Abstract
How predators vary search patterns in response to prey predictability is poorly known. For example, marine invertebrates may be predictable but of low energy value, while fish may be of higher energy value but unpredictable at large (pelagic schools) or small (solitary benthics) spatial scales. We investigated the search patterns of the thick-billed murre (Uria lomvia), an Arctic seabird feeding on invertebrates, pelagic fish, or benthic fish. Foraging ranges at the Coats Island colony are generally smaller (<240 min per trip) than at larger colonies, and many birds specialize in foraging tactics and diet. Underwater search times for benthic fish were higher than for pelagic fish or invertebrates while above-water search times for pelagic fish were higher than for benthic fish or invertebrates. There were few stops during trips. Total trip time, flying time, number of flights, and number of dives were intercorrelated and increased with prey energy content, suggesting that longer trips involved fewer prey encounters due to selection of higher-quality, but rarer, prey items. Flight times were not Lévy-distributed and seabirds may have used area-restricted searches. The high degree of specialization, apparent absence of information center effects, and reduced above-water searching times may be linked to the relatively small colony size and the resulting short commuting distances to feeding areas, leading to greater prey predictability. We concluded that prey predictability over various scales affected predator search patterns.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Predators are continually presented with choices for how to move (or not move) to maximize prey capture rates (Ropert-Coudert et al. 2004; Ohashi et al. 2007; Garthe et al. 2007; Catania et al. 2008). These choices form the “search pattern” of the predator. Search patterns are particularly important in marine ecosystems, where predators must search in three dimensions (Sjoberg and Ball 2000; Simpkins et al. 2001; Davoren et al. 2003a, b) and where prey items (e.g., fish) are patchily distributed, meaning that search patterns represent decisions on whether to search a given patch or move on to another patch (Mori et al. 2002, Grémillet et al. 2004; Weimerskirch et al. 2005a, b). During the breeding season, search patterns may be under particularly strong selection, as adult marine mammals and birds minimize time away from offspring while maximizing energy delivered to offspring (Wilson et al. 2005; Garthe et al. 2007; Jaquemet et al. 2007).
Search patterns will be partially dependent on the spatial and temporal predictability of prey aggregations. Marine animals searching over small spatial and temporal scales, where prey aggregations are likely to persist between foraging bouts, may use memory to return to the same patches that were successful on previous foraging bouts (Davoren et al. 2003a, b; Weimerskirch 2007; Woo et al. 2008). Marine animals searching over larger spatial and temporal scales, where prey aggregations are unlikely to persist between foraging bouts, are less likely to return to the same patches and require other sources of information to locate prey items (Fauchald et al. 2000; Weimerskirch et al. 2005a, b; Weimerskirch 2007; Mallory et al. 2008). For example, unsuccessful or naïve foragers may obtain information about food sources by following successful foragers (Information Center Hypothesis or ICH, sensu Ward and Zahavi 1973; Birkhead 1985; Buckley 1997a, b), providing an explanation for why colonial breeding increases foraging efficiency only when food occurs in a few dense but ephemeral patches (Barta and Szép 1992). Information transfer may occur away from the colony because animals often gather some distance from a colony before departing on foraging flights (“information halo” sensu Burger 1997) because outgoing birds can cue to the directions of incoming birds (Gaston and Nettleship 1981) or because outgoing birds cue in on the location of actively foraging animals far away from the colony (“local enhancement”). To illustrate, murres usually descend to the water within a 1-km area of the colony (“splashdown area”) after a brooding shift and time spent in this area may be an opportunity for information transfer (“information halo”; Burger 1997).
Because animals are able to locate prey or feeding aggregations that occur within visual range, predators are expected to move directly to patches that are within this fixed radius (“local enhancement”) but randomly if there are no visible patches within that radius. Search patterns obeying this concept could result in a Lévy search pattern, characterized by flight distances that follow an inverse-square power law (Reynolds 2005; Bénichou et al. 2006). Animals as diverse as albatrosses (Viswanathan et al. 1996), bumblebees (Viswanathan et al. 1999), monkeys (Ramos-Fernández et al. 2004), whales (Austin et al. 2004), plankton (Bartumeus et al. 2003), fishing fleets (Bertrand et al. 2005, 2007), and deer (Viswanathan et al. 1999, Mårell et al. 2002) have been reported to obey Lévy search patterns. However, reanalysis of the data from three studies showed that flight times actually followed an exponential distribution, implying that food patches were encountered randomly (Edwards et al. 2007).
Excluding animals feeding on sessile prey (Halsey et al. 2003; Heath et al. 2006, 2007), predators on pelagic and benthic prey have very different search patterns (Womble and Sigler 2006; Gende and Sigler 2006; Elliott et al. 2008c). Pelagic prey items congregate in enormous concentrations, but these concentrations may be extremely dispersed and their location may vary over large spatial and temporal scales (Gende and Sigler 2006; Womble and Sigler 2006; Cotté et al. 2007). Thus, for pelagic foragers, locating patches may be difficult, but once a patch is located capturing prey items is easy (Irons 1998; Weimerskirch et al. 2005a, b; Weimerskirch 2007). Benthic prey items often occur in much smaller concentrations, but they persist at the same location owing to habitat requirements (e.g., reefs, sandy bottoms, kelp forests) over large spatial and temporal scales (Davoren et al. 2003a, b; Womble and Sigler 2006; Gende and Sigler 2006). Thus, for benthic foragers, locating patches may be easy, but once a patch is located finding an accessible prey item may be difficult. Spawning pelagic fish, which may be both highly abundant and highly predictable, creating “hot spots” (Davoren et al. 2003a, b, 2006; Womble and Sigler 2006; Gende and Sigler 2006), are an exception to this generalization.
The search patterns of marine predators tend to be characterized by long travel times interspersed by area-restricted searches over small spatial scales (Weimerskirch et al. 2005a, b; Pinaud and Weimerskirch 2007; Cotté et al. 2007), potentially resulting in Lévy search patterns (Plank and James 2008). Foraging site fidelity is greater for those seabirds feeding at relatively small spatial and temporal scales because prey is more predictable at smaller scales (Weimerskirch 2007). For example, Grémillet et al. (2004, 2005) showed that prey encounter rates are exceptionally high for a seabird foraging on benthic prey over small spatial scales, suggesting little need for searching. Many high-latitude seabirds show a high degree of foraging site fidelity (Irons 1998; Hamer et al. 2001; Garthe et al. 2007).
