Abstract
TDP-43 is present in a high proportion of aged brains that do not meet criteria for frontotemporal lobar degeneration (FTLD). We determined whether there are distinct TDP-43 types in non-FTLD brains. From a cohort of 553 brains (Braak neurofibrillary tangle (NFT) stage 0–VI), excluding cases meeting criteria for FTLD, we identified those that had screened positive for TDP-43. We reviewed 14 different brain regions in these TDP-43 positive cases and classified them into those with “typical” TDP-43 immunoreactive inclusions (TDP type-α), and those in which TDP-43 immunoreactivity was adjacent to/associated with NFTs in the same neuron (TDP type-β). We compared pathological, genetic (APOE4, TMEM106B and GRN variants), neuroimaging and clinical data between types, as well as compared neuroimaging between types and a group of TDP-43 negative cases (n = 309). Two-hundred forty-one cases were classified as TDP type-α (n = 131, 54%) or TDP type-β (n = 110, 46%). Type-α cases were older than type-β at death (median 89 years vs. 87 years; p = 0.02). Hippocampal sclerosis was present in 78 (60%) type-α cases and 16 (15%) type-β cases (p < 0.001). Type-α cases showed a pattern of widespread TDP-43 deposition commonly extending into temporal, frontal and brainstem regions (84% TDP-43 stage 4–6) while in type-β cases deposition was predominantly limbic, located in amygdala, entorhinal cortex and subiculum of the hippocampus (84% TDP-43 stages 1–3) (p < 0.001). There was a difference in the frequency of TMEM106B protective (GG) and risk (CC) haplotypes (SNP rs3173615 encoding p.T185S) in type-α cases compared to type-β cases (GG/CG/CC: 8%/42%/50% vs. 24%/49%/27%; p = 0.01). Type-α cases had smaller amygdala (− 10.6% [− 17.6%, − 3.5%]; p = 0.003) and hippocampal (− 14.4% [− 21.6%, − 7.3%]; p < 0.001) volumes on MRI at death compared to type-β cases, although both types had smaller amygdala and hippocampal volumes compared to TDP-43 negative cases (− 7.77%, − 21.6%; p < 0.001). These findings demonstrate that there is distinct heterogeneity of TDP-43 deposition in non-FTLD brains.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
TAR DNA binding protein of 43 kDa (TDP-43) was first reported to be associated with frontotemporal lobar degeneration (FTLD) and amyotrophic lateral sclerosis in 2006 [2, 41]. It was later shown to be present in 17–74% of brains of aged individuals and those with Alzheimer’s disease [1, 3, 4, 9, 12, 14, 29, 35, 56]. Today, TDP-43 has become important to our understanding of age-related cognitive impairment and Alzheimer’s disease [15, 21]. TDP-43 has been shown to be associated with the presence of dementia, more severe cognitive impairment in general at death and overtime and loss of episodic memory even after accounting for other pathologies that have also been linked to these clinical features [5, 24,25,26, 57]. TDP-43 has been shown to be associated with smaller hippocampal volumes and faster rates of hippocampal atrophy in aging and Alzheimer’s disease [17, 24, 26]. TDP-43 has also been linked to the apolipoprotein epsilon e4 allele (APOE ε4) [15, 53, 58] and the transmembrane protein 106B (TMEM106B) [46] in aging and Alzheimer’s disease.
Over a decade ago we observed some cases with unusual histological characteristics of TDP-43 deposition in aging and Alzheimer’ disease [1]. Most commonly, we observed cases of TDP-43 deposition with morphological features that were “Typical” and similar to those described in one type of FTLD [1]. Specifically, we noted the presence of TDP-43 immunoreactive neuronal cytoplasmic inclusions, neuronal intranuclear inclusions and dystrophic neurites (Fig. 1) throughout different brain regions that are characteristic of FTLD-TDP type-A [32]. We did not appreciate characteristic features of any of the other FTLD-TDP types [30, 32] at that time. Interestingly, we also noted cases where TDP-43 deposition appeared distinct. In these cases we observed TDP-43 immunoreactivity that was located adjacent to tau-immunoreactive neurofibrillary tangles (NFTs) (Fig. 1) [1]. We showed with confocal microscopy that in such cases there was indeed co-localization of TDP-43 with phosphorylated tau [1]. In addition, with electron microscopy, we showed that TDP-43 immunoreactivity was associated with tightly bundled, paired helical filaments [1]. Interestingly, in these cases, often we did not see some of the classic features of TDP-43 that is characteristic of FTLD-TDP type-A, such as neuronal intranuclear inclusions. To date, it remains unknown whether our observations are consistent with true heterogeneity of TDP-43 deposition in non-FTLD brains, i.e. whether there are at least two distinct TDP-43 types; one with typical inclusions, and the other linked to the presence of NFTs.
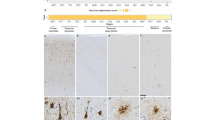
The pathological features of TDP-43 including those of TDP-43 type-β in which TDP-43 immunoreactivity is associated with neurofibrillary tangles. a Pick body-like, dense neuronal cytoplasmic inclusions (NCI) in perirhinal cortex, layer II. b NCI and neurites in amygdala; inset (asterisk) shows TDP-43 deposits co-mingled with neurofibrillary tangle (see also cells with asterisks in c and e). c Perivascular (v capillary) globular astrocytic inclusion (arrow) in amygdala. d Spindly neurites in subpial space (ca corpora amylacea in glia limitans astrocytes). e Pleomorphic TDP-43 pathology in amygdala. f Skein-like NCI and perivascular globular inclusion (arrow, v capillary). g Pleomorphic neuronal and neuritic pathology in amygdala
In this study, we determine whether there is evidence supporting the notion that there may indeed be at least two distinct pathological types of TDP-43 deposition in non-FTLD brains. We hypothesize that there would be evidence supporting the existence of two distinct pathological types.
