Abstract
Purpose
To evaluate low-dose non-enhanced CT (ldCT) and full-dose contrast-enhanced CT (ceCT) in integrated 18F-fluorodeoxyglucose (FDG) PET/CT studies for restaging of uterine cancer.
Methods
A group of 100 women who had undergone treatment for uterine cervical (n=55) or endometrial cancer (n=45) underwent a conventional PET/CT scans with ldCT, and then a ceCT scan. Two observers retrospectively reviewed and interpreted the PET/ldCT and PET/ceCT images in consensus using a three-point grading scale (negative, equivocal, or positive) per patient and per lesion. Final diagnoses were obtained by histopathological examination, or clinical follow-up for at least 6 months.
Results
Patient-based analysis showed that the sensitivity, specificity and accuracy of PET/ceCT were 90% (27/30), 97% (68/70) and 95% (95/100), respectively, whereas those of PET/ldCT were 83% (25/30), 94% (66/70) and 91% (91/100), respectively. Sensitivity, specificity and accuracy did not significantly differ between two methods (McNemar test, p=0.48, p=0.48, and p=0.13, respectively). There were 52 sites of lesion recurrence: 12 pelvic lymph node (LN), 11 local recurrence, 8 peritoneum, 7 abdominal LN, 5 lung, 3 supraclavicular LN, 3 liver, 2 mediastinal LN, and 1 muscle and bone. The grading results for the 52 sites of recurrence were: negative 5, equivocal 0 and positive 47 for PET/ceCT, and negative 5, equivocal 4 and positive 43 for PET/ldCT, respectively. Four equivocal regions by PET/ldCT (local recurrence, pelvic LN metastasis, liver metastasis and muscle metastasis) were correctly interpreted as positive by PET/ceCT.
Conclusion
PET/ceCT is an accurate imaging modality for the assessment of uterine cancer recurrence. Its use reduces the frequency of equivocal interpretations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Despite continuing advances in surgical and nonsurgical therapeutic strategies, cancer recurrence and distant metastasis after initial treatment are often a major problem for women with gynaecological cancer. Early and accurate detection of recurrence in patients with uterine cancer has an important influence on therapy, and selection of appropriate treatment strategies can be expected to have a significant impact on overall survival [1, 2].
In the late 1990s, positron emission tomography (PET) with 18F-fluorodeoxyglucose (FDG), which exploits the increased utilization of glucose by malignant cells and thereby their high uptake of glucose, has opened a new field in clinical imaging and is widely used for staging, restaging, therapeutic response monitoring and prognostication in patients with various cancers. PET makes it possible to diagnose cancer recurrence and distant metastasis in the preclinical stage before it becomes evident by conventional imaging modalities such as CT and MRI. However, PET does not provide anatomical information and precise localization of any suspicious lesions may be difficult. Recently, an integrated PET/CT system, in which a full-ring detector clinical PET scanner and multidetector row helical CT scanner are combined, has made it possible to acquire both metabolic and anatomical imaging data using a single device in a single diagnostic session, and provides precise anatomical localization of suspicious areas of increased FDG uptake and rules out false-positive PET findings [3].
Today, different approaches are adopted for PET/CT scanning, with the CT protocol positioned between two possible extremes: (1) CT is used as a fast transmission source for attenuation correction and approximate anatomical mapping and is performed with a low radiation dose (“low-dose CT”), and (2) CT is used for attenuation correction and diagnostic purposes, being performed with a standard radiation dose and administration of intravenous contrast agent (“diagnostic CT”).
There have been several studies of FDG PET/CT [4–11] that have shown its usefulness for restaging uterine cancer and all of these studies except one [10] used low-dose non-enhanced CT (ldCT) for the CT component. However, the one study that used full-dose contrast-enhanced CT (ceCT) did not compare the diagnostic difference between PET/ceCT and PET/ldCT, and therefore the usefulness of intravenous contrast medium in PET/CT examination was strictly not clarified [10]. The purpose of the present study was to compare the diagnostic performance of PET/ceCT and PET/ldCT for restaging patients previously treated for uterine cancer, and to investigate the clinical value of intravenous contrast agent in the PET/CT scan.