Here, we investigate the search patterns of an arctic seabird, the thick-billed murre (Uria lomvia), at a colony where it feeds on both pelagic and benthic prey items (Gaston and Bradstreet 1993; Gaston et al. 2003; Hipfner et al. 2006). Most individuals forage relatively close to the colony relative to other colonies (<40 km; Elliott et al. 2008c). Consistent with the idea that birds foraging over small spatial scales have greater specialization, murres at this site show a high degree of fidelity to foraging strategies (dive depth, flight time, dive shape), locations, and prey types regardless of whether the prey items are consumed by adults or chicks (Woo et al. 2008). At large colonies (foraging ranges >100 km), murres stop periodically on the outbound trip to sample the environment (Benvenuti et al. 1998; Falk et al. 2000, 2002). In contrast, due to the smaller spatial scale of our study colony, we predicted that there would be few stops on outbound trips. As previous investigators had shown an inverse power law frequency distribution for total trip times (Davoren and Montevecchi 2003; Davoren et al. 2003a, b), we predicted that flight times would follow a power law distribution. We also predicted that birds returning with pelagic prey items would have more flight bouts and less bottom time than birds returning with benthic prey items, reflecting more above-water search time and less underwater search time. Finally, we examine the importance of an ICH-type mechanism for information exchange at this colony by describing behavior in splashdown areas relative to at-sea searching behavior as well as the synchrony of departures and arrivals of adults at the colony, the latter suggesting the type of following behavior predicted by the ICH (Ward and Zahavi 1973; Birkhead 1985; Götmark 1990). Another approach is to assess whether incoming directions were significantly clumped over time (see Davoren et al. 2003a,b), but this was not possible as virtually all birds feed to the west of the colony (Elliott et al. 2008c).
Materials and methods
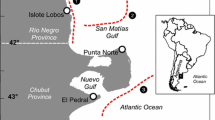
Our observations were made at the Coats Island thick-billed murre (U. lomvia) colony (30,000 breeding pairs; 62° 57′ N, 82° 00′ W), Nunavut, Canada (Gaston et al. 2003, 2005; Hipfner et al. 1997; Gaston and Hipfner 2006a, b) during the breeding seasons 2000–2007. At least three continuous (24 or 48 h) observational sessions of breeding sites were carried out from a blind situated on the Q study plot, within 5 m of the birds (Hipfner et al. 2006). We did not conduct feeding watches when it was too dark to see deliveries (roughly 0100–0200 hours in late July; 2300–0400 in mid-August) because chicks are rarely fed at this time (Gaston and Bradstreet 1993; Gaston et al. 2003). During these observation sessions, prey items delivered to chicks were identified whenever possible.
Time–depth–temperature recorders
In conjunction with these observations, murres were caught with a noose pole (2004: n = 23; 2005: n = 33; 2006: n = 80; 2007: n = 37; total foraging trips = 729). Handling time was always less than 10 min and usually less than 5 min. We secured LOTEK 1100LTD time–depth–temperature recorders (TDRs; Lotek Marine Technology, St. John’s, Newfoundland, Canada) with duct tape to plastic bands that were attached to the legs of murres (murres do not use their legs for underwater propulsion). The TDRs were cylindrical (mass = 4.5 g; diameter = 1 cm; length = 3.3 cm; sampling interval = 3 s; absolute error ± 2 m) and attached parallel to the leg with the rounded end facing toward the body and the pressure sensor facing toward the foot. We corrected for drift using a custom-built Excel macro and dives shallower than 3 m were ignored (Elliott and Gaston 2009). Whereas back-mounted TDRs are known to impact murre provisioning rates, trip duration, mass loss, and dive behavior (Tremblay et al. 2005; Hamel et al. 2004; Paredes et al. 2004; Elliott et al. 2007, 2008b), our smaller leg-mounted devices had no measurable effect on any of these parameters (Elliott et al. 2007, 2008b, c). We used the temperature log from the TDR to determine whether the bird was on the water, in the air, or at the colony (Tremblay et al. 2005; Elliott et al. 2007, 2008c). Because we also knew when the bird arrived at the colony from the continuous watches, we were able to calibrate the temperature log in many cases. A summary date file of TDR trips appears as ESM Fig. S1.
Prey type
During feeding watches, prey items delivered to chicks were visually identified whenever possible. Size was estimated in relation to the length of the white streak on the bill (approximately 5 cm, Gaston et al. 2003; Hipfner et al. 2006). Prey energy content was calculated from species-specific energy densities and mass–length regressions developed at Coats Island (Elliott and Gaston 2008). By assuming that the last dive or dive bout prior to a prey delivery represented the foraging behavior associated with that prey item, we were able to determine dive depth, dive shape, and flight time associated with each prey item. Support for the assumption that prey items were captured on the final dive is provided by the observation that the final dive prior to prey delivery tends to be shorter, but no deeper, than other dives, suggesting that the final dive represents a premature abortion following a successful prey capture event (Elliott et al. 2008a, b, c). We categorized prey items into pelagic (Arctic cod Boreogadus saida and sand lance Ammodytes sp.), benthic (sculpin, shannies, blennies, and poachers), or invertebrate (shrimp, squid, and amphipods) groupings. Sand lance were considered to be pelagic because they were generally captured after u-shaped dives (Fig. 2b in Elliott et al. 2008c—mislabeled as “U-shaped” in the legend), and because they were almost always zero- or one-age class, which are usually captured in pelagic schools (Robards et al. 1999; Elliott et al. 2008c; Elliott and Gaston 2008). Fish captured during u-shaped dives would also be considered pelagic based on classification schemes using wiggles (Halsey et al. 2007). We separated invertebrates from fish because invertebrates are superabundant, requiring little above-water or underwater searching compared with fish (Elliott et al. 2008c; Elliott and Gaston 2008). Capelin (Mallotus villosus) was not categorized because they can occur as either benthic or pelagic schools (Davoren et al. 2003a, b; Hedd et al. 2009). Furthermore, both capelin and invertebrates were usually captured after V-shaped dives, meaning that searching presumably also occurs during transit to and from the surface and that our values for bottom time likely underestimate total underwater search time; auks sometimes capture pelagic prey from below (Burger et al. 1993).
Synchrony of arrivals and departures and splashdown behavior
To test the ICH, we examined arrival, departure, and splashdown behavior. We put more emphasis on arrival synchrony than departure synchrony because most birds depart on foraging trips from the splashdown area rather than the colony and, thus, synchrony of departures from the colony may not reflect synchrony of departures for feeding (Gaston and Nettleship 1981; Burger 1997). To test for synchrony of arrivals and departures, we used the 4-h period between 0600 and 1000 hours because no autocorrelation occurred during this interval. Time intervals between successive arrivals and successive departures were calculated for plot Q during 1997–2007. The frequencies of occurrence of time intervals were plotted against the interval lengths to give the simulated distributions of time intervals between successive arrivals and time intervals between successive departures. To determine if these distributions showed some clumping of arrivals or departures, a computer model simulating random arrivals and departures was created using Visual Basic. This model generated random numbers between zero and 240 representing bird arrival or departure times in a 4-h period (240 min in 4 h). The model generated a specific number of random numbers set as the mean number of events (either arrivals or departures) observed to occur within a 4-h period. These numbers were ordered ascendantly and the intervals between successive simulated arrivals or departures were calculated. This procedure was repeated 200 times to generate the expected distribution of intervals between arrivals or departures, assuming independent behavior. We also calculated the dispersion (ratio of standard deviation to the mean) for feeding intervals for individual murres (Hipfner et al. 2006).
