Abstract
Purpose of Review
Radionuclide myocardial perfusion imaging (MPI) continues to be the most reliable modality for diagnosis of hemodynamically significant coronary artery disease (CAD). The application of radionuclide MPI using single photon emission computed tomography (SEPCT) and positron emission tomography (PET) for CAD is reviewed, with emphasis on diagnosis and risk stratification.
Recent Findings
Contemporary studies have reported the diagnostic and prognostic value of novel imaging protocols, employing stress-first or stress-only approach. In addition, the superior diagnostic value of PET has been established with a role of assessment of myocardial blood flow to improve risk stratification. The utility of MPI in special populations, such as the elderly, women, and diabetic patients has also been recently evaluated. Furthermore, multicenter studies have reported a similar diagnostic and prognostic value of radionuclide MPI compared with other functional and anatomical techniques for CAD.
Summary
Radionuclide MPI with SPECT and PET are efficacious for diagnosis and prognosis of CAD. Its universal application in varied patient populations highlights its excellent clinical effectiveness.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Once a problem of only western and developed countries, coronary artery disease (CAD) has now become a common cause of mortality and morbidity worldwide [1]. Furthermore, caring for patients with known or suspected CAD poses tremendous economic pressure on healthcare resources, not only due to costs related to testing and treatment, but also those associated with loss of productivity in afflicted individuals [1]. Therefore, it is expected that more efficient use of resources and focus on improving cardiovascular outcomes in such patients will be prioritized as we continue to move from volume- to value-based care [2]. In addition, there is currently growing emphasis on maximizing the yield of diagnostic testing, such that an imaging modality is not only expected to confirm the presence or absence of a certain diagnosis, but also to guide post-test decision making in ways that will improve patients’ prognosis and quality of life [3].
There are several objectives to be achieved when evaluating patients presenting with chest pain or other anginal-equivalent symptoms. Determining the etiology of symptoms and particularly discerning whether they can be attributed to underlying CAD is arguably the principal goal of care in these patients. Once the diagnosis of CAD is confirmed, the next goal would be to stratify those with disease according to their risk of future adverse cardiovascular events based on the anatomical or physiological severity extent of the disease. Finally, this information will also be used to guide referral for more invasive cardiac procedures, such as coronary angiography and revascularization, based on the likelihood that patients will derive more benefit from revascularization compared to medical therapy alone.
Even when obstructive CAD is not identified as the culprit for symptoms, this encounter represents a unique opportunity to assess patients’ cardiovascular prognosis and to implement appropriate preventative therapies for those at risk. Detecting markers of “hidden cardiovascular risk”, such as incidental subclinical coronary atherosclerosis or coronary microvascular dysfunction, may be used to guide the initiation of therapies, including aspirin and statins, which have been shown to mitigate risk of cardiac events.
To this end, radionuclide myocardial perfusion imaging (MPI) is a commonly utilized imaging test for the evaluation of patients with known or suspected CAD. There is currently a large body of evidence supporting the significant role of MPI in the diagnosis and management of patients with suspected ischemic heart disease, whether performed using single photon emission computed tomography (SPECT) and positron emission tomography (PET) technologies. In this report, we aim to provide an overview of diagnostic and prognostic implications of both SPECT and PET MPI in patients with CAD.
Role of PET and SPECT in the Diagnosis of CAD
MPI forms the backbone for diagnosis of CAD and for the assessment of physiological significance of anatomical stenosis. This reliance on MPI is based on decades of prospective observational data highlighting its ability to provide reliable assessment of the presence and severity of CAD, across a wide range of population subgroups. Pooled data from a large number of observational studies suggest the sensitivity and specificity of exercise and pharmacological SPECT MPI to be 87 to 89% and 73 to 75%, respectively [4]. A high normalcy rate of 91% is again indicative of the exceptional diagnostic value of SPECT MPI to detect normal test results among those with a low likelihood of CAD. The diagnostic performance of SPECT MPI is further improved by the use of attenuation correction. While most of the evidence has been on the diagnostic performance of MPI with SPECT, recent data from PET suggests a similarly high diagnostic sensitivity and specificity [5]. Addition of myocardial flow reserve (normal > 2), which is an established advantage of PET, confers a sensitivity and specificity of 96% and 80%, respectively, with an overall accuracy of 92%.
Due to higher spatial resolution and inherent ability to correct for scatter and soft tissue attenuation, PET has better image quality, higher interpretive certainty, and superior sensitivity and specificity for detection of obstructive CAD compared with conventional SPECT [6,7,8]. Moreover, PET MPI exposes patients to significantly lower radiation dose compared with most conventional SPECT protocols due to the inherent characteristics of PET tracers [9]. Such technical advantages have supported growth of cardiac PET in contemporary nuclear laboratories. Despite its diagnostic performance, underestimating the true extent of obstructive epicardial coronary disease (e.g., situations with balanced ischemia), and the inability to determine microvascular dysfunction remain major limitations of stress MPI with conventional SPECT [10, 11]. Several perfusion and non-perfusion markers have been proposed to improve detection of left main or three-vessel coronary disease angiographically when combined with relative perfusion assessment [12]. Additionally, as the use of PET for cardiac applications continues to grow, several PET-derived markers have also been described to improve the diagnostic yield of stress perfusion imaging. A preserved left ventricular ejection fraction reserve with PET—defined as an increase in left ventricular ejection fraction between rest and peak hyperemic stress—is associated with a low likelihood of finding angiographically extensive coronary disease [13•]. Unlike PET-derived LVEF reserve, however, a negative LVEF reserve with SPECT does note carry the same diagnostic utility [14, 15•], underscoring the differences in the timing of gated image acquisition between PET and SPECT. Myocardial blood flow reserve is another PET-derived marker that improves discrimination between patients with and without left main or three-vessel disease, and finding extensive CAD is unlikely in patients with intact coronary vasomotor function [16•, 17]. The field of dynamic SPECT with solid state detector cameras is rapidly evolving, and assessment of MBF could be a reality with SPECT in the near future. Table 1 summarizes the contemporary perfusion and non-perfusion findings with PET and SPECT that suggest the presence of left main or three-vessel CAD.