Methods
Subjects
We identified all cases in a consecutive autopsy series of prospectively enrolled participants in the Mayo Clinic Alzheimer’s Disease Research Center, Mayo Clinic Alzheimer’s Disease Patient Registry, or the Mayo Clinic Study of Aging, that had a brain autopsy, between Jan 1, 1999, and Dec 31, 2012 and had been screened for the presence of TDP-43 in the amygdala and/or hippocampus. Individuals with a pathological diagnosis of any type of FTLD were excluded [8, 33]. That is, cases where degeneration of the frontal and/or temporal lobes, either on gross examination or histologically showing microvacuolation, neuronal loss and astrogliosis most prominent in laminar II or transcortical, were the most characteristic feature. From a total of 553 non-FTLD cases that had been screened for TDP-43, a total of 244 cases had screened positive, and 309 (which we will refer to as controls for this study) had screened negative.
Pathological analysis
All 553 cases and controls had undergone pathological examination according to the recommendations of the Consortium to Establish a Registry for Alzheimer’s disease (CERAD) [37], and each individual had been assigned a Braak NFT stage 0–VI [6] and a CERAD neuritic plaque stage 0–3 [37]. The presence of Lewy bodies in amygdala, brainstem regions or neocortical regions was documented, as was the presence of vascular lesions (micro-infarcts, lacunar infarcts (< 1 cm), large infarcts (≥ 1 cm) and amyloid angiopathy). Hippocampal sclerosis (HpScl) was diagnosed on the basis of neuronal loss in the subiculum and CA1 regions of the hippocampus out of proportion to the observable burden of extracellular NFT pathology, according to consensus recommendations [10, 43]. The 244 cases that had screened positive for TDP-43 had additional brain sections screened for the presence/absence of TDP-43 and as a result were rendered a TDP-43 stage of 1–6 [19, 20]. To accomplish staging, we screened amygdala, entorhinal cortex, subiculum, dentate gyrus of the hippocampus, occipitotemporal cortex, inferior temporal cortex, basal forebrain, insula, ventral striatum, basal ganglia, superior middle frontal cortex, substantia nigra, midbrain tectum and inferior olive [19, 20]. Staging was rendered as follows: stage 1 = TDP-43 deposition was limited to the amygdala; stage 2 = TDP-43 extending into the subiculum or entorhinal cortex; stage 3 = extension into the dentate gyrus of the hippocampus or occipitotemporal cortex; stage 4 = extension into basal forebrain, insula, ventral striatum or inferior temporal cortex; stage 5 = extension into brainstem regions (substantia nigra, midbrain tectum or inferior olive) and stage 6 = extension into basal ganglia or frontal cortex. The TDP-43 staging scheme has been validated [48].
This study was approved by the Mayo Clinic Institutional Review Board. Before death, all participants or their proxies had provided written consent for brain autopsy examination.
Determination of TDP-43 type
To determine TDP-43 type we reviewed the morphological features of TDP-43 in all regions where TDP-43 was present (i.e. range: 1–14 regions) in all 244 cases [19, 20]. Cases were categorized into those with typical, well-described, TDP-43 inclusions such as neuronal cytoplasmic inclusions, neuronal intranuclear inclusions and dystrophic neurites, without NFT-associated TDP-43 in any region (designated TDP-43 type-α) and those where TDP-43 was observed adjacent to NFTs in multiple cells in at least 1 brain region screened (designated TDP-43 type-β) (Fig. 1). We chose to designate the two variants as type-α and type-β to avoid confusion with the TDP-43 type 1, 2, 3 and TDP-43 type A–E typing of FTLD-TDP [8] and to be specific for TDP-43 deposition in non-FTLD brains. Categorization was based on consensus of three experts in degenerative neuropathology (DWD, KAJ, MEM).
Clinical and MRI outcomes
The 244 cases were categorized as cognitively unimpaired, having mild cognitive impairment [42], dementia of the Alzheimer’s type [36], or a non-Alzheimer’s dementia at the last visit before death. For each participant, determination of cognitively normal status was based on consensus agreement between the study coordinator, examining physician and neuropsychologist who evaluated the participant, taking into account education, prior occupation, visual or hearing deficits and reviewing all other participant clinical information. Clinical measures that were captured during life included the Mini-Mental State Examination [11], Clinical Dementia Rating Scale Sum of Boxes [34], Wechsler Memory Scale-Revised Logical Memory II [52], Boston Naming Test [28], Control Oral word Association Test [31], Wechsler Adult Intelligence Scale-R Block Design [52], Auditory Verbal Learning Test (AVLT) Delayed Recall [44] and Trail Making Test Parts A and B [47]. Of the 244 cases, 187 had completed a volumetric head MRI during life. If more than one MRI had been performed, we analyzed the MRI closest to death. Regional volumes were calculated using FreeSurfer version 5.3.0. We assessed volumes of the amygdala, hippocampus, lateral temporal cortex (inferior, middle, superior temporal cortex), parietal (inferior parietal, supramarginal and isthmus cingulate cortex), and lateral superior frontal cortex, and outputted total intracranial volume (TIV) to allow for the correction of head size [54].
Genetic analyses
Genotypes of the two variants in the TMEM106B and GRN genes were generated by the Taqman method (Invitrogen, Carlsbad, CA, USA). Assays were ordered for rs3173615 (TMEM106B) and rs5848 (GRN) and genotyping was performed following manufacturer’s protocol on 133 cases with available DNA using a QuantStudio 7 and Taqman Genotyper software 1.4.0. APOE genotyping was performed for all 244 cases as previously described [9].
Statistical analysis
We compared demographic, pathological, clinical, genetic and neuroimaging variables between TDP-43 type-α and TDP-43 type-β variants using Fisher’s Exact or Wilcoxon rank sum tests where appropriate. We also fitted a linear regression on log-transformed volume adjusting for age at death, TIV and Braak stage to assess for relationships between volumes and TDP-43 type (type-α and type-β), as well as between volumes and TDP-43 type vs. the 309 control cases without TDP-43 (i.e. TDP-43 negative cases or TDP-43 stage 0).