To test whether birds were lingering in the splashdown area to obtain information on likely feeding areas, we compared the duration of the splashdown period with the subsequent outbound flight duration, number of flights, and duration of between-dive flights, assuming that uncertainty in prey location increases with these parameters and that they are therefore indices for above-water searching. For example, birds accessing predictable food sources (e.g., invertebrates) would not need information transfer during the splashdown period and, thus, would remain in this area for much shorter durations than those accessing unpredictable sources (e.g., pelagic fish). We also compared time in the splashdown area between birds whose breeding sites were located below other birds (and which therefore become dirtier and presumably require longer periods of cleaning) and those breeding on sites where they generally remained unsoiled.
Searching metrics
In contrast with other studies showing that genders eat different prey (Jones et al. 2002, Lewis et al. 2002, Paredes et al. 2006), chick prey items are usually larger than adult prey items (Hobson 1993; Davoren and Burger 1999; Wilson et al. 2004) leading to bimodal foraging distributions (Ropert-Coudert et al. 2004; Steen et al. 2007), murres at Coats Island show few gender differences, no difference between adult and chick prey items and have highly specialized foraging behavior across trips (Woo et al. 2008), meaning that we did not need to distinguish between searching for self-feeding and searching for chick provisioning. Nonetheless, self-feeding dives are shorter than chick-provisioning dives (Elliott et al. 2008b). Foraging behavior of thick-billed murres is stereotyped for a given prey item and occurs along three major axes (Elliott et al. 2008c), representing prey depth (measured as dive depth), benthic or pelagic foraging (dive shape), and distance from the colony (flight time). As all other foraging variables are closely correlated with one of these parameters, variability in foraging behavior can be monitored by including only these three measures. Metrics for underwater searching were bottom time per dive and number of dives per bout (Table 1). Bottom time per dive was defined as time spent within 90% of maximum depth. Unlike murres that catch invertebrates and capelin, those taking Arctic cod and sand lance usually have u- or U-shaped dives, with a clear bottom phase, so it is unlikely that searching occurs during transit (Elliott et al. 2008c). Metrics for above-water searching were outbound flight time, number of flights per trip, total trip time, total flying time, the percentage of trips with flights between dive bouts, the duration of flights between dive bouts, and the difference between outbound and inbound flight times (Table 2). For both searching metrics, we used the residual of the metric on energy content for all statistical analyses to control for the effect of prey energy value.
To examine whether murres followed a Lévy search pattern, we examined the shape of the log-frequency and log-flight time relationship (Reynolds 2005; Bénichou et al. 2006). Specifically, we used the maximum likelihood method presented by Edwards et al. (2007) to compare the statistical support (∆AIC value) for a linear versus shifted gamma distribution with exponential tail. Considering the tail to start at a (e.g., for x ≥ a), the power law f(x) and exponential g(x) tails have probability density functions:
Maximum likelihood estimators for λ and μ can be solved using Eqs. 5 and 6 from Edwards et al. (2007). We used the methodology presented therein on both all data pooled and only the data within a single individual, for all individuals with at least 30 recorded flights.
Results
Splashdown behavior and arrival/departure synchrony
Most brooding shifts were followed by a period on the water with no diving activity (Fig. 1). The majority (97%) of shifts longer than 120 min were followed by splashdown periods, while no shift shorter than 30 min was followed by splashdown. Thus, splashdown duration increased with shift duration (t 146 = 4.93, p < 0.0001, r 2 = 0.17, ESM Fig. S2), but not for shifts longer than 120 min (t 98 = 0.90, p = 0.37, r 2 = 0.01); birds did not use the splashdown after foraging trips without at-colony shifts. Time spent in the splashdown area was generally short (8.0 ± 0.4 min) and duration was not related to above-water search behavior, such as number of flight bouts (t 146 = 0.85, p = 0.40, r 2 = 0.00), duration of between-dive flight bouts (t 146 = 0.39, p = 0.83, r 2 = 0.00), and outbound flight duration (t 146 = 0.79, p = 0.43, r 2 = 0.00). Birds on ledges with no birds above them had significantly shorter splashdown durations (6.5 ± 2.9 min) than birds on ledges with birds above them (8.9 ± 2.8 min, t 135 = −3.05, p = 0.001). We also found that 25% of birds returned to the colony for 3–98 min (24 ± 19 min) following splashdown. Birds that spent less time in the splashdown were not more likely to return to the colony (t 22 = 0.72, p = 0.51).
Arrival (χ 2 = 17,042, df = 49, p < 0.00001) and departure times (χ 2 = 9,302, df = 49, p < 0.00001) were significantly different from the associated Poisson distribution, with many more close arrival and departure times than anticipated from the Poisson distribution. Nonetheless, arrivals (χ 2 = 54.4, df = 49, p = 0.28, ESM Fig. S3a) and departures (χ 2 = 60.1, df = 49, p = 0.13, ESM Fig. S3b) were not significantly different from the randomly generated function. Arrivals (χ 2 = 56.8, df = 49, p = 0.24) and departures (χ 2 = 63.2, df = 49, p = 0.10, ESM Fig. S3b) were also not significantly different from the exponential distribution. This was also true for arrivals with benthic (χ 2 = 10.1, df = 6, p = 0.12) and pelagic prey items (χ 2 = 14.0, df = 6, p = 0.03) at the Bonferroni-corrected α value of 0.01. Across all feeding watches 1999–2007, the average dispersion in feeding intervals = σ:μ = 0.63 ± 0.02.
Underwater searching
Bottom time per dive and the number of dives in the final dive bout increased with prey energy content (Fig. 2). After accounting for prey energy content, bottom time per dive was higher for benthic than for pelagic (t 34 = −3.67, p = 0.0004) and invertebrate prey items (t 174 = −6.51, p < 0.0001, Table 1). The number of dives during the final bout was also higher for benthic than for pelagic (t 22 = 2.17, p = 0.02) and invertebrate (t 285 = 9.25, p < 0.0001, Table 1) prey items. In total, birds spent 88% more bottom time per dive bout when searching for benthic prey items than when searching for pelagic prey items and four times more bottom time per dive bout searching for benthic prey items than when searching for invertebrate prey items.
