Abstract
An extensive population of the burrowing crab, Neohelice granulata, inhabits the intertidal areas of the Bahía Blanca estuary, a moderately polluted temperate ecosystem located to the southwest of the province of Buenos Aires, Argentina. In order to determine the metal-accumulating ability of this species, concentrations of Cd, Cu, Pb, Ni, Zn, Mn, Cr, and Fe in soft tissues of adult specimens were measured. Subsequently, the bioconcentration factors (BCFs) of all heavy metals were determined using levels of concentrations previously obtained in intertidal sediments. The results showed concentrations above the detection limit in soft tissues of male and female crabs for all metals except Pb and Cr. BCF > 1 were obtained for Cd, Cu, and Zn, indicating that these metals are accumulated and biomagnified. However, BCF values < 1 were found for the rest of the metals (Mn, Ni, and Fe). The findings of metal accumulation in soft tissues of N. granulata is of great importance taking into account that this is a key species within this temperate ecosystem, playing a major role in the transference of pollutants to higher trophic levels.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Aquatic invertebrates accumulate trace metals in their tissues both from the environment as well as via food sources. Although these organisms require certain levels of several metals as copper (Cu), zinc (Zn), manganese (Mn), and iron (Fe) playing essential roles in metabolism, excessive amounts of metals may have toxic effects (Rainbow 1997, 2002; Wang and Fisher 1999).
Marine and estuarine sediments act as a sink for a wide variety of metals sometimes reaching high concentrations. These pollutants are known to concentrate in fine grain sediments since fine particles, such as clay and colloidal materials, are generally surface-active and contain organic matter and Fe/Mn oxide surface coatings, playing an important role controlling the deposition of metals in sediments (Ip et al. 2007). To interpret the toxicological responses of organisms, it is necessary to understand the relationship between the concentration of chemicals in the environment (e.g., water, sediment, or air) and the concentration of chemicals in an organism, or in the target organ(s) of an organism, in contact with that environment (Gobas and Morrison 2000).
Some benthic crustaceans can use the sediment not only to live in it but also as part of their diet; therefore, it is expected that the concentrations of contaminants in the sediment and in the tissues of these organisms have a certain degree of similarity (Marsden and Rainbow 2004). Studies of metal bioaccumulation from sediments have proved to be useful tools for assessing ecological risks from metal-contaminated sediment and researching the influences on bioavailability and mobility of metals in sediments (Ingersoll et al. 1994; Sundaray et al. 2011; Yang et al. 2012).
Neohelice granulata (Brachyura, Varunidae) is a semi-terrestrial estuarine crab widely distributed along the Atlantic coast of South America from southern Brazil (23°S) to the northern Argentinean Patagonia (41°S) (Spivak et al. 1994). This benthic organism is intimately associated with the sediment of both the supratidal and the intertidal zone, being found in the salt marshes vegetated by the Spartina densiflora, S. alterniflora, and Sarcocornia perennis as well as in tidal flats (Spivak et al. 1997; Escapa et al. 2008). The feeding of crabs differs according to where they are found. In the salt marshes, they are herbivorous–detritivorous while those found in the tidal flats are mainly deposit feeders (Iribarne et al. 1997). The incorporation of metals and other contaminants is intimately related to the close association of these organisms with benthos, which facilitates the income of metals and other contaminants present in sediments. This species is one of the most abundant macroinvertebrates in the estuary (Boschi 1964; Spivak 1997) and is a fundamental link in the trophic web since all stages of the life cycle of this species are important items in the diet of other species such as birds and fish among others. In this sense, N. granulata is a key species of this ecosystem, being a fundamental link in the transfer of contaminants through the food chain.
In Argentina, the Bahía Blanca estuary is considered a moderately polluted environment linked to an intense anthropogenic activity on the north shoreline that receives the contribution of diverse pollutants (Arias et al. 2010; Cifuentes et al. 2016), Biancalana et al. 2012, Oliva et al. 2015). There is a discharge of wastewater into the estuary without appropriate treatment, which comes from several urban settlements (Bahía Blanca, Punta Alta, General Cerri, 380,000 inhabitants) and industries (processing of chemicals, oil and plastic). In the inner area of the estuary, there are three sewage treatment plants in the city of Bahía Blanca, capable to treat approximately 215.000 m3/day. One of the purification plants, called the “Third Basin,” was recently installed in the most internal area of the estuary. The treatment plant of greater volume denominated “The big basin of Bahía Blanca” (181.440 m3/day) owns only a preliminary treatment, and one of the localities of Ingeniero White is out of service. There is also a continuous atmospheric contribution of substances from the use of fossil fuels, fumes, particles in suspension, and products of industrial and urban activities (Cifuentes et al. 2016). Another source of contaminants to the estuary is agricultural activity through the continental runoff of fertilizers (Perillo et al. 2001; Botté et al. 2010).
