Abstract
More than two million people tear their anterior cruciate ligament (ACL) each year, and ACL reconstruction occupies a significant proportion of everyday orthopedic practice, being one of the most commonly performed sports medicine surgical procedures. Patients with postoperative symptoms are frequently imaged to monitor ligament grafts and to identify complications. Given the number of patients undergoing ACL reconstruction, knowledge of the potential complications of this surgery is essential for radiologists. This article provides a review of imaging of ACL reconstruction procedures and the potential complications specific to this surgery.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The anterior cruciate ligament (ACL) injury is a common sports injury with over two million cases occurring every year. The number of ACL injuries has undoubtedly increased over the years with the greater participation of young adults in sporting activities [1]. Initial ground contact flatfooted or with the hindfoot, knee abduction and increased hip flexion may be risk factors for anterior cruciate ligament injury as well as oxidative stress and collagen disorders [2]. In females, the laxness of the anterior cruciate ligament (ACL) is strictly related to estrogen levels across phases of menstrual cycle [3]. ACL reconstruction occupies a significant proportion of everyday orthopedics, being one of the most commonly performed sports medicine procedures. Nevertheless, cell-based therapy just used in other pathologies should be useful in treatment of ACL injuries [4, 5].
The aim of the surgical treatment would be to restore function, minimize symptoms, improve quality and minimize the risk of future complications [6, 7]. Therefore, it is imperative for radiologists to be familiar not only with the normal postoperative appearance but also with the possible complications after surgery that can be diagnosed with imaging.
Surgical procedures
The most commonly used methods for ACL reconstruction are autologous bone-patella tendon-bone graft and autologous four-strand hamstring graft. The bone-patella tendon-bone graft is composed of the middle third of the patellar tendon along with a bone fragment from the distal pole of the patella and the tibial tubercle. The four-strand hamstring graft is made of distal semitendinosus and gracilis tendons that are sutured together. They replace the anteromedial bundle of native ACL. Some authors suggest the use of a double-bundle graft to replicate a more physiologic function of ACL and to provide greater biomechanical stability [8–10].
Tunnels
Anatomic techniques include reconstruction of the femoral and tibial tunnel. The key factor for a correct and long-lasting ACL reconstruction is an accurate position which assures graft isometry independently of the knee movements [11].
Femoral tunnel
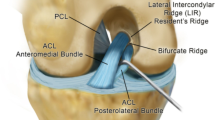
Surgical bony landmarks used are: the footprint of the femoral attachment of the ACL and the lateral intercondylar ridge (resident’s ridge), commonly located anteriorly to the femoral attachment of the ACL [12, 13].
Althouth today there are several dynamic and static imaging modalities, the first technique used for immediate postoperative examination is radiography in frontal and lateral views in maximum extension [14–21]. To establish optimal placement of the femoral tunnel Bernard and Hertel’s grid is used in lateral view. The optimal placement for deep-shallow direction and for high-low direction is a ratio, respectively, of 24 and of 28% [22, 23]. Another study suggests that only 3D computed tomography (CT) is able to depict the femoral tunnel in both directions, compared to radiography and magnetic resonance imaging (MRI) [24, 25]. In a frontal radiographic view, there is an additional measurement, the femoral angle, between the femoral tunnel and a line drawn along the diaphysis. This measures approximately 39°, but, if it is <17°, it is possible for rotatory instability of the knee to occur [26]. On MRI, the femoral tunnel is evaluated on both the coronal and sagittal planes. Its correct positioning on sagittal MRI images is located at the intersection of two lines: the first line drawn along the posterior cortex of the femur and the second line tangent to the roof of the intercondylar region (Fig. 1). On coronal images, the femoral tunnel should open superiorly above the lateral femoral condyle at the 10–11 o’ clock position in the right knee and the 1–2 o’ clock position in the left knee, considering a clock face imposed with the center in the intercondylar notch (Fig. 2) [11, 27, 28].
