Abstract
Bryophytes exhibit a decline in species richness with latitude across the sub-Antarctic islands, Antarctic Peninsula and Antarctic continent, but not within the Antarctic continent itself. We analyzed diversity and biogeographic patterns of bryophytes at intra-regional scale across the Ross Sector of continental Antarctica, also comparing the “coast” and “slope” provinces within this region, and placed these patterns in the context of bryophyte biogeography across Antarctica. Our study area included 63 sites along a transect through Victoria Land and the Transantarctic Mountains. Distributions of bryophyte species were collated from recent field surveys, the Antarctic Plant Database and the literature. Data analyses included rarefaction, hierarchical classification, multivariate analyses and description of richness trends by latitude bands. Despite an almost linear climatic gradient, bryophyte diversity in the Ross Sector is not influenced by latitude, and patterns differ depending on the scale of analysis. At local scale, diversity “hot spots” appear to be related to favorable local microclimatic conditions. At intra-regional scale, site location in the coast or slope province is the most effective predictor of bryophyte diversity. The site clustering within each province is consistent with precipitation and biogeographic separation of two sub-regions due to important dispersal barriers, as also reported for the microarthropod fauna. At continental scale, bryophyte diversity patterns among sectors suggest a continent–Antarctic Peninsula separation, consistent with the Gressitt Line, suggesting a common feature in the evolutionary history of the vegetation and invertebrate fauna. The high similarities of the floras of adjacent continental sectors suggest a potential route for bryophyte dispersal along the coast of continental Antarctica.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Poleward declines in species richness remain among the most widespread macro-ecological patterns described, although with stronger supporting evidence at regional than at local scale (Hillebrand 2004; Mittelbach et al. 2007). The origins and drivers of the latitudinal diversity gradient are debated (e.g., Mittelbach et al. 2007). Local and regional processes may critically affect the dynamics of latitudinal diversity gradients, due to species-specific and habitat-specific responses to the impacts of climate change and anthropogenic land use (e.g., Frank et al. 2007). Across most of the planet, with the exception of Antarctica, direct and appreciable human impacts are pervasive. Therefore, this remote continent provides the opportunity to assess diversity trends and patterns over a large area where species dynamics are influenced predominantly by natural processes.
The relationships between latitude and potential drivers of diversity have received significant attention for vascular plants (e.g., Qian and Ricklefs 2007; Rozzi et al. 2008 and references therein), while only a few studies have focused on bryophytes, and these either do not extend to or do not consider high latitudes in detail (Shaw et al. 2005; Rozzi et al. 2008). Terrestrial vegetation communities in Antarctica are predominantly composed of cryptogams (bryophytes and lichens). The greatest diversity of bryophytes is found on the Antarctic Peninsula (Peat et al. 2007; Ochyra et al. 2008). At a global scale, bryophytes do not show clear gradients with latitude, with the highest diversity levels being measured in the Southern Hemisphere and only intermediate levels present at tropical latitudes (Shaw et al. 2005). Along a 25° latitudinal gradient in Chile, Rozzi et al. (2008) demonstrated that bryophyte species richness was inversely related to latitude, reaching a maximum of about 800 species in the Magellan ecoregion (around 52°S). Beyond the region addressed by that study, there is a clear decline in bryophyte species richness from the Magellan ecoregion to Antarctica, where diversity declines to 111–115 species on the Antarctic Peninsula and Scotia arc (60–72°S) and to only 24–27 species on the Antarctic continent (south of 67°S) (Peat et al. 2007; Ochyra et al. 2008). Thus, a large-scale decline in species richness with latitude is clear for bryophytes when comparing the southern elements of South America, Africa and Australia with Antarctica, and this pattern is clear also at regional scale, considering the sub-Antarctic islands, Antarctic Peninsula and Antarctic continent separately (Peat et al. 2007; Ochyra et al. 2008).
Due to the continent’s long-term isolation and extreme climatic conditions, Antarctic terrestrial ecosystems include some of the simplest communities known. Even within this region, there are relatively few examples where extended latitudinal and environmental gradients can be hypothesized to coincide (Balks et al. 2006; Peat et al. 2007; Howard-Williams et al. 2010). Within the Antarctic continent, the Victoria Land (VL) coastline with the Ross Sea, and its southwards extension through the Transantarctic Mountains (all within the geographic Ross Sector), potentially provide one such example and ideal conditions to assess the existence and the potential drivers of latitude-related gradients. Peat et al. (2007) completed a continent-wide analysis of patterns of cryptogam diversity across the Antarctica, based on data from documented herbarium specimens and the literature, and confirmed a large-scale decrease in diversity with increasing latitude for the Antarctic Peninsula, with a much stronger trend apparent for lichens than for mosses. In VL (represented by a transect from the Queen Maud Range in the Transantarctic Mountains to Mount Gordon), no evidence of latitudinal gradients in either group has been identified, despite a relatively large amount of survey data across a range of sampling locations (Adams et al. 2006; Peat et al. 2007). Moreover, Green et al. (2011) report the existence of a “hot spot” of cryptogam diversity at an extreme southern location (Beardmore Glacier, 84°S) in the Transantarctic Mountains which could indicate a refugial location pre-dating the present Ross Ice Shelf extension. However, previous studies have not attempted detailed analyses of intra-regional diversity patterns along this latitudinal transect.