Above-water searching
Over all foraging trips, the outbound trip was ~13 min, followed by a 17-min inbound trip, with 42% of trips having at least one flight between dive bouts (average of one between-dive flights per trip), which averaged about 8 min (Table 2). Overall, the total duration of flight per trip was 37 min (Table 2). Once invertebrates were excluded, typical foraging trips consisted of a 15-min outbound trip, followed by a 20-min inbound trip, with 55% of trips having at least one flight between dive bouts for a total flight time per trip of 44 min. Virtually all (>99%) stops were followed by diving, showing that underwater searching usually followed above-water searching. Total flight time, inbound and outbound flight time, number of stops per trip, number of dives in the final dive bout, and total trip time were all highly correlated (Fig. 3; Table 3). The weakest relationships were between outbound flight time and other parameters (Table 3). All parameters except outbound flight time also increased with prey energy content, but the best correlation was with inbound flight time (Fig. 2).
After accounting for prey energy content, the following measures varied among prey types: inbound flight time, outbound flight time, the difference between inbound and outbound flight time, the number of flights per trip, the percent of trips with flights between dives, and total flight time (Table 2). The duration of flight bout between dive bouts did not vary among prey types. In all cases where above-water searching parameters varied among prey type, search times were greater for pelagic and benthic fish than for invertebrates. Furthermore, after accounting for prey energy content, total flight time was higher for pelagic than benthic (t 34 = 3.11, p = 0.007) and invertebrate prey items (t 44 = −13.06, p < 0.0001, Table 2). The number of stops per trip was also higher for pelagic than benthic (t 34 = 2.61, p = 0.01) and invertebrate prey items (t 44 = 6.71, p < 0.0001, Table 2). Similarly, inbound flight time was greater for pelagic than benthic (t 34 = 2.77, p = 0.009) and invertebrate prey items (t 44 = 137.57, p < 0.0001, Table 2).
There was no difference in the number of flights (within-individual paired t 52 = 1.48, p = 0.15), total trip duration (t 52 = 0.80, p = 0.43), inbound flight duration (t 52 = 1.05, p = 0.30), or total flight duration (t 52 = 1.89, p = 0.07) between trips that were taken after short stays at the colony (<10 min) and those taken following extended periods at the colony (>2 h). Flight durations (μ = 2.66, λ = 0.090, ΔAIC = 12.0, N = 2,083) followed a shifted gamma distribution with exponential tail and there was no support for a Lévy search pattern (Fig. 4). ΔAIC values were higher for an exponential tail rather than power law tail for all individual murres considered separately. An exponential distribution implies random distribution of flight durations with constant low probability for long enough flight durations.
Frequency distributions for ln-transformed flight durations (all flights, including outbound and inbound; subset: all flight durations) for thick-billed murres at Coats Island 2004-2007 (N = 2083 flights). The thin line represents the maximum likelihood shifted gamma function and the thick line represents a potential Lévy search pattern for comparison
Discussion
Search patterns varied among birds collecting different prey types, with birds increasing both above-water and underwater search effort for more energetically profitable items (Fig. 2). Longer foraging trips involved more underwater and above-water search time (Fig. 3). Presumably, trips were extended to increase the opportunity for encountering more energetically profitable, but less predictable, prey items (Figs. 2 and 3). On longer trips, birds apparently spent more time and covered a larger area both underwater and above-water, increasing the opportunity for encounters with prey that were spatially and temporally unpredictable. Inbound flight duration, and presumably foraging radius, increased with trip time across the entire range of trip times, in contrast to other seabirds (cf. Hamer et al. 2000; Daunt et al. 2002; Weimerskirch et al. 2005a, b). Longer trips meant higher time and energetic costs. Consequently, on longer trips, birds presumably pursued only more valuable prey items and were less likely to give up and return to the colony, leading to extended underwater and above-water search times.
For animals with widely spaced and dispersed prey items, prey encounters occur randomly (e.g., some albatrosses; Weimerskirch et al. 2005a, b). However, for most animals, prey occurs in patches and the optimal strategy is to conduct an area-restricted search following prey encounters (Weimerskirch 2007). For murres, benthic specialists return to the same habitat features repeatedly while pelagic foragers use local enhancement to locate pelagic fish schools (Fauchald and Erikstad 2002; Davoren et al. 2003a,b; Woo et al. 2008). As pelagic fish schools do not persist over short time scales, murres feeding on pelagic schools likely use area-restricted searches based on memory of past prey encounters to find feeding flocks, leading to longer above-water search times than for birds returning to known habitat features (Fauchald et al. 2000; Davoren et al. 2003a,b). Many birds may have “chosen” a single foraging location and probable prey type prior to leaving the colony, as evidenced by the relatively small number of stops per trip (Table 2). In support, there is a high degree of individual specialization diet, foraging behavior, and foraging location (Elliott et al. 2008c; Woo et al. 2008).
Our results add to the growing body of literature showing that marine predators modulate their prey capture strategy for different prey types (Garthe et al. 2000; Estes et al. 2003; Tremblay et al. 2005; Ropert-Coudert et al. 2006; Tinker et al. 2006; Yeates et al. 2007; Zavalaga et al. 2007; Deagle et al. 2008; Paredes et al. 2008; Enstipp et al. 2007; Miller and Trivelpiece 2008). For example, European shags (Phalacrocorax aristotelis) feeding on gunnels in rocky habitats swam solitarily and rapidly along the bottom while shags feeding on sand lance in sandy habitats foraged in groups and slowly probed the ground (Watanuki et al. 2008). For murres, pelagic specialists were more likely to give up on a given patch, while benthic specialists were more likely to remain at the same patch and extend each visit (dive, dive bout) at the same patch (Tables 1 and 2). Foraging trips preceding deliveries of benthic prey items were characterized by more underwater (many dives per bout, more bottom time per dive) and less above-water (many flights per trip, more total flying time) searching than those preceding the delivery of pelagic prey items (Tables 1 and 2). We suggest that it may be difficult to locate patches of pelagic prey but once the patch is located it is easy to capture prey items while for benthic prey, it may be easy to locate patches, but once a patch is located it may be difficult to capture prey (Womble and Sigler 2006; Gende and Sigler 2006; Hedd et al. 2009). For pelagic capelin and invertebrates, differences in underwater searching include searching throughout the descent or ascent phase, as shown by the V-shaped dive shape and slower descent rate (Elliott et al. 2008c). The differences in search patterns between pelagic and benthic items extends the dichotomy between these two prey types apparent in foraging behavior, with pelagic prey items associated with V-shaped dives, slower descent rates, more variable dive depths within a bout and shorter dives for a given depth (Elliott et al. 2008b, c).
