Abstract
Purposeof Review
In this review, we will summarize the effects of these perioperative anesthetics and anesthetic interventions on the immune system and tumorigenesis as well as address the related clinical evidence on cancer-related mortality and recurrence.
Recent Findings
Cancer remains a leading cause of morbidity and mortality worldwide. For many solid tumors, surgery is one of the major therapies. Unfortunately, surgery promotes angiogenesis, shedding of circulating cancer cells, and suppresses immunity. Hence, the perioperative period has a close relationship with cancer metastases or recurrence. In the perioperative period, patients require multiple anesthetic management including anesthetics, anesthetic techniques, and body temperature control. Preclinical and retrospective studies have found that these anesthetic agents and interventions have complex effects on cancer outcomes. Therefore, well-planned, prospective, randomized controlled trials are required to explore the effects of different anesthetics and techniques on long-term outcomes after cancer surgery.
Summary
Due to the conflicting effects of anesthetic management on cancer recurrence, further preclinical and clinical trials are required and beneficial to the development of systemic cancer therapies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cancer is a major contributor to global morbidity and mortality despite advances in prevention, diagnosis, and treatment. According to the World Health Organization and the American Medical Association, cancer is becoming a major global health concern since it is the second leading cause of death after cardiovascular disease, resulting in 10 million deaths in 2020 [1], and the overall incidence of new cancer cases is almost 19.3 million worldwide [2]. Moreover, many patients experienced delayed diagnosis and treatment (including surgery) because of the unprecedented impacts of the COVID-19 pandemic on public health care systems. These delays may have a significant effect on cancer prognosis.
Despite advances in chemotherapy, radiotherapy, and immunotherapy, surgery (under anesthesia) is still an important component of modern cancer treatment that helps millions of people live healthier, more productive lives. However, the relationship between anesthesia and cancer recurrence remains a controversial issue because surgical stress and intraoperative anesthesia can impair the host immune system [3]. A number of theories explain how the surgical procedure may promote cancer recurrence after excision and the adverse impact of surgical stress on the body’s innate tumor defense mechanisms [4, 5] and the inadvertent seeding of tumor cells during the perioperative period [6, 7••, 8]. Anesthetics can also suppress the immune response by directly affecting cell-mediated immunity (CMI) or by activating the hypothalamic–pituitary–adrenal axis (HPA) and the sympathetic nervous system (SNS) [9]. The impairment of CMI may reactivate micrometastases that are already disseminated at the time of surgery [10], increasing the frequencies of cancer recurrence and distant metastasis. The stimulation of the HPA induces the release of neuroendocrine mediators, such as catecholamines, prostaglandin E2 (PGE2), cytokines, and cortisol, which in turn attenuate the activity of immune cells [11•]. Other neuroendocrine mediators, such as IL-6 and matrix metalloproteinases (MMPs), are also secreted and play critical roles in the regulation of tumor growth and angiogenesis [10].
The role of anesthesia in cancer recurrence has received considerable attention in recent decades, and there have been a number of retrospective studies and much preclinical research into this topic. An increasing number of preclinical findings suggest that general anesthetics have the potential to influence critical hallmarks of cancer that are involved in tumorigenesis and metastasis [12]. For example, intravenous anesthetics, such as propofol, and inhalational volatile anesthetics, such as sevoflurane, are two major classes of general anesthetics often utilized in clinical practice. Sevoflurane has been demonstrated to promote proliferation, migration, invasion, and angiogenesis in a variety of cancer cell types, whereas propofol may antagonize these same pathways [13,14,15]. Several retrospective studies have demonstrated an association between inhalational anesthesia and lower rates of recurrence-free survival in cancer patients who undergo elective surgery than in those who receive propofol-based anesthesia [16, 17]. Additionally, perioperative anesthetic management, including different anesthetic techniques and body temperature management, also has a close relationship with cancer prognosis [18•, 19••]. Based on current preclinical findings, every component of these procedures has a possible link to the development of immune dysfunctions that influence cancer metastasis and recurrence. However, only some small-scale retrospective studies have investigated the association between perioperative anesthetic management and cancer recurrence, which highlights the need for further prospective and randomized controlled trials (RCTs).
With the aim of optimizing the current clinical strategy and unraveling the controversial findings on exactly how surgery/anesthetics/anesthetic intervention-induced immunosuppression leads to an increase in cancer-related recurrence during the perioperative period, we provide a comprehensive review of the important role of the perioperative period in cancer treatment and the profound influence of anesthesia on cancer recurrence.
Effects of the Immune System on Cancer Recurrence
To understand the effects of surgery and anesthetics on cancer recurrence, the first step is to understand the basics of cancer biology. The microenvironment of tumors consists of cancer cells, different kinds of inflammatory cells, and inflammatory mediators. Both tumor and inflammatory cells secrete a complex array of chemical and protein signaling molecules that promote cancer development and metastasis in both an autocrine and paracrine manner. It should be emphasized that the immune cells that have been recruited may not have the typical protective response that leads to tumor cell eradication. The secretion of proinflammatory cytokines by these immune cells and the tumor itself may shift the balance in favor of tumor progression [20]. As a result, a tumor can be viewed as an organism that establishes its own complex physical and chemical connections with the host immune system. The majority of the molecular and cellular processes involved in this process remain to be fully elucidated.
Both the regression and development of cancer cells are influenced by the immune system. The innate and adaptive systems form the functional and effective immune system. Epithelial barriers, granulocytes, macrophages, natural killer (NK) cells, and dendritic cells make up the innate immune system, which is the first-line defense against invading organisms and tumor cells. The adaptive immune system serves to eliminate threats that have evaded the innate immune system. It comprises humoral immunity, antibody-mediated response, and CMI [21]. CMI, which is mediated by numerous kinds of T cells, is the principal defense against tumor cell invasion among these three components of immunity. Activated T cells are categorized by the cytokines that they secrete. The cytokines released by T-helper (Th) 1 cells promote inflammation, stimulate B cells, activate macrophages, and promote the development of cytotoxic T cells. IgE production and eosinophil are both stimulated by the cytokines that are secreted by Th2 cells [22]. Another important proinflammatory cytokine is IL-1, which is generated mostly by monocytes and macrophages. It is also a key regulator of inflammation and immune responses [23].
It has long been recognized that cancer can occur at sites of inflammation and injury caused by surgery, and it is known that inflammatory cells and mediators play a key role in cancer recurrence. Inflammatory cells, including leukocytes (neutrophils, monocytes, eosinophils, and basophils) and lymphocytes (T cells, B cells, and NK cells), are present early in the neoplastic process and play an important role in the tumor microenvironment by secreting an array of cytokines and chemokines, including IL-6, TNF-α, IL-1β, and PGE2. During the “elimination phase,” when a cancer-free state should be achieved, they are the essential players in recognizing and eliminating cancer cells [24••]. If tumor cells survive after the “elimination phase,” they may enter an “equilibrium” state, in which the host’s adaptive immune response keeps them dormant and prevents further tumor progression. In the final stage, called the “escape phase,” tumor cells exhibit apparent clinical growth as they evade the control of the host’s immunity and its capacity to create an immunosuppressive state by producing various cytokines, such as vascular endothelial growth factor (VEGF) and transforming growth factor-β (TGF-β) [25]. Moreover, DNA damage and somatic mutations may lead to cancer formation, which is a process that has been termed “initiation”; this process can persist in cells indefinitely until a second injury or “trigger” occurs. Inflammation, injury, irritants, or a host of other exposures may all act as “triggers.” This kind of “trigger”-related process results in the recruitment of inflammatory cells and finally constitutes a positive feedback loop, which leads to decreased cellular proliferation [26•].
The Perioperative Period as a Critical Window for Cancer Recurrence
The perioperative period is divided into three phases: the preoperative period (a few preoperative hours), the intraoperative period, and the postoperative period (several days after surgery). During the intraoperative period, general anesthesia consists of the administration of intravenous anesthetics (e.g., thiopental or propofol) for induction, followed by muscle relaxants and endotracheal intubation, then volatile anesthetics (e.g., sevoflurane) and opioids, which are applied for maintenance and pain control. The perioperative period triggers significant physiologic disruptions because it is a time of maximum vulnerability for patients with cancer. In the setting of perioperative inflammation, immunosuppression, and increased concentrations of catecholamines and angiogenesis, the seeding of circulating cancer cells in distant organs, as well as the growth of dormant tumors and micrometastasis, can be facilitated [27].
Given the critical role of the perioperative period of surgery, it is necessary to clarify its impact on cancer recurrence. It was first hypothesized a century ago that surgery promotes local recurrence and the distant spread of cancer [28]. Several theories have been proposed to explain this phenomenon, the most notable of which involve minimal levels of residual disease, the dissemination of tumor cells at the time of surgery, and, possibly, a switch from tumor dormancy to proliferation [29, 30]. Recent studies have investigated the possible influence of the metabolic, neuroendocrine, inflammatory, and immunological changes that occur perioperatively and are connected to or induced by anesthesia. Indeed, there is some evidence that anesthetics and other perioperative factors have the potential to affect long-term outcomes after cancer surgery [31]. According to these findings, general anesthetics, except propofol, can impair various immune functions of macrophages, dendritic cells, T lymphocytes, and NK cells [32]. Additionally, although opioid analgesia is the foundation of cancer pain relief, it can also promote tumor growth by inhibiting immune function and increasing angiogenesis [33]. As a result, opioid-sparing analgesia may be used to maintain the normal function of NK cells and reduce the metastatic spread of cancer [34]. As regional anesthesia (RA)/analgesia can block both afferent neural transmissions from the central nervous system and the descending efferent signals, it may attenuate surgery-stimulated adverse effects by minimizing the neuroendocrine stress response induced by SNS activation. Furthermore, regional analgesia inhibits the release of endogenous opioids; thus, the occurrence of opioid-induced immune suppression may be reduced [35]. With the combination of regional and general anesthesia (GA)/analgesia, the amount of general anesthetics required can be significantly reduced, which is probably accompanied by reduced immunosuppression as well as lower requirements for the use of opioids for postoperative pain relief [3]. However, further RCTs are required to substantially verify this theory.
Mechanisms of Cancer Recurrence in the Perioperative Period
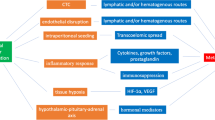
The mechanisms underlying postsurgical cancer recurrence are complicated and poorly understood. Following the intended curative surgical resection of a primary tumor, cancer may recur at several sites due to a variety of mechanisms [18•]. There are four possible mechanisms that can cause cancer recurrence. The first is local recurrence at the tumor resection site, which occurs due to the proliferation of residual cells. The second is lymph node metastasis caused by the release of tumor cells into the lymphatic system before or during the procedure. The third method involves the seeding of distant organs by circulating tumor cells (CTCs) released before or during the procedure. The last involves seeding within a body cavity (e.g., peritoneal spread) [36].
The risk that individual cancer cells will seed in tissue and that the cancer will progress to a clinically significant metastatic disease is partly influenced by intraoperative dissemination, potentially by anesthesia and other perioperative events [37]. To survive in the hostile circulatory system by avoiding detection and elimination by marginalized leukocytes, few CTCs exist in the “slow circulation points” of the pulmonary and hepatic capillaries so that a complex tissue microenvironment involving the interrelations of surrounding noncancerous stromal cells, immune system cells, extracellular matrix, chemokines, cytokines, and myriad other factors may be established [38, 39]. Once immune escape is accomplished, inflammatory mediators can boost the efficiency of local colonization by aiding in the destruction of the endothelial glycocalyx, combined with endothelial denudation resulting in the formation of a premetastatic niche [40]. This precursor state comprises clusters of bone marrow-derived cells that populate and precondition the extracellular environment, allowing subsequent CTC infiltration and colony expansion [41]. Hypoxic environments established at the surgical resection sites, as well as the activation of platelets that release chemokines that attract bone marrow-derived cells, may also aid in the development of the premetastatic niche [42]. These pathological changes can drive the process known as “epithelial-to-mesenchymal transition,” whereby epithelial cancer cells develop a mesenchymal phenotype facilitating cellular motility and thus triggering angiogenic and metastatic cascades [43]. Therefore, dissemination, immune escape, and angiogenesis form an entire pathological process that finally induces cancer recurrence. We will further discuss each process below.