In the current study, we analyze patterns of bryophyte species richness along a south–north latitudinal transect from the Queen Maud Range (84°S, Transantarctic Mountains) to the northern part of VL (70°S). As only one liverwort species (Cephaloziella varians) occurs in continental Antarctica, in this paper we focused on mosses, because these are the only abundant and diverse bryophytes represented.
We aimed to:
-
(a)
Identify, at intra-regional-scale, diversity and biogeographic patterns of bryophytes both across the entire Ross Sector and through separately comparing the “coast” and “slope” biogeographic provinces of the continental Antarctica within this sector (sensu Ochyra et al. 2008) and
-
(b)
place these patterns in the context of bryophyte biogeography across Antarctica, including consideration of their potential mechanisms of colonization.
Materials and methods
Study area
The study area is located in the Ross Sector of eastern continental Antarctica, within which cryptogam distributional data were obtained from 63 sites located between 84.62°S and 70.02°S (Fig. 1; additional data given in Online Resource 1). Within this region, the sites were classified into two recognized provinces of the continental Antarctic biogeographic zone (sensu Ochyra et al. 2008): the coastal province (including 47 sampling locations from southern to northern Victoria Land) and the slope province (16 locations from the Queen Maud Range to northern Victoria Land). These sites include all available locations where vegetation has been sampled and described in the Ross Sector. Inevitably, in this region, access to sampling locations is severely constrained by logistic opportunities and the typical harsh operating conditions. All sites are isolated by water or permanent ice and are also geographically remote from each other, reducing any problems of spatial auto-correlation and pseudo-replication, as the minimum distance between neighboring sites was 3 km and, in most cases, >10 km. At most study locations (>95 %), a full floristic inventory was available (with a mean investigated area ≥1 km2). In the remaining cases, the sample location represents the coordinates of single herbarium specimens.
a Location of the study area; b study site locations in the Ross Sector; c areas of the Ross Sector with different levels of snow precipitation (accumulation 1955–2004, mm/year, redrawn from Monaghan et al. 2006) in relation to site clustering along the coast (ellipses with gray bars) and slope provinces (ellipses with black bars). Legend: A precipitation >200 mm/year, B precipitation 100–200 mm/year, C precipitation 50–100 mm/year, D precipitation 25–50 mm/year
The macroscopic vegetation of VL is exclusively composed of cryptogams (Adams et al. 2006). At a regional scale, this flora is reasonably well known (APD reference database; Cannone 2006; Cannone and Seppelt 2009; Seppelt et al. 2010; Green et al. 2011). The total number of bryophyte species recorded in the continental Antarctica is 24 (23 species and 1 variety), with 14 (13 species and 1 variety) of these recorded in the Ross Sector (Ochyra et al. 2008). Due to particular difficulties with the taxonomic identity of the “silver” Bryum group (Hills et al. 2010), we refer all materials here to Bryum argenteum.
The climate of the continental Antarctica is extremely harsh, with a gradient of decreasing temperatures and increasing aridity from the coast to the slope provinces. The coastal province is characterized by mean annual air temperature of c. −12 to −15 °C (northern VL) and c. −17 to −20 °C (southern VL) (Ochyra et al. 2008) and a snow precipitation gradient from 100 to 300 mm/year (Monaghan et al. 2006). In the slope province, the climate is extremely cold and arid, with mean air temperature ranging from −20 to −30 °C in the McMurdo Dry Valleys area and from −35 to −50 °C in the southern Transantarctic Mountains (Ochyra et al. 2008) and snow precipitation less than 100–200 mm/year (Monaghan et al. 2006). Permafrost is continuous with an overlying active layer (maximum summer thawing depth) of variable depth though generally <1 m (Guglielmin 2006; Adlam et al. 2010).
The first continental-scale ice sheets formed on Antarctica around 34 Ma (e.g., Strand et al. 2003; Sugden and Denton 2004; Barker et al. 2007). The pulsating nature and the warm-based conditions of the East Antarctic Ice Sheet (EAIS) are well known and documented between 33–34 and 14 Ma (e.g., Troedson and Smellie 2002). Despite episodes of extensive ice cover, at least some terrestrial habitat must have remained exposed throughout this period as coastal areas with vegetation ranging from low woodland (Nothofagus) to permafrost tundra persisted at least until the sharp Mid-Miocene cooling (e.g., Lewis et al. 2008). After this strong cooling event, the largest remaining ice-free areas (e.g., McMurdo Dry Valleys) experienced very limited landscape changes. After the Last Glacial Maximum (LGM, occurred in the Ross Sector between 18 and 27 ky BP, e.g., Ingolfsson et al. 1998), coastal areas deglaciated progressively from north to south, reaching approximately the contemporary state around 6 ky BP (e.g., Hall et al. 2004).
Methods
To describe climatic gradients across the latitudinal transect, we utilized data from all the available automatic weather stations (AWS) located in the coastal region of VL (from 83.1°S to 71.9°S, at elevations between 5 and 310 m a.s.l.), providing continuous year-round air temperature records (SCAR READER Project www.antarctica.ac.uk/met/READER/climate.html, and PNRA database www.climantartide.it). For each AWS, we computed for the year 2002 the mean annual (MAAT) and seasonal air temperatures (summer, T_DJF; fall, T_MAM; winter, T_JJA; spring, T_SON). Due to the well-known difficulties in measuring precipitation in Antarctica and the lack of available data, our analysis was limited to air temperature data. Regional trends of precipitation (snow accumulation) are derived from existing literature (e.g., Monaghan et al. 2006).
