Abstract
Recently, there has been a renewed interest in primary repair of proximal anterior cruciate ligament (ACL) tears. Magnetic resonance imaging (MRI) plays an important role in preoperative patient selection and in postoperative ligament assessment. Knowledge of the imaging factors that make patients candidates for primary ACL repair, namely proximal tear location and good tissue quality, can help radiologists provide information that is meaningful for surgical decision making. Furthermore, an understanding of the surgical techniques can prevent misinterpretation of the postoperative MRI. This article reviews preoperative MRI characterization of ACL injuries, techniques of arthroscopic primary ACL repair surgery and examples of postoperative MRI findings.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Anterior cruciate ligament (ACL) injuries are common and can have a significant impact on a patient’s activity level and ability to participate in sports activities [1, 2]. The rate of ACL reconstruction surgery within the first year after injury has significantly increased across all ages in the last 20 years [1]. This is thought to be secondary to a combination of unsatisfying outcomes of conservative treatment, with low return to sport rates and a high incidence of progressive meniscal and cartilage loss, in combination with improved surgical techniques and postoperative rehabilitation [3,4,5,6,7].
Primary repair was the first surgical treatment of ACL injuries and was commonly performed in the 1970s and 1980s using an open arthrotomy technique [8,9,10,11]. Although short-term results were promising, many authors found mid and long-term results to be inconsistent [12,13,14]. This led to a shift away from primary repair to augmented primary repair and ultimately to ACL reconstruction [15,16,17].
In 1991, however, at the end of the primary repair era, Sherman et al. reviewed their disappointing mid-term results and noted that the outcomes were better in patients with proximal tears [13]. Indeed, a recent systematic review found that the outcomes of primary repair of proximal tears were significantly better than those of mid-substance tears in historical studies [18]. This data, along with advancements in minimally invasive surgery (i.e. arthroscopy) and modern rehabilitation (i.e. focus on early range-of-motion), has led to a renewed interest in primary repair [19,20,21,22]. In 2015, DiFelice reported the first outcomes of arthroscopic primary repair of proximal ACL tears and showed excellent outcomes at 3.5-year follow-up [23]. Some authors have therefore advocated a treatment algorithm for ACL injuries that is based primarily on tear location and remnant tissue quality [19, 24]. Advocates for primary repair suggest that patients with proximal tears and sufficient tissue quality can be treated with arthroscopic primary repair, and the early data on arthroscopic primary repair is promising [23, 25,26,27,28].
In light of these recent developments, it is important for radiologists to understand preoperative MRI characteristics that make patients potential candidates for primary ACL repair, and to understand the normal and abnormal postoperative imaging appearance after primary ACL repair. Therefore, the aim of this article is to describe the (I) preoperative MRI characteristics that make patients candidates for primary ACL repair, (II) currently performed surgical techniques in primary ACL repair, and (III) normal and abnormal imaging appearance after primary ACL repair.
Repair versus reconstruction
ACL reconstruction is most commonly performed arthroscopically using autograft or allograft tissue, and is known to have low graft failure and high return to sport rates [29,30,31,32,33]. The current reconstruction procedures, however, have limitations. Following bone-patellar tendon-bone autograft, patients frequently experience persistent anterior knee pain and kneeling pain [34,35,36]. Following hamstring autograft patients may experience donor site pain, weakness, and nerve injury [37]. The use of allograft tissue comes with an increased risk of revision surgery, which is particularly high in younger patients [38]. Recent biomechanical research has also shown that ACL reconstruction does not restore normal joint kinematics [39, 40] and may not prevent the early development of osteoarthritis after an ACL injury [41, 42]. Furthermore, revision surgery is often complicated after reconstruction due to pre-existing tunnels, tunnel malpositioning and/or tunnel widening, and the unavailability of autograft tissue [43,44,45]. As a result, outcomes of revision surgery are inferior to those of primary ACL reconstruction [46,47,48].
Arthroscopic primary ACL repair has theoretical advantages over ACL reconstruction surgery and has therefore been increasingly performed. Experimental studies suggest that primary ACL repair, with preservation of the native ligament, may more effectively restore normal joint kinematics and slow the progression of degenerative changes after an ACL injury [49,50,51,52]. Also, when the ACL is primarily repaired, there are usually no tunnels drilled and no graft tissue harvested which allows for a faster recovery with fewer complications when compared to reconstruction surgery [53]. Finally, primary ACL repair can be easily converted to a reconstruction if the primary ACL repair happens to fail in the future: no surgical bridges are burned with the repair procedure.