Intraoperative Dissemination: a Trigger for Cancer Recurrence
The intraoperative dissemination of tumor cells can occur through the lymphatic, hematogenous, and/or transcoelomic (a route of tumor metastasis across a body cavity or organ surface including the pleural or peritoneal surfaces) routes. CTCs are detectable in the majority of patients with solid tumors [44], and an increased number of CTCs has been confirmed to be linked with a poor prognosis in patients with a variety of tumor types [45, 46]. In particular, it has been demonstrated that CTC numbers increase following surgery for breast [47], lung [48], and colorectal [49] cancers. Thus, although the evidence that high numbers of CTCs correlate with inferior patient outcomes across all tumor types is currently inconclusive [50], there are reasonable concerns that tumor cell release after surgery leads to metastatic colonization.
Even after the removal of the primary tumor, disseminated cancer cells may survive and retain the ability to invade via lymphatic vessels because of surgical disruption, which has been observed by real-time fluorescence imaging [51]. For example, the number of CTCs was detected to increase fourfold on average in the sentinel lymph nodes of a cohort of 414 patients following breast cancer surgery [52]. By analyzing mouse models and tissue samples from patients with pancreatic ductal adenocarcinoma, it was shown that dormant disseminated cancer cells that lacked a cell surface molecule that elicits T-cell-mediated attacks account for both quiescence and resistance to immune elimination since they are unable to relieve endoplasmic reticulum stress [53•]. Accordingly, the outgrowth of disseminated cells into macrometastases requires not only high levels of surgical stress but also the suppression of systemic immunity [53•]. Additionally, the dissemination of tumor cells can be induced directly by surgical procedures. For example, the use of laparoscopic ports can result in port-site recurrences that may cause intra-abdominal spread. This phenomenon has been reported following surgery for gastrointestinal [41], gynecological [54], urological [55], and thoracic [56] malignancies and, alarmingly, was reported to occur in more than 10% of patients following the resection of incidentally diagnosed gall bladder cancer [57].
Anesthetics used during surgery also have a potential role in promoting metastatic dissemination. In murine models of breast cancer, sevoflurane led to significantly more lung metastasis than propofol [58]. Interestingly, sevoflurane can increase IL-6 levels, which in turn leads to the activation of signal transducer and activator of transcription (STAT)-3 as well as the subsequent infiltration of myeloid cells into the lung [58]. According to this finding, anesthetics can promote cancer metastasis by altering the tumor microenvironment through cytokines [59].
Immune Escape: a Maladaptive Immune Response for Cancer Recurrence
A number of studies in both preclinical models and patient samples have converged on the theme that primary cancers and metastases apply a range of strategies to avoid detection and destruction by the immune system, and the majority of which are activated in the aftermath of surgery. Tissue damage is usually accompanied by localized inflammation, hypoxia, and acidosis, which can also influence infiltrating immune cells by promoting the activity of protumor M2-like macrophages and by suppressing antitumor immune responses [60] under the influence of inflammatory mediators, such as PGE2. PGE2 is a lipid mediator, and its action is mediated by PGE2 receptors (EP1-4). Both EP2 and EP4 are Gs-coupled receptors that signal through adenylate cyclase-dependent cAMP/PKA/CREB pathways [61]. The effects of PGE2 related to immunosuppression include the inhibition of neutrophil, NK, and T-cell mitogenesis [62]. Tumor cells can shed cell surface ligands to evade recognition by immune cells, including NK cells, with the consequent impairment of NK-cell-mediated cytolytic activity [63]. Such mechanisms reveal the temporary development of a tumor-promoting milieu surrounding the surgical wound or at sites of micrometastases that might increase the risk of recurrence [64, 65].
Following surgery, there is a protracted period of immunosuppression. This counterbalancing phenomenon, referred to as the resolution phase of inflammation, has evolved to contain the intensity of the acute inflammatory response but might also contribute to perioperative vulnerability to cancer recurrence. Accordingly, a compensatory anti-inflammatory response aroused by the surgical response can also lead to the dysregulation of CMI with subsequent immunosuppression [66]. Furthermore, prostaglandin signaling regulates lymphatic vessel dilatation and, therefore, may enable cancer metastasis [67]. This cytokine-mediated imbalance results in a shift toward the protumoral Th2 profile, which favors tumor growth by disrupting CMI [68].
The secretion of cortisol and catecholamines is also a major trigger caused by surgical stress and anesthetics [69]. The action of cortisol is to diffuse through the cellular membrane to bind the intracellular glucocorticoid receptor. This complex translocates into the cell nucleus, where it interacts with glucocorticoid-responsive elements to transcribe different factors, such as NF-κB, which enable cortisol to inhibit or promote the production of inflammatory cytokines directly [70]. β-adrenoreceptors have been found in breast, prostate, lung, esophageal, and liver cancer cells [71,72,73,74,75]. Once epinephrine or norepinephrine activates β-adrenergic signaling, the intracellular concentration of cyclic adenosine monophosphate (cAMP) is increased to directly modulate cancer cell growth, proliferation, invasiveness, angiogenesis, and metastasis [76]. One critical characteristic of cancer cells is the formation of invadopodia, which are used to degrade and facilitate migration through the extracellular matrix [77]. The activation of β-adrenoreceptors promotes an increase in the number of invadopodia, which correlates with enhanced tumor invasion in breast cancer models [78]. Hence, minimizing surgical stress and limiting subsequent immunosuppression might reduce patient vulnerability and hamper cancer metastasis after surgery.
Epithelial-to-Mesenchymal Transition: a Major Cellular Mechanism of Cancer Recurrence
It has been shown that metastatic cancer cells migrate individually both in vivo and in vitro [79]. In humans, it is believed that seeding requires the joint action of a cluster of tumor cells moving together [80], which involves epithelial-mesenchymal transition (EMT). EMT is the transdifferentiation process through which transformed epithelial cells obtain the capacity to invade, resist stress, and disseminate [81]. Specifically, cancer cell clusters can retain and require epithelial gene expression and can transition between distinct epithelial differentiation states to accomplish the proliferative versus migratory components of metastasis. The transition from one state to another is governed by several kinds of growth factors [82] and signaling pathways [83]. Spontaneous EMT in primary tumor cells shifts among different intermediate stages with different invasive, metastatic, and differentiation characteristics [84]. Tumor cells with a combination of epithelial and mesenchymal phenotypes are more effective in circulation and colonization at the secondary site, which favors the development of metastasis [84].
The initiation of the EMT process is considered responsive to specific environmental stimuli during cancer surgery [85]. Moreover, recent studies have implicated that using anesthetics can induce EMT and facilitate tumor metastasis during the perioperative period. Ischemia/reperfusion injury (IRI) often occurs during surgeries involving hepatocellular carcinoma (HCC) and liver transplantation [86, 87]. Hypoxia and inflammation can upregulate lipocalin2 (LCN2) levels to induce EMT in many cancers, which promotes tumor cell survival, proliferation, and metastasis [88, 89]. Latent EMT programs can be activated through the crosstalk that occurs between various immune cells that accumulate during the surgery and neighboring carcinoma cells. For example, CD8 + cytotoxic T cells are typically involved in the immunosurveillance and immunoediting of carcinomas. Moreover, pancreatic ductal epithelial cells lose the expression of E-cadherin following coculture with activated T cells in vitro, resulting in a spindle-shaped mesenchymal morphology accompanied by the expression of vimentin and ZEB1 [90]. In cancer patients, EMT programs are important for tissue regeneration and repair during wound healing after surgery. With the help of EMT, nonmotile epithelial cells are able to migrate across a wound site, proliferate, and then revert to the epithelial state to restore the integrity of the epithelial barrier as part of a process known as “re-epithelialization” [91, 92]. However, when the deregulated activity of EMT is induced by the overexpression of SLUG in human keratinocytes in vitro, it can cause the increased levels of desmosomal disruption and tumor spreading that are typically observed during local recurrence [93]. Regarding anesthetics, propofol was found to normalize EMT by inhibiting SLUG expression, resulting in an increase in the occurrence of apoptosis and a reduction in the growth and invasion of pancreatic cancer cells [94•].
The Perioperative Effects of Anesthetics on Cancer Recurrence
Inhalational Anesthetics
Volatile anesthetics are widely used during oncological surgery. Preclinical research suggests that volatile agents can promote cancer progression by direct and indirect mechanisms. First, volatile anesthetics can directly modify intracellular signals involved in key aspects of cancer cell behavior, including proliferation, invasion, migration, and sensitivity to chemotherapeutic agents. For example, isoflurane at 1.2% has been found to promote proliferation and migration while reducing levels of apoptosis in glioblastoma stem cells by promoting the overexpression of hypoxia-inducible factor (HIF) [95••, 96]. In non-small cell lung cancer, isoflurane (at 1%, 2%, and 3%) promoted proliferation in a concentration-dependent manner and invasion and invasiveness via Akt-mTOR signaling [97]. Desflurane at 10.3% induces EMT and metastasis through dysregulation of a well-known tumor suppressor called the miR-34/LOXL3 axis in a colorectal cancer cell line [98]. Using 2% sevoflurane for 6 h in vitro can increase the survival of breast cancer cells via modulation of intracellular Ca2 + homeostasis [99]. Second, sevoflurane can suppress CMI, which indirectly promotes tumor cell proliferation and angiogenesis. Sevoflurane, isoflurane, and desflurane induce the apoptosis of T lymphocytes and upregulate the expression of HIF-1α both in vitro and in vivo [100, 101]. It has also been shown that sevoflurane increases the levels of MMP-3 and MMP-9 in patients undergoing breast cancer surgery [102]. Third, inhalation anesthetics may cause distant metastasis by the activation of the HPA axis and the SNS by releasing neuroendocrine mediators, such as cortisol, catecholamines, and PGE2 [103].
In addition to these preclinical findings cited above, retrospective clinical studies have also widely discussed the effects of volatile and propofol-based anesthesia on cancer outcomes. Nevertheless, these studies have either shown that inhalational anesthetics are associated with an increased risk of cancer recurrence and the decreased overall survival (OS) or have shown no difference in different cancer outcomes between the two groups that were analyzed [16, 17, 104, 105•, 106, 107].
In summary, volatile anesthetics regulate important functions of cancer cells. The conflicting results of the pro and antitumoral effects on cancer cells might be explained by differences in experimental conditions, such as type of cell line, incubation time (range from 30 min to 6 h), type, and especially the concentration of volatile anesthetics (range between 0.5 and 10%). For instance, some studies treated cancer cells with extremely high concentrations that are unlikely to be employed in clinical practice, and perhaps, the “antitumortumoural” effect is related to the use of toxic concentrations of volatile anesthetics [108].
Intravenous Anesthetics
Similar to inhaled anesthetics, propofol is hypothesized to have both antitumor and tumor-promoting effects [109, 110]. Propofol inhibits the invasion and migration of breast tumors directly by altering the expression of MMPs, which are enzymes that play important roles in the degradation of extracellular proteins and EMT [111], by inhibiting the NF-κB pathway in vitro [112]. In another in vitro study, propofol inhibited the migration, but not the proliferation, of both ER-positive and ER-negative breast cancer cells mediated by decreasing the expression of neuroepithelial transforming gene-1, which is important for enhanced migration [113]. Tumor endothelial cells (TECs) [114], which are located in the inner surface of the blood vessels of the tumor stroma, have close associations with tumor progression in angiogenesis, metastasis, and colonization [115]. Propofol can reduce the expression of adhesion molecules (E-selectin, ICAM-1, and VCAM-1) and glycolysis proteins (GLUT1, HK2, and LDHA) in TECs, leading to an inhibitory effect on tumor metastasis [116]. It has also been found that aerobic glycolysis in colorectal cancer cells can be directly disrupted by propofol via inactivation of the NMDAR-CAMKII-ERK pathway [117]. The antitumoral effects of propofol on cancer progression also entail indirect mechanisms underlying immunosuppression, such as the potentiation of NK-cell cytotoxicity and reduced activation of the inflammatory response. For instance, in the peripheral blood of patients with colon cancer, propofol increased the expression levels of activated p30 and p44 in NK cells, which can promote the activation and proliferation of NK cells [118]. Additionally, in the peripheral blood of patients with esophageal squamous cell carcinoma, propofol enhanced the expression of cytotoxic effector molecules, such as granzyme B and IFN-γ, indicating enhanced NK cytotoxicity [119]. Regarding the cytokine profile, propofol downregulates the levels of proinflammatory cytokines, such as IL-1β, IL-6, and TNF-α [120], and inhibits PGE2 and COX activity [121]. Surprisingly, propofol can decrease NET formation through the inhibition of p-ERK without disrupting neutrophil killing capacity [122, 123].
