Abstract
Purpose
The purpose of this study was to evaluate the accuracy of integrated positron emission tomography (PET) and computed tomography (CT) for the identification of suspected recurrent endometrial cancer after treatment.
Methods
Thirty-one women (median age, 53 years) with endometrial cancer treated by primary staging laparotomy who had [18F]fluorodeoxyglucose (FDG) PET/CT performed for suspected recurrence were retrospectively reviewed. The findings of the PET/CT scans were compared, with the histological examination after a surgical biopsy in 20 cases and with clinical follow-up in 11 cases to determine the diagnostic accuracy of PET/CT.
Results
Twelve (38.7%) of the 31 patients had a documented recurrence by surgical biopsy or clinical follow-up, and 19 (61.3%) had no evidence of recurrence. Of the 12 patients with recurrent disease, nine (75.0%) women were confirmed to have a recurrence by surgical biopsy. A close correlation was found between the PET/CT and histological or clinical analyses (κ = 0.933, p < 0.001). The overall sensitivity, specificity, accuracy, positive predictive value, negative predictive value, and accuracy of PET/CT were 100, 94.7, 92.3, 100, and 96.8%, respectively. The PET/CT results modified the diagnostic or treatment plan in seven (22.6%) patients, resulting in five (16.1%) patients undergoing previously unplanned therapeutic procedures and eliminating previously planned diagnostic procedures in two (6.5%) patients. Patients with negative PET/CT scans showed significantly better progression-free survival than those with positive scans (p = 0.015).
Conclusion
Integrated PET/CT appears to be highly sensitive, specific, and accurate as a post-therapy surveillance modality for endometrial cancer in well-selected patients. The PET/CT might be used to improve patient surveillance and prognosis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Guidelines for the optimal post-therapy surveillance of patients treated for endometrial cancer have not yet been introduced [1]. Conventional surveillance consists of a physical examination, evaluation of serum tumor markers, and imaging studies. However, these methods are not very accurate and do not exactly identify sites of recurrence [2]. Diagnosis of recurrent cancer by computed tomography (CT) or magnetic resonance imaging (MRI) is based on the detection of new lesions or changes in the size of a known lesion caused by cancer growth [3, 4]. Despite the widespread use of these conventional imaging modalities, accuracy for the detection of tumor recurrence has been low [5–7]. Positron emission tomography (PET), with fluorodeoxyglucose (FDG), has been widely used for monitoring cancer patients; it has been demonstrated to have high sensitivity but average specificity, with a limited ability to provide information on the exact location of an identified lesion [8–15].
Integrated PET/CT offers the combined benefits of anatomic and functional imaging; it has been used to localize areas of increased FDG uptake with improved anatomic specificity [12, 16–19]. The initial oncology results of this combined anatomic and functional technology have been promising [20–24]. To date, however, its value in the post-therapy surveillance of endometrial cancers has not been established. Thus, the purpose of this retrospective study was to evaluate the accuracy of integrated PET/CT for the identification of suspected recurrent endometrial carcinoma after treatment, with the use of histological analysis and clinical follow-up for comparison.
Materials and methods
Patients
For this retrospective study, data from well-selected patients with endometrial cancer from November 2003 to June 2007 were evaluated. Forty-three consecutive patients with endometrial cancer, who underwent primary cytoreductive surgery followed by adjuvant treatment if necessary, were included in the study. The adjuvant treatments after surgery consisted of platinum-based combination chemotherapy (n = 6), radiation therapy (n = 2), or concurrent chemoradiation (n = 18). We performed a PET/CT scan when patients had the following findings: (1) Symptoms that were suspicious of a recurrence; (2) new lesions identified on surveillance imaging studies; (3) elevated serum tumor markers with or without abnormal imaging studies; (4) abnormal results on physical or cytological examination on routine surveillance; and (5) patient request of a surveillance PET/CT scan for fear of recurrence without evidence of disease. The patient exclusion criteria were contraindications to PET scanning, such as a blood glucose level higher than 140 mg/dl, a history of diabetes, and claustrophobia. Before being enrolled, all patients provided informed consent in accordance with the regulations of the institutional review board that approved our study.
