Abstract
A slit-lamp examination is an indispensable and essential clinical evaluation method in ophthalmology, but, it is qualitative subjective. To complement its weaknesses in making a quantitative evaluation of flare intensity and number of cells in the aqueous humor in the eye, we invented the laser flare-cell photometer in 1988. The instrument enables a non-invasive quantitative evaluation of flare intensity and number of cells in the aqueous with good accuracy and repeatability as well as maneuverability equal to slit-lamp microscopy. The instrument can elucidate the pathophysiology in the blood-aqueous barrier (BAB) function in a variety of ocular disorders. The accuracy of the instrument makes it possible to investigate not only the pathophysiology of intraocular disorders but also the effects of various drugs and surgical procedures in BAB. The instrument does not only lighten the burden on patients in clinical examinations and study but it also helps minimize the sacrifice of experimental animals and improves the reliability of the results by minimizing inter-individual variations through its good repeatability. Here I shall relate how the instrument has been applied to clinical and basic studies in ophthalmology and what novel knowledge its application contributed to pathophysiology in ophthalmology.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A slit-lamp microscope and a funduscope are indispensable to an ophthalmological examination. These are non-invasive methods that enable us to detect various conditions in the eye. Based on the principle of visual function defined by Hering comprising light perception, colour sensation and visual acuity according to a definition of minimum spatial resolution, expert ophthalmologists can evaluate and assess most ocular disorders by using these instruments and a tonometer following an examination of visual acuity. However, slit-lamp microscopy and funduscopy are both subjective and qualitative. Therefore, there are inter-individual variations in assessment results. Various ophthalmic instruments have been invented to complement slit-lamp microscopy and funduscopy.
Ophthalmologists use slit-lamp microscopy to evaluate not only anatomical features but also pathophysiological findings such as inflammatory processes in the anterior segment of the eye. The blood-ocular barrier is composed of a blood-aqueous barrier (BAB) and a blood-retinal barrier. The BAB is located at the non-pigmented epithelium of the ciliary body and the capillary wall of the iris and pathophysiological processes in the eye cause disruptions to its barrier functions. When an inflammation occurs in the anterior segment of the eye, it causes a deterioration of the BAB function, resulting in leakage of serum protein and blood cells into the aqueous humor. The level of deterioration of the BAB function depends on the severity of the inflammatory activity in the anterior segment of the eye, the more intense the inflammatory reaction, the larger the increase of proteins and blood cells in the aqueous. The increase in proteins and blood cells in the aqueous can be assessed by measuring the increase of flare and density of cells by slit-lamp microscopy. Semi-quantitative grading classification of flare and cells using slit-lamp microscopy is reported and clinically used. In this method, the grading scale for flare and cells is step-wise and primarily subjective. To discriminate minute differences in flare intensity is difficult, yet with slit-lamp microscopy assessment of absence or presence of cells in the aqueous is relatively easy. Fluorophotometry was invented as a complementary method to flare assessment by slit-lamp microscopy, and is used as a clinical tool. Fluorophotometry can be applied to evaluate not only physiological parameters such as aqueous humor dynamics and permeability of the corneal endothelial cell layer but also any deterioration in the BAB functions and corneal endothelial permeability. However, the molecular weight of sodium fluorescein is far smaller than that of serum protein composed of albumin and globulin. Therefore, the results obtained by fluorophotometry are an index but not the equivalent of flare intensity measured by slit-lamp microscopy. Furthermore, fluorophotometry cannot evaluate cells in the aqueous. A further drawback is, that in spite of the various possible administrative methods: intravenous injection, oral uptake and topical electrophoresis; it is unable to continuously measure the fluorescein up to its disappearance as well as potential adverse events due to the administration of fluorescein.
A number of studies tried to develop an objective and quantitative method. They achieved good results in vitro or under limited experimental conditions, but could not be applied clinically. To become clinically applicable method, several important factors must be considered: a minimally or non-invasive method like slit-lamp microscopy, good repeatability and accuracy with adequate maneuverability and bio-compatibility including a tolerable short examination time.
The principle of flare and cell observation by slit-lamp microscopy is the light scattering phenomena due to proteins and cells in the aqueous. With the support of grants-in-aid for Scientific Research from the Ministry of Education and Science of Japan (renamed as The Ministry of Education, Culture, Sports, Science and Technology), my colleagues and I invented a method to determine the flare intensity in the aqueous in vivo and published our preliminary report in Japanese in 1987 [1]. Then we introduced the laser flare-cell photometer (flare-cell photometer) in 1988 which enables an objective and quantitative determination of flare intensity and number of cells in the aqueous on the basis of the same principle as slit-lamp microscopy [2]. This article reviews the principle of the laser flare-cell photometer and its significance in clinical and experimental studies.
Development of laser flare-cell photometer and related studies
Principle and set-up of the laser flare-cell photometer
Principle and set-up
Any clinical examination method has to have a clear purpose and will necessarily have limitations resulting from its operating principles. Obtained data should be treated carefully according to the guiding principles and specific resolution of the instruments. All such instruments should be equipped with a calibration method. Based on the original laser flare-cell photometer (FC-1000) (Fig. 1a), several modified types have been developed (Fig. 1b). The underlying principles of these instruments are the same as those of the original one. The following description of the instrument is cited from the original paper (1988) [2].
Setup of laser flare-cell photometer (FC-1000 and FM-500). FC-1000. a The instrument is comprised of an optical unit, power unit and computer unit. b FM-500. Three units of FC-1000 are integrated into a conventional slit-lamp microscope. The laser system and photomultiplier system are within the slit-lamp microscope and the computer system is mounted in the table plate
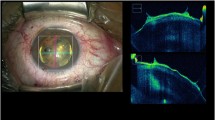
The principle of flare and cell detection by slit-lamp is the light scattering phenomena in the aqueous, which can be determined by Debye’s equation. It indicates the relationship between the intensity of scattered light and incident light. According to this equation, both intensities of scattered light and incident light can be determined simply by the concentration of solvent and its molecular weight in the aqueous solution, provided the various parameters such as the angle between the axis of incident light and axis of detector of scattered light, the wave length and the intensity of the incident light are prefixed. Thus, the instrument is comprised of a He–Ne laser beam as the incident light and digital photon counting photomultiplier as the detector of intensity of the scattered light, both of which are mounted on a slit-lamp microscope, set at an angle of 90° and aligned in the anterior chamber (Fig. 2a). The maximum power of the He–Ne laser is 50 µW, within the safety limits for ocular tissues and the diameter of the focused beam is 20 µm measured in the air. The intensity of the laser beam is monitored by an internally equipped system and its fluctuation is fed back to detected signals. The sampling window size of the photomultiplier is 0.3 × 0.5 mm in the air and positioned in the center of the laser beam (Fig. 2b). A detected scattered light signal which has fluctuation noise is analyzed by a newly developed computer program. The power of the laser beam and alignment of sampling window can be calibrated using a separately developed device. Total examination time of laser flare-cell photometry is programed to last 1 s.
Block diagram and montage image of the sampling window in the anterior chamber [2]. a Block diagram: Laser beam system is irradiated through a Galvano mirror into the anterior chamber as shown in montage image c. The laser power is 25 μW and the focused beam diameter is 20 µm in the air. Scattered light in the aqueous is detected by the digital photomultiplier. The angle between the laser beam and axis of the photomultiplier is set at 90°. The system control and analysis of detected scattered light intensity is operated by a computer program. b Montage image of laser beam and sampling window in the anterior chamber of the eye
Flare measurement
Flare is derived from the serum protein which components’ differ depending on the break-down of the BAB function. To minimize signal contamination by background laser scattering due to reflection from the iris, crystalline lens, intraocular lens or fundus, we programed the laser beam to scan vertically, for a length of 0.6 mm, covering the sampling window (Fig. 3). A single photon sampling time is 1 ms and 10 consecutive samplings are treated by the method of a running mean. The unit of measurement is photon count/ms (PC/ms).
Laser beam scan for flare measurements [2]. a Schematic drawing of optical scanner and sampling window. Laser beam is scanned from the bottom upward vertically for the length of 0.6 mm and 0.5 s. b Schematic drawing of component scattered light. S’o, S’’o: out of sampling window = background light scatter. Si: light scatter in sampling window = flare + background
Signals detected by the photomultiplier are divided into three as shown in Fig. 3. Si denotes the scattered light intensity recorded when the laser beam passes in the sampling window, and S’o and S’’o denote scattered light intensity when the beam passes above and below the sampling window. Si is theoretically composed of both scattered laser light intensity from protein in the aqueous, which is denoted as Sp, and background laser scattering, either S’o or S’’o. The background laser scattering is subtracted by a computer program from Si, using the following equation:
When the values for S’o and S’’o are markedly different, the sampling window is not positioned in the background with a homogeneous light scattering condition. When the difference between the values for S’o and S’’o exceeds 20% of that for Si, the measurement datum is abandoned and a repeated measurement will be performed. Generally, more than 5 separate measurements in a session are recommended. The measurement data in each session is automatically averaged. Inadequate data can be deleted from the averaged results.
Cell measurement
The laser beam scans two dimensionally, 0.6 × 0.25 mm, in a sampling window using simultaneously operating Galvano mirrors for 0.5 s, making an optical cube: 0.6 × 0.25 × 0.5 = 0.075 mm3/0.5 s which forms the cell measurement unit (Fig. 4). Based on an in vitro experiment using a solution containing latex particles, we determined the optimized peak that corresponds to cells in the aqueous solution. Whenever a peak in a cell exceeded 4 photon counts per 400 µs we considered it as a peak and counted the number of peaks using a computer program.
Laser beam scan for cell measurement [2]. Two dimensional beam scan using simultaneously operated Galvano mirror: 0.25 × 0.6 mm. Optically produced volume: 0.25 × 0.6 × 0.5 = 0.075 mm3/0.5 s
Validation of flare and cell measurement
Flare measurement
Figure 5 shows the results the correlation between flare measurements (Photon counts: PC/ms) and concentration of bovine serum albumin solution. There is a significant correlation between both and their dynamic range is large when compared using the Lowry method. Based on the regression curve between flare value (PC/ms) and bovine serum albumin, flare values can be converted to albumin equivalent concentrations.
Plasma samples taken from two normal subjects (Samples A and B) were diluted with distilled water from 1/50 to 1/104 times. Bovine serum albumin (Sigma) solutions were prepared using distilled water, ranging from 1.0 mg/100 mL to 1000 mg/100 mL [2]. Test sample solution in a disposable plastic cuvette (Evergreen) was measured using the flare measurement mode. The results are shown in Fig. 6. The correlation between values for photon counts and concentrations of human plasma or bovine serum albumin show a significant linear function with a wide range. Similar results are also reported, e.g., albumin equivalent concentration converted from the correlation between albumin concentration and flare values in vitro was higher than true aqueous protein concentration in severe inflammation in the anterior chamber of the eye [3–6].
Correlation between human plasma solution and bovine serum albumin solution [2]. A and B indicate human serum samples and the thick solid line indicates bovine serum albumin. The protein concentration is the same in both plasma samples (7.7 g/100 mL), but the percentiles of albumin/globulin in sample A was 63.2/36.8% and in B 68.0/32.0%. Molecular weight of globulin is larger than that of albumin, thus the regression line for sample A is higher than that for sample B and both regression lines are higher and steeper than the line for bovine serum albumin. Sample A: Y = 0.54 + 1.06X [r = 0.998, P = 0.00), Sample B: Y = 0.10 + 1.12X (r = 0.987, P = 0.00), bovine serum albumin: Y = 0.809X–0.54 (r = 0.989, P = 0.00)]. These phenomena agree with Debye’s rule. [Bar: mean ± S.D.]
