Abstract
In 2017, the Journal of Nuclear Cardiology published many high-quality articles. In this review, we will summarize a selection of these articles to provide a concise review of the main advancements that have recently occurred in the field. In the first article of this 2-part series, we focused on publications dealing with positron emission tomography, computed tomography, and magnetic resonance. This review will place emphasis on myocardial perfusion imaging using single-photon emission computed tomography summarizing advances in the field including prognosis, safety and tolerability, the impact of imaging on management, and the use of novel imaging protocols.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Each year, we summarize a selection of articles that appeared in the prior year in the Journal to provide the reader with a concise review of the main advancement in the field.1,2,3,4,5,6 In the first part of this 2-part series, we addressed advancements in positron emission tomography, computed tomography, and magnetic resonance.7 Since SPECT is of major interest to our readers, we have dedicated this review entirely to the advancements in SPECT that were published in the Journal of Nuclear Cardiology in 2017.
Prognostic Value of MPI
A large body of literature has established the prognostic value of myocardial perfusion imaging (MPI),8,9 Recent advances in imaging have allowed for faster imaging with lower radiation exposure using cadmium-zinc-telluride (CZT) camera technology. Lima et al from Rio de Janeiro, Brazil10 reported on the prognostic data of 3,554 patients (selected from a total of 6128 patients based on propensity matching) who underwent either a 2-day MPI using traditional Anger camera or a 1-day MPI using a CZT camera. Abnormal perfusion was more prevalent in patients who underwent traditional MPI (27.4% vs. 21.6%, P < 0.001). While the rate of annualized hard events was the same between the two groups in patients with abnormal perfusion (3.3% vs. 3.2%, P = NS), it was lower in those with normal perfusion with CZT vs. Anger camera (0.5% vs. 1.0%, P < 0.0). These data are reassuring regarding the prognostic value obtained using lower radiation exposure and shorter protocols with CZT cameras.11
Yao et al reported on the outcomes of older patients (≥ 70 years) undergoing adenosine triphosphate MPI confirming the prognostic value of vasodilator MPI in an elderly population with suspected coronary artery disease (CAD).12 Saab and Hage reviewed the different vasodilators used as stress agents in MPI.13 Other studies evaluated the accuracy of MPI for the detection of multi-vessel CAD,14,15 the prevalence of myocardial ischemia in patients with nonobstructive CAD,16,17,18 and the ability of conventional SPECT to measure myocardial blood flow,19,20 which have implications for prognosis and risk stratification.
Patients with end-stage renal disease (ESRD) are at increased risk of cardiovascular events.21 Doukky et al reported on the prognostic data provided by regadenoson MPI in patients with ESRD followed prospectively in the ASSAUGE and ASSUAGE-CKD trials.22 Abnormal myocardial perfusion (summed stress score ≥ 4) was associated with increased risk of cardiac death, myocardial infarction (MI), or late coronary revascularization (> 90 days post MPI) with an adjusted hazard ratio of 1.80 (95% CI 1.03-3.14, P = 0.039) (Figure 1). Further, a stepwise increase in the severity of perfusion abnormality was associated with a stepwise increase in cardiac events during follow-up. In an accompanying editorial, Miller and Schwartz elaborate on the implications of these findings on the evaluation of ESRD patients prior to renal transplantation 23. In a separate study, Abuzeid et al reported on the outcome of ESRD after renal transplantation in relation to MPI findings.24 Abnormal myocardial perfusion was associated with increased risk of cardiovascular events after adjustment for relevant covariates. These data are useful to extend the value of MPI in risk stratification to the postrenal transplant population, but questions remain regarding the best treatment strategy in this patient population.25

Reproduced with permission from22
Impact of regadenoson-induced perfusion abnormalities on cardiac outcomes adjusted for clinical covariates, depicting Cox proportional hazards survival plots adjusted for age, gender, diabetes, dyslipidemia, smoking, and history of coronary artery disease. SPECT single-photon emission computed tomography, MPI myocardial perfusion imaging, ESRD end-stage renal disease, CD cardiac death, MI myocardial infarction, CR coronary revascularization, Late CR coronary revascularization occurring > 90 days post-MPI, HR hazard ratio, and CI 95% confidence interval.
