Abstract
Little is known in continental Antarctic about patterns of abundance, diversity, and succession of microfungi within moss profiles consisting of live, senescent, and dead tissues in different stages of decomposition. In the present study, vertical patterns of the abundance and diversity of microfungi and their relationship with chemical compositions were examined within moss colonies collected from coastal outcrops in the Lützow–Holm Bay area (Queen Maud Land), East continental Antarctica. Total and darkly pigmented hyphal length, the richness of molecular operational taxonomic units (MOTUs) of microfungi, and the occurrence of Phoma herbarum and Pseudogymnoascus pannorum increased with the depth of moss profiles. The content of organic chemical components and nitrogen in moss tissues decreased, whereas ash content increased with the depth of moss profiles. Relative amount of recalcitrant compounds and total carbohydrates did not significantly differ among the vertical layers. The downward increase of the microfungal richness and occurrence in the Antarctic moss profiles without MOTU replacement was consistent with the directional-nonreplacement model of succession, indicative of the high environmental resistance, which represents the sum of the adverse factors hindering the success of species establishment. This contrasted with the fungal succession in arctic moss profiles, which accorded with the directional-replacement model, in which species replacement took place due to modification of habitat and competition. More hostile environmental conditions than those in the Arctic characterized the fungal succession and limited the fungal decomposition of moss in continental Antarctica.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mosses are major components of terrestrial ecosystems of Antarctica and contribute importantly to the crucial budgets of carbon and nutrients via primary production and concomitant decomposition and accumulation of moss tissues in soil (Kanda and Inoue 1994; Ochyra et al. 2008; Cannone et al. 2013). The accumulated live, senescent, and dead moss tissues, termed the Bryosphere (Lindo and Gonzalez 2010), harbors a variety of fungi and other organisms driving carbon and nutrient dynamics in the Bryosphere (Davey and Currah 2006; Osono and Trofymow 2012). Previous studies have examined the accumulation and decomposition of terrestrial moss mainly in maritime and peninsular Antarctica (Baker 1972; Fenton 1980; Davis 1981), but less in continental Antarctica. Such bias in study location holds true for the study of Antarctic moss-associated fungi, which has been conducted mainly in maritime and peninsula Antarctica (Stevens et al. 2007; Zhang et al. 2013; Arenz et al. 2014; Yu et al. 2014), and less in continental Antarctica (McRae and Seppelt 1999; Tosi et al. 2002; Ruisi et al. 2007). Very little is known about Antarctic patterns of abundance, diversity, and succession of microfungi within moss profiles consisting of live, senescent, and dead tissues in different stages of decomposition.
The purpose of the present study was to examine vertical patterns of the abundance and diversity of microfungi and their relationship with chemical compositions within moss profiles collected from coastal outcrops at five locations in the Lützow–Holm Bay area (Queen Maud Land), East continental Antarctica. Colonies of Bryum pseudotriquetrum (Hedw.) P. Gaertn., B. Mey. & Scherb./B. archangelicum Bruch & Schimp. complex were collected and divided into four layers according to the color and texture. The changes in organic chemical and nutrient contents in the profiles are indicative of the pattern of decomposition and accumulation of organic matter. Investigating the microfungal abundance and composition in the profiles would help to identify possible factor(s) affecting the microfungal succession in decomposing tissues and the roles of microfungi in the decomposition processes.
Materials and methods
Study area
Samples were collected at five sites in two ice-free regions (Langhovde and Skarvsnes) in the Lützow-Holm Bay area in East Antarctica (Table 1; Fig. 1). These sites are along a creek or a lake and did not appear to be affected by colonies of large animals. Samples were collected during the 51st Japanese Antarctic Research Expedition (JARE-51) from December 2009 to January 2010. At each site, five blocks (each sized 2 × 2 cm, approximately 6–10 cm below the surface) of Bryum pseudotriquetrum/B. archangelicum complex, one of the dominant moss species in the regions (Kanda and Inoue 1994; Fig. 2a), were collected, making a total of 25 moss blocks. The blocks were divided into 4 layers (denoted as layers G, R, B, and BT from the upper to the lower) according to color and texture (Fig. 2b), making a total of 100 samples. Moss stems (2 cm total length) were chosen from within the sample with tweezers sterilized with flame and 70% ethanol, preserved in a sterilized vial (1.5 mm3 volume), and stored at −20 °C to be used for fungal isolation. The remaining moss samples were divided into two subsamples and weighed for fresh weight. One subsample was preserved in a vinyl bag and stored at −20 °C to be used for hyphal length estimation. The other subsample was preserved in paper bags and dried at room temperature to be used for chemical analyses. These samples were taken back to the laboratory in Japan for further processing.