In order to predict TDP status we fitted a three-category multinomial logistic regression model to estimate the odds of TDP negative (stage 0) vs. type-α vs. type-β using four predictors: age at death, total intracranial volume, amygdala volume and hippocampal volume.
All analyses were performed with R statistical software version 3.1.2 (R Foundation for Statistical Computing, Vienna, Austria). p < 0.05 was considered statistically significant.
Results
Of the 244 cases with TDP-43, 131 (54%) met criteria for TDP-43 type-α and 110 (46%) met criteria for TDP-43 type-β. Three cases with TDP-43 immunoreactive inclusions were excluded from further analysis as they were found to have predominantly long, thick dystrophic neurites in the entorhinal cortex and subiculum of the hippocampus, reminiscent of FTLD-TDP type-C pathology [32]. There were no significant differences in demographic features between both groups except for age at death where those with TDP-43 type-α were slightly older on average than those with type-β (median [range] of 89 [70,104] years vs. 87 [56,105] years) (p = 0.02) (Table 1).
Pathological findings
Those designated TDP-43 type-α were much more likely to have HpScl (60%) compared to those designated TDP-43 type-β (15%) (p < 0.001) and were more likely to have a higher TDP-43 stage compared to those with TDP-43 type-β (type-α = 84% stage 4–6 vs. type-β = 84% stage 1–3; p < 0.001) (Table 2). As an example, there was a 25-fold higher odds of TDP-43 type-β in TDP-43 stage 1 compared to higher stages (95% CI 7–158) (p < 0.0001). In contrast, there was a 22-fold higher odds of TDP-43 type-α in TDP-43 stage 6 vs. lower stages (95% CI 5–400) (p = 0.003). There were only two TDP type-α cases with amygdala only TDP-43 (stage 1). At the other end of the staging scheme, there was only one TDP-43 type-β case with frontal lobe or basal ganglia involvement (stage 6) (Fig. 2). TDP-43 type-β pathology only involved the inferior temporal lobe in a single case. That is, although type-β cases showed TDP-43 extended to stages 4–6 in 18 cases, only one case (6%) showed TDP-43 involvement of the inferior temporal lobe. This was distinctly different from type-α cases where the inferior temporal lobe was affected in 50% of cases at stages 4–6. We also observed that all five cases that did not conform to the staging scheme and instead showed “skipped regions” had been classified as TDP-43 type-β (Fig. 2). These cases did not conform due to involvement of the inferior olive without involvement of regions in the preceeding two stages. Figure 3 shows mosaic plots—graphical representation of two-way tables where the area of each “tile” depends on the number of individuals in a subgroup—that highlight the strong relationships between TDP-43 type and both HpScl and TDP-43 stage, showing that cases with HpScl, and cases with high TDP-43 stages, are more likely to be TDP-43 type-α. Although there was a difference in Braak NFT stage (TDP-43 type-β was associated with higher Braak NFT stage compared to TDP-43 type-α; p = 0.03), Fig. 3 shows that for Braak stages IV–VI, which accounted for 91% of the data in TDP-43 type-α and 99% in TDP-43 type-β, there was no difference in types. There were no differences in the frequency of Lewy bodies, neuritic plaques or vascular disease between TDP-43 type-α and type-β.
Mosaic plots shows the percentages of hippocampal sclerosis (HpScl), TDP-43 and Braak NFT stages by TDP-43 type. These plots are graphical representations of two-way tables where the area of a “tile” is related to the number of individuals in the table cell. The figure demonstrates the striking association of TDP-43 type-α with HpScl and higher TDP-43 stage. For Braak NFT stages, there is no difference between TDP-43 type-α and type-β at Braak NFT stages IV–VI where > 90% of data exists. Orange color represents TDP-43 type-α; blue color represents TDP-43 Type-β
Clinical and imaging findings
We found no significant differences in the final clinical diagnoses rendered before death, or on clinical measures between TDP-43 type-α and type-β cases, although there was a trend for TDP-43 type-α cases to perform more poorly on the AVLT delayed recall measure and on the Trail Making Test B compared to TDP-43 type β cases (300 s vs. 240 s; p = 0.07) (Table 3). On neuroimaging, TDP-43 type-α had significantly smaller hippocampal volumes (4.7 [1.6, 8.1] vs. 5.2 [3.1, 7.9]; p = 0.005), compared to TDP-43 type β cases; however, TDP-43 type β cases had smaller parietal (37.1 [25.7, 52.2] vs. 38.9 [23.8, 52.6]; p = 0.01) and lateral superior frontal (31.8 [23.4, 42.5] vs. 32.2 [21.5, 46.4]; p = 0.04) volumes (Table 3). However, after adjusting for age at death, TIV, and Braak stage, we found TDP-43 type-α cases to have significantly smaller hippocampal and amygdala volumes compared to TDP-43 type-β cases. Our adjusted analysis showed that cases with TDP-43 type-α had an estimated − 10.6% (− 3.5%, − 17.6%) lower amygdala volume compared to type-β (p = 0.003) (Fig. 4). For hippocampus, cases with TDP-43 type-α had − 14.4% (− 7.3%, − 21.6%) lower volume than TDP-43 type-β (p < 0.001).
We investigated the neuroimaging data further by fitting linear regressions adjusting for age at death, total intracranial volume, and Braak stage. Our analysis showed that cases with TDP-43 type-α had an estimated − 11% (− 4%, − 18%; p = 0.003) smaller amygdala volume than TDP-43 type-β cases. For hippocampi, TDP-43 type-α cases had − 14% (− 7%, − 22%; p < 0.001) smaller volume than TDP-43 type-β cases. Plotted values are regression estimates and 95% confidence intervals
Given that we have previously identified a difference in hippocampal and amygdala volumes between TDP-43 positive and TDP-43 negative cases with TDP-43 positive cases having smaller volumes, and now find TDP-43 type-α cases to have smaller volumes than TDP-43 type-β cases, we compared both TDP-43 types (type-α and type-β) to the group of 309 cases without TDP-43. We found smaller amygdala volumes in both TDP-43 type-α and type-β cases compared to TDP-43 negative controls (type-α = − 18.4% [− 23.0%, − 13.8%] and type-β = − 8.40% [− 13.4%, − 3.44%]; p < 0.001), with similar results for hippocampal volumes for both TDP-43 type-α (− 21.6% [− 26.0%, − 17.1%]; p < 0.001) and type-β (− 7.77 [− 12.6%, − 2.96%]; p = 0.002) (Fig. 5; Table 4). We also found TDP-43 type-α cases to have smaller lateral temporal lobe volumes (− 5.59% [− 8.90%, − 2.28%]; p < 0.001) and TDP-43 type-β cases to have smaller superior frontal lobe volumes (− 5.37 [− 8.91%, − 1.83%]; p = 0.003) compared to TDP-43 negative controls.
