Abstract
The tomato leafminer, Tuta absoluta (Lepidoptera: Gelechiidae), is considered to be a major pest that damages tomato (Solanum lycopersicum L; Solanaceae) crops in South American, European, and Mediterranean countries. This insect species is polyphagous (i.e., feeds on many types of food); hence, it could also develop on other cultivated host plants, principally solanaceous plants, such as potato (S. tuberosum L.; Solanaceae) and eggplant (S. melongena L.; Solanaceae). Therefore, we tested the hypothesis that host plant choice by adult T. absoluta is influenced by plant volatile organic compounds and larval host plant experience. One tomato cultivar (cv.) ‘Moneymaker’ and three potato cv. ‘Charlotte’ ‘Bintje,’ and ‘Nicola’ were tested. Using a flying tunnel, we observed that females reared on tomato preferred flying toward tomato and, to a lesser extent, potato cv. ‘Charlotte.’ These preferences might be explained by the high release of terpenes by these two plants. When conducting oviposition choice assays, we found no preference between tomato and potato in the number of eggs laid by females that had been previously reared on either host plant. This study indicates that the host finding behavior of T. absoluta is mediated by solanaceous volatiles, while oviposition behavior appears to depend on additional stimuli. These results provide baseline information for use in the development of new control strategies against T. absoluta using semiochemicals and plant breeding.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plants have evolved a range of strategies to manipulate the behavior of their insect partners, with chemical communication being used as the main strategy in plant–insect interactions (Witzany 2008; Schiestl 2010). Plants emit volatile organic compounds (VOCs) from different above (stems, leaves, flowers, and fruits) and belowground (roots) organs into the atmosphere and soil, respectively. These compounds have important roles in mediating complex interactions between plants and other organisms in the same ecosystem, particularly insects (Maffei 2010; Tholl et al. 2006; Dudareva et al. 2013; Unsicker et al. 2009; Dudareva et al. 2006; Gfeller et al. 2013). VOCs are involved in the attraction of insect pollinators and seed dispersers (Peñuelas and Llusià 2004; Maffei 2010). In addition, they are involved in direct and indirect plant defense against herbivores by repelling the attacking insect (Kessler and Baldwin 2001; De Moraes et al. 2001; Verheggen et al. 2013) and by attracting natural enemies that prey-on or parasitize herbivorous insects (Tholl et al. 2006; Dicke et al. 2003; Kessler and Baldwin 2001). VOCs also protect plants against pathogens (Huang et al. 2012) and are involved in plant–plant communication (Beyaert and Hilker 2014; Dudareva et al. 2013). Herbivorous insects often use these volatiles as cues in long-range orientation to locate plants to feed on or sites for eggs deposition (McCormick et al. 2012; Beyaert and Hilker 2014; Harrewijn et al. 1995).
Consequently, insect management strategies based on plant VOCs involve the development of cultivars that have enhanced direct and/or indirect defense mechanisms (Schnee et al. 2006; Kang et al. 2010; Harmel et al. 2007), the use of polyculture (i.e., agricultural practices using multiple crops in the same place) (Jones and Gillett 2005; George et al. 2013; Stamps and Linit 1997), intercropping (Xie et al. 2012; Zhou et al. 2013), or the extraction of plant volatiles to develop organic pesticides or products that lure insects (Shrivastava et al. 2010; Bleeker et al. 2009; Kaplan 2012). While the chemical composition of VOCs is very complex, the majority of these compounds belong to terpenoid, fatty acid derivative, or phenolic families (Dudareva et al. 2006; Maffei 2010).
Recently, Proffit et al. (2011) showed that terpenoid VOCs are involved in the attraction and oviposition of the tomato leafminer, Tuta absoluta (Lepidoptera: Gelechiidae), indicating the potential of using tomato volatiles for the biocontrol of this pest. New and effective environmentally sound control strategies (e.g., the use of natural enemies, resistant cultivars, or plant volatiles) are required to control T. absoluta, which is considered to be one of the major pests that damage tomato crops in South American, European, and Mediterranean countries (EPPO/OEPP 2005; Desneux et al. 2010, 2011). The tomato leafminer originates from South America, but was accidently introduced to Spain in 2006. Subsequently, this species has spread rapidly north to the Netherlands, south to Senegal, and east to Iran (Pfeiffer et al. 2013; Desneux et al. 2010, 2011). The main host of T. absoluta is tomato (Solanum lycopersicum L.; Solanaceae); however, other cultivated host plants, principally solanaceous species, may also be attacked, including potato (S. tuberosum L.; Solanaceae), eggplant (S. melongena L.; Solanaceae), sweet pepino (S. muricatum L.; Solanaceae), and tobacco (Nicotiana tabacum L.; Solanaceae) (Pereyra and Sánchez 2006; Galarza 1984; Garcia and Espul 1987). The ability of T. absoluta to develop on different potato varieties under laboratory (Pereyra and Sánchez 2006; Caparros Megido et al. 2013) and outdoor (Morrison et al. 2011; Unlu 2012) conditions is now well established. Thus, this pest could probably spread from tomato crops to nearby potato crops. Generalist species, like T. absoluta, are able to detect and interpret a wider range of cues produced by plant volatiles.