Locations of the ice-free coastal outcrops from which the moss samples were collected. Detailed location maps are given in Tanabe et al. (2017)
Hyphal length estimation
Hyphal lengths in moss profiles were estimated using the agar film method of Jones and Mollison (1948) but with several modifications (Osono et al. 2006b). One gram of moss tissues was taken from the frozen subsample for hyphal length estimation for each profile. Total hyphal length was calculated as the sum of the lengths of hyaline hyphae stained with fluorescent brightener and of darkly pigmented hyphae. The details of the methods are described in Osono et al. (2006b).
Isolation of fungi
Fungi were isolated from moss stems with a modified washing method (Osono et al. 2008). Moss stems were washed five times with 10 ml of sterilized 0.005% Aerosol-OT (di-2-ethylhexyl sodium sulfosuccinate) solution and then rinsed with sterilized water three times in a sterile test tube using a vertical type automatic mixer (S-100; Taitec Co., Ltd, Japan). The rinsed stems were transferred to a sterile filter paper in 9-cm Petri dishes and dried for 30 min. The stems were then placed on the surface of corn meal agar (Nissui Pharmaceutical Co., Ltd, Japan) in 100 mg/l chloramphenicol plates, and then the plates were incubated at 15 °C in the dark. The incubated plates were observed microscopically nine times at 1-week intervals. Any fungal colonies appearing on the plate were isolated, transferred to a plate containing a 1:1 mixture of corn meal agar and malt extracted agar supplemented with yeast extract (Nissui Pharmaceutical Co., Ltd, Japan), incubated, and identified by observing micromophological characteristics.
Molecular analysis of fungi
Fungal isolates were transferred to fresh malt extract agar overlaid with a cellophane membrane, and genomic DNA was extracted from the mycelia following the modified CTAB method (Hirose et al. 2013). Molecular analysis was performed as described by Tateno et al. (2015). Polymerase chain reactions (PCR) were performed for genomic DNA extracted from mycelia using a Quick Taq HS DyeMix (Toyobo, Osaka, Japan). Each PCR reaction contained a 50 µl mixture [21 µl distilled water, 25 µl master mix, 3 µl ca. 0.5 ng µl−1 template DNA, and 0.5 µl of each primer (final, 0.25 µM)]. To amplify the region including the rDNA ITS and 28S rDNA D1-D2 domain by PCR, the primer pair its1f (Gardes and Bruns 1993) and LR3 (Vilgalys and Hester 1990) was used. Each DNA fragment was amplified using a PCR thermal cycler (DNA Engine; Bio-Rad, Hercules, CA, USA) using the following thermal cycling conditions. The first cycle consisted of 5 min at 94 °C, followed by 35 cycles of 30 s at 94 °C, 30 s at 50 °C for annealing, 1 min at 72 °C, and a final incubation for 10 min at 72 °C. The reaction mixture was then cooled at 4 °C for 5 min. PCR products were purified with a QIAquick PCR Purification Kit (Qiagen, Germany) according to the manufacturer’s instructions.
Purified PCR products were sequenced by FASMAC Co., Ltd. (Kanagawa, Japan). Sequencing reactions were performed in a Gene Amp PCR System 9700 (Applied Biosystems, USA) using a BigDye Terminator V3.1 (Applied Biosystems), following the protocols supplied by the manufacturer. The fluorescent-labeled fragments were purified from the unincorporated terminators using an ethanol precipitation protocol. The samples were resuspended in formamide and subjected to electrophoresis in an ABI 3730xl sequencer (Applied Biosystems).
The rDNA ITS sequences of the fungal isolates were compared with sequences available in the GenBank database using BLASTN searches (Altschul et al. 1997). We defined molecular operational taxonomic units (MOTUs) within the rDNA ITS sequences dataset by clustering the sequences with the BLASTclust program provided online by the Max Planck Institute (http://toolkit.tuebingen.mpg.de/blastclust#) based on 97% sequence similarity and 90% coverage criteria. Occurrence of MOTU was recorded as presence-absence in each sample. The sequences of MOTUs determined in this study were deposited in the DNA Data Bank of Japan (DDBJ) (Accession: LC085207–LC085220) (Table 2).
