Abstract
The Magellanic penguin Spheniscus magellanicus has recently colonized two new coastal sites (Islote Lobos and El Pedral), increasing the number of colonies in northern Patagonia, Argentina. Assuming foraging parameters during the breeding season to be valid short-term indicators of population health, we studied several foraging parameters of penguins from the two new established sites and from an older and well-established colony (Punta Norte) also localized in the north of Patagonia. Penguins from the recently formed colonies performed shorter foraging trips and visited waters closer to their colony than birds from Punta Norte, with penguins from Punta Norte spending almost twice the time at sea spent by penguins from El Pedral. Penguins from Punta Norte also spent more time underwater, spent a higher proportion of the complete diving cycle searching for prey, performed more wiggles (indicative of prey capture) and had fewer estimated prey captures per unit time underwater than penguins from Islote Lobos and El Pedral. This information suggests that, given no limitations on nest-site availability and no prevalence of predators, the good foraging conditions around El Pedral and Islote Lobos may be implicated in the increase in these two relatively new colonies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Foraging is an important component of animal behavior, and its enhanced efficiency is assumed to increase fitness (Stephens and Krebs 1986). For the particular case of central place foragers such as seabirds, both the amount of time and energy allocated for foraging are particularly constrained during the breeding season (Orians and Pearson 1979) when parents have to obtain enough food for self-provisioning as well as for their brood and do so at appropriate rates (Stearns 1992).
In addition, seabirds exploit a heterogeneous environment and are expected to adjust their behavior in order to cope with different scenarios of prey abundance and availability (Pyke 1984; Hunt 1990; Fauchald and Erikstad 2002). This capacity of buffering changes in prey availability through modifications in their foraging behavior makes seabirds good models to monitor feeding conditions around breeding areas (Jodice et al. 2006; Hamer et al. 2006; Harding et al. 2007). Moreover, the quantification of foraging parameters during the breeding season has proven to be a powerful short-term indicator of breeding performance in several seabird species (Lewis et al. 2001, 2006; Grémillet et al. 2006; Sala et al. 2012a, 2014a).
Penguins are highly specialized diving seabirds whose foraging behavior has been extensively studied thanks to the advances in electronic technology (e.g., Wilson 1995; Burger and Shaffer 2008). The use of miniaturized highly sensitive sensors has allowed researchers to study in detail the foraging movements of these birds and even examine food consumption rates (Wilson et al. 2007; Sala et al. 2012b). The latter has been possible because feeding penguins display marked undulations in their depth profiles (presented graphically as depth against time) that can be used as a powerful proxy for foraging success (Simeone and Wilson 2003; Bost et al. 2007; Hanuise et al. 2010; Sala et al. 2012b).
The Magellanic penguin Spheniscus magellanicus is an important top predator of the Southern Hemisphere oceans, being ranked 20th in terms of projected global annual food consumption of all seabird species (Brooke 2004). This species breeds at mainland and island colonies on both coasts of South America as well as on the Malvinas/Falkland Islands (Williams 1995; Schiavini et al. 2005). Several studies have been performed on the foraging behavior of this species along the Patagonia coast (e.g., Wilson et al. 2005; Boersma and Rebstock 2009; Boersma et al. 2009; Sala et al. 2012a, b, 2014a, b), and at least four of these have revealed important relationships between several aspects of Magellanic penguin foraging effort and population growth rates (Boersma and Rebstock 2009; Sala et al. 2012a, b, 2014a). For example, Magellanic penguins breeding at colonies with lower population growth rates have been observed to make longer trips, travel greater distances and have higher diving effort than those coming from colonies with higher growth rates and these differences have been attributed to disparities in food availability around breeding sites (Sala et al. 2012a, b, 2014a).
Over the last decade, Magellanic penguins have colonized two new coastal sites, increasing the number of colonies along the north of Patagonia (Schiavini et al. 2005). These two new colonies, known as El Pedral and Islote Lobos (Fig. 1), are increasing at fast rates with a lambda value of 1.91 and 2.63 for Islote Lobos and El Pedral, respectively (for more information, see Pozzi et al. 2015), probably due to enhanced foraging conditions although neither site has, as yet, been examined with regard to foraging behavior. Punta Norte, an older and bigger colony of north Patagonia, has also increased during the last years (λ = 1.21); however, its rate of increase is lower than that of Islote Lobos and El Pedral (Pozzi et al. 2015). A recent work suggests that the steady increase in northern Patagonian colonies would be principally associated with the immigration of young birds from southern colonies (Pozzi et al. 2015).
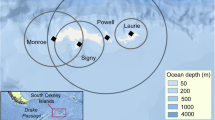
Map showing the localization of the three colonies where Magellanic penguins (Spheniscus magellanicus) were instrumented with GPS devices and the main oceanographic features in the study site (indicated with red dashed lines). The oceanographic features shown are 1—Coastal upwelling events (Pisoni et al. 2014), 2—Thermohaline front (Piola and Scasso 1988) and 3—Península Valdés tidal front (Tonini et al. 2013)
In this work, we examine several foraging parameters (e.g., foraging range, diving behavior and capture rates) of Magellanic penguins breeding at Islote Lobos, El Pedral and Punta Norte, the three northernmost colonies along the Magellanic penguin breeding distributional range at the Patagonian coast. Our specific aim was to determine whether there were differences between the foraging behavior of penguins from the recently established colonies and that of animals from the older colony of Punta Norte. Behind these objectives underlies the hypothesis that foraging effort shall be a proxy of population growth rate.
