Abstract
Concerns related to higher levels of acrylamide in processed carbohydrate-rich foods, especially in fried potato products, are well known. This article provides updates on various aspects of acrylamide in processed potato products including mechanisms of acrylamide formation and health risks due to its intake. Levels of reducing sugars in potatoes are considered as a main factor contributing towards the formation of acrylamide in processed potato products. Useful approaches in lowering the levels of reducing sugars such as use of suitable varieties, storage methods, storage temperature and duration of storage are described and discussed. Importance and practical utility of various steps before and during the processing that can contribute in reducing the final concentration of acrylamide are highlighted. Progress made and present status of potato processing industry in India are part of this article. The article describes varietal improvement and spread of short-term and long-term storage technologies in India and their contribution towards round the year availability of processing-grade potatoes to the processing industries and how all this has helped in achieving reduced levels of acrylamide in chips and French fries. Outcome and implications of cold-induced sweetening tolerance in potatoes are presented along with other management practices and strategies that can lower the acrylamide levels in processed potato products. Future lines of work have been suggested to make the consumption of fried potato products safer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acrylamide (C3H5NO, molar mass 71.08 dalton, other names; ethylenecarboxamide, prop-2-enamide or 2-propenamidemes) is an odourless crystalline solid which is highly soluble in water (2004 g/l of water at 25 °C) and organic solvents. Mostly, acrylamide is used to synthesize polyacrylamide, which is used as water-soluble thickeners for waste water treatment, gel electrophoresis (SDS-PAGE), paper making, ore processing and in preparation of permanent press fabrics. Some acrylamide is used in the manufacturing of dyes and other monomers. The categorization of acrylamide as a potential carcinogen (Group 2A) by IARC (1994) has been a cause of concern. Researchers of Swedish National Food Administration (SNFA) and the University of Stockholm discovered that acrylamide is present in high concentrations in carbohydrate-rich foods such as French fries and potato crisps (Rosen and Hellenas 2002; Tareke et al. 2002). EC (2002) and WHO (2002) have listed acrylamide as Category 2 of mutagens. This finding has lead to a huge surge in interest and research in this area. Studies have established that acrylamide is a byproduct and it is formed when carbohydrate-rich foods are fried, baked or roasted at high temperatures above 120 °C (Pedreschi 2007). Among different food products, highest levels of acrylamide have been reported in French fries, potato chips and other potato products where frying, deep-fat frying or oven-cooking is involved (Tareke et al. 2000; Weisshaar 2004). Tareke et al. (2002) pointed out that although human exposure to acrylamide is primarily occupational, in view of above finding general public may also be exposed to acrylamide by consuming heated starchy foods including potato processed products in their diet. This article provides update with respect to the status of international research on various aspects of acrylamide in processed potato products with special reference to the work done in India. Article elaborates how the varietal improvement and spread of short-term and long-term storage technologies in India have contributed towards round the year availability of potato for table and processing purposes and also how this has affected the issue of acrylamide in processed potato products. Importance and scope of reducing acrylamide content by different strategies during pre-processing and processing of potatoes are also highlighted. Lastly, implication of cold-induced sweetening (CIS) tolerance is discussed along with future lines of research work for making the consumption of fried potato products safer.
Mechanism of acrylamide formation
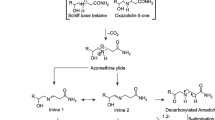
Maillard reaction (Maillard 1912) is mainly responsible for the formation of acrylamide in starchy foods (Mottram et al. 2002; Everts 2012; NTP 2013). This reaction involves condensation of the amino acid (primarily asparagine) with reducing sugars, i.e. fructose or glucose on heating at temperatures more than 120 °C (Fig. 1) (Stadler et al. 2002; Mottram et al. 2002; Everts 2012). During frying, heat transferred from the hot oil increases the internal energy of potato strip till the surface temperature reaches more than the boiling point of water (103–104 °C). After this, a large amount of incoming energy is used for moisture evaporation. At temperatures of <150 °C, the energy input to potato strip is limited which prevents its surface from reaching the temperatures above 120 °C within 10 min of frying. However, when the oil temperature is high (>170 °C) the energy input becomes sufficient for rapid moisture evaporation and as a result the surface temperature increases to 120 °C or more, a favourable condition for the formation of acrylamide (Gokmen et al. 2006a).
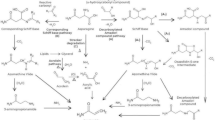
The acrylamide synthesis pathway is not completely clear, but it has been shown that acrylamide formation has close relationship with the Maillard reaction (Stefano and Avellone 2014). Hence, the Maillard reaction is considered as the principal mechanism behind the formation of acrylamide in fried potatoes. In the Maillard reaction, melanoidins are the main end products. They are brown polymers and have significant effect on colour, quality, taste and acceptability of food products (Gokmen and Senyuva 2006a; NTP 2013). Thus the Maillard reaction leads to browning and formation of acrylamide in processed food products. Acrylamide formation begins at temperatures of about 120 °C and it increases sharply at temperatures between 160 and 180 °C (Mottram et al. 2002; Matthaus et al. 2004). Besides potato, carbohydrate-rich foods on frying generate acrylamide (Tareke et al. 2002). In addition to Maillard reaction, decarboxylated asparagine (3-aminopropionamide), when heated, can also generate acrylamide in the absence of reducing sugars (Zyzak et al. 2003; Zamora et al. 2009). Further, degradation of amino acids with ammonia can produce acrylamide via thermal decomposition. Other amino acids like aspartic acid, cysteine and glutamine are also known to produce low amounts of acrylamide (Sohn and Ho 1995; Becalski et al. 2004). Acrylamide is reported to be present in number of food products which are prepared by either frying or roasting of any commodity rich in starch or carbohydrates. Levels of acrylamide present in different types of processed potato products are presented in Table 1. Potatoes are high in asparagine and reducing sugars, and are commonly prepared for consumption by frying or baking. All these factors contribute to the formation of acrylamide in processed potato products. This also explains the reason for relatively high levels of acrylamide in fried/heated potato products (Stefano and Avellone 2014). In a study carried out on the fried snacks of Southern India, it has been reported that potato chips have mean acrylamide content of 1456.5 μg kg−1 (range 82.0–4245.5 μg kg−1) (Shamla and Nisha 2014). In a number of studies it has been found that there is a positive correlation between acrylamide content in chips and chip colour score (Kotsiou et al. 2013; Elmore et al. 2015). Lower the colour score lower will be the content of acrylamide in the chips or French fries.
Health risks due to acrylamide intake
Most of the acrylamide taken in with food is absorbed in humans. Since acrylamide molecule is small and hydrophilic (highly soluble in water; 2004 g/l of water at 25 °C), it passively diffuses throughout the body (Friedman 2003). Acrylamide is absorbed through unbroken skin, mucous membranes of gastrointestinal tract and lungs (Klaassen et al. 1986; Merck 1989). Due to this, almost all tissues are theoretically targets of acrylamide-mediated carcinogenesis (Janneke 2008). On 4 June, 2015 European Food Safety Authority (EFSA) published its first full risk assessment of acrylamide in food (EFSA 2015). Experts from EFSA’s Panel on Contaminants in the Food Chain (CONTAM) reconfirmed that acrylamide in the food items has the potential to increase the risk of developing cancer in all age groups (EFSA 2015). Acrylamide is reported to cause cancer (Shipp et al. 2006) and damage to nervous system in humans and animals is one of the non-cancerous effects linked to acrylamide (LoPachin and Lehning 1994; LoPachin 2004). Details of mechanisms involved in acrylamide-mediated neurotoxicity are described by LoPachin and Gavin (2012). According to them, acrylamide causes a direct inhibitory effect on presynaptic function and it also reduces neurotransmission at central and peripheral synapses by disturbing signalling pathways. This results in cumulative neurotoxicity in exposed humans and laboratory animals. Acrylamide is a soft electrophile that can preferentially form adducts with soft nucleophilic sites on macromolecules. Further, acrylamide is a type-2 alkene that can interact with endogenous unsaturated aldehydes leading to development of diabetes, atherosclerosis, Alzheimer’s diseases and other pathogenic conditions due to cellular oxidative stress (reviewed by LoPachin and Gavin 2012). Acrylamide is also a developmental and reproductive toxin (Dearfield et al. 1988; Costa et al. 1992; Manson et al. 2005) with carcinogenic and mutagenic properties, experimentally shown in mammalian system under in vitro and in vivo conditions (Dearfield et al. 1995; Friedman et al. 1995; EU Scientific Committee Report 2002; EFSA 2005, 2015). Acrylamide causes increase in micronucleus formation in peripheral blood cells. In rat and human systems, it causes cancer when administered orally and increases tumours in the ovary, breast, bladder, prostate, lung, gastrointestinal tract, brain, nervous system, oral cavity, peritoneum, thyroid gland, mammary gland, uterus and clitoris (Rice 2005; Hogervorst et al. 2007, 2008, 2010; Besaratinia and Pfeifer 2007; Larsson et al. 2009a, b, c, d; Pedersen et al. 2010; Lyn-Cook et al. 2011; Pelucchi et al. 2011). Acrylamide also reacts with glutathione and this may (1) alter the redox status of cells, (2) gene transcription, (3) interference with DNA repair or (4) bring about hormonal imbalance (Besaratinia and Pfeifer 2007). Acceptable daily intake (ADI) limit of acrylamide is 1.0 μg kg−1 of body weight day−1 (WHO 2002; Grob et al. 2003; Cummins et al. 2006).
Two years of study by National Toxicology Program (NTP), USA, on acrylamide revealed clear evidence of carcinogenic activity based on tumour in multiple sites (NTP 2013). This study was carried out by NTP based on the demand for such study by the US Food and Drug Administration (FDA). FDA actually wanted high-quality data from animal studies to exactly know about the potential risk to humans due to dietary exposure to acrylamide. After this report by NTP, FDA is currently developing guidelines for industry to bring down the acrylamide levels in food products (see advisories at http://www.fda.gov/Food/FoodborneIllnessContaminants/ChemicalContaminants/ucm151000.htm).
Glycidamide is a major metabolite of acrylamide. When acrylamide is consumed through food, the body converts it to glycidamide (NTP 2013). Acrylamide and its epoxide metabolite glycidamide [produced by cytochrome P4502E1 (CYP2E1)] are clastogenic and glycidamide can forms DNA adducts (Doerge et al. 2005; Hogervorst et al. 2008; EFSA 2015). As already stated above for non-genotoxic pathways, acrylamide reacts with glutathione and in this way it can not only alter the redox status of cells but also the gene transcription. In addition to this, it may interfere with hormonal balances and repair of DNA (Besaratinia and Pfeifer 2007). The genotoxic action of glycidamide (Gamboa da Costa et al. 2003; Ghanayem et al. 2005; Doerge et al. 2005) is currently considered as the mechanism of carcinogenic action of acrylamide in cancer risk assessments. Toxicological studies with the main metabolite of acrylamide, i.e. glycidamide have shown that the metabolite is carcinogenic and genotoxic (NTP 2013). Acrylamide probably accounts for neurotoxicity while the glycidamide may be critical for the carcinogenic and genotoxic responses in animals and humans (Codex Alimentarius Commission 2004; Ghanayem et al. 2005; Rice 2005; Burley et al. 2010; Anonymous 2010; LoPachin and Gavin 2012).
Levels of reducing sugars in potato is a main factor contributing towards the formation of acrylamide in processed potato products
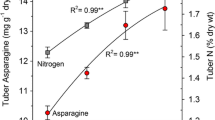
Reducing sugars (glucose and fructose) and free asparagines are the major determinants of acrylamide formation in potatoes (Haase et al. 2003; Amrein et al. 2003; Becalski et al. 2004; Matsuura-Endo et al. 2006; Halford et al. 2012a, b). Since the concentration of asparagine is already high in potatoes (0.5–3.0% on dry weight basis), it is the concentration of reducing sugars which is considered as rate-limiting factor for acrylamide formation in fried potato products (Pedreschi et al. 2014). Sugar concentration in fact exhibits a strong correlation with the amount of acrylamide formed in processed potato products (Biedermann et al. 2002; Amrein et al. 2004; Matthaus et al. 2004; Gokmen et al. 2007; Mestdagh et al. 2008). These findings re-emphasize the importance of ongoing practice of selecting varieties with low levels of reducing sugars. Out of two reducing sugars, glucose and fructose, it is the fructose (as carbonyl source) which has been found more effective in forming acrylamide. Basically, it is the chemical reactivity and physical state of sugars that play role in acrylamide formation. The melting point of fructose (126 °C) is lower than that of glucose (157 °C) (Claeys et al. 2005; Robert et al. 2004; Gokmen and Senyuva 2006a), therefore, fructose is more reactive than glucose towards the formation of acrylamide on heating (Gokmen and Papazoglu 2008). Changes in the levels of substrates (reducing sugars and free amino acids) of acrylamide are also reported to be influenced by nitrogen and sulphur fertilization (Elmore et al. 2008; Muttucumaru et al. 2013, 2014).
Based on the correlation studies, many research reports have highlighted the relationship between levels of reducing sugars present in the tuber and acrylamide formation in crisps (Ohara-Takada et al. 2005; Matsuura-Endo et al. 2006; Viklund et al. 2008; Elmore et al. 2015). In practice, a limit of reducing sugars ranging from 150 to 200 mg per 100 g of tuber FW (fresh weight) is used as an indicator for suitability of potatoes for processing (Pedreschi 2007). In addition to potato variety, temperature prevailing during storage of potatoes play a major role in determining the levels of reducing sugars present in potatoes and their suitability for processing (Kumar et al. 2004; Ezekiel et al. 2007a, b). Therefore, temperature during potato storage influences the level of acrylamide formed in the processed products prepared from stored potatoes. More details on this aspect are described later in this article.
Approaches useful in reducing acrylamide content in potato products
Use of suitable varieties
At a given point of time, sugar contents in potatoes are reported to be influenced more by variety and storage conditions than any other factor (Olsson et al. 2004; Amrein et al. 2004; Becalski et al. 2004; Kumar et al. 2004; Viklund et al. 2008; Knutsen et al. 2009; Whittaker et al. 2010; Halford et al. 2012a, b). Potato variety has a significant effect on acrylamide formation (Williams 2005; De Meulenaer et al. 2008; Elmore et al. 2015). Natural variation in DNA at candidate loci is reported to be associated with potato chip colour (Li et al. 2008). The trait of acrylamide producing potential and chip colour of potato is known to segregate in breeding population with significant variations (Manivel et al. 2007; Shepherd et al. 2010). Besides the levels of reducing sugars and amino acids, phenolic compounds and other antioxidants also influence the formation of acrylamide (Trelka 2010; Zhu et al. 2010; Ou et al. 2010; Jin et al. 2013). De Wilde et al. (2006) reported the influence of fertilization (preharvest factor) on acrylamide formation during frying. In a study by Elmore et al. (2015), 20 UK varieties were stored for 2 and 6 months at 8 °C. Before processing into chips varieties were analysed for the levels of precursors involved in the formation of acrylamide. Results showed very large varietal differences in the total free amino acids (73–137 mmol kg−1 dry weight), asparagine (14–29% of the total free amino acids) and total reducing sugars (3.7–520 m mol kg−1 dry weight). Acrylamide content in chips ranged from 131 to 5360 µg kg−1 of chips. The study thereby concluded that varietal selection and varietal improvement are of great importance in minimizing the levels of acrylamide in processed potato products.
