Abstract
Key message
Studying seed oil metabolism.
Abstract
The seeds of higher plants represent valuable factories capable of converting photosynthetically derived sugars into a variety of storage compounds, including oils. Oils are the most energy-dense plant reserves and fatty acids composing these oils represent an excellent nutritional source. They supply humans with much of the calories and essential fatty acids required in their diet. These oils are then increasingly being utilized as renewable alternatives to petroleum for the chemical industry and for biofuels. Plant oils therefore represent a highly valuable agricultural commodity, the demand for which is increasing rapidly. Knowledge regarding seed oil production is extensively exploited in the frame of breeding programs and approaches of metabolic engineering for oilseed crop improvement. Complementary aspects of this research include (1) the study of carbon metabolism responsible for the conversion of photosynthetically derived sugars into precursors for fatty acid biosynthesis, (2) the identification and characterization of the enzymatic actors allowing the production of the wide set of fatty acid structures found in seed oils, and (3) the investigation of the complex biosynthetic pathways leading to the production of storage lipids (waxes, triacylglycerols). In this review, we outline the most recent developments in our understanding of the underlying biochemical and molecular mechanisms of seed oil production, focusing on fatty acids and oils that can have a significant impact on the emerging bioeconomy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In seed plants, production of seeds interrupts the life cycle, linking two sporophytic generations and allowing dispersion and survival of plants. Seeds originate from the fertilized ovule in Angiosperms. Double fertilization of the embryo sac initiates the development of the embryo and the endosperm. These zygotic tissues are protected by a seed coat comprising several cell layers of maternal origin. The coordinated growth of these tissues of distinct origins leads to the formation of seed structures containing all the genetic material and nutrients required to establish the next generation. Seed development comprises two major phases: early embryo morphogenesis is followed by a maturation phase (Vicente-Carbajosa and Carbonero 2005). Storage compounds are accumulated during maturation. Their biosynthesis is developmentally controlled by a complex network of transcription factors termed master regulators of the maturation program that were shown to regulate the transition between vegetative phases of development and seed maturation (Baud and Lepiniec 2010; Devic and Roscoe 2016; Fatihi et al. 2016). Seed reserves play an important role in the acquisition of desiccation tolerance by maturing seeds. They also significantly impact the subsequent seed germination and seedling establishment success by providing the materials required to support growth until the developing seedling becomes autotrophic.
Main seed storage compounds usually consist of storage proteins, carbohydrates (starch or β-glucans), and storage lipids like waxes or triacylglycerols (TAGs). Depending on plant species, the relative proportions of these components vary greatly, but a source of nitrogen (N) and a source of carbon (C) are usually associated. Oils are composed of long-chain hydrocarbons that are one of the most highly reduced and energy-dense carbon compounds produced by nature. Oxidation of oils in germinating seeds thus releases more than twice as much energy as the oxidation of storage carbohydrates or proteins on a per g basis (Graham 2008). These molecules are also important sources of calories for human and animal nutrition, as well as petroleum alternatives for the chemical industry and for biofuels (Aznar-Moreno and Durrett 2017b).
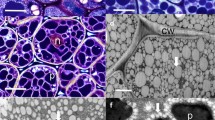
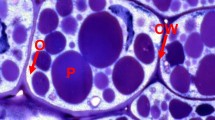
TAGs, the main components of plant oils, are composed of three fatty acids (FAs) esterified to glycerol. They are stored in subcellular structures termed oil bodies or oleosomes that comprise a matrix of TAGs surrounded by a phospholipid monolayer (Miquel et al. 2014; Shimada et al. 2017). Depending on the species considered, the abundance and location of olesomes in seeds vary greatly. Some oilseed species like soybean (Glycine max), rapeseed (Brassica napus), linseed (Linum usitatissimum), and sunflower (Helianthus annuus) predominantly store oil in embryo tissues that occupy most of the space available in between the integuments of the seed. On the contrary, oil is mostly accumulated in the large endosperm tissue observed in seeds of castor bean (Ricinus communis) for example. Additionally to mechanisms controlling cell proliferation and degeneration within the developing endosperm, and therefore the relative importance of this tissue within the seed (Ingram 2017; Olsen 2004; Sreenivasulu and Wobus 2013), transcription factors specifically induced in the maturing endosperm further control the partitioning of reserves like oil between zygotic tissues (Barthole et al. 2014). Interestingly, recent methodological developments in lipidomics, microscopy, and imaging have provided us with high-resolution spatial maps of lipids in the different tissues of maturing oilseeds. These observations have revealed gradients of oils within zygotic tissues as well as contrasting FA compositions of oils between the different regions of oil-storing tissues (Borisjuk et al. 2013; Horn and Chapman 2014). These observations reveal both the influence of environmental parameters (e.g., light, temperature, oxygen availability) on oil metabolism and the existence of local developmental regulations that are not well understood.
Driving incoming carbon into the fatty acid biosynthetic pathway
Photosynthetically assimilated carbon is transported into seeds through the phloem, mostly in the form sucrose. A first important aspect of the regulation of oil metabolism in early-maturing seeds lies in the activation of metabolic pathways driving incoming carbon into the FA biosynthetic network at the expense of competitive pathways using carbon to synthesize starch, amino acids, storage β-glucans, or cellulose. Following sucrose breakdown, hexoses are converted to hexose-phosphates that can be metabolized through any of the above-mentioned pathways (Fig. 1).
Overview of central metabolism in maturing oilseeds. This simplified scheme of carbon metabolism presents the various biosynthetic pathways using sucrose as a precursor for the production of cellulose, starch, and fatty acids in oilseeds. Arrows represent net fluxes of carbon; the arrow thicknesses are proportional to net fluxes of carbon. For the sake of clarity, amino acid biosynthetic pathways have been omitted. 1, Sucrose synthase; 2, invertase; 3, cellulose synthase; 4, UDP-glucose pyrophosphorylase; 5, hexokinase; 6, fructokinase; 7, ADP-glucose pyrophosphorylase; 8, phosphoglucomutase; 9, phosphoglucose isomerase; 10, phosphofructokinase; 11, aldolase; 12, glyceraldehyde-3-phosphate dehydrogenase; 13, phosphoglycerate kinase; 14, phosphoglycerate mutase; 15, enolase; 16, pyruvate kinase; 17, NADP-dependent glyceraldehyde-3-phosphate dehydrogenase; 18, starch synthase; 19, starch branching enzyme; 20, phosphoenolpyruvate carboxylase; 21, malate dehydrogenase; 22, NADP-dependent malic enzyme; 23, pyruvate dehydrogenase; 24, acetyl-coenzyme A carboxylase; 25, malonyl-coenzyme A:acyl carrier protein S-malonyltransferase; 26, fatty acid synthase; 27, ADP-glucose/ADP transporter; 28, glucose-6-phosphate/phosphate transporter; 29, triose-phosphate/phosphate transporter; 30, phosphoenolpyruvate/phosphate transporter; 31, sodium-dependent pyruvate transporter; 32, malate/α-ketoglutarate transporter. Abbreviations: AcCoA, acetyl-coenzyme A; ADPGlc, adenosine diphosphate glucose; DHAP, dihydroxyacetone-3-phosphate; Fru, fructose; Fri1,6P, fructose-1,6-bisphophate; Fru6P, fructose-6-phosphate; GAP, glyceraldehyde-3-phosphate; Glc, glucose; Glc1P, glucose-1-phosphate; Glc6P, glucose-6-phosphate; MaCoA, malonyl-coenzyme A; MAL, malate; OAA, oxaloacetate; PEP, phosphoenolpyruvate; 1,3PG, 1,3-bisphosphoglycerate; 2PGA, 2-phosphoglycerate; 3PGA, 3-phosphoglycerate; PYR, pyruvate; UDPGlc, uridine diphosphate glucose
Storage β-glucans are polysaccharides that consist of unbranched and unsubstituted chains of β-D-glucosyl residues stored in the endosperm walls of some cereals (Hrmova et al. 2002; Guillon et al. 2011). UDP-glucose appears to be the glucose donor supplying synthesis of these polysaccharides (Marcotuli et al. 2016). Cellulose synthase-like genes are believed to be involved in the biosynthesis of these β-glucans, and their controlled transcriptional activation may play a key role in determining β-glucan concentration in developing grains (Houston et al. 2014; Wong et al. 2015). In oilseeds, this pathway is usually inactive. On the contrary, cellulose, a linear polymer of (1 → 4) β-D-glucosyl residues synthesized from UDP-glucose by cellulose synthases and representing the principal structural component of plant cell walls, is produced in every type of seed. Carbon supplied to the seed is thus partially incorporated into cell wall via cellulose biosynthesis, and this biosynthetic pathway competes with the conversion of carbon supplies into reserves like oil (Alonso et al. 2007).
The synthesis of starch begins with the enzyme ADP-glucose pyrophosphorylase (AGPase), which catalyzes the reaction of glucose-1-phosphate with ATP to produce the activated glucosyl donor ADP-glucose (Cross et al. 2004). ADP-glucose is then used as a substrate for the synthesis of starch through the complex interplay between starch synthase (elongation of the glucose polymer), and starch branching and debranching enzymes (Zeeman et al. 2010). The exact molecular processes that control starch metabolism in seeds largely remain to be elucidated (Zhang et al. 2016). Nevertheless, coordinated transcriptional activation of several genes coding for biosynthetic enzymes participating in the pathway has been reported during grain filling in wheat or maize for instance (Cao et al. 2016; Mukherjee et al. 2015; Xiao et al. 2016). The recent discovery and characterization of transcriptional activators of the pathway such as OsbZIP58 (Wang et al. 2013), ZmbZIP91 (Chen et al. 2016a), ZmDof3 (Qi et al. 2017), ZmMYB14 (Xiao et al. 2017), and ZmNAC36 (Zhang et al. 2014a) further reinforce the hypothesis of important transcriptional controls governing starch storage in the endosperm of cereal grains in particular (Zheng and Wang 2015).
In oilseeds, hexose-phosphates are efficiently metabolized through the glycolysis, and pyruvate is the primary precursor for FA synthesis in the plastid. The cytosolic glycolysis pathway is commonly accepted as the major route for the metabolism of hexose-phosphates in many oilseeds. In seeds of the Brassicaceae (B. napus, Arabidopsis thaliana), a complete glycolytic pathway was also described in the plastid. This plastidic glycolysis pathway is interconnected with the oxidative pentose phosphate pathway (OPPP) and, in some species, with a ribulose-1,5-bisphophate carboxylase/oxygenase (RuBisCO) shunt fixing CO2 apart from the Calvin cycle (Schwender et al. 2004) (for a complete review, see Baud and Lepiniec 2010). Several transporters of the plastid envelope link the cytosolic and plastidial glycolysis pathways. Hexose-phosphates, triose-phosphates, as well as end-products of the glycolysis like phosphoenolpyruvate or even pyruvate can be actively transported (Furumoto et al. 2011; Lee et al. 2017). However, import of phosphoenolpyruvate from the cytosol to the plastid and its subsequent conversion to pyruvate by plastidial pyruvate kinase is generally thought to be a major route for providing metabolic precursors for FA production. In some plant species, a minor part of the carbon for FA synthesis is attributable to the import of malate and the subsequent conversion of this compound in pyruvate by NADP-dependent malic enzyme.
The oxidative decarboxylation of plastidial pyruvate by the pyruvate dehydrogenase complex (PDC) produces acetyl-CoA that is used in turn by acetyl-CoA carboxylase (ACC) to form malonyl-CoA (Li-Beisson et al. 2010). ACC has long been considered as a key regulatory step for the FA biosynthetic process. Activity of this heteromeric complex is fine-tuned by a complex set of post-translational regulatory mechanisms integrating different parameters (light and redox status, carbon and energy availability, concentration of end-products of the pathway; for detailed reviews on ACC’s regulatory network, see Salie and Thelen 2016; Troncoso-Ponce et al. 2016b; Xu and Shanklin 2016). Malonyl-CoA produced by plastidial ACC constitutes the carbon donor for each cycle of the FA biosynthetic process. Before entering the process, the malonyl group is transferred from CoA to a protein cofactor named acyl carrier protein (ACP) by a malonyl-CoA:ACP S-malonyltransferase. Production of saturated FAs (Fig. 2) is performed in a stepwise manner by the FA synthase (FAS) of type II (Fig. 3). This multi-subunit complex consisting of monofunctional enzymes uses acetyl-CoA as a starting unit and malonyl-ACP as the elongator. The malonyl-thioester enters in a series of condensation reactions catalyzed by β-ketoacyl-ACP synthase (KAS). 3-Ketoacyl-ACP thus obtained is then reduced by a β-ketoacyl-ACP reductase (KAR), yielding 3-hydroxyacyl-ACP that is subjected to dehydration by the enzyme hydroxyacyl-ACP dehydratase (HAD). The enoyl-ACP obtained is finally reduced by the enzyme enoyl-ACP reductase (EAR). Following their synthesis, acyl groups are hydrolyzed by acyl-ACP thioesterases that release free FAs (Moreno-Pérez et al. 2012). After their transport through the plastid envelope (Li et al. 2015a, 2016), they are activated to CoA esters by long-chain acyl-CoA synthetases (LACS) (Zhao et al. 2010) prior to their transport into the ER (Kim et al. 2013), where they can be used for the assembly of storage lipids.
Fatty acid elongation. The scheme depicts the different pathways involved in the synthesis and elongation of fatty acids, in the plastids and in the endoplasmic reticulum. Growing fatty acid chains bound to acyl carrier proteins in the plastids are presented in blue, free fatty acids released by thioesterases in black, and acyl-coenzyme A in green. 26, Fatty acid synthase; 33, thioesterase; 34, fatty acid transporter; 35, long-chain acyl-coenzyme A synthetase; 36, acyl-coenzyme A transporter; 37, acyl-CoA elongase
Extensive transcriptomic analyses have provided detailed expression patterns for lipid biosynthetic genes in various oilseed species (Abdullah et al. 2016; Dussert et al. 2013; Li et al. 2015b; Troncoso-Ponce et al. 2011; Huang et al. 2017; Venglat et al. 2011). In early-maturing seeds, a coordinated transcriptional activation of actors of the glycolytic and FA biosynthetic pathways can be observed. These genes exhibit a bell-shaped pattern of expression during the course of seed maturation (Baud and Lepiniec 2009). Interestingly, rates of FA production in early-maturing seeds increase in a manner proportional to the transcript levels of genes encoding corresponding biosynthetic enzymes, thus highlighting the importance of transcriptional regulations for the control of FA production in these storage organs. Their concerted transcriptional activation is controlled by the WRINKLED1 (WRI1) transcription factors that bind AW boxes present in their promoter sequences (Maeo et al. 2009). These transcription factors of the APETALA2/ethylene-responsive element binding (AP2/EREBP) family have been characterized in a wide range of oilseeds and their function appears highly conserved (Guerin et al. 2016; Liu et al. 2009; Ma et al. 2013; Qu et al. 2012; Shen et al. 2010; To et al. 2012). Upon binding to DNA, transcription factors recruit the Mediator complex and, in turn, the Pol II complex to initiate transcription. In A. thaliana, WRI1 was shown to interact with the MED15 subunit of the Mediator complex that mediates activation of FA biosynthetic genes (Kim et al. 2016). Considering that wri1 knockout mutants maintain a basal level of glycolytic and FA biosynthetic gene transcriptional activity, the transcriptional machinery governing these pathways certainly includes additional transcription factors able to activate the pathways in the absence of WRI1. A direct transactivation of certain targets of WRI1 by master regulators of seed maturation was proposed (Wang et al. 2007; Mu et al. 2008). However, complementary studies would be required to elucidate the molecular mechanisms underpinning these transcriptional regulations.
