Abstract
The construction of dams may lead to dramatic changes in fish assemblages due to the loss of lotic habitat caused by impoundment, which have been well documented. However, knowledge of the temporal variation of fish assemblages in the transitional zone, which is an ecotone between lotic and lentic environments of the reservoir, is still very rare. In the present study, fishes in the transitional zone of the Three Gorges Reservoir were sampled from 1997 to 2002 and from 2006 to 2009 to investigate interannual variations of fish assemblages. The results showed that 9 families, composed of 77 species, were found in the transitional zone with Cyprinidae as the dominant group. By cluster analysis (CLUSTER) and non-metric multidimensional scaling analysis (MDS), assemblages were separated into two groups at a Bray-Curtis similarity value of 77.26%, representing the pre-impoundment period and post-impoundment period. Following analysis of similarity percentages of species contributions (SIMPER), shifts in abundances of Coreius guichenoti, Rhinogobio cylindricus and Coreius heterodon, etc. contributed most to the difference between the two groups. Surprisingly, contrasting to the drastic changes in lacustrine region, the fish assemblage in the transitional zone appeared to be relatively stable, since the ten core species caught were consistently recorded every year, and no significant species replacement occurred during our study period. Besides, the moderate index of persistence indicated persistence of the fish assemblage as well, in spite of the shift of some species. Based on the results of ordination and time-lag analysis, the fish assemblage in the transitional zone showed no directional change. We conclude that fish assemblage in the transitional zone of the Three Gorges Reservoir was stable across the surveyed years. Based on our results, we propose that maintenance of dynamic transitional zones in reservoirs will be useful for the preservation of fishes, particularly for the endemic species inhabiting the upper Yangtze.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dams are major threats to aquatic ecosystems and have been shown to change the flow regime, water quality, habitat conditions and aquatic biota of the rivers (Baxter 1977; Dudgeon 2000; Nilsson et al. 2005). Considerable attention has been given to the impacts on fish assemblages of downstream reaches (De Jalon et al. 1994; Quinn and Kwak 2003; Merona et al. 2005) and upstream reaches (Gido et al. 2000; Quist et al. 2005; Guenther and Spacie 2006). Remarkable changes in fish assemblages have been found after the impoundment of reservoirs. For instance, in the downstream reach of a large Ozark river, Quinn and Kwak (2003) found changes in fish assemblage composition 30 years after impoundment of Beaver Dam: warm-water fish species dominated in the pre-impoundment surveys and coldwater species dominated 30 years after impoundment. Quist et al. (2005) identified dramatic changes in the fish assemblages in both upstream and downstream reaches after impoundment of the Laramie River by Grayrocks Reservoir.
Less consideration has been given to fish assemblages in the transitional zones of the reservoirs. Habitats in reservoirs vary longitudinally, allowing the division of three distinct zones: riverine, transitional and lacustrine (Thornton et al. 1990). Studies on the fish assemblages in reservoirs suggest that transitional zone, which is an ecotone between the river upstream and the lacustrine zone of the reservoir, is important for the management of reservoirs (Carvalho et al. 1998; Oliveira et al. 2004, 2005; Agostinho et al. 2008). The transitional zone has relatively higher fish density and diversity than others possibly by reason of its higher light penetration and primary productivity (Carvalho et al. 1998; Oliveira et al. 2004; Santos et al. 2010; Terra et al. 2010). However, one of the characteristics of the transition zone is its dynamic nature (Thornton et al. 1990). This zone may consist of static water, gently flowing water and running water and may be at the highest risk of experiencing water quality problems within the three zones (Lind et al. 1993; Scott et al. 2009), and the fish assemblages could be most seriously influenced by frequent flow regulation. Thus, fish assemblages in the transitional zone of a reservoir may exhibit large variations. However, empirical data on temporal variations of fish assemblages in the complex zones are scarce.