Chemical analyses of moss
The subsamples for chemical analyses were oven-dried to a constant mass at 40 °C to determine oven-dry mass. Moisture content was calculated gravimetrically according to the equation: moisture content (%) = amount of water/oven-dry mass × 100. The subsamples were then ground in a laboratory mill to pass a 0.5-mm screen and used for chemical analyses according to the methods described in detail in Osono et al. (2006a). Acid-unhydrolyzable residue (AUR, also known as the acid-insoluble residue or ‘Klason lignin’ fraction) and extractive (EXT) contents were measured by sulfuric acid digestion and alcohol-benzene extraction, respectively (King and Heath 1967). Acid-unhydrolyzable residue contains not only true lignin of plant origin but also condensed tannin, phenolic compounds, carboxylic compounds, and alkyl compounds such as cutin (Preston et al. 1997). It should be noted that no ‘true’ lignin has been found in moss, but other phenolic cell wall material has been found (Erickson and Miksche 1974). Total carbohydrate (TCH) content was measured by a phenol–sulfuric acid method (Dubois et al. 1956). Total carbon and nitrogen contents were measured by automatic gas chromatography (NC analyzer SUMIGRAPH NC-900, Sumitomo Chemical Co., Ltd, Osaka, Japan). The contents were expressed in mg g−1 ash-free dry material.
Lignocellulose index (LCI) is a useful indicator of the availability of organic chemical compounds for decomposer organisms (Osono and Takeda 2005). The LCI denotes the amount of carbohydrates relative to lignocellulose matrix in plant tissues and is calculated as: LCI = TCH content/(TCH content + AUR content).
Statistical analysis
We first used generalized linear models (GLMs) to evaluate the difference in total hyphal length, darkly pigmented hyphal length, the proportion of darkly pigmented hyphal length with respect to the total hyphal length, MOTU richness, and the occurrence of major fungal MOTUs using moss layer and sampling site as predictor variables. Major MOTUs were defined as those that occurred on more than 10 out of 100 moss samples tested. We then used GLMs to evaluate the difference in chemical contents in moss tissues using moss layer and sampling site as predictor variables. Finally, factors affecting the hyphal lengths, MOTU richness, and the occurrence of major MOTUs were analyzed with additional GLMs with moss layer and sampling site as categorical predictors, and chemical contents as continuous predictors. The error structure of the GLM was Gaussian (hyphal lengths), Poisson (MOTU richness), or binomial (occurrence of major MOTUs). The GLM function in the R software package for Macintosh 3.1.2 (http://www.R-project.org) was used to perform the analysis, with the glht function of the R multcomp package for multiple comparisons with Tukey’s test, and an automatic stepwise model selection with Akaike information criterion (AIC) was performed to find the most parsimonious model, using the minimum AIC as the best-fit estimator.
Results
Hyphal length
Total and darkly pigmented hyphal lengths in moss ranged on average from 1256 to 5406 m/g dry material and from 35 to 1818 m/g, respectively, and were significantly greater in layers R, B, and BT than in layer G (Fig. 3a, b; Table 3). The proportion of darkly pigmented hyphal length with respect to the total hyphal length ranged from 2 to 43% on average and was the highest in layer B and was significantly higher in layers R and BT than in layer G (Fig. 3c; Table 3). Total hyphal length was not significantly different among the five sampling sites (Fig. 3a; Table 3), while darkly pigmented hyphal length and its proportion relative to the total hyphal length were significantly higher at site #5 than at the other four sites (Fig. 3b, c; Table 3).
Total hyphal length (a), the length of darkly pigmented hyphae (b), and the proportion of darkly pigmented hyphal length relative to the total hyphal length (c) in moss profiles, with respect to the vertical layers (left) and sites (right). The same letters indicate that there is not a significant difference at the 5% level by Tukey’s HSD test. Bars indicate standard errors (n = 5)
Richness and occurrence of fungi
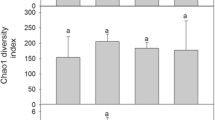
A total of 72 isolates were obtained from 53 (53%) out of the 100 moss samples from five sites, with zero to three isolates (0.7 isolates on average) per sample (Table 4). These fungal isolates were classified into 14 MOTUs (Table 2). The most frequent MOTU was Phoma herbarum Westend., accounting for 31% (31 samples) of the 100 samples, followed by Pseudogymnoascus pannorum (Link) Minnis & D.L. Lindner (15%). The other 12 MOTUs constituted 6% (Dothideomycetes sp.1), 4% (Phaeosphaeria sp.1), or less (1–3% of the samples; Cadophora malorum (Kidd & Beaum.) W. Gams, Cladophialophora minutissima M.L. Davey & Currah, Cryptococcus victoriae M.J. Montes, Belloch, Galiana, M.D. García, C. Andrés, S. Ferrer, Torr.-Rodr. & J. Guinea, Phaeosphaeria sp.2, Tetracladium sp., and five unidentified species of Leotiomycetes).