Regarding tumor-promoting effects, in vitro studies have shown that propofol significantly promotes apoptosis in breast cancer cells followed by the downregulation of miR-24 expression, upregulation of p27 expression, and cleaved caspase-3 expression [124]. The expression levels of pro-apoptotic proteins, such as Bax, Bak, and cytochrome C, are increased, followed by the activation of the caspase cascades via an intrinsic apoptotic signaling pathway induced by propofol [125]. In addition, HIF-1 activation and the activation of related downstream genes, such as VEGF, were suppressed by propofol in an in vitro study using macrophage cells. This process is expected to inhibit the systemic inflammatory response to surgery [126]. In particular, propofol has a causal link with breast cancer recurrence. Propofol can accelerate the migration of breast cancer cells in association with the activation of the GABAA receptor [127] and promote the proliferation of human breast cancer cells related to the inhibition of p53 and activation of nuclear factor E2-related factor-2 in vitro [128].
Consistent with these preclinical data, the findings of retrospective studies that are presented in Table 1 are contradictory. However, recently, an increasing number of retrospective studies have shown that improved survival associated with propofol anesthesia is more pronounced in gastric cancer surgery. Nevertheless, further large-scale, high-quality RCTs are warranted to confirm the relationship between different anesthetic choices and cancer outcomes. Recent basic and clinical evidence raises the possibility that total intravenous anesthesia with propofol is appropriate for use as the standard anesthetic agent for all cancer surgeries.
Opioids
Opioid analgesics, as the fundamental pain relief approach, are commonly used within the perioperative period to supplement general anesthetic agents in both the induction and maintenance of anesthesia. There is some conflicting evidence from experimental studies investigating the role of opioids in tumor growth and metastasis. Most animal studies have found that certain opioids cause immunosuppression and, in turn, increase tumor recurrence postoperatively. In particular, morphine has largely been shown to suppress NK-cell cytotoxicity and T-cell proliferation [129, 130]. Similarly, it has been shown that fentanyl suppresses the activity of NK cells and promotes the apoptosis of lymphocytes and macrophages [131, 132]. However, other studies resulted in contradictory findings and instead proposed the effects of morphine on resistance to tumor metastasis by the downregulation of the reciprocal proangiogenic interaction between macrophages and breast cancer cells [133, 134]. A recent retrospective cohort clinical study of 1679 patients with stage I–III colorectal cancer showed no association between fentanyl use and oncological recurrence [135]. Alternatively, tramadol has been shown to have immunostimulatory properties by enhancing NK-cell cytotoxicity [136].
The mu-opioid receptors (MORs) are the major subtype of opioid receptors. MORs have been shown to be overexpressed in certain cancers. Mathew and colleagues in their report supported the fact that MOR is linked with tumor metastasis and found that there was less growth progression of lung carcinoma in MOR knockout mice [137]. Morphine has been found to not stimulate tumor initiation; however, it was found to stimulate the growth of existing breast tumors via MOPs in an experimental study [138]. Furthermore, the investigators concluded that patients who were given propofol anesthesia had a higher level of NK-cell expression than patients given general anesthesia with opioid analgesia. Conclusively, this report implies that opioids may modulate immune function mediated by MORs in breast cancer tissue [139]. Previous clinical evidence shows that the expression of MOR is associated with tumor grade and prognosis. Prostate cancer tissue exhibits more intense expression of MOR during staining, and in turn, patients with higher expression have worse oncologic outcomes [140]. MOR is also expressed at low levels in patients with low-grade (G1, G2) or low-stage (T1, T2) hepatocellular carcinoma [141], which is consistent with the results of a long-term retrospective study [142•]. Therefore, it is reasonable that there are several retrospective and prospective studies showing that intraoperative administration of high-dose opioids, including morphine and sufentanil, significantly reduced the survival rate and the median survival length in prostate cancer patients [143,144,145].
Local Anesthetics (LAs)
Several theories regarding the possible mechanisms of the observed potential beneficial effects of the LA results are stated below. First, it is well known that using RAs and LAs can reduce the use of opioids or volatile anesthetics during the perioperative period [146], which indirectly prevents the possible negative effects of general anesthetics on cancer recurrence. Second, there is strong evidence that LAs and RAs are able to reduce perioperative inflammation and the stress response induced by surgery [147, 148] and preserve the function of NK cells as one of the most important factors for the detection and destruction of CTCs [149, 150]. This systemic effect of the LA has a possible positive impact on perioperative processes leading to antitumor micrometastases by reducing the levels of CTCs in peripheral circulation, thus allowing prolonged (at least recurrence-free) survival. Third, lidocaine, bupivacaine, and ropivacaine, as usually used in LA, have been found to reduce mesenchymal stem cell proliferation in vitro, and the activation of transcription pathways related to the initiation of neoplasia and metastasis was also found to be inhibited [151]. It has been shown that the cytotoxic effects of T-lymphoma cells can be regulated by LAs in vitro, which is correlated with their lipophilicity and potency [152]. Apoptosis was also observed at lower concentrations of LAs, while necrosis was seen at higher concentrations. LAs have also been reported to alter the DNA methylation status of certain cancer cell types in a time- and dose-dependent manner to eventually reactivate tumor suppressor genes [153].
Recently, a systematic review showed that LAs, including lidocaine, ropivacaine, and levobupivacaine, present promising and consistent results regarding the anticancer influence of LAs on breast cancer [154•]. Therefore, regardless of the LA applied to avoid the possible pro-tumor effects of surgical stress response and general anesthesia, they can also reduce cancer recurrence involving the prevention of cancer cell proliferation, migration, and invasion. The etiology of these effects is likely multifactorial. In vitro and in vivo studies have proposed numerous mechanisms centered on NaV1.5 channels [155], Ras homolog gene family member A [156, 157], the cell cycle [158], endothelial growth factor receptor [159], calcium influx [160, 161], microRNA and mitochondria, in combination with hyperthermia and transient receptor potential melastatin 7 channels [162, 163]. Lidocaine has a shorter half-life than other LAs and is less toxic. As a result, it is the only amide LA compatible with intravenous administration. Interestingly, some findings in laboratory conditions have raised the possibility that intravenous infusion of lidocaine may be a safe and inexpensive way to provide significant benefits in long-term cancer outcomes [164]. It can potentially affect multiple biological pathways to act as an anti-inflammatory agent, immune cell modulator, and/or direct inhibitor of cancer cells [164]. Based on the promising laboratory data, accumulating prospective and retrospective clinical trials also support the beneficial anticancer effects of perioperative lidocaine treatment. In a recent retrospective study of 2239 patients undergoing resection of pancreatic carcinomas conducted by Zhang et al., those who received perioperative i.v. lidocaine exhibited significantly better OS at 1 and 3 years, while disease-free survival was unaffected [165••].
Others
Although intravenous and inhalational anesthetics have profound effects on cancer outcomes, we cannot ignore the effects of some perioperative anesthetic adjuvants. Ketamine is an NMDA receptor antagonist widely used in cancer treatment during the perioperative period due to its strong analgesic effects at subanesthetic doses. Increasing numbers of findings indicate that ketamine can modulate immune function through three major mechanisms [166]. First, ketamine has anti-inflammatory effects mediated by the inhibition of the expression of proinflammatory cytokines, such as IL-6 and TNF-α, during the early postoperative period [167]. Second, similar to other analgesics, ketamine significantly interferes with NK-cell cytotoxicity, thus increasing the susceptibility to tumor metastasis [14]. Third, ketamine can disrupt the balance of different T-cell populations, which inhibits antitumor immune function and is associated with cancer recurrence and poor survival in a dose-dependent manner [168]. A retrospective study also showed that ketamine is not associated with improved oncological outcomes [169].
Given that nonsteroidal anti-inflammatory drugs (NSAIDs) have opioid-sparing properties, they are another important adjuvant frequently administered in the perioperative period for analgesia. As COX inhibitors, NSAIDs decrease the expression of both cyclooxygenase-1 (COX-1) and COX-2 enzymes. Therefore, the overexpression of COX-2 increases the amount of PGE2, which further upregulates the expression levels of immunosuppressive IL-10. Similar to other oncogenes, COX-2 is overexpressed in colorectal carcinomas and adenomas as well as mammary tumors and plays a carcinogenic role [170, 171]. Indeed, COX-2 inhibitors are actively used in breast cancer [172], and overexpression of COX-2 favors breast tumor growth and increases the risk of cancer relapse by stimulating epithelial cell proliferation, inhibiting apoptosis, stimulating angiogenesis, suppressing immunity, and increasing the production of mutagens [173]. Three clinical studies have indicated that the use of NSAIDs may decrease the risk of cancer recurrence and increase disease-free survival. In a retrospective study of 327 women, a lower cancer recurrence rate was shown when ketorolac was given before cancer surgery than when other opioid-based analgesics were used [169]. In a follow-up study of the same patient population, it was found that the use of ketorolac could extend disease-free survival in the first few years after surgery with nearly no occurrence of relapse within the early months [174]. In another retrospective study of 720 breast cancer patients from a single-center cohort, the intraoperative use of NSAIDs (ketorolac or diclofenac) was associated with improved disease-free survival and overall survival [175]. As a result, the favorable antitumor effects of NSAIDs suggest their broader application to cancer treatment.
Dexmedetomidine (DEX) is becoming widely used to attenuate the stress response and reduce opioid requirements. DEX is a highly selective α2 adrenoreceptor agonist and has multiple pharmacologic effects, including hypnosis, analgesia, sedation, and anxiolysis. α2 adrenergic receptors are expressed on both immune cells and tumors, which indicates that DEX may affect the balance of the immune system and tumorigenesis [176, 177]. According to a meta-analysis of patient studies by Wang et al., the intraoperative use of DEX infusion increases the numbers of NK cells, B cells, and CD4 + T cells and the ratios of CD4 + /CD8 + and Th1/Th2 cells, while the number of CD8 + T cells is significantly decreased [178]. These data indicated that DEX can protect the immune function of surgical patients. This indication corroborates a previous study, which showed that DEX did not significantly inhibit T-cell proliferation or IL-2 production [179]. A recent study also showed that perioperative DEX had no favorable impacts on NK-cell activity or inflammatory responses in uterine cancer surgery patients [180]. However, Levon et al. tested the effects of DEX on tumor growth and found that the size of breast, lung, and colon cancer was significantly larger in mice that received DEX [181, 182•, 183]. Consistent with the animal study, the study of 1404 patients by Cata et al. showed that intraoperative dexmedetomidine administration was associated with worsened overall survival after surgery for non-small cell lung cancer [184]. However, a randomized controlled study should be conducted to confirm the results of these studies.
Tramadol is an atypical opioid analgesic that has shown antitumor effects in breast cancer cells in vitro and in vivo [185•, 186]. The mechanism by which tramadol exerts these effects involves suppression of colony formation, cell cycle arrest, and the induction of apoptosis via extracellular signal-regulated kinases by decreasing 5-hydroxytryptamine 2B receptor and transient receptor potential vanilloid-1 expression, as demonstrated by in vitro experiments [185•]. In vivo, tramadol administration decreased the expression of inflammatory cytokines, such as IL-6 and TNF-α, which are involved in tumor growth and invasion [186]. Additionally, tramadol functions similarly to morphine but may produce the opposite effect and instead increase the activation of NK cells in patients undergoing surgical tumor resection [33, 187]. Furthermore, a retrospective analysis of 2588 patients showed that tramadol use was associated with decreased rates of breast cancer recurrence and improved survival after breast cancer surgery [185•].