MRI is necessary for evaluation of the height of the femoral tunnel, but it is less useful to assess depth, if compared with radiography or CT. Correct positioning of the femoral tunnel is fundamental for perfect isometry and to avoid unsuccessful surgery. The femoral tunnel can be drilled with an anteromedial approach and a transtibial approach. With the AM approach, the femoral tunnel is drilled through the anteromedial portal, while the knee is in maximum flexion between 125° and 130°. With the transtibial technique, the femoral tunnel is drilled through the tibial tunnel. The transtibial approach recreates a femoral tunnel more vertically on the coronal plane, in a non-anatomic placement corresponding to a high position in the notch in the anatomic footprint of native ACL. Therefore, this technique has a residual pivot shift and has a worse outcome compared to the anteromedial portal. The AM portal recreates a femoral tunnel more obliquely on the coronal plane, in a low position in the notch in the anatomic footprint of native ACL. This creates greater rotatory stability [28]. Currently, this approach is the most frequently used.
Tibial tunnel
Correct positioning of the tibial tunnel is oriented parallel to the Blumensaat line on sagittal images on MRI or at lateral radiograph. This is a line drawn along the intercondylar roof; so the proximal portion should be located posterior to the intersection of the Blumensaat line with the tibia and distal portion lying near the tibial tuberosity (Fig. 3) [11]. Techniques used to assess the tibial tunnel are lateral radiography or midsagittal MR imaging with the knee in full extension. In addition, the tibial tunnel placement is reported as a ratio in the anteroposterior direction and its value is between 27 and 60% [29]. Other studies show that the best means to evaluate the tibial tunnel are radiography and CT-MIP.
Fixation techniques
Fixation techniques play a key role for optimal functional stability after ACL reconstruction. For anchorage of grafts interference screws, fixation devices such as endobutton or transfixation devices such as a cross-pin are used. The interference screws are considered the best method to use, although some studies have reported screw divergence or graft damage [30]. The endobutton is a device that is pushed through a small hole in the bone and deploys as a T; a transfixation device, such as the cross-pin device, is placed in femoral condyles perpendicular to the long axis of the graft. In biomechanical analysis, the cross-pin is considered a technique that provides pullout strength comparable to other fixation devices. In early postoperative follow-up, it is crucial to perform a radiography to localize the endobutton or interference screw. Cross-pins are made of bio-absorbable material and radiolucent, so in this case MRI is necessary [31].
Neoligament
In the immediate postoperative period, on sagittal MRI, the graft appears as a thick band of homogeneous low signal intensity on short TE sequences [8]. After ACL reconstruction, a gradual process of proliferation of synovial tissue and vascularization of tendons, defined as “ligamentization,” is responsible for an increased signal of grafts in all MRI sequences (Fig. 4). The time of “ligamentization” is different for patellar and hamstring tendons. Patellar tendon ends the process at 24 months, while the hamstring ends at 12 months returning its MRI characteristics of a band of low signal intensity. The presence of normal increased signal in a double-bundle graft, due to four separate strands, may be misdiagnosed as evidence of a graft tear; however, in a double-bundle graft it is oriented along the fibers, while a probable tear is perpendicular to the graft [32]. An increased intrasubstance signal graft can be seen after ACL reconstruction at long-term follow-up on intermediate-weighted and T2-weighted MRI images. Saupe et al. demonstrate that these signal changes do not necessarily correlate with clinical instability or functional limitations, but are probably due to variations of density of the fiber bundles or structural alteration of the collagen graft, or degenerative changes [33].
Complications
Graft tear
Graft tear is reported as the first cause of graft failure; it is most commonly due to a new injury, above all in the sporting population, but it could represent the result of a graft impingement [34].
A graft tear can be partial or complete and clinically manifests with knee instability. On MRI, a partial graft tear appears as a focal area of increased signal intensity on T2-weighted MR images with some fibers remaining intact (Fig. 5a, b). A complete graft tear is defined by the absence of intact fibers with an increased signal on T2-weighted sequences (Fig. 6). Secondary signs maybe anterior tibial translation and uncovering of the posterior horn of the lateral meniscus [35].
Tunnel malposition and graft impingement
A too anterior position of the femoral tunnel is one of the most common technical mistakes related to bone drilling, which may cause a failure of ACL reconstruction [36]. A femoral tunnel drilled too anteriorly may cause a tightened graft when the knee flexes, graft stretching and laxity of the graft on knee extended (Fig. 7 prendi caso di cicco). The placement of the femoral tunnel too posteriorl leads to a tightened graft in knee extension and laxity on flexion.
