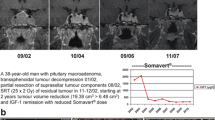
Abstract
Purpose Treatment of acromegaly has undergone important progress in the last 20 years mainly due to the development of new medical options and advances in surgical techniques. Pituitary surgery is usually first-line therapy, and medical treatment is indicated for persistent disease, while radiation (RT) is often used as third-line therapy. The benefits of RT (tumor volume control and decreased hormonal secretion) are hampered by the long latency of the effect and the high risk of adverse effects. Stereotactic RT methods have been developed with the aim to provide more precise targeting of the tumor with better control of the radiation dose received by the adjacent brain structures. The purpose of this review is to present the updates in the efficacy and safety of pituitary RT in acromegalic patients, with an emphasis on the new stereotactic radiation techniques. Methods A systematic review was performed using PubMed and articles/abstracts and reviews detailing RT in acromegaly from 2000 to 2016 were included. Results Stereotactic radiosurgery and fractionated stereotactic RT (FSRT) for patients with persistent active acromegaly after surgery and/or during medical therapy provide comparable high rates of tumor control, i.e. stable or decrease in size of the tumor in 93–100% of patients at 5–10 years and endocrinological remission in 40–60% of patients at 5 years. Hypofractionated RT is an optimal option for tumors located near the optic structures, due to its lower toxicity for the optic nerves compared to single-dose radiosurgery. The rate of new hypopituitarism varies from 10 to 50% at 5 years and increases with the duration of follow-up. The risk for other radiation-induced complications is usually low (0–5% for new visual deficits, cranial nerves damage or brain radionecrosis and 0–1% for secondary brain tumors) and risk of stroke may be higher in FSRT. Conclusion Although the use of radiotherapy in patients with acromegaly has decreased with advances in medical treatments, it remains an effective treatment option after unsuccessful surgery and/or resistance or unavailability of medical therapy. Long-term studies evaluating secondary morbidity and mortality rate after the new stereotactic techniques are needed, in order to evaluate their potential brain-sparing effect.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acromegaly is a severe disease responsible for disabling symptoms, comorbidities and shortened life-span if left untreated [1]. Primary treatment usually involves neurosurgery and, in selected cases, medical therapy. External pituitary irradiation (RT) has been used in the therapy of patients with acromegaly for more than 100 years, initially as an adjuvant to neurosurgery. According to the most recent guidelines, RT is suggested for patients with residual tumor mass following surgery, and if medical therapy is unavailable, unsuccessful, or not tolerated [2].
The method with the longest therapeutical experience is conventional radiotherapy (CRT). CRT is administered by a linear accelerator (4–8 MeV) with a total dose of 40–45 Gy, fractionated in at least 20 sessions. A single rotational field, two opposing fields, or a three field technique are generally used, focusing single beams of high-energy radiation onto a small treatment zone area [3]. A five field technique has also been described [4], but is rarely used in clinical practice. CRT achieves long-term tumor growth control in 80–100% of patients and eventually induces GH/IGF1 normalization in 60–80% [5–11]. However its benefits are hampered by the very slow onset of effects (5–15 years until maximal benefit) and the high risk of adverse effects in long-term: hypopituitarism in 30–80% of patients [5, 6, 8–14], radiation-induced optic neuropathy in 0–5%, cranial nerve deficit and brain necrosis in 0–3%, second brain tumors in up to 2% at 10–20 years [14, 15], cerebrovascular accidents in 4% at 5 years up to 21% at 20 years [16], psychocognitive impairments [17, 18]; all leading to an increase in mortality (1.6–2.2 times higher mortality rate [19–22]) in CRT treated patients, mainly due to cerebrovascular disease (standardized mortality ratio 4.4 [23]–7.1 [20]). The adverse consequences of CRT have been attributed to the radiation of healthy surrounding tissues.
Stereotactic techniques have been developed since 1950, including stereotactic radiosurgery (SRS) and fractionated stereotactic radiotherapy (FSRT). They deliver a precise high radiation dose to a defined target with a steep dose gradient at the tumor margin, thus limiting the irradiation and the damage to the adjacent brain structures.
Stereotactic radiosurgery is usually performed using photons, as in gamma knife, cyberknife and linear accelerator, or protons.
Gamma knife (GK) consists of an array of 192 or 201 cobalt-60 sources arranged in a hemisphere and focused with a collimator helmet on a single or multiple points named isocenters. The patient wears a rigid metal helmet fixed on the skull and the irradiation is delivered as a single fraction. The dose is usually prescribed at the 50% isodose to obtain the maximum dose at the center of each pinpointed target and the prescribed dose at tumor margins [24]. A mean dose to which each tissue is exposed can also be calculated. Doses delivered to the tumor margin are higher for secreting adenomas (18–35 Gy) than for nonfunctioning pituitary adenomas (10–20 Gy) [3].
Cyberknife (CK) combines a mobile linear accelerator mounted on a robotic arm with an image-guided robotic system. The patient is fixed in a more comfortable thermoplastic mask and the dose can be delivered as a single-fraction (usually) or in 3–5 fractions, a technique called hypofractionated SRS [25, 26].
Linear accelerator (LINAC) utilizes X-rays which are derived from colliding accelerated electrons with a target metal. The treatment is delivered using multiple arcs or beams shaped with a multileaf collimator. Modified LINAC have an improved frameless stereotactic fixation system (infrared and radiographic imaging with a Novalis Tx accelerator or cone-beam CT) and allow single or hypofractionated SRS (3–5 fractions) [27, 28].
Fractionated stereotactic radiotherapy (FSRT) usually denominates an improved conventional RT in which a similar total dose of 45–55 Gy is delivered by a LINAC in 25–33 daily fractions. The patient is immobilized in a frameless stereotactic mask with an accuracy of 1–2 mm and a similar planning system as in SRS is used [3], resulting in more localized irradiation as compared with conventional RT.
