Abstract
Endocannabinoids and opiates have regulatory role in some physiological functions in mammals but their interaction(s) have not been studied in avian. This survey is designed to investigate interaction of these systems on feeding behavior in neonatal chickens. In experiment 1, chicken intracerebroventricular (ICV) injected with saline, DAMGO (µ-opioid receptors agonist, 125 pmol), SR141716A (CB1 receptors antagonist, 6.25 µg) and SR141716A + DAMGO. In experiment 2, saline, DAMGO, AM630 (CB2 receptors antagonist, 1.25 µg) and DAMGO + AM630. Experiments 3–6 followed the procedure similar to experiments 1 and 2, except DPDPE (δ-opioid receptors agonist, 40 pmol) and U-50488H (κ-opioid receptors agonist, 30 nmol) instead of DAMGO were used. In experiment 7, saline, Naloxone (opioid receptors antagonist, 5 µg), 2-AG (CB1 receptors agonist, 2 µg), Naloxone + 2-AG were used. Experiment 8 was similar to experiment 7, except CB65 (CB2 receptors agonist, 1.25 µg) used instead of 2-AG. Cumulative food intake was recorded until 120 min post injection. Data provided that, ICV injection of DAMGO decreased food intake and its effect amplified by CB1 and CB2 receptors antagonist (P < 0.001). DPDPE increased food intake and CB2 receptors antagonist blocked DPDPE-induced hyperphagia (P < 0.001). U-50488H-induced feeding but its effect did not alter via CB1 and CB2 receptors antagonist (P > 0.05). Hyperphagia-induced by CB1 and CB2 receptors agonist amplified by naloxone (P < 0.001). Perhaps there is interaction between endocannabinoid and opioidergic systems on appetite regulation in chicken.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
To date much progress has been done to identify mediatory effect of neurotransmitters on feeding behavior in mammals but food intake regulation in domestic poultry is not fully studied (Zendehdel and Hassanpour 2014). It is well documented that central feeding behavior is not regulated via a single neuropeptide and a wide distributed neural network interacts with diversity of neurotransmitters on feeding status (Irwin et al. 2008; Zendehdel et al. 2013a, 2014a). Cannabinoids (CBs) were originally characterized as psychoactive ingredients of Cannabis sativa plant or marijuana (∆9-tetrahydrocannabinol, THC) (Novoseletsky et al. 2011). The isolation of CBs had important progress in neuropharmacology. To date two CB receptors have been identified which are belong to G-protein coupled receptors (GCPRs). CB1 receptors are mainly expressed in presynaptic terminals of inhibitory and excitatory nerves in the central nervous system (CNS) in both mammals and birds (Novoseletsky et al. 2011; Sharkey et al. 2014). CB2 receptors frequently found on cells and organs of the immune system in the peripheral nervous system (PNS) but also expressed in the brain (Onaivi et al. 2012). Pharmacological information revealed that CB receptors mainly expressed in prefrontal cortex (PFC), amygdala, septohippocampal system and arcuate nucleus (ARC) (Tambaro et al. 2013). It is suggested CBs have prominent role on feeding regulation which agonists stimulate appetite while antagonists have opposite effect in humans, rodents (Chen et al. 2006; Wiley et al. 2012) and poultry (Novoseletsky et al. 2011).
Opioids are known to be inhibitory neurotransmitters and three opioid receptor subtypes (μ, κ and δ) have been identified; all are members of the GPCR (Chen et al. 2006; Le Merrer et al. 2009). Opioid receptors are expressed in several parts of the hypothalamus, nucleus accumbens (NAcc), amygdala, ventral tegmental area (VTA) and nucleus tractus solitaries (NTS) which have regulatory role in pain perception, respiration, immune system and food intake (Barnes et al. 2006; Kozlov et al. 2013). Activation of central μ-opioid receptors increases food consumption (Pritchett et al. 2010). Central injection of DAMGO and DPDPE (µ- and δ-receptors agonists, respectively) amplified eating in mammals (Taha, 2010; Kaneko et al. 2012). There is evidence that food-intake regulation mechanisms are different among mammals and birds (Zendehdel and Hassanpour, 2014). For instance, ICV injection of µ-opioid receptors decreases food intake while δ- and κ-opioid receptors increase food intake in chicks (Bungo et al. 2005). Interactions between opioids and other neurotransmitters have not been fully explored (Pritchett et al. 2010). It is suggested that neurological interaction exist between opioids and CBs which share a diversity of pharmacological properties e.g. hypothermia, sedation, hypotension and anti-nociception (Pickel et al. 2004). Increasing evidences suggest pro-dynorphins and pro-enkephalins regulate endocannabinoid level in animal’s brain. These phenomena have supported the possible existence of functional linkage between two mechanisms which CBs and opioids mimic endogenous ligands, through the GPCRs, CB and opioid receptors (Manzanares et al. 2005). Pre-treatment with ∆9-THC increases enkephalins levels in the NAcc of mice (Ibegbu et al. 2011). It seems, these two systems interact via µ- and κ-opioid receptors (Braida et al. 2001; Ibegbu et al. 2011). Combined central injection of opioid receptor antagonist and CB1 receptors agonists produced a synergistic inhibitory effect on feeding behavior in rat. However, direct molecular and cellular mechanism underlying this interconnection is not fully elicited (Chen et al. 2006).