Recent results regarding the regulation of WRI1 in A. thaliana suggest that this actor integrates a variety of information that are essential for the successful accumulation of storage lipids in developing seeds (Marchive et al. 2014). Transcriptional activation of WRI1 by master regulators of the seed maturation program like LEAFY COTYLEDON1 (LEC1) and LEC2 first ensures that FA biosynthesis is triggered in a coordinated manner with other maturation-related programs. Post-translational regulations of WRI1 by various molecular mechanisms involving 14-3-3 proteins (Ma et al. 2016), kinases (Zhai et al. 2017), and the ubiquitine proteasome pathway (Chen et al. 2013) may then contribute to integrate signal inputs like sugar and energy availability.
Interestingly, transient expression in leaves of Nicotiana benthamiana of five different WRI1 homologs originating from different species followed by transcriptome sequencing not only confirmed the ability of these transcription factors to activate the FA biosynthetic pathway in this context but also revealed a concomitant repression of the starch biosynthetic pathway and an activation of starch degrading enzymes (Grimberg et al. 2015). Whether this negative regulation of starch accumulation was directly orchestrated by WRI1 is not known. In contrast with the results of the above-mentioned study, gene co-expression network analysis carried out in the mesocarp of oil palm revealed positive correlations between the actors of FA biosynthesis and the actors of starch biosynthesis and degradation (Guerin et al. 2016). These apparent discrepancies suggest that the complex interplay between starch and FA metabolism might well vary between tissues and/or between species. In several oilseeds of the Brassicaceae, transient starch deposition was described in early-maturing embryos (Baud et al. 2008). The role of this transient accumulation of starch at the onset of oil storage remains a matter of debate. Enzymatic and expression data have established that the changes in starch content throughout embryo development reflect the net balance between synthetic and degradative capacity rather than a synthetic phase followed by a degradative phase (da Silva et al. 1997; White et al. 2000). Further analyses will be required to better understand the interactions between these pathways.
The negative correlation between oil and protein contents observed by breeders in seeds of soybean and Brassica (Eskandari et al. 2013; Mao et al. 2013; Mahmood et al. 2006) suggests that corresponding biosynthetic pathways interfere too. This negative correlation reflects a competition for space to store end-products of these pathways since most embryo cells in mature seeds are filled with storage compounds. To a certain extent, these observations also illustrate how the two pathways compete for carbon resources (Kanai et al. 2015). Biosynthesis of seed storage proteins massively uses amino acids imported from the mother plant or derived from these amino acids through transamination reactions. However, approaches of metabolic flux analyses carried out in various oilseeds have unraveled that a minor part of 3-carbon sugars, pyruvate, and acetyl-CoA derived from imported sucrose could serve as precursors for de novo amino acid biosynthesis in maturing seeds (Schwender et al. 2006; Iyer et al. 2008; Lonien and Schwander 2009). The mechanisms regulating carbon partitioning between these different pathways remain poorly understood.
Seeds as reservoirs for a variety of fatty acids
A huge variety of fatty acid structures
Plant oil production is dominated by a few established species and the majority of these food oils are comprised of only five FAs: palmitic acid (C16:0), stearic acid (C18:0), oleic acid (C18:1Δ9c), linoleic acid (C18:2Δ9c,12c), and α-linolenic acid (C18:3Δ9c,12c,15c). In striking contrast, seed plants collectively display enormous variation in the FAs they synthesize and store into seeds. More than 300 different FA structures have been reported to occur in plant seeds (https://plantfadb.org) and more structures might yet be discovered in plant families never analyzed so far for FA composition. These unusual FAs present differences in chain length (shorter than 16 carbons or longer than 18), position or number of double bonds, or due to the addition of hydroxyl, epoxyl, ketone, acetylenic, cyclopropyl, and other functional groups (Napier 2007) (Fig. 2). The biological function of most of these unusual FAs is unknown, even though a role in defense against pests and pathogens has been put forward (Schultz et al. 1996; Cahoon et al. 2003). Many of these unusual FAs have a great potential in the chemical industry due to the interesting physicochemical properties inherent in their original structure. Although some of these FAs can be present in very high amount in the seed oils from wild species, the low yields and poor agronomical performance of these species usually preclude their large-scale commercial production. Attempts made to alter oil composition of oilseed crops through bioengineering to produce these unusual FAs and enable the development of new and productive crops that can serve as renewable sources of industrial feedstock were extensively described in excellent reviews (Aznar-Moreno and Durrett 2017b; Beaudoin et al. 2014; Carlsson et al. 2011; Dyer et al. 2008; Napier 2007; Vanhercke et al. 2013).
Variation in chain length
FA structures present variation in chain length. Elongation of acyl chains by the FAS complex involves three different KAS. The initial condensation reaction of acetyl-CoA and malonyl-ACP is catalyzed by KAS isoform III, yielding a four-carbon product. Subsequent condensations (up to C16:0-ACP) require a second KAS isoform, KASI, whereas the final elongation of C16:0-ACP to C18:0-ACP is catalyzed by a third condensing enzyme, KASII. In most species, only C16 and C18 long-chain FAs are released from the FAS complex, so that C16 and C18 FAs are the predominant FAs found in most seed oils. Down-regulation of KASII in seeds by biotechnological means can significantly increase C16 levels at the expense of C18 (Liu et al. 2017).
The synthesis of medium-chain FAs (MCFAs), including caprylic acid (C8:0), capric acid (C10:0), lauric acid (C12:0) (Fig. 2), and myristic acid (C14:0) is a variation on typical de novo FA synthesis that occurs in seeds that accumulate primarily C16 and C18 FAs. Whereas hydrolysis of long-chain acyl-ACP is catalyzed primarily by FatA (C18-ACP) and FatB thioesterases (C16-ACP), divergent FatB enzymes with substrate specificities for saturated FAs with chain length less than C16 are responsible for the synthesis of MCFAs (Kim et al. 2015b). Once the free MCFA is formed, it is exported from the plastid and acylated to glycerol, yielding medium-chain TAGs. These MCFAs are stored in seeds of members of the genera Litsea and Cuphea for example. MCFAs are important for industrial production of detergents, soaps, cosmetics, surfactants, and lubricants (Dyer et al. 2008). Laurate is currently derived from plants of the Arecaceae family (Cocos nucifera and Elaeis guineensis). Introduction of thioesterase genes with specificities toward medium-chain acyl-ACP from different Cuphea species or from California bay (Umbellularia californica) was successfully achieved in several Brassicaceae species (A. thaliana, B. napus, Camelina sativa) (Hu et al. 2017). One of the earliest successes in engineering unusual FA composition in oilseeds consisted in the production of lauric acid (Dyer et al. 2008; Wiberg et al. 2000).
Very long-chain FAs (VLCFAs) contain more than 18 carbon atoms. They are synthesized from long-chain acyl-CoA by the endoplamic reticulum (ER)-associated FA elongase (FAE) complex (Fig. 3) (Haslam and Kunst 2013). Each cycle of elongation adds two carbon units to the acyl chain and involves four reactions. Malonyl-CoA and long-chain acyl-CoA are first condensed by a β-ketoacyl-CoA synthase (KCS) (Chen et al. 2011; Jasinski et al. 2012; Mietkiewska et al. 2007). The resulting 3-ketoacyl-CoA is then reduced by the action of a β-ketoacyl-CoA reductase (KCR) (Beaudoin et al. 2009), yielding 3-hydroxyacyl-CoA that is subsequently dehydrated to 2-enoyl-CoA by a β-hydroxyacyl-CoA dehydratase (HCD). A second reduction reaction performed by an enoyl-CoA reductase (ECR) forms the elongated acyl-CoA (Salas et al. 2005). KCS is the rate-limiting enzyme for seed VLCFA production. The enzyme plays a key role in determining the chain length of FAs. VLCFAs are common components of plant seed oils in a number of plant families including Brassicaceae, Limnanthaceae and Tropaeolaceae. Erucic acid (C22:1Δ13c) (Fig. 2), obtained by elongation of oleic acid by the FAE complex, is the major VLCFA in the seed oil from Crambe abyssinica and HEAR (high erucid acid rapeseed) B. napus cultivars. A major derivative of erucic acid is erucamide, which is used as a surface-active additive in coatings and in the production of plastic films as an antiblock or slip agent (Mietkiewska et al. 2007). High concentrations of erucic acid being nutritionally undesirable, LEAR (low erucid acid rapeseed) B. napus cultivars have also been produced for food purposes, the low erucic acid phenotype of these varieties being due to variations in the sequence or expression level of the KCS-coding gene FAE1 (Roscoe et al. 2001; Yan et al. 2015).
Monounsaturated fatty acids
If the FAS complex preferentially produces C18:0-ACP, only limited amounts of stearic acid (C18:0) are exported from the plastids, so that this FA seldom accumulates in seed oils. Molecules of C18:0-ACP are efficiently desaturated by a stromal Δ9 stearoyl-ACP desaturase (SAD) of the acyl-ACP desaturase (AAD) family, so that oleic acid (C18:1Δ9cis) (Fig. 2) is preferentially exported to the ER (Fig. 4). In most plant oils, monounsaturated FAs (MUFAs) usually consist of oleic acid and its elongated derivatives gondoic acid (C20:1Δ11c) and erucic acid (C22:1Δ13c) (Fig. 2). Oleic acid, one of the most widely distributed FA in seed oils, represents an important component of human diet. Aside from food and feed uses, MUFA also have significant industrial potential. They are relatively resistant to oxidation and impart better stability for direct use of the oil in products like biolubricants or biodiesel (Durrett et al. 2008). Cleavage of their double bonds by chemical processing then yields precursors highly demanded by the chemical industry for the production of various polyamides (nylons) (Vanhercke et al. 2013). High-oleic compositions have been obtained in various oilseed crops using either conventional breeding or bioengineering. A particularly successful approach of metabolic engineering aimed at increasing the proportion of oleic acid in seed oil consisted in the concomitant inhibition, by seed-specific RNAi-mediated gene silencing, of C16:0 release from the FAS complex by FatB and of further desaturation of oleic acid by FAD2 (see below) (Vanhercke et al. 2013).
Fatty acid desaturation. The scheme depicts the different pathways involved in the biosynthesis of unsaturated fatty acids in seeds. Fatty acid chains bound to acyl carrier proteins in the plastids are presented in blue, free fatty acids released by thioesterases in black, acyl-coenzyme A in green, fatty acids bound to phosphatidylglycerol in orange, and fatty acids bound to phosphatidylcholine in purple. 26, Fatty acid synthase; 33, thioesterase; 34, fatty acid transporter; 35, long-chain acyl-coenzyme A synthetase; 36, acyl-coenzyme A transporter; 37, acyl-CoA elongase; 38, acyl–acyl carrier protein desaturase; 39, acyl-coenzyme A desaturase; 40, Δ12 fatty acid desaturase; 41, Δ15 fatty acid desaturase; 42, phosphatidylglycerol lipase. Abbreviations: PC, phosphatidylcholine; PG, phosphatidylglycerol
In some plant species, variant AAD isoforms have been characterized that vary from the archetypal Δ9 SAD in their substrate specificity or regioselectivity (Guy et al. 2011). For example, Δ9 palmitoyl-ACP desaturases (PAD) prefer C16:0-ACP as a substrate and produce palmitoleic acid (C16:1Δ9c), that can be further elongated to form vaccenic acid (C18:1Δ11c) and paullinic acid (C20:1Δ13c) (Fig. 2) (Bryant et al. 2016; Cahoon et al. 1998; Troncoso-Ponce et al. 2016a, b). These omega-7 MUFAs are abundant in seed oils of milkweed (Asclepias syriaca), macadamia (Macadamia integrifolia), and cat’s claw vine (Doxantha unguis-cati). Transcriptional activation of PAD-coding genes by the MYB115 and MYB118 transcription factors in the maturing endosperm of A. thaliana seeds was recently shown to be responsible for the tissue-specific accumulation of omega-7 MUFAs in these seeds (Troncoso-Ponce et al. 2016a). Omega-7 MUFAs sourced from sea buckthorn (Hippophae rhamnoides) berries are increasingly exploited for their positive effects on health (Wu et al. 2012). These particular MUFAs also have great industrial potential. Olefin metathesis constitutes a powerful tool for polymer chemistry and ethenolytic metathesis of omega-7 FAs from plant oils could potentially provide a competitive source of 1-octene to make linear low-density polyethylene. Due to the high economic value of plant omega-7, there is an increasing interest in either finding new natural plant sources with potentially better agronomical performances or in improving existing oilseed crops for better omega-7 production. Approaches of metabolic engineering relying on the overexpression of PAD genes have been successfully achieved in A. thaliana and in crops like C. sativa, B. napus, and soybean (Glycine max) (Bondaruk et al. 2007; Nguyen et al. 2010, 2015; Ettaki et al. 2018).
Other unusual MUFAs synthesized by divergent AAD isoforms have been described in seed oils. For example, petroselinic acid (C18:1Δ6c) is abundant in seeds of Daucus carota and Coriandrum sativum. A Δ4 PAD identified in C. sativum produces C16:1Δ4c that is further elongated to C18:1Δ6c by a specialized KAS isoform (Cahoon et al. 1992; Mekhedov et al. 2001). In seeds of Thunbergia alata, a Δ6 PAD produces sapienic acid (C16:1Δ6c) (Cahoon et al. 1994). Petroselenic and sapienic acid can be split by ozonolysis to yield adipic acid and lauric acid (C12:0) or capric acid (C10:0). Adipic acid is one of the building blocks in 6,6-nylon. A high-yielding crop with high amounts of these unusual MUFAs in seed oil would be an economically favorable alternative feedstock for the production of adipic acid (Carlsson et al. 2011). Despite the identification of the desaturases involved in the biosynthesis of these MUFAs, classical approaches of metabolic engineering by overexpression of the corresponding genes yielded low amounts of these FAs of interest in seed oils. A set of enzymes with particular specificities may be required to efficiently channel these unusual MUFAs into TAGs.
In seeds of meadowfoam (Limnanthes alba), a different mechanism of unusual MUFA biosynthesis involves a presumptive ER-bound fatty acyl-CoA desaturase. Arachidic acid (C20:0) serves as a substrate for Δ5 desaturation by this enzyme that produces C20:1Δ5c (Fig. 4) (Cahoon et al. 2000). The close position of the double bond to the carboxy terminus results in original chemical and physical properties of this very long-chain MUFA. Seed oil of Limnanthes is therefore desirable for use in cosmetics, surfactants, and lubricants. Metabolic engineering of soybean by co-expression of specialized KCS and acyl-CoA desaturase from Limnanthes resulted in the accumulation of C20:1Δ5c in the seed oil of transgenic lines (Jadhav et al. 2005).
Polyunsaturated fatty acids
Linoleic acid (C18:2Δ9c,12c) and α-linolenic acid (C18:3Δ9c,12c,15c) (Fig. 2) are the most common polyunsaturated FAs (PUFAs) found in seed oils. These important dietary PUFAs are synthesized by membrane-bound FA desaturases that sequentially desaturate oleic acid (Fig. 4). The main pathway leading to the production of linoleic and α-linolenic acid in oilseeds starts in the ER with the incorporation of oleoyl-CoA into membrane phosphatidylcholine (PC) where it is desaturated to linoleic acid by the Δ12 FA desaturase 2 (FAD2) (Kang et al. 2011; Zeng et al. 2017). Linoleoyl-PC can be further desaturated by the Δ15 FA desaturase FAD3, yielding α-linolenic acid (O’Neill et al. 2011). Plastidial membrane-bound desaturases were long known to be major contributors to PUFA biosynthesis in leaves: After incorporation into plastidial glycerolipids, oleic acid can be desaturated to linoleic acid by the Δ12 FA desaturase FAD6, yielding linoleic acid that can be further desaturated by the Δ15 FA desaturases FAD7/8 (Román et al. 2015). Interestingly, a plastid phosphatidylglycerol (PG) lipase was recently shown to hydrolyze polyunsaturated acyl groups from PG in seeds of A. thaliana (Wang et al. 2017). The acyl groups thus released are then exported from the chloroplast to be used for the biosynthesis of storage lipids in the ER.