Preoperative evaluation and patient selection
Patients are first evaluated clinically with a history and physical exam. The majority of ACL injuries are noncontact injuries and patients often report feeling their leg give out, hearing a pop and noticing rapid onset swelling [54,55,56]. Two physical exam maneuvers, the Lachman test and the pivot shift test are valuable in assessment of a possible ACL tear. A recent meta-analysis showed the Lachman test to have 87% sensitivity and a very low negative likelihood ratio, making it an effective test for ruling out a possible ACL tear [57]. The pivot shift test has been shown to have a high specificity and positive likelihood ratio [57].
Patients with a clinical suspicion (abnormal Lachman test and/or pivot shift) for ACL tear are referred for MRI. MRI has been shown to be accurate for diagnosing many intra-articular injuries of the knee with the greatest sensitivity and specificity often found for ACL injuries [58]. A recent meta-analysis showed MRI to be 92% sensitive and 99% specific in the identification of ACL injuries compared with arthroscopic correlation [58]. Historically, the main focus of the MRI was (1) to verify the suspected ACL injury and characterize as a complete or partial tear, if possible, and (2) to carefully evaluate the knee for other injuries that may have an effect on surgical management, surgical outcome, or progression to osteoarthritis of the knee. Recently, however, there has been increased focus on the accurate characterization of the ACL tear location and potentially the tissue quality of the ligament remnant [19]. Surgeons use this information to dictate surgical management and make a preoperative assessment as to whether the patient will be a candidate for primary ACL repair [19, 23, 59, 60].
Tear location
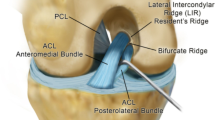
ACL tears at our institution are classified as one of five types which are a modification of the Sherman classification (Table 1, Fig. 1) [13]. A type I tear is a femoral avulsion with more than 90% of the distal ACL left intact (Fig. 2). A type II tear is a proximal tear with 75–90% of the distal ACL left intact. A type III tear is a tear in the middle 50% of the ACL (mid-substance). A type IV tear is a distal tear with 10–25% of the distal ACL left intact and a type V tear is a tibial avulsion with less than 10% of the distal ACL left intact at the tibial insertion. Type V tears can be classified as soft tissue avulsions (type VA) or bony avulsions (type VB). This grading scale has been shown to be reproducible with good inter and intraobserver reliability and has been shown to be helpful in predicting whether patients will be candidates for primary ACL repair [24, 60]. In a recent MRI analysis of 353 complete ACL tears, 16% were classified as type I tears and 27% were classified as type II tears [24]. In a recent study of preoperative MRIs of 130 patients with ACL injuries who went on to surgery, 90% of patients with type I ACL tears and 46% of patients with type II tears on the preoperative MRI were repaired primarily. Evaluation of the femoral attachment on MRI in the standard sagittal extended position can be limited due to volume averaging, which may explain some of the discrepancies between remnant tissue length on imaging and ultimate treatment outcome [61, 62]. MRI can also be limited in assessing for partial ACL tears and in differentiation between complete and partial tears [63,64,65]. While not routinely obtained, there are some who feel that sagittal oblique and coronal oblique imaging can provide a more accurate assessment of the integrity of the femoral attachment [61, 66]. In our experience, we find the axial plane most helpful among standard imaging planes for assessing the ligament at the femoral attachment.
Type I ACL tear with good tissue quality. Preoperative MRI and arthroscopic images of a 42-year-old man who suffered an injury while playing paddle tennis. a–b Sagittal and coronal proton density weighted images showing a complete femoral avulsion ACL tear with very little remnant tissue left on the femoral footprint (white arrow). The distal ACL has homogeneous low signal, often indicating good tissue quality. c Arthroscopic image showing probing of the torn ACL with no remnant tissue left at the femoral footprint (white arrow). d Arthroscopic image after primary ACL repair. Notice the presence of sutures running through the repaired ligament, which can lead to a heterogeneous appearance of the repaired ligament on MRI
Tissue quality
Tissue quality is generally described at our institution as one of the following grades: good (when nearly all fibers are running in the same direction and the signal is homogeneous (Fig. 2), fair (when some of the fibers are running in the same direction and the signal is mildly heterogeneous), or poor (when most fibers are running in different directions and the signal is heterogeneous) (Fig. 3) [60]. Though there is very little literature on grading tissue quality using this scale, a recent retrospective study using this method showed that it can help predict which patients will be candidates for primary ACL repair. In the study, 100% of patients with type 1 tears and good tissue quality on the preoperative MRI had their ACLs repaired primarily after arthroscopic evaluation. In contrast 0% of patients with type 1 tears and fair or poor tissue quality on the preoperative MRI had their ACLs repaired primarily. In patients with type II tears on the preoperative MRI, 88% with good tissue quality and only 13% of those with fair or poor tissue quality were repaired primarily [60].