The Effects of Anesthetic Techniques on Cancer Recurrence
Whether anesthetic techniques during potentially curative intraoperative periods influence cancer recurrence is a question that needs to be addressed to reveal the associations between different anesthetic managements and cancer outcomes. There are two major anesthetic approaches that may potentially affect cancer recurrence risk: (1) the use of RA, including neuraxial (epidural or spinal) and paravertebral blocks, and (2) the use of general anesthesia, including intravenous and inhalation anesthesia. Over the past decade, RA has been hypothesized to lower the surgical stress response and immunosuppression, reduce the need for volatile anesthesia, and minimize pain and opioid needs in the perioperative period, hence decreasing the activation of pro-tumor pathways and enhancing long-term oncological outcomes. Additionally, RA preserves the function of the immune system and has a direct inhibitory effect on cancer cells [188, 189]. For example, when comparing the use of regional techniques with the use of general anesthesia in cancer patients in RCTs, substantially lower levels of NK-cell and T-cell activity were observed in those who received both than in individuals who only received general anesthesia [190, 191]. As a result, several retrospective studies and meta-analyses have shown that the application of RA to supplement GA is closely associated with better OS than the use of GA plus opioid analgesia, at least during the treatment of certain types of cancer, such as breast, colon, lung, and prostate cancers [192,193,194,195,196].
In this review, we summarized over 5 years of clinical evidence regarding the effects of various RAs on cancer outcomes. This review is almost exclusively from retrospective studies (Table 2). Interpretation of retrospective analyses should be inherently cautious because of potential selection bias. According to this summary, retrospective findings are not enough to prove the positive effects of RA due to conflicting results, while all the RCTs have reached similar conclusions. Specifically, some RCTs investigating RA and cancer recurrence enrolled more than 2100 women with primary breast cancer. Patients were randomly assigned to receive either RA (preferentially paravertebral block) with propofol sedation or sevoflurane/opioid-based general anesthesia. It has been shown that there is no difference in disease-free survival or cancer recurrence [197••]. The effect of combined epidural-general was also investigated in a large multicenter RCT including patients (n = 1712) with major noncardiac thoracic or abdominal surgery. The median follow-up time was after 5 years, and the combined epidural-general anesthesia and general anesthesia group had similar rates of mortality, cancer-specific survival, and relapse-free survival (RFS) [198]. The most recent RCT included 400 patients who were undergoing video-assisted thoracoscopic lung cancer resection and compared the effects of the use of combination epidural-general anesthesia against the use of general anesthesia alone. The primary outcome was RFS. Secondary outcomes were OS and cancer-specific survival. The median follow-up time was 32 months. The results indicated that the use of epidural anesthesia during major lung surgery did not result in better RFS, cancer-specific survival, or OS than the use of general anesthesia alone [199]. Other RCTs focusing on the effects of RA on colon and prostate cancer surgery also failed to demonstrate any benefits in cancer outcomes [200, 201]. Given that these studies are appropriately powered and the results seem compelling, the influence of RA on cancer recurrence might be negligible or not existent. It can be speculated that RA probably fails to produce a robust immunomodulatory or anti-inflammatory effect or that the concentrations of LAs in micrometastatic niches are too low to produce significant antitumor effects [102, 202]. Although there is insufficient evidence to support the use of regional techniques at this time for their perceived benefit in terms of cancer recurrence, RA continues to play a fundamental role in the developing subspecialty of oncology anesthesia, which is focused on individualized, perioperative management in an attempt to minimize morbidity, accelerate recovery, and promote the progression to the next stage of oncological therapy.
The Effects of Perioperative Body Temperature on Cancer Recurrence
Body temperature is a sensitive indicator of whether the internal environment of the body is in a stable state. Body temperature acts as a critical regulator to influence the response to cancer. However, the mechanisms have only recently been investigated. Increasing in vitro and in vivo studies found that elevated temperatures can generally promote the activation, function, and delivery of immune cells, while reduced temperatures inhibit these processes [203]. In particular, adaptive immunity can be modulated by temperature, including antigen-presenting cells and CD4 + /CD8 + T cells [203]. Moreover, in patients undergoing open surgery for gastric cancer, Yi Yang et al. found that maintaining a body temperature close to normal could preserve immune functions [204]. Accordingly, the picture that is emerging is that temperature can have varying impacts on cancer outcomes. Given that perioperative systemic hypothermia is commonly encountered and even a few degrees of perioperative hypothermia can have immunosuppressive consequences, it is necessary to explore the association between hypothermia and cancer recurrence. In a rat model of colon cancer, tumor retention was found to be promoted by perioperative hypothermia [205], and severe hypothermia (3–7 °C decrease from the normal body temperature) markedly suppressed NK-cell activity and jeopardized host resistance to experimental mammary metastasis [206]. However, these results have not been replicated in human studies. In a reanalysis of data from a previous cohort of 852 patients, where intraoperative core body temperature was defined as a median intraoperative temperature of < 36 °C, consistent with the existing consensus definition [207], there was no significant increase in the incidence of cancer recurrence or death from metastasis following radical cystectomy. Yucel et al. also drew a similar conclusion [208•]. However, there are several conflicting findings that demonstrate the causal relationship between hypothermia and cancer prognosis. For example, intraoperative hypothermia may be a significant predictor of recurrence and survival in muscle-invasive bladder cancer [209] and rectal cancer [210].
According to a clinical study by Zheng et al. of stage III gastric cancer patients, a high postoperative body temperature could significantly reduce the 5-year disease-free survival [211]. Therefore, treating hyperthermia (also called thermal therapy or thermotherapy) is becoming one of the cancer treatment methods used to reduce cancer recurrence. Indeed, accumulating evidence indicates that physiological responses to high body temperature can enhance the microenvironment’s ability to resist tumors through temperature-sensitive checkpoints that regulate tumor vascular perfusion and metabolism [212]. In contrast to prior research, a recent in vitro study indicated that cancer cells can resist higher temperatures than normal cells by not activating caspase 3 [213]. As a result, the influence of thermal stimuli on the tumor environment and the antitumor immune response remain incompletely understood.
Conclusion and Future Perspectives
Recently, many of the retrospective clinical trials highlighted in this review have definitively demonstrated the profound impacts of perioperative events on cancer recurrence. For example, most retrospective trials have found that TIVA is a better anesthetic choice for cancer surgery according to the increased OS and RFS. However, several RCTs have shown that epidural and paravertebral nerve blocks are unable to modify cancer prognosis. For other anesthetic agents/techniques, there is still a critical lack of clinical evidence confirming the association with cancer recurrence during the perioperative period. The preclinical and clinical studies provided in this review exhibited conflicting findings on the effects of anesthesia on the immune response and cancer growth. Therefore, large-cohort prospective clinical trials are required to explore the effects of different anesthetics and techniques on long-term outcomes after cancer surgery. Furthermore, such trials will be beneficial to the development of systemic cancer therapies, which will enable us to optimize perioperative cancer treatment.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ahmad FB, Anderson RN. The leading causes of death in the US for 2020. JAMA - J Am Med Assoc. 2021;325(18):1829–30.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71(1):7–33.
Kim R. Anesthetic technique and cancer recurrence in oncologic surgery: unraveling the puzzle. Cancer Metastasis Rev. 2017;36(1):159–77.
Tsuchiya Y, Sawada S, Yoshioka I, et al. Increased surgical stress promotes tumor metastasis. Surg. 2003;133(5):547–55.
Boomsma MF, Garssen B, Slot E, et al. Breast cancer surgery-induced immunomodulation. J Surg Oncol. 2010;102(6):640–8.
Camara O, Kavallaris A, Noschel H, Rengsberger M, Jorke C, Pachmann K. Seeding of epithelial cells into circulation during surgery for breast cancer: the fate of malignant and benign mobilized cells. World J Surg Oncol. 2006;4(1):1–7.
Eschwège P, Blanchet P, Benoit G, et al. Haematogenous dissemination of prostatic epithelial cells during radical prostatectomy. Lancet. 1995;346(8989):1528–30. In this study, they confirmed that radical prostatectomy can cause hematogenous dissemination of prostatic epithelial cells in the general circulation, which was related to the emergence of metastatic disease.
Mori M, Mimori K, Ueo H, et al. Molecular detection of circulating solid carcinoma cells in the peripheral blood: the concept of early systemic disease. Int J Cancer. 1996;68(6):739–43.
Alsina E, Matute E, Ruiz-Huerta DA, Gilsanz F. The effects of sevoflurane or remifentanil on the stress response to surgical stimulus. Curr Pharm Des. 2014;20(34):5449–68.
Kim R. Effects of surgery and anesthetic choice on immunosuppression and cancer recurrence. J Transl Med. 2018;16(1):8.
Cole SW, Nagaraja AS, Lutgendorf SK, Green PA, Sood AK. Sympathetic nervous system regulation of the tumour microenvironment. Nat Rev Cancer. 2015;15(9):563–72. This review highlights that multiple cellular and molecular pathways through SNS activation influence tumor initiation, the development and conditioning of the metastatic niche, or the responses to anti-tumor therapy.
Alam A, Rampes S, Patel S, Hana Z, Ma D. Anesthetics or anesthetic techniques and cancer surgical outcomes: a possible link. Korean J Anesthesiol. 2021;74(3):191–203.
Huang H, Benzonana LL, Zhao H, et al. Prostate cancer cell malignancy via modulation of HIF-1α pathway with isoflurane and propofol alone and in combination. Br J Cancer. 2014;111(7):1338–49.
Melamed R, Bar-Yosef S, Shakhar G, Shakhar K, Ben-Eliyahu S. Suppression of natural killer cell activity and promotion of tumor metastasis by ketamine, thiopental, and halothane, but not by propofol: mediating mechanisms and prophylactic measures. Anesth Analg. 2003;97(5):1331–9.
Mammoto T, Mukai M, Mammoto A, et al. Intravenous anesthetic, propofol inhibits invasion of cancer cells. Cancer Lett. 2002;184(2):165–70.
Wigmore TJ, Mohammed K, Jhanji S. Long-term survival for patients undergoing volatile versus IV anesthesia for cancer surgery: a retrospective analysis. Anesthesiol. 2016;124(1):69–79.
Jun IJ, Jo JY, Kim JI, Chin JH, Kim WJ, Kim HR, Lee EH, Choi IC. Impact of anesthetic agents on overall and recurrence-free survival in patients undergoing esophageal cancer surgery: a retrospective observational study. Scientific reports. 2017;7(1):1–9.
Hiller JG, Perry NJ, Poulogiannis G, Riedel B, Sloan EK. Perioperative events influence cancer recurrence risk after surgery. Nat Rev Clin Oncol. 2018;15(4):205–18. This review discusses the relationship between perioperative events and cancer recurrence, which shows the critical role of the perioperative period in cancer treatment.
Horowitz M, Neeman E, Sharon E, Ben-Eliyahu S. Exploiting the critical perioperative period to improve long-term cancer outcomes. Nat Rev Clin Oncol. 2015;12(4):213–26. This review raises a novel concept that the perioperative timeframe can be transformed from a prominent facilitator of metastatic progression to a window of opportunity for arresting and/or eliminating residual disease, potentially improving long-term survival rates in patients with cancer.
Raman D, Baugher PJ, Thu YM, Richmond A. Role of chemokines in tumor growth. Cancer Lett. 2007;256(2):137–65.
Tedore T. Regional anaesthesia and analgesia: relationship to cancer recurrence and survival. Br J Anaesth. 2015;115:ii34–45.
Schmitt N, Ueno H. Regulation of human helper T cell subset differentiation by cytokines. Curr Opin Immunol. 2015;34:130–6.
Dinarello CA. Interleukin-1. Cytokine Growth Factor Rev. 1997;8(4):253–65.
Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Sci. 2011;331(6024):1565–70. This review proposes a unifying conceptual framework called “cancer immunoediting,” which shows that the immune system can not only suppress tumor growth by destroying cancer cells or inhibiting their outgrowth but also promote tumor progression either by selecting tumor cells that are more fit to survive in an immunocompetent host or by establishing conditions within the tumor microenvironment that facilitate tumor outgrowth.
Sekandarzad MW, Van Zundert AAJ, Lirk PB, Doornebal CW, Hollmann MW. Perioperative anesthesia care and tumor progression. Anesth Analg. 2017;124(5):1697–708.
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420(6917):860–7. The review gives the insights that inflammation cells orchestrate tumor microenvironment and are indispensable participants in the neoplastic process, fostering proliferation, survival, and migration.
Wang Y, Qu M, Qiu Z, et al. Surgical stress and cancer progression: new findings and future perspectives. Curr Oncol Rep: Published online June; 2022.
Tyzzer EE. Factors in the production and growth of tumor metastases. J Med Res. 1913;28(2):309–32.