Fungal MOTU richness and the occurrences of P. herbarum and P. pannorum increased significantly from layer G to layer BT (Fig. 4a–c; Table 3). Fungal MOTU richness was significantly higher at site #19 than at site #20 (Fig. 4a; Table 3). Phoma herbarum occurred at a significantly higher frequency at site #4 than at site #6, and it was not isolated from moss samples at site #20 (Fig. 4b; Table 3). Pseudogymnoascus pannorum occurred at a significantly higher frequency at site #19 than at the other four sites (Fig. 4c; Table 3).
MOTU richness (a) and the occurrence of Phoma herbarum (b) and Pseudogymnoascus pannorum (c) in moss profiles, with respect to the vertical layers (left) and sites (right). The same letters indicate that there is not a significant difference at the 5% level by Tukey’s HSD test. Bars indicate standard errors (n = 5)
Chemical composition of moss tissues
Generally, the contents of AUR, TCH, EXTR, total carbon, and total nitrogen decreased significantly from layer G to layer BT, whereas ash content increased significantly from layer G to layer BT (Fig. 5a, b, d–g; Table 3). The LCI was not significantly different among the four layers (Fig. 5c; Table 3), while the C/N ratio was the highest in layer R and was significantly higher in layers G and B than in layer BT (Fig. 5h; Table 3). The chemical contents were significantly different among the sites, with the AUR and TCH contents higher at sites #4 and #5 than at the other sites, and N content was in the order of: site #4 > sites #5 and #6 > sites #19 and #20 (Fig. 5a, b, d, e, f, g; Table 3). The LCI was highest at site #19 and was significantly higher at sites #4, #6, and #20 than at site #5 (Fig. 5c; Table 3). C/N ratio was significantly higher at sites #5 and #20 than at sites #4 and #6 (Fig. 5h; Table 3).
Contents of acid-unhydrolyzable residue (a) and total carbohydrates (b), lignocellulose index (c), the contents of extractives (d), ash (e), carbon (f), and nitrogen (g), and carbon to nitrogen ratio (h) in moss profiles, with respect to the vertical layers (left) and sites (right). The same letters indicate that there is not a significant difference at the 5% level by Tukey’s HSD test. Bars indicate standard errors (n = 5)
Stepwise model selections resulted in significant effects of moss layer and/or sampling location on hyphal lengths, MOTU richness, and the occurrence of major MOTUs (Table 5). Of the chemical contents, EXT and nitrogen contents were selected as predictor variables for total hyphal length but had χ 2 values that were not statistically significant (P > 0.05) (Table 5). Ash and nitrogen contents were selected as predictor variables for MOTU richness, of which ash content had a significant χ 2 value and a positive regression coefficient (Table 5). Carbon and water contents were selected as predictor variables for the occurrence of P. herbarum, and carbon content had a significant χ 2 value and a negative regression coefficient (Table 5). Carbon content was selected as a predictor variable for the occurrence of P. pannorum that had a significant χ 2 value and a negative regression coefficient (Table 5).
Discussion
The total hyphal lengths in moss tissues (Fig. 3) were within the range in previous reports for dead plant tissues and soil in the Antarctic and Arctic (i.e., up to 7000 m g−1) (Dowding and Widden 1974; Miller and Laursen 1974; Robinson et al. 1996; Osono et al. 2014). Bailey and Wynn-Williams (1982) reported a similar value of the mean hyphal length (2783 m g−1) for Mountain moss (surface 0–1.3 cm) in Signy Island, Antarctica. Our previous study in the Arctic also found similar levels of total hyphal lengths, with mean values ranging from 104 to 6310 m g−1 within the profiles (up to 18 cm depth) of two moss species (Osono et al. 2012). Total hyphal length increased significantly in the region of moss senescence from layer G to R, whereas it showed no significant net change from layer R to BT (Fig. 3). This suggests that the hyphal ingrowth was restricted in layer G, possibly due to environmental constraints, such as the antifungal activity of live moss tissues, which can impede fungal invasion (Lehtonen et al. 2009), or UV radiation (Tosi et al. 2005).