However, experience from previous life stages might also be used to facilitate host choice (Bernays 2001; Anderson et al. 2013). For instance, the plant species on which a mother develops may affect the composition of her eggs, which subsequently affects the growth and development of her progeny (Hopkins 1917; Mousseau and Fox 1998). In this study, we investigated the ability of adult T. absoluta to adjust host plant choice by using larval host plant experience and plant VOCs.
Materials and methods
Plant and insect cultures
Tomato (Solanum lycopersicum ‘Moneymaker,’ ‘Pyros F1’) and potato (Solanum tuberosum ‘Charlotte,’ ‘Nicola,’ and ‘Bintje’) plants were cultivated in a greenhouse at 25 ± 5 °C in plastic pots (20 cm diameter × 20 cm height) that were filled with loam (VP113BIO; Peltracom, Belgium) and grown under a 16L:8D (16 h light and 8 h dark) photoperiod. The plants were watered once every 2 days.
In July 2011, 200 third instars of the tomato leafminer, T. absoluta, were collected from a commercial tomato plantation located in Saint-Andiol (France) and were subsequently kept under laboratory conditions at 24 ± 1 °C, 60–70 % relative humidity (RH), and under a 16L:8D photoperiod. Four separate T. absoluta colonies were reared and bred on tomato (‘Pyros F1’) and potato (‘Charlotte,’ ‘Nicola,’ or ‘Bintje’) crops within net cages (each cage 46.5 cm × 46.5 cm × 46.5 cm). Caterpillars were provided with fresh plants three times a week, until pupation. At least five generations were raised on each type of host plant before starting the experiments, to eliminate the effect of food reserves in the eggs or on the emerging larvae (Hasan and Ansari 2011). We maintained a 1:1 female/male ratio in the colonies, to avoid the parthenogenetic reproduction of the females and a consequent decrease in fitness (Caparros Megido et al. 2012).
Collection of plant volatiles
Entire plants (Tomato ‘Moneymaker’ and potato ‘Charlotte,’ ‘Nicola,’ or ‘Bintje,’ 4 weeks old with a 107 BBCH [Biologische Bundesanstalt, Bundessortenamt und CHemische Industrie] code, number of tested plants = 4) were separately placed into 40-l glass jars that were closed with a glass lid. Before volatile collection, each glass and Teflon® pipe used to connect the experimental setup was washed with Extran® solution (MA 01; Merck, Darmstadt, Germany). In addition, each plant pot was wrapped in aluminum foil to avoid contamination from the pot and the soil. A pull air pump (Escort Elf® Pump, MSA, Pittsburgh, PA, USA) provided constant airflow from the bottom to the top of the jar at 1 l/min, after being charcoal-filtered with a carbon trap (Carbon Cap 75, Whatman, Maidstone, England). Volatiles were collected from each plant over a 4-h period, after a 45-min acclimation period. To collect volatile organic compounds (VOCs) released into the plant headspace, a 45-mg HayeSep Q glass trap (80/100 mesh; Hayes Separation Inc., Houston, TX, USA) was connected to the setup with a Teflon® pipe connecting the pump and the glass jar. A second trap was connected inline, to ensure the trap has not reached breaking point.
Before use, all of the traps were washed with 600 μl dichloromethane and 600 μl n-hexane (95 % purity; Sigma-Aldrich, St. Louis, MO, USA). After volatile collection, the traps were immediately eluted with 200 μl n-hexane, after which and n-butyl-benzene (100 ng) was added to each sample as the internal standard. The samples were stored in closed vials wrapped with a Teflon strip at −80 °C before analysis.
Extracts were analyzed by gas chromatography coupled with mass spectrometry (GC–MS) using an Agilent Technologies (Santa Clara, CA, USA) model 6890n GC System/5973 Mass Selective Detector. Aliquots (1 µl) were injected with a splitless injector maintained at 220 °C. The column (HP-5MS, 30 m × 0.25 mm × 0.25 μm) was maintained at 35 °C for 2 min, before being heated to 100 °C at a constant rate of 5 °C/min. The column was then heated to 260 °C at a constant rate of 20 °C/min. The quantification of compounds was realized by comparing their areas with the internal standard and a response factor of 1, using Chemstation software (Agilent Technologies, Palo Alto, CA, USA).
The laboratory was maintained at 20 ± 1 °C, 65 ± 5 % RH, and a 16L:8D photoperiod under cool white light-emitting diode (LED) lights (77 μmol/sqm/s). All conditions were monitored using an automatic datalogger (HOBO RH/TEMP 8 K; Onset Computer Corporation, Bourne, MA, USA).
Chemical identification
The components were identified by comparing the recorded mass spectra with the National Institute of Standards and Technology (NIST) and Wiley spectral databases. Further identification was carried out by calculating non-isothermal Kovats retention indices and by injecting saturated n-alkane standard solution (C7–C30 1,000 μg/ml in hexane, Supelco, Belgium) under the same chromatographic conditions. Identification was confirmed by the injection of available commercial standards (termed STD in Table 1). As chromatographic conditions were identical for the analyses of both the collected compounds and the standards, compound identification was confirmed by comparing their retention data and mass spectra with those of the commercially available reference compounds.