Too anterior femoral tunnel. Sagittal fat-suppressed intermediate-weighted images of the extended knee showing a femoral tunnel (arrowhead) localized anterior to the intersection of the tangent to the posterior cortical femoral borderline and tangent to the intercondylar notch; the graft looks wavy because of its laxity (arrows)
Roof impingement is the second cause of graft failure. The main cause of roof impingement is the too anterior positioning of the tibial fixation tunnel (Fig. 8). The tibial tunnel should be parallel to the line of the roof of the notch (Blumensaat line) in the sagittal plane, opening behind the intersection of this line with the tibial plateau, with an extended knee [37, 38]. Coronally, it should open opposite the intercondylar notch [12]. If the tibial tunnel is positioned too far anteriorly, the graft comes into contact with the roof of the intercondylar notch during knee extension [38]. On sagittal MR images, the impinged graft appears posteriorly bowed from the anterior margin of the intercondylar notch; increased signal intensity on T1- and T2-weighted images is evident on the anterior two-thirds of the graft [11, 27] (Fig. 9). Sometimes, it may be difficult to distinguish between a graft tear or graft impingement: a signal hyperintensity almost equal to fluid on T2-weighted images involving partial or full thickness of the graft, associated with clinical instability, is strongly suggestive of a tear of the graft [1].
Less commonly, the tibial tunnel is displaced too far laterally so the graft impinges on the sidewall of the intercondylar notch. If the tibial tunnel is too medially and vertically positioned, the graft comes into contact with the posterior cruciate ligament (PCL), limiting knee flexion [39] (Fig. 10a, b). Graft impingement may be treated surgically with arthroscopic “notchplasty” consisting of resection of a small fragment of bone from the medial wall of the lateral femoral condyle, while an interference screw is inserted in the anterior aspect of the tibia to correct the too anterior position [12, 38]. Other causes of graft impingement may be anterior translation of the tibia, and osteophytes from the intercondylar notch (Fig. 11), from the tibial plateau and from the margins of the tibial tunnel which are indicative for roof plasty and notchoplasty [35].
Anterior knee laxity
A complication after ACL reconstruction is anterior laxity due to postoperative thickening of the posterior capsule causing anterior tibial translation. This complication is commonly assessed with clinical examination, but MRI suggestive findings can be present. In this case, it is possible to demonstrate the presence of anterior translation of the tibia of more than 7 mm compared to the femur (Fig. 12). Positive findings on MRI do not always correlate with an arthrometric test, because of their low sensitivity and a low predictive positive value. In this case, clinical examination has greater sensitivity. Negative findings on MRI associated with a negative clinical examination exclude the diagnosis of anterior laxity, because of high specificity and a high negative predictive value of MRI signs [40, 41].
Cystic degeneration of the graft
This is a late complication, also known as ganglion cyst formation, and may be responsible for pain and limitation of motion, but is not related to graft failure. On MRI, it appears as a large cystic lesion between the graft fibers which result intact, thus being distinguished from graft rupture (Fig. 13). Cystic lesion is usually localized adjacent to the tibial or femoral tunnel, which may be enlarged with formation of fluid collections; fluid may extend through the tibial tunnel into the joint space or distally into the soft tissue anterior to the tibial tubercle [42].
Arthrofibrosis
Arthrofibrosis occurs in 1–10% of cases after ACL reconstruction and is the second cause of loss of terminal extension of the knee. The focal form, also known as “Cyclope lesion” because of its arthroscopic appearance, is a nodular lesion characterized by synovial hyperplasia and excessive production of fibro-proliferative tissue which may also contain osseous or cartilaginous tissue. It is typically localized on the anterior portion of the intercondylar notch, appearing on MRI as a well-circumscribed nodule, usually of 10–15 mm, with variable or intermediate signal on all sequences (Fig. 14a, b) [43–45]. It has been reported that a partially torn ACL graft with fibers lying in the anterior intercondylar notch may mimic focal anterior arthrofibrosis on MRI, also called “Pseudocyclops lesion” [46]. A diffuse form of arthrofibrosis is possible, involving anterior and posterior compartments between the ACL graft and joint capsule and the infrapatellar fat pad; it has similar signal intensity to focal form and is associated with capsular thickening [47] (Fig. 15). Usually, the diffuse form is confused with fibrotic scars related to previous arthroscopy; however, fibrotic scars show a linear hypointense strand and not a mass-like characteristic of diffuse arthrofibrosis [31].