Cell measurements
We chose two latex particles (average particle diameter; 2.02 and 2.95, The Dow Chemical Company, Hayward, CA, USA) which reflected the laser beam at a strength equal to the blood cells in our preliminary observations. Each latex particle was dissolved in bovine serum albumin solution with different concentrations in order to examine the effect of particle numbers in the albumin concentration and vice versa.
An albumin solution without latex particles in concentrations higher than 500 mg/100 mL emits peaked signals resembling those for particles. Therefore, the number of peaks found in such albumin solution without latex particles is subtracted from the number of peaks detected in an albumin solution of the same concentration with latex particles. Figure 7 shows the relationship between the number of peaks and the number of latex particles in the solution [2]. Figure 8 shows a similar study using human peripheral blood cells and lymphocytes.
a, b In vitro study using different latex particles in albumin solution to verify the cell count mode (Latex particle diameter; a 2.02 µm, b 2.95 µm) [2]. The average number of peaks correlates significantly with the pre-determined number of latex particles (r = 0.994, P < 0.001 for particle diameter 2.02 µm, r = 0.985, P < 0.003 for particle diameter 2.95 µm). However, as the number of latex particles in the solution decreased, the detected number of peaks tends to show larger variations. [Bar: mean ± S.D.]
In vitro study with human peripheral blood cells using the cell count mode [7]. Peripheral blood cells [●] and lymphocytes [○] were sampled and counted using a hemocytometer (Burker–Turk) and a microscope. Diluted solution was adjusted using bovine serum albumin solution. There is a significant correlation between number of cells determined by the cell count mode (cells/0.075 mm3) and predetermined cell densities in the diluted solution. [Bar: mean ± S.D.]
In vivo measurements
In our earlier studies to validate the reliability of the instrument, in vivo measurements were conducted as follows.
A first group comprised of 31 healthy volunteers, with a mean age of 23 ± 0.8 (±S.D.) years was assembled. They underwent topical administration of mydriatic eyedrops in one eye and no treatment in the fellow eye. A second group was comprised of 20 older subjects (mean age 70 ± 8.5 years) with age-related cataracts ranging from incipient to immature, but no other ocular disorders. They underwent measurements without mydriatic eyedrops [2].
In the healthy young adults, average photon counts (/ms) were 4.2 ± 1.0 in the eyes without mydriatics and 3.9 ± 1.0 in the eyes with mydriatics, but there was no significant difference. In the second group, average photon counts were 6.2 ± 2.5 which was significantly higher than in the eyes of the young adults without mydriatics (P < 0.005). Peak signals corresponding to cells were not detected in any eye.
Clinical validation study and novel pharmacological knowledge in cataract surgery
We used the new laser flare-cell photometer to investigate various pathophysiological findings in the eye [7, 8]. We evaluated the effects of topical indomethacin, a non-steroidal anti-inflammatory drug (NSAID), on postoperative inflammation in cataract surgery. The study was performed based on the results of a 1976 study that assessed the efficacy of topical indomethacin in preventing miosis through the inhibition of prostaglandins’ biosynthesis during soft cataract surgery. It was the first report of topical NSAIDs in ophthalmology [9]. In that study, conducted in 1977, we used slit-lamp microscopy, pachometry and infrared photography to reveal the efficacy of topical indomethacin in suppressing postoperative inflammation in intracapsular cataract extraction [10]. The results of the study regarding pre- and postoperative treatment with indomethacin demonstrated the suppressive effects of the NSAID on the postoperative breakdown of the BAB function, resulting in the suppression of an increase of both flare and cells in the aqueous. Furthermore, topical indomethacin could suppress the postoperative increase of central corneal thickness and miosis. Following this report, topical NSAID treatment either with or without topical steroids in cataract surgery has been prescribed as a routine regimen.
Taking into consideration that the above studies were performed using a slit-lamp microscope, we evaluated postoperative inflammation in cataract surgery using the laser flare-cell photometer with almost the same regimen, although the method of cataract surgery shifted from intracapsular cataract extraction alone to planned extracapsular cataract extraction (p-ECCE) with an implantation of posterior chamber intraocular lens (PC-IOL). The results gave us new information regarding the pharmacological effects of NSAIDs on the inflammatory biomechanism and pathophysiology in postoperative response [7]. The subjects underwent routine topical steroid treatment postoperatively and, depending on the regimen were divided into two groups; one group with and the second without topical indomethacin pre- and post-treatment. The subjects undergoing indomethacin treatment did not develop any fibrin reaction. The subjects without topical indomethacin were divided into two subgroups, one with and the other without fibrin reaction [11]. According to the laser flare-cell photometry, topical indomethacin can suppress postoperative increase of flare, but not the increase of cells (Fig. 9). These results do not agree with our previous results of 1977 [10]. This postoperative dissociation between flare and increase of cells can be explained by the inflammatory process relationship between arachidonic acid cascade and indomethacin [12, 13]. There are two major pathways of inflammatory chemical mediators; cyclooxygenase and lipoxygenase associated pathways. Cyclooxygenase plays a role in the biosynthesis of prostaglandins, thromboxane and others. Prostaglandins are active in the dilatation of vascular endothelium and cause leakage of serum protein into the aqueous humor. Lipoxygenase is involved in the biosynthesis of leukotrienes. NSAIDs can inhibit the cyclooxygenase pathway, but not the lipoxygenase pathway. Leukotriene B4 helps promote the production of inflammatory cells. The difference in effects of NSAIDs on lipoproteins in inflammatory response results in the differences in postoperative responses in flare and cells in cataract surgery.
The postoperative course in lens implant surgery with planned extracapsular cataract extraction (p-ECCE) [7]. a Flare, b Cell. According to the laser flare-cell photometry, a steep increase of flare and cells on the first postoperative day followed gradual decrease of flare and cells is observed in every case. The patients undergoing indomethacin treatment [■] show a significantly lower aqueous protein concentration compared with the patients undergoing routine postoperative topical steroid treatment [○: without postoperative fibrin reaction; ● with postoperative fibrin reaction]. However, in cases with fibrin reaction [●], once the decreased flare inverts to rise to develop fibrin in the aqueous which can be observed by slit-lamp microscopy. However, cases with fibrin reaction keep a relatively higher number of cells without marked fluctuations. Those different postoperative clinical courses suggest a different recovery process of the blood-aqueous barrier (BAB) function. [Bar: mean ± S.D.]. ■: Topical Indomethacin group. Control (Indomethacin (−) group ○: Fibrin (−) subgroup ●: Fibrin (+) subgroup
In an uneventful case, the breakdown of the BAB function due to surgical trauma recovers following postoperative regimen, but in cases with fibrin reaction, severe disruption of the BAB function disturbs the recovering process. There might be various reasons for this, such as host’s vulnerability, or IOL materials which are bio-incompatible. Recent development of sophisticated surgical instruments and manipulation including micro-incision techniques and anti-inflammatory drug regimen, make it possible to minimize postoperative inflammation and adverse events. The laser flare-cell photometer has contributed to the study of postoperative pathophysiology as described later.
Additional clinical studies using flare-cell photometer
We applied the instrument to clinical situations. It is reported that mydriatic eyedrops’ treatment decreases flare values significantly [14], although our preliminary study showed that mydriasis caused a tendency of lowering the eyedrops’ effect on flare [2]. It is also reported that flare and cell measurements can be applicable to Caucasian people regardless of iris colour, or fundus pigmentation [15]. Mydriasis had lowering effects on flare values but increased cell counts [15, 16]. The lowering effect of mydriasis on flare is considered to be the result of an enlargement in the volume of aqueous humor. It is believed that the increase in cells caused by mydriasis is not an inflammatory reaction but due to the dispersion of iris pigments. The flare values showed no difference between the right and left eyes in individuals, but increased with age as shown in Fig. 10 [14, 17]. Cells in the aqueous did not change with aging except for the 8th decade. On the other hand, the BAB function does deteriorate with age [4]. It is also revealed that flare values show diurnal variations and are in inverted pattern with intraocular pressure not only in humans, as shown in Fig. 11 [18], but also in rabbits. This phenomenon reflects the diurnal changes in aqueous humor production, because the volume of the anterior chamber is constant without change.
Change of aqueous protein concentration with age (Revised with permission from: [14]). Aqueous protein concentration increases with the increase in age. [Bar: mean ± S.D.]
Relationship between diurinal variations of intraocular pressure and aqueous protein concentration [18]. Flare value shows diurnal variations and inverted pattern with intraocular pressure. ●: Flare value ○: Intraocular pressure (P value: *<0.05, **<0.01, + <0.001). [Bar: mean ± S.D.]
Relationship between grading assessment using slit-lamp microscopy [19] and laser flare-cell photometry was studied in subjects with various types of uveitis [20, 21]. Flare assessment by slit-lamp microscopy is subjective and qualitative, thus the differences between the two methods can result in a methodological difference between an objective, quantitative evaluation and a subjective, qualitative one. In terms of cell assessment, slit-lamp microscopy is helpful in determining the absence or presence of cells in the aqueous, since both volume and duration of examination are arbitrary. On the other hand, both volume and duration of examination are fixed and limited in laser flare-cell photometry, thus, a result obtained by laser flare-cell photometry is quantitative with a limitation (0.075 mm3/0.5 s). Similar studies were performed later and confirm our results [22, 23].
Modification of the instrument
Following the original laser-flare cell photometer, 4 models were developed; model FC-2000 can evaluate both flare and cells with enlarged measurement volume from 0.075 mm3 to 0.5 mm3/0.5 s, whereas other models only evaluate flare measurements (FM-500, FM-600 and FM-700); except for FM-600, in which the set-up is similar to a conventional slit-lamp microscope and it can also function as a slit-lamp microscope (Fig. 12). Each model has the same performance power and specifications of flare measurement as the original model, FC-1000. Comparative clinical studies between FC-1000 and the other models were performed and equivalent efficacy was evaluated [6, 24, 25]. A new program was developed for the FC-2000 to exclude noise in cell measurements. Objective volume was divided into 128 strands, and the strands which were determined to have noise contamination were excluded from the final results [26]. Although it is relatively difficult to secure and align a large measurement volume in the anterior chamber of the FC-2000 compared with the FC-1000, it still works well.
Relationship between grading assessment using slit-lamp microscopy and laser flare-cell photometry in subjects with various types of uveitis [21]. a Flare, b Cells. Within the same grading score regarding flare assessment by slit-lamp microscopy, a large inter-individual variation is found and the variation is more pronounced in the groups with higher grading score. On the other hand, some cells are nondetectable by the laser flare-cell photometer even when cell grading is more than 1+ by slit-lamp microscopy
Comparative studies in flare values between laser-flare photometry and other methods
Many studies agree that there is a significantly good correlation between flare values measured by laser flare photometry and globulin/albumin concentration in eyes with endogenous uveitis [27–29] as well as aqueous albumin concentration in rabbit eyes [30]. A comparative study of the flare values with the results of a fluorophotometric study to assess the BAB function was also performed [28, 31]. Although both the flare-cell photometry and the fluorophotometry could effectively assess the BAB function in postsurgical inflammation in patients undergoing cataract surgery, in a clinical setting the flare-cell photometer was decidedly superior with distinct advantages.
Clinical studies using flare-cell photometers in various ophthalmological fields
Cataract surgery and related studies
In cataract surgery, the flare-cell photometer has been applied to investigate pathophysiology including effects of drug treatment and surgical manipulation. There is another study of bio-compatibility of intraocular lens (IOL) products and determination of limits of residual endotoxin concentrations in IOL. The results reveal that the flare-cell photometer contributed to the development of the sophisticated cataract surgery with IOL implantation practiced at present.
