Abstract
The distinct origin and hydrological characteristics of reservoirs, shaped by fluctuating water levels and seasonal variations, play a crucial role in determining aquatic species assemblages across diverse limnological zones. This study aimed to analyze fish and crayfish assemblages in the nearshore area of a canyon-shaped Mediterranean reservoir, seeking to identify seasonal and spatial convergent patterns and infer the factors influencing them. Samplings were conducted at five stations along the longitudinal profile of the reservoir at depths around 7 m using gillnets and hoops nets. A seasonal pattern emerged regarding species abundance and biomass, with higher values recorded during the warmer period. Additionally, a spatial trend was observed, indicating higher species abundance in the middle part of the reservoir and lower abundance near the dam, particularly during the colder period. Water transparency, temperature, and the distance from the dam were identified as the most significant factors affecting species assemblages in terms of abundance and biomass. This research provides valuable insights into the intricate interplay between environmental factors, seasonal changes, and the assemblages of aquatic species in a Mediterranean reservoir, contributing to our understanding of its ecological dynamics.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The unique origin and hydrological characteristics of reservoirs, which share similarities with both rivers and lakes (Gido et al. 2009), can have a significant impact on aquatic organism assemblages. The act of damming disrupts river continuity, creating physical barriers that constrain the movement of fish both upstream and downstream. This disruption particularly affects the migration routes of long-distance migratory species, such as those belonging to the genus Anguilla and Salmo, leading to population isolation and extirpation (Hughes et al. 2005; Araújo et al. 2008).
Furthermore, damming alters the hydrological conditions in the river valley, leading to shorter retention times, higher water level fluctuation, and increased organic loads. These alterations favor the dominance of lentic species over lotic ones (Kubečka 1993; Irz et al. 2002; Hughes et al. 2005). Consequently, the fish assemblage that originally inhabited the river undergoes substantial modifications. Additionally, reservoirs with a canyon-shaped structure, featuring steep banks that result in restricted littoral areas and a limited diversity of habitats (Veselý et al. 2020), also impact the prevailing fish fauna.
However, even years after the reservoir construction, which initially led to the described significant changes, species assemblages continue to differ spatially and temporally. In general, fish are not randomly distributed within a water body. Instead, their assemblages result from a complex interplay of environmental factors, including physical and chemical conditions (such as water depth, temperature, oxygen levels, proximity to inlets/outlets) (Godinho et al. 1998; Vašek et al. 2004; Prchalová et al. 2008), biotic interactions (such as competition, predation, human activities) (Prchalová et al. 2008), preferences for specific microhabitats, spawning seasons, food availability, and the presence of cover and structure (such as submerged rocks, vegetation, artificial structures) (Choi and Kim 2020). Water quality also plays a crucial role in shaping their communities (Dustin and Vondracek 2017; Hatcher et al. 2019). Similarly, crayfish distribution in reservoirs is influenced by factors such as predation, resource, and shelter availability, as well as physicochemical parameters and their intricate interactions (Veselý et al. 2020).
In reservoirs, fish distribution can vary spatially along the longitudinal profile of the reservoir (i.e., among the present limnological zones: dam zone, middle zone, and upstream zone), vertically (along the water column), or among the littoral, profundal, and pelagic zones (Prchalová et al. 2008, 2009; Yang et al. 2012; Becker et al. 2016). The dam zone (or lacustrine zone), characterized by greater depth, exhibits more lake-like features, the upstream zone displays mainly riverine characteristics, and the middle zone is marked by intermediate environmental conditions, thus supporting differences in fish assemblages. Vertical distribution is influenced by thermal stratification, with fish favoring surface depths with adequate dissolved oxygen concentrations while avoiding layers with lower temperatures and hypoxia beneath the thermocline (Vašek et al. 2004; Järvalt et al. 2005; Prchalová et al. 2008). Concerning the littoral and profundal zones, while conditions vary, a common pattern is the movement of fish to shallower shoreline areas for spawning in spring, as well as for nesting and feeding (García-Berthou 2001; Winfield 2004; Penne and Pierce 2008; Vander Zanden et al. 2011). Conversely, species favoring warmer waters, such as catfish, may become more active in warmer, shallower areas as temperature rises (Alp et al. 2004; Ferreira 2019; Santos 2021). In autumn, fish may exhibit increased feeding activity in preparation for winter, with predatory species like pike or pikeperch becoming more active (Vehanen and Lahti 2003; Říha et al. 2022). During winter, most fish species are likely found in deeper areas where temperatures are more stable (Vehanen and Lahti 2003; Penne and Pierce 2008). Concerning crayfish, their habitat utilization extends across both littoral areas and the deepest zones. The selection of these habitats is influenced by environmental suitability and the fulfillment of their ecological needs (Reynolds et al. 2013; Veselý et al. 2020).
The research explores the composition of fish and crayfish species in the nearshore zone of a canyon-shaped Mediterranean reservoir, seeking spatiotemporal patterns and trends and investigating the primary environmental factors influencing them. This knowledge is crucial for understanding ecological dynamics of these reservoirs, providing valuable insights for environmental management and design. Such understanding is instrumental in guiding spatial planning for conservation or protection areas, aligning with the overarching goals of environmental sustainability and preservation (Hughes et al. 2005; Wang et al. 2006).