Behavioral assays
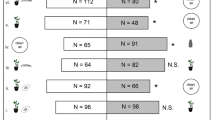
A flying tunnel (232.5 cm × 46.5 cm × 46.5 cm) was used to study the preferences of T. absoluta toward different solanaceous plants (tomato ‘Moneymaker,’ potato ‘Bintje,’ ‘Nicola,’ and ‘Charlotte,’ 4 weeks old with a 107 BBCH code) (Fig. 1). This enclosure was divided into three parts: a central zone for insect release and two zones at opposite sites containing the plants. Sixteen newly emerged T. absoluta adults were placed in the neutral zone of the experimental area at a distance of 116.5 cm from each plant. After 48 h, the number of males, females, and eggs laid (under a binocular magnifier) in each zone of the experimental setup was counted. For the egg counts, only the aerial parts of the plants were checked. Each plant combination was randomly tested five times (for a total of 80 insects tested per dual choice assay), and the same combinations were tested with insects reared on tomato and potato plants. The experiments were carried out under laboratory conditions (20 ± 1 °C, 65 ± 5 % RH, and a 16L:8D photoperiod under cool white LED lights [77 μmol/sqm/s]). These conditions were monitored using an automatic datalogger (HOBO RH/TEMP 8 K; Onset Computer Corporation, Bourne, MA, USA).
Flying tunnel (232.5 cm × 46.5 cm × 46.5 cm) used to study the preferences of T. absoluta toward different solanaceous plants (tomato cv. Moneymaker, potato cv. ‘Bintje,’ ‘Nicola,’ and ‘Charlotte’). This enclosure is divided into three parts, a central zone for the insect release and two zones containing plants, and situated on the two opposite sites
Statistical analyses
Principal component analysis (PCA) was performed on a correlation matrix for the visual comparison of the VOC profiles from the four different solanaceous plant cultivars. One-way ANOVAs were applied to compare the mean quantities of each VOC released by the four tested cultivars. Chi-square tests of independence were used to analyze the behavioral assays, by comparing the choice percentages of males and females and by comparing the number of eggs laid by females on each studied cultivar (the results of the five behavioral assay replicates were pooled together). All tests were performed using Minitab® v.16 software (http://www.minitab.com/fr-FR/default.aspx).
Results
Analysis of plant volatiles
Gas chromatographic analyses performed on plant volatile extracts were used to draft the VOC profile of each plant cultivar (Fig. 2). A total of 33 compounds were found in the headspace of all of the tested plants, of which 31 were identified (Table 1). Compounds commonly associated with the earth’s atmosphere (e.g., toluene and benzene), as well as compounds associated with the analytical system (e.g., siloxanes or phthalates), were excluded from the list (Jansen et al. 2009). In addition, volatile chemicals identified only in one of the four replicates were not included. The terpenoids (principally mono- and sesquiterpenes) were the most abundant of these compounds. Tomato released larger amounts of β-phellandrene (compound number 10 in Table 1, F 3,15 = 9.04, P = 0.002) and α-humulene (24, F 3,15 = 33.51, P < 0.001) compared to the three potato cultivars, and was the only tested plant to release sabinene (2), myrcene (4), δ-2-carene (5), α-phellandrene (6), and α-terpinene (9) (Table 1, Fig. 2). In comparison, potato cv. ‘Charlotte’ released larger amounts of β-caryophyllene (20, F 3,15 = 9.69, P = 0.002), (E)-β-farnesene (22, F 3,15 = 10.29, P < 0.001), germacrene-d (26, F 3,15 = 13.72, P < 0.001), bicyclogermacrene (27, F 3,15 = 12.42, P < 0.001), β-sesquiphellandrene (28, F 3,15 = 4.89, P = 0.019), and germacrene-d-4-ol (31, F 3,15 = 8.62, P = 0.003) compared to all other plants, and released eight compounds that were absent from the other three plants, including 1,4-cineole (8), α-cubebene (17), α-copaene (18), α-gurjunene (19), dodecanol (25), palustrol (30), caryophyllene oxide (32), and an unidentified sesquiterpene (33) (Table 1; Fig. 2). ‘Nicola’ and ‘Bintje’ produced a similar volatile blend, except for an unidentified sesquiterpene (23), which was only found in ‘Bintje.’ The potato cv. ‘Nicola’ did not release any additional compounds compared to the other cultivars. The most abundant compound released in the headspace of tomato and potato cv. ‘Bintje’ was β-phellandrene (54.6 % of the total amount of emitted compounds by this cultivar) and germacrene-d (19.4 %), respectively. β-caryophyllene was the most abundant volatile in potato cv. ‘Nicola’ (25.8 %) and ‘Charlotte’ (31.1 %). Finally, the total amount of VOCs released by tomato was significantly higher compared to the three potato cultivars (F 3,15 = 8.20, P = 0.003).
Chromatograms of VOCs emitted by four solanaceous plants (the number on peaks refers to the compound number given in Table 1, IS internal standard)
PCA was used to visually compare the volatile emissions of the tested plants (Fig. 3). This analysis confirmed the observed differences in the volatile profiles of the tested plants. Tomato and potato cv. ‘Charlotte’ had clearly different VOC emissions compared to the other two plants. In comparison, potato cv. ‘Nicola’ and ‘Bintje’ released a similar volatile blend, as shown on the first (47.8 %) and the second axis (28.2 %) of the PCA.