As described above, the effects of indomethacin pre-and post-operative treatment on pathophysiology in cataract surgery was a clinically important contribution, i.e., it plays a key role in the suppression of postoperative inflammation through the cyclooxygenase pathway in the inflammatory arachidonic acid cascade. This inflammatory pathophysiology in cataract surgery is mainly due to cyclooxygenase rather than lipoxygenase. If the surgical trauma is minimized, topical NSAIDs are more likely to suppress postoperative inflammation than topical steroid treatment. Therefore, clinical studies using the laser flare-cell photometer for various topical NSAIDs and determination of their optimal usage such as drug concentration were performed to get government approval in Japan. Figure 13 demonstrates an example of a multi-centered clinical study of efficacy of topical ketorolac treatment on postoperative inflammation. Clinical studies with a similar protocol were also performed [32–45]. It is also reported that smaller incisions cause less postoperative inflammation than larger ones [37, 46].
Multi-center clinical study of efficacy of ketorolac on postoperative inflammation in cataract surgery (Revised with permission from [207] a Flare (albumin equivalent concentration) b Cell counts. The patients undergoing cataract surgery with posterior intraocular lens implantation were divided into 3 groups; one group underwent routine postoperative steroid treatment, the other two groups underwent topical ketorolac 0.1 or 0.5% in addition to the routine regimen. Topical 0.5% ketorolac treatment is more potent in suppressing the postoperative increase of flare. Ketorolac group ○: 0.1%, ■: 0.5%, ●: Control group (Vehicle). [Bar: mean ± S.D.]
Miyake revealed the pathophysiology of unfavorable cystoid macular edema (CME) after cataract surgery with the help of funduscopy and fluorescein angiography [47]. He reports that the suppression of the inflammatory response or flare values in the anterior chamber of the eye played a key role in prevention of postoperative CME. The conclusion is that application of topical NSAIDs and the development of a surgical method which helps minimize surgical trauma can reduce the incidence of CME by applying the conclusions stemming from Miyake’s work. Other studies report similar results, i.e., flare values showed a correlation with postoperative best corrected visual acuity but not central macular thickness determined by OCT, but high flare values could predict the possibility of the development of CME in cataract surgery [48]. It is also reported that there exists a pharmacological suppressive effect on the development of CME [37, 49–54].
The effects of surgical methods [55–62] and neodymium-doped yttrium aluminum garnet (Nd: YAG) capsulotomy [63] as well as the duration of phacoemulsification [64] on postoperative inflammation in cataract surgery were also studied. The recently developed small-incision cataract surgery can minimize postoperative inflammatory responses. However, it causes an acute deterioration of the BAB function within the first postoperative 48 h, with inter-individual variations in the inflammatory responses within 1 day postoperatively [40].
Bio-compatibility of the components of different types of IOL materials or design [65–67] and its effects on eyes of pseudoexfoliation syndrome [68–70], newly accommodative IOLs [70] and Phakic IOLs [71] or irrigation solution [72, 73] were also investigated. Heparin-coated IOLs could reduce cellular deposits on IOLs but their postoperative inflammatory response was similar to other non-coated lens materials as shown in Table 1 [7, 74, 75].
One cause of toxic lens syndrome is residual endotoxins in IOLs. The limit of residual endotoxin concentration in IOLs was determined using intracameral injections of endotoxins in Dutch rabbits that were measured with the laser flare-cell photometer (Figs. 14, 15) [76]. Following this study, the standard of residual endotoxin concentrations in IOLs was changed by the International Organization for Standardization and by the Food and Drug Administration in the U.S.A.
Change of aqueous flare after intracameral injection of endotoxin solution [76]. Endotoxin solution (5.0E U/mL, 50 μL) was injected intracamerally using a microsyringe with 30G-needle. Same amount of limulus amebocyte lysate (LAL) Reagent Water (LRW) solution was injected into the fellow eye as a control. Both endotoxin solution and LRW solution cause the initial increase of flare, following which flare values decrease gradually till 24 h after treatment. LRW solution causes mechanical deterioration of BAB function. The difference in flare values between endotoxin and LRW solutions is a true inflammatory response caused by endotoxin. The area under the curve (AUC) is calculated as total inflammatory reaction caused by the intracamerally injected endotoxin. ●: Endotoxin solution (5.0 EU/mL, 50 μL) ○: Control (LRW) solution. [Bar: mean ± S.D.]
AUC for flare and number of cells caused by endotoxin and LRW solution [76]. Amount of injected endotoxin solution was determined by the endotoxin concentration time weight difference in the microsyringe between before and after the intracameral injection. Injected volume of LRW solution was also determined by the same method, and average AUC for flare and number of cells were calculated. The regression curves of AUC for flare and number of cells are; y = 110175x + 127126 (R2 = 0.4153, P = 0.000161)and y = 110175x + 127126 (R2 = 0.4153, P = 0.000161), respectively. Based on the results by LRW solution and endotoxin solution, 0.2 EU is assessed to be an appropriate safe residual concentration to avoid breakdown of BAB. ●: Endotoxin solution, ○: LRW solution
Endogenous intraocular inflammation
Slit-lamp microscopy plays an important role in assessment and diagnosis of endogenous intraocular inflammatory diseases (uveitis) by detecting flare and cells in the aqueous. The permeability of BAB differs in its pathophysiologic status, since BAB has the structure of a biological sieve [77]. The worse the deterioration of the BAB function, the more plasma protein and cells leak into the aqueous humor [5, 78]. In normal conditions the main protein in the aqueous is pre-albumin; however, once the aqueous protein concentration exceeds 700 mg/100 mL the plasma albumin begins to leak, and then the globulin leaks as well [79]. However, with slit microscopy alone it is difficult to discover minute differences in aqueous protein changes. At the same time, using slit microscopy, expert ophthalmologists can differentiate cells in the aqueous into red blood cells, white blood cells, lymphocytes and pigments from the iris by their shape, size and the warm current pattern, making it an indispensable diagnostic tool [80]. The flare-cell photometer can count the number of particles in the aqueous, but cannot distinguish their roles as slit-lamp microscopy can. Furthermore, the flare-cell photometer can give a quantitative assessment, but has the limitation of small measurement volume and time. Therefore, cell count data obtained by a flare-cell photometer should be verified by findings obtained by slit-lamp microscopy. It is noteworthy that the diagnostic power of the flare-cell photometer is similar to slit-lamp microscopy and a definitive diagnosis of intraocular inflammation should be performed using specified diagnostic methods [81, 82].
Although it is reported that the flare-cell photometer is not used by experts of uveitis [83–85], many studies report that the flare-cell photometer has advantages in its quantitative and objective assessment [22, 86–88]. Furthermore, the increase in flare values persists longer than the presence of cells in the aqueous, indicating that a breakdown of BAB persists longer than the disappearance of cells as detected by slit-lamp microscopy [89–91].
Hence, the flare-cell photometer is a complementary tool to a slit-lamp microscope. However, it has also been used to detect various pathophysiologic facts and clinical courses in uveitis as is reported [92].
Juvenile idiopathic arthritis or chronic anterior uveitis
There are several reports in which clinical findings including drug responses, recurrence [93] and risk of complications in juvenile idiopathic arthritis or chronic anterior uveitis in children [94] were revealed by flare-cell photometer.
Vogt-Koyanagi-Harada disease
Differences in clinical responses between Vogt-Koyanagi-Harada disease patients with initial-onset and patients with recurrence are reported [95, 96]. Furthermore, a close relationship between serum IL-23 and aqueous flare and cells [97] as well as clinical characteristics are also reported [98].
Behçet’s disease
In Japan, Behçet’s disease is a major concern in uveitis of the posterior segment and recent clinical publications deal with the efficacy of molecularly-targeted drugs [99–101]. Clinical studies of Behçet’s disease report that flare values correlate well with retinal vascular leakage determined by fluorescein angiography [102] and treatment response (Fig. 16). Flare values reflect the degree of severity of Behçet’s disease or sarcoidosis but the treatment effects of steroids on flare values are not remarkable in patients with toxoplasmosis or birdshot disease. It was concluded that the sensitivity of the flare-cell photometry is good for the clinical evaluation and follow-up in various types of uveitis of the posterior segment [103].
Clinical follow-up of a patient with Behçet’s disease. The patient developed 2 attacks with a short interval between. Both increases of flare and cells in the aqueous after the first attack (arrow) show a reducing pattern toward the second attack. Following the second attack, they increase suddenly. Hence, flare or cell measurements cannot reliably predict the development of an attack. Flare values are converted to albumin equivalent protein concentrations. The increased inflammatory findings decrease with time. Arrow: attack of intraocular inflammation, ●: flare, ○: cell, ■: visual acuity
Fuchs’ syndrome
In eyes with Fuchs’ syndrome, flare and cell values are higher than their non-affected fellow eyes, and they correlate with the degree of iris depigmentation and keratic precipitates [104]. But their increase is not so manifest compared with other types of anterior uveitis [105]. In the diagnosis of Fuchs’ syndrome, it is interesting that diffuse medium-sized round keratic precipitates, low flare readings, iris stromal atrophy without hypochromia and vitreous opacities in the absence of macular edema are more important findings than heterochromia [106].
Sjögren’s syndrome
In a comparative study between patient groups with Sjögren’s syndrome and with rheumatoid arthritis and normal subjects, an elevation of flare values in the patient groups was observed and the degree of the damage to BAB is reported to relate with ocular surface inflammation and dryness evaluated by conjunctival cytology and levels of serum albumin in tears [107].
Sympathetic ophthalmia and acute retinal necrosis
It is reported that flare-cell photometry is useful in determining treatment doses for patients with sympathetic ophthalmia who underwent cyclosporine and prednisolone therapy [108], or patients with acute retinal necrosis who underwent anti-viral treatment [109] (Fig. 17). Although not yet accepted as such, this diagnostic criteria of acute retinal necrosis was recently reported in Japan [110].
Clinical course of acute retinal necrosis. After paracentesis to confirm the diagnosis of acute retinal necrosis, both flare and cells increase transiently. After the vitrectomy, they decrease with a combination of systemic anti-viral and steroid treatment. *arrow: paracentesis, **arrow: vitrectomy, ●: Flare, ○: cell
Cytomegalovirus (CMV) retinitis
Pathophysiologic and clinical aspects of CMV retinitis and AIDS were investigated with flare-cell photometers [111–113]. Flare values can be a useful index reflecting the severity of chorioretinal disorders. In an immunocompetent patient with iridocyclitis with ocular hypertension without retinitis caused by CMV, reduction of aqueous flare correlated with the reduction of virus copies in aqueous humor [114]. Comparative study of characteristics in clinical findings among Herpes simplex virus, Varicella zoster virus and Cytomegalovirus is reported and this data proved useful to differential diagnosis [115].
Human leukocyte antigen (HLA)-B-27 related acute anterior uveitis
Clinical findings in HLA-B-27 related acute anterior uveitis were studied using the flare-cell photometer, optical coherent tomography and a positive correlation between central retinal thickness and initial flare values in the affected eye was reported [116].
Evaluation of treatment efficacy
Studies regarding the efficacy and adverse effects of routine medication in patients with uveitis [42, 117, 118] report that posterior sub-Tenon steroid triamcinolone acetonide injections for the treatment of posterior ocular inflammation can suppress flare values, but it may cause adverse events such as intraocular hypertension and development of cataracts which exceed the efficacy of the treatment [119].
Cataract surgery in patients with uveitis
Cataract surgery in patients with uveitis is important. Studies using the flare-cell photometer indicate that useful data regarding indication including optimal time of surgery was acquired, and pre-and post-surgical regimen and postoperative clinical courses could be evaluated [68, 120–124].