Data on bryophyte species occurrence at each site in this region were obtained from recent field surveys (Cannone 2006; Cannone and Seppelt 2009; Cannone unpublished data), documented herbarium specimens (British Antarctic Survey Herbarium database, http://www.antarctica.ac.uk/bas_research/data/collections/plant.php), Antarctic Plant Database (APD: http://www.antarctica.ac.uk/Resources/BSD/PlantDatabase/index.html) and additional recent literature (see APD; Ochyra et al. 2008; Seppelt et al. 2010; Green et al. 2011). This generated 47 locations from the coastal and 16 locations from the slope provinces. As there is much taxonomic uncertainty in the identification of the two varieties of B. argenteum (see Hills et al. 2010), we considered it as a single species for the purposes of this study. All records of Grimmia reflexidens Mull. Hal. were referred to Coscinodon reflexidens (Mull. Hal.) Ochyra comb. nov. (Ochyra 2004).
Data analyses
The influence of latitude on the mean annual and seasonal air temperatures (as a proxy for the main climatic conditions across the transect) was assessed by linear regression performed by the software Statistica®.
Distribution patterns of bryophyte species were examined using incidence data (presence/absence) at different spatial scales: (a) local scale (considering the total species diversity at each study site); (b) intra-regional scale (comparing the coast and slope provinces within the Ross Sector); (c) regional scale (considering the entire Ross Sector); and (d) continental scale (comparing the different geographic sectors of Antarctica; after Ochyra et al. 2008).
To assess the sufficiency of sampling effort, we analyzed the relationship between the number of sampling locations and the number of species recorded (rarefaction curves, after Peat et al. (2007)) (a) for the coastal province, (b) for the slope province and (c) for the whole study area. The rarefaction curves were computed using the software EstimateS® using sample-based rarefaction curves with 95 % confidence intervals.
The distribution patterns and characteristics of the bryophyte flora of the Ross Sector were analyzed by means of hierarchical classification (dendrogram, carried out using weighted averages among groups, using Manhattan distances, without data transformation prior to analysis, and performed by the software Statistica®), integrated by multivariate statistics (PCA, providing indirect ecological information). For PCA, we excluded species records associated with sites of geothermal activity, as these are linked to particular ecological conditions and are not representative of either the Ross Sector or the continental Antarctica. We also performed DCA (detrended correspondence analysis, again providing indirect ecological information) on the same dataset. As the results provided by DCA were less clear than those from PCA, we present only the latter here. Analyses of the relationships between bryophyte occurrence and environmental factors (location in the coast vs. slope province, latitude and elevation) were performed using redundancy analysis (RDA). For the RDA, we used only the most widespread species (only the species occurring in at least 10 % of the sampling sites, Cannone et al. 2004) among the coast and slope provinces to avoid potential artefacts due to rare species occurrence. Both PCA and RDA (and DCA) were performed using the software CANOCO (Ter Braak and Smilauer 1998).
For practical logistic reasons, sampling intensity was not constant across latitude, as the areas located between 77°S and 74°S, more accessible from research stations, contained a larger number of sampling sites with respect to all the other latitude bands. We found no evidence that the more intensive study of these sites influenced the patterns found (data not shown).
The richness trends of bryophyte species and higher taxa (genus, family, order) were computed by latitude bands (following Peat et al. 2007) for (a) the Ross Sector, (b) the coast province and (c) the slope province. We also compared species biogeography (following the biogeographic elements recognized by Ochyra et al. 2008) between the coast and slope provinces.
The similarity of the bryophyte floristic composition was quantified using the Jaccard similarity index applied across (a) the latitude bands of the Ross Sector and (b) all the standard but arbitrary geographic sectors of Antarctica.
Results
Abiotic factors and latitude
Mean annual air temperature showed a statistically significant decreasing trend with increasing latitude (r 2 = 0.77, b = −1.21, p < 0.01, equating to a decrease of 1.2 °C/latitude), as did seasonal mean air temperatures, with the steepest gradients in winter (r 2 = 0.77, b = −1.73, p < 0.01) and autumn (r 2 = 0.75, b = −1.26, p < 0.01) over spring (r 2 = 0.76, b = −1.00, p < 0.01) and summer (r 2 = 0.58, b = −0.81, p < 0.01), confirming the existence of a latitude-driven climate gradient across the study transect.
Bryophyte patterns
A total of 18 bryophyte species were recorded in the Ross Sector, with 17 species occurring in the coast province (Online Resource 2), 10 in the slope province (Online Resource 3) and 8 species common between the two provinces. Rarefaction curves confirmed that the sampling effort was sufficient in the coast province, slope province and whole Ross Sector (Fig. 2), allowing further analyses.