Several studies have been conducted on the concentration of metals in this species inhabiting the Bahía Blanca estuary (Ferrer 2001; Ferrer et al. 2006; Simonetti et al. 2012, 2013). However, a relationship between the concentrations of metals in the sediment and in these organisms, namely bioconcentration factor (BCF), has not yet been established. Bioconcentration is the process of accumulation by which the concentration of a certain chemical in aquatic organisms exceeds the levels found in the environment (i.e. water or sediment) (Gobas and Morrison 2000; Mountouris et al. 2002). In this sense, the aim of this study was to assess the Cd, Cu, Pb, Zn, Mn, Ni, Cr, and Fe bioconcentration factors in the burrowing crab N. granulata from the Bahía Blanca estuary. Therefore, heavy metals were determined in the fine sediments and soft tissues of the crabs, to establish which metals and bioconcentrated in the tissues of these benthic invertebrates, trying to understand a potential trophic transfer through the corresponding food web.
Materials and methods
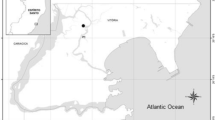
The Bahia Blanca Estuary is located in the southwestern Atlantic Ocean (38°45′–39°40’S and 61°45′–62°30’W). Is a mesotidal coastal plain estuary extended over 2300 km2, formed by several tidal channels, islands, and extensive tidal flats with patches of low salt marshes dominated by crab caves of the burrowing crab Neohelice granulata (Piccolo et al. 2008).
Sampling of males and females of N. granulata was seasonally performed from April 2014 to September 2015 in two locations within the Bahía Blanca estuary (Fig. 1). Cuatreros Port (CP) located in the inner part of the estuary receives the contribution of the Sauce Chico River and the Saladillo de García stream, which drain an area of approximately 1600 km2 that involve large extensions of agricultural fields (Perillo et al. 2001; Limbozzi and Leitao 2008). Rosales Port (RP), located in the middle part of the estuary, is influenced by Punta Alta city (~ 60 thousand inhabitants) and its sewage outfall, which discharges into the estuary with no treatment.
Samples of intertidal superficial sediment (n = 3 for each site) for determination of metal concentrations were taken with PVC cores (100 mm i.d.; 150 mm long). Afterwards, they were kept in polyethylene bags, carried to the laboratory in refrigerated boxes and stored up to maximum of 48 h at 4 °C. Sampling was done in the morning at low tide. In the laboratory, sediment samples were oven dried at 60 ± 5 °C for 4 days. Then, samples were ground in a porcelain mortar and they were sifted through stainless steel meshes until fine particles (< 63 μm) were obtained. Finally, they were stored in plastic desiccators until their analytical treatment.
Crabs were caught using a crab trap with bait during high tide. Only adult males and females (i.e., maximum width carapace > 15 mm for males and > 17 mm for females, according to Luppi et al. (2004)) were selected. Individuals of each sex were placed in two plastic boxes with estuarine water and were transported to the laboratory where they were washed with distilled water and preserved at − 20 °C until analysis.
The soft tissues obtained by dissection of the crab samples were pooled (five or six specimens) according to sex and location and placed in Petri dishes in an oven at 50 °C until constant weight was reached. Then, dried samples were grinded in a porcelain mortar for homogenization. Acid digestion of crab tissues and fine sediment was performed according to the methodology described by Botté et al. (2010). Samples of 500 ± 50 mg were mineralized with 5 ml concentrated HNO3 (65%, Merck) and 1 ml concentrated HClO4 (70–72%, Merck) and placed in a heated glycerin bath at 120 ± 10 °C until complete mineralization. The residue was completed with diluted HNO3 (0.7%) up to 10 ml into centrifuge tubes. All glassware and equipment used for dissection and drying of samples were cleaned with diluted nitric acid (5% v/v) to prevent contamination.