An analysis between 309 TDP-43 negative cases (reference group) and TDP-43 type-α and type-β. After adjusting for age at death, TIV, and Braak stage, we found volume differences in the amygdala and hippocampus between both TDP-43 type-α and type-β cases, lateral temporal for type-α and superior frontal for type-β compared to TDP-43 negative cases. Plotted values are regression estimates and 95% confidence intervals
Genetic findings
There was no difference in APOE allele frequency or the GRN rs5848 haplotype frequencies between type-α and type-β (Table 5). There was, however, a difference in the TMEM106B rs3173615 haplotype frequencies. Specifically, there was a difference in the frequency of TMEM106B protective (GG) and risk (CC) haplotypes (single nucleotide polymorphism, SNP, rs3173615 encoding p.T185S) in type-α cases compared to type-β cases (GG/CG/CC: 8%/42%/50% vs. 24%/49%/27%; p = 0.01).
Predicting TDP-43 status and type
Figure 6 shows the relationships between age, amygdala volume and hippocampal volumes as predictors of TDP-43 status and type. We found that a 10-year increase in age was associated with a 1.8-fold increase in the odds of type-α relative to being TDP-43 negative (95% CI 1.3–2.5; p < 0.001) and a 1.6-fold increase in the odds of type-α relative to being type-β (95% CI 1.1–2.3; p = 0.02). Relative to those who were TDP-43 negative, lower amygdala and lower hippocampal volumes were associated with increased odds of both types-α and type-β. Specifically, a 0.5 cm3 decrease in amygdala volume was associated with 1.7-fold increase in the odds of type-α vs. TDP-43 negative (95% CI 1.0–2.8; p = 0.04) and a 1.9-fold increase in the odds of type-β vs. TDP-43 negative (95% CI 1.2–3.1; p = 0.01). In terms of hippocampal volume, a 1 cm3 decrease in hippocampal volume was associated with a 2.7-fold increase in the odds of type-α relative to TDP-43 negative (95% CI 1.8–4.0; p < 0.001) and a 1.5-fold increase in the odds of type-β vs. TDP-43 negative (95% CI 1.0–2.1; p = 0.03). Further, the same decrease of 1 cm3 in the hippocampus was associated with a 1.8-fold increase in the odds of type-α relative to type-β (95% CI 1.2–2.9; p = 0.008).
The relationships between age at MRI scan and amygdala volume (left plot), age at MRI scan and hippocampal (HP) volume (middle plot) and hippocampal volume and amygdala volume (right plot). TDP-43 negative cases are shaded grey, TDP-43 type-α cases are shaded red and TDP-43 type-β cases are shaded blue. The plots show that in general for a given age those with the smallest amygdala and hippocampal volumes are TDP-43 type-α. It also shows that there is strong correlation between amygdala and hippocampal volumes
Discussion
In this study we assess whether there is heterogeneity in TDP-43 deposition in non-FTLD brains. We found significant pathological differences in HpScl and TDP-43 stage, and significant genetic difference in TMEM106B haplotype frequencies, between TDP-43 type-α and type-β cases. We also found type-α cases to be associated with smaller hippocampi and amygdala volumes compared to type-β cases and both types to have smaller volumes (amygdala and hippocampi), compared to TDP-43 negative cases.
The FTLDs are classified first by abnormal protein deposition and then sub-classified within protein category. FTLD-TDP has been shown to have five different types (type A–E) [30, 32], each with distinct histological features and with some types having distinct clinical and neuroimaging characteristics. We now show that, similar to FTLD-TDP, non-FTLD TDP-43 deposition also has distinct types. TDP-43 type-α is pathologically characterized by the presence of neuronal cytoplasmic inclusions, dystrophic neurites, neuronal intranuclear inclusions, and more widespread TDP-43 deposition, with the majority of cases having frontotemporal neocortex deposition. Furthermore, HpScl was a feature of TDP-43 type-α. Therefore, TDP-43 type-α is pathologically similar to FTLD-TDP type A [32]; although the burden of TDP-43 deposition is distinctly less in non-FTLD cases (personal observation). TDP-43 type-β is pathologically distinct from type-α with the main pathological characteristic being TDP-43 co-localized with NFTs in the same neuron. It is, therefore, not surprising that TDP-43 type-β was uncommon in cases of low Braak stage. Out of 110 type-β cases, 109 (99%) occurred in cases with Braak stage IV–VI. Therefore, TDP-43 type-β is much more likely to be identified in cases of high probability Alzheimer’s disease [13] where there is extensive NFT distribution. With-that-said, there is no evidence that TDP-43 type-β is more strongly associated with Alzheimer’s disease. In fact, both types were equally associated with a high frequency of the APOE ε4 genotype, and there was no difference in the CERAD neuritic plaque stage between them. It is certainly possible that type-α and type-β lie on a spectrum, but it appears less likely to be the situation that one type is a precursor to the other type, particularly given that we have also identified genetic differences between them.