The risk of visual complications is proportional with the radiation dose that reaches the optic nerves, and this dose is larger when RT is delivered in a single session, compared to fractionated sessions. Therefore, the use of single-session SRS is usually indicated to relatively small tumors (<3 cm) located more than 3 mm away from the optic structures [2, 29], while hypofractionated SRS can be used in perioptic tumors [28, 30]. CRT and FSRT are usually indicated in large pituitary tumors, including those with invasion of the optic nerves. Currently there is limited experience with hypofractionated SRS as compared to FSRT.
The advances in medical therapy and surgical techniques in acromegaly for the last 20 years were followed by a progressive decline in the use of RT. A recent analysis of the Spanish national registry of acromegaly shows that RT use declined over four decades, from 62.8% of patients treated prior to 1980, to 11.9% in 2000 (p < 0.001) [31]. A similar decrease was shown in a Greek center, from 57.8% of patients treated with RT before 1990 to 16.8% after 1990, p < 0.001) [32]. Since RT is used more restricted for aggressive or drug resistant tumors, biochemical cure nowadays is potentially lower than previously reported. However, it is important to keep in mind that RT may ultimately lead to cure of GH hypersecretion. The purpose of this review is to present the updates in the efficacy and safety of pituitary RT in acromegalic patients, with an emphasis on the new stereotactic radiation techniques.
Methods
An online search for journal articles relevant to the topic was conducted using the PubMed Database from 2000 up to 2016 by entering combinations of the MeSH terms “acromegaly,” “radiosurgery,” “radiation,” “radiotherapy,” “fractionated,” “Gamma Knife,” “Cyberknife,” and “proton beam.” Articles were limited to the English language. Cited references within articles were also searched for relevancy to the topic. Combined data from multiple studies are presented as weighted means.
Results
Stereotactic radiosurgery (SRS) in acromegaly
Efficacy
In Table 1, 35 SRS studies including 1868 patients are detailed: 26 studies using GK [33–58], 4 with LINAC SRS [59–62], 3 with CK [25, 26, 63] and 2 with proton SRS [64, 65]. The median doses delivered to the tumor margin ranged from 15 to 35 Gy.
Local tumor control
Tumor control, i.e. stable or reduction in size, is 93–100% in 33 published studies including 1746 patients with acromegaly treated with SRS (Table 1); weighted mean tumor control was 98% at a median follow up (mFU) of 59 months, similar to CRT-induced tumor control (Table 4); tumor shrinkage occured in about 50–75% of cases [35, 41, 43, 45].
Biochemical control
In SRS, the remission rate is most probably 44–52% at 5 years at a median dose of 23.5 Gy. In 35 studies on SRS (Table 1) including 1868 acromegalic patients, the weighted mean biochemical control rate of the disease was 44.3% at a mFU of 59 months (18–114 months) [25, 26, 33–65]. In ten studies (including 700 patients) [35, 38, 40, 41, 43, 45, 49, 53, 61, 64] where a Kaplan Meyer estimate of disease control was available and more stringent criteria for normalization were used (i.e. GH < 1 ng/mL and normal age-corrected IGF-1), at 5 years the biochemical control reached 52% [24] and median time to normalization ranged from 12 to 144 months (median weighted 41.5 months).
Even in studies with ≥5 years of mFU there is a large variability of the normalization rate for GH and IGF-1 serum levels, from 12 to 68% of patients (weighted mean 47.4%), increasing up to 47–86% at 10 years [35, 41, 49, 55] (Table 1). This variability may be due to different criteria used to define GH/IGF-1 normalization, different FU duration, pre-irradiation levels of GH /IGF-1, tumor size, use of RT as primary or post-surgery therapy and use of concomitant medical therapies in some of the studies. Overall, biochemical control may seem higher in SRS than in CRT (52% versus 36% at 5 years in a recent meta-analysis), but it did not reach statistical significance, possibly due to a shorter follow up [66] (Table 3).
In addition, 10 to 30% of the patients who were uncontrolled on medical therapy prior to SRS reach GH/IGF1 normalization with medical therapy after SRS [38, 57].
Favorable prognostic factors for hormonal remission after SRS include a higher margin radiation dose, higher maximum dose, and lower initial GH/IGF-1 level [41, 43]. Densely granulated GH-secreting tumors have a similar response to SRS as sparsely granulated tumors [67].
At similar mFU of 55–60 months, a median dose <20 Gy achieved remission in 31% of 216 patients, doses of 20–25 Gy in 47% of 1196 patients and doses >25 Gy in 33% of 390 patients [24].
Some authors [40, 47, 68], but not all [41, 42, 56, 59] have shown that use of somatostatin analogs may decrease the success of SRS (eg, remission rate was 59% in patients off suppressive medications compared with 37% in patients receiving a suppressive medication, most commonly octreotide, at the time of GK treatment) [47]. Therefore temporary withdrawal of the medical treatment before and during RT was suggested [2].
After RT, the use of medical treatment is recommended [1], with yearly interruptions of 1–3 months (depending on the type of medication) to monitor the efficacy of radiation therapy [56].
Recurrence after GK SRS was evaluated in a retrospective analysis of 272 patients with nonfunctioning pituitary adenoma (NFPA) and 271 patients with a hormone secreting-pituitary adenoma, including 148 with GH-secreting adenomas [69]. The mFU after GK was 78 months and disease recurrence occurred in 2.7% of acromegalic patients, less frequent than in patients with NFPA and other hormone secreting adenomas (10% and 5%). In smaller studies, endocrine relapse was described in 1.2–6% of patients with acromegaly [41, 47] at 26–50 months after SRS-induced remission [47].