Corneal surgery
Corneal surgery is classified into two groups regarding pathophysiologic aspects affecting the BAB function: penetrating keratoplasty (PKP) and corneal refractive surgery such as laser in situ-keratomileusis (LASIK).
PKP has two major phases, postoperative clinical course and rejection episodes [125]. In terms of the postoperative course (Fig. 18), increased flare values shortly after surgery tend mostly to decrease, which is believed to be the result of direct damage to the BAB function during surgery due to drastic changes in the homeostasis of the anterior structure and physiology. In triple procedure; PKP and cataract surgery (ECCE) with IOL implantation, the postoperative increase of flare and cells was higher than in PKP alone [126]. It is noteworthy that, when compared with normal eyes, high flare values in treated eyes remained high longer than those in post-cataract surgery. It cannot be inferred whether the duration of these subclinical high flare values is due to host-graft immunological reaction or not. In cases of rejection, increases of flare and cells in the aqueous could be noted by the appearance of keratic precipitates as well as observed by slit-lamp microscopy. Flare-cell photometry could monitor effects of steroid therapy on rejection [125], but could not predict the rejection in advance. Subjective symptoms and a careful slit-lamp examination are the preferred methods in determining the onset of rejection.
Flare values after penetrating keratoplasty. Increased flare values are distributed in early postoperative period, although some cases are plotted in the normal flare range which is shown by shaded zone. Furthermore, there are cases which show flare higher than normal range even 40 weeks after the surgery. It is not known whether this persistent increase of flare values might be caused by a subclinical immune reaction in the anterior chamber or anatomical changes. In general, it is difficult to determine flare and cells in the aqueous in the presence of graft edema or Descemet’s folding. Shaded zone: flare value within normal range
In keratorefractive surgery, uneventful radial keratotomy showed a transient increase of flare on postoperative day 1 and then returned to the preoperative level; but cases of perforation showed an increase of flare values for the first postoperative week [127–129]. It is thought that the perforation causes a similar pathophysiologic response to paracentesis.
There are different reports relating to LASIK; one claims that it caused an increase in flare postoperatively and returned to preoperative levels within a week [130]. This flare change might correlate with the depth of photo-abrasion. Another report says it did not cause significant changes in postoperative flare regardless of the amount of abrasion depth [131]. It is reported that intracorneal ring segments did not affect flare values either [130].
Glaucoma (pharmacological and surgical studies)
Glaucoma is a major concern in ophthalmology, since the incidence of patients with glaucoma is high and it constitutes a major cause of blindness. Both the physiology and pharmacodynamics of the aqueous humor play an important role in the diagnosis of glaucoma. Therefore, the study of aqueous humor dynamics is important to the understanding of the etiology of glaucoma.
From the information above we can infer that in healthy eyes diurnal variations of the intraocular pressure demonstrate an inverted pattern with flare values. Provided the BAB function is normal and stable under the fixed volume anterior chamber, flare values are determined by the ratio of aqueous protein and secretion of aqueous humor. An increase of aqueous humor secretion causes the increase in intraocular pressure and a relative decrease of flare values, and vice versa [18, 132]. Although there are two major routes for aqueous outflow, conventional and uveoscleral outflow, the pharmacodynamics regarding glaucoma drugs can be assessed by comparative measurement of diurnal changes of intraocular pressure and flare with and without medication in the same individual [133–135].
Clinical investigation of pathophysiology in eyes with glaucoma
The major topical glaucoma medication at present is prostaglandin analogues and its effects including preservatives on the BAB function were investigated [136–140]. The pathophysiology of patients with pseudoexfoliation syndrome (PEX) was also investigated and revealed that there was a breakdown in the BAB function, since a significant increase of flare was detected in eyes with PEX compared with eyes with PEX (−) [141–143]. It is also reported that there is a difference in the increase in flare after trabeculectomy between eyes with PEX and POAG without PEX [144, 145]. An involvement of inflammatory process is approved in PEX. It is also reported that there is a positive correlation between the increase in flare and decrease in corneal endothelial cell density [146].
Effects of surgical methods
Effects of surgical methods in glaucoma on intraocular pressure and BAB were investigated [144, 145]; i.e., argon laser trabeculoplasty causes more intense postoperative inflammation than diode laser trabeculoplasty [147] and the difference in postoperative inflammatory reaction between deep sclerectomy with collagen implants and conventional trabeculectomy was also investigated [148].
Here, the quantitative assessment of flare values plays a key role in comparative studies and can elucidate the pathophysiology in glaucomatous eyes better than qualitative flare assessment by slit-lamp microscopy.
Diabetes mellitus, retinal vascular disorders and the BAB function
The principal etiology of diabetes mellitus involves microvascular disorders that affect the blood-ocular barrier. The deterioration of the blood-retinal barrier in diabetic retinopathy is detected by the extent of non-perfusion areas and the amount of fluorescein leakage revealed by fluorescein angiography. Diabetic iridopathy in patients with progressive diabetic retinopathy can be a complication involving BAB [149]. The relationship between the BAB function and diabetes mellitus was investigated [150–158], and it was concluded that the assessment of flare values by the flare-cell photometer can provide one predictor of diabetic retinopathy. In patients with diabetes mellitus, retinal photocoagulation [159] or cataract surgery [160, 161] can cause worse deterioration of the BAB function than in patients without diabetes mellitus. It is also reported that intravitreal injections of either dexamethasone or triamcinolone during vitrectomy in patients with PDR can reduce postoperative inflammation in the anterior segment of the eye [162, 163]. Furthermore, triamcinolone-assisted vitrectomy could improve surgical procedure though the facilitation of visibility of the vitreous body [162].
Effects of intravitreal injection treatment
Intravitreal anti-vascular endothelial growth factor (anti-VEGF) injections are common in various refractory macular diseases. The effects of intravitreal injections on the BAB function were investigated. Intravitreal injection of bevacizumab to treat neovascular age-related macular degeneration did not show a significant increase of flare, rather, there was a slight reduction. This is believed to be due to the anti-inflammatory effect of the anti-VEGF qualities of the medication [164].
Retinal vascular disorders
Although it is not known whether retinal vascular disorders such as retinal venal or arterial occlusion affect the BAB function, some comparative studies between affected eyes and non-affected fellow eyes or control eyes reveal that retinal vascular disorders affect the BAB function [165–168].
Chorioretinal diseases
The relationship between the BAB function and chorioretinal diseases has been studied extensively including in rhegmatogenous retinal detachment [169, 170] choroideremia [171], retinal pigment epitheliopathy [172], retinitis pigmentosa [173–176] and both malignant and benign uveal tumors [177].
It can be concluded from these studies that a deterioration of the BAB function occurs in eyes with progressive chorioretinal diseases.
Other clinical studies of effects of ophthalmic treatment on BAB function
Strabismus surgery
Blood supply through the extraocular muscle is important in maintaining the physiology of the anterior segment of the eye. Standard surgical procedure in strabismus surgery on either one or two extraocular muscles does not show significant postoperative changes in flare values [178–180].
Pharmacology
Preservatives in eye drops such as benzalkonium chloride (BAK) have potent cytotoxic effects on the ocular surface. Artificial tears containing BAK cause a significant increase of flare, indicating an effect on the BAB function [140, 181].
Systemic disorders and BAB function
Psoriasis is an unknown etiologic systemic inflammatory disease classified into vulgaris and other types. Flare in the aqueous of patients with psoriasis showed higher values than in healthy subjects regardless of the type of disease. Furthermore, flare intensity increases with the length in disease duration and severity, indicating that psoriasis caused subclinical disruption of the BAB function [182].
According to studies of flare in the aqueous of patients with Graves’ ophthalmopathy who underwent orbital decompression, extraocular muscle or eye lid surgery, orbital decompression surgery caused large increases of flare for 3 months during the first postoperative week. Extraocular muscle surgery showed peak flare values on the first postoperative day that then returned to the preoperative levels. It is also reported that standard strabismus surgery did not cause postoperative increases in flare values. It is not known whether BAB in patients with Grave’s disease is susceptible to surgical invasion or not. Lid surgery had no significant effect on postoperative flare levels [183].
In patients with a first acute anterior unilateral uveitic attack associated with seronegative spondyloarthropathy or ankylosing spondylitis as a subgroup, there was a close correlation between macular thickness detected by optical coherence tomography and inflammation by flare measurement [184].
Although patients with asymptomatic hepatitis C showed impairment of the BAB and thickened choroids, choroidal thickness increased with the increase of subclinical inflammation in the aqueous. Furthermore, Patients with significant liver fibrosis showed the highest flare values and the thickest choroids, indicating that BAB might be affected by hepatic disorders [185]. On the other hand, in patients with various intestinal inflammatory diseases such as Crohn’s disease, ulcerative colitis, and disorders of undetermined nature, when taking into account the differences in age, the levels of flare values in the aqueous are reported to be within the normal range [186].
Animal experiment
The laser flare-cell photometer makes a great contribution to animal experiments in two major aspects. Not only toxic effects but also pharmacological effects of various substances on the BAB function are investigated using animals. Quantitative evaluation of responses in BAB requires aspirated samples of aqueous humor. Aspiration of aqueous humor itself causes a breakdown of the BAB function [187, 188], therefore, the study protocol needs one animal for each sample. This requires a large number of sacrificed animals and involves potential deterioration of reliability due to inter-individual variations. Laser flare-cell photometry enables non-invasive multiple examinations within a short time range without anesthetization [76, 189]. Serial examinations can minimize inter-individual variations and give good reproducible results. In rabbit experiments, pigmented rabbits are preferred to albino rabbits in order to avoid noise reflexes from the fundi.
So far various drugs such as endotoxins, NSAIDs [190, 191], steroids [192, 193], vascular endothelial growth factors [194], glaucoma drugs [195–197] were studied in eyes with [198–203] or without prostaglandin pretreatment, or other inflammatory drugs inducing experimental uveitis in rabbit [204].
At the same time, the effects of anterior segment ischemic models [205] as well as retinal photocoagulation [206] on the BAB function were also investigated.
Conclusion
Laser flare-cell photometers enable a noninvasive and quantitative determination of the BAB functions. The application of the instrument revealed novel findings through the evaluation of flare and cells in the aqueous which could not be obtained by slit-lamp microscopic evaluations. Especially, a quantitative flare evaluation with good repeatability which cannot be evaluated by slit-lamp microscopy came to play a key role in the assessment of intraocular inflammation. Furthermore, in animal experiments, the repeatable applicability of the instrument in individuals can improve the accuracy of data by minimizing inter-individual variations, resulting in reduction in the number of animals in experiments.
References
Sawa M, Okubo A, Okuno Y, Shimizu H. Determination of aqueous protein concentration by laser slit-lamp microscope. Nippon Ganka Gakkai Zasshi. 1987;91:102–6 (in Japanese).
Sawa M, Tsurimaki Y, Tsuru T, Shimizu H. New quantitative method to determine protein concentration and cell number in aqueous in vivo. Jpn J Ophthalmol. 1988;32:132–42.
Kronfeld PC. The protein content of the aqueous humor in man. Am J Ophthalmol. 1941;24:1121–31.
Krause U, Raunio V. The protein of the pathologic human aqueous humor. Ophthalmologica. 1970;16:280–7.
Zirm M. Proteins in aqueous humor. Adv Ophthalmol. 1980;40:100–72.
Shah SM, Spalton DJ, Taylor JC. Correlations between laser flare measurements and anterior chamber protein concentrations. Invest Ophthalmol Vis Sci. 1992;33:2878–84.