Metal concentrations (Cd, Cu, Pb, Ni, Zn, Mn, Cr, and Fe) were determined by inductively coupled plasma-optical emission spectroscopy (ICP OES Optima 2100 DV Perkin Elmer). The method detection limit (MDL) was calculated as MDLm = t (n-1, 1-α = 0.99) x Sm, where m corresponds to each metal and S corresponds to the standard deviation of 15 method blanks (EPA 2016). The MDLs, expressed in μg g−1, were as follows: Cd 0.017, Cu 0.136, Pb 0.209, Zn 0.276, Mn 1.854, Ni 0.214, Cr 0.185, and Fe 2.674.
Analytical quality was checked against certified reference material (pond sediment flour R.M.No. 82) provided by The National Institute for Environmental Studies (NIES) from Tsukuba University (Japan). The recovery percentages for all analyzed metals were as follows: Cu 71%, Pb 105.6%, Zn 107.5%, Mn 82.3%, Ni 105.1%, Cr 91.5%, and Fe 76%. All concentrations are expressed in parts per million (μg/g) on a dry weight (dw) basis. For samples that were lower than the applied analytical MDL, a value of one half the detection limit was assigned (Jones and Clarke 2005). These samples were considered in the dataset for statistical treatment only if they represented less than 40% of the total samples. Otherwise, only descriptive statistics was performed using the detectable values.
Two-way analysis of variance (ANOVA) was performed to analyze possible interactions between sites and sexes. Statistical analyses were carried out with appropriate software (IBM SPSS Statistics 22.0 and Infostat version 2016- Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina).
We used bioconcentration factors (BCFs) to analyze the relationship between heavy metals in crabs and sediment (Mountouris et al. 2002; Mendoza- Carranza et al. 2016). It was calculated as BCF = Corg/Csed, where Corg is the concentration of metal in the organism and Csed is the concentration of the same metal in the environment, intertidal superficial fine sediment in this case.
The results of heavy metal concentrations found in the intertidal sediments are analyzed in detail in Simonetti et al. (2017).
Result and discussion
Heavy metals in burrowing crabs (Neohelice granulata) and sediments as well as bioconcentration data are shown in Table 1. The concentration of all the metals (except Mn) in the fine sediment was significantly higher (p < 0.005) in RP than in CP (Simonetti et al. 2017). However, the concentrations found in burrowing crabs at both sites did not exhibit the same behavior.
Of the eight metals determined, Pb and Cr were found in concentrations below the detection limit in all crab tissue samples (Table 1) which coincide with previous studies of this species in the estuary (Ferrer 2001; Simonetti et al. 2012, 2013). These results place these organisms in a lower range than those registered for crabs from other parts of the world (Table 2). The availability of metals to be bioaccumulated is dependent upon the concentration of the element within the sediment as well as its geochemical behavior and physical-chemical conditions within that matrix. On the other hand, for crustaceans, several intrinsic factors, for instance age, size, stage in the molt cycle and reproductive cycle, may influence bioaccumulation process (Marsden and Rainbow 2004; Bjerregaard et al. 2005; Chen et al. 2005). Although recent information on the geochemical partitioning of metals in sediments of the Bahía Blanca estuary is not available, previous studies by Botté et al. (2010) in CP found, for potentially bioavailable fractions (i.e., PBF = F1 + F2, according to Marcovecchio and Ferrer (2005)), 55.6% for Pb and 16% for Cr. For RP, the study of Grecco et al. (2011) determined 62.4% for Pb and 18.7% for Cr. This suggests a moderate bioavailability for Pb whereas for Cr, a large part of this metal is found in non-bioavailable fractions, which could explain the results for Cr in crabs from both sites. Taking into account the greater potential bioavailability of Pb in the sediment, all the crab samples however showed concentrations below the limit of detection. The concentrations of Pb below MDL in soft tissues of Neohelice granulata could be explained considering both the facility of this metal to produce complex with organic matter in estuarine environments (Town and Filella 2002; Shank et al. 2004; Louis et al. 2009) as well as the high charge of organic matter as recorded within the Bahía Blanca estuary (Marcovecchio and Freije 2004; Marcovecchio et al. 2009). A second possible explanation could be that a detoxification of this metal is occurring. Previous studies indicate that molting as a mechanism for detoxification may vary depending upon the particular element and crustacean species (Bergey and Weis 2007). Moreover, Pb is known to be readily absorbed in calcium carbonate skeletons (Ahmed et al. 2011). Ferrer (2001), working with metals in soft tissues and shells of this same species in the Bahía Blanca estuary, found that for Pb, concentrations above the limit of detection were only obtained in shells of males and females, whereas in soft tissues of both sexes, Pb levels were not detectable. Based on this evidence, this species would be eliminating the Pb from its tissues through the molt cycles.