Behavioral assays
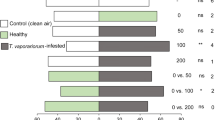
None of the four solanaceous plants attracted adult (males and females) T. absoluta that had been reared on potato, nor males that had been reared on tomato (Table 2). Females reared on tomato made a choice in all tested combinations of host plant and were preferentially attracted by tomato plants, followed by potato cv. ‘Charlotte,’ ‘Nicola,’ and finally ‘Bintje.’ Regardless of the plant variety that the insects were reared on, females laid more eggs on the most attractive plant between the two proposed plants, even if, in some cases, this attraction was not statistically significant (Table 2).
Discussion
Tuta absoluta is considered to be a polyphagous species (Desneux et al. 2010). For example, it is known to develop and reproduce on different tomato and potato cultivars under laboratory conditions, in greenhouses, and on agricultural land (Caparros Megido et al. 2013; Proffit et al. 2011; Unlu 2012; Gharekhani and Salek-Ebrahimi 2013; Ecole et al. 2001; Oliveira et al. 2009; Giustolin et al. 2001). During the plant-host location phase, T. absoluta females detect small differences in the odor blend of tomato cultivars and adapt their oviposition behavior according to plant volatile cues (Proffit et al. 2011). In this study, the attraction of T. absoluta to four different solanaceous plant cultivars was evaluated in a bioassay. This study shows that tomato-reared T. absoluta females were more attracted to tomato plants compared to potato plants (in the order of ‘Charlotte,’ ‘Nicola,’ and ‘Bintje’). This result was expected because tomato is considered to be the ideal host for T. absoluta development (Pereyra and Sánchez 2006; Miranda et al. 1998). In addition, it is known that moth females preferentially oviposit on plant species on which they previously developed (Moreau et al. 2008; Olsson et al. 2006).
Host plant choice is important for Lepidoptera fitness (Thompson 1988; Thompson and Pellmyr 1991). The neonate larvae of many species are unable to move to an alternative food source and are dependent on their mother’s choices (Feeny et al. 1983; Portier 1949). Classically, both males and females are attracted to the odor blends released by plants, which signal food sources, mating sites, or larval host plants (Trona et al. 2010). However, in this study, T. absoluta reared on potato plants did not exhibit any preferences in dual choices with potato and tomato plants. These insects might be simultaneously attracted by the plant on which they were exposed during their entire development (potato) and by the volatile blend of the plant that their ancestors were reared on (tomato) as the appropriate food source.
The larval host plant did not influence host acceptance (adult oviposition) in the current study, because both tomato- and potato-reared females laid similar amounts of eggs on both plants offered during the experiments. It has been long hypothesized that female oviposition preferences are directly influenced by larval experience (Hopkins 1917). However, the results of the current study indicate that while T. absoluta larval experience might influence adult female host searching behavior, it only has a weak influence on oviposition behavior. This finding might be explained by the fact that, during host searching, females only rely on olfactory cues, whereas, during oviposition, females are exposed to a variety of cues, including plant volatiles, contact chemicals, and visual signals, which help determine the characteristics of the plant (e.g., secondary metabolite production, phenology, tissue hardness, and defense mechanisms) to confirm their choice (Olsson et al. 2006; Awmack and Leather 2002; Costa et al. 2009).
Tuta absoluta males that were reared on either tomato or potato plants did not show any preferences toward any of the four tested cultivars. In non-feeding species, like T. absoluta, males primarily use olfaction to detect conspecific female sex pheromones, whereas females primarily rely on olfactory cues to locate and identify appropriate larval substrate for oviposition (Ramaswamy 1988; Olsson et al. 2006). Moreover, while plant odors might influence the male response to female sex pheromones (Trona et al. 2010), males rarely exhibit adaptive behavior in response to host plant volatiles, as shown for females (Anderson et al. 2013).
This study confirmed that the four tested cultivars produced plant volatiles with different compositions and quantities of compounds. These differences might explain the host searching behavior response exhibited by females during the behavioral assays. Tomato was clearly the most preferred plant by T. absoluta females, followed by potato cv. ‘Charlotte,’ ‘Nicolas,’ and ‘Bintje.’ Tomato and potato cv. ‘Charlotte’ appeared to be the most attractive plants in the behavioral assays. The tomato cultivar primarily released a blend of monoterpenes, with large amounts of α-pinene, sabinene, myrcene, δ-2-carene, α-phellandrene, δ-3-carene, β-phellandrene, and one sesquiterpene (β-caryophyllene). In comparison, potato cv. ‘Charlotte’ primarily released a blend of sesquiterpenes, with large amounts of β-caryophyllene, (E)-β-farnesene, germacrene-d, and germacrene-d-4-ol. These findings partially support previous studies on tomato (‘Moneymaker’) (Reisenman et al. 2013; Jansen et al. 2009; Fraser et al. 2003) and potato (‘Surprise,’ ‘Princess,’ ‘Agria,’ and ‘Désirée’) (Bolter et al. 1997; Laothawornkitkul et al. 2010; Karlsson et al. 2009; Gosset et al. 2009) cultivars. Tomato also released a small amount of α-terpinene and α-humulene, while ‘Charlotte’ released different minor compounds, such as 1,4-cineole, α-cubebene, α-copaene, α-gurjunene, dodecanol, bicyclogermacrene, β-sesquiphellandrene, palustrol, and caryophyllene oxide.