Poor tissue quality. Preoperative MRI and arthroscopic images of a 21-year-old female who sustained a pole vault injury. a Sagittal proton density image showing a femoral avulsion tear of the proximal ACL (type I). There is heterogeneous signal within the torn distal ligament, which often indicates poor tissue quality (white arrow). There is also abnormal laxity of the ligament, which is collapsed in the intercondylar notch with decreased inclination. b Probing of the ligament during arthroscopy reveals frayed margins (white arrow) and poor tissue quality (arrowhead), making this patient a poor candidate for primary ACL repair
Chronic tears
An important finding that can be mischaracterized as an intact ligament on MRI is chronic complete tear of the ACL with scarring of the ACL to the PCL [63]. This is of importance as there is some thought that scarring of the ACL to the PCL prevents retraction of the ligament. Some patients have been shown to do well following primary ACL repair up to 11 years after initial injury when this finding is present [59, 67, 68].
Surgical techniques
After physical examination and preoperative imaging verify a complete tear of the ACL, all patients are consented for primary ACL repair with possible need for augmentation or for complete ACL reconstruction. The preferred surgical technique of the senior author for primary repair has been described previously in the orthopedic literature [23, 25, 27].
Suturing of ligament
During arthroscopy, the ligament is assessed for tear location and tissue quality and analyzed to see if the distal remnant length is sufficient for primary repair. The distal remnant should be of sufficient length to reach the femoral wall and the remnant should be of sufficient quality to tolerate suture passage and tensioning. The anteromedial (AM) and posterolateral (PL) bundles are identified and sutured separately. Beginning distally, sutures are passed in an alternating, interlocking Bunnell-type pattern towards the avulsed proximal end for both the AM and PL bundles. A small anterior notchplasty is then performed to improve visualization and the femoral footprint is roughened to provide bleeding and stimulate healing.
Transosseous pullout technique
With the pullout technique, small drill holes are made in the femur from the lateral femoral cortex to the insertion sites of the AM and PL bundle. Suture of the AM and PL bundle are then carried through the AM and PL drill holes, respectively, and tied over a button on the lateral femoral condyle (Fig. 4).
Transosseous button fixation. Postoperative MRI of a 22-year-old man who sustained an ACL injury while playing basketball. The patient underwent arthroscopic primary ACL repair with transosseous button fixation (pullout technique). MRI was performed 3 years after surgery. a–b Sequential sagittal proton density images showing primary ACL repair with securing of the sutures at the femoral footprint by transosseous button fixation (white arrows). c Coronal proton density image showing securing of the repair suture by a button overlying the lateral distal femoral cortex (white arrows). d Coronal proton density image showing ideal placement of the repair suture within the intercondylar notch at the femoral footprint (white arrows)
Suture anchor technique
With the suture anchor technique, holes are drilled, punched or tapped (depending on bone quality) into the femoral footprint of both the AM and PL bundle. Both bundles are then tensioned towards the proximal wall using two suture anchors (Fig. 5). This technique is similar to suture anchor repair of the rotator cuff. In patients at increased risk of re-injury such as high level athletes, patients with questionable tissue quality, or high school girls with lax knees and valgus alignment [69,70,71,72], the repair technique is reinforced with internal suture augmentation [26, 73]. With this technique, the AM suture anchor is loaded with the tape suture, which functions as a checkrein protection for ligament healing in the early phases of rehabilitation. A drill hole is made starting at the anteromedial tibial cortex and runs superiorly up into the anterior part of the ACL tibial insertion. The tape is then channeled distally along the ligament, through the drill hole, and exits at the anteromedial cortex of the tibia where it is fixed with another suture anchor (Fig. 6).
Suture anchor repair. Postoperative MRI of a 57-year old man who sustained an ACL injury while playing softball. The patient was treated with arthroscopic suture anchor primary repair and this MRI was performed 6 months postoperatively. a–c Sequential sagittal and coronal proton density weighted images showing suture anchor placement at the ACL femoral footprint (black arrow in a, white arrow in c). Notice that there are no femoral or tibial tunnels used in this procedure
Internal bracing. Postoperative MRI of a 26 year-old man who sustained an ACL injury while playing basketball. The patient was treated with arthroscopic primary ACL repair with suture anchors and additional internal bracing. Postoperative MRI was performed at 1.2-year follow-up. a–b Sequential sagittal proton density images show internal bracing with FiberTape running through (white arrow) and fixed within a tibial drill hole (black arrows). The internal bracing provides added support for the repaired ACL ligament in the early phases of rehabilitation
Postoperative rehabilitation
Postoperatively, patients are placed in a locked hinged knee brace. The brace is locked in extension until volitional quadriceps control is regained. The brace is worn for the first 4 weeks with weight bearing as tolerated while patients start range-of-motion exercises immediately after surgery. After 2 weeks, formal physical therapy is initiated. The patient is often followed with multiple clinical scales including the Lysholm, Modified Cincinnati, Tegner, SANE, IKDC, and KT-1000 [23].
