Abstract
Alkaline phosphomonoesterase (ALP) mainly originates from soil microbial secretion and plays a crucial role in the turnover of soil phosphorus (P). To examine the response of ALP-encoding microbial communities (analysed for the biomarker of the ALP gene, phoD) of soils and derivative soil fractions to different fertilisation regimes, soil samples were collected from a long-term experimental field (over 35 years). The different organic P (Po) pools of soil fractions and the ALP activity of soil were also determined. Compared with chemical-only fertilised soils, the ALP activity was 232–815% higher in organic-amended soils, and the highest enzyme activity was observed in the organic-only fertilised treatment. The abundance of the phoD gene harbouring in soil fractions, determined by quantitative PCR (qPCR), was affected by different fertilisations. The highest abundance of the phoD gene was generally detected in the 2–63-μm-sized fraction (silt), but most phoD-encoding microbial species were associated to the 0.1–2-μm-sized fraction (clay) in the chemical-only fertilised soil. The contents of labile Po (LPo), moderately labile Po (MLPo) and fulvic acid-associated Po (FAPo) were significantly correlated with the phoD gene abundance, whereas only LPo content was significantly correlated with the ALP activity. The dominant phoD-encoding phylas were Actinobacteria and Proteobacteria, according to a high-throughput sequencing. Bradyrhizobium, a N2-fixer identified as a phoD-encoding genus, showed the highest abundance in fertilised soils. The abundance of Bradyrhizobium, Streptomyces, Modestobacter, Lysobacter, Frankia and Burkholderia increased with the organic-only amendment and was significantly correlated with the ALP activity. According to structure equation models (SEM), pH and LPo content significantly and directly affected the ALP activity; the soil organic C (Corg) content was related to composition and abundances of phoD-harbouring microbial communities; since both microbial properties were correlated to the ALP activity, the Corg content was indirectly related to the ALP activity. In conclusion, soil management practices can be used to optimise the contents of soil available P and the organic P with regulation of soil ALP activity and the community composition of corresponding microbes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phosphorus (P) deficiencies can limit plant growth and crop yield (Welsh et al. 2009). Most of the P applied to agro-ecosystems is retained in the soil as mineral-associated P, which is often not available to plants (Waldrip et al. 2011), whereas when mobilised, it can reach waterways with potential risks of P eutrophication (Withers and Haygarth 2007). To reduce these problems and to compensate for the insufficient resources of rock phosphate used for fertiliser production (Cordell et al. 2009), it is important to carefully consider the agronomical use of the “legacy phosphorus” or accumulated P, which includes both inorganic P (Pi) and organic P (Po).
The Po content of soil can range from 30 to 50% of the total soil P (Dalal 1978) and can be a P source for plants and microbes if it is mineralised to Pi by phosphatases (Tarafdar and Claassen 1988; Richardson 2001), which include phosphomonoesterases (hydrolysing phosphoric monoesters), phosphodiesterases (hydrolysing phosphoric diester), phosphoric triester hydrolases, polyphosphate hydrolases etc. Both acid (ACP) and alkaline phosphomonoesterase (ALP; commonly termed phosphatases) activities have been studied in soils to evaluate the organic P mineralisation to inorganic P (Nannipieri et al. 2011, 2012) since they are considered to play a main role in this process (Tarafdar and Jungk 1987). The primary producers of acid phosphomonoesterases are plant roots and soil microbes, whereas alkaline phosphomonoesterases only originate from soil microbes and fauna (Tarafdar and Claassen 1988; Spohn and Kuzyakov 2013a). However, the origin of these enzymes in soil is poorly known and understanding it may give insights on environmental factors and agricultural management, for example the type and rate of fertilisation, affecting the mineralisation of organic P to inorganic P. The ALP-encoding genes are grouped into three distinct families (phoA, phoD and phoX) based on their sequence similarities and substrate specificities (Kathuria and Martiny 2011). About 32% of soil prokaryotes contained at least one of these ALP-encoding genes (Zimmerman et al. 2013), which are classified as COG1785, COG3540 and COG3211, respectively, according to the Cluster of Orthologous Groups (COG) categorisation (Kageyama et al. 2011). Of these enzyme-encoding genes, the phoD gene is considered the key ALP gene in soil (Tan et al. 2013).
Long-term fertilisation influences soil nutrient cycling and nutrient availability by altering the physical, chemical and biological properties of soil, including effects on activity, biomass and composition of microbial communities responsible for soil functions. Agricultural management can affect both composition and abundances of phoD microbial community (Fraser et al. 2015a, b), but it is not known how the long-term fertilisation affects the distribution of the microbial communities across the soil particle size fractions (PSFs) and the relation of these communities with different soil Po fractions. This knowledge may get insight on the mechanisms responsible for the mineralisation of the different organic P fractions to plant available P.
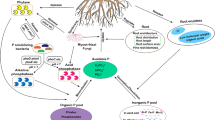
In this study, we hypothesised that the different long-term fertilisation regimes would change the different soil organic P fractions, the abundance and composition of ALP-encoding microbial communities and ALP activity. Moreover, as changes in the distribution of organic matter in soil particle size fractions (PSFs) are well established under different fertilisation regimes (Nicolás et al. 2012; Yu et al. 2012; Ling et al. 2014), we also hypothesised that the abundance of phoD-harbouring microbes among the different PSFs may be affected by different fertilisation practises. The abundance and composition of phoD-harbouring microbial communities were determined by quantitative PCR (qPCR) and Illumina MiSeq sequencing. The phoD was chosen as the biomarker of ALP gene and multivariate statistics, e.g. redundancy analysis, structure equation models (SEM) etc., were employed to analyse the obtained data. Soil samples were collected from a long-term (started in 1981) field experiment with different fertilisation regimes.
Materials and methods
Site description and sample collection
The long-term field experiment cropped to corn–wheat rotation was established in 1981 in Suixi County, Anhui Province, China (116° 45′ E, 33° 37′ N). The mean annual temperature of this region is approximately 15.0 °C, and the mean annual precipitation is approximately 870 mm. The soils of this Province are vertisols developed in fluvial and lacustrine deposits and are also classified as a lime concretion black soil, according to the Chinese soil classification system (Zhang et al. 2014b). At the beginning of the experiment, the main soil physicochemical properties were pH 7.6; SOM, 10.22 g kg−1; total N, 0.78 g kg−1; total P, 0.47 g kg−1; and available P, 2.5 mg kg−1. This experiment included the following five treatments with four replicated plots (30 m2 per plot) for each treatment: (1) CK, no fertiliser added; (2) NPK, only mineral NPK (N, 525 kg ha−1 year−1; P2O5, 210 kg ha−1 year−1; and K2O, 210 kg ha−1 year−1); (3) MNPK, mineral NPK (N, 262.5 kg ha−1 year−1; P2O5, 105 kg ha−1 year−1; and K2O, 105 kg ha−1 year−1) plus 3750 kg composted bean cake ha−1 year−1: (4) HMNPK, mineral NPK (N, 420 kg ha−1 year−1; P2O5, 168 kg ha−1 year−1; and K2O, 168 kg ha−1 year−1) plus 6000 kg composted bean cake ha−1 year−1; and (5) M, only composted bean cake (7500 kg ha−1 year−1). Only three replicates of each treatment were considered in this study. The N application rates of the M, NPK and MNPK treatments were equal to 525 kg N ha−1 year−1 by summing organic N and inorganic N inputs, whereas 840 kg N ha−1 year−1 were applied in the HMNPK treatment with an organic N to inorganic N ratio of 1:1. The mineral NPK fertilisers were urea, superphosphate and potassium chloride, whereas composted bean cake contained 30–40% organic matter, 6–7% total N, 1–3% P2O5, 2–3% K2O and 10–15% water. Crop straw residues were removed after harvest each year.
