Abstract
Coronary computed tomography angiography (CCTA) has become a well-established diagnostic tool for the assessment of coronary artery stenosis. In particular, the high negative predictive value of almost a 100 % in patients with an intermediate likelihood of coronary artery disease permits reliable exclusion of coronary artery stenosis in patients with normal CCTA. Despite of the high diagnostic accuracy, CCTA is able to visualize not only the coronary artery stenosis but the coronary artery plaque. Differentiation of the coronary plaque composition is crucial to identify plaque conditions being prone to rupture. CCTA provides a high accuracy for plaque detection and yields important diagnostic information on the characterization of coronary artery plaques and the quantification of coronary artery stenosis.
Access provided by Autonomous University of Puebla. Download reference work entry PDF
Similar content being viewed by others
Keywords
- Coronary computed tomography angiography
- Coronary artery disease
- Plaque imaging
- Plaque characterization
- Coronary artery stenosis
- Histopathology
Key Facts of Coronary Computed Tomography Angiography
-
Coronary computed tomography angiography has a high diagnostic accuracy for the assessment of coronary artery stenosis particularly to exclude substantial stenosis in patients with atypical chest pain and an intermediate likelihood of coronary artery disease.
-
Coronary computed tomography angiography reliable depicts all kinds of coronary artery plaques.
-
The depiction rate of coronary computed tomography angiography depends on the observer’s experience, the stage of plaque progression, and the presence of calcifications.
-
Characterization of coronary artery plaques is feasible by coronary computed tomography angiography and permits identification of vulnerable plaques.
-
The quantification of coronary artery stenosis by coronary computed tomography angiography correlates with that from quantitative catheter angiography, but grading systems for the degree of coronary artery stenosis are recommendable for practical reasons.
Definitions
Cardiac computed tomography
Cardiac computed tomography is a diagnostic tool in which data acquisition of a computed tomography system is synchronized to the heartbeat by the ECG signal in order to suppress motion artifacts by the beating heart.
Catheter coronary angiography
Catheter coronary angiography is a cardiology diagnostic test and procedure in which a catheter is placed in the coronary artery and the heart chambers to visualize the coronary artery lumen and the blood flow.
Dual-energy computed tomography
Dual-energy imaging is a special computed tomography imaging method which is supplied by systems from different vendors. The cornerstone of dual-energy imaging is to perform computed tomography at two different energy levels. As a result, materials can be differentiated by differences in Compton scattering and photoeffect at both energy levels, and therefore dual-energy imaging adds to the diagnostic information from computed tomography imaging.
Dual-source computed tomography
Dual-source computed tomography is a special system, in which in opposition to the traditional single-source computed tomography, the scanner is composed of two X-ray tubes and two detectors are arranged in a 90° angular offset. The benefit of this special arrangement is a substantial increase in temporal resolution and the ability to perform dual-energy imaging.
Intravascular ultrasound
Intravascular ultrasound is an invasive cardiological method in which a special catheter with an ultrasound probe attached to the end of the catheter is placed in the coronary artery in order to visualize diseases in the coronary artery wall and visualization of coronary artery plaques.
Stary classification scheme
The Stary classification scheme is a histopathological grading system for atheromatous plaques, which has been proposed by an American Heart Association consensus group headed by Herbert Stary. This scheme divides the coronary artery plaques by the different plaque components into eight different types and differentiates early from advanced-stage plaques.
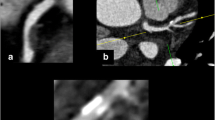
Atherosclerotic cardiovascular disease is one of the leading causes of morbidity and mortality in developed countries. Coronary artery disease is characterized by the development of plaques due to the accumulation of lipid or calcified deposits in the coronary artery wall (Enrico et al. 2009; Olgac et al. 2009). Acute myocardial infarction is most frequently the result of rupture of coronary atherosclerotic plaque with subsequent formation of an intraluminal thrombus (Arbustini et al. 1999; Thim et al. 2010; Henzler et al. 2011; Precht 2013). Procedures that can improve the detection, the determination of the plaque composition, and the plaque quantification are essential. Traditionally, imaging of coronary artery disease was based on catheter coronary angiography. Unfortunately, this method is limited by the luminographic visualization of the coronary arteries and the inability to visualize coronary atherosclerotic plaques (Fig. 1) (Raggi et al. 2005). Therefore, intravascular ultrasound (IVUS) is considered as the clinical reference modality for most accurate characterization and detection of coronary atherosclerotic plaques, regarding plaque morphology and quantification of arterial stenosis (Raggi et al. 2005). Despite its advantages compared with catheter coronary angiography, the application of IVUS for risk stratification in larger patient populations is limited due to its invasive nature. Hence a noninvasive, widely available technique for the detection and characterization of the content of coronary atherosclerotic plaques is desired. Today, computed tomography (CT) is the preferred cross-sectional imaging modality for detection, characterization and quantification of coronary artery stenosis .
Coronary Plaque Detection with CT
Technical Requirements
Coronary computed tomographic angiography (CCTA) is nowadays a clinically accepted technique for noninvasive and accurate assessment of coronary artery stenosis and coronary plaques (Dettmer et al. 2013; Alkadhi et al. 2008; Leber et al. 2006; Leschka et al. 2005). CCTA not only permits noninvasive detection and quantification of coronary artery stenosis but also enables the visualization and characterization of the atherosclerotic plaque (Fig. 1) (Feuchtner et al. 2012; Dey et al. 2010)
Initial CT studies using conventional single-energy CT systems with 4-slice and 16-slice technology have been limited by the relatively low spatial and temporal resolution (Becker et al. 2003; Halliburton et al. 2006). In 2004, 64-slice CT systems have become available for the first time and proofed to be robust enough for cardiac CT imaging in daily clinical practice (Fig. 2). These CT systems allow for an examination of the entire heart within a short breath-hold time of less than 10 s. Due to improvements in spatial resolution, 64-slice CT systems yield a detailed evaluation of small-sized branches of less than 1 mm in diameter (Leber et al. 2005a; Leschka et al. 2005; Mollet et al. 2005; Raff et al. 2005; Ehara et al. 2006; Nikolaou et al. 2006; Ong et al. 2006; Pugliese et al. 2006; Ropers et al. 2006). However, despite the improved temporal resolution, 64-slice CT systems still need a low and stable heart rate for optimal image quality (Leschka et al. 2006), rendering a heart rate control by the means of beta-blockers prior to CCTA frequently required. The remaining challenges of temporal resolution and a resulting high detector coverage speed have led to the development of the latest CT systems: 256-/320-slice CT and dual-source CT. These CT systems are capable of covering the entire heart within a single heartbeat. While the 256-/320-slice CT systems mainly focus on a large detector coverage, the dual-source CT system mainly focuses on a high temporal resolution: it is composed of two X-ray tubes, and two corresponding detectors are mounted onto the rotating gantry with an angular offset of 90° (Flohr et al. 2006) thus reducing the rotation angle needed to acquire the required projection data for image reconstruction from half a rotation in a single-source CT to a quarter of a rotation in a dual-source CT and thereby almost doubling the temporal resolution (Flohr et al. 2006; Leschka et al. 2007; Matt et al. 2007).