Postoperative imaging
While the normal and abnormal imaging appearance after ACL reconstruction has been well described [74,75,76], there are no descriptions of the normal MRI imaging appearance after primary ACL repair in the literature. Radiologists must therefore be familiar with primary ACL repair technique to avoid MRI misinterpretation. In ACL reconstruction a femoral tunnel is drilled to secure either the bone-patella-bone or the hamstring graft. Small femoral tunnels can be used in primary ACL repair if transosseous button fixation is used to secure the sutures to the femoral footprint (Fig. 4); however, they are not required if suture anchors are used as described previously (Fig. 5). If internal bracing is performed, a tibial tunnel will be present (Fig. 6) but this tunnel will be smaller than the traditional tunnel made during ACL reconstruction.
As previously described, the suture anchors are ideally placed at the anatomic femoral insertion site of the AM and PL bundles. Following the ACL on the sagittal images can verify that the suture anchors enter at the posterior aspect of the MRI equivalent of Blumensat’s line as it intersects a line drawn along the posterior cortex of the femoral shaft (Fig. 7) [77, 78]. The suture anchors should ideally enter the femur at the anterior superior aspect of the femoral footprint [79, 80].
Heterogeneous signal within repaired ligament. Postoperative MRI of a 17 year-old boy who sustained an ACL injury while playing rugby. He was treated with arthroscopic primary ACL repair with suture anchors and a postoperative MRI was performed at 1.2-year follow up. a Sagittal proton density weighted image shows heterogeneous signal within the repaired ligament near the femoral attachment (white arrow). b Sagittal STIR image showing hyperintense signal within the repaired ligament (white arrow), which is secondary to sutures running longitudinally through the ligament and ligament healing. c Arthroscopic image at the surgery. The ACL is re-approximated to the femoral wall (white arrow) and sutures are running through the ligament (white asterisk)
Initial imaging after the procedure may show the native ligament to be heterogeneous in signal, probably secondary to the sutures running through the ligament and to postoperative edema (Fig. 7). In the early postoperative period, this appearance can simulate a tear (Fig. 8a). Increased signal can persist over time, often over a year, before the ligament becomes homogeneously low in signal similar to the normal, native ligament (Fig. 8b). It is unclear why the heterogeneous signal persists for such a long time after surgery, though it is probably due to altered ligament architecture around the sutures. It is important to note that while ACL reconstruction grafts go through a period of “ligamentization,” during which new linear high signal can be seen within the substance of the ligament as the properties of the tendon graft are altered [74, 81, 82], repaired native ligaments do not undergo this process and any new high signal within the repaired ligament should be viewed with suspicion.
Changing signal within repaired ligament. Postoperative MRI of a 50-year-old female who sustained an ACL injury while skiing. She was treated with arthroscopic primary ACL repair and MRI was performed at 1-year (a) and 5-year follow up (b). a A repaired ligament 1 year after repair shows heterogeneous signal (white arrow) which mimics a torn ligament on a sagittal proton density image. b The previous heterogeneous signal is no longer present and the ligament is now homogeneously low in signal on a sagittal proton density image (white arrow)
Re-injury will often be suspected clinically based on a history of the knee giving out or a positive pivot shift or Lachman test on physical exam. In the experience of the senior author, most re-injuries of the repaired ACL are re-ruptures in the mid-substance or proximal third of the ligament below the repair construct (Fig. 9). Imaging evaluation should include a thorough search for primary and secondary signs of ACL injury as in a native ligament tear. Further research is warranted to identify ways to best image and detect re-injury after primary ACL repair, especially given the limitations of already altered ligament signal on MRI in the early post operative period.