Use of appropriate storage method, temperature and duration
Content of sugars and free amino acids in stored potato tubers is strongly influenced by storage conditions mainly the temperature and duration of storage (Kumar et al. 2004, 2005; Ezekiel and Dahiya 2004; Paul et al. 2005; Ohara-Takada et al. 2005; De Wilde et al. 2005; Matsuura-Endo et al. 2006; Mehta and Ezekiel 2006; Ezekiel et al. 2007a, b; Paul and Ezekiel 2007; Kyriacou et al. 2009). In contrast to normal cold storage of potatoes at 2–4 °C (RH 90–95%), storage at 8–12 °C (RH 85–90%) results in lower accumulation of reducing sugars in potatoes (Ezekiel et al. 2005a, b) which make them suitable for processing. The rate of respiration by tubers at 8–12 °C is lower than that at 4 °C (Burton 1989), therefore the respiratory losses in tubers stored at 8–12 °C are lower than that at 4 °C. Storing of potatoes at 2–4 °C however results in cold-induced sweetening (CIS) leading to a large accumulation of reducing sugars in potatoes (Sonnewald 2001; Blenkinsop et al. 2002; Ezekiel et al. 2005a; Mehta et al. 2010; Kumar 2011). So, potatoes stored at this temperature become unsuitable for processing because higher levels of reducing sugars finally result in brown or dark-brown colour of fried products mainly due to higher degree of Maillard reaction and formation of higher levels of acrylamide (Ezekiel et al. 2003, 2007a). Therefore, storage of potatoes at 8–12 °C is widely accepted as the optimum storage temperature for maintaining lower or acceptable levels of reducing sugars in potatoes (Ezekiel et al. 2005a, 2007b). Since storage at 8–12 °C induces sprouting on potatoes, they are treated with isopropyl N-(3-chlorophenyl) carbamate (CIPC) (Ezekiel and Singh 2007; Ezekiel et al. 2007b; Mehta et al. 2007, 2010; Wustman et al. 2011; Paul et al. 2014). CIPC is a sprout suppressant (Marth and Schultz 1952; Burton et al. 1992; Blenkinsop et al. 2002; Ezekiel et al. 2005a, b; Mahajan et al. 2008; Singh and Ezekiel 2010; Paul and Ezekiel 2013), which is applied in the form of a fog treatment (Ezekiel et al. 2005a, b). For long-term storage of potatoes (4–8 months; from March to October), two CIPC treatments are required [each @ 18 g (a. i.) per metric tonne of potatoes] (Kumar and Pandey 2008; Brajesh and Ezekiel 2010).
On-farm or traditional methods of potato storage such as pits and heaps have proved to be highly useful in supplying potatoes to the processing industry for 2–3 months after the harvest of potato crop (Paul et al. 2005; Mehta and Ezekiel 2010; Wustman et al. 2011; Paul and Ezekiel 2013; Gautam et al. 2013; Ezekiel 2015). In these storage methods, high storage temperature (21–32 °C with RH ranges from 51 to 95%) helped in maintaining the lower concentration of reducing sugars in potatoes (Paul and Ezekiel 2003a, 2005; Kumar et al. 2005; Ezekiel et al. 2005a). Generally, potatoes stored by traditional methods have acceptable weight loss and chip colour (Paul and Ezekiel 2003a, b, 2004a, b). Studies on traditional methods of potato storage done at various locations in India indicated that after 90 days of storage levels of reducing sugars can decrease up to 30% (Kumar et al. 2005; Paul et al. 2005; Mehta and Ezekiel 2010). This reduced level of reducing sugars results in lower chip colour score, which is an indicator of low levels of acrylamide in the processed potatoes (Gokmen et al. 2007; CPRI Annual Report 2011–2012). Kufri Chipsona-1, a processing variety could be stored up to 110 days under traditional storage methods (CIPC was used as sprout suppressant) with acceptable chip colour (Singh and Ezekiel 2005; Mehta et al. 2007). The levels of reducing sugars decreased from its initial value of 155 to 34 mg per 100 g of tuber FW at the end of storage period and the residue level of CIPC was also found to be below the permissible level of 10 mg per kg of tuber FW (Singh and Ezekiel 2005). Use of various sprout suppressants other than CIPC have been reviewed (Mehta and Ezekiel 2006; Paul et al. 2016a) and a few (maleic hydrazide and glyphosate) have been tested in India as preharvest treatment (Paul and Ezekiel 2006a, b; Mehta and Ezekiel 2010; Mamani Moreno et al. 2012; Paul et al. 2014). Alcohols, acetaldehyde, 2,4-dichlorophenoxy acetic acid ethyl ester, triadimefon, γ radiation, 1,4-DMN, essential oils like caraway, mentha oil (Burton et al. 1992; Tayler et al. 1996; Paul and Ezekiel 2002, 2003c, 2004a; Mehta and Kaul 2002; Ezekiel et al. 2008; Campbell et al. 2010; Chauhan et al. 2011; Olsen et al. 2011; Rezaee et al. 2011; Gomez-Castillo et al. 2013; Paul et al. 2016a) have been tried for their efficacy and potential as alternatives of CIPC during storage of potatoes.
There is a significant effect of storage duration, particularly longer storage duration, on the content of acrylamide detected in potato chips (Halford et al. 2012a; Muttucumaru et al. 2014). Largest increases in acrylamide levels were observed after 6 months of storage, while the increase was small from potatoes stored up to 6 months (Halford et al. 2012a). Similar conclusions were drawn in a recent study with 20 UK-grown varieties of potatoes by Elmore et al. (2015) and they recommended that potatoes for processing should be used within the optimal storage window of 2–6 months (Elmore et al. 2015).
Steps before processing
Reconditioning is the process of storing potatoes at elevated temperature (10–20 °C at RH 75–90%) for a period of 2–3 weeks. This results in a decrease in the levels of reducing sugars (Coffin et al. 1987; Sukumaran and Verma 1993; EFSA 2003; Grob et al. 2003; Kumar et al. 2004; Kumar and Ezekiel 2005; Singh et al. 2008; Mestdagh et al. 2008; Truong et al. 2013).
Steps during processing
Enzymatic browning
Enzymatic browning does not produce acrylamide because this browning is not mediated by Maillard reaction. Here discolouration takes place due to the oxidation of phenolic compounds by the enzyme polyphenol oxidase. Quinones are formed as a result of this enzymatic reaction and they impart brownish colour. This enzymatic browning is usually reduced by inhibiting the action of above enzyme by high temperature dip (which results in inactivation of enzyme). Technically, this step is known as blanching and it involves dipping of cut slices into hot water at temperature of 60–80 °C for 2–3 min. This step not only reduces the enzymatic browning significantly, but it also removes (washout) some proportion of reducing sugars and asparagine from the tubers (Grob et al. 2003; Pedreschi et al. 2004, 2014; Marwaha et al. 2008; Viklund et al. 2010 ). Blanching not only helps in reducing the extent of enzymatic browning, but also the extent of Maillard reaction. This finally results in reduction in the formation of acrylamide during frying. Dipping in normal water has also been found useful. Pedreschi et al. (2004) found 32 and 25% decrease in glucose content in the potato slices when soaked for 90 and 40 min in distilled water, respectively. Earlier to this report, Grob et al. (2003) documented reduction of 6% in glucose and 12% in fructose by 30 min of washing of potato slices in cold water.
pH
Acrylamide formation in food is dependent on pH because the rate of Maillard reaction is strongly dependent on the pH of the reaction environment. The pH around 7.0 is considered as optimum for the formation of acrylamide in food. At pH below 7.0, acrylamide formation is inhibited (Brown 2003; Baardseth et al. 2006). Lowering the pH of the potatoes by various approaches including treatment with citric acid (0.5–1.0%, for about 20 min) results in lowering the levels of acrylamide (Jung et al. 2003; Rydberg et al. 2003; Anonymous 2003; Taubert et al. 2004; Surdyk et al. 2004; Kita et al. 2004; Gama-Baumgartner et al. 2004; Low et al. 2006).
Water activity
Acrylamide formation in food is also dependent on water activity. Acrylamide formation takes place only when the water activity of food is below 0.8 and rate of acrylamide formation is reported to be very high at the water activity value of 0.4 or less than this (Hoenicke and Gatermann 2004, 2005). Removal of acrylamide from heated foods (bakery products and potato chips) is reported to enhance with the rise of water activity (Sadd et al. 2008).
Temperature during frying
One of the straight ways to lower the levels of acrylamide is to avoid visual browning when frying or baking (WHO 2002). Acrylamide formation is less in the beginning of the frying process, because in the beginning the moisture level of the food item is high (a condition not favourable for the formation of acrylamide). But, as the frying proceeds, the acrylamide level increases exponentially (due to higher rate of dehydration or sharp decrease in moisture levels (now conditions become favourable for the Maillard reaction to take place) (Gokmen and Papazoglu 2008). Due to this, acrylamide formation increases sharply towards the end of the frying process (Xu et al. 2014) especially when the frying temperature is also high (above 175 °C). It is thereby suggested that the initial frying temperature should not exceed 170 °C (Matthaus et al. 2004) and further reduction in the formation of acrylamide can be achieved by lowering the temperature to 150 °C towards the end of the frying period. This necessitates for better control of frying temperatures. So, it is necessary to have reliable and accurate temperature control system inbuilt in the frying equipments. In view of higher cost of such equipments (with precise temperature controlling system), they are more expected to be the part of commercial or industrial setup and preparations (Qu et al. 2008; Medeiros Vinci et al. 2011).
Optimum time of frying
Overcooked French fries show a very high level of acrylamide (>10,000 µg kg−1). This clearly indicates the impact of frying temperature and frying time on acrylamide formation (WHO 2002; Gokmen et al. 2006a). Matthaus et al. (2004) also reported that in French fries the amount of acrylamide increases with the temperature and the frying time (especially at the temperature more than 175 °C). On the other hand, with the same frying time but at the temperature of 150 °C there is little or no effect on the formation of acrylamide in potato slices. After frying at 190 °C for 3 min, acrylamide content was about 40 times higher than that of frying at 150 °C for 9 min (Pedreschi et al. 2005). This study also reports that after frying for 7 min (at 150 °C) the acrylamide content of potato slices was about 500 µg kg−1 and after frying for 3.5 min (at 190 °C) the level was 4500 µg kg−1. The increase in acrylamide with the time follows a linear function, while a non-linear relationship exists with the temperature of frying. Further it was noted that after frying for 10 min (at 170 °C) the acrylamide content in French fries was about 800 µg kg−1 and this increased to about 3700 µg kg−1 after frying for 10 min (at 190 °C). The above study thus showed that a reduction in the processing temperature results in lower concentrations of acrylamide in the products. Romani et al. (2008) studied acrylamide content in French fries fried at fixed initial oil temperature of 180 °C for different time durations (3, 4, 5, 6, 7, 8 and 9 min). It has been found that after about 4 min of frying when the temperature of potato surface reaches 120 °C and that of oil reaches 140 °C; the increase of frying time becomes a key factor in deciding the rate of formation of acrylamide. Thus, the above studies have demonstrated that higher temperature and longer frying time contribute to higher levels of acrylamide during processing. Based on the colour, oil content and acrylamide content, the best culinary product can be obtained after 5 min of frying at temperature of 180 °C. It was concluded by Williams (2005) and Fiselier et al. (2006) that it would be prudent for manufacturers to search for a time/temperature combination at which acrylamide levels may be reduced without unduly affecting quality and such solutions are likely to be product specific.
Type of oil
It was noted that the type of frying oil used for frying has little influence on the formation of acrylamide (Becalski et al. 2003; Mestdagh et al. 2005). However, a recent report by Zhang et al. (2015) reported higher levels of acrylamide in French fries when frying was done in oils with higher heat transfer coefficient in comparison to the oils with lower heat transfer coefficient. Another report (Anonymous 2009) showed that among different types of oils, potatoes fried in sunflower oil exhibited highest levels of acrylamide formation.
Home versus commercially produced chips and French fries
Individual or home cooking is likely to have higher levels of acrylamide than the commercially produced chips or French fries. This highlights the importance of the steps such as reconditioning, soaking, blanching and pre-frying in reducing the levels of sugars and asparagine and these steps are either not carried out or difficult to do in home environment (Cummins et al. 2006). For home cooking, chip colour may be aimed at white or golden yellow instead of brown and frying temperatures should not be higher than 175 °C (EU 2003; Food Drink Europe 2011).
Extruded product
In a study on extrusion of blends of potato flour and semolina it was observed that both, the formation and extrusion processing conditions influence the acrylamide formation. The moisture content of the flour sample and the die temperature had a strong influence on acrylamide formation than the screw speed (Mulla et al. 2011a).
Present status of potato and potato processing industry in India
With the average productivity of 21.1 metric tonnes per hectare, India produced 41.55 million metric tonnes of potatoes from 1.973 million hectares of area in 2014 and ranked second in the world (FAOSTAT 2015; NHB 2014a). India contributes about 11.37% of the global potato production of 365.44 million metric tonnes. In India about 89.5% of the total potato production comes from the Northern plains (also referred as Indo-Gangetic plains) (Paul et al. 2014). The situations and problems linked to the storage of potatoes in temperate countries of the world are different from that of sub-temperate, sub-tropical and tropical countries. This is because the harvest of potatoes in the former is followed by cool winter months, whereas in the latter harvest is followed by hot summer months. Potato crop in India is mainly grown during winters. Out of total annual production of potato in India, about 68% are utilized for table purpose, 7.5% for processing, 8.5% is used as seed for next year planting and remaining 16% represent a loss, mainly the postharvest loss (Gupta et al. 2014). Gupta et al. (2014) reported that about 65% of the total potato produce in India are kept in cold stores. Remaining 35% of potatoes are stored under ambient (non-refrigerated conditions) by using traditional/on-farm storage methods. As per a recent survey published by NHB (2014b), total number of cold stores in India is about 5367 and they can provide storage capacity up to 26.85 million metric tonnes. Out of these, around 3414 cold stores stock raw potatoes.
Two major aspects that determine the progress of processing sector of potatoes in India are the availability and spread of (1) potatoes suitable for processing and (2) short-term and long-term potato storage methods. Chips, French fries and other processed products made from varieties which are not suitable for processing or even from processing-grade potatoes but not stored properly result in products of inferior quality along with high degree of browning due to higher levels of reducing sugars. The above two aspects are described and discussed below.
