Abstract
The tomato leafminer Tuta absoluta (Lepidoptera: Gelechiidae) is a devastating pest of cultivated tomato Solanum lycopersicum throughout South and Central America and Europe. We aimed to characterize the behavioral mechanisms and the chemical cues involved in host selection of T. absoluta females by chemical analysis of tomato leaf volatiles, wind tunnel attraction assays, and oviposition bioassays. Tomato leaf odor elicited in mated females upwind orientation flight followed by landing as well as egg-laying, demonstrating the essential role of plant volatiles in T. absoluta host-finding behavior. In wind tunnel and oviposition choice experiments, T. absoluta females significantly preferred tomato S. lycopersicum over wild tomato Solanum habrochaites, which is resistant to larval feeding. This indicates that leaf volatiles provide information on the suitability of plants as larval hosts. Mated females also discriminated three cultivars of S. lycopersicum according to their volatile profiles. Headspace collections from leaves of these three cultivars contained large amounts of β-phellandrene, followed by limonene, 2-carene, and (E)-β-caryophyllene, which together accounted for more than 70% of tomato foliage headspace. Most leaf volatiles were released by all three cultivars, but they showed significant differences with respect to the presence of a few minor compounds and blend proportion. This is an initial study of the volatile signatures that mediate attraction and oviposition of tomato leafminer T. absoluta in response to its main host, tomato.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plants release a variety of volatile organic compounds that play multiple roles in interactions with other plants and animals (Dudareva et al., 2004; Knudsen et al., 2006; Pichersky et al., 2006). Insect herbivores exploit these volatiles to locate their host plants at a distance, for feeding, mating, and egg-laying (Linn et al., 2003; Bengtsson et al., 2006; Tasin et al., 2006; Pinero and Dorn, 2009; Cha et al., 2008; Schmidt-Busser et al., 2009; Sole et al., 2010). In addition to olfaction, vision and contact chemoreception (taste) could play a role at a short distance (Renwick and Chew, 1994; Calatayud et al., 2008; Pontes et al., 2010). Knowledge of the chemicals and mechanisms that mediate host plant location by insect herbivores is essential for our understanding of plant—insect relationships, and also will lead to the development of novel tools for insect management (Szendrei and Rodriguez-Saona, 2010; Witzgall et al., 2010).
Plant volatiles are products of diverse metabolic pathways, but many are derived from the isoprenoid or terpenoid pathways (Sacchettini and Poulter, 1997; Degenhardt et al., 2009). Solanaceous plants, like tomato, produce a suite of terpenes that likely serve as defense agents against herbivores (Kennedy, 2003; Bleeker et al., 2009; Kang et al., 2010). The constitutive headspace of undamaged plants varies with genotype, phenological stage, and environmental conditions. It is conceivable that insects use the volatile signals that correlate with this variation to distinguish the most suitable hosts (Bengtsson et al., 2001; Vallat and Dorn, 2005; Karlsson et al., 2009).
The tomato leafminer Tuta absoluta (Meyrick) (Lepidoptera, Gelechiidae) feeds on several solanaceous species, and preferentially on tomato Solanum lycopersicum (Siqueira et al., 2000). It is a serious threat to commercial tomato production in South and Central America. Since its introduction, it has become an economically important insect in Europe (Torres et al., 2001; Desneux et al., 2010). It is anticipated that T. absoluta will even spread from Central America into Mexico and the USA. Control of T. absoluta relies on multiple insecticide sprays, but resistance to these chemicals is growing (Siqueira et al. 2000; Lietti et al., 2005). Synthetic sex pheromones are used to monitor population levels, but also for control by mass trapping of males in greenhouses (Salas, 2004; Witzgall et al., 2010).
Tomato plants are infested at any developmental stage, with females ovipositing preferentially on leaves. After hatching, larvae feed on leaf parenchyma tissue, on tender portions of the stems (especially axillary buds), and in developing and mature fruit, causing bud drop, fruit malformation, fruit rot, and a drastic reduction in leaf area (Torres et al., 2001). No tomato cultivars are entirely resistant to T. absoluta, but not all cultivars are equally susceptible (Bogorni et al., 2003; Oliveira et al., 2009). In addition, the wild tomato species S. habrochaites (syn. Lycopersicon hirsutum) is more resistant to T. absoluta and several other tomato herbivores than S. lycopersicum (Leite et al., 2001; Kennedy, 2003).
The aim of this study was to determine the role of tomato volatiles in host finding and oviposition behavior of tomato leafminer T. absoluta. We identified the volatiles released from leaves of three different cultivars of tomato S. lycopersicum and investigated the question whether tomato leafminer uses these volatiles for the discrimination of suitable larval hosts.