Demicheli R, Retsky MW, Hrushesky WJM, Baum M, Gukas ID. The effects of surgery on tumor growth: a century of investigations. Ann Oncol. 2008;19(11):1821–8.
van der Bij GJ, Oosterling SJ, Bögels M, et al. Blocking α2 integrins on rat CC531s colon carcinoma cells prevents operation-induced augmentation of liver metastases outgrowth. Hepatol. 2007;47(2):532–43.
Cassinello F, Prieto I, Del Olmo M, Rivas S, Strichartz GR. Cancer surgery: how may anesthesia influence outcome? J Clin Anesth. 2015;27(3):262–72.
Kurosawa S. Anesthesia in patients with cancer disorders. Curr Opin Anaesthesiol. 2012;25(3):376–84.
Sacerdote P, Bianchi M, Gaspani L, et al. The effects of tramadol and morphine on immune responses and pain after surgery in cancer patients. Anesth Analg. 2000;90(6):1411–4.
Ben-Eliyahu S, Page GG, Yirmiya R, Shakhar G. Evidence that stress and surgical interventions promote tumor development by suppressing natural killer cell activity. Int J Cancer. 1999;80(6):880–8.
Chae BK, Lee HW, Sun K, Choi YH, Kim HM. The effect of combined epidural and light general anesthesia on stress hormones in open heart surgery patients. Surg Today. 1998;28(7):727–31.
Wall T, Sherwin A, Ma D, Buggy DJ. Influence of perioperative anaesthetic and analgesic interventions on oncological outcomes: a narrative review. Br J Anaesth. 2019;123(2):135–50.
Neeman E, Ben-Eliyahu S. Surgery and stress promote cancer metastasis: new outlooks on perioperative mediating mechanisms and immune involvement. Brain Behav Immun. 2012;22:139–46.
Casey SC, Amedei A, Aquilano K, et al. Cancer prevention and therapy through the modulation of the tumor microenvironment. Semin Cancer Biol. 2015;35:S199–223.
Benish M, Melamed R, Rosenne E, et al. The marginating-pulmonary immune compartment in mice exhibits increased NK cytotoxicity and unique cellular characteristics. Immunol Res. 2014;58(1):28–39.
Hiratsuka S, Watanabe A, Aburatani H, Maru Y. Tumour-mediated upregulation of chemoattractants and recruitment of myeloid cells predetermines lung metastasis. Nat Cell Biol. 2006;8(12):1369–75.
Green BL, Marshall HC, Collinson F, et al. Long-term follow-up of the Medical Research Council CLASICC trial of conventional versus laparoscopically assisted resection in colorectal cancer. Br J Surg. 2013;100(1):75–82.
Sceneay J, Chow MT, Chen A, et al. Primary tumor hypoxia recruits CD11b+/Ly6Cmed/ Ly6G+ immune suppressor cells and compromises NK cell cytotoxicity in the premetastatic niche. Cancer Res. 2012;72(16):3906–11.
Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial–mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15(3):178–96.
Nagrath S, Sequist LV, Maheswaran S, et al. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature. 2007;450(7173):1235–9.
Rahbari NN, Aigner M, Thorlund K, et al. Meta-analysis shows that detection of circulating tumor cells indicates poor prognosis in patients with colorectal cancer. Gastroenterol. 2010;138(5):1714-1726.e13.
Hardingham JE, Grover P, Winter M, Hewett PJ, Price TJ, Thierry B. Detection and clinical significance of circulating tumor cells in colorectal cancer—20 years of progress. Mol Med. 2015;21:S25–31.
Brown DC, Purushotham AD, Birnie GD, George WD. Detection of intraoperative tumor cell dissemination in patients with breast cancer by use of reverse transcription and polymerase chain reaction. Surg. 1995;117(1):96–101.
Hashimoto M, Tanaka F, Yoneda K, et al. Significant increase in circulating tumour cells in pulmonary venous blood during surgical manipulation in patients with primary lung cancer. Interact Cardiovasc Thorac Surg. 2014;18(6):775–83.
Peach G, Kim C, Zacharakis E, Purkayastha S, Ziprin P. Prognostic significance of circulating tumour cells following surgical resection of colorectal cancers: a systematic review. Br J Cancer. 2010;102(9):1327–34.
Martin OA, Anderson RL, Narayan K, MacManus MP. Does the mobilization of circulating tumour cells during cancer therapy cause metastasis? Nat Rev Clin Oncol. 2017;14(1):32–44.
Hayashi K, Jiang P, Yamauchi K, et al. Real-time imaging of tumor-cell shedding and trafficking in lymphatic channels. Cancer Res. 2007;67(17):8223–8.
Tvedskov TF, Jensen MB, Kroman N, Balslev E. Iatrogenic displacement of tumor cells to the sentinel node after surgical excision in primary breast cancer. Breast Cancer Res Treat. 2012;131(1):223–9.
Pommier A, Anaparthy N, Memos N, et al. Unresolved endoplasmic reticulum stress engenders immune-resistant, latent pancreatic cancer metastases. Sci. 2018;360(6394):4eaao4908. In this article, they found that adaptive immune response can select single disseminated cancer cells, in which the endoplasmic reticulum stress response accounted for both quiescence and resistance to immune elimination. Moreover, the suppression of systemic immunity enabled the outgrowth of disseminated cancer cells to macrometastases.
Kadar N. Port-site recurrences following laparoscopic operations for gynaecological malignancies. BJOG An Int J Obstet Gynaecol. 1997;104(11):1308–13.
Song J, Kim E, Mobley J, et al. Port site metastasis after surgery for renal cell carcinoma: harbinger of future metastasis. J Urol. 2014;192(2):364–8.
Downey RJ, McCormack P, LoCicero J III. Dissemination of malignant tumors after video-assisted thoracic surgery: a report of twenty-one cases. J Thorac Cardiovasc Surg. 1996;111(5):954–60.
Berger-Richardson D, Chesney TR, Englesakis M, Govindarajan A, Cleary SP, Swallow CJ. Trends in port-site metastasis after laparoscopic resection of incidental gallbladder cancer: a systematic review. Surg (United States). 2017;161(3):618–27.
Li R, Huang Y, Lin J. Distinct effects of general anesthetics on lung metastasis mediated by IL-6/JAK/STAT3 pathway in mouse models. Nat Commun. 2020;11(1):1–10.
Fares J, Fares MY, Khachfe HH, Salhab HA, Fares Y. Molecular principles of metastasis: a hallmark of cancer revisited. Signal Transduct Target Ther. 2020;5(1):1–17.
Colegio OR, Chu NQ, Szabo AL, et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature. 2014;513(7519):559–63.
Agard M, Asakrah S, Morici LA. PGE(2) suppression of innate immunity during mucosal bacterial infection. Front Cell Infect Microbiol. 2013;3:45.
Martinet L, Jean C, Dietrich G, Fournié JJ, Poupot R. PGE2 inhibits natural killer and γδ T cell cytotoxicity triggered by NKR and TCR through a cAMP-mediated PKA type I-dependent signaling. Biochem Pharmacol. 2010;80(6):838–45.
Pietra G, Manzini C, Rivara S, et al. Melanoma cells inhibit natural killer cell function by modulating the expression of activating receptors and cytolytic activity. Cancer Res. 2012;72(6):1407–15.
Mirnezami A, Mirnezami R, Chandrakumaran K, Sasapu K, Sagar P, Finan P. Increased local recurrence and reduced survival from colorectal cancer following anastomotic leak: systematic review and meta-analysis. Ann Surg. 2011;253(5):890–9.
Lu ZR, Rajendran N, Lynch AC, Heriot AG, Warrier SK. Anastomotic leaks after restorative resections for rectal cancer compromise cancer outcomes and survival. Dis Colon Rectum. 2016;59(3):236–44.
Hogan BV, Peter MB, Shenoy HG, Horgan K, Hughes TA. Surgery induced immunosuppression. Surgeon. 2011;9(1):38–43.
Karnezis T, Shayan R, Caesar C, et al. VEGF-D Promotes tumor metastasis by regulating prostaglandins produced by the collecting lymphatic endothelium. Cancer Cell. 2012;21(2):181–95.
Ellyard JI, Simson L, Parish CR. Th2-mediated anti-tumour immunity: friend or foe? Tissue Antigens. 2007;70(1):1–11.
Finnerty CC, Mabvuure NT, Ali A, Herndon DN. The surgically induced stress response. J Parenter Enter Nutr. 2013;37:21S-29S.
Quatrini L, Ugolini S. New insights into the cell- and tissue-specificity of glucocorticoid actions. Cell Mol Immunol. 2021;18(2):269–78.
Braadland PR, Ramberg H, Grytli HH, Taskén KA. β-Adrenergic receptor signaling in prostate cancer. Front Oncol. 2015;4:375.
Choy C, Raytis JL, Smith DD, et al. Inhibition of β2- adrenergic receptor reduces triple-negative breast cancer brain metastases: the potential benefit of perioperative β-blockade. Oncol Rep. 2016;35(6):3135–42.
Huang Q, Tan Q, Mao K, Yang G, Ma G, Luo P. The role of adrenergic receptors in lung cancer. Am J Cancer Res. 2018;8(11):2227–37.
Liu J, Qu L, Wan C, Xiao M, Ni W, Jiang F, Fan Y, Lu C, Ni R. A novel β2-AR/YB-1/β-catenin axis mediates chronic stress-associated metastasis in hepatocellular carcinoma. Oncogenesis. 2020;9(9):1–14.
Liu X, Wu WKK, Yu L, et al. Epinephrine stimulates esophageal squamous-cell carcinoma cell proliferation via β-adrenoceptor-dependent transactivation of extracellular signal-regulated kinase/cyclooxygenase-2 pathway. J Cell Biochem. 2008;105(1):53–60.
Zhang H, Kong Q, Wang J, Jiang Y, Hua H. Complex roles of cAMP–PKA–CREB signaling in cancer. Exp Hematol Oncol. 2020;9(1):1–13.
Gould CM, Courtneidge SA. Regulation of invadopodia by the tumor microenvironment. Cell Adhes Migr. 2014;8(3):226–35.
Creed SJ, Le CP, Hassan M, Pon CK, Albold S, Chan KT, Berginski ME, Huang Z, Bear JE, Lane JR, Halls ML, Ferrari D, Nowell CJ, Sloan EK. β2-adrenoceptor signaling regulates invadopodia formation to enhance tumor cell invasion. Breast Cancer Res. 2015;17(1):1–12.
Clark AG, Vignjevic DM. Modes of cancer cell invasion and the role of the microenvironment. Curr Opin Cell Biol. 2015;36:13–22.
Cheung KJ, Ewald AJ. A collective route to metastasis: seeding by tumor cell clusters. Sci. 2016;352(6282):167–9.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74.
Katsuno Y, Lamouille S, Derynck R. TGF-β signaling and epithelial–mesenchymal transition in cancer progression. Curr Opin Oncol. 2013;25(1):76–84.
De CB, Berx G. Regulatory networks defining EMT during cancer initiation and progression. Nat Rev Cancer. 2013;13(2):97–110.
Pastushenko I, Brisebarre A, Sifrim A, Fioramonti M, Revenco T, Boumahdi S, Van Keymeulen A, Brown D, Moers V, Lemaire S, De Clercq S, Minguijón E, Balsat C, Sokolow Y, Dubois C, De Cock F, Scozzaro S, Sopena F, Lanas A, D'Haene N, Salmon I, Marine JC, Voet T, Sotiropoulou PA, Blanpain C. Identification of the tumour transition states occurring during EMT. Nature. 2018;556(7702):463–68.
Klymkowsky MW, Savagner P. Epithelial-mesenchymal transition: a cancer researcher’s conceptual friend and foe. Am J Pathol. 2009;174(5):1588–93.
Nagai S, Yoshida A, Facciuto M, et al. Ischemia time impacts recurrence of hepatocellular carcinoma after liver transplantation. Hepatol. 2015;61(3):895–904.
Kornberg A, Witt U, Kornberg J, Friess H, Thrum K. Extended ischemia times promote risk of hcc recurrence in liver transplant patients. Dig Dis Sci. 2015;60(9):2832–9.
Rodvold JJ, Mahadevan NR, Zanetti M. Lipocalin 2 in cancer: when good immunity goes bad. Cancer Lett. 2012;316(2):132–8.