Materials and methods
Study area
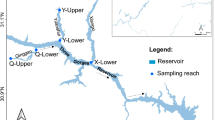
The Polyphytos Reservoir is located in the municipality of Kozani, in the northwest of Greece. Established in 1975 by the Public Power Corporation, it was created by damming the Aliakmon River to facilitate electrification through hydroelectric power. Multiple reservoirs (Ilariona, Polyphytos, Sfikias, Asomaton, and Ag. Varvaras) have been installed along the main course of the Aliakmon River, disrupting its continuity and modifying hydrological conditions over extensive sections (Fig. 1).
The drainage basin of the reservoir encompasses an area of 5630 km2. The reservoir has a narrow and elongated shape (canyon-shaped), with a maximum width of 2.5 km, and its length varies between 22 and 31 km, depending on the balance of inflows and outflows. Its useful capacity is 1220 × 106 m3, its maximum operational level is at + 291 m a.s.l., its surface area is 74.1 km2, and its length is 4.5 km (Ministry of Environment and Energy, 2017b). Seasonally, the water level fluctuates by approximately 15 m. The average depth is approximately 26.2 m, with a maximum depth of 91 m. The water renewal time is estimated at 1.5 times per year. It is a warm monomictic reservoir. During the summer period, it displays thermal stratification, with the oxygen concentration in the thermocline lower than in the hypolimnion. The epilimnion extends up to a depth of 10 m, while the thermocline zone has a width of 7 m. It is characterized as mesotrophic with tendencies toward eutrophication. The richness and abundance of aquatic macrophytes in its littoral zone are low due to high water level fluctuations.
Samplings
A series of four seasonal samplings (spring to winter 2022) was carried out at Polyphytos Reservoir across five stations along its longitudinal axis (Fig. 1). These sampling points were strategically positioned in depths around 7 m, in close proximity to the reservoir’s littoral zone, owing to its abrupt bathymetric features. Specifically, stations positions were moved a few meters each season, so that water depths remained approximately constant.
Water temperature (WT, °C), dissolved oxygen concentrations (DO, mg/l) in the surface and the reservoir’s bottom, pH, electrical conductivity (EC, μS/cm), salinity (Sal, ppt), and total dissolved solids (TDS, mg/l) were measured in the surface water, at each sampling station, using a YSI ProDSS (digital sampling system) multiparameter probe. Additionally, water transparency (m) was determined using a Secchi disk.
Samplings of fish and crayfish were conducted using benthic gill nets and hoop nets (traps). The gill nets were 25 m long and 6 m high, made of yellow thread, consisting of ten different segments with mesh sizes ranging from 6.5 to 60 mm. The hoop nets were 8 m long, with dimensions of 40 cm × 22 cm and a net mesh size of 9 mm. No bait was used in the traps to attract aquatic organisms. One fishing tool of each type (i.e., one gillnet and one trap) was placed at each station. The fishing gear was retrieved the next morning (i.e., the fishing duration was approximately 20 h).
Catches were identified at the species level. The total length (mm) and weight (g) of each fish were measured, while only weight was recorded for the crayfish.
Data analysis
Comparisons of environmental variables and catch abundances (specimens’ number and biomass) among seasons were performed. After checking for homoscedastic and normality’s parametric statistical test requirements, the non-parametric Kruskal–Wallis test was used to check for differences in parameters’ values (estimated from five sampling stations) among seasons.
In addition, a Spearman correlation analysis was conducted using values of the physicochemical parameters, distance from the dam (measured as the straight distance of each station from the dam using Google earth, imagery date: 15/7/2023), species number, and total catches (fish and crayfish) in terms of abundance and biomass to detect multicollinearity among parameters.
A subset of independent variables that were not highly correlated to each other was selected to be included in further analysis. Specifically, those were water temperature, Secchi disc depth (transparency), conductivity, and the distance of the sampling station from the dam. All the above analyses were conducted using IBM SPSS Statistics (version 27). The direct gradient canonical correspondence analysis (CCA) was then used for the evaluation of the variability in assemblage structure in relation to the selected environmental factors. Both abundance and biomass species data were used, specifically catches per season and sampling station. The significance of the CCA result was assessed through an analysis of variance (ANOVA) designed for CCA using “vegan” package of R-studio (Oksanen et al. 2007). All data used for the above analyses were log(x + 1) transformed.
Results
Water’s physicochemical parameters
Descriptive statistics of the physicochemical parameters measured in the water per season are presented in Table 1. The highest mean water temperature was recorded during the summer sampling, while the lowest in winter. Dissolved oxygen concentrations remained consistently high throughout the year exceeding 8 mg/l, at both the surface and bottom of the reservoir. Conductivity and pH values did not show significant variations among seasons. In autumn, the highest salinity (Sal) and total dissolved solid (TDS) values were recorded, reaching up to two- to threefold compared to other seasons. Secchi disk depth values ranged from 2 to 5 m, with the highest mean transparency values recorded in spring (4.2 ± 0.374 m) and the lowest in autumn (2.82 ± 0.220 m). Specifically, statistically significant differences (p < 0.001) were detected among seasons for temperature, DO, Sal, TDS, and Secchi depth, but not for EC and pH.