Other differential diagnoses of arthrofibrosis include villonodular synovitis and synovial chondromatosis as an intra-articular foreign body, although it is uncommon after ligament surgery [48]. There is a higher prevalence of arthrofibrosis in patients who have had an ACL reconstruction within 4 weeks after the trauma and in those who had signs of knee inflammation at the time of surgery [49]. Arthrofibrosis can be treated with arthroscopic debridement and manipulation.
Tunnel cyst
The formation of tunnel cyst after ACL reconstruction has been reported in 2.2% of cases, femoral tunnel cysts being less common than tibial tunnel cysts [12, 50].
Cystic degeneration of the tibial tunnel can be associated with a widening of the tunnel; more than 13 mm which is the normal admitted value observed during the first 18 months after ACL reconstruction [36] (Fig. 16). Its pathogenesis is unclear; some cases report the presence of a communication between the tunnel and pretibial cyst probably due to a graft failure (Fig. 17). In other cases, there was not a reported communication and the cause was found in an adverse reaction to material used for the screw [poly (l-lactic acid)—PLLA], with consequent formation of a foreign body. In this last case, patients are symptomatic for inflammatory tissue, and MRI findings on T2-weighted and STIR consist of the presence of multiloculated cystic formation in the pretibial region, in the intercondylar notch or in the popliteal fossa with reactive marrow edema surrounding the tibial tunnel cyst [51].
Septic arthritis
Is a rare complication, affecting less than 0.5% of patients, usually caused by Staphylococcus aureus. MR imaging may support the clinical suspicion demonstrating the presence of joint effusion, synovial hyperplasia, osteomyelitis with bone erosion, edema of bone marrow and soft tissue, and soft tissue abscesses (Fig. 18a, b). Less commonly, a sinus tract can be seen as a linear channel extending from bone to skin surface and characterized by low signal intensity on T1-weighted and high signal intensity on short tau inversion recovery (STIR) images [52].
Osteoarthritis
Even if long-term squeal of ACL reconstruction has not yet been defined, such ligament reconstruction does not protect the knee joint from osteoarthritis; on the contrary, ACL reconstruction could be a risk factor for tibiofemoral osteoarthritis, as a long-term complication, 2–15 years after surgery, because of the potential alteration of the natural symmetry of the knee, causing compression of articular cartilage and changes in joint biomechanics [53]. Ostheoarthritic changes may appear on the tibiofemoral or patellofemoral joint, in case of inability of surgery to restore a biomechanical function. Reported risk factors for osteoarthritis seem to be meniscal injury, an interval more than 6 months from injury to reconstruction, and an age over 25 years at the time of surgery [54].
Patients may be symptomatic for pain and limited extension or may not reveal symptoms, resulting positive for radiographic or MRI findings [55].
Intra-articular bodies
A rare complication after ACL reconstruction is the presence of intra-articular bodies, composed of articular cartilage or cortical bone (Fig. 19a, b). This represents another potential cause of decreased range of motion after ACL reconstruction. On MRI, they have an intermediate to low signal on T2-weighted sequences or high intensity and signal equal to that of bone marrow on T1-weighted sequences. They can be secondary to a chondral injury occurred during initial trauma [56].
Donor site abnormalities
Such abnormalities are more frequent when the patellar tendon is used as graft material. On MRI, it can appear as a pathological area with non-tendinous-like signal, thickening of a tendon or a scar formation in the central part. Patellar entrapment syndrome is a cause of limited motion with loss of extension or flexion: it is related to adherence of the rotula to adjacent infrapatellar fat pad [57].