Ten soil cores (5 cm diameter) were collected from each plot at a depth of 0–20 cm in March 2016, carefully composited as a single sample, gently broken apart along the natural break points and then moist sieved (<2 mm) with removal of visible plant matter and organic debris. Each sample was then divided into two parts: one part was stored at 4 °C for biochemical and chemical analyses, and the other part was stored at −20 °C for DNA extraction.
Soil chemical and biochemical analyses
Soil properties were analysed as previously described (Chu and Grogan 2010; Ling et al. 2014). Soil pH was measured with a soil-water (1:2.5, w/v) slurry using a compound electrode (PE-10; Sartorious, Germany). Soil organic C (Corg) and total N (TN) were measured with an elemental analyser (Vario MAX; Elementar, Germany). Soil NO3 −-N and exchangeable NH4 +-N were extracted with 0.01 M CaCl2 and then detected on a continuous flow analytical system (Santt System; Skalar, Holland). Soil dissolved organic C (DOC) was extracted with 0.5 M K2SO4 and then determined with a TOC analyser (ELEMENTER, Germany). Soil Ca2+ was extracted with 1 M Mg(NO3)2 and determined by an inductively coupled plasma mass spectrometry (ICP-MS; Perkin Elmer Nexion 300×, USA). Available potassium (AK) was determined in 1 M ammonium acetate extracts with flame photometry (FP640: INASA). Soil total P was digested by HF-HClO4 and ultimately determined via molybdenum-blue colorimetry (Xun et al. 2016). Soil available P (AP) was extracted with 0.5 M NaHCO3 and determined using the ammonium molybdate ascorbic method (Zhong et al. 2010).
Fractionation procedure for soil particles
The size fractionation was carried out according to Stemmer et al. (1998). Briefly, fresh soils (140 g equivalent dry weight for each sample) were suspended in 400 ml of distilled water and the soil-water suspension was dispersed by low-energy sonication (output energy of 0.2 kJ/g). Subsequently, the suspensions were fractionated by a combination of wet sieving and repeated centrifugation (3900×g) to avoid disruption of micro-aggregates. The following four fractions were obtained for each sample: coarse sand fraction (200–2000 μm), fine-sand fraction (63–200 μm), silt fraction (2–63 μm) and clay fraction (0.1–2 μm). The fractions were freeze-dried, and then DNA was extracted from each fraction of the different fertilised soils (Ling et al. 2014). The average recovery of all fractions from the soil ranged from 92 to 95%.
Fractionation procedure for soil organic P
Soil organic P (Po) was fractionated according to the sequential extraction procedure by Bowman and Bowman and Cole (1978) and modified by Slazak et al. (2010). These were the obtained pools: (1) labile P pool (LPo), extracted from soil with 0.5 M NaHCO3 solution (pH = 8.5); (2) moderately labile P pool (MLPo), extracted from the sediment of the first extraction with 1.0 M HCl; (3) more resistant Po pool (FAPo), extracted from the sediment of the second extraction with 0.5 M NaOH; and (4) highly resistant Po pool (HAPo), the P content of the residue remaining after all extractions. According to Slazak et al. (2010) and Tiecher et al. (2012), FAPo was primarily associated with fulvic acid and the HAPo was primarily associated with humic acids. The sum of the four fractions was the total Po (TPo).
Potential ALP activity assay
ALP activity was estimated by measuring the release of p-nitrophenol (PNP) from p-nitrophenyl phosphate (PNPP) as described by Tabatabai and Bremner (1969). One gramme of fresh soil was slightly shaken with 0.5 ml toluene for 15 min, and then incubated in modified universal buffer (pH 11.0) containing the p-nitrophenol phosphate (p-NPP; Sigma-Aldrich, USA) for 1 h at 37 °C. Reactions were stopped with 0.5 M NaOH, and solutions were filtered with Whatman 42 paper. The formation of p-nitrophenol was determined at 420 nm (Sakurai et al. 2008; Fraser et al. 2015a, b). The ALP activity was expressed as microgram per h per g of soil (Cui et al. 2015).
DNA extraction
Total genomic soil DNA was extracted from 0.25 g of soil (dry weight equivalent) using a Power Soil® DNA Isolation Kit (MoBio, Carlsbad, CA, USA), according to the manufacturer’s protocol. The DNA extracts were purified with a Wizard DNA Clean-Up System (Axygen Bio, USA), as recommended by the manufacturer. The DNA was stored at −80 °C until analysis.
Quantitative PCR analysis of phoD genes
The phoD gene was amplified with primers ALPS-F730 (5′-CAGTGGGACGACCACGAGGT-3′) and ALPS-1101 (5′GAGGCCGATCGGC-ATGTCG-3′) (Sakurai et al. 2008; Acuña et al. 2016; Lagos et al. 2016), and the size of amplicon is 371 bp. Each sample involved three technical replicates for the phoD gene amplification, which was carried out in an ABI 7500 Cycle Real-time PCR System (Applied Biosystems, Germany); 25 μl of solution contained 12.5 μl of SYBR® Premix Ex Taq (2×; Tli RNaseH Plus), 0.5 μl of ROX Reference Dye II (50×; TAKARA, BIO, INC, Japan), 0.5 μl of each primer and 1 μl of template and ddH2O added to bring to a volume. Cycling conditions were as follows: 95 °C for 30 s, followed by 40 cycles at 95 °C for 5 s and at 60 °C for 34 s. The plasmid for the standard curve was constructed according to Fraser et al. (2015a, b). The standard curve was prepared using serial 10-fold dilutions, and the number of gene copies was calculated by measuring the concentration of the plasmid and the number of base pairs. Amplification efficiencies ranged from 96 to 98% with R 2 values of 0.9992.