Ligament re-injury after primary repair. Postoperative MRI of a 16-year-old female who initially sustained an ACL injury during soccer. She returned to soccer but at 10-month follow-up, she re-ruptured her repaired ligament. a Sagittal proton density image shows no ligament fibers reaching the femoral attachment and hyperintense signal (white arrow) at the expected location of the ACL fibers. b Arthroscopic image confirms re-injury of the repaired ligament with no fibers reaching the femoral attachment. The tear was in the proximal third of the ligament and most of the fibers are retracted distally (white arrow). The internal brace was still intact (white asterisk)
Conclusion
Early results of arthroscopic primary repair of proximal ACL tears with good tissue quality are promising. As this surgery is more commonly performed, radiologists should be familiar with the potential role of tear location and tissue quality on surgical management. Radiologists should also be familiar with the expected postoperative appearance of primary ACL repair surgery to prevent mischaracterization of normal postoperative findings. Further research is necessary to identify ways to best detect ligament re-injury in the early postoperative period.
References
Sanders TL, Maradit Kremers H, Bryan AJ, et al. Incidence of anterior cruciate ligament tears and reconstruction: a 21-year population-based study. Am J Sports Med. 2016;44(6):1502–7.
Hawkins RJ, Misamore GW, Merritt TR. Followup of the acute nonoperated isolated anterior cruciate ligament tear. Am J Sports Med. 1984;14(3):205–10.
Sanders TL, Pareek A, Kremers HM, et al. Long-term follow-up of isolated ACL tears treated without ligament reconstruction. Knee Surgery, Sport Traumatol Arthrosc. 2016:1–8.
Kruse LM, Gray B, Wright RW. Rehabilitation after anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(19):1737–48.
Finsterbush A, Frankl U, Matan Y, Mann G. Secondary damage to the knee after isolated injury of the anterior cruciate ligament. Am J Sports Med. 1990;18(5):475–9.
Davarinos N, O’Neill BJ, Curtin W. A brief history of anterior cruciate ligament reconstruction. Adv Orthop Surg. 2014;2014:1–6.
Murrell G, Maddali S, Horovitz L, Oakley SP, Warren RF. The effects of time course after anterior cruciate ligament injury in correlation with meniscal and cartilage loss. Am J Sports Med. 2001;29(1):9–14.
Robson A. Ruptured cruciate ligaments and their repair by operation. Ann Surg. 1903;37(5):716–8.
Warren RF. Primary repair of the anterior cruciate ligament. Clin Orthop Relat Res. 1983;172:65–70.
Marshall JL, Warren RF, Wickiewicz TL. Primary surgical treatment of anterior cruciate ligament lesions. Am J Sports Med. 1982;10(2):103–7.
Sherman MF, Bonamo JR. Primary repair of the anterior cruciate ligament. Clin Sports Med. 1988;7(4):739–50.
Feagin JAJ, Curl WW. Isolated tear of the anterior cruciate ligament: 5-year follow-up study. Am J Sports Med. 1976;4(3):95–100.
Sherman MF, Lieber L, Bonamo JR, Podesta L, Reiter I. The long-term followup of primary anterior cruciate ligament repair. Defining a rationale for augmentation. Am J Sports Med. 1991;19(3):243–55.
Engebretsen L, Benum P, Sundalsvoll S. Primary suture of the anterior cruciate ligament: a 6-year follow-up of 74 cases. Acta Orthop Scand. 1989;60(5):561–4.
Harilainen A, Myllynen P. Treatment of fresh tears of the anterior cruciate ligament: a comparison of primary suture and augmentation with carbon fibre. Injury. 1987;18(6):396–400.
Jones KG. Results of use of the central one-third of the patellar ligament to compensate for anterior cruciate ligament deficiency. Clin Orthop Relat Res. 1980;147:39–44.
Hefti F, Gachter A, Jenny H, Morscher E. Replacement of the anterior cruciate ligament: a comparative study of four different methods of reconstruction. Arch Orthop Trauma Surg. 1982;100(2):83–94.
van der List JP, DiFelice GS. Role of tear location on outcomes of open primary repair of the anterior cruciate ligament: a systematic review of historical studies. Knee. 2017;24(5):898–908.
van der List JP, DiFelice GS. Preservation of the anterior cruciate ligament: a treatment algorithm based on tear location and tissue quality. Am J Orthop (Belle Mead NJ). 2016;45(7):E393–405.
Taylor SA, Khair MM, Roberts TR, DiFelice GS. Primary repair of the anterior cruciate ligament: a systematic review. Arthroscopy. 2015;31(11):2233–47.
van Eck CF, Limpisvasti O, ElAttrache NS. Is there a role for internal bracing and repair of the anterior cruciate ligament? A systematic literature review. Am J Sports Med [Internet]. 2017. https://doi.org/10.1177/0363546517717956
van der List JP, DiFelice GS. Primary repair of the anterior cruciate ligament: a paradigm shift. Surgeon. 2017;15(3):161–8.