Repeated SRS irradiation after CRT or FSRT is possible in selected cases, leading to hormonal normalization in 45–90% of 23 published cases with acromegaly [70, 71]. A higher rate of neurological complications (16%) [70], of visual defects (in 3 of 5 re-irradiated patients) [47] and of hypopituitarism occurred in re-treated patients [70]. It was suggested that 50% of the original radiation dose, recalculated as a single-fraction dose, remains active in occulomotor nerve [70] and 40% in the optic nerve [72].
SRS side effects
Radiation-induced hypopituitarism is the main side-effect of SRS, occurring in 0–66% of patients with acromegaly (weighted mean 22%) at mFU of 60.5 months (around 20–40% in half of the studies with a mFU ≥ 4 years and <20% in a third of those studies) (Table 1). The risk is apparently lower than in CRT treated patients (33%) [66] (Tables 3, 4), but shows the same increasing occurrence over time as in patients treated with CRT [55]. Since hypopituitarism can appear anytime between 1 and 10–15 years after RT [40], yearly assessment of pituitary function is recommended in RT treated patients [2].
Predictive factors reported for SRS-induced hypopituitarism are: margin dose, suprasellar extension, invasion in the cavernous sinus, prior craniotomy, pretreatment pituitary gland function, tumour volume and the rigorousness and length of endocrine FU [37, 40, 55, 57, 73]. Hypophysopexy, a surgical pituitary transposition, may reduce the radiation dose to the normal pituitary gland in cases of residual tumor within the cavernous sinus [74].
Radiation-induced optic neuropathy occurs in 0–4.2% of patients usually during the first 3 years after SRS [35, 47]. Maximum point doses <8–10 Gy to the optic nerves and chiasm are recommended for single-fraction SR [75]. However, in most patients without pre-existing cranial nerve injury, systemic comorbidities (e.g., diabetes and hypertension) or prior irradiation, the maximum dose tolerated by the optic apparatus is likely 10–12 Gy, if applied to small portions (2–4%) of the optic pathways [76].
Cranial neuropathies and brain radionecrosis have been reported in 0–5% of patients when marginal doses ≥20 Gy are used [57, 60].
The risk to develop a new brain tumor after SRS appears to be low after mFU of 60 months (0% in the large majority of studies) (Table 1), but longer FU studies are needed to elucidate this effect.
Cerebrovascular disease and mortality after SRS have not been systematically studied, but in 35 SRS studies (Table 1) [25, 26, 33–65], cerebrovascular events have been described in only two studies and consisted of one case of coronary artery stenosis (2.2% of the patients) and 2 transient ischaemic attacks at 72 and 132 months (5.7% of the patient series). In a retrospective series of 42 acromegalic patients cured after CRT (31) or GK (11) compared to 56 patients cured by surgery alone, no difference was observed between irradiated and non-irradiated groups regarding major cardio or cerebrovascular events (10% vs 6% after mFU 16.5 years). In these cured patients, no differences were found between CRT and GK subgroups [77].
These data support the expected brain-sparring effect of SRS, compared to CRT with up to 21% cerebrovascular events reported after 20 years, but longer FU prospective studies are still needed. The cerebrovascular disease rate in CRT treated patients was influenced by the radiation dose and related to atherogenesis in the vascular lining due to radiotoxicity [16]. Published studies support the potential for SRS to cause less vasculopathy than CRT since it irradiates less healthy brain tissue.
More recent studies on mortality failed to confirm an increased risk in patients irradiated with newer techniques [78, 79]. Data from the Danish National registry show that the elevated mortality risk in patients with acromegaly ( HR= 1.3, 95% CI: 1.0-1.7) is uninfluenced by treatment modality [80]. Furthermore, a study including 806 patients with a non-functioning pituitary adenoma from the Dutch National Registry of Growth Hormone Treatment in Adults reported that the frequency of secondary intracranial tumors and mortality did not differ between irradiated and non-irradiated subjects [81]. It is plausible that modern RT techniques, medical treatments which improve the biochemical control, a more careful management of comorbidities or all of the above might influence the life expectancy and the cerebrovascular morbidity in acromegalic patients, even in the irradiated ones [20, 79].
Other effects on neuropsychological performance and quality of life in patients with acromegaly treated with stereotactic RT have not been studied.
Types of SRS
GammaKnife
Most published studies used GK in patients with acromegaly [33–58] with a reported tumor control of 93–100% and biochemical normalization of 46% (17–65%) of 1536 patients, at a mFU of 58 months. New hypopituitarism occured in 22% of patients (2–58%). In a large retrospective study of 136 acromegalic patients treated with GK and followed-up for a median of 61.5 months, 65.4% of the patients achieved remission (mean time to remission 27.5 months). The actuarial remission rates at 2, 4, 6, and 8 years after SRS were 32, 64.5, 73, and 83%, respectively, when normalization criteria were normal age- and gender-matched IGF1 or GH in OGTT < 1 ng/mL, off any medication [43]. New pituitary hormone deficiency occurred in 43 patients (32%) [43].
Cyberknife
The results for 67 acromegalic patients (15 radiated in ≤3 fractions and 52 in >3 fractions with a total dose of 20–25 Gy) showed tumor control in 92–100% and biochemical control in 22.5% of patients (17–44%), after a mFU of 33 months [25, 26, 63]. The apparently lower efficacy of CK is due to a recent study (Iwata et al) which included many large perioptic tumors and used stringent criteria (GH < 1 ng/mL and normal IGF-1) [26] (Table 1). The other 2 studies, including 15 patients, had similar control rates with GK. Hypopituitarism occurred in 0–33% of patients, and no grade 2 or more visual deficits were recorded.