Omega-6 and omega-3 PUFAs found in edible oils are essential for normal human growth and development. Due to increasing world population, there is an increasing interest in searching for plant oils rich in long-chain omega-3 PUFAs such as α-linolenic acid, that is abundant in linseed oil for example (Rao et al. 2008). A further interesting long-chain omega-3 PUFA is stearidonic acid (C18:4Δ6c,9c,12c,15c) present only in species of few plant families (mainly the Boraginaceae and the Primulaceae) (Guil-Guerrero et al. 2017; Kuhnt et al. 2012; Surette 2013). Stearidonic acid is synthesized from α-linolenic acid by a Δ6 FA desaturase that contains an N-terminal cytochrome b5 domain. Overexpression of the corresponding cDNA in seeds of A. thaliana or linseed was sufficient to trigger accumulation of stearidonic acid in transgenic seeds (Ruiz-Lopez et al. 2009).
Finally, dietary very long-chain omega-3 PUFAs that mostly originate from fish oils have a proven role in reducing the risk of cardiovascular disease (Mozaffarian 2008). Decreasing availability of fish oils due to overfishing led plant lipid biotechnologists to develop seed oils enriched in very long-chain omega-3 PUFAs. For example, efficient metabolic engineering of transgenic C. sativa producing the nutritionally important eicosapentaenoic acid (EPA; C20:5Δ5c,8c,11c,14c,17c) and docosahexaenoic acid (DHA; C22:6Δ4c,7c,10c,13c,16c,19c) has been reported (Ruiz-Lopez et al. 2015; Usher et al. 2017). A variety of strategies have been used to introduce the very long-chain omega-3 PUFA metabolic pathways in oil crops, mainly by expressing desaturase and elongase genes involved in different biosynthetic routes for EPA and DHA accumulation (Haslam et al. 2013).
Non-methylene-interrupted fatty acids, conjugated fatty acids
While common PUFAs of seed oils contain cis-double bonds that are separated by a single methylene group, some plant species produce unusual PUFAs with non-methylene-interrupted double bonds. A first category of non-methylene-interrupted PUFAs (NMI-PUFAs) consists of Δ5c polymethylene interrupted FAs. These PUFAs bear the first double bond on C-5 (Δ5c unsaturation) that is separated by several methylene units from the next double bond (Hammann et al. 2015). For example, the Δ5 desaturase from L. douglasii catalyzing the synthesis of C20:1Δ5c was shown to also use C22:1Δ13c as a substrate to form C22:2Δ5c,13c (Jadhav et al. 2005). Other representatives of Δ5c NMI-PUFAs are taxoleic acid (C18:2Δ5c,9c), pinolenic acid (C18:3Δ5c,9c,12c), sciadonic acid (C20:3Δ5c,11c,14c), and juniperonic acid (C20:4Δ5c,11c,14c,17c) that are present in seed oils of a great variety of gymnosperm species (Wollf et al. 2001; Wolff and Christie 2002; Hammann et al. 2015) and in some species of the Ranunculaceae family (Aitzetmuller 1995). Two ‘front end’ Δ5 desaturases that use acyl-CoA substrates to form Δ5c NMI-PUFAs were identified in Anemone leveillei (Sayanova et al. 2007). Co-expression of one of these enzymes with an elongase in transgenic A. thaliana seeds yielded production of sciadonic acid and juniperonic acid, albeit at low levels (Sayanova et al. 2007). Biomedical benefits of Δ5c NMI-PUFA-containing oils including triglyceride-lowering effect and anti-inflammatory properties have been established by several studies (Asset et al. 1999; Chen et al. 2012).
Conjugated FAs are NMI-PUFAs in which at least one pair of double bonds are separated by only one single bond (lack of methylene interruption). Conjugated linolenic acid (CLN) isomers are found in the seed oils of various plant species belonging to the Cucurbitaceae, Punicaceae, Bignoniaceae, Rosaceae, Chrysobalanaceae, Lythraceae, Balasaminaceae, and Euphorbiaceae as either C18 trienes or C18 tetraenes (Rawat et al. 2012). Examples of these conjugated FAs include α-eleostearic acid (C18:3Δ9c,11t,13t) (Fig. 2) found in bitter gourd (Momordica charantia), white mahlab (Prunus mahaleb) and tung tree (Aleurites fordii), β-eleostearic acid (C18:3Δ9t,11t,13t) in pomegranate (Punica granatum) and bitter gourd, punicic acid (C18:3Δ9c,11t,13c) in pomegranate, snakeground (Trichosanthes kirilowii), and Cayaponia africana, calendic acid (C18:3Δ8t,10t,12c) in pot marigold (Calendula officinalis), jacaric acid (C18:3Δ8c,10t,12c) in Jacaranda mimosifolia, catalpic acid (C18:3Δ9t,11t,13c) in Catalpa bignonioidesi and Catalpa ovata, and α-parinaric acid (C18:4Δ9c,11t,13t,15c) in Parinarum laurinum and Impatiens balsamina (Dulf et al. 2013; Sbihi et al. 2014). These conjugated FAs are synthesized from linoleic acid (C18:2Δ9c,12c) and α-linolenic acid (C18:3Δ9c,12c,15c) by enzymes termed conjugases (FADX) that are divergent forms of Δ12 oleate desaturase FAD2 (Cahoon et al. 2001; Qiu et al. 2001; Rawat et al. 2012). Most of the FADX are bifunctional and possess both desaturase and conjugase activities. Depending on the isoform considered, production of conjugated double bonds results from the modification of preexisting Δ9- (to produce calendic acid from linoleic acid for example) or Δ12-double bonds (to produce punicic acid from linoleic acid for example). Exploration of structure–function relationships in this class of enzymes by domain swapping and site-directed mutagenesis has shown that a small number of residues at key positions in the vicinity of the active site can have a profound impact on catalytic outcome of the enzymes (Rawat et al. 2012).
Oils rich in conjugated FAs are important medicinally as a source of nutraceuticals (Chen et al. 2016b; Dulf et al. 2013; Shabbir et al. 2017). Then, conjugated double bonds increases the rate of oxidation of conjugated FAs relative to PUFAs with methylene-interrupted double bonds, making seed oils rich in conjugated FAs well suited for use as drying agents in paints and inks. If the tung tree is commercially cultivated for production of its seed oil, the other plant species known to produce conjugated FAs do not have agronomic traits allowing large-scale production. Biotechnological efforts have therefore been directed toward the production of conjugated FAs in seeds of transgenic A. thaliana, as proof of concept, then in transgenic oilseed crops. The transfer of conjugases thus realized allowed producing α-eleostearic acid, calendic acid, or punicic acid in host seeds (Cahoon et al. 2006; Mietkiewska et al. 2014; Song et al. 2017). The amounts of conjugated FAs thus produced were much lower than what is found in seeds of plants that naturally produce conjugated FAs though. What is more, the physiology of seeds was dramatically altered as a consequence of the accumulation of likely disruptive conjugated FAs in phospholipids (Cahoon et al. 2006).
Hydroxylated fatty acids
Another important class of unusual FAs found in seed oils are hydroxylated FAs (HFAs). Castor (Ricinus communis) produces ricinoleic acid (Δ12–OH–C18:1Δ9c) (Fig. 2), and members of the Physaria and Paysonia genera accumulate oils with high amounts of lesquerolic acid (Δ14–OH–C20:1Δ11c), densipolic acid (Δ12–OH–C18:2Δ9c,15c), ricinoleic acid, and auricolic acid (Δ14–OH–C20:2Δ11c,17c) (Horn et al. 2016; Kim and Chen 2015; Yamamoto et al. 2008). Hydroxylated FAs are synthesized in the ER membrane by the addition of a hydroxyl group to the Δ12 position of oleic acid (C18:1Δ9c) esterified to PC (Smith et al. 2003). The Δ12-hydroxylases catalyzing this reaction are homolog to FAD2. Some Δ12 hydroxylases are bifunctional and possess both desaturase and hydroxylase activities. Site-directed mutagenesis experiments have demonstrated that subtle changes near the active site of the enzyme play a key role in influencing desaturation and hydroxylation product partitioning (Broun et al. 1998; Broadwater et al. 2002). Hydroxylated products of the Δ12 hydroxylase can be further desaturated and elongated in the ER (Lee et al. 2015; Horn et al. 2016; Smith et al. 2003).
Hydroxylated FAs are high-value, widely used feedstocks for industrial products, including greases, polymers, paints, coatings, and high-quality lubricants. Despite the commercial importance of castor oil, castor is considered to be unsuitable for large-scale agricultural production due to the presence of a potent toxin (ricin) and highly allergenic 2S albumins in its seeds. Improvement of castor seed for elimination of the toxic components and breeding efforts and research to improve the agronomic performance of other plant species of the Physaria and Paysonia genera producing hydroxylated FAs have been proposed. Considering that cultivation of the above-mentioned species will be limited to hot regions, development of transgenic oilseed crops for production of ricinoleic acid in high amounts in the temperate climates is desirable. Numerous attempts for expressing various Δ12 hydroxylases in A. thaliana and other Brassicaeae plants have therefore been reported (Lee et al. 2015; Snapp et al. 2014). Collectively, the accumulation of hydroxylated FAs in transgenic plants has been relatively disappointing. Research efforts are now concentrating on the elucidation of the metabolism of hydroxylated FAs in the gene-donor species to overcome metabolic bottlenecks limiting hydroxylated FAs accumulation in host seeds (Bates 2016; Horn et al. 2016; Kim et al. 2011; Kim and Chen 2015; van Erp et al. 2011).
Epoxygenated fatty acids
The most studied epoxygenated FA is vernolic acid (Δ12,13–O–C18:1Δ9c) (Fig. 2). This unusual FA is enriched in the seed oils of several Asteraceae genera, including Crepis, Vernonia, and Stokesia. Important amounts of vernolic acid were also detected in seeds of certain Euphorbiaceae species like Euphorbia lagascae and Bernardia pulchella. Coronaric acid (Δ9,10–O–C18:1Δ12c) was found in several Acacia species, in Lactuta sativa and Xeranthemum coronarium. Alchornoic acid (Δ14,15–O–C20:1Δ11c) and Δ9,10–O–C18:0 were observed in Alchornea cordifolia and Tragopogon porrifolius, respectively. The epoxy group of vernolic acid results from the insertion of an oxygen atom at the Δ12 desaturation of PC-bound linoleic acid (C18:2Δ9c,12c) (Cahoon et al. 2002). Divergent classes of enzymes can catalyze this reaction. In Crespis palestina and Vernonia galamensis, the Δ12 epoxygenase is a member of the di-iron dioxygenase family of plant membrane-bound FAD2 desaturases (Rezzonico et al. 2004). In E. lagascae, a cytochrome P450-type epoxidase is involved in the formation of the epoxy group (Cahoon et al. 2002). Thus, two distinct reaction centers carry out the same reaction but within different protein environments (Billault et al. 2012).
Because of the unique chemical properties associated with the Δ12-epoxy group, vernolic acid-enriched seed oils have a number of potential industrial applications. They can be used as a plasticizer of polyvinyl chloride, as a component of adhesives and paints or as a precursor in the manufacture of nylon. Transgenic production of epoxygenated FAs by heterologous expression of single epoxygenase genes in A. thaliana, Nicotiana tabacum, and G. max can result in accumulation of these FAs of interest in seed oils (Li et al. 2010; Rezzonico et al. 2004). However, in contrast to the native plants, the majority of the transgenic plants accumulate only low amounts of these FAs (Cahoon et al. 2002). Transgenic production of epoxygenated FAs could be improved by increasing the availability of the substrate of epoxygenases (by co-expression of the epoxygenase with a typical FAD2 enzyme in a fad3 fae1 mutant background) (Rezzonico et al. 2004) and by favoring the channeling of the epoxygenated FAs synthesized into storage lipids (Li et al. 2010, 2012).
Acetylenic fatty acids
Acetylenic bonds are present in various compounds, including FAs found in some seeds. Crepenynic acid (C18:2Δ9c,12a) (Fig. 2) is found in seed oils of plant species from Compositae, Caesalpinioideae, and Rubiaceae. Ximenynic (C18:2Δ9a,11t) and stearolic acids (C18:1Δ9a) were described in members of the Santalaceae family (Okada et al. 2013). A variant FAD2 desaturase termed acetylenase was shown to catalyze the insertion of a triple bond at the Δ12 position of linoleic acid (C18:2Δ9c,12c), forming crepenynic acid in Crepis alpina (Carlsson et al. 2004; Nam and Kappock 2007). In addition to producing crepenynic acid, the acetylenase was shown to produce both cis and trans isomers of C18:2 (Carlsson et al. 2004). Many different plants contain an acetylenase gene (Cahoon et al. 2003), but these acetylenases do not necessarily participate to the production of acetylenic FAs stored in oil. These acetylenic FAs and their biologically active polyacetylenic derivatives having potent antimicrobial, insecticidal, and antifungal properties, they are indeed synthesized in various plant tissues (Cao et al. 2013). Acetylenic FAs like crepenynic acid also represent interesting raw material for the production of high-quality coatings or cold weather ester-type lubricant for example (Lee et al. 1998).
Cyclopropane and cyclopropene fatty acids
Cyclopropane (CPA) FAs contain 3-membered carbocyclic ring. The carbocyclic group is introduced by the action of a cyclopropane FA synthase (CPS). The enzyme adds a methylene group derived from S-adenosylmethionine across the double bond of a monounsaturated FA esterified to PC (Bao et al. 2002; Yu et al. 2011). CPA FAs can be desaturated by a cyclopropane desaturase to form cyclopropene (CPE) FAs. These infrequent carbocyclic FAs are found in Malvaceae, Sterculiaceae, Bonbaceae, Tilaceae, Gnetaceae, and Sapindaceae (Yu et al. 2011). Sterculic acid (Δ9,10-cpe-C19:1) is usually the prevalent CPE FA, but malvalic acid (Δ8,9-cpe-C18:1), present in cotton (Gossypium hirsutum) seeds, can be a significant component. In Litchi chinensis seed oil, high amounts of the CPA FA dihydrosterculic acid (Δ9,10-cpa-C19:0) (Fig. 2) are accumulated. Carbocyclic FA are desirable for numerous industrial applications. For example, hydrogenation of CPA FAs such as dihydrosterculic acid results in ring opening and produces methyl-branched FAs ideally suited for use in lubricants. Oils with high levels of CPE FAs self-polymerize at elevated temperatures and therefore have potential applications as feedstocks for the production of coatings and polymers (Yu et al. 2011). For these reasons, approaches of metabolic engineering were implemented to produce carbocyclic FAs of interest in A. thaliana and C. sativa (Yu et al. 2011, 2017).
Cyclopentene fatty acids
Cyclopentene FAs like hydnocarpic (C16:1cy) (Fig. 2), chaulmoogric (C18:1cy), and gorlic acids (C18:2cyΔ6c) were described in seed oils of two tribes of the Flacourtiaceae family, Oncobeae and Pangieae. Due to their anti-inflammatory properties, some of these oils have been used in medicine for the treatment of leprosy until 1940 (Lima et al. 2005). A biosynthetic pathway involving cyclopentenylglycine transamination and oxidative decarboxylation to form aleprolic acid, which then serves as a starter molecule for cyclopentene FA synthesis by chain lengthening has been proposed (Cramer and Spener 1977). However, the actors participating in this pathway have not been identified so far in plants.
Different types of storage lipids
Wax esters
Whatever the structure of reserve lipids found in seeds, they all derive from fatty acyl-CoA thioesters and are synthesized by ER-localized enzymes. Wax esters are a class of highly hydrophobic neutral lipids that serve numerous functions in plants including carbon storage in seeds (Iven et al. 2013). The desert shrub Simmondsia chinensis (jojoba) is known to accumulate wax esters as seed storage lipids. These esters of long-chain FAs and long-chain fatty alcohols are synthesized by two enzymatic reactions (Fig. 5). Alcohol-forming fatty acyl-CoA reductase (FAR) firstly reduces acyl-CoA to corresponding alcohols and a wax synthase (WS) then catalyzes the esterification reaction of a fatty acyl-CoA with fatty alcohol (Miklaszewska and Banas 2016).