In the ARC nucleus of hypothalamus, neuropeptide Y (NPY)/agouti related protein (AgRP) and pro-opiomelanocortin (POMC) and cocaine/amphetamine regulated transcript (CART) are the main sites which regulates appetite (Zendehdel et al. 2013b). Opioid receptors involve in orexigenic effect of NPY and AgRP neurons in the ARC (Barnes et al. 2006) where orexigenic effect of NPY mediates via µ-opioid receptors in meat type chickens (Dodo et al. 2005). Presumably, opioid and CB receptors in the NAcc interact with each other to modulate feeding (Skelly et al. 2010). No report was found on involvement of CBergic system on opioid-induced feeding behavior in neonatal chicken. So, the current research was conducted to specifically examine the role of ICV injection of CB receptors agonists and antagonists (CB1 and CB2) on opioid receptors (µ, δ and κ)-induced feeding in neonatal chickens.
Materials and Methods
Animals
To assume effect of CBergic system on opioid-induced feeding behavior, a batch of 384 1 day-old layer-type chickens (Seamorgh Co. Iran) were kept for 2 days as flocks. Then birds randomly allocated into their individual cages and kept at temperature of 30 ± 1 °C with 50 ± 2 % humidity until 5 days of age (Olanrewaju et al. 2006). During the study, animals provided mesh diet contains 21 % crude protein and 2850 kcal/kg of metabolizable energy (Animal Science Research Institute Co. Iran). Chickens had ad libitum access to food and fresh water during the study. Just 3-h prior the injections, animals were food deprived (FD3) but given free access to water. All birds received ICV injections at 5 days of age. Animal handling and experimental procedures were performed according to the Guide for the Care and Use of Laboratory animals by the National Institutes of Health (USA) and the current laws of the Iranian government for animal care.
Experimental Drugs
SR141716A (selective CB1 receptors antagonist), AM630 (selective CB2 receptors antagonist), DAMGO (µ-opioid receptors agonist), DPDPE (δ-opioid receptors agonist), U-50488H (κ-opioid receptors agonist), 2-AG (CB1 receptors agonist), CB65 (CB2 receptors agonist,), Naloxone and Evans blue were purchased from Sigma Co. (Sigma, USA). Drugs except CB1 receptors agonist (2-AG) were dissolved in 0.1 % Evans blue solution which was prepared in either 0.85 % saline. 2-AG first dissolved in absolute dimethyl sulfoxide (DMSO) then diluted with 0.85 % saline containing Evans blue at a ratio of 1/250. The control group in 2-AG treated experiment received DMSO containing saline diluted in mentioned ration.
ICV Injection Procedures
Former to start experiments, chickens were weighed and based on their body weight distributed into treatment groups. So, that the average weight between treatment groups was as uniform as possible. To assume interaction of CBergic and opioidergic systems on food intake in this study, eight experiments were designed, each include four treatment groups within 12 replicates in each group (n = 48 birds per experiment). ICV injections were done using a microsyringe (Hamilton, Switzerland) without anesthesia in accordance to Davis et al. (1979) and Furuse et al. (1997). In this method, head of the chicken was held with an acrylic device while the bill holder was 45° and calvarium parallel to the surface of table (Van Tienhoven and Juhasz 1962). In a plate a hole was drilled which the skull over the right lateral ventricle immediately overlaid via this plate. In this technique microsyringe inserts into the right ventricle using the hole and tip of the needle penetrated 4 mm beneath the skin of the skull. There is no ICV injection induced physiological stress using this method in neonatal birds (Saito et al. 2005). At the end of the each experiment, accuracy of placement of the injection in the ventricle was verified by presence of Evans blue followed by slicing the frozen brain tissue. In each group, 12 birds received injections but just data of those individuals were used for analysis where dye was present into their lateral ventricle (n = 9–12 chickens/group). All experimental procedures were done from 9:00 a.m. until 1:00 p.m.