Previous studies have already characterized the VOCs released by the tomato cv. ‘Moneymaker,’ in addition to recording the presence of some alcohol and aldehyde compounds, which were not observed in the volatile collection of the current study (Fraser et al. 2003; Jansen et al. 2009; Reisenman et al. 2013). Differences in the volatile profile might be explained by the use of cut plants, rather than entire plants (as in the current study), in the previous studies. It is known that mechanical damage in plants could induce an immediate release of VOCs that are chemically different to those induced by undamaged plants, including alcohols, aldehydes, and acetates (Turlings et al. 1998). In addition, the phenological stage of the plant might also influence the composition of VOCs being released and explain the different volatile blends that were observed (Beyaert and Hilker 2014; Najar-Rodriguez et al. 2010). Published studies on the VOCs released by ‘Charlotte,’ ‘Nicola,’ and ‘Bintje’ are not available. However, in addition to the classical set of terpenes found in potatoes, Karlsson et al. (2009) also identified different alcohols, aldehydes, and benzenoids. These differences in the volatile profile might, again, be explained by the use of cut plants, rather than entire plants. For instance, Bolter et al. (1997) studied entire potato plants (‘Surprise’) and obtained very similar volatile profiles to those presented in our study. These results imply that entire plants provide more representative profiles of the natural volatile blend released by plants compared to cut plants.
Minimal differences were obtained for the volatile profile of the tested plants in the current study compared to previous studies. Twenty-two of the identified compounds were highly released by two most attractive plants (i.e., tomato and potato cv. ‘Charlotte’). These compounds could be considered as major candidates in the research of VOCs that induce the long-range attraction of T. absoluta females. It has already been confirmed that some of the highly released compounds (i.e., sabinene, myrcene, β-caryophyllene, (E)-β-farnesene, germacrene-d, and germacrene-d-4-ol), as well as some of the minor compounds (i.e., α-cubebene and α-copaene), elicit antennal responses in Tecia solanivora, which is a potato pest belonging to the Gelechiidae family (Karlsson et al. 2009).
To confirm the effect of one specific chemical (or blend of the 22 mentioned compounds) on the attraction of T. absoluta, further electrophysiological and behavioral assays are required. Such assays should focus on both the major and minor compounds, because minor constituents in a blend might also play an important role in host plant location (Najar-Rodriguez et al. 2010; Tasin et al. 2011). In conclusion, this study confirmed that T. absoluta females use volatile cues to select and oviposit on certain plants. Thus, while this species is likely to use tomato plants preferentially, pest control schemes should be developed to prevent this species transferring to certain potato cultivars.
References
Adams RP (2007) Identification of essential oil components by gas chromatography/mass spectrometry. Illinois, USA, 804 p
Anderson P, Sadek MM, Larsson M, Hansson BS, Thöming G (2013) Larval host plant experience modulates both mate finding and oviposition choice in a moth. Anim Behav 85(6):1169–1175. doi:10.1016/j.anbehav.2013.03.002
Awmack CS, Leather SR (2002) Host plant quality and fecundity in herbivorous insects. Annu Rev Entomol 47(1):817–844
Azuma H, Thien LB, Toyota M, Asakawa Y, Kawano S (1997) Distribution and differential expression of (E)-4,8-dimethyl-1,3,7-nonatriene in leaf and floral volatiles of Magnolia and Liriodendron taxa. J Chem Ecol 23(11):2467–2478
Bernays EA (2001) Neural limitations in phytophagous insects: Implications for diet breadth and evolution of host affiliation. Annu Rev Entomol 46:703–727
Beyaert I, Hilker M (2014) Plant odour plumes as mediators of plant–insect interactions. Biol Rev 89(1):68–81
Bleeker PM, Diergaarde PJ, Ament K, Guerra J, Weidner M, Schütz S, de Both MTJ, Haring MA, Schuurink RC (2009) The role of specific tomato volatiles in tomato-whitefly interaction. Plant Physiol 151(2):925–935
Bolter CJ, Dicke M, Van Loon JJA, Visser JH, Posthumus MA (1997) Attraction of Colorado potato beetle to herbivore-damaged plants during herbivory and after its termination. J Chem Ecol 23(4):1003–1023
Caparros Megido R, Haubruge E, Verheggen FJ (2012) First evidence of deuterotokous parthenogenesis in the tomato leafminer, Tuta absoluta (Meyrick) (Lep., Gelechiidae). J Pest Sci 85(4):409–412