Novel Radionuclide MPI Protocols for Diagnosis of CAD
Novel MPI protocols for diagnosing CAD and for evaluating acute chest pain in the emergency department (ED) have been recently introduced, that allow for improved laboratory throughput, while maintaining the diagnostic and prognostic value of MPI. Since PET MPI is inherently fast, with very short tracer half-life and a significantly lower radiation exposure, these protocols specifically apply to SPECT MPI.
Stress-First or Stress-Only MPI
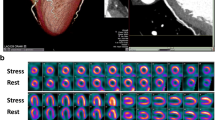
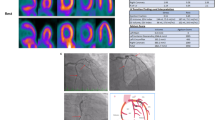
Numerous studies have recently reported the application of stress-first (SF) or stress-only (SO) SPECT MPI for CAD. Chang et al. reported a retrospective analysis of a large cohort of 16,854 patients who had normal SPECT MPI by either a SF or s stress/rest protocol [18]. Approximately 48% of these patients underwent a SF protocol, and over an average follow up period of 4.7 years, there were no significant difference in the adjusted annualized mortality rates among those who underwent SF (2.6%) vs. those who underwent stress/rest imaging (2.9%), p = 0.89. Additionally, there were no difference in outcomes among the two groups by gender, diabetes, known CAD, and Duke Treadmill score. Another study by Duvall et al. studied the prognostic value of a normal SO SPECT MPI protocol among patient with low pre-test risk of CAD [19]. Among 1673 patients with a normal SO SPET MPI, the annualized all-cause mortality was < 1% over an average follow-up period of 40 months. This was similar to a similarly low-risk cohort of patients who underwent conventional rest/stress protocol (p = 0.94). Representative images of patients with an abnormal SO SPECT MPI are shown in Fig. 1.
Abnormal stress-only SPECT myocardial perfusion imaging. Patient was a 58-year-old male, with hypertension and no prior history of coronary artery disease, who complained of with chest pain. Stress-only SPECT myocardial perfusion imaging was performed with 30 mCi of technetium-99 m following treadmill exercise. Patient exercised for 8 min, achieving 85% maximum predicted heart rate, with non-exercise limiting angina. Exercise ECG was negative for ischemia. Gated stress perfusion images show a severe apical perfusion defect, for which the patient was referred for a coronary angiogram that showed critical stenosis of the left anterior descending coronary artery (red arrow). Stress-AC: stress imaging with attenuation correction
MPI for Acute Chest Pain
Accurate triage of patients presenting to the emergency department (ED) with chest pain is essential for risk assessment and adequate patient management. In majority of the patients presenting to the ED, the diagnosis of ACS is not always conclusive, and radionuclide imaging can help in triaging of these patients. In patients presenting to the emergency with chest pain, who are suspected of having an acute coronary syndrome, rest imaging during pain can be employed to detect acute myocardial infarction or ischemia [20]. In this protocol, a standard high-dose of Tc-99 m is injected at rest during chest pain or soon after resolution (within 30 min), with gated SPECT imaging performed within 30–60 min. A completely normal-gated SPECT study virtually excludes the presence of myocardial ischemia. Evaluation of patients with chest pain in the ED with resting SPECT MPI has shown excellent sensitivity of MPI (92%) for myocardial ischemia, which was significantly greater than that conferred by the first troponin level (39%) [21]. A clinical trial of patients with acute chest pain in the ED has highlighted the utility of rest MPI limit hospitalization and facilitate early discharge from the ED. [22] SPECT MPI with Tc-99 m is particularly well suited for acute chest pain imaging, due to its 6-h half-life and no significant redistribution (unlike Thallium-201) allowing perfusion to be “fixed” at the time of injection with flexibility in timing of imaging.
Given the development of ultrafast SPECT systems employing cadmium-zinc-telluride technology, these novel protocols have received significant traction for their ability to improve laboratory throughput by eliminating the need for additional imaging, with significant radiation dose reduction [23]. Even with a standard Anger camera, a SF/SO protocol or acute chest pain imaging has the ability to reduce radiation dose to around 9 mSv considering 30 mCi as a standard high-dose for MPI.
Risk Stratification of with PET and SPECT
Prognostic Value of Perfusion-Based Findings
Several studies have shown the association between the presence and magnitude of myocardial ischemia the risk of cardiac death and non-fatal myocardial infarction, with both PET and SPECT [24, 25•]. An abnormal SPECT MPI portends a 5 to 6-fold increase in the rate of cardiac death and non-fatal myocardial infarction compared with a normal study, whether patients underwent exercise (0.7% vs. 4.3%, respectively) or pharmacologic (1.8% vs. 10%) SPECT MPI [26]. Similar findings were also shown across the spectrum of cardiovascular risk, including patients with or without CAD, diabetes, and chronic kidney disease. Data from the PET multicenter registry showed a twofold increase in the hazards of cardiac death in patients with a mildly abnormal PET MPI (HR 2.3, 95%CI [1.4–3.8]; p = 0.001), and almost a fivefold increase in those who had a severely abnormal PET MPI (HR 4.9, 95%CI [2.5–9.6]; p < 0.0001) [25•].