Single nucleotide polymorphisms (SNP) in TMEM106B are associated with an increased risk of FTLD-TDP [50]. The SNP rs3173615, which is in linkage disequilibrium with SNP rs1990622, dictates a change from threonine to serine at position 185 (p.T185S). It has been shown that subjects with FTLD-TDP are less likely to have two copies of the minor TMEM106B allele (GG haplotype) compared to cognitively normal controls (6% GG in FTLD-TDP vs. 19% GG in normal controls) [49]. Hence, the GG haplotype is considered protective. In our aged non-FTLD cohort, we found the TMEM106B GG haplotype frequency to be 8% in type-α cases, similar to the frequency reported in FTLD-TDP, and 24% in type-β cases, similar to the frequency reported in normal controls. This finding further supports TDP-43 type-α being related to FTLD-TDP, and TDP-43 type-α and β being distinct. Future genetic studies are needed to compare TMEM106B haplotype frequency across the FTLD-TDP types and TDP-43 type-α and type-β.
We found three cases with TDP-43 that did not conform to either type-α or type-β designation. In these three cases, TDP-43 deposition was characterized by the presence of long, thick dystrophic neurites in the neocortex and Pick body-like inclusions in the dentate gyrus of the hippocampus. These features were identical to the features that characterizes FTLD-TDP type C [22, 32] which is strongly associated with a clinical diagnosis of semantic dementia [40]. None of our three cases were clinically diagnosed with frontotemporal dementia, including semantic dementia. Therefore, whether such cases represent a third variant of TDP-43 in non-FTLD brains or whether they are cases of FTLD-TDP type C remains to be determined. We did not come across cases with TDP-43 pathological features similar to those described in FTLD-TDP type B, D or E pathology. FTLD-TDP type B has many TDP-43 immunoreactive pre-inclusions and is strongly associated with clinical motor neuron disease [18, 22, 32] and genetically with C9ORF72 mutations [38]. FTLD-TDP type-D is associated with TDP-43 immunoreactive neuronal intranuclear inclusions only, inclusion body myopathy, Paget disease of the bone and mutations in the valosin-containing protein gene [51]. FTLD-TDP type E is associated with TDP-43 immunoreactive granulofilamentous neuronal inclusions, abundant grains, and oligodendroglia inclusions and a very rapid clinical course [30]. Both FTLD-TDP type D and type E are extremely rare entities and none of the accompanying features of FTLD-TDP type B, D, E (motor neuron disease, myopathy, Paget’s disease of the bone and rapid course) were a feature of any of our cases. Hence, it is no surprise that we did not identify any such cases in our cohort.
One of the most striking differences that we observed between TDP-43 type-α and type-β was in the distribution of TDP-43. We observed that 85% of the type-α cases had widespread TDP-43 deposition beyond limbic areas which differed from type-β cases where approximately 85% of the cases showed a limbic-predominant pattern. In fact, we found a difference even when both types were stage 4. Specifically, while stage 4 type-α cases commonly involved the inferior temporal lobe, the inferior temporal lobe was never involved in any stage 4 type-β cases. In fact, the inferior temporal lobe was only one involved in 1 stage 4–6 type-β case. This is strong evidence for type-β to truly have an association with limbic regions while type-α shows more of an association with the fronto-temporal neocortices and hence should not be considered limbic predominant.
Another difference we observed was with HpScl that was found to be a feature of TDP-43 type-α, not so much type-β. This is a significant finding given that HpScl has been intricately linked to TDP-43 [1, 24, 26, 39]. Interestingly, HpScl is most common in FTLD-TDP type A compared to FTLD-TDP type B and FTLD-TDP type C [22] although it does occur in cases with frontotemporal dementia with motor neuron disease [16] which typically shows FTLD-TDP type B pathology [22]. Many studies have struggled with separating the effects of TDP-43 from the effects of HpScl and have had to perform mediation analyses to try to do so [17, 26, 39, 58], similar to comparing TDP-43 cases with and without HpScl. The findings from this study support an argument for all these prior studies basically separating the effect of TDP-43 type-α from the effect of TDP-43 type-β. We, therefore, propose that future studies conduct TDP-43 typing, and assess for the effect of each type, instead of, or in addition to, conducting mediation analyses. While it is likely that HpScl is playing a role in explaining why TDP-43 type-α cases have smaller hippocampal volumes than TDP-43 type-β cases, it does not explain the difference observed in amygdala volume. It is possible that the more severe medial temporal volume loss in TDP-43 type-α could also be due to differences in TDP-43 burden or biology. For the latter, it is possible that there is a difference in the ratio of TDP-43 species (TDP-43 fragments vs. full-length TDP-43) in type-α vs. type-β with species having variable effects on neuronal loss and atrophy [59].
We present evidence that the regional volumetric differences observed in this study could be useful to help predict TDP-43 status and type during life. We found that older age and smaller volumes of the amygdala and hippocampus all helped predict TDP-43 positivity, although hippocampal volumes performed the best when also predicting TDP-43 type and, therefore, could prove to be the most useful biomarker. In our cohort, cases with 1 cm3 smaller hippocampi were almost twice as likely to be type-α as type-β. Given our TMEM106B haplotype findings, it is possible that prediction could be further improved when considering both hippocampal volumes and TMEM106B haplotype.
In our previous reports demonstrating a pattern of possible TDP-43 spread, i.e. TDP-43 staging scheme, we observed some cases that did not fit the TDP-43 staging scheme [19, 20]. In these non-conforming cases we noted that there were skipped regions (i.e. there were no involvement of regions from stages 3 or 4, yet there was TDP-43 deposition in the inferior olive but no other region from stage 5). We have now identified such cases as being TDP-43 type-β. The reason for involvement of the inferior olive without involvement other stage 3–5 regions is unclear, but these may be unique cases that should be investigated further. It is possible that involvment of the medulla in some cases reflects a different process.
There was a trend for TDP-43 type-α cases to perform poorer on the Trail-Making-Test B, a test of executive function, which would suggest that TDP-43 type-α may be having a greater effect on frontal systems. However, this observation requires further analysis since it was only trend level significance and since TDP-43 type-β cases, not type-α cases, showed a stronger association with smaller frontal lobe volumes compared to controls. We only found a trend for difference in one of our measures of loss of episodic memory between TDP-43 type-α and type-β cases. It likely did not reach significance since cases with TDP-43 type-α and type-β performed at floor level, with average test scores of 0 on both measures of episodic memory loss. This would support the notion that although the presence of TDP-43, independent of type, is strongly associated with loss of episodic memory, TDP-43 type-α may have a stronger association with episodic memory loss.