Species assemblages
In total, 5102 individuals of fish and crayfish were caught using gillnets and hoop nets, with a total weight of 127.17 Kg. The presence of nine fish species belonging to six families and one species of crayfish, Pontastacus leptodactylus (Family Astacidae), was recorded (Fig. 2). The most abundant species based on the number of individuals was Rutilus rutilus, followed by Perca fluviatilis (Fig. 2a). Based on biomass, the most abundant species was Rutilus rutilus, followed by Alburnus thessalicus (Fig. 2b). Species such as Cyprinus carpio, Esox lucius, Silurus glanis, Squalius vardarensis, and Vimba melanops had relatively low specimen counts (≥ 10). A total of 153 crayfish were captured using both gillnets and hoop nets, with approximately 95% of this species caught by hoop nets. In contrast, gillnet fishing contributed more substantially to the overall fish catch.
The highest number of species was recorded in the catches during summer (nine species), while the lowest was observed in winter (six species) (Fig. 3). The highest total catch in terms of numerical abundance and biomass occurred in summer, followed by spring, with the lowest catches observed in winter (Fig. 3). Species abundance and number did not differ significantly (p > 0.05) across seasons. The opposite, however, was observed for biomass (p < 0.05).
Seasonal (left) and spatial (right) distribution of catches estimated in terms of a species, b numerical abundance, and c biomass in Polyphytos Reservoir during the sampling campaign in 2022. Mean values (x), median values (represented by horizontal lines), 50% of the values (box), and the highest and lowest values (outer lines) are provided. The numbers above the boxes indicate the total number of species, numerical abundance, and biomass catch per season or sampling station
The highest number of species was recorded at sampling station S3, while the highest catches and biomass were observed at sampling station S2, indicating that both parameters were higher in the middle section of the reservoir. In contrast, the smallest values were estimated from the catches in the “lower” section of the reservoir concerning its longitudinal profile, specifically at stations S4 and S5, which were closer to the dam (Fig. 3).
In general, all species exhibited lower abundance and biomass catches during winter (Fig. 4), except for E. lucius, which showed its highest catch during that period, and R. rutilus, whose catches during winter remained at the same level as in autumn. C. carpio, A. thessalicus, and R. rutilus had their highest catches during spring. P. fluviatilis recorded the highest catch during summer as crayfish P. leptodactylus, while S. glanis was not recorded in winter catches and exhibited the highest catch in summer.
Factors affecting species assemblages
Spearman correlation revealed significant correlations between water temperature and DO, pH, Sal, TDS as long as abundance, biomass, and species number, proving that the seasonal shift is a major factor affecting the species assemblages of this nearshore zone (Table 2). However, the distance of the sampling station from the dam (distance from dam) was not correlated with either species number nor abundance or biomass data.
The first two CCA dimensions (CCA1 and CCA2) indicated that environmental variables accounted for approximately 26.6% of the total variation in species assemblages concerning abundance data and 24.8% concerning biomass data (Table 3). In addition, ANOVA revealed a statistically significant relationship between species assemblages and the environmental variables used, considering both numerical abundances and biomass data (Fig. 5). Specifically, the CCA models showed that the variables most significantly affecting species abundance were the Secchi disc (indicating water transparency) in the first axis (CCA1) and the distance from the dam in CCA2 axis (Table 3, Fig. 5a). On the other hand, for species biomass, Secchi depth (CCA1) and temperature (CCA2) had the most pronounced impact (Table 3, Fig. 5b).
Plot of CCA analysis showing the correspondence of the main environmental parameters (temperature (temp) and distance from the dam (distance), vectors) with a abundance and b biomass of species assemblages in Polyphytos Reservoir. S1–S5: the sampling stations, while seasons are represented by s, spring; su, summer; a, autumn; w, winter
Stations located in the middle section of the reservoir, farther from the dam, consistently exhibit higher abundances of A. thessalicus throughout the year (Fig. 5a). The peak in water transparency occurred during spring, correlated with increased numerical catches of C. carpio, especially in the lower part (S3_s, S4_s, and S5_s) of the reservoir where the deepest areas are located (Fig. 5a). Conversely, lower water transparency values recorded during summer and autumn samplings in stations S4 and S1 were associated with higher numerical catches of species S. vardarensis, L. gibbosus, and V. melanops.
Since abundance serves as a proxy for biomass, most of the observations noted in the CCA abundance model were corroborated by the CCA biomass model (Fig. 5b). However, the CCA model using biomass data showcased a correlation of higher catches of species L. gibbosus, S. glanis, and S. vardarensis with the warmer temperatures of the summer period, while higher biomass catches of species E. lucius were evident during the winter period marked by lower temperatures, particularly in the upper part of the reservoir (S1_w and S1_s) (Fig. 5b).