Intra-regional distribution patterns
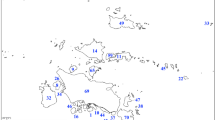
For the entire Ross Sector, at the local scale, the hierarchical classification (Fig. 3) identified two main branches: (a) a branch including sites mostly located at 77°S (with only one site at 72°S and one at 76°S), where the bryophyte flora was dominated by a high frequency of B. argenteum (100 %) and Bryum pseudotriquetrum (87.5 %), along with Hennediella heimii (75 %), Syntrichia sarconeurum (62.5 %) and Didymodon brachyphyllus (44 %); b) a branch including sites again dominated by a high frequency of B. argenteum (63 %), occurring with S. sarconeurum (56 %), Schistidium antartici (32 %), Ceratodon purpureus (30 %) and S. magellanica (22 %), and with the companion species Coscinodon lawianus, Grimmia plapodia, Orthogrimmia sessitana and Bryum archangelicum. The latter branch included sites characterized by lowest diversity (with only one or two species) and also included the two geothermally influenced sites (Mt. Melbourne and Mt. Rittmann), providing the only records of Campylopus pyriformis and Pohlia nutans. As these two species are representative of particular and unusual ecological conditions and not of the main environmental gradients of Antarctica, they were excluded from further analyses. The dendrogram shows that the sites were classified based on their floristic composition, rather than by latitude.
Hierarchical classification by dendrogram of the bryophyte communities of 63 sites of the Ross Sector. Legend for site acronyms: FP Finger Point, KP Kar Plateau, TV Taylor Valley, CH Cape Hallett, Gneiss Gneiss Point, BotB Botany Bay, BlueG Blue Glacier, CR Cape Ross, DI Dunlop Island, Turk Turk’s Head, MCM McMurdo, Armt Armitage Glacier, MP Marble Point, FL Finger Point/the Flatiron, Fryx Fryxell Lake, Hoobs Hobbs Glacier, CK Cape King, HP Harrow Peaks, GO Gondwana, Adare Cape Adare, SN Starr Nunatak, CS Cape Sastrugi, CC Crater Cirque, AI Apostrophe Island, PI Prior Island, CratH Crater Hill/Hut Point, EP Edmonson Point, GI Gregory Island, Kyff Mount Kyffin, Brat Bratina Island, Bird Bird Island, Bern Cape Bernacchi, Miers Miers Valley, RennG Rennick Glacier, BeafI Beaufort Island, GC Commonwealth Glacier, Dail Dailey Island, SC Simpson Crags, Patter Mount Patterson, Mmelb. Mount Melbourne, Rittm Mount Rittmann, Kukri Kukri Hills, Delta Delta Bluff, Mgord Mount Gordon, Harc Mount Harcourt, Penny Lake Penny, CommRan Commonwealth Range, Mbrown Mount Browning, BC Boulderclay, CP Cape Phillips, II Inexpressible Island, LI Lamplugh Island, Engl Mount England, Croz. Cape Crozet, Royd Cape Royd, TF Tarn Flat, Suess Mount Suess, McK Cape McKay, CastR Castle Ridge, Terror Mount Terror, BraGla Barrett Glacier
PCA demonstrated that the study sites were located along a gradient of species richness, with the less diverse sites (<3 species) toward the left and the richer (≥4 species) toward the right of the ordination, and clustered on the basis of their floristic composition, rather than latitude (Fig. 4). The first two axes explained 45.1 % of the species cumulative variance. In particular, the upper part of the ordination included a cluster with B. argenteum, B. pseudotriquetrum, H. heimii and Didymodon brachyphyllus, while the species Syntrichia magellanica, S. sarconeurum, Ceratodon purpureus and Schistidium antarctici clustered in the lower part of the ordination. Both classification and ordination showed that sites in the slope province generally exhibited lower species diversity (mean of 2 species for each site and a total of 10 species) than those of the coast province (mean of 3.8 species and a total of 17 species).
Biplot (species and sites) showing the results of PCA of the bryophyte species and the 62 investigated sites of the Ross Sector (70°–84°S). Legend for Species abbreviations: Bry.arch Bryum archangelicum, Bry.arg Bryum argenteum, Bry.pse Bryum pseudotriquetrum, Cer.pur Ceratodon purpureus, Cosc.law Coscinodon lawianus, Cosc.ref Coscinodon reflexidens, Dyd.bra Dydimodon brachyphyllus, Gri.pla Grimmia plagiopodia, Hen.ant Hennediella antartica, Hen.hei Hennediella heimii, Ort.sess Orthogrimmia sessitana, Schi.ant Schistidium antartici, Schi.urn Schistidium urnulaceum, Syn.mag Syntrichia magellanica, Syn.sar Syntrichia sarconeurum. For site acronyms, please refer to the legend of Fig. 2
The RDA analysis (with the first two axes explaining the 85.9 % of cumulative variance in species–environment relations) again demonstrated that sample location (coast vs. slope province) was more effective than latitude or elevation in classifying both species and sites (Fig. 5). The cluster of coastal sites was characterized by the occurrence of Syntrichia magellanica, a species exclusive to the coast province, and by a high frequency of species distributed between 70°S and 78°S (B. argenteum, H. heimii, Dydimodon brachyphyllus). The coastal sites were aligned following latitude and could be further grouped into two sub-clusters, one including sites mainly located between 70°S and 75°S (northern coastal sub-cluster) and a second group including only sites located between 76°S and 78°S (southern coastal sub-cluster). There was a correspondence between site clustering, floristic composition and dominance (higher frequency of Syntrichia magellanica in the northern coastal cluster and of H. heimii and D. brachyphyllus in the southern coastal cluster). However, there was no clear relationship between site clustering and bryophyte diversity as, within each cluster, site diversity varied widely.