Storage lipid biosynthesis. The scheme depicts the different pathways involved in the biosynthesis of the different storage lipids found in oilseeds. Examples of wax ester (tetracosanoyl arachidate), acetyl-TAG (1,2-di-oleoyl-3-acetyl-sn-glycerol), and TAG structures (1,2,3-tri-oleoyl-sn-glycerol) are presented. 35, Long-chain acyl-coenzyme A synthetase; 43, fatty acyl-coenzyme A reductase; 44, wax synthase; 45, glycerol-3-phosphate dehydrogenase; 46, acyl-coenzyme A:sn-glycerol-3-phosphate acyltransferase; 47, acyl-coenzyme A:lysophosphatidic acid acyltransferase; 48, phosphatidic acid phosphohydrolase; 49, acyl-coenzyme A:1,2-diacyl-sn-glycerol acyltransferase; 50, CDP-choline:1,2-diacyl-sn-glycerol choline phosphotransferase; 51, phosphatidylcholine:1,2-diacyl-sn-glycerol choline phosphotransferase; 52, phospholipid:1,2-diacyl-sn-glycerol acyltransferase; 53, phospholipase A2; 54, acyl-coenzyme A:lysophosphatidylcholine acyltransferase. Abbreviations: DHAP, dihydroxyacetone phosphate; DAG, diacylglycerol; FA, fatty acid; G3P, glycerol-3-phosphate; LPA, lysophosphatidic acid; LPC, lysophosphatidylcholine; PA, phosphatidic acid; TAG, triacylglycerol
Wax esters have favorable properties for industrial applications and serve as high-pressure lubricants in all sorts of machineries (Biermaan et al. 2011; Carlsson et al. 2011). Traditionally obtained from the whaling industry, then synthesized from petrochemicals, wax esters have been produced so far from natural resources that are now diminishing. There is an increasing interest in the renewable production of wax esters in plant seed oils. However, species naturally accumulating wax esters in their seeds like jojoba are not suitable for large-scale cultivation. What is more the very long-chain wax esters produced in jojoba seeds are not suitable for technical applications such as high-pressure lubrication. Metabolic engineering of established oilseed crops has therefore been proposed for the renewable production of wax esters with tailored species composition. Different Brassicaceae species (A. thaliana, C. sativa, Crambe abyssinica, Brassica carinata) were successfully engineered for the production of wax esters (Heilmann et al. 2012; Ruiz-Lopez et al. 2017; Zhu et al. 2016). First attempts relied on the expression of FAR and WS from jojoba in seeds of the host and yielded jojoba-like wax esters in which the majority of acyl chains are longer than 20 carbons (Lardizabal et al. 2000; Zhu et al. 2016). More recently, expression of FAR and WS from other species together with approaches of systematic metabolic engineering resulted in the production of wax esters with reduced chain lengths more suitable for high-pressure and liquid lubrication purposes (Heilmann et al. 2012; Ruiz-Lopez et al. 2017).
Triacylglycerols
Triacylglycerol (TAG) is composed of three fatty acyl groups esterified to a glycerol backbone at the sn-1, sn-2 and sn-3 positions. The conversion of dihydroxyacetone phosphate (DHAP) to glycerol-3-phosphate (G3P) by glycerol-3-phosphate dehydrogenase (G3PDH) provides glycerol backbones (Fig. 5). Two acyl-CoA molecules are then used for the sequential sn-1 and sn-2 acylations of G3P. The first acylation reaction, catalyzed by acyl-CoA:sn-glycerol-3-phosphate acyltransferase (GPAT), produces lysophosphatidic acid (LPA) (Shockey et al. 2016). The acylation of LPA by acyl-CoA:lysophosphatidic acid acyltransferase (LPAAT) yields in turn phosphatidic acid (PA) (Maisonneuve et al. 2010). GPAT and LPAAT are stereo-specific with regard to acylation of the glycerol backbone and selective in terms of acyl-CoA donors (Kim et al. 2015a). PA is converted to 1,2-diacyl-sn-glycerol (DAG) by the action of phosphatidic acid phosphatase (PAP) (Eastmond et al. 2010). In the straightforward Kennedy pathway, the final acyl-CoA-dependent acylation is catalyzed by acyl-CoA:1,2-diacyl-sn-glycerol acyltransferase (DGAT), the only enzymatic reaction of the pathway exclusively committed to TAG biosynthesis. Two families of membrane-bound DGAT, DGAT1 and DGAT2, have been described (Aymé et al. 2014, 2015; Aznar-Moreno et al. 2015; Chen et al. 2017; Yu et al. 2008). They both utilize acyl-CoAs but share essentially no homology. DGAT2 were shown to have a role in catalyzing the acylation of unusual FAs such as hydroxylated FAs, epoxygenated FAs, or conjugated FAs into TAGs (Burgal et al. 2008; Li et al. 2010).
Recent studies have shown that incorporation of newly synthesized FAs into TAGs in seeds is much more complicated than simply the linear Kennedy pathway (Wang et al. 2012). DAG is an important branch point between storage and membrane lipid synthesis. The de novo DAG pool originating from G3P through the Kennedy pathway is rapidly converted to PC and then reconverted back to DAG. This second, PC-derived DAG pool is eventually channeled to TAGs (Bates et al. 2009; Bates and Browse 2011, 2013; Bates 2016). Acyl flux through PC allows accumulating PC-modified FAs in TAGs. Molecules of DAG can be converted into PC by CDP-choline:1,2-diacyl-sn-glycerol choline phosphotransferase (CPT). Although the CPT reaction is reversible, the importance of CPT in PC-derived DAG production in oilseeds remains unclear. More recently, a second enzyme was shown to participate in the interconversion of DAG and PC. Phosphatidylcholine:1,2-diacyl-sn-glycerol choline phosphotransferase (PDCT) efficiently catalyzes the transfer of phosphocholine head group between DAG and PC. PDCT activity provides a major route for desaturated acyl chains to be returned to the DAG pool (Lu et al. 2009). Finally, an acyl-CoA independent mechanism of TAG synthesis provides another way to move FAs from PC to TAG. Phospholipid:1,2-diacyl-sn-glycerol acyltransferase (PDAT) catalyzes the production of TAG through transfer of an acyl moiety from PC to DAG (Marmon et al. 2017; Pan et al. 2013; Zhang et al. 2009). This reaction may transfer unusual FAs from PC into TAG in some oleaginous species (Kim et al. 2011). The relative flux of FAs into TAG through either the DGAT or PDAT activities remains unclear in most oilseed species though.
Aside from the utilization of PC-derived DAG as the substrate for TAG synthesis and the direct transfer of FAs from PC to DAG producing TAG, acyl-editing mechanisms represent another possible route for moving unusual or polyunsaturated FAs from PC into the acyl-CoA pool for eventual TAG synthesis. Acyl editing is a PC-deacylation and lysophosphatidylcholine (LPC)–reacylation cycle allowing exchanges of FAs between PC and the acyl-CoA pool without net PC synthesis or degradation (Bates et al. 2013). This acyl-editing cycle called Lands cycle firstly involves phospholipase A2 (PLA2) that hydrolyzes acyl groups at the sn-2 position of PC, thus producing LPC and liberated FAs available for subsequent activation by LACS. Acyl-CoA thus obtained can in turn serve as substrate for the acyl-CoA-dependent acyltransferases of the Kennedy pathway. Reacylation of LPC is mediated by acyl-CoA:lysophosphatidylcholine acyltransferase (LPCAT), producing a new PC molecule different in fatty acyl chains (Wang et al. 2012). The reverse LPCAT reaction, though less thermodynamically favorable, is likely to occur in planta (Xu et al. 2012; Lager et al. 2013), but the relative amount of acyl editing by Lands cycle versus reversible LPCAT remains unclear (Bates 2016).
Vegetable oils, composed mainly of TAGs, are important nutritional and industrial commodities. To overcome ever-growing demands for these oils, increasing seed oil content and the yield of oil per unit area of land is of paramount importance. Genetic improvement of oil yields has shown fairly steady progress in oilseed crops (Weselake et al. 2009). After decades of genetic improvement driven by classical breeding programs, elucidation of the molecular bases of oil biosynthesis has provided knowledge and molecular tools useful for further increasing oil yields through marker-assisted selection or metabolic engineering. Plant lipid biotechnologists have firstly demonstrated that significant increase in seed oil content could be achieved through the manipulation of single genes (Chen et al. 2015; Hatanaka et al. 2016). Recent progress in molecular and synthetic biology has then led to the emergence of a second generation of constructs allowing multigene engineering (Aznar-Moreno and Durrett 2017a; Chhikara et al. 2017; Liu et al. 2015b; van Erp et al. 2014). Enzymes catalyzing the acyl-CoA-dependent acylation of the glycerol backbone have constituted privileged targets for biotechnological approaches (Weselake et al. 2009). Increasing the production of building blocks for TAG assembly, including G3P and FAs, was also considered. For this purpose, overexpression of G3PDH (Vigeolas et al. 2007) or FA biosynthetic genes (Roesler et al. 1997; Marchive et al. 2014) was implemented. Finally, protecting TAGs from degradation during the course of seed maturation through the seed-specific suppression of the SUGAR-DEPENDENT1 (SDP1) TAG lipase also contributed to enhance seed oil yields (Kelly et al. 2013).
While most oils accumulated in oilseed crops contain just five basic FA structures, there is a riche diversity of FA structures present in seed oils of non-crop species, many of which have potential usage in industry (Dyer et al. 2008). Many attempts have been made to modify the FA composition of seed oils so as to produce unusual FAs at commercially viable levels in oilseed crops, with varying degrees of success (Napier and Graham 2010). These approaches have underlined the importance of substrate preference of acyltransferases in the utilization of unusual FAs in transgenic oilseeds (Aznar-Moreno and Durrett 2017b; Snyder et al. 2009). For example, accumulation of medium-chain, saturated FAs in seed oils of transgenic Brassicaceae was significantly improved when co-expressing specialized acyltransferases together with medium-chain acyl-ACP thioesterases (Iskandarov et al. 2017; Knutzon et al. 1999; Wiberg et al. 2000). Specialized acyltransferases are not the only limitation to the successful engineering of crops producing desired seed oils though. Given the importance of mechanisms of acyl editing via phospholipids in FA substrate trafficking and utilization, understanding the control of acyl flux through various branches of the lipid metabolic network (Bates 2016), the substrate specificity and regulations of the enzymatic actors involved will be critical to develop more efficient engineering strategies.
Acetyl-triacylglycerols
3-Acetyl-1,2-diacyl-sn-glycerols (acetyl-TAGs) are unusual TAGs with an acetate group at the sn-3 position instead of a FA (Bansal and Durrett 2016). Acetyl-TAG molecules are abundant in the seeds of Celastraceae, Balsaminaceae, Lardizabalaceae, Ranunculaceae and Rosaceae species. A diacylglycerol acetyltransferase (DAcT) uses acetyl-CoA to acetylate DAG to form acetyl-TAG (Fig. 5) (Durrett et al. 2010). DAcT belongs to a small, plant-specific subfamily of the membrane-bound O-acyltransferases (MBOAT) that acylate different lipid substrates (Tran et al. 2017).
The presence of the sn-3 acetyl group confers useful physical, chemical, and nutritional properties to acetyl-TAGs. For example, reduced viscosity and lower melting point compared to regular TAGs make acetyl-TAGs attractive for biofuel and lubricant production (Aznar-Moreno and Durrett 2017b). Transgenic lines overexpressing the Euonymus alatus DAcT in their seeds produced important amounts of acetyl-TAGs (Durrett et al. 2010; Liu et al. 2015a). Inhibition of pathways competing for DAG substrates thanks to the RNAi suppression of DGAT1, for example, led to further increases in the amount of acetyl-TAGs accumulated.
Triacylglycerol-estolides
Acylglycerols containing more than three FAs have been found in seeds of a number of plant species like Mallotus philippensis, Trewia nudiflora, Cardamine impatiens, Sapium sebiferum, Lesquerella lyrata (Smith et al. 2013; Zhang et al. 2012). They are usually associated with the synthesis of FAs containing a hydroxyl group (Lin et al. 2006). The hydroxyl moiety of these FAs enables the attachment by ester linkage of additional FAs. The resulting molecule is usually referred to as FA-estolide. When these ester linked FAs are attached to a glycerol backbone, the molecule is termed a TAG-estolide. The pathways of biosynthesis of natural TAG-estolides are unknown. Considering the attractive potential of these molecules for industrial applications (e.g., as a source of biodegradable lubricants) and the high cost of production of synthetic TAG-estolides, there is a considerable interest in finding an abundant, natural source of these products. Alternatively, the elucidation of the biosynthetic pathways for the production of these TAG-estolides would pave the way for the metabolic engineering of estolide production in existing oilseed crops (Zhang et al. 2014b).
Conclusions and perspectives
Seed oils produced by domesticated oilseed crops, mostly in the form of TAGs, are primarily and increasingly used for nutritional applications as a consequence of population growth and changing diets. The demand for seed oils for production of biofuels and chemical feedstocks is increasing too in the light of rising environmental concerns. The use of unusual FAs of interest may represent an important step in the transition from a crude oil-based society to a more sustainable economy (Dyer and Mullen 2005). Considering that agricultural lands are finite, increasing seed oil productivity represents a first important challenge. Plant breeders and plant lipid biotechnologists already take advantage of the knowledge acquired regarding storage lipid biosynthetic pathways to increase yields of oil per unit area of land in crop species (Barthole et al. 2012; Marchive et al. 2014; Snyder et al. 2009; Weselake et al. 2009). Although it is very unlikely that seed oils will significantly contribute to replace fossil oil for energy, the prospects of doing so for some material and chemicals for the industry are much more favorable (Carlsson et al. 2011). This implies to produce entirely novel oils so that their compositions meet end-use requirements. Tailoring plant oils to have high purity of single desirable FA will rely on approaches of plant biotechnology. Despite the cloning of many genes involved in the biosynthetic pathways for unusual FAs of interest, attempts at engineering economic levels of specialty industrial FAs by transferring these genes in oilseed crops have so far met with only limited success (Vanhercke et al. 2013). This illustrates our incomplete understanding of the FA biosynthesis and accumulation pathways as well as our inability to anticipate bottlenecks to acyl flux within engineered oilseeds. The emergence of new technologies allowing the ready availability of transcript profiles, the monitoring of carbon and acyl fluxes within maturing seeds, the high-resolution spatial mapping of lipid populations within seed tissues, and the modular cloning of genes of interest will certainly allow us to deepen our knowledge of oil metabolism and to develop new engineering strategies to produce the designer oilseeds able to replace petrochemicals. Ideally, these novel non-food technical oil crops should be able to grow with minimal inputs on marginal lands so as not to compete with food crops.