While the prognostic value of myocardial perfusion abnormalities with MPI has been extensively studied (see the following articles regarding the variability of different software programs used for quantitative assessment of perfusion abnormalities),26,27 nonperfusion abnormalities on MPI also provide useful prognostic data.28 Multiple studies have demonstrated the robust association of a blunted heart-rate response to vasodilator stress and subsequent outcomes.29,30,31,32,33,34 Gomez et al demonstrated that integrating the heart-rate response to vasodilator stress with MPI interpretation improves risk stratification in ESRD patients.35 In particular, patients with normal perfusion, left ventricular ejection fraction (LVEF), and heart-rate response had the best outcomes, while those with abnormal traditional MPI findings and a blunted heart-rate response had the worse outcomes 36. In contrast, and while a drop in blood pressure during exercise stress is associated with worse outcomes, Witbrodt et al demonstrated that the blood pressure response to vasodilator stress does not provide useful prognostic data.37,38 Another MPI variable that has been used in conjunction with perfusion for risk stratification is transient ischemic dilation.39,40 Jameria et al validated cutoffs for transient ischemic dilation using upright imaging on a CZT camera.41,42
Safety and Tolerability
The safety and tolerability of regadenoson and other stress agents have received increased scrutiny over the last few years.43,44 The safety of regadenoson in patients with elevated cardiac markers have not been demonstrated. Rai et al evaluated the safety of different stress agents in 703 (51% with regadenoson) patients who had elevated troponin levels ≤ 7 days prior to testing .45 The composite outcome of death, nonfatal MI, heart failure, stroke, ventricular arrhythmias, atrial fibrillation/flutter, or atrioventricular block requiring intervention within 24 h of testing occurred in 1.6% of patients undergoing regadenoson stress compared to 1.0% with dipyridamole, 1.4% with adenosine, 11.1% with dobutamine, and 3.3% with exercise. The high event rates in patients receiving dobutamine or undergoing exercise should be interpreted with caution given the small sample size of these subsets.46 However, the risk appears to be low in patients undergoing vasodilator stress with no signal of increased risk with any particular vasodilator agent. Another patient population that lacked data on safety of vasodilator stress is the one of severe aortic stenosis. This is of particular interest in the current era with the increasing use of percutaneous aortic valve replacement as a treatment strategy for severe aortic stenosis with percutaneous coronary intervention in those with coexisting CAD. Hussain et al reported on the safety of vasodilator stress (45% regadenoson, 31% dipyridamole, and 24% adenosine) in 95 cases with severe aortic stenosis.47 Common symptoms accompanying vasodilator stress included dyspnea (17%), headache (7%), flushing (6%), and nausea (4%). None of the cases experienced advanced heart block, sustained atrial flutter/fibrillation, ventricular tachycardia or ventricular fibrillation, unstable angina, peri-procedure MI, or death. A significant drop in blood pressure (> 20 mm Hg) occurred in 45% of patients with no significant difference between the stress agents, and this was not accompanied by symptoms of hypotension. In the subset of patients who underwent angiography, the diagnostic performance of MPI was acceptable. These findings suggest that vasodilator stress is well tolerated in patients with severe aortic stenosis although significant drop in blood pressure is common and should be anticipated.48
A known complication associated with the use of adenosine, dipyridamole, and regadenoson for MPI is atrioventricular (AV) block.49 Massalha et al reported on the incidence of conduction abnormalities among 2010 patients who underwent dipyridamole stress testing.50 At baseline, 17% of patients had conduction abnormalities although patients with second or third degree AV block were excluded. After dipyridamole infusion, 0.8% of patients developed a transient change in AV conduction and/or severe sinus bradycardia. A single patient developed sinus arrest that lasted several seconds causing syncope which was treated with aminophylline and atropine. Another patient with atrial fibrillation at baseline developed asystole that lasted for 10 seconds and was treated with aminophylline. Second or third degree AV block occurred in 8 (0.4%) patients. The rate of AV block was higher in patients with baseline conduction abnormalities than in those without (3.1% vs. 0.3%). Subbiah and Patil summarized the rate of clinically significant arrhythmias seen with the different vasodilator stress agents.51 Also, a recent meta-analysis of 34 studies that included data on 22,957 patients who underwent adenosine or regadenoson MPI reported an estimated incidence of AVB of 8.58% (95% CI 5.55-12.21%) with adenosine vs. 0.30% (0.04-0.82%, P < 0.001) with regadenoson.52 In this meta-analysis, the estimated incidence of high-grade AV block (second or third degree) was 5.21% (2.81-8.30%) with adenosine vs. 0.05% (< .001-0.19%, P < 0.001) with regadenoson. These data demonstrate that AV block is infrequent but occurs at a higher frequency with nonselective adenosine agonist, such as adenosine, as compared with selective A2A agonists, such as regadenoson.