Device complications
Interference screws, pins and other types of graft fixation materials can be a cause of complications or lead to failed surgery. The most common complication is the fracture of the device or/and intra- or extrarticular migration [58] (Fig. 20). In early postoperative healing, cross-pins can fracture with a breach of the femoral cortex and migration of fragments, often inside or posteriorly to the joint. Cross-pins can also protrude beyond the proximal cortex of the lateral femoral condyle, becoming prominent. Another possible complication is partial or complete resorption of the pin. These complications are better visualized with MRI on the coronal and sagittal planes. MRI findings positive for mobilization devices should be correlated with clinical findings of joint instability or pain, as demonstrated by Studler et al. [32]. In this study, a negative value of cross-pin angle (on the axial plane with respect to the transepicondilar line) was significantly associated with cross-pin fractures.
Endobutton evaluation is made with radiography in anteroposterior (AP), latero-lateral (LL) and intercondylar notch view. The normal position of the endobutton is seating on the femoral cortex. When the distance between the femoral cortex and the endobutton is >1 mm (evaluated by AP radiography with a tangent line to the femoral cortex), the endobutton is considered floated. The most frequent complications related to endobutton are migration, dropping into the knee joint and hanging into the femoral tunnel [59]. In double-bundle reconstructions, there is a major incidence of migration of the posterolateral bundle [56].
Certain biomaterials used can cause a severe inflammatory reaction within the bone structures and also possibly in the neighboring soft tissues because of the immune reaction triggered leading to manifestations such as bone marrow edema, synovitis as well as bone resorption around the bone plugs [32] (Fig. 21).
Conclusion
ACL reconstruction is one of the most commonly performed interventions of everyday orthopedic procedures, particularly because of the increasing number of sports injuries. Given the increasing number of patients undergoing ACL reconstruction, it is fundamental for radiologists to be familiar with these procedures and their complications.
References
White LM, Kramer J, Recht MP (2005) MR imaging evaluation of the postoperative knee: ligaments, menisci, and articular cartilage. Skelet Radiol 34:431–452
Nurzynska D, Di Meglio F, Castaldo C, Latino F, Romano V, Miraglia R, Guerra G, Brunese L, Montagnani S (2012) Flatfoot in children: anatomy of decision making. Ital J Anat Embriol 117(2):98–106
Khowailed IA, Petrofsky J, Lohman E, Daher N, Mohamed O (2015) 17β-estradiol induced effects on anterior cruciate ligament laxness and neuromuscular activation patterns in female runners. J Womens Health (Larchmt) 24(8):670–680
Hirzinger C, Tauber M, Korntner S, Quirchmayr M, Bauer HC, Traweger A, Tempfer H (2014) ACL injuries and stem cell therapy. Arch Orthop Trauma Surg 134(11):1573–1578
Bernuzzi G, Petraglia F, Pedrini MF, Filippo MD, Pogliacomi F, Verdano MA, Costantino C (2014) Use of platelet-rich plasma in the care of sports injuries: our experience with ultrasound-guided injection. Blood Transfus 12(SUPPL.1):s229–s234
De Filippo M, Corsi A, Evaristi L, Bertoldi C, Sverzellati N, Averna R, Crotti P, Bini G, Tamburrini O, Zompatori M, Rossi C (2011) Critical issues in radiology requests and reports. Radiol Med 116(1):152–162. doi:10.1007/s11547-010-0587-z (Epub 2010 Sep 17. English, Italian)
Renström PA (2013) Eight clinical conundrums relating to anterior cruciate ligament (ACL) injury in sport: recent evidence and a personal reflection. Br J Sports Med 47:367–372. doi:10.1136/bjsports-2012-091623