Sawa M. Clinical application of laser flare-cell meter. Jpn J Ophthalmol. 1990;34:346–63.
Sawa M. Development of non-invasive clinical examination methods for the anterior segment of the eye and their clinical significance. Nihon Ganka Gakkai Zasshi. 2011;115:177–211 (in Japanese).
Sawa M, Masuda K. Topical indomethacin in soft cataract aspiration. Jpn J Ophthalmol. 1976;20:514–9.
Mochizuki M, Sawa M, Masuda K. Topical indomethacin in intracapsular extraction of senile cataract. Jpn J Ophthalmol. 1977;21:215–26.
Miyake K, Maekubo K, Miyake Y. Cause and therapy of fibrin reaction after posterior chamber lens implantation. J Ophthalmic Surg (Jpn). 1988;1:153–60 (in Japanese).
Bhattarcherjee P, Eakins KE. Inhibition of prostaglandin synthetase system in ocular tissue by indomethacin. Br J Ophthalmol. 1974;50:227–30.
Masuda K. Anti-inflammatory agent: Nonsteroidal anti-inflammatory drugs. In: Ed Sears MI, editor. Pharmacology of the Eye. Berlin: Springer; 1984. p. 539–51.
Oshika T, Kato S. Change in aqueous flare and cell after mydriasis. Jpn J Ophthalmol. 1989;33:271–8.
Onodera T, Gimbel HV, DeBroff BM. Aqueous flare and cell number in healthy eyes of Caucasians. Jpn J Ophthalmol. 1993;37:445–51.
Shah SM, Spalton DJ, Smith SE. Measurement of aqueous cells and flare in normal eyes. Br J Ophthalmol. 1991;75:348–52.
El-Harazi SM, Ruiz RS, Feldman RM, Chuang AZ, Villanueva G. Quantitative assessment of aqueous flare: the effect of age and pupillary dilation. Ophthalmic Surg Lasers. 2002;33:379–82.
Oshika T, Araie M, Masuda K. Diurnal variation of aqueous flare in normal human eyes measured with laser flare-cell meter. Jpn J Ophthalmol. 1988;32:143–50.
Hogan MJ, Kimura SJ, Thygeson P. Signs and symptoms of uveitis, I Anterior uveitis. Am J Ophthalmol. 1959;47:155–70.
Ohara K, Okubo A, Miyazawa A, Miyamoto T, Sasaki H, Oshima F. Aqueous flare and cell measurement using laser in endogenous patients. Jpn J Ophthalmol. 1989;33:265–70.
Oshika T, Nishi M, Mochizuki M, Nakamura M, Kawashima H, Iwase K, et al. Quantitative assessment of aqueous flare and cells in uveitis. Jpn J Ophthalmol. 1989;33:279–87.
Bernasconi O, Papadia M, Herbort CP. Sensitivity of laser flare photometry compared to slit-lamp cell evaluation in monitoring anterior chamber inflammation in uveitis. Int Ophthalmol. 2010;30:495–500.
Agrawal R, Keane PA, Singh J, Saihan Z, Kontos A, Pavesio CE. Comparative analysis of anterior chamber flare grading between clinicians with different levels of experience and semi-automated laser flare photometry. Ocul Immunol Inflamm. 2014;26:1–10.
Shimizu H, Tsurimaki Y, Sawa M, Nakanishi T, Usui M, Osanai T, et al. Clinical comparative study of laser flare meter (FM-500) with laser flare-cell meter (FC-1000). Ganka Rinsyou Iho. 1993;87:63–7 (in Japanese).
Nguyen NX, Martus P, Küchle M. A clinical comparative study of tyndallometry using 2 laser flare meters. Klin Monbl Augenheilkd. 1996;209:89–93 (in German).
Sawa M, Kamura Y, Masuda K, Fujino Y, Merino G, Shimizu H, et al. Clinical evaluation of flare-cell meter Mark II (FC-2000). Ganka Rinsho Iho. 1997;91:1194–8 (in Japanese).
Küchle M. Laser tyndallometry in anterior segment diseases. Curr Opin Ophthalmol. 1994;5:110–6.
Shah SM, Spalton DJ, Allen RJ, Smith SE. A comparison of the laser flare cell meter and fluorophotometry in assessment of the blood-aqueous barrier. Invest Ophthalmol Vis Sci. 1993;34:3124–30.
Chiou AG, Florakis GJ, Herbort CP. Correlation between anterior chamber IgG/albumin concentrations and laser flare photometry in eyes with endogenous uveitis. Ophthalmologica. 1998;212:275–7.
Mori M, Inamochi K, Araie M. Evaluation of albumin concentration in rabbit anterior chamber with laser flare-cell meter. Curr Eye Res. 1995;14:593–9.
Schalnus R, Ohrloff C. Quantification of blood-aqueous barrier function using laser flare measurement and fluorophotometry–a comparative study. Lens Eye Toxic Res. 1992;9:309–20.
Shah SM, McHugh JD, Spalton DJ. The effects of subconjunctival betamethasone on the blood aqueous barrier following cataract surgery: a double-blind randomised prospective study. Br J Ophthalmol. 1992;76:475–8.
Hessemer V, Schmitt K. Monotherapy vs combination therapy with topical prednisolone and indomethacin. Ophthalmologe. 1995;92:31–4 (in German).
Nicolás T, Benítez del Castillo JM, Díaz D, Castillo A, García-Sánchez J. Effects of subconjunctival methylprednisolone on the blood aqueous barrier following cataract surgery. Int Ophthalmol. 1995;19:235–8.
Hessemer V, Schmitt K, Jacobi A. Anti-inflammatory effects and aqueous humor concentration of various nonsteroidal anti-inflammatory drugs in extracapsular cataract surgery. Klin Monbl Augenheilkd. 1996;208:161–6 (in German).
Chang DF, Wong V. Two clinical trials of an intraocular steroid delivery system for cataract surgery. Trans Am Ophthalmol Soc. 1999;97:261–74.
Miyake K, Masuda K, Shirato S, Oshika T, Eguchi K, Hoshi H, et al. Comparison of diclofenac and fluorometholone in preventing cystoid macular edema after small incision cataract surgery: a multicentered prospective trial. Jpn J Ophthalmol. 2000;44:58–67.
Herbort CP, Jauch A, Othenin-Girard P, Tritten JJ, Fsadni M. Diclofenac drops to treat inflammation after cataract surgery. Acta Ophthalmol Scand. 2000;78:421–4.
Struck HG, Bariszlovich A. Comparison of 0.1% dexamethasone phosphate eye gel (Dexagel) and 1% prednisolone acetate eye suspension in the treatment of post-operative inflammation after cataract surgery. Graefes Arch Clin Exp Ophthalmol. 2001;239:737–42.
Findl O, Amon M, Petternel V, Kruger A. Early objective assessment of intraocular inflammation after phacoemulsification cataract surgery. J Cataract Refract Surg. 2003;29:2143–7.
Wadood AC, Armbrecht AM, Aspinall PA, Dhillon B. Safety and efficacy of a dexamethasone anterior segment drug delivery system in patients after phacoemulsification. J Cataract Refract Surg. 2004;30:761–8.
Meacock WR, Spalton DJ, Bender L, Antcliff R, Heatley C, Stanford MR, et al. Steroid prophylaxis in eyes with uveitis undergoing phacoemulsification. Br J Ophthalmol. 2004;88:1122–4.
Arcieri ES, Santana A, Rocha FN, Guapo GL, Costa VP. Blood-aqueous barrier changes after the use of prostaglandin analogues in patients with pseudophakia and aphakia: a 6-month randomized trial. Arch Ophthalmol. 2005;123:186–92.
Saari KM, Nelimarkka L, Ahola V, Loftsson T, Stefánsson E. Comparison of topical 0.7% dexamethasone-cyclodextrin with 0.1% dexamethasone sodium phosphate for postcataract inflammation. Graefes Arch Clin Exp Ophthalmol. 2006;244:620–6.
Petternel V, Findl O, Kruger A, Schauersberger J, Amon M. Effect of tropicamide on aqueous flare before and after cataract surgery. J Cataract Refract Surg. 2000;26:382–5.
Laplace O, Goldschild M, De Saint Jean M, Guepratte N, Baudouin C. Evaluation by laser flare meter of the inflammatory response after cataract surgery. J Fr Ophthalmol. 1998;21:265–9 (in French).
Miyake K. Prevention of cystoid macular edema after lens extraction by topical indomethacin. I. A preliminary report. Albrecht Graefes Arch Clin Exp Ophthalmol. 1977;203:81–8.
Sourdille P, Santiago PY. Optical coherence tomography of macular thickness after cataract surgery. J Cataract Refract Surg. 1999;25:256–61.
Miyake K, Ota I, Maekubo K, Ichihashi S, Miyake S. Latanoprost accelerates disruption of the blood-aqueous barrier and the incidence of angiographic cystoid macular edema in early postoperative pseudophakias. Arch Ophthalmol. 1999;117:34–40.
Ursell PG, Spalton DJ, Whitcup SM, Nussenblatt RB. Cystoid macular edema after phacoemulsification: relationship to blood–aqueous barrier damage and visual acuity. J Cataract Refract Surg. 1999;25:1492–7.
Miyake K, Nishimura K, Harino S, Ota I, Asano S, Kondo N, et al. The effect of topical diclofenac on choroidal blood flow in early postoperative pseudophakias with regard to cystoid macular edema formation. Invest Ophthalmol Vis Sci. 2007;48:5647–52.
Dieleman M, Wubbels RJ, van Kooten-Noordzij M, de Waard PW. Single perioperative subconjunctival steroid depot versus postoperative steroid eyedrops to prevent intraocular inflammation and macular edema after cataract surgery. J Cataract Refract Surg. 2011;37:1589–97.
Miyake K, Ota I, Miyake G, Numaga J. Nepafenac 0.1% versus fluorometholone 0.1% for preventing cystoid macular edema after cataract surgery. J Cataract Refract Surg. 2011;37:1581–8.
Ersoy L, Caramoy A, Ristau T, Kirchhof B, Fauser S. Aqueous flare is increased in patients with clinically significant cystoid macular oedema after cataract surgery. Br J Ophthalmol. 2013;97:862–5.
Sourdille P. Lensectomy-vitrectomy indications and techniques in cataract surgery. Curr Opin Ophthalmol. 1997;8:56–9.
Willwerth AE, Kremer T, Hermeking H, Schmitz P, Gerke E. Disruption of the blood-aqueous barrier after implantation of sclera-fixed posterior chamber lenses. Early postoperative phase and long-term outcome. Ophthalmologe. 1997;94:24–9 (in German).
Laurell CG, Zetterström C, Philipson B, Syrén-Nordqvist S. Randomized study of the blood-aqueous barrier reaction after phacoemulsification and extracapsular cataract extraction. Acta Ophthalmol Scand. 1998;76:573–8.
Chee SP, Ti SE, Sivakumar M, Tan DT. Postoperative inflammation: extracapsular cataract extraction versus phacoemulsification. J Cataract Refract Surg. 1999;25:1280–5.
Dick HB, Schwenn O, Krummenauer F, Krist R, Pfeiffer N. Inflammation after sclerocorneal versus clear corneal tunnel phacoemulsification. Ophthalmology. 2000;107:241–7.
Amino K, Yamakawa R. Long-term results of out-of-the-bag intraocular lens implantation. J Cataract Refract Surg. 2000;26:266–70.
Stifter E, Menapace R, Kriechbaum K, Vock L, Luksch A. Effect of primary posterior continuous curvilinear capsulorhexis with and without posterior optic buttonholing on postoperative anterior chamber flare. J Cataract Refract Surg. 2009;35:480–4.