Materials and methods
Patients
A group of 100 patients (age range 32–88 years, mean 60 years) who had undergone treatment for histopathologically proven uterine cervical (n=55) or endometrial cancer (n=45) underwent PET/CT scans with intravenous contrast agent for suspected recurrence at our institution between December 2007 and February 2009 were included in this study with the approval of the institutional review board. Informed consent was obtained from each patient after the procedures were fully explained. Further details of these patients and their demographic data are listed in Table 1. Recurrence was suspected on the basis of physical examination, elevated tumour marker levels, abnormal CT and/or MR imaging findings, and an abnormal Papanicolaou (Pap) smear.
FDG-PET/CT study
Whole-body imaging was performed using a combined PET/CT scanner (Discovery ST Elite-Performance, GE Healthcare, Waukesha, WI). The CT scan covered a region ranging from the meatus of the ear to the mid-thigh. The technical parameters of the 16-detector row helical CT scanner were: pitch 6 (high-speed mode), gantry rotation speed 0.6 s and slice thickness 3.75 mm. The PET component of the combined imaging system allowed simultaneous acquisition of 47 transaxial PET images with an interslice spacing of 3.75 mm in one bed position and provided an image from the meatus of the ear to the mid-thigh with seven or eight bed positions. The transaxial field of view and axial field of view of the PET images reconstructed for fusion were 70 cm and 15.9 cm, respectively, with a matrix size of 128×128. To avoid artefacts caused by the urinary tract, patients were asked to drink 500 ml of water 1 to 2 h prior to image acquisition, and to void just before the start of acquisition. No urinary bladder catheterization was used. After at least 4 h of fasting, patients received an intravenous injection of 3.33 MBq/kg body weight of 18F-FDG. The blood glucose levels were checked in all patients before FDG injection and no patients showed a blood glucose level of more than 160 mg/dl.
About 50 min later, initially a ldCT scan was performed at 140 kV and 40 mA with the normal expiration position for attenuation correction of PET images. A whole-body emission PET scan was performed immediately after the ldCT scan, with a 2-min acquisition per bed position using a three-dimensional acquisition mode. If the body mass index exceeded 30 kg/m2, the acquisition time for the PET scan was 2.5 min. Finally, a diagnostic ceCT scan was performed with the same axial coverage at 120 kV, 350 mA, and 27.0 mm/rotation, during breath hold with the normal expiration position, similar to the ldCT scan. Iodinated contrast agent (Iopamiron Inj, Syringe; Bayer Schering Pharma, Berlin, Germany) containing 300 or 370 mg of iodine was administered intravenously via a power injector at a rate of 2.0–2.5 ml/s to a total volume of 80–100 ml, and the scan was started 100 s after injection. For image fusion, 3.75-mm slices were reconstructed. The ldCT, ceCT and PET images were transferred to a commercially available workstation (Xeleris, GE Healthcare) to access all the data. Attenuation-corrected PET images were reconstructed with an ordered-subset expectation maximization iterative reconstruction algorithm called VUE Point Plus. No oral contrast agent was administered.
Image analysis
Two experienced radiologists (reader A and reader B with 4 and 6 years of experience in PET/CT, respectively) who had knowledge of neither the other imaging results nor the clinical data retrospectively interpreted the PET/ceCT images and PET/ldCT images in consensus using a three-point scale (negative, equivocal or positive) per patient and per lesion. These two datasets were reviewed after a minimum interval of 1 month to avoid decision threshold bias due to reading-order effects. Two experienced radiologists (reader C and reader D with 8 and 18 years of experience in CT, respectively) who had knowledge of neither the other imaging results nor the clinical data retrospectively interpreted ceCT images in consensus using a three-point scale per patient and per lesion. Diagnostic ability was determined on a patient basis and on the basis of nine lesion sites: supraclavicular lymph node (LN), mediastinal and hilar LN, abdominal LN (including paraaortic, mesenteric and retrocrural LN), pelvic LN, lung, liver, peritoneum, muscle and bone, and pelvic local recurrence. On PET/ceCT and PET/ldCT, recurrent or metastatic lesions were diagnosed when abnormal focal FDG uptake observed on PET images corresponded to an abnormal mass on CT. LNs with increased glucose uptake were deemed positive for metastatic spread even if their short-axis diameter was less than 1 cm. Conversely, LNs with no detectable tracer uptake were deemed negative for metastatic spread, even if their short-axis diameter was more than 1 cm. Semiquantitative analysis was not done in this study. This method of PET/CT image analysis was based on previous studies [4–7, 9, 10]. On ceCT, LNs with a short-axis diameter of 10 mm or more, between 6 and 9 mm, and 5 mm or less were defined as malignant, equivocal and benign, respectively. Furthermore, the presence of a central unenhanced area suggesting central necrosis was considered a sign of malignancy, and the presence of peripheral low attenuation suggesting a fatty hilum within a LN was considered a benign sign, regardless of node size [12].
