Abstract
This study aimed to evaluate environmental influences on fish distribution and to assess the extent to which concepts in river ecology accommodate levels of spatio-temporal heterogeneity of fish assemblages in a 1,080-km long tropical river. A total of 25 sites were sampled between November 2002 and March 2003 in two seasons (summer/wet versus winter/dry). A thermal gradient separating the upper reaches from the lower reaches was detected. The middle-upper reaches showed higher conductivity and lower dissolved oxygen and pH levels compared with the other reaches. Although some significant associations were found between some fish abundance and environmental variables, the most abundant species (Tilapia rendalli, Geophagus brasiliensis, and Oligosarcus hepsetus) occurred in most sites and under most environmental conditions. Fish community structure varied more in space (longitudinal) than through time (seasonal). The community in the lower reach species was more diverse in comparison with the other reaches. Differences in the fish assemblage structure among the longitudinal river sections appear to have been influenced by the effects of damming, and seem to be partially consistent with the Serial Discontinuity Concept, which views dams as discontinuities within the river continuum. Only the lower river reach showed seasonal differences in the fish community structure, attributable to the influence of flooding. Management plans and biodiversity conservation will benefit by considering the effects of dam disruption and flood increased connectivity to the lotic systems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fish assemblages are structured by biotic, abiotic, and historical factors. Typically, the distribution and abundance of native fishes in rivers exhibit longitudinal zonation from upstream to downstream (Hughes & Gammon, 1987; Bhat, 2004; Habit et al., 2006). Species also assort themselves along environmental gradients, with species diversity increasing downstream. Lowe-McConnell (1975) explained this pattern as being a reflection of habitat diversity, which also increases along the upstream to downstream axis. Fausch et al. (1984) suggested that fish assemblages change gradually with stream order. Greater fish species diversity downstream could also be the result of the increased richness of detritus and plankton there (Lowe-McConnell 1975). In contrast, Matthews (1986) suggests that riverine fish assemblages respectively change abruptly or gradually because of abrupt or gradual changes in the physicochemical habitat. Usually, fish assemblages in rivers show longitudinal zonation and the relationship between assemblages composition and physicochemical variability continues to be actively studied. Understanding the influences on fish assemblage structure, therefore, requires analysis of factors influencing assemblages of many types over a broad range of space or environmental conditions.
Vannote et al. (1980), introducing the River Continuum Concept (RCC), viewed streams as having gradients in physical conditions from the headwaters to the mouth, which influence fish species distribution and thus the communities thereof. Overlaying this pattern, the Serial Discontinuity Concept (SDC) (Stanford & Ward, 2001) predicts that dams or other anthropogenic variables (i.e., pollution, erosion, etc.) should disrupt the underlying continuum, causing longitudinal shifts in the river’s abiotic and biotic parameters and processes. The SDC implies that channel networks can be divided into discrete regions within which the community structure and dynamics differentially respond to various disturbance regimes. Therefore, if the predictions of the RCC (e.g., a higher proportion of insectivore, frugivore, and herbivore feeding guilds in the upper reaches, shifting to a predominance of carnivore, omnivore, and detritivore feeding guilds in the lower reaches) are not met, and distinct fish assemblages down in the river coincide with the dam disruption of the natural continuum of the river, it is reasonable to expect that the predictions of SDC can be at least partially applied.
The River Continuum Concept devoted little attention to the lateral dimension or to processes pertaining to floodplain rivers, issues that are now considered essential (Ward et al., 2001). Nonetheless, flood-related seasonal variation is expected to have a significant impact on fish distributions in lotic systems (Wootton, 1990). How discharge variability influences the community structure may be modified by the availability of shelters from high and low flows (Schlosser & Angermeier, 1990). Flooding is considered as an essential ecological interaction between the river channel and its associated floodplain (Junk et al., 1989). Flooding allows access to a greater diversity of floodplain habitat structures than would be available in the main channel, providing for a more diverse flora and fauna. During a flood, aquatic organisms migrate onto the floodplain to use the newly available habitats and resources, and assemblages are expected to change between dry and wet seasons.
Environmental variables (e.g., temperature, dissolved oxygen, conductivity, and pH) characterize aquatic environments, and fish community composition varies in response to these parameters. Ostrand & Wilde (2002) suggested that the assemblage structure is determined more by average or persistent differences in environmental conditions among sites than by seasonal variation in environmental conditions. Matthews et al. (1992), comparing large water quality and fish abundance datasets, found spatial distribution of stream fishes of Arkansas to be significantly related to an aggregate of water-quality conditions. Understanding how environmental variables (and their spatial and temporal variations) shape fish community structure is an important issue for environmental managers. For temperate rivers, numerous studies have tested how environmental changes influence fish assemblages (e.g., Matthews et al., 1988; Fausch & Bramblett, 1991; Brown, 2000). However, for tropical systems, there have been few such studies.
In this study, we assess whether and how fish species distributions vary down the length of the 1,080-km-long Rio Paraíba do Sul, there being major landscape changes from the headwaters to the estuary. Furthermore, we document spatial and seasonal differences in the community structure, and investigate how certain environmental variables influence the occurrence of the most abundant and dominant fish species. Our evaluation of whether fish assemblages differed longitudinally allowed us to test aspects of the Serial Discontinuity Concept, specifically (i) does fish community structure change along longitudinal and temporal river gradients, (ii) do dams disrupt longitudinal fish assemblage structure, and (iii) how does fish abundance relate to longitudinal gradients in environmental variables? In doing so, we test the degree to which ecological models fit the fish distributions and community structuring that exist, and thereby assess the extent to which concepts in river ecology accommodate levels of spatio-temporal heterogeneity of fish assemblages. In addition, we address whether the presence of dams alters the river’s seasonal pattern of variation in environmental conditions and how fishes distribute themselves in response.
Materials and methods
Study region
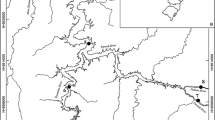
The Rio Paraíba do Sul in southeastern Brazil is a 9th order river (length: 1,080 km; watershed area 57,000 km2) draining one of the most important industrial regions in the country (states of São Paulo, Minas Gerais, and Rio de Janeiro). Its watershed (Fig. 1) is located between parallels 20°26′ and 23°38′ South and meridians 41°00′ and 46°30′ West. As one of Brazil’s largest tropical rivers, marked variation in the landscape occurs along its length (Araújo et al., 2003; Pinto et al., 2006). The upper reach drains relatively well-preserved high altitude areas. The middle-upper and middle-lower reaches contain water of decreased quality due to indiscriminate land use (agricultural, industrial, and urban) (Pfeiffer et al., 1986); these reaches receive large amounts of untreated sewage and industrial effluents. The lower reaches receive less waste, and the river here is wider and flanked by a broad floodplain. Typical winter and summer flows are 109 m3 s−1 and 950 m3 s−1, respectively. Annual rainfall ranges from 100 to 300 cm, with the average generally over 200 cm (Carvalho & Torres, 2002). The drainage basin’s climate is mesothermic, with hot and wet summers and dry winters (Barbiere & Kronemberger, 1994). Four hydropower dams interrupt the river’s flow at locations that divide the river into four segments: the upper, middle-upper, middle-lower, and lower reaches (Fig. 1). It is within these geographic units (in sensu Bizerril, 1999) that we conducted our geomorphic and environmental characterizations.