Materials and methods
Field work was conducted during early chick-rearing between mid-November and the first week of December 2011 at three Magellanic penguin colonies: Islote Lobos (41°26ʹS, 65°02ʹW), Punta Norte/San Lorenzo (42°04ʹS, 63°49ʹW) and El Pedral (42°56ʹS, 64°20ʹW) (Fig. 1). Islote Lobos is the most northerly Magellanic penguin colony along the Patagonia coast. It is situated on the west coast of the San Matías Gulf and has increased from 22 breeding pairs in 2002 to approximately 4750 in 2011 (Schiavini et al. 2005; Pozzi et al. 2015). Punta Norte/San Lorenzo is localized on the southern coast of San Matías Gulf. This colony is 170 km from Islote Lobos and holds 1,35,000 breeding pairs (Pozzi et al. 2015) (Fig. 1). El Pedral is a small, new colony situated in the coast of the Nuevo Gulf (Fig. 1). Its first nests were observed in 2009, and in 2011, this colony held 175 breeding pairs (Pozzi et al. 2015). This breeding site is situated 160 km south from Punta Norte/San Lorenzo colony.
A total of twenty-four Magellanic penguins (eight from each colony comprising a total of eight males and 16 females) brooding two chicks of less than 1 week old were equipped with GPS loggers (GPS-TDlog, Earth and Ocean Technologies; 96 × 39 × 27 mm (L × W × H), comprising ~6.5 % of the cross-sectional area of the bird’s body). Each bird was removed from its nest, sexed by its bill depth and length (Gandini et al. 1992; Bertellotti et al. 2002) and equipped with a logger on the lower back using TESA tape (Wilson et al. 1997). The instruments were programmed to record latitude and longitude every 1 s and depth every 2 s. The horizontal accuracy of the positional fixes was better than 5 m for 90 % of the fixes (GPS-TDlog Manual), while depth data were recorded with an absolute accuracy better than 0.3 m. The weight of male and female was 4501 ± 402 and 3708 ± 355 g, respectively (Bertellotti et al. 2002); thus, the weight of the devices was 1.7 ± 0.15 % of male’s body mass and 2 ± 0.2 % of female’s body mass. The instrumentation procedure was completed in less than 5 min, and birds were quickly returned to their nests. Penguins were allowed to forage for a single trip before the devices were retrieved. All birds carrying devices continued to feed the chicks normally while they were instrumented. The Magellanic penguin is a slightly dimorphic species (Gandini et al. 1992), and no differences have been reported in the foraging parameters between sexes during the chick-rearing period (Boersma and Rebstock 2009; Raya Ray et al. 2012). Thus, data from females and males were pooled together.
Spatial analysis
Spatial analyses were conducted using the open-source statistical package R version 2.13.0 (R Development Core Team 2011). The total home range size of each colony was determined using the minimum convex polygon method (function mcp, package adehabitatHR, Calenge 2006). Kernel analyses to assess habitat use from GPS locations (Worton 1989) were performed using the function kernel UD from the library adehabitatHR. We used a smoothing parameter h = 3000 with a cell size of 1000 m to determine kernel contours of 50, 75 and 95 %. Distribution maps were produced for the complete data set of the foraging trips.
Foraging trip analysis
For each foraging trip, the following variables were calculated: (1) total trip length (km), defined as the total cumulative horizontal distance between all positional fixes along the track; (2) total trip duration (h), obtained by calculation of the time elapsed between the first and last position recorded in the water; and (3) the maximum distance from the colony (km), defined by the maximum straight-line distance between the bird and its nest site. Each penguin foraging trip was divided into three segments, separated according to comparisons of running mean speeds over 10-min periods with the overall mean swimming speed recorded for the daylight hours of the relevant trip (i.e., between 4:00 a.m. and 10:00 p.m.). This methodology has been extensively used and assumes that penguins decrease their horizontal speed while they forage and travel faster when commuting to their feeding grounds and on their way back to the colony (e.g., Boersma et al. 2009; Raya Rey et al. 2010; Sala et al. 2012a). The three trip segments were defined as follows: (1) outbound phase, the travel period between the colony and the foraging area; (2) foraging phase, the period while the penguin forages; and (3) the inbound phase, the period between the end of the foraging phase and the return to the colony. The classification of each fix into one of the trip phases was obtained by an algorithm developed with the open-source statistical package R 2.13.0 (R Development Core Team 2011). For each penguin, the path length and the mean horizontal speed of each of the three phases were calculated. Foraging phase fixes were classified as belonging to day (≥4 am and <10 pm) or night hours (≥10 pm and <4 am). This classification allowed us to calculate the amount of time each penguin spent foraging during daylight hours. Foraging trip parameters were calculated using the standard or base R packages plus the library chron.
Data analysis on diving behavior
Penguin diving behavior was analyzed using the program MTDIVE (Jensen Software System), specially designed to detect automatically the three characteristic phases of a dive (descent, bottom and ascent phase) based on changes in the rate of descent/ascent. All submersions deeper than 1.5 m were considered as dives (Sala et al. 2012b). Each dive was classified as belonging to one of the previously mentioned trip phases (i.e., outbound, foraging or inbound). Moreover, foraging dives were classified as having been performed during daylight or night hours. For each trip, the following variables were calculated: (1) the total amount of time spent underwater during foraging daytime dives; (2) the total amount of time spent in the bottom phase of foraging daytime dives; and (3) the total number of foraging daytime dives. For each foraging daytime dive, we calculated (1) its total duration (defined as the total time the bird spent underwater during an immersion), (2) the maximum reached depth, (3) the bottom duration (defined in Sala et al. 2012b), (4) the post-dive interval (defined as the surface period between two consecutive dives) and (5) the dive efficiency [defined as the bottom duration/(dive duration + post-dive interval)]. To explore the relationship between the ratio of maximum dive duration and minimum pause and depth, foraging daytime dives were classified into 1-m bins of dive depth (e.g., 5–6 m). For each 1-m-depth interval, the ratio between maximum dive duration and the minimum pause duration was calculated.