Modified LINAC SRS
In 193 patients with acromegaly this technique induced 95–97% tumor control rate and biochemical remission in 39.4% of patients (23–68%) after mFU of 69 months (54–98) [59–62]. Remission criteria were GH < 2–2.5 ng/mL and/or normal IGF-1. Time to remission was 12–43 months and new hypopituitarism occurred in 20.5% (12–46%) of patients. Visual impairment, cranial nerve deficits and symptomatic brain necrosis occurred in up to 3% of cases and only 1 patient with intracranial malignancy was reported, but it was associated with extracranial metastatic malignancy and was not considered induced by RT [60].
The current data suggest that all the photon SRS techniques achieve similar results.
Proton beam SRS
In 70 acromegalic patients it induced tumor control in 95–100% and biochemical remission in about 50–67% of patients at 5 years [64, 65]. Median time to remission was 30.5 months [65]. The actuarial 3-year and 5-year rates of development of new hypopituitarism were 45 and 62%, median time to deficiency was 40 months [64]. There were no radiation-induced tumors or visual defects. These data suggest that proton SRS achieves similar results with photons SRS.
Hypofractionated SRS
This technique has been used with good results and few visual side-effects in patients with perioptic tumors (within 2–3 mm from the optic nerves or chiasm) for which single session SRS is not suitable. Iwata et al [26] administered CK in 52 patients with GH-secreting pituitary adenomas in a schedule of 3 fractions of 7 Gy each (marginal doses 17–27 Gy) or 5 fractions of 5 Gy each (marginal doses 20–32 Gy), with a mFU of 60 months. The 2 radiation schedules had similar efficacy. The 5- year overall survival and local control was 100%, while stringent hormonal control (random GH < 1 ng/mL or <0.4 ng/mL in OGTT and normal age and sex-adjusted IGF1) was achieved in 17.3% of patients. New hypopituitarism occurred in one patient (2%) but no other major complications [26]. Similar radiation regimens applied to 40 various perioptic pituitary tumors showed 97.5% tumor control and no new hypopituitarism or visual defects at a mFU of 38.5 months [30]. Liao [28] applied 3 fractions with a total dose of 21 Gy with a modified LINAC system to 34 various perioptic pituitary tumors and achieved tumor control in 100% at a mean FU of 37 months, with transient post-treatment diplopia in 1 patient (3%).
A dose–response model for visual pathway tolerance to SRS delivered in 1–5 fractions for perioptic tumors has been recently published [82]. Based on a retrospective evaluation of 262 patients with perioptic tumors, the model suggests a less than 1% incidence of radiation induced optic neuropathy in patients treated with an optic apparatus pathway maximum point dose of 12 Gy in one, 19.5 Gy in three, and 25 Gy in five fractions.
Hypofractionated GK in two or three fractions with a mean margin dose of 7.2 Gy (range 5–8 Gy) was used in ten patients with giant pituitary tumors (adenoma size >4 cm, five functional tumors), after failed surgery. Tumor control was achieved in 100% of cases [83]. In 60% a tumor shrinkage occured during the mFU of 31 months. Hypopituitarism occurred in 10% (one patient) after two fractionated SRS in 5 years.
Fractionated stereotactic radiotherapy (FSRT)
Few reports, including 261 patients, studied the outcome of FSRT in acromegaly, using median total dose of 49 Gy (range 45–54 Gy) [84–91] (Table 2).
Efficacy
Local tumor control was achieved in 97% (92–100%) of patients after mFU of 71 months, similar to SRS or CRT. Tumor shrinkage occurred in 48–53% [85, 86]
Biochemical control varied from 18 to 75%, with a weighted mean of 35% at mFU of 71 months in published series, most of them using stringent GH/IGF1 control criteria. Diallo et al [85] reported 34 acromegalic patients treated by FSRT with a total dose of 50 Gy, mean FU of 12 years. Hormonal remission rate was 25% at 5 years, 43% at 10 years, and 50% at 15 years. Mean time to normalization was 28 months.
In a prospective series of 35 patients with acromegaly treated with SRS (21) or FSRT (12) according to the radiation toxicity risk, after a mFU of 8 years biochemical cure (combined methods) was achieved in 23% and the 5-year local control was 97.1% [62].
In a series of 34 giant GH-secreting tumors, none cured after surgery, from 12 patients who were treated with FSRT (5), CRT (5) and SRS (2), only one patient achieved GH/IGF1 control 1 year following SRS. Seven other irradiated patients were controlled after association with medical treatment [92].
Side effects
Hypopituitarism developed in 29.4% of patients at mFU of 71 months [84–91]. Visual defects occurred in 0–5%, secondary brain tumor (meningioma) was recorded in 1 patient (1.8%), no cranial neuropathies or brain necrosis were reported. In our series of 77 acromegalic patients treated with FSRT with a mFU of 73 months (6–264 months), clinical or imaging signs for stroke occurred in 9% of patients [86], similar with the 8.2% rate in the study of Kim et al [87], while in the study of Diallo et al [85] the rate for stroke was 0% at more than 10 years of FU (Table 2).
Overall, FSRT seems to have a similar efficacy with SRS and risk rate for hypopituitarism and neuropathies, but the risk of stroke seems higher and should be further evaluated (Table 4).
Comparison between SRS and fractionated RT
In Table 4 comparative data, expressed as weighted means from published studies, are shown for the RT methods used in acromegaly. Earlier reports suggested that the declining of serum GH concentration after GK SRS is faster compared with fractionated conventional RT [59, 60, 93]; others did not confirm this finding [44, 45, 51, 56, 61]. The greatest effect occurs within the first 2 years from SRS [40, 59].