A more detailed chronology on the technical development of CT for cardiac imaging could be found in a recent review article (Leschka et al. 2009).
Depiction Rate of Coronary Artery Plaques with CT
A huge number of studies investigated the diagnostic performance of CCTA in the detection of coronary artery stenosis in comparison to catheter angiography (Table 1) (Leschka et al. 2009). All of the studies, irrespective of the CT scanner type used, demonstrated a high negative predictive value of CT approaching 100 %, but the rate of not-evaluative coronary artery segments highly depends on the CT system used with up to 32 % of coronary artery segments being not-evaluative when using 4-slice CT (Morgan-Hughes et al. 2003) and as low as 0 % being not-evaluative on 64-slice CT (Leschka et al. 2005) and dual-source CT (Tsiflikas et al. 2009). In general, the average specificity for patients with significant coronary artery stenosis has increased, while the number of not-evaluative segments decreased with modern CT technology. In addition, coronary artery plaques in proximal and mid-segments are depicted with a higher sensitivity than in distal segments (Leschka et al. 2009).
Despite of these encouraging results on the high diagnostic performance of CCTA, one must consider that those clinical studies commonly center on the detection of substantial coronary artery stenosis (i.e., luminal diameter narrowing of more than 50 %), while the CT detection rate of early stage coronary plaques causing a non-substantial coronary artery stenosis which may represent precursors of future coronary artery stenosis is less frequently investigated.
In a phantom study using a 64-slice dual-source CT, the detection rate for coronary plaques of any histopathologic subtype was 79 %, while the detection of advanced-stage plaques was 100 %. Nevertheless, although the CT system used in the mentioned study (Leschka et al. 2010) had a spatial resolution of 0.4 × 0.4 × 0.4 mm3, the size of early stage atherosclerotic plaques still was below the resolution of the CT scanner, so none of the very early stage plaques (i.e., Stary I) and only 17 % of Stary II plaques could be identified by CT.
Influence of Observer Experience on Plaque Detection with CT
An important factor affecting the reliability and accuracy of coronary artery plaque detection with CCTA is the observer’s experience in cardiac imaging. Saur et al. (2010) conducted a study involving three readers with different levels of expertise in cardiac CT imaging. All three readers evaluated 50 CCTA data sets twice regarding the presence or absence of coronary artery plaques. Afterward, a consensus reading was performed in which all three readers jointly determined the presence of coronary artery plaques, which was then defined as the reference standard of the study. The observer’s variability for plaque depiction varied between κ = 0.582 for the least experienced reader and κ = 0.892 for the observer with the highest experience in cardiac imaging. Furthermore, with an increasing level of experience, intraobserver variability for coronary plaque detection and the time required for interpreting the cardiac CT study significantly decreased.
The accuracy of coronary artery plaque detection depends on the observer’s experience, and the intraobserver variability and evaluation time decrease with increasing observer experience (Saur et al. 2010).
Coronary Plaque Characterization with CT
Data from patients with acute coronary syndrome have shown that two-thirds of culprit plaques were angiographically non-obstructive (i.e., less than 50 % luminal stenosis) (Ambrose et al. 1985). As such, the composition of the atherosclerotic lesion rather than the degree of stenosis is currently considered to be the most important determinant for the biomechanical stability of coronary atherosclerotic lesions and for acute clinical events (Pasterkamp et al. 2000). Albeit coronary calcification is associated with worse cardiovascular prognosis, the presence of calcification does not decrease the biomechanical stability of the plaque (Huang et al. 2001). In contrast, a thin fibrous cap and a large lipid pool are important determinants of increased risk for plaque rupture (Huang et al. 2001). The ability to recognize such plaques vulnerable to rupture is important for the detection of this risk and the possible prevention of acute coronary events.
In CCTA, plaque elements can be characterized by their CT attenuation profiles, which are determined by the physical properties of the contained elements (atomic number and density) and by the delivered tube voltage and tube current. A CT classification of coronary plaque structures has been performed according to the presence or absence of calcifications (Achenbach et al. 2004), by density measurements of the plaque (Nikolaou et al. 2003), or by both methods combined (Kopp et al. 2001; Becker et al. 2003; Schroeder et al. 2004a; Halliburton et al. 2006).
Histopathological Classification
Atherosclerotic lesions are often classified according to Stary (grades I–VIII) in histopathology (Stary 2000). An atherosclerotic plaque is classified as type I when containing small isolated groups of macrophages with intracellular fat accumulation; as type II when multiple foam cell layers are formed; as type III when isolated extracellular lipid pools are present; as type IV when the predominant tissue in the plaque is fat (histologically known as lipid core) and there is no detectable fibrotic component located toward the lumen; as type V when the predominant tissue in the plaque is fat with a fibrotic plaque component located toward the lumen (histologically known as fibrous cap) (Fig. 4); as type VI when the lumen adjacent to the plaque contains thrombotic material or the plaque surface is interrupted, indicating a complicated plaque; as type VII when the predominant tissue of the plaque is calcified; and as type VIII when the non-calcified tissue component is predominantly fibrotic (Leschka et al. 2010).
The plaque composition determines the risk of acute coronary events (Motoyama et al. 2007).