Incremental Prognostic Value of Coronary Flow Reserve
The ability to quantify myocardial blood flow in absolute terms (ml/min/g) has led to increased interest in cardiac applications of PET imaging technology, not only for the diagnostic benefits mentioned earlier but also for the incremental prognostic value provided with assessment of coronary vasomotor function [27]. Coronary flow reserve (CFR)—defined as the ratio of peak-hyperemic myocardial blood flow to that at rest—is a physiologic marker of the coronary microcirculatory status which can now be routinely assessed in clinical practice in addition to relative perfusion assessment [28•]. PET-derived CFR measurements are feasible with available PET tracers, including 82Rb, N-13 ammonia and O-15 water, and flow measurements obtained can be processed across available software packages [29].
CFR is a stronger predictor of cardiac death compared with peak hyperemic myocardial blood flow [30•, 31]. PET-determined CFR has also been shown in several studies to provide incremental risk stratification and to predict adverse cardiovascular events, independent of age, gender, cardiovascular risk factors, presence, and extent of ischemia (Fig. 2) [32]. Low CFR was associated with a threefold increase in the risk of cardiac death, non-fatal myocardial infarction, or late revascularization among patients who underwent N-13 ammonia PET MPI, irrespective of the presence or absence of relative perfusion defects [33]. Similarly, CFR with 82Rb PET MPI was independently predictive of future major adverse cardiac events with comparable estimates of risk [34,35,36,37]. The prognostic utility of PET-derived CFR was also demonstrated in other patient cohorts, including women, patients with diabetes, chronic kidney disease, and those with elevated markers of myocardial injury [38•, 39•, 40,41,42,43].
Association between PET-derived coronary flow reserve and risk of cardiovascular events. CFR: coronary flow reserve. (Adapted with permission from Springer Nature from: Al Badarin et al. Heart Failure Reviews 2017;22 (4):441–53) [32]
Role of Revascularization in Improving Survival in Patients with Ischemia
Currently, available evidence supports that the burden of ischemia might be beneficial in guiding revascularization decisions in patients undergoing SPECT MPI [44]. Albeit observational and retrospective in nature, those data have provided evidence that patients with greater extent of ischemia (> 10–12.5%) are likely to benefit from referral to early revascularization compared with those with milder degrees of ischemia who fared better with medical therapy. Similar data with PET are available and suggest the threshold at which patients derive survival benefit with revascularization might be lower. This concept is being tested in the randomized International Study of Comparative Health Effectiveness with Medical and Invasive Approaches (ISCHEMIA) trial which has recently finished enrollment, awaiting the release of its results next year.
Prognostic Value of MPI in Special Populations
Elderly Patients
Elderly patients constitute a high-risk population, likely due to age-related atherosclerotic changes as a natural course of disease, and also due to concomitant comorbidities. Studies have shown age to be one of the strongest predictors of myocardial ischemia among the elderly when compared with the burden of other CAD risk factors [45]. Though increasing age has strong association with anatomical CAD [46], MPI has the ability to risk stratify them and has shown a similarly low occurrence of adverse outcomes among elderly with a negative SPECT MPI [47]. The negative predictive value of a normal SPECT MPI among the elderly was reported to provide a similar short-term warranty period for the elderly when compared with younger patients, with the exception of elderly diabetic women. A recent PET MPI study evaluated the prognostic value of PET among the elderly [48]. In this study, there was an increase in all-cause mortality among the elderly (> 65 years) with an increasing burden of ischemia and percent LV stress perfusion defect (Fig. 3). This sequential increase in mortality did not however reach statistical significance among the very elderly (> 85 years) due to a small population size (n = 248). The relatively younger population included patients between 65 and 84 years of age (n = 3085), which per Diamond and Forrester criteria are the subgroup with the highest pretest likelihood of CAD [49], thus underscoring the prognostic value of PET MPI in a high-risk elderly population.
Annual mortality among elderly, stratified by the degree of left ventricular perfusion abnormalities. LV, left ventricular. (Based on data from Erthal et al. [48])
Women
Despite a relatively lower pretest likelihood of CAD and a lower burden of anatomical CAD when compared with men of similar age and symptoms, women tend to have worse prognosis from CAD. Though studies have not consistently reported under testing among women to be the rationale for this discrepancy [50], perfusion imaging among women is more challenging, with a high rate of both false positive and false negative studies. This could be due to breast attenuation and partial volume effect from smaller ventricles in women. Despite these limitations, MPI allows for robust diagnosis and prognosis from CAD among women, with SPECT MPI providing a comparable diagnostic accuracy among women [51]. A meta-analysis has previously reported superior sensitivity of Thallium SPECT MPI vs. ETT alone 78% vs. 61% among women with intermediate to high pretest likelihood of CAD [52]. A large meta-analysis reported an excellent prognostic value of SPECT MPI among women with known or suspected CAD. After a mean follow-up of 3 years, women with known or suspected CAD with a normal SPECT MI experienced a 99% event-free survival (similar to men) [53]. Additionally, an abnormal SPECT MPI provided incremental risk stratification over ETT variables alone, wherein increasing severity of SPECT abnormality were associated with a graded increase in adverse cardiovascular events among women. SPECT MPI better risk stratified women when compared with men: area under the curve for women vs. men 0.84 vs. 0.71, for cardiac death and non-fatal MI. Furthermore, left ventricular ejection fraction, which can be simultaneously obtained from routine gated SPECT MPI acquisitions, further identifies a greater risk of cardiac death and non-fatal MI among women with severe inducible ischemia [54]. Sharir et al. reported a significantly greater 3-year event rate among women with severe ischemia and LVEF < 51% (40%) when compared with those with LVEF > 51% (10%). Furthermore, the greater diagnostic accuracy of PET MPI may confer an additional prognostic advantage and may also reduce the number of indeterminate studies. Studies have shown that additional risk stratification among women can be provided by assessment of MBF, which is a unique advantage of PET MPI. In a cohort of symptomatic patients referred for invasive coronary angiography, an impaired CFR identified a low frequency of multivessel CAD among women when compared with men [40]. Despite a lower pretest probability of CAD and a lower burden of anatomical CAD, women experienced a greater risk of cardiovascular events as compared with men. There is now growing burden of evidence to suggest that a very low CFR may explain the discordance between the lower prevalence of anatomically severe CAD and ischemic events among women, which are likely a function of endothelial dysfunction that can be effectively imaged with PET MPI.