Prey capture estimation
Undulations in the dive profile, commonly known as “wiggles,” have been reported to indicate prey capture events (Simeone and Wilson 2003) and have been adopted as such by a number of authors (see above). Using parameters defined in Sala et al. 2012b, a single wiggle was defined as two or three serial points of inflexion (SPI) of >0.5 m/s and each wiggle was taken to represent a prey capture event. For each animal, we calculated the total amount of wiggles performed during daytime foraging dives. Since penguins mostly feed on the bottom diving phase (Simeone and Wilson 2003; Wilson et al. 2010), the number of wiggles per bottom time of a dive was considered as a measure of “catch per unit effort” (CPUE) (Sala et al. 2012b). A CPUE value was calculated for each foraging daytime dive.
Speed measurements
To investigate whether penguins changed their rate of horizontal movement in relation to catch-per-unit-effort (CPUE) metrics, we calculated the horizontal speed between the beginning and end of each dive and considered this as a measure of the traveling speed in two dimensions. For this, first we obtained the position (lat, long) of the beginning and end of each dive and calculated the distance between both positions. The horizontal speed was obtained from the division of distance by dive duration. The horizontal speed was only estimated for those dives where we could immediately measure the exact position at the beginning and end (to within 10 s). This analysis was performed with the complete set of dives (not only for foraging daytime dives).
Statistical analysis
Differences between colonies in foraging trip parameters were tested using ANOVA or Kruskal–Wallis depending on normality. When a significant difference was detected, Tukey’s post hoc test or Mann–Whitney pair-wise comparisons were performed, respectively. In order to control for pseudoreplication, linear mixed effect models (LMM) fitted by maximum likelihood (ML) were used for all dive analyses. In these analyses, colony was set as a fixed factor, while bird identity was set as a random factor. Where data were not normally distributed, generalized mixed effect models (GLMM) with Poisson distribution were performed. Because maximum dive depth has an important effect on dive duration, bottom duration, pause duration, dive efficiency and horizontal speed, the analysis of these factors included the effect of dive depth as a covariate (Tremblay and Cherel 2000).
For those foraging trip parameters analyzed by means of LMM or GLMM (e.g., where there were several measurements per animal), the results presented in Tables 1 and 2 were obtained in the following way. First, we obtained a mean value per parameter per individual. Then these mean values were used to calculate a grand mean per colony, the standard deviation and range. To study the effect of the CPUE on the horizontal speed of penguins from the three colonies, the former was divided into three categories: (1) values of CPUE below 1; (2) values between 1 and less than 2; and (3) values equal to or higher than 2. CPUE values were classified into these categories in order to have a similar amount of dives in each one of them.
LMMs were performed using the function lme from the package nlme, while GLMMs were run using the function lmer from the package lme4. In all analyses, we applied a backward selection procedure, removing nonsignificant terms from the model one by one and comparing the models with progressively simplified fixed effects using the anova function from the package stats (Crawley 2007). Post hoc testing was performed using the function glht from the library multicomp. All statistical analyses were performed using the open-source statistical package R version 2.13.0 (R Development Core Team 2011) with a level of significance of P < 0.05. Results are shown as mean ± standard deviation (SD).
Results
Of a total of 24 instrumented animals, we recorded 21 complete foraging trips constituting 11,053 dives. One of the penguins from El Pedral could not be recaptured, and two of the devices from this colony failed to record data. Thus, the following results were obtained from the analysis of the foraging trips performed by eight penguins from Punta Norte, eight penguins from Islote Lobos and five penguins from El Pedral.
At sea distribution and foraging areas
During the early chick-rearing period, penguins from Punta Norte exploited an area six times larger than that used by penguins from Islote Lobos and ten times larger than the area visited by penguins from El Pedral (4879, 794 and 468 km2, Punta Norte, Islote Lobos and El Pedral, respectively). Birds from Islote Lobos foraged principally in an area located northeast of the colony in the waters of the San Matías Gulf (Fig. 2). This area is characterized by the presence of upwelling events and high primary productivity (Fig. 1). Penguins from Punta Norte exploited areas to the northeast and southeast of the colony in the Atlantic Ocean. They also foraged to the north and northwest of the colony in waters of the San Matías Gulf (Fig. 2). During the austral summer, an important thermohaline front develops in this area around 41°50ʹS (from west to east) (Fig. 1). Positions at sea from the five penguins from El Pedral revealed that they foraged north and northwest of the colony in waters of the Nuevo Gulf (Fig. 2).