Triplot showing the results of redundancy analysis (RDA) carried out using 9 bryophyte species, 63 sites and 4 environmental descriptors for the Ross Sector. For species and site acronyms, refer to the legend of Fig. 4. Environmental descriptors: Coast coastal province, Slope slope province, Elevat elevation, Latit latitude
Intra-regional and regional diversity patterns
Bryophyte diversity at the local scale was not related to latitude. The highest species number was recorded at 74°S (Harrow Peak), and the richer sites mainly occurred between 72°S (Cape Hallett) and 77°S (several sites) (Fig. 6). In most cases, the richest sites exhibited similar floristic composition, as confirmed also by the Jaccard similarity index (data not shown), independent of their location across the latitudinal transect.
No latitudinal diversity relationship was apparent at either regional or intra-regional scale (coast vs. slope province). Rather, a bimodal distribution was apparent, although with different patterns seen when comparing the entire Ross Sector with the coast and slope provinces, as confirmed by the diversity analysis by latitude bands (Fig. 7). The coast province exhibited the highest diversity at 77°S and a second lower peak at 74°S, with two site clusters (northern coastal sites at 70–75°S and southern coastal sites at 76–78°S), as also seen in the floristic composition (Fig. 3, 4). The slope province showed the highest diversity at 77°S and a second lower peak at 83°S, with two site clusters—the northern slope sites (74–78°S) and the southern slope sites (83–84°S) (Figs. 3, 4).
A group of five ubiquitous species was distributed across the entire transect (70–84°S) (B. pseudotriquetrum, Ceratodon purpureus, Grimmia plagiopodia, Schistidium antarctici and S. sarconeurum), while Schistidium urnulaceum occurred only at 83°S. A number of species were not recorded at latitudes higher than 78°S, with four only occurring north of 75°S (Coscinodon lawianus, Orthogrimmia sessitana, B. archangelicum and B. dichotomum). Patterns of genus-level occurrence (Table 1) exhibited trends similar to those seen with species (especially in the slope province), as in most cases only one or two species are recorded for each genus, except Bryum which includes four different species. The higher taxa were constant from 83°S to 72°S and decreased at both ends of the transect (Table 1).
Intra-regional biogeographic elements
In both slope and coast provinces, the species pool included four biogeographic elements: (a) Antarctic endemic species (four in the Ross Sector, with only one species, Coscinodon lawianus, endemic to continental Antarctica, the other three occurring also in the maritime Antarctic), (b) species restricted to the Southern Hemisphere, (c) bipolar species and (d) cosmopolitan species (Table 2). Within each element, there was no trend with latitude in either the slope or the coastal province.
Bryophyte diversity and biogeography at continental scale
We extended our analyses at continental scale including the recognized geographic sectors of the Antarctic continent. Species richness showed no clear trend across the different continental sectors (Fig. 8). The highest diversity was recorded in the Ross Sector (18 species) and the lowest in the Scotia Sector (3 species), although this sector is close to the Antarctic Peninsula (maritime Antarctic) where bryophyte diversity is much higher (110–115 species). Among the 24–27 species reported for continental Antarctica, there is a common pool of species occurring in most sectors (C. reflexidens, Bryum dichotomum, B. pseudotriquetrum, H. heimii, Ortogrimmia sessitana, S. sarconeurum). Very few species occur only in one sector of continental Antarctica and also in the maritime Antarctic, including Andreaea gainii and A. regularis (Byrd Sector), B. archangelicum and C. pyriformis (Ross Sector), Dryptodon fuscoluteus and Funaria hygrometrica (Wilkes Sector), Plagiothecium cavifolium (Maud Sector), and Pohlia wilsonii (Enderby Sector).
Bryophyte diversity (total species number) and species biogeography (using the same four categories adopted for the Ross Sector, after Ochyra et al. 2008) among the different arbitrary geographic sectors of the continental Antarctica. Species biogeography: BIP bipolar, COSMO cosmopolitan, END Antarctic endemic, S_HEM Southern Hemisphere
The Jaccard similarity index computed across the sectors of coastal continental Antarctica (Table 3) indicates that the highest similarity occurs between neighboring sectors (Maud–Enderby, Enderby–Wilkes, Wilkes–Ross, Ross–Byrd) or among non-neighboring but connected sectors (Byrd–Wilkes through Ross and Maud–Wilkes through Enderby). The Scotia Sector shows very low similarity with all other sectors. Furthermore, the maritime Antarctic shows low floristic similarity with the various sectors of continental Antarctica.
Discussion
The Ross Sector provides a unique opportunity to assess bryophyte diversity patterns along latitudinal and environmental gradients in continental Antarctica and to place these in the context of general climatic, ecological, historic and biogeographic factors.