Persistence and stability are important temporal elements of ecological assemblages. Findings about persistence and stability of assemblages are fundamental to our understanding of structure and function in natural ecological systems (Meffe and Berra 1988). Following Holling (1973) and Connell and Sousa (1983), persistence is often defined as constancy in community composition, and stability refers to the constancy in relative species abundance. Although theses terms have often been confused and used interchangeably, both should be considered separately when evaluating temporal change in natural assemblages (Meffe and Minckley 1987). Persistence and stability of fish assemblages structure across temporal scales have been investigated in many aquatic ecosystems (Ross et al. 1985; Meffe and Minckley 1987; Matthews et al. 1988; Oberdorff et al. 2001; Pyron et al. 2006; Hued et al. 2010). For example, in the middle Wabash River, USA, Pyron et al. (2006) found that the fish assemblage was unstable over a period of 25 years. In Suquía River Basin, Argentina, Hued et al. (2010) identified a persistent and stable fish assemblage, even though the deterioration of water quality was recorded. Up to now, few studies have attempted to examine temporal variation of fish assemblages in large reservoirs of large rivers.
The Three Gorges Reservoir (TGR) located in the upper reaches of the Yangtze River, China, is one of the largest hydropower station in the world, with the capacity of 3.93 × 1010 m3, and the surface area of 1080 km2 (Huang et al. 2006). According to the schedule, the water filling of TGR was completed through three filling events, which were conducted in 2003, 2006 and 2009, respectively. It was predicted that the impoundment would severely threaten the rare and endemic fishes in the upper Yangtze River because of the inundating of the original spawning areas and habitats of fluvial fishes (Park et al. 2003; Xie 2003). Unfortunately, only a few studies have dealt with the impacts of reservoir construction on the fish assemblages (Duan et al. 2002; Wu et al. 2007; Gao et al. 2010), which found the immediate change of fish assemblages after the impoundment. According to the schedule, after the planned last filling in 2009, the water level was risen to 175 m ASL and subsequently the water level would fluctuate between 145 m ASL (flood season) and 175 m ASL (the remaining months of a year) (Fig. 1). A large transitional zone was created in the TGR, where the water level alternated between 145 m ASL and 175 m ASL. At present, the dynamic of fish assemblages in this zone is poorly known.
To investigate the dynamic processes of fish assemblages potentially impacted by the reservoir regulation, fish assemblage surveys were conducted in the transitional zone during pre-impoundment and post-impoundment periods. We aimed to analyze the temporal variations of fish assemblages, estimate the impacts of impoundment and flow regulation on the fish assemblages, test their persistence and stability, and give suggestions for fish conservation in the future.
Materials and methods
Study site and fish sampling
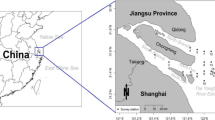
Samplings were conducted in Mudong reach (29°34′N, 106°50′E) of the Yangtze River, which is about 560 km upstream from the Three Gorges Dam, and inundated after the second impoundment in 2006, where is a typical transitional zone (Fig. 2a, b). According to the regulation scheme of the TGR, after the first filling in June 2003, the water level in front of the dam reached 135 m ASL. In October 2006, the second filling was finished with the water level increasing to 156 m ASL. In the trial filling in October 2008, the water level reached 172 m ASL (Fig. 3). The investigations were conducted each year from 1997 to 2002 and 2006 to 2009. The fishes were collected twice each year in May-June and September-October. The duration of each sampling was 15 to 20 days. All analyses were based on pooled data for the spring and fall catches.
Variations of the water level ASL in front of the Three Gorges Dam since 2003. The straight line represents the altitude of Mudong reach. After the second filling, Mudong reach was inundated in the non-flood season (the shaded areas) and remained lotic environment in the flood seasons (areas filled with oblique lines). Dotted lines represent the sampling periods since 2006
Our study area is about 30 km reach where the local fishers prefer to fishing and could set the nets in the whole reach based on their long-time experiences for more catches. We investigated at least ten fishing boats using multi-mesh gill nets with 100–130 m long and 1.2–1.4 m high. There were four mesh-sizes of gill nets ranging from 50 to 120 mm. Most of the boats were investigated every day during the sampling period. Fishing of each boat usually lasts about 12 h each day, following the fishermen’s experience. The fish specimens were collected at 6:00–7:00 every morning. The fishes were then identified to species, counted in numbers, and weighted in individual body weight. Each fish was weighted to the nearest 1 g.