The final diagnosis was obtained from the results of histopathological examination after surgery or biopsy, or clinical follow-up of at least 6 months (range 6–20 months, mean 14 months) on the basis of tumour marker levels, ceCT findings and PET/ceCT findings. Cases were classified as recurrence and/or metastasis if: (1) the present study revealed highly suspected recurrence without pathological evidence, and the patient underwent chemotherapy resulting in a decrease or disappearance in size and/or FDG uptake in the follow-up study; and (2) the follow-up study revealed tumour recurrence by CT and/or PET/CT in a place where a tiny lesion without FDG uptake was imaged at the time of the initial (present) study.
Statistical analysis
Patient-based and lesion site-based analyses of the PET/ceCT results were performed based on the consensus verdict in general, compared with the PET/ldCT and ceCT findings. Sensitivity, specificity and accuracy were calculated defining equivocal interpretations as negativity for the sensitivity and positivity for the specificity to evaluate the diagnostic ability with certainty. Differences between the two imaging modalities (PET/ceCT vs. PET/ldCT and PET/ceCT vs. ceCT) were tested with the McNemar test. P values less than 0.05 were considered to indicate statistical significance.
Results
Patient-based analysis
In 30 (30.0%) of the 100 patients, recurrence and/or distant metastasis was confirmed by pathological examination and a clinical follow-up study and imaging. In the other 70 patients (70.0%), no recurrence was confirmed by the clinical follow-up study of tumour markers and imaging for at least 6 months. The grading results for detecting the 30 patients with recurrence were: negative 3, equivocal 0 and positive 27 for PET/ceCT; negative 3, equivocal 2 and positive 25 for PET/ldCT; and negative 7, equivocal 2 and positive 21 for ceCT (Table 2). Two patients with equivocal findings on PET/ldCT (one patient with local recurrence, and the other with liver metastasis) were correctly interpreted as positive on PET/ceCT. There were three patients with false-negative findings on both PET/ceCT and PET/ldCT, of whom two had a tiny local recurrence and one had tiny pelvic LN metastasis.
The imaging results in the 70 patients without recurrence were: negative 68, equivocal 0 and positive 2 for PET/ceCT; negative 66, equivocal 2 and positive 2 for PET/ldCT; and negative 66, equivocal 2 and positive 2 for ceCT. Two patients had false-positive findings on PET/ceCT, one focus being physiological FDG uptake in the ovary that was misinterpreted as pelvic LN metastasis and the other focus being physiological FDG uptake in the liver that was misinterpreted as liver metastasis. Two patients had false-positive findings on PET/ldCT, one focus being physiological FDG uptake in the ovary that was misinterpreted as pelvic LN metastasis and the other focus being physiological FDG uptake in the intestine that was misinterpreted as peritoneal dissemination. Two patients had equivocal findings on PET/ldCT, one focus being physiological FDG uptake in the liver that was misinterpreted as liver metastasis and the other focus being physiological uptake in the inner iliac vessel that was misinterpreted as pelvic LN metastasis.
In the patient-based analysis the sensitivity, specificity and accuracy of PET/ceCT were 90.0% (27/30), 97.1% (68/70) and 95.0% (95/100), respectively, and of PET/ldCT were 83.3% (25/30), 94.3% (66/70) and 91.0% (91/100), and of ceCT were 70.0% (21/30), 94.3% (66/70) and 87.0% (87/100), respectively. Although PET/ceCT offered a little better sensitivity, specificity and accuracy than PET/ldCT, the sensitivity, specificity and accuracy did not significantly differ between the two imaging modalities (McNemar test, p=0.4795, p=0.4795, and p=0.1336, respectively). PET/ceCT offered significantly better sensitivity and accuracy than ceCT (McNemar test, p=0.0412 and p=0.0133, respectively) and there was no significant difference in specificity (p=0.4795).
For patients with cervical cancer, patient-based sensitivity, specificity and accuracy of PET/ceCT were 88.2% (15/17), 97.4% (37/38) and 94.5% (52/55), respectively, and for patients with endometrial cancer, those of PET/ceCT were 92.3% (12/13), 96.9% (31/32) and 95.6% (43/45), respectively.