Availability and supply system of suitable and processing-grade potato varieties
About 25 years back, suitable raw material for chips and French fries was not available in India. But today, potato processing industry in India has witnessed a sea change with availability of quality raw material round the year especially for chips (Ezekiel et al. 2005a, b; Pandey et al. 2006a, 2008a, b; Brajesh and Ezekiel 2010; Marwaha et al. 2010; Mehta and Ezekiel 2010; Mehta et al. 2010; Paul and Ezekiel 2013). CPRI (Central Potato Research Institute, an ICAR institute) has developed and released six new potato varieties suitable for processing viz. Kufri Chipsona-1 (Gaur et al. 1998), Kufri Chipsona-2 (Gaur et al. 1999), Kufri Chipsona-3 (Pandey et al. 2006b), Kufri Himsona (Pandey et al. 2008c), Kufri Chipsona-4 (CPRI Annual Report 2009– 2010) and Kufri Frysona (Singh 2010) during the last 15 years. Out of these newly released potato varieties for processing only Kufri Chipsona-1 is being grown on considerable area. Other, four potato varieties have not become as popular as Kufri Chipsona-1 due to several reasons including keeping quality, crop duration and tuber characteristics. So, Kufri Chipsona-1 along with other earlier released Indian varieties namely Kufri Jyoti, Kufri Lauvkar and Kufri Chandramukhi have helped in fulfilling the needs of potato processing industries in India for wide range of products like chips, French fries, flakes and other items including laccha, dehydrated chips, flour and starch. Important features and quality parameters of some of indigenously developed and exotic being grown on large area are presented in Table 2. It is important to note that depending upon the location and cultural practices followed, there exist wide variations in the levels of reducing sugars and dry matter in different varieties (Table 2) (Ezekiel et al. 1999a, b; Marwaha et al. 2007; Pandey et al. 2009; Singh et al. 2009).
Acrylamide content in potato chips prepared from freshly harvested tubers of Indian potato varieties varies widely (CPRI Annual Report 2009–2010). Minimum concentrations of acrylamide were found in processing varieties Kufri Chipsona-2 (40 µg kg−1 of chips) and Kufri Chipsona-1 (109 µg kg−1) (Table 3). Other varieties such as Kufri Jyoti, Kufri Lauvkar and Kufri Chandramukhi which are also being used for processing at commercial scale had comparatively lower concentrations of acrylamide (ranging from 108 to 306 µg kg−1). While, varieties which are considered unsuitable for processing namely Kufri Anand, Kufri Girdhari, Kufri Sutlej, Kufri Lalima and Kufri Pukhraj, etc., contained very high concentrations of acrylamide in them (more than 1000 µg kg−1) (Table 3). There was a significant and positive relationship between acrylamide content and chip colour (CPRI Annual Report 2009–2010). Mulla et al. (2011b) determined the content of reducing sugars, asparagine and acrylamide in eight Indian potato varieties (Kufri Chipsona-2, Kufri Anand, Kufri Sutlej, Kufri Bahar, Kufri Surya, Kufri Lauvkar, Kufri Jyoti and Kufri Badshah). Levels of all these constituents were significantly influenced by storage temperature (4 and 14 °C), duration of storage (2, 4 and 6 months) and treatment (with and without irradiation). Variety Kufri Chipsona-2 showed lowest levels of reducing sugars (68.07 ± 5.65 mg 100 g−1 FW) and acrylamide (416.75 ± 15.25 μg kg−1 of chips). Variety Kufri Lauvkar showed lowest levels of asparagine content (207.44 ± 12.22 mg 100 g−1 FW). At storage temperature of 14 °C, levels of acrylamide in non-irradiated potatoes of Kufri Chipsona-2 changed from 416.4 ± 7.8 (initial value), 451.8 ± 5.3 (2 months), 511.9 ± 4.6 (4 months) and 548.8 ± 1.8 (6 months). Similar values for irradiated potatoes were 416.4 ± 7.8 (initial value), 432.7 ± 4.6 (2 months), 469.3 ± 1.7 (4 months) and 502.6 ± 2.8 (6 months) (Mulla et al. 2011b). Comparison of different studies [Mulla et al. 2011b and CPRI Annual Report 2009–2010 (Table 3)] for content of acrylamide present in potato chips of different Indian varieties showed wide variations. This could be due to differences in the procedures followed for determination of acrylamide.
Determination of acrylamide contents in French fries prepared from freshly harvested potato tubers of different varieties (Singh 2013) showed that concentrations of acrylamide were minimum in processing varieties namely Kufri Chipsona-1, Kufri Chipsona-2, Kufri Chipsona-3, Kufri Himsona, Kufri Frysona and Kufri Chandramukhi varying from 63 to 101 μg kg−1. These varieties also had lower concentrations of reducing sugars. However, the varieties which are generally considered unsuitable for processing such as Kufri Arun, Kufri Anand, Kufri Giriraj, Kufri Ashoka, etc., had significantly higher concentrations of acrylamide (364–3686 μg kg−1). A positive correlation between the content of acrylamide and reducing sugars and also with colour score was observed (CPRI Annual Report 2011–2012; Singh 2013).
The complete cycle for round the year availability of processing-grade potatoes in India is presented in Fig. 2. Kufri Chipsona-1 along with other earlier released Indian varieties namely Kufri Jyoti, Kufri Lauvkar and Kufri Chandramukhi are being used by potato processing industries in India (Patel et al. 2007; Singh et al. 2009; Wustman et al. 2011; Vivan Fernandes 2015; Virendra Pandit 2015a). In addition to these Indian varieties, exotic varieties such as Lady Rosetta and Atlantic are grown in India for chipping industry (Wustman et al. 2011; Virendra Pandit 2015b) and Shepody, Kennebec and Santana are grown in Gujarat state for French fries (Vivan Fernandes 2015; Virendra Pandit 2015a). The strides made in Gujarat state in potato processing are praise worthy. At present, around 7.5% of total area under potato is occupied by the processing varieties both of Indian and exotic origin. Out of total processing-grade potato produced in Gujarat, 65% is used for making chips, while the remaining 35% is used for preparation of French fries.
The complete cycle for the availability of processing-grade potatoes in India. Months and the duration between months represent the time period for which potatoes are at present available for processing and table purposes. Varieties with names beginning with K (Kufri) are Indian varieties while Lady Rosetta and Atlantic are imported varieties grown for chipping industry and Shepody, Kennebec and Santana are again imported varieties grown for French fries. Although a number of potato varieties such as Kufri Chipsona 1, 2, 3, and 4 are available for processing, only those potato varieties (Indian and Exotic) which are being cultivated in considerable area in different states have been shown here
From the month of May and onwards, industries utilize potatoes stored at 10–12 °C with CIPC treatment (Fig. 2). But, after July/August, stored potatoes start accumulating higher amount of reducing sugar (due to prolonged storage) and this makes these potatoes unsuitable for processing. The period after July/August was considered critical due to non-availability of potatoes from plains. This problem is now solved in view of the availability of potatoes suitable for processing from the hilly regions of the country (Fig. 2). Hills have a long planting and harvesting season for potato depending upon the altitude where the crop is grown. So, now hills are the source of potatoes suitable for processing from July to September (Fig. 2).
The above described system of potato availability and supply chain have enabled a number of potato processors in India including ITC Ltd. (Bingo), Potato King, GP Foods, Marino Industries Ltd., Century Lamination Co., Haldiram, Balaji Wafers, Himalaya International Ltd, Bikano Namkeen, Ace Food, Arumugam Industries, Golden Fries, Vimpy International, Vista Foods, Kakaji Namkeen, Mota Chips, Shivdeep Foods, Vimal Oil and Foods, Prakash Snacks, Twinkle Chips, Budhani Brothers, Chintamani Food and Snacks, Kishlay Foods, Faber Leather, etc., to produce quality potato products, mainly the potato chips. Multinational companies such as Pepsi (Lays, Lehar, Uncle Chips) and McCains have also set up potato processing units in the country (Rana and Pandey 2007; Pandey et al. 2009; Marwaha et al. 2010; Wustman et al. 2011; Virendra Pandit 2015a, b). In addition to above referred organized sector, high proportion of potato in India is also processed by unorganized sector (Rana and Pandey 2007). Farmers in the identified areas (suitable for growing processing quality potatoes) have entered into contract farming of processing-grade potatoes to the processing industries. This arrangement has ensured timely availability of required quality raw materials to processors along with assured profitable returns to the farmers (Rana and Pandey 2007; Ganguly 2013; Virendra Pandit 2015a, b; Vivan Fernandes 2015). In addition to this, contract farming has also helped in avoiding gluts leading to distress sale by farmers.
Although the above Indian varieties contributed towards the development of processing sector in India, potato processors use Atlantic (USA, released in 1968) and also Lady Rosetta (the Netherlands, released in 1988) as raw material for potato chips (Wustman et al. 2011). Lady Rosetta is preferred especially in Gujarat (Wustman et al. 2011; Virendra Pandit 2015b). Chips prepared from this variety have low levels of acrylamide, i.e. 340 and 655 µg kg−1 even after 2 and 6 months of storage, respectively, and these values are far below the European Commission (EC) indicative value of 1000 µg of acrylamide per kg of chips (EC 2011based technology has been standard; Elmore et al. 2015).
Availability and spread of short-term and long-term potato storage methods
Work on standardization, gradual improvements and multi-location testing of traditional storage methods and trials in modern potato stores were carried out in India during the last 15 years (Paul and Ezekiel 2003a, 2004b, 2013; Ezekiel et al. 2003, 2005a, 2007a, b; Kumar et al. 2005; Marwaha et al. 2005; Kumar and Ezekiel 2006; Mehta et al. 2010; Wustman et al. 2011; Sandhu et al. 2014; Ezekiel 2015). CIPC-based technology has been standardized and recommended for storing potatoes at 10–12 °C with 85–90% RH (Ezekiel et al. 2003, 2005a, b, 2007a, b; Ezekiel and Singh 2007; Singh and Ezekiel 2010). Presently this technology is used for large-scale storage of processing potatoes for 5–6 months (with the use of CIPC as sprout suppressant). This technology has proved profitable to the farmers, cold store owners and traders not only for processing potatoes but for storing table potatoes as well. In addition to this, this technology has played a pivotal role in enabling round the year availability of required quantity of suitable potatoes for potato processing industry (Fig. 2). Potatoes stored by above technology fetch higher price in the market due to better quality and processing traits. In view of economic benefits and also as an effective way of better postharvest management of potato this technology has witnessed rapid progress and spread across the country in the past (Paul and Ezekiel 2013). This is also evident from the fact that in the year 2011, 390 cold stores exclusively practised this technology for storing 1.065 million metric tonnes of potatoes at 10–12 °C with CIPC treatment (Paul and Ezekiel 2013). This figure has increased to more than 2.5 million metric tonnes in the year 2015 (Paul et al. 2016b). Further improvement in the above technology is achieved by regulating the level of CO2 (carbon dioxide) during the period of storage. Besides maintaining the temperature at 10–12 °C (with 85–90% RH), CO2 levels are maintained lower than 0.5% or 5000 ppm (Paul and Ezekiel 2013). With two CIPC treatments during the storage period, processing potatoes can be stored up to 6 months and table potatoes can be stored up to 9 months (Paul and Ezekiel 2013). Problems of age-related sweetening (senescence sweetening) and decay (rottage) were also found to be low. Several cold stores in different states such as Gujarat, Uttar Pradesh, West Bengal, Madhya Pradesh, Haryana and Punjab are now using this improved technology (Paul and Ezekiel 2013).
In addition to the above large-scale modern storage methods, different types of non-refrigerated traditional storage methods (heaps, pits, spreading tubers on floor, a dark room as such or with provision of evaporative passive cooling, storing in bins, ventilated wooden or bamboo structures and hanging potatoes in bamboo baskets) are also used by farmers for short-term storage of potatoes (Ezekiel et al. 1999a, b; Paul et al. 2002a, b; Paul and Ezekiel 2004b; Wustman et al. 2011; Gautam et al. 2013; Ezekiel 2015). These methods are cost-effective and helpful in extending the time period for marketing (Ezekiel et al. 2005a; Paul and Ezekiel 2013). With the gradual improvements in these on-farm storage methods of potato storage and integration of time-bound schedule of crop production and storage-related activities (Paul et al. 2002a, b; Paul and Ezekiel 2003a, b, 2004b, 2005; Kumar et al. 2005; Mehta et al. 2007) these methods have become more popular among the farmers of low or medium income groups. Today, thousands of metric tonnes of potatoes are being stored by these on-farm methods in states like Madhya Pradesh, Punjab, Gujarat, Uttar Pradesh, Karnataka and Maharashtra (Wustman et al. 2011; Paul and Ezekiel 2013; Ezekiel 2015).
Glut of potatoes at the time of harvest, low utilization of available potatoes for processing, non-availability of storage space in cold stores, unequal distribution of cold stores in India (about 70% of the total storage capacity exists in six states of India), high cost of cold storage (Rs 10–15 per 50 kg bag of potatoes for 1 month), poor management during cold storage, outdated cold storage facilities and operation of cold stores as stand-alone unit (majority of cold stores in India are not networked) contribute to substantial postharvest losses (Wustman et al. 2011; Paul and Ezekiel 2013; NHB 2014b). This situation demands a better postharvest management of potatoes including increased utilization of potatoes for processing (Wustman et al. 2011; CPRI 2013a, b). As already stated only 7.5% of total potato production in India is processed. This fraction is quite low in comparison to the figures of 75% in the Netherlands (40% into food products and 30% for making potato starch) and 40–50% in USA (French fries, chips and other processed potato products) (Wustman et al. 2011). The present scenario needs to be changed with major policy decisions and an urgent initiative for modernization of cold stores in India on priority basis. This step will ensure better monitoring of temperature, RH, air circulation and levels of CO2. This in turn will improve the efficiency of cold stores and processing quality of stored potatoes besides contributing for energy efficiency and higher effectiveness of CIPC as sprout suppressant.
Colour of processed potato products as an indicator of acrylamide content
In many studies a significant positive correlation has been reported between acrylamide content and chip colour score (Gokmen and Senyuva 2006b; Pedreschi et al. 2006; Gokmen et al. 2007; Serpen and Gokmen 2009; Halford et al. 2012a, b). For baked potato discs (made from potato mash consisting of potato powder (30%), oil (10%), flour (10%), and water (50%), it was observed that browning ratio higher than 45% and moisture content lower than 17% result in dark brown or almost burnt products. In these cases, no correlation was noticed between the selected variable and acrylamide content. However, a good linear correlation can be observed with the acrylamide formation when the browning ratio falls in the range of 0–45%. This suggests that the browning ratio may be considered as a reliable indicator of acrylamide concentration (Kotsiou et al. 2013). Acrylamide formation in low-fat potato snacks was also found to be correlated with the colour development (Majcher and Jelen 2007). In a recent study by Elmore et al. (2015), chips which were brownest were found to be the highest in acrylamide content while those which were relatively pale were low in acrylamide. These studies have made it clear that dark-coloured chips/French fries are not only disliked by consumers but can also be harmful to health due to higher levels of acrylamide. It was suggested that computer-based digital colour images of fried potato chips and French fries can be used for the estimation of acrylamide levels (Gokmen et al. 2006b, 2007). Further progress in this area in fact made use of computer-based optic-electronic combination to successfully demonstrate the sorting and selective removal of dark fried potato products (Gokmen et al. 2007; Pedreschi 2007; Gokmen and Mogol 2010; Food Drink Europe 2011; Medeiros Vinci et al. 2011). These technological advancements will be highly beneficial in safeguarding the limits of acrylamide in processed potato products for the benefits of consumers. Here, it is also important to mention that European Commission (EC 2011) has set indicative value for acrylamide in chips as 1000 µg per kg of chips and the similar value for ready-to-eat French fries is 600 (600 µg per kg of French fries). Kotsiou et al. (2013) have reported that the indicative value of 1000 µg kg−1 corresponds to a browning ratio of less than 8% and moisture content of fried potato products more than 23.5%.