Gill nets, like any fishing gear, have their inherent biases; however, they provide useful estimates of changes in relative abundance (Hubert 1996). Small fish may not be efficiently sampled using the 50–120 mm gill nets. Therefore, our investigations were restricted to relatively large species, and do not provide information on assemblage structure of small bodied fish species. But this bias should not interfere with the conclusions of this study since the bias was the same for all sampling years. In terms of the aim of our study, it was therefore believed that the data were sufficient enough to reflect the actual conditions of the fish assemblages in this transitional zone.
The data of the water level in front of the dam were provided by China Three Gorges Project Corporation.
Data analysis
Variation of fish species composition was examined using cluster analysis (CLUSTER) based on Bray-Curtis similarity matrix calculated on fourth root transformed interannual relative abundance data. Only species making up more than 0.1% of the total catch were included in the analysis, because rare species typically have a minor influence on results of multivariate statistics and can be perceived as outliers in ordinations (Gauch 1982). To construct a configuration of the sampling years according to fish assemblages, non-metric multidimensional scaling (MDS) ordination analysis was carried out. Stress coefficients were treated as critical values to test goodness-of-fit of a given MDS model in two dimensions (Clarke and Warwick 2001). The significance of differences among fish assemblages was tested using the analysis of similarity (ANOSIM). The Similarity Percentage procedure (SIMPER) was used to identify those species most responsible for the dissimilarity between year-groups (Clarke and Warwick 2001). All the multivariate analyses were performed with the PRIMER 5 software package, including modules ‘CLUSTER’, ‘MDS’, ‘ANOSIM’, and ‘SIMPER’ (Clarke and Warwick 2001).
We measured the persistence of fish assemblage through an index of species turnover rate (Diamond and May 1977; Järvinen 1979; Meffe and Minckley 1987). The index was defined as:
where C and E were the number of species that colonized or went extinct between sample years, and S1 and S2 were number of species that were present during each sampling years. Turnover rates were determined between all pairs of sampling years, and a mean turnover rate (\( \overline T \)) was calculated as the average of all k-1 adjacent sampling years. An index of persistence was calculated as 1- \( \overline T \)where zero indicated no persistence and one indicated complete persistence (Meffe and Minckley 1987).
Principal Component Analysis (PCA) was used to examine the temporal trajectories of assemblage composition. Linear ordination method was selected because preliminary detrended correspondence analyses showed turnovers <2SD, which is the recommended criterion for choosing linear versus uni-modal ordination models (ter Braak and Smilauer 2002). The PCA was performed on relative abundance data by species using Canoco version 4.5 (ter Braak and Smilauer 2002). Fish data were square-root transformed prior to analysis.
Although ordination analyses can be used to assess general patterns of temporal change in community data, ordinations do not provide statistical inferences as to whether or not compositional change is truly directional (Wildi 1988). Time-lag analysis provides a useful quantitative measurement of the rate and pattern of temporal dynamics in communities over time (Collins et al. 2000). Therefore, in the present study, time-lag regression was fitted to test directional change in an assemblage during the sampling periods following the method of Collins et al. (2000). The test for directional change was performed by first calculating Bray-Curtis dissimilarity matrix for the species × time data matrix. Bray-Curtis dissimilarity measures were used because they result in more normal frequency distributions than Euclidean distance (Pyron et al. 2006). The dissimilarity values were then regressed against time lags in years, which could produce three theoretical patterns (Collins et al. 2000). For instance, a data set sampled over 10 years would have nine 1-year time lags, eight 2-year time lags and seven 3-year time lags, etc., for a total of 45 time lags over the entire period. A regression line that is significant, positive and linear implies the assemblage is unstable because it is undergoing directional change. A regression that is not significant implies a stable assemblage. A negative, significant and linear slope implies an unstable assemblage with convergence to one of the early sample period.