Diabetic Patients
Diabetes is often referred to as CAD equivalent and is considered to portend a high global risk for CAD. Studies have shown that silent myocardial ischemia occurs with high frequency among asymptomatic diabetic patients [55], and this myocardial ischemia on SPECT MPI is associated with a high risk of mortality and adverse cardiovascular events [56]. Though assessment of ischemia by stress MPI is generally not indicated in asymptomatic patients, it may be considered in patients with diabetes (high global CAD risk) as suggested by clinical guidelines [57]. Coronary endothelial dysfunction is often present in patients with diabetes and can be assessed by measuring myocardial blood flow on PET MPI. A recent study evaluated the role of coronary flow reserve (CFR) in risk stratification of patients with and without diabetes [41]. In a large study of 2783 patients (42% with diabetes), Murthy et al. quantified CFR using Rubidium-82 PET MPI. While CFR predicted an increased rate of cardiac death in both diabetics and nondiabetics, diabetic patients with impaired CFR but no known CAD had similar cardiac death rate when compared with nondiabetic patients with CAD (2.8% vs. 2%, p = 0.3). Interestingly, diabetic patients with preserved CFR and no known CAD had a similar cardiac death rate when compared with patients with no known CAD or nondiabetics (0.3% vs. 0.5%, p = 0.65). While relative MPI allows for diagnosis of CAD among diabetic patients, identification of impaired myocardial blood flow with PET MPI can further identify a high-risk subgroup of diabetic patients that have a similarly poor prognosis as compared with those patients with overt CAD.
Post-MPI Resource Utilization
In the evolving era of value-based care, it is exceedingly critical to understand how a diagnostic modality affects downstream resource use and how it informs post-test management decisions by providers. To this end, several studies have evaluated patterns of post-MPI angiography and revascularization with PET and SPECT as an indicator of how these tests alter downstream management of patients who undergo MPI. Merhige and colleagues have shown in a retrospective observational study that the use of PET was associated with lower rates of angiography (0.13% vs. 0.34%, respectively; P < 0.0001) and revascularization (0.06% vs. 0.13%, respectively; P < 0.0001) compared with SPECT in patients with intermediate pretest likelihood, without an increase in rates of short-term cardiovascular events [58]. Another group of investigators, however, have shown discordant results with higher rates of angiography and revascularization at 60 days post-MPI after PET implementation compared with historical controls from a preceding SPECT-only era [59]. Similarly, rates of post-MPI angiography and revascularization after PET were higher in the Study of Myocardial Perfusion and Coronary Anatomy Imaging Roles in Coronary Artery Disease registry (SPARC) that prospectively examined management patterns following a variety of non-invasive modalities used in patients without CAD [60]. The cause for this apparent discordance between results is unclear, but differences in study design and referral patterns between the modalities may potentially be relevant.
Diagnostic and Prognostic Performance of SPECT and PET MPI Compared with Other Imaging Modalities
Several studies have compared the performance of MPI for diagnosing CAD with other modalities. Though exercise treadmill stress alone is recommended for individuals at the lowest risk of CAD, its reported sensitivity and specificity of 68% and 77%, respectively, are significantly inferior to that of MPI [61]. Additionally, studies have shown that ischemia on MPI can be present in patients with no evidence of ECG ischemia on treadmill exercise, and this ischemia on MPI is associated with an increased risk of all-cause mortality [62]. When compared to stress perfusion with cardiac magnetic resonance (CMR), non-attenuation-corrected SPECT MPI has been shown to have similar specificity and positive predictive value for CAD [63]. Additionally, when comparing outcomes, such as invasive angiography rates following stress perfusion imaging, there were no differences in downstream resource utilization and cost with either stress CMR or stress SPECT MPI. Though a direct comparison between PET and CMR has not been performed, a pooled analysis of 37 CMR studies and 15 PET studies reported a superior AUC for PET than CMR for CAD [64]. While the diagnostic accuracy of SPECT MPI and stress echocardiography is generally considered to be comparable, and both have a similarly high negative predictive value, a markedly abnormal SPECT MPI identifies a significantly higher risk of mortality vs. a normal SPECT MPI [65]. While stress echocardiography provides a similar prognostic stratification [66], the magnitude of discrimination of risk between a normal and a markedly abnormal stress echocardiogram is substantially lower. Additionally, vasodilator perfusion stress imaging is not routinely performed with echocardiography in contemporary clinical practice, which is a limitation for patients in whom medicines such as beta-blockers cannot be withheld. Cardiac computed tomography angiography (CCTA), by virtue of anatomical imaging, has excellent sensitivity and comparable specificity for diagnosing CAD when compared with MPI; it does involve administration of iodinated contrast and the need for heart rate control for adequate image quality. The PACIFIC trial reported a superior diagnostic accuracy of PET MPI for detection of hemodynamically significant CAD, when compared with both SPECT and CCTA [67]. In addition, the recently published ReASSESS study reported the diagnostic accuracy of fractional flow reserve derived from computed tomography (FFR-CT) with that of conventional SPECT MPI [68•]. Though the sensitivity of FFR-CT was greater, SPECT MPI had a greater specificity for obstructive CAD, with no difference in overall accuracy. Additionally, the need for CCTA images to be analyzed by a third party to derive the FFR results makes it costlier with additional time and resources required for diagnosis, when there is no additional diagnostic benefit. The PROMISE (Prospective Multicenter Imaging Study for Evaluation of Chest Pain) trial evaluated the occurrence of major adverse cardiovascular events (MACE) among individuals randomized to either cardiac computed tomography angiography (CCTA) first or functional testing first strategy [69•]. Although MPI was not independently evaluated, 2/3rd of the patient in the functional testing arm underwent MPI. Results from the PROMISE trial showed no difference in the occurrence of MACE among those randomized to CCTA vs. functional testing, though more patients randomized to CCTA underwent invasive coronary angiography (ICA).