Efforts to select/modify genotypes with better cold-induced sweetening (CIS) tolerance and reduced acrylamide formation
Cold-induced sweetening (CIS) tolerance
Storage at 8–12 °C with CIPC has reduced the problem of reducing sugar accumulation to a large extent in stored potatoes meant for processing. But at the same time, use of CIPC has some limitations and it is linked with issues pertaining to health and environmental safety (Balaji et al. 2006; Sakaliene et al. 2009; Sihtmaee et al. 2010; Smith and Bucher 2012; El-Awady Aml et al. 2014; Cools et al. 2014; Mohammed et al. 2014; Paul et al. 2014). In the present scenario, there is continuous demand for the lowest possible levels of chemical residues in potatoes and processed potato products. Therefore, it is necessary to identify/develop new varieties of potato that can show high level of tolerance or resistance to CIS under prolonged storage period of 6–9 months at 2–4 °C (Marwaha et al. 2010; Paul et al. 2016a; Mohammed et al. 2015). Development of processing potato varieties that can resist the CIS has become a priority area of research all over the world. Progress made so far in this area at international and national levels is discussed below.
Biochemical and molecular controls of CIS in potato were elaborated by Sowokinos (2001a). After this, considerable progress has been made in understanding the CIS at biochemical and molecular levels by following different approaches including (1) silencing of vacuolar invertase (Ye et al. 2010; Bhaskar et al. 2010; Brummell et al. 2011; Wu et al. 2011; Wiberley-Bradford et al. 2014; Zhu et al. 2014); (2) enhancing expression of apoplastic invertase inhibitor gene INHap (Greiner et al. 1999; Brummell et al. 2011; McKenzie et al. 2013); (3) lowering the expression of UDP-glucose pyrophosphorylase (Sowokinos 2001b; Gupta et al. 2008); (4) lowering amylose content in tubers (Jansky and Fajardo 2014); (5) enhancing expression of amylase inhibitor gene SbAI (Zhang et al. 2014); (6) enhancing the expression of RING finger gene (SbRFP1) (Chen et al. 2012; Zhang et al. 2013) and a new isoform of thioredoxin h group in potato (SbTRXh) (He et al. 2012) and (7) looking for allelic variation in the expression of apoplastic invertase, apoplastic invertase inhibitor and UDP-glucose pyrophosphorylase genes and using them as markers in molecular breeding programme (Gupta et al. 2008; Baldwin et al. 2011).
Broadly, three approaches were followed in India towards improvement in tolerance to CIS. First was to identify genetic resources with low temperature sweetening tolerance trait and then their subsequent uses in the development of varieties with tolerance to CIS. One tuberosum accession CP3103 and 2 andigena accessions (JEX/A 663 and JEX/A 785) have been identified as cold chippers and presently they are being used as parents in the ongoing breeding programme (Marwaha et al. 2008; Luthra et al. 2009). In addition to this, a hexaploid wild clone SS1735-02 (possessing tolerance to CIS up to 6 months of storage) was identified by Bhardwaj et al. (2013). Secondly, efforts were made to make the existing processing varieties cold tolerant through transgenic approach. This was attempted in two different ways: (1) reducing vacuolar acid invertase (INV) by over expression of tobacco invertase inhibitor gene (Nt-Inhh) in varieties Kufri Chipsona-1, Kufri Jyoti and Kufri Badshah. The transgenics produced showed acceptable chip colour even after cold storage (Pandey et al. 2009). (2) Silencing of INV gene expression at the post-transcriptional level through artificial microRNA (amiRNA), RNA interference (RNAi), or ribonuclease P (RNase P)-mediated silencing. In addition to this, reduction of UDP-glucose pyrophosphorylase (UGPase) was also attempted. This was done by silencing the gene either via introduction of intron containing inverted repeat gene construct against UGPase promoter (silencing at the transcriptional level) or through the introduction of amiRNAs against UGPase gene (silencing at the post-transcriptional level) (Pandey et al. 2009). RNAi, amiRNA and RNase P-mediated suppression of invertase activity has generated transgenic lines in variety Kufri Chipsona-1. These lines exhibited good cold chipping attributes even after 3 months of cold storage at 4 °C (CPRI Annual Report 2009–2010, 2010–2011). In yet another study, positive attempts have been made in identifying allelic variations in the expression of asparagine synthase, vacuolar invertase, apoplastic invertase, apoplastic invertase inhibitor and UDP-glucose pyrophosphorylase genes (Yadav et al. 2012). In future, combinations of these allelic variations can be used as markers in molecular breeding programme for varietal improvement. Although considerable work has been done and some transgenics have also been raised exhibiting tolerance to CIS, final and acceptable product that can pass through acceptability, popularity and commercial angle is still awaited.
Transgenic potato with reduced acrylamide
As per a recent report on potatopro.com (Anonymous 2016), the food regulator of Australia and New Zealand (Food Standards Australia New Zealand or FSANZ) has called for reviewing genetically modified (GM) potato which produces less of acrylamide when fried. This GM potato (named as Innate potato) is designed by Simplot (SPC International), wherein expression of four of the potato’s own genes is reduced. Last year, this GM potato has already been approved for its production and processing in US.
Additional management practices/strategies to lower acrylamide content in processed potato products
It is now clear that the level of acrylamide can be reduced by reducing the concentration of reducing sugars in potatoes. Further, it has also been shown that out of two types of reducing sugars, i.e. glucose and fructose, it is the fructose which is more actively involved in the reaction leading to the formation of acrylamide during frying (Gokmen and Papazoglu 2008). So, efforts should be made to reduce the level of fructose either through physiological breeding or biotechnological interventions. Asparaginase (enzyme which reduces the levels of free asparagine) if added before cooking leads to reduction in the acrylamide formation (Zyzak et al. 2003; Ciesarova et al. 2006; Pedreschi et al. 2011). New frying method such as vacuum frying is reported to reduce the acrylamide formation (Granda et al. 2004; Granda and Moreira 2005). It has also been shown that potato slices when treated with cations in non-transition state such as Ca2+ can reduce the acrylamide formation in foods during heating (Lindsay and Jang 2005). Likewise, out of various additives, calcium chloride reduced formation of acrylamide formation (up to 65%) in extruded snacks, prepared from blends of semolina and potato flour (Mulla et al. 2011a). Cations basically prevent the formation of Schiff base, a key intermediate responsible for acrylamide formation (Gokmen and Senyuva 2007). For French fries, lowering of surface area-to-volume ratio (SVR) results in lower levels of acrylamide formation. It is therefore recommended to cut thicker strips (Haase 2006; Food Drink Europe 2011). Based on the growing experience of researchers and food industry a guidance document referred as “Toolbox” has been created to bring down the levels of acrylamide in processed products (Codex 2009; Food Drink Europe 2011; Anonymous 2011; Food Drink Europe Acrylamide Toolbox 2013). These guidelines cover mitigation strategies applicable at different stages during manufacturing, including raw material, ingredient (recipe), and food processing (heating).
Recent past has shown incremental trend in the awareness on human health safety and environmental issues. Hence, today there is higher concern on the issue of acrylamide in processed potato products (Fohgelberg et al. 2005; Deryck 2007; Pedersen et al. 2010; Anonymous 2010; Lyn-Cook et al. 2011; Lin et al. 2011; EFSA 2011; EC 2011; WHO 2011; LoPachin and Gavin 2012; Medeiros Vinci et al. 2012; Riboldi et al. 2014; EFSA 2015). But at the same time it can also be said that scientific progress has contributed towards better assessment (Grob 2007; Besaratinia and Pfeifer 2007; EFSA 2011), monitoring (CIAA 2009; EC 2009; Serpen and Gokmen 2009; Gokmen and Mogol 2010; Biedermann et al. 2010; EC 2011) and availability of strategies to lower the levels of acrylamide in processed potato products (Gerendas et al. 2007; Gobel and Kliemant 2007; Anese et al. 2009; Mestdagh et al. 2009; Pedreschi 2009; Pedreschi et al. 2010, 2011; US Patent 2010; Cheng et al. 2010; Bassama et al. 2010; Anese et al. 2010; Medeiros Vinci et al. 2010, 2011, 2012; Lineback et al. 2012; Troise and Fogliano 2013; Zhang et al. 2015). Advancements in different areas pertaining to potato crop improvement, crop production, storage methods, pre- and post-processing protocols and associated technologies and other management practices will help in achieving still higher level of food safety standards for processed potato products. This statement is supported by the fact that during the year 2002–2011, modifications in cooking practices and improvements in management of potato storage in Europe resulted in about 53% decrease in the acrylamide present in potato crisps (Powers et al. 2013).
Acrylamide exposure varies and it depends primarily on eating habits of the peoples and the method by which foods are processed and prepared (Svensson et al. 2003; Konings et al. 2003; Dybing and Sanner 2003; FDA 2004; Boon et al. 2005; Wilson et al. 2009, 2010; Borda and Alexe 2011; EFSA 2011). Generally, daily consumption of fried potato products, baked food, roasted coffee and ready-to-eat breakfast cereals contribute to the acrylamide exposure (Gokmen and Papazoglu 2008). Since acrylamide is present in a very wide range of food items which are consumed on a daily basis, the above-mentioned health concern applies to all consumers but based on the body weight, children fall in the most exposed group (EFSA 2015). Due to food habit and life style, European people are expected to get exposed to higher levels of acrylamide as they consume lots of chips, French fries, coffee and baked food like cake, bread, biscuits (Boon et al. 2005; Larsson et al. 2009a, b, c, d; Burley et al. 2010; Lin et al. 2011; EFSA 2014). But at the same time availability of food products with higher levels of acrylamide due to inferior raw material and or technological limitations during processing in developing and poor countries is also a cause of worry. Acrylamide is present in varieties of food besides in chips and French fries (Swedish National Food Administration 2002a, b; Codex Alimentarius Commission 2004; Gokmen and Senyuva 2006a). So, the threat of acrylamide causing cancer, neurotoxicity and other health issues should not be viewed with respect to the consumption of chips and French fries only.
Conclusions
In the last decade, India has made visible progress not only in the production and productivity of potatoes, but also in channelizing the surplus potatoes into processing. In India, market for processed potato products is growing at the rate of 15–20% annually (Virendra Pandit 2015a). Potato chips are the major processed product in India and French fries, potato flakes and Alu Bhujia are the other processed potato products in the country. In India, presently, Gujarat is the state where processing varieties are grown on a large scale to meet the requirement of potato processing industries for chips and French fries. But still, processing varieties account for less than 10% of the total potato production in the state (Vivan Fernandes 2015). At national level, more than 2.8 million metric tonnes of potatoes are processed and this is about 7.5% of the total potato production in India (Gupta et al. 2014). This figure is expected to increase further to more than 10% by 2020 in view of growing demand for diverse potato products. The expected demand for processing potatoes in India will be about 25 million metric tonnes by 2050 (CPRI 2013b). This demand is expected to be met from increase in area as well as productivity of potato in India. Development of suitable varieties, increase in cold storage capacity, efficient storage technologies, setting up of more number of small and big processing units in country will contribute in enhancing the growth of potato processing sector in India. Further, the government of India proposes to set up 30 mega food parks in the country. Additionally, Big Mac is investing in India to open 250 new restaurants in the next 3–5 years and its potato supplier, McCain Foods India has plan to double the contract farming area for potato in Gujarat state (Virendra Pandit 2015a; Vivan Fernandes 2015). Today, potato chips and French fries available in the Indian market or food courts are of international standards. This indicates minimum and acceptable levels of acrylamide in these products. With the advancements in different aspects of potato production and processing, still higher level of food safety standards can be met for processed potato products.
Future perspectives
Today, at global level, potato is the 4th major food crop after rice, wheat and maize. Potato crop is being grown in nearly 155 countries and more than a billion people worldwide consume potatoes (FAOSTAT 2015). In future, potatoes will continue to play a major role in achieving food and nutritional security (Pandey et al. 2005; IPY 2008, Lutaladio and Castaidi 2009; Topcu et al. 2010; Singh and Rana 2013). Efforts made in breeding for the development of potato varieties that can tolerate CIS even under prolonged storage (up to 9 months) at low temperature (4 °C) are expected to yield good results in the near future. In addition to this, considerable molecular and biochemical work done has also enhanced our understanding of CIS leading to the possibility of developing new varieties/transgenics. This will help in lowering acrylamide content in processed potato products. It would be desirable to make use of pre-breeding, traditional and marker-assisted breeding for the development of varieties tolerant to CIS. But, at the same time it is also true that storing the potatoes for longer duration at 4 °C instead of 8–12 °C will increase the demand for electricity. As per an estimate, storing potatoes at 4 °C is about 40% costlier than storing them at 8–12 °C (CPRI 2013a). Due to higher cost and problems pertaining to the continuous availability and accessibility of electricity, this storage option may be less preferred in developing and third world countries. Storage of potatoes at 8–12 °C will require effective and judicious use of sprout suppressant/s. There will also be a demand for the use of more safe, effective and natural sprout suppressants that can be either used alone or as supplement to the existing sprout suppressant (CIPC) to keep the residue levels of sprout suppressants in the potatoes within acceptable levels. Other future lines of research work should include extending the dormancy period, from the present duration of 2 to about 4 months or even more. This will improve storability and also assist in maintaining the quality as required for processing. Developing varieties that have lower levels of reducing sugars at harvest and after short- and long-term storage should be among the top priorities.
Author contribution statement
VP and RE contributed by research and writing while, RP contributed in writing of manuscript.