Caparros Megido R, Brostaux Y, Haubruge E, Verheggen FJ (2013) Propensity of the Tomato Leafminer, Tuta absoluta (Lepidoptera: Gelechiidae), to develop on four potato plant varieties. Am J Potato Res 90(3):255–260
Costa JF, Cosio W, Cardenas M, Yábar E, Gianoli E (2009) Preference of quinoa moth: Eurysacca melanocampta Meyrick (Lepidoptera: Gelechiidae) for two varieties of quinoa (Chenopodium quinoa Willd.) in olfactometry assays. Chil J Agric Res 69(1):71–78
De Moraes CM, Mescher MC, Tumlinson JH (2001) Caterpillar-induced nocturnal plant volatiles repel conspecific females. Nature 410(6828):577–579
Desneux N, Wajnberg E, Wyckhuys KAG, Burgio G, Arpaia S, Narváez-Vasquez CA, González-Cabrera J, Ruescas DC, Tabone E, Frandon J, Pizzol J, Poncet C, Cabello T, Urbaneja A (2010) Biological invasion of European tomato crops by Tuta absoluta: ecology, geographic expansion and prospects for biological control. J Pest Sci 83(3):197–215
Desneux N, Luna MG, Guillemaud T, Urbaneja A (2011) The invasive South American tomato pinworm, Tuta absoluta, continues to spread in Afro-Eurasia and beyond: the new threat to tomato world production. J Pest Sci 84(4):403–408
Dicke M, Van Poecke RMP, De Boer JG (2003) Inducible indirect defence of plants: from mechanisms to ecological functions. Basic Appl Ecol 4(1):27–42
Dudareva N, Negre F, Nagegowda DA, Orlova I (2006) Plant volatiles: recent advances and future perspectives. Crit Rev Plant Sci 25(5):417–440
Dudareva N, Klempien A, Muhlemann JK, Kaplan I (2013) Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol 198(1):16–32
Ecole CC, Picanço MC, Guedes RNC, Brommonschenkel SH (2001) Effect of cropping season and possible compounds involved in the resistance of Lycopersicon hirsutum f. typicum to Tuta absoluta (Meyrick) (Lep., Gelechiidae). J Appl Entomol 125(4):193–200. doi:10.1046/j.1439-0418.2001.00525.x
EPPO/OEPP (2005) Tuta absoluta: Data sheets on quarantine pests. http://www.eppo.int/QUARANTINE/insects/Tuta_absoluta/DS_Tuta_absoluta.pdf. Accessed 18 Aug 2012
Feeny PP, Rosenberry L, Carter M (1983) Chemical aspects of oviposition behavior in butterflies. In: Ahmad S (ed) Host-seeking behavior and mechanisms. Academic Press, London, pp 27–76
Flavornet (2013) Compilation of aroma compounds found in human odor space. http://www.flavornet.org/flavornet.html
Fraser AM, Mechaber WL, Hildebrand JG (2003) Electroantennographic and behavioral responses of the sphinx moth Manduca sexta to host plant headspace volatiles. J Chem Ecol 29(8):1813–1833. doi:10.1023/A:1024898127549
Galarza J (1984) Laboratory assessment of some solanaceous plants as possible food plants of the tomato moth Scrobipalpula absoluta. Idia 421(424):30–32
Garcia MF, Espul JC (1987) Bioecologia de la polilla del tomate (Scrobipalpula absoluta) en Mendoza, Republica Argentina. Revista de Investigaciones Agropecuarias 18:135–146
Gbolade AA, Dzamic AM, Ristic MS, Marin PD (2009) Erratum: Essential oil composition of centratherum punctatum from Nigeria (Chemistry of Natural Compounds (2009) 45:1 (118–119). doi:10.1007/s10600-009-9231-3). Chem Nat Compd 45(2):296
George DR, Collier RH, Whitehouse DM (2013) Can imitation companion planting interfere with host selection by Brassica pest insects? Agric For Entomol 15(1):106–109
Gfeller A, Laloux M, Barsics F, Kati DE, Haubruge E, du Jardin P, Verheggen FJ, Lognay G, Wathelet JP, Fauconnier ML (2013) Characterization of volatile organic compounds emitted by barley (Hordeum vulgare L.) roots and their attractiveness to wireworms. J Chem Ecol 39(8):1129–1139. doi:10.1007/s10886-013-0302-3
Gharekhani GH, Salek-Ebrahimi H (2013) Evaluating the damage of Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae) on some cultivars of tomato under greenhouse condition. Arch Phytopathol Plant Prot. doi:10.1080/03235408.2013.811800
Giustolin TA, Vendramim JD, Alves SB, Vieira SA, Pereira RM (2001) Susceptibility of Tuta absoluta (Meyrick) (Lep., Gelechiidae) reared on two species of Lycopersicon to Bacillus thuringiensis var. kurstaki. J Appl Entomol 125(9–10):551–556
Gosset V, Harmel N, Göbel C, Francis F, Haubruge E, Wathelet JP, Du Jardin P, Feussner I, Fauconnier ML (2009) Attacks by a piercing-sucking insect (Myzus persicae Sultzer) or a chewing insect (Leptinotarsa decemlineata Say) on potato plants (Solanum tuberosum L.) induce differential changes in volatile compound release and oxylipin synthesis. J Exp Bot 60(4):1231–1240. doi:10.1093/jxb/erp015