The upper reach is located at relatively high altitudes (between 500 and 1,800 m above sea level) and have variable slopes (average = 4.9 m km−1). The streams confluent with the main river range from 1st to 3rd order and the river here drains 4,000 km2 (National Waters Authority, 2003).
The middle-upper reach, where the main river transitions from 4th to 5th order, is accompanied by floodplain and meander areas with marginal lagoons, and has a gentle slope (0.19 m km−1). At the junction with the middle-lower reach, the river drains 11,677 km2. The climate here is mesothermic, with hot and wet summers and dry winters (Barbiere & Kronemberger, 1994).
The middle-lower reach is geomorphologically more variable. The river here transitions from 5th to 8th order, drains 31,580 km2, and has an average slope of 1.0 m km−1. The substrates here are unconsolidated to semi-consolidated sand, gravel, silt, and clay, with basalt outcroppings, low mountains, low-nutrient soils, fragments of semi-deciduous seasonal rain forest, and poor cropland areas.
The lower reach (average slope = 0.22 m km−1) is primarily floodplain that increases in extent toward a deltaic estuary. In this reach, the river becomes 9th order, having drained 57,000 km2 at its confluence with the sea (Marengo & Alves, 2005).
Two dams (forming the Paraibuna and Santa Branca reservoirs, respectively) are located between the upper and the middle-upper reaches. Another dam (forming Funil reservoir) separates the middle-upper and middle-lower reaches. A final dam (forming Ilha dos Pombos reservoir) is located between the middle-lower and lower reaches (Fig. 1). Each of these dams completely disrupts the river flow, although the latter dam has a fish-ladder, which is opened during flood periods.
Fish sampling
Locations
Twenty-five total sampling sites, covering the length of the main channel of the Paraíba do Sul and its main tributaries, were sampled. Sites were chosen on the basis of accessibility, similarity in habitat types, and to maximize the diversity of habitat types (pools, riffles and rapids) at each site. We sampled 24 of these sites in the summer/wet season (three upper, three middle-upper, 13 middle-lower, and five lower reaches), and 19 sites in the winter/dry season (three upper, three middle-upper, eight middle-lower, and five lower reaches). Most sites were sampled in both periods (between November 2002 and March 2003), but a few of them were replaced or eliminated, where conditions prevented obtaining a standardized sample. The number of samples varied across the river reaches because some areas were less accessible and some reaches were longer.
Nets
At each site, we collected fish using 20 cast net throws, 20 mesh trays lifts, and 22 gill net sets. This combination of capture methodologies was employed to document as much of the fish diversity as possible. The cast nets were 4 m in diameter with 2-cm mesh. The mesh trays were 80 cm in diameter with 1-mm mesh.
The experimental gill nets were 30 m by 2 m with mesh panels ranging from 2.5 to 6.5 cm (seven nets were 2.5 cm mesh, eight were 4.5 cm, and seven were 6.5 cm). The nets were deployed in the afternoon and retrieved in the following morning after approximately 16 h. Because our fishing effort was standardized, we could calculate a catch per unit effort (CPUE), value simply by totaling the number of fish collected by all of our nets at each site.
Temperature, conductivity, pH, and dissolved oxygen were measured for every location and time we sampled. All these variables were measured at 3 to 4 points, each 200 m apart, at 50 cm sub-surface and about 3 m far from the margins, using a HORIBA W-21 multiparametric probe. These variables were measured at 8, 14 and 18 h in different sampling sites and the overall average was used to represent the site. All captured fish were fixed in 10% formalin for 48 h, and then transferred to 70% alcohol. Each individual was identified to a species, measured (total length, mm), and weighed (g). Information from the literature was used to assign each species in a feeding guild. Fish diets vary largely according to season, and almost all species also perform distinct shifts during life history as a result of ontogenetic changes in resource use. In spite of this limitation, the identification and analysis of the guild structure played a fundamental role in the understanding of the underlying mechanisms responsible for their community organization and structuring (e.g., Vannote et al., 1980; Orth & Maughan, 1984; Bhat, 2004). The gut contents of some species were examined to confirm their feeding habits and guild designations. Voucher specimens were deposited in the fish collection of the Fish Ecology Laboratory of the University Federal Rural of Rio de Janeiro.
Data analysis
Impoundment of the four reaches along the Rio Paraíba do Sul may have resulted in the changes to the pre-existing fish community structure. Therefore, we initially tested for spatial (along the length of the river) and temporal differences in the community structure among the four reaches and two seasons. Species richness among the river reaches was compared for each season by using rarefaction of individuals. The individual-based rarefaction curves representing the means of repeated re-sampling of all pooled individuals (Gotelli & Colwell, 2001) were computed by using EstimateS v. 7.5.2 (Colwell, 2000). We follow by square-root transforming our fish CPUE data to meet the assumptions of multivariate normality and to moderate the influence of extremes in species abundance. The transformed data were then used to create a Bray–Curtis dissimilarity matrix calculated for all pair-wise sample comparisons (Thorne et al., 1999).
Next, we used a non-parametric permutation-based one-way analysis of similarity (ANOSIM; Clarke, 1993) to test for differences in the fish community structure among the reaches (when averaged across the seasons), and among the seasons (when averaged across the reaches). Analysis of similarity is analogous to univariate Analysis of Variance (ANOVA), in that it tests for significant differences among groups. However, unlike ANOVA, ANOSIM is executed on a similarity matrix rather than the raw data; significance is based on comparisons of this matrix to random permutations of the matrix (Clarke & Warwick, 1994), with the degree of dissimilarity associated with each factor being measured by an R statistic (comparable to the F statistic of ANOVA).
Nonmetric multidimensional scaling (MDS) was then used to identify groupings of observations, and a similarity breakdown procedure (SIMPER; Clarke & Warwick, 1994) was used both to identify the species that contributed most to any among-group dissimilarity and to quantify and rank species that on average contribute strongly to assemblage structuring (Clarke, 1993). The procedure also allowed us to quantify the average contribution each species made to the overall measure of dissimilarity between reaches within season and between seasons within reaches. In order to accomplish these ends, the procedure uses the standard deviation of the Bray–Curtis dissimilarity matrix, attributed to a species, for all species pairs and compares that with the average contribution of a species to the dissimilarity.
A principal component analysis (PCA; correlation matrix) was performed on the log-transformed environmental variables to facilitate detection of any spatial/seasonal patterning. Next, we used ANOVA followed by “a posteriori” Tukey means separation tests to compare our environmental data among the reaches and seasons.