References
Amrein TM, Bachmann S, Noti A, Biedermann M, Barbosa MF, Biedermann-Brem S, Grob K, Keiser A, Realini P, Escher F, Amado R (2003) Potential of acrylamide formation, sugars, and free asparagine in potatoes: a comparison of cultivars and farming systems. J Agric Food Chem 51:5556–5560
Amrein TM, Schonbachler B, Rohner F, Lukac H, Schneider H, Keiser A, Escher F, Amado R (2004) Potential for acrylamide formation in potatoes: data from the 2003 harvest. Eur Food Res Technol 219:572–578
Anese M, Suman M, Nicoli MC (2009) Technological strategies to reduce acrylamide levels in heated foods. Food Eng Rev 1:169–179
Anese M, Suman M, Nicoli MC (2010) Acrylamide removal from heated foods. Food Chem 119:791–794
Anonymous (2003) Information on ways to lower the levels of acrylamide formed in food. Note of the meeting of experts on industrial contaminants in food. Acrylamide workshop, 20–21 October, 2003. http://ec.europa.eu/food/food/chemicalsafety/contaminants/acryl_guidance.pdf. Accessed 10 Jan 2015
Anonymous (2009) Effect of frying oil on acrylamide formation in potatoes. Dartmouth under graduate. J Sci. http://dujs.dartmouthedu/winter-2009/research-effects-of-frying-oil-on. Accessed 15 April 2015
Anonymous (2010) Toxicological review of acrylamide (CAS No. 79-06-1), EPA/635/R-0. http://niehs.nih.gov/ntp/roc/eleventh/profiles/s003acry.pdf. Accessed 15 April 2015
Anonymous (2011) A “Toolbox” for the reduction of acrylamide in fried potato products/French fries. http://ec.europa.eu/food/food/chemicalsafety/contaminants/acrylamide/frenchfries-EN-final.pdf; http://www.fooddrinkeurope.eu/publication/fooddrinkeurope-updates-industry-wide-toolbox-to-help-manufacturers-further/. Accessed 8 Sept 2015
Anonymous (2016) Food regulator Australia and New Zealand is reviewing Simplot’s GMO potato. http://potatopro.com/news/2016/food-regulator-australia-and-new-zealand-reviewing-simplots-gm. Accessed 30 Aug 2016
Baardseth P, Blom H, Skrede G, Mydland LT, Skrede A, Slinde E (2006) Lactic acid fermentation reduces acrylamide formation and other Maillard reactions in French fries. J Food Sci 71:C28–C33
Balaji V, Chandra S, Goswami DA, Das SK, Mandal TK, Chakraborty AK, Bhattacharyya A (2006) Toxicokinetics, metabolism, and microsomal studies of chlorpropham in rats. Toxicol Environ Chem 88:527–539
Baldwin SJ, Dodds KG, Auvray B, Genet RA, Macknight RC, Jacobs JME (2011) Association mapping of cold-induced sweetening in potato using historical phenotypic data. Ann Appl Biol 158:248–256
Bassama J, Brat P, Bohuon P, Boulanger R, Gunata Z (2010) Study of acrylamide mitigation in model system: effect of pure phenolic compounds. Food Chem 123:558–562
Becalski A, Lau BPY, Lewi D, Seaman SW (2003) Acrylamide in foods: occurrence, source and modeling. J Agric Food Chem 51:802–808
Becalski A, Lau BPY, Lewis D, Seaman SW, Hayward S, Sahagian M, Ramesh M, Leclerc Y (2004) Acrylamide in French fries: influence of free amino acids and sugars. J Agric Food Chem 52:3801–3806
Besaratinia A, Pfeifer GP (2007) A review of mechanisms of acrylamide carcinogenicity. Carcinogenesis 28:519–528
Bhardwaj V, Luthra SK, Dalamu Singh BP, Kumar V, Dinesh Kumar, Sharma S (2013) SS1735-02 (IC0594469; INGR13048) a hexaploid wild potato clone (Solanum demissum) with high resistance against late blight and low cold induced sweetening even after 6 months of cold storage (2–4 °C). Indian J Plant Genet Resour 26:260–261
Bhaskar PB, Wu L, Busse JS, Whitty BR, Hamernik AJ, Jansky SH, Buell CR, Bethke PC, Jiang J (2010) Suppression of the vacuolar invertase gene prevents cold-induced sweetening in potato. Plant Physiol 154:939–948
Biedermann M, Noti A, Bidermann-Bren S, Mozzetti V, Grob K (2002) Experiments on acrylamide formation and possibilities to decrease the potential of acrylamide formation in potatoes. Mitt Lebensm Hyg 93:668–687
Biedermann M, Grundbock F, Fiselier K, Biedermann S, Burgi C, Grob K (2010) Acrylamide monitoring in Switzerland, 2007–2009: results and conclusions. Food Addi Contam Part A-Chem Anal Control Expo Risk Assess 27:1352–1362
Blenkinsop RW, Copp LJ, Yada RY, Marangoni AG (2002) Effect of chlorpropham (CIPC) on carbohydrate metabolism of potato tubers during storage. Food Res Int 35:651–655
Boon PE, de Mul A, van der Voet H, van Donkersgoed G, Brette M, van Klaveren JD (2005) Calculations of dietary exposure to acrylamide. Mutat Res 580:143–155
Borda T, Alexe P (2011) Acrylamide levels in food. Romanian J Food Sci 1:3–15
Brajesh S, Ezekiel R (2010) Isopropyl n-(3-chlorophenyl) carbamate (CIPC) residues in potatoes stored in commercial cold stores in India. Potato Res 53:111–120
Brown R (2003) Formation, occurrence and strategies to address acrylamide in food. FDA Food Advisory Committee Meeting on Acrylamide, February 24–45, 2003. University of Maryland, College Park, Maryland. http://www.cfsan.fda.gov/~dms/acrybrow.html. Accessed 8 May 2015
Brummell DA, Chen RKY, Harris JC, Zhang H, Hamiaux C, Kralicek AV, McKenzie MJ (2011) Induction of vacuolar invertase inhibitor mRNA in potato tubers contributes to cold-induced sweetening resistance and includes spliced hybrid mRNA variants. J Exp Bot 62:3519–3534
Burley VJ, Greenwood DC, Hepworth SJ, Fraser LK, de Kok TM, van Breda SG, Kyrtopoulos SA, Botsivali M, Kleinjans J, McKinney PA, Cade JE (2010) Dietary acrylamide intake and risk of breast cancer in the UK women’s cohort. Br J Cancer 103:1749–1754
Burton WG (1989) The potato, 3rd edn. Longman Scientific and Technical Publishers, Essex
Burton WG, van Es A, Hartmans KJ (1992) The physics and physiology of storage. In: Paul H (ed) The potato crop. Chapman and Hall, London, pp 608–727
Campbell MA, Gleichsner A, Alsbury R, Horvath D, Suttle J (2010) The sprout inhibitors chlorpropham and 1,4-dimethylnaphthalene elicit different transcriptional profiles and do not suppress growth through a prolongation of the dormant state. Plant Mol Biol 73:181–189
Chauhan SS, Prakash O, Padalia RC, Vivekanand Pant AK, Mathela CS (2011) Chemical diversity in Mentha spicata: antioxidant and potato sprout inhibition activity of its essential oils. Nat Prod Commun 6:1373–1378
Chen X, Song B, Liu J, Yang J, He T, Lin Y, Zhang H, Xie C (2012) Modulation of gene expression in cold-induced sweetening resistant potato species Solanum berthaultii exposed to low temperature. Mol Genet Genomics 287:411–421
Cheng KW, Shi JJ, Ou SY, Wang MF, Jiang Y (2010) Effects of fruit extracts on the formation of acrylamide in model reactions and fried potato crisps. J Agric Food Chem 58:309–312
CIAA (2009) The CIAA acrylamide toolbox Rev. 12 http://www.ciaa.be/documents/brochures/ac_toolbox_20090216.pdf. Accessed 8 May 2015
Ciesarova Z, Kiss E, Boegl P (2006) Impact of l-asparaginase on acrylamide content in potato products. J Food Nutr Res 45:141–146
Claeys W, De Vleeschouwer K, Hendrickx ME (2005) Effect of amino acids on acrylamide formation and elimination kinetics. Biotechnol Prog 21:1525–1530
Codex (2009) Code of practice for the reduction of acrylamide in foods (CAC/RCP 67-2009). http://www.codexalimentarius.net/download/standards/11258/CXP_067e.pdf. Accessed 15 May 2015
Codex Alimentarius Commission (2004) Discussion paper on acrylamide. Joint FAO/WHO food standard programme. Codex committee on food additives and contaminants. Thirty-sixth Session, Rotterdam, The Netherlands, 22–26 March 2004
Coffin RH, Yada RY, Parkin KL, Grodzinski B, Stanley DW (1987) Effect of low temperature storage on sugar concentrations and chip color of certain processing potato cultivars and selections. J Food Sci 52:639
Cools K, del Carmen AM, Terry LA (2014) Controlling sprouting in potato tubers using ultraviolet-C irradiance. Postharvest Biol Technol 98:106–114
Costa LG, Deng H, Greggotti C, Manzo L, Faustman EM, Bergmark E, Calleman CJ (1992) Comparative studies on the neuro and reproductive toxicity of acrylamide and its epoxide metabolite glycidamide in the rat. Neurotoxicology 13:219–224
CPRI Annual Report (2009–2010) Central Potato Research Institute (CPRI), Shimla, India, pp 21, 35, 41–42,118–119
CPRI Annual Report (2010–2011) Central Potato Research Institute (CPRI), Shimla, India, pp 53–54
CPRI Annual Report (2011–2012) Central Potato Research Institute (CPRI), Shimla, India, pp 13, 112–113
CPRI (2013a) Significant outputs and outcomes during last five years. http://cpri.ernet.in/res_ach/res_ach.pdf. Accessed 27 Aug 2015
CPRI (2013b) Vision—2050. Central Potato Research Institute (CPRI), Shimla, India
Cummins E, Butler F, Brunton N, Gormley R (2006) Factors affecting acrylamide formation in processed potato products—a simulation approach. IUFoST. doi:10.1051/IUFoST:20060719. http://iufost.edpsciences.org. Accessed 22 Feb 2015
De Meulenaer B, De Wilde T, Mestdagh F, Govaert Y, Ooghe W, Fraselle S, Demeulemeester K, Van Peteghem C, Calus A, Degroodt J-M, Verhe R (2008) Comparison of potato varieties between seasons and their potential for acrylamide formation. J Sci Food Agric 88:313–318
De Wilde T, De Meulenaer B, Mestdagh F, Govaert Y, Vandeburie S, Ooghe W, Fraselle S, Demeulemeester K, Van Peteghem C, Calus A, Degroodt J-M, Verhe R (2005) Influence of storage practices on acrylamide formation during potato frying. J Agric Food Chem 53:6550–6557
De Wilde T, De Meulenaer B, Mestdagh F, Govaert Y, Vandeburie S, Ooghe W, Fraselle S, Demeulemeester K, Van Peteghem C, Calus A, Degroodt JM, Verhe R (2006) Influence of fertilization on acrylamide formation during frying of potatoes harvested in 2003. J Agric Food Chem 54:404–408
Dearfield KL, Abernathy CO, Ottley MS, Brantner JH, Hayes PF (1988) Acrylamide: its metabolism, developmental and reproductive effects, genotoxicity, and carcinogenicity. Mutat Res 195:45–77
Dearfield KL, Douglas GR, Ehling UH, Moore MM, Sega GA, Brusick DJ (1995) Acrylamide: a review of its genotoxicity and an assessment of heritable genetic risk. Mutat Res 330:71–99
Deryck DP (2007) Acrylamide in fried, baked or roasted foods may increase risk of cancer in susceptible individuals. Internet J Food Saf 9:14–16
Doerge DR, da Costa GG, McDaniel LP, Churchwell MI, Twaddle NC, Beland FA (2005) DNA adducts derived from administration of acrylamide and glycidamide to mice and rats. Mutat Res 580:131–141
Dybing E, Sanner T (2003) Risk assessment of acrylamide in foods. Toxicol Sci 75:7–15
EC (2009) Food contaminants—acrylamide brochures. http://www.ec.europa.eu/food/food/chemicalsafety/contaminants/acrylamide_en.htm. Accessed 18 Oct 2015
EC (2011) European Commission Recommendation of 10 January 2011 on investigations into the levels of acrylamide in food. http://www.ec.europa.eu/food/food/chemicalsafety/contaminants/recommendation_10012011_acrylamide_food_en.pdf. Accessed 18 Oct 2015
EFSA (2003) Workshop on acrylamide formation in food. Report of the workshop. AF 06.04.2004–2010, 17 Nov Brussels
EFSA (2005) Opinion of the scientific committee on a request from EFSA related to a harmonized approach for risk assessment of substances which are both genotoxic and carcinogenic (Request No. EFSA-Q-2004-020). EFSA J 282:1–31. http://www.efsa.europa.eu/sites/default/files/scientific_output/files/main_documents/282.pdf. Accessed 17 June 2015
EFSA (2011) Scientific Report of EFSA—results on acrylamide levels in food from monitoring years 2007–2009 and exposure assessment. EFSA J 9:2133 [48 pp]. http://www.efsa.europa.eu/sites/default/files/scientific_output/files/main_documents/2133.pdf. Accessed 22 Oct 2015
EFSA (2014) Draft scientific opinion on acrylamide in food. http://www.efsa.europa.eu/en/consultations/call/140701.htm. Accessed 22 Oct 2015
EFSA (2015) Acrylamide. http://www.efsa.europa.eu/en/topics/topic/acrylamide. Accessed 25 Oct 2015
El-Awady Aml A, Moghazy AM, Gouda AEA, Elshatoury RSA (2014) Inhibition of sprout growth and increase storability of processing potato by antisprouting agent. Trends Hortic Res 4:31–40
Elmore JS, Parker JK, Halford NG, Muttucumaru N, Mottram DS (2008) Effects of plant sulphur nutrition on acrylamide and aroma compounds in cooked wheat. J Agric Food Chem 56:6173–6179
Elmore S, Briddon A, Dodson A, Muttucumaru N, Halford N, Mottram D (2015) Acrylamide in potato crisps prepared from 20 UK-grown varieties: effects of variety and tuber storage time. Food Chem 182:1–8
EU (2003) Information on ways to lower the levels of acrylamide formed in food (note of the meeting of experts on industrial contaminants in food: acrylamide workshop, 20–21 Oct 2003. http://europa.eu.int/comm/food/food/chemicalsafety/contaminants/acryl_guidance.pdf. Accessed 17 June 2015
EU Scientific Committee Report (2002) Report from the EU Scientific Committee of the Norwegian Food Control Authority: Risk assessment of acrylamide intake from foods with special emphasis on cancer risk, 6 June 2002. http://www.snt.no/nytt/tema/Akrylamid/acrylamide.pdf. Accessed 11 May 2015
European Commission (2011) Final commission recommendation of 10.1.2011 on investigations into the levels of acrylamide in food, Brussels
Everts S (2012) The Maillard reaction turns 100. Chem Eng News 90:58–60
Ezekiel R (2015) Potato. In: Chadha KL, Pal RL (eds) Managing postharvest quality and losses in horticultural crops. Daya Publishing House, New Delhi, pp 727–755
Ezekiel R, Dahiya PS (2004) Storage behaviour and processing quality of potatoes stored in heaps and pits in the Malwa region (MP). Potato J 31:21–28
Ezekiel R, Singh B (2007) Effect of cooking and processing on CIPC residue concentrations in potatoes and processed potato products. Potato Res 50:175–184
Ezekiel R, Paul V, Singh J, Shekhawat GS (1999a) Potato storage in India. Indian Farm 49:21–25
Ezekiel R, Verma SC, Sukumaran NP, Shekhawat GS (1999b) A guide to potato processors in India. Technical Bulletin No. 48. CPRI, Shimla, p 38
Ezekiel R, Singh B, Kumar D, Paul V (2003) Processing quality and CIPC residue in potatoes stored at 10–12 °C in commercial cold stores applying CIPC. Indian J Plant Physiol (Special Issue) 489–494