DiFelice GS, Villegas C, Taylor S. Anterior cruciate ligament preservation: early results of a novel arthroscopic technique for suture anchor primary anterior cruciate ligament repair. Arthroscopy. 2015;31(11):2162–71.
van der List JP, Mintz DN, DiFelice GS. The location of anterior cruciate ligament tears: a prevalence study using magnetic resonance imaging. Orthop J Sport Med. 2017;5(6). https://doi.org/10.1177/2325967117709966.
van der List JP, DiFelice GS. Preservation of the anterior cruciate ligament: surgical techniques. Am J Orthop (Belle Mead NJ). 2016;45(7):E406–14.
Anthony IC, Mackay G. Anterior cruciate ligament repair revisited. Preliminary results of primary repair with internal brace ligament augmentation: a case series. Orthop Muscular Syst. 2015;4(2). https://doi.org/10.4172/2161-0533.1000188.
DiFelice GS, van der List JP. Arthroscopic primary repair of proximal anterior cruciate ligament tears. Arthrosc Tech. 2016;5(5):e1057–61.
van der List J, DiFelice G. Arthroscopic primary repair of proximal anterior Cruciate ligament tears: no deterioration at mid-term follow-up. Arthrosc J Arthrosc Relat Surg [Internet]. 2017;33(6):e7. https://doi.org/10.1016/j.arthro.2017.04.038
Tibor L, Chan PH, Funahashi TT, Wyatt R, Maletis GB, Inacio MCS. Surgical technique trends in primary ACL reconstruction from 2007 to 2014. J Bone Joint Surg Am. 2016;98(13):1079–89.
Samuelsen BT, Webster KE, Johnson NR, Hewett TE, Krych AJ. Hamstring autograft versus patellar tendon autograft for ACL reconstruction: is there a difference in graft failure rate? a meta-analysis of 47,613 patients. Clin Orthop Relat Res. 2017;475(10):2459–68.
Gabler CM, Jacobs CA, Howard JS, Mattacola CG, Johnson DL. Comparison of graft failure rate between autografts placed via an anatomic anterior cruciate ligament reconstruction technique: a systematic review, meta-analysis, and meta-regression. Am J Sports Med. 2016;44(4):1069–79.
Irarrázaval S, Kurosaka M, Cohen M, Fu FH. Anterior cruciate ligament reconstruction. J ISAKOS Jt Disord Orthop Sport Med. 2016;1(1):38–52.
van Eck CF, Lesniak BP, Schreiber VM, Fu FH. Anatomic single- and double-bundle anterior cruciate ligament reconstruction flowchart. Arthroscopy. 2010;26(2):258–68.
Sajovic M, Vengust V, Komadina R, Tavcar R, Skaza K. A prospective, randomized comparison of semitendinosus and gracilis tendon versus patellar tendon autografts for anterior cruciate ligament reconstruction: five-year follow-up. Am J Sports Med. 2006;34(12):1933–40.
Beaufils P, Gaudot F, Drain O, Boisrenoult P, Pujol N. Mini-invasive technique for bone patellar tendon bone harvesting: its superiority in reducing anterior knee pain following ACL reconstruction. Curr Rev Musculoskelet Med. 2011;4(2):45–51.
Kartus J, Magnusson L, Stener S, Brandsson S, Eriksson BI, Karlsson J. Complications following arthroscopic anterior cruciate ligament reconstruction: a 2–5-year follow-up of 604 patients with special emphasis on anterior knee pain. Knee Surgery, Sport Traumatol Arthrosc. 1999;7(1):2–8.
Wittstein JR, Wilson JB, Moorman CT III. Complications related to hamstring tendon harvest. Oper Tech Sports Med. 2017;14(1):15–9.
Kaeding CC, Aros B, Pedroza A, Pifel E, Amendola A, Andrish JT, et al. Allograft versus autograft anterior cruciate ligament reconstruction: predictors of failure from a MOON prospective longitudinal cohort. Sports Health. 2010;3(1):73–81.
Kaiser J, Vignos MF, Liu F, Kijowski R, Thelen DG. MRI assessments of cartilage mechanics, morphology and composition following reconstruction of the anterior cruciate ligament. Clin Biomech. 2016;34:38–44.
Imhauser C, Mauro C, Choi D, et al. Abnormal tibiofemoral contact stress and its association with altered kinematics after center-center anterior cruciate ligament reconstruction: an in vitro study. Am J Sports Med. 2013;41(4):815–25.
Simon D, Mascarenhas R, Saltzman BM, Rollins M, Bach BR, MacDonald P. The relationship between anterior cruciate ligament injury and osteoarthritis of the knee. Adv Orthop [Internet]. 2015. https://doi.org/10.1155/2015/928301.