Illumina MiSeq high-throughput sequencing and data analysis
The diversity and composition of ALP-harbouring bacterial communities were assessed using high-throughput sequencing technique. Universal primers ALPS-F730 and ALPS-1101 were selected for PCR amplification of the phoD gene. Despite few biases, this primer can give accurate phylogenetic and taxonomic information (Tan et al. 2013; Ragot et al. 2015). Personal Biotechnology Co., Ltd. (Shanghai, China) conducted the sequencing using an Illumina MiSeq platform. Overlapping paired-end reads were assembled using PEAR software (Zhang et al. 2013). Primers and poor-quality sequences were removed using cut adapt software (Martin 2011). The sequences were converted to amino acid sequences using the Framebot tool in the RDP function gene pipeline (http://fungene.cme.msu.edu/FunGenePipeline/); sequences that did not correspond to the phoD gene or had a termination codon in the middle were removed. In total, 641,489 high-quality sequences were obtained from 15 samples, and the number of sequences per sample ranged from 25,295 to 70,381 (mean = 42,765). Each sample was rarefied to the identical number of reads (10,000) for downstream analyses. The UPARSE pipeline was used to cluster sequences into centroid operational taxonomic units (OTUs) (Edgar 2013). Sequences with 80% similarity were assigned to OTUs, and 941 OTUs were obtained; this resulted in reads that were mapped back to OTUs using USEARCH software. A representative sequence was selected from each OTU, and the RDP function gene database was used to assign taxonomic data to each representative sequence. MOTHUR software (Schloss et al. 2009) was used to calculate the percentages of shared OTUs between samples and to analyse α- and β-diversity; the similarity tree was visualised using the iTOL portal (Letunic and Bork 2006). All sequences were deposited in the NCBI Sequence Read Archive (SRA) database (accession number SRX2010902).
Statistical analyses
Statistical analyses were performed using the IMB SPSS statistical software package version 20 (IBM Corporation, New York, USA). Data were analysed with one-way ANOVA, and Fisher’s least significant difference (p < 0.05) was used to compare the mean for each variable. Shannon diversity and Chao richness were calculated considering OTU tables, and redundancy analysis (RDA) was performed by the R statistical software package (version 2.15.0) using vegan packages to explore the relationships between the composition of ALP-encoding microbial community and soil properties. The phylogenetic tree was generated using the neighbour-joining method based on representative ALP partial nucleotide sequence matches using MEGA7.0 software. The abundance of the identified genus (A) was calculated as follows:
where QA is the total abundance of the phoD gene measured by qPCR and RA is the relative abundance of each identified genus.
SEM were constructed to test for indirect or direct relationships among abundance, diversity and composition of the phoD-harbouring communities, potential explanatory variables and ALP activities in a multivariate approach using AMOS software (IBM SPSS AMOS 20.0.0). SEM are better than other statistical analyses, e.g. multiple regressions, because directions can be assigned to several relationships yielding multiple explanatory as well as multiple response variables in one model (Grace 2006). The RDA first axis scores of samples were used as indicators of the community composition of phoD-harbouring microbes. The fit of a model to the data was tested using the maximum likelihood (χ 2) goodness-of-fit test, p value and the root mean square error of approximation (RMSEA).
Results
Effects of different fertilisation treatments on soil properties
The soil physicochemical properties were significantly affected by fertilisation regimes. Soil pH values increased in M and HMNPK treatments but deceased in the NPK treatment, compared with the CK and MNPK. Soil EC values increased in all treatments compared with the CK. The percentage of fine-sand fraction increased significantly in soils treated with organic fertiliser compared with the NPK and CK treatments, whereas soils only treated with chemical fertilisers contained the highest percentage of coarse sand. Little variation in the percentages of silt and clay fractions was found among treatments (Table 1). The concentrations of NO3 −-N, exchangeable NH4 +-N and available K increased with fertilisation. The highest concentration of exchangeable NH4 +-N (9.56 mg/kg) was in the NPK-treated soil whereas that of NO3 −-N was in the HMNPK-treated soil (29.61 mg/kg). Compared with other treatments, the concentrations of Corg, TN and DOC were relatively higher in HMNPK-treated soils (29.17 g/kg, 2.07 g/kg and 86.44 mg/kg, respectively), follow by M-treated soils (26.24 g/kg, 1.84 g/kg and 79.50 mg/kg, respectively). The contents of Ca2+ in the M and CK soils (5.38 and 5.75 g/kg, respectively) were much higher than those in the other treated soils (Table 1).
Effects of different fertilisation treatments on soil ALP activity
The highest ALP activity (54.57 μg PNP g−1 soil h−1) was in the M soil, followed by that of the CK soil (47.39 μg PNP g−1 soil h−1). The NPK soil had the lowest ALP activity (5.96 μg PNP g−1 soil h−1), which was almost 9.2-fold lower than that in the M soil. The ALP activity of the MNPK and HMNPK soils decreased by 11% and 27%, respectively, compared with that of the CK soil (Fig. 1).
Potential alkaline phosphatase (ALP) activity measured in soils amended with different long-term fertilisation treatments. Data points represent treatment means (n = 3), and different letters indicate significant differences (p < 0.05). CK no fertiliser application, M application of single organic fertiliser, NPK application of single chemical fertiliser, MNPK mixed application of organic and chemical fertilisers, HMNPK mixed application of organic and chemical fertilisers with higher N rate
Effects of different fertilisation treatments on phoD gene abundance of soil and relative fractions (PSFs)
The trend of phoD gene abundance was similar to that of soil ALP activity (Fig. 1), being the highest in the M soil (8.94 × 107 copies g−1 soil) and the lowest was in the NPK soil (19.44-fold lower than that in M) (Fig. 2a). The distribution of the phoD gene in soil fractions was also affected by the different long-term fertilisation regimes. The highest abundance of the phoD gene was detected in the soil silt fraction (1.12 × 107–4.77 × 107 copies g−1 soil) and the lowest in the soil coarse sand fraction (2.39 × 105–2.96 × 106 copies g−1 soil; Fig. 2b). In all soils, excluding the NPK soil, most of the phoD-harbouring microbes commonly colonised the silt fraction, accounting for 62–71% of the total abundance in bulk soil (Fig. 2c). In the NPK soil, the percentage of phoD abundance associated to the clay fraction (47%) was higher than that associated to the silt fraction (40%) (Fig. 2c).
Abundances of the bacterial phoD gene estimated by gene copy numbers, as quantified by qPCR. a Total phoD gene copy numbers in each treatment, b phoD gene copy numbers in different soil fractions of each treatment and c relative abundance of phoD genes summing copy numbers of the different soil fractions. Data points represent treatment means (n = 3), and different letters indicate significant differences (p < 0.05) between treatments: 0.1–2 μm, clay; 2–63 μm, silt; 63–200 μm, fine sand; and 200–2000 μm, coarse sand. CK no fertiliser application, M application of single organic fertiliser, NPK application of single chemical fertiliser, MNPK mixed application of organic and chemical fertilisers, HMNPK mixed application of organic and chemical fertilisers with higher N rate
Effects of different fertilisation treatments on soil P fractions
The content of AP was the highest in the chemical fertiliser-treated soils (NPK, MNPK and HMNPK) (Fig. 3a), and the total P (TP) content was higher in all fertilised soils than in the control (CK), with the highest content (0.74 g/kg) in the HMNPK soil. The contents of soil AP and TP in the organic-only fertilised soil were intermediate between the CK and chemically fertilised soils. Moreover, the percentages of inorganic P and organic P of soils depended on the different fertilisation regimes (Fig. 3b). The percentage of organic P contents were 81% and 73% in the CK and M soils, respectively, being higher than those (32–49%) of the NPK-, MNPK- and HMNPK-treated soils (Fig. 3b).