Result
Fish species composition
We collected a total of 58 086 individuals, representing 77 species of 9 families from 1997 to 2002 and 2006 to 2009. The assemblage was totally dominated by species belonging to Cyprinidae (62%), Bagridae (13%), and Cobitidae (10%), and the numerically abundant species included Coreius heterodon (34%), Coreius guichenoti (24%), Rhinogobio ventralis (14%) and Rhinogobio cylindricus (10%). Interannual number of fish species was recorded between 29 and 46 species (mean = 34.1, SD = 4.8).
Interannual variations of fish assemblage
Cluster analysis showed the fish assemblages fell into two groups while similarity level was set at 77.26% (Fig. 4a). Group I consisted of 6 years in the pre-impoundment period (from 1997 to 2002). Group II included 4 years in the post-impoundment period (from 2006 to 2009). The clustering of the two groups was further confirmed by a MDS two-dimensional plot that showed no overlap between groups (Fig. 4b). The overall ordinations were strong with low stress values of 0.11. The result of the MDS ordination analysis was supported by the ANOSIM test (Global R = 0.655, P < 0.05).
Ten species, Coreius heterodon, C. guichenoti, Rhinogobio ventralis, R. cylindricus, Pelteobagrus vachelli, Leiocassis longirostris, R. typus, Leptobotia elongate and Cyprinus carpio were present in all years. These ten taxa accounted for 95% of total species abundance during the study period. SIMPER analysis showed that 18 species contributed to 90% of the dissimilarity (Table 1). Of the 18 “discriminating” species, mean annual relative abundances of Pelteobagrus nitidus, Rhinogobio cylindricus and Coreius heterodon were generally higher in the post-impoundment period. Mean annual relative abundances of Xenophysogobio boulengeri, Leiocassis longirostris, Gobiobotia filifer, R. ventralis, Saurogobio gymnocheilus, Botia reevesae, Pseudolaubuca sinensis, Silurus meridionalis, S. dabryi, Carassius auratus, C. guichenoti, L. crassilabris, P. fulvidraco, P. vachelli and Jinshaia abbreviata were greater in the pre- impoundment period.
Persistence and stability
The index of persistence is based on species presence in a community and for this study it equals to 0.68 (zero indicates no persistence and one indicates complete persistence), indicating that there were few changes in species composition over time.
The Principal Component Analysis of the fish assemblage in the transitional zone showed no clear patterns of directional change throughout the ten years (Fig. 5). Time-lag analysis was used to quantify temporal dynamics of fish assemblage in the transitional zone. The slope of the time-lag regression line was not significantly different from zero and the overall regression was not significant (P > 0.05) (Fig. 6), indicating that the fish assemblage was stable.
Discussion
Impacts of impoundment of TGR on fish assemblage in the transitional zone
It is widely recognized that the shift in fish assemblage is an inevitable impact of the impoundment (Agostinho et al. 2008). Previous studies also revealed clear changes in fish assemblages in the TGR due to the impoundment (Wu et al. 2007; Gao et al. 2010). The impoundment of TGR showed various degrees of impacts on the fish assemblages between the transitional zone and the lacustrine zone. Duan et al. (2002) and Wu et al. (2007) investigated the fish resources of the Three Gorges Reservoir during the pre-impoundment period (1997–2000) and post-impoundment period (2005–2006) respectively. They found that fish compositions of Mudong reach (the transitional zone) and Wanzhou reach (the lacustrine zone) were similar before the impoundment (Duan et al. 2002). However, after the impoundment, locating in middle part of the TGR, Wanzhou reach was dominated by lentic species. In contrast, Mudong reach was still dominated by lotic species due to its transitional environmental condition. Our results suggested that the impoundment of the TGD had evidently altered the structure of the fish assemblage in the transitional zone. Several species have shifted in their relative abundance over time. The most notable species was C. guichenoti, which accounted for 27.96% of the abundance measured by number of individuals before the impoundment, but dropped to 15.19% after the impoundment. Conversely, C. heterodon increased from 25.74% to 43.69%, and R. cylindricus increased from 6.94% to 19.80%. The reduced abundance of C. guichenoti in the transitional zone could be attributed to their lotic preferences, which was in accordance with earlier studies (Wu et al. 2007). They might not be adapted to the slow flow after the impoundment and moved more upstream to lotic habitats (Institute of hydrobiology, unpubl. data).