Song E-K, Seon J-K, Yim J-H, Woo S-H, Seo H-Y, Lee K-B. Progression of osteoarthritis after double- and single-bundle anterior cruciate ligament reconstruction. Am J Sports Med. 2013;41(10):1–7.
Cheatham SA, Johnson DL. Anticipating problems unique to revision ACL surgery. Sports Med Arthrosc. 2013;21(2):129–34.
Maak TG, Voos JE, Wickiewicz TL, Warren RF. Tunnel widening in revision anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2010;18(11):695–706.
Kamath GV, Redfern JC, Greis PE, Burks RT. Revision anterior cruciate ligament reconstruction. Am J Sports Med. 2010;39(1):199–217.
Wright RW, Gill CS, Chen L, et al. Outcome of revision anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(6):531–6.
Andriolo L, Filardo G, Kon E, et al. Revision anterior cruciate ligament reconstruction: clinical outcome and evidence for return to sport. Knee Surg Sport Traumatol Arthrosc. 2015;23(10):2825–45.
Grassi A, Ardern CL, Muccioli G, Neri MP, Marcacci M, Zaffagnini S. Does revision ACL reconstruction measure up to primary surgery ? A meta-analysis comparing outcomes and radiographic results. Br J Sports Med. 2016;50:716–24.
Fleming BC, Carey JL, Spindler KP, Murray MM. Can suture repair of ACL transection restore normal anteroposterior laxity of the knee? An ex vivo study. J Orthop Res. 2008;26(11):1500–5.
Murray MM, Fleming BC. Use of a bioactive scaffold to stimulate anterior cruciate ligament healing also minimizes posttraumatic osteoarthritis after surgery. Am J Sports Med. 2013;41(8):1762–70.
Murray MM. Current status and potential for primary ACL repair. Clin Sports Med. 2009;28(1):51–61.
Gao F, Zhou J, He C, et al. A morphologic and quantitative study of mechanoreceptors in the remnant stump of the human anterior cruciate ligament. Arthroscopy. 2016;32(2):273–80.
van der List JP, DiFelice GS. Range of motion and complications following primary repair versus reconstruction of the anterior cruciate ligament. Knee. 2017;24(4):798–807.
Boden BP, Dean GS, Feagin JAJ, Garrett WEJ. Mechanisms of anterior cruciate ligament injury. Orthopedics. 2000;23(6):573–8.
Heard WMR, VanSice WC, Savoie FH 3rd. Anterior cruciate ligament tears for the primary care sports physician: what to know on the field and in the office. Phys Sports Med. 2015;43(4):432–9.
Spindler KP, Wright RW. Anterior cruciate ligament tear. N Engl J Med. 2008;359(20):2135–42.
Huang W, Zhang Y, Yao Z, Ma L. Clinical examination of anterior cruciate ligament rupture: a systematic review and meta-analysis. Acta Orthop Traumatol Turc. 2016;50(1):22–31.
Smith C, McGarvey C, Harb Z, et al. Diagnostic efficacy of 3-T MRI for knee injuries using arthroscopy as a reference standard: a meta-analysis. Am J Roentgenol. 2016;207(2):369–77.
van der List JP, DiFelice GS. The role of ligament repair in anterior cruciate ligament surgery. In: Mascarenhas R, Bhatia S, Lowe WR, editors. Ligamentous injuries of the knee, 1st ed. Houston: Nova Science Publishing; 2016. p. 199–220.
van der List JP, DiFelice GS. Preoperative magnetic resonance imaging predicts eligibility for arthroscopic primary anterior cruciate ligament repair. Knee Surg Sport Traumatol Arthrosc. 2017. https://doi.org/10.1007/s00167-017-4646-z.
Pereira ER, Ryu KN, Ahn JM, Kayser F, Bielecki D, Resnick D. Evaluation of the anterior cruciate ligament of the knee: comparison between partial flexion true sagittal and extension sagittal oblique positions during MR imaging. Clin Radiol. 1998;53(8):574–8.
Ng WHA, Griffith JF, Hung EHY, Paunipagar B, Law BKY, Yung PSH. Imaging of the anterior cruciate ligament. World J Orthop. 2011;2(8):75–84.
Van Dyck P, Vanhoenacker FM, Gielen JL, et al. Three tesla magnetic resonance imaging of the anterior cruciate ligament of the knee: can we differentiate complete from partial tears? Skelet Radiol. 2011;40(6):701–7.
Van Dyck P, Vanhoenacker FM, Lambrecht V, et al. Prospective comparison of 1.5 and 3.0-T MRI for evaluating the knee menisci and ACL. J Bone Joint Surg Am. 2013;95(10):916–24.