A Mantel test was then used to identify correlations between the species-by-sample and environmental variable-by-sample data matrices, this being further evidence of species abundance being influenced by environmental attributes. In addition, we then tested for the existence of species–environment relationships using a Spearman rank correlation procedure (P < 0.05) and the 20 most abundant fish species (i.e., those which contributed more than 1% of the overall catch). We restricted consideration here to the most abundant species because they tended to occur across more of the sites and dates, facilitating detection of any underlying correlations.
Results
Fish assemblages
In total, we collected 8,570 fish from nine orders, 29 families, 55 genera, and 81 species. The 20 species which exceeded 1% of the total catch together amounted to 87.7% of the individuals and 75.0% of their total weight (Table 1). The total number of recorded species ranged from 34 to 35 species in the upper and middle-upper reaches to 53 to 58 species in the middle-lower and lower reaches, respectively.
Of the 20 most abundant species, only three were absent from the upper reach (P. vivipara, G. albescens, and L. castaneus), six were absent from the middle-upper reach (P. lineatus, P. fur, G. albescens, A. giton, L. castaneus, and C. lacustris), and two were absent from the lower reach (P. reticulata and P. fur) (Table 2). A total of 11 of the 81 species were non-native, and 13 were marine species that occurred only in the lower river reach (Table 1). Two introduced species (T. rendalli and P. reticulata) ranked as the first and third most abundant species comprising 15.0 and 6.4% of the total fish abundance, respectively, with the former being distributed in all river reaches.
The rarefaction curves for the summer/wet season showed higher number of species compared with the winter/dry season (Fig. 2). During the wet season, a more marked increase in number of species from the middle-upper reach to the lower was shown. The individual rarefaction curve for the lower reach lied well above the corresponding curve for the middle-lower and the middle-upper reaches. On the other hand, during the dry season, the lower reach curve dropped relatively to the middle-lower reach curve but it still lied above it. The upper reach curves had the lowest number of species and did not reach an asymptote.
A total of 12 out of the 20 most abundant species occurred in all the river reaches. Only five species were exclusive to the upper reach; five species were restricted to the middle-upper reach; nine species occurred only in the middle-lower reach; 18 species (most marine) occupied just the lower reach. Although some species were widely distributed all over the river reaches, the catches of some species were greater in certain specific reaches. For example, catches of A. parahybae and R. quelen in the dry season, and of A. facetum and P. maculatus in the wet season came mainly from the upper reach. H. littorale and P. maculatus came mainly from the middle-upper reach, and O. hepsetus and G. brasiliensis from the middle-lower and lower reaches.
Fish community structure differed substantially across the river reaches during both the wet and dry seasons (Table 3), although differences between the upper and middle-upper reaches were negligible. Across the reaches, our between-season comparison proved non-significant (R = 0.052; P > 0.25), indicating that the community structure remained relatively unaffected. However, within the reaches, a significant seasonal difference in the community structure was found for the lower reach (R = 0.352; P < 0.005).
Our non-metric multidimensional scaling analysis revealed substantial overlap in the community structuring of the upper and middle-upper reaches. However, the community structuring of the upper + middle-upper, middle-lower, and lower reaches were fairly distinct. Seasonal differences were apparent only in the lower reach, whereas the remaining reaches showed considerable overlap between the seasons (Fig. 3).
Spatially, Geophagus brasiliensis was the most common single species, being widely distributed throughout all the river reaches. According to our SIMPER analyses, the assemblages were highly dominated by G. brasiliensis in all reaches except for the middle-upper reach (Table 4). G. brasiliensis and O. hepsetus were typical of the middle-lower and lower reaches, contributing respectively to 12.6 and 13.7% of the similarity in middle-lower reach, and 11.3 and 12.8% in the lower reach. P. lineatus was more abundant in the lower reach, contributing to significantly higher abundances and similarity in the wet season (average abundance = 53.5 ind./sample; average similarity = 10.8%).
Analyses of similarities percentage (SIMPER) indicated that variability of the community structure was more pronounced in the upper (average similarity = 26.4%) and lower reaches (average similarity = 36.9%) than in the middle-upper (average similarity = 50.5%) and middle-lower reaches (average similarity = 48.8%). This spatial trend of the community structure was consistent for both seasons (Table 4). The upper reach consistently contained G. brasiliensis in the wet season (33.9%), but A. parahybae and R. quelen in the dry season. In contrast, the middle-upper reach contained H. littorale and P. maculatus in both seasons (Table 4). The lower reach was populated by O. hepsteus, H. littorale, and P. lineatus in the wet season (average similarity = 38.4%), but by P. vivipara and G. brasiliensis in the dry season (average similarity = 45.9%).
On the basis of species richness, the upper river reach was populated mainly by fish belonging to the omnivorous and detritivorous feeding guilds, although the number of species here was the lowest (Table 5). The number of detritivorous species was also high in the middle-upper and lower reaches. The numbers of omnivorous, invertivorous, and carnivorous species were highest in the middle-lower and lower reaches (Table 5). In other words, our catches of invertivores and carnivores increased from upstream to downstream, whereas the omnivores and detritivores were distributed more evenly down the length of the river. Numerically, the number of individuals also changed longitudinally: herbivorous individuals dominated the upper reach; omnivorous, carnivorous, and invertivorous individuals dominated the middle-lower reaches; detritivorous individuals were dominant in the lower reach.
Environmental influences on fish distribution
Temperature varied spatially, increasing from the upper to the lower reaches, with the among-reach (F = 21.93; P < 0.001) and between-season (F = 9.94; P = 0.003) differences being significant.
Dissolved oxygen and pH exhibited a similar spatial pattern, although only the among-reach differences were significant (F = 17.96; P < 0.001 and F = 6.75; P < 0.001, respectively). The dissolved oxygen values were significantly lower in the middle-upper reach compared to the others. The pH values were highest in the lower reaches, and lowest in the upper and middle-upper reaches.
Conductivity also varied significantly among the reaches (F = 11.64; P < 0.001), but was higher in the middle-upper reaches and lowest in the upper reaches, with no difference between the seasons being evident (Table 6).
A significant correlation was found between the matrix of species-by-sample data and the associated environmental variables (temperature, dissolved oxygen, pH, and conductivity) according to the Mantel test (r = 0.21; P = 0.006) and was considered consistent with the hypothesis that community organization depends, at least partially, on environmental factors at the basin-wide scale.
The first axis of our principal components analysis (PCA 1; Fig. 4) separated middle-upper reach sites having high conductivity, low dissolved oxygen, and low pH values (lower right) and middle-lower and lower reaches sites with the reverse conditions (lower left). The second axis of our principal components analysis (PCA 2; Fig. 4) separated upper reach sites (top of diagram) from the remainder by virtue of their higher dissolved oxygen values and lower values for the remaining parameters. A thermal gradient separating the upper reaches from the lower reaches was detected by the second axis.