Conrad-Hengerer I, Hengerer FH, Al Juburi M, Schultz T, Dick HB. Femtosecond laser-induced macular changes and anterior segment inflammation in cataract surgery. J Refract Surg. 2014;30:222–6.
Altamirano D, Mermoud A, Pittet N, van Melle G, Herbort CP. Aqueous humor analysis after Nd: YAG laser capsulotomy with the laser flare-cell meter. J Cataract Refract Surg. 1992;18:554–8.
Kruger AJ, Schauersberger J, Abela-Formanek C, Schild G, Kellner C, Kolodjaschna J, et al. Effect of duration of phacoemulsification on postoperative inflammation—a retrospective study. Klin Monbl Augenheilkd. 2001;218:204–8 (in German).
Schauersberger J, Kruger A, Abela C, Müllner-Eidenböck A, Petternel V, Svolba G, et al. Course of postoperative inflammation after implantation of 4 types of foldable intraocular lenses. J Cataract Refract Surg. 1999;25:1116–20.
Hollick EJ, Spalton DJ, Ursell PG. Surface cytologic features on intraocular lenses: can increased biocompatibility have disadvantages? Arch Ophthalmol. 1999;117:872–8.
Nguyen NX, Tomida I, Küchle M. Blood-aqueous barrier after phacoemulsification with posterior chamber lens implantation; foldable acrylate lens vs PMMA lens–a clinical study on 46 eyes. Klin Monbl Augenheilkd. 2000;217:215–8 (in German).
Schumacher S, Nguyen NX, Küchle M, Naumann GO. Quantification of aqueous flare after phacoemulsification with intraocular lens implantation in eyes with pseudoexfoliation syndrome. Arch Ophthalmol. 1999;117:733–5.
Abela-Formanek C, Amon M, Schauersberger J, Schild G, Kruger A. Postoperative inflammatory response to phacoemulsification and implantation of 2 types of foldable intraocular lenses in pseudoexfoliation syndrome. Klin Monbl Augenheilkd. 2000;217:10–4 (in German).
Nguyen NX, Langenbucher A, Huber S, Seitz B, Küchle M, Erlangen Accommodative Intraocular Lens Study Group. Short-term blood-aqueous barrier breakdown after implantation of the 1CU accommodative posterior chamber intraocular lens. J Cataract Refract Surg. 2002;28:1189–94.
Pérez-Santonja JJ, Iradier MT, Benítez del Castillo JM, Serrano JM, Zato MA. Chronic subclinical inflammation in phakic eyes with intraocular lenses to correct myopia. J Cataract Refract Surg. 1996;22:183–7.
Kruger A, Amon M, Abela-Formanek C, Schild G, Kolodjaschna J, Schauersberger J. Effect of heparin in the irrigation solution on postoperative inflammation and cellular reaction on the intraocular lens surface. J Cataract Refract Surg. 2002;28:87–92.
Findl O, Amon M, Kruger A, Petternel V, Schauersberger J. Effect of cooled intraocular irrigating solution on the blood-aqueous barrier after cataract surgery. J Cataract Refract Surg. 1999;25:566–8.
Pande M, Shah SM, Spalton DJ. Correlations between aqueous flare and cells and lens surface cytology in eyes with poly(methyl methacrylate) and heparin-surface-modified intraocular lenses. J Cataract Refract Surg. 1995;21:326–30.
Shah SM, Spalton DJ. Comparison of the postoperative inflammatory response in the normal eye with heparin-surface-modified and poly(methyl methacrylate) intraocular lenses. J Cataract Refract Surg. 1995;21:579–85.
Sakimoto A, Sawa M, Oshida T, Sugaya S, Hirono T, Ishimori A. Minimum endotoxin concentration causing inflammation in the anterior segment of rabbit eyes. Jpn J Ophthalmol. 2009;53:425–32.
Davson H. Physiology of the eye. London: Churchill Livingstone; 1980. p. 9–81.
Goodner EK. Aqueous humor studies in uveitis. In: Aronson SB, editor. Clinical Methods in Uveitis. St Louis: Mosby; 1968. p. 197–9.
Dernouchamps JP. The proteins of the aqueous humor. Doc Ophthalmol. 1982;53:193–248.
The Standardization of Uveitis Nomenclature (SUN) Working Group. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am J Ophthalmol. 2005;140:509–16.
Ladas JG, Wheeler NC, Morhun PJ, Rimmer SO, Holland GN. Laser flare-cell photometry: methodology and clinical applications. Surv Ophthalmol. 2005;50:27–47.
Mochizuki M, Sugita S, Kamoi K, Takase H. A new era of uveitis: impact of polymerase chain reaction in intraocular inflammatory diseases. Jpn J Ophthalmol. 2016. doi:10.1007/s10384-016-0474-9.
Wakefield D, Herbort CP, Tugal-Tutkun I, Zierhut M. Controversies in ocular inflammation and immunology laser flare photometry. Ocul Immunol Inflamm. 2010;18:334–40.
Zierhut M, Heiligenhaus A, deBoer J, Cunningham ET, Tugal-Tutkun I. Controversies in juvenile idiopathic arthritis-associated uveitis. Ocul Immunol Inflamm. 2013;21:167–79.
Yeo TH, Ilangovan S, Keane PA, Paves C, Agrawa R. Discrepancies in assessing anterior chamber activity among uveitis specialists. Jpn J Ophthalmol. 2016;60:206–11.
Küchle M, Nguyen NX, Naumann GO. Tyndallometry with the laser flare cell meter in intraocular inflammation. Ophthalmologe. 1994;91:219–23 (in German).
Herbort CP, Guex-Crosier Y, de Ancos E, Pittet N. Use of laser flare photometry to assess and monitor inflammation in uveitis. Ophthalmology. 1997;104:64–71.
de Ancos E, Pittet N, Herbort CP. Quantitative measurement of inflammation in HLA-B27 acute anterior uveitis using the Kowa FC-100 laser flare-cell meter. Klin Monbl Augenheilkd. 1994;204:330–3 (in French).
Yang P, Fang W, Huang X, Zhou H, Wang L, Jiang B. Alterations of aqueous flare and cells detected by laser flare-cell photometry in patients with Behcet’s disease. Int Ophthalmol. 2010;30:485–9.
Tappeiner C, Heinz C, Roesel M, Heiligenhaus A. Elevated laser flare values correlate with complicated course of anterior uveitis in patients with juvenile idiopathic arthritis. Acta Ophthalmol. 2011;89:e521–7.
Grajewski RS, Zurek-Imhoff B, Roesel M, Heinz C, Heiligenhaus A. Favourable outcome after cataract surgery with IOL implantation in uveitis associated with juvenile idiopathic arthritis. Acta Ophthalmol. 2012;90:657–62.
Ladas JG, Yu F, Loo R, Davis JL, Coleman AL, Levinson RD, et al. Relationship between aqueous humor protein level and outflow facility in patients with uveitis. Invest Ophthalmol Vis Sci. 2001;42:2584–8.
Wenkel H, Nguyen NX, Schönherr U, Küchle M. Laser tyndallometry and monitoring of treatment in 20 children with juvenile uveitis. Klin Monbl Augenheilkd. 2000;217:323–8 (in German).
Tappeiner C, Heinz C, Roesel M, Heiligenhaus A. Elevated laser flare values correlate with complicated course of anterior uveitis in patients with juvenile idiopathic arthritis. Klin Monbl Augenheilkd. 2000;217:323–8 (in German).
Iwahashi C, Okuno K, Hashida N, Nakai K, Ohguro N, Nishida K. Incidence and clinical features of recurrent Vogt–Koyanagi–Harada disease in Japanese individuals. Jpn J Ophthalmol. 2015;59:157–63.
Fang W, Zhou H, Yang P, Huang X, Wang L, Kijlstra A. Longitudinal quantification of aqueous flare and cells in Vogt–Koyanagi–Harada disease. Br J Ophthalmol. 2008;92:182–5.
Jiang S, Liu X, Luo L, Qu B, Huang X, Xu L, et al. Elevated serum IL-23 correlates with intraocular inflammation after cataract surgery in patients with Vogt-Koyanagi-Harada disease. Br J Ophthalmol. 2010;94:1078–82.
Yang P, Ren Y, Li B, Fang W, Meng Q, Kijlstra A. Clinical characteristics of Vogt–Koyanagi–Harada syndrome in Chinese patients. Ophthalmology. 2007;114:606–14.
Kawaguchi T, Kawazoe Y, Kamoi K, Miyanaga M, Takase H, Sugita S, et al. Clinical course of patients with Behçet’s uveitis following discontinuation of infliximab therapy. Jpn J Ophthalmol. 2014;58:75–80.
Kaburaki T, Namba K, Sonoda K, Kezuka T, Keino H, Fukuhara T, et al. The Ocular Behçet’s Disease Research Group of Japan. Behçet’s disease ocular attack score 24: evaluation of ocular disease activity before and after initiation of infliximab. Jpn J Ophthalmol. 2014;58:120–30.
Kuroyanagi K, Sakai T, Kohno H, Okano K, Akiyama G, Aoyagi R, et al. Association between the major histocompatibility complex and clinical response to infliximab therapy in patients with Behçet uveitis. Jpn J Ophthalmol. 2015;59:401–8.
Tugal-Tutkun I, Cingü K, Kir N, Yeniad B, Urgancioglu M, Gül A. Use of laser flare-cell photometry to quantify intraocular inflammation in patients with Behçet uveitis. Graefes Arch Clin Exp Ophthalmol. 2008;246:1169–77.
Guex-Crosier Y, Pittet N, Herbort CP. Sensitivity of laser flare photometry to monitor inflammation in uveitis of the posterior segment. Ophthalmology. 1995;102:613–21.
Fang W, Zhou H, Yang P, Huang X, Wang L, Kijlstra A. Aqueous flare and cells in Fuchs syndrome. Eye (Lond). 2009;23:79–84.
Küchle M, Nguyen NX. Analysis of the blood aqueous barrier by measurement of aqueous flare in 31 eyes with Fuchs’ heterochromic uveitis with and without secondary open-angle glaucoma. Klin Monbl Augenheilkd. 2000;217:159–62 (in German).
Tugal-Tutkun I, Güney-Tefekli E, Kamaci-Duman F, Corum I. A cross-sectional and longitudinal study of Fuchs uveitis syndrome in Turkish patients. Am J Ophthalmol. 2009;148:510–5.
Cellini M, Versura P, Leonetti P, Frigato M, Manfredini E, Malavolta N, et al. Ocular surface and intraocular inflammation are related in SS-I and rheumatoid arthritis patients. Rheumatol Int. 2007;27:853–7.
Nguyen NX, Küchle M, Naumann GO. Tyndallometry in monitoring therapy of sympathetic ophthalmia. Klin Monbl Augenheilkd. 1994;204:33–6 (in German).
Nguyen NX, Amann T, Küchle M. Laser-tyndallometry in eyes with acute retinal necrosis syndrome. Erlanger Laser-tyndallometry study group. Klin Monbl Augenheilkd. 1998;213:197–200 (in German).
Takase H, Okada AA, Goto H, Mizuki N, Namba K, Ohguro N, et al. Development and validation of new diagnostic criteria for acute retinal necrosis. Jpn J Ophthalmol. 2015;59:14–20.
Althaus C, Best J, Hintzmann A, Schimkat M, Hudde T, Cepin A, et al. Endothelial precipitates and laser flare photometry in patients with acquired immunodeficiency syndrome: a screening test for cytomegalovirus retinitis? Ger J Ophthalmol. 1996;5:443–8.