Persistence and stability
According to Connell and Sousa (1983), assessments of persistence and stability would be more meaningful if: (1) the disturbing force could cause a potentially disrupt structure of the system; (2) populations are examined over at least one complete turnover of the individuals. We were sure both these conditions were met in this study. First, the transitional zone of the TGR has been disturbed by artificial flow regulation. In addition, the 13-yr scale of this study was long enough to encompass at least one turnover of individuals in almost all the species studied (Ding 1994).
When investigating temporal assemblage change, a single analytical tool may restrict the breadth of understanding and interpretation. It is useful to employ more than one technique when assessing assemblage change (Phillips and Johnston 2004). Thus we use different community metrics in our present study. All metrics employed in this study indicated that fish assemblage structure was relatively stable in the study area. Comparing with the index in other reports (e.g., Meffe and Minckley 1987; Meffe and Berra 1988), which were above 0.9, indicating high persistence in their fish assemblages, the index of persistence (equaled to 0.68) in our study was moderate, indicating that the species composition of our study area was relatively constant. Both lentic and lotic species may co-exist in the transitional zone, using the habitat simultaneously, thus this zone may have a high number of rare species (Oliveira et al. 2003) and result in the moderate persistence index. We followed the analytical method of Collins et al. (2000) to test for directional change in the fish assemblages. The regression line was not significant, and the ordination of the fish assemblage showed no clear patterns of directional change, which implied a stable fish assemblage.
Despite the varied environment, the fish assemblages in the transitional zone of the TGR tended to have certain resilience and re-establish themselves in the long term, which leads us to reject our hypothesis that fish assemblages in the transitional zone of a reservoir may exhibit large variations. For instance, the ten species were recorded every year and remained the most abundant; also no significant species replacement occurred during the sampling years. This apparent stability in the face of harsh environmental variation has already been reported in many systems (Ross et al. 1985; Matthews et al. 1988; Meffe and Berra 1988; Hued et al. 2010). Several factors may contribute to the persistence and stability of fish assemblage in the transitional zone. For example, successful recruitment of some species may lead to the persistence and stability of the fish assemblage. The transitional zone provided important nursery areas for the juveniles of many fishes with drifting fertilized eggs (e.g. C. guichenoti, R. ventralis, C. heterodon, G. filifer, X. boulengeri, S. dabryi) in the upper Yangtze River probably because of its abundance of food and relatively high water velocity (Yang et al. 2010), which might be useful for the completion of their life cycles.
Additionally, regulation of flow regime may play a major role in structuring the fish assemblage. In stable environments, biological interactions are thought to be the main determinants of community structure (Schlosser 1987). In unstable environments, physical interactions are thought to be the main determinants of community structure (Schlosser 1987; Beesley and Prince 2010). Species-habitat level interactions also have been reported as major drivers of fish assemblage organization in many studies (Marsh-Matthews and Matthews 2000; Peres-Neto 2004; Bhat and Magurran 2007). The transitional zone remained lotic environment in the flood seasons since 2006, for the water level of the TGR remained at about 145 m (ASL) during these periods. Species evolving in a large river ecosystem and normally experiencing harsh river environment seem to be adapted to the highly variable river environment (Gido et al. 2000). Most species in our study appear to have some degree of resistance to the fluctuations in water level. Some species might move upstream during the non-flood periods and return to the transitional zone during flood periods, which is in accordance with the reason proposed by Ross et al. (1985) that a high mobility may permit a local refuge-seeking behavior and the return of the species after the perturbation.