Methods and Materials
Insect Rearing
A laboratory colony of tomato leafminer T. absoluta originated from pupae and larvae collected in a commercial tomato plantation located in Viçosa, Minas Gerais (Brazil). The colony was maintained at 24 ± 1°C, 70 ± 10% r.h., and a 12:12 h L:D photoperiod. Adults were fed with sugar water and provided with tomato (cv. Aromata) leaves for egg-laying. Leaf stems were cut and placed through a hole in the transparent plastic lid of a 500-ml recipient containing water and a nutrient solution. Larvae were provided with fresh leaves every 2nd d, and rolls of corrugated paper were added for pupation. Pupae were sexed and placed in plastic cages for eclosion (100 × 40 × 40 cm).
Plant Material
For behavioral experiments and volatile analysis, three tomato Solanum lycopersicum Mill. (Solanaceae) cultivars were used: cv. Santa Clara and cv. Carmen, which are known to be highly susceptible, and cv. Aromata, which is less susceptible (Svalöf-Weibull Trädgård AB, Hammenhög, Sweden) (Bogorni et al., 2003; Oliveira et al., 2009; Lima et al. unpublished data). In addition, the wild tomato species S. habrochaites, which is resistant to T. absoluta, was used for wind tunnel and oviposition bioassays. Tomato seeds were pre-cultivated in rock wool cubes in the greenhouse for 4 week (r.h.: 75%, light intensity: 200 μmolm−2 s - 1). Plants were transplanted to a hydroponic system and fertilized daily with a nutrient solution. Standard values (in mM) for the composition of the nutrient solution were: 6.0 Ca(NO3)2, 8.0 KNO3, 5.0 MgSO4, 1.0 NH4NO3, 2.3 KH2PO4 and for micronutrients: 0.4 Fe–EDTA, 0.01 MnSO4, 0.025 H3BO3, 0.005 ZnSO4, 0.00075 CuCl2,2 H2O, 0.0005 Na2MoO4,and pH 5.8 ± 0.1.
Flight Behavior Bioassay
Female attraction to plants was tested in a wind tunnel (flight section 63 × 63 × 200 cm; Tasin et al., 2006). Filtered air (24 cylinders with active charcoal, 14.5 × 32.5 cm, Camfil, Trosa, Sweden) was blown into the tunnel by a centrifugal fan (Fischbach GmbH, Neunkirchen, Germany) at 25 cm/s. Outcoming air was aspirated by another fan and cleaned by two additional sets of charcoal filters. The flight section was lit diffusely from the side at 10 lux, and the room was kept at 23 ± 2°C and 40–60% r. h. Cut tomato plants (ca. 35 cm high) were placed into vials with water in the center of the upwind end of the flight tunnel. Two h before the end of the photophase, females were placed individually into cylindrical plastic containers. They were kept until testing in the wind tunnel room. Moth behavior was scored for upwind oriented flight in the center of the wind tunnel (over at least 60 cm from the downwind end) and for approaching tomato plants. By using this method, we tested (1) the attraction of 2-d-old mated (N = 190) and virgin (N = 150) females to S. lycopersicum cv. Aromata, and (2) the attraction of mated female (N = 150) to S. lycopersicum cv. Santa Clara and S. habrochaites.
Oviposition Bioassays
Experiments were done with 2-d-old mated females during 24 hr. Females were individually kept in a glass tube (15.0 × 3.0 cm) covered on both sides with a plastic film. To keep the females in one side of the tube, it was divided in half with a nylon grid. The female and the tomato leaf were put on the same (N = 48) or different sides of the tube (N = 48). This experiment was done with the three tomato S. lycopersicum cultivars. After 24 h, the eggs laid on the glass wall and the nylon grid were counted.
In choice tests, mated females were kept in a plastic container (12 × 10 cm) either with one leaf of cv. Aromata, cv. Carmen and cv. Santa Clara (N = 81 females), or with one leaf of Santa Clara and wild tomato S. habrochaites (N = 37 females), and the leaves were scored for eggs after 24 h.
Plant Volatile Analysis
Freshly cut plants of the three cultivars of S. lycopersicum were confined separately in a 2-L glass jar that was closed with a ground-glass fitting. The cut end of the branch was held in a 10-ml vial of water. A charcoal-filtered airstream was pulled over the plant material from the bottom to the top of the jar, and over a 35-mg Super Q trap (80/100 mesh; Alltech, Deerfield, IL, USA) which was held between plugs of glass-wool in a 4 × 40 mm glass tube. Before use, traps were rinsed sequentially with 3 ml methanol, ether, and redistilled hexane, after 15 min treatments in ultrasonic baths in ether and hexane, respectively. The air flow was 150 ml/min, exchanging the headspace in the jar 4.5 times/h. Collections were done for 24 h, at 20–22°C and 10–30 lux. The charcoal filter for incoming air and the Super Q trap for outcoming air were connected with glass fittings to the jar. All glassware was heated to 350°C for 10 h before use. After volatile collections, traps were extracted immediately with 0.5 ml hexane (redistilled; LabScan, Malmö, Sweden). Sample volumes were reduced to 50–60 μl, at ambient temperature in Francke-vials with an elongated tip (5 cm × 2 mm i.d.). Samples were stored in sealed glass capillary tubes at −18°C (Bengtsson et al., 2001).