CT Classification of Coronary Plaques According to Calcified Components
The qualitative approaches classify coronary plaques as non-calcified, mixed, or calcified (Becker et al. 2003; Achenbach et al. 2004) (Fig. 3) or as predominantly lipid rich, intermediate, or predominantly calcified (Schroeder et al. 2004a). Becker et al. (2003) reported a moderate correlation of this descriptive classification scheme compared to the histopathological Stary classification. In particular, calcified plaques were found even in early stage Stary type III plaques. Moreover, plaques were classified as being non-calcified by CT, although being predominantly calcified type VII plaques. In contrast, Leschka et al. (2010) reported that none of the Stary type VII plaques were classified as non-calcified. Mixed plaque composition was found in Stary type IV–VIII and in 15 % of type VII plaques, whereas purely calcified plaques were not only present in type VII but also in type V and type VI plaques. Non-calcified plaques were found in all Stary types with the exception of type VII plaques. Thus, a qualitative CT classification as non-calcified might be useful to identify early stage plaques, whereas CT classification as mixed or calcified is highly predictive of advanced plaque stage. Nevertheless, a descriptive classification that is based on the presence of calcified components oversimplifies the histologic structure of coronary artery plaques and does not permit the identification of vulnerable plaques such as the fibrous cap atheroma (Huang et al. 2001; Leschka et al. 2010).
Classification of Coronary Plaques According to Density
The quantitative CT classification scheme for characterization of plaques is based on density measurements for distinguishing lipid, fibrotic, and calcified components (Kopp et al. 2001; Becker et al. 2003; Nikolaou et al. 2003; Schroeder et al. 2004b; Halliburton et al. 2006). A CT density threshold of 60 HU for distinguishing between predominantly lipid-rich and intermediate plaques and of 120 HU for distinguishing between fibrotic and calcified components has been proposed (Schroeder et al. 2004b).
Areas of plaque with density values <60 HU as measured by coronary computed tomography angiography are associated with an increased likelihood of lipid-core plaque in histology (Puchner et al. 2014).
Unfortunately, the measurement of plaque attenuation can be affected by several factors. Plaque density measurements can also be compromised by the presence of iodinated contrast agent in the arterial lumen (Cademartiri et al. 2005; Halliburton et al. 2006; Pohle et al. 2007). Image noise can compromise the mean HU values particularly of non-calcified plaque components, preventing accurate discrimination of lipid and fibrotic tissues on the basis of density measurements (Barreto et al. 2008). As a consequence, there is a large overlap of CT density values of lipid-rich and fibrous coronary artery plaques in CT, and discrimination of both plaque types by CT density is – if at all – only valid when considering a lipid-rich plaque at a CT density of less than 30 HU (Fig. 4).
In a recent study using dual-source CT (Leschka et al. 2010), only the predominantly calcified plaques of Stary type VII could be clearly distinguished from other plaque types due to the high atomic number of calcium. The average CT density of early stage Stary type II–III plaques was significantly lower than that of advanced-stage Stary IV–VIII plaques due to the increase of calcified components in advanced-stage plaques with a corresponding increase in the average CT density. However, a differentiation of Stary plaques except the calcified Stary VII plaques by the CT density value is not feasible.
A qualitative CT classification of plaque composition differentiating non-calcified, mixed, and calcified components as well as a quantitative classification that is based on the CT density measurements distinguishes between early and advanced-stage plaques (Leschka et al. 2010).
CT classification of coronary artery plaques on the basis of the presence of calcification and average CT density allows for the identification of Stary type VII plaques with a high diagnostic accuracy (Leschka et al. 2010).
Identification of Vulnerable Plaques by CT
The accurate assessment of plaque composition is of utmost importance, because atherosclerotic plaques that are prone to rupture typically have a lipid-rich necrotic core and a thin fibrous cap (Mann and Davies 1996; Virmani et al. 2000, 2002, 2003, 2006). Studies using single-source CT scanners found a significant overlap of the attenuation ranges of necrotic cores of rupture-prone plaques and of fibrous plaques, which limited the results (Sun et al. 2008; Hur et al. 2009).
Studies have described a napkin-ringlike attenuation pattern of atherosclerotic plaques on coronary CT images in patients with acute coronary syndrome, potentially representing a culprit lesion. The pattern is characterized by a core with low CT attenuation surrounded by a rim-like area of higher CT attenuation (Tanaka et al. 2008; Goldstein et al. 2009; Kashiwagi et al. 2009; Narula and Achenbach 2009; Maurovich-Horvat et al. 2010). Histopathologically, the lesion is characterized by a necrotic core, which is consistent with the low attenuation core of the plaque, and a significant amount of fibrous plaque tissue, which is consistent with the high attenuation rim on CT images (Fig. 5).
The napkin-ring sign is considered a CT signature of high-risk coronary atherosclerotic plaque (Maurovich-Horvat et al. 2010).
CT images have been reconstructed using filtered back projection algorithms since the inception of the modality. In recent years, iterative reconstruction algorithms have become popular and have been implemented in CCTA. Iterative reconstruction is based on multiple iteration steps taking into account the scanner geometry and noise statistics, and it provides improved image reconstruction with reduced scatter noise at the cost of higher computation time or it may be used to reduce the radiation exposure of CCTA. Several studies have shown that iterative reconstruction algorithms can improve image quality and reduce the radiation dose without increasing the image noise level (Moscariello et al. 2011; Scheffel et al. 2012; Schuhbaeck et al. 2013). It could be demonstrated that iterative reconstruction algorithms may improve the feasibility and accuracy of plaque and stenosis quantification (Moscariello et al. 2011; Scheffel et al. 2012; Fuchs et al. 2013; Morsbach et al. 2013; Schuhbaeck et al. 2013). In 2014 Puchner et al. (2014) investigated the effect of different iterative reconstruction algorithms on the assessment of atherosclerotic plaque composition, using the histopathologic plaque classification as reference standard. They could demonstrate an improved diagnostic accuracy of iterative reconstruction algorithms for the detection of vulnerable atherosclerotic plaques with a lipid-rich core (Puchner et al. 2014).
Iterative reconstruction improves detection of high-risk lipid-core plaques and thus may lead to improved management and risk stratification of patients with coronary artery disease (Puchner et al. 2014).