Lesion site-based analysis
Nine lesion sites, including the retrovesical region, lung, liver, muscle, peritoneum, supraclavicular LN, mediastinal and hilar LN, abdominal LN, and pelvic LN, were evaluated in each patient. There were 52 sites of lesion recurrence: 12 in pelvic LNs, 11 in the retrovesical regions, 8 in the peritoneum, 7 in paraaortic LNs, 5 in the lung, 3 in the liver, 3 in supraclavicular LNs, 2 in mediastinal and hilar LNs, and 1 in muscle. The grading results for detecting the 52 recurrent regions were: negative 5, equivocal 0 and positive 47 for PET/ceCT; negative 5, equivocal 4 and positive 43 for PET/ldCT; and negative 14, equivocal 6 and positive 32 for ceCT (Table 3). Four regions interpreted as equivocal on PET/ldCT (local recurrence, pelvic LN metastasis, liver metastasis, and muscle metastasis) were correctly interpreted as positive on PET/ceCT. Five regions were interpreted as false-negative on both PET/ceCT and PET/ldCT: two local recurrence, one pelvic LN metastasis, one paraaortic LN metastasis, and one peritoneum. Overall sensitivity for LN metastasis including supraclavicular LNs, mediastinal and hilar LNs, abdominal LNs and pelvic LNs, on PET/ceCT, PET/ldCT and ceCT were 91.7% (22/24), 87.5% (21/24) and 58.3% (14/24), respectively. Two representative cases are shown in Figs. 1 and 2.
A 56-year-old woman with local recurrence. a Low-dose non-enhanced CT image shows no abnormal findings. b PET/ldCT image shows abnormal FDG uptake corresponding to a soft-tissue density mass, suggestive of local recurrence. The image was interpreted equivocal. c Full-dose contrast-enhanced CT image shows an enhanced soft-tissue density mass in the resected region. The image was interpreted positive. d PET/ceCT image shows abnormal FDG uptake corresponding to an enhanced soft-tissue density mass, and therefore the image was interpreted as local recurrence (positive). Histopathological examination of the surgical specimen revealed local recurrence
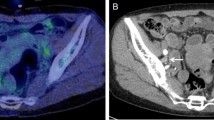
A 41-year-old woman with bilateral pelvic LN metastases. a Low-dose non-enhanced CT image shows no abnormal findings. b PET/ldCT image shows abnormal FDG uptake corresponding to two small nodules near the bilateral inner iliac vessels, suggestive of bilateral pelvic LN metastases (arrows). The image was interpreted as equivocal. c Full-dose contrast-enhanced CT image shows bilateral small iliac LNs 7 and 8 mm in short axis diameter with mild enhancement (arrows). The image was interpreted as equivocal. d PET/ceCT image shows abnormal FDG uptake corresponding to bilateral small inner iliac LNs and therefore bilateral pelvic LN metastases was interpreted (arrows). Histopathological examination of the surgical specimen revealed bilateral pelvic LN metastases
Discussion
To our knowledge, this is the first study to have investigated the additional diagnostic value of integrated PET/ceCT over PET/ldCT for diagnosis of uterine cancer recurrence. Our results showed that patient-based sensitivity, specificity and accuracy of PET/ceCT were 90%, 97% and 95%, respectively, whereas those of PET/ldCT were 83%, 94% and 91%, respectively. The difference, although not statistically significant, between the two methods in sensitivity, specificity and accuracy occurred because of the way “equivocal” was defined as positive or negative as described in the Image analysis section. Further study in a large population is required to obtain more evidence. Moreover, with PET/ceCT, the proportion of equivocal findings on the per patient basis decreased by 4.0% (from 4 of 100 patients to 0 of 100) and on the per lesion basis decreased by 7.7% (from 4 of 52 lesions to 0 of 52). PET/ceCT led to fewer or no equivocal interpretations and accurate diagnostic performance with more certainty. As in our series, conventional PET/CT scans without intravenous contrast agent often leads to some equivocal interpretations. Depending on the level of clinical suspicion, equivocal interpretations may require further evaluation with additional imaging studies such as ceCT and MRI, and biopsy. This may be associated with additional costs for the health-care system and an emotional burden on the patient involved. In contrast, the use of iodine-based intravenous contrast material allows more accurate and confident interpretations, and PET/ceCT reduces the frequency of equivocal interpretations, and therefore minimizes the burden on the patient. The significant decrease in the number of equivocal interpretations afforded by PET/ceCT, associated with consistently high accuracy in image interpretation, is therefore of immediate clinical relevance. Although PET/ceCT is a real “one-stop-shop” examination, the use of intravenous contrast agent in the PET/CT scan may not be justified for clinical routine examinations in the view of the prevalence and cost.