Odor samples were analyzed on a gas-chromatograph coupled with a mass-spectrometer (GC-MS). Volatile compounds in trap eluents were injected in aliquots of 2 μl into a Hewlett- Packard 5970 B MS instrument, with electron impact ionization at 70 eV, and interfaced to an HP 5890 GC (Hewlett- Packard, Palo Alto, CA, USA). The GC was equipped with a fused silica capillary column (30 m × 0.25 mm) coated with DB-wax (df = 0.25 μm; J&W Scientific, Folsom, CA, USA) using helium as carrier gas (35 cm/s). The temperature program was from 35°C (hold 5 min) at 8°C/min to 230°C (hold 5 min). The Kovats retention index (KI) of each compound was calculated. Tentative identification of compounds was based on comparison of matching of mass spectra with the NIST 98 MS and Wiley databases and a reference library built by measuring commercially available standards, and comparison of KIs on similar columns. Identification of some components was confirmed by comparison with mass spectra and GC retention data with those of standards.
Statistical Analysis
All statistical analysis was done using R (version 2.8.0; R Development Core Team, www.R-project.org) following the method of Crawley (2005).
The effect of either mating status or test plant (S. lycopersicum and S. habrochaites), on the proportion of females, grouped per day, that were attracted to and that landed on the plant was tested with a Generalized Linear Modelling (GLM) under quasibinomial distribution.
Similarly, the effect of contact with the host plant and the effect of the tomato cultivar (Carmen, Santa Clara, Aromata) on the total number of eggs laid in the non-choice bioassays were tested using a GLM under quasipoisson distribution. In the choice experiments, the effect of S. lycopersicum cultivars or tomato species on the proportion of eggs laid on leaves was tested using a GLM with quasibinomial distribution. In all analyses, a full model was fitted, from which terms were deleted in a stepwise fashion to obtain the minimal adequate model (MAM). Significance (P < 0.05) was assessed by testing the change in deviance after the removal of a term from the model. Differences among treatment levels were examined by contrast analysis. Candidate similarity levels were amalgamated to compose a new model, which was then compared with the previous un-amalgamated model. The new, simplified model was accepted if it did not differ from the previous, more complex model. Amalgamation proceeded until all treatment levels were checked. Models were checked by residual analyses, and eventually corrected for overdispersion as well as for the correctness of the assumed distribution (Crawley, 2005).
Headspace composition was compared among samples by multivariate analyses using R with Vegan and labdsv packages (Oksanen et al., 2008; Roberts, 2010). Non-metric multidimensional scaling (NMDS) was used to detect similarities among samples (using amounts relative to total peak area). Data were square-root transformed and standardized before calculating Bray–Curtis similarities (Bray and Curtis, 1957). In oviposition bioassays, T. absoluta prefers cv. Santa Clara and cv. Carmen over cv. Aromata with no difference between Santa Clara and Carmen. Thus, based on T. absoluta oviposition preference, variation in odor composition among tomato cultivars and between cv. Aromata vs. cvs. Santa Clara and Carmen (pooled), was tested for significance using permutational multivariate analysis of variance (permanova) with the Bray–Curtis dissimilarity measurement and 10000 permutations (McArdle and Anderson, 2001).
Differences in the occurrence and abundance of compounds in the headspace of the cv. Aromata vs. the cvs. Santa Clara/Carmen were determined according to the method of Dufrêne and Legendre (1997) by calculating ‘indicator compound’ (e.g., Proffit et al., 2009). For each compound, an observed indicator value (IV) was calculated. The deviation of the observed IV from a random distribution of IVs was tested using a Monte Carlo test with 10000 randomizations. Indicator compounds have an observed IV significantly different from the random IV, and designate the most characteristic compounds in headspace samples of a group, either because of their occurrence in different samples and/or because of their relative abundance.
Results
Wind Tunnel Experiments
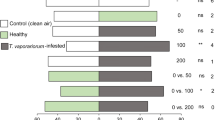
Mated tomato leafminer T. absoluta females flew upwind over 180 cm towards tomato plants (45% of the females tested) and landed (42%) on these plants. In contrast, unmated females showed no attraction response to tomato (N = 17; for flight, F 1,15 = 192.63, P < 0.001; for landing, F 1,15 = 122.09, P < 0.001; Fig. 1).
Proportion of mated and virgin females of tomato leafminer Tuta absoluta flying upwind and landing on tomato leaves (Solanum lycopersicum cv. Aromata) in the wind tunnel (mean ± SE/day). For both behavioral responses, differences between mated (N = 190) and virgin females (N = 150) are significant (N = 17 days, GLM, P < 0.05)
Oviposition Experiments with Tomato Cultivars
In non-choice tests, the cultivar had a significant effect on oviposition (df = 2, χ 2 = 132.71, P < 0.001; Fig. 2a and b). Tomato leafminer females laid more eggs in response to cvs. Santa Clara and Carmen as compared cv. Aromata (df = 1, χ 2 = 110.39, P < 0.001 and df = 1, χ 2 = 95.75, P < 0.001), both with and without leaf contact: the interaction between leaf contact and cultivar was not significant (df = 2, χ 2 = 3.38, P = 0.82).