Cd concentrations in the fine sediment of CP and RP were mostly below the limit of detection (Table 1). Similarly, concentrations of this metal in crab tissues from RP were in all cases below the detection limit. In contrast, even though levels of this metal in crabs from CP were not high, all the samples were found above the limit of detection, in both females and males, and these concentrations are within the values found in the literature, except when is compared with Sesarma mederi crab (Chaiyara et al. 2013) (Table 2). It is evident that there is some other source of Cd from which the crabs would be incorporating this metal. A possible explanation could be the atmospheric deposition, which has been suggested by several authors as a possible source of heavy metals to the system (Galloway et al. 1982; Wong et al. 2003; Sabin et al. 2005; Kumar Sharma et al. 2008), but so far, there are no studies within this estuary proving the entry through atmosphere. A second possibility could be from agricultural sources. The agricultural and livestock activities carried out in the area make intensive use of fertilizers, herbicides, and other agrochemicals. In these farming activities, some herbicides such as glyphosate, that is a chelating agent for heavy metals and organic cations, could potentially affect the bioavailability, toxicity, and bioaccumulation of heavy metals when applied directly into the aquatic ecosystems (Tsui et al. 2005).
According to Vassiliki and Konstantina (1984), a BCF value of < 1 is expected for most of the metals; otherwise, bioconcentration of metals by organisms will occur. The results of this study showed a BCF < 1 for Pb, Cr, Ni, Mn, and Fe (Table 1). However, the BCF obtained for Cd (i.e., in crabs from CP), Cu, and Zn were greater than 1. In the case of Cd, the high BCF was due to greater concentrations found in the organisms than in the sediment, which allows to infer that there is some degree of active accumulation of this metal in the soft tissues of N. granulata. On the other hand, previous studies on this species in the Bahía Blanca estuary have shown high concentrations of Cu and Zn, two essential metals for these organisms (Ferrer 2001; Simonetti et al. 2012, 2013). Cu is an integral part of the respiratory pigment hemocyanin of decapod crustaceans and Zn is associated with the activity of several enzymes involved in various metabolic pathways and is also an activator of other enzyme systems (White and Rainbow 1985, Rainbow 1997, MacFarlane et al. 2000; Harris et al. 2018). The concentrations of Cu found in N. granulata tended to be higher than the crabs of other coastal areas around the world; meanwhile, Zn concentrations were within the range of concentrations described in the literature (Table 2). Although the metabolic requirements for this species are still unknown, taking into account the concentrations of Cu and Zn found in sediment and organisms, it could be inferred that, as with Cd, there is a certain level of accumulation of these metals in the soft tissues of N. granulata.
Considering that the BCF does not have the same meaning for organisms if the metals that are being bioconcentrated are essential or not essential, it is worth noting that those metals that presented BFC > 1 in this study were Cu, Zn (i.e., essentials), and Cd (i.e., non-essential). Although non-essential metals are toxic to organisms even in traces, it is important to note that the essential metals can also produce toxic effects if the levels to which they are accumulated are excessively high. In this sense, it is necessary to determine the metabolic requirements of these metals in N. granulata to be able to establish if bioconcentration levels found are potentially toxic for this species.
Differences between sites and/or sexes were analyzed (i.e., two-way ANOVA for Cu, Zn, Mn, and Fe; student t test for Cd and Ni). Figure 2 shows the differences between sites and sexes for each metal. Although it was not possible to perform a statistical analysis for sites, the figure shows clear differences for Cd and Ni. Meanwhile, significant differences between sites were only found for Mn, with concentrations significantly greater in crabs from RP (Mn: 64.20 ± 16.93 μg/g dw vs 47.51 ± 16.69 μg/g dw, p = 0.002).