One future approach for coronary plaque characterization might be the use of dual-energy CT (Obaid et al. 2014). In dual-energy CT, both tubes of the dual-source CT system are operated with different tube voltage thereby allowing a material differentiation by the means of differences in photo effect and Compton scattering (Johnson 2012). Obaid et al. (2014) demonstrated that the lipid-rich necrotic core has a lower atomic number than calcium in the plaque and a lower density than fibrous tissue and results in both the lowest attenuation values and the smallest change in attenuation at different energies (Obaid et al. 2014). Using the attenuation at two different energies, it turned out that the dual-energy index of a necrotic core is significantly lower than that of fibrous plaques and calcified plaques, and, importantly, there is no significant overlap in the dual-energy indices of the necrotic core and the fibrous plaque (Obaid et al. 2014). There is a significant change in attenuation of the fibrous plaque at different X-ray energies but not of the lipid-rich necrotic core (Fig. 6) (Obaid et al. 2014).
Dual-energy coronary CT angiography imaging may improve differentiation of necrotic core and fibrous plaque (Obaid et al. 2014).
Coronary Plaque Quantification with CT
Quantification of coronary artery stenosis by CT can be performed as diameter stenosis or as area stenosis (Fig. 7). In our experience, diameter stenosis in CCTA correlates better with the invasive quantitative coronary angiography, whereas area stenosis in CCTA correlates better with intravascular ultrasound and histopathology. Some studies have tried to use a quantitative approach for determining percent stenosis and comparing these absolute values to quantitative coronary angiography (Cury et al. 2005, 2006; Hoffmann et al. 2005; Dragu et al. 2006; Busch et al. 2007; Achenbach 2008). Although the degree of stenosis detected by CCTA and conventional invasive angiography correlated, the relationship showed substantial scatter and limits of agreement typically ranged from 20 % to 40 % (Cury et al. 2005, 2006; Hoffmann et al. 2005; Dragu et al. 2006; Busch et al. 2007; Achenbach 2008). Therefore, applying a grading system for coronary artery stenosis is recommendable for practical reasons (Table 2). Cheng et al. (2008) proposed the use of a categorical 5-point score to grade the stenosis in CCTA and could demonstrate a significant agreement with quantitative conventional invasive coronary angiography. Cury et al. (2006) used a 3-point score system with a wide category of moderate stenosis, ranging from 41 % to 70 %. In daily clinical practice, we prefer the classification proposed by Goldstein et al. (2007) who included a separate category for total coronary occlusion, which can be an important distinction from high-grade stenosis.
Schematic drawing of stenosis measurement by CT. (a) Diameter stenosis is quantified by measuring the minimal luminal diameter in the stenosis and comparison with the average diameter of normal-appearing coronary segments proximal and distal to the stenosis. (b) Area stenosis is quantified by measuring the luminal area in the stenosis and comparison with the average luminal area of normal-appearing coronary segments proximal and distal to the stenosis
Quantitative catheter angiography is the gold standard for coronary artery stenosis quantification. Early reports on stenosis grading in CCTA using 64-slice CT in comparison to catheter angiography demonstrated only a minor correlation between both modalities (Leber et al. 2006). Modern CT systems such as dual-source CT provide accurate correlation of stenosis degree using angiography as reference and are more reliable in the prediction of high-grade lesions reaching >70 % stenosis (Dragu et al. 2008).
Dettmer et al. (2013) investigated the influence of the histopathological plaque type on the stenosis degree grading in CCTA. They found that the overall stenosis degree measurement significantly correlated between CT and histology and that the luminal narrowing of non-calcified plaques is underestimated while the stenosis caused by calcified plaques is overestimated by CCTA.
CT systematically overestimates the degree of stenosis in calcified plaques and underestimates the degree of stenosis in non-calcified plaques, while quantification is accurate in mixed plaques (Dettmer et al. 2013).
Schroeder et al. (2004a) correlated the histopathological Stary classification of atherosclerotic plaques with 16-slice CT, but a classification beyond the three-tier system non-calcified, mixed, and calcified was not possible and the degree of stenosis was not assessed. Dettmer et al. (2013) using a 64-slice dual-source CT found no differences in stenosis degree measurements between CT and histopathology for the Stary type III–VI and type VIII plaques, while they found a significant difference between the two modalities for the early non-calcified lesion Stary type II and the heavy calcified lesion Stary type VII. The stenosis degree measured by CT was significantly overestimated for Stary type VII plaques (mean difference −9 % ± 10 %) and significantly underestimated for Stary type II plaques (mean difference −14 % ± 9 %). The underestimation of early lesions is due to the limited spatial resolution, while it is known that calcifications lead to an overestimation of a stenosis. Both histopathology and CT found a significantly higher stenosis degree in advanced-stage plaques (Stary type IV–VIII) compared to early stage plaques (Stary II and III).
Modern CT reliably depicts advanced-stage coronary artery plaques with an overall good correlation of stenosis degree compared to histopathology. However, the degree of stenosis is systematically overestimated in calcified plaques and underestimated in non-calcified plaques.
Quantification of stenosis degree by CT is only accurate for mixed plaques (Dettmer et al. 2013).
Conclusion
Owing to technical developments in the past years, CCTA has advanced from a research tool to that of an increasingly used diagnostic modality in clinical practice. Modern CT systems not only permit accurate detection and quantification of coronary artery plaques but visualize the underlying coronary artery plaque and provide information on the plaque composition. From the clinical point of view, CCTA may be particularly useful for depiction of vulnerable plaques. Novel developments such as dual-energy CT may improve the detection of rupture-prone plaques.
Potential Applications to Prognosis, Other Diseases, or Conditions
The noninvasive identification of coronary atherosclerotic plaques and the determination of the plaque composition are some of the ultimate goals of coronary imaging. Improvements in cardiac CT imaging have rendered this imaging modality to a fundamental diagnostic tool in cardiology and can significantly improve the risk stratification of patients with suspected coronary artery disease and may improve the prognosis of coronary artery disease by early and focused therapy (Mowatt et al. 2008; Gao et al. 2011).