Discussion
Physicochemical parameters
The study revealed significant seasonal variations in water physicochemical parameters that align with the prevailing climatic conditions of the Mediterranean region, explicitly the distinct shifts between wet winter and dry summer periods (Lionello et al. 2006). The DO concentrations were found to be high throughout the year, both at the surface and at the bottom of the lake. However, the occurrence of anoxic conditions in the deeper layers of the lake during the summer period cannot be ruled out, a phenomenon common in eutrophic Mediterranean systems. Conductivity values were within the range commonly recorded in Greek lakes (200–600 μS/cm) (Kagalou and Leonardos 2009). Also, the measured pH values were within the range typical for most lakes (6 < pH < 9) and in the alkaline zone. They were also within the range considered optimal for most aquatic organisms (6.5 < pH < 8.2) and within the limits of Directive 44/2006/EC (pH 6–9 for both salmonids and cyprinids). The highest estimated salinity value (0.4 psu) was close to the upper limit characterizing low salinity inland waters (0.5 psu). Total dissolved solid (TDS) concentrations were within the range commonly found in inland waters (20–1000 mg/L), particularly close to the values encountered in calcareous regions (200–400 mg/L) (Boyd and Boyd 2015). TDS concentrations in inland waters typically depend on geological and climatic factors, including the composition of the region’s rocks, inflows from streams, precipitation, and even human-made sources (Sherrard et al. 1987). Higher salinity and TDS concentrations were recorded during the autumn sampling, a phenomenon that cannot be attributed to a specific factor. The Secchi disk values, indicative of water transparency were relatively high compared to measurements in Mediterranean systems. In fact, these values were similar to those observed in natural deep Greek lakes, such as Trichonis and the transboundary Great Prespa (Kagalou and Leonardos 2009). The lower water transparency values recorded during the autumn may be attributed to higher phytoplankton concentrations during this period. It is noteworthy that, based on the observed phytoplankton succession pattern in Mediterranean systems (Moustaka‐Gouni et al. 2014), the phytoplankton community experiences a minimum during spring and continuous growth from summer to autumn and reaches maximum biomass levels in the autumn.
Species composition
Among the fish species recorded in this study, A. thessalicus, S. vardarensis, and V. melanops are endemic to the Balkans. The remaining species, except L. gibbosus, are native to Europe and parts of Asia, having a relatively wide distribution. Notably, E. lucius, S. glanis, C. carpio, P. fluviatilis, and R. rutilus hold commercial significance and are actively targeted for fishing. In contrast, L. gibbosus is an introduced species of American origin that has established populations in various European systems (Cucherousset et al. 2009). Additionally, P. leptodactylus is an introduced crustacean (Perdikaris et al. 2017) with high economic importance in local fisheries.
Noteworthy is the absence of certain confirmed species in the reservoir, such as Carassius gibelio, intentionally targeted by local fishermen in recent years for export to Romania and Turkey. Except overfishing, other factors, such as regular desiccation or sudden water level fluctuations, often associated with fish harvesting or drainage, could significantly influence the abundance of this species (Ferincz et al. 2016). Additionally, Barbus macedonicus, due to its rheophilic nature, might have sporadic presence in Polyphytos Reservoir. The absence of some small-bodied fish species like Gambusia holbrooki and Rhodeus amarus, preferring the littoral zone, could be attributed to the limitations of the capturing method. Their small size might require a more suitable technique, such as electrofishing gear to sweep sections of the littoral zone (Petriki 2015).
Dam constructions have been identified as significant factors in altering riverine environments through inundation, hydrologic alteration, and fragmentation. These alterations lead to shifts in species assemblages (Kubečka 1993). Particularly, following dam construction, fish species assemblages typically show dominance by limnophilic species that prefer stagnant waters. Rheophilic species may coexist but usually in smaller populations, particularly in the transitional zone (the upper section along the longitudinal axis), where the hydrological regime resembles that of a river, or in adjacent tributaries flowing into the lake (Prchalová et al. 2008, 2009). This adaptation is evident in catches from Polyphytos Reservoir where the dominant species were R. rutilus, P. fluviatilis, and A. thessalicus. This observed pattern aligns with findings in many Greek lakes, where species of the genus Rutilus or Leucos tend to dominate, accompanied by a significant presence of P. fluviatilis and Alburnus species (Petriki 2015). In addition, it aligns with the eutrophic status of the reservoir that favors the dominance of cyprinids (Olin et al. 2002).
In general, native species dominated the catches; thus, the frequently reported pattern of shifting from native-dominated stream fishes to non-native invasive-dominated fish assemblages (Clavero and Hermoso 2010) following the dam construction and driven by intentional and accidental introductions of non-natives to support fisheries was not observed here.
The disparity between the study’s catch and professional fishermen (personal observations) is attributed to the latter’s use of larger nets with significantly larger dimensions and mesh sizes exceeding 60 mm. These larger mesh sizes are employed by lake fishermen targeting C. carpio and C. gibelio to capture only commercially sized fish. In contrast, the study utilized nets with mesh sizes ranging from 6.25 to 60 mm to provide a more comprehensive capture of the fish community structure.
Spatiotemporal patterns
Variations in the spatiotemporal distribution of aquatic fauna can be attributed to a range of factors, including abiotic, biotic, and anthropogenic elements. These factors encompass the water body’s bathymetric features that generate habitat heterogeneity, water depth, littoral zone structure, substrate composition, and more (Vašek et al. 2004; Prchalová et al. 2008; Miranda 2011). However, water temperature and water transparency, indicative of water productivity, stand out as fundamental factors influencing aquatic assemblages, particularly those of fish (Quirós 1995; Fischer and Eckmann 1997; Gelós et al. 2010; Rosso et al. 2010; Specziár et al. 2013; Bunnell et al. 2021).
The influence of water temperature on fish assemblages primarily manifests through its impact on ecosystem and species metabolism, nutritional requirements, and activity levels, ultimately leading to higher gillnetting efficiency (Linlokken and Haugen 2006; Lall and Tibbetts 2009). Consistent with findings from other studies (Bobori and Salvarina 2010; Gelós et al. 2010), fish species richness and abundance of fish and crayfish were higher during spring and summer and lower during winter. This behavior can be attributed to species’ responses to seasonal changes.