Magone MT, Nussenblatt RB, Whitcup SM. Elevation of laser flare photometry in patients with cytomegalovirus retinitis and AIDS. Am J Ophthalmol. 1997;124:190–8.
Herbort CP, Chave JP. Cicatrization of cytomegalovirus retinitis following introduction of highly active anti-retroviral therapy: uveitis as a possible indicator of good ocular prognosis. Graefes Arch Clin Exp Ophthalmol. 1998;236:795–7.
Kawaguchi T, Sugita S, Shimizu N, Mochizuki M. Kinetics of aqueous flare, intraocular pressure and virus-DNA copies in a patient with cytomegalovirus iridocyclitis without retinitis. Int Ophthalmol. 2007;27:383–6.
Takase H, Kubono R, Terada Y, Imai A, Fukuda S, Tomita M, et al. Comparison of the ocular characteristics of anterior uveitis caused by herpes simplex virus, varicella-zoster virus, and cytomegalovirus. Jpn J Ophthalmol. 2014;58:473–82.
Balaskas K, Ballabeni P, Guex-Crosier Y. Retinal thickening in HLA-B27-associated acute anterior uveitis: evolution with time and association with severity of inflammatory activity. Invest Ophthalmol Vis Sci. 2012;53:6171–7.
Kiuchi Y, Okada K, Ito N, Hayashida Y, Fukui K, Ohnishi T, Ishimoto I, Saitoh Y. Effect of a single drop of latanoprost on intraocular pressure and blood-aqueous barrier permeability in patients with uveitis. Kobe J Med Sci. 2002;48:153–9.
Sabzevari A, Adibkia K, Hashemi H, Hedayatfar A, Mohsenzadeh N, Atyabi F, et al. Polymeric triamcinolone acetonide nanoparticles as a new alternative in the treatment of uveitis: in vitro and in vivo studies. Eur J Pharm Biopharm. 2013;84:63–71.
Lafranco Dafflon M, Tran VT, Guex-Crosier Y, Herbort CP. Posterior sub-Tenon’s steroid injections for the treatment of posterior ocular inflammation: indications, efficacy and side effects. Graefes Arch Clin Exp Ophthalmol. 1999;237:289–95.
Strobel J, Stoltenberg I. In vivo examination of aqueous humor for cells and protein in clinically silent anterior uveitis before surgical interventions. Fortschr Ophthalmol. 1990;87:305–7 (in German).
Tran VT, Guex-Crosier Y, Herbort CP. Effect of cataract surgery with intraocular lens implantation on inflammation in chronic uveitis: a longitudinal laser flare photometry study. Can J Ophthalmol. 1998;33:264–9.
Nguyen NX, Kuchle M, Naumann GO. Quantification of blood-aqueous barrier breakdown after phacoemulsification in Fuchs’ heterochromic uveitis. Ophthalmologica. 2005;219:21–5.
Roesel M, Tappeiner C, Heinz C, Koch JM, Heiligenhaus A. Comparison between intravitreal and orbital floor triamcinolone acetonide after phacoemulsification in patients with endogenous uveitis. Am J Ophthalmol. 2009;147:406–12.
Roesel M, Heinz C, Koch JM, Heiligenhaus A. Comparison of orbital floor triamcinolone acetonide and oral prednisolone for cataract surgery management in patients with non-infectious uveitis. Graefes Arch Clin Exp Ophthalmol. 2010;248:715–20.
Küchle M, Nguyen NX, Naumann GO. Aqueous flare following penetrating keratoplasty and in corneal graft rejection. Arch Ophthalmol. 1994;112:354–8.
Nguyen NX, Langenbucher A, Seitz B, Graupner M, Cursiefen C, Küchle M, et al. Blood-aqueous barrier breakdown after penetrating keratoplasty with simultaneous extracapsular cataract extraction and posterior chamber lens implantation. Graefes Arch Clin Exp Ophthalmol. 2001;239:114–7.
Vita RC, Campos M, Belfort Jr R, Paiva ER. Alterations in blood-aqueous barrier after corneal refractive surgery. Cornea. 1998;17:158–62.
Sen HN, Uusitalo R, Laatikainen L. Subclinical inflammation after laser in situ keratomileusis in corneal grafts. J Cataract Refract Surg. 2002;28:782–7.
El-Harazi SM, Chuang AZ, Yee RW. Assessment of anterior chamber flare and cells after laser in situ keratomileusis. J Cataract Refract Surg. 2001;27:693–6.
Pisella PJ, Albou-Ganem C, Bourges JL, Debbasch C, Limon S. Evaluation of anterior chamber inflammation after corneal refractive surgery. Cornea. 1999;18:302–5.
Nguyen NX, Seitz B, Langenbucher A, Küchle M. Quantification of blood-aqueous barrier breakdown after photorefractive keratectomy for myopia. Graefes Arch Clin Exp Ophthalmol. 1999;237:113–6.
Oshika T, Sakurai M, Araie MA. study on diurnal fluctuation of blood-aqueous barrier permeability to plasma proteins. Exp Eye Res. 1993;56:129–33.
Mori M, Sakurai M, Araie M. Topical timolol and blood-aqueous barrier permeability to protein in human eyes. Nihon Ganka Gakkai Zasshi. 1992;96:1418–22 (in Japanese).
Mori M, Araie M. Effect of apraclonidine on blood-aqueous barrier permeability to plasma protein in man. Exp Eye Res. 1992;54:555–9.
Mori M, Sakurai M, Araie M. Effect of topical epinephrine on permeability of blood-aqueous barrier in human eyes. Jpn J Ophthalmol. 1992;36:342–7.
Lindén C, Nuija E, Alm A. Effects on IOP restoration and blood-aqueous barrier after long-term treatment with latanoprost in open angle glaucoma and ocular hypertension. Br J Ophthalmol. 1997;81:370–2.
Diestelhorst M, Roters S, Krieglstein GK. The effect of latanoprost 0.005% once daily versus 0.0015% twice daily on intraocular pressure and aqueous humour protein concentration in glaucoma patients. A randomized, double-masked comparison with timolol 0.5%. Graefes Arch Clin Exp Ophthalmol. 1997;235:20–6.
Martin E, Martinez-de-la-Casa JM, Garcia-Feijoo J, Troyano J, Larrosa JM, Garcia-Sanchez J. A 6-month assessment of bimatoprost 0.03% vs timolol maleate 0.5%: hypotensive efficacy, macular thickness and flare in ocular-hypertensive and glaucoma patients. Eye (Lond). 2007;21:164–8.
Arcieri ES, Pierre Filho PT, Wakamatsu TH, Costa VP. The effects of prostaglandin analogues on the blood aqueous barrier and corneal thickness of phakic patients with primary open-angle glaucoma and ocular hypertension. Eye (Lond). 2008;22:179–83.
Stevens AM, Kestelyn PA, De Bacquer D, Kestelyn PG. Benzalkonium chloride induces anterior chamber inflammation in previously untreated patients with ocular hypertension as measured by flare meter: a randomized clinical trial. 12. Acta Ophthalmol. 2012;90:e221–4.
Küchle M, Nguyen NX, Horn F, Naumann GO. Quantitative assessment of aqueous flare and aqueous ‘cells’ in pseudoexfoliation syndrome. Acta Ophthalmol (Copenh). 1992;70:201–8.
Küchle M, Nguyen NX, Hannappel E, Beck W, Ho ST, Naumann GO. Tyndallometry with the laser flare cell meter and biochemical protein determination in the aqueous humor of eyes with pseudoexfoliation syndrome. Ophthalmologe. 1994;91:578–84 (in German).
Küchle M, Nguyen NX, Hannappel E, Naumann GO. The blood-aqueous barrier in eyes with pseudoexfoliation syndrome. Ophthalmic Res. 1995;27(Suppl 1):136–42.
Nguyen NX, Küchle M, Martus P, Naumann GO. Quantification of blood-aqueous barrier breakdown after trabeculectomy: pseudoexfoliation versus primary open-angle glaucoma. J Glaucoma. 1999;8:18–23.
Ayala M, Chen E. Comparison of selective laser trabeculoplasty (SLT) in primary open angle glaucoma and pseudoexfoliation glaucoma. Clin Ophthalmol. 2011;5:1469–73.
Wang L, Yamasita R, Hommura S. Corneal endothelial changes and aqueous flare intensity in pseudoexfoliation syndrome. Ophthalmologica. 1999;213:387–91.
Moriarty AP, McHugh JD, Spalton DJ, Ffytche TJ, Shah SM, Marshall J. Comparison of the anterior chamber inflammatory response to diode and argon laser trabeculoplasty using a laser flare meter. Ophthalmology. 1993;100:1263–7.
Chiou AG, Mermoud A, Jewelewicz DA. Post-operative inflammation following deep sclerectomy with collagen implant versus standard trabeculectomy. Graefes Arch Clin Exp Ophthalmol. 1998;236:593–6.
Inoue M, Azumi A, Shirabe H, Yamamoto M. Iridopathy in eyes with proliferative diabetic retinopathy: detection of early stage of rubeosis iridis. Ophthalmologica. 1998;212:15–8.
Küchle M, Schönherr U, Nguyen NX, Steinhäuser B, Naumann GO. Quantitative measurement of aqueous flare and aqueous “cells” in eyes with diabetic retinopathy. Ger J Ophthalmol. 1992;1:164–9.
Schalnus R, Ohrloff C. The blood-retina barrier and blood-aqueous humor barrier in type I diabetic patients without retinopathy. Determination of permeability using fluorophotometry and laser flare measurements. Klin Monbl Augenheilkd. 1993;202:281–7 (in German).
Moriarty AP, Spalton DJ, Moriarty BJ, Shilling JS, Ffytche TJ, Bulsara M. Studies of the blood-aqueous barrier in diabetes mellitus. Am J Ophthalmol. 1994;15(117):768–71.
Tost F, Heilmann P, Lautenschläger C. Aqueous flare measurement with a laser flare cellmeter in eyes with diabetic retinopathy. Ophthalmologica. 1995;209:56–9.
Nguyen NX, Schönherr U, Küchle M. Aqueous flare and retinal capillary changes in eyes with diabetic retinopathy. Ophthalmologica. 1995;209:145–8.
Blum M, Müller UA, Höche A, Hunger-Dathe W, Stein G, Strobel J. Flare measurement and albuminuria in type I diabetics. Klin Monbl Augenheilkd. 1998;212:80–3 (in German).
Funatsu H, Yamashita H, Noma H, Mimura T, Yamashita T, Hori S. Increased levels of vascular endothelial growth factor and interleukin-6 in the aqueous humor of diabetics with macular edema. Am J Ophthalmol. 2002;133:70–7.
Jandrasits K, Krepler K, Wedrich A. Aqueous flare and macular edema in eyes with diabetic retinopathy. Ophthalmologica. 2003;217:49–52.
Noma H, Mimura T, Yasuda K, Shimura M. Role of inflammation in diabetic macular edema. Ophthalmologica. 2014;232:127–35.
Moriarty AP, Spalton DJ, Shilling JS, Ffytche TJ, Bulsara M. Breakdown of the blood-aqueous barrier after argon laser panretinal photocoagulation for proliferative diabetic retinopathy. Ophthalmology. 1996;103:833–8.
Kruger AJ, Schauersberger J, Petternel V, Amon M. Inflammation after phacoemulsification in patients with type 2 diabetes mellitus without retinopathy: prospective study. J Cataract Refract Surg. 1999;25:227–32.
Gatinel D, Lebrun T, Le Toumelin P, Chaine G. Aqueous flare induced by heparin-surface-modified poly(methyl methacrylate) and acrylic lenses implanted through the same-size incision in patients with diabetes. J Cataract Refract Surg. 2001;27:855–60.