Abbreviations
- CCTA:
-
Coronary computed tomography angiography
- CT:
-
Computed tomography
- HU:
-
Hounsfield units
- IVUS:
-
Intravascular ultrasound
References
Achenbach S. Quantification of coronary artery stenoses by computed tomography. JACC Cardiovasc Imaging. 2008;1(4):472–4.
Achenbach S, Moselewski F, Ropers D, Ferencik M, Hoffmann U, MacNeill B, Pohle K, Baum U, Anders K, Jang IK, Daniel WG, Brady TJ. Detection of calcified and noncalcified coronary atherosclerotic plaque by contrast-enhanced, submillimeter multidetector spiral computed tomography: a segment-based comparison with intravascular ultrasound. Circulation. 2004;109(1):14–7.
Alkadhi H, Scheffel H, Desbiolles L, Gaemperli O, Stolzmann P, Plass A, Goerres GW, Luescher TF, Genoni M, Marincek B, Kaufmann PA, Leschka S. Dual-source computed tomography coronary angiography: influence of obesity, calcium load, and heart rate on diagnostic accuracy. Eur Heart. 2008;J29(6):766–76.
Ambrose JA, Winters SL, Arora RR, Haft JI, Goldstein J, Rentrop KP, Gorlin R, Fuster V. Coronary angiographic morphology in myocardial infarction: a link between the pathogenesis of unstable angina and myocardial infarction. J Am Coll Cardiol. 1985;6(6):1233–8.
Arbustini E, Dal Bello B, Morbini P, Burke AP, Bocciarelli M, Specchia G, Virmani R. Plaque erosion is a major substrate for coronary thrombosis in acute myocardial infarction. Heart. 1999;82(3):269–72.
Barreto M, Schoenhagen P, Nair A, Amatangelo S, Milite M, Obuchowski NA, Lieber ML, Halliburton SS. Potential of dual-energy computed tomography to characterize atherosclerotic plaque: ex vivo assessment of human coronary arteries in comparison to histology. J Cardiovasc Comput Tomogr. 2008;2(4):234–42.
Becker CR, Nikolaou K, Muders M, Babaryka G, Crispin A, Schoepf UJ, Loehrs U, Reiser MF. Ex vivo coronary atherosclerotic plaque characterization with multi-detector-row CT. Eur Radiol. 2003;13(9):2094–8.
Busch S, Johnson TR, Nikolaou K, von Ziegler F, Knez A, Reiser MF, Becker CR. Visual and automatic grading of coronary artery stenoses with 64-slice CT angiography in reference to invasive angiography. Eur Radiol. 2007;17(6):1445–51.
Cademartiri F, Mollet NR, Runza G, Bruining N, Hamers R, Somers P, Knaapen M, Verheye S, Midiri M, Krestin GP, de Feyter PJ. Influence of intracoronary attenuation on coronary plaque measurements using multislice computed tomography: observations in an ex vivo model of coronary computed tomography angiography. Eur Radiol. 2005;15(7):1426–31.
Caussin C, Daoud B, Ghostine S, Perrier E, Habis M, Lancelin B, Angel CY, Paul JF. Comparison of lumens of intermediate coronary stenosis using 16-slice computed tomography versus intravascular ultrasound. Am J Cardiol. 2005;96(4):524–8.
Cheng V, Gutstein A, Wolak A, Suzuki Y, Dey D, Gransar H, Thomson LE, Hayes SW, Friedman JD, Berman DS. Moving beyond binary grading of coronary arterial stenoses on coronary computed tomographic angiography: insights for the imager and referring clinician. JACC Cardiovasc Imaging. 2008;1(4):460–71.
Cury RC, Pomerantsev EV, Ferencik M, Hoffmann U, Nieman K, Moselewski F, Abbara S, Jang IK, Brady TJ, Achenbach S. Comparison of the degree of coronary stenoses by multidetector computed tomography versus by quantitative coronary angiography. Am J Cardiol. 2005;96(6):784–7.
Cury RC, Ferencik M, Achenbach S, Pomerantsev E, Nieman K, Moselewski F, Abbara S, Jang IK, Brady TJ, Hoffmann U. Accuracy of 16-slice multi-detector CT to quantify the degree of coronary artery stenosis: assessment of cross-sectional and longitudinal vessel reconstructions. Eur J Radiol. 2006;57(3):345–50.
Dettmer M, Glaser-Gallion N, Stolzmann P, Glaser-Gallion F, Fornaro J, Feuchtner G, Jochum W, Alkadhi H, Wildermuth S, Leschka S. Quantification of coronary artery stenosis with high-resolution CT in comparison with histopathology in an ex vivo study. Eur J Radiol. 2013;82(2):264–9.
Dey D, Schepis T, Marwan M, Slomka PJ, Berman DS, Achenbach S. Automated three-dimensional quantification of noncalcified coronary plaque from coronary CT angiography: comparison with intravascular US. Radiology. 2010;257(2):516–22.
Dragu R, Rispler S, Ghersin E, Gruberg L, Lessick J, Litmanovich D, Aronson D, Hammerman H, Ofer A, Engel A, Beyar R. Contrast enhanced multi-detector computed tomography coronary angiography versus conventional invasive quantitative coronary angiography in acute coronary syndrome patients-correlation and bias. Acute Card Care. 2006;8(2):99–104.
Dragu R, Kerner A, Gruberg L, Rispler S, Lessick J, Ghersin E, Litmanovich D, Engel A, Beyar R, Roguin A. Angiographically uncertain left main coronary artery narrowings: correlation with multidetector computed tomography and intravascular ultrasound. Int J Cardiovasc Imaging. 2008;24(5):557–63.
Ehara M, Surmely JF, Kawai M, Katoh O, Matsubara T, Terashima M, Tsuchikane E, Kinoshita Y, Suzuki T, Ito T, Takeda Y, Nasu K, Tanaka N, Murata A, Suzuki Y, Sato K. Diagnostic accuracy of 64-slice computed tomography for detecting angiographically significant coronary artery stenosis in an unselected consecutive patient population: comparison with conventional invasive angiography. Circ. 2006;J70(5):564–71.
Enrico B, Suranyi P, Thilo C, Bonomo L, Costello P, Schoepf UJ. Coronary artery plaque formation at coronary CT angiography: morphological analysis and relationship to hemodynamics. Eur Radiol. 2009;19(4):837–44.