Conservation implications
Findings about the contributions of biotic and abiotic factors in structuring fish assemblages have important implications for conservation and management of habitats under the effect of human activities (Bhat and Magurran 2007). Our results support the view that flow regime had a large impact on the fish assemblage. Human-induced threats related to hydroelectric development lead to the decreasement of endemic fish resources in the upper Yangtze River (He et al. 2011). Fortunately, the endemic fishes in the upper reaches of the Yangtze River, such as C. guichenoti, R. ventralis, L. elongate, and R. cylindricus, continue to account for a certain proportion in the transitional zone. The transitional zone could play an important role in protecting the endemic species, and should be kept in a reservoir.
Exotic fish species can remarkably influence fish community structures when they are introduced to reservoirs (Han et al. 2008), and the introduction of exotic species to reservoirs has been reported by many authors (e.g. Gido et al. 2000; Oliveira et al. 2005). In the TGR, some exotic species (for example, Ameiurus melas, Tinca tinca, and Ictalurus punctatus) also had successfully colonized the lacustrine zone according to Gao et al. (2010). However, no exotic fishes have been recorded in the transitional zone of the TGR during our study period. Native fishes exhibit different habitat requirements and respond to temporal variation in flow in a different manner than nonnative fishes (Marchetti and Moyle 2001). The harsh nature of the transitional zone most likely creates an environment unsuitable for introduced species.
According to the integrated plan for the Yangtze River basin, a large scale of cascade hydropower plants will be established in the main channel and tributaries in the upper reaches. After these projects carried out, most regions of the upper reaches will become cascade reservoirs. The cumulative effects of the cascade reservoirs may have great impacts on fish species (Chen et al. 2006). We suggest that, while setting reservoir regulation scheme, to maintain a dynamic transitional zone in reservoir will be useful for the preservation of fishes, particularly for the endemic species inhabiting the upper Yangtze.
References
Agostinho AA, Pelicice FM, Gomes LC (2008) Dams and the fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Braz J Biol 68:1119–1132
Baxter RM (1977) Environmental effects of dams and impoundments. Annu Rev Ecol Syst 8:255–283
Beesley LS, Prince J (2010) Fish community structure in an intermittent river: the importance of environmental stability, landscape factors and within-pool habitat descriptors. Mar Freshw Res 61:605–614
Bhat A, Magurran AE (2007) Does disturbance affect the structure of tropical fish assemblages? A test using null models. J Fish Biol 70:623–629
Carvalho ED, Silva VFB, Fujihara CY, Henry R, Foresti F (1998) Diversity of fish species in the River Paranapanema-Jurumirim Reservoir transition region (São Paulo, Brazil). Ital J Zoolog 65:325–330
Chen J, Huang W, Zhang H (2006) A preliminary approach on impacts of hydropower development in Upper Yangtze River on ecological environment. Water Resour Dev Res 6:10–13 (in Chinese)
Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation, 2nd edn. PRIMER-E Ltd, Plymouth
Collins SL, Micheli F, Hartt L (2000) A method to determine rates and patterns of variability in ecological communities. Oikos 91:285–293
Connell JH, Sousa WP (1983) On the evidence needed to judge ecological stability or persistence. Am Nat 121:789–824
De Jalon DG, Sanchez P, Camargo JA (1994) Downstream effects of a new hydropower impoundment on macrophyte, macroinvertebrate and fish communities. Regul Rivers Res Manage 9:253–261