Foraging behavior
Overall trip characteristics are shown in Table 1. Penguins from the three studied colonies travelled between 33.3 and 254 km, with the longest foraging trips being performed by birds from Punta Norte (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 11.7, P < 0.05; Table 1). Penguins from Punta Norte reached the furthest distance from the colony (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 10.1, P < 0.05; Table 1). The total time spent at sea ranged between 10.7 and 49.8 h and was greater for birds from Punta Norte than for birds breeding at El Pedral (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 6.5, P < 0.05; Table 1). The mean distances travelled to reach the foraging areas and to return to the colony were similar between penguins from the three studied colonies (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 3.0, P > 0.05; \(\chi_{(2)}^{2}\) = 4.4, P > 0.05, respectively; Table 1). During the foraging phase, penguins from Punta Norte covered more distance than penguins from Islote Lobos and El Pedral (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 11.4, P < 0.05; Table 1). Penguins from Punta Norte travelled almost twice as fast as penguins from Islote Lobos and El Pedral during the three trip phases (GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 21.3, P < 0.001; GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 28.3, P < 0.05 and GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 24.1, P < 0.001 for the outbound, foraging and inbound phase, respectively; Table 1).
Diving behavior
There were substantial differences between colonies in the mean amount of time spent foraging during daylight hours, with penguins from Punta Norte spending almost twice the time spent by penguins from El Pedral (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 6.2, P < 0.05; Table 2).
Penguins from Punta Norte spent more time underwater than penguins from the other two colonies (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 7.7, P < 0.05; Table 2). Once underwater, the total time spent at the bottom was also higher for penguins from Punta Norte than for penguins from Islote Lobos and El Pedral (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 10.9, P < 0.05; Table 2). There was, however, no difference between colonies in the number of foraging dives performed per trip (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 2.7, P = 0.1; Table 2). The grand mean of dive depth for daytime foraging immersions was 38 ± 15 m with the deepest dive registered from a bird at Islote Lobos at 148 m. Penguins from the three sites dived in average to similar depths (GLMM, likelihood ratio test, \(\chi_{(2)}^{2}\) = 3.1, P = 0.2; Table 2); however, there were differences in the depth frequency distribution of foraging dives (Fig. 3). Penguins from Islote Lobos and Punta Norte performed 78 and 85 % of their daytime foraging dives in the first 50 m of the water column, respectively. However, penguins from Islote Lobos reached depths greater than penguins from Punta Norte (Fig. 3). Animals from El Pedral showed a bimodal distribution of the maximum depth reached during foraging dives with a first peak in the first 20 m of the water column and a second one at depths between 70 and 80 m (Fig. 3).
Both mean dive duration and mean bottom times were longer for penguins from Punta Norte than birds from Islote Lobos and El Pedral (GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 47.7, P < 0.01; LMM L ratio = 27.1, P < 0.001; Table 2); however, no differences were observed in the mean pause duration (GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 1.3, P = 0.5; Table 2). Accordingly, penguins from Punta Norte had a higher diving efficiency than those from Islote Lobos and El Pedral, spending a higher proportion of the complete diving cycle at the bottom phase of their dives (GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 17.9, P < 0.001; Table 2). The total amount of wiggles (i.e., indicative of prey capture) performed during daytime foraging dives was highest in birds from Punta Norte; however, penguins from this colony had the lowest CPUE (Kruskal–Wallis test, \(\chi_{(2)}^{2}\) = 8.2, P = 0.02; GLMM, likelihood ratio test χ 2 = 19.3, P < 0.001, respectively, Table 2).
The ratio between maximum dive duration and minimum pause duration diminished as maximum depth increased, and it was always higher for penguins from Punta Norte than for penguins from Islote Lobos and El Pedral (depth effect: LMM, L ratio = 309, P < 0.001; colony effect: LMM, L ratio = 97.5, P < 0.001; Fig. 4).
Horizontal speed versus CPUE
The horizontal speed of dives from Islote Lobos was not affected neither by the maximum depth reached during a dive (GLMM, likelihood ratio test \(\chi_{(1)}^{2}\) < 0.001, P = 0.9) nor by the CPUE (GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 1.8, P = 0.4) (y = 0.05e5.10−7x, y = −0.0003e4.7.10−7x and y = −0.05e4.7.10−7x for values of 0–1 CPUE, between 1 and 2 CPUE and equal or higher than 2 CPUE, respectively) (Fig. 5a). At Punta Norte, however, the horizontal speed was affected by the maximum depth reached during a particular dive, deeper dives being associated with lower horizontal speeds (GLMM, likelihood ratio test \(\chi_{(1)}^{2}\) = 9.7, P = 0.002). The horizontal speed by penguins from this colony was also affected by the CPUE (GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 7.9, P = 0.02), and for a particular diving depth, penguins moved slower in the horizontal dimension as the CPUE increased (Fig. 5b). The relationship between horizontal speed and maximum dive depth was y = 0.35e−0.0041x, y = 0.29e−0.0041x and y = 0.18e−0.0041x for values of 0–1 CPUE, between 1 and 2 CPUE and equal or higher than 2 CPUE, respectively (Fig. 5b). For penguins from El Pedral, the horizontal speed diminished as the maximum dive depth increased (GLMM, likelihood ratio test \(\chi_{(1)}^{2}\) = 5.6, P = 0.02); however, it was not affected by CPUE (GLMM, likelihood ratio test \(\chi_{(2)}^{2}\) = 4.9, P = 0.08) (y = 0.1e−0.003x, y = 0.06e−0.003x and y = −0.04e−0.003x for values of 0–1 CPUE, between 1 and 2 CPUE and equal or higher than 2 CPUE, respectively) (Fig. 5c).