Regarding individual taxa, seven abundant species (G. brasiliensis, P. vivipara, O. hepsetus, P. fur, H. auroguttatus, and H. affinis) were widely distributed throughout the river reaches (Tables 1, 2), but their CPUE values were not correlated with any of the environmental variables (Table 7). In contrast, temperature was positively associated with our L. castaneus, C. lacustris, P. lineatus, and A. giton catch abundances, and negatively associated with our R. quelen and A. parahybae catches (Table 7). Higher dissolved oxygen concentrations were positively associated with our catches of P. lineatus and A. giton, whereas the reverse was true for P. maculatus, H. littorale and P. reticulata. Higher pH values accompanied higher catches of C. lacustris and P. lineatus, whereas catches of P. maculatus, A. parahybae, and G. albescens were greater at lower pH values. Greater catches of H. littorale, C. gilbert, A. bimaculatus, P. maculatus, P. reticulata, T. rendalli, and P. vivipara were accompanied by higher conductivity values.
Discussion
Longitudinal assemblage differences
Fish species richness in the Rio Paraíba do Sul exhibited a gradual downstream increase, as demonstrated by the individual-based rarefaction curves. Fish assemblages differed in measured species richness among the reaches and between the two seasons. Communities may differ in measured species richness because of differences in underlying species richness, the shape of the relative abundance distribution, or the number of individuals counted or collected (Denslow, 1995). Differences in the number of individuals counted may themselves reflect biologically meaningful patterns of resource availability in the Paraíba do Sul reaches. However, effects of different sampling efforts among the reaches due to differences in the reach size could be a confounding factor to be considered. Overall, our findings match with the general pattern for fish communities in riverine systems, i.e., that species richness, diversity, and abundance gradually increase from upstream to downstream (Welcomme, 1985; Bayley & Li, 1994; Cowx & Welcomme, 1998; Vila-Gispert et al., 2002). In addition, the wet season has more species richness compared with the dry season, attributable to the higher number of microhabitat and available resources due to higher water levels.
Some species additions and replacements occurred, with the most apparent change occurring in the lower reaches, where some marine species were captured. P. reticulata occurred in the upper, middle-upper, and middle-lower reaches, while its co-generic P. vivipara occurred in middle-upper, middle-lower, and lower reaches. The abundant L. castanaeus and G. albescens occurred in the middle-lower and lower reaches. P. maculatus was widely distributed throughout all reaches, but its co-generic P. fur was recorded in middle-lower and upper reaches. Additionally, two abundant introduced species (Tilapia rendali and P. reticulata) were distributed in the whole system. It is well known that the introduction of exotic species has harmful effects on native fish fauna (e.g., Godinho & Ferreira, 1998). Non-native species in large numbers replacing the natural fish fauna has been associated to low integrity of the aquatic systems. Kennard et al. (2005) found that the potentially strong impact that many alien fish species can have on the biological integrity of natural aquatic ecosystems suggest that some alien species (particularly species from the family Poeciliidae) can represent a reliable “first cut” indicator of river health. In Paraiba do Sul river, effects of deforestation, habitat alteration, and pollution are evident throughout the system.
The natural longitudinal changes in how large river fish communities are structured are attributed to the degree to which the geomorphic structure of the river and its floodplain differ from upstream to downstream (Hughes & Gammon, 1987; Schlosser & Angermeier, 1990; Oberdorff et al., 1993). Along a longitudinal extent, gradient changes in habitat features may reflect species addition, species replacement, or changing relative abundances (Gorman & Karr, 1978; Boys & Thoms, 2006). In general, upper river reaches are geologically young, occupy a narrow valley, and flow swiftly because of the high gradient. In contrast, the lower river reaches are typically older, exhibit a low gradient, and occupy an alluvial floodplain (Starret, 1971). The differences in the habitat structure and complexity associated with these longitudinal differences may, in part, contribute to how and how much the upper and lower river fish communities differ.
According to the River Continuum Concept (Vannote et al. 1980), species from the upper river reaches would be expected to depend mainly on exogenous sources of food provided by the riparian vegetation and its associated fauna (i.e., they would belong to the insectivore, frugivore, and herbivore feeding guilds). In contrast, fishes from lower river reaches would be expected to rely on foods either produced directly in the river itself or those that flow down unused from upstream reaches (i.e., because the riparian vegetation makes a proportionally lower trophic contribution downstream, and there is comparatively more organic matter deposited in the sediment here, the fishes would tend to belong to the carnivore, omnivore, and detritivore feeding guilds). Contrary to these expectations, we found that omnivorous and detritivorous fishes dominated the Rio Paraíba do Sul as a whole, as Mazzoni & Lobón-Cerviá (2000) found in another tropical river in southeastern Brazil; in addition, there are few species (herbivores, invertivores, and frugivores) in the upper reaches that depended on riparian vegetation. Furthermore, in the Rio Paraíba do Sul’s upper and middle-lower reaches, omnivores were most abundant, whereas the complex habitats of the middle-lower and lower reaches were inhabited by species of all the trophic guilds we considered. Consequently, trophic guild structuring of the fish assemblages of the Rio Paraiba do Sul appear not to conform to the predictions of the River Continuum Concept.
One reason for this lack of agreement may stem from the headwaters of the Paraíba do Sul being comprised by grasslands and meadows that provided little in the way of riparian canopy, the existence of which the River Continuum Concept presumes. In addition, the RCC model was derived with relatively pristine temperate streams in mind; it may require adaptation before it can be applied to the more complex systems presented by large tropical rivers, where anthropogenic influence/alterations are a potential confounding factor. We also recognize that more detailed studies of the feeding habits and preferences of the species we encountered would contribute to more precise assignments of species to their respective feeding guilds.
Dam effects on assemblage structure
Compared to unregulated rivers, where we would expect a gradual downstream increase in species richness (Vannote et al., 1980), damming establishes physical barriers, which impede fish movement (especially upstream), contributing to population isolation. In contrast, changes in the fish community down the Rio Paraíba do Sul seem to be partially explained by the Serial Discontinuity Concept, with the dams providing the discontinuities. The fact that a different fish assemblage occurred in each of the four river reaches into which dams divided the Rio Paraíba do Sul suggests the applicability of the Serial Discontinuity Concept (SDC): the dams disrupt the natural continuum of the river, and appear to have caused upstream–downstream shifts in abiotic and biotic parameters and processes (Ward & Stanford, 1995; Mérona et al., 2005). The observed spatial differences in the fish community structure were likewise considered as a potential consequence of dams in Pegg & Pierce’s (2002) study. However, given the lack of reference or pre-impoundment information concerning the biophysical gradients that occurred along each reach of the Paraíba do Sul, the river’s large size, and the degree to which tributaries influence the mainstem, a rigorous test of the SDS’s predictions here is compromised. Furthermore, the extent to which anthropogenic effects have affected habitats here may have overwhelmed what underlying patterns researchers might seek to detect. The mode of dam operation (e.g., surface versus deep release and continuous versus regulated flow) is yet another confounding factor. However, given our knowledge of how dams and reservoirs alter lotic fish assemblages (Bowen et al., 1996; Agostinho et al., 2000; Pringle et al., 2000; Schmutz et al. 2000; Tiemann et al., 2004), we believe that further study of how dams have affected the Paraíba do Sul is warranted.