Whereas Coats murres averaged two or fewer stops (one or fewer between-dive flights) during foraging (Table 2), murres at larger colonies showed many more stops during foraging (Benvenuti et al. 1998; Falk et al. 2000, 2002). For example, at a large colony in Iceland (>650,000 pairs of seabirds including >400,000 pairs of murres, compared with 30,000 pairs of murres and fewer than 100 other seabirds at Coats Island) where foraging radii and trip durations were large, trips averaged 7.8 stops and varied between four and 12 stops (Benvenuti et al. 1998; excluding data without inbound trip information). Similarly, the dispersion of feeding rates (σ:μ = 0.63) is similar to that found in the past (σ:μ = 0.60; Hipfner et al. 2006). Feeding rates are underdispersed at Coats Island compared to a nearby large colony (Digges Island, 300,000 breeding pairs; σ:μ = 0.67), meaning that adults are better able to regulate feeding rates (presumably because searching is less time-consuming) at Coats Island compared to larger colonies (Hipfner et al. 2006).
The relatively small amount of above-water searching at Coats Island, where foraging trips are short in both distance and duration, is consistent with the idea that foraging site fidelity is greater for seabirds that feed at relatively small spatial and temporal scales because prey are more predictable over smaller scales, at least in nontropical environments (Weimerskirch 2007). Specialization in diet and foraging strategy is high for many high-latitude seabirds (Irons 1998; Hamer et al. 2001; Garthe et al. 2007; Elliott et al. 2008c, Woo et al. 2008), including murres at another small colony in Svalbard (Mehlum et al. 2001), due to high prey encounter rates (Grémillet et al. 2004). Nonetheless, our observation of periodic sampling of the environment (stops), which has also been observed for murres elsewhere (Benvenuti et al. 1998, 2002; Falk et al. 2000, 2002), showing that some above-water searching does occur and that specialization does not mean complete foraging site fidelity. Outbound flights were generally shorter than inbound flights (Fig. 3), suggesting that stops must occur progressively further out on the outbound journey. Virtually all stops were followed by diving. Thus, as with other marine predators, search patterns were characterized by long travel times interspersed by area-restricted searches over small spatial scales (Weimerskirch et al. 2005a, b; Pinaud and Weimerskirch 2007; Cotté et al. 2007) but where, in this case, the area-restricted searches were dives.
We did not find any evidence for a Lévy search pattern (Fig. 4). Lévy search patterns are characterized by flight distances following a power law relationship between log-frequency and log-flight time (Reynolds 2005; Bénichou et al. 2006; cf. ESM Fig. S3). Many marine animals have been reported to obey Lévy search patterns (Viswanathan et al. 1996; Bartumeus et al. 2003; Austin et al. 2004; but see Edwards et al. 2007). An exponential distribution, such as we observed, implies random distribution of flight durations with constant low probability for long enough flight durations (Edwards et al. 2007). Theoretical models show that a distribution very similar to what we observe can occur when birds use small steps if prey is found and large steps if prey is not found (“area-restricted search”; Plank and James 2008).
Information transfer appeared to be relatively unimportant at the Coats Island colony because (1) foraging site fidelity was high (Woo et al. 2008); (2) naïve birds (birds leaving the colony for the first time) did not require more above-water search time than experienced birds; (3) time spent in the splashdown area did not reduce above-water searching time, with 25% of birds returning to the colony before making a final departure from the area; (4) there was no evidence of synchronized arrivals or departures. Earlier studies showing synchronization of arrivals in seabirds (e.g., Götmark 1990; Burger 1997) may have incorrectly applied the Poisson distribution instead of the exponential distribution. Rather than being a major site of information transfer, the main purpose of the splashdown period appeared to be to cleanse the feathers, as birds occupying sites that were likely to be soiled by feces spent 37% longer at the splashdown area than birds occupying sites where they were less likely to be soiled. Although we suggest that birds locate prey largely by returning to known locations or by searching at small spatial scales, either independently, via network foraging or by local enhancement, there are still several ways that information transfer could play a role at our colony (Burger 1997). For example, Gaston and Nettleship (1981) estimated that outgoing flocks would encounter incoming flocks every 280 m and Davoren et al. (2003a, b) reported that commuting paths of murres at sea were obvious, possibly keeping murres on track to food sources, and the splashdown may aid in the selection of commuting route while also playing a role in cleansing (Burger 1997). Presumably, at larger colonies where foraging ranges are greater, especially at colonies where rapidly changing ice conditions mean that the location of food sources change over a time scale of hours or where increased presence of fog means that visibility is low (Gaston and Nettleship 1981), ICH effects—as well as greater above-water searching—may be more important and reduced ability to buffer changes in prey abundance (Harding et al. 2007).
In conclusion, thick-billed murres varied their search strategies in response to the predictability of the target prey item. Underwater search times for benthic fish were 90% higher than for pelagic fish and 300% higher than for invertebrates while above-water searching for pelagic fish involved 20% more flights and total flying time than for benthic fish and 90% more flights and 180% more total flying time than for invertebrates. The absence of dietary differences between adult and chick prey items (Woo et al. 2008) and the unimodal distribution of trip durations suggest that birds searched for similar targets during both self-feeding and chick provisioning. The presence of a high degree of specialization in diet and foraging behavior and the lower above-water searching compared to larger colonies suggest that less energy may be spent searching at small spatial and temporal foraging scales due to greater predictability of prey items. At large (between colonies), medium (above-water), and small (underwater) scales, the predictability of prey items plays a strong role in determining the search patterns of a marine predator.