Food Intake Measurement Procedure
In this study, eight experiments were designed, each with four treatment groups: A, B, C and D, groups (n = 48 in each experiment). In experiment 1, 3 h-fasted chickens were ICV injected as follows: (A) saline, (B) DAMGO (µ-opioid receptors agonist, 125 pmol), (C): SR141716A (selective CB1 receptors antagonist, 6.25 µg), (D): co-injection of SR141716A + DAMGO. Birds in experiment 2, injected with (A) saline, (B) DAMGO (125 pmol), (C): AM630 (selective CB2 receptors antagonist, 1.25 µg), (D): co-injection of DAMGO + CB2 receptors antagonist. Birds in experiments 3–6 followed the procedure similar to experiment 1 and 2, except that chickens received ICV injection of DPDPE (δ-opioid receptors agonist, 40 pmol) and U-50488H (κ-opioid receptors agonist, 30 nmol) instead of DAMGO. In experiment 7, (A) saline, (B) Naloxone (non-selective opioid antagonist, 5 µg), (C): 2-AG (selective CB1 receptors agonist, 2 µg), (D): co-injection of Naloxone + 2-AG. In experiment 8, followed the procedure similar to experiment 7 except that birds received ICV injection of CB65 (selective CB2 receptors agonist, 1.25 µg) instead of 2-AG. All injections were done in a volume of 10 µl. In all experiments, control group ICV injected with 10 µl saline containing Evans blue. Each chick was injected once only. Right away after injection, chickens were returned to their individual cages and provided ad libitum food (pre-weighed) and water. Cumulative food intake was recorded at 30, 60 and 120 post injection. Food consumption was calculated as percent of body weight (to adjust differences between body weights) (% BW). Injection dosages were calculated based on previous (Steinman et al. 1987; Bungo et al. 2004, 2005; Khan et al. 2009; Emadi et al. 2011) and our pilot studies (un-published).
Statistical Analysis
Cumulative food intake (% BW) was analyzed by two-way analysis of variance (ANOVA) for repeated measurement using SPSS 16.0 for Windows and is presented as mean ± SEM. For treatment showing a main effect by ANOVA, means have compared by Duncan test. P < 0.05 was considered as significant differences between treatments.
Results
Effect of CBergic and opioidergic systems on cumulative food intake in FD3 neonatal layer-type chickens is presented in Figs. 1, 2, 3, 4, 5, 6, 7, and 8. According to the results, ICV injection of sub effective dose of selective CB1 (SR141716A, 6.25 µg) and CB2 (AM630, 1.25 µg,) receptors antagonists had no significant effect on food intake (% BW) compared to control group in experiments 1–6 (P > 0.05). So, in this study we used from sub effective dose for CB1 and CB2 receptors antagonists.
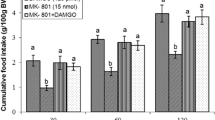
Effect of ICV injection of SR141716A (6.25 µg), DAMGO (125 pmol), and their combination on cumulative food intake (% BW) in neonatal chickens. SR141716A: selective CB1 receptors antagonist, DAMGO: µ-opioid receptors agonist. There are significant differences between groups with different superscripts in a column (a, b and c; P < 0.05)
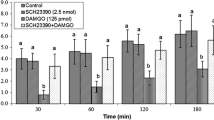
Effect of ICV injection of AM630 (1.25 µg), DAMGO (125 pmol), and their combination on cumulative food intake (% BW) in neonatal chickens. AM630: selective CB2 receptors antagonist, DAMGO: µ-opioid receptors agonist. There are significant differences between groups with different superscripts in a column (a, b and c; P < 0.05)
Effect of ICV injection of SR141716A (6.25 µg), DPDPE (40 pmol) and their combination on cumulative food intake (% BW) in neonatal chickens. SR141716A: selective CB1 receptors antagonist, DPDPE (δ-opioid receptors agonist). There are significant differences between groups with different superscripts in a column (a and b; P < 0.05)
Effect of ICV injection of AM630 (1.25 µg), DPDPE (40 pmol) and their combination on cumulative food intake (% BW) in neonatal chickens. AM630: selective CB2 receptors antagonist, DPDPE (δ-opioid receptors agonist). There are significant differences between groups with different superscripts in a column (a and b; P < 0.05)
Effect of ICV injection of SR141716A (6.25 µg), U-50488H (30 nmol) and their combination on cumulative food intake (% BW) in neonatal chickens. SR141716A: selective CB1 receptors antagonist, U-50488H (κ-opioid receptors agonist). There are significant differences between groups with different superscripts in a column (a and b; P < 0.05)
Effect of ICV injection of AM630 (1.25 µg), U-50488H (30 nmol) and their combination on cumulative food intake (% BW) in neonatal chickens. AM630: selective CB2 receptors antagonist, U-50488H (κ-opioid receptors agonist). There are significant differences between groups with different superscripts in a column (a and b; P < 0.05)
Effect of ICV injection of 2-AG (2 µg), Naloxone (5 µg) and their combination on cumulative food intake (% BW) in neonatal chickens. 2-AG: CB1 receptors agonist, Naloxone: non-selective opioid receptors antagonist. There are significant differences between groups with different superscripts in a column (a, b and c; P < 0.05)
Effect of ICV injection of CB65 (1.25 µg), Naloxone (5 µg) and their combination on cumulative food intake (% BW) in neonatal chickens. CB65: CB2 receptors agonist, Naloxone: non-selective opioid receptors antagonist. There are significant differences between groups with different superscripts in a column (a, b and c; P < 0.05)
As seen, ICV injection of DAMGO (µ-opioid receptors agonist, 125 pmol) significantly decreased food intake compared to control group until 120 min post injection [F(l,34) = 157.01, P < 0.001]. The dose of DAMGO was selected based on our pilot study, because it was found to induce a decrease in food intake without affecting other non-ingestive behavioral parameters such as sedation. Also, co-administration of SR141716A and DAMGO amplified hypophagic effect of DAMGO in chickens [F(l,34) = 190.72, P < 0.001] (Fig. 1).