Seasonal effects on assemblage structure
Habitat diversity is high in river-floodplain systems because of sediment deposition in the floodplain forms bars, levees, swales, ox-bows, backwaters, and side channels (Lorenz et al. 1997). Habitat complexity is likewise expected to be greatest in the usually meandering channels of rivers traversing piedmont and coastal plain areas (Stanford & Ward 2001). Nonetheless, even though the middle-upper reach (slope = 0.19 m km−1) of the Rio Paraíba do Sul also possesses a floodplain, seasonal differences in the fish community structure were shown only for the lower reach (slope = 0.22 m km−1): the summer/wet season assemblage was dominated by O. hepsetus, H. littorale, and P. lineatus, whereas the winter/dry season assemblage was dominated by P. vivipara and G. brasiliensis. Given its floodplain, the lack of seasonal differences in the fish community of the middle-upper reach suggests that damming has somehow homogenized environmental attributes there. A similar effect has been reported by Ward & Stanford (1995). Seasonal flooding (lateral connectivity) in the Rio Paraíba do Sul now only occurs in the lower river reach, and it is only there that the anticipated seasonal changes in the fish community structure were observed.
Environmental effects on species abundance
Overall, environmental variables we examined may influence how fishes are distributed in the Rio Paraíba do Sul (see below), given the significant correlation we found between our species CPUE and environmental data, and the co-occurrence of fluctuations in our catches and in the levels of certain environmental variables. However, we recognize that additional factors not addressed in this study (habitat characteristics, channel geomorphology, and biotic interactions) probably influence fish community composition and structure as well.
Temperature
Of the Rio Paraíba do Sul’s most abundant species, the captures of only six appear to have been significantly associated with temperature variation: during the dry season in the upper reach, R. quelen and A. parahybae were mainly captured when temperatures were lowest; during the wet season in the lower reach, P. lineatus, L. castaneus, C. lacustris, and A. giton were mainly captured in lower reach, temperatures here generally being higher than elsewhere in the river. These findings are consistent with the well-established notion that the differing thermal tolerances of different species affect their distributions (Petts, 2000) and thus fish community composition. However, it is unlikely that differing thermal tolerances are the primary factor influencing the fish distributions in the Rio Paraíba do Sul. Although heightened temperatures are typically accompanied by lower dissolved oxygen levels (McKinsey & Chapman, 1998), the effects of the latter are mitigated on the Paraíba do Sul by the presence of several small waterfalls and riffles, which increase oxygenation.
Conductivity
Conductivity is a useful water quality indicator, given that increments are associated with increased organic matter degradation and the ion inputs associated with water pollution (Vega et al., 1998; Fialho et al., 2008). For the Rio Paraíba do Sul, heightened conductivity and lowered dissolved oxygen were not expected to occur in the middle-upper reaches. However, a heavily industrialized area near Site 6 carries appreciable quantities of organic and industrial effluent into the river, yielding the unexpectedly high conductivity and low dissolved oxygen readings we recorded in this section of the river.
For fishes, disturbed areas like Site 6 constitute a hostile environment for more sensitive species, causing them to decrease in abundance or disappear, and leaving them to become dominated by a few tolerant generalist species, which increase in abundance (Karr 1981; Karr et al. 1986; Soto-Galera et al. 1998). In the Rio Paraíba do Sul, the cyprinodontiform species P. reticulata and P. caudimaculatus were considered highly tolerant and indicators of low water quality (Pinto et al., 2006); P. reticulata and P. vivipara and the silurids H. littorale and P. maculatus were typical of the middle-upper reach, where pollution was the greatest. Similarly, Ganasan & Hughes (1998) found that degraded sites in the Khan River near Indore City, India, were populated primarily by three tolerant species (Lebistes reticulata, Channa punctatus, and Heteropneustus fossilis). Cyprinodontiforms and the silurids H. littorale and P. maculatus are probably more adapted to thrive in poor environmental conditions of the middle-upper reach.
Dissolved oxygen
For tropical fishes, low dissolved oxygen levels rarely limit their distributions because they have evolved the capacity to tolerate or accommodate them (Kramer et al., 1978). In addition, the flowing nature of rivers rarely allows critically low or anoxic oxygen levels to develop. For the Rio Paraíba do Sul, dissolved oxygen levels tended to be higher in the upper reach, where rapids caused turbulence and the human modification of the adjacent landscape was minimal. The middle-lower reaches of the Rio Paraíba do Sul also maintained their oxygenation levels during the wet season, perhaps because the increased flow allowed for greater mixing and re-oxygenation (Maier, 1978).
Overall, the four examined environmental variables were not primary factors for the distribution of fish communities and the distributional patterns probably depend on historical biogeographical criteria, as dispersal barriers, migratory behavior, and top-down factors as predation and interspecific competition. An understanding of the mechanisms determining the spatial segregation of species is of great importance in conservation and management of aquatic resources. Only by examining factors at multiple scales and over a broad range of environmental conditions, it will be possible to detect patterns of influence on composition, richness, and other aspects of the assemblage structure. The study is a step to reach these aims and hopes to provide a basis for management strategies in tropical rivers.
References
Agostinho, A. A., S. M. Thomaz, C. V. Minte-Vera & K. O. Winemiller, 2000. Biodiversity in the high Parana River floodplain. In Gopal, B., W. J. Junk & J. A. Davis (eds), Biodiversity in Wetlands: Assessment, Function and Conservation. Backhuys. Leiden, The Netherlands: 89–118.
Almeida, V. L. L., E. K. Resende, M. S. Lima & C. J. A. Ferreira, 1993. Diet and feeding of Prochilodus lineatus Characiformes, Prochilodontidae in the lowlands of Miranda-Aquidauana, Mato Grosso do Sul State, Brazil. Revista Unimar 15: 125–141.
Andrade, P. M. & F. M. S. Braga, 2005. Diet and feeding of fish from Grande River, located below the Volta Grande reservoir, MG-SP. Brazilian Journal of Biology 65: 377–385.
Araújo, F. G., C. C. Andrade, R. N. Santos, A. F. G. N. Santos & L. N. Santos, 2005. Spatial and seasonal changes in the diet of Oligosarcus hepsetus (Characiformes, Characidae) in a Brazilian Reservoir. Brazilian Journal of Biology 65: 1–8.
Araújo, F. G., I. Fichberg, B. C. T. Pinto & M. G. Peixoto, 2003. A preliminary index of biotic integrity for monitoring the condition of the rio Paraíba do Sul, Southeast Brazil. Environmental Management 32: 516–526.
Barbiere, E. B. & D. M. P. Kronemberger, 1994. Climatologia do litoral sul-sudeste do estado do Rio de Janeiro. Caderno Geociências 12: 57–73.