References
Austin D, Bowen WD, McMillan JI (2004) Intraspecific variation in movement patterns: modeling individual behaviour in a large marine predator. Oikos 105:15–30
Barta Z, Szép T (1992) The role of information transfer under different food patterns: a simulation study. Behav Ecol 3:318–324
Bartumeus F, Peters F, Pueyo S, Marrasé C, Catalan J (2003) Helical Lévy walks: adjusting searching statistics to resource availability in microzooplankton. Proc Natl Acad Sci U S A 100:12771–12775
Bénichou O, Loverdo C, Moreau M, Voituriez R (2006) Two-dimensional intermittent search processes: an alternative to Lévy flight strategies. Phys Rev E 74:020102
Benvenuti S, Bonadonna F, Dall’Antonia L, Gudmundsson GA (1998) Foraging flights of breeding thick-billed murres (Uria lomvia) as revealed by bird-borne direction recorders. Auk 115:57–66
Benvenuti S, Dall’Antonia L, Falk K (2002) Diving behaviour differs between incubating and brooding Brunnich’s guillemots, Uria lomvia. Polar Biol 25:474–478
Bertrand S, Burgos JM, Gerlotto F, Atiquipa J (2005) Lévy trajectories of Peruvian purse-seiners as an indicator of the spatial distribution of anchovy (Engraulis ringens). ICES J Mar Sci 62:477–482
Bertrand S, Bertrand A, Guevara-Carrasco R, Gerlotto F (2007) Scale-invariant movements of fishermen: the same foraging strategy as natural predators. Ecol Appl 17:331–337
Birkhead TR (1985) Coloniality and social behaviour in the Atlantic Alcidae. In: Nettleship DN, Birkhead TR (eds) The Atlantic Alcidae. Academic, London, pp 355–382
Buckley NJ (1997a) Experimental tests of the information-center hypothesis with black vultures (Coragyps atratus) and turkey vultures (Cathartes aura). Behav Ecol Sociobiol 41:267–279
Buckley NJ (1997b) Spatial-concentration effects and the importance of local enhancement in the evolution of colonial breeding in seabirds. Am Nat 146:1091–1112
Burger AE (1997) Arrival and departure behaviour of common murres at colonies: evidence for an information halo? Col Waterbirds 20:55–65
Burger AE, Wilson RP, Garnier D, Wilson MPT (1993) Diving depths, diet and underwater foraging of rhinoceros auklets in British Columbia. Can J Zool 71:2528–2540
Catania KC, Hare JF, Campbell KL (2008) Water shrews detect movement, shape, and smell to find prey underwater. Proc Natl Acad Sci 105:571–576
Cotté C, Park YH, Guinet C, Bost CA (2007) Movements of foraging king penguins through marine mesoscale eddies. Proc Roy Soc Lond B 274:2385–2391
Daunt F, Benvenuti S, Harris MP, Dall’Antonia L, Elston DA, Wanless S (2002) Foraging strategies of the black-legged kittiwake Rissa tridactyla at a North Sea colony: evidence for a maximum foraging range. Mar Ecol Prog Ser 245:239–247
Davoren GK, Burger AE (1999) Differences in prey selection and behaviour during self-feeding and chick provisioning in rhinoceros auklets. Anim Behav 58:853–863
Davoren GK, Montevecchi WA (2003) Consequences of foraging trip duration on provisioning behaviour and fledging condition of common murres Uria aalge. J Avian Biol 34:44–53
Davoren GK, Montevecchi WA, Anderson JT (2003a) Search strategies of a pursuit-diving marine bird and the persistence of prey patches. Ecol Monogr 73:463–481
Davoren GK, Montevecchi WA, Anderson JT (2003b) The influence of fish behaviour on the search strategies of common murres Uria aalge in the Northwest Atlantic. Mar Ornithol 31:123–131
Davoren GK, Anderson JT, Montevecchi WA (2006) Habitat utilization by spawning capelin Mallotus villosus off the northeast coast of Newfoundland. Can J Fish Aquat Sci 63:268–284
Deagle BE, Gales NJ, Hindell MA (2008) Variability in foraging behaviour of chick-rearing macaroni penguins Eudyptes chrysolophus and its relation to diet. Mar Ecol Prog Ser 359:295–309
Edwards AM, Phillips RA, Watkins NW, Freeman MP, Murphy EJ, Afanasyev V, Buldyrev SV, da Luz MGE, Raposo EP, Stanley HE, Viswanathan GM (2007) Revisiting Lévy flight search patterns of wandering albatrosses, bumblebees and deer. Nature 449:1044–1048
Elliott KH, Gaston AJ (2008) Mass–length relationships and energy density of fish collected from thick-billed murre ledges in the Canadian Arctic 1981–2007. Mar Ornith 36:35–34
Elliott KH, Gaston AJ (2009) Accuracy of depth recorders. Waterbirds 32:183–191
Elliott KH, Davoren GK, Gaston AJ (2007) The influence of buoyancy and drag on the dive behaviour of an arctic seabird, the thick-billed murre. Can J Zool 85:352–361
Elliott KH, Davoren GK, Gaston AJ (2008a) Time allocation by a deep-diving bird reflects energy gain and prey type. Anim Behav 75:1301–1310
Elliott KH, Davoren GK, Gaston AJ (2008b) Time allocation by a deep-diving bird reflects energy expenditure. Anim Behav 75:1311–1317
Elliott KH, Woo K, Gaston AJ, Benvenuti S, Dall’Antonia L, Davoren GK (2008c) Seabird foraging behaviour indicates prey type. Mar Ecol Prog Ser 354:289–303
Enstipp MR, Grémillet D, Jones DR (2007) Investigating the functional link between prey abundance and seabird predatory performance. Mar Ecol Prog Ser 331:267–279
Estes JA, Riedman ML, Staedler MM, Tinker MT, Lyon BE (2003) Individual variation in prey selection by sea otters: patterns, causes and implications. J Anim Ecol 72:144–155
Falk K, Benvenuti S, Dall'antonia L, Kampp K, Ribolini A (2000) Time allocation and foraging behaviour of chick-rearing Brunnich's guillemots Uria lomvia in high-arctic Greenland. Ibis 142:82–92
Falk K, Benvenuti S, Dall'antonia L, Gilchrist G, Kampp K (2002) Foraging behaviour of thick-billed murres breeding in different sectors of the North Water polynya: an inter-colony comparison. Mar Ecol Prog Ser 231:293–302
Fauchald P, Erikstad KE (2002) Scale-dependent predator–prey interactions: the aggregative response of seabirds to prey under variable prey abundance and patchiness. Mar Ecol Prog Ser 231:279–291
Fauchald P, Erikstad KE, Skarsfjord H (2000) Scale-dependent predator–prey interactions: the hierarchical spatial distribution of seabirds and prey. Ecology 81:773–783
Garthe S, Benvenuti S, Montevecchi WA (2000) Pursuit plunging by Northern Gannets (Sula bassana) feeding on capelin (Mallotus villosus). Proc Roy Soc Lond B 267:1717–1722
Garthe S, Montevecchi WA, Chapdelaine G, Rail JF, Hedd A (2007) Contrasting foraging tactics by northern gannets (Sula bassana) breeding in different oceanographic domains with different prey fields. Mar Biol 151:687–694