Relationship between horizontal speed and maximum dive depth for dives from Islote Lobos, Punta Norte and El Pedral. On each panel, the three relationships shown correspond to dives with CPUE values between 0 and 1 (blue line), dives with CPUE values between 1 and 2 (green line) and dives with CPUE values equal or higher than 2 (red line)
Discussion
Previous studies performed on Magellanic penguins breeding along the Patagonian coast of Argentina have revealed significant differences in the foraging behavior of the species along its latitudinal range. These differences have been principally attributed to birds adjusting their behavior to varying conditions of prey abundance and type (Wilson et al. 2005; Boersma et al. 2009; Sala et al. 2012a, b, 2014a). Our study reinforces these findings, noting substantive differences in the foraging behavior between penguins from the three studied colonies. In contrast to previous studies though, our approach was performed on a much smaller spatial scale and with a smaller sample size. Despite the particularly small sample size at Punta Norte, our results are in line with that obtained for the same colony during previous years (Sala et al. 2012a, 2014a). Even though penguins from Islote Lobos may be ingesting Patagonian redfish Sebastes oculatus during their deepest dives (Ciancio et al. unpublished work), over the studied region penguins would be principally consuming the same prey species, Argentinean anchovy Engraulis anchoita (Scolaro et al. 1999; Wilson et al. 2005). Therefore, differences in the foraging behavior of penguins from Islote Lobos, El Pedral and Punta Norte would be principally related to dissimilar scenarios of prey abundance and distribution.
Differences in apparent prey densities around the three colonies could be linked to different intraspecific competition around each breeding site. Ashmole’s halo hypothesis (Ashmole 1963) predicts that if resources are evenly distributed, seabirds from larger colonies should have to travel further to find food than birds from smaller colonies. Several works have supported this idea (Lewis et al. 2001; Gaston 2004; Gaston et al. 2007); however, recent studies on the foraging behavior of Magellanic penguins along the Patagonian coast did not find a positive relationship between colony size and trip length, maximum foraging range or trip duration and this has been explained by the substantial heterogeneity in marine productivity along the Patagonian shelf (Boersma et al. 2009; Sala et al. 2012a). The contrast between our results and those previously found could be at least partially explained by the fact that our study was performed at a smaller spatial scale, possibly comprising a less heterogeneous area. In addition, in marked contrast to previous studies (Boersma et al. 2009; Sala et al. 2012a), data from the three colonies were collected during the same breeding season, eliminating the effect of interannual variability. Finally, the size difference between the studied colonies (180, 4750 and 135,000 breeding pairs for El Pedral, Islote Lobos and Punta Norte, respectively) (Schiavini et al. 2005; Pozzi et al. 2015) exceeds, by far, the difference in size between the colonies studied in previous works (e.g., Sala et al. 2012a).
Penguins from the three breeding sites concentrated their foraging effort in specific areas. Birds from Islote Lobos fed relatively near to the colony along the western coast of the San Matías Gulf. Upwelling events that carry subsurface, nutrient-rich waters to the upper layer and frequently lead to high primary productivity are common in this area (Pisoni et al. 2014) (Fig. 1). Prey supply could be increased in these zones of enhanced local primary production, thereby generating food-rich waters for penguins (Acha et al. 2004). Some of the areas exploited along the San Matías Gulf by penguins from Punta Norte also coincide with the presence of an oceanographic feature, in this case a thermohaline front. This front located during the austral summer around 41°50ʹS (from west to east) divides the gulf into saltier and warmer waters to the north and fresher and less salty waters to the south (Piola and Scasso 1988) (Fig. 1). The formation of this front attracts dense shoals of fish (Ocampo-Reinaldo et al. 2013), providing productive feeding grounds for penguins. Birds from El Pedral may be also exploiting waters with high densities of fish. Previous studies performed along the Nuevo Gulf revealed high concentrations of anchovy in the southern coast of the gulf (Degrati 2011), just where penguins from this study fed.
Comparisons of foraging parameters between colonies revealed that penguins from Punta Norte moved at higher horizontal speeds than birds from El Pedral and Islote Lobos, coinciding with the lowest CPUE values and implying that birds were working harder. Lower net rates of energy gain should tie in with longer foraging trips since birds have to spend more time looking for food and paying for the extra energy used to find it (e.g., Ballance et al. 2009; Sala et al. 2012a). Our results were in line with this: Penguins from Punta Norte not only made the longest foraging trips but also spent more time per dive underwater and more (depth-corrected) time at the bottom, which would increase the probability of finding prey (Wilson et al. 1996). All this would point to higher rates of energy expenditure, presumably because this strategy is necessary to maintain appropriate provisioning rates (Ropert-Coudert et al. 2004; Sala et al. 2014b). The overall higher energy expenditure would also presumably necessitate higher total amounts of prey caught per trip, which is what we observed, manifest in penguins from Punta Norte exhibiting more wiggles per foraging trip than birds from the other two colonies. Considering the negative relationship between the foraging effort and the rate of population change found in the Magellanic penguin (Sala et al. 2012a, 2014b) and in other seabird species (e.g., Lewis et al. 2006), we would expect Punta Norte colony to have the lowest rate of population change of the three studied sites. This is in fact what was recently showed by Pozzi et al. 2015.
Air-breathing divers must balance the time between the surface and the feeding site at depth in order to maximize the proportion of time spent foraging (Kramer 1988; Wilson 2003). Extended pause durations can be an optimal strategy if the rate of prey acquisition in a given patch is high (Mori 1998a, b, 1999) although theoretical treatises on this assume that the patch is not temporarily limited while some work on penguins indicates that it is (e.g., Wilson et al. 2010). However, under less favorable scenarios, and particularly if patch size is only available for a short time (as would be the case, for example, where a small shoal of fish at depth could only be relocated if the dive–pause were minimal—cf. Wilson et al. 2011), divers are expected to minimize the time spent on the surface in order to increase the probability of encountering another prey patch (Wilson and Wilson 1995). This might explain the differential results in dive–pause ratios in birds from Punta Norte compared to penguins from Islote Lobos and El Pedral.