In a study comparing the results of GK treatment in 32 patients with acromegaly followed at our center (50% isodose 16–22 Gy, mFU 41 months) to those of FSRT in 77 patients (total dose 45–50 Gy, mFU 73 months) [86], the cumulative probability of GH normalization <1 ng/mL was higher in GK treated patients (13% at 2 years and 29% at 4 years, compared with FSRT: 5% at 2 years and 15% at 5 years [86], p < 0.05); after SRS the median nadir GH in OGTT was lower after 2 years (2.5 ± 1.8 ng/mL vs 4.8 ± 8.2 ng/mL, p < 0.05) and the rate of GH decrease was higher at 6 months (45 ± 20% vs 38 ± 59% respectively, p < 0.05), but not afterwards [86].
A recent systematic review and meta-analysis comparing the outcomes of SRS and fractionated RT (including FSRT and CRT) in acromegaly analyzed 30 eligible studies including 2464 patients, with a FU between 12–240 months [66]. Compared to RT, SRS was associated with a nonsignificant trend of higher IGF-I-based remission rate (52 vs 37%, P = 0.14) or GH-based remission (49 vs 36%) at the latest FU period. The length of FU did not significantly affect remission rate.
In the RT group, treatment-naïve patients had similar remission rates compared with patients who had received a prior treatment (surgery, SRS, medical). SRS had a lower incidence of hypopituitarism than RT with borderline statistical significance (32 vs 51%, p = 0.05), the difference being largely due to hypogonadism. No comparison was reported for brain necrosis, headache, and secondary malignancy outcomes after either intervention. The authors concluded that SRS may be more efficient than fractionated RT, but the strength of evidence was very low due to the noncomparative nature of the research, increased risk of bias, imprecision, and substantial heterogeneity among studies [66].
Indeed, data presented in Table 4 also suggest a slightly increased benefit of SRS compared mainly to CRT, regarding biochemical control and the risk for radiation-induced hypopituitarism and cerebrovascular disease; visual deficits are similar in SRS and both fractionated RT subtypes. The risk for radionecrosis and second brain tumors may be slightly lower after SRS and FSRT than after CRT, but longer FU studies are needed in order to elucidate these effects.
Conclusion
Although the use of radiotherapy in patients with acromegaly has decreased with the advances in medical and surgical treatments, it remains an effective treatment option in patients with unsuccessful surgery and/or with intolerance, lack of response, or unavailability of medical therapy in selected countries. SRS may be potentially more effective than conventional RT regarding biochemical remission. There have been reports that SRS has a lower rate of induced hypopituitarism, but long term FU is limited and the large heterogeneity of the studies makes the comparison difficult. Long-term studies evaluating cerebrovascular disease and mortality rate after the new stereotactic techniques are needed, in order to evaluate their brain-sparing effects.
References
Melmed S, Colao A, Barkan A et al (2009) Guidelines for acromegaly management: an update. J Clin Endocrinol Metab 94(5):1509–1517
Katznelson L, Laws ER Jr, Melmed S et al (2014) Acromegaly: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 99(11):3933–3951
Minniti G, Clarke E, Scaringi C, Enrici RM (2016) Stereotactic radiotherapy and radiosurgery for non-functioning and secreting pituitary adenomas. Rep Pract Oncol Radiother 21(4):370–378
Parhar PK, Duckworth T, Shah P et al (2010) Decreasing temporal lobe dose with five-field intensity-modulated radiotherapy for treatment of pituitary macroadenomas. Int J Radiat Oncol Biol Phys 78(2):379–384
Barrande G, Pittino-Lungo M, Coste J et al (2000) Hormonal and metabolic effects of radiotherapy in acromegaly: long-term results in 128 patients followed in a single center. J Clin Endocrinol Metab 85(10):3779–3785
Biermasz NR, Dulken HV, Roelfsema F (2000) Postoperative radiotherapy in acromegaly is effective in reducing GH concentration to safe levels. Clin Endocrinol 53(3):321–327
Brada M, Rajan B, Traish D et al (1993) The long-term efficacy of conservative surgery and radiotherapy in the control of pituitary adenomas. Clin Endocrinol 38(6):571–578
Epaminonda P, Porretti S, Cappiello V, Beck-Peccoz P, Faglia G, Arosio M (2001) Efficacy of radiotherapy in normalizing serum IGF-I, acid-labile subunit (ALS) and IGFBP-3 levels in acromegaly. Clin Endocrinol 55(2):183–189
Jenkins PJ, Bates P, Carson MN, Stewart PM, Wass JA (2006) Conventional pituitary irradiation is effective in lowering serum growth hormone and insulin-like growth factor-I in patients with acromegaly. J Clin Endocrinol Metab 91(4):1239–1245
Minniti G, Jaffrain-Rea ML, Osti M et al (2005) The long-term efficacy of conventional radiotherapy in patients with GH-secreting pituitary adenomas. Clin Endocrinol 62(2):210–216
Powell JS, Wardlaw SL, Post KD, Freda PU (2000) Outcome of radiotherapy for acromegaly using normalization of insulin-like growth factor I to define cure. J Clin Endocrinol Metab 85(5):2068–2071
Jallad RS, Musolino NR, Salgado LR, Bronstein MD (2007) Treatment of acromegaly: is there still a place for radiotherapy? Pituitary 10(1):53–59
Gonzalez B, Vargas G, Espinosa-de-los-Monteros AL, Sosa E, Mercado M (2011) Efficacy and safety of radiotherapy in acromegaly. Arch Med Res 42(1):48–52
Cozzi R, Barausse M, Asnaghi D, Dallabonzana D, Lodrini S, Attanasio R (2001) Failure of radiotherapy in acromegaly. Eur J Endocrinol 145(6):717–726