Contents of the different soil P fractions: a content of soil NaHCO3-extractable available P in original soil; b concentrations of organic P, inorganic P and total P of original soil; c content of labile organic P (LPo); d content of moderately labile organic P (MLPo); e content of fulvic acid-associated organic P (FAPo); and f content of humic acid-associated organic P (HAPo). CK no fertiliser application, M application of single organic fertiliser, NPK application of single chemical fertiliser, MNPK mixed application of organic and chemical fertilisers, HMNPK mixed application of organic and chemical fertilisers with higher N rate. Numbers on top of (b) represent the percentage of total P present as organic P. Data points represent treatment means (n = 3), and different letters indicate significant differences (p < 0.05)
Compared with the CK treatment, the contents of MLPo, FAPo and HAPo (59.88–81.54 mg/kg, 88.20–123.62 mg/kg and 45.62–59.91 mg/kg, respectively) significantly increased in organic amended treatments (M, MNPK and HMNPK). Notably, the content of LPo was not significantly different between CK (80.24 mg/kg) and M (83.19 mg/kg) treatments, but it was higher than those of the chemical-fertilised (NPK, MNPK and HMNPK) soils. The lowest content of MLPo was in the NPK treated soil, whereas the lowest contents of FAPo and HAPo were observed in the CK soil. Both MLPo and FAPo contents increased by the only-organic amendment, compared with the chemical-only fertilisation (Fig. 3). The content of HAPo (54.96 mg/kg) was significantly higher (Fig. 3f) in the NPK than the CK soil. The highest percentage soil total Po present as LPo occurred in the CK treatment (32.05%), but the highest percentages of total Po present as FAPo and HAPo were displayed in the NPK soil and that present as MLPo in the MNPK soil. The fertilised soils (M, NPK, MNPK and HMNPK) have higher percentage of FAPo than other Po fractions (35.5%, 40.35%, 30.36% and 42.64%, respectively). The percentage soil total Po present as HAPo was the lowest in the CK and M soils (Fig. S1).
Relationship of soil P status with phoD gene abundance and ALP activity
Significant positive correlation was observed between the phoD gene abundance and ALP activity (Fig. S2a; p < 0.01, r 2 = 0.856). The phoD gene abundance was significantly correlated with the LPo (Fig. S3a; p < 0.01, r 2 = 0.551), MLPo (Fig. S3b; p < 0.01, r 2 = 0.403) and FAPo (Fig. S3c; p < 0.05, r 2 = 0.272) contents but not with the HAPo content (Fig. S3d; p = 0.875, r 2 = 0.002). The ALP activity was significantly correlated with the LPo content (Fig. S3e; p < 0.01, r 2 = 0.840) but not with MLPo (Fig. S3f), FAPo (Fig. S3g) or HAPo (Fig. S3h) contents. In addition, a negative relationship was observed between the phoD gene abundance and available P content (Fig. S2b; p < 0.05, r 2 = 0.272) and ALP activity with both AP (Fig. S2c; p = 0.01, r 2 = 0.526) and total P (Fig. S2d; p = 0.01, r 2 = 0.398) contents.
Effects of different fertilisation treatments on the composition of microbial community harbouring the phoD gene
The rarefaction curves of all treated soils nearly reached a plateau, and all samples rarefied to 10,000 sequences; this suggests that the read number per sample was sufficient to capture the overall richness. The species richness and alpha diversity of M and CK soils were lower than those of the other fertilised soils (NPK, MNPK and HMNPK), as shown by the rarefaction curves, Shannon index and Chao index (Fig. S4; Table 2). The first two axes of redundancy analysis (RDA) explained 43.56% and 17.45% of the total variation. The phoD gene communities of CK and M soils were separated from the other treated soils (NPK, MNPK and HMNPK) along the first axis. The gene community compositions of NPK and CK soils were separated from those of the other organic-fertilised soils (M, MNPK and HMNPK) along the second axis (Fig. 4). According to the Monte Carlo permutation test (permutation = 9999), the ALP activity, pH, EC and AP; LPo; TP; Ca2+; Corg; DOC; and TN contents were represented with long arrows and were significantly correlated with the composition of the phoD microbial communities (Table S1).
Ordination plots by the redundancy analysis (RDA) used to explore the relationships between the ALP-encoding microbial community and selected soil properties. The arrows indicate the lengths and angles between explanatory and response variables and reflect their correlations. Samples from different long-term fertilisation treatments were marked with different colours. Symbols of fertiliser regimes are as described in Table 1. CK no fertiliser application, M application of single organic fertiliser, NPK application of single chemical fertiliser, MNPK mixed application of organic and chemical fertilisers, HMNPK mixed application of organic and chemical fertilisers with higher N rate. Numbers behind the treatments indicate the replications
Effects of different fertilisation treatments on the dominant phoD-harbouring microbial communities
Most of the OTUs were affiliated with the bacterial kingdom. Whereas, only a few OTUs were affiliated with the fungal kingdom. The dominant phoD-harbouring bacterial phyla in all treatments were Actinobacteria and Proteobacteria, and the dominant genera were Actinoplanes, Micromonospora, Streptomyces, Bradyrhizobium, Burkholderia and Lysobacter (>1%) (Fig. S5). Bradyrhizobium was the dominant phoD-harbouring genus in the different fertilised soils, particularly in the M, CK and MNPK soils (accounting for 57%, 53% and 51% of the total, respectively). The relative abundance of Streptomyces was higher in the organic-fertilised soils (range, 15–20%) than in the NPK and CK soils. The Sphingomonas and Thauera relative abundances were the highest in the NPK (12% of the total) and in the HMNPK soil (10%), respectively (Fig. S5).
The dominant species (OTUs; >10%) were used to construct the phylogenetic tree (Fig. 5). The OTU24880, OTU16861 and OTU22270 were closely related to Bradyrhizobium, and the OTU5587 was closely related to Mesorhizobium. The OTU4187 and OTU3540 were closely related to Sphingomonas and Actinomyces, respectively. The OTU26581 and OTU31653 were closely related to Frankia, and OTU7961 and OTU9863 to Streptomyces. The other OTUs were not closely related to a species but were located in either the Proteobacteria or the Actinobacteria cluster (Fig. 5).