References
Abdullah HM, Akbari P, Paulose B, Schnell D, Qi W, Park Y, Pareek A, Dhankher P (2016) Transcriptome profiling of Camelina sativa genes involved in triacylglycerol biosynthesis and accumulation in the developing seeds. Biotechnol Biofuels 9:136. https://doi.org/10.1186/s13068-016-0555-5
Aitzetmuller K (1995) Fatty acid patterns of Ranunculaceae seed oils: phylogenetic relationships. Pl Syst Evol 9:229–240
Alonso AP, Goffman FD, Ohlrogge JB, Shachar-Hill Y (2007) Carbon conversion efficiency and central metabolic fluxes in developing sunflower (Helianthus annuus L.) embryos. Plant J 52:296–308. https://doi.org/10.1111/j.1365-313x.2007.03235.x
Asset G, Staels B, Wolff RL, Bauge E, Madj Z, Fruchart JC, Dallongeville J (1999) Effects of Pinus pinaster and Pinus koraiensis seed oil supplementation on lipoprotein metabolism in the rat. Lipids 34:39–44
Aymé L, Baud S, Dubreucq B, Joffre F, Chardot T (2014) Function and localization of the Arabidopsis thaliana diacylglycerol acyltransferase DGAT2 expressed in yeast. PLoS ONE 9:e92237. https://doi.org/10.1371/journal.pone.0092237
Aymé L, Jolivet P, Nicaud JM, Chardot T (2015) Molecular characterization of the Elaeis guineensis medium-chain fatty acid diacylglycerol acyltransferase DGAT1-1 by heterologous expression in Yarrowia lipolytica. PLoS ONE 18:e0143113. https://doi.org/10.1371/journal.pone.0143113
Aznar-Moreno JA, Durrett TP (2017a) Simultaneous targeting of multiple gene homeologs to alter seed oil production in Camelina sativa. Plant Cell Physiol 58:1260–1267. https://doi.org/10.1093/pcp/pcx058
Aznar-Moreno JA, Durrett TP (2017b) Metabolic engineering of unusual lipids in the synthetic biology era. Plant Sci 263:126–131. https://doi.org/10.1016/j.plantsci.2017.07.007
Aznar-Moreno J, Denolf P, Van Audenhove K, De Bodt S, Engelen S, Fahy D, Wallis JG, Browse J (2015) Type 1 diacylglycerol acyltransferases of Brassica napus preferentially incorporate oleic acid into triacylglycerol. J Exp Bot 66:6497–6506. https://doi.org/10.1093/jxb/erv363
Bansal S, Durrett TP (2016) Rapid quantification of low-viscosity acetyl-triacylglycerols using electrospray ionization mass spectrometry. Lipids 51:1093–1102. https://doi.org/10.1007/s11745-016-4179-0
Bao X, Katz S, Pollard M, Ohlrogge J (2002) Carbocyclic fatty acids in plants: biochemical and molecular genetic characterization of cyclopropane fatty acid synthesis of Sterculia foetida. Proc Natl Acad Sci USA 99:7172–7177. https://doi.org/10.1073/pnas.092152999
Barthole G, Lepiniec L, Rogowsky PM, Baud S (2012) Controlling lipid accumulation in cereal grains. Plant Sci 185–186:33–39. https://doi.org/10.1016/j.plantsci.2011.09.002
Barthole G, To A, Marchive C, Brunaud V, Soubigou-Taconnat L, Berger N, Dubreucq B, Lepiniec L, Baud S (2014) MYB118 represses endosperm maturation in seeds of Arabidopsis. Plant Cell 26:3519–3537. https://doi.org/10.1105/tpc.114.130021
Bates PD (2016) Understanding the control of acyl flux through the lipid metabolic network of plant oil biosynthesis. Biochim Biophys Acta 1861:1214–1225. https://doi.org/10.1016/j.bbalip.2016.03.021
Bates PD, Browse J (2011) The pathway of triacylglycerol synthesis through phosphatidylcholine in Arabidopsis produces a bottleneck for the accumulation of unusual fatty acids in transgenic seeds. Plant J 68:387–399. https://doi.org/10.1111/j.1365-313x.2011.04693.x
Bates PD, Durrett TP, Ohlrogge JB, Pollard M (2009) Analysis of acyl fluxes through multiple pathways of triacylglycerol synthesis in developing soybean embryos. Plant Physiol 150:55–72. https://doi.org/10.1104/pp.109.137737
Bates PD, Stymne S, Ohlrogge J (2013) Biochemical pathways in seed oil synthesis. Cur Opin Plant Biol 16:358–364. https://doi.org/10.1016/j.pbi.2013.02.015
Baud S, Lepiniec L (2009) Regulation of de novo fatty acid synthesis in maturing oilseeds of Arabidopsis. Plant Physiol Biochem 47:448–455. https://doi.org/10.1016/j.plaphy.2008.12.006
Baud S, Lepiniec L (2010) Physiological regulation of seed oil production. Prog Lipid Res 49:235–249. https://doi.org/10.1016/j.plipres.2010.01.001
Baud S, Dubreucq B, Miquel M, Rochat C, Lepiniec L (2008) Storage reserve accumulation in Arabidopsis: metabolic and developmental control of seed filling. Arabidopsis Book 6:e0113. https://doi.org/10.1199/tab.0113
Beaudoin F, Wu X, Li F, Haslam RP, Markham JE, Zheng H, Napier JA, Kunst L (2009) Functional characterization of the Arabidopsis β-ketoacyl-coenzyme A reductase candidates of the fatty acid elongase. Plant Physiol 150:1174–1191. https://doi.org/10.1104/pp.109.137497
Beaudoin F, Sayanova O, Haslam RP, Bancroft I, Napier JA (2014) Oleaginous crops as integrated production platforms for food, feed, fuel and renewable industrial feedstock-Manipulation of plant lipid composition via metabolic engineering and new opportunities from association genetics for crop improvement and valorisation of co-products. OCL 21:D606. https://doi.org/10.1051/ocl/2014042
Biermann U, Bornscheuer U, Meier MAR, Metzger JO, Schäfer HJ (2011) Oils and fats as renewable raw materials in chemistry. Angew Chem Int Ed Engl 50:3854–3871. https://doi.org/10.1002/anie.201002767
Billault I, Le Du A, Ouethrani M, Serhan Z, Lesot P, Robins RJ (2012) Probing substrate-product relationships by natural abundance deuterium 2D NMR spectroscopy in liquid-crystalline solvents: epoxidation of linoleate to vernoleate by two different plant enzymes. Anal Bioanal Chem 402:2985–2998. https://doi.org/10.1007/s00216-012-5748-6
Bondaruk M, Johnson S, Degafu A, Boora P, Bilodeau P, Morris J, Wiehler W, Foroud N, Weselake R, Shah S (2007) Expression of a cDNA encoding palmitoyl-acyl carrier protein desaturase from cat’s claw (Doxantha unguis-cati L.) in Arabidopsis thaliana and Brassica napus leads to accumulation of unusual unsaturated fatty acids and increased stearic acid content in the seed oil. Plant Breed 126:186–194. https://doi.org/10.1111/j.1439-0523.2007.01316.x
Borisjuk L, Rolletschek H, Neuberger T (2013) Nuclear magnetic resonance imaging of lipid in living plants. Prog Lipid Res 52:465–487. https://doi.org/10.1016/j.plipres.2013.05.003
Broadwater JA, Whittle E, Shanklin J (2002) Desaturation and hydroxylation: residues 148 and 324 of Arabidopsis FAD2, in addition to substrate chain length, exert a major influence in partitioning of catalytic specificity. J Biol Chem 277:15613–15620
Broun P, Shanklin J, Whittle E, Somerville C (1998) Catalytic plasticity of fatty acid modification enzymes underlying chemical diversity of plant lipids. Science 282(1315):1317. https://doi.org/10.1074/jbc.m200231200
Bryant FM, Munoz-Ascarate O, Kelly AA, Beaudoin F, Kurup S, Eastmond PJ (2016) ACYL-ACYL CARRIER PROTEIN DESATURASE2 and 3 are responsible for making omega-7 fatty acids in the Arabidopsis aleurone. Plant Physiol 172:154–162. https://doi.org/10.1104/pp.16.00836
Burgal J, Shockey J, Chaofu Lu, Dyer J, Larson T, Graham I, Browse J (2008) Metabolic engineering of hydroxyl fatty acid production in plants: RcDGAT2 drives dramatic increases in ricinoleate levels in seed oil. Plant Biotechnol J 6:819–831. https://doi.org/10.1111/j.1467-7652.2008.00361.x
Cahoon EB, Shanklin J, Ohlrogge JB (1992) Expression of a coriander desaturase results in petroselinic acid production in transgenic tobacco. Proc Natl Acad Sci USA 89:11184–11188
Cahoon EB, Becker CK, Shanklin J, Ohlrogge JB (1994) cDNAs for isoforms of the ∆9-stearoyl-acyl carrier protein desaturase from Thunbergia alata endosperm. Plant Physiol 106:807–808
Cahoon EB, Shah S, Shanklin J, Browse J (1998) A determinant of substrate specificity predicted from the acyl-acyl carrier protein desaturase of developing cat’s claw seed. Plant Physiol 117:593–598
Cahoon EB, Marillia E-F, Stecca KL, Hall SE, Taylor DC, Kinney AJ (2000) Production of fatty acid components of meadowfoam oil in somatic soybean embryos. Plant Physiol 124:243–251
Cahoon EB, Ripp KG, Hall SE, Kinney AJ (2001) Formation of conjugated ∆8, ∆10-double bonds by ∆12-oleic-acid desaturase-related enzymes. J Biol Chem 276:2637–2643. https://doi.org/10.1074/jbc.m009188200
Cahoon EB, Ripp KG, Hall SE, McGonigle B (2002) Transgenic production of epoxy fatty acids by expression of a cytochrome P450 enzyme from Euphorbia lagascae seed. Plant Physiol 128:615–624. https://doi.org/10.1104/pp.010768
Cahoon EB, Schnurr JA, Huffman EA, Minto RE (2003) Fungal responsive fatty acid acetylenases occur widely in evolutionarily distant plant families. Plant J 34:671–683
Cahoon EB, Dietrich CR, Meyer K, Damude HG, Dyer JM, Kinney AJ (2006) Conjugated fatty acids accumulate to high levels in phospholipids of metabolically engineered soybean and Arabidopsis seeds. Phytochemistry 67:1166–1176. https://doi.org/10.1016/j.phytochem.2006.04.013
Cao S, Zhou X-R, Wood CC, Green AG, Singh SP, Liu L, Liu Q (2013) A large functionally diverse family of Fad2 genes in safflower (Carthamus tinctorius L.). BMC Plant Biol 13:5. https://doi.org/10.1186/1471-2229-13-5
Cao H, He M, Zhu C, Yuan L, Dong L, Bian Y, Zhang W, Yan Y (2016) Distinct metabolic changes between wheat embryo and endosperm during grain development revealed by 2D-DIGE-based integrative proteome analysis. Proteomics 16:1515–1536. https://doi.org/10.1002/pmic.201500371
Carlsson AS, Thomaeus S, Hamberg M, Stymne S (2004) Properties of two multifunctional plant fatty acid acetylenase/desaturase enzymes. Eur J Biochem 271:2991–2997. https://doi.org/10.1111/j.1432-1033.2004.04231.x
Carlsson AS, Yilmaz JL, Green AG, Stymne S, Hofvander P (2011) Replacing fossil oil with fresh oil–with what and for what? Eur J Lipid Sci Technol 113:812–831. https://doi.org/10.1002/ejlt.201100032
Chen Y, Kelly EE, Masluk RP, Nelson CL, Cantu DC, Reilly PJ (2011) Structural classification and properties of ketoacyl synthases. Protein Sci 20:1659–1667. https://doi.org/10.1002/pro.712
Chen S-J, Huang W-C, Yang T-T, Lu J-H, Chuang L-T (2012) Incorporation of sciadonic acid into cellular phospholipids reduces pro-inflammatory mediators in murine macrophage through NF-κB and MAPK signaling pathways. Food Chem Toxicol 50:3687–3695. https://doi.org/10.1016/j.fct.2012.07.057
Chen L, Lee JH, Weber H, Tohge T, Witt S, Roje S, Fernie AR, Hellmann H (2013) Arabidopsis BPM proteins function as substrate adaptors to a CULLIN3-based E3 ligase to affect fatty acid metabolism in plants. Plant Cell 25:2253–2264. https://doi.org/10.1105/tpc.112.107292
Chen S, Lei Y, Xu X, Huang J, Jiang H, Wang J, Cheng Z, Zhang J, Song Y, Liao B, Li Y (2015) The peanut (Arachis hypogaea L.) gene AhLPAT2 increases the lipid content of transgenic seeds. PLoS ONE 10:e0136170. https://doi.org/10.1371/journal.pone.0136170
Chen J, Qiang Y, Cao Y, Wei B, Zheng L, Xiao Q, Xie Y, Gu Y, Li Y, Huang H, Wang Y, Hou X, Long T, Zhang J, Liu H, Liu Y, Yu G, Huang Y (2016a) ZmbZIP91 regulates expression of starch synthesis-related genes by binding to ACTCAT elements in their promoters. J Exp Bot 67:1327–1338. https://doi.org/10.1093/jxb/erv527
Chen G-C, Su H-M, Lin Y-S, Tsou P-Y, Chyan J-H, Chao P-M (2016b) A conjugated fatty acid present at high levels in bitter melon seed favorably affects lipid metabolism in hepatocytes by increasing NAD+/NADH ratio and activating PPARα, AMPK and SIRT1 signaling pathway. J Nutr Biochem 33:28–35. https://doi.org/10.1016/j.jnutbio.2016.03.009
Chen G, Xu Y, Siloto RMP, Caldo KMP, Vanhercke T, El Tanchy A, Niesner N, Chen Y, Mietkiewska E, Weselake RJ (2017) High-performance variants of plant diacylglycerol acyltransferase 1 generated by directed evolution provide insights into structure function. Plant J 92:167–177. https://doi.org/10.1111/tpj.13652
Chhikara S, Abdullah HM, Akbari P, Schnell D, Dhankher OP (2017) Engineering Camelina sativa (L) Crantz for enhanced oil and seed yields by combining diacylglycerol acyltransferase1 and glycerol-3-phosphate dehydrogenase expression. Plant Biotechnol J. https://doi.org/10.1111/pbi.12847
Cramer U, Spener F (1977) Biosynthesis of cyclopenteny fatty acids. Eur J Biochem 74:495–500
Cross JM, Clancy M, Shaw JR, Greene TW, Schmidt RR, Okita TW, Hannah LC (2004) Both subunits of ADP-glucose pyrophosphorylase are regulatory. Plant Physiol 135:137–144. https://doi.org/10.1104/pp.103.036699
da Silva PMFR, Eastmond PJ, Hill LM, Smith AM, Rawsthorne S (1997) Starch metabolism in developing embryos of oilseed rape. Planta 203:480–487
Devic M, Roscoe T (2016) Seed maturation: Simplification of control networks in plants. Plant Sci 252:335–346. https://doi.org/10.1016/j.plantsci.2016.06.013
Dulf FV, Pamfil D, Baciu AD, Pintea A (2013) Fatty acid composition of lipids in pot marigold (Calendula officinalis L.) seed genotypes. Chem Cent J 7:8. https://doi.org/10.1186/1752-153x-7-8
Durrett TP, Benning C, Ohlrogge JB (2008) Plant triacylglycerols as feedstocks for the production of biofuels. Plant J 54:593–607. https://doi.org/10.1111/j.1365-313x.2008.03442.x
Durrett TP, McClosky DD, Tumaney AW, Elzinga DA, Ohlrogge J, Pollard M (2010) A distinct DGAT with sn-3 acyltransferase activity that synthesizes unusual, reduced-viscosity oils in Eunymus and transgenic seeds. Proc Natl Acad Sci USA 107:9464–9469. https://doi.org/10.1073/pnas.1001707107