Intra-regional and regional floristic and diversity patterns
Diversity patterns depend on the scale of analysis, because the mechanisms affecting species richness are scale sensitive (Rahbek 2005), with stronger and steeper gradients at the regional than at the local scales (Hillebrand 2004). Within the Ross Sector, we identified several “hot spots” of bryophyte diversity, but no significant relationship with latitude (Fig. 6). Despite the existence of an almost linear climatic gradient with latitude, the lack of any associated pattern in bryophyte diversity is consistent with Peat et al. (2007). The hot spots identified appear to be related to the availability of favorable local microclimatic conditions. Continental Antarctic terrestrial bryophyte communities are mainly restricted to low-lying areas with adequate free water (e.g., Wasley et al. 2006). In our study, sites with the highest diversity were located at different latitudes along the coastal province and exhibited similar local conditions relating to aspect (mainly N, NE, E), availability of free water during the summer and relatively high ground water content (range 3–30 %; e.g., Cannone et al. 2008). At such sites, direct bryophyte colonization on moraine or alluvial deposits is favored by warmer microclimatic conditions due to sheltered locations and favorable microrelief (Seppelt et al. 2010). Similar trends are also exhibited by soil-inhabiting springtails in the Ross Sector, with the distribution of each species being fragmented, reflecting the availability of suitable habitat (e.g., Torricelli et al. 2010).
At intra-regional scale, bryophyte diversity is higher throughout the coast province than in the slope province. Within each, the sites are divided into two sub-clusters although this clustering is based on site floristic composition and not on their overall diversity (Fig. 5). This site clustering is compatible with the distribution patterns of individual bryophyte species within the Ross Sector, with some species recorded between 70 and 78°S and others between 70 and 75°S. Only one species (Schistidium urnulaceum) is exclusively recorded at the high latitude of 83°S, while the remaining five species occur across the entire regional transect. This clustering into northern and southern sites along the coast province is consistent with the two regions proposed by Adams et al. (2006), based on biogeographic separation among some biota due to important dispersal barriers (such as the Darwin Glacier at 79°S and the Drygalsky Ice Tongue at 75°S) (e.g., Stevens et al. 2006; Demetras et al. 2010). Similar distribution patterns were described for the microarthropod fauna along a transect from the Queen Maud Mountains to northern VL, with a boundary detected at 75°S corresponding with the Drygalsky Ice Tongue (see Adams et al. 2006). Demetras et al. (2010) suggested the existence of a major barrier to mite dispersal located north of Darwin Glacier (79°S), which could also coincide with the Ross Ice Shelf (78°S). The barriers we identify for vegetation may not coincide with physical barriers to dispersal identified for microfauna, but rather with physiologic constraints linked to water availability. Our site clustering within the Ross Sector shows considerable overlap with the precipitation gradient described by Monaghan et al. (2006; referring to the period 1955–2004), which identified three sub-zones (Fig. 1), (a) from the Transantarctic Mountains to the inner Dry Valleys (50–100 mm/year), (b) from the Dry Valleys to Drygalsky Ice Tongue (100–200 mm/year) and (c) from north of Drygalsky Ice Tongue to Cape Hallett (200–300 mm/year).
The bryophyte diversity patterns identified at intra-regional level may be related to the exposure age of the substrata, with survival in refugial locations such as nunataks allowing long-term persistence and regional re-colonization during inter-glacial periods. Such scenarios have been proposed for springtails, mites and chironomids in this region or elsewhere in Antarctica (reviewed by Adams et al. 2006; Convey et al. 2008), as well as local extinction of biota during glacial periods, in most cases linked to EAIS dynamics. The bryophyte diversity peaks at 77°S (Dry Valleys) in both provinces could be related to the longer duration of surface exposure available for colonization and development in this area, estimated to be at least about 13.85 My (Lewis et al. 2008).
Antarctic diversity and biogeography at continental scale
At continental scale, the Ross Sector shows the highest bryophyte diversity (18 species) of all continental sectors. However, no recorded species are exclusive to this sector, whose diversity also includes the largest numbers of both bipolar and Antarctic endemic species (Fig. 8). The higher diversity of the Ross Sector compared with the other continental Antarctic sectors is not associated with latitude (the ice-free areas of this sector extend to the highest latitudes, and the mean latitude of this sector is greater than that of the other sectors) or with precipitation (Monaghan et al. 2006). Another potentially important influence on diversity is likely to be the available ice-free area. The Ross Sector includes the largest ice-free area (>21,000 km2), followed by the Enderby Sector (7,363 km2, 13 bryophyte species). However, the Scotia Sector is only slightly smaller (6,314 km2) but includes the lowest recorded bryophyte diversity (3 species). These figures suggest that while the available ice-free area may influence diversity levels, it is not sufficient to explain the observed diversity trends at the continental scale. Species distribution and the similarity in floristic composition among sectors (Table 3) emphasize that the Scotia Sector is the poorest and most distinct sector, even though it is geographically closest to the rich Antarctic Peninsula. This may suggest that also bryophytes are influenced by the existence of an important and ancient barrier, the Gressitt Line, a boundary identified at the base of the Antarctic Peninsula and indicative of ancient and different evolutionary origins of elements of the biota occurring on either side (Chown and Convey 2007).
The relatively high similarities of the bryophyte floras of adjacent continental sectors (Maud–Enderby, Enderby–Wilkes, Wilkes–Ross, Ross–Byrd) or connected sectors (such as Byrd–Wilkes through Ross and Maud–Wilkes through Enderby) suggest the existence of a potential route for species dispersal along the coast of continental Antarctica. Further support is provided by very low floristic similarity between the bryophyte flora of the maritime Antarctic and most sectors of the continental Antarctica other than the Ross and Byrd Sectors, which may also suggest that species dispersal has occurred from the Antarctic Peninsula westward along the coast of the continental Antarctica.