The safety and tolerability of repeat consecutive doses (100, 200, or 400 µg or placebo, 10 minutes apart) of regadenoson was assessed in a randomized, repeat-dose, placebo controlled study in 36 healthy subjects.53 There was no consistent pattern of effects on systolic blood pressure associated with repeat-dose administration of regadenoson. Repeat dosing was associated with small reductions in diastolic blood pressure and transient increases in heart rate. There were a total of 27 adverse effects and no serious adverse effects. There was no obvious pattern in the incidence of adverse effects with respect to sequential doses. This study demonstrates that repeat dosing of regadenoson is well tolerated and provides reassurance for repeat dosing in case of intravenous infiltration of the drug or if the radiotracer was unavailable.54 Based on the pharmacodynamics effects of regadenoson, Thomas et al suggest a reasonable re-dosing interval of 2.5 hours.54
Aminophylline, a nonselective adenosine receptor antagonist, is often used to counter the adverse effects of regadenoson, adenosin, resp dipyridamole. However, in case of shortage of aminophylline, an alternative reversal agent is needed. Doran et al compared the use of IV aminophylline (100 mg) with IV (60 mg) and oral caffeine (coffee or diet cola) in patients undergoing regadenoson MPI.55 While IV caffeine provided rapid reversal of regadenoson-induced adverse events, PO caffeine was inferior with respect to complete or predominant reversal. In order to allow for almost complete extraction of the radiotracer, the reversal agent should not be administered at less than 2 minutes from regadenoson administration and preferably after 3 minutes.56 In a separate study, Fughhi et al demonstrated that 75 mg IV aminophylline at 2 minutes following regadenoson does not seem to substantially interfere with the effects of regadenoson on myocardial perfusion by MPI.57,58
Regadenoson and Exercise
Regadenoson, or other pharmacologic agents, is used in lieu of exercise in patients who are unable to exercise to an adequate level. Exercise stress is the preferred modality since it provides prognostic data related to exercise tolerance and hemodynamic changes accompanying activity. It may be difficult to predict whether some patients can achieve adequate exercise making it necessary to switch from exercise to pharmacologic stress. The EXERRT trial assessed the noninferiority and safety of regadenoson administration during recovery from inadequate exercise compared with administration without exercise.59 The study randomized 1,147 patients who were unable to achieve adequate exercise stress to regadenoson at 3 minutes during recovery (Reg-recov) vs. 1 hour after exercise (Reg-rest). All patients underwent a regadenoson-only MPI at least 1 day later (MPI 2). The agreement rate between MPI 1 and MPI 2 was not different between the two groups implying that administering regadenoson during recovery does not alter the interpretation of the images compared to administering it at rest (Figure 2).60 An important limitation of this study is high prevalence of no or minimal ischemia on imaging confounding the results. The study also showed that administration of regadenoson during recovery results in a higher target-to-background ratio. This did not result in improved image quality, since image quality was deemed good/excellent in the majority of patients in both groups. Nevertheless, subdiaphragmatic activity was significant lower for Reg-recov. The study also suggested the safety of this approach when careful monitoring is applied. Overall, adverse events were not different between the two groups although flushing and headaches were numerically less for Reg-recov. As expected, systolic blood pressure decreased with regadenoson administration and was more pronounced in the Reg-recov group, but severe hypotension (systolic blood pressure < 90 mm Hg) was uncommon (< 4%) and not different between groups. Serious adverse events occurred more frequently in Reg-recov (0.9%) than for MPI 2 (0.4%) and for the Reg-rest (0.2%). Importantly, two patients in the Reg-recov group experienced acute coronary syndrome (one with myocardial infarction) although both patients exhibited ischemic ST changes and symptoms prior to regadenoson administration. The authors recommend ‘careful monitoring for symptoms and ECG changes’ during recovery and, if these are present, administration of the tracer and proceeding with MPI without regadenoson.

Reproduced with permission from59
Summed stress scores and summed difference scores agreement rates.
Janvier et al investigated whether addition of isometric exercise (handgrip started 2 minutes before regadenoson injection and continued to 5-7 minutes after injection) improves the side-effect profile of regadenoson while providing better image quality in a small proof-of-concept study.61 Patients who performed handgrip exercise reported fewer side-effects and experienced a drop in blood pressure less frequently (drop in blood pressure by > 10 mm Hg of 45% vs. 77.5%, P = 0.12). Further, this was associated with improved image quality.62 These results, although preliminary, indicate the feasibility of low-level exercise for patients undergoing regadenoson MPI and the potential of such an approach to minimize side-effects and improve image quality using a simple approach.63
Impact of Imaging on Management
The benefit of coronary revascularization in patients with stable CAD on hard cardiovascular outcomes is not well established.64 Serial MPI imaging can be used to assess changes in myocardial perfusion over time and in response to interventions such as coronary revascularization.65,66,67 Nudi et al evaluated the impact of revascularization on myocardial ischemia in 3631 patients undergoing serial MPI of whom 27% underwent revascularization (Figure 3).68 In patients with moderate or severe ischemia at baseline, coronary revascularization was associated with a higher prevalence of no, minimal, or mild ischemia on follow-up imaging compared with medical therapy alone (80% vs. 43%, P < 0.001). These results were supported by multivariable-adjusted, propensity score-adjusted, and propensity score-matched analyses.

Reproduced with permission from 68
Prevalent changes in maximal ischemia scores (MISs) between baseline and repeat myocardial perfusion scintigraphy (MPS) comparing medical therapy (Med Rx) vs. coronary revascularization (Revasc). Negative values indicate an overall decrease (i.e., improvement) in MIS, and positive values an overall increase (i.e., worsening) in MIS.
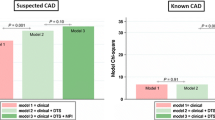
Several studies have compared an anatomic approach towards chest pain with computed tomography coronary angiography (CTCA) with a functional approach using imaging stress test.69,70 The PERFECT trial randomized 411 patients who were admitted form the emergency room with chest pain with negative initial cardiac marker and ECG to CCTA vs. imaging stress test (MPI or stress echocardiography). The two approaches were comparable with respect to time-to-discharge, initiation of cardiac medications, downstream noninvasive cardiac testing, and subsequent hospitalizations.71 However, the CTCA arm was associated with significantly higher rates of invasive angiography (11% vs. 2%, P = 0.001) and PCI (6% vs. 0%). Karthikeyan et al reported on an international, multicenter, randomized controlled trial (IAEASPECT/CTA study) which randomized 303 patients with suspected CAD to an initial strategy of CTCA vs. MPI.72,73 Patients undergoing stress MPI as the initial test were half as likely (adjusted OR 0.51, 95% CI 0.28-0.91, P = 0.023) as those undergoing CTCA to have further downstream noninvasive or invasive cardia testing within 6 months. Both studies, while performed in diverse populations, point to the lower utilization of downstream testing when a functional approach is used.