Neighbour-joining phylogenetic tree that represents the phylogenetic relationships of the 15 OTUs (>10%) to the most closely related identified sequences obtained from the NCBI was inferred using the maximum likelihood method based on the Tamura-Nei model for a neighbour-joining tree. The percentage of trees in which the associated taxa clustered together is shown next to the branches. A bootstrap analysis was performed with 1000 trials. The bar indicates an estimated sequence divergence
The abundance of each dominant genus, which was calculated considering the relative abundance and qPCR data, was significantly shifted in soils by different fertilisation regimes (Fig. S6). All 12 dominant genera, except Sphingomonas, had lower abundance in the NPK soil than in the other fertilised soils. Micromonospora, Modestobacter, Streptomyces, Lysobacter and Bradyrhizobium were dominant genera in the M soil (1.82 × 106 copies g−1 soil, 5.90 × 106 copies g−1 soil, 1.39 × 107 copies g−1 soil, 6.56 × 106 copies g−1 soil and 4.83 × 107 copies g−1 soil, respectively). Notably, the highest abundance of Frankia occurred in the CK soil (3.34 × 106 copies g−1 soil), and the abundance of Burkholderia was higher in CK (3.52 × 106 copies g−1 soil) and M (2.83 × 106 copies g−1 soil) soil than in the other treated soils (Fig. S6).
Correlations of dominant genera abundances with ALP activity and soil properties
A significantly positive relationship was observed between ALP activity and abundances of Bradyrhizobium (p < 0.01, r 2 = 0.939), Modestobacter (p < 0.05, r 2 = 0.360), Frankia (p < 0.05, r 2 = 0.462), Streptomyces (p < 0.01, r 2 = 0.645), Lysobacter (p < 0.01, r 2 = 0.913) and Burkholderia (p < 0.01, r 2 = 0.747), and the abundances of these genera were the highest in the M or CK soils (Figs. S6 and S7).
According to the Pearson’s correlations (Table S2), the abundances of all dominant genera were significantly correlated with pH, except for those of Thauera and Frankia. The abundances of Modestobacter, Frankia, Bradyrhizobium, Burkholderia and Lysobacter were positively correlated with the LPo content. And the abundances of Frankia, Bradyrhizobium, Burkholderia and Lysobacter were positively correlated with the Ca2+ content but negatively correlated with the AP content. The abundances of Frankia were negatively correlated with TN, DOC and Corg contents, and these soil properties were positively correlated with Rhodanobacter abundance. Among all investigated soil properties, the abundance of Micromonospora, Streptomyces and Sphingomonas was only significantly correlated with soil pH (Table S2).
SEM pathways of fertilisation regimes indicate effects on soil ALP activity via soil properties
The SEMs explained 98% of the variance in soil ALP activities (Fig. 6), which depended on pH and contents of AP, DOC, LPo and Corg as affected by the different fertilisation regimes. Particularly, ALP activity was directly mediated by both soil pH (path coefficient = 0.480) and LPo content (path coefficient = 0.42) and probably indirectly by AP, Corg and DOC contents (Fig. 6). The abundance of the phoD gene was directly affected by the AP content, which affected the ALP activity as reported above. Probably, soil Corg content affected ALP activity indirectly via direct effect on composition of the phoD gene community or AP content. Finally, soil DOC content directly affected total bacterial abundance (total bacterial abundance is shown in Fig. S8) and then mediated the abundance and composition of the phoD gene microbial community; probably, this resulted in the ultimate regulation of ALP activity. Although 72% of the total variance in the α-diversity of phoD-harbouring microbes was explained by soil pH and DOC content, these soil properties did not play a significant role in the regulation of ALP activity. Nevertheless, biotic factors, including composition and abundance of the phoD gene communities directly affected ALP activity, with the path coefficients of −0.18 and 0.13, respectively (Fig. 6).
Structural equation model (SEM) of the influences of phoD gene composition and abundance, pH and labile organic P (LPo) content on ALP activity, as mediated by total bacterial abundance, dissolved organic C (DOC), Corg, and available P (AP) in the soil. Numbers following the included variables show the explained percentage of its variance by its predictors. Numbers on arrows are standardised path coefficients. Green and grey arrows indicate significant and unsignificant effects, respectively, with the thickness representing the extent of influence. The models fit the data well. Model fits are given in Table S3, with the significance levels provided in Table S4. The total bacterial population is shown in Fig. S8
Discussion
The abundance of the phoD gene increased significantly by organic-only fertilisation but decreased by chemical-only fertilisation when compared with the control. The organic fertiliser, rich in C substrates but low in available P, probably favoured the growth of some phoD-harbouring species, thus increasing the abundance of the phoD genes and ALP activity. However, the organic fertilisation decreased the diversity of the phoD genes in soil. Fraser et al. (2015b) also found fewer OTUs and higher ALP activity in organically managed soils than in conventionally managed soils. In addition, it is well established that low available P favour the synthesis of phosphatases (Nannipieri et al. 2011). On the other hand, the long-term, chemical-only fertilisation might have developed a low C resource environment, which might have favoured antagonistic bacteria (Bakker et al. 2013) with the increase in the competitive interactions of microbes for C substrate. As previously demonstrated, greater proportions of antagonistic bacteria are positively correlated with bacterial diversity (Schlatter et al. 2015). Therefore, although the soil had high phoD gene diversity, the ALP activity remained low, probably regulated by available P content and also associated with thew low phoD-harbouring microbial abundance.
Significant differences in the soil phoD bacterial community composition were observed with different fertilisation regimes. Our results confirmed that changes in the composition of phoD microbial communities can occur under organic fertilisation (Sakurai et al. 2008; Tan et al. 2013). The soil pH was the primary driver of the composition of phoD-harbouring communities in arable and grassland soils (Ragot et al. 2015; Wang et al. 2012); in addition, Ca2+, as a cofactor for the activity of ALP synthesised by the phoD gene (Kageyama et al. 2011), may affect indirectly the composition of phoD-harbouring microbial communities (Wu et al. 2007). Indeed, the Ca2+ content of soil with the contents of Po, Corg, DOC and pH had significant effects on the phoD microbial community composition, based on the Monte Carlo test (Table S1). However, this was not observed by the SEM model probably because only the first RDA axis score was considered to represent the community composition and Ca2+ content might affect community composition via the other axes.
The dominant phoD-harbouring microbes were Actinobacteria and Proteobacteria (Lagos et al. 2016). Both phyla together to Cyanobacteria have been already reported to be the dominant phoD-harbouring in soils subjected to different phosphate fertilisation regimes (Tan et al. 2013; Chaudhry et al. 2012; Chhabra et al. 2012). In all studied soils, the dominant genera of phoD-harbouring microbes were Bradyrhizobium and Streptomyces, thus conferming that the dominance of the phoD-harbouring genera does not change in soils by changing environmental conditions (Ragot et al. 2015). Particularly, the abundances of Actinoplanes, Micromonospora, Nocardioides, Rhodanobacter, Streptomyces and Thauera increased in organic fertilised soils. The abundances of Modestobacter, Frankia, Bradyrhizobium, Burkholderia and Lysobacter were significantly and positively affected by the LPo content, negatively affected by the AP content and positively correlated with the Ca2+ content, with the exception of Modestobacter. The abundances of Micromonospora, Streptomyces and Sphingomonas were only sensitive to the variation in soil pH. All of these results obviously suggest that the particular taxa within a phylum differ in their ecological characteristics (Spohn et al. 2015).