Optimal foraging theory predicts that animals should spend more time on higher quality “patches” (McNair 1982; Stephens and Krebs 1986). Our study of a corollary of this, traveling speed, in diving birds is complicated by dive depth because vertical displacement increases in deeper dives (Petersen et al. 2006; Boersma and Rebstock 2009; Raya Rey et al. 2010; Sala et al. 2012a) due to greater dive angles (Wilson et al. 1996; Ropert-Coudert et al. 2001). Thus, deeper dives are generally correlated with lower horizontal speeds (Cotté et al. 2007; Wilson et al. 2005; Boersma and Rebstock 2009; Raya Rey et al. 2010; Sala et al. 2012a). Once prey is detected, predators are expected to increase their search effort, this being linked to the high probability of encountering other prey items nearby (Fauchald and Tveraa 2003; Weimerskirch et al. 2007). This would be especially the case for those predators feeding on prey that form aggregations such as penguins feeding on schools of pelagic fish. However, lower horizontal speeds were associated with higher derived prey abundance only at Punta Norte. The differences between sites in the relationship between the horizontal speed and CPUE could be linked to dissimilarities in the way anchovy shoals are distributed along the three foraging areas. Under a scenario of scattered shoals, we would expect penguins to remain more time and move slowly once a shoal is found and travel quickly between shoals. However, under a situation where fish is relatively more homogenously distributed, it might not be as necessary to diminish speed as CPUE increases. Although there are no studies that compare how anchovy is distributed along the areas where penguins from this study foraged, our results suggest more scattered shoals of fish in the vicinity of Punta Norte than around Islote Lobos and El Pedral. A detailed analysis on the relationship between penguin’s horizontal movement and prey abundance and distribution would be crucial since it may be used to predict certain prey characteristics around an area by the use of speed information. Moreover, for penguins preying on a commercial fish species such as Magellanic penguins feeding on Argentinean anchovy, the existence of a relationship between speed data and prey distribution and abundance would allow the former to be used as an additional tool for fish stock assessment and management.
Our results suggest that there would appear to be differences in the abundance and distribution of anchovy around the breeding sites. This can be inferred by analyzing general foraging trip parameters and also by studying the relationship between horizontal movement and CPUE. Moreover, our results show that it is crucial to continue monitoring the foraging effort of Magellanic penguins. Despite a reduced sample size, specially at Punta Norte where 135,000 breeding pairs reproduce (Pozzi et al. 2015), our results suggest that the good foraging conditions around El Pedral and Islote Lobos, together with nest-site suitability and availability and no prevalence of predators, would promote the increase in these two relatively new colonies. Monitoring the breeding output and foraging success of both colonies in relation to resource distribution and environmental variability would help elucidate to what extent food availability is responsible of the northward breeding range expansion of this penguin species.
References
Acha EM, Mianzan HW, Guerrero RA, Favero M, Bava J (2004) Marine fronts at the continental shelves of austral South America: physical and ecological processes. J Mar Syst 44:83–105
Ashmole NP (1963) The regulation of numbers of tropical oceanic birds. Ibis 103:458–473
Ballance LT, Ainley DG, Ballard G, Barton K (2009) An energetic correlate between colony size and foraging effort in seabirds, an example of the Adélie penguin Pygoscelis adeliae. J Avian Biol 40:279–288
Bertellotti M, Tella JL, Godoy JA, Blanco G, Forero MG, Donázar JA, Ceballos O (2002) Determining sex of magellanic penguins using molecular procedures and discriminant functions. Waterbirds 25:479–484
Boersma PD, Rebstock GA (2009) Foraging distance affects reproductive success in Magellanic penguins. Mar Ecol Prog Ser 375:263–275
Boersma PD, Rebstock GA, Frere E, Moore SE (2009) Following the fish: penguins and productivity in the South Atlantic. Ecol Monogr 79(1):59–76
Bost C, Handrich Y, Butler P, Fahlman A, Halsey LG, Woakes A, Ropert-Coudert Y (2007) Changes in dive profiles as an indicator of feeding success in king and Adélie penguins. Deep Sea Res II 54:248–255
Brooke ML (2004) The food consumption of the word’s seabirds. Proc R Soc B (Suppl) 27:S246–S248
Burger AE, Shaffer SA (2008) Application of tracking and data-logging technology in research and conservation of seabirds. Auk 125:253–264
Calenge C (2006) The package “Adehabitat” for the R software: a tool for the analysis of space and habitat use by animals. Ecol Model 197:516–519
Cotté C, Park YH, Guinet C, Bost CA (2007) Movements of foraging king penguins through marine mesoscale eddies. Proc Biol Sci 274:2385–2391
Crawley MJ (2007) The R book. Wiley, West Sussex, England