Oviposition rate (mean ± SE) of mated tomato leafminer Tuta absoluta females on three tomato cultivars Aromata, Santa Clara, and Carmen, in (a) a non-choice experiment without leaf contact (N = 48), (b) a non-choice experiment with leaf contact (N = 48), and (c) a choice experiment with leaf contact (N = 81). For each experiment, treatments represented by filled and empty bars are significantly different (GLM with contrast, P < 0.05)
A choice test (Fig. 2c) confirmed the effect of the cultivar on oviposition (F 1,78 32.08, P < 0.001). Here again, the females preferred Santa Clara and Carmen over Aromata (F 1,78 = 22.96, P < 0.001 and F 1,78 = 36.11, P < 0.001, respectively). In both non-choice and choice tests, there was no difference in the preference between Santa Clara and Carmen (df = 1, χ 2 = 0.56, P = 0.79, and F 1,78 = 1.53, P = 0.22, respectively). Females laid significantly more eggs when in contact with tomato leaves of S. lycopersicum cv. Aromata, Carmen and Santa Clara, compared to when they were exposed only to leaf headspace (df = 1, χ 2 = 766.55, P < 0.001; Fig. 2).
Comparison Between Cultivated and Wild Tomato
Both in the wind tunnel as well as in oviposition bioassays, T. absoluta preferred cultivated tomato S. lycopersicum over wild tomato S. habrochaites (F 1,20 = 109.30, P < 0.001; F 1,72 = 46.42, P < 0.001, respectively). The proportion of insects preferring S. lycopersicum was similar in both experiments (Fig. 3).
Upwind attraction (N = 22 days, mean ± SE/day) and oviposition choice tests (N = 37, mean ± SE) of mated tomato leafminer Tuta absoluta with cultivated Solanum lycopersicum cv. Santa Clara and wild tomato Solanum habrochaites. The effect of the tomato species was significant in both experiments (GLM, P < 0.05)
Chemical Analysis of Tomato Headspace
We found 52 volatile compounds in the headspace collection of cv. Aromata, Santa Clara, and Carmen (Table 1). Terpenoid compounds largely dominated the tomato leaf headspace. Beta-phellandrene was the most abundant compound by far and represented more than half of the total volatile emission. In addition, limonene, 2-carene, as well as (E)-β-caryophyllene were major compounds in tomato headspace of the three cultivars.
Although most volatiles were released by all three cultivars (Table 1), leaf headspace composition differed among them (Permanova F 2,19 = 5.95, P < 0.001; Fig. 4), headspace differed between Aromata and Santa Clara/Carmen (Permanova F 1,20 = 2.23, P = 0.001). According to a Monte Carlo test, 26 of the 52 compounds were good indicators of the leaf headspace of either Aromata or Santa Clara/Carmen (Table 1), most of which are minor terpenoid compounds, with a mean leaf headspace proportion of < 1%. Aromata did not produce extra compounds compared to the two other cultivars.
Non-metric multidimensional scaling (NMDS) plot visualizing the samples of three tomato cultivars, Aromata, Santa Clara, and Carmen based on the similarity (Bray-Curtis distance) of their leaf headspace composition, rotated by principal component, so that the variance of points is maximized on the first dimension (stress = 0.15). Samples are grouped (dashed lines) according to tomato leafminer Tuta absoluta oviposition preference. The centroids of cv. Aromata vs. the preferred cvs. Carmen/Santa Clara are the focal points of the solid lines
In contrast, Carmen and Santa Clara released 6 compounds that were absent from the headspace of Aromata: carvacrol, α-copaene, (Z)-limonene oxide, an unidentified monoterpene (unknown 4), an unidentified sesquiterpene (unknown 11), and one unindentified compound (unknown 18). In addition, 9 other compounds that were characteristic for Santa Clara/Carmen were present in smaller amounts in Aromata leaf headspace. On the other hand, 10 compounds were characteristic for Aromata and more abundant than in Santa Clara and Carmen headspace (Table 1).
Discussion
Combined headspace analysis and behavioral assays show that tomato leaf volatiles are essential cues for host finding and oviposition in tomato leafminer T. absoluta (Figs. 1, 2 and 3). The females detected small variations in volatile signatures of the different tomato cultivars (Table 1, Fig. 4), and the ensueing behavioral response (Figs. 2 and 3) matched the suitability of tomato cultivars and species as larval hosts (Leite et al., 1999, 2001; Oliveira et al., 2009).