Bradyrhizobium, a free-living and symbiotic N2-fixer, had the highest relative abundance in all treated soils, suggesting that this genus might play an important role in coupling soil N and P cycle. Some alpha-Proteobacteria (e.g. Bradyrhizobium) respond to P stress by increasing ALP activity and P transport rates (Smart et al. 1984; Alniemi et al. 1997; Sakurai et al. 2008). Moreover, Dick et al. (1988) found that ALP is highly correlated with total N content. Therefore, future work should clarify the coupling between N cycling and P turnover under various conditions (e.g. AP-limiting) as mediated by Bradyrhizobium, which might not only contribute to the increases in ALP activity and P transport rates but also to modify soil N pools.
Because of differences in mineralogical composition and organic matter content, soil fractions provide different surface properties and microenvironments, which may affect the adsorption of chemicals and select for distinct microbial communities (Hemkemeyer et al. 2015). The highest abundance of the phoD gene was generally detected in the soil silt fraction, but the fertilisation regimes affected the abundance of the phoD-harbouring microbes within the different soil fractions (Fig. 2). The most abundant phoD-encoding microbes were harboured in the soil clay fraction in the chemical-only fertilisation treatment, which was unexpected because SOM in the soil clay fraction is suggested to be more recalcitrant than in the other soil fractions (Huang et al. 2010; Chung et al. 2008). However, phosphorylation of soil organic compounds is one of the strategies for increasing the recalcitrance of organic compounds by microbes (Yagil and Beacham 1975). Generally, plant phosphatases only function to mineralise organic P, whereas microbial phosphatases and other microbial enzymes are involved in organic C mineralisation to make both P and C available to microbial cells from Po (Steenbergh et al. 2011; Spohn and Kuzyakov 2013b; Heuck et al. 2015). Probably, microbes of long-term, chemical-only fertilised soils are under C-limited conditions and might release phosphomonoesterase not only to mineralise organic P but also to render organic-phosphorylated compounds available as a C source after dephosphorylation. If this hypothesis is true, enriched phoD-harbouring microbes in soil clay fraction under long-term chemical-only fertilisation may accelerate the dephosphorylation of SOM allocated to clay particles thus increasing degradation of SOM protected in clay, and soil ALP activity can play a role in regulating the stabilisation of soil C by clay particles. Further studies are required to understand the potential links between composition and function of phoD-harbouring microbial communities associated with different soil fractions under the influence of long-term fertilisation (Hemkemeyer et al. 2015) and the contributions of phosphomonoesterase activities to soil C stabilisation in different soil fractions.
Soil ALP activity was negatively correlated with AP (Acuña et al. 2016) but positively correlated with Po, thus conforming that available P represses the synthesis of phosphomonoesterases (Sakurai et al. 2008; Nannipieri et al. 2011; Zhang et al. 2012; Fraser et al. 2015a, b; Wei et al. 2014; Cui et al. 2015). In our study, organic amendment increased the contents of soil MLPo, FAPo and HAPo, and that of LPo significantly increased in organic-only amended soil. Whereas, the chemical fertiliser inputs decreased the LPo and MLPo concentration (Fig. 3). Probably, the content of AP were sufficient to microorganisms in the chemical-fertilised soil, and thus they did not need to decompose the stable Po (FAPo and HAPo) whose content increased with the fertilisation. Cui et al. (2015) found that soil LPo and MLPo contents showed a significant relationship with the phoD gene community composition, whereas in this study, the phoD gene abundance was significantly correlated with LPo, MLPo and FAPo contents, whereas the ALP activity was only positively correlated with the LPo content (Fig. S3) probably because LPo is the Po fraction more easily hydrolysed by the enzyme than the other Po fractions. The most recalcitrant Po fraction, HAPo, likely had no role in affecting either the abundance of phoD-harbouring microbes or ALP activity. Further research should also investigate the link between other phosphatases (i.e. acid phosphomonoesterase, phytase and phosphodiesterase) and the MLPo, FAPo and HAPo fractions.
Both acid and alkaline phosphomonoesterase activities can be used as indicators of the potential organic P mineralisation in soil (Chen 2003). Soil ALP activity was significantly affected by the different long-term fertilisation regimes, being increased by the addition of organic matter as already reported (Saha et al. 2008; Sakurai et al. 2008; Nannipieri et al. 2011; Zhang et al. 2014a), but decreased by the addition of mineral P (Saha et al. 2008; Spohn et al. 2015; Marklein and Houlton 2012). However, contradictory reports concerning the relationships between ALP activity and some soil properties have been reported. Dick et al. (1988) and Saha et al. (2008) found a positive correlation between phosphomonoesterase activity and SOM content, whereas others showed no relationship (Brockett et al. 2012; Fraser et al. 2015a, b; Senwo et al. 2007). By using the SEM model, we have observed that both soil pH (path coefficient = 0.480) and LPo (path coefficient = 0.42; Fig. 6) contents directly mediated ALP activity. Other studies showed that pH and LPo content were the primary driving factors affecting ALP activity; it is well established that ALP activity prevails over acid phosphomonoesterase activity at alkaline pH values and the opposite occurs at acid pH values (Dick et al. 2000; Nannipieri et al. 2011; Wei et al. 2014; Cui et al. 2015). In addition, we also observed indirect effects of soil Corg, DOC and AP contents on ALP activity via regulation of other biotic and abiotic variables caused by the different fertilisation regimes (Fig. 6).
According to the SEM, ALP activity was directly regulated by the phoD microbial abundance and composition. It has been known already that the phoD gene abundances and ALP activity are significantly correlated in soil under different management systems (Fraser et al. 2015b), and that the variation in the composition of the phoD gene induced by fertilisation regimes can affect ALP activity (Tan et al. 2013; Chaudhry et al. 2012; Chhabra et al. 2012). Here, we have observed that the abundances of Bradyrhizobium, Streptomyces, Modestobacter, Lysobacter, Frankia and Burkholderia were higher in organic-treated than in chemical-only fertilised soils, and they were significantly correlated with soil ALP activity. Probably, these genera may be more efficiently involved in the synthesis of the enzyme upon organic fertilisation than others (Figs. S6 and S7). To verify this, hypothesis research on expression of the relative enzyme-encoding genes is needed.
In conclusion, different fertilisation regimes notably changed the abundance and composition of ALP gene (phoD) harbouring microbial communities and ALP activities mainly by affecting soil properties as pH and contents of AP, DOC, LPo and Corg. The SEM showed that the changes in soil pH and LPo content induced by the different fertilisations were the primary driving forces affecting the ALP activity directly, whereas the other abiotic factors probably affected the ALP activity indirectly by altering the composition and abundance of phoD-harbouring microbes. Soil organic P fractions played an important role in shaping the composition of ALP-encoding microbial community and ALP activity. The highest number of the phoD gene was commonly associated to the soil silt fraction. It is hypothesised that the phoD microbial communities of the clay particles of the chemical-only fertilised soil may contribute to the degradation of the recalcitrant organic P compounds associated with clay particles by dephosphorylating these compounds. The obtained data confirmed the hypothesis that the abundance phoD gene differed in soil fractions of the different fertilised soils. Actinobacteria and Proteobacteria were identified as the dominant phoD-encoding phyla. The Bradyrhizobium, as a N2-fixer, was identified as an abundant phoD-encoding bacteria in all treated soils. Further studies should address the temporal variability and the expression of phoD-encoding microbial communities, and both determinations of mRNA and synthesised proteins are needed to confirm the relationship between phoD microbial communities and ALP activity.