For years the field has struggled with the question of whether TDP-43 depositing in the brains of old-age individuals, particularly those with high TDP-43 stage, should be considered FTLD-TDP. Currently, a diagnosis of FTLD requires evidence of a pathological process targeting the frontal and temporal lobes out of proportion to the rest of the brain. In fact, FTLD is pathologically diagnosed when there is gross and histological evidence of focal/distinct degeneration of the frontal and/or temporal lobes [8, 33]. We did not find gross or histological evidence of a lobar degeneration of the frontal and temporal lobes in these cases, and the deposition of TDP-43 (type-α and type-β) was much less than what is typically observed in FTLD-TDP. We did find TDP-43 type-α and type-β to be associated with frontal and temporal lobe atrophy, but this was not striking, and amygdala and hippocampal atrophy was much more striking. We have also previously shown that even cases with high TDP-43 stage do not show clinical features of FTLD [20, 27] or much frontal and temporal atrophy on MRI [20] which is in sharp contrast to the striking degree of frontotemporal atrophy observed in FTLD-TDP cases [45, 55]. All these findings would not support TDP-43 type-α and type-β being an FTLD. With-that said, we are aware of FTLD cases that are not associated with striking and predominant frontal and temporal lobe atrophy or degeneration on pathology [23]. Furthermore, mutations in the C9ORF72 gene, for example, can be associated with FTLD-TDP pathology with little-to-no frontotemporal atrophy [40], and mutations in the GRN gene can be associated with FTLD-TDP with prominent parietal lobe atrophy [40]. Alzheimer’s disease has been shown to be associated with prominent frontal lobar degeneration [7]. Therefore, maybe the real issue is that the phenotype of FTLD needs to be readdressed.
Another question the field has struggled with is whether TDP-43 in non-FTLD brains is biologically similar to or different from the TDP-43 in FTLD-TDP. This study was not designed to, and hence cannot answer this biological question. What we can say is that there were morphological similarities between FTLD-TDP type-A and TDP-43 type-α that are not obviously distinguishable, at least at the light microscopic level. What is next needed to address this question are molecular, biochemical and additional genetic analyses of TDP-43 type α, type β and the FTLD-TDP types.
In summary, in this study we show that there are distinct TDP-43 variants in non-FTLD brains, including those with Alzheimer’s disease. Further investigation is now needed to determine whether there are biochemical differences between both types and how these two types are associated with FTLD-TDP.
References
Amador-Ortiz C, Lin WL, Ahmed Z, Personett D, Davies P, Duara R et al (2007) TDP-43 immunoreactivity in hippocampal sclerosis and Alzheimer’s disease. Ann Neurol 61:435–445. https://doi.org/10.1002/ana.21154
Arai T, Hasegawa M, Akiyama H, Ikeda K, Nonaka T, Mori H et al (2006) TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun 351:602–611. https://doi.org/10.1016/j.bbrc.2006.10.093
Arai T, Mackenzie IR, Hasegawa M, Nonoka T, Niizato K, Tsuchiya K et al (2009) Phosphorylated TDP-43 in Alzheimer’s disease and dementia with Lewy bodies. Acta Neuropathol 117:125–136. https://doi.org/10.1007/s00401-008-0480-1
Arnold SJ, Dugger BN, Beach TG (2013) TDP-43 deposition in prospectively followed, cognitively normal elderly individuals: correlation with argyrophilic grains but not other concomitant pathologies. Acta Neuropathol 126:51–57. https://doi.org/10.1007/s00401-013-1110-0
Boyle PA, Yang J, Yu L, Leurgans SE, Capuano AW, Schneider JA et al (2017) Varied effects of age-related neuropathologies on the trajectory of late life cognitive decline. Brain 140:804–812. https://doi.org/10.1093/brain/aww341
Braak H, Braak E (1991) Neuropathological staging of Alzheimer-related changes. Acta Neuropathol 82:239–259
Brun A (1987) Frontal lobe degeneration of non-Alzheimer type. I. Neuropathology. Arch Gerontol Geriatr 6:193–208
Cairns NJ, Bigio EH, Mackenzie IR, Neumann M, Lee VM, Hatanpaa KJ et al (2007) Neuropathologic diagnostic and nosologic criteria for frontotemporal lobar degeneration: consensus of the Consortium for Frontotemporal Lobar Degeneration. Acta Neuropathol 114:5–22. https://doi.org/10.1007/s00401-007-0237-2
Davidson YS, Raby S, Foulds PG, Robinson A, Thompson JC, Sikkink S et al (2011) TDP-43 pathological changes in early onset familial and sporadic Alzheimer’s disease, late onset Alzheimer’s disease and Down’s syndrome: association with age, hippocampal sclerosis and clinical phenotype. Acta Neuropathol 122:703–713. https://doi.org/10.1007/s00401-011-0879-y
Dickson DW, Davies P, Bevona C, Van Hoeven KH, Factor SM, Grober E et al (1994) Hippocampal sclerosis: a common pathological feature of dementia in very old (> or = 80 years of age) humans. Acta Neuropathol 88:212–221
Folstein MF, Robins LN, Helzer JE (1983) The Mini-Mental State Examination. Arch Gen Psychiatry 40:812
Geser F, Robinson JL, Malunda JA, Xie SX, Clark CM, Kwong LK et al (2010) Pathological 43-kDa transactivation response DNA-binding protein in older adults with and without severe mental illness. Arch Neurol 67:1238–1250. https://doi.org/10.1001/archneurol.2010.254