Feuchtner G, Loureiro R, Bezerra H, Rocha-Filho JA, Sarwar A, Pflederer T, Marwan M, Petranovic M, Raffel CO, Brady TB, Jang IK, Achenbach S, Cury RC. Quantification of coronary stenosis by dual source computed tomography in patients: a comparative study with intravascular ultrasound and invasive angiography. Eur J Radiol. 2012;81(1):83–8.
Flohr TG, McCollough CH, Bruder H, Petersilka M, Gruber K, Suss C, Grasruck M, Stierstorfer K, Krauss B, Raupach R, Primak AN, Kuttner A, Achenbach S, Becker C, Kopp A, Ohnesorge BM. First performance evaluation of a dual-source CT (DSCT) system. Eur Radiol. 2006;16(2):256–68.
Fuchs TA, Fiechter M, Gebhard C, Stehli J, Ghadri JR, Kazakauskaite E, Herzog BA, Husmann L, Gaemperli O, Kaufmann PA. CT coronary angiography: impact of adapted statistical iterative reconstruction (ASIR) on coronary stenosis and plaque composition analysis. Int J Cardiovasc Imaging. 2013;29(3):719–24.
Gao D, Ning N, Guo Y, Ning W, Niu X, Yang J. Computed tomography for detecting coronary artery plaques: a meta-analysis. Atherosclerosis. 2011;219(2):603–9.
Goldstein JA, Gallagher MJ, O’Neill WW, Ross MA, O’Neil BJ, Raff GL. A randomized controlled trial of multi-slice coronary computed tomography for evaluation of acute chest pain. J Am Coll Cardiol. 2007;49(8):863–71.
Goldstein JA, Grines C, Fischell T, Virmani R, Rizik D, Muller J, Dixon SR. Coronary embolization following balloon dilation of lipid-core plaques. JACC Cardiovasc Imaging. 2009;2(12):1420–4.
Halliburton SS, Schoenhagen P, Nair A, Stillman A, Lieber M, Murat Tuzcu E, Geoffrey Vince D, White RD. Contrast enhancement of coronary atherosclerotic plaque: a high-resolution, multidetector-row computed tomography study of pressure-perfused, human ex-vivo coronary arteries. Coron Artery Dis. 2006;17(6):553–60.
Henzler T, Porubsky S, Kayed H, Harder N, Krissak UR, Meyer M, Sueselbeck T, Marx A, Michaely H, Schoepf UJ, Schoenberg SO, Fink C. Attenuation-based characterization of coronary atherosclerotic plaque: comparison of dual source and dual energy CT with single-source CT and histopathology. Eur J Radiol. 2011;80(1):54–9.
Hoffmann MH, Shi H, Schmitz BL, Schmid FT, Lieberknecht M, Schulze R, Ludwig B, Kroschel U, Jahnke N, Haerer W, Brambs HJ, Aschoff AJ. Noninvasive coronary angiography with multislice computed tomography. JAMA. 2005;293(20):2471–8.
Huang H, Virmani R, Younis H, Burke AP, Kamm RD, Lee RT. The impact of calcification on the biomechanical stability of atherosclerotic plaques. Circulation. 2001;103(8):1051–6.
Hur J, Kim YJ, Lee HJ, Nam JE, Choe KO, Seo JS, Choi DH, Kim JS, Choi BW. Quantification and characterization of obstructive coronary plaques using 64-slice computed tomography: a comparison with intravascular ultrasound. J Comput Assist Tomogr. 2009;33(2):186–92.
Johnson TR. Dual-energy CT: general principles. AJR Am J Roentgenol. 2012;199(5 Suppl):S3–8.
Kashiwagi M, Tanaka A, Kitabata H, Tsujioka H, Kataiwa H, Komukai K, Tanimoto T, Takemoto K, Takarada S, Kubo T, Hirata K, Nakamura N, Mizukoshi M, Imanishi T, Akasaka T. Feasibility of noninvasive assessment of thin-cap fibroatheroma by multidetector computed tomography. JACC Cardiovasc Imaging. 2009;2(12):1412–9.
Kopp AF, Schroeder S, Baumbach A, Kuettner A, Georg C, Ohnesorge B, Heuschmid M, Kuzo R, Claussen CD. Non-invasive characterisation of coronary lesion morphology and composition by multislice CT: first results in comparison with intracoronary ultrasound. Eur Radiol. 2001;11(9):1607–11.
Leber AW, Knez A, Becker A, Becker C, Reiser M, Steinbeck G, Boekstegers P. Visualising noncalcified coronary plaques by CT. Int J Cardiovasc Imaging. 2005a;21(1):55–61.
Leber AW, Knez A, von Ziegler F, Becker A, Nikolaou K, Paul S, Wintersperger B, Reiser M, Becker CR, Steinbeck G, Boekstegers P. Quantification of obstructive and nonobstructive coronary lesions by 64-slice computed tomography: a comparative study with quantitative coronary angiography and intravascular ultrasound. J Am Coll Cardiol. 2005b;46(1):147–54.
Leber AW, Becker A, Knez A, von Ziegler F, Sirol M, Nikolaou K, Ohnesorge B, Fayad ZA, Becker CR, Reiser M, Steinbeck G, Boekstegers P. Accuracy of 64-slice computed tomography to classify and quantify plaque volumes in the proximal coronary system: a comparative study using intravascular ultrasound. J Am Coll Cardiol. 2006;47(3):672–7.
Leschka S, Alkadhi H, Plass A, Desbiolles L, Grunenfelder J, Marincek B, Wildermuth S. Accuracy of MSCT coronary angiography with 64-slice technology: first experience. Eur Heart. 2005;J26(15):1482–7.
Leschka S, Wildermuth S, Boehm T, Desbiolles L, Husmann L, Plass A, Koepfli P, Schepis T, Marincek B, Kaufmann PA, Alkadhi H. Noninvasive coronary angiography with 64-section CT: effect of average heart rate and heart rate variability on image quality. Radiology. 2006;241(2):378–85.
Leschka S, Scheffel H, Desbiolles L, Plass A, Gaemperli O, Valenta I, Husmann L, Flohr TG, Genoni M, Marincek B, Kaufmann PA, Alkadhi H. Image quality and reconstruction intervals of dual-source CT coronary angiography: recommendations for ECG-pulsing windowing. Invest Radiol. 2007;42(8):543–9.