As mainly poikilothermic organisms, fish experience body temperature variations associated with the external environment (Lall and Tibbetts 2009). Thus, as water temperature declines along the seasonal shift from autumn to winter, they reduce their metabolism and, consequently, their mobility to lower their energy cost which is depicted in gillnet catches (Linlokken and Haugen 2006). This allows them to endure extended periods without feeding and helps increase their survival rates (Banet et al. 2022). In response, fish may aggregate in deeper areas to overwinter, where water temperature fluctuations are smaller, and temperatures are generally higher than those prevailing in shallower areas. This behavior has been widely observed in cyprinid fish but also salmonids (Vašek et al. 2004; Prchalová et al. 2008). Therefore, the observed decrease in fish catches during these colder seasons can be attributed to the fish’s migration to deeper areas. On the contrary, during the warmer periods of the year, such as spring and summer, fish tend to inhabit shallower waters and are often found near the littoral zone, particularly during the spring breeding period.
The highest abundance of species C. carpio during spring period may attribute to its reproductive behavior as it prefers to spawn in the littoral zone (Froese and Pauly 2023). Contrastingly, the peak catches of S. glanis during summer and E. lucius during winter samplings can be attributed to the increased mobility of these species during their respective spawning periods (Alp et al. 2004; Yağcı et al. 2009). In Polyphytos Reservoir, these spawning periods extend from June to August for S. glanis and from January to May for E. lucius, according to information obtained through personal communication with commercial fishermen.
The higher catches of L. gibbosus and S. vardarensis during summer also suggest that these species may exhibit increased activity in warmer months, likely due to improved feeding opportunities, favorable reproductive conditions, higher metabolic rates, or a better adaptation or preference for environments with lower water transparency. It is worth noting that lower water transparency values were recorded during summer and autumn samplings.
The influence of turbidity on fish assemblages largely depends on species’ visual adaptations and life strategies, particularly their ability to thrive in environments with varying turbidity levels (Michael et al. 2021). Specifically, for species that rely on vision to detect prey, water transparency affects reaction distance, encounter rates, and ultimately, consumption rates (Lehtiniemi et al. 2005; Turesson and Brönmark 2007; Figueiredo et al. 2016). Therefore, water transparency can impose changes in fish assemblages and even fisheries production through a variety of ecological mechanisms, including predator avoidance, reaction distances, and foraging efficiency (Figueiredo et al 2016; Bunnell et al. 2021). Conversely, fish species can act as ecosystem engineers by altering water clarity dynamics through trophic cascade effects conducted by predation or nutrient control, which can significantly influence water clarity (e.g., Quirós 1995; Rowe 2007; Rosso et al. 2010). This truth underscores many biomanipulation techniques where deliberate alteration of fish populations within aquatic ecosystems, typically lakes, is attempted to improve ecological conditions or restore ecosystem balance (Drenner and Hambright 1999).
Moreover, the highest catches of the crayfish P. leptodactylus during the summer period were likely due to the approaching mating period in September (Kubec et al. 2019). Generally, during winter, crayfish tend to seek shelter during the day and are primarily active during the night and twilight hours. With higher water temperatures in spring and summer and increased availability of food sources, crayfish exhibit greater activity (Alvanou et al. 2022). Due to this heightened mobility, as observed in commercial catches, the fishing season for many crayfish species is extended from mid-summer to autumn to protect berried females, which may carry eggs or hatchlings until mid-July (Jussila and Mannonen 2004).
The higher number of species, along with higher numerical and biomass catches, in the middle section of Polyphytos Reservoir aligns with findings from relevant studies in canyon-shaped reservoirs, indicating a gradient in species numbers, abundance, and biomass along the reservoir’s longitudinal axis (Oliveira et al. 2004; Vašek et al. 2004; Prchalová et al. 2008). This pattern is likely attributed to canyon-shaped reservoirs’ gradient in productivity along the same axis, evident through higher nutrient concentrations and phyto- and zooplankton densities (Prchalová et al. 2008). As suspected, fish abundance and biomass align with this gradient, benefiting from the richest resources that occur in more eutrophic environments (Jeppesen et al. 2005; Garcia et al. 2006). Additionally, this gradient could be attributed to the higher heterogeneity of the environment in the riverine and transitional zones that tend to support more native and rare species (Oliveira et al. 2003, 2005; Gao et al. 2010). The suspected preference of the A. thessalicus species for the middle part of the reservoir, assuming that this section is indeed more productive compared to the lower part, aligns with the preferences observed in the same genus species, which tend to prey upon zooplankton and spawn in tributaries (Vašek et al. 2004).
The environmental variables explained part of the variability of species distribution. Indeed, more complex mechanisms, such as competition and predation, shape the species assemblages in these nearshore areas. However, given the limited information available on the seasonal and spatial distribution of fishes in Mediterranean reservoirs (Moutopoulos et al. 2023), and even less for canyon-shaped ones (Vašek et al. 2004, 2006; Prchalová et al. 2009), let alone the ongoing lack of knowledge regarding habitat use and the preferences of various aquatic species, the significance of the present study becomes evident.