Meyers AB, Haims AH, Menn K, Moukaddam H (2010) Imaging of anterior cruciate ligament repair and its complications. AJR Am J Roentgenol 194:476–484. doi:10.2214/AJR.09.3200
Kiekara T, Järvelä T, Huhtala H, Paakkala A (2012) MRI of double-bundle ACL reconstruction: evaluation of graft findings. Skelet Radiol 41:835–842. doi:10.1007/s00256-011-1285-1
Casagranda BU, Maxwell NJ, Kavanagh EC, Towers JD, Shen W, Fu FH (2009) Normal appearance and complications of double-bundle and selective-bundle anterior cruciate ligament reconstructions using optimal MRI techniques. AJR Am J Roentgenol 192:1407–1415. doi:10.2214/AJR.08.1185
Bencardino JT, Beltran J, Feldman MI, Rose DJ (2009) MR imaging of complications of anterior cruciate ligament graft reconstruction. Radiographics 29:2115–2126. doi:10.1148/rg.297095036
Rayan F, Nanjayan SK, Quah C, Ramoutar D, Konan S, Haddad FS (2015) Review of evolution of tunnel position in anterior cruciate ligament reconstruction. World J Orthop 18(6):252–262. doi:10.5312/wjo.v6.i2.252
Ferretti M, Ekdahl M, Shen W, Fu FH (2007) Osseous landmarks of the femoral attachment of the anterior cruciate ligament: an anatomic study. Arthroscopy 2:1218–1225
Barile A, Conti L, Lanni G, Calvisi V, Masciocchi C (2013) Evaluation of medial meniscus tears and meniscal stability: weight-bearing MRI vs arthroscopy. Eur J Radiol 82(4):633–639
Masciocchi C, Conti L, D’Orazio F, Conchiglia A, Lanni G, Barile A (2012) Errors in musculoskeletal MRI. In: Errors in Radiology. Springer, Milan, pp 209–217
Pinto A, Reginelli A, Pinto F, Sica G, Scaglione M, Berger FH, Romano L, Brunese L (2014) Radiological and practical aspects of body packing. Br J Radiol 87(1036):20130500
Dejour H, Bonnin M (1994) Tibial translation after anterior cruciate ligament rupture. Two radiological tests compared. Bone Joint J 76(5):745–749
Faletti R, Cassinis MC, Fonio P, Grasso A, Battisti G, Bergamasco L, Gandini G (2013) Diffusion-weighted imaging and apparent diffusion coefficient values versus contrast-enhanced MR imaging in the identification and characterisation of acute pyelonephritis. Eur Radiol 23(12):3501–3508
Stijak L, Herzog RF, Schai P (2008) Is there an influence of the tibial slope of the lateral condyle on the ACL lesion? Knee surg sports traumatol arthrosc 16(2):112–117
Zappia M, Cuomo G, Martino MT, Reginelli A, Brunese L (2016) The effect of foot position on Power Doppler Ultrasound grading of Achilles enthesitis. Rheumatol Int 36(6):871–874
Regini E, Mariscotti G, Durando M, Ghione G, Luparia A, Campanino PP, Bianchi CC, Bergamasco L, Fonio P, Gandini G (2014) Radiological assessment of breast density by visual classification (BI-RADS) compared to automated volumetric digital software (Quantra): implications for clinical practice. Radiol Med 119(10):741–749
Parkar AP, Adriaensen ME, Strand T, Inderhaug E, Harlem T, Solheim E (2013) How to read post-operative radiographs and CT scans after single-bundle anterior cruciate ligament reconstruction. Skelet Radiol 42:1489–1500. doi:10.1007/s00256-013-1686-4
Bernard M, Hertel P, Hornung H, Cierpinski T (1997) Femoral insertion of the ACL. Radiographic quadrant method. Am J Knee Surg 10:14–21 (discussion 21-2)
Parkar AP, Adriaensen ME, Fischer-Bredenbeck C, Inderhaug E, Strand T, Assmus J, Solheim E (2015) Measurements of tunnel placements after anterior cruciate ligament reconstruction—a comparison between CT, radiographs and MRI. Knee 22:574–579. doi:10.1016/j.knee.2015.06.011
Mariani S, La Marra A, Arrigoni F, Necozione S, Splendiani A, Di Cesare E, Barile A, Masciocchi C (2015) Dynamic measurement of patello-femoral joint alignment using weight-bearing magnetic resonance imaging (WB-MRI). Eur J Radiol 84(12):2571–2578