Effect of ICV injection of AM630 (1.25 µg, selective CB2 receptors antagonist) and DAMGO on cumulative food intake (% BW) in FD3 neonatal layer type chickens is presented in Fig. 2. According to the data, there was no significant effect on food intake after ICV injection of CB2 receptors antagonist (1.25 µg) [F(l,34) = 4.701, P > 0.05]. Furthermore, ICV injection of 125 pmol of DAMGO had hypophagic effect compared to control group (P < 0.001). Co-administration of AM630 and DAMGO amplified hypophagic effect of DAMGO in neonatal layer type chickens compared to control group [F(l,34) = 140.02, P < 0.001].
According to the results of experiment 3, DPDPE (40 pmol) significantly increased food intake in chickens compared to control group [F(l,34) = 181.47, P < 0.001] and hyperphagic effect of DPDPE (40 pmol) was not significantly attenuated by co-injection of DPDPE and selective CB1 receptors antagonist) [F(l,34) = 1.508, P > 0.05] (Fig. 3). Likewise, hyperphagic effect of DPDPE (40 pmol) significantly blocked by co-administration of DPDPE + AM630 (selective CB2 receptors antagonist) [F(l,34) = 195.02, P < 0.0001] (Fig. 4).
As seen in experiment 5, U-50488H (κ-opioidergic receptors agonist, 30 nmol) significantly increased food intake but hyperphagic effect of U-50488H did not change by co-administration of U-50488H + SR141716A (CB1 receptors antagonist) compared to control group in birds [F(l,34) = 6.739, P > 0.05] (Fig. 5).
According to the obtained data from experiment 6, the significant reinforced effect of U-50488H on food intake was similar to co-injection of U-50488H and AM630 (selective CB2 receptors antagonist) [F(l,34) = 4.006, P > 0.05] (Fig. 6).
In this study, food intake significantly increased via CB1 receptors agonist (2-AG, 2 µg) compared to control group [F(l,34) = 143.18, P < 0.001]. Also, Naloxone (non-selective opioid antagonist, 5 µg) had no effect on food intake while co-administration of 2-AG + Naloxone sginificantly amplified heperphagic effect of CB1 receptors agonist [F(l,34) = 157.013, P < 0.001] (Fig. 7).
In experiment 8, administration of CB65 (CB2 receptors agonist, 1.25 µg) improved food intake compared to control group in chicken [F(l,34) = 131.64, P < 0.001] and its effect augumented via co-administration of CB65 + Naloxone [F(l,34) = 124.84, P < 0.001] (Fig. 8).
Discussion
The present survey provides support for physiologic relevant interactions between CBergic and opioidergic systems on food intake in FD3 neonatal layer-type chickens. Despite psychoactive constituent of cannabis sativa (marijuana), Δ9-THC for the first time isolated in 1964 and specific CB receptors identified in 1980 but role of CBergic system is not fully elicited. To date several researches have done on role of CBergic system. Now evidences imply that endocannabinoids include in several behavior and emotional states, such as food intake, pain, anxiety and go fourth (López 2010). In the CNS, endocannabinoids produced from arachidonic acid on the cell membranes. CB1 receptor is identified in the terminals of central, peripheral neurons and glial cells; however they are expressed in the peripheral tissue (D’Addario et al. 2014). It is reported CBergic system modulates food intake in neonatal broilers (Emadi et al. 2011). ICV injection of low to mid-dose of SR141716A (selective CB1 receptors antagonist) or AM251 (CB1 cannabinoid receptors inverse agonist) has anorectic effect in rat (Chen et al. 2006). In this study, we used sub-effective dose of antagonists which blocks receptor without effect on food intake to assay possible interaction(s) of CBergic system on opioidergic-induced food intake.
µ-opioid receptors impress orexigenic effect on feeding behavior through nucleus accumbens (NAc) and NTS in rodents (Browning et al. 2006; Fichna et al. 2007; Zheng et al. 2007). Central appetite regulatory mechanisms in chicken are somewhat dissimilar than mammals. Controversial results exist about the effect of µ-opioid receptors on food intake in chickens. It is reported DAMGO inhibits feeding behavior in neonatal broiler (Bungo et al. 2004, 2005). In comparison, central µ-opioid receptors have orexigenic role in broilers (Khan et al. 2009). Our results revealed μ-opioid receptors have anorexigenic effect in layer-type chicken. Discrepancy between layer and broiler may be ascribed to genetic selection (Bungo et al. 2004; Zendehdel et al. 2014b) where genetic selection for growth in broilers altered their brain appetite regulation centers and/or pathways (Denbow 1994).