Van Dyck P, de Smet E, Veryser J, et al. Partial tear of the anterior cruciate ligament of the knee: injury patterns on MR imaging. Knee Surg Sport Traumatol Arthrosc. 2012;20(2):256–61.
Kosaka M, Nakase J, Toratani T, et al. Oblique coronal and oblique sagittal MRI for diagnosis of anterior cruciate ligament tears and evaluation of anterior cruciate ligament remnant tissue. Knee. 2017;21(1):54–7.
Crain EH, Fithian DC, Paxton EW, Luetzow WF. Variation in anterior cruciate ligament scar pattern: does the scar pattern affect anterior laxity in anterior cruciate ligament-deficient knees? Arthroscopy. 2005;21(1):19–24.
van der List JP, DiFelice GS. Successful arthroscopic primary repair of a chronic anterior cruciate ligament tear 11 years following injury. HSS J. 2017;13(1):90–5.
Wiggins AJ, Grandhi RK, Schneider DK, Stanfield D, Webster KE, Myer GD. Risk of secondary injury in younger athletes after anterior cruciate ligament reconstruction: a systematic review and meta-analysis. Am J Sports Med. 2016;44(7):1861–76.
Webster KE, Feller JA. Exploring the high reinjury rate in younger patients undergoing anterior cruciate ligament reconstruction. Am J Sports Med. 2016;44(11):2827–32.
Allen MM, Pareek A, Krych AJ, et al. Are female soccer players at an increased risk of second anterior cruciate ligament injury compared with their athletic peers? Am J Sports Med. 2016;44(10):2492–8.
Larruskain J, Lekue JA, Diaz N, Odriozola A, Gil SM. A comparison of injuries in elite male and female football players: a 5-season prospective study. Scand J Med Sci Sports. 2017. https://doi.org/10.1111/sms.12860.
Mackay GM, Blyth MJG, Anthony I, Hopper GP, Ribbans WJ. A review of ligament augmentation with the internal brace. Surg Technol Int. 2015;26:239–55.
Bencardino JT, Beltran J, Feldman MI, Rose DJ. MR imaging of complications of anterior cruciate ligament graft reconstruction. Radiographics. 2009;29(7):2115–26.
Casagranda BC, Maxwell NJ, Kavanagh EC, Towers JD, Shen W, Fu FH. Normal appearance and complications of double-bundle and selective-bundle anterior cruciate ligament reconstructions using optimal MRI techniques. Am J Roentgenol. 2009;192(5):1407–15.
Meyers AB, Haims AH, Menn K, Moukaddam H. Imaging of anterior cruciate ligament repair and its complications. Am J Roentgenol. 2010;194(2):476–84.
Ferretti M, Ekdahl M, Shen W, Fu FH. Osseous landmarks of the femoral attachment of the anterior cruciate ligament: an anatomic study. Arthroscopy. 2007;23(11):1218–25.
Edwards A, Bull AMJ, Amis AA. The attachments of the anteromedial and posterolateral fibre bundles of the anterior cruciate ligament : part 2: femoral attachment. Knee Surg Sport Traumatol Arthrosc. 2008;16(1):29–36.
Śmigielski R, Zdanowicz U, Drwięga M, Ciszek B, Williams A. The anatomy of the anterior cruciate ligament and its relevance to the technique of reconstruction. Bone Joint J. 2016;98–B(8):1020–6.
Nawabi DH, Tucker S, Schafer KA, et al. ACL fibers near the lateral intercondylar ridge are the most load bearing during stability examinations and isometric through passive flexion. Am J Sports Med. 2016;44(10):1–9.
Claes S, Verdonk P, Forsyth R, Bellemans J. The “ligamentization” process in anterior cruciate ligament reconstruction: what happens to the human graft? A systematic review of the literature. Am J Sports Med. 2011;39(11):2476–83.
Pauzenberger L, Syré S, Schurz M. “Ligamentization” in hamstring tendon grafts after anterior cruciate ligament reconstruction: a systematic review of the literature and a glimpse into the future. Arthroscopy. 2013;29(10):1712–21.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The senior author is a paid consultant for Arthrex and receives research support from Arthrex.
The other authors have no disclosures.
Additional information
Dr. DiFelice is a paid consultant for Arthrex and receives research support from Arthrex.
Rights and permissions
About this article
Cite this article
Daniels, S.P., van der List, J.P., Kazam, J.J. et al. Arthroscopic primary repair of the anterior cruciate ligament: what the radiologist needs to know. Skeletal Radiol 47, 619–629 (2018). https://doi.org/10.1007/s00256-017-2857-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-017-2857-5