Illingworth KD, Hensler D, Working ZM, Macalena JA, Tashman S, Fu FH (2011) A simple evaluation of anterior cruciate ligament femoral tunnel position: the inclination angle and femoral tunnel angle. Am J Sports Med 39:2611–2618. doi:10.1177/0363546511420128
Papakonstantinou O, Chung CB, Chanchairujira K et al (2003) Complications of anterior cruciate ligament reconstruction: MR imaging. Eur Radiol 13:1106–1117
Tomczak RJ, Hehl G, Mergo PJ, Merkle E, Rieber A, Brambs HJ (1997) Tunnel placement in anterior cruciate ligament reconstruction: MRI analysis as an important factor in the radiological report. Skelet Radiol 26:409–413
Hantes ME, Zachos VC, Liantsis A, Venouziou A, Karantanas AH, Malizos KN (2009) Differences in graft orientation using the transtibial and anteromedial portal technique in anterior cruciate ligament reconstruction: a magnetic resonance imaging study. Knee Surg Sports Traumatol Arthrosc 17:880–886. doi:10.1007/s00167-009-0738-8
Stäubli HU, Rauschning W (1994) Tibial attachment area of the anterior cruciate ligament in the extended knee position. Anatomy and cryosections in vitro complemented by magnetic resonance arthrography in vivo. Knee Surg Sports Traumatol Arthrosc 2:138–146
Brand J Jr, Weiler A, Caborn DN, Brown CH Jr, Johnson DL (2000) Graft fixation in cruciate ligament reconstruction. Am J Sports Med 28:761–774
Studler U, White LM, Naraghi AM, Tomlinson G, Kunz M, Kahn G, Marks P (2010) Anterior cruciate ligament reconstruction by using bioabsorbable femoral cross pins: MR imaging findings at follow-up and comparison with clinical findings. Radiology 255:108–116. doi:10.1148/radiol.09091119
Recht MP, Kramer J (2002) MR imaging of the postoperative knee: a pictorial essay. Radiographics 22:765–774
Saupe N, White LM, Chiavaras MM, Essue J, Weller I, Kunz M, Hurtig M, Marks P (2008) Anterior cruciate ligament reconstruction grafts: MR imaging features at long-term follow-up—correlation with functional and clinical evaluation. Radiology 249:581–590. doi:10.1148/radiol.2492071651
Galal A, Abdul-Maksoud S, Al-Kandary S, Abdul-Salam S, Awad A (2011) Anterior cruciate ligament reconstruction: magnetic resonance imaging and factors influencing outcome. Egypt J Radiol Nucl Med 42:193–200
Horton LK, Jacobson JA, Lin J, Hayes CW (2000) MR imaging of anterior cruciate ligament reconstruction graft. AJR Am J Roentgenol 175:1091–1097
Trojani C, Sbihi A, Djian P, Potel JF, Hulet C, Jouve F, Bussière C, Ehkirch FP, Burdin G, Dubrana F, Beaufils P, Franceschi JP, Chassaing V, Colombet P, Neyret (2011) Causes for failure of ACL reconstruction and influence of meniscectomies after revision. Knee Surg Sports Traumatol Arthrosc 19:196–201. doi:10.1007/s00167-010-1201-6
Fineberg MS, Zarins B, Sherman OH (2000) Practical considerations in anterior cruciate ligament replacement surgery. Arthroscopy 16:715–724
Muneta T, Yamamoto H, Ishibashi T, Asahina S, Murakami S, Furuya K (1995) The effects of tibial tunnel placement and roofplasty on reconstructed anterior cruciate ligament knees. Arthroscopy 11:57–62
Howell SM (1998) Principles for placing the tibial tunnel and avoiding roof impingement during reconstruction of a torn anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc 6(Suppl 1):S49–S55
Naraghi AM, Gupta S, Jacks LM, Essue J, Marks P, White LM (2012) Anterior cruciate ligament reconstruction: MR imaging signs of anterior knee laxity in the presence of an intact graft. Radiology 263:802–810. doi:10.1148/radiol.12110779
Bergin D, Morrison WB, Carrino JA, Nallamshetty SN, Bartolozzi AR (2004) Anterior cruciate ligament ganglia and mucoid degeneration: coexistence and clinical correlation. AJR Am J Roentgenol 182:1283–1287
Muellner T, Kdolsky R, Grossschmidt K, Schabus R, Kwasny O, Plenk H Jr (1999) Cyclops and cyclopoid formation after anterior cruciate ligament reconstruction: clinical and histomorphological differences. Knee Surg Sports Traumatol Arthrosc 7:284–289