Long Range Attraction Cues
Mated T. absoluta females responded to tomato leaves by upwind oriented flight, followed by landing (Fig. 1). Host volatiles have been shown to elicit attraction of several other moths (Tasin et al., 2006; Arab et al., 2007; Masante-Roca et al., 2007; Knudsen et al., 2008; Pinero and Dorn, 2009; Sole et al., 2010).
Only mated females were attracted to tomato leaves (Fig. 1). In most herbivorous insects, host-searching behavior is based not only on the perception of suitable olfactory information, but also on the insect’s internal state. Physiological changes such as ovarian maturation, egg production, and mating status can influence the response of insect females to host cues (Thompson and Pellmyr, 1991; Yan et al., 1999; Masante-Roca et al., 2007). For example, mated grapevine moth Lobesia botrana or Egyptian cotton leafworm Spodoptera littoralis females are attracted to their larval host plants, not unmated females (Masante-Roca et al., 2007; Saveer et al. unpublished data).
Oviposition Cues
Tomato leaf volatiles alone elicited an oviposition response in T. absoluta (Fig. 2a). Moreover, ovipositing females discriminated among different tomato cultivars and between cultivated and wild tomato on the basis of volatile cues (Figs. 2 and 3). However, leaf contact significantly increased the number of eggs laid (Fig. 2a and b), and leaf surface morphology and chemistry is accordingly of importance for oviposition. Tuta absoluta females lay eggs uniformly on the under- and upper-side of leaves that are covered with trichomes that provide chemical and mechanical stimuli (Torres et al., 2001).
Tomato leafminer females lay their eggs directly on leaves (Torres et al., 2001). Accordingly, there is no spatial separation between the oviposition site and plant part that releases the chemical signal that elicits host-finding. In contrast, two closely related species from potato, Tecia solanivora and Phthorimaea operculella, oviposit in the soil close to the plant, rather than on leaves or stems (Fenemore, 1988; Horgan et al., 2007; Karlsson et al., 2009).
Variation of Volatile Profile Among Tomato Cultivars
The volatile profile of tomato leaves of the cultivars Aromata, Santa Clara, and Carmen was dominated by monoterpenes, in particular β-phellandrene (major compound), limonene, and 2-carene, and the sesquiterpene (E)-β-caryophyllene (Table 1), which is in accordance with headspace analysis of other tomato cultivars (Buttery et al., 1987; Zhang et al., 2008). Nonetheless, overall headspace composition of Aromata differed significantly from Santa Clara and Carmen (Fig. 4), due to differences in blend proportions of minor compounds and due to the absence of several compounds, mostly terpenes, in Aromata (Table 1). Our oviposition bioassays shows that T. absoluta females were able to detect this inter-cultivar variation in leaf headspace, since they laid more eggs in response to headspace from Santa Clara and Carmen than Aromata (Fig. 2).
Tomato Leaf Headspace as an Indicator of Host Suitability
Herbivorous moths are attracted to host plant volatiles at specific blend ratios in laboratory assays (Fraser et al., 2003; Tasin et al., 2006). However, more recent studies in Oriental fruit moth Cydia molesta and grapevine moth Lobesia botrana show that females are attracted to plant volatile blends that show considerable variation with respect to proportions and composition (Tasin et al., 2007, 2010; Najar-Rodriguez et al. 2010). A response to a broad spectrum of plant volatile blends could represent an adaptation to differences in volatile signatures from genotypes and to variation during phenological changes and would accordingly facilitate host finding even in specialist herbivores.
This is contrasted by the differential response of tomato leafminer to comparatively small variations in tomato headspace components and proportions (Table 1, Fig. 2). A tentative explanation for discrimination of minor blend variations is that the females are less tolerant and respond strongly to those compounds that are direct cues of host plant suitability. Trichome-borne terpenoids, which account for much of the difference between the cultivars Aromata, Santa Clara, and Carmen (Table 1) have been suggested to influence host plant selection and fitness in tomato insects (Bleeker et al., 2009, 2011; Kang et al., 2010).
The idea that volatile cues provide information of host suitability at a distance is further substantiated by attraction and egg-laying choice tests with wild tomato S. habrochaites, where females clearly preferred cultivated over wild tomato (Fig. 3). Leaf headspace of wild tomato S. habrochaites was dominated by β-ocimene, β-myrcene, and undecan-2-one (Smith et al., 1996; Bleeker et al., 2009). Glandular trichomes of wild tomato also contain many sesquiterpenes, including caryophyllene, curcumene, elemene, humulene, and zingiberene, that play a role in tomato resistance against beet armyworm and whitefly (Eigenbrode et al., 1994; Smith et al., 1996; van der Hoeven et al., 2000; Bleeker et al., 2009, 2011). Resistance of wild tomato to tomato leafminer has been attributed to tridecan-2-one, undecan-2-one, and zingiberene, which have not been found in the three cultivars studied here (Leite et al., 1999; Azevedo et al., 2003).