Even PET/ceCT could not detect several tiny lesions in our series. The spatial resolution of PET scans is insufficient for the detection of small lesions. With spatial resolutions of 4 to 6 mm for currently available PET and PET/CT systems, the detection of microscopic lesions remains challenging. Improving the spatial resolution and sensitivity of PET and PET/CT scanners and developing new, more specific radioactive tracers may help to overcome this limitation in the future.
With regard to posttherapy surveillance, in several studies integrated PET/ldCT has shown a sensitivity of 90–100%, a specificity of 81–100% and an accuracy of 87–97% in the clinical diagnosis of uterine cervical cancer [4, 5, 9, 11] and endometrial cancer [5–8], and in one study integrated PET/ceCT has shown a sensitivity of 91%, a specificity of 94% and an accuracy of 92% in the clinical diagnosis of uterine cervical cancer and endometrial cancer [10]. Our results were almost the same. In the previous studies, false-negative findings included local recurrence and paraaortic LN metastasis, and false-positive findings included local recurrence, lung metastasis, bone metastasis, peritoneal dissemination, mediastinal and hilar LN metastasis, and axillary LN metastasis [4–11].
The use of CT intravenous contrast agents in PET/CT is still controversial. Some have argued that CT image data should be used only for attenuation correction of PET, for reduction of acquisition times, to prevent overestimation of PET attenuation factors by intravenous contrast media, and to localize hypermetabolic lesions with a low radiation dose (“low-dose CT”) [13–15], whereas others advocate the need to perform full-dose, contrast-enhanced, and high-resolution CT (“diagnostic CT”) [16, 17]. As an integrated PET/CT scanner has been installed in many institutions, an increasing number of reports describing the clinical usefulness of iodine-based contrast material for PET/CT scanning have appeared. There have been several reports of the superiority PET/ceCT over PET/ldCT in staging malignant lymphoma [18], staging and therapy planning of non-small-cell lung cancer [19], evaluating the preoperative nodal status of rectal cancer [20], evaluating the nodal status of pelvic and retroperitoneal lymphatic pathways in malignant lymphoma [21], assessing the resectability of pancreatic cancer [22], restaging ovarian cancer [23], and restaging colon cancer [24, 25]. A recent study has demonstrated that although there is an increase in standardized uptake values in normal and pathological regions of high contrast agent concentration when ceCT is used for attenuation, this increase is clinically insignificant, and ceCT can be used in combination with PET in the evaluation of patients with cancer [26]. We used ldCT for attenuation correction in our series to prevent overestimation of PET attenuation factors by intravenous contrast media because when ceCT is used for attenuation, the problem of high radiation exposure can occur. Further study in a larger patient population is needed to elucidate the efficacy, radiation exposure, and cost-effectiveness of PET/ceCT.
This study had certain limitations. First, the gold standard for any analysis would be histological confirmation of the findings. However, clinical follow-up is a valid way to evaluate diagnostic accuracy and response to therapy, and it would have been unethical to investigate all PET/CT-detected lesions using invasive procedures. Positive findings are easy to confirm, but negative findings only mean that it is not possible to acquire positive findings during the follow-up period, making it uncertain whether the findings are truly negative. Therefore, sensitivity in this series may have been overestimated. Second, the study population was relatively small. Therefore, further studies are required to validate the present results in a larger population. Third, we did not strictly compare PET/CT with contrast agent and PET/CT without contrast agent, but compared PET plus low-dose CT without contrast agent and PET plus full-dose CT with contrast agent. It would have been more appropriate to compare PET plus full-dose CT without contrast agent and PET plus full-dose CT with contrast agent in order to investigate the clinical value of intravenous contrast agent. Fourth, no oral contrast materials were used in this series. Adding an oral contrast agent would probably have helped to better delineate normal bowel activity and demonstrate pathological intraabdominal activity (peritoneal implantation). Fifth, the follow-up period (minimum 6 months) was relatively short which may have been one reason for the relatively low percentage recurrence (30%) in our series. Furthermore, some true-negative findings could have been found to be false-negative if the follow-up period had been longer.