Harmel N, Almohamad R, Fauconnier ML, Jardin PD, Verheggen F, Marlier M, Haubruge E, Francis F (2007) Role of terpenes from aphid-infested potato on searching and oviposition behavior of Episyrphus balteatus. Insect Sci 14(1):57–63
Harrewijn P, Minks AK, Mollema C (1995) Evolution of plant volatile production in insect-plant relationships. Chemoecology 5–6(2):55–73
Hasan F, Ansari MS (2011) Effects of different brassicaceous host plants on the fitness of Pieris brassicae (L.). Crop Prot 30(7):854–862
Hopkins AD (1917) A discussion of C.G. Hewitt’s paper on ‘Insect Behavior’. J Econ Entomol 10:92–93
Huang M, Sanchez-Moreiras AM, Abel C, Sohrabi R, Lee S, Gershenzon J, Tholl D (2012) The major volatile organic compound emitted from Arabidopsis thaliana flowers, the sesquiterpene (E)-β-caryophyllene, is a defense against a bacterial pathogen. New Phytol 193(4):997–1008. doi:10.1199/tab.0039
Jansen RMC, Hofstee JW, Wildt J, Verstappen FWA, Bouwmeester HJ, Posthumus MA, Van Henten EJ (2009) Health monitoring of plants by their emitted volatiles: trichome damage and cell membrane damage are detectable at greenhouse scale. Ann Appl Biol 154(3):441–452. doi:10.1111/j.1744-7348.2008.00311.x
Jennings W, Shibamoto T (1980) Qualitative analysis of flavour and fragrance volatiles by glass-capillary gas chromatograph. New York
Jones GA, Gillett JL (2005) Intercropping with sunflowers to attract beneficial insects in organic agriculture. Fla Entomol 88(1):91–96
Kang JH, Liu G, Shi F, Jones AD, Beaudry RM, Howe GA (2010) The tomato odorless-2 mutant is defective in trichome-based production of diverse specialized metabolites and broad-spectrum resistance to insect herbivores. Plant Physiol 154(1):262–272
Kaplan I (2012) Attracting carnivorous arthropods with plant volatiles: the future of biocontrol or playing with fire? Biol Control 60(2):77–89
Karlsson MF, Birgersson G, Prado AMC, Bosa F, Bengtsson M, Witzgall P (2009) Plant odor analysis of potato: response of guatemalan moth to above- and belowground potato volatiles. J Agric Food Chem 57(13):5903–5909
Kessler A, Baldwin IT (2001) Defensive function of herbivore-induced plant volatile emissions in nature. Science 291(5511):2141–2144
Laothawornkitkul J, Jansen RMC, Smid HM, Bouwmeester HJ, Muller J, van Bruggen AHC (2010) Volatile organic compounds as a diagnostic marker of late blight infected potato plants: a pilot study. Crop Prot 29(8):872–878
Maffei ME (2010) Sites of synthesis, biochemistry and functional role of plant volatiles. S Afr J Bot 76(4):612–631
McCormick AC, Unsicker SB, Gershenzon J (2012) The specificity of herbivore-induced plant volatiles in attracting herbivore enemies. Trends Plant Sci 17(5):303–310
Miranda MMM, Picanço M, Zanuncio JC, Guedes RNC (1998) Ecological life table of Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Biocontrol Sci Technol 8(4):597–606
Moreau J, Rahme J, Benrey B, Thiery D (2008) Larval host plant origin modifies the adult oviposition preference of the female European grapevine moth Lobesia botrana. Naturwissenschaften 95(4):317–324. doi:10.1007/s00114-007-0332-1
Morrison N, Walker A, Baxter I, Harveay-Samuel T, Hdidi A, Alphey L (2011) Investigating the potential of genetic control as part of an IPM approach for Tuta absoluta. In: International symposium on management of Tuta absoluta, Agadir (Marocco), 2011. http://archives.eppo.int/MEETINGS/2011_conferences/tuta/38_Morrison/index.html
Mousseau TA, Fox CW (1998) The adaptive significance of maternal effects. Trends Ecol Evol 13(10):403–407. doi:10.1016/S0169-5347(98)01472-4
Najar-Rodriguez AJ, Galizia CG, Stierle J, Dorn S (2010) Behavioral and neurophysiological responses of an insect to changing ratios of constituents in host plant-derived volatile mixtures. J Exp Biol 213(19):3388–3397. doi:10.1242/jeb.046284
Oliveira FA, da Silva DJH, Leite GLD, Jham GN, Picanço M (2009) Resistance of 57 greenhouse-grown accessions of Lycopersicon esculentum and three cultivars to Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Sci Hortic 119(2):182–187. doi:10.1016/j.scienta.2008.07.012
Olsson POC, Anderbrant O, Löfstedt C (2006) Experience influences oviposition behaviour in two pyralid moths, Ephestia cautella and Plodia interpunctella. Anim Behav 72(3):545–551. doi:10.1016/j.anbehav.2005.10.023
Peñuelas J, Llusià J (2004) Plant VOC emissions: making use of the unavoidable. Trends Ecol Evol 19(8):402–404
Pereyra PC, Sánchez NE (2006) Effect of two solanaceous plants on developmental and population parameters of the tomato leaf miner, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Neotrop Entomol 35(5):671–676