Guo L, Li SY, Ji FY, et al. Role of Angptl4 in vascular permeability and inflammation. Inflamm Res. 2014;63(1):13–22.
Goebel L, Grage-Griebenow E, Gorys A, Helm O, Genrich G, Lenk L, Wesch D, Ungefroren H, Freitag-Wolf S, Sipos B, Röcken C, Schäfer H, Sebens S. CD4+ T cells potently induce epithelial-mesenchymal-transition in premalignant and malignant pancreatic ductal epithelial cells-novel implications of CD4+ T cells in pancreatic cancer development. Oncoimmunology. 2015;4(4):e1000083.
Stone RC, Pastar I, Ojeh N, et al. Epithelial-mesenchymal transition in tissue repair and fibrosis. Cell Tissue Res. 2016;365(3):495–506.
Savagner P, Arnoux V. Epithelio-mesenchymal transition and cutaneous wound healing. Bull Acad Natl Med. 2009;193(9):1981–92.
Savagner P, Kusewitt DF, Carver EA, et al. Developmental transcription factor slug is required for effective re-epithelialization by adult keratinocytes. J Cell Physiol. 2005;202(3):858–66.
Gao X, Mi Y, Guo N, et al. The mechanism of propofol in cancer development: an updated review. Asia Pac J Clin Oncol. 2020;16(2):e3–11. In this review, they summarized the preclinical trials to show the anti-tumor effects of propofol on cancer development by inhibiting epithelial-to-mesenchymal transition.
Benzonana LL, Perry NJS, Watts HR, et al. Isoflurane, a commonly used volatile anesthetic, enhances renal cancer growth and malignant potential via the hypoxia-inducible factor cellular signaling pathway in vitro. Anesthesiol. 2013;119(3):593–605. The first well-established preclinical trial shows that the exposure of human renal cancer cells to isoflurane (0.5–2% for 2h) promotes cancer cell growth and migration in vitro and hence enhances malignant potential.
Zhu M, Li M, Zhou Y, et al. Isoflurane enhances the malignant potential of glioblastoma stem cells by promoting their viability, mobility in vitro and migratory capacity in vivo. Br J Anaesth. 2016;116(6):870–7.
Zhang W, Shao X. Isoflurane promotes non-small cell lung cancer malignancy by activating the akt-mammalian target of rapamycin (mTOR) signaling pathway. Med Sci Monit. 2016;22:4644–50.
Ren J, Wang X, Wei G, Meng Y. Exposure to desflurane anesthesia confers colorectal cancer cells metastatic capacity through deregulation of miR-34a/LOXL3. Eur J Cancer Prev. 2021;30(2):143–53.
Deng X, Vipani M, Liang G, Gouda D, Wang B, Wei H. Sevoflurane modulates breast cancer cell survival via modulation of intracellular calcium homeostasis. BMC Anesthesiol. 2020;20(1):1–9.
Loop T, Dovi-Akue D, Frick M, et al. Volatile anesthetics induce caspase-dependent, mitochondria-mediated apoptosis in human T lymphocytes in vitro. Anesthesiol. 2005;102(6):1147–57.
Tavare AN, Perry NJS, Benzonana LL, Takata M, Ma D. Cancer recurrence after surgery: direct and indirect effects of anesthetic agents. Int J Cancer. 2012;130(6):1237–50.
Deegan CA, Murray D, Doran P, et al. Anesthetic technique and the cytokine and matrix metalloproteinase response to primary breast cancer surgery. Reg Anesth Pain Med. 2010;35(6):490–5.
Ecimovic P, McHugh B, Murray D, Doran P, Buggy DJ. Effects of sevoflurane on breast cancer cell function in vitro. Anticancer Res. 2013;33(10):4255–60.
Lee JH, Kang SH, Kim Y, Kim HA, Kim BS. Effects of propofol-based total intravenous anesthesia on recurrence and overall survival in patients after modified radical mastectomy: a retrospective study. Korean J Anesthesiol. 2016;69(2):126–32.
Wu ZF, Lee MS, Wong CS, et al. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in colon cancer surgery. Anesthesiol. 2018;129(5):932–41. A retrospective cohort study of 1158 patients shows that propofol anesthesia for colon cancer surgery is associated with better overall survival than desflurane anesthesia irrespective of tumor–node–metastasis stage.
Kim MH, Kim DW, Kim JH, Lee KY, Park S, Yoo YC. Does the type of anesthesia really affect the recurrence-free survival after breast cancer surgery? Oncotarget. 2017;8(52):90477–87.
Enlund M, Berglund A, Andreasson K, Cicek C, Enlund A, Bergkvist L. The choice of anaesthetic-sevoflurane or propofol-and outcome from cancer surgery: a retrospective analysis. Ups J Med Sci. 2014;119(3):251–61.
Ramirez MF, Cata JP. Anesthesia techniques and long-term oncological outcomes. Front Oncol. 2021;11(December):1–13.
Li R, Liu H, Dilger JP, Lin J. Effect of propofol on breast cancer cell, the immune system, and patient outcome. BMC Anesthesiol. 2018;18(1):77.
Wang J, Cheng CS, Lu Y, et al. Novel findings of anti-cancer property of propofol. Anticancer Agents Med Chem. 2018;18(2):156–65.
Gialeli C, Theocharis AD, Karamanos NK. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J. 2011;278(1):16–27.
Li Q, Zhang L, Han Y, Jiang Z, Wang Q. Propofol reduces MMPs expression by inhibiting NF-κB activity in human MDA-MB-231 cells. Biomed Pharmacother. 2012;66(1):52–6.
Ecimovic P, Murray D, Doran P, Buggy DJ. Propofol and bupivacaine in breast cancer cell function in vitro - role of the NET1 gene. Anticancer Res. 2014;34(3):1321–31.
Hida K, Maishi N, Torii C, Hida Y. Tumor angiogenesis—characteristics of tumor endothelial cells. Int J Clin Oncol. 2016;21(2):206–12.
Hergenreider E, Heydt S, Tréguer K, et al. Atheroprotective communication between endothelial cells and smooth muscle cells through miRNAs. Nat Cell Biol. 2012;14(3):249–56.
Qi J, Wu Q, Zhu X, et al. Propofol attenuates the adhesion of tumor and endothelial cells through inhibiting glycolysis in human umbilical vein endothelial cells. Acta Biochim Biophys Sin (Shanghai). 2019;51(11):1114–22.
Physiology C. Erratum: Propofol disrupts aerobic glycolysis in colorectal cancer cells via inactivation of the NMDAR-CAMKII-ERK pathway (Cellular Physiology and Biochemistry. Cell Physiol Biochem. 2018;46(2):492–504. https://doi.org/10.1159/000488617.
Liu D, Sun X, Du Y, Kong M. Propofol promotes activity and tumor-killing ability of natural killer cells in peripheral blood of patients with colon cancer. Med Sci Monit. 2018;24:6119–28.
Zhou M, Dai J, Zhou Y, et al. Propofol improves the function of natural killer cells from the peripheral blood of patients with esophageal squamous cell carcinoma. Exp Ther Med. 2018;16(1):83–92.
Ding XW, Sun X, Shen XF, et al. Propofol attenuates TNF-α-induced MMP-9 expression in human cerebral microvascular endothelial cells by inhibiting Ca(2+)/CAMK II/ERK/NF-κB signaling pathway. Acta Pharmacol Sin. 2019;40(10):1303–13.
Inada T, Hirota K, Shingu K. Intravenous anesthetic propofol suppresses prostaglandin E2 and cysteinyl leukotriene production and reduces edema formation in arachidonic acid-induced ear inflammation. J Immunotoxicol. 2015;12(3):261–5.
Chen MS, Lin WC, Yeh HT, Hu CL, Sheu SM. Propofol specifically reduces PMA-induced neutrophil extracellular trap formation through inhibition of p-ERK and HOCl. Life Sci. 2019;221:178–86.
Meier A, Chien J, Hobohm L, Patras KA, Nizet V, Corriden R. Inhibition of human neutrophil extracellular trap (NET) production by propofol and lipid emulsion. Front Pharmacol. 2019;10:323.
Yu B, Gao W, Zhou H, et al. Propofol induces apoptosis of breast cancer cells by downregulation of miR-24 signal pathway. Cancer Biomark. 2018;21(3):513–9.
Wang H, Zhao L, Wu J, Hong J, Wang S. Propofol induces ROS-mediated intrinsic apoptosis and migration in triple-negative breast cancer cells. Oncol Lett. 2020;20(1):810–6.
Tanaka T, Takabuchi S, Nishi K, et al. The intravenous anesthetic propofol inhibits lipopolysaccharide-induced hypoxia-inducible factor 1 activation and suppresses the glucose metabolism in macrophages. J Anesth. 2010;24(1):54–60.
Garib V, Lang K, Niggemann B, Zänker KS, Brandt L, Dittmar T. Propofol-induced calcium signalling and actin reorganization within breast carcinoma cells. Eur J Anaesthesiol. 2005;22(8):609–15.
Chao M, Linlin S, Juan W, Li D, Liu Y, Cui X. Propofol induces proliferation partially via downregulation of p53 protein and promotes migration via activation of the Nrf2 pathway in human breast cancer cell line MDA-MB-231. Oncol Rep. 2017;37(2):841–8.
Lu H, Zhang H, Weng ML, et al. Morphine promotes tumorigenesis and cetuximab resistance via EGFR signaling activation in human colorectal cancer. J Cell Physiol. 2021;236(6):4445–54.
Das J, Kumar S, Khanna S, Mehta Y. Are we causing the recurrence-impact of perioperative period on long-term cancer prognosis: review of current evidence and practice. J Anaesthesiol Clin Pharmacol. 2014;30(2):153–9.
Beilin B, Shavit Y, Hart J, et al. Effects of anesthesia based on large versus small doses of fentanyl on natural killer cell cytotoxicity in the perioperative period. Anesth Analg. 1996;82(3):492–7.
Shavit Y, Ben-Eliyahu S, Zeidel A, Beilin B. Effects of fentanyl on natural killer cell activity and on resistance to tumor metastasis in rats: dose and timing study. NeuroImmunoModulation. 2004;11(4):255–60.
Khabbazi S, Nassar ZD, Goumon Y, Parat MO. Morphine decreases the pro-angiogenic interaction between breast cancer cells and macrophages in vitro. Sci Rep. 2016;6(1):1–10.
Koodie L, Yuan H, Pumper JA, et al. Morphine inhibits migration of tumor-infiltrating leukocytes and suppresses angiogenesis associated with tumor growth in mice. Am J Pathol. 2014;184(4):1073–84.
Tai YH, Wu HL, Chang WK, Tsou MY, Chen HH, Chang KY. Intraoperative fentanyl consumption does not impact cancer recurrence or overall survival after curative colorectal cancer resection. Sci Rep. 2017;7(1):1–8.
Gaspani L, Bianchi M, Limiroli E, Panerai AE, Sacerdote P. The analgesic drug tramadol prevents the effect of surgery on natural killer cell activity and metastatic colonization in rats. J Neuroimmunol. 2002;129(1–2):18–24.
Mathew B, Lennon FE, Siegler J, et al. The novel role of the mu opioid receptor in lung cancer progression: a laboratory investigation. Anesth Analg. 2011;112(3):558–67.
Byrne K, Levins KJ, Buggy DJ. Can anesthetic-analgesic technique during primary cancer surgery affect recurrence or metastasis? Can J Anesth Can d’anesthésie. 2016;63(2):184–92.
Nguyen J, Luk K, Vang D, et al. Morphine stimulates cancer progression and mast cell activation and impairs survival in transgenic mice with breast cancer. Br J Anaesth. 2014;113(1):i4–13.
Lec PM, Lenis AT, Golla V, et al. The role of opioids and their receptors in urological malignancy: a review. J Urol. 2020;204(6):1150–9.
Li Y, Li G, Tao T, et al. The μ-opioid receptor (MOR) promotes tumor initiation in hepatocellular carcinoma. Cancer Lett. 2019;453:1–9.
Zhang H, Sun M, Zhou D, et al. Increased mu-opioid receptor expression is associated with reduced disease-free and overall survival in laryngeal squamous cell carcinoma. Br J Anaesth. 2020;125(5):722–9. This clinical trial retrospectively shows that high mu-opioid receptor expression is associated with poor prognosis in patients with laryngeal squamous cell carcinoma.