Understanding the seasonal variations in fish assemblages and the potential mobility patterns of aquatic species provides valuable insights into their ecological strategies and adaptations to changing environmental conditions. Further research and monitoring efforts could help elucidate the underlying mechanisms driving these observed patterns and their implications for the overall dynamics of the reservoir ecosystem. Such understanding is crucial for environmental management and design, as it can guide spatial planning for conservation or protection areas.
Conclusions
In conclusion, fish and crayfish assemblages in Polyphytos Reservoir exhibited spatiotemporal trends, with more pronounced and heterogeneous catches observed during warmer seasons. The highest species numbers and catches were noted in the middle section of the reservoir, contrasting with the deeper areas closer to the dam. Water transparency, temperature, and distance from the dam were identified as the most significant factors influencing species assemblages. These findings provide valuable insights into the dynamics that shape seasonal and spatial patterns in fish assemblages in Mediterranean reservoirs. It is noteworthy that despite reservoirs being common aquatic habitats in Europe, there is limited quantitative information and ecological knowledge on the fish and crustacean assemblages inhabiting them.
Data availability
The data supporting the findings of this study are available within the paper. For additional information, please contact the corresponding author.
References
Alp A, Kara C, Büyükçapar HM (2004) Reproductive biology in a native European catfish, Silurus glanis L. 1758, population in Menzelet Reservoir. Turk J Vet Anim Sci 28:613–622
Alvanou MV, Papadopoulos DK, Lattos A, Georgoulis I, Feidantsis K, Apostolidis ΑP et al (2022) Biology, distribution, conservation status and stocking perspective of freshwater crayfish in Greece: an updated review. Aquac Res 53:5115–5128
Araújo FG, Pinto BCT, Teixeira TP (2008) Longitudinal patterns of fish assemblages in a large tropical river in southeastern Brazil: evaluating environmental influences and some concepts in river ecology. Hydrobiologia 618:89–107
Banet NV, Fieberg J, Sorensen PW (2022) Migration, homing and spatial ecology of common carp in interconnected lakes. Ecol Freshw Fish 31:164–176
Becker B, Galhardo BDOS, Macedo DR, Hughes RM, Callisto M, Santos GB (2016) Influence of limnological zones on the spatial distribution of fish assemblages in three Brazilian reservoirs. J Limnol 75:156–168
Bobori DC, Salvarina I (2010) Seasonal variation of fish abundance and biomass in gillnet catches of an East Mediterranean lake: Lake Doirani. J Environ Biol 31:995–1000
Boyd CE, Boyd CE (2015) Dissolved solids. Water Quality: An Introduction 71–100
Bunnell DB, Ludsin SA, Knight RL, Rudstam LG, Williamson CE, Höök TO et al (2021) Consequences of changing water clarity on the fish and fisheries of the Laurentian Great Lakes. Can J Fish Aquat Sci 78:1524–1542
Choi JY, Kim SK (2020) Effects of aquatic macrophytes on spatial distribution and feeding habits of exotic fish species Lepomis macrochirus and Micropterus salmoides in shallow reservoirs in South Korea. Sustainability 12(4):1447
Clavero M, Hermoso V (2010) Reservoirs promote the taxonomic homogenization of fish communities within river basins. Biodivers Conserv 20:41–57
Cucherousset J, Copp GH, Fox MG, Sterud E, van Kleef HH, Verreycken H, Záhorská E (2009) Life-history traits and potential invasiveness of introduced pumpkinseed Lepomis gibbosus populations in northwestern Europe. Biol Invasions 11:2171–2180
Drenner RW, Hambright KD (1999) Biomanipulation of fish assemblages as a lake restoration technique. Arch Hydrobiol 146:129–165
Dustin DL, Vondracek B (2017) Nearshore habitat and fish assemblages along a gradient of shoreline development. N Am J Fish Manag 37:432–444
Ferincz Á, Horváth Z, Staszny Á, Ács A, Kováts N, Vad CF, Csaba J, Sütő S, Paulovits G (2016) Desiccation frequency drives local invasions of non-native gibel carp (Carassius gibelio) in the catchment of a large, shallow lake (Lake Balaton, Hungary). Fish Res 173:37–44
Ferreira MAMF (2019) European catfish (Silurus glanis) movements and diet ecology in a newly established population in the Tagus drainage. Dissertation, Universidade de Lisboa (Portugal)
Figueiredo BR, Mormul RP, Chapman BB, Lolis LA, Fiori LF, Benedito E (2016) Turbidity amplifies the non-lethal effects of predation and affects the foraging success of characid fish shoals. Freshw Biol 61:293–300
Fischer P, Eckmann R (1997) Seasonal changes in fish abundance, biomass and species richness in the littoral zone of a large European lake, Lake Constance, Germany. Arch Hydrobiol 139:433–448
Froese R, Pauly D (2023) FishBase. World Wide Web electronic publication. Available from: www.fishbase.orghttps://doi.org/10.1023/A:1021132426514