Conclusions
Radionuclide MPI is a robust technique for the diagnosis of CAD, to guide therapeutic decision making and to determine the risk of adverse outcomes. Though the field of radionuclide MPI is mature, it continues to evolve, which is evident from the application of new imaging protocols, development of more efficient tracers, along with advances in camera technology resulting in reduced radiation exposure. Given that radionuclide MPI can be universally employed, without any contraindications, as opposed to other imaging modalities for CAD, its clinical applicability remains unmatched. A comparison of the clinical effectiveness of various imaging modalities is provided in Table 2.
Abbreviations
- CAD:
-
Coronary artery disease
- MPI:
-
Myocardial perfusion imaging
- PET:
-
Positron emission tomography
- SPECT:
-
Single photon emission computed tomography
- SF:
-
Stress-first
- SO:
-
Stress-only
- ED:
-
Emergency department
- MBF:
-
Myocardial blood flow
- CFR:
-
Coronary flow reserve
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Benjamin EJ, Virani SS, Callaway CW, Chang AR, Cheng S, Chiuve SE, et al. Heart disease and stroke statistics—2018 update: a report from the American Heart Association. Circulation. 2018;137(12):e67–e492. https://doi.org/10.1161/CIR.0000000000000558.
Durand DJ, Lewin JS, Berkowitz SA. Medical-imaging stewardship in the accountable care era. N Engl J Med. 2015;373(18):1691–3. https://doi.org/10.1056/NEJMp1507703.
Shaw LJ, Blankstein R, Jacobs JE, Leipsic JA, Kwong RY, Taqueti VR, et al. Defining quality in cardiovascular imaging: a scientific statement from the American Heart Association. Circ Cardiovasc Imaging. 2017;10(12).
Klocke FJ, Baird MG, Lorell BH, Bateman TM, Messer JV, Berman DS, et al. ACC/AHA/ASNC guidelines for the clinical use of cardiac radionuclide imaging--executive summary: a report of the American College of Cardiology/American Heart Association task force on practice guidelines (ACC/AHA/ASNC Committee to revise the 1995 guidelines for the clinical use of cardiac radionuclide imaging). J Am Coll Cardiol. 2003;42(7):1318–33.
Fiechter M, Ghadri JR, Gebhard C, Fuchs TA, Pazhenkottil AP, Nkoulou RN, et al. Diagnostic value of 13N-ammonia myocardial perfusion PET: added value of myocardial flow reserve. J Nucl Med. 2012;53(8):1230–4.
Mc Ardle BA, Dowsley TF, deKemp RA, Wells GA, Beanlands RS. Does rubidium-82 PET have superior accuracy to SPECT perfusion imaging for the diagnosis of obstructive coronary disease?: A systematic review and meta-analysis. J Am Coll Cardiol. 2012;60(18):1828–37.
Parker MW, Iskandar A, Limone B, Perugini A, Kim H, Jones C, et al. Diagnostic accuracy of cardiac positron emission tomography versus single photon emission computed tomography for coronary artery disease: a bivariate meta-analysis. Circ Cardiovasc Imaging. 2012;5(6):700–7.
Bateman TM, Heller GV, McGhie AI, Friedman JD, Case JA, Bryngelson JR, et al. Diagnostic accuracy of rest/stress ECG-gated Rb-82 myocardial perfusion PET: comparison with ECG-gated Tc-99m sestamibi SPECT. J Nucl Cardiol. 2006;13(1):24–33.
Bateman TM. Advantages and disadvantages of PET and SPECT in a busy clinical practice. J Nucl Cardiol. 2012;19(Suppl 1):S3–11.
Beller GA. Underestimation of coronary artery disease with SPECT perfusion imaging. J Nucl Cardiol. 2008;15(2):151–3.
Berman DS, Kang X, Slomka PJ, Gerlach J, de Yang L, Hayes SW, et al. Underestimation of extent of ischemia by gated SPECT myocardial perfusion imaging in patients with left main coronary artery disease. J Nucl Cardiol. 2007;14(4):521–8.
Nakanishi R, Gransar H, Slomka P, Arsanjani R, Shalev A, Otaki Y, et al. Predictors of high-risk coronary artery disease in subjects with normal SPECT myocardial perfusion imaging. J Nucl Cardiol. 2016;23(3):530–41.
• Dorbala S, Vangala D, Sampson U, Limaye A, Kwong R, Di Carli MF. Value of vasodilator left ventricular ejection fraction reserve in evaluating the magnitude of myocardium at risk and the extent of angiographic coronary artery disease: a 82Rb PET/CT study. J Nucl Med. 2007;48(3):349–58. In this study, the association of a negative LVEF reserve with extensive/severe epicardial coronary disease was shown for the first time.