Degrati M (2011) Patrón de actividad y estrategias de alimentación del delfín oscuro (Lagenorhynchus obscurus) en el norte de Patagonia. Doctoral thesis. Universidad Nacional del Comahue, Río Negro
Fauchald P, Erikstad KE (2002) Scale-dependent predator-prey interactions: the aggregative response of seabirds to prey under variable prey abundance and patchiness. Mar Ecol Prog Ser 231:279–291
Fauchald P, Tveraa T (2003) Using first-passage time in the analysis of area-restricted search and habitat selection. Ecology 84:282–288
Gandini PA, Frere E, Holik TM (1992) Implicancias de las diferencias en el tamaño corporal entre colonias para el uso de medidas morfométricas como métodos de sexado en Spheniscus magellanicus. Hornero 13:211–213
Gaston AJ (2004) Seabirds: a natural history. T. & A.D. Poyser, London, UK, p 222
Gaston AJ, Ydenberg RC, Smith GEJ (2007) Ashmole´s halo and population regulation in seabirds. Mar Ornithol 35:119–126
Grémillet D, Pichegru L, Siorat F, Georges JY (2006) Conservation implications of the apparent mismatch between population dynamics and foraging effort in French northern gannets from the English Channel. Mar Ecol Prog Ser 319:15–25
Hamer KC, Lewis S, Wanless S, Phillips RA, Sherrat TN, Humphreys EM, Hennicke J, Garthe S (2006) Use of gannets to monitor prey availability in the northeast Atlantic Ocean: colony size, diet and foraging behaviour. In: Boyd IL, Wanless S, Camphuysen CJ (eds) Top predators in marine ecosystems, their role in monitoring and management. Cambridge University Press, Cambridge, pp 191–210
Hanuise N, Bost CA, Huin W, Auber A, Halsey LG, Handrich Y (2010) Measuring foraging activity in a deep-diving bird: comparing wiggles, oesophageal temperatures and beak-opening angles as proxies of feeding. J Exp Biol 213:3874–3880
Harding AMA, Piatt JF, Schmutz JA, Shultz MT, van Pelt TI, Kettle AB, Speckman SG (2007) Prey density and the behavioral flexibility of a marine predator: the common murre (Uria aalge). Ecology 88:2024–2033
Hunt GL (1990) The pelagic distribution of marine birds in a heterogeneous environment. Polar Res 8:43–54
Jodice PGR, Rody DD, Turco KR, Suryan RM, Irons DB, Piatt JF, Shultz MT, Roseneau DG, Kettle AB, Anthony JA (2006) Assessing the nutritional stress hypothesis: relative influence of diet quantity and quality on seabird productivity. Mar Ecol Prog Ser 325:267–279
Kramer DL (1988) The behaviour ecology of air breathing by aquatic animals. Can J Zool 66:89–94
Lewis S, Sherratt TN, Hamer KC, Wanless S (2001) Evidence of intra-specific competition for food in a pelagic seabird. Nature 412:816–819
Lewis S, Gremillet D, Daunt F, Ryan PG, Crawford RJM, Wanless S (2006) Using behavioural and state variables to identify proximate causes of population change in a seabird. Oecologia 147:606–614
McNair JM (1982) Optimal giving-up time and the marginal value theorem. Am Naturalist 119:511–529
Mori Y (1998a) Optimal choice of foraging depth in divers. J Zool 245:279–283
Mori Y (1998b) The optimal patch use in divers: optimal time budget and the number of dive cycles during bout. J Theor Biol 190:187–199
Mori Y (1999) The optimal allocation of time and respiratory metabolism over the dive cycle. Behav Ecol 10:155–160
Ocampo-Reinaldo M, González R, Williams G, Storero LP, Romero MA, Narvarte M, Gagliardini DA (2013) Spatial patterns of the Argentine hake Merluccius hubbsi and oceanographic processes in a semi-enclosed Patagonian ecosystem. Mar Biol Res 9(4):394–406
Orians GH, Pearson NE (1979) On the theory of central place foraging. In: Horn DJ, Mitchell RD, Stairs GR (eds) Analysis of ecological systems. Ohio State University Press, Columbus, pp 154–177
Petersen SL, Ryan PG, Gremillet D (2006) Is food availability limiting African Penguins Spheniscus demersus at Boulders? A comparison of foraging effort at mainland and island colonies. Ibis 148:14–26
Piola AR, Scasso LM (1988) Circulación en el Golfo San Matías. Geoacta 15(1):33–51
Pisoni JP, Rivas A, Piola AR (2014) Satellite remote sensing reveals coastal upwelling events in the San Matías Gulf- Northern Patagonia. Remote Sens Environ 152:270–278
Pozzi LM, Garcìa Borboroglu P, Boersma PD, Pascual MA (2015) Building a Metapopulation perspective for the Magellanic Penguin in Argentina. PLoS ONE. doi:10.1371/journal.pone.0119002
Pyke GH (1984) Optimal foraging theory: a critical review. Annu Rev Ecol Evol S 15:523–575
R Development Core Team I (2011) R: A language and environment for statistical computing. In R Foundation for Statistical Computing. Vienna, Austria: ISBN 3-900051-07-0, URL http://www.R-project.org/
Raya Ray A, Pütz K, Scioscia G, Lüthi B, Schiavini A (2012) Sexual differences in the foraging behaviour of Magellanic Penguins related to stage of breeding. Emu 112:90–96
Raya Rey A, Bost CA, Schiavini A, Pütz C (2010) Foraging movements of Magellanic Penguins Spheniscus magellanicus in the Beagle Channel, Argentina, related to tide and tidal currents. J Ornithol 151:933–943
Ropert-Coudert Y, Kato A, Baudat J, Bost CA, Le Maho Y, Naito Y (2001) Time/depth usage of Adélie penguins: an approach based on dive angles. Polar Biol 24:467–470