An ASNC Consensus Statement summarized the evidence base supporting the use of MPI in the clinical evaluation of women presenting with symptoms of stable ischemic heart disease.74 The Journal also published the ACC/AATS/AHA/ASE/ASNC/SCAI/SCCT/STS 2017 Appropriate Use Criteria for Coronary Revascularization in Patients With Stable Ischemic Heart Disease75 which was accompanied by a Guidelines in Review concise summary.76
Stress-Only, Early Imaging, and Reduction in Radiation Exposure
Stress-only imaging is increasingly being used to decrease radiation exposure, shorten MPI time, reduce cost, and streamline patient evaluations.77 Chaudhry et al compared the accuracy of technologist determination of the need of rest imaging to the reference gold standard of nuclear cardiologist determination.78 Based on the reference gold standard nuclear cardiologist assessment, 83% of patients did not require rest imaging. Technologists correctly classified 92% of patients as either needing or not needing rest imaging, while quantitative automated software correctly classified only 72% studies primarily due to incorrectly requesting rest images in 24% of patients. In a simulated model whereby the computer or technologist could correct for the other’s incorrect classification, 97% stress-first images were correctly classified. Using such an approach would greatly facilitate the use of stress-only imaging at sites where nuclear cardiologists are not available to review the images in real time and make a determination for the need of rest images. A separate study demonstrated the value of attenuation correction in decreasing the need for rest imaging in stress-first MPI using CZT cameras with no effect on long-term outcomes.79,80
Typically, imaging is delayed for 30-45 minutes after tracer injection. Earlier imaging would shorten the time required for completing the MPI protocol providing convenience to the patient and improved throughput to the stress laboratory.81 Early imaging may also allow for assessment of LVEF reserve providing additional prognostic data.82 In two separate studies, Katsikis et al and Meyer et al demonstrate the feasibility of early imaging using traditional Anger cameras and CZT cameras.83,84
The use of CZT cameras can result in significant reduction in radiation exposure. Nevertheless, since traditional Anger cameras are widely used, there is significant interest in advancements in imaging that can lead to reduction in radiation using software upgrades such as resolution recovery.85 Lecchi et al investigated how a reduction in counts impacts imaging in normal weight and obese individuals using Bright View gamma camera and Astonish algorithm.86 The investigators altered acquisition time to decrease count statistics simulating a lower dose. They reported that this software allowed for a reduction in acquisition time up to 25% in normal weight subjects and up to 50% in obsess subjects using objective quantitative analysis of perfusion and function.
Phase Analysis
Phase analysis of MPI can provide an assessment of mechanical dyssynchrony.87,88 Zafrir et al evaluated the relationship between mechanical dyssynchrony by phase analysis and cardiac outcomes in patients scheduled to undergo either ICD or CRT-D.89 In multivariate analysis, phase standard deviation was an independent predictor for cardiac death. Their data suggest that phase analysis may be used as an alternative indication for ICD implantation in the future.90 Chiang et al demonstrated that cardiac reverse remodeling after CRT is associated with the prevalence of ventricular arrhythmias.91,92 In a separate study of 1244 patients with CAD undergoing MPI, Hess et al showed that mechanical dyssynchrony by phase analysis was independently associated with mortality and was a stronger predictor of death than electrical dyssynchrony as assessed by QRS duration.93 Thus, assessment of mechanical dyssynchrony may be of benefit in patients undergoing MPI in addition to perfusion and LV function.94
Abbreviations
- AV:
-
Atrioventricular
- CAD:
-
Coronary artery disease
- CTCA:
-
Computed tomography coronary angiography
- CZT:
-
Cadmium-zinc-telluride
- ESRD:
-
End-stage renal disease
- LVEF:
-
Left ventricular ejection fraction
- MI:
-
Myocardial infarction
- MPI:
-
Myocardial perfusion imaging
References
Hage FG, AlJaroudi WA. Review of cardiovascular imaging in the journal of nuclear cardiology in 2016: Part 2 of 2-myocardial perfusion imaging. J Nucl Cardiol. 2017;24:1190–9.
AlJaroudi W, Hage FG. Review of cardiovascular imaging in the Journal of Nuclear Cardiology in 2016. Part 1 of 2: Positron emission tomography, computed tomography and magnetic resonance. J Nucl Cardiol. 2016;2017:649–56.
Hage FG, AlJaroudi WA. Review of cardiovascular imaging in the Journal of Nuclear Cardiology in 2015-Part 2 of 2: Myocardial perfusion imaging. J Nucl Cardiol. 2016;23:493–8.
AlJaroudi WA, Hage FG. Review of cardiovascular imaging in the journal of nuclear cardiology in 2015. Part 1 of 2: Plaque imaging, positron emission tomography, computed tomography, and magnetic resonance. J Nucl Cardiol. 2015;2016:122–30.
Hage FG, AlJaroudi WA. Review of cardiovascular imaging in The Journal of Nuclear Cardiology in 2014: Part 2 of 2: Myocardial perfusion imaging. J Nucl Cardiol. 2015;22:714–9.
AlJaroudi WA, Hage FG. Review of cardiovascular imaging in The Journal of Nuclear Cardiology in 2014: Part 1 of 2: Positron emission tomography, computed tomography, and neuronal imaging. J Nucl Cardiol. 2015;22:507–12.