Ezekiel R, Mehta A, Singh B, Kumar D, Kumar NR, Paul V, Das M (2005a) CIPC [isopropyl N-(3-chlorophenyl) carbamate] for sprout suppression in potatoes during storage. Technical Bulletin No. 69, Central Potato Research Institute (CPRI), Shimla, HP, India
Ezekiel R, Singh B, Kumar D, Paul V, Das M (2005b) Storage of potatoes at 10–12 °C with CIPC treatment in commercial cold stores. Potato J 32:211–212
Ezekiel R, Rani M, Kumar D (2007a) Chipping quality of potatoes stored at 8–20 °C under controlled conditions. Potato J 34:174–179
Ezekiel R, Singh B, Kumar D, Mehta A (2007b) Processing qualities of potato varieties grown at two locations and stored at 4, 10 and 12 °C. Potato J 34:164–173
Ezekiel R, Singh B, Datta PS (2008) Chipping quality of γ-irradiated potatoes of three Indian cultivars stored at 8, 12 and 16 °C. J Food Sci Technol 43:36–43
FAOSTAT (2015) http://faostat3.fao.org/download/Q/QC/E. Accessed 20 Nov 2015
FDA (2004) FDA releases acrylamide data and final acrylamide action plan. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2004/ucm108270.htm. Accessed 22 Oct 2015
Fiselier K, Bazzocco D, Gama-Baumgartner F, Grob K (2006) Influence of the frying temperature on acrylamide formation in French fries. Eur Food Res Technol 222:414–419
Fohgelberg P, Rosen J, Hellenas KE, Abramsson-Zetterberg L (2005) The acrylamide intake via some common baby food for children in Sweden during their first year of life- an improved method for analysis of acrylamide. Food Chem Toxicol 43:951–959
Food Drink Europe (2011) Food Drink Europe Acrylamide Toolbox. http://fooddrinkeurope.eu/uploads/publications_documents/Toolboxfinal260911.pdf. Accessed 1 Nov 2015
Food Drink Europe Acrylamide Toolbox (2013) http://ec.europa.eu/food/food/chemicalsafety/contaminants/toolbox_acrylamide_201401_en.pdf. Accessed 1 Nov 2015
Friedman M (2003) Chemistry, biochemistry, and safety of acrylamide. A review. J Agric Food Chem 51:4504–4526
Friedman MA, Dulak LH, Stedham MA (1995) A lifetime oncogenicity study in rats with acrylamide. Fundam Appl Toxicol 27:95–105
Gama-Baumgartner F, Grob K, Biedermann M (2004) Citric acid to reduce acrylamide formation in French fries and roasted potatoes. Mitteilungen aus Lebensmitteluntersuchung und Hygiene 95:110–117
Gamboa da Costa G, Churchwell MI, Hamilton LP, Von Tungeln LS, Beland FA, Marques MM, Doerge DR (2003) DNA adduct formation from acrylamide via conversion to glycidamide in adult and neonatal mice. Chem Res Toxicol 16:1328–1337
Ganguly U (2013) Contract farming: Can it be a vehicle for inclusive growth? Review and synthesis of various case studies on contract farming in India. http://www.vlsa.glsl.org/sites/default/files/gla/document/Final%20Report-CF%20-%202013.pdf. Accessed 25 Nov 2015
Gaur PC, Pandey SK, Singh SV, Devendra Kumar, Marwaha RS, Dinesh Kumar (1998) Kufri Chipsona-1: a potato variety for processing. J Indian Potato Assoc 25:115–118
Gaur PC, Singh SV, Pandey SK, Kumar Marwaha RS, Devendra Kumar Dinesh (1999) Kufri Chipsona-2: a new potato variety for chipping. Curr Sci 76:722–724
Gautam IP, Sharma MD, Khatri BB, Thapa RB, Shrestha K (2013) Storability and chips quality of chemical treated potatoes under ordinary condition. J Basic Appl Sci 9:1–10
Gerendas J, Heuser F, Sattelmacher B (2007) Influence of nitrogen and potassium supply on contents of acrylamide precursors in potato tubers and on acrylamide accumulation in French fries. J Plant Nutr 30:1499–1516
Ghanayem BI, Witt KL, Kissling GE, Tice RR, Recio L (2005) Absence of acrylamide-induced genotoxicity in CYP2E1-null mice: evidence consistent with a glycidamide-mediated effect. Mutat Res 578:284–297
Gobel A, Kliemant A (2007) The German minimization concept for acrylamide. Food Addit Contam 24:82–90
Gokmen V, Mogol BA (2010) Computer vision-based image analysis for rapid detection of acrylamide in heated foods. Qual Assur Saf Crops Foods 2:203–207
Gokmen V, Papazoglu TK (2008) Acrylamide formation in food during thermal processing with a focus on frying. Food Bioprocess Technol 1:35–42
Gokmen V, Senyuva HZ (2006a) Study of colour and acrylamide formation in coffee, wheat flour and potato chips during heating. Food Chem 99:238–243
Gokmen V, Senyuva HZ (2006b) A simplified approach for the kinetic characterization of acrylamide formation in fructose—asparagine model system. Food Addit Contam 23:348–354
Gokmen V, Senyuva HZ (2007) Acrylamide formation is prevented by divalent cations during the Maillard reaction. Food Chem 103:196–203
Gokmen V, Palazoglu TK, Senyuva HZ (2006a) Relation between the acrylamide formation and time–temperature history of surface and core regions of French fries. J Food Eng 77:972–976
Gokmen V, Senyuva HZ, Dulek B, Cetin AE (2006b) Computer vision based analysis of potato chips—a tool for rapid detection of acrylamide level. Mol Nutr Food Res 50:805–810
Gokmen V, Akbudak B, Serpen A, Acar J, Turan ZM, Eris A (2007) Effects of controlled atmosphere storage and low-dose irradiation on potato tuber components affecting acrylamide and color formations upon frying. Eur Food Res Technol 224:681–687
Gomez-Castillo D, Cruz E, Iguaz A, Arroqui C, Virseda P (2013) Effect of essential oils on sprout suppression and quality of potato cultivars. Postharvest Biol Technol 82:15–21
Granda C, Moreira RG (2005) Kinetics of acrylamide formation during traditional and vacuum frying of potato chips. J Food Process Eng 28:478–493
Granda C, Moreira RG, Tichy SE (2004) Reduction of acrylamide formation in potato chips by low-temperature vacuum frying. J Food Sci 69:405–411
Greiner S, Rausch T, Sonnewald U, Herbers K (1999) Ectopic expression of a tobacco invertase inhibitor homolog prevents cold-induced sweetening of potato tubers. Nat Biotechnol 17:708–711
Grob K (2007) Options for legal measures to reduce acrylamide contents in the most relevant foods. Food Addit Contam 24:71–81
Grob K, Biedermann M, Biedermann B, Noti A, Imhof D, Amrein T, Pfefferl A, Bazzocco D (2003) French fries with less than 100 μg/kg acrylamide. A collaboration between cooks and analysts. Eur Food Res Technol 217:185–194
Gupta SK, Sowokinos JR, Hahn In-Su (2008) Regulation of UDP-glucose pyrophosphorylase isozyme UGP5 associated with cold-sweetening resistance in potatoes. J Plant Physiol 165:679–690
Gupta VK, Luthra SK, Singh BP (2014) Potato processing varieties: Present status and future thrusts. National Seminar on “postharvest management and processing of potato for increasing food security in India” 22 Sept 2014. UAS Campus, Dharwad, Karnataka, India. https://www.researchgate.net/publication/273120681_Potato_processing_varieties_Present_status_and_future_thrusts. Accessed 10 Nov 2015
Haase NU, Matthaeus B, Vosmann K (2003) Acrylamide formation in foodstuffs—minimizing strategies for potato crisps. Dtsch Lebensm-Rundsch 99:87–90
Halford NG, Muttucumaru N, Powers SJ, Gillatt PN, Hartley L, Elmore JS, Mottram DS (2012a) Concentrations of free amino acids and sugars in nine potato varieties: effects of storage and relationship with acrylamide formation. J Agric Food Chem 60:12044–12055
Halford NG, Curtis TY, Muttucumaru N, Postles J, Elmore JS, Mottram DS (2012b) The acrylamide problem: a plant and agronomic science issue. J Exp Bot 63:2841–2851
He T, Song B, Liu J, Chen X, Ou Y, Lin Y, Zhang H, Xie C (2012) A new isoform of thioredoxin h group in potato, SbTRXh1, regulates cold-induced sweetening of potato tubers by adjusting sucrose content. Plant Cell Rep 31:1463–1471
Hoenicke K, Gatermann R (2004) Stability of acrylamide in food during storage. Czech J Food Sci 22:355–356
Hoenicke K, Gatermann R (2005) Studies on the stability of acrylamide in food during storage. J AOAC Int 88:268–273
Hogervorst JG, Schouten LJ, Konings EJ, Goldbohm RA, van den Brandt PA (2007) A prospective study of dietary acrylamide intake and the risk of endometrial ovarian, and breast cancer. Cancer Epidemiol Biomark Prev 16:2304–2313
Hogervorst JG, Schouten LJ, Konings EJ, Goldbohm RA, van den Brandt PA (2008) Dietary acrylamide intake and the risk of renal cell, bladder, and prostate cancer. Am J Clin Nutr 87:1428–1438
IARC (International Agency for Research on Cancer) (1994) Acrylamide. IARC monographs on the evaluation of the carcinogenic risk of chemicals to humans, 60, IARC, Lyon, France, pp 389–433
IPY (2008) International year of the potato 2008. http://www.potato2008.org. Accessed 21 Oct 2015
Jansky SH, Fajardo DA (2014) Tuber starch amylose content is associated with cold-induced sweetening in potato. Food Sci Nutr 2:628–633
Jin C, Wu X, Zhang Y (2013) Relationship between antioxidants and acrylamide formation: a review. Food Res Int 51:611–620
Jung MY, Choi DS, Ju JW (2003) A novel technique for limitation of acrylamide formation in fried and baked corn chips and in French fries. J Food Sci 68:1287–1290
Kita A, Brathen E, Knutsen S, Wicklund T (2004) Effective ways of decreasing acrylamide content in potato crisps during processing. J Agric Food Chem 52:7011–7016
Klaassen CD, Amdur MO, Doull J (1986) Casarett and Doull’s toxicology: the basic science of poisons, 3rd edn. Macmillan, New York
Knutsen SH, Dimitrijevic S, Molteberg EL, Segtnan VH, Kaaber L, Wicklund T (2009) The influence of variety, agronomical factors and storage on the potential for acrylamide formation in potatoes grown in Norway. LWT-Food Sci Technol 42:550–556
Konings EJ, Baars AJ, van Klaveren JD, Spanjer MC, Rensen PM, Hiemstra M, van Kooij JA, Peters PW (2003) Acrylamide exposure from foods of the Dutch population and an assessment of the consequent risks. Food Chem Toxicol 41:1569–1579
Kotsiou K, Tasioula-Margari M, Fiore A, Gokmen V, Fogliano V (2013) Acrylamide formation and colour development in low-fat baked potato products as influenced by baking conditions and oil type. Eur Food Res Technol 236:843–851
Kumar D (2011) Cold induced sweetening development in Indian potato (Solanum tuberosum L.) varieties. Indian J Biochem Biophys 48:123–127
Kumar D, Ezekiel R (2005) Changes in sugar content and processing quality of potatoes during storage and reconditioning. J Food Sci Technol 42:400–404
Kumar D, Ezekiel R (2006) Heap storage - An economical and environmentally friendly technology for storing chipping potatoes under sub-tropical climate. J Food Sci Technol 43:555–557
Kumar A, Pandey SK (2008) Impact of potato research in India. In: Souvenir global potato conference, pp 9–12
Kumar D, Singh BP, Kumar P (2004) An overview of the factors affecting sugar content of potatoes. Ann Appl Biol 145:247–256
Kumar D, Paul V, Ezekiel R (2005) Chipping quality of potatoes stored in heap and pits in sub-tropical plains of India. Hortic Sci (Prague) 32:23–30
Kyriacou MC, Ioannides IM, Gerasopoulos D, Siomos AS (2009) Storage profiles and processing potential of four potato (Solanum tuberosum L.) cultivars under three storage temperature regimes. J Food Agric Environ 7:31–37
Larsson SC, Akesson A, Wolk A (2009a) Long-term dietary acrylamide intake and breast cancer risk in a prospective cohort of Swedish women. Am J Epidemiol 169:376–381
Larsson SC, Akesson A, Wolk A (2009b) Dietary acrylamide intake and prostate cancer risk in a prospective cohort of Swedish men. Cancer Epidemiol Biomark Prev 18:1939–1941
Larsson SC, Akesson A, Wolk A (2009c) Long-term dietary acrylamide intake and risk of epithelial ovarian cancer in a prospective cohort of Swedish women. Cancer Epidemiol Biomark Prev 18:994–997
Larsson SC, Hakansson N, Akesson A, Wolk A (2009d) Long-term dietary acrylamide intake and risk of endometrial cancer in a prospective cohort of Swedish women. Int J Cancer 124:1196–1199
Li L, Paulo MJ, Strahwald J, Lubeck J, Hofferbert HR, Tacke E, Junghans H, Wunder J, Draffehn A, van Eeuwijk F, Gebhardt C (2008) Natural DNA variation at candidate loci is associated with potato chip color, tuber starch content, yield and starch yield. Theor Appl Genet 116:1167–1181
Lin YL, Lagergren J, Lu YX (2011) Dietary acrylamide intake and risk of esophageal cancer in a population-based case-control study in Sweden. Int J Cancer 128:676–681
Lindsay RC, Jang S (2005) Model systems for evaluating factors affecting acrylamide formation in deep fried foods. Adv Exp Med Biol 561:329–341
Lineback DR, Coughlin JR, Stadler RH (2012) Acrylamide in foods: a review of the science and future considerations. Annu Rev Food Sci Technol 3:15–35
LoPachin RM (2004) The changing view of acrylamide neurotoxicity. Neurotoxicology 25:617–630
LoPachin RM, Gavin T (2012) Molecular mechanism of acrylamide neurotoxicity: lessons learned from organic chemistry. Environ Health Perspect 120:1650–1957
Lopachin RM, Lehning EJ (1994) Acrylamide induced distal axon degeneration. A proposed mechanism of action. Neurotoxicology 15:247–260
Low MY, Koutsidis G, Parker JK, Elmore JS, Dodson AT, Mottram DS (2006) Effect of citric acid and glycine addition on acrylamide and flavor in a potato model system. J Agric Food Chem 54:5976–5983
Lutaladio N, Castaidi L (2009) Potato: the hidden treasure. J Food Comp Anal 22:491–493
Luthra SK, Gopal J, Kumar D, Singh BP, Pandey SK (2009) Solanum wild and cultivated species as source of resistance to cold induced sweetening. Potato J 36:115–120
Lyn-Cook LE, Tareke E, Word B, Starlard-Davenport A, Lyn-Cook BD, Hammons GJ (2011) Food contaminant acrylamide increases expression of Cox-2 and nitric oxide synthase in breast epithelial cells. Toxicol Ind Health 27:11–18
Mahajan BVC, Dhatt AS, Sandhu KS, Garg A (2008) Effect of CIPC (isopropyl-N (3-chlorophenyl) carbamate) on storage and processing quality of potato. J Food Agric Environ 6:34–38
Maillard LC (1912) Action of amino acids on sugars. Formation of melanoidins in a methodical way. Comptes rendus de l’Academie des Sci 154:66–68
Majcher MA, Jelen HH (2007) Acrylamide formation in low-fat potato snacks and its correlation with colour development. Food Addit Contam 24:337–342