Leschka S, Stolzmann P, Alkadhi H. Recent developments in coronary computed tomography imaging. Imaging Med. 2009;1(1):103–14.
Leschka S, Seitun S, Dettmer M, Baumuller S, Stolzmann P, Goetti R, Scheffel H, Feuchtner G, Wunnicke K, Wildermuth S, Oehlschlegel C, Marincek B, Jochum W, Alkadhi H. Ex vivo evaluation of coronary atherosclerotic plaques: characterization with dual-source CT in comparison with histopathology. J Cardiovasc Comput Tomogr. 2010;4(5):301–8.
Mann JM, Davies MJ. Vulnerable plaque. Relation of characteristics to degree of stenosis in human coronary arteries. Circulation. 1996;94(5):928–31.
Matt D, Scheffel H, Leschka S, Flohr TG, Marincek B, Kaufmann PA, Alkadhi H. Dual-source CT coronary angiography: image quality, mean heart rate, and heart rate variability. AJR Am J Roentgenol. 2007;189(3):567–73.
Maurovich-Horvat P, Hoffmann U, Vorpahl M, Nakano M, Virmani R, Alkadhi H. The napkin-ring sign: CT signature of high-risk coronary plaques? JACC Cardiovasc Imaging. 2010;3(4):440–4.
Mollet NR, Cademartiri F, van Mieghem CA, Runza G, McFadden EP, Baks T, Serruys PW, Krestin GP, de Feyter PJ. High-resolution spiral computed tomography coronary angiography in patients referred for diagnostic conventional coronary angiography. Circulation. 2005;112(15):2318–23.
Morgan-Hughes GJ, Marshall AJ, Roobottom CA. Multislice computed tomographic coronary angiography: experience in a UK centre. Clin Radiol. 2003;58(5):378–83.
Morsbach F, Desbiolles L, Plass A, Leschka S, Schmidt B, Falk V, Alkadhi H, Stolzmann P. Stenosis quantification in coronary CT angiography: impact of an integrated circuit detector with iterative reconstruction. Invest Radiol. 2013;48(1):32–40.
Moscariello A, Takx RA, Schoepf UJ, Renker M, Zwerner PL, O’Brien TX, Allmendinger T, Vogt S, Schmidt B, Savino G, Fink C, Bonomo L, Henzler T. Coronary CT angiography: image quality, diagnostic accuracy, and potential for radiation dose reduction using a novel iterative image reconstruction technique-comparison with traditional filtered back projection. Eur Radiol. 2011;21(10):2130–8.
Motoyama S, Kondo T, Sarai M, Sugiura A, Harigaya H, Sato T, Inoue K, Okumura M, Ishii J, Anno H, Virmani R, Ozaki Y, Hishida H, Narula J. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J Am Coll Cardiol. 2007;50(4):319–26.
Mowatt G, Cook JA, Hillis GS, Walker S, Fraser C, Jia X, Waugh N. 64-Slice computed tomography angiography in the diagnosis and assessment of coronary artery disease: systematic review and meta-analysis. Heart. 2008;94(11):1386–93.
Narula J, Achenbach S. Napkin-ring necrotic cores: defining circumferential extent of necrotic cores in unstable plaques. JACC Cardiovasc Imaging. 2009;2(12):1436–8.
Nikolaou K, Sagmeister S, Knez A, Klotz E, Wintersperger BJ, Becker CR, Reiser MF. Multidetector-row computed tomography of the coronary arteries: predictive value and quantitative assessment of non-calcified vessel-wall changes. Eur Radiol. 2003;13(11):2505–12.
Nikolaou K, Knez A, Rist C, Wintersperger BJ, Leber A, Johnson T, Reiser MF, Becker CR. Accuracy of 64-MDCT in the diagnosis of ischemic heart disease. AJR Am J Roentgenol. 2006;187(1):111–7.
Obaid DR, Calvert PA, Gopalan D, Parker RA, West NE, Goddard M, Rudd JH, Bennett MR. Dual-energy computed tomography imaging to determine atherosclerotic plaque composition: a prospective study with tissue validation. J Cardiovasc Comput Tomogr. 2014;8(3):230–7.
Olgac U, Poulikakos D, Saur SC, Alkadhi H, Kurtcuoglu V. Patient-specific three-dimensional simulation of LDL accumulation in a human left coronary artery in its healthy and atherosclerotic states. Am J Physiol Heart Circ Physiol. 2009;296(6):H1969–82.
Ong AT, Serruys PW, Mohr FW, Morice MC, Kappetein AP, Holmes Jr DR, Mack MJ, van den Brand M, Morel MA, van Es GA, Kleijne J, Koglin J, Russell ME. The SYNergy between percutaneous coronary intervention with TAXus and cardiac surgery (SYNTAX) study: design, rationale, and run-in phase. Am Heart. 2006;J151(6):1194–204.
Pasterkamp G, Falk E, Woutman H, Borst C. Techniques characterizing the coronary atherosclerotic plaque: influence on clinical decision making? J Am Coll Cardiol. 2000;36(1):13–21.
Petranovic M, Soni A, Bezzera H, Loureiro R, Sarwar A, Raffel C, Pomerantsev E, Jang IK, Brady TJ, Achenbach S, Cury RC. Assessment of nonstenotic coronary lesions by 64-slice multidetector computed tomography in comparison to intravascular ultrasound: evaluation of nonculprit coronary lesions. J Cardiovasc Comput Tomogr. 2009;3(1):24–31.
Pohle K, Achenbach S, Macneill B, Ropers D, Ferencik M, Moselewski F, Hoffmann U, Brady TJ, Jang IK, Daniel WG. Characterization of non-calcified coronary atherosclerotic plaque by multi-detector row CT: comparison to IVUS. Atherosclerosis. 2007;190(1):174–80.
Precht H. Optimisation of post mortem cardiac computed tomography compared to optical coherence tomography and histopathology – technical note. J Forensic Radiol Imaging. 2013;2(2014):85–90.