References
Acuña JJ, Durán P, Lagos LM (2016) Bacterial alkaline phosphomonoesterase in the rhizospheres of plants grown in chilean extreme environments. Biol Fert Soils 52:1–11
Alniemi TS, Summers ML, Elkins JG, Kahn ML, Mcdermott TR (1997) Regulation of the phosphate stress response in rhizobium meliloti by phoB. Appl Environ Microb 63:4978–4981
Bakker MG, Otto-Hanson L, Lange AJ, Bradeen JM, Kinkel LL (2013) Plant monocultures produce more antagonistic soil streptomyces, communities than high-diversity plant communities. Soil Biol Biochem 65:304–312
Bowman RA, Cole CV (1978) An exploratory method for fractionation of organic phosphorus from grassland soils. Soil Sci 125:95–101
Brockett BFT, Prescott CE, Grayston SJ (2012) Soil moisture is the major factor influencing microbial community structure and enzyme activities across seven biogeoclimatic zones in western Canada. Soil Biol Biochem 4:9–20
Chaudhry V, Rehman A, Mishra A, Chauhan PS, Nautiyal CS (2012) Changes in bacterial community structure of agricultural land due to long-term organic and chemical amendments. Microb Ecol 64:450–460
Chen H (2003) Phosphatase activity and P fractions in soils of an 18-year-old Chinese fir (Cunninghamia lanceolata) plantation. Forest Ecol Manag 178:301–310
Chhabra S, Brazil D, Morrissey J, Burke J, O’Gara F, Dowling DN (2012) Fertilization management affects the alkaline phosphatase bacterial community in barley rhizosphere soil. Biol Fert Soils 49:31–39
Chu H, Grogan P (2010) Soil microbial biomass, nutrient availability and nitrogen mineralization potential among vegetation-types in a low arctic tundra landscape. Plant Soil 329:411–420
Chung H, Grove JH, Six J (2008) Indications for soil carbon saturation in a temperate agroecosystem. Soil Sci Soc Am J 72:1132–1139
Cordell D, Drangert JO, White S (2009) The story of phosphorus: global food security and food for thought. Global Environ Chang 19:292–305
Cui H, Zhou Y, Gu Z, Zhu H, Fu S, Yao Q (2015) The combined effects of cover crops and symbiotic microbes on phosphatase gene and organic phosphorus hydrolysis in subtropical orchard soils. Soil Biol Biochem 82:119–126
Dalal RC (1978) Organic phosphorus. Adv Agron 29:83–117
Dick RP, Rasmussen PE, Kerle EA (1988) Influence of long-term residue management on soil enzyme activities in relation to soil chemical properties of a wheat-fallow system. Biol Fert Soils 6:159–164
Dick WA, Cheng L, Wang P (2000) Soil acid and alkaline phosphatase activity as pH adjustment indicators. Soil Biol Biochem 32:1915–1919
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998
Fraser T, Lynch DH, Entz MH, Dunfield KE (2015a) Linking alkaline phosphatase activity with bacterial phoD gene abundance in soil from a long-term management trial. Geoderma 257:115–122
Fraser TD, Lynch DH, Bent E, Entz MH, Dunfield KE (2015b) Soil bacterial phoD, gene abundance and expression in response toapplied phosphorus and long-term management. Soil Biol Biochem 88:137–147
Grace JB (2006) Structural equation modeling and natural systems. Cambridge University Press, Cambridge
Hemkemeyer M, Christensen BT, Martens R, Tebbe CC (2015) Soil particle size fractions harbour distinct microbial communities and differ in potential for microbial mineralisation of organic pollutants. Soil Biol Biochem 90:255–265
Heuck C, Weig A, Spohn M (2015) Soil microbial biomass C:N:P stoichiometry and microbial use of organic phosphorus. Soil Biol Biochem 85:119–129
Huang S, Peng X, Huang Q (2010) Soil aggregation and organic carbon fractions affected by long-term fertilization in a red soil of subtropical China. Geoderma 154:364–369
Kageyama H, Tripathi K, Rai AK, Chaum S, Waditee-Sirisattha R, Takabe T (2011) An alkaline phosphatase/phosphodiesterase, PhoD, induced by salt stress and secreted out of the cells of Aphanothece halophytica, a halotolerant cyanobacterium. Appl Environ Microb 77:5178–5183
Kathuria S, Martiny AC (2011) Prevalence of a calcium-based alkaline phosphatase associated with the marine cyanobacterium prochlorococcus and other ocean bacteria. Environ Microb 13:74–83
Lagos LM, Acuna JJ, Maruyama F, Ogram A, Mora MDLL, Jorquera MA (2016) Effect of phosphorus addition on total and alkaline phosphomonoesterase-harboring bacterial populations in ryegrass rhizosphere microsites. Biol Fert Soils 52:1–13
Letunic I, Bork P (2006) Interactive tree of life (iTOL): an online tool for phylogenetic tree display and annotation. Bioinformatics 23:127–128
Ling N, Sun YM, Guo JJ, Zhu P, Peng C, Yu GH, Ran W, Guo SW, Shen QR (2014) Response of the bacterialdiversity and soil enzyme activity in particle-size fractions of Mollisol after different fertilization in a long-term experiment. Biol Fert Soils 50:901–911
Marklein AR, Houlton BZ (2012) Nitrogen inputs accelerate phosphorus cycling rates across a wide variety of terrestrial ecosystems. New Phytol 193:696–704
Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet Journal 17(1). doi:10.14806/ej.17.1.200
Nannipieri P, Giagnoni L, Landi L, Renella G (2011) Role of phosphatase enzymes in soil. In: Bünemann EK et al (eds) Phosphorus in action, soil biology, vol 26, pp 215–243
Nannipieri P, Giagnoni L, Renella G, Puglisi E, Ceccanti B, Masciandaro G, Fornasier F, Moscatelli MC, Marinari S (2012) Soil enzymology: classical and molecular approaches. Biol Fertil Soils 48:743–762
Nicolás C, Hernández T, García C (2012) Organic amendments as strategy to increase organic matter in particle-size fractions of a semi-arid soil. Appl Soil Ecol 57:50–58
Ragot SA, Huguenin-Elie O, Kertesz MA, Frossard E, Bünemann EK (2015) Total and active microbial communities and phoD as affected by phosphate depletion and pH in soil. Plant Soil 408:15–30
Richardson AE (2001) Prospects for using soil microorganisms to improve the acquisition of phosphorus by plants. Aust J Plant Physiol 28:897–906
Saha S, Prakash V, Kundu S, Kumar N, Lal Mina B (2008) Soil enzymatic activity as affected by long term application of farm yard manure and mineral fertilizer under rainfed soybean-wheat system in N-W Himalaya. Eur J Soil Biol 44:309–315
Sakurai M, Wasaki J, Tomizawa Y, Shinano T, Osaki M (2008) Analysis of bacterial communities on alkaline phosphatase genes in soil supplied with organic matter. Soil Sci Plant Nutr 54:62–71