Pfeiffer DG, Muniappan R, Sall D, Diatta P, Diongue A, Dieng EO (2013) First record of Tuta absoluta (Lepidoptera: Gelechiidae) in Senegal. Fla Entomol 96(2):661–662
Pherobase (2013) Database of pheromones and semiochemicals. http://pherobase.com
Portier P (1949) La biologie des Lépidoptères, vol 23. Paris
Proffit M, Birgersson G, Bengtsson M, Reis R Jr, Witzgall P, Lima E (2011) Attraction and oviposition of Tuta absoluta females in response to tomato leaf volatiles. J Chem Ecol 37(6):565–574
Ramaswamy SB (1988) Host finding by moths: sensory modalities and behaviours. J Insect Physiol 34(3):235–249. doi:10.1016/0022-1910(88)90054-6
Reisenman CE, Riffell JA, Duffy K, Pesque A, Mikles D, Goodwin B (2013) Species-specific effects of herbivory on the oviposition behavior of the moth Manduca sexta. J Chem Ecol 39(1):76–89. doi:10.1007/s10886-012-0228-1
Schiestl FP (2010) The evolution of floral scent and insect chemical communication. Ecol Lett 13(5):643–656. doi:10.1111/j.1461-0248.2010.01451.x
Schnee C, Köllner TG, Held M, Turlings TCJ, Gershenzon J, Degenhardt J (2006) The products of a single maize sesquiterpene synthase form a volatile defense signal that attracts natural enemies of maize herbivores. Proc Natl Acad Sci USA 103(4):1129–1134
Shrivastava G, Rogers M, Wszelaki A, Panthee DR, Chen F (2010) Plant volatiles-based insect pest management in organic farming. Crit Rev Plant Sci 29(2):123–133
Stamps WT, Linit MJ (1997) Plant diversity and arthropod communities: implications for temperate agroforestry. Agrofor Syst 39(1):73–89
Tasin M, Betta E, Carlin S, Gasperi F, Mattivi F, Pertot I (2011) Volatiles that encode host-plant quality in the grapevine moth. Phytochemistry 72(16):1999–2005. doi:10.1016/j.phytochem.2011.06.006
Tholl D, Boland W, Hansel A, Loreto F, Röse USR, Schnitzler JP (2006) Practical approaches to plant volatile analysis. Plant J 45(4):540–560. doi:10.1029/2001JD000576
Thompson JN (1988) Evolutionary ecology of the relationship between oviposition preference and performance of offspring in phytophagous insects. Entomol Exp Appl 47(1):3–14
Thompson JN, Pellmyr O (1991) Evolution of oviposition behavior and host preference in Lepidoptera. Annu Rev Entomol 36:65–89
Trona F, Casado D, Coracini M, Bengtsson M, Ioriatti C, Witzgall P (2010) Flight tunnel response of codling moth Cydia pomonella to blends of codlemone, codlemone antagonists and pear ester. Physiol Entomol 35(3):249–254. doi:10.1111/j.1365-3032.2010.00737.x
Turlings TCJ, Tumlinson JH, Heath RR, Proveaux AT, Doolittle RE (1991) Isolation and identification of allelochemicals that attract the larval parasitoid, Cotesia marginiventris (Cresson), to the microhabitat of one of its hosts. J Chem Ecol 17(11):2235–2251
Turlings TCJ, Lengwiler UB, Bernasconi ML, Wechsler D (1998) Timing of induced volatile emissions in maize seedlings. Planta 207(1):146–152. doi:10.1007/s004250050466
Unlu L (2012) Potato: a new host plant of Tuta absoluta Povolny (Lepidoptera: Gelechiidae) in Turkey. Pak J Zool 44(4):1183–1184
Unsicker SB, Kunert G, Gershenzon J (2009) Protective perfumes: the role of vegetative volatiles in plant defense against herbivores. Curr Opin Plant Biol 12(4):479–485
Verheggen FJ, Haubruge E, De Moraes CM, Mescher MC (2013) Aphid responses to volatile cues from turnip plants (Brassica rapa) infested with phloem-feeding and chewing herbivores. Arthropod-Plant Interact 7(5):567–577. doi:10.1007/s11829-013-9272-1
Witzany G (2008) The biosemiotics of plant communication. Am J Semiot 24(1–3):39–56
Xie HC, Chen JL, Cheng DF, Zhou HB, Sun JR, Liu Y, Francis F (2012) Impact of wheatmung bean intercropping on English grain aphid (Hemiptera: Aphididae) populations and its natural enemy. J Econ Entomol 105(3):854–859. doi:10.1603/EC11214
Zhou HB, Chen JL, Liu Y, Francis F, Haubruge E, Bragard C, Sun JR, Cheng DF (2013) Influence of garlic intercropping or active emitted volatiles in releasers on aphid and related beneficial in wheat fields in China. J Integr Agric 12(3):467–473. doi:10.1016/S2095-3119(13)60247-6
Acknowledgments
This research was funded by the Service Public de Wallonie (SPW–DGO3, Project D31-1263).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jarmo Holopainen.
Rights and permissions
About this article
Cite this article
Caparros Megido, R., De Backer, L., Ettaïb, R. et al. Role of larval host plant experience and solanaceous plant volatile emissions in Tuta absoluta (Lepidoptera: Gelechiidae) host finding behavior. Arthropod-Plant Interactions 8, 293–304 (2014). https://doi.org/10.1007/s11829-014-9315-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-014-9315-2