Diamond JM, May RM (1977) Species turnover rates on islands: dependence on census interval. Science 197:266–270
Ding RH (1994) The fishes of Sichuan. Sichuan Publishing House of Science and Technology, Chengdu (in Chinese)
Duan XB, Chen DQ, Liu SP, Chi CG, Yang RH (2002) Studies on status of fishery resources in Three Gorges Reservoir reaches of the Yangtze River. Acta Hydrobiol Sin 26:605–611 (in Chinese)
Dudgeon D (2000) The ecology of tropical Asian rivers and streams in relation to biodiversity conservation. Annu Rev Ecol Evol Syst 31:239–263
Gao X, Zeng Y, Wang JW, Liu HZ (2010) Immediate impacts of the second impoundment on fish communities in the Three Gorges. Environ Biol Fish 87:163–173
Gauch HG (1982) Multivariate analysis in community ecology: Cambridge University Press
Gido KB, Matthews WJ, Wolfinbarger WC (2000) Long-term changes in a reservoir fish assemblage: stability in an unpredictable environment. Ecol Appl 10:1517–1529
Guenther CB, Spacie A (2006) Changes in fish assemblage structure upstream of impoundments within the upper Wabash River basin, Indiana. Trans Am Fish Soc 135:570–583
Han M, Fukushima M, Fukushima T (2008) Species richness of exotic and endangered fishes in Japan’s reservoirs. Environ Biol Fish 83:409–416
He YF, Wang JW, Lek S, Cao WX, Lek-Ang S (2011) Structure of endemic fish assemblages in the upper Yangtze River Basin. River Res Appl 27:59–75
Holling CS (1973) Resilience and stability of ecological systems. Annu Rev Ecol Evol Syst 4:1–23
Huang ZL, Li YL, Chen YC, Li JX, Xing ZG (2006) Water quality prediction and water environmental carrying capacity calculation for three gorges reservoir. China Water Power Press, Beijing (in Chinese)
Hubert WA (1996) Passive capture techniques. In: Murphy BR, Willis DW (eds) Fisheries techniques, 2nd edn. American Fisheries Society, Bethesda, pp 157–192
Hued AC, Dardanelli S, Bistoni MA (2010) Temporal and spatial variability of fish assemblages in a river basin with an environmental degradation gradient. Community Ecol 11:41–50
Järvinen O (1979) Geographical gradients of stability in European land bird communities. Oecologia 38:51–69
Lind OT, Terrell TT, Kimmel BL (1993) Problems in reservoir trophic-state classification and implications for reservoir management. In: Straskraba M, Tundisi JG, Duncan A (eds) Comparative reservoir limnology and water quality management. Kluwer, Netherlands, pp 57–67
Marchetti MP, Moyle PB (2001) Effects of flow regime on fish assemblages in a regulated California stream. Ecol Appl 11:530–539
Marsh-Matthews E, Matthews WJ (2000) Geographic, terrestrial and aquatic factors: which most influence the structure of stream fish assemblages in the midwestern United States? Ecol Freshw Fish 9:9–21
Matthews WJ, Cashner RC, Gelwick FP (1988) Stability and persistence of fish faunas and assemblages in three midwestern streams. Copeia 4:945–955
Meffe GK, Berra TM (1988) Temporal characteristics of fish assemblage structure in an Ohio stream. Copeia 3:684–691
Meffe GK, Minckley WL (1987) Persistence and stability of fish and invertebrate assemblages in a repeatedly disturbed Sonoran Desert stream. Am Midl Nat 117:177–191
Merona B, Vigouroux R, Tejerina-Garro FL (2005) Alteration of fish diversity downstream from Petit-Saut Dam in French Guiana. Implication of ecological strategies of fish species. Hydrobiologia 551:33–47
Nilsson C, Reidy CA, Dynesius M, Revenga C (2005) Fragmentation and flow regulation of the world's large river systems. Science 308:405
Oberdorff T, Hugueny B, Vigneron T (2001) Is assemblage variability related to environmental variability? An answer for riverine fish. Oikos 93:419–428
Oliveira EF, Goulart E, Minte-Vera CV (2003) Patterns of dominance and rarity of fish assemblage along spatial gradients in the Itaipu reservoir, Paraná, Brazil. Acta Sci Anim Sci 1:71–78