Gao X, Zeng Y, Wang J, Liu H (2010) Immediate impacts of the second impoundment on fish communities in the Three Gorges Reservoir. Environ Biol Fish 87:163–173
García-Berthou E (2001) Size-and depth-dependent variation in habitat and diet of the common carp (Cyprinus carpio). Aquat Sci 63:466–476
Garcia XF, Diekmann M, Brämick U, Lemcke R, Mehner T (2006) Correlations between typeindicator fish species and lake productivity in German lowland lakes. J Fish Biol 68(4):1144–1157
Gelós M, Teixeira-de Mello F, Goyenola G, Iglesias C, Fosalba C, García-Rodríguez F et al (2010) Seasonal and diel changes in fish activity and potential cascading effects in subtropical shallow lakes with different water transparency. Hydrobiologia 646:173–185
Gido KB, Schaefer JF, Falke JA (2009) Convergence of fish communities from the littoral zone of reservoirs. Freshw Biol 54:1163–1177
Godinho FN, Ferreira MT, Castro MIP (1998) Fish assemblage composition in relation to environmental gradients in Portuguese reservoirs. Aquat Living Resour 11:325–334
Hatcher HR, Miranda LE, Colvin ME, Coppola G, Lashley MA (2019) Fish assemblages in a Mississippi reservoir mudflat with low structural complexity. Hydrobiologia 841:163–175
Hughes RM, Rinne JN, Calamusso B. (eds.) (2005) Historical changes in large river fish assemblages of the Americas. American Fisheries Society Symposium 45. American Fisheries Society, Bethesda, MD
Irz P, Laurent A, Messad S (2002) Influence of site characteristics on fish community patterns in French reservoirs. Ecol Freshw Fish 11:123–136
Järvalt A, Krause T, Palm A (2005) Diel migration and spatial distribution of fish in a small stratified lake. Hydrobiologia 547:197–203
Jeppesen E, Jensen JP, Soendergaard M, Lauridsen TL (2005) Response of fish and plankton to nutrient loading reduction in eight shallow Danish lakes with special emphasis on seasonal dynamics. Freshw Biol 50(10):1616–1627
Jussila J, Mannonen A (2004) Crayfisheries in Finland, a short overview. Bull Fr Pêche Piscic 372–373:263–273
Kagalou I, Leonardos Ι (2009) Typology, classification and management issues of Greek lakes: implication of the Water Framework Directive (2000/60/EC). Environ Monit Assess 150:469–484
Kubec J, Kouba A, Buřič M (2019) Communication, behaviour, and decision making in crayfish: a review. Zoologischer Anzeiger 278:28–37
Kubečka J (1993) Succession of fish communities in reservoirs of Central and Eastern Europe. In: Straškraba M, Tundisi JG, Duncan A (eds) Comparative reservoir limnology and water quality management. Springer, Dordrecht, pp 153–168
Lall SP, Tibbetts SM (2009) Nutrition, feeding, and behavior of fish. Vet Clin North Am Exot Anim Pract 12:361–372
Lehtiniemi M, Engström-Öst J, Viitasalo M (2005) Turbidity decreases anti-predator behaviour in pike larvae, Esox lucius. Environ Biol Fish 73:1–8
Linlokken A, Haugen TO (2006) Density and temperature dependence of gill net catch per unit of effort for perch, Perca fluviatilis, and roach, Rutilus rutilus. Fish Manag Ecol 13:261–269
Lionello P, Malanotte-Rizzoli P, Boscolo R, Alpert P, Artale V, Li L et al (2006) The Mediterranean climate: an overview of the main characteristics and issues. In: Lionello P, Malanotte-Rizzoli P, Boscolo R (eds) Mediterranean climate variability. Elsevier, Amsterdam, pp 1–26
Michael SC, Patman J, Lutnesky MM (2021) Water clarity affects collective behavior in two cyprinid fishes. Behav Ecol Sociobiol 75:120
Miranda LE (2011) Depth as an organizer of fish assemblages in floodplain lakes. Aquat Sci 73:211–221
Moustaka-Gouni M, Michaloudi E, Sommer U (2014) Modifying the PEG model for Mediterranean lakes–no biological winter and strong fish predation. Freshw Biol 59:1136–1144
Moutopoulos DK, Korakis A, Katselis G (2023) Changes of the ichthyofauna in the impoundment of the Aoos Springs, Greece. Acta Zool Bulg 75:225–233
Oksanen J, Kindt R, Legendre P, O’Hara B, Stevens MHH, Oksanen MJ, MASS Suggests (2007) The Vegan Package. Commun Ecol Package 10:631–637
Olin M, Rask M, Ruuhljärvi J, Kurkilahti M, Ala-Opas P, Ylönen O (2002) Fish community structure in mesotrophic and eutrophic lakes of southern Finland: the relative abundances of percids and cyprinids along a trophic gradient. J Fish Biol 60:593–612
Oliveira EF, Goulart E, Minte-Vera CV (2003) Patterns of dominance and rarity of fish assemblage along spatial gradients in the Itaipu Reservoir, Paraná, Brazil. Acta Sci Biol Sci 25:71–78
Oliveira EF, Goulart E, Minte-Vera CV (2004) Fish diversity along spatial gradients in the Itaipu Reservoir, Paraná, Brazil. Braz J Biol 64:447–458
Oliveira EF, Minte-Vera CV, Goulart E (2005) Structure of fish assemblages along spatial gradients in a deep subtropical reservoir (Itaipu Reservoir, Brazil-Paraguay border). Environ Biol Fish 72:283–304
Penne CR, Pierce CL (2008) Seasonal distribution, aggregation, and habitat selection of common carp in Clear Lake, Iowa. Trans Am Fish Soc 137:1050–1062