In conclusion, integrated PET/ceCT is an accurate imaging modality for the assessment of uterine cancer recurrence. Its use reduces the frequency of equivocal interpretations.
References
Larson DM, Copeland LJ, Stringer CA, Gershenson DM, Malone JM Jr, Edwards CL. Recurrent cervical carcinoma after radical hysterectomy. Gynecol Oncol 1988;30:381–7.
Irvin WP, Rice LW, Berkowitz RS. Advances in the management of endometrial adenocarcinoma. A review. J Reprod Med 2002;47:173–90.
Beyer T, Townsend DW, Brun T, Kinahan PE, Charron M, Roddy R, et al. A combined PET/CT scanner for clinical oncology. J Nucl Med 2000;41:1369–79.
Chung HH, Jo H, Kang WJ, Kim JW, Park NH, Song YS, et al. Clinical impact of integrated PET/CT on the management of suspected cervical cancer recurrence. Gynecol Oncol 2007;104:529–34.
Sironi S, Picchio M, Landoni C, Galimberti S, Signorelli M, Bettinardi V, et al. Post-therapy surveillance of patients with uterine cancers: value of integrated FDG PET/CT in the detection of recurrence. Eur J Nucl Med Mol Imaging 2007;34:472–9. doi:10.1007/s00259-006-0251-y.
Kitajima K, Murakami K, Yamasaki E, Hagiwara S, Fukasawa I, Inaba N, et al. Performance of FDG-PET/CT in the diagnosis of recurrent endometrial cancer. Ann Nucl Med 2008;22:103–9. doi:10.1007/s12149-007-0087-y.
Park JY, Kim EN, Kim DY, Suh DS, Kim JH, Kim YM, et al. Clinical impact of positron emission tomography or positron emission tomography/computed tomography in the posttherapy surveillance of endometrial carcinoma: evaluation of 88 patients. Int J Gynecol Cancer 2008;18:1332–8.
Chung HH, Kang WJ, Kim JW, Park NH, Song YS, Chung JK, et al. The clinical impact of [18F]FDG PET/CT for the management of recurrent endometrial cancer: correlation with clinical and histological findings. Eur J Nucl Med Mol Imaging 2008;35:1081–8. doi:10.1007/s00259-007-0687-8.
Kitajima K, Murakami K, Yamasaki E, Domeki Y, Kaji Y, Sugimura K. Performance of FDG-PET/CT for diagnosis of recurrent uterine cervical cancer. Eur Radiol 2008;18:2040–7. doi:10.1007/s00330-008-0979-9.
Kitajima K, Murakami K, Yamasaki E, Domeki Y, Kaji Y, Morita S, et al. Performance of integrated FDG-PET/contrast-enhanced CT in the diagnosis of recurrent uterine cancer: comparison with PET and enhanced CT. Eur J Nucl Med Mol Imaging 2009;36:362–72. doi:10.1007/s00259-008-0956-1.
Mittra E, El-Maghraby T, Rodriguez CA, Quon A, McDougall IR, Gambhir SS, et al. Efficacy of 18F-FDG PET/CT in the evaluation of patients with recurrent cervical carcinoma. Eur J Nucl Med Mol Imaging 2009;36:1952–9. doi:10.1007/s00259-009-1206-x.
Kitajima K, Murakami K, Yamasaki E, Kaji Y, Fukasawa I, Inaba N, et al. Diagnostic accuracy of integrated FDG-PET/contrast-enhanced CT in staging ovarian cancer: comparison with enhanced CT. Eur J Nucl Med Mol Imaging 2008;35:1912–20. doi:10.1007/s00259-008-0890-2.
Cohade C, Wahl RL. Applications of positron emission tomography/computed tomography image fusion in clinical positron emission tomography: clinical use, interpretation methods, diagnostic improvements. Semin Nucl Med 2003;33:228–37.
Coleman RE, Delbeke D, Guiberteau MJ, Conti PS, Royal HD, Weinreb JC, et al. Concurrent PET/CT with an integrated imaging system: intersociety dialogue from the joint working group of the American College of Radiology, the Society of Nuclear Medicine, and the Society of Computed Body Tomography and Magnetic Resonance. J Nucl Med 2005;46:1225–39.