Minniti G, Traish D, Ashley S, Gonsalves A, Brada M (2005) Risk of second brain tumor after conservative surgery and radiotherapy for pituitary adenoma: update after an additional 10 years. J Clin Endocrinol Metab 90(2):800–804
Brada M, Burchell L, Ashley S, Traish D (1999) The incidence of cerebrovascular accidents in patients with pituitary adenoma. Int J Radiat Oncol Biol Phys 45(3):693–698
Lecumberri B, Estrada J, Garcia-Uria J et al (2015) Neurocognitive long-term impact of two-field conventional radiotherapy in adult patients with operated pituitary adenomas. Pituitary 18(6):782–795
van der Klaauw AA, Biermasz NR, Hoftijzer HC, Pereira AM, Romijn JA (2008) Previous radiotherapy negatively influences quality of life during 4 years of follow-up in patients cured from acromegaly. Clin Endocrinol 69(1):123–128
Bex M, Abs R, T’Sjoen G et al (2007) AcroBel–the Belgian registry on acromegaly: a survey of the ‘real-life’ outcome in 418 acromegalic subjects. Eur J Endocrinol 157(4):399–409
Colao A, Vandeva S, Pivonello R et al (2014) Could different treatment approaches in acromegaly influence life expectancy? A comparative study between Bulgaria and Campania (Italy). Eur J Endocrinol 171(2):263–273
Mestron A, Webb SM, Astorga R et al (2004) Epidemiology, clinical characteristics, outcome, morbidity and mortality in acromegaly based on the Spanish acromegaly registry (Registro Espanol de Acromegalia, REA). Eur J Endocrinol 151(4):439–446
Sherlock M, Reulen RC, Alonso AA et al (2009) ACTH deficiency, higher doses of hydrocortisone replacement, and radiotherapy are independent predictors of mortality in patients with acromegaly. J Clin Endocrinol Metab 94(11):4216–4223
Ayuk J, Clayton RN, Holder G, Sheppard MC, Stewart PM, Bates AS (2004) Growth hormone and pituitary radiotherapy, but not serum insulin-like growth factor-I concentrations, predict excess mortality in patients with acromegaly. J Clin Endocrinol Metab 89(4):1613–1617
Minniti G, Osti MF, Niyazi M (2016) Target delineation and optimal radiosurgical dose for pituitary tumors. Radiat Oncol 11(1):135
Roberts BK, Ouyang DL, Lad SP et al (2007) Efficacy and safety of CyberKnife radiosurgery for acromegaly. Pituitary 10(1):19–25
Iwata H, Sato K, Nomura R et al (2016) Long-term results of hypofractionated stereotactic radiotherapy with CyberKnife for growth hormone-secreting pituitary adenoma: evaluation by the Cortina consensus. J Neurooncol 128(2):267–275
Gevaert T, Boussaer M, Engels B et al (2013) Evaluation of the clinical usefulness for using verification images during frameless radiosurgery. Radiother Oncol 108(1):114–117
Liao HI, Wang CC, Wei KC et al (2014) Fractionated stereotactic radiosurgery using the Novalis system for the management of pituitary adenomas close to the optic apparatus. J Clin Neurosci 21(1):111–115
Petrovich Z, Jozsef G, Yu C, Apuzzo ML (2003) Radiotherapy and stereotactic radiosurgery for pituitary tumors. Neurosurg Clin N Am 14(1):147–166
Puataweepong P, Dhanachai M, Hansasuta A et al. The clinical outcome of hypofractionated stereotactic radiotherapy with CyberKnife robotic radiosurgery for perioptic pituitary adenoma. Technol Cancer Res Treat 2016;15(6):NP10–NP15
Sesmilo G, Gaztambide S, Venegas E et al (2013) Changes in acromegaly treatment over four decades in Spain: analysis of the Spanish acromegaly registry (REA). Pituitary 16(1):115–121
Karapanou O, Tzanela M, Christoforaki M et al (2016) Therapeutic trends and outcome of acromegaly: a single center experience over a 40-year period. Hormones 15(3):368–376
Zhang N, Pan L, Wang EM, Dai JZ, Wang BJ, Cai PW (2000) Radiosurgery for growth hormone-producing pituitary adenomas. J Neurosurg 93(Suppl 3):6–9
Wan H, Chihiro O, Yuan S (2009) MASEP gamma knife radiosurgery for secretory pituitary adenomas: experience in 347 consecutive cases. J Exp Clin Cancer Res 28:36
Vik-Mo EO, Oksnes M, Pedersen PH et al (2007) Gamma knife stereotactic radiosurgery for acromegaly. Eur J Endocrinol 157(3):255–263
Sicignano G, Losa M, Del VA et al (2012) Dosimetric factors associated with pituitary function after gamma knife surgery (GKS) of pituitary adenomas. Radiother Oncol 104(1):119–124
Sheehan JP, Pouratian N, Steiner L, Laws ER, Vance ML (2011) Gamma Knife surgery for pituitary adenomas: factors related to radiological and endocrine outcomes. J Neurosurg 114(2):303–309
Ronchi CL, Attanasio R, Verrua E et al (2009) Efficacy and tolerability of gamma knife radiosurgery in acromegaly: a 10-year follow-up study. Clin Endocrinol 71(6):846–852
Poon TL, Leung SC, Poon CY, Yu CP (2010) Predictors of outcome following Gamma Knife surgery for acromegaly. J Neurosurg 113(Suppl):149–152
Pollock BE, Jacob JT, Brown PD, Nippoldt TB (2007) Radiosurgery of growth hormone-producing pituitary adenomas: factors associated with biochemical remission. J Neurosurg 106(5):833–838
Losa M, Gioia L, Picozzi P et al (2008) The role of stereotactic radiotherapy in patients with growth hormone-secreting pituitary adenoma. J Clin Endocrinol Metab 93(7):2546–2552
Liu X, Kano H, Kondziolka D et al (2012) Gamma knife radiosurgery for clinically persistent acromegaly. J Neurooncol 109(1):71–79
Lee CC, Vance ML, Xu Z et al (2014) Stereotactic radiosurgery for acromegaly. J Clin Endocrinol Metab 99(4):1273–1281
Kobayashi T, Mori Y, Uchiyama Y, Kida Y, Fujitani S (2005) Long-term results of gamma knife surgery for growth hormone-producing pituitary adenoma: is the disease difficult to cure? J Neurosurg 102:119–123
Jezkova J, Marek J, Hana V et al (2006) Gamma knife radiosurgery for acromegaly-long-term experience. Clin Endocrinol 64(5):588–595.