Barbieri, J. G. & E. P. Santos, 1980. Dinâmica da nutrição de Geophagus brasiliensis (Quoy & Gaimard, 1824) na represa do Lobo, Campina - São Paulo, Brasil. Ciência e Cultura 32: 87–89.
Basile-Martins, M. A., M. N. Cipoli & H. M. Godinho, 1983. Alimentação do mandi Pimelodus maculatus Lacepede, 1803 (Osteichthyes, Pimelodidae) de trechos dos rios Jaguari e Piracicaba, São Paulo, Brasil. Boletim Instituto Pesca 13: 17–19.
Batchelor, G. R., 1978. Aspects of the biology of Tilapia rendalli in the Doorndraai Dam, Transvaal. Journal of the Limnological Society of Southern Africa 4: 65–68.
Bayley, P. B. & H. W. Li, 1994. Riverine fishes. In Calow, P. & G. Petts (eds), The River Handbook: Hydrological and Ecological Principles. Blackwell Science, Boston: 251–281.
Bhat, A., 2004. Patterns in the distribution of freshwater fishes in rivers of Central Western Ghats, India and their associations with environmental gradients. Hydrobiologia 529: 83–97.
Bizerril, C. R. S. F., 1999. The icthyofauna of the Rio Paraíba do Sul watershed. Biodiversity and biogeographics patterns. Brazilian Archives of Biology and Technology 42: 233–250.
Bowen, Z. H., M. C. Freeman & D. L. Watson, 1996. Index of biotic integrity applied to a flow-regulated river system. Proceedings of the Annual Conference of the Southeastern Association of Fish and Wildlife Agencies 50: 26–37.
Boys, C. A. & M. C. Thoms, 2006. A large-scale, hierarchical approach for assessing habitat associations of fish assemblages in large dryland rivers. Hydrobiologia 572: 11–31.
Braga, F. M. S., 2005. Feeding and condition factor of characidiin fishes in Ribeirão Grande system, southeastern Brazil. Acta Scientiarum. Maringá 27: 271–276.
Brown, L. R., 2000. Fish communities and their associations with environmental variables, lower San Joaquin River drainage, California. Environmental Biology of Fishes 57: 251–269.
Câmara, M. R. & S. Chellappa, 1996. Regime alimentar e adaptações morfo-anatômicas do trato digestivo dos Ciclídeos (Pisces: Cichlidae). Boletim do DOL/CB/UFRN- Departamento de Oceanografia e Limnologia/Centro de Biociências/Universidade Federal do Rio Grande do Norte 9: 59–66.
Carvalho, C. E. V. & J. P. M. Torres, 2002. The ecohydrology of the Paraíba do Sul river, Southeast Brazil. In McClain, M. E. (ed.), The ecohydrology of South American Rivers and Wetlands. The IAHS Series of Special Publications. Venice, Italy: 179–191.
Castillo-Rivera, M., A. Kobelkowsky & A. M. Chavez, 2000. Feeding biology of the flatfish Citharichthys spilopterus (Bothidae) in a tropical estuary of Mexico. Journal of Applied Ichthyology 16: 73–78.
Castro, R. M. C. & L. Casatti, 1997. The fish fauna from a small forest stream of the Upper Parana River Basin, southern Brazil. Ichthyological Exploration of Freshwater 7: 337–352.
Clarke, K. R., 1993. Non-parametric multivariate analysis of changes in community structure. Australian Journal of Ecology 18: 117–143.
Clarke, K. R. & R. M. Warwick, 1994. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation. Plymouth Marine Laboratories, Plymouth: 859.
Colwell, R. K. 2000. EstimateS: Statistical Estimation of Species Richness and Shared Species from Samples (Software and User’s Guide), Version 7.5.2. http://viceroy.eeb.uconn.edu/estimates.
Costa, W. J. E., 1987. Feeding habits of a fish community in a tropical coastal stream, rio Mato Grosso, Brazil. Studies on Neotropical Fauna and Environment 22: 145–153.
Coutinho, A. B., E. F. Albuquerque, T. Aguiaro & C. W. C. Branco, 2000. Alimentação de Hyphessobrycon bifasciatus Ellis, 1911 (Pisces, Characiformes, Characidae) na lagoa Cabiúnas (Macaé, RJ). Acta Limnologica Brasiliensis 12: 45–54.
Cowx, I. G. & R. L. Welcomme, 1998. Rehabilitation of rivers for fish. Food and Agriculture Organization of the United Nations (FAO), Rome: 260.
Denslow, J., 1995. Disturbance and diversity in tropical rain forest: the diversity effect. Ecological Applications 5: 962–968.
Fausch, K. D. & R. G. Bramblett, 1991. Disturbance and fish communities in intermittent tributaries of Western Great Plains river. Copeia 1991: 659–674.
Fausch, K. D., J. R. Karr & P. R. Yant, 1984. Regional application of an index of biotic integrity based on stream fish communities. Transactions of the American Fisheries Society 113: 39–55.
Fialho, A. P., L. G. Oliveira, F. L. Tejerina-Garro & B. Mérona, 2008. Fish-habitat relationship in a tropical river under anthropogenic influences. Hydrobiologia 598: 315–324.
Figueiredo, J. L. & N. A. Menezes, 1978. Manual de peixes marinhos do sudeste do Brasil. II. Teostei (1). Muzeu de Zoologia, Universidade de São Paulo, São Paulo: 110.
Figueiredo, J. L. & N. A. Menezes, 1980. Manual de peixes marinhos do sudeste do Brasil. III. Teleostei (2). Muzeu de Zoologia, Universidade de São Paulo, São Paulo: 90.
Figueiredo, J. L. & N. A. Menezes, 2000. Manual de peixes marinhos do sudeste do Brasil. VI. Teleostei (5). Museu de Zoologia, Universidade de São Paulo, São Paulo: 116.
Ganasan, V. & R. M. Hughes, 1998. Application of an index of biological integrity (IBI) to fish assemblages of the rivers Klan and Kshipra (Madhya Pradesh), India. Freshwater Biology 40: 367–383.
Godinho, F. N. & M. T. Ferreira, 1998. The relative influence of exotic species and environmental factors on an Iberian native fish community. Environmental Biology of Fishes 51: 41–51.
Gorman, O. T. & J. R. Karr, 1978. Habitat structure and stream fish communities. Ecology 59: 507–515.
Gotelli, N. J. & R. K. Colwell, 2001. Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecology Letters 4: 379–391.
Habit, E., M. C. Belk, R. C. Tuckfield & O. Parra, 2006. Response of the fish community to human-induced changes in the Biobío River in Chile. Freshwater Biology 51: 1–11.
Harpaz, S., T. Slosman & R. Segev, 2005. Effect of feeding guppy fish fry (Poecilia reticulata) diets in the form of powder versus flakes. Aquaculture Research 36: 996–1000.
Hartz, S. M., C. M. Silveira & G. Barbieri, 1996. Alimentação das espécies de Astyanax Baird & Girard, 1854 ocorrentes na Lagoa Caconde, RS, Brasil (Teleostei, Characidae). Revista Unimar 18: 269–281.