Ferro A, Petretta M, Acampa W, Fiumara G, Daniele S, Petretta MP, et al. Post-stress left ventricular ejection fraction drop in patients with diabetes: a gated myocardial perfusion imaging study. BMC Cardiovasc Disord. 2013;13:99.
• Gomez J, Golzar Y, Fughhi I, Olusanya A, Doukky R. The significance of post-stress decrease in left ventricular ejection fraction in patients undergoing regadenoson stress gated SPECT myocardial perfusion imaging. J Nucl Cardiol. 2018;25(4):1313–23. This observational study did not show a diagnositc value of the decline in ejection fraction following vasodilator SPECT, for severe coronary artery disease.
• Naya M, Murthy VL, Taqueti VR, Foster CR, Klein J, Garber M, et al. Preserved coronary flow reserve effectively excludes high-risk coronary artery disease on angiography. J Nucl Med. 2014;55(2):248–55. Provides evidence for the utility of assessment of myocardial blood flow for high risk coroanry artery disease.
Ziadi MC, Dekemp RA, Williams K, Guo A, Renaud JM, Chow BJ, et al. Does quantification of myocardial flow reserve using rubidium-82 positron emission tomography facilitate detection of multivessel coronary artery disease? J Nucl Cardiol. 2012;19(4):670–80.
Chang SM, Nabi F, Xu J, Raza U, Mahmarian JJ. Normal stress-only versus standard stress/rest myocardial perfusion imaging: similar patient mortality with reduced radiation exposure. J Am Coll Cardiol. 2010;55(3):221–30.
Duvall WL, Wijetunga MN, Klein TM, Razzouk L, Godbold J, Croft LB, et al. The prognosis of a normal stress-only Tc-99m myocardial perfusion imaging study. J Nucl Cardiol. 2010;17(3):370–7.
Heller GV, Stowers SA, Hendel RC, Herman SD, Daher E, Ahlberg AW, et al. Clinical value of acute rest technetium-99m tetrofosmin tomographic myocardial perfusion imaging in patients with acute chest pain and nondiagnostic electrocardiograms. J Am Coll Cardiol. 1998;31(5):1011–7.
Kontos MC, Jesse RL, Anderson FP, Schmidt KL, Ornato JP, Tatum JL. Comparison of myocardial perfusion imaging and cardiac troponin I in patients admitted to the emergency department with chest pain. Circulation. 1999;99(16):2073–8.
Udelson JE, Beshansky JR, Ballin DS, Feldman JA, Griffith JL, Handler J, et al. Myocardial perfusion imaging for evaluation and triage of patients with suspected acute cardiac ischemia: a randomized controlled trial. JAMA. 2002;288(21):2693–700.
Depuey EG, Mahmarian JJ, Miller TD, Einstein AJ, Hansen CL, Holly TA, et al. Patient-centered imaging. J Nucl Cardiol. 2012;19(2):185–215.
Shaw LJ, Iskandrian AE. Prognostic value of gated myocardial perfusion SPECT. J Nucl Cardiol. 2004;11(2):171–85.
• Dorbala S, Di Carli MF, Beanlands RS, Merhige ME, Williams BA, Veledar E, et al. Prognostic value of stress myocardial perfusion positron emission tomography: results from a multicenter observational registry. J Am Coll Cardiol. 2013;61(2):176–84. Large multicenter observational study showing that ischemia and scar on PET MPI provides incremental prognostic value for cardiac and all-cause mortality.
Navare SM, Mather JF, Shaw LJ, Fowler MS, Heller GV. Comparison of risk stratification with pharmacologic and exercise stress myocardial perfusion imaging: a meta-analysis. J Nucl Cardiol. 2004;11(5):551–61.
Taqueti VR, Di Carli MF. Clinical significance of noninvasive coronary flow reserve assessment in patients with ischemic heart disease. Curr Opin Cardiol. 2016;31(6):662–9.
• Murthy VL, Bateman TM, Beanlands RS, Berman DS, Borges-Neto S, Chareonthaitawee P, et al. Clinical quantification of myocardial blood flow using PET: joint position paper of the SNMMI cardiovascular council and the ASNC. J Nucl Med. 2018;59(2):273–93. This document provides clinicians with an overview of technical aspect and clinical applications of myocardial blood flow.
Nesterov SV, Deshayes E, Sciagra R, Settimo L, Declerck JM, Pan XB, et al. Quantification of myocardial blood flow in absolute terms using (82)Rb PET imaging: the RUBY-10 study. JACC Cardiovasc Imaging. 2014;7(11):1119–27.
• Gupta A, Taqueti VR, van de Hoef TP, Bajaj NS, Bravo PE, Murthy VL, et al. Integrated noninvasive physiological assessment of coronary circulatory function and impact on cardiovascular mortality in patients with stable coronary artery disease. Circulation. 2017;136(24):2325–36. Study showing a stronger predictive value of coronary flow reserve, as opossed to maximal myocardial blood flow, for predicting cardiovascualr death.
Murthy VL, Lee BC, Sitek A, Naya M, Moody J, Polavarapu V, et al. Comparison and prognostic validation of multiple methods of quantification of myocardial blood flow with 82Rb PET. J Nucl Med. 2014;55(12):1952–8.
Al Badarin F, Aljizeeri A, Almasoudi F, Al-Mallah MH. Assessment of myocardial blood flow and coronary flow reserve with positron emission tomography in ischemic heart disease: current state and future directions. Heart Fail Rev. 2017;22(4):441–53.