This initial study of the attraction and oviposition response of tomato leafminer T. absoluta in response to its primary host tomato will support the identification of the odor template that encodes host recognition, and the development of new methods to control this important tomato pest worldwide. Tomato volatiles can be used directly for trapping mated females, and the differential behavioral response to tomato cultivars indicates the potential of plant breeding for improved resistance. While a complete description of the bioactive compounds is a considerable undertaking, the wind tunnel bioassay is already available for efficient screening of plant genotypes.
References
Arab, A., Trigo, J. R., Lourenção, A. L., Peixoto, A. M., Ramos, F., and Bento, J. M. S. 2007. Differential attractiveness of potato tuber volatiles to Phthorimaea operculella (Gelechiidae) and the predator Orius insidiosus (Anthocoridae). J. Chem. Ecol. 33:1845–1855.
Azevedo, S.M., Faria, M.V., Maluf, W.R., De Oliveira, A.C.B., and De Freitas, J.A. 2003. Zingiberene-mediated resistance to the South American tomato pinworm derived from Lycopersicon hirsutum var. hirsutum. Euphytica 134:347–351.
Bengtsson, M., Backman, A. C., Liblikas, I., Ramirez, M. I., Borg-Karlson, A. K., Ansebo, L., Anderson, P., Lofqvist, J., and Witzgall, P. 2001. Plant odor analysis of apple: antennal response of codling moth females to apple volatiles during phenological development. J. Agric. Food Chem. 49:3736–3741.
Bengtsson, M., Jaastad, G., Knudsen, G., Kobro, S., Backman, A. C., Pettersson, E., and Witzgall, P. 2006. Plant volatiles mediate attraction to host and non-host plant in apple fruit moth, Argyresthia conjugella. Entomol. Exp. Appl. 118:77–85.
Bleeker, P. M., Diergaarde, P. J., Ament, K., Guerra, J., Weidner, M., Schutz, S., De Both, M. T. J., Haring, M. A., and Schuurink, R. C. 2009. The role of specific tomato volatiles in tomato-whitefly interaction. Plant Physiol. 151:925–935.
Bleeker, P. M., Diergaarde, P., Ament, K., Schütz, S., Johne, B., Dijkink, J., Hiemstra, H., de Gelder, R., de Both, M. T. J., Sabelis, M. W., Haring, M. A., and Schuurink, R. C. 2011. Tomato-produced 7-epizingiberene and R-curcumene act as repellents to whiteflies. Phytochemistry 72:68–73.
Bogorni, P. C., Silva, R. A., and Carvalho G. S. 2003. Leaf mesophyll consumption by Tuta absoluta (Meyrick, 1971) in three cultivars of Lycopersicon esculentum Mill. Cienc. Rural 33:7–11.
Bray, J. R. and Curtis, J. T. 1957. An ordination of the upland forest communities of Southern Wisconsin. Ecol. Monogr. 27:326–349.
Buttery, R. G., Ling, L. C., and Light, D. M. 1987. Tomato leaf volatile aroma components. J. Agric. Food Chem. 35:1039–1042.
Calatayud, P. A., Guenego, H., Ahuya, P., Wanjoya, A., Ru, B. L., Silvain, J. F., and Frerot, B. 2008. Flight and oviposition behaviour of the African stem borer, Busseola fusca, on various host plant species. Entomol. Exp. Appl. 129:348–355.
Cha, D. H., Nojima, S., Hesler, S. P., Zhang, A., Linn, C. E., Roelofs, W. L., and Loeb, G. M. 2008. Identification and field evaluation of grape shoot volatiles attractive to female grape berry moth (Paralobesia viteana). J. Chem. Ecol. 34:1180–1189.
Crawley, M. J. C. 2005. The R Book. Wiley, Chichester.
Degenhardt, J., Kollner, T. G., and Gershenzon, J. 2009. Monoterpene and sesquiterpene synthases and the origin of terpene skeletal diversity in plants. Phytochemistry 70:1621–1637.
Desneux, N., Wajnberg, E., Wyckhuys, K. A. G., Burgio, G., Arpaia, S., Narvaez-Vasquez, C. A., Gonzalez-Cabrera, J., Ruescas, D. C., Tabone, E., Frandon, J., Pizzol, J., Poncet, C., Cabello, T., and Urbaneja, A. 2010. Biological invasion of European tomato crops by Tuta absoluta: ecology, geographic expansion and prospects for biological control. J. Pest Sci. 83:197–215.
Dudareva, N., Pichersky, E., and Gershenzon, J. 2004. Biochemistry of plant volatiles. Plant Physiol. 135:1893–1902.
Dufrêne, M. and Legendre, P. 1997. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecol. Monogr. 67:345–366.
Eigenbrode, S. D., Trumble, J. T., Millar, J. G., and White, K. K. 1994. Topical toxicity of tomato sesquiterpenes to the beet armyworm and the role of these compounds in resistance derived from an accession of Lycopersicon hirsutum f. typicum. J. Agric. Food Chem. 42:807–810.