Schaefer NG, Hany TF, Taverna C, Taverna C, Seifert B, Stumpe KD, et al. Non-Hodgkin lymphoma and Hodgkin disease: coregistered FDG-PET and CT at staging and restaging – Do we need contrast-enhanced CT? Radiology. 2004;232:823–9. doi:10.1148/radiol.2323030985.
Antoch G, Freudenberg LS, Beyer T, Bockisch A, Debatin JF. To enhance or not to enhance? 18F-FDG and CT contrast agents in dual-modality 18F-FDG PET/CT. J Nucl Med 2004;45:56–65.
Pfannenberg AC, Aschoff P, Brechtel K, Muller M, Klein M, Bares R, et al. Value of contrast-enhanced multiphase CT in combined PET/CT protocols for oncological imaging. Br J Radiol 2007;80:437–45. doi:10.1259/bjr/34082277.
Rodriguez-Vigil B, Gomez-Leon N, Pinilla I, Hernandez-Maraver D, Coya J, Martin-Curto L, et al. PET/CT in lymphoma: Prospective study of enhanced full-dose PET/CT versus unenhanced low-dose PET/CT. J Nucl Med. 2006;47:1643–8.
Pfannenberg AC, Aschoff P, Brechtel K, Muller M, Bares R, Paulsen F, et al. Low dose non-enhanced CT versus standard dose contrast-enhanced CT in combined PET/CT protocols for staging and therapy planning in non-small cell lung cancer. Eur J Nucl Med Mol Imaging 2007;34:36–44. doi:10.1007/s00259-006-0186-3.
Tateishi U, Maeda T, Morimoto T, Miyake M, Arai Y, Kim EE. Non-enhanced CT versus contrast-enhanced CT in integrated PET/CT studies for nodal staging of rectal cancer. Eur J Nucl Med Mol Imaging 2007;34:1627–34.
Morimoto T, Tateishi U, Maeda T, Arai Y, Nakajima Y, Kim EE. Nodal status of malignant lymphoma in pelvic and retroperitoneal lymphatic pathways: comparison of integrated PET/CT with or without contrast enhancement. Eur J Radiol 2008;67:508–13.
Strobel K, Heinrich S, Bhure U, Soyka J, Veit-Haibach P, Pestalozzi BC, et al. Contrast-enhanced 18F-FDG PET/CT: 1-stop-shop imaging for assessing the respectability of pancreatic cancer. J Nucl Med 2008;49:1408–13.
Kitajima K, Murakami K, Yamasaki E, Domeki Y, Kaji Y, Fukasawa I, et al. Performance of integrated FDG-PET/contrast-enhanced CT in the diagnosis of recurrent ovarian cancer: comparison with integrated FDG-PET/non-contrast-enhanced CT and enhanced CT. Eur J Nucl Med Mol Imaging 2008;35:1439–48. doi:10.1007/s00259-008-0776-3.
Soyka JD, Veit-Haibach P, Strobel K, Breitenstein S, Tschopp A, Mende KA, et al. Staging pathways in recurrent colorectal carcinoma: is contrast-enhanced 18F-FDG PET/CT the diagnostic tool of choice? J Nucl Med 2008;49:354–61.
Kitajima K, Murakami K, Yamasaki E, Domeki Y, Kaji Y, Suganuma M, et al. Performance of integrated FDG-PET/contrast-enhanced CT in the diagnosis of recurrent colorectal cancer: comparison with integrated FDG-PET/non-contrast-enhanced CT and enhanced CT. Eur J Nucl Med Mol Imaging 2009;36:1388–96. doi:10.1007/s00259-009-1081-5.
Mawlawi O, Erasmus JJ, Munden RF, Pan T, Knight A, Macapinlac HA, et al. Quantifying the effect of IV contrast media on integrated PET/CT: clinical evaluation. AJR Am J Roentgenol 2006;186:308–19. doi:10.2214/AJR.04.1740.
Acknowledgments
We thank Hiroyoshi Okajima, Keita Miyamoto, Eiji Takeda, and Kazuhiro Kubo for their excellent technical assistance and generous support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kitajima, K., Suzuki, K., Nakamoto, Y. et al. Low-dose non-enhanced CT versus full-dose contrast-enhanced CT in integrated PET/CT studies for the diagnosis of uterine cancer recurrence. Eur J Nucl Med Mol Imaging 37, 1490–1498 (2010). https://doi.org/10.1007/s00259-010-1440-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-010-1440-2