Jane JA Jr, Vance ML, Woodburn CJ, Laws ER Jr (2003) Stereotactic radiosurgery for hypersecreting pituitary tumors: part of a multimodality approach. Neurosurg Focus 14(5):e12
Jagannathan J, Sheehan JP, Pouratian N, Laws ER Jr, Steiner L, Vance ML (2008) Gamma knife radiosurgery for acromegaly: outcomes after failed transsphenoidal surgery. Neurosurgery 62(6):1262–1269
Izawa M, Hayashi M, Nakaya K et al (2000) Gamma knife radiosurgery for pituitary adenomas. J Neurosurg 93(Suppl 3):19–22
Iwai Y, Yamanaka K, Yoshimura M, Kawasaki I, Yamagami K, Yoshioka K (2010) Gamma knife radiosurgery for growth hormone-producing adenomas. J Clin Neurosci 17(3):299–304
Hayashi M, Chernov M, Tamura N et al (2010) Gamma Knife robotic microradiosurgery of pituitary adenomas invading the cavernous sinus: treatment concept and results in 89 cases. J Neurooncol 98(2):185–194
Gutt B, Wowra B, Alexandrov R et al (2005) Gamma-knife surgery is effective in normalising plasma insulin-like growth factor I in patients with acromegaly. Exp Clin Endocrinol Diabetes 113(4):219–224
Grant RA, Whicker M, Lleva R, Knisely JP, Inzucchi SE, Chiang VL (2014) Efficacy and safety of higher dose stereotactic radiosurgery for functional pituitary adenomas: a preliminary report. World Neurosurg 82(1–2):195–201
Franzin A, Spatola G, Losa M, Picozzi P, Mortini P (2012) Results of gamma knife radiosurgery in acromegaly. Int J Endocrinol 2012:342034
Erdur FM, Kilic T, Peker S, Celik O, Kadioglu P (2011) Gammaknife radiosurgery in patients with acromegaly. J Clin Neurosci 18(12):1616–1620
Cohen-Inbar O, Ramesh A, Xu Z, Vance ML, Schlesinger D, Sheehan JP (2016) Gamma knife radiosurgery in patients with persistent acromegaly or Cushing’s disease: long-term risk of hypopituitarism. Clin Endocrinol 84(4):524–531
Castinetti F, Taieb D, Kuhn JM et al (2005) Outcome of gamma knife radiosurgery in 82 patients with acromegaly: correlation with initial hypersecretion. J Clin Endocrinol Metab 90(8):4483–4488
Castinetti F, Nagai M, Morange I et al (2009) Long-term results of stereotactic radiosurgery in secretory pituitary adenomas. J Clin Endocrinol Metab 94(9):3400–3407
Attanasio R, Epaminonda P, Motti E et al (2003) Gamma-knife radiosurgery in acromegaly: a 4-year follow-up study. J Clin Endocrinol Metab 88(7):3105–3112
Yan JL, Chang CN, Chuang CC et al (2013) Long-term follow-up of patients with surgical intractable acromegaly after linear accelerator radiosurgery. J Formos Med Assoc 112(7):416–420
Wilson PJ, De-Loyde KJ, Williams JR, Smee RI (2013) Acromegaly: a single centre’s experience of stereotactic radiosurgery and radiotherapy for growth hormone secreting pituitary tumours with the linear accelerator. J Clin Neurosci 20(11):1506–1513
Voges J, Kocher M, Runge M et al (2006) Linear accelerator radiosurgery for pituitary macroadenomas: a 7-year follow-up study. Cancer 107(6):1355–1364
Bostrom JP, Kinfe T, Meyer A et al (2015) Treatment of acromegaly patients with risk-adapted single or fractionated stereotactic high-precision radiotherapy: High local control and low toxicity in a pooled series. Strahlenther Onkol 191(6):477–485
Cho CB, Park HK, Joo WI, Chough CK, Lee KJ, Rha HK (2009) Stereotactic Radiosurgery with the CyberKnife for Pituitary Adenomas. J Korean Neurosurg Soc 45(3):157–163
Wattson DA, Tanguturi SK, Spiegel DY et al (2014) Outcomes of proton therapy for patients with functional pituitary adenomas. Int J Radiat Oncol Biol Phys 90(3):532–539
Petit JH, Biller BM, Coen JJ et al (2007) Proton stereotactic radiosurgery in management of persistent acromegaly. Endocr Pract 13(7):726–734
Abu Dabrh AM, Asi N, Farah WH et al (2015) Radiotherapy versus radiosurgery in treating patients with acromegaly: a systematic review and meta-analysis. Endocr Pract 21(8):943–956
Lee CC, Vance ML, Lopes MB, Xu Z, Chen CJ, Sheehan J (2015) Stereotactic radiosurgery for acromegaly: outcomes by adenoma subtype. Pituitary 18(3):326–334
Landolt AM, Haller D, Lomax N et al (2000) Octreotide may act as a radioprotective agent in acromegaly. J Clin Endocrinol Metab 85(3):1287–1289
Losa M, Spatola G, Albano L et al. Frequency, pattern, and outcome of recurrences after gamma knife radiosurgery for pituitary adenomas. Endocrine 2016
Landolt AM, Lomax N, Scheib SG, Girard J (2006) Gamma Knife surgery after fractionated radiotherapy for acromegaly. J Neurosurg 105:31–36
Swords FM, Monson JP, Besser GM et al (2009) Gamma knife radiosurgery: a safe and effective salvage treatment for pituitary tumours not controlled despite conventional radiotherapy. Eur J Endocrinol 161(6):819–828
Flickinger JC, Deutsch M, Lunsford LD (1989) Repeat megavoltage irradiation of pituitary and suprasellar tumors. Int J Radiat Oncol Biol Phys 17(1):171–175