Fenemore, P. G. 1988. Host-plant location and selection by adult potato moth, Phthorimaea operculella (Lepidoptera, Gelechiidae) - a review. J. Insect Physiol. 34:175–177.
Fraser, A. M., Mechaber, W. L., and Hildebrand, J. G. 2003. Electroantennographic and behavioral responses of the sphinx moth Manduca sexta to host plant headspace volatiles. J. Chem. Ecol. 29:1813–1833.
Horgan, F. G., Quiring, D. T., Lagnaoui, A., and Pelletier, Y. 2007. Variable responses of tuber moth to the leaf trichomes of wild potatoes. Entomol. Exper. Appl. 125:1–12.
Kang, J. H., Liu, G. H., Shi, F., Jones, A. D., Beaudry, R. M., and Howe, G. A. 2010. The tomato odorless-2 mutant is defective in trichome-based production of diverse specialized metabolites and broad-spectrum resistance to insect herbivores. Plant Physiol. 154:262–272.
Karlsson, M. F., Birgersson, G., Prado, A. M. C., Bosa, F., Bengtsson, M., and Witzgall, P. 2009. Plant odor analysis of potato: response of Guatemalan moth to above- and belowground potato volatiles. J. Agric. Food Chem. 57:5903–5909.
Kennedy, G. G. 2003. Tomato, pests, parasitoids, and predators: Tritrophic interactions involving the genus Lycopersicon. Annu. Rev. Entomol. 48:51–72.
Knudsen, J. T., Eriksson, R., Gershenzon, J., and Ståhl, B. 2006. Diversity and distribution of floral scent. Bot. Rev. 72:1–120.
Knudsen, G. K., Bengtsson, M., Kobro, S., Jaastad, G., Hofsvang, T., and Witzgall, P. 2008. Discrepancy in laboratory and field attraction of apple fruit moth Argyresthia conjugella to host plant volatiles. Physiol. Entomol. 33:1–6.
Leite, G. L. D., Picanco, M., Della, L., and Moreira, M. D. 1999. Role of canopy height in the resistance of Lycopersicon hirsutum f. glabratum to Tuta absoluta (Lep.; Gelechiidae). J. Appl. Entomol. 123:459–463.
Leite, G. L. D., Picanco, M., Guedes, R. N. C., and Zanuncio, J. C. 2001. Role of plant age in the resistance of Lycopersicon hirsutum f. glabratum to the tomato leafminer Tuta absoluta (Lepidoptera: Gelechiidae). Sc. Hortic. 89:103–113.
Lietti, M. M. M., Botto, E., and Alzogaray, R. A. 2005. Insecticide resistance in Argentine populations of Tuta absoluta (Meyrick) (Lepidoptera : Gelechiidae). Neotrop. Entomol. 34:113–119.
Linn, C. E., Feder, J. L., Nojima, S., Dambroski, H. R., Berlocher, S. H., and Roelofs, W. L. 2003. Fruit odor discrimination and sympatric host race formation in Rhagoletis. Proc. Natl. Acad. Sci. USA 100:11490–11493.
Masante-Roca, I., Anton, S., Delbac, L., Dufour, M. C., and Gadenne, C. 2007. Attraction of the grapevine moth to host and non-host plant parts in the wind tunnel: effects of plant phenology, sex, and mating status. Entomol. Exp. Appl. 122:239–245.
McArdle, B. H. and Anderson, M. J. 2001. Fitting multivariate models to community data: A comment on distance-based redundancy analysis. Ecology 82:290–297.
Najar-Rodriguez, A. J., Galizia, C. G., Stierle, J., and Dorn, S. 2010. Behavioral and neurophysiological responses of an insect to changing ratios of constituents in host plant-derived volatile mixtures. J. Exp. Biol. 213:3388–3397.
Oksanen, J., Kindt, R., Legendre, P., O'Hara, B., Gavin, L., Simpson, G. L., and Stevens, M.H.H. 2008. Vegan: community ecology package. R package version 1.11-4. http://cran.r-project.org/, http://vegan.r-forge.r-project.org/.
Oliveira, F. A., Da Silva, D. J. H., Leite, G. L. D., Jham, G. N., and Picanco, M. 2009. Resistance of 57 greenhouse-grown accessions of Lycopersicon esculentum and three cultivars to Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Scientia Horticulturae 119:182–187.
Pichersky, E., Sharkey, T. D., and Gershenzon, J. 2006. Plant volatiles: a lack of function or a lack of knowledge? Tr. Plant Sci. 11:421–421.
Pinero, J. C. and Dorn, S. 2009. Response of female oriental fruit moth to volatiles from apple and peach trees at three phenological stages. Entomol. Exp. Appl. 131:67–74.
Pontes, W. J. T., Lima, E. R., Cunha, E. G., De Andrade, P. M. T., Lobo, A. P., and Barros, R. 2010. Physical and chemical cues affect oviposition by Neoleucinodes elegantalis. Physiol. Entomol. 35:134–139.
