11 PET/CT for the primary evaluation of localized renal mass: a prospective study. J Nucl Med 62:773–778
Mittlmeier LM, Unterrainer M, Rodler S et al (2021) 18F-PSMA-1007 PET/CT for response assessment in patients with metastatic renal cell carcinoma undergoing tyrosine kinase or checkpoint inhibitor therapy: preliminary results. Eur J Nucl Med Mol Imaging 48:2031–2037
Rowe SP, Gorin MA, Hammers HJ et al (2015) Imaging of metastatic clear cell renal cell carcinoma with PSMA-targeted 18F-DCFPyL PET/CT. Ann Nucl Med 29:877–882
Rhee H, Blazak J, Tham CM et al (2016) Pilot study: use of gallium-68 PSMA PET for detection of metastatic lesions in patients with renal tumour. EJNMMI Res 6:76
Meyer AR, Carducci MA, Denmeade SR et al (2019) Improved identification of patients with oligometastatic clear cell renal cell carcinoma with PSMA-targeted 18F-DCFPyL PET/CT. Ann Nucl Med 33:617–623
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
Lee SI, Catalano OA, Dehdashti F (2015) Evaluation of gynecologic cancer with MR imaging, 18F-FDG PET/CT, and PET/MR imaging. J Nucl Med 56:436–443
Ponisio MR, Fowler KJ, Dehdashti F (2016) The emerging role of PET/MR imaging in gynecologic cancers. PET Clin 11:425–440
Sarabhai T, Schaarschmidt BM, Wetter A et al (2018) Comparison of 18F-FDG PET/MRI and MRI for pre-therapeutic tumor staging of patients with primary cancer of the uterine cervix. Eur J Nucl Med Mol Imaging 45:67–76
Kitajima K, Suenaga Y, Ueno Y et al (2014) Fusion of PET and MRI for staging of uterine cervical cancer: comparison with contrast-enhanced (18)F-FDG PET/CT and pelvic MRI. Clin Imaging 38:464–469
Tsuyoshi H, Tsujikawa T, Yamada S et al (2021) Diagnostic value of 18F-FDG PET/MRI for revised 2018 FIGO staging in patients with cervical cancer. Diagnostics (Basel) 11:202
Tsuyoshi H, Tsujikawa T, Yamada S et al (2020) Diagnostic value of [18F]FDG PET/MRI for staging in patients with ovarian cancer. EJNMMI Res 10:117
Anner P, Mayerhöfer M, Wadsak W et al (2018) [18F]FDG-PET/CT and MRI for initial pelvic lymph node staging in patients with cervical carcinoma: The potential usefulness of [18F]FDG-PET/MRI. Oncol Lett. https://doi.org/10.3892/ol.2018.7775
Kitajima K, Suenaga Y, Ueno Y et al (2013) Value of fusion of PET and MRI for staging of endometrial cancer: comparison with 18F-FDG contrast-enhanced PET/CT and dynamic contrast-enhanced pelvic MRI. Eur J Radiol 82:1672–1676
Ironi G, Mapelli P, Bergamini A et al (2022) Hybrid PET/MRI in staging endometrial cancer: Diagnostic and predictive value in a prospective cohort. Clin Nucl Med 47:e221–e229
Graham MM, Gu X, Ginader T et al (2017) 68Ga-DOTATOC imaging of neuroendocrine tumors: A systematic review and metaanalysis. J Nucl Med 58:1452–1458
Beiderwellen KJ, Poeppel TD, Hartung-Knemeyer V et al (2013) Simultaneous 68Ga-DOTATOC PET/MRI in patients with gastroenteropancreatic neuroendocrine tumors: initial results. Invest Radiol 48:273–279
Sawicki LM, Deuschl C, Beiderwellen K et al (2017) Evaluation of 68Ga-DOTATOC PET/MRI for whole-body staging of neuroendocrine tumours in comparison with 68Ga-DOTATOC PET/CT. Eur Radiol 27:4091–4099
Jawlakh H, Velikyan I, Welin S, Sundin A (2021) 68Ga-DOTATOC-PET/MRI and11C-5-HTP-PET/MRI are superior to68Ga-DOTATOC-PET/CT for neuroendocrine tumour imaging. J Neuroendocrinol. https://doi.org/10.1111/jne.12981
Berzaczy D, Giraudo C, Haug AR et al (2017) Whole-body 68Ga-DOTANOC PET/MRI versus 68Ga-DOTANOC PET/CT in patients with neuroendocrine tumors: a prospective study in 28 patients. Clin Nucl Med 42:669–674
Alshammari A (2019) Impact of integrated whole body 68Ga PET/MR imaging in comparison with 68Ga PET/CT in lesions detection and diagnosis of suspected neuroendocrine tumours. Am J Intern Med 7:102
Seith F, Schraml C, Reischl G et al (2018) Fast non-enhanced abdominal examination protocols in PET/MRI for patients with neuroendocrine tumors (NET): comparison to multiphase contrast-enhanced PET/CT. Radiol Med 123:860–870
Fuin N, Pedemonte S, Catalano OA et al (2017) PET/MRI in the presence of metal implants: completion of the attenuation map from PET emission data. J Nucl Med 58:840–845
Attenberger U, Catana C, Chandarana H et al (2015) Whole-body FDG PET-MR oncologic imaging: pitfalls in clinical interpretation related to inaccurate MR-based attenuation correction. Abdom Imaging 40:1374–1386
Gassert FG, Rübenthaler J, Cyran CC, et al (2021) 18F FDG PET/MRI with hepatocyte-specific contrast agent for M staging of rectal cancer: a primary economic evaluation. European Journal of Nuclear Medicine and Molecular Imaging
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
This article was conceptualized by OAC, FSF, MH, SM, HK, and AH performed the literature search and data analysis; FSF, MH, and OAC drafted the manuscript; and SM and OAC critically revised the work.
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare no competing interests.
Ethics approval
This is a review study. This article does not contain any studies with human or animal subjects performed by the any of the authors. The Institutional Review Board has confirmed that no ethical approval is required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Furtado, F.S., Hesami, M., Mcdermott, S. et al. The synergistic effect of PET/MRI in whole-body oncologic imaging: an Expert Review. Clin Transl Imaging 11, 351–364 (2023). https://doi.org/10.1007/s40336-023-00577-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40336-023-00577-1