Steele GL, Dudek AZ, Gilmore GE, et al. Impact of pain, opioids, and the mu-opioid receptor on progression and survival in patients with newly diagnosed stage IV pancreatic cancer. Am J Clin Oncol Cancer Clin Trials. 2020;43(8):591–7.
Forget P, Tombal B, Scholtès JL, et al. Do intraoperative analgesics influence oncological outcomes after radical prostatectomy for prostate cancer? Eur J Anaesthesiol. 2011;28(12):830–5.
Akbari M, Naghibzadeh-Tahami A, Khanjani N, et al. Opium as a risk factor for bladder cancer: a population-based case-control study in Iran. Arch Iran Med. 2015;18(9):567–71.
MacFater WS, Rahiri JL, Lauti M, Su’a B, Hill AG. Intravenous lignocaine in colorectal surgery: a systematic review. ANZ J Surg. 2017;87(11):879–85.
Ortiz MP, de Godoy MC M, Schlosser RS, et al. Effect of endovenous lidocaine on analgesia and serum cytokines: double-blinded and randomized trial. J Clin Anesth. 2016;35:70–7.
Herroeder S, Pecher S, Schönherr ME, et al. Systemic lidocaine shortens length of hospital stay after colorectal surgery. Ann Surg. 2007;246(2):192–200.
Cata JP, Bauer M, Sokari T, et al. Effects of surgery, general anesthesia, and perioperative epidural analgesia on the immune function of patients with non-small cell lung cancer. J Clin Anesth. 2013;25(4):255–62.
Ramirez MF, Tran P, Cata JP. The effect of clinically therapeutic plasma concentrations of lidocaine on natural killer cell cytotoxicity. Reg Anesth Pain Med. 2015;40(1):43–8.
Lucchinetti E, Awad AE, Rahman M, et al. Antiproliferative effects of local anesthetics on mesenchymal stem cells: potential implications for tumor spreading and wound healing. Anesthesiol. 2012;116(4):841–56.
Werdehausen R, Braun S, Fazeli S, et al. Lipophilicity but not stereospecificity is a major determinant of local anaesthetic-induced cytotoxicity in human T-lymphoma cells. Eur J Anaesthesiol. 2012;29(1):35–41.
Lirk P, Berger R, Hollmann MW, Fiegl H. Lidocaine time- and dose-dependently demethylates deoxyribonucleic acid in breast cancer cell lines in vitro. Br J Anaesth. 2012;109(2):200–7.
Matos AC, Marques IA, Pires AS, Valentim A, Abrantes AM, Botelho MF. The potential effect of lidocaine, ropivacaine, levobupivacaine and morphine on breast cancer pre-clinical models: a systematic review. Int J Mol Sci. 2022;23(3):1894.
Makarova M, Rycek L, Hajicek J, Baidilov D, Hudlicky T. Tetrodotoxin: History, biology, and synthesis. Angew Chemie. 2019;657(1–3):51–8.
Castelli V, Piroli A, Marinangeli F, et al. Local anesthetics counteract cell proliferation and migration of human triple-negative breast cancer and melanoma cells. J Cell Physiol. 2019;284(12):8114–26.
Pang QY, Duan LP, Jiang Y, Liu HL. Comparison of outcomes after breast cancer surgery between inhalational and propofol-based intravenous anaesthesia: a systematic review and meta-analysis. J Pain Res. 2021;14:2165–77.
Sun H, Sun Y. Lidocaine inhibits proliferation and metastasis of lung cancer cell via regulation of miR-539/EGFR axis. Artif Cells, Nanomedicine Biotechnol. 2019;47(1):2866–74.
Xing W, Chen DT, Pan JH, et al. Lidocaine Induces apoptosis and suppresses tumor growth in human hepatocellular carcinoma cells in vitro and in a xenograft model in vivo. Anesthesiol. 2017;126(5):868–81.
Xuan W, Zhao H, Hankin J, Chen L, Yao S, Ma D. Local anesthetic bupivacaine induced ovarian and prostate cancer apoptotic cell death and underlying mechanisms in vitro. Sci Rep. 2016;6(1):1–12.
Liu H, Dilger JP, Lin J. The role of transient receptor potential melastatin 7 (TRPM7) in cell viability: a potential target to suppress breast cancer cell cycle. Cancers. 2020;12(1):131.
Leng T, Lin S, Xiong Z, Lin J. Lidocaine suppresses glioma cell proliferation by inhibiting TRPM7 channels. Int J Physiol Pathophysiol Pharmacol. 2017;9(2):8–15.
Raff AB, Thomas CN, Chuang GS, et al. Lidocaine-induced potentiation of thermal damage in skin and carcinoma cells. Lasers Surg Med. 2019;51(1):88–94.
Wall TP, Buggy DJ. Perioperative intravenous lidocaine and metastatic cancer recurrence - a narrative review. Front Oncol. 2021;11(August):1–16.
Zhang H, Yang L, Zhu X, et al. Association between intraoperative intravenous lidocaine infusion and survival in patients undergoing pancreatectomy for pancreatic cancer: a retrospective study. Br J Anaesth. 2020;125(2):141–8. This retrospective study included 2239 patients suggesting that intraoperative intravenous lidocaine infusion is associated with improved overall survival in patients undergoing pancreatectomy.
Ackerman RS, Luddy KA, Icard BE, Piñeiro Fernández J, Gatenby RA, Muncey AR. The effects of anesthetics and perioperative medications on immune function: a narrative review. Anesth Analg. 2021;133(3):676–89.
Beilin B, Rusabrov Y, Shapira Y, et al. Low-dose ketamine affects immune responses in humans during the early postoperative period. Br J Anaesth. 2007;99(4):522–7.
Hou M, Zhou NB, Li H, et al. Morphine and ketamine inhibit immune function of gastric cancer patients by increasing percentage of CD4(+)CD25(+)Foxp3(+) regulatory T cells in vitro. J Surg Res. 2016;203(2):306–12.
Forget P, Vandenhende J, Berliere M, et al. Do intraoperative analgesics influence breast cancer recurrence after mastectomy? A retrospective analysis Anesth Analg. 2010;110(6):1630–5.
Howe LR, Subbaramaiah K, Chung WJ, Dannenberg AJ, Brown AM. Transcriptional activation of cyclooxygenase-2 in Wnt-1-transformed mouse mammary epithelial cells. Cancer Res. 1999;59(7):1572–7.
Howe LR, Subbaramaiah K, Brown AM, Dannenberg AJ. Cyclooxygenase-2: a target for the prevention and treatment of breast cancer. Endocr Relat Cancer. 2001;8(2):97–114.
Yakar I, Melamed R, Shakhar G, et al. Prostaglandin E 2 suppresses NK activity in vivo and promotes postoperative tumor metastasis in rats. Ann Surg Oncol. 2003;10(4):469–79.
Singh-Ranger G, Salhab M, Mokbel K. The role of cyclooxygenase-2 in breast cancer: review. Breast Cancer Res Treat. 2008;109(2):189–98.
Retsky M, Rogers R, Demicheli R, et al. NSAID analgesic ketorolac used perioperatively may suppress early breast cancer relapse: particular relevance to triple negative subgroup. Breast Cancer Res Treat. 2012;134(2):881–8.
Forget P, Bentin C, Machiels JP, Berliere M, Coulie PG, De Kock M. Intraoperative use of ketorolac or diclofenac is associated with improved disease-free survival and overall survival in conservative breast cancer surgery. Br J Anaesth. 2014;113(1):i82–7.
Yuki K. The immunomodulatory mechanism of dexmedetomidine. Int Immunopharmacol. 2021;97(April):107709.
Montejano J, Jevtovic-Todorovic V. Anesthesia and cancer, friend or foe? A narrative review Front Oncol. 2021;11(December):1–7.
Wang K, Wu M, Xu J, et al. Effects of dexmedetomidine on perioperative stress, inflammation, and immune function: systematic review and meta-analysis. Br J Anaesth. 2019;123(6):777–94.
Yuki K, Soriano SG, Shimaoka M. Sedative drug modulates T-cell and lymphocyte function-associated antigen-1 function. Anesth Analg. 2011;112(4):830–8.
Cho JS, Seon K, Kim MY, Kim SW, Yoo YC. Effects of perioperative dexmedetomidine on immunomodulation in uterine cancer surgery: a randomized, controlled trial. Front Oncol. 2021;11(November):1–9.
Bruzzone A, Piñero CP, Castillo LF, et al. α 2-Adrenoceptor action on cell proliferation and mammary tumour growth in mice. Br J Pharmacol. 2008;155(4):494–504.
Lavon H, Matzner P, Benbenishty A, et al. Dexmedetomidine promotes metastasis in rodent models of breast, lung, and colon cancers. Br J Anaesth. 2018;120(1):188–96. In this study, they confirmed that dexmedetomidine increased tumor-cell retention and growth of metastases in rodent models of breast, lung, and colon cancers.
Szpunar MJ, Burke KA, Dawes RP, Brown EB, Madden KS. The antidepressant desipramine and α2-adrenergic receptor activation promote breast tumor progression in association with altered collagen structure. Cancer Prev Res. 2013;6(12):1262–72.
Cata J, Singh V, Lee B, et al. Intraoperative use of dexmedetomidine is associated with decreased overall survival after lung cancer surgery. J Anaesthesiol Clin Pharmacol. 2017;33(3):317–23.
Kim MH, Oh JE, Park S, et al. Tramadol use is associated with enhanced postoperative outcomes in breast cancer patients: a retrospective clinical study with in vitro confirmation. Br J Anaesth. 2019;123(6):865–76. This research provided both preclinical and retrospective clinical evidence to suggest that tramadol appears anti-tumor effects on breast cancer by inhibiting proliferation and induction of apoptosis.
Kim MH, Lee JR, Kim KJ, et al. Identification for antitumor effects of tramadol in a xenograft mouse model using orthotopic breast cancer cells. Sci Rep. 2021;11(1):1–8.
Edinoff AN, Kaplan LA, Khan S, et al. Full opioid agonists and tramadol: pharmacological and clinical considerations. Anesthesiol pain Med. 2021;11(4):e119156.
Pérez-González O, Cuéllar-Guzmán LF, Soliz J, Cata JP. Impact of regional anesthesia on recurrence, metastasis, and immune response in breast cancer surgery: a systematic review of the literature. Reg Anesth Pain Med. 2017;42(6):751–6.
Zhang Y, Peng X, Zheng Q. Ropivacaine inhibits the migration of esophageal cancer cells via sodium-channel-independent but prenylation-dependent inhibition of Rac1/JNK/paxillin/FAK. Biochem Biophys Res Commun. 2018;501(4):1074–9.
Wang L, Liang S, Chen H, Xu Y, Wang Y. The effects of epidural anaesthesia and analgesia on T lymphocytes differentiation markers and cytokines in patients after gastric cancer resection. BMC Anesthesiol. 2019;19(1):1–7.
Kun L, Tang L, Wang J, Yang H, Ren J. Effect of combined general/epidural anesthesia on postoperative nk cell activity and cytokine response in gastric cancer patients undergoing radical resection. Hepatogastroenterol. 2014;61(132):1142–7.
Chen WK, Miao CH. The effect of anesthetic technique on survival in human cancers: a meta-analysis of retrospective and prospective studies. PLoS One. 2013;8(2):e56540.
Sun X, Yang C, Li K, Ding S. The impact of anesthetic techniques on survival for patients with colorectal cancer: evidence based on six studies. Hepatogastroenterol. 2015;62(138):299–302.
Lee BM, Singh Ghotra V, Karam JA, Hernandez M, Pratt G, Cata JP. Regional anesthesia/analgesia and the risk of cancer recurrence and mortality after prostatectomy: a meta-analysis. Pain Manag. 2015;5(5):387–95.
Jang D, Lim CS, Shin YS, Ko YK, Park SI, Song SH, Kim BJ. A comparison of regional and general anesthesia effects on 5 year survival and cancer recurrence after transurethral resection of the bladder tumor: a retrospective analysis. BMC Anesthesiol. 2015;16(1):1–6.
Zimmitti G, Soliz J, Aloia TA, et al. Positive impact of epidural analgesia on oncologic outcomes in patients undergoing resection of colorectal liver metastases. Ann Surg Oncol. 2016;23(3):1003–11.