Perdikaris C, Konstantinidis E, Georgiadis C, Kouba A (2017) Freshwater crayfish distribution update and maps for Greece: combining literature and citizen-science data. Knowl Manag Aquat Ecosyst 418:51
Petriki O (2015) Development of a fish-based multi-metric index for the assessment of the ecological quality of Greek lakes. Dissertation, Aristotle University of Thessaloniki
Prchalová M, Kubečka J, Vašek M, Peterka J, Sed’a J, Jůza T et al (2008) Distribution patterns of fishes in a canyon-shaped reservoir. J Fish Biol 73:54–78. https://doi.org/10.1111/j.1095-8649.2008.01880.x
Prchalová M, Kubečka J, Čech M, Frouzová J, Draštík V, Hohausová E et al (2009) The effect of depth, distance from dam and habitat on spatial distribution of fish in an artificial reservoir. Ecol Freshw Fish 18:247–260
Quirós R (1995) The effects of fish assemblage composition on lake water quality. Lake Reserv Manage 11:291–298
Reynolds J, Souty-Grosset C, Richardson A (2013) Ecological roles of crayfish in freshwater and terrestrial habitats. Freshw Crayfish 19:197–218
Říha M, Rabaneda-Bueno R, Jarić I, Souza AT, Vejřík L, Draštík V et al (2022) Seasonal habitat use of three predatory fishes in a freshwater ecosystem. Hydrobiologia 849:3351–3371
Rosso JJ, Sosnovsky A, Rennella AM, Quirós R (2010) Relationships between fish species abundances and water transparency in hypertrophic turbid waters of temperate shallow lakes. Int Rev Hydrobiol 95:142–155
Rowe DK (2007) Exotic fish introductions and the decline of water clarity in small North Island, New Zealand lakes: a multi-species problem. Hydrobiologia 583:345–358
Santos GS (2021) Activity patterns and tridimensional space use by the European catfish (Silurus glanis) in Belver reservoir. Doctoral dissertation, Universidade de Lisboa (Portugal)
Sherrard JH, Moore DR, Dillaha TA (1987) Total dissolved solids: determination, sources, effects, and removal. J Environ Educ 18:19–24
Specziár A, György ÁI, Erős T (2013) Within-lake distribution patterns of fish assemblages: the relative roles of spatial, temporal and random environmental factors in assessing fish assemblages using gillnets in a large and shallow temperate lake. J Fish Biol 82:840–855
Turesson H, Brönmark C (2007) Predator–prey encounter rates in freshwater piscivores: effects of prey density and water transparency. Oecologia 153:281–290
Vander Zanden MJ, Vadeboncoeur Y, Chandra S (2011) Fish reliance on littoral–benthic resources and the distribution of primary production in lakes. Ecosystems 14:894–903
Vašek M, Kubečka J, Matěna J, Seďa J (2006) Distribution and diet of 0+ fish within a canyon-shaped European reservoir in late summer. Int Rev Hydrobiol 91(2):178–194
Vašek M, Kubečka J, Peterka J, Čech M, Draštík V, Hladík M et al (2004) Longitudinal and vertical spatial gradients in the distribution of fish within a canyon-shaped reservoir. Int Rev Hydrobiol 89:352–362
Vehanen T, Lahti M (2003) Movements and habitat use by pikeperch (Stizostedion lucioperca (L.)) in a hydropeaking reservoir. Ecol Freshw Fish 12:203–215
Veselý L, Ercoli F, Ruokonen TJ, Bláha M, Kubec J, Buřič M et al (2020) The crayfish distribution, feeding plasticity, seasonal isotopic variation and trophic role across ontogeny and habitat in a canyon-shaped reservoir. Aquat Ecol 54:1169–1183
Wang L, Seelbach PW, Lyons J (2006) Effects of levels of human disturbance on the influence of catchment, riparian, and reach-scale factors on fish assemblages. In: Hughes RM, Wang L, Seelbach PW (eds) Landscape influences on stream habitats and biological assemblages. American Fisheries Society, Bethesda, MD, pp 199–219
Winfield IJ (2004) Fish in the littoral zone: ecology, threats and management. Limnologica 34:124–131
Yağcı M, Alp A, Uysal R, Yeğen V, Yağcı A (2009) Reproduction properties of pike (Esox lucius L. 1758) population in the Ișıklı Dam Lake (Çivril-Denizli/Turkey). J Fish Sci 3:220–230
Yang S, Gao X, Li M, Ma B, Liu H (2012) Interannual variations of the fish assemblage in the transitional zone of the Three Gorges Reservoir: persistence and stability. Environ Biol Fish 93:295–304
Funding
Open access funding provided by HEAL-Link Greece. The research forms part of the “Innovation in fisheries using intelligent molecular approaches” project, co-funded by the European Maritime and Fisheries Fund and the Hellenic Ministry of Rural Development and Food.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The research received approval from the relevant committee in Greece for the conducted fish samplings.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Petriki, O., Ntislidou, C., Petrocheilou, D. et al. Disentangling determinants of nearshore fish and crayfish assemblages in a canyon-shaped Mediterranean reservoir. Environ Biol Fish 107, 551–565 (2024). https://doi.org/10.1007/s10641-024-01550-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-024-01550-1