AlJaroudi WA, Hage FG. Review of cardiovascular imaging in the Journal of Nuclear Cardiology 2017. Part 1 of 2: Positron emission tomography, computed tomography, and magnetic resonance. J Nucl Cardiol. 2017;2018:320–30.
Shaw LJ, Hage FG, Berman DS, Hachamovitch R, Iskandrian A. Prognosis in the era of comparative effectiveness research: where is nuclear cardiology now and where should it be? J Nucl Cardiol. 2012;19:1026–43.
Hage FG, Ghimire G, Lester D, McKay J, Bleich S, El-Hajj S, et al. The prognostic value of regadenoson myocardial perfusion imaging. J Nucl Cardiol. 2015;22:1214–21.
Lima R, Peclat T, Soares T, Ferreira C, Souza AC, Camargo G. Comparison of the prognostic value of myocardial perfusion imaging using a CZT-SPECT camera with a conventional anger camera. J Nucl Cardiol. 2017;24:245–51.
Henzlova MJ, Duvall WL. What do we know? What do we need to know? J Nucl Cardiol. 2017;24:252–4.
Yao Z, Zhu H, Li W, Chen C, Wang H, Shi L, et al. Adenosine triphosphate stress myocardial perfusion imaging for risk stratification of patients aged 70 years and older with suspected coronary artery disease. J Nucl Cardiol. 2017;24:429–33.
Saab R, Hage FG. Vasodilator stress agents for myocardial perfusion imaging. J Nucl Cardiol. 2017;24:434–8.
Gimelli A, Liga R, Duce V, Kusch A, Clemente A, Marzullo P. Accuracy of myocardial perfusion imaging in detecting multivessel coronary artery disease: A cardiac CZT study. J Nucl Cardiol. 2017;24:687–95.
Sabharwal N, Lahiri A. Multi-vessel disease and CZT SPECT. Comparison with coronary angiography. J Nucl Cardiol. 2017;24:696–7.
Gimelli A, Liga R, Pasanisi EM, Casagranda M, Marzullo P. Myocardial ischemia in the absence of obstructive coronary lesion: The role of post-stress diastolic dysfunction in detecting early coronary atherosclerosis. J Nucl Cardiol. 2017;24:1542–50.
Dakik HA. Non-invasive imaging in suspected coronary artery disease: Choosing the right test from the first time. J Nucl Cardiol. 2017;24:523–4.
Songy B. Detection of non-obstructive coronary artery disease: Is post-stress diastolic dysfunction assessed by myocardial perfusion imaging a useful tool? J Nucl Cardiol. 2017;24:1551–4.
Shrestha U, Sciammarella M, Alhassen F, Yeghiazarians Y, Ellin J, Verdin E, et al. Measurement of absolute myocardial blood flow in humans using dynamic cardiac SPECT and (99 m)Tc-tetrofosmin: Method and validation. J Nucl Cardiol. 2017;24:268–77.
Slomka P, Berman DS, Germano G. Myocardial blood flow from SPECT. J Nucl Cardiol. 2017;24:278–81.
Parikh K, Appis A, Doukky R. Cardiac imaging for the assessment of patients being evaluated for kidney or liver transplantation. J Nucl Cardiol. 2015;22:282–96.
Doukky R, Fughhi I, Campagnoli T, Wassouf M, Ali A. The prognostic value of regadenoson SPECT myocardial perfusion imaging in patients with end-stage renal disease. J Nucl Cardiol. 2017;24:112–8.
Miller EO, Schwartz RG. Cardiovascular risk assessment with regadenoson SPECT MPI in patients with end-stage renal disease is safe, effective, and well tolerated: Does it matter? J Nucl Cardiol. 2017;24:119–21.
Abuzeid W, Iwanochko RM, Wang X, Kim SJ, Husain M, Lee DS. Prognostic impact of SPECT-MPI after renal transplantation. J Nucl Cardiol. 2017;24:295–303.
Kan K, Bangalore S. Cardiovascular risk stratification after renal transplant: Is SPECT-MPI the answer? J Nucl Cardiol. 2017;24:304–7.
Ather S, Iskandrian AE, Hage FG. Sources of variability in the measurement of perfusion defect size using commercially available software programs: Are there gender differences? J Nucl Cardiol. 2017;24:1089–93.
Ather S, Iqbal F, Gulotta J, Aljaroudi W, Heo J, Iskandrian AE, et al. Comparison of three commercially available softwares for measuring left ventricular perfusion and function by gated SPECT myocardial perfusion imaging. J Nucl Cardiol. 2014;21:673–81.
Bajaj NS, Singh S, Farag A, El-Hajj S, Heo J, Iskandrian AE, et al. The prognostic value of non-perfusion variables obtained during vasodilator stress myocardial perfusion imaging. J Nucl Cardiol. 2016;23:390–413.
Andrikopoulou E, Hage FG. Heart rate response to regadenoson: Making the case for its value in clinical practice. J Nucl Cardiol. 2016;23:575–80.
Hage FG, Dean P, Iqbal F, Heo J, Iskandrian AE. A blunted heart rate response to regadenoson is an independent prognostic indicator in patients undergoing myocardial perfusion imaging. J Nucl Cardiol. 2011;18:1086–94.