This study provides the first demonstration of hypophagia induced by central administration of μ-opioid receptors agonist mediates by selective CB1 and CB2 receptors antagonists in chicks (Fig. 1). CB1 receptors in high density are expressed on presynaptic nerve terminals of both inhibitory and excitatory nerves, based on the brain region (Sharkey et al. 2014). CB1 densities are identified in the amygdala and hypothalamus. CB2 predominantly found in the peripheral tissues like spleen and leukocytes (López 2010). Interestingly, CB2 is also present in the brain (D’Addario et al. 2014). Cannabinoid agonists may activate a comparative greater number of G-proteins per occupied receptor in these brain regions (López 2010).
Numerous hypotheses have been postulated to explain the mechanisms underlying the interaction between CBergic and μ-opioid receptors. Anatomical evidence suggesting both receptors involved in feeding regulation centers in the brain (Chen et al. 2006). In keeping with these observations, orexigenic mediator, NPY, increase via CB1 receptors. CBergic system interacts by several anorexigenic and orexigenic neurotransmitters in the hypothalamus in regulation of energy expenditure and food intake (D’Addario et al. 2014). Both types of receptors are coupled to adenylyl cyclase activity, decrease in cAMP production through activation of Gi proteins (Chen et al. 2006). Postsynaptic depolarization increase endocannabinoid synthesis binds to CB receptors on the presynaptic membrane and activates G protein, inhibits Ca2+ influx and terminates to decrease neurotransmitter release (López 2010). CB1 and μ-opioid receptor interacts in the NAcc through a mechanism involving Gαinhibitory3 protein (Gαi3) (Yao et al. 2006). It is reported food intake increases via μ-opioid receptor in rat and co-injection of naloxone (μ-opioid receptor antagonist) and selective CB1 receptor antagonist suppressed food intake (Kirkham and Williams 2001). Perhaps, a systemic cannabinoid–opioid interaction may exist on appetite regulation (Chen et al. 2006). There are differences in the activities of neurotransmitters such as ghrelin (Zendehdel and Hassanpour, 2014), leptin and adiponectin (Novoseletsky et al. 2011) on feeding behavior regulation between mammals and avian. Suggested mechanism for rewarding effect of CBergic system on µ-receptor-induced feeding is that CBergic system mediates endogenous opioid systems via dopaminergic system (Braida et al. 2001; Solinas and Goldberg, 2005) where mesolimbic incentive dopaminergic pathways are modulated by endocannabinoids and opioidergic reward circuits. So, these actions underlie the orexigenic potency of CBs (Bodnar 2012). We think, further researches needed to identify accuracy of this hypothesis in birds.
In this study, DPDPE (δ-opioid receptors agonist) significantly reinforced cumulative food intake. It is reported that following 12-h food deprivation, a peripheral injection of AM630 (selective CB2 receptors antagonist) increased food intake in rat (Onaivi et al. 2008). Perhaps, CB2 receptors act on the immune system and indirectly regulate feeding behavior by alter in the digestive system function (Emadi et al. 2011). As observed, co-administration of δ-opioid receptor agonist and CB1 receptors antagonist was not able to diminish hyperphagic effect of DPDPE while this effect minimized via CB2 receptors antagonist. δ-opioid receptor agonist elevates food intake in neonatal meat-type chicken (Bungo et al. 2004; Yanagita et al. 2008; Khan et al. 2009) and rats (Levine 2006; Kaneko et al. 2012). In normal physiological conditions, central prostaglandins D2 (PGD2) and NPY system play deniable role in appetite and this interaction is via opioidergic system. Central injection of naltrindole (δ-opioid receptors agonist) activates cyclooxygenase-2 (COX-2) which terminates to produce PGD2, act on its specific receptor, DP1 (GPCR subtype) and stimulates NPY level via DP1-possessing NPY neurons in the CNS. Presumably, δ-opioid-induced orexigenic activity is mediated via PGD2–NPY system (Kaneko et al. 2012). Hypothalamic endocannabinoid is necessary for orexigenic effect of NPY (Di Marzo et al. 2001). However, less information exist on interaction of CBergic system on δ-opioid-induced food intake. Stimulation of CB2 receptors suppresses microglial activation. Perhaps this effect mediates through microglia. There is no evidence for expression or function of CB2 receptors in the microglia and neurons in chicken (Emadi et al. 2011). In a sole study, Fowler et al. (2001) reported CB2-like protein exists in CNS of neonatal chick and it is disappear in adult chicken. We think much more research needs to clarify role of CB2 receptors on feeding behavior in layer-type chicken. As seen, hyperphagic effect of U-50488H (κ-opioid receptors agonist) was not attenuted via both CB1 or CB2 receptor antagonists. It is reported that central injection of U-50488H elicits hyperphagia in broiler chickens (Bungo et al. 2004). Presumably, κ-opioid induced food intake is not mediates via CBergic system in layer-type chicken.