Dussert S, Guerin C, Andersson M, Joët T, Tranberger TJ, Pizot M, Sarah G, Omore A, Durand-Gasselin T, Morcillo F (2013) Comparative transcriptome analysis of three oil palm fruit and seed tissues that differ in oil content and fatty acid composition. Plant Physiol 162:1337–1358. https://doi.org/10.1104/pp.113.220525
Dyer JM, Mullen RT (2005) Development and potential of genetically engineered oilseeds. Seed Sci Res 15:255–267. https://doi.org/10.1079/ssr2005216
Dyer JM, Stymne S, Green AG, Carlsson AS (2008) High-value oils from plants. Plant J 54:640–655. https://doi.org/10.1111/j.1365-313x.2008.03430.x
Eastmond PJ, Quettier A-L, Kroon JTM, Craddock C, Adams N, Slabas AR (2010) PHOSPHATIDIC ACID PHOSPHOHYDROLASE 1 and 2 regulate phospholipid synthesis at the endoplasmic reticulum in Arabidopsis. Plant Cell 22:2796–2811. https://doi.org/10.1105/tpc.109.071423
Eskandari M, Cober ER, Rajcan I (2013) Genetic control of soybean seed oil: II. QTL and genes that increase oil concentration without decreasing protein or with increased seed yield. Theor Appl Genet 126:1677–1687. https://doi.org/10.1007/s00122-013-2083-z
Ettaki H, Troncoso-Ponce MA, To A, Barthole G, Lepiniec L, Baud S (2018) Overexpression of MYB115, AAD2, or AAD3 in Arabidopsis thaliana seeds yields contrasting omega-7 contents. PLoS ONE 13:e0192156. https://doi.org/10.1371/journal.pone.0192156
Fatihi A, Boulard C, Bouyer D, Baud S, Dubreucq B, Lepiniec L (2016) Deciphering and modifying LAFL transcriptional regulatory network in seed for improving yield and quality of storage compounds. Plant Sci 250:198–204. https://doi.org/10.1016/j.plantsci.2016.06.013
Furumoto T, Yamaguchi T, Ohshima-Ichie Y, Nakamura M, Tsuchida-Iwata Y, Shimamura M, Ohnishi J, Hata S, Gowik U, Westhoff P, Bräutigam A, Weber AP, Izui K (2011) A plastidial sodium-dependent pyruvate transporter. Nature 476:472–475. https://doi.org/10.1038/nature10250
Graham IA (2008) Seed storage oil mobilization. Annu Rev Plant Biol 59:115–142. https://doi.org/10.1146/annurev.arplant.59.032607.092938
Grimberg Å, Carlsson AS, Marttila S, Bhalerao R, Hofvander P (2015) Transcriptional transitions in Nicotiana benthamiana leaves upon induction of oil synthesis by WRINKLED1 homologs from diverse species and tissues. BMC Plant Biol 15:192. https://doi.org/10.1186/s12870-015-0579-1
Guerin C, Joët T, Serret J, Lashermes P, Vaissayre V, Agbessi MDT, Beulé T, Severac D, Amblard P, Tregear J, Durand-Gasselin T, Morcillo F, Dussert S (2016) Gene coexpression network analysis of oil biosynthesis in an interspecific backcross of oil palm. Plant J 87:423–441. https://doi.org/10.1111/tpj.13208
Guil-Guerrero J, Gomez-Mercado F, Ramos-Bueno RP, Gonzales-Fernandez MJ, Urrestarazu M, Rincon-Cervera MA (2017) Sardinian Boraginaceae are new potential sources of gamma-linolenic acid. Food Chem 218:435–439. https://doi.org/10.1016/j.foodchem.2016.09.111
Guillon F, Bouchet B, Jamme F, Robert P, Quemener B, Barron C, Larre C, Dumas P, Saulnier L (2011) Brachypodium distachyon grain: characterization of endosperm cell walls. J Exp Bot 62:1001–1015. https://doi.org/10.1093/jxb/erq332
Guy EJ, Whittle E, Moche M, Lengqvist J, Lindqvist Y, Shanklin J (2011) Remote control of regioselectivity in acyl-acyl carrier protein-desaturase. Proc Natl Acad Sci USA 108:16594–16599. https://doi.org/10.1073/pnas.1110221108
Hammann S, Schröder M, Schmidt C, Vetter W (2015) Isolation of two Δ5 polymethylene interrupted fatty acids from Podocarpus falcatus by countercurrent chromatography. J Chromatogr A 1394:89–94. https://doi.org/10.1016/j.chroma.2015.03.042
Haslam T, Kunst L (2013) Extending the story of very-long-chain fatty acid elongation. Plant Sci 210:93–107. https://doi.org/10.1016/j.plantsci.2013.05.008
Haslam RP, Ruiz-Lopez N, Eastmond P, Moloney M, Sayanova O, Napier JA (2013) The modification of plant oil composition via metabolic engineering–better nutrition by design. Plant Biotechnol J 11:157–168. https://doi.org/10.1111/pbi.12012
Hatanaka T, Serson W, Li R, Armstrong P, Yu K, Pfeiffer T, Li X-L, Hildebrand D (2016) A Vernonia diacylglycerol acyltransferase can increase renewable oil production. J Agric Food Chem 64:7188–7194. https://doi.org/10.1021/acs.jafc.6b02498
Heilmann M, Iven T, Ahmann K, Hornung E, Stymne S, Feussner I (2012) Production of wax esters in plant seed oils by oleosomal cotargeting of biosynthetic enzymes. J Lipid Res 53:2153–2161. https://doi.org/10.1194/jlr.m029512
Horn PJ, Chapman KD (2014) Lipidomics in situ: insights into plant lipid metabolism from high resolution spatial maps of metabolites. Prog Lipid Res 54:32–52. https://doi.org/10.1016/j.plipres.2014.01.003
Horn PJ, Liu J, Cocuron J-C, McGlew K, Thrower NA, Larson M, Lu C, Alonso AP, Ohlrogge J (2016) Identification of multiple lipid genes with modifications in expression and sequence associated with the evolution of hydroxy fatty acid accumulation in Physaria fendleri. Plant J 86:323–348. https://doi.org/10.1111/tpj.13163
Houston K, Russell J, Schreiber M, Halpin C, Oakey H, Washington JM, Booth A, Shirley N, Burton RA, Fincher GB, Waugh R (2014) A genome wide association scan for (1,3;1,4)-β-glucan content in the grain of contemporary 2-row Spring and Winter barleys. BMC Genom 15:907. https://doi.org/10.1186/1471-2164-15-907
Hrmova M, De Gori R, Smith BJ, Fairweather JK, Varghese JN, Fincher GB (2002) Structural basis for broad substrate specificity in higher plant beta-D-glucan glucohydrolases. Plant Cell 14:1033–1052
Hu Z, Wu Q, Dalal J, Vasani N, Lopez HO, Sederoff HW, Qu R (2017) Accumulation of medium-chain, saturated fatty acyl moieties in seed oils of transgenic Camelina sativa. PLoS ONE 12:e0172296. https://doi.org/10.1371/journal.pone.0172296
Huang R, Huang Y, Sun Z, Huang J, Wang Z (2017) Transcriptome analysis of genes involved in lipid biosynthesis in the developing embryo of pecan (Carya illinoinensis). J Agric Food Chem 65:4223–4236. https://doi.org/10.1021/acs.jafc.7b00922
Ingram GC (2017) Dying to live: cell elimination as a developmental strategy in angiosperm seeds. J Exp Bot 68:785–796. https://doi.org/10.1093/jxb/erw364
Iskandarov U, Silva JE, Kim HJ, Andersson M, Cahoon RE, Mockaitis K, Cahoon EB (2017) A specialized diacylglycerol acyltransferase contributes to the extreme medium-chain fatty acid content of Cuphea seed oil. Plant Physiol 174:97–109. https://doi.org/10.1104/pp.16.01894
Iven T, Herrfurth C, Hailmann M, Hofvander P, Stymne S, Zhu L-H, Feussner I (2013) Wax ester profiling of seed oil by nano-electrospray ionization tandem mass spectrometry. Plant Methods 9:24. https://doi.org/10.1186/1746-4811-9-24
Iyer VV, Sriram G, Fulton DB, Zhou R, Westgate ME, Shanks JV (2008) Metabolic flux maps comparing the effects of temperature on protein and oil biosynthesis in developing soybean cotyledons. Plant Cell Environ 31:506–517. https://doi.org/10.1111/j.1365-3040.2008.01781.x
Jadhav A, Marillia E-F, Babic V, Giblin EM, Cahoon EB, Kinney AJ, Mietkiewska E, Brost JM, Taylor DC (2005) Production of 22:2∆5,∆13 and 20:1∆5 in Brassica carinata and soybean breeding lines via introduction of Limnanthes genes. Mol Breed 15:157–167
Jasinski S, Lécureuil A, Miquel M, Loudet O, Froissard M, Guerche P (2012) Natural variation in seed long chain fatty acid content is controlled by a new isoform of KCS18 in Arabidopsis thaliana. PLoS ONE 7:e49261. https://doi.org/10.1371/journal.pone.0049261
Kanai M, Mano S, Kondo M, Hayashi M, Nishimura M (2015) Extension of oil biosynthesis during the mid-phase of seed development enhances oil content in Arabidopsis seeds. Plant Biotechnol J 14:1241–1250. https://doi.org/10.1111/pbi.12489
Kang J, Snapp AR, Lu C (2011) Identification of three genes encoding microsomal oleate desaturases (FAD2) from the oilseed crop Camelina sativa. Plant Physiol Biochem 49:223–229. https://doi.org/10.1016/j.plaphy.2010.12.004
Kelly AA, Shaw E, Powers SJ, Kurup S, Eastmond PJ (2013) Suppression of the SUGAR-DEPENDENT1 triacylglycerol lipase family during seed development enhances oil yield in oilseed rape (Brassica napus L.). Plant Biotechnol J 11:355–361. https://doi.org/10.1111/pbi.12021
Kim HU, Chen GQ (2015) Identification of hydroxyl fatty acid and triacylglycerol metabolism-related genes in lesquerella through seed transcriptome analysis. BMC Genom 16:230. https://doi.org/10.1186/s12864-015-1413-8
Kim HU, Lee K-R, Go YS, Jung JH, Suh M-C, Kim JB (2011) Endoplasmic reticulum-located PDAT1-2 from castor bean enhances hydroxyl fatty acid accumulation in transgenic plants. Plant Cell Physiol 52:983–993. https://doi.org/10.1093/pcp/pcr051
Kim S, Yamaoka Y, Ono H, Kim H, Shim D, Maeshima M, Martinoia E, Cahoon EB, Nishida I, Lee Y (2013) AtABCA9 transporter supplies fatty acids for lipid synthesis to the endoplasmic reticulum. Proc Natl Acad Sci USA 110:773–778. https://doi.org/10.1073/pnas.1214159110
Kim HJ, Silva JE, Iskandarov U, Andersson M, Cahoon RE, Mockaitis K, Cahoon EB (2015a) Structurally divergent lysophosphatidic acid acyltransferases with high selectivity for saturated medium chain fatty acids from Cuphea seeds. Plant J 84:1021–1033. https://doi.org/10.1111/tpj.13063
Kim HJ, Silva JE, Vu HS, Mockaitis K, Nam J-W, Cahoon EB (2015b) Toward production of jet fuel functionality in oilseeds: identification of FatB acyl-acyl carrier protein thioesterases and evaluation of combinatorial expression strategies in Camelina seeds. J Exp Bot 66:4251–4265. https://doi.org/10.1093/jxb/erv225
Kim MJ, Jang I-C, Chua N-H (2016) The mediator complex MED15 subunit mediates activation of downstream lipid-related genes by the WRINKLED1 transcription factor. Plant Physiol 171:1951–1964. https://doi.org/10.1104/pp.16.00664
Knutzon DS, Hayes TR, Wyrick A, Xiong H, Davies HM, Voelker TA (1999) Lysophosphatidic acid acyltransferase from Coconut endosperm mediates the insertion of laurate at the sn-2 position in triacylglycerols in lauric rapeseed oil and can increase total laurate levels. Plant Physiol 120:739–746
Kuhnt K, Degen C, Jaudszus A, Jahreis G (2012) Searching for health beneficial n-3 and n-6 fatty acids in plant seeds. Eur J Lipid Sci Technol 114:153–160. https://doi.org/10.1002/ejlt.201100008
Lager I, Yilmaz JL, Zhou X-R, Jasieniecka K, Kazachkov M, Wang P, Zou J, Weselake R, Smith MA, Bayon S, Dyer JM, Shockey JM, Heinz E, Green A, Banas S, Stymne S (2013) Plant acyl-CoA:lysophosphatidylcholine acyltransferases (LPCATs) have different specificities in their forward and reverse reactions. J Biol Chem 288:36902–36914. https://doi.org/10.1074/jbc.m113.521815
Lardizabal KD, Metz JG, Sakamoto T, Hutton WC, Pollard MR, Lassner MW (2000) Purification of a jojoba embryo wax synthase, cloning of its cDNA, and production of high levels of wax in seeds of transgenic Arabidopsis. Plant Physiol 122:645–655
Lee M, Lenman M, Banas A, Bafor M, Singh S, Schweizer M, Nilsson R, Liljenberg C, Dahlqvist A, Gummeson P-O, Sjödahl S, Green A, Stymne S (1998) Identification of non-heme diiron proteins that catalyze triple bond and epoxy group formation. Science 280:915–918
Lee K-R, Chen GQ, Kim HU (2015) Current progress towards the metabolic engineering of plant seed oil for hydroxyl fatty acids production. Plant Cell Rep 34:603–615. https://doi.org/10.1007/s00299-015-1736-6
Lee EJ, Oh M, Hwang JU, Li-Beisson Y, Nishida I, Lee Y (2017) Seed-specific overexpression of the pyruvate transporter BASS2 increases oil content in Arabidopsis seeds. Front Plant Sci 8:194. https://doi.org/10.3389/fpls.2017.00194
Li R, Yu K, Hatanaka T, Hildebrand DF (2010) Vernonia DGATs increase accumulation of epoxy fatty acids in oil. Plant Biotechnol J 8:184–195. https://doi.org/10.1111/j.1467-7652.2009.00476.x
Li R, Yu K, Wu Y, Tateno M, Hatanaka T, Hildebrand DF (2012) Vernonia DGATs can complement the disrupted oil and protein metabolism in epoxygenase-expressing soybean seeds. Metab Eng 14:29–38. https://doi.org/10.1016/j.ymben.2011.11.004
Li N, Gügel IL, Giavalisco P, Zeisler V, Schreiber L, Soll J, Philippar K (2015a) FAX1, a novel membrane protein mediating plastid fatty acid export. PLoS Biol 13:e1002053. https://doi.org/10.1371/journal.pbio.1002053
Li SS, Wang LS, Shu QY, Wu J, Chen LG, Shao S, Yin DD (2015b) Fatty acid composition of developing tree peony (Paeonia section Moutan DC.) seeds and transcriptome analysis during seed development. BMC Genom 16:208. https://doi.org/10.1186/s12864-015-1429-0
Li N, Xu C, Li-Beisson Y, Philippar K (2016) Fatty acid and lipid transport in plant cells. Trends Plant Sci 21:145–158. https://doi.org/10.1016/j.tplants.2015.10.011
Li-Beisson Y, Shorrosh B, Beisson F, Andersson MX, Arondel V, Bates PD, Baud S, Bird D, DeBono A, Durrett TP, Franke RB, Graham IA, Katayama K, Kelly AA, Larson T, Markham JE, Miquel M, Molina M, Nishida I, Rowland O, Samuels L, Schmid KM, Wada H, Welti R, Xu C, Zallot R, Ohlrogge J (2010) Acyl-lipid metabolism. Arabidopsis Book 8:e0133. https://doi.org/10.1199/tab.0133
Lima JA, Oliveira AS, de Miranda ALP, Rezende CM, Pinto AC (2005) Anti-inflammatory and antinociceptive activities of an oil fraction of the seeds of Carpotroche brasiliensis (Raddi) (Flacourtiaceae). Braz J Med Biol Res 38:1095–1103. https://doi.org/10.1590/S0100-879X2005000700013