Group Working (1998) Consensus report of the Working Group on: “Molecular and Biochemical Markers of Alzheimer’s Disease”. The Ronald and Nancy Reagan Research Institute of the Alzheimer’s Association and the National Institute on Aging Working Group. Neurobiol Aging 19:109–116
Hu WT, Josephs KA, Knopman DS, Boeve BF, Dickson DW, Petersen RC et al (2008) Temporal lobar predominance of TDP-43 neuronal cytoplasmic inclusions in Alzheimer disease. Acta Neuropathol 116:215–220. https://doi.org/10.1007/s00401-008-0400-4
Josephs KA (2018) Fitting TDP-43 into the APOE epsilon4 and neurodegeneration story. Lancet Neurol 17:735–737. https://doi.org/10.1016/S1474-4422(18)30288-6
Josephs KA, Dickson DW (2007) Hippocampal sclerosis in tau-negative frontotemporal lobar degeneration. Neurobiol Aging 28:1718–1722. https://doi.org/10.1016/j.neurobiolaging.2006.07.010
Josephs KA, Dickson DW, Tosakulwong N, Weigand SD, Murray ME, Petrucelli L et al (2017) Rates of hippocampal atrophy and presence of post-mortem TDP-43 in patients with Alzheimer’s disease: a longitudinal retrospective study. Lancet Neurol 16:917–924. https://doi.org/10.1016/S1474-4422(17)30284-3
Josephs KA, Hodges JR, Snowden JS, Mackenzie IR, Neumann M, Mann DM et al (2011) Neuropathological background of phenotypical variability in frontotemporal dementia. Acta Neuropathol 122:137–153. https://doi.org/10.1007/s00401-011-0839-6
Josephs KA, Murray ME, Whitwell JL, Parisi JE, Petrucelli L, Jack CR et al (2014) Staging TDP-43 pathology in Alzheimer’s disease. Acta Neuropathol 127:441–450. https://doi.org/10.1007/s00401-013-1211-9
Josephs KA, Murray ME, Whitwell JL, Tosakulwong N, Weigand SD, Petrucelli L et al (2016) Updated TDP-43 in Alzheimer’s disease staging scheme. Acta Neuropathol 131:571–585. https://doi.org/10.1007/s00401-016-1537-1
Josephs KA, Nelson PT (2015) Unlocking the mysteries of TDP-43. Neurology 84:870–871. https://doi.org/10.1212/WNL.0000000000001322
Josephs KA, Stroh A, Dugger B, Dickson DW (2009) Evaluation of subcortical pathology and clinical correlations in FTLD-U subtypes. Acta Neuropathol 118:349–358. https://doi.org/10.1007/s00401-009-0547-7
Josephs KA, Whitwell JL, Jack CR, Parisi JE, Dickson DW (2006) Frontotemporal lobar degeneration without lobar atrophy. Arch Neurol 63:1632–1638. https://doi.org/10.1001/archneur.63.11.1632
Josephs KA, Whitwell JL, Knopman DS, Hu WT, Stroh DA, Baker M et al (2008) Abnormal TDP-43 immunoreactivity in AD modifies clinicopathologic and radiologic phenotype. Neurology 70:1850–1857. https://doi.org/10.1212/01.wnl.0000304041.09418.b1
Josephs KA, Whitwell JL, Tosakulwong N, Weigand SD, Murray ME, Liesinger AM et al (2015) TAR DNA-binding protein 43 and pathological subtype of Alzheimer’s disease impact clinical features. Ann Neurol 78:697–709. https://doi.org/10.1002/ana.24493
Josephs KA, Whitwell JL, Weigand SD, Murray ME, Tosakulwong N, Liesinger AM et al (2014) TDP-43 is a key player in the clinical features associated with Alzheimer’s disease. Acta Neuropathol 127:811–824. https://doi.org/10.1007/s00401-014-1269-z
Jung Y, Dickson DW, Murray ME, Whitwell JL, Knopman DS, Boeve BF et al (2014) TDP-43 in Alzheimer’s disease is not associated with clinical FTLD or Parkinsonism. J Neurol 261:1344–1348. https://doi.org/10.1007/s00415-014-7352-5
Kaplan E, Goodglass H, Weintraub S (1978) The Boston Naming Test. Veterans Administration Medical Center, Portland
Kwong LK, Uryu K, Trojanowski JQ, Lee VM (2008) TDP-43 proteinopathies: neurodegenerative protein misfolding diseases without amyloidosis. Neurosignals 16:41–51. https://doi.org/10.1159/000109758
Lee EB, Porta S, Michael Baer G, Xu Y, Suh E, Kwong LK et al (2017) Expansion of the classification of FTLD-TDP: distinct pathology associated with rapidly progressive frontotemporal degeneration. Acta Neuropathol 134:65–78. https://doi.org/10.1007/s00401-017-1679-9
Lezak M (1995) Neuropsychological assessment. Oxford University Press, Oxford
Mackenzie IR, Neumann M, Baborie A, Sampathu DM, Du Plessis D, Jaros E et al (2011) A harmonized classification system for FTLD-TDP pathology. Acta Neuropathol 122:111–113. https://doi.org/10.1007/s00401-011-0845-8
Mackenzie IR, Neumann M, Bigio EH, Cairns NJ, Alafuzoff I, Kril J et al (2009) Nomenclature for neuropathologic subtypes of frontotemporal lobar degeneration: consensus recommendations. Acta Neuropathol 117:15–18. https://doi.org/10.1007/s00401-008-0460-5
Mattis S (1988) Dementia rating scale: professional manual. Psychological Assessment Resources, Odessa
McAleese KE, Walker L, Erskine D, Thomas AJ, McKeith IG, Attems J (2017) TDP-43 pathology in Alzheimer’s disease, dementia with Lewy bodies and ageing. Brain Pathol 27:472–479. https://doi.org/10.1111/bpa.12424
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH et al (2011) The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7:263–269. https://doi.org/10.1016/j.jalz.2011.03.005
Mirra SS, Heyman A, McKeel D, Sumi SM, Crain BJ, Brownlee LM et al (1991) The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer’s disease. Neurology 41:479–486