Puchner SB, Ferencik M, Maurovich-Horvat P, Nakano M, Otsuka F, Kauczor HU, Virmani R, Hoffmann U, Schlett CL. Iterative image reconstruction algorithms in coronary CT angiography improve the detection of lipid-core plaque – a comparison with histology. Eur Radiol. 2014;21:318–24.
Pugliese F, Mollet NR, Runza G, van Mieghem C, Meijboom WB, Malagutti P, Baks T, Krestin GP, deFeyter PJ, Cademartiri F. Diagnostic accuracy of non-invasive 64-slice CT coronary angiography in patients with stable angina pectoris. Eur Radiol. 2006;16(3):575–82.
Raff GL, Gallagher MJ, O’Neill WW, Goldstein JA. Diagnostic accuracy of noninvasive coronary angiography using 64-slice spiral computed tomography. J Am Coll Cardiol. 2005;46(3):552–7.
Raggi P, Taylor A, Fayad Z, O’Leary D, Nissen S, Rader D, Shaw LJ. Atherosclerotic plaque imaging: contemporary role in preventive cardiology. Arch Intern Med. 2005;165(20):2345–53.
Ropers D, Rixe J, Anders K, Kuttner A, Baum U, Bautz W, Daniel WG, Achenbach S. Usefulness of multidetector row spiral computed tomography with 64- × 0.6-mm collimation and 330-ms rotation for the noninvasive detection of significant coronary artery stenoses. Am J Cardiol. 2006;97(3):343–8.
Saur SC, Alkadhi H, Stolzmann P, Baumuller S, Leschka S, Scheffel H, Desbiolles L, Fuchs TJ, Szekely G, Cattin PC. Effect of reader experience on variability, evaluation time and accuracy of coronary plaque detection with computed tomography coronary angiography. Eur Radiol. 2010;20(7):1599–606.
Scheffel H, Stolzmann P, Schlett CL, Engel LC, Major GP, Karolyi M, Do S, Maurovich-Horvat P, Hoffmann U. Coronary artery plaques: cardiac CT with model-based and adaptive-statistical iterative reconstruction technique. Eur J Radiol. 2012;81(3):e363–9.
Schroeder S, Kuettner A, Leitritz M, Janzen J, Kopp AF, Herdeg C, Heuschmid M, Burgstahler C, Baumbach A, Wehrmann M, Claussen CD. Reliability of differentiating human coronary plaque morphology using contrast-enhanced multislice spiral computed tomography: a comparison with histology. J Comput Assist Tomogr. 2004a;28(4):449–54.
Schroeder S, Kuettner A, Wojak T, Janzen J, Heuschmid M, Athanasiou T, Beck T, Burgstahler C, Herdeg C, Claussen CD, Kopp AF. Non-invasive evaluation of atherosclerosis with contrast enhanced 16 slice spiral computed tomography: results of ex vivo investigations. Heart. 2004b;90(12):1471–5.
Schuhbaeck A, Achenbach S, Layritz C, Eisentopf J, Hecker F, Pflederer T, Gauss S, Rixe J, Kalender W, Daniel WG, Lell M, Ropers D. Image quality of ultra-low radiation exposure coronary CT angiography with an effective dose <0.1 mSv using high-pitch spiral acquisition and raw data-based iterative reconstruction. Eur Radiol. 2013;23(3):597–606.
Stary HC. Natural history and histological classification of atherosclerotic lesions: an update. Arterioscler Thromb Vasc Biol. 2000;20(5):1177–8.
Sun J, Zhang Z, Lu B, Yu W, Yang Y, Zhou Y, Wang Y, Fan Z. Identification and quantification of coronary atherosclerotic plaques: a comparison of 64-MDCT and intravascular ultrasound. AJR Am J Roentgenol. 2008a;190(3):748–54.
Sun Z, Lin C, Davidson R, Dong C, Liao Y. Diagnostic value of 64-slice CT angiography in coronary artery disease: a systematic review. Eur J Radiol. 2008b;67(1):78–84.
Tanaka A, Shimada K, Yoshida K, Jissyo S, Tanaka H, Sakamoto M, Matsuba K, Imanishi T, Akasaka T, Yoshikawa J. Non-invasive assessment of plaque rupture by 64-slice multidetector computed tomography – comparison with intravascular ultrasound. Circ J. 2008;72(8):1276–81.
Thim T, Hagensen MK, Wallace-Bradley D, Granada JF, Kaluza GL, Drouet L, Paaske WP, Botker HE, Falk E. Unreliable assessment of necrotic core by virtual histology intravascular ultrasound in porcine coronary artery disease. Circ Cardiovasc Imaging. 2010;3(4):384–91.
Tsiflikas I, Brodoefel H, Reimann AJ, Thomas C, Ketelsen D, Schroeder S, Kopp AF, Claussen CD, Burgstahler C, Heuschmid M. Coronary CT angiography with dual source computed tomography in 170 patients. Eur J Radiol. 2009;44:159–67.
Virmani R, Kolodgie FD, Burke AP, Farb A, Schwartz SM. Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler Thromb Vasc Biol. 2000;20(5):1262–75.
Virmani R, Burke AP, Kolodgie FD, Farb A. Vulnerable plaque: the pathology of unstable coronary lesions. J Interv Cardiol. 2002;15(6):439–46.
Virmani R, Burke AP, Kolodgie FD, Farb A. Pathology of the thin-cap fibroatheroma: a type of vulnerable plaque. J Interv Cardiol. 2003;16(3):267–72.
Virmani R, Burke AP, Farb A, Kolodgie FD. Pathology of the vulnerable plaque. J Am Coll Cardiol. 2006;47(8 Suppl):C13–8.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media Dordrecht
About this entry
Cite this entry
Leschka, S., Waelti, S., Wildermuth, S. (2016). Comparing Cardiac Computed Tomography and Histology in Coronary Artery Stenosis. In: Patel, V., Preedy, V. (eds) Biomarkers in Cardiovascular Disease. Biomarkers in Disease: Methods, Discoveries and Applications. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-7678-4_12
Download citation
DOI: https://doi.org/10.1007/978-94-007-7678-4_12
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-7677-7
Online ISBN: 978-94-007-7678-4
eBook Packages: Biomedical and Life SciencesReference Module Biomedical and Life Sciences