According to the literature, there is no consensus regarding sex as a biological intrinsic factor that may influence the bioaccumulation of metals in crustaceans. Several studies with various species of crabs found a lack of relationship between sex and the accumulation of metals in the tissues. Examples include the researches of Kannan et al. (1995) working with the marine crab Tachypleus tridentatus, Olusegun et al. (2009) with the crab Callinectes amnicola, or of Ayas (2013) studying adult blue swimmer crabs (Portunus pelagicus), among others. Meanwhile, in the case of the shrimp Pleoticus muelleri, Jeckel et al. (1996) found sex differences for Cu and Zn. Chen et al. (2005) also found differences between sexes for the accumulation of certain metals in the rock crab Thalamita crenata.
In this study, significant differences between sexes were found for Cd (RP only), Cu, Mn, and Ni in both sites (p < 0.003 in all four metals) being superior in males than females in all cases (Fig. 2). Previous studies in crabs of CP showed no differences between sexes for any of these metals (Simonetti et al. 2012, 2013). However, in those studies, the sample size was quite small, so that the lack of differences could be related to that factor. For RP, to date, there are no data published in journals on the metal concentrations in crabs; thus, there are no previous data to compare the results obtained in this study.
Conclusions
The results of this study showed concentrations above the detection limit in soft tissues of male and female crabs for six of the eight analyzed metals. This suggests that metals would be bioconcentrated in the tissues of these benthic invertebrates. On the other hand, when the concentrations of metals in the sediment were taking into account, BCF > 1 was obtained only for Cd and the essential metals Cu and Zn so it can be inferred that there is a certain level of bioconcentration of these metals in N. granulata from the Bahía Blanca estuary. In this study, differences between sexes for the concentration of Cd, Cu, Mn, and Ni have been recorded. It would therefore be advisable to keep them separate in future studies.
The knowledge of metal accumulation in soft tissues of N. granulata is of great importance taking into account that this is a key species within the Bahía Blanca estuary, playing a major role in the transference of pollutants to higher trophic levels. In this sense, it is worth noting that these are the first results obtained for this species in Rosales Port; therefore, they will be of great utility for subsequent studies of environmental monitoring.
References
Ahmed K, Mehedi Y, Haque R, Mondol P (2011) Heavy metal concentrations in some macrobenthic fauna of the Sundarbans mangrove forest, south west coast of Bangladesh. Environ Monit Assess 177(1):505–514
Arias AH, Botello AV, Tombesi N, Ponce-Velez G, Freije J, Marcovecchio JE (2010) Presence, distribution, and origins of polycyclic aromatic hydrocarbons (PAHs) in sediments from Bahía Blanca estuary, Argentina. Environ Monit Assess 160:301–314
Ayas D (2013) Effects of gender and season on potentially toxic metal levels in muscles of adult blue swimmer crabs (Portunus pelagicus) from the northeastern Mediterranean Sea. J Mar Biol Oceanogr 2(2):2–4
Ayas D, Ozogul Y (2011) The effects of sex and seasonality on the metal levels of different muscle tissues of mature Atlantic blue crabs (Callinectes sapidus) in Mersin Bay, north-eastern mediterranean. Int J Food Sci Technol 46:2030–2034
Bergey LL, Weis JS (2007) Molting as a mechanism of depuration of metals in the fiddler crab, Uca pugnax. Mar Environ Res 64(5):556–562
Biancalana MF, Menéndez MC, Berazategui AA, Fernández Severini MD, Hoffmeyer MS (2012) Sewage pollution effects on mesozooplankton structure in a shallow temperate estuary. Environ Monit Assess 184(6):3901–3913