Xu Z, Lee VM, Schlesinger D, Sheehan JP (2013) Hypopituitarism after stereotactic radiosurgery for pituitary adenomas. Neurosurgery 72(4):630–637
Taussky P, Kalra R, Coppens J, Mohebali J, Jensen R, Couldwell WT (2011) Endocrinological outcome after pituitary transposition (hypophysopexy) and adjuvant radiotherapy for tumors involving the cavernous sinus. J Neurosurg 115(1):55–62
Mayo C, Martel MK, Marks LB, Flickinger J, Nam J, Kirkpatrick J (2010) Radiation dose-volume effects of optic nerves and chiasm. Int J Radiat Oncol Biol Phys 76(3 suppl):28–35
Leavitt JA, Stafford SL, Link MJ, Pollock BE (2013) Long-term evaluation of radiation-induced optic neuropathy after single-fraction stereotactic radiosurgery. Int J Radiat Oncol Biol Phys 87(3):524–527
Ronchi CL, Verrua E, Ferrante E et al (2011) Long-term effects of radiotherapy on cardiovascular risk factors in acromegaly. Eur J Endocrinol 164(5):675–684
Sattler MG, van Beek AP, Wolffenbuttel BH et al (2012) The incidence of second tumours and mortality in pituitary adenoma patients treated with postoperative radiotherapy versus surgery alone. Radiother Oncol 104(1):125–130
Mercado M, Gonzalez B, Vargas G et al (2014) Successful mortality reduction and control of comorbidities in patients with acromegaly followed at a highly specialized multidisciplinary clinic. J Clin Endocrinol Metab 99(12):4438–4446
Dal J, Feldt-Rasmussen U, Andersen M et al (2016) Acromegaly incidence, prevalence, complications and long-term prognosis: a nationwide cohort study. Eur J Endocrinol 175(3):181–190
van Varsseveld NC, van Bunderen CC, Ubachs DH et al (2015) Cerebrovascular events, secondary intracranial tumors, and mortality after radiotherapy for nonfunctioning pituitary adenomas: a subanalysis from the Dutch national registry of growth hormone treatment in adults. J Clin Endocrinol Metab 100(3):1104–1112
Hiniker SM, Modlin LA, Choi CY et al (2016) Dose-response modeling of the visual pathway tolerance to single-fraction and hypofractionated stereotactic radiosurgery. Semin Radiat Oncol 26(2):97–104
Zhao K, Liu X, Liu D et al (2016) Fractionated Gamma Knife surgery for giant pituitary adenomas. Clin Neurol Neurosurg 150:139–142
Colin P, Jovenin N, Delemer B et al (2005) Treatment of pituitary adenomas by fractionated stereotactic radiotherapy: a prospective study of 110 patients. Int J Radiat Oncol Biol Phys 62(2):333–341
Diallo AM, Colin P, Litre CF et al (2015) Long-term results of fractionated stereotactic radiotherapy as third-line treatment in acromegaly. Endocrine 50(3):741–748
Gheorghiu ML, Purice M, Poiana C, Coculescu M. Efficacy of pituitary radiotherapy on growth hormone (GH) secretion in patients with acromegaly. Abstract book for the American Association of Clinical Endocrinologists’ 21st Annual Meeting and Clinical Congress, May 23–27, Philadelphia, 2012, A152. 2012
Kim MY, Kim JH, Oh YK, Kim E (2016) Long-term outcomes of surgery and radiotherapy for secreting and non-secreting pituitary adenoma. Radiat Oncol J 34(2):121–127
Milker-Zabel S, Zabel A, Huber P, Schlegel W, Wannenmacher M, Debus J (2004) Stereotactic conformal radiotherapy in patients with growth hormone-secreting pituitary adenoma. Int J Radiat Oncol Biol Phys 59(4):1088–1096
Minniti G, Traish D, Ashley S, Gonsalves A, Brada M (2006) Fractionated stereotactic conformal radiotherapy for secreting and nonsecreting pituitary adenomas. Clin Endocrinol 64(5):542–548.
Patt H, Jalali R, Yerawar C et al (2016) High-precision conformal fractionated radiotherapy is effective in achieving remission in patients with acromegaly after failed transphenoidal surgery. Endocr Pract 22(2):162–172
Roug S, Rasmussen AK, Juhler M et al (2010) Fractionated stereotactic radiotherapy in patients with acromegaly: an interim single-centre audit. Eur J Endocrinol 162(4):685–694
Shimon I, Jallad RS, Fleseriu M, Yedinak CG, Greenman Y, Bronstein MD (2015) Giant GH-secreting pituitary adenomas: management of rare and aggressive pituitary tumors. Eur J Endocrinol 172(6):707–713
Landolt AM, Haller D, Lomax N et al (1998) Stereotactic radiosurgery for recurrent surgically treated acromegaly: comparison with fractionated radiotherapy. J Neurosurg 88(6):1002–1008
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that she has no conflict of interest regarding this manuscript. This article does not contain any direct studies with human participants or animals performed by the author, as it was a review
Rights and permissions
About this article
Cite this article
Gheorghiu, M.L. Updates in outcomes of stereotactic radiation therapy in acromegaly. Pituitary 20, 154–168 (2017). https://doi.org/10.1007/s11102-016-0783-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-016-0783-5