Hage FG, Dean P, Bhatia V, Iqbal F, Heo J, Iskandrian AE. The prognostic value of the heart rate response to adenosine in relation to diabetes mellitus and chronic kidney disease. Am Heart J. 2011;162:356–62.
Andrikopoulou E, AlJaroudi WA, Farag A, Lester D, Patel H, Iskandrian AE, et al. The reproducibility and prognostic value of serial measurements of heart rate response to regadenoson during myocardial perfusion imaging. Eur J Nucl Med Mol Imaging. 2016;43:1493–502.
Bellam N, Veledar E, Dorbala S, Di Carli MF, Shah S, Eapen D, et al. Prognostic significance of impaired chronotropic response to pharmacologic stress Rb-82 PET. J Nucl Cardiol. 2014;21:233–44.
Uzendu AI, Iskandrian A, Hage FG. The heart rate response to regadenoson in patients with atrial fibrillation. J Nucl Cardiol. 2017. https://doi.org/10.1007/s12350-017-1051-4.
Gomez J, Fughhi I, Campagnoli T, Ali A, Doukky R. Impact of integrating heart rate response with perfusion imaging on the prognostic value of regadenoson SPECT myocardial perfusion imaging in patients with end-stage renal disease. J Nucl Cardiol. 2017;24:1666–71.
Dakik HA. Abnormal heart rate response with vasodilator stress myocardial perfusion imaging: Relevance to clinical practice. J Nucl Cardiol. 2017;24:1672–3.
Witbrodt B, Goyal A, Kelkar AA, Dorbala S, Chow BJW, Di Carli MF, et al. Prognostic significance of blood pressure response during vasodilator stress Rb-82 positron emission tomography myocardial perfusion imaging. J Nucl Cardiol. 2017;24:1966–75.
Reyes E, Hage FG. The blood pressure response to vasodilator stress does not provide independent prognostic information. J Nucl Cardiol. 2017;24:1976–8.
Lester D, El-Hajj S, Farag AA, Bhambhvani P, Tauxe L, Heo J, et al. Prognostic value of transient ischemic dilation with regadenoson myocardial perfusion imaging. J Nucl Cardiol. 2016;23:1147–55.
Golzar Y, Olusanya A, Pe N, Dua SG, Golzar J, Gidea C, et al. The significance of automatically measured transient ischemic dilation in identifying severe and extensive coronary artery disease in regadenoson, single-isotope technetium-99 m myocardial perfusion SPECT. J Nucl Cardiol. 2015;22:526–34.
Jameria ZA, Abdallah M, Dwivedi A, Washburn E, Khan N, Khaleghi M, et al. Computer derived transient ischemic dilation ratio for identifying extensive coronary artery disease using a CZT camera and imaging in the upright position. J Nucl Cardiol. 2017;24:1702–8.
Slomka PJ, Berman DS, Germano G. Normal limits for transient ischemic dilation with (99 m)Tc myocardial perfusion SPECT protocols. J Nucl Cardiol. 2017;24:1709–11.
Hage FG. Regadenoson for myocardial perfusion imaging: Is it safe? J Nucl Cardiol. 2014;21:871–6.
Hage FG, Iskandrian AE. Serious complications associated with regadenoson administration for myocardial perfusion imaging: a commentary. J Nucl Cardiol. 2014;21:877–9.
Rai M, Ahlberg AW, Marwell J, Chaudhary W, Savino JA 3rd, Alter EL, et al. Safety of vasodilator stress myocardial perfusion imaging in patients with elevated cardiac biomarkers. J Nucl Cardiol. 2017;24:724–34.
Doukky R, Golzar Y. Safety of stress testing in patients with elevated cardiac biomarkers: Are all modalities created equal? J Nucl Cardiol. 2017;24:735–7.
Hussain N, Chaudhry W, Ahlberg AW, Amara RS, Elfar A, Parker MW, et al. An assessment of the safety, hemodynamic response, and diagnostic accuracy of commonly used vasodilator stressors in patients with severe aortic stenosis. J Nucl Cardiol. 2017;24:1200–13.
Parastatidis I, Lerakis S. The use of vasodilator myocardial perfusion imaging in severe aortic stenosis: Is it time for a new prospective study? J Nucl Cardiol. 2017;24:1214–5.
Andrikopoulou E, Hage FG. Adverse effects associated with regadenoson myocardial perfusion imaging. J Nucl Cardiol. 2018. https://doi.org/10.1007/s12350-018-1218-7.
Massalha S, Reizberg I, Israel O, Kapeliovich M, Sholy H, Koskosi A, et al. Conduction abnormalities during dipyridamole stress testing. J Nucl Cardiol. 2017;24:405–9.
Subbiah R, Patil PV. Arrhythmias in vasodilator stress testing. J Nucl Cardiol. 2017;24:410–2.
Andrikopoulou E, Morgan CJ, Brice L, Bajaj NS, Doppalapudi H, Iskandrian AE, et al. Incidence of atrioventricular block with vasodilator stress SPECT: A meta-analysis. J Nucl Cardiol. 2017. https://doi.org/10.1007/s12350-017-1081-y.
Townsend R, Desai A, Rammelsberg D, Kowalski D, Simmons N, Kitt TM. Safety and tolerability of intravenous regadenoson in healthy subjects: A randomized, repeat-dose, placebo-controlled study. J Nucl Cardiol. 2017;24:57–65.