Bjerregaard P, Bjørn L, Nørum U, Pedersen KL (2005) Cadmium in the shore crab Carcinus maenas: seasonal variation in cadmium content and uptake and elimination of cadmium after administration via food. Aquat Toxicol 72:5–15
Bordon ICAC, Sarkis JES, Tomas ARG, Scalco A, Lima M, Hortellani MA, Andrade NP (2012) Assessment of Metal Concentrations in Muscles of the Blue Crab, Callinectes danae S., from the Santos Estuarine System. Bull Environ Contam Toxicol 89:484–488
Boschi EE (1964) Los crustáceos decápodos Brachyura del litoral bonaerense (R. A.). Bol Inst Biol Mar 6:1–75
Botté SE, Freije RH, Marcovecchio JE (2010) Distribution of several heavy metals in tidal flats sediments within Bahía Blanca estuary (Argentina). Water Air Soil Pollut 210:371–388
Chaiyaraa R, Ngoendeeb M, Kruatrachue M (2013) Accumulation of Cd, Cu, Pb, and Zn in water, sediments, and mangrove crabs (Sesarma mederi) in the upper Gulf of Thailand. ScienceAsia 39:376–383
Chen MH, Chen CY, Chou HY, Wen TC (2005) Gender and size effects of metal bioaccumulation on the rock crab, Thalamita crenata, in Dapeng Bay, southwestern Taiwan. Mar Pollut Bull 50(4):463–469
Cifuentes O, Escudero D, Medus S, Devoto B (2016) Cadmium found in the Bahía Blanca estuary. Int Educ Sci Res J 2(10):61–79
EPA- United States Environmental Protection Agency (2016) Definition and procedure for the determination of the method detection limit revision 2, EPA 821-R-16-006, 8pp
Escapa M, Perillo GME, Iribarne O (2008) Sediment dynamics modulated by burrowing crab activities in contrasting SW Atlantic intertidal habitats. Estuar Coast Shelf Sci 80:365–373
Ferrer LD (2001) Estudio de los diversos metales pesados en sedimentos del estuario de Bahía Blanca y sus efectos tóxicos sobre el cangrejo Chasmagnathus granulata. Doctoral thesis, Universidad Nacional del Sur, Bahía Blanca, Provincia de Buenos Aires
Ferrer LD, Andrade S, Asteasuain R, Marcovecchio JE (2006) Acute toxicities of four metals on the early life stages of the crab Chasmagnathus granulata from Bahia Blanca estuary, Argentina. Ecotoxicol Environ Saf 65:209–217
Galloway JN, Thronton JD, Norton SA, Volchok HL, McLean RAN (1982) Trace metals in atmospheric deposition: a review and assessment. Armos Environ 16(1):1677–1700
Gobas FAPC, Morrison HA (2000) Bioconcentration and biomagnification in the aquatic environment. In: Mackay D, Boethling RS (eds) Handbook of property estimation methods for chemicals: environmental health sciences. CRC Press, pp 189–197
Grecco LE, Gómez EA, Botté SE, Marcos ÁO, Marcovecchio JE, Cuadrado DG (2011) Natural and anthropogenic heavy metals in estuarine cohesive sediments: geochemistry and bioavailability. Ocean Dyn 61(2–3):285–293
Harris JM, Vinobaba1 P, Kularatne RKA, Kankanamge CE (2018) Heavy metal bioaccumulation and Fulton’s K condition indices in Scylla serrata (Forskål) in relation to sex. Int J Environ Sci Technol 1-10. https://doi.org/10.1007/s13762-018-1653-7
Ingersoll CG, Brumbauch WG, Dwyer FJ, Kemble NE (1994) Bioaccumulation of metals by Hyalella azteca exposed to contaminated sediments from the upper Clark Fork river, Montana. Environ Toxicol Chem 13(12):2013–2020
Ip CCM, Li XD, Zhang G, Wai OWH, Li YS (2007) Trace metal distribution in sediments of the Pearl River estuary and the surrounding coastal area, South China. Environ Pollut 147:311–323
Iribarne O, Bortolus A, Botto F (1997) Between-habitat differences in burrow characteristics and trophic modes in the southwestern Atlantic burrowing crab Chasmagnathus granulata. Mar Ecol Prog Ser 155:132–145
Jeckel WH, Roth RR, Ricci L (1996) Patterns of trace-metal distribution in tissues of Pleoticus muelleri (Crustacea: Decapoda: Solenoceridae). Mar Biol 125:297–306
Jones RP, Clarke JU (2005) Analytical chemistry detection limits and the evaluation of dredged sediment. ERDC/TN EEDP-04–36. United States Army Engineer Research and Development Center, Vicksburg