Darkly pigmented hyphal lengths have rarely been investigated in the Antarctic and Arctic regions, despite the adaptive importance of hyphal pigmentation and melanization under environmental stresses such as low temperature, desiccation, and exposure to UV radiation (Butler and Day 1998). The lengths of darkly pigmented hyphae and its proportions relative to the total length in the present study were similar to those reported for Arctic mosses (Osono et al. 2012) and dead vascular plant tissues and forest floor materials in arctic tundra and temperate forests (Osono et al. 2006b, 2011, 2014). Darkly pigmented hyphae are attributable to those of dematiaceous species, such as Cadophora and Cladophialophora (Table 2). Colonization of moss tissues by these fungal species may have contributed to some extent to the accumulation of darkly pigmented hyphae in moss profiles. Alternatively, melanized hyphae simply accumulated from layer G to B because of slower degradation and turnover of melanized hyphae than hyaline hyphae (Fernandez and Koide 2014). The present study was not successful in elucidating possible effects of chemical composition on darkly pigmented hyphae (Table 5). The reasons for the decrease in the proportion of darkly pigmented hyphal length with respect to the total hyphal length in layer BT (Fig. 3c) remain unclear, but one possibility would be less extreme fluctuations in temperature and moisture in the lowermost layer than in the upper layers (Osono et al. 2012), which could enhance the survival of hyaline hyphae in the lowermost layer.
The present study found 14 MOTUs (at the 97% similarity threshold), which was within the range of previous reports for the richness of microfungi associated with moss and dead plant tissues from the Antarctic (10–28 species or OTUs; McRae and Seppelt 1999; Tosi et al. 2002; Hirose et al. 2013; Zhang et al. 2013; Yu et al. 2014) and Arctic (Osono et al. 2012, 2014) but appeared to be lower than in temperate mosses (Thormann et al. 2004; Osono and Trofymow 2012). It was also similar to the levels of microfungal richness in Antarctic soils (Malosso et al. 2006; Bridge and Newsham 2009; Rao et al. 2012). The increase of MOTU richness from the upper layer G to the lowermost layer BT (Fig. 4a) was related to the increase of ash content (Fig. 5e; Table 5), suggesting the gradual mixing of moss tissues with soil might partly account for the occurrence of fungi. The lower richness in the upper profile may also have been related to the more extreme fluctuations in temperature and moisture and to the antifungal activity of live moss tissues, as discussed above.
Phoma herbarum and P. pannorum are common components of fungal assemblages of moss in continental Antarctica (McRae and Seppelt 1999; Tosi et al. 2002). The adaptation of P. herbarum to Antarctic soil conditions, which are characterized by low temperature, high thermal variation, and freeze–thaw cycles, is probably related to its ability to produce exopolysaccharide (Selbmann et al. 2002). These fungal species are known as psychrotrophic or psychrophilic, and are known to also be distributed in Arctic regions (Robinson 2001; Domsch et al. 2007; Osono et al. 2012). Hirose et al. (2016) found contrasting patterns in the geographical distributions of P. herbarum and P. pannorum in the Lützow-Holm Bay and Amunzen Bay areas of continental Antarctica. That is, P. herbarum was distributed widely in these areas and tended to occur more frequently in moss tissues with higher total nitrogen content, whereas P. pannorum exhibited a localized distribution.
The downward increase of P. herbarum and P. pannorum was related to the decrease of total carbon content (Table 5), implying the fungal decomposition of organic materials in moss tissues. Indeed, these two species were capable of utilizing polymericcarbohydrates, such as cellulose and starch (Domsch et al. 2007; Leung et al. 2011). The C/N ratio in moss tissues decreased from layer R to layer BT (Fig. 5h). Such a decrease of C/N ratio during decomposition has often been attributed to the incorporation of nitrogen in fungal hyphae (Berg and Söderström 1979; Osono and Takeda 2001) and/or the secondary formation of recalcitrant compounds registered as AUR, possibly caused by the activity of decomposer fungi (Berg 1986; Osono and Takeda 2004; Fukasawa et al. 2009). In the present study, neither fungal hyphae (Fig. 3a) nor AUR accumulation (Fig. 3c) could account for the decrease of C/N ratio from layer R to BT. Alternative explanations for the decrease of C/N ratio may include the roles of other microbes, such as bacteria, and/or soil animals (Davis 1981; Lindo and Gonzalez 2010), but further studies are needed to test these speculations.