The high levels of floristic similarity among non-adjacent continental sectors may support the existence of refugia, from which dispersal could have occurred during inter-glacial periods. The distribution of the only continental Antarctic endemic, Coscinodon lawianus (Willis) Ochyra, is consistent with this hypothesis, occurring in all sectors except Wilkes and Byrd. While Scotia, Maud and Enderby are adjacent sectors, Ross is not directly connected through the coast to these sectors, and the only potential connection is through the isolated ranges and nunataks between the Scotia Sector, the Transantarctic Mountains and the Ross Sector.
The Ross Sector exhibits the highest Antarctic endemism rate (including all four endemic species known from continental Antarctica) among the continental Antarctic sectors. However, the rate of endemism may not be informative in reconstructing the evolution of the bryophyte flora in Antarctica, as bryophytes show atypical patterns of endemism compared to other groups of Antarctic biota (Chown and Convey 2007; Convey et al. 2009). The Ross Sector also includes the largest proportion of bipolar species. Species with highly disjunct distributions may demonstrate the long-term success of natural dispersal strategies (e.g., Barnes et al. 2006), while the occurrence of endemic species may indicate in situ evolution (Stevens et al. 2006).
Both overall and site-specific bryophyte diversities along the Ross Sector transect are considerably lower than values found anywhere in the Antarctic Peninsula and associated island archipelagoes (maritime Antarctic) (Ochyra et al. 2008), despite these two latitudinal transects being of similar length and geographic extent. Underlying these inter-regional differences, it is notable that although the maritime Antarctic shares a similar climatic gradient (~1.1 °C/latitude), it has much higher MAAT (ranging from −2 °C at the South Sandwich Islands, 56–60°S, to −20 °C in the southeastern Antarctic Peninsula) than that of the Queen Maud Range–Victoria Land transect. Such large differences in bryophyte species diversity between the Antarctic Peninsula and continental Antarctica may be underlain by the climatic differences between these two regions. However, the greater geographic insulation of the Antarctic continent may contribute to explaining the differences in diversity. The role of geographic isolation is evident when comparing patterns of bryophyte species diversity between continental Antarctica, the Antarctic Peninsula, the wider maritime Antarctic and the sub-Antarctic islands. Among the 246 species recorded in the sub-Antarctic islands, only 15 occur in continental Antarctica (these accounting for c. 62 % of the entire flora of continental Antarctica), 36 in the Antarctic Peninsula and 61 in the wider maritime Antarctic. The more pronounced reduction in bryophyte species diversity in continental Antarctica could therefore be due to a combination of historical evolutionary origins (longer geographic isolation) and contemporary ecological conditions (harsher climate), which are not mutually exclusive.
References
Adams B, Bardgett RD, Ayres E et al (2006) Diversity and distribution of victoria land biota. Soil Biol Biochem 38:3003–3018
Adlam LS, Balks MR, Seybold CA, Campbell DI (2010) Temporal and spatial variation in active layer depth in the McMurdo Sound Region, Antarctica. Antarct Sci 22:45–52
Balks MR, Cummings V, Green TGA et al (eds) (2006) The latitudinal Gradient Project (LGP). Antarct Sci 18:198
Barker PF, Diekmann B, Escutia C (2007) Onset of Cenozoic Antarctic glaciation. Deep-Sea Res Part II Top Stud Oceanogr 54:2293–2307
Barnes DKA, Hodgson DA, Convey P et al (2006) Incursion and excursion of Antarctic biota: past, present and future. Glob Ecol Biogeogr 15:121–142
Cannone N (2006) A network for monitoring terrestrial ecosystems along a latitudinal gradient in continental Antarctica. Antarct Sci 18:549–560
Cannone N, Seppelt RD (2009) A preliminary floristic classification of Northern and Southern Victoria Land vegetation (continental Antarctica). Antarct Sci 20:553–562
Cannone N, Guglielmin M, Gerdol R (2004) Relationships between vegetation patterns and periglacial landforms in north-western Svalbard. Polar Biol 4:1–22
Cannone N, Wagner D, Hubberten HW, Guglielmin M (2008) Biotic and abiotic factors influencing soil properties across a latitudinal gradient in Victoria Land, Antarctica. Geoderma 144:50–65
Chown SL, Convey P (2007) Spatial and temporal variability across life’s hierarchies in the terrestrial Antarctic. Philos Trans R Soc B 362:2307–2331
Convey P, Gibson J, Hillenbrand CD et al (2008) Antarctic terrestrial life—challenging the history of the frozen continent? Biol Rev 83:103–117
Convey P, Stevens MI, Hodgson DA et al (2009) Exploring biological constraints on the glacial history of Antarctica. Quat Sci Rev 28:3035–3048
Demetras NJ, Hogg ID, Banks JC, Adams BJ (2010) Latitudinal distribution and mitochondrial DNA (COI) variability of Stereotydeus spp. (Acari: Prostigmata) in Victoria Land and the central Transantarctic Mountains. Antarct Sci 22:749–756