Of particular interest of present study was that co-administration of CB1 or CB2 agonist with naloxone significantly amplified hyperphagic effect of CB1 receptor agonist. It is reported CBergic-induced food intake is attenuated by naloxone in rat where co-injection of naloxone and CB1 receptors antagonist synergistically depress food intake in rat (Williams and Kirkham 2002). On the other hand, central injection of naloxone (10 µg) elevated food intake in neonatal layer-type chicken (Steinman, et al. 1987). Interestingly, peripheral but not central administration of naloxone attenuated food intake in domestic fowl (McCormack and Denbow 1990). Naloxone attenuated food consumption through production of malaise or impairment of motor function where high levels of naloxone lead to overt symptoms of illness, discomfort or motor problems (McCormack and Denbow 1990). In the present study, we used sub-effective dose of naloxone (5 µg) (Steinman et al. 1987) which blocked the opioid receptors without effect on food intake to investigate opioid receptors interconnection with CBergic receptors (Soria-Gomez et al. 2007). Naloxone precipitates opioid-like withdrawal syndrome in CB-dependent mice (Ghozland et al. 2002). Opioid and CB receptors in the NAcc interact with each other to modulate feeding (Skelly et al. 2010). Therefore, presumably neurotransmitter release not only modulates through CBergic system but it may also influence the expression of feeding related neuropeptides (Emadi et al. 2011). Based on literature, food intake diminished via µ-opioid receptors in chicken which is dissimilar to rodents (Bungo et al. 2004). In this study, food intake increased with co-administration of naloxone and CB1 and CB2 receptors agonists in birds. To our knowledge we think blockade of opioidergic receptors might amplify hyperphagic effect of CBergic system in chicks. Most research on central food intake regulation has done in rat models whereas considering few investigations done in birds. Actually, there was no similar research to compare our results on mediatory effect of naloxone on cannabinoid-induced food intake as poultry model. It is reported, central role of opioidergic system is somewhat different in avian and mammalian (Zendehdel and Hassanpour, 2014). These controversial data might be the consequence of injection methods and species difference (Meade and Denbow 2001). To conclude new finding of current study can be used as base information on regulatory role of CBs on effect of opioidergic system on food intake in neonatal chicken. Further research required to clarify any direct interaction of cellular and molecular signaling pathways in the interconnection between CBergic and opioidergic systems on feeding behavior in avian.
References
Barnes MJ, Holmes G, Primeaux SD, York DA, Bray GA (2006) Increased expression of mu opioid receptors in animals susceptible to diet-induced obesity. Peptides 27:3292–3298
Bodnar RJ (2012) Endogenous opiates and behavior: 2011. Peptides 38:463–522
Braida D, Pozzi M, Parolaro D, Sala M (2001) Intracerebral self-administration of the cannabinoid receptor agonist CP 55,940 in the rat: interaction with the opioid system. Eur J Pharmacol 413:227–234
Browning KN, Zheng Z, Gettys TW, Travagli RA (2006) Vagal afferent control of opioidergic effects in rat brainstem circuits. J Physiol 575:761–776
Bungo T, Kawamura K, Izumi T, Dodo K, Ueda H (2004) Feeding responses to µ-, δ- and κ-opioid receptor agonists in the meat-type chick. Pharmacol Biochem Behav 78:707–710
Bungo T, Kawamura K, Izumi T, Dodo K, Ueda H (2005) Effects of various µ-, δ- and κ-opioid ligands on food intake in the meat-type chick. Physiol Behav 85:519–523
Chen RZ, Frassetto A, Fong TM (2006) Effects of the CB1 cannabinoid receptor inverse agonist AM251 on food intake and body weight in mice lacking μ-opioid receptors. Brain Res 1108:176–178
D’Addario C, Micioni Di Bonaventura MV, Puccia M, Romano A, Gaetani S, Ciccocioppo R, Cifani C, Maccarrone M (2014) Endocannabinoid signaling and food addiction. Neurosci Biobehav Rev 47:203–224
Davis JL, Masuoka DT, Gerbrandt LK, Cherkin A (1979) Autoradiographic distribution of l-proline in chicks after intracerebral injection. Physiol Behav 22:693–695
Denbow DM (1994) Peripheral regulation of food intake in poultry. J Nutr 124:1349S–1354S