Ropert-Coudert Y, Wilson RP, Daunt F, Kato A (2004) Patterns of energy acquisition by a central place forager: benefits of alternating short and long foraging trips. Behav Ecol 15:824–830
Sala JE, Wilson RP, Frere E, Quintana F (2012a) Foraging effort in Magellanic penguins in coastal Patagonia, Argentina. Mar Ecol Prog Ser 464:273–287
Sala JE, Wilson RP, Quintana F (2012b) How much is too much? Assessment of prey consumption by Magellanic penguins in Patagonian colonies. PLoS ONE 7
Sala JE, Wilson RP, Quintana F (2014a) Foraging effort in Magellanic penguins: balancing the energy books for survival? Mar Biol. doi:10.1007/s00227-014-2581-9
Sala JE, Wilson RP, Frere E, Quintana F (2014b) Flexible foraging for finding fish: variable diving patterns in Magellanic Penguins Spheniscus magellanicus from different colonies. J Ornithol. doi:10.1007/s10336-014-1065-5
Schiavini A, Yorio P, Gandini P, Raya Rey A, Boersma PD (2005) Los pingüinos de las costas argentinas: estado poblacional y conservacion. Hornero 20:5–23
Scolaro JA, Wilson RP, Laurenti S, Kierspel M, Gallelli H, Upton J (1999) Feeding preferences of the magellanic penguin over its breeding range in Argentina. Waterbirds 22:104–110
Simeone A, Wilson RP (2003) In-depth studies of Magellanic penguin (Spheniscus magellanicus) foraging: can we estimate consumption by perturbations in the dive profile? Mar Biol 143:825–831
Stearns SC (1992) The evolution of life history. Oxford University Press, New York, USA
Stephens DW, Krebs JR (1986) Foraging theory. Princeton University Press, Princeton, New Jersey
Tonini MH, Palma ED, Piola AR (2013) A numerical study of gyres, thermal fronts and seasonal circulation in austral semi-enclosed gulfs. Cont Shelf Res 65:97–110
Tremblay Y, Cherel Y (2000) Benthic and pelagic dives: a new foraging behaviour in rockhopper penguins. Mar Ecol Prog Ser 204: 257–267
Weimerskirch H, Pinaud D, Pawlowski F, Bost CA (2007) Does prey capture induce area restricted search? A fine-scale study using GPS in a marine predator, the Wandering Albatross. Am Nat 170:734–743
Williams TD (1995) The penguins. Oxford University Press, Oxford
Wilson RP (1995) The foraging ecology of penguins. In: Williams TD (ed) The penguins. Oxford University Press, Oxford, pp 81–106
Wilson RP (2003) Penguins predict their performance. Mar Ecol Prog Ser 249:305–310
Wilson RP, Wilson MP (1995) The foraging behaviour of the African penguins. In: Dann P, Norman I, Reilly P (eds) The Penguins: ecology and management. Surrey Beatty & Sons, Sydney, pp 244–265
Wilson RP, Culik BM, Peters G, Bannasch R (1996) Diving behaviour of Gentoo penguins, Pygoscelis papua; factors keeping dive profiles in shape. Mar Biol 126:153–162
Wilson RP, Putz K, Charrassin JB, Lage J (1997) Long-term attachment of transmitting and recording devices to penguins and other seabirds. Wildl Soc Bull 25:101–106
Wilson RP, Scolaro JA, Gremillet D, Kierspel MAM et al (2005) How do Magellanic penguins cope with variability in their access to prey. Ecol Monogr 75:379–401
Wilson RW, Jackson S, Thor Straten M (2007) Rates of food consumption in free-living magellanic penguins Spheniscus magellanicus. Mar Ornithol 35:109–111
Wilson R, Shepard ELC, Gómez Laich A, Frere E, Quintana F (2010) Pedalling downhill and freewheeling up; a penguin perspective on foraging. Aquatic Biol 8:193–202
Wilson RP, McMahon CR, Quintana F, Frere E, Scolaro A, Hays GC, Bradshaw C (2011) N-dimensional animal energetic niches clarify behavioural options in a variable marine environment. J Exp Biol 214:646–656
Worton BJ (1989) Kernel methods for estimating the utilization in home-range studies. Ecology 70:164–168
Acknowledgments
This research was funded by grants from the Wildlife Conservation Society, Consejo Nacional de Investigaciones Científicas y Técnicas de la República Argentina (CONICET) and Agencia de Promoción Científica y Tecnológica to F. Quintana and by a Rolex Award for Enterprise awarded to R. P. Wilson. We would like to thank the Conservation Agencies of Chubut and Río Negro for the permits to work in the different protected areas and the Centro Nacional Patagónico (CENPAT-CONICET) for institutional and logistical support. A. We also thank Javier Ciancio and Juan Pablo Pisoni for their valuable help, Patricia Dell´Arciprete for her technical assistance and two anonymous reviewers for their valuable comments on an earlier version of the manuscript. During the development of the present study, A. Gómez-Laich was supported by a postdoctoral fellowship from the Consejo Nacional de Investigaciones Científicas y Técnicas de la República Argentina (CONICET).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Garthe.
Reviewed by M. Berlincourt and an undisclosed expert.
Rights and permissions
About this article
Cite this article
Gómez-Laich, A., Wilson, R.P., Sala, J.E. et al. Moving northward: comparison of the foraging effort of Magellanic penguins from three colonies of northern Patagonia. Mar Biol 162, 1451–1461 (2015). https://doi.org/10.1007/s00227-015-2681-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-015-2681-1