Bradley DM, Bergman AG, Dillingham MF (2000) MR imaging of cyclops lesions. AJR Am J Roentgenol 174:719–726
Jackson DW, Schaefer RK (1990) Cyclops syndrome: loss of extension following intra-articular anterior cruciate ligament reconstruction. Arthroscopy 6:171–178
Simpfendorfer C, Miniaci A, Subhas N, Winalski CS, Ilaslan H (2015) Pseudocyclops: two cases of ACL graft partial tears mimicking cyclops lesions on MRI. Skelet Radiol 44:1169–1173. doi:10.1007/s00256-015-2100-1
Sanders TG (2002) MR imaging of postoperative ligaments of the knee. Semin Musculoskelet Radiol 6:19–33
Kulczycka P, Larbi A, Malghem J, Thienpont E, Vande Berg B, Lecouvet F (2015) Imaging ACL reconstructions and their complications. Diagn Interv Imaging 96:11–19. doi:10.1016/j.diii.2014.04.007
Mayr HO, Weig TG, Plitz W (2004) Arthrofibrosis following ACL reconstruction: reasons and outcome. Arch Orthop Trauma Surg 124:518–522
Deie M, Sumen Y, Ochi M, Murakami Y, Fujimoto E, Ikuta Y (2000) Pretibial cyst formation after anterior cruciate ligament reconstruction using auto hamstring grafts: two case reports in a prospective study of 89 cases. Magn Reson Imaging 18:973–977
Ghazikhanian V, Beltran J, Nikac V, Feldman M, Bencardino JT (2012) Tibial tunnel and pretibial cysts following ACL graft reconstruction: MR imaging diagnosis. Skelet Radiol 41:1375–1379
Williams RJ 3rd, Laurencin CT, Warren RF, Speciale AC, Brause BD, O’Brien S (1997) Septic arthritis after arthroscopic anterior cruciate ligament reconstruction. Diagnosis and management. Am J Sports Med 25:261–267
Tsuda E, Okamura Y, Ishibashi Y, Otsuka H, Toh S (2001) Techniques for reducing anterior knee symptoms after anterior cruciate ligament reconstruction using a bone-patellar tendon-bone autograft. Am J Sports Med 29:450–456
Seon JK, Song EK, Park SJ (2006) Osteoarthritis after anterior cruciate ligament reconstruction using a patellar tendon autograft. Int Orthop 30:94–98
Culvenor AG, Cook JL, Collins NJ, Crossley KM (2013) Is patellofemoral joint osteoarthritis an under-recognised outcome of anterior cruciate ligament reconstruction? A narrative literature review. Br J Sports Med 47:66–70. doi:10.1136/bjsports-2012-091490
Wu H, Webber C, Fuentes CO, Bensen R, Beattie K, Adachi JD, Xie X, Jabbari F, Levy DR (2007) Prevalence of knee abnormalities in patients with osteoarthritis and anterior cruciate ligament injury identified with peripheral magnetic resonance imaging: a pilot study. Can Assoc Radiol J 58:167–175
Kartus J, Lindahl S, Köhler K, Sernert N, Eriksson BI, Karlsson J (1999) Serial magnetic resonance imaging of the donor site after harvesting the central third of the patellar tendon. A prospective study of 37 patients after arthroscopic anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 7:20–24
Bush-Joseph CA, Bach BR Jr (1998) Migration of femoral interference screw after anterior cruciate ligament reconstruction. Am J Knee Surg 11:32–34
Uchida R, Mae T, Matsumoto N, Kuroda S, Toritsuka Y, Shino K (2014) The effect of cortical button location on its post-operative migration in anatomical double-bundle anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 22:1047–1054. doi:10.1007/s00167-013-2458-3
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
For this type of article, formal consent from a local ethics committee is not required.
Patient consent
Informed consent was obtained from all patients before diagnostic examination.
Rights and permissions
About this article
Cite this article
Zappia, M., Capasso, R., Berritto, D. et al. Anterior cruciate ligament reconstruction: MR imaging findings. Musculoskelet Surg 101 (Suppl 1), 23–35 (2017). https://doi.org/10.1007/s12306-017-0460-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12306-017-0460-5