Mamani Moreno C, Stadler T, Da Silva AA, Barbosa LCA, De-Queiroz MELR (2012) Determination of maleic hydrazide residues in garlic bulbs by HPLC. Talanta 89:369–376
Manivel P, Pandey SK, Singh SV, Kumar D (2007) Pattern of chip colour segregation in some crosses of potato. Potato J 34:49–50
Manson J, Brabec MJ, Buelke-Sam J, Carlson GP, Chapin RE, Favor JB, Fischer LJ, Hattis D, Lees PS, Perreault-Darney S, Rutledge J, Smith TJ, Tice RR, Working P (2005) NTP-CERHR expert panel report on the reproductive and developmental toxicity of acrylamide. Birth Defects Res (Part B) Dev Reprod Toxicol 74:17–113
Marth PC, Schultz ES (1952) A new sprout inhibitor for potato tubers. Am Potato J 29:268–272
Marwaha RS, Pandey SK, Singh SV, Minhas JS (2005) Chipping performance of advanced potato hybrids and new varieties before and after on-farm storage. Potato J 32:141–142
Marwaha RS, Pandey SK, Singh SV, Kumar D (2007) Yield, chipping and nutritive qualities of spring grown potatoes in north-western plating. Potato J 34:61–62
Marwaha RS, Kumar D, Singh SV, Pandey SK (2008) Influence of blanching of slices of potato varieties on chipping quality. J Food Sci Technol 45:364–367
Marwaha RS, Pandey SK, Kumar Dinesh, Singh SV, Kumar Parveen (2010) Potato processing scenario in India: industrial constraints, future projections, challenges ahead and remedies—a review. J Food Sci Technol 47:137–156
Matsuura-Endo C, Ohara-Takada A, Chuda Y, Ono H, Yada H, Yoshida M, Kobayashi A, Tsuda S, Takigawa S, Noda T, Yamauchi H, Mori M (2006) Effects of storage temperature on the contents of sugars and free amino acids in tubers from different potato cultivars and acrylamide in chips. Biosci Biotechnol Biochem 70:1173–1180
Matthaus B, Haase NU, Vosmann K (2004) Factors affecting the concentration of acrylamide during deep-fat frying of potatoes. Eur J Lipid Sci Technol 106:793–801
McKenzie MJ, Chen RK, Harris JC, Ashworth MJ, Brummell DA (2013) Posttranslational regulation of acid invertase activity by vacuolar invertase inhibitor affects resistance to cold-induced sweetening of potato tubers. Plant Cell Environ 36:176–185
Medeiros Vinci R, Mestdagh F, De Muer N, Van Peteghem C, De Meulenaer B (2010) Effective quality control of incoming potatoes as an acrylamide mitigation strategy for the French fries industry. Food Addit Contam Part A-Chem Anal Control Expos Risk Assess 27:417–425
Medeiros Vinci R, Mestdagh F, Van Poucke C, Kerkaert B, de Muer N, Denon Q, Van Peteghem C, De Meulenaer B (2011) Implementation of acrylamide mitigation strategies on industrial production of French fries: challenges and pitfalls. J Agric Food Chem 59:898–906
Medeiros Vinci R, Mestdagh F, De Meulenaer B (2012) Acrylamide formation in fried potato products—present and future, a critical review on mitigation strategies. Food Chem 133:1138–1154
Mehta A, Ezekiel R (2006) Potato storage: needs, present scenario, emerging technology and future strategies—a critical appraisal. J Food Sci Technol 43:453–466
Mehta A, Ezekiel R (2010) Non-refrigerated storage of potatoes. Potato J 37:87–99
Mehta A, Kaul HA (2002) Evaluation of menthol and menthe oils as potato sprout inhibitors. J Indian Potato Assoc 29:107–112
Mehta A, Singh B, Kumar D, Ezekiel R (2007) Evaluation of CIPC spray for sprout inhibition in potatoes under traditional storage methods. Potato J 34:69–70
Mehta A, Singh B, Ezekiel R, Kumar D (2010) Effect of CIPC on sprout inhibition and processing quality of potatoes stored under traditional storage system. Indian Potato Res 53:1–15
Merck (1989) The Merck Index, 11th edn. Merck & Company, Inc., Rahway
Mestdagh FJ, De Meulenaer B, Van Poucke C, Detavernier C, Cromphout C, Van Peteghem C (2005) Influence of oil type on the amounts of acrylamide generated in a model system and in French fries. J Agric Food Chem 53:6170–6174
Mestdagh F, De Wilde T, Castelein P, Nemeth O, Van Peteghem C, De Meulenae B (2008) Impact of the reducing sugars on the relationship between acrylamide and Maillard browning in French fries. Eur Food Res Technol 227:69–76
Mestdagh F, Van Peteghem C, De Meulenaer B (2009) A farm-to-fork approach to lower acrylamide in fried potatoes. In: Yee N, Bussel WT (eds) IV potatoes (food 3, special issue 2 edn). Global Science Books, pp 66–75
Mills C, Mottram DS, Wedzicha BL (2009) Acrylamide. In: Stadler RH, Lineback DR (eds) Process-induced food toxicants. Wiley, Hoboken, pp 23–50
Mohammed NMS, Flowers TH, Duncan HJ (2014) Development and validation of an HPLC method for the analysis of chlorpropham and 3-choroaniline in potato extract. Chromatogr Res Int. http://www.hindawi.com/journals/cri/contents/. Accessed 14 July 2015 (article ID 108694, 5 pp)
Mohammed NMS, Flowers TH, Duncan HJ (2015) HPLC-UV method for analysis of potato sprout inhibitor chlorpropham and its metabolite 3-chloroaniline in potatoes. IOSR J Environ Sci Toxicol Food Technol 9:78–85
Mottram DS, Wedzicha BL, Dodson AT (2002) Acrylamide is formed in the Maillard reaction. Nature 419:448–449
Mulla MZ, Bharadwaj VR, Annapure US, Singhal RS (2011a) Effect of formulation and processing parameters on acrylamide formation: a case study on extrusion of blends of potato flour and semolina. LWT-Food Sci Technol 44:1643–1648
Mulla MZ, Bharadwaj VR, Annapure US, Variyar PS, Sharma A, Singhal RS (2011b) Acrylamide content in fried chips prepared from irradiated and non-irradiated stored potatoes. Food Chem 127:1668–1672
Muttucumaru N, Powers SJ, Elmore JS, Mottram DS, Halford NG (2013) Effects of nitrogen and sulfur fertilization on free amino acids, sugars and acrylamide-forming potential in potato. J Agric Food Chem 61:6734–6742
Muttucumaru N, Powers SJ, Briddon A, Elmore JS, Mottram DS, Halford NG (2014) Evidence for the complex relationship between the concentrations of free amino acids, sugars and acrylamide-forming potential in potato. Ann Appl Biol 164:286–300
NHB (2014a) Indian Horticulture Database - 2014. Ministry of Agriculture, Government of India, 85 Institutional Area, Sector-18, Gurgaon, 122 015. http://www.nhb.gov.in/area-pro/NHB_Database_2015.pdf. Accessed 15 Nov 2015
NHB (2014b) All India cold storage capacity and technology—baseline study, insights from Hansa Research Group Pvt. Ltd—final report. http://nhb.gov.in/default.aspx; http://nhb.gov.in/PDFViwer.aspx?enc=3ZOO8K5CzcdC/Yq6HcdIxH2DHFRzps8/Lv2GqoSPwtI= Accessed 15 Nov 2015
NTP (2013) National Toxicology Program (USA)—acrylamide. https://www.niehs.nih.gov/health/assets/docs_a_e/acrylamide_fact_sheet_508.pdf. Accessed 11 May 2016
Ohara-Takada A, Matsuura-Endo C, Chuda Y, Ono H, Yada H, Yoshida M, Kobayashi A, Tsuda S, Takigawa S, Noda T, Yamauchi H, Mori M (2005) Change in content of sugars and free amino acids in potato tubers under short-term storage at low temperature and the effect on acrylamide level after frying. Biosci Biotechnol Biochem 69:1232–1238
Olsen N, Frazier MJ, Ingham R, Keeling J (2011) The feasibility of irradiation as a phytosanitary tool for sprout control and nematode destruction in potato tubers for export, final report 2010–2011. Report of research findings to the US National Potato Council, Washington, DC
Olsson K, Svensson R, Roslund C (2004) Tuber components affecting acrylamide formation and colour in fried potato: variation by variety, year, storage temperature and storage time. J Sci Food Agric 84:447–458
Ou SY, Shi JJ, Huang CH, Zhang GW, Teng JW, Jiang Y, Yang B (2010) Effect of antioxidants on elimination and formation of acrylamide in model reaction systems. J Hazard Mater 182:863–868
Pandey SK, Singh SV, Sarkar D (2005) Potato (Solanum tuberosum L.) for sustaining food and nutrition security in developing world. Indian J Agric Sci 75:3–18
Pandey SK, Sarkar D, Singh SV (2006a) Potato processing in India: today and tomorrow. Potato J 33:11–19
Pandey SK, Singh SV, Kumar D, Manivel P, Marwaha RS, Kumar P, Singh BP (2006b) Kufri Chipsona-3: a high yielding potato variety for chipping with defect free tubers. Potato J 33:26–34
Pandey SK, Singh JP, Gopal J (2008a) Potato varieties and cropping system in India. Potato J 35:103–110
Pandey SK, Singh SV, Gaur PC, Marwaha RS, Kumar Dinesh, Kumar Parveen Singh BP (2008a) Chipsona varieties: a success story. Technical Bulletin No. 89, Central Potato Research Institute, Shimla, 171 001 (HP), India
Pandey SK, Singh SV, Kumar D, Manivel P, Marwaha RS, Kumar P, Singh BP, Gupta VK (2008c) Kufri Himsona: a chipping cultivar for hill regions of India. Potato J 35:1–8
Pandey SK, Marwaha RS, Kumar D, Singh SV (2009) Indian Potato processing story: industrial limitations, challenges ahead and vision for the future. Potato J 36:1–13
Patel NH, Patel RN, Singh SV, Pandey SK, Patel JM, Patel SB (2007) Performance of exotic and Indian potato varieties in Gujarat for processing into French fries. Potato J 34:51–52
Paul V, Ezekiel R (2002) Suppression of potato sprout growth by alcohols, acetaldehyde and 2,4-dichlorophenoxy acetic acid ethyl ester at higher temperatures. J Indian Potato Assoc 29:119–122
Paul V, Ezekiel R (2003a) Improved heap method of potato storage for central Indo-Gangetic plains. J Indian Potato Assoc 30:159–160
Paul V, Ezekiel R (2003b) Relationship between tuber size and storage behaviour in two potato cultivars during on-farm storage. J Indian Potato Assoc 30:161–162
Paul V, Ezekiel R (2003c) Suppression of sprout growth of potato (Solanum tuberosum L.) tubers by triadimefon. J Plant Biol 30:353–356
Paul V, Ezekiel R (2004a) Evaluation of heap and pit methods of potato storage in the central Indo-Gangetic Plains. Indian J Agric Sci 74:665–668
Paul V, Ezekiel R (2004b) Effect of triadimefon on ion leakage in ageing potato tubers during storage at higher temperature. Indian J Plant Physiol 9:192–194
Paul V, Ezekiel R (2005) Changes in temperature and relative humidity in heap and pit during storage of potatoes. Potato J 32:205–206
Paul V, Ezekiel R (2006a) Sprout suppression of potato (Solanum tuberosum L.) tubers stored at 18 °C by pre and post-harvest application of sub-lethal doses of glyphosate. Indian J Plant Physiol 21:300–305
Paul V, Ezekiel R (2006b) Inhibition of potato sprout growth by pre-harvest foliar application of glyphosate. Potato J 33:56–61
Paul V, Ezekiel R (2007) Changes in sugar levels and membrane permeability during storage in potato (Solanum tuberosum L.) tubers previously stored in cold store. Indian J Agric Sci 77:32–35
Paul V, Ezekiel R (2013) Scientific storage options crucial for potatoes. Agric Today (Year Book), The National Agriculture Magazine, pp 134–137
Paul V, Ezekiel R, Shekhawat GS (2002a) Traditional methods of potato storage in changing scenario. Indian Farm 52(11–13):18–19
Paul V, Ezekiel R, Singh J, Shekhawat GS (2002a) Evaluation of on-farm storage methods of potato in Indo-Gangetic plains. In: Paul Khurana SM, Shekhawat GS, Pandey SK, Singh BP (eds) Potato, global research and development, Proceeding of global conference on potato, 6–11 Dec 1999, New Delhi, vol II. Malhotra Publishing House, New Delhi, pp 1080–1085
Paul V, Kumar D, Ezekiel R (2005) Effect of crop maturity and on-farm storage on sugar content of potato (Solanum tuberosum L.) tubers. Indian J Plant Physiol 3:287–291
Paul V, Ezekiel R, Pandey R, Kumar D (2014) Potential of glyphosate as a sprout suppressant of potato (Solanum tuberosum L.) tubers during storage. Indian J Plant Physiol 19:293–305
Paul V, Ezekiel R, Pandey R (2016a) Sprout suppression on potato: need to look beyond CIPC for more effective and safer alternatives. J Food Sci Technol 53:1–18
Paul V, Ezekiel R, Pandey R (2016b) Cold storage in India: present scenario and future directions. Process Food Ind 19:25–28
Pedersen GS, Hogervorst JGF, Schouten LJ, Konings EJM, Goldbohm RA, van den Brandt PA (2010) Dietary acrylamide intake and estrogen and progesterone receptor-defined postmenopausal breast cancer risk. Breast Cancer Res Treat 122:199–210
Pedreschi F (2007) The canon potato science: 49. Acrylamide. Potato Res 50:411–413
Pedreschi F (2009) Acrylamide formation and reduction in fried potatoes. In: Ortega-Rivas E (ed) Processing effects on safety and quality of foods, vol 310. Taylor and Francis, USA, pp 231–251
Pedreschi F, Kaack K, Granby K (2004) Reduction of acrylamide formation in potato slices during frying. Lebensmittel-Wissenschaft und Technologie 37:679–685
Pedreschi F, Moyano P, Kaack K, Granby K (2005) Color changes and acrylamide formation in fried potato slices. Food Res Int 38:1–9
Pedreschi F, Kaack K, Granby K (2006) Acrylamide content and color development in fried potato strips. Food Res Int 39:40–46
Pedreschi F, Granby K, Risum J (2010) Acrylamide mitigation in potato chips by using NaCl. Food Bioprocess Technol 3:917–921
Pedreschi F, Mariotti S, Granby K, Risum J (2011) Acrylamide reduction in potato chips by using commercial asparaginase in combination with conventional blanching. LWT-Food Sci Technol 44:1473–1476
Pedreschi F, Mariotti MS, Granby K (2014) Current issues in dietary acrylamide: formation, mitigation and risk assessment. J Sci Food Agric 94:9–20
Pelucchi C, La Vecchia C, Bosetti C, Boyle P, Boffetta P (2011) Exposure to acrylamide and human cancer: a review and meta-analysis of epidemiologic studies. Ann Oncol 22:1487–1499
Petersen BJ, Tran N (2005) Exposure to acrylamide: placing exposure in context. Adv Exp Med Biol 561:63–76
Powers SJ, Mottram DS, Curtis A, Halford NG (2013) Acrylamide concentrations in potato crisps in Europe from 2002 to 2011. Food Addit Contam Part A-Chem Anal Control Expos Risk Assess 30:1493–1500
Qu SY, Lin Q, Zhang YP, Huang CH, Sun X, Fu L (2008) Reduction of acrylamide formation by selected agents in fried potato crisps on industrial scale. Innov Food Sci Emerg Technol 9:116–121
Rana RK, Pandey SK (2007) Processing quality potatoes in India: India analysis of indigenous demand. Process Food Ind 10:26–35