Kannan K, Yasunaga Y, Iwata H, Ichihashi H, Tanabe S, Tatsukawa R (1995) Concentrations of heavy metals, organochlorines, and organotins in horseshoe crab, Tachypleus tridentatus, from Japanese coastal waters. Arch Environ Contam Toxicol 28:40–47
Kumar Sharma R, Agrawal M, Marshall FM (2008) Atmospheric deposition of heavy metals (Cu, Zn, Cd and Pb) in Varanasi City, India. Environ Monit Assess 142:269–278
Limbozzi F, Leitao TE (2008) Characterization of Bahía Blanca main existing pressures and their effects on the state indicators for surface and groundwater quality. In: Neves R, Baretta JW, Mateus M (eds) Perspectives on integrated coastal zone management in South America. IST Press, Lisboa
Louis Y, Garnier C, Lenoble V, Omanović D, Mounier S, Pizěta I (2009) Characterisation and modelling of marine dissolved organic matter interactions with major and trace cations. Mar Environ Res 67:100–107
Luppi TA, Spivak ED, Bas CC, Anger K (2004) Molt and growth of an estuarine crab, Chasmagnathus granulatus (Brachyura: Varunidae), in Mar Chiquita Lagoon, Argentina. J Appl Ichthyol 20:333–344
MacFarlane GR, Booth DJ, Brown KR (2000) The semaphore crab, Heloecius cordiformis: bioindication potential for heavy metals in estuarine systems. Aquat Toxicol 50:153–166
Marcovecchio JE, Ferrer L (2005) Distribution and geochemical partitioning of heavy metals in sediments of the Bahía Blanca estuary, Argentina. J Coast Res 21:826–834
Marcovecchio JE, Freije RH (2004) Efectos de la intervención antrópica sobre sistemas marinos costeros: El estuario de Bahía Blanca. Anales de la Academia Nacional de Ciencias Exactas, Físicas y Naturales (ANCEFN). Argentina 56:115–132
Marcovecchio JE, Spetter CV, Botté SE, Delucchi F, Arias AH, Fernández Severini MD, Negrin V, Popovich C, Freije RH (2009) Inorganic nutrients and organic matter tidal time-scale variation in a mesotidal estuary: Bahıa Blanca, Argentina. Chem Ecol 25(6):453–465
Marsden ID, Rainbow PS (2004) Does the accumulation of trace metals in crustaceans affect their ecology-the amphipod example? J Exp Mar Biol Ecol 300:373–408
Mendoza- Carranza M, Sepúlveda-Lozada A, Dias-Ferreira C, Geissen V (2016) Distribution and bioconcentration of heavy metals in a tropical aquatic food web: a case study of a tropical estuarine lagoon in SE Mexico. Environ Pollut 210:155–165
Mountouris A, Voutsas E, Tassios D (2002) Bioconcentration of heavy metals in aquatic environments: the importance of bioavailability. Mar Pollut Bull 44:1136–1141
Na CK, Park HJ (2012) Distribution of heavy metals in tidal flat sediments and their bioaccumulation in the crab Macrophthalmus japonicas in the coastal areas of Korea. Geosciences J 16(2):153–164
Oliva AL, Quintas PY, La Colla NS, Arias AH, Marcovecchio JE (2015) Distribution, sources, and potential Ecotoxicological risk of polycyclic aromatic hydrocarbons in surface sediments from Bahía Blanca estuary, Argentina. Arch Environ Contam Toxicol 69(2):163–172
Olusegun AO, Olukemi TO, Olukemi MB (2009) Heavy metal distribution in crab (Callinectes amnicola) living on the shores of Ojo Rivers, Lagos, Nigeria. Environmentalist 29(1):33–36
Perillo GME, Piccolo C, Parodi E, Freije RH (2001) Bahía Blanca estuary, Argentina. In: Seeliger U, Kjerfve B (eds) Coastal marine ecosystems of Latin America, ecological studies, vol 144. Springer, Berlin, pp 205–217. https://doi.org/10.1007/978-3-662-04482-7
Perry H, Isphording W, Trigg C, Riedel R (2015) Heavy metals in red crabs, Chaceon quinquedens, from the Gulf of Mexico. Mar Pollut Bull 101:845–851
Piccolo MC, Perillo GME, Melo W (2008) The Bahía Blanca estuary: an integrated overview of its geomorphology and dynamics. In: Neves R, Baretta JW, Mateus M (eds) Perspectives on integrated coastal zone management in South America. IST Press, Lisboa, pp 219–229