Sakamoto T, Miyazaki M, Hisatomi T, Nakamura T, Ueno A, Itaya K, et al. Triamcinolone-assisted pars plana vitrectomy improves the surgical procedures and decreases the postoperative blood-ocular barrier breakdown. Graefes Arch Clin Exp Ophthalmol. 2002;240:423–9.
Chalam KV, Malkani S, Shah VA. Intravitreal dexamethasone effectively reduces postoperative inflammation after vitreoretinal surgery. Ophthalmic Surg Lasers Imaging. 2003;34:188–92.
Kiss C, Michels S, Prager F, Weigert G, Geitzenauer W, Schmidt-Erfurth U. Evaluation of anterior chamber inflammatory activity in eyes treated with intravitreal bevacizumab. Retina. 2006;26:877–81.
Nguyen NX, Küchle M. Aqueous flare and cells in eyes with retinal vein occlusion–correlation with retinal fluorescein angiographic findings. Br J Ophthalmol. 1993;77:280–3.
Asano S, Miyake K, Miyake S, Ota I. Relationship between blood-aqueous barrier disruption and ischemic macular edema in patients with branch or central retinal vein occlusion: effects of sub-tenon triamcinolone acetonide injection. J Ocul Pharmacol Ther. 2007;23:577–84.
Noma H, Mimura T, Tatsugawa M, Shimada K. Aqueous flare and inflammatory factors in macular edema with central retinal vein occlusion: a case series. BMC Ophthalmol. 2013;11(13):78. doi:10.1186/1471-2415-13-78.
Noma H, Mimura T, Shimada K. Role of inflammation in previously untreated macular edema with branch retinal vein occlusion. BMC Ophthalmol. 2014;18(14):67.
Veckeneer M, Van Overdam K, Bouwens D, Feron E, Mertens D, Peperkamp E, et al. Randomized clinical trial of cryotherapy versus laser photocoagulation for retinopexy in conventional retinal detachment surgery. Am J Ophthalmol. 2001;132:343–7.
Amann T, Nguyen NX, Küchle M. Tyndallometry and cell count in the anterior chamber in retinal detachment. Klin Monbl Augenheilkd. 1997;210:43–7 (in German).
Chen MS, Chang CC, Ho TC, Tsai TH, Fan IM, Hou PK. Blood-aqueous barrier function in a patient with choroideremia. J Formos Med Assoc. 2010;109:167–71.
Chen MS, Chang CC, Tsai TH, Fan IM, Hou PK. Reticular dystrophy of the retinal pigment epithelium. J Formos Med Assoc. 2007;106:490–4.
Küchle M, Nguyen NX, Schalnus R, Freissler K, Lüchtenberg M, Müller M. Quantification of disorders of the blood-aqueous humor barrier in retinitis pigmentosa–initial results. Klin Monbl Augenheilkd. 1994;204:211–6 (in German).
Küchle M, Nguyen NX, Martus P, Freissler K, Schalnus R. Aqueous flare in retinitis pigmentosa. Graefes Arch Clin Exp Ophthalmol. 1998;236:426–33.
Murakami Y, Yoshida N, Ikeda Y, Nakatake S, Fujiwara K, Notomi S, et al. Relationship between aqueous flare and visual function in retinitis pigmentosa. Am J Ophthalmol. 2015;159:958–63.
Strobbe E, Cellini M, Fresina M, Campos EC. ET-1 plasma levels, aqueous flare, and choroidal thickness in patients with retinitis pigmentosa. J Ophthalmol. 2015;2015:292615. doi:10.1155/2015/292615.
Küchle M, Nguyen NX, Naumann GO. Quantitative assessment of the blood-aqueous barrier in human eyes with malignant or benign uveal tumors. Am J Ophthalmol. 1994;15(117):521–8.
Sanchez E, Klainguti G, Herbort CP. Surgery of strabismus and rupture of the blood-aqueous barrier (ischemia of the anterior segment). Klin Monbl Augenheilkd. 1998;212:296–8 (in French).
Neugebauer A, Kaszli FA, Diestelhorst M, Rüssmann W. Do standard procedures in squint surgery influence the blood-aqueous barrier? Ophthalmic Surg Lasers. 1997;28:570–3.
Kaszli FA, Diestelhorst M, Esser P, Rüssmann W. Disorder of the blood-aqueous barrier after eye muscle operation in children. Ophthalmologe. 1996;93:325–7 (in German).
Abe RY, Zacchia RS, Santana PR, Costa VP. Effects of benzalkonium chloride on the blood-aqueous and blood-retinal barriers of pseudophakic eyes. J Ocul Pharmacol Ther. 2014;30:413–8.
Okamoto F, Sato T, Umebayashi Y, Ohtsuka F, Hommura S. Quantitative evaluation of aqueous flare in psoriasis using a laser flare-cell meter. Nihon Ganka Gakkai Zasshi. 1997;101:487–91 (in Japanese).
Kłysik A, Kozakiewicz M. Blood-aqueous barrier integrity in patients with Graves’ ophthalmopathy (GO), before and after rehabilitative surgery. Eye (Lond). 2015;29:542–51.
Szepessy Z, Barsi A, Németh J. Macular Changes Correlate with the Degree of Acute Anterior Uveitis in Patients with Spondyloarthropathy. Ocul Immunol Inflamm. 2014;20:1–6.
Strobbe E, Cellini M, Campos EC. Aqueous flare and choroidal thickness in patients with chronic hepatitis C virus infection: a pilot study. Ophthalmology. 2013;120:2258–63.
Verbraak FD, Schreinemachers MC, Tiller A, van Deventer SJ, de Smet MD. Prevalence of subclinical anterior uveitis in adult patients with inflammatory bowel disease. Br J Ophthalmol. 2001;85:219–21.
Stjernschantz J, Uusitalo R, Palkama A. The aqueous proteins of the rat in normal eye and after aqueous withdrawal. Exp Eye Res. 1973;16:215–21.
Unger WG, Cole DF, Hammond B. Disruption of the blood-aqueous barrier following paracentesis in the rabbit. Exp Eye Res. 1975;20:255–70.
Nussenblatt RB, Calogero D, Buchen SY, Leder HA, Goodkin M, Eydelman MB. Rabbit intraocular reactivity to endotoxin measured by slit-lamp biomicroscopy and laser flare photometry. Ophthalmology. 2012;119:e19–23.
Hayasaka Y, Hayasaka S, Zhang XY, Nagaki Y. Effects of topical corticosteroids and nonsteroidal anti-inflammatory drugs on prostaglandin e2-induced aqueous flare elevation in pigmented rabbits. Ophthalmic Res. 2003;35:341–4.
Abe T, Hayasaka Y, Zhang XY, Hayasaka S. Effects of intravenous administration of FR122047 (a selective cyclooxygenase 1 inhibitor) and FR188582 (a selective cyclooxygenase 2 inhibitor) on prostaglandin-E2-induced aqueous flare elevation in pigmented rabbits. Ophthalmic Res. 2004;36:321–6.
Ogawa T, Ohara K, Shimizu H, Terai T, Watanabe N. Effects of a combined use of steroidal and nonsteroidal anti-inflammatory drugs on E. coli endotoxin-induced uveitis in pigmented rabbits. Jpn J Ophthalmol. 1995;39:353–9.
Yanagisawa S, Hayasaka S, Zhang XY, Hayasaka Y, Nagaki Y, Kitagawa K. Effect of topical betaxolol on the acute rise of aqueous flare induced by highly selective agonists for prostaglandin E2 receptor subtypes in pigmented rabbits. Ophthalmic Res. 2002;34:48–50.
Chen MS, Chang CC, Lin CP, Wang PC, Lin LR, Hou PK, et al. Role of vascular endothelial growth factor in the breakdown of the blood-aqueous barrier after retinal laser photocoagulation in pigmented rabbits. J Ocul Pharmacol Ther. 2012;28:83–8.
Watanabe K, Hayasaka S, Hiraki S, Matsumoto M, Kadoi C, Nagaki Y. Effects of alpha(2)-adrenergic agonists on lipopolysaccharide-induced aqueous flare elevation in pigmented rabbits. Jpn J Ophthalmol. 2001;45:221–6.
Zhang XY, Hayasaka S, Hayasaka Y, Yanagisawa S, Nagaki Y. Effects of isopropyl unoprostone, latanoprost, and prostaglandin E(2) on acute rise of aqueous flare in pigmented rabbits. Ophthalmic Res. 2002;34:90–3.
Hayasaka Y, Hayasaka S, Zhang XY, Nagaki Y. Effects of topical antiglaucoma eye drops on prostaglandin E(2)-induced aqueous flare elevation in pigmented rabbits. Invest Ophthalmol Vis Sci. 2002;43:1142–5.
Kitagawa K, Hayasaka S, Watanabe K, Nagaki Y. Aqueous flare elevation induced by transcorneal application of highly selective agonists for prostaglandin E2 receptor subtypes in pigmented rabbits: effect of tetramethylpyrazine. Prostaglandins Other Lipid Mediat. 2001;65:189–98.
Hiraki S, Zhang XY, Hayasaka S. Effects of a nitric oxide synthase inhibitor on prostaglandin-induced aqueous flare elevation in pigmented rabbits. Ophthalmic Res. 1996;28:260–4.
Kosaka T, Mishima HK, Kiuchi Y, Kataoka K. The effects of prostaglandins on the blood-ocular barrier. Jpn J Ophthalmol. 1995;39:368–76.
Nagaki Y, Hayasaka S, Zhang XY, Hayasaka Y, Nakamura N, Terasawa K. Effects of topical instillation of traditional herbal medicines, herbal extracts, and their components on prostaglandin E2-induced aqueous flare elevation in pigmented rabbits. Jpn J Ophthalmol. 2003;47:249–53.
Kitagawa K, Hayasaka S, Nagaki Y, Watanabe K. Effects of tetramethylpyrazine on prostaglandin E(2)- and prostaglandin E(2) receptor agonist-induced disruption of blood-aqueous barrier in pigmented rabbits. Jpn J Ophthalmol. 2001;45:227–32.
Nakamura-Shibasaki M, Latief MA, Ko JA, Funaishi K, Kiuchi Y. Effects of topical adrenergic agents on prostaglandin E2-induced aqueous flare and intraocular pressure elevation in pigmented rabbits. Jpn J Ophthalmol. 2016;60:95–102.
Chen J, Fujino Y, Takahashi T. Experimental uveitis induced by intravitreal or intravenous lipoteichoic acid in rabbits. Jpn J Ophthalmol. 1999;43:368–74.
Inoue M, Shirabe H, Yamamoto M. Blood-aqueous barrier disruption in experimental anterior segment ischemia in rabbit eyes. Ophthalmic Res. 1999;31:213–9.
Inoue M, Tsukahara Y, Shirabe H, Yamamoto M. Disruption of the blood-aqueous barrier following retinal laser photocoagulation and cryopexy in pigmented rabbits. Ophthalmic Res. 2001;33:37–41.
Sawa M, et al. Ganka Rinsho Iho. 1993; 87: 2648–59. (in Japanese).
Zeng L, Sawa M, et al. Rinshoganka. 1990; 44: 1997–2001. (in Japanese).
Acknowledgements
The author thanks Dr. Yutaka Iwasaki for his generous support to prepare figures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
M. Sawa, Editor-in-Chief of Jpn J Ophthalmol, Inventor of Laser Flare-Cell Photometer.
About this article
Cite this article
Sawa, M. Laser flare-cell photometer: principle and significance in clinical and basic ophthalmology. Jpn J Ophthalmol 61, 21–42 (2017). https://doi.org/10.1007/s10384-016-0488-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-016-0488-3