Schlatter DC, Bakker MG, Bradeen JM, Kinkel LL (2015) Plant community richness and microbial interactions structure bacterial communities in soil. Ecology 96:134–142
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, Van Horn DJ, Weber CF (2009) Introducing mothur: opensource, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microb 75:7538–7541
Senwo ZN, Ranatunga TD, Tazisong IA, Taylor RW, He Z (2007) Phosphatase activity of ultisols and relationship to soil fertility indices. J Food Agric Environ 5(1):262–266
Slazak A, Freese D, Matos EDS, Hüttl RF (2010) Soil organic phosphorus fraction in pine–oak forest stands in northeastern Germany. Geoderma 158:156–162
Smart JB, Dilworth MJ, Robson AD (1984) Effect of phosphorus supply on phosphate uptake and alkaline phosphatase activity in rhizobia. Arch Microbiol 140:281–286
Spohn M, Kuzyakov Y (2013a) Distribution of microbial- and root-derived phosphatase activities in the rhizosphere depending on P availability and C allocatione coupling soil zymography with 14C imaging. Soil Biol Biochem 67:106–113
Spohn M, Kuzyakov Y (2013b) Phosphorus mineralization can be driven by microbial need for carbon. Soil Biol Biochem 61:69–75
Spohn M, Treichel NS, Cormann M, Schloter M, Fischer D (2015) Distribution of phosphatase activity and various bacterial phyla in the rhizosphere of Hordeum vulgare L. depending on P availability. Soil Biol Biochem 89:44–51
Steenbergh AK, Bodelier PLE, Hoogveld HL, Slomp CP, Laanbroek HJ (2011) Phosphatases relieve carbon limitation of microbial activity in Baltic Sea sediments along a redox-gradient. Limnol Oceanogr 56:2018–2026
Stemmer M, Gerzabek MH, Kandeler E (1998) Organic matter and enzyme activity in particle-size fractions of soils obtained after low-energy sonication. Soil Biol Biochem 30:9–17
Tabatabai MA, Bremner JM (1969) Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1:301–307
Tan H, Matthieu B, Mooij MJ, Rice O, Morrissey JP, Dobson A, Griffiths B, O'Gara F (2013) Long-term phosphorus fertilisation increased the diversity of the total bacterial community and the phoD phosphorus mineraliser group in pasture soils. Biol Fert Soils 49:661–672
Tarafdar JC, Claassen N (1988) Organic phosphorus compounds as a phosphorus source for higher plants through the activity of phosphatase produced by plant roots and microorganisms. Biol Fert Soils 5:308–312
Tarafdar JC, Jungk A (1987) Phosphatase activity in the rhizosphere and its relation to the depletion of soil organic phosphorus. Biol Fert Soils 3(4):199–204
Tiecher T, Santos DR, Calegari A (2012) Soil organic phosphorus forms under different soil management systems and winter crops, in a long term experiment. Soil Till Res 124:57–67
Waldrip HM, He Z, Erich MS (2011) Effects of poultry manure amendment on phosphorus uptake by ryegrass, soil phosphorus fractions and phosphatase activity. Biol Fert Soils 47(4):407–418
Wang Y, Zhang F, Marschner P (2012) Soil pH is the main factor influencing growth and rhizosphere properties of wheat following different pre-crops. Plant Soil 360:271–286
Wei K, Chen Z, Zhu A, Zhang J, Chen L (2014) Application of 31P NMR spectroscopy in determining phosphatase activities and P composition in soil aggregates influenced by tillage and residue management practices. Soil Till Res 138:35–43
Welsh C, Tenuta M, Flaten DN, Thiessen-Martens JR, Entz MH (2009) High yielding organic crop management decreases plant-available but not recalcitrant soil phosphorus. Agron J 101:1027–1035
Withers PJA, Haygarth PM (2007) Agriculture, phosphorus and eutrophication: a European perspective. Soil Use Manage 23:1–4
Wu JR, Shien JH, Shieh HK, Hu CC, Gong SR, Chen LY, Chang PC (2007) Cloning of the gene and characterization of the enzymatic properties of the monomeric alkaline phosphatase (PhoX) from Pasteurella multocida strain X-73. FEMS Microbiol Lett 267:113–120
Xun W, Zhao J, Xue C, Zhang G, Ran W, Wang B (2016) Significant alteration of soil bacterial communities and organic carbon decomposition by different long-term fertilization management conditions of extremely low-productivity arable soil in South China. Environ Microb 18:1907–1917
Yagil EZRA, Beacham IR (1975) Uptake of adenosine 50-monophosphate by Escherichia coli. J Bacteriol 121:401–405
Yu HY, Ding WX, Luo JF, Donnison A, Zhang JB (2012) Long-term effect of compost and inorganic fertilizer on activities of carbon-cycle enzymes in aggregates of an intensively cultivated sandy loam. Soil Use Manage 28:347–360
Zhang A, Chen Z, Zhang G, Chen L, Wu Z (2012) Soil phosphorus composition determined by 31P NMR spectroscopy and relative phosphatase activities influenced by land use. Eur J Soil Biol 52:73–77
Zhang J, Kobert K, Flouri T, Stamatakis A (2013) PEAR: a fast and accurate Illumina paired-end reAd mergeR. Bioinformatics 30:614–620
Zhang G, Chen Z, Zhang A, Chen L, Wu Z, Ma X (2014a) Phosphorus composition and phosphatase activities in soil affected by long-term application of pig manure and inorganic fertilizers. Commun Soil Sci Plan 45:1866–1876
Zhang WL, Xu AG, Zhang RL, Ji HJ (2014b) Review of soil classification and revision of China soil classification system. Sci Agric Sin 47:3214–3230
Zhong W, Gu T, Wang W, Zhang B, Lin X, Huang Q, Shen W (2010) The effects of mineral fertilizer and organic manure on soil microbial community and diversity. Plant Soil 326:511–522
Zimmerman AE, Martiny A, Allison SD (2013) Microdiversity of extracellular enzyme genes among sequenced prokaryotic genomes. ISME J 7:1187–1199
Acknowledgements
This work was financially supported by the China Science and Technology Ministry (2015CB150500, 2013CB127403), Youth Innovation Fund of Anhui Academy of Agricultural Sciences (15B0204) and Jiangsu Science and Technology Department (BK20150059).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 356 kb)
Rights and permissions
About this article
Cite this article
Luo, G., Ling, N., Nannipieri, P. et al. Long-term fertilisation regimes affect the composition of the alkaline phosphomonoesterase encoding microbial community of a vertisol and its derivative soil fractions. Biol Fertil Soils 53, 375–388 (2017). https://doi.org/10.1007/s00374-017-1183-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-017-1183-3