Sessler DI, Pei L, Huang Y, et al. Recurrence of breast cancer after regional or general anaesthesia: a randomised controlled trial. Lancet. 2019;394(10211):1807–15. This study is the first well-established large-scale multicentre randomized controlled trial that shows regional anesthesia-analgesia does not reduce breast cancer recurrence after potentially curative surgery compared with volatile anesthesia and opioids
Starnes-Ott K, Goravanchi F, Meininger JC. Anesthetic choices and breast cancer recurrence: a retrospective pilot study of patient, disease, and treatment factors. Crit Care Nurs Q. 2015;38(2):200–10.
Xu ZZ, Li HJ, Li MH, Huang SM, Li X, Liu QH, Li J, Li XY, Wang DX, Sessler DI. Epidural anesthesia-analgesia and recurrence-free survival after lung cancer surgery: a randomized trial. Anesthesiology. 2021;135(3):419–32.
Myles PS, Peyton P, Silbert B, Hunt J, Rigg JRA, Sessler DI. Perioperative epidural analgesia for major abdominal surgery for cancer and recurrence-free survival: randomised trial. BMJ. 2011;342(7800):746.
Tsui BCH, Rashiq S, Schopflocher D, et al. Epidural anesthesia and cancer recurrence rates after radical prostatectomy. Can J Anesth. 2010;57(2):107–12.
O’Riain SC, Buggy DJ, Kerin MJ, Watson RWG, Moriarty DC. Inhibition of the stress response to breast cancer surgery by regional anesthesia and analgesia does not affect vascular endothelial growth factor and prostaglandin E 2. Anesth Analg. 2005;100(1):244–9.
Appenheimer MM, Evans SS. Temperature and adaptive immunity. Handb Clin Neurol. 2018;156:397–15.
Shao L, Pang N, Yan P, et al. Control of body temperature and immune function in patients undergoing open surgery for gastric cancer. Bosn J Basic Med Sci. 2018;18(3):289–96.
Kurz A, Sessler DI, Lenhardt R. Perioperative normothermia to reduce the incidence of surgical-wound infection and shorten hospitalization. N Engl J Med. 1996;334(19):1209–15.
Ben-Eliyahu S, Shakhar G, Rosenne E, Levinson Y, Beilin B. Hypothermia in barbiturate-anesthetized rats suppresses natural killer cell activity and compromises resistance to tumor metastasis: a role for adrenergic mechanisms. Anesthesiol. 1999;91(3):732–40.
Hooper VD, Chard R, Clifford T, et al. ASPAN’s evidence-based clinical practice guideline for the promotion of perioperative normothermia: second edition. J perianesthesia Nurs Off J Am Soc PeriAnesthesia Nurses. 2010;25(6):346–65.
Yücel Y, Barlan M, Lenhardt R, Kurz A, Sessler DI. Perioperative hypothermia does not enhance the risk of cancer dissemination. Am J Surg. 2005;189(6):651–5. In this study, they found that mild perioperative hypothermia had no effects on cancer-free and overall survival rates.
Morozumi K, Mitsuzuka K, Takai Y, et al. Intraoperative hypothermia is a significant prognostic predictor of radical cystectomy especially for stage II muscle-invasive bladder cancer. Med (United States). 2019;98(2):E13962.
Yu H, Luo Y, Peng H, et al. The predicting value of postoperative body temperature on long-term survival in patients with rectal cancer. Tumor Biol. 2015;36(10):8055–63.
Zheng HL, Lu J, Li P, et al. Effect of high postoperative body temperature on long-term prognosis in patients with gastric cancer after radical resection. World J Surg. 2019;43(7):1756–65.
Repasky EA, Evans SS, Dewhirst MW. Temperature matters! And why it should matter to tumor immunologists. Cancer Immunol Res. 2013;1(4):210–6.
Tang X, Cao F, Ma W, et al. Cancer cells resist hyperthermia due to its obstructed activation of caspase 3. Reports Pract Oncol Radiother. 2020;25(3):323–6.
Miao L, Lv X, Huang C, Li P, Sun Y, Jiang H. Long-term oncological outcomes after oral cancer surgery using propofol-based total intravenous anesthesia versus sevoflurane-based inhalation anesthesia: a retrospective cohort study. Danan D, ed. PLoS One. 2022;17(5):e0268473.
Wu WW, Zhang WH, Zhang WY, et al. The long-term survival outcomes of gastric cancer patients with total intravenous anesthesia or inhalation anesthesia: a single-center retrospective cohort study. BMC Cancer. 2021;21(1):1–12.
Huang YH, Wu ZF, Lee MS, et al. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in glioblastoma surgery. PLoS One. 2021;16(8):1–13.
Makito K, Matsui H, Fushimi K, Yasunaga H. Volatile versus total intravenous anesthesia for cancer prognosis in patients having digestive cancer surgery. Anesthesiology. 2020;133(4):764–73.
Huang NC, Lee MS, Lai HC, et al. Propofol-based total intravenous anesthesia improves survival compared to desflurane anesthesia in gastric cancer surgery: a retrospective analysis. Medicine (Baltimore). 2020;99(25):e20714.
Pfail JL, Katims AB, Gul Z, et al. Can anesthetics affect bladder cancer recurrence? Total intravenous versus volatile anesthesia in patients undergoing robot-assisted radical cystectomy: a single institution retrospective analysis. Urol Oncol Semin Orig Investig. 2021;39(4):233.e1-233.e8.
Lai HC, Lee MS, Liu YT, et al. Propofol-based intravenous anesthesia is associated with better survival than desflurane anesthesia in pancreatic cancer surgery. PLoS ONE. 2020;15(5):1–14.
Chang CY, Wu MY, Chien YJ, Su IM, Wang SC, Kao MC. Anesthesia and long-term oncological outcomes: a systematic review and meta-analysis. Anesth Analg. 2021;132(3):623–34.
Cata JP, Nguyen LT, Ifeanyi-Pillette IC, et al. An assessment of the survival impact of multimodal anesthesia/analgesia technique in adults undergoing cytoreductive surgery with hyperthermic intraperitoneal chemotherapy: a propensity score matched analysis. Int J Hyperth. 2019;36(1):369–75.
Lai HC, Lee MS, Lin KT, Chan SM, Chen JY, Lin YT, Wu ZF. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in intrahepatic cholangiocarcinoma surgery. Medicine. 2019;98(51):e18472
Oh TK, Kim HH, Jeon YT. Retrospective analysis of 1-year mortality after gastric cancer surgery: total intravenous anesthesia versus volatile anesthesia. Acta Anaesthesiol Scand. 2019;63(9):1169–77.
Yoo S, Lee HB, Han W, et al. Total intravenous anesthesia versus inhalation anesthesia for breast cancer surgery: a retrospective cohort study. Anesthesiol. 2019;130(1):31–40.
Lai HC, Lee MS, Lin C, et al. Propofol-based total intravenous anaesthesia is associated with better survival than desflurane anaesthesia in hepatectomy for hepatocellular carcinoma: a retrospective cohort study. Br J Anaesth. 2019;123(2):151–60.
Hong B, Lee S, Kim Y, Lee M, Youn AM, Rhim H, Hong SH, Kim YH, Yoon SH, Lim C. Anesthetics and long-term survival after cancer surgery-total intravenous versus volatile anesthesia: a retrospective study. BMC Anesthesiol. 2019;19(1):1–10.
Yap A, Lopez-Olivo MA, Dubowitz J, et al. Anesthetic technique and cancer outcomes: a meta-analysis of total intravenous versus volatile anesthesia. Can J Anesth. 2019;66(5):546–61.
Yan T, Zhang GH, Wang BN, Sun L, Zheng H. Effects of propofol/remifentanil-based total intravenous anesthesia versus sevoflurane-based inhalational anesthesia on the release of VEGF-C and TGF-β and prognosis after breast cancer surgery: a prospective, randomized and controlled study. BMC Anesthesiol. 2018;18(1):1–9.
Zheng X, Wang Y, Dong L, et al. Effects of propofol-based total intravenous anesthesia on gastric cancer: a retrospective study. Onco Targets Ther. 2018;11:1141–8.
Oh TK, Kim K, Jheon S, Lee J, Do SH, Hwang JW, Song IA. Long-term oncologic outcomes for patients undergoing volatile versus intravenous anesthesia for non-small cell lung cancer surgery: a retrospective propensity matching analysis. Cancer control. 2018;25(1):1073274818775360.
Cata JP, Hagan KB, Bhavsar SDO, et al. The use of isoflurane and desflurane as inhalational agents for glioblastoma surgery. A survival analysis J Clin Neurosci. 2017;35:82–7.
Xu Q, Shi NJ, Zhang H, Zhu YM. Effects of combined general-epidural anesthesia and total intravenous anesthesia on cellular immunity and prognosis in patients with non-small cell lung cancer: a comparative study. Mol Med Rep. 2017;16(4):4445–54.
Du YT, Li YW, Zhao BJ, Guo XY, Feng Y, Zuo MZ, Fu C, Zhou WJ, Li HJ, Liu YF, Cheng T, Mu DL, Zeng Y, Liu PF, Li Y, An HY, Zhu SN, Li XY, Li HJ, Wu YF, Wang DX, Sessler DI. Long-term survival after combined epidural-general anesthesia or general anesthesia alone: follow-up of a randomized trial. Anesthesiology. 2021;135(2):233–45.
Gao H, Meng XY, Wang HQ, et al. Association between anaesthetic technique and oncological outcomes after colorectal carcinoma liver metastasis resection. Int J Med Sci. 2019;16(2):337–42.
Tai YH, Chang WK, Wu HL, Chan MY, Chen HH, Chang KY. The effect of epidural analgesia on cancer progression in patients with stage IV colorectal cancer after primary tumor resection: a retrospective cohort study. PLoS One. 2018;13(7):e0200893.
Koumpan Y, Jaeger M, Mizubuti GB, et al. Spinal anesthesia is associated with lower recurrence rates after resection of nonmuscle invasive bladder cancer. J Urol. 2018;199(4):940–6.
Tseng JH, Cowan RA, Afonso AM, et al. Perioperative epidural use and survival outcomes in patients undergoing primary debulking surgery for advanced ovarian cancer. Gynecol Oncol. 2018;151(2):287–93.
Cata JP, Bhavsar S, Hagan KB, et al. Scalp blocks for brain tumor craniotomies: a retrospective survival analysis of a propensity match cohort of patients. J Clin Neurosci. 2018;51:46–51.
Karmakar MK, Samy W, Lee A, et al. Survival analysis of patients with breast cancer undergoing a modified radical mastectomy with or without a thoracic paravertebral block: a 5-year follow-up of a randomized controlled trial. Anticancer Res. 2017;37(10):5813–20.
Wang Y, Wang L, Chen H, Xu Y, Zheng X, Wang G. The effects of intra- and post-operative anaesthesia and analgesia choice on outcome after gastric cancer resection: a retrospective study. Oncotarget. 2017;8(37):62658–65.
Choi WJ, Baek S, Joo EY, et al. Comparison of the effect of spinal anesthesia and general anesthesia on 5-year tumor recurrence rates after transurethral resection of bladder tumors. Oncotarget. 2017;8(50):87667–74.
Lee EK, Ahn HJ, Zo JI, Kim K, Jung DM, Park JH. Paravertebral block does not reduce cancer recurrence, but is related to higher overall survival in lung cancer surgery: a retrospective cohort study. Anesth Analg. 2017;125(4):1322–8.
Zheng L, Hagan KB, Villarreal J, Keerty V, Chen J, Cata JP. Scalp block for glioblastoma surgery is associated with lower inflammatory scores and improved survival. Minerva Anestesiol. 2017;83(11):1137–45.
Funding
This research was supported by the Wu Jieping Medical Foundation (No. 320.6750.2020–21-19), the Yangfan Plan of Shanghai Science and Technology Commission (No. 22YF1439600), and the China Postdoctoral Science Foundation (No. 2022M710771).
Author information
Authors and Affiliations
Contributions
All authors wrote, revised, and finalized the manuscripts for publication.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical collection on Anesthesiology and Critical Care
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xia, Sh., Zhou, D., Ge, F. et al. Influence of Perioperative Anesthesia on Cancer Recurrence: from Basic Science to Clinical Practice. Curr Oncol Rep 25, 63–81 (2023). https://doi.org/10.1007/s11912-022-01342-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11912-022-01342-9