The vertical patterns of abundance and richness of fungi and chemical changes in the Antarctic moss profiles exhibited interesting similarities and contrasts to those of the Arctic ones (Table 6). Osono et al. (2012) conducted similar investigations using arctic moss profiles of Hylocomium splendens (Hedw.) W.P. Schimp. and Racomitrium lanuginosum (Hedwig) Bridel on Ellesmere Island, Canada (80°N). The hyphal length, the microfungal richness, and chemical changes were generally similar between Antarctic and Arctic moss profiles (Table 6). Meanwhile, marked differences were found for fungal succession and lignocellulose decomposition between Antarctica and the Arctic (Table 6). Species composition of microfungi changed successively downward within the Arctic moss profiles, concomitantly with the decrease of species richness and the relative decrease of TCH, indicating the competition for carbohydrates and competitive exclusion between microfungal species. In contrast, a cumulative increase of microfungal MOTUs with no significant changes in LCI characterized the succession in Antarctic moss profiles. These two patterns of fungal succession and resource utilization are analogous to the models of primary succession in polar deserts proposed by Svoboda and Henry (1987). Svoboda and Henry (1987) summarized plant succession in marginal arctic environments and presented three models of succession: a directional-replacement model in low resistance environments, a directional-nonreplacement model in high resistance environments, and a nondirectional-nonreplacement model in extremely resistant environments. The richness and occurrence of microfungi increased in the Antarctic moss profiles, but there was little evidence of MOTU replacement (Table 4), in the chronosequence, indicating directional-nonreplacement succession and the high environmental resistance, which represent the sum of the adverse factors hindering the success of species establishment. In contrast, the fungal succession in Arctic moss profiles is consistent with the directional-replacement model, in which species replacement takes place due to competition. The contrasting patterns of fungal succession suggest that more hostile environmental conditions in continental Antarctica than in the Arctic characterize the pattern of succession in decomposing moss tissues and limit the roles of microfungi in the decomposition processes.
To our knowledge, the present study is the first to determine the detailed picture of the vertical pattern of fungal occurrence within moss profiles (i.e., the Bryosphere) of continental Antarctica, with reference to hyphal length and culturable fungal assemblages in relation to chemical changes in decomposing moss tissues. Mosses are a major component of poorly vegetated, ice-free coastal outcrops in continental Antarctica, and studies of their diversity, distribution, and biomass have been conducted (Kanda and Inoue 1994; Ochyra et al. 2008; Cannone et al. 2013). Our dataset provides useful insights into the abundance and diversity of fungi, potential factors affecting them, and possible roles of fungi in chemical changes in moss profiles in continental Antarctica.
References
Altschul SF, Madden TL, Schaffer AA, Zhang JH, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Arenz BE, Blanchette RA, Farrell RL (2014) Fungal diversity in Antarctic soils. In: Cowan DA (ed) Antarctic terrestrial microbiology. Springer, Berlin, pp 35–53
Bailey AD, Wynn-Williams DD (1982) Soil microbiological studies at Signy Island, South Orkney Islands. Br Antarct Surv Bull 51:167–191
Baker JH (1972) The rate of production and decomposition of Chorisodontium aciphyllum (Hook. f. & Wils.) Broth. Br Antarct Surv Bull 27:123–129
Berg B (1986) Nutrient release from litter and humus in coniferous forest soils—a mini review. Scand J For Res 1:359–369
Berg B, Söderström B (1979) Fungal biomass and nitrogen in decomposing Scots pine needle litter. Soil Biol Biochem 11:339–341
Bridge PD, Newsham KK (2009) Soil fungal community composition at Mars Oasis, a southern maritime Antarctic site, assessed by PCR amplification and cloning. Fungal Ecol 2:66–74
Butler MJ, Day AW (1998) Fungal melanins: a review. Can J Microbiol 44:1115–1136
Cannone N, Convey P, Guglielmin M (2013) Diversity trends of bryophytes in continental Antarctica. Polar Biol 36:259–271