Lin J-T, Arcinas A, Harden LR, Fagerquist CK (2006) Identification of (12-ricinoleoylricinoleoyl)diricinoleoylglycerol, an acylglycerol containing four acyl chains, in castor (Ricinus communis L.) oil by LC-ESI-MS. J Agric Food Chem 54:3498–3504. https://doi.org/10.1021/jf060150e
Liu J, Hua W, Zhan G, Wei F, Wang X, Liu G, Wang H (2009) Increasing seed mass and oil content in transgenic Arabidopsis by the overexpression of wri1-like gene from Brassica napus. Plant Physiol Biochem 48:9–15. https://doi.org/10.1016/j.plaphy.2009.09.007
Liu J, Rice A, McGlew K, Shaw V, Park H, Clemente T, Pollard M, Ohlrogge J, Durrett TP (2015a) Metabolic engineering of oilseed crops to produce high levels of novel acetyl glyceride oils with reduced viscosity, freezing point and calorific value. Plant Biotechnol J 13:858–865. https://doi.org/10.1111/pbi.12325
Liu F, Xia Y, Wu L, Fu D, Hayward A, Luo J, Yan X, Xiong X (2015b) Enhanced seed oil content by overexpressing genes related to triacylglyceride synthesis. Gene 557:163–171. https://doi.org/10.1016/j.gene.2014.12.029
Liu Q, Wu M, Zhang B, Shrestha P, Petrie J, Green AG, Singh SP (2017) Genetic enhancement of palmitic acid accumulation in cotton seed oil through RNAi down-regulation of ghKAS2 encoding β-ketoacyl-ACP synthase II (KASII). Plant Biotch J 15:132–143
Lonien J, Schwender J (2009) Analysis of metabolic flux phenotypes for two Arabidopsis mutants with severe impairment in seed storage lipid synthesis. Plant Physiol 151:1617–1634. https://doi.org/10.1104/pp.109.144121
Lu C, Xin Z, Ren Z, Miquel M, Browse J (2009) An enzyme regulating triacylglycerol composition is encoded by the ROD1 gene of Arabidopsis. Proc Natl Acad Sci USA 106:18837–18842. https://doi.org/10.1073/pnas.0908848106
Ma W, Kong Q, Arondel V, Kilaru A, Bates PD, Thrower NA, Benning C, Ohlrogge JB (2013) WRINKLED1, a ubiquitous regulator in oil accumulating tissues from Arabidopsis embryos to oil palm mesocarp. PLoS ONE 7:e68887. https://doi.org/10.1371/journal.pone.0068887
Ma W, Kong Q, Mantyla JJ, Yang Y, Ohlrogge JB, Benning C (2016) 14-3-3 protein mediates plant seed oil biosynthesis through interaction with AtWRI1. Plant J 88:228–235. https://doi.org/10.1111/tpj.13244
Maeo K, Tokuda T, Ayame A, Mitsui N, Kawai T, Tsukogashi H, Ishiguro S, Nakamura K (2009) An AP2-type transcription factor, WRINKLED1, of Arabidopsis thaliana binds to the AW-box sequence conserved among proximal upstream regions of genes involved in fatty acid synthesis. Plant J 60:476–487. https://doi.org/10.1111/j.1365-313x.2009.03967.x
Mahmood T, Rahman MH, Stringam GR, Yeh F, Good AG (2006) Identification of quantitative trait loci (QTL) for oil and protein contents and their relationships with other seed quality traits in Brassica juncea. Theor Appl Genet 113:1211–1220. https://doi.org/10.1007/s00122-006-0376-1
Maisonneuve S, Bessoule J-J, Lessire R, Delseny M, Roscoe TJ (2010) Expression of rapeseed microsomal lysophosphatidic acid acyltransferase isozymes enhances seed oil content in Arabidopsis. Plant Physiol 152:670–684. https://doi.org/10.11104/pp.109.148247
Mao TT, Jiang ZF, Han YP, Teng WL, Zhao X, Li WB (2013) Identification of quantitative trait loci underlying seed protein and oil contents of soybean across multi-genetic backgrounds and environments. Plant Breed 132:630–641
Marchive C, Nikovics K, To A, Lepiniec L, Baud S (2014) Transcriptional regulation of fatty acid production in higher plants: molecular bases and biotechnological outcomes. Eur J Lipid Sci Technol 116:1332–1343. https://doi.org/10.1002/ejlt.201400027
Marcotuli I, Houston K, Schwerdt JG, Waugh R, Fincher GB, Burton RA, Blanco A, Gadaleta A (2016) Genetic diversity and genome wide association study of β-glucan content in tetraploid wheat grains. PLoS ONE 11:e0152590. https://doi.org/10.1371/journal.pone.0152590
Marmon S, Sturtevant D, Herrfurth C, Chapman K, Stymne S, Feussner I (2017) Two acyltransferases contribute differently to linolenic acid levels in seed oil. Plant Physiol 173:2081–2095. https://doi.org/10.1104/pp.16.01865
Mekhedov S, Cahoon EB, Ohlrogge J (2001) An unusual seed-specific 3-ketoacyl-ACP synthase associated with the biosynthesis of petroselinic acid in coriander. Plant Mol Biol 47:507–518
Mietkiewska E, Brost JM, Giblin EM, Bartin DL, Taylor DC (2007) Cloning and functional characterization of the fatty acid elongase 1 (FAE1) genre from high erucic Crambe abyssinica cv. Prophet. Plant Biotechnol J 5:636–645. https://doi.org/10.1111/j.1467-7652.2007.00268.x
Mietkiewska E, Liles R, Wickramarathna A, Sahibollah AF, Greer MS, Chen G, Weselake RJ (2014) Combined transgenic expression of Punica granatum conjugase (FADX) and FAD2 desaturase in high linoleic acid Arabidopsis thaliana mutants leads to increased accumulation of punicic acid. Planta 240:575–583. https://doi.org/10.1007/s00425-014-2109-z
Miklaszewska M, Banas A (2016) Biochemical characterization and substrate specificity of jojoba fatty acyl-CoA reductase and jojoba wax synthase. Plant Sci 249:84–92. https://doi.org/10.1016/j.plantsci.2016.05.009
Miquel M, Trigui G, d’Andréa S, Kelemen Z, Baud S, Berger A, Deruyffelaere C, Trubuil A, Lepiniec L, Dubreucq B (2014) Specialization of oleosins in oil body dynamics during seed development in Arabidopsis seeds. Plant Physiol 164:1866–1878. https://doi.org/10.1104/pp.113.233262
Moreno-Pérez A, Venegas-Calerón M, Vaistij FE, Salas JJ, Larson TR, Gercés R, Graham IA, Martínez-Force E (2012) Reduced expression of FatA thioesterases in Arabidopsis affects the oil content and fatty acid composition of the seeds. Planta 235:629–639. https://doi.org/10.1007/s00425-011-1534-5
Mozaffarian D (2008) Fish and n-3 fatty acids for the prevention of fatal coronary heart disease and sudden cardiac death. Am J Clin Nutr 87:19915–19965
Mu J, Tan H, Zheng Q, Fu F, Liang Y, Zhang J, Yang X, Wang T, Chong K, Wang X-J, Zuo J (2008) LEAFY COTYLEDON1 is a key regulator of fatty acid biosynthesis in Arabidopsis. Plant Physiol 148:1042–1054. https://doi.org/10.1104/pp.108.126342
Mukherjee S, Liu A, Deol KK, Kulichikhin K, Stasolla C, Brûlé-Babel A, Ayele BT (2015) Transcriptional coordination and abscisic acid mediated regulation of sucrose transport and sucrose-to-starch metabolism related genes during grain filling in wheat (Triticum aestivum L.). Plant Sci 240:143–160. https://doi.org/10.1016/j.plantsci.2015.09.010
Nam J-W, Kappock TJ (2007) Cloning and transcriptional analysis of Crepis alpina fatty acid desaturases affecting the biosynthesis of crepenynic acid. J Exp Bot 58:1421–1432. https://doi.org/10.1093/jxb/erm005
Napier JA (2007) The production of unusual fatty acids in transgenic plants. Annu Rev Plant Biol 58:295–319. https://doi.org/10.1146/annurev.arplant.58.032806.103811
Napier JA, Graham IA (2010) Tailoring plant lipid: designer oilseeds come of age. Curr Opin Plant Biol 13:330–337. https://doi.org/10.1016/j.pbi.2010.01.008
Nguyen FT, Mishra G, Whittle E, Pidkowich MS, Bevan SA, Merlo AO, Walsh TA, Shanklin J (2010) Metabolic engineering of seeds can achieve levels of ω-7 fatty acids comparable with the highest levels found in natural plant sources. Plant Physiol 154:1897–1904. https://doi.org/10.1104/pp.110.165340
Nguyen HT, Park H, Koster KL, Cahoon RE, Nguyen HTM, Shanklin J, Clemente TE, Cahoon EB (2015) Redirection of metabolic flux for higher levels of omega-7 monounsaturated fatty acid accumulation in camelina seeds. Plant Biotechnol J 13:38–50. https://doi.org/10.1111/pbi.12233
O’Neill CM, Baker D, Bennett G, Clarke J, Bancroft I (2011) Two high linolenic mutants of Arabidopsis thaliana contain megabase-scale genome duplications encompassing the FAD3 locus. Plant J 68:912–918. https://doi.org/10.1111/j.1365-313x.2011.04742.x
Okada S, Zhou X-R, Damcevski K, Gibb N, Wood C, Hamberg M, Haritos VS (2013) Diversity of Δ12 fatty acid desaturases in Santalaceae and their role in production of seed oil acetylenic fatty acids. J Biol Chem 45:32405–32413. https://doi.org/10.1074/jbc.m113.511931
Olsen O-A (2004) Nuclear endosperm development in cereals and Arabidopsis thaliana. Plant Cell 16:S214–S227. https://doi.org/10.1105/tpc.017111
Pan X, Siloto RMP, Wickramarathna AD, Mietjiewska E, Weselake RJ (2013) Identification of a pair of phospholipid:diacylglycerol acyltransferases from developing flax (Linum usitatissimum L.) seed catalyzing the selective production of trilinolenin. J Biol Chem 33:24173–24188. https://doi.org/10.1074/jbc.m113.475699
Qi X, Li S, Zhu Y, Zhao G, Zhu D, Yu J (2017) ZmDof3, a maize endosperm-specific Dof protein gene, regulates starch accumulation and aleurone development in maize endosperm. Plant Mol Biol 93:7–20. https://doi.org/10.1007/s11103-016-0543-y
Qiu X, Reed DW, Hong H, Mac Kenzie SL, Covello PS (2001) Identification and analysis of a gene from Calendula officinalis encoding a fatty acid conjugase. Plant Physiol 125:847–855
Qu J, Ye J, Geng Y-F, Sun Y-W, Gao S-Q, Zhang B-P, Chen W, Chua N-H (2012) Dissecting functions of KATANIN and WRINKLED1 in cotton fiber development by virus-induced gene silencing. Plant Physiol 160:738–748. https://doi.org/10.1104/pp.112.198564
Rao S, Abdel-Reheem M, Bhella R, McCracken C, Hildebrand D (2008) Characteristics of high alpha-linolenic acid accumulation in seed oils. Lipids 43:749–755. https://doi.org/10.1007/s11745-008-3207-0
Rawat R, Yu X-H, Sweet M, Shanklin J (2012) Conjugated fatty acids: residues 111 and 115 influence product partitioning of Momordica charantia conjugase. J Biol Chem 287:16230–16237. https://doi.org/10.1074/jbc.m111.325316
Rezzonico E, Moire L, Delessert S, Poirier Y (2004) Level of accumulation of epoxy fatty acid in Arabidopsis thaliana expressing a linoleic acid Δ12-epoxygenase is influenced by the availability of the substrate linoleic acid. Theor Appl Genet 109:1077–1082. https://doi.org/10.1007/s00122-004-1721-x
Roesler K, Shintani D, Savage L, Boddupalli S, Ohlrogge J (1997) Targeting of the Arabidopsis homomeric acetyl-coenzyme A carboxylase to plastids of rapeseeds. Plant Physiol 113:75–81
Román Á, Hernández ML, Soria-Gracia Á, López-Gomollón S, Lagunas B, Picorel R, Martínez-Rivas JM, Alfonso M (2015) Non-redundant contribution of the plastidial FAD8 ω-3 desaturase to glycerolipid unsaturation at different temperatures in Arabidopsis. Mol Plant 8:1599–1611. https://doi.org/10.1016/j.molp.2015.06.004
Roscoe TJ, Lessire R, Puyaubert J, Renard M, Delseny M (2001) Mutations in the fatty acid elongation 1 gene are associated with loss of β-ketoacyl-CoA synthase activity in low erucic acid rapeseed. FEBS Lett 492:107–111
Ruiz-Lopez N, Haslam RP, Venegas-Caleron M, Larson TR, Graham IA, Napier JA, Sayanova O (2009) The synthesis and accumulation of stearidonic acid in transgenic plants: a novel source of ‘heart-healthy’ omega-3 fatty acids. Plant Biotechnol J 7:704–716. https://doi.org/10.1111/j.1467-7652.2009.00436.x
Ruiz-Lopez N, Haslam RP, Usher S, Napier JA, Sayanova O (2015) An alternative pathway for the effective production of the omega-3 long-chain polyunsaturates EPA and ETA in transgenic oilseeds. Plant Biotechnol J 13:1264–1275. https://doi.org/10.1111/pbi.12328
Ruiz-Lopez N, Broughton R, Usher S, Salas JJ, Haslam RP, Napier JA, Beaudoin F (2017) Tailoring the composition of novel wax esters in the seeds of transgenic Camelin sativa through systematic metabolic engineering. Plant Biotechnol J 15:837–849. https://doi.org/10.1111/pbi.12679
Salas JJ, Mortinez-Force E, Garces R (2005) Very long chain fatty acid synthesis in sunflower kernels. J Agric Food Chem 53:2710–2716. https://doi.org/10.1021/jf047939e
Salie MJ, Thelen JJ (2016) Regulation and structure of the heteromeric acetyl-CoA carboxylase. Biochim Biophys Acta 1861:1207–1213. https://doi.org/10.1016/j.bbalip.2016.04.004
Sayanova O, Haslam R, Venegas Caleron M, Napier JA (2007) Cloning and characterization of unusual fatty acid desaturases from Anemone leveillei: identification of an acyl-Coenzyme A C20 Δ5-desaturase responsible for the synthesis of sciadonic acid. Plant Physiol 144:455–467. https://doi.org/10.1104/pp.107.098202
Sbihi H, Nehdi IA, Al-Resayes SI (2014) Characterization of white mahlab (Prunus mahaleb L.) seed oil: a rich source of α-eleostearic acid. J Food Sci 79:C795–801. https://doi.org/10.1111/1750-3841.12467
Schultz DJ, Cahoon EB, Shanklin J, Craig R, Cox-Foster DL, Mumma RO, Medford JI (1996) Expression of a Δ9 14:0-acyl carrier protein fatty acid desaturase gene is necessary for the production of ω5 anacardic acids found in pest-resistant geranium (Pelargonium xhortorum). Proc Natl Acad Sci USA 93:8771–8775
Schwender J, Goffman F, Ohlrogge JB, Shachar-Hill Y (2004) Rubisco without the Calvin cycle improves the carbon efficiency of developing green seeds. Nature 432:779–782
Schwender J, Shachar-Hill Y, Ohlrogge JB (2006) Mitochondrial metabolism in developing embryos of Brassica napus. J Biol Chem 281:34040–34047. https://doi.org/10.1074/jbc.m606266200
Shabbir MA, Khan MR, Saeed M, Pasha I, Khalil AA, Siraj N (2017) Punicic acid: a striking health substance to combat metabolic syndromes in humans. Lipids Health Dis 16:99. https://doi.org/10.1186/s12944-017-0489-3
Shen B, Allen WB, Zheng P, Li C, Glassman K, Ranch J, Nubel D, Tarczynski C (2010) Expression of ZmLEC1 and ZmWRI1 increases seed oil production in maize. Plant Physiol 153:980–987. https://doi.org/10.1104/pp.110.157537