Di Marzo VGS, Wang L, Liu J, Batkai S, Jarai Z, Fezza F, Miura GI, Palmiter RD, Sugiura T, Kunos G (2001) Leptin regulated endocannabinoids are involved in maintaining food intake. Nature 410:822–825
Dodo K, Izumi TH, Ueda H, Bungo T (2005) Response of neuropeptide Y-induced feeding to µ-, δ- and κ-opioid receptor antagonists in the neonatal chick. Neurosci Lett 373:85–88
Emadi L, Jonaidi H, Hosseini Amir Abad E (2011) The role of central CB2 cannabinoid receptors on food intake in neonatal chicks. J Comp Physiol A 197:1143–1147
Fichna J, Janecka A, Costentin J, Do Rego J (2007) The endomorphin system and its evolving neurophysiological role. Pharmacol Rev 59:88–123
Fowler CJ, Nilsson O, Andersson M, Disney G, Jacobsson SO, Tiger G (2001) Pharmacological properties of cannabinoid receptors in the avian brain: similarity of rat and chicken cannabinoid1 receptor recognition sites and expression of cannabinoid2 receptor-like immunoreactivity in the embryonic chick brain. Pharmacol Toxicol 88:213–222
Furuse M, Matsumoto M, Saito N, Sugahara K, Hasegawa S (1997) The central corticotropin-releasing factor and glucagon-like peptide-1 in food intake of the neonatal chick. Eur J Pharmacol 339:211–214
Ghozland S, Matthes HW, Simonin F, Filliol D, Kieffer BL, Maldonado R (2002) Motivational effects of cannabinoids are mediated by μ-opioid and 6-opioid receptors. J Neurosci 22(3):1146–1154
Ibegbu AO, Mullaney I, Fyfe L, MacBean D (2011) The roles of opioid receptors and agonists in health and disease conditions. Br J Pharmacol Toxicol 2(2):84–91
Irwin N, Hunter K, Frizzell N, Flatt PR (2008) Antidiabetic effects of sub-chronic administration of the cannabinoid receptor (CB1) antagonist, AM251, in obese diabetic (ob/ob) mice. Eur J Pharmacol 581:226–233
Kaneko K, Yoshikawa M, Ohinata K (2012) Novel orexigenic pathway prostaglandin D2-NPY system-involvement in orally active orexigenic δ opioid peptide. Neuropeptides 46:353–357
Khan MSI, Ohkubo T, Masuda N, Tachibana T, Ueda H (2009) Central administration of metastin increases food intake through opioid neurons in chicks. Comp Biochem Physiol Part A 153:209–212
Kirkham TC, Williams CM (2001) Synergistic effects of opioid and cannabinoid antagonists on food intake. Psychopharmacol 153:267–270
Kozlov AP, Nizhnikov M, Kramskaya TA, Varlinskaya EI, Spear NE (2013) Mu-opioid blockade reduces ethanol effects on intake and behavior of the infant rat during short-term but not long-term social isolation. Pharmacol Biochem Behav 103:773–782
Le Merrer J, Becker JAJ, Befort K, Kieffer BL (2009) Reward processing by the opioid system in the brain. Physiol Rev 89:1379–1412
Levine AS (2006) The animal model in food intake regulation: examples from the opioid literature. Physiol Behav 89:92–96
López HH (2010) Cannabinoid–hormone interactions in the regulation of motivational processes. Hormones Behav 58:100–110
Manzanares J, Ortiz S, Oliva JM, Perez-Rial S, Palomo T (2005) Interactions between cannabinoid and opioid receptor systems in the mediation of ethanol effects. Alcohol Alcohol 40(1):25–34
McCormack JF, Denbow DM (1990) Central versus peripheral opioid regulation of ingestive behavior in the domestic fowl. Pharmacol Biochem Behav 96(1):211–216
Meade S, Denbow M (2001) Feeding, drinking, and temperature response of chickens to intracerebroventricular histamine. Physiol Behav 73:65–73
Novoseletsky N, Nussinovitch A, Friedman-Einat M (2011) Attenuation of food intake in chicks by an inverse agonist of cannabinoid receptor 1 administered by either injection or ingestion in hydrocolloid carriers. Gen Comp Endocrinol 170:522–527
Olanrewaju HA, Thaxton JP, Dozier WA, Purswell J, Roush WB, Branton SL (2006) A review of lighting programs for broiler production. Int J poultry Sci 5(4):301–308
Onaivi ES, Carpio O, Ishiguro H, Schanz N, Uhl GR, Benno R (2008) Behavioral effects of CB2 cannabinoid receptor activation and its influence on food and alcohol consumption. Ann N Y Acad Sci 1139:426–433
Onaivi ES, Ishiguro H, Gu S, Liu QR (2012) CNS effects of CB2 cannabinoid receptors: beyond neuro-immuno-cannabinoid activity. J Psychopharmacol 26:92–103
Pickel V, Chan J, Kash T, Rodriguez J, MacKie K (2004) Compartment-specific localization of cannabinoid 1 (CB1) and μ-opioid receptors in rat nucleus accumbens. Neuroscience 127(1):101–112
Pritchett CE, Pardee AL, McGuirk SR, Will MJ (2010) The role of nucleus accumbens adenosine–opioid interaction in mediating palatable food intake. Brain Res 1306:85–92