Rezaee M, Almassi M, Farahani AM, Minaei S, Khodadadi M (2011) Potato sprout inhibition and tuber quality after postharvest treatment with gamma irradiation on different dates. J Agric Sci Technol 13:829–841
Riboldi BP, Vinhas ÁM, Moreira JD (2014) Risks of dietary acrylamide exposure: a systematic review. Food Chem 157:310–322
Rice JM (2005) The carcinogenicity of acrylamide. Mutat Res 580:3–20
Robert F, Vuataz G, Pollien P, Saucy F, Alonso MI, Bauwens I, Blank I (2004) Acrylamide formation from asparagine under low-moisture Maillard reaction conditions. 1. Physical and chemical aspects in crystalline model systems. J Agric Food Chem 52:6837–6842
Romani S, Bacchiocca M, Rocculi P, Rosa MD (2008) Effect of frying time on acrylamide content and quality aspects of French fries. Eur Food Res Technol 226:555–560
Rosen J, Hellenas K (2002) Analysis of acrylamide in cooked foods by liquid chromatography tandem mass. Analyst 127:880–882
Rydberg P, Erikson S, Takreke E, Karlsson P, Ehrenberg L, Tornqvist M (2003) Investigations of factors that influence the acrylamide content of heated foodstuffs. J Agric Chem 51:7012–7018
Sadd PA, Hamlet GH, Liang L (2008) Effectiveness of methods for reducing acrylamide in bakery products. J Agric Food Chem 56:6154–6161
Sakaliene O, Koskinen WC, Blazauskiene G, Petroviene I (2009) Level and fate of chlorpropham in potatoes during storage and processing. J Environ Sci Health (Part B) 44:1–6
Sandhu KS, Marwaha RS, Kumar P (2014) Processing attributes of potato varieties stored at ambient conditions of north-western plains. Potato J 41:86–90
Serpen A, Gokmen V (2009) Evaluation of the Maillard reaction in potato crisps by acrylamide, antioxidant capacity and color. J Food Compos Anal 22:589–595
Shamla L, Nisha P (2014) Acrylamide in deep-fried snacks of India. Food Addit Contam 7:220–225
Shepherd LVT, Bradshaw JE, Dale MFB, McNicol JW, Pont SDA, Mottram DS, Davies HV (2010) Variation in acrylamide producing potential in potato: segregation of the trait in a breeding population. Food Chem 123:568–573
Shipp A, Lawrence G, Gentry R, McDonald T, Bartow H, Bounds J, Macdonald N, Clewell H, Allen B, Van Landingham C (2006) Acrylamide: review of toxicity data and dose-response analyses for cancer and non-cancer effects. Crit Rev Toxicol 36:481–608
Sihtmaee M, Mortimer M, Kahru A, Blinova I (2010) Toxicity of five anilines to crustaceans, protozoa and bacteria. J Serbian Chem Soc 75:1291–1302
Singh M (2010) Projection of potato export from India: a Markov chain approach. Potato J 37:48–55
Singh BP (2013) Final report of NAIP sub-project “value chain on potato and potato products” CPRI-Campus, Modipuram, UP, India
Singh B, Ezekiel R (2005) Isopropyl N-(3-chlorophenyl) carbamate (CIPC) residues in potatoes treated with fog and dust formulations during storage. Potato J 32:213–214
Singh B, Ezekiel R (2010) Isopropyl N-(3-chlorophenyl) Carbamate (CIPC) Residues in Potatoes Stored in Commercial Cold Stores in India. Potato Res 53(2):111–120
Singh BP, Rana RK (2013) Potato for food and nutritional security in India. Indian Farm 63:37–43
Singh B, Ezekiel R, Kumar D, Kumar S (2008) Reducing sugar content and chipping quality of tubers of potato cultivars after storage and reconditioning. Potato J 35:23–30
Singh SV, Marwaha RS, Kumar D, Kumar P, Pandey SK (2009) Suitability of potato varieties grown in north-eastern Indian plants for processing. Potato J 36:25–34
Smith MJ, Bucher G (2012) Tools to study the degradation and loss of the N-phenyl carbamate chlorpropham—comprehensive review. Environ Int 49:38–50
Sohn M, Ho CT (1995) Ammonia generation during thermal degradation of amino acids. J Agric Food Chem 43:3001–3003
Sonnewald U (2001) Control of potato tuber sprouting. Trends Plant Sci 6:333–335
Sowokinos JR (2001a) Biochemical and molecular control of cold-induced sweetening in potatoes. Am J Potato Res 78:221–236
Sowokinos JR (2001b) Allele and isozyme patterns of UDP-glucose pyrophosphorylase as a marker for cold-sweetening resistance in potatoes. Am J Potato Res 78:57–64
Stadler R, Blank I, Varga N, Robert F, Hau JR, Guy P, Robert M, Riediker S (2002) Acrylamide from Maillard reaction products. Nature 419:449–450
Stefano VD, Avellone G (2014) Food contaminants. J Food Stud 3:88–103
Sukumaran NP, Verma SC (1993) Storage and processing. In: Chadha KL, Grewal JS (eds) Advances in horticulture, vol 7. Potato Malhotra Publishing House, New Delhi, pp 701–732
Surdyk N, Rosen J, Andersson R, Aman P (2004) Effects of asparagine, fructose, and baking conditions on acrylamide content in yeast-leavened wheat bread. J Agric Food Chem 52:2047–2051
Svensson K, Abramsson L, Becker W, Glynn A, Hellenas K-E, Lind Y, Rosen J (2003) Dietary intake of acrylamide in Sweden. Food Chem Toxicol 41:1581–1586
Swedish National Food Administration (2002a) Acrylamide is formed during the preparation of food and occurs in many foodstuffs. Press release from Livsmedelsverket, Swedish National Food Administration, 24 April 2002. http://www.slv.se/engdefault.asp. Accessed 11 Jan 2015
Swedish National Food Administration (2002b) Analysis of acrylamide in food. http://www.slv.se/acrylamide. Accessed 11 Jan 2015
Tareke E, Rydberg P, Karlsson P, Tornqvist M, Eriksson S (2000) Acrylamide—a cooking carcinogen? Chem Res Toxicol 13:517–522
Tareke E, Rydberg P, Karlsson P, Eriksson S, Tornqvist M (2002) Analysis of acrylamide, a carcinogen formed in heated foodstuffs. J Agric Food Chem 51:4998–5006
Taubert D, Harlfinger S, Henkes L, Berkels R, Schomig E (2004) Influence of processing parameters on acrylamide formation during frying of potatoes. J Agric Food Chem 52:2735–2739
Tayler PN, Gussin EJ, Leck K (1996) Control of sprouting in potatoes from applications made under commercial conditions. In: Proceedings of 13th triennial conference of the European Association for Potato Research. Pudoc, Wageningen, pp 589–590. PSS 25
Topcu Y, Uzundumlu AS, Guler IO (2010) Economic effectiveness analyses of potato farms: the case of Erzurum province, Turkey. Sci Res Essays 5:2560–2566
Trelka R (2010) Antioxidant capacity of potato chips and snapshot trends in acrylamide content in potato chips and cereals on the Canadian market. Food Addit Contam Part A-Chem Anal Control Expos Risk Assess 27:1193–1198
Troise AD, Fogliano V (2013) Reactants encapsulation and Maillard reaction. Trends Food Sci Technol 33:63–74
Truong VD, Pascua Y, Reynolds R (2013) Processing treatments for reducing the acrylamide level in sweet potato French fries. J Agric Food Chem 62:310–316
US Patent (2010) US 2010/0080872 A1—low acrylamide French fry and preparation process. United States. http://www.google.co.in/patents/US20100080872. Accessed 15 Jan 2015
Viklund G, Olsson K, Sjoholm I, Skog K (2008) Variety and storage conditions affect the precursor content and amount of acrylamide in potato crisps. J Sci Food Agric 88:305–312
Viklund G, Olsson K, Sjoholm I, Skog K (2010) Acrylamide in crisps: effect of blanching studied on long-term stored potato clones. J Food Compos Anal 23:194–198
Virendra Pandit (2015a) McDonald’s potato supplier to double crop area in Gujarat. BusinessLine, 24 Sept 2015. http://www.thehindubusinessline.com/economy/agri-business/mcdonalds-potato-supplier-to-double-crop-area-in-gujarat/article3931837.ece. Accessed 10 Nov 2015
Virendra Pandit (2015b) In Gujarat, the red potato is no humble spud. BusinessLine, 13 March 2015. http://www.thehindubusinessline.com/economy/agri-business/in-gujarat-the-red-potato-is-no-humble-spud/article4504956.ece. Accessed 10 Nov 2015
Vivan Fernandes (2015) Gujarat contract farmers smile as potato price crash. Financial Express, 17 Feb 2015. http://www.smartindianagriculture.in/gujarat-contract-farmers-insulated-from-potato-price-crash/. Accessed 10 Nov 2015
Weisshaar R (2004) Acrylamide in heated potato products–analytics and formation routes. Eur J Lipid Sci Technol 106:786–792
Whittaker A, Marotti I, Dinelli G, Calamai L, Romagnoli S, Manzelli M, Palchetti E, Vecchio V, Benedettelli S (2010) The influence of tuber mineral element composition as a function of geographical location on acrylamide formation in different Italian potato genotypes. J Sci Food Agric 90:1968–1976
WHO (2002) FAO/WHO consultation on the health implications of acrylamide in food. Summary report of a meeting held in Geneva, 25–27 June 2002. http://www.who.int/foodsafety/publications/chem/en/acrylamide_full.pdf. Accessed 24 July 2015
WHO (2011) Technical report series No. 959—evaluation of certain contaminants in food. Seventy-second report of the joint FAO/WHO expert committee on food additives. http://apps.who.int/iris/bitstream/10665/44514/1/WHO_TRS_959_eng.pdf. Accessed 24 July 2015
Wiberley-Bradford AE, Busse JS, Jiang J, Bethke PC (2014) Sugar metabolism, chip color, invertase activity, and gene expression during long-term cold storage of potato (Solanum tuberosum) tubers from wild-type and vacuolar invertase silencing lines of Katahdin. BMC Res Notes 7:801. doi:10.1186/1756-0500-7-801. http://www.biomedcentral.com/1756-0500/7/801. Accessed 27 Oct 2015
Williams JSE (2005) Influence of variety and processing conditions on acrylamide levels in fried potato crisps. Food Chem 90:875–881
Wilson KM, Balter K, Adami HO, Gronberg H, Vikstrom AC, Paulsson B, Tornqvist M, Mucci LA (2009) Acrylamide exposure measured by food frequency questionnaire and hemoglobin adduct levels and prostate cancer risk in the cancer of the prostate in Sweden study. Int J Cancer 124:2384–2390
Wilson KM, Mucci LA, Rosner BA, Willett WC (2010) A prospective study on dietary acrylamide intake and the risk for breast, endometrial, and ovarian cancers. Cancer Epidemiol Biomark Prev 19:2503–2515
Wu L, Bhaskar PB, Busse JS, Zhang RF, Bethke PC, Jiang J (2011) Developing cold-chipping potato varieties by silencing the vacuolar invertase gene. Crop Sci 51:981–990
Wustman R, Haverkort A, Zhang X, Rathee G (2011) An overview of the potato sector in India and prospects of Indo and Dutch cooperation. Project No. 3250214811. Applied Plant Research (Praktijkonderzoek Plant and Omgeving), part of Wageningen UR Business Unit Arable Farming, Multifunctional Agriculture and Field Production of Vegetables. Wageningen. http://edepot.wur.nl/192686. Accessed 24 July 2015
Xu Y, Cui B, Ran R (2014) Risk assessment, formation, and mitigation of dietary acrylamide: current status and future prospects. Food Chem Toxicol 69:1–12
Yadav SK, Lekshmy S, Verma RK, Santhosh VV, Sairam RK, Chinnusamy V (2012) Molecular analysis of cold induced sweetening in potato. Poster presented at National Seminar on “Physiological and molecular approaches for development of climate resilient crops”, 12–14 Dec 2012, Acharya N.G. Ranga Agricultural University, Rajendranagar, Hyderabad, India
Ye J, Roshani S, Pradeep S, Rommens CM (2010) Tuber-specific silencing of the acid invertase gene substantially lowers the acrylamide-forming potential of potato. J Agric Food Chem 58:12162–12167
Zamora R, Delgado RM, Hidalgo FJ (2009) Conversion of 3-aminopropionamide and 3-alkylaminopropionamides into acrylamide in model systems. Mol Nutr Food Res 53:1512–1520
Zhang H, Liu X, Liu J, Ou Y, Lin Y, Li M, Song B, Xie C (2013) A novel RING finger gene, SbRFP1, increases resistance to cold-induced sweetening of potato tubers. FEBS Lett 587:749–755
Zhang H, Liu J, Hou J, Yao Y, Lin Y, Ou Y, Song B, Xie C (2014) The potato amylase inhibitor gene SbAI regulates cold-induced sweetening in potato tubers by modulating amylase activity. Plant Biotechnol J 12:984–993
Zhang H, Zhang H, Cheng L, Wang L, Qian H (2015) Influence of deep-frying using commercial oils on acrylamide formation in French fries. Food Addit Contam Part A 32:1083–1088
Zhu F, Cai YZ, Ke J, Corke H (2010) Compositions of phenolic compounds, amino acids and reducing sugars in commercial potato varieties and their effects on acrylamide formation. J Sci Food Agric 90:2254–2262
Zhu X, Richael C, Chamberlain P, Busse JS, Bussan AJ, Jiang J, Bethke PC (2014) Vacuolar invertase gene silencing in potato (Solanum tuberosum L.) improves processing quality by decreasing the frequency of sugar-end defects. PLoS One 9:e93381. doi:10.1371/journal.pone.0093381
Zyzak DV, Sanders RA, Stojanovic M, Tallmadge DH, Eberhart BL, Ewald DK, Gruber DC, Morsch TR, Strothers MA, Rizzi GP, Villagran MD (2003) Acrylamide formation mechanism in heated foods. J Agric Food Chem 51:4782–4787
Acknowledgements
Authors wish to convey their sincere thanks to ICAR-Central Potato Research Institute (CPRI), Shimla; ICAR-Indian Agricultural Research Institute (IARI), New Delhi, and the ICAR, the parent organization, under the Ministry of Agriculture and Farmers Welfare, Government of India. Financial support provided by CPRI for the Institute Research Programme “Development of efficient potato storage methods [P1-1999/16-IPR-F-60/0210] under the Mega Project “Evaluation and improvement of traditional and modern potato storage methods” is acknowledged. We further wish to acknowledge the financial support received from the Ministry of Food Processing Industries under the project “Demonstration and training to potato processing industries of improved storage technology for potatoes meant for processing purposes” and National Horticulture Board (NHB) and Agricultural and Processed Food Products Export Development Authority (APEDA) for the project “Monitoring of quality of CIPC-treated potatoes stored at 10–12 °C for export and processing”. We put forward our apologies for not being able to cite all the relevant literature due to limitation of space.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Communicated by A. K. Kononowicz.
Rights and permissions
About this article
Cite this article
Paul, V., Ezekiel, R. & Pandey, R. Acrylamide in processed potato products: progress made and present status. Acta Physiol Plant 38, 276 (2016). https://doi.org/10.1007/s11738-016-2290-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-016-2290-8