Herzog BA, Husmann L, Valenta I, Gaemperli O, Siegrist PT, Tay FM, et al. Long-term prognostic value of 13N-ammonia myocardial perfusion positron emission tomography added value of coronary flow reserve. J Am Coll Cardiol. 2009;54(2):150–6.
Fukushima K, Javadi MS, Higuchi T, Lautamaki R, Merrill J, Nekolla SG, et al. Prediction of short-term cardiovascular events using quantification of global myocardial flow reserve in patients referred for clinical 82Rb PET perfusion imaging. J Nucl Med. 2011;52(5):726–32.
Farhad H, Dunet V, Bachelard K, Allenbach G, Kaufmann PA, Prior JO. Added prognostic value of myocardial blood flow quantitation in rubidium-82 positron emission tomography imaging. Eur Heart J Cardiovasc Imaging. 2013;14(12):1203–10.
Murthy VL, Naya M, Foster CR, Hainer J, Gaber M, Di Carli G, et al. Improved cardiac risk assessment with noninvasive measures of coronary flow reserve. Circulation. 2011;124(20):2215–24.
Ziadi MC, Dekemp RA, Williams KA, Guo A, Chow BJ, Renaud JM, et al. Impaired myocardial flow reserve on rubidium-82 positron emission tomography imaging predicts adverse outcomes in patients assessed for myocardial ischemia. J Am Coll Cardiol. 2011;58(7):740–8.
• Taqueti VR, Everett BM, Murthy VL, Gaber M, Foster CR, Hainer J, et al. Interaction of impaired coronary flow reserve and cardiomyocyte injury on adverse cardiovascular outcomes in patients without overt coronary artery disease. Circulation. 2015;131(6):528–35. Coronary flow reserve predicted cardiovascular death and heart failure admission, independent of angiographic coronary artery disease.
• Taqueti VR, Hachamovitch R, Murthy VL, Naya M, Foster CR, Hainer J, et al. Global coronary flow reserve is associated with adverse cardiovascular events independently of luminal angiographic severity and modifies the effect of early revascularization. Circulation. 2015;131(1):19–27. Provides evidence of the link between imparied coronary flow reserve in predictking the risk from coroanry artery disease among women.
Taqueti VR, Shaw LJ, Cook NR, Murthy VL, Shah NR, Foster CR, et al. Excess cardiovascular risk in women relative to men referred for coronary angiography is associated with severely impaired coronary flow reserve, not obstructive disease. Circulation. 2017;135(6):566–77.
Murthy VL, Naya M, Foster CR, Gaber M, Hainer J, Klein J, et al. Association between coronary vascular dysfunction and cardiac mortality in patients with and without diabetes mellitus. Circulation. 2012;126(15):1858–68.
Murthy VL, Naya M, Foster CR, Hainer J, Gaber M, Dorbala S, et al. Coronary vascular dysfunction and prognosis in patients with chronic kidney disease. JACC Cardiovasc Imaging. 2012;5(10):1025–34.
Shah NR, Charytan DM, Murthy VL, Skali Lami H, Veeranna V, Cheezum MK, et al. Prognostic value of coronary flow reserve in patients with dialysis-dependent ESRD. J Am Soc Nephrol. 2016;27(6):1823–9.
Hachamovitch R, Hayes SW, Friedman JD, Cohen I, Berman DS. Comparison of the short-term survival benefit associated with revascularization compared with medical therapy in patients with no prior coronary artery disease undergoing stress myocardial perfusion single photon emission computed tomography. Circulation. 2003;107(23):2900–7.
Malhotra S, Sharma R, Kliner DE, Follansbee WP, Soman P. Relationship between silent myocardial ischemia and coronary artery disease risk factors. J Nucl Cardiol. 2013;20(5):731–8.
Bamberg F, Mayrhofer T, Ferencik M, Bittner DO, Hallett TR, Janjua S, et al. Age- and sex-based resource utilisation and costs in patients with acute chest pain undergoing cardiac CT angiography: pooled evidence from ROMICAT II and ACRIN-PA trials. Eur Radiol. 2018;28(2):851–60.
Hachamovitch R, Hayes S, Friedman JD, Cohen I, Shaw LJ, Germano G, et al. Determinants of risk and its temporal variation in patients with normal stress myocardial perfusion scans: what is the warranty period of a normal scan? J Am Coll Cardiol. 2003;41(8):1329–40.
Erthal F, Hossain A, Dorbala S, Shaw LJ, Di Carli MF, Merhige ME, et al. Is there an age when myocardial perfusion imaging may no longer be prognostically useful? Circ Cardiovasc Imaging. 2018;11(7):e007322.
Diamond GA, Forrester JS. Analysis of probability as an aid in the clinical diagnosis of coronary-artery disease. N Engl J Med. 1979;300(24):1350–8.
Doukky R, Hayes K, Frogge N. Appropriate use criteria for SPECT myocardial perfusion imaging: are they appropriate for women? J Nucl Cardiol. 2016;23(4):695–705.
Sanders GD, Patel MR, Chatterjee R, Ross AK, Bastian LA, Coeytaux RR, et al. Noninvasive technologies for the diagnosis of coronary artery disease in women: future research needs: identification of future research needs from comparative effectiveness review No 58. AHRQ Future Research Needs Papers. Rockville (MD). 2013.
Kwok Y, Kim C, Grady D, Segal M, Redberg R. Meta-analysis of exercise testing to detect coronary artery disease in women. Am J Cardiol. 1999;83(5):660–6.