Proffit, M., Chen, C., Soler, C., Bessiere, J. M., Schatz, B., and Hossaert-Mckey, M. 2009. Can chemical signals, responsible for mutualistic partner encounter, promote the specific exploitation of nursery pollination mutualisms? The case of figs and fig wasps. Entomol. Exp. Appl. 131:46–57.
Renwick, J. A. A. and Chew, F. S. 1994. Oviposition behavior in Lepidoptera. Annu. Rev. Entomol. 39:377–400.
Roberts, D. W. 2010. Laboratory for dynamic synthetic vegephenonenology (LabDSV) version 1.4-1. http://ecology.msu.montana.edu/labdsv/R
Sacchettini, J. C. and Poulter, C. D. 1997. Biochemistry - Creating isoprenoid diversity. Science 277:1788–1789.
Salas, J. 2004. Capture of Tuta absoluta (Lepidoptera: Gelechiidae) in traps baited with its sex pheromone. Rev. Colomb. Entomol. 30:75–78.
Schmidt-Busser, D., von Arx, M., and Guerin, P. M. 2009. Host plant volatiles serve to increase the response of male European grape berry moths, Eupoecilia ambiguella, to their sex pheromone. J. Comp. Physiol. A 195:853–864.
Siqueira, H. A. A., Guedes, R. N. C., and Picanco, M. C. 2000. Insecticide resistance in populations of Tuta absoluta (Lepidoptera: Gelechiidae). Agricult. Forest Entomol. 2:147–153.
Smith, R. M., Marshall, J. A., Davey, M. R., Lowe, K. C., and Power, J. B. 1996. Comparison of volatiles and waxes in leaves of genetically engineered tomatoes. Phytochemistry 43:753–758.
Sole, J., Sans, A., Riba, M., and Guerrero, A. 2010. Behavioural and electrophysiological responses of the European corn borer Ostrinia nubilalis to host-plant volatiles and related chemicals. Physiol. Entomol. 35:354–363.
Szendrei, Z. and Rodriguez-Saona, C. 2010. A meta-analysis of insect pest behavioral manipulation with plant volatiles. Entomol. Exp. Appl. 134:201–210.
Tasin, M., Backman, A. C., Bengtsson, M., Ioriatti, C., and Witzgall, P. 2006. Essential host plant cues in the grapevine moth. Naturwissenschaften 93:141–144.
Tasin, M., Bäckman, A.-C., Coracini, M., Casado, D., Ioriatti, C., and Witzgall, P. 2007. Synergism and redundancy in a plant volatile blend attracting grapevine moth females. Phytochemistry 68:203–209.
Tasin, M., Bäckman, A.-C., Anfora, G., Carlin, S., Ioriatti, C., and Witzgall, P. 2010. Attraction of female grapevine moth to common and specific olfactory cues from 2 host plants. Chem. Senses 35:57–64.
Thompson, J. N. and Pellmyr, O. 1991. Evolution of oviposition behavior and host preference in Lepidoptera. Annu. Rev. Entomol. 36:65–89.
Torres, J. B., Faria, C. A., Evangelista, W. S., and Pratissoli, D. 2001. Within-plant distribution of the leaf miner Tuta absoluta (Meyrick) immatures in processing tomatoes, with notes on plant phenology. Int. J. Pest Manag. 47:173–178.
Vallat, A., and Dorn, S. 2005. Changes in volatile emissions from apple trees and associated response of adult female codling moths over the fruit-growing season. J. Agric. Food Chem. 53:4083–4090.
Van der Hoeven, R. S., Monforte, A. J., Breeden, D., Tanksley, S. D., and Steffens, J. C. 2000. Genetic control and evolution of sesquiterpene biosynthesis in Lycopersicon esculentum and L. hirsutum. Plant Cell 12:2283–2294.
Witzgall, P., Kirsch, P., and Cork, A. 2010. Sex pheromones and their impact on pest management. J. Chem. Ecol. 36:80–100.
Yan, F. M., Bengtsson, M., and Witzgall, P. 1999. Behavioral response of female codling moths, Cydia pomonella, to apple volatiles. J. Chem. Ecol. 25:1343–1351.
Zhang, P. Y., Chen, K. S., He, P. Q., Liu, S. H., and Jiang, W. F. 2008. Effects of crop development on the emission of volatiles in leaves of Lycopersicon esculentum and its inhibitory activity to Botrytis cinerea and Fusarium oxysporum. J. Integr. Plant Biol. 50:84–91.
Acknowledgements
This study was supported by the Swedish University of Agricultural Sciences (SLU) and by the Swedish Foundation for International Cooperation in Research and Higher Education (STINT) and EL was supported by FAPEMIG.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Proffit, M., Birgersson, G., Bengtsson, M. et al. Attraction and Oviposition of Tuta absoluta Females in Response to Tomato Leaf Volatiles. J Chem Ecol 37, 565–574 (2011). https://doi.org/10.1007/s10886-011-9961-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-011-9961-0