Hughes, R. M. & J. R. Gammon, 1987. Longitudinal changes in fish assemblages and water quality in the Willamette River, Oregon. Transactions of the American Fisheries Society 116: 196–209.
Junk, J. W., P. B. Bayley & R. E. Sparks, 1989. The flood pulse concept in river-floodplain systems, In Dodge, D. P. (ed.), Proceedings of the International Large River Symposium. Canadian Journal of Fisheries and Aquatic Sciences – Special Publication 106: 110–127.
Karr, J. R., 1981. Assessment of biotic integrity using fish communities. Fisheries 6: 21–27.
Karr, J. R., K. D. Fausch, P. L. Angermeier, P. R. Yant & I. J. Schlosser, 1986. Assessing biological integrity in running waters a method and its rationale. Illinois Natural History Survey Special Publication 5, Urbana: 28.
Kennard, M. J., A. H. Arthington, B. J. Pusey & B. D. Harch, 2005. Are alien fish a reliable indicator of river health? Freshwater Biology 50: 174–193.
Kramer, D. L., C. C. Lindsey, G. E. E. Moodie & E. D. Stevens, 1978. The fishes and the aquatic environment of the central Amazon Basin, with particular reference to respiratory patterns. Canadian Journal Zoology 56: 717–725.
Lolis, A. A. & I. F. Andrian, 1996. Alimentação de Pimelodus maculatus Lacépède, 1803 (Siluriformes, Pimelodidae) na planície de inundação do alto rio Paraná, Brasil. Boletim Instituto Pesca 23: 187–2002.
Lorenz, C. M., G. M. Van Dijk, A. G. M. Van Hattum & W. P. Cofino, 1997. Concepts in river ecology: implications for indicator development. Regulated Rivers: Research & Management 13: 501–516.
Lowe-McConnell, R. H., 1975. Fish Communities in Tropical Freshwaters: Their Distribution, Ecology and Evolution. Longman, London: 377.
Maier, M. H., 1978. Considerações sobre características limnológicas de ambientes lóticos. Boletim do Instituto de Pesca 5: 75–90.
Marengo, J. A. & L. M. Alves, 2005. Tendências hidrológicas da bacia do rio Paraíba do Sul. Revista Brasileira de Meteorologia 20: 215–226.
Matthews, W. J., 1986. Fish faunal ‘breaks’ and stream order in the eastern and central United States. Environmental Biology of Fishes 17: 81–92.
Matthews, W. J., R. C. Cashner & F. P. Gelwich, 1988. Stability and persistence of fish fauna and assemblage in three midwestern stream. Copeia 1988: 945–955.
Matthews, W. J., J. D. Hough & H. W. Robinson, 1992. Similarities in fish distribution and water quality patterns in stream of Arkansas: congruence of multivariate analyses. Copeia 1992: 296–305.
Mazzoni, R. & J. Lobón-Cerviá, 2000. Longitudinal structure, density and production rates of a neotropical stream fish assemblage: the river Ubatiba in the Serra do Mar, southeast Brazil. 2000. Ecography 23: 588–602.
McKinsey, D. M. & L. J. Chapman, 1998. Dissolved oxygen and fish distribution in a Florida spring. Environmental Biology of Fishes 53: 211–223.
Menezes, N. A. & J. L. Figueiredo, 1980. Manual de peixes marinhos do sudeste do Brasil. IV. Teleostei (3). Muzeu de Zoologia, Universidade de São Paulo, São Paulo: 96.
Menezes, N. A. & J. L. Figueiredo, 1985. Manual de peixes marinhos do sudeste do Brasil. V. Teleostei (4). Muzeu de Zoologia, Universidade de São Paulo, São Paulo: 105.
Menin, E., 1989. Anatomia funcional da cavidade bucofaringeana de Gymnotus carapo Linnaeus, 1758 (Siluriformes, Gymnotoidei, Gymnotidae). Revista Ceres 36(207): 422–434.
Menin, E. & O. M. Mimura, 1991. Anatomia funcional da cavidade bucofaringiana de Hoplias malabaricus (Bloch, 1974) (Characiformes, Erythrinidae). Revista Ceres 38: 240–255.
Mérigoux, S. & D. Ponton, 1998. Body shape, diet and ontogenetic diet shifts in young fish of the Sinnamary River, French Guiana, South America. Journal of Fish Biology 52: 556–569.
Mérona, B., R. Vigouroux & F. L. Tejerina-Garro, 2005. Alteration of fish diversity downstream from Petit-Saut Dam in French Guiana. Implication of ecological strategies of fish species. Hydrobiologia 551: 33–47.
Mol, J. H., 1995. Ontogenetic diet shifts and diet overlap among three closely related neotropical armoured catfishes. Journal of Fish Biology 47: 788–807.
National Waters Authority - ANA, 2003. Paraíba do Sul Project. Available: http://pbs.ana.gov.br (October 2007).
Nomura, H., 1976. Fecundidade e hábitos alimentares da piava, Leporinus copelandii Steindachner, 1875 do rio Mogi-Guassu, SP. (Osteichthyes, Anostomidae). Revista Brasileira de Biologia 26: 269–273.
Oberdorff, T., E. Guilbert & J. C. Lucchetta, 1993. Patterns of fish species richness in the Seine River basin, France. Hydrobiology 259: 157–167.
Orth, D. J. & O. E. Maughan, 1984. Community structure and seasonal changes in standing stocks of fish in a warm-water stream. American Midland Naturalist 112: 369–378.
Ostrand, K. G. & G. R. Wilde, 2002. Seasonal and spatial variation in a prairie stream-fish assemblage. Ecology of Freshwater Fish 11: 137–149.
Oyakawa, O. T., A. Akama, K. C. Mautari & J. C. Nolasco, 2006. Peixes de mata Atlântica nas unidades de conservação do vale do Rio Ribeira de Iguape no estado de São Paulo. Editora Neotrópica, São Paulo: 201.
Pegg, M. A. & C. L. Pierce, 2002. Fish community structure in the Missouri and lower Yellowstone rivers in relation to flow characteristics. Hydrobiologia 479: 155–167.
Pelicice, F. M. & A. A. Agostinho, 2006. Feeding ecology of fishes associated with Egeria spp. patches in a tropical reservoir, Brazil. Ecology of Freshwater Fish 15: 10–19.
Petts, G. E., 2000. A perspective on the abiotic processes sustaining the ecological integrity of running waters. Hydrobiologia 422/423: 15–27.
Pfeiffer, W. C., M. Fiszman, O. Malm & J. M. Azcue, 1986. Monitoring heavy metals pollution by the critical pathway analysis in the Paraíba do Sul River. The Science of the Total Environment 58: 73–79.
Pinto, B. C. T., F. G. Araújo & R. M. Hughes, 2006. Effects of landscape and riparian condition on a fish index of biotic integrity in a large southeastern Brazil river. Hydrobiologia 556: 69–83.