Thomas GS, Jolly AF, Safani M. When to re-dose regadenoson? J Nucl Cardiol. 2017;24:66–8.
Doran JA, Sajjad W, Schneider MD, Gupta R, Mackin ML, Schwartz RG. Aminophylline and caffeine for reversal of adverse symptoms associated with regadenoson SPECT MPI. J Nucl Cardiol. 2017;24:1062–70.
Jolly AF, Thomas GS. Intravenous caffeine: An alternative to aminophylline to reverse adverse effects during regadenoson myocardial perfusion imaging. J Nucl Cardiol. 2017;24:1071–4.
Fughhi I, Campagnoli T, Ali A, Doukky R. Impact of a regimented aminophylline administration protocol on the burden of regadenoson-induced ischemia detected by SPECT myocardial perfusion imaging. J Nucl Cardiol. 2017;24:1571–8.
Daya HA, Hage FG. Effect of aminophylline administration on the diagnostic yield of vasodilator myocardial perfusion imaging. J Nucl Cardiol. 2017;24:1579–82.
Thomas GS, Cullom SJ, Kitt TM, Feaheny KM, Ananthasubramaniam K, Gropler RJ, et al. The EXERRT trial: “EXErcise to Regadenoson in Recovery Trial”: A phase 3b, open-label, parallel group, randomized, multicenter study to assess regadenoson administration following an inadequate exercise stress test as compared to regadenoson without exercise for myocardial perfusion imaging using a SPECT protocol. J Nucl Cardiol. 2017;24:788–802.
Mahmarian JJ. Regadenoson stress during low-level exercise: The EXERRT trial-does it move the needle? J Nucl Cardiol. 2017;24:803–8.
Janvier L, Pinaquy J, Douard H, Karcher G, Bordenave L. A useful and easy to develop combined stress test for myocardial perfusion imaging: Regadenoson and isometric exercise, preliminary results. J Nucl Cardiol. 2017;24:34–40.
Ceyrat Q, Mesguich C, Janvier L, Douard H, Bordenave L, Pinaquy JB. The impact of combination of regadenoson and isometric exercise on image quality of myocardial perfusion scintigraphy. J Nucl Cardiol. 2017;24:993–7.
Kansal P, Holly TA. A helping hand for regadenoson tests. J Nucl Cardiol. 2017;24:41–2.
Kureshi F, Abdallah MS. ISCHEMIA, to revascularize or not to revascularize. J Nucl Cardiol. 2017;24:1699–701.
Iskandrian AE, Roth CP, Hage FG. Serial imaging and outcome prediction. J Nucl Cardiol. 2016;23:117–21.
El-Hajj S, AlJaroudi WA, Farag A, Bleich S, Manaoragada P, Iskandrian AE, et al. Effect of changes in perfusion defect size during serial regadenoson myocardial perfusion imaging on cardiovascular outcomes in high-risk patients. J Nucl Cardiol. 2016;23:101–12.
Iskandrian AE, Hage FG, Shaw LJ, Mahmarian JJ, Berman DS. Serial myocardial perfusion imaging: defining a significant change and targeting management decisions. JACC Cardiovasc Imaging. 2014;7:79–96.
Nudi F, Di Belardino N, Versaci F, Pinto A, Procaccini E, Neri G, et al. Impact of coronary revascularization vs medical therapy on ischemia among stable patients with or suspected coronary artery disease undergoing serial myocardial perfusion scintigraphy. J Nucl Cardiol. 2017;24:1690–8.
Bittencourt MS, Blankstein R. Coronary computed tomography angiography: How should we act on what we find? J Nucl Cardiol. 2017;24:1279–81.
Peix A. Functional versus anatomical approach in stable coronary artery disease patients: Perspective of low- and middle-income countries. J Nucl Cardiol. 2017;24:518–22.
Uretsky S, Argulian E, Supariwala A, Agarwal SK, El-Hayek G, Chavez P, et al. Comparative effectiveness of coronary CT angiography vs stress cardiac imaging in patients following hospital admission for chest pain work-up: The Prospective First Evaluation in Chest Pain (PERFECT) Trial. J Nucl Cardiol. 2017;24:1267–78.
Karthikeyan G, Guzic Salobir B, Jug B, Devasenapathy N, Alexanderson E, Vitola J, et al. Functional compared to anatomical imaging in the initial evaluation of patients with suspected coronary artery disease: An international, multi-center, randomized controlled trial (IAEA-SPECT/CTA study). J Nucl Cardiol. 2017;24:507–17.
Morgan CJ. Statistical issues associated with terminating a clinical trial due to slow enrollment. J Nucl Cardiol. 2017;24:525–6.
Taqueti VR, Dorbala S, Wolinsky D, Abbott B, Heller GV, Bateman TM, et al. Myocardial perfusion imaging in women for the evaluation of stable ischemic heart disease-state-of-the-evidence and clinical recommendations. J Nucl Cardiol. 2017;24:1402–26.