Qiu (2015) Bioaccumulation of heavy metals both in wild and mariculture food chains in Daya Bay, South China. Est Coast Shelf Sci 163:7–14
Rainbow PS (1997) Trace metal accumulation in marine invertebrates: marine biology or marine chemistry. J Mar Biol Assoc U K 77:195–210
Rainbow PS (2002) Trace metal concentrations in aquatic invertebrates: why and so what? Environ Pollut 120:497–507
Sabin LD, Lim JH, Stolzenbach KD, Schiff KC (2005) Contribution of trace metals from atmospheric deposition to stormwater runoff in a small impervious urban catchment. Water Res 39:3929–3937
Shank GC, Skrabal SA, Whitehead RF, Kieber RJ (2004) Strong copper complexation in an organic-rich estuary: the importance of allochthonous dissolved organic matter. Mar Chem 88:21–39
Simonetti P, Botté SE, Fiori SM, Marcovecchio JE (2012) Heavy-metal concentrations in soft tissues of the burrowing crab Neohelice granulata in Bahía Blanca estuary, Argentina. Arch Environ Contam Toxicol 62:243–253
Simonetti P, Botté SE, Fiori SM, Marcovecchio JE (2013) Burrowing crab (Neohelice granulata) as a potential bioindicator of heavy metals in the Bahía Blanca estuary, Argentina. Arch Environ Contam Toxicol 64:110–118
Simonetti P, Botté SE, Macrovecchio JE (2017) Occurrence and spatial distribution of metals in intertidal sediments of a temperate estuarine system (Bahía Blanca, Argentina). Environ Earth Sci 76:636
Spivak ED (1997) Cangrejos estuariales del Atlantico sudoccidental (25°-41°S) (Crustacea: Decapoda: Brachyura). Invest Mar Valparaıso 25:105–120
Sundaray SK, Nayak BB, Lin S, Bhatta D (2011) Geochemical speciation and risk assessment of heavy metals in the river estuarine sediments—a case study: Mahanadi basin, India. J Hazard Mater 186(2–3):1837–1846
Town RM, Filella M (2002) Implications of natural organic matter binding heterogeneity on understanding lead (II) complexation in aquatic systems. Sci Total Environ 300:143–154
Tsui MTK, Wang WX, Chu LM (2005) Influence of glyphosate and its formulation (Roundup®) on the toxicity and bioavailability of metals to Ceriodaphnia dubia. Environ Pollut 138:59–68
Vassiliki K, Konstantina AD (1984) Transfer factors of heavy metals in aquatic organisms of different trophic levels. Bull Environ Contam Toxicol 33:423–429
Wang WX, Fisher NS (1999) Delineating metal accumulation pathways for marine invertebrates. Sci Total Environ 237(238):459–472
White SL, Rainbow PS (1985) On the metabolic requirements for copper and zinc in molluscs and crustaceans. Mar Environ Res 16:215–229
Wong CSC, Li XD, Zhang G, Qi SH, Peng XZ (2003) Atmospheric deposition of heavy metals in the Pearl River Delta, China. Atmos Environ 37(6):767–776
Yang Y, Chen F, Zhang L, Liu J, Wu S, Kang M (2012) Comprehensive assessment of heavy metal contamination in sediment of the Pearl River estuary and adjacent shelf. Mar Pollut Bull 64:1947–1955
Acknowledgments
We thank N. Buzzi, F. García, and J. Pons for their collaboration both in the field work and in the laboratory and W. Melo for providing the map and collaborating with figures elaboration. We also thank the IADO executive directors and Chemical Oceanography Area’s staff.
Funding
This investigation was supported by the National Council for Scientific and Technological Research (CONICET; PIP No. D-738/2010 and Resol. No. 4541/12), the National Agency for Promotion of Science and Technology (ANPCyT: PICT 2012–1383, 2012–2794, 2014–3364, and 2015–2076), and the Science and Technical Secretary of the Universidad Nacional del Sur (PGI 24/ZB59), all from Argentina.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Simonetti, P., Botté, S.E. & Marcovecchio, J.E. Heavy metal bioconcentration factors in the burrowing crab Neohelice granulata of a temperate ecosystem in South America: Bahía Blanca estuary, Argentina. Environ Sci Pollut Res 25, 34652–34660 (2018). https://doi.org/10.1007/s11356-018-3404-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-3404-1