Ponton, D. & S. Mérigoux, 2001. Description and ecology of some early life stages of fishes in the River Sinnamary (French Guiana, South America). Folia Zoology 50: 1–116.
Pringle, C. M., M. C. Freeman & B. J. Freeman, 2000. Regional effects of hydrologic alterations on riverine macrobiota in the new world: tropical-temperate comparisons. BioScience 50: 807–823.
Randall, J. E., 1967. Food habits of reef fishes of the West Indies. Studies in Tropical Oceanography 5: 665–847.
Roberts, T. R., 1970. Scale-eating American characoid fishes, with special reference to Probolodus heterostomus. Proceedings of the California Academy of Sciences 20: 383–390.
Rodrigues, S. S. & E. Menin, 2006. Anatomia da cavidade bucofaringeana de Salminus brasiliensis (Cuvier, 1817) (Pisces, Characidae, Salmininae). Biotemas 19: 41–50.
Rojas-Beltran, R., 1989. Quelques aspects de l’ecologie alimentaire de trois mâchoirans (Teleostei, Siluriformes, Ariidae) de la Guyane. Cybium 13: 181–187.
Sabino, J. & R. M. C. Castro, 1990. Alimentação, período de atividade e distribuição espacial dos peixes de um riacho da floresta atlântica (sudeste do Brasil). Brazilian Journal of Biology 50: 23–36.
Sazima, I., 1983. Scale-eating in characoids and other fishes. Environmental Biology of Fishes 9: 87–101.
Sazima, I., 1986. Similarities in feeding behaviour between some marine and freshwater fishes in two tropical communities. Journal of Fish Biology 29: 53–65.
Schlosser, I. J. & P. L. Angermeier, 1990. The influence of environmental variability, resource abundance, and predation on juvenile cyprinid and centrarchid fishes. Polskie Archiwum Hydrobiologii 37: 265–284.
Schmutz, S., M. Kaufmann, B. Vogel, M. Jungwirth & S. Muhar, 2000. A multi-level concept for fish-based, river-type-specific assessment of ecological integrity. Hydrobiologia 423: 279–289.
Sierra, L. M., R. Claro & O. A. Popova, 1994. Alimentacion y relaciones tróficas. In Claro, R. (ed.), Ecología de los Peces Marinos de Cuba. Instituto de Oceanología. Academia de Ciencias de Cuba and Centro de Investigaciones de Quintana Roo, Mexico: 263–284.
Silvano, R. A. M., O. T. Oyakawa, B. D. Amaral & A. Begossi, 2001. Peixes do alto Rio Juruá (Amazonas, Brasil). Editora da Universidade de São Paulo, Brazil.
Soto-Galera, E., E. Daz-Pardo, E. Lopez-Lopez & J. Lyons, 1998. Fish as indicators of environmental quality in the Rio Lerma Basin, Mexico. Aquatic Ecosystem Health & Management 1: 267–276.
Spataru, P., W. J. A. R. Viveen & M. Gophen, 1987. Food composition of Clarias gariepinus (= C. lazera) (Cypriniformes, Clariidae) in Lake Kinneret (Israel). Hydrobiologia 144: 77–82.
Stanford, J. A. & J. V. Ward, 2001. Revisiting the serial discontinuity concept. Regulated Rivers: Research & Management 17: 303–310.
Starret, W. C., 1971. Man and the Illinois River. In Oglesby, R. T., C. A. Carlson & J. A. McCann (eds), River ecology and the impacts of man. Academic Press, New York: 131–169.
Talde, C. M., A. C. Mamaril & M. L. D. Palomares, 2004. The diet composition of some economically important fishes in the three floodplain lakes in Agusan Marsh wildlife sanctuary in the Philippines. Sri Lanka Journal of Aquatic Science 9: 45–56.
Teixeira, R. L., 1994. Abundance, reproductive period, and feeding habits of eleotrid fishes in estuarine habitats of north-east Brazil. Journal of Fish Biology 45: 749–761.
Thorne, R. J., W. P. Williams & Y. Cao, 1999. The influence of data transformations on biological monitoring studies using macroinvertebrates. Water Research 33: 343–350.
Tiemann, J. S., D. P. Gillette, M. L. Wildhaber & D. R. Edds, 2004. Effects of lowhead dams on riffle-dwelling fishes and macroinvertebrates in a midwestern river. Transactions of the American Fisheries Society 133: 705–717.
Vannote, R. L., G. W. Minshall, J. R. Sedell & C. E. Cushing, 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37: 130–137.
Vega, M., R. Pardo, E. Barrado & L. Debán, 1998. Assessment of seasonal and polluting effects on the quality of river water by exploratory data analysis. Water Research 32: 3581–3592.
Vila-Gispert, A., E. García-Berthou & R. Moreno-Amich, 2002. Fish zonation in a Mediterranean stream: effects of human disturbances. Aquatic Sciences 64: 163–170.
Ward, J. V. & J. A. Stanford, 1995. The serial discontinuity concept: extending the model to floodplain rivers. Regulated Rivers: Research and Management 10: 159–168.
Ward, J. V., U. Tockner, U. Uehlinger & F. Malard, 2001. Understanding natural patterns and processes in river corridors as the basis for effective river restoration. Regulated Rivers: Research and Management 17: 311–323.
Welcomme, R. L., 1985. Rivers fisheries. FAO Technical paper 262. Food and Agriculture Organization of the United Nations (FAO), Rome: 330.
Weliange, W. S. & U. S. Amarasinghe, 2003. Accounting for diel feeding periodicity in quantifying food resource partitioning in fish assemblages in three reservoirs of Sri Lanka. Asian Fisheries Science 16: 203–213.
Williams, J. D., K. O. Winemiller, D. C. Taphorn & L. Balbas, 1998. Ecology and status of piscivores in Guri, an oligotrophic tropical reservoir. North American Journal of Fisheries Management 18: 274–285.
Wootton, R. J., 1990. Ecology of teleost fishes. Chapman & Hall, London: 404.
Acknowledgements
We thank Bianca de Freitas Terra, Eduardo Oliveira Estiliano, Daniel Gracia, and Paulo Cesar Silva for help with the fieldwork. Dr. Joel Trexler provided comments that helped to improve the manuscript. The programme CNPq-CT-HIDRO—Brazilian Agency for Scientific Development and Research supplied initial financial support for this project (Proc. 550047-2002-6). The Rio de Janeiro Foundation for Research Development—FAPERJ—Rio de Janeiro Agency for Research (Cientísta do Nosso Estado—Proc. E-26/152.463/2002) funded the second part of the field research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: J. Trexler
Rights and permissions
About this article
Cite this article
Araújo, F.G., Pinto, B.C.T. & Teixeira, T.P. Longitudinal patterns of fish assemblages in a large tropical river in southeastern Brazil: evaluating environmental influences and some concepts in river ecology. Hydrobiologia 618, 89–107 (2009). https://doi.org/10.1007/s10750-008-9551-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-008-9551-5