Gaston AJ, Bradstreet MSW (1993) Intercolony differences in the summer diet of thick-billed murres in the eastern Canadian Arctic. Can J Zool 71:1831–1840
Gaston AJ, Hipfner JM (2006a) Adult Brünnich’s guillemots Uria lomvia balance body condition and investment in chick growth. Ibis 148:106–113
Gaston AJ, Hipfner JM (2006b) Body mass changes in Brünnich’s guillemots Uria lomvia with age and breeding state. J Avian Biol 37:101–109
Gaston AJ, Nettleship DN (1981) The thick-billed murres of Prince Leopold Island. Canadian Wildlife Service, Ottawa, p 351
Gaston AJ, Woo K, Hipfner JM (2003) Trends in forage fish populations in northern Hudson Bay since 1981, as determined from the diet of nestling thick-billed murres Uria lomvia. Arctic 56:227–233
Gaston AJ, Gilchrist HG, Hipfner JM (2005) Climate change, ice conditions and reproduction in an Arctic nesting marine bird: Brünnich’s guillemot (Uria lomvia L.). J Anim Ecol 74:832–841
Gende SM, Sigler MF (2006) Persistence of forage fish ‘hot spots’ and its association with foraging Steller sea lions (Eumetopias jubatus) in southeast Alaska. Deep Sea Res II 53:432–441
Götmark F (1990) A test of the information-centre hypothesis in a colony of sandwich terns Sterna sandvicensis. Anim Behav 39:487–495
Grémillet D, Kuntz G, Delbart F, Mellet M, Kato A, Robin JP, Chaillon PE, Gendner JP, Lorentsen SH, Le Maho Y (2004) Linking the foraging performance of a marine predator to local prey abundance. Funct Ecol 18:793–801
Grémillet D, Kuntz G, Woakes AJ, Gilbert C, Robin JP, Le Maho Y, Butler PJ (2005) Year-round recordings of behavioural and physiological parameters reveal the survival strategy of a poorly-insulated diving endotherm during the Arctic winter. J Exp Biol 208:4231–4241
Halsey LG, Woakes AJ, Butler PJ (2003) Testing optimal foraging models for air-breathing divers. Anim Behav 65:641–653
Halsey LG, Bost CA, Handrich Y (2007) A thorough and quantified method for classifying seabird diving behaviour. Pol Biol 30:991–1004
Hamel NJ, Parrish JK, Conquest LL (2004) Effects of tagging on behaviour, provisioning, and reproduction in the common murre (Uria aalge), a diving seabird. Auk 121:1161–1171
Hamer KC, Phillips RA, Wanless S, Harris MP, Wood AG (2000) Foraging ranges, diets and feeding locations of gannets Morus bassanus in the North Sea: evidence from satellite telemetry. Mar Ecol Prog Ser 200:257–264
Hamer KC, Phillips RA, Hill JK, Wanless S, Wood AG (2001) Contrasting foraging strategies of gannets Morus bassanus at two North Atlantic colonies: foraging trip duration and foraging area fidelity. Mar Ecol Prog Ser 224:283–290
Harding AMA, Piatt JF, Schmutz JA, Kettle AB, Shultz M, Van Pelt TI, Kettle AB, Speckman SG (2007) Prey density and the behavioural flexibility of a marine predator: the common murre (Uria aalge). Ecology 88:2024-2035
Heath JP, Gilchrist HG, Ydenberg RC (2006) Regulation of stroke patterns and swim speed across a range of current velocities: diving by Common Eiders wintering in polynyas in the Canadian arctic. J Exp Biol 209:3974–3983
Heath JP, Gilchrist HG, Ydenberg RC (2007) Can diving models predict patterns of foraging behaviour? Diving by common eiders in an arctic polynya. Anim Behav 73:877–884
Hedd A, Regular PM, Montevecchi WA, Buren AD, Burke CM, Fifield DA (2009) Going deep: Common murres dive into frigid water for aggregated, persistent and slow-moving capelin. Mar Biol 156:741–751
Hipfner JM (1997) The effects of parental quality and timing of breeding on the growth of nestling thick-billed murres. Condor 99:353–360
Hipfner JM, Gaston AJ, Smith BD (2006) Regulation of provisioning rate in the thick-billed murre (Uria lomvia). Can J Zool 84:931–938
Hobson KA (1993) Trophic relationships among high Arctic seabirds: insights from tissue-dependent stable isotope models. Mar Ecol Prog Ser 95:7–18
Irons DB (1998) Foraging area fidelity of individual seabirds in relation to tidal cycles and flock feeding. Ecology 79:647–655
Jaquemet S, Le Corre M, Quartly GD (2007) Ocean control of the breeding regime of the sooty tern in the southwest Indian Ocean. Deep-Sea Res II 54:130–142
Jones IL, Fraser GS, Rowe S, Carr X, Taylor P (2002) Different patterns of parental effort during chick-rearing by female and male thick-billed murres (Uria lomvia) at a low Arctic colony. Auk 119:1064–1074
Lewis S, Benvenuti S, Dall’Antonia L, Griffiths R, Money L, Sherratt TN, Wanless S, Hamer KC (2002) Sex-specific foraging behaviour in a monomorphic seabird. Proc Roy Soc Lond B 269:1687–1693
Mallory ML, Gaston AJ, Forbes MR, Gilchrist HG, Cheney B, Lewis S, Thompson PM (2008) Flexible incubation rhythm in northern fulmars: a comparison between oceanographic zones. Mar Biol 154:1031–1040
Mårell A, Ball JP, Hofgaard A (2002) Foraging and movement paths of female reindeer: insights from fractal analysis, correlated random walks, and Lévy flights. Can J Zool 80:854–865
Mehlum F, Watanuki Y, Takahashi A (2001) Diving behaviour and foraging habitats of Brünnich’s guillemots (Uria lomvia) breeding in the high-Arctic. J Zool Lond 255:413–423
Miller AK, Trivelpiece WZ (2008) Chinstrap penguins alter foraging and diving behavior in response to the size of their principle [sic] prey, Antactic krill. Mar Biol 154:201–208
Mori Y, Takahashi A, Mehlum F, Watanuki Y (2002) An application of optimal diving models to diving behaviour of Brünnich’s guillemots. Anim Behav 64:739–745
Ohashi K, Thomson JD, D’Souza D (2007) Trapline foraging by bumble bees: IV. Optimization of route geometry in the absence of competition. Behav Ecol 18:1–11
Paredes R, Jones IL, Boness DJ (2004) Reduced parental care, compensatory behaviour and reproductive costs experienced by female and male thick-billed murres equipped with data loggers. Anim Behav 69:197–208
Paredes R, Jones IL, Boness DJ (2006) Parental roles of male and female thick-billed murres and razorbills at the Gannet Islands, Labrador. Behaviour 143:451–481
Paredes R, Jones IL, Boness DJ, Tremblay Y, Renner M (2008) Sex-specific differences in diving behaviour of two sympatric Alcini species: thick-billed murres and razorbills. Can J Zool 86:610–622