Oliveira EF, Goulart E, Minte-Vera CV (2004) Fish diversity along spatial gradients in the Itaipu Reservoir, Paraná, Brazil. Braz J Biol 64:447–458
Oliveira EF, Minte-Vera CV, Goulart E (2005) Structure of fish assemblages along spatial gradients in a deep subtropical reservoir (Itaipu Reservoir, Brazil-Paraguay border). Environ Biol Fish 72:283–304
Park YS, Chang JB, Lek S, Cao WX, Brosse S (2003) Conservation strategies for endemic fish species threatened by the Three Gorges Dam. Conser Biol 17:1748–1758
Peres-Neto PR (2004) Patterns in the co-occurrence of fish species in streams: the role of site suitability, morphology and phylogeny versus species interactions. Oecologia 140:352–360
Phillips BW, Johnston CE (2004) Fish assemblage recovery and persistence. Ecol Freshw Fish 13:145–153
Pyron M, Lauer TE, Gammon JR (2006) Stability of the Wabash River fish assemblages from 1974 to 1998. Freshw Biol 51:1789–1797
Quinn JW, Kwak TJ (2003) Fish assemblage changes in an Ozark river after impoundment: a long-term perspective. Trans Am Fish Soc 132:110–119
Quist MC, Hubert WA, Rahel FJ (2005) Fish assemblage structure following impoundment of a Great Plains river. West North Am Naturalist 65:53–63
Ross ST, Matthews WJ, Echelle AA (1985) Persistence of stream fish assemblages: effects of environmental change. Am Nat 126:24–40
Santos ABL, Terra BF, Araújo FG (2010) Influence of the river flow on the structure of fish assemblage along the longitudinal gradient from river to reservoir. Zoologia 27:732–740
Schlosser IJ (1987) A conceptual framework for fish communities in small warmwater streams. In: Matthews WJ, Heinz DC (eds) Community and evolutionary ecology of North American stream fishes. University of Oklahoma Press, Norman, pp 17–24
Scott JT, Stanley JK, Doyle RD, Forbes MG, Brooks BW (2009) River–reservoir transition zones are nitrogen fixation hot spots regardless of ecosystem trophic state. Hydrobiologia 625:61–68
ter Braak CJF, Smilauer P (2002) CANOCO reference manual and CanoDraw for windows user's guide: Software for canonical community ordination (version 4.5). Microcomputer Power, Ithaca
Terra BF, Santos ABI, Araújo FG (2010) Fish assemblage in a dammed tropical river: an analysis along the longitudinal and temporal gradients from river to reservoir. Neotrop Ichthyol 8:599–606
Thornton KW, Kimmel BL, Payne FE (1990) Reservoir limnology: Ecological perspectives. John Wiley, New York
Wildi O (1988) Linear trend in multi-species time series. Vegetatio 77:51–56
Wu Q, Duan XB, Xu SY, Xiong CX, Chen DQ (2007) Studies on fishery resources in the Three Gorges Reservoir of the Yangtze River. Freshw Fish 37:70–75 (in Chinese)
Xie P (2003) Three-Gorges Dam: risk to ancient fish. Science 302:1149–1151
Yang SR, Gao X, Ma BS, Kong Y, Liu HZ (2010) Seasonal dynamics of fish assemblages in Mudong reach of the Three Gorges Reservoir of the Yangtze River. Chin J Appl Environ Biol 16:555–560 (in Chinese)
Acknowledgments
This paper is the result of many years of hard work by a great number of people. We gratefully acknowledge all colleagues for their help with the collection of the survey data. Thanks are given to Martin Reichard and three anonymous referees for their valuable comments on the manuscript. This study was funded by an Innovation Project of the Chinese Academy of Sciences (KZCX1-YW-08-01), and the Three Gorges Project eco-environmental monitoring system (SX [2009]-019).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, S., Gao, X., Li, M. et al. Interannual variations of the fish assemblage in the transitional zone of the Three Gorges Reservoir: persistence and stability. Environ Biol Fish 93, 295–304 (2012). https://doi.org/10.1007/s10641-011-9936-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-011-9936-6