Murray ME, Bieniek KF, Banks Greenberg M, DeJesus-Hernandez M, Rutherford NJ, van Blitterswijk M et al (2013) Progressive amnestic dementia, hippocampal sclerosis, and mutation in C9ORF72. Acta Neuropathol 126:545–554. https://doi.org/10.1007/s00401-013-1161-2
Nag S, Yu L, Capuano AW, Wilson RS, Leurgans SE, Bennett DA et al (2015) Hippocampal sclerosis and TDP-43 pathology in aging and Alzheimer disease. Ann Neurol 77:942–952. https://doi.org/10.1002/ana.24388
Neary D, Snowden JS, Gustafson L, Passant U, Stuss D, Black S et al (1998) Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology 51:1546–1554
Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT et al (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314:130–133. https://doi.org/10.1126/science.1134108
Petersen RC (2004) Mild cognitive impairment as a diagnostic entity. J Intern Med 256:183–194. https://doi.org/10.1111/j.1365-2796.2004.01388.x
Rauramaa T, Pikkarainen M, Englund E, Ince PG, Jellinger K, Paetau A et al (2013) Consensus recommendations on pathologic changes in the hippocampus: a postmortem multicenter inter-rater study. J Neuropathol Exp Neurol 72:452–461. https://doi.org/10.1097/NEN.0b013e318292492a
Rey A (1964) L’examen clinique en psychologie. Presses Universitaires de France, Paris
Rohrer JD, Geser F, Zhou J, Gennatas ED, Sidhu M, Trojanowski JQ et al (2010) TDP-43 subtypes are associated with distinct atrophy patterns in frontotemporal dementia. Neurology 75:2204–2211. https://doi.org/10.1212/WNL.0b013e318202038c
Rutherford NJ, Carrasquillo MM, Li M, Bisceglio G, Menke J, Josephs KA et al (2012) TMEM106B risk variant is implicated in the pathologic presentation of Alzheimer disease. Neurology 79:717–718. https://doi.org/10.1212/WNL.0b013e318264e3ac
Spreen O, Strauss E (1998) Compendium of neuropsychological tests, second edition: administration, norms and commentary. Oxford University Press, Oxford
Tan RH, Kril JJ, Fatima M, McGeachie A, McCann H, Shepherd C et al (2015) TDP-43 proteinopathies: pathological identification of brain regions differentiating clinical phenotypes. Brain 138:3110–3122. https://doi.org/10.1093/brain/awv220
van Blitterswijk M, Mullen B, Nicholson AM, Bieniek KF, Heckman MG, Baker MC et al (2014) TMEM106B protects C9ORF72 expansion carriers against frontotemporal dementia. Acta Neuropathol 127:397–406. https://doi.org/10.1007/s00401-013-1240-4
Van Deerlin VM, Sleiman PM, Martinez-Lage M, Chen-Plotkin A, Wang LS, Graff-Radford NR et al (2010) Common variants at 7p21 are associated with frontotemporal lobar degeneration with TDP-43 inclusions. Nat Genet 42:234–239. https://doi.org/10.1038/ng.536
Watts GD, Wymer J, Kovach MJ, Mehta SG, Mumm S, Darvish D et al (2004) Inclusion body myopathy associated with Paget disease of bone and frontotemporal dementia is caused by mutant valosin-containing protein. Nat Genet 36:377–381. https://doi.org/10.1038/ng1332
Wechsler D (1987) Wechsler Memory Scale-Revised (Manual). Psychological Corporation, San Antonio
Wennberg A, Tosalkulwong N, Lesnick T, Murray ME, Whitwell JL, Liesinger AM et al (2018) Association of apolipoprotein E epsilon 4 with transactive response DNA binding protein 43. JAMA Neurol 75:1347–1354
Whitwell JL, Crum WR, Watt HC, Fox NC (2001) Normalization of cerebral volumes by use of intracranial volume: implications for longitudinal quantitative MR imaging. AJNR Am J Neuroradiol 22:1483–1489
Whitwell JL, Jack CR Jr, Parisi JE, Senjem ML, Knopman DS, Boeve BF et al (2010) Does TDP-43 type confer a distinct pattern of atrophy in frontotemporal lobar degeneration? Neurology 75:2212–2220. https://doi.org/10.1212/WNL.0b013e31820203c2
Wilson AC, Dugger BN, Dickson DW, Wang DS (2011) TDP-43 in aging and Alzheimer’s disease—a review. Int J Clin Exp Pathol 4:147–155
Wilson RS, Yu L, Trojanowski JQ, Chen EY, Boyle PA, Bennett DA et al (2013) TDP-43 pathology, cognitive decline, and dementia in old age. JAMA Neurol 70:1418–1424. https://doi.org/10.1001/jamaneurol.2013.3961
Yang HS, Yu L, White CC, Chibnik LB, Chhatwal JP, Sperling RA et al (2018) Evaluation of TDP-43 proteinopathy and hippocampal sclerosis in relation to APOE epsilon4 haplotype status: a community-based cohort study. Lancet Neurol 17:773–781. https://doi.org/10.1016/S1474-4422(18)30251-5
Zhang YJ, Xu YF, Cook C, Gendron TF, Roettges P, Link CD et al (2009) Aberrant cleavage of TDP-43 enhances aggregation and cellular toxicity. Proc Natl Acad Sci USA 106:7607–7612. https://doi.org/10.1073/pnas.0900688106
Acknowledgements
This study was funded by the following grants from the US National Institutes of Health (National Institute on Aging): R01 AG037491, P50 AG16574, U01 AG006786 and R35 NS097261 (National Institute of Neurological Disorders and Stroke). We thank the families of the patients who donated their brains to science and thus allowed completion of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Josephs, K.A., Murray, M.E., Tosakulwong, N. et al. Pathological, imaging and genetic characteristics support the existence of distinct TDP-43 types in non-FTLD brains. Acta Neuropathol 137, 227–238 (2019). https://doi.org/10.1007/s00401-018-1951-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-018-1951-7