Davey ML, Currah RS (2006) Interactions between mosses (Bryophyta) and fungi. Can J Bot 84:1509–1519
Davis RC (1981) Structure and function of two Antarctic terrestrial moss communities. Ecol Monogr 51:125–143
Domsch KH, Gams W, Anderson TH (2007) Compendium of soil fungi, 2nd edn. IHW-Verlag, Eching
Dowding P, Widden P (1974) Some relationships between fungi and their environment in tundra regions. In: Holding AJ, Heal OW, MacLean SF Jr, Flangan PW (eds) Soil organisms and decomposition in tundra. University of Alaska, Tundra Biome Steering Committee, Fairbanks, pp 123–150
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith E (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Erickson M, Miksche GE (1974) On the occurrence of lignin or polyphenols in some mosses and liverworts. Phytochemistry 13:2295–2299
Fenton JHC (1980) The rate of peat accumulation in Antarctic moss banks. J Ecol 68:211–228
Fernandez CW, Koide RT (2014) Initial melanin and nitrogen concentrations control the decomposition of ectomycorrhizal fungal litter. Soil Biol Biochem 77:150–157
Fukasawa Y, Osono T, Takeda H (2009) Effects of attack of saprobic fungi on twig litter decomposition by endophytic fungi. Ecol Res 24:1067–1073
Gardes M, Bruns TD (1993) ITS primer with enhanced specificity for basidiomycetes: application to the identification of mycorrhizae and rust. Mol Ecol 21:113–118
Hirose D, Tanabe Y, Uchida M, Kudoh S, Osono T (2013) Microfungi associated with withering willow wood in ground contact near Syowa Station, East Antarctica for 40 years. Polar Biol 36:919–924
Hirose D, Hobara S, Matsuoka S, Kato K, Tanabe Y, Uchida M, Kudoh S, Osono T (2016) Diversity and community assembly of moss-associated fungi in ice-free coastal outcrops of continental Antarctica. Fungal Ecol 24:94–101
Jones PCT, Mollison JE (1948) A technique for the quantitative estimation of soil microorganisms. J Gen Microbiol 2:54–69
Kanda H, Inoue M (1994) Ecological Monitoring of moss and lichen vegetation in the Syowa station area, Antarctica. Proc NIPR Symp Polar Biol 7:221–231
King HGC, Heath GW (1967) The chemical analysis of small samples of leaf material and the relationship between the disappearance and composition of leaves. Pedobiologia 7:192–197
Lehtonen MT, Akita M, Kalkkinen N, Ahola-Iivarinen E, Rönnholm G, Somervuo P, Thelander M, Valkonen JPT (2009) Quickly-released peroxidase of moss in defense against fungal invaders. New Phytol 183:432–443
Leung GL, Robson GD, Robinson CH (2011) Characterisation of cold-tolerant fungi from a decomposing High Arctic moss. Soil Biol Biochem 43:1975–1979
Lindo Z, Gonzalez A (2010) The Bryosphere: an integral and influential component of the Earth’s biosphere. Ecosystems 13:612–627
Malosso E, Waite IS, English L, Hopkins DW, O’Donnell AG (2006) Fungal diversity in maritime Antarctic soils determined using a combination of culture isolation, molecular fingerprinting and cloning techniques. Polar Biol 29:552–561
McRae CF, Seppelt RD (1999) Filamentous fungi of the Windmill Islands, continental Antarctica. Effect of water content in moss turves on fungal diversity. Polar Biol 22:389–394
Miller OK Jr, Laursen GA (1974) Belowground fungal biomass on US tundra biome sites at Barrow, Alaska. In: Holding AJ, Heal OW, MacLean SF Jr, Flangan PW (eds) Soil organisms and decomposition in tundra. University of Alaska, Tundra Biome Steering Committee, Fairbanks, pp 151–158
Ochyra R, Lewis-Smith RI, Bednarek-Ochyra H (2008) The illustrated moss flora of Antarctica. Cambridge University Press, Cambridge
Osono T, Takeda H (2001) Organic chemical and nutrient dynamics in decomposing beech leaf litter in relation to fungal ingrowth and succession during three year decomposition processes in a cool temperate deciduous forest in Japan. Ecol Res 16:649–670
Osono T, Takeda H (2004) Accumulation and release of nitrogen and phosphorus in relation to lignin decomposition in leaf litter of 14 tree species in a cool temperate forest. Ecol Res 19:593–602
Osono T, Takeda H (2005) Limit values for decomposition and convergence process of lignocellulose fraction in decomposing leaf litter of 14 tree species in a cool temperate forest. Ecol Res 20:51–58
Osono T, Trofymow JA (2012) Microfungal diversity associated with Kindbergia oregana in successional forests of British Columbia. Ecol Res 27:35–41