Patel MR, Calhoon JH, Dehmer GJ, Grantham JA, Maddox TM, Maron DJ, et al. ACC/AATS/AHA/ASE/ASNC/SCAI/SCCT/STS 2017 appropriate use criteria for coronary revascularization in patients with stable ischemic heart disease: A Report of the American College of Cardiology Appropriate Use Criteria Task Force, American Association for Thoracic Surgery, American Heart Association, American Society of Echocardiography, American Society of Nuclear Cardiology, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Computed Tomography, and Society of Thoracic Surgeons. J Nucl Cardiol. 2017;24:1759–92.
Abu Daya H, Hage FG. Guidelines in review: ACC/AATS/AHA/ASE/ASNC/SCAI/SCCT/STS 2017 appropriate use criteria for coronary revascularization in patients with stable ischemic heart disease. J Nucl Cardiol. 2017;24:1793–9.
Mahmarian JJ. Implementation of stress-only imaging: What will it take? J Nucl Cardiol. 2017;24:821–5.
Chaudhry W, Hussain N, Ahlberg AW, Croft LB, Fernandez AB, Parker MW, et al. Multicenter evaluation of stress-first myocardial perfusion image triage by nuclear technologists and automated quantification. J Nucl Cardiol. 2017;24:809–20.
van Dijk JD, Mouden M, Ottervanger JP, van Dalen JA, Knollema S, Slump CH, et al. Value of attenuation correction in stress-only myocardial perfusion imaging using CZT-SPECT. J Nucl Cardiol. 2017;24:395–401.
Pazhenkottil AP, Kaufmann PA, Gaemperli O. Attenuation correction in stress-only myocardial perfusion imaging. J Nucl Cardiol. 2017;24:402–4.
Taillefer R. Myocardial perfusion imaging with 99mTc-labeled radiopharmaceuticals: How fast can a stress-rest same-day imaging protocol be completed? J Nucl Cardiol. 2017;24:1328–31.
Liga R, Gimelli A. Detection of ischemia with early myocardial perfusion imaging: You see more if you watch before. J Nucl Cardiol. 2017;24:1157–60.
Meyer C, Weinmann P. Validation of early image acquisitions following Tc-99 m sestamibi injection using a semiconductors camera of cadmium-zinc-telluride. J Nucl Cardiol. 2017;24:1149–56.
Katsikis A, Theodorakos A, Kouzoumi A, Kitziri E, Georgiou E, Koutelou M. Fast myocardial perfusion imaging with (99 m)Tc in challenging patients using conventional SPECT cameras. J Nucl Cardiol. 2017;24:1314–27.
Slomka P, Germano G. Optimizing radiation dose and imaging time with conventional myocardial perfusion SPECT: Technical aspects. J Nucl Cardiol. 2017;24:888–91.
Lecchi M, Martinelli I, Zoccarato O, Maioli C, Lucignani G, Del Sole A. Comparative analysis of full-time, half-time, and quarter-time myocardial ECG-gated SPECT quantification in normal-weight and overweight patients. J Nucl Cardiol. 2017;24:876–87.
Salimian S, Thibault B, Finnerty V, Gregoire J, Harel F. Phase analysis of gated blood pool SPECT for multiple stress testing assessments of ventricular mechanical dyssynchrony in a tachycardia-induced dilated cardiomyopathy canine model. J Nucl Cardiol. 2017;24:145–57.
Germano G, Van Kriekinge SD. Measuring mechanical cardiac dyssynchrony in the 3-D era. J Nucl Cardiol. 2017;24:158–61.
Zafrir N, Bental T, Strasberg B, Solodky A, Mats I, Gutstein A, et al. Yield of left ventricular dyssynchrony by gated SPECT MPI in patients with heart failure prior to implantable cardioverter-defibrillator or cardiac resynchronization therapy with a defibrillator: Characteristics and prediction of cardiac outcome. J Nucl Cardiol. 2017;24:122–9.
Yoshinaga K. Predicting cardiac events using ventricular dyssynchrony in patients who received implantable cardioverter defibrillators: Are more treatment options required? J Nucl Cardiol. 2017;24:130–3.
Chiang KF, Hung GU, Tsai SC, Cheng CM, Chang YC, Lin WY, et al. Impact of cardiac reverse remodeling after cardiac resynchronization therapy assessed by myocardial perfusion imaging on ventricular arrhythmia. J Nucl Cardiol. 2017;24:1282–8.
Malhotra S. Myocardial substrate after cardiac resynchronization therapy and the risk of ventricular arrhythmias. J Nucl Cardiol. 2017;24:1289–91.
Hess PL, Shaw LK, Fudim M, Iskandrian AE, Borges-Neto S. The prognostic value of mechanical left ventricular dyssynchrony defined by phase analysis from gated single-photon emission computed tomography myocardial perfusion imaging among patients with coronary heart disease. J Nucl Cardiol. 2017;24:482–90.
Zafrir N. Left ventricular mechanical dyssynchrony in patients with coronary artery disease. J Nucl Cardiol. 2017;24:491–3.
Disclosure
Dr. Fadi G. Hage reports research grant support from Astellas Pharma. Wael A. AlJaroudi has no conflicts of interest related to this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hage, F.G., AlJaroudi, W.A. Review of cardiovascular imaging in the Journal of Nuclear Cardiology in 2017. Part 2 of 2: Myocardial perfusion imaging. J. Nucl. Cardiol. 25, 1390–1399 (2018). https://doi.org/10.1007/s12350-018-1266-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-018-1266-z