Metz LD, Beattie M, Hom R, Redberg RF, Grady D, Fleischmann KE. The prognostic value of normal exercise myocardial perfusion imaging and exercise echocardiography: a meta-analysis. J Am Coll Cardiol. 2007;49(2):227–37.
Sharir T, Kang X, Germano G, Bax JJ, Shaw LJ, Gransar H, et al. Prognostic value of poststress left ventricular volume and ejection fraction by gated myocardial perfusion SPECT in women and men: gender-related differences in normal limits and outcomes. J Nucl Cardiol. 2006;13(4):495–506.
Wackers FJ, Young LH, Inzucchi SE, Chyun DA, Davey JA, Barrett EJ, et al. Detection of silent myocardial ischemia in asymptomatic diabetic subjects: the DIAD study. Diabetes Care. 2004;27(8):1954–61.
Zhang L, Li H, Zhang S, Jaacks LM, Li Y, Ji L. Silent myocardial ischemia detected by single photon emission computed tomography (SPECT) and risk of cardiac events among asymptomatic patients with type 2 diabetes: a meta-analysis of prospective studies. J Diabetes Complicat. 2014;28(3):413–8.
Wolk MJ, Bailey SR, Doherty JU, Douglas PS, Hendel RC, Kramer CM, et al. ACCF/AHA/ASE/ASNC/HFSA/HRS/SCAI/SCCT/SCMR/STS 2013 multimodality appropriate use criteria for the detection and risk assessment of stable ischemic heart disease: a report of the American College of Cardiology Foundation appropriate use criteria task force, American Heart Association, American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Computed Tomography, Society for Cardiovascular Magnetic Resonance, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2014;63(4):380–406.
Merhige ME, Breen WJ, Shelton V, Houston T, D'Arcy BJ, Perna AF. Impact of myocardial perfusion imaging with PET and (82)Rb on downstream invasive procedure utilization, costs, and outcomes in coronary disease management. J Nucl Med. 2007;48(7):1069–76.
Knight S, Min DB, Le VT, Meredith KG, Dhar R, Biswas S, et al. Implementation of a cardiac PET stress program: comparison of outcomes to the preceding SPECT era. JCI Insight. 2018;3(9).
Hachamovitch R, Nutter B, Hlatky MA, Shaw LJ, Ridner ML, Dorbala S, et al. Patient management after noninvasive cardiac imaging results from SPARC (study of myocardial perfusion and coronary anatomy imaging roles in coronary artery disease). J Am Coll Cardiol. 2012;59(5):462–74.
Lee TH, Boucher CA. Clinical practice. Noninvasive tests in patients with stable coronary artery disease. N Engl J Med. 2001;344(24):1840–5.
Malhotra S, Sharma R, Khanna M, Soman P. SPECT imaging provides long-term prognostic value over exercise electocardiography alone in patients with a normal baseline electrocardiogram. J Am Coll Crdiol. 2015;65(10S):A1241.
Greenwood JP, Maredia N, Younger JF, Brown JM, Nixon J, Everett CC, et al. Cardiovascular magnetic resonance and single-photon emission computed tomography for diagnosis of coronary heart disease (CE-MARC): a prospective trial. Lancet. 2012;379(9814):453–60.
Jaarsma C, Leiner T, Bekkers SC, Crijns HJ, Wildberger JE, Nagel E, et al. Diagnostic performance of noninvasive myocardial perfusion imaging using single-photon emission computed tomography, cardiac magnetic resonance, and positron emission tomography imaging for the detection of obstructive coronary artery disease: a meta-analysis. J Am Coll Cardiol. 2012;59(19):1719–28.
Hachamovitch R, Berman DS, Kiat H, Cohen I, Cabico JA, Friedman J, et al. Exercise myocardial perfusion SPECT in patients without known coronary artery disease: incremental prognostic value and use in risk stratification. Circulation. 1996;93(5):905–14.
Marwick TH, Case C, Vasey C, Allen S, Short L, Thomas JD. Prediction of mortality by exercise echocardiography: a strategy for combination with the duke treadmill score. Circulation. 2001;103(21):2566–71.
Danad I, Raijmakers PG, Driessen RS, Leipsic J, Raju R, Naoum C, et al. Comparison of coronary CT angiography, SPECT, PET, and hybrid imaging for diagnosis of ischemic heart disease determined by fractional flow reserve. JAMA Cardiol. 2017;2(10):1100–7.
• Sand NPR, Veien KT, Nielsen SS, Norgaard BL, Larsen P, Johansen A, et al. Prospective comparison of FFR derived from coronary CT angiography with SPECT perfusion imaging in stable coronary artery disease: the ReASSESS study. JACC Cardiovasc Imaging. 2018;11(11):1640–50. Comparable accuracy of CT derived FFR versus SPECT MPI for diagnosing coronary artery disease.
• Douglas PS, Hoffmann U, Patel MR, Mark DB, Al-Khalidi HR, Cavanaugh B, et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med. 2015;372(14):1291–300. PROMISE trial comparing cardiac computed angiography first approach to function testing for diagnosis of coronary artery disease.
Funding
Dr. Al Badarin is financially supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health under Award Number T32HL110837. Dr. Malhotra is financially supported by the Becker Fund for Heart Research, Community Foundation of Greater Buffalo.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Firas J. Al Badarin and Saurabh Malhotra declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Disclaimer
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Nuclear Cardiology
Rights and permissions
About this article
Cite this article
Al Badarin, F.J., Malhotra, S. Diagnosis and Prognosis of Coronary Artery Disease with SPECT and PET. Curr Cardiol Rep 21, 57 (2019). https://doi.org/10.1007/s11886-019-1146-4
Published:
DOI: https://doi.org/10.1007/s11886-019-1146-4