Shimada TL, Hayashi M, Hara-Nishimura I (2017) Membrane dynamics and multiple functions of oil bodies in seeds and leaves. Plant Physiol. https://doi.org/10.1104/pp.17.01522
Shockey J, Regmi A, Cotton K, Adhikari N, Browse J, Bates PD (2016) Identification of Arabidopsis GPAT9 (At5g60620) as an essential gene involved in triacylglycerol biosynthesis. Plant Physiol 170:163–179. https://doi.org/10.1104/pp.15.01563
Smith MA, Moon H, Chowrira G, Kunst L (2003) Heterologous expression of a fatty acid hydroxylase gene in developing seeds of Arabidopsis thaliana. Planta 217:507–516. https://doi.org/10.1007/s00425-003-1015-6
Smith MA, Zhang H, Forseille L, Purves RW (2013) Characterization of novel triacylglycerol estolides from the seed oil of Mallotus philippensis and Trewia nudiflora. Lipids 48:75–85. https://doi.org/10.1007/s11745-012-3721-y
Snapp AR, Kang J, Qi X, Lu C (2014) A fatty acid enzyme from Physaria fendleri increases hydroxyl fatty acid accumulation in transgenic oilseeds of Camelina sativa. Planta 240:599–610. https://doi.org/10.1007/s00425-014-2122-2
Snyder CL, Yurchenko OP, Siloto RMP, Chen X, liu Q, Mietkiewska E, Weselake RJ (2009) Acyltransferase action in the modification of seed oil biosynthesis. New Biotechnol 26:11–16. https://doi.org/10.1016/j.nbt.2009.05.005
Song Z, Mietkiewska E, Weselake RJ (2017) The linin promoter is highly effective in enhancing punicic acid production in Arabidopsis. Plant Cell Rep 36:447–457. https://doi.org/10.1007/s00299-016-2094-8
Sreenivasulu N, Wobus U (2013) Seed-development programs: a systems biology-based comparison between dicots and monocots. Annu Rev Plant Biol 64:189–217. https://doi.org/10.1146/annurev-arplant-050312-120215
Surette ME (2013) Dietary omega-3 PUFA and health: stearidonic acid-containing seed oils as effective and sustainable alternatives to traditional marine oils. Mol Nutr Food Res 57:748–759. https://doi.org/10.1002/mnfr.201200706
To A, Joubès J, Barthole G, Lécureuil A, Scagnelli A, Jasinski S, Lepiniec L, Baud S (2012) WRINKLED transcription factors orchestrate tissue-specific regulation of fatty acid biosynthesis in Arabidopsis. Plant Cell 24:5007–5023. https://doi.org/10.1105/tpc.112.106120
Tran TNT, Shelton J, Brown S, Durrett TP (2017) Membrane topology and identification of key residues of EaDAcT, a plant MBOAT with unusual substrate specificity. Plant J 92:82–94. https://doi.org/10.1111/j.1365-2443.2008.01212.x
Troncoso-Ponce MA, Kilaru A, Cao X, Durrett TP, Fan J, Jensen JK, Thrower NA, Pauly M, Wilkerson C, Ohlrogge JB (2011) Comparative deep transcriptional profiling of four developing oilseeds. Plant J 68:1014–1027. https://doi.org/10.1111/j.1365-313x.2011.04751.x
Troncoso-Ponce MA, Barthole G, Tremblais G, To A, Miquel M, Lepiniec L, Baud S (2016a) Transcriptional activation of two delta-9 palmitoyl-ACP desaturase genes by MYB115 and MYB118 is critical for biosynthesis of omega-7 monounsaturated fatty acids in the endosperm of Arabidopsis seeds. Plant Cell 28:2666–2682. https://doi.org/10.1105/tpc.16.00612
Troncoso-Ponce MA, Nikovics K, Marchive C, Lepiniec L, Baud S (2016b) New insights on the organization and regulation of the fatty acid biosynthetic network in the model higher plant Arabidopsis thaliana. Biochimie 120:3–8. https://doi.org/10.1016/j.biochi.2015.05.013
Usher S, Han L, Haslam RP, Michaelson LV, Sturtevant D, Aziz M, Chapman KD, Sayanova O, Napier JA (2017) Tailoring seed oil composition in the real world: optimizing omega-3 long chain polyunsaturated fatty acid accumulation in transgenic Camelina sativa. Sci Rep 7:6570. https://doi.org/10.1038/s41598-017-06838-0
van Erp H, Bates PD, Burgal J, Shockey J, Browse J (2011) Castor phospholipid:diacylglycerol acyltransferase facilitates efficient metabolism of hydroxy fatty acids in transgenic Arabidopsis. Plant Physiol 155:683–693. https://doi.org/10.1104/pp.110.167239
van Erp H, Kelly AA, Menard G, Eastmond PJ (2014) Multigene engineering of triacylglycerol metabolism boosts seed oil content in Arabidopsis. Plant Physiol 165:30–36. https://doi.org/10.1104/pp.114.236430
Vanhercke T, Wood CC, Stymne S, Singh SP, Green AG (2013) Metabolic engineering of plant oils and waxes for use as industrial feedstocks. Plant Biotechnol J 11:197–210. https://doi.org/10.1111/pbi.12023
Venglat P, Xiang D, Qiu S, Stone SL, Tibivhe C, Cram D, Alting-Mees M, Nowak J, Cloutier S, Deyholos M, Bekkaoui F, Sharpe A, Wang E, Rowland G, Selvaraj G, Datla R (2011) Gene expression analysis of flax seed development. BMC Plant Biol 11:74. https://doi.org/10.1186/1471-2229-11-74
Vicente-Carbajosa J, Carbonero P (2005) Seed maturation: developing an intrusive phase to accomplish a quiescent state. Int J Dev Biol 49:645–651. https://doi.org/10.1387/ijdb.052046jc
Vigeolas H, Waldeck P, Zant T, Geigenberger P (2007) Increasing seed oil content in oil-seed rape (Brassica napus L.) by over-expression of a yeast glycerol-3-phosphate dehydrogenase under the control of a seed-specific promoter. Plant Biotechnol J 5:431–441. https://doi.org/10.1111/j.1467-7652.2007.00252.x
Wang H, Guo J, Lambert KN, Lin Y (2007) Developmental control of Arabidopsis seed oil biosynthesis. Planta 226:773–783. https://doi.org/10.1007/s00425-007-0524-0
Wang L, Shen W, Kazachkov M, Chen G, Chen Q, Carlsson AS, Stymne S, Weselake R, Zou J (2012) Metabolic interactions between the Lands cycle and the Kennedy pathway of glycerolipid synthesis in Arabidopsis developing seeds. Plant Cell 24:4652–4669. https://doi.org/10.1105/tpc.112.104604
Wang JC, Xu H, Zhu Y, Liu QQ, Cai XL (2013) OsbZIP58, a basic leucine zipper transcription factor, regulates starch biosynthesis in rice endosperm. J Exp Bot 64:3453–3466. https://doi.org/10.1093/jxb/ert187
Wang K, Froehlich JE, Zienkiewicz A, Lynn Hersh H, Benning C (2017) A plastid phosphatidylglycerol lipase contributes to the export of acyl groups from plastids for seed oil biosynthesis. Plant Cell 29:1678–1696. https://doi.org/10.1105/tpc.17.00397
Weselake RJ, Taylor DC, Rahman MH, Shah S, Laroche A, McVetty PBE, Harwood JL (2009) Increasing the flow of carbon into seed oil. Biotechnol Adv 27:866–878. https://doi.org/10.1016/j.biotechadv.2009.07.001
White JA, Todd J, Newman T, Focks N, Girke T, de Ilarduya Martinez, Jaworski JG, Ohlrogge JB, Benning C (2000) A new set of Arabidopsis expressed sequence tags from developing seeds. The metabolic pathway from carbohydrate to seed oil. Plant Physiol 124:1582–1594
Wiberg E, Edwards P, Byrne J, Stymne S, Dehesh K (2000) The distribution of caprylate, caprate and laurate in lipids from developing and mature seeds of transgenic Brassica napus L. Planta 212:33–40. https://doi.org/10.1007/s004250000361
Wolff RL, Christie WW (2002) Structures, practical sources (gymnosperm seeds), gas-liquid chromatography data (equivalent chain lengths) and mass spectrometric characteristics of all cis Δ5-olefinic acids. Eur J Lipid Sci Technol 104:234–244
Wollf RL, Lavialle O, Pedrono F, Pasquier E, Deluc LG, Marpeau AM, Angers P, Aitzetmuller K (2001) Fatty acid composition of Pinaceae as taxonomic markers. Lipids 36:439–451
Wong SC, Shirley NJ, Little A, Khoo KHP, Schwerdt J, Fincher GB, Burton RA, Mather DE (2015) Differential expression of the HvCslF6 gene late in grain development may explain quantitative differences in (1,3;1,4)-β-glucan concentration in barley. Mol Breeding 35:20. https://doi.org/10.1007/s11032-015-0208-6
Wu Y, Li R, Hildebrand DF (2012) Biosynthesis and metabolic engineering of palmitoleate production, an important contributor to human health and sustainable industry. Prog Lipid Res 41:340–349. https://doi.org/10.1016/j.plipres.2012.05.001
Xiao Y, Thatcher S, Wang M, Wang T, Beatty M, Zastrow-Hayes G, Li L, Li J, Li B, Yang X (2016) Transcriptome analysis of near-isogenic lines provides molecular insights into starch biosynthesis in maize kernel. J Int Plant Biol 58:713–723. https://doi.org/10.1111/jipb.12455
Xiao Q, Wang Y, Du J, Li H, Wei B, Wang Y, Li Y, Yu G, Liu H, Zhang J, Liu Y, Hu Y, Huang Y (2017) ZmMYB14 is an important transcription factor involved in the regulation of the activity of the ZmBT1 promoter in starch biosynthesis. FEBS J 284:3079–3099. https://doi.org/10.1111/febs.14179
Xu C, Shankli J (2016) Triacylglycerol metabolism, function, and accumulation in plant vegetative tissues. Annu Rev Plant Biol 67:179–206. https://doi.org/10.1146/annurev-arplant-043015-111641
Xu J, Carlsson AS, Francis T, Zhang M, Hoffman T, Giblin ME, Taylor DC (2012) Triacylglycerol synthesis by PDAT1 in the absence of DGAT1 activity is dependent on re-acylation of LPC by LPCAT2. BMC Plant Biol 12:4. https://doi.org/10.1186/1471-2229-12-4
Yamamoto K, Kinoshita A, Shibahara A (2008) Ricinoleic acid in common vegetable oils and oil seeds. Lipids 43:457–460. https://doi.org/10.1007/s11745-008-3154-9
Yan G, Li D, Cai M, Gao G, Chen B, Xu K, Li J, Li F, Wang N, Qiao J, Li H, Zhang T, Wu X (2015) Characterization of FAE1 in the zero erucic germplasm of Brassica rapa L. Breed Sci 65:257–264. https://doi.org/10.1270/jsbbs.65.257
Yu K, Li R, Hatanaka T, Hildebrand D (2008) Cloning and functional analysis of two type 1 diacylglycerol acyltransferases from Vernonia galamensis. Phytochemistry 69:1119–1127. https://doi.org/10.1016/j.phytochem.2007.11.015
Yu X-H, Rawat R, Shanklin J (2011) Characterization and analysis of the cotton cyclopropane fatty acid synthase family and their contribution to cyclopropane fatty acid synthesis. BMC Plant Biol 11:97. https://doi.org/10.1186/1471-2229-11-97
Yu X-H, Cahoon RE, Horn PJ, Shi H, Prakash RR, Cai Y, Hearney M, Chapman KD, Cahoon EB, Schwender J, Shanklin J (2017) Identification of bottlenecks in the accumulation of cyclic fatty acids in camelina seed oil. Plant Biotechnol J. https://doi.org/10.1111/pbi.12839
Zeeman SC, Kossman J, Smith AM (2010) Starch: its metabolism, evolution, and biotechnological modification in plants. Annu Rev Plant Biol 61:209–234. https://doi.org/10.1146/annurev-arplant-042809-112301
Zeng F, Roslinsky V, Cheng B (2017) Mutations in the promoter, intron and CDS of two FAD2 generate multiple alleles modulating linoleic acid level in yellow mustard. Sci Rep 7:8284. https://doi.org/10.1038/s41598-017-08317-y
Zhai Z, Liu H, Shanklin J (2017) Phosphorylation of WRINKLED1 by KIN10 results in its proteasomal degradation, providing a link between energy homeostasis and lipid biosynthesis. Plant Cell 29:871–889. https://doi.org/10.1105/tpc.17.00019
Zhang M, Fan J, Taylor DC, Ohlrogge JB (2009) DGAT1 and PDAT1 acyltransferases have overlapping functions in Arabidopsis triacylglycerol biosynthesis and are essential for normal pollen and seed development. Plant Cell 21:3885–3901. https://doi.org/10.1105/tpc.109.071795
Zhang H, Olson DJH, Van D, Purves RW, Smith MA (2012) Rapid identification of triacylglycerol-estolides in plant and fungal oils. Ind Crops Prod 37:186–194. https://doi.org/10.1016/j.indcrop.2011.10.026
Zhang J, Chen J, Yi Q, Hu Y, Liu H, Liu Y, Huang Y (2014a) Novel role of ZmaNAC36 in co-expression of starch synthetic genes in maize endosperm. Plant Mol Biol 84:359–369. https://doi.org/10.1007/s11103-013-0153-x
Zhang H, Smith MA, Purves RW (2014b) Optimization of triacylglycerol-estolide analysis by matrix-assisted laser desorption/ionization-mass spectrometry. J Am Oil Chem Soc 91:905–915. https://doi.org/10.1007/s11746-014-2437-y
Zhang L, Ren Y, Lu B, Yang C, Feng C, Liu Z, Chen J, Ma W, Wang Y, Yu X, Wang Y, Zhang Y, Wang Y, Liu S, Wu F, Zhang X, Guo X, Bao Y, Jiang L, Wan J (2016) FLOURY ENDOSPERM7 encodes a regulator of starch synthesis and amyloplast development essential for peripheral endosperm development in rice. J Exp Bot 67:633–647. https://doi.org/10.1093/jxb/erv469
Zhao L, Katavic V, Li F, Haughn GW, Kunst L (2010) Insertional mutant analysis reveals that the long-chain acyl-CoA synthetase 1 (LACS1), but not LACS8, functionally overlaps with LACS9 in Arabidopsis oil biosynthesis. Plant J 64:1048–1058. https://doi.org/10.1111/j.1365-313X.2010.04396.x
Zheng Y, Wang Z (2015) The cereal starch endosperm development and its relationship with other endosperm tissues and embryo. Protoplasma 252:33–40. https://doi.org/10.1007/s00709-014)0687-z
Zhu L-H, Krens F, Smith MA, Li X, Qi W, van Loo EN, Iven T, Feussner I, Nazarenus TJ, Huai D, Taylor DC, Zhou X-R, Green AG, Shockey J, Klasson KT, Mullen RT, Huang B, Dyer JM, Cahoon EB (2016) Dedicated industrial oilseed crops as metabolic engineering platforms for sustainable industrial feedstock oroduction. Sci Rep 6:22181. https://doi.org/10.1038/srep22181
Acknowledgements
We are grateful to A. To, B. Dubreucq, M. Miquel, and L. Lepiniec for helpful discussions about oil accumulation in seeds. We apologize to those researchers whose work we were not able to cite in this review. The Institut Jean-Pierre Bourgin benefits from the support of the Labex Saclay Plant Sciences-SPS (ANR-10-LABX-0040-SPS).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by L. Lepiniec, H. North, G. Ingram.
A contribution to the special issue ‘Seed Biology’.
Rights and permissions
About this article
Cite this article
Baud, S. Seeds as oil factories. Plant Reprod 31, 213–235 (2018). https://doi.org/10.1007/s00497-018-0325-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-018-0325-6