Frank KT, Petrie B, Shackell NL (2007) The ups and downs of trophic control in continental shelf ecosystems. Trends Ecol Evol 22:236–242
Green TGA, Sancho LG, Turk R et al (2011) High diversity of lichens at 84°S, Queen Maud Mountains, suggests preglacial survival of species in the Ross Sea region, Antarctica. Polar Biol 34:1211–1220
Guglielmin M (2006) Ground surface temperature (GST), active layer and permafrost monitoring in continental Antarctica. Permafr Periglac 17:133–143
Hall BL, Baroni C, Danton GH (2004) Holocene relative sea-level history of the Southern Victoria Land coast, Antarctica. Glob Planet Change 42:241–263
Hillebrand H (2004) On the generality of the latitudinal diversity gradient. Am Nat 163:192–211
Hills SFK, Stevens MI, Gemmill CEC (2010) Molecular support for Pleistocene persistence of the continental Antarctic moss Bryum argenteum. Antarct Sci 22:721–726
Howard-Williams C, Hawes I, Gordon S (2010) The environmental basis of ecosystem variability in Antarctica: research in the latitudinal gradient project. Antarct Sci 22:591–602
Ingolfsson O, Hjort C, Berkman PA et al (1998) Antarctic glacial history since the Last Glacial Maximum: an overview of the record on land. Antarct Sci 10:326–344
Lewis AR, Marchant DR, Ashworth G et al (2008) Mid-Miocene cooling and the extinction of tundra in continental Antarctica. Proc Natl Acad Sci USA 105:10676–10680
Mittelbach GG, Schemske DW, Cornell HV et al (2007) Evolution and the latitudinal diversity gradient: speciation, extinction and biogeography. Ecol Lett 10:315–331
Monaghan AJ, Bromwich DH, Wang SH (2006) Recent trends in Antarctic snow accumulation from Polar MM5. Philos Trans R Soc A 364:1683–1708
Ochyra R (2004) Antipodal mosses: XIV. On the taxonomic status of Grimmia lawiana (Bryopsida: Grimmiaceae) from the continental Antarctic. Pol Polar Res 25:111–122
Ochyra R, Smith RIL, Bednarek-Ochyra H (2008) The illustrated moss flora of Antarctica. Cambridge University press, Cambridge, p 685
Peat HJ, Clarke A, Convey P (2007) Diversity and biogeography of the Antarctic flora. J Biogeogr 34:132–146
Qian H, Ricklefs RE (2007) A latitudinal gradient in large-scale beta diversity for vascular plants in North America. Ecol Lett 10:737–744
Rahbek C (2005) The role of spatial scale and the perception of large-scale species-richness patterns. Ecol Lett 8:224–239
Rozzi R, Armesto JJ, Goffinet B et al (2008) Changing lenses to assess biodiversity: patterns of species richness in sub-Antarctic plants and implications for global conservation. Front Ecol Environ 6:131–137
Seppelt RD, Türk R, Green TGA et al (2010) Lichen and moss communities of Botany Bay, Granite Harbour, Ross Sea, Antarctica. Antarct Sci 22:691–702
Shaw AJ, Cox CJ, Goffinet B (2005) Global patterns of moss diversity: taxonomic and molecular inferences. Taxon 54:337–352
Stevens MI, Greenslade P, Hogg ID, Sunnucks P (2006) Examining Southern hemisphere springtails: could any have survived glaciation of Antarctica? Mol Biol Evol 23:874–882
Strand K, Passchier S, Näsi J (2003) Implications of quartz grain microtextures for onset Eocene/Oligocene glaciation in Prydz Bay, ODP site 1166, Antarctica. Palaeogeogr, Palaeocl 198:101–111
Sugden DE, Denton GH (2004) Cenozoic landscape evolution of the Convoy Range to Mackay Glacier area, Transantarctic Mountains: onshore to offshore synthesis. Geol Soc Am Bull 116:840–857
Ter Braak CJF, Smilauer P (1998) CANOCO reference manual and user’s guide to CANOCO for windows: software for canonical community ordination (version 4). Centre for Biometry, Wageningen, p 351
Torricelli G, Frati F, Convey P et al (2010) Population structure of Friesea grisea (Collembola, Neanuridae) in the Antarctic Peninsula and Victoria Land: evidence for local genetic differentiation of pre-Pleistocene origin. Antarct Sci 22:757–765
Troedson AL, Smellie JL (2002) The Polonez Cove formation of King George Island, Antarctica: stratigraphy, facies and implications for mid-Cenozoic cryosphere development. Sedimentology 49:277–301
Wasley J, Robinson SA, Lovelock CE, Popp M (2006) Some like it wet—biological characteristics underpinning tolerance of extreme water stress events in Antarctic bryophytes. Funct Plant Biol 33:443–455
Acknowledgments
We are very grateful to the PNRA (Progetto Nazionale Ricerche in Antartide) for funding and logistical support. We thank Dr Helen Peat for providing data from the BAS Antarctic Plant Database. Anonymous referees are thanked for their constructive comments. This paper also contributes to the BAS core “Ecosystems” and the SCAR “Evolution and Biodiversity in the Antarctic” programmes.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cannone, N., Convey, P. & Guglielmin, M. Diversity trends of bryophytes in continental Antarctica. Polar Biol 36, 259–271 (2013). https://doi.org/10.1007/s00300-012-1257-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-012-1257-5