Osono T, Hobara S, Koba K, Kameda K (2006a) Reduction of fungal growth and lignin decomposition in needle litter by avian excreta. Soil Biol Biochem 38:1623–1630
Osono T, Hirose D, Fujimaki R (2006b) Fungal colonization as affected by litter depth and decomposition stage of needle litter. Soil Biol Biochem 38:2743–2752
Osono T, Ishii Y, Hirose D (2008) Fungal colonization and decomposition of Castanopsis sieboldii leaf litter in a subtropical forest. Ecol Res 23:909–917
Osono T, Hobara S, Hishinuma T, Azuma JI (2011) Selective lignin decomposition and nitrogen mineralization in forest litter colonized by Clitocybe sp. Eur J Soil Biol 47:114–121
Osono T, Ueno T, Uchida M, Kanda H (2012) Abundance and diversity of fungi in relation to chemical changes in arctic moss profiles. Polar Sci 6:121–131
Osono T, Matsuoka S, Hirose D, Uchida M, Kanda H (2014) Fungal colonization and decomposition of leaves and stems of Salix arctica on deglaciated moraines in high-Arctic Canada. Polar Sci 8:207–216
Preston CM, Trofymow JA, Sayer BG, Niu J (1997) 13C nuclear magnetic resonance spectroscopy with cross-polarization and magic-angle spinning investigation of the proximate-analysis fractions used to assess litter quality in decomposition studies. Can J Bot 75:1601–1613
Rao S, Chan Y, Lacap DC, Hyde KD, Pointing SB, Farrell RL (2012) Low-diversity fungal assemblage in an Antarctic Dry Valleys soil. Polar Biol 35:567–574
Robinson CH (2001) Cold adaptation in Arctic and Antarctic fungi. New Phytol 151:341–353
Robinson CH, Borisova OB, Callaghan TV, Lee JA (1996) Fungal hyphal length in litter of Dryas octopetala in a high-Arctic polar semi-desert, Svalbard. Polar Biol 16:71–74
Ruisi S, Barreca D, Selbmann L, Zucconi L, Onofri S (2007) Fungi in Antarctica. Rev Environ Sci Biotechnol 6:127–141
Selbmann L, Onofri S, Fenice M, Federici F, Petruccioli M (2002) Production and structural characterization of the exopolysaccharide of the Antarctic fungus Phoma herbarum CCFEE 5080. Res Microbiol 153:585–592
Stevens MI, Hunger SA, Hills SFK, Gemmill CEC (2007) Phantom hitch-hikers mislead estimates of genetic variation in Antarctic mosses. Plant Syst Evol 263:191–201
Svoboda J, Henry GHR (1987) Succession in marginal arctic environments. Arc Alp Res 19:373–384
Tanabe Y, Yasui S, Osono T, Uchida M, Kudoh S, Yamamuro M (2017) Abundant deposits of nutrients inside lakebeds of Antarctic oligotrophic lakes. Polar Biol 40:603–613
Tateno O, Hirose D, Osono T, Takeda H (2015) Beech cupules share endophytic fungi with leaves and twigs. Mycoscience 56:252–256
Thormann MN, Currah RS, Bayley SE (2004) Patterns of distribution of microfungi in decomposing bog and fen plants. Can J Bot 82:710–720
Tosi S, Casado B, Gerdol R, Caretta G (2002) Fungi isolated from Antarctic mosses. Polar Biol 25:262–268
Tosi S, Onofri S, Brusoni M, Zucconi L, Vishniac H (2005) Response of Antarctic soil fungal assemblages to experimental warming and reduction of UV radiation. Polar Biol 28:470–482
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246
Yu NH, Kim JA, Jeong MH, Cheong YH, Hong SG, Jung JS, Koh YJ, Hur JS (2014) Diversity of endophytic fungi associated with bryophyte in the maritime Antarctic (King George Island). Polar Biol 37:27–36
Zhang T, Zhang YQ, Liu HY, Wei YZ, Li HL, Su J, Zhao LX, Yu LY (2013) Diversity and cold adaptation of culturable endophytic fungi from bryophytes in the Fildes Region, King George Island, maritime Antarctica. FEMS Microb Ecol 341:52–61
Acknowledgements
We thank Dr. Y. Motoyoshi and members of JARE-51 for their assistance during the expedition; Dr. H. Kanda and Dr. A.S. Mori for useful discussions; Dr. K. Kato for helpful identification of moss species; and Dr. Elizabeth Nakajima for critical reading of the manuscript. This study was partially supported by the National Institute of Polar Research through General Collaboration Projects No. 26-28 to T.O. and by a Japan Society for the Promotion of Science KAKENHI Grant (No. 70370096 to M.U.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hirose, D., Hobara, S., Tanabe, Y. et al. Abundance, richness, and succession of microfungi in relation to chemical changes in Antarctic moss profiles. Polar Biol 40, 2457–2468 (2017). https://doi.org/10.1007/s00300-017-2157-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-017-2157-5