Saito ES, Kaiya H, Tachibana T, Tomonaga S, Denbow DM, Kangawa K, Furuse M (2005) Inhibitory effect of ghrelin on food intake is mediated by the corticotropin-releasing factor system in neonatal chicks. Regul Pept 125:201–208
Sharkey KA, Darmani NA, Parker LA (2014) Regulation of nausea and vomiting by cannabinoids and the endocannabinoid system. Eur J Pharmacol 722:134–146
Skelly MJ, Guy EG, Howlett AC, Pratt WE (2010) CB1 receptors modulate the intake of a sweetened-fat diet in response to mu-opioid receptor stimulation of the nucleus accumbens. Pharmacol Biochem Behav 97:144–151
Solinas M, Goldberg SR (2005) Motivational effects of cannabinoids and opioids on food reinforcement depend on simultaneous activation of cannabinoid and opioid systems. Neuropsychopharmacol 30:2035–2045
Soria-Gomez E, Matias I, Rueda-Orozco PE, Cisneros M, Petrosino S, Navarro L, Di Marzo V, Prospero-Garcia O (2007) Pharmacological enhancement of the endocannabinoid system in the nucleus accumbens shell stimulates food intake and increases c-Fos expression in the hypothalamus. Br J Pharmacol 151:1109–1116
Taha SA (2010) Preference or fat? Revisiting opioid effects on food intake. Physiol Behav 100:429–437
Tambaro S, Tomasi ML, Bortolato M (2013) Long-term CB1 receptor blockade enhances vulnerability to anxiogenic-like effects of cannabinoids. Neuropharmacol 70:268–277
Van Tienhoven A, Juhasz LP (1962) The chicken telencephalon, diencephalon and mesencephalon in sterotaxic coordinates. J Comp Neurol 118:185–197
Wiley JL, Marusich JA, Zhang Y, Fulp A, Maitra R, Thomas BF, Mahadevan A (2012) Structural analogs of pyrazole and sulfonamide cannabinoids: effects on acute food intake in mice. Eur J Pharmacol 695:62–70
Williams CM, Kirkham TC (2002) Reversal of ∆9-THC hyperphagia by SR141716 and naloxone but not dexfenfluramine. Pharmacol Biochem Behav 71:333–340
Steinman JL, Fujikawa DG, Wasterlain CG, Cherkin A, Morley JE (1987) The effects of adrenergic, opioid and pancreatic polypeptidergic compounds on feeding and other behaviors in neonatal leghorn chicks. Peptides 8:585–592
Yanagita K, Shiraishi J, Fujita M, Bungo T (2008) Effects of N-terminal fragments of β-endorphin on feeding in chicks. Neurosci Lett 442:140–142
Yao L, McFarland K, Fan P, Jiang Z, Ueda T, Diamond I (2006) Adenosine A2a blockade prevents synergy between mu-opiate and cannabinoid CB1 receptors and eliminates heroin-seeking behavior in addicted rats. Proc Natl Acad Sci 103:7877–7882
Zendehdel M, Hassanpour S (2014) Ghrelin-induced hypophagia is mediated by the β2 adrenergic receptor in chicken. J Physiol Sci 64:383–391
Zendehdel M, Mokhtarpouriani K, Babapour V, Baghbanzadeh A, Pourrahimi M, Hassanpour S (2013a) The effect of serotonergic system on nociceptin/orphanin FQ induced food intake in chicken. J Physiol Sci 63:271–277
Zendehdel M, Mokhtarpouriani K, Hamidi F, Montazeri R (2013b) Intracerebroventricular injection of ghrelin produces hypophagia through central serotonergic mechanisms in chicken. Vet Res Commun 37(1):37–41
Zendehdel M, Hamidi F, Hassanpour S (2014a) The effect of histaminergic system on nociceptin/orphanin FQ induced food intake in chicken. Int J Pept Res Ther. doi:10.1007/s10989-014-9445-5
Zendehdel M, Hasani K, Babapour V, Seyedali Mortezaei S, Khoshbakht Y, Hassanpour S (2014b) Dopamine-induced hypophagia is mediated by D1 and 5HT-2c receptors in chicken. Vet Res Commun 38:11–19
Zheng H, Patterson LM, Berthoud HR (2007) Orexin signaling in the ventral tegmental area is required for high-fat appetite induced by opioid stimulation of the nucleus accumbens. J Neurosci 27:11075–11082
Conflict of interest
Morteza Zendehdel, Shahin Hassanpour, Vahab Babapour, Saeed Charkhkar and Mahshid Mahdavi declare that they have no conflict of interest.
Human and Animal Rights
All experiments executed in according to the Guide for the Care and Use of Laboratory Animals and approved by the institutional animal ethical committee.
Informed Consent
This manuscript does not contain any studies with human subjects performed by any of authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zendehdel, M., Hassanpour, S., Babapour, V. et al. Interaction Between Endocannabinoid and Opioidergic Systems Regulates Food Intake in Neonatal Chicken. Int J Pept Res Ther 21, 289–297 (2015). https://doi.org/10.1007/s10989-015-9457-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-015-9457-9