Pinaud D, Weimerskirch H (2007) At-sea distribution and scale-dependent foraging behaviour of petrels and albatrosses: a comparative study. J Anim Ecol 76:9–19
Plank MJ, James A (2008) Optimal foraging: Lévy pattern or process? J R Soc Interface 5:1077–1086
Ramos-Fernández G, Mateos JL, Miramontes O, Cocho G (2004) Lévy walk patterns in the foraging movements of spider monkeys (Ateles geoffroyi). Behav Ecol Sociobiol 55:223–230
Reynolds AM (2005) Scale-free movement patterns arising from olfactory-driven foraging. Phys Rev E 72:041928
Robards MD, Willson MF, Armstrong RH, Piatt JF (1999) Sand lance: a review of biology and predator relations and annotated bibliography. Research Paper PNW-RP-521. Pacific Northwest Research Station, Portland, p 327
Ropert-Coudert Y, Wilson RP, Daunt F, Kato A (2004) Patterns of energy acquisition by penguins: benefits of alternating short and long foraging trips. Behav Ecol 15:824–830
Ropert-Coudert Y, Grémillet D, Kato A (2006) Swim speed of free-ranging great cormorants. Mar Biol 249:415–422
Simpkins MA, Kelly BP, Wartzok D (2001) Three-dimensional analysis of search behaviour by ringed seals. Anim Behav 62:67–72
Sjoberg M, Ball JP (2000) Grey seal, Halichoerus grypus, habitat selection around haulout sites in the Baltic Sea: bathymetry or central-place foraging? Can J Zool 78:1661–1667
Steen H, Vogedes D, Broms F, Falk-Petersen S, Berge J (2007) Little auks (Alle alle) breeding in a High Arctic fjord system: bimodal foraging strategies as a response to poor food quality? Pol Res 26:118–125
Tinker MT, Costa DP, Estes JA, Wieringa N (2006) Individual dietary specialization and dive behaviour in the California sea otter: using archival time-depth data to detect alternative foraging strategies. Deep Sea Res II 54:330–342
Tremblay Y, Cook TR, Cherel Y (2005) Time budget and diving behaviour of chick-rearing Crozet shags. Can J Zool 83:971–982
Viswanathan GM, Afanasyev V, Buldyrev SV, Murphy EJ, Prince PA, Stanley HE (1996) Lévy flight search patterns of wandering albatrosses. Nature 381:413–415
Viswanathan GM, Buldyrev SV, Havlin S, da Luz MGE, Raposo EP, Stanley HE (1999) Optimizing the success of random searches. Nature 401:911–914
Ward P, Zahavi A (1973) The importance of certain assemblages of birds as “information centres” for food finding. Ibis 115:517–534
Watanuki Y, Daunt F, Takahashi A, Newell M, Wanless S, Sato K, Miyazaki N (2008) Microhabitat use and prey capture of a bottom-feeding top predator, the European shag, shown by camera loggers. Mar Ecol Prog Ser 356:283–293
Weimerskirch H (2007) Are seabirds foraging for unpredictable resources? Deep Sea Res II 54:211–223
Weimerskirch H, Gault A, Cherel Y (2005a) Prey distribution and patchiness: factors in foraging success and efficiency of wandering albatrosses. Ecology 86:2611–2622
Weimerskirch H, Le Corre M, Ropert-Coudert Y, Kato A, Marsac F (2005b) The three-dimensional flight of red-footed boobies: adaptations to foraging in a tropical environment? Proc R Soc Lond B 272:53–61
Wilson LJ, Daunt F, Wanless S (2004) Self-feeding and chick-provisioning diet differ in the common guillemot Uria aalge. Ardea 92:197–207
Wilson RP, Scolaro JA, Grémillet D, Kierspel MAM, Laurenti S, Upton J, Gallelli H, Quintana F, Frere E, Müller G, Straten MT, Zimmer I (2005) How do magellanic penguins cope with variability in their access to prey? Ecol Monogr 75:379–401
Womble JN, Sigler MF (2006) Seasonal availability of abundant, energy-rich prey influences the abundance and diet of a marine predator, the Steller sea lion Eumetopias jubatus. Mar Ecol Prog Ser 325:281–293
Woo K, Elliott KH, Davidson M, Gaston AJ, Davoren GK (2008) Individual specialization in diet by a generalist predator reflects specialization in foraging behaviour. J Anim Ecol 77:1082–1091
Yeates LC, Williams TM, Fink TL (2007) Diving and foraging energetics of the smallest marine mammal, the sea otter (Enhydra lutris). J Exp Biol 210:1960–1970
Zavalaga CB, Benvenuti S, Dall’Antonia L, Emslie SD (2007) Diving behavior of blue-footed boobies Sula nebouxii in northern Peru in relation to sex, body size and prey type. Mar Ecol Prog Ser 336:291–303
Acknowledgements
We thank B. Addison, K. Ashbrook, M. Barrueto, A. Hargreaves, M. Hipfner, S. Jacobs, G. Lancton, A. Moody, J. Provencher, A. Ronson, K. Skebo, P. Smith, K. Woo, and P. Woodward for help in the field. A. Fromevitch, K. Hedges, and P. Turko helped with data compilation. J. Nakoolak kept us safe from bears. A. Burger, D. Gillis, and an anonymous reviewer provided excellent comments on an earlier version of the manuscript. KHE benefited from funding provided by NSERC Postgraduate (M) Award, NSERC Northern Research Internship, Andrew Taylor Northern Research Grant, Mountain Equipment Co-op Studentship, Arctic Institute of North America Grant-in-aid, Malcolm Ramsay Award, Frank M. Chapman Memorial Fund, Society of Canadian Ornithologists/Bird Studies Canada Taverner Award, and the Northern Scientific Training Program through the Department of Indian Affairs and Northern Development. The Canadian Wildlife Service Migratory Birds Division, 2007–2008 International Polar Year, the Polar Continental Shelf Project, and the University of Manitoba also funded this project. R. Armstrong at the Nunavut Research Institute, M. Mallory at the Canadian Wildlife Service Northern Research Division, and C. Eberl at the Canadian Wildlife Service in Ottawa provided logistical support. All procedures were approved under the guidelines of the Canadian Committee for Animal Care (Protocol Number F04-030).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Brown
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Table S1
(MHT 529 kb)
ESM
Time at splashdown (±SE) increases with time at the colony prior to departure.
ESM
Frequency distribution for (a) arrival and (b) departure times using actual data (columns) and simulations (lines) for thick-billed murres at Coats Island 1999-2007. (DOC 49.0 kb)
Rights and permissions
About this article
Cite this article
Hamish Elliott, K., Bull, R.D., Gaston, A.J. et al. Underwater and above-water search patterns of an Arctic seabird: reduced searching at small spatiotemporal scales. Behav Ecol Sociobiol 63, 1773–1785 (2009). https://doi.org/10.1007/s00265-009-0801-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-009-0801-y