PET/CT technique
The patients were studied using a dedicated PET/CT system (Philips, Gemini). All patients were requested to fast for 4 h before undergoing PET/CT. The blood sugar levels were checked to ensure that there was no hyperglycemia; this is important because FDG uptake in cancer cells is reduced if there are competing unlabeled glucose molecules. Approximately 900 ml of a barium sulfate solution [Readi-cat (1.3% weight-volume barium sulfate suspension); E-Z-EM, Westbury, NY, USA] was administered orally 1 h before imaging to opacify the bowel for the CT portion of the study. In addition, 15–20 mCi (555–740 MBq; 0.22 mCi/kg body weight) of FDG was administered intravenously 1 h before imaging. Patients sat quietly in a dimly lit room during the uptake phase and were asked to void just before the imaging. During the scanning, the patients were supine with their arms raised above the head in the fused PET/CT scanner with a single gantry and table. The CT and PET scans were obtained with the patient in quiet respiration.
The CT was performed before the PET, and the resulting data were used to generate an attenuation correction map for the PET. Five-millimeter-thick sections were obtained at 80 mA (but adjusted for body thickness) and 140 kVp from the skull base to the mid-thigh. The images were reconstructed with a 512 × 512 matrix and a 50-cm field of view. For fusion with the PET data, the images were also reconstructed with a 128 × 128 matrix.
Next, the PET was performed on a dedicated PET scanner with a 5-min emission acquisition per imaging level. The images were acquired in a caudo-cranial direction from the mid-thigh to the skull base. The CT transmission map was used for attenuation correction, and the PET images were reconstructed with a 128 × 128 matrix, an ordered subset expectation maximum iterative reconstruction algorithm (two iterations, 28 subsets), an 8-mm Gaussian filter, and a 50-cm field of view.
Image analysis
All studies were interpreted retrospectively and were reviewed with knowledge of the patient’s clinical history. This included the results of previous imaging studies performed from the time of the initial diagnosis to the time when suspicion of a recurrence arose. Two experienced nuclear medicine physicians who were aware of each patient’s clinical history initially interpreted the findings together.
The PET study was read as negative when no areas of abnormal FDG uptake were seen. Every focus of increased FDG uptake was recorded and classified as malignant, equivocal, or benign based on the shape, size, and intensity of the uptake. A site of increased FDG uptake was defined as benign, and unrelated to cancer, when it was located in an area of known physiologic uptake of the tracer or in a known nonmalignant process. When a focal FDG uptake, with intensity higher than that of the surrounding tissues, was seen in areas unrelated to physiological or benign processes, it was defined as malignant. Any other area of increased FDG uptake that could not be clearly characterized was defined as equivocal. A PET study showing at least one site of abnormal FDG uptake characterized as malignant was defined as positive. PET studies with all lesions defined as equivocal or benign were interpreted as equivocal or negative.
Recurrent tumor was diagnosed when the abnormal focal uptake, observed on the PET images, corresponded to an abnormal soft-tissue mass in the pelvis and peritoneum observed on the CT images. Calculation of maximal standardized uptake value (SUVmax) was as followed: SUVmax = C max × TBW/IA [C max: activity concentration in the voxel of highest tumor activity (Bq/ml), TBW: total body weight (kg), IA: injected activity (kBq)]. According to previous reports [18, 25], SUVmax greater than 3.0 was considered to indicate a malignancy.
Clinical evaluation
Recurrent disease was confirmed in all cases with either tissue biopsy or the demonstration of progressive disease by serial imaging studies such as CT, MRI, or PET/CT. Clinical proof of no recurrent disease consisted of a negative physical and gynecological examination, a negative tissue biopsy, and negative findings on serial follow-up imaging studies.
To confirm the lesion detected on PET/CT imaging, surgical biopsy and CT- or ultrasound-guided biopsy was performed to obtain tissue for histological evaluation in cases where it was considered necessary for patient management. Progressive elevation of serum tumor markers (CA-125) accompanied by an increase in lesion size or the appearance of new lesions was considered a disease recurrence.
Statistical analysis
Calculation of the sensitivity, specificity, accuracy, and positive and negative predictive values for tumor detection by PET/CT was performed. These findings were then compared to the results of the histological analysis after biopsy or clinical follow-up. For the purposes of statistical analysis, a true-positive lesion was a lesion seen on the PET/CT images and found to be positive for tumor tissue by histological analysis or clinical follow-up. A false-positive lesion was a lesion seen on the PET/CT images but found to be negative for tumor tissue by histological analysis or clinical follow-up. A true-negative lesion was when no lesion was seen on the PET/CT images, and the results of the histological analysis were negative for tumor tissue or clinical follow-up after 2 months. A false-negative lesion was a lesion that was missed on image analysis but was found to be positive for neoplastic tissue by histological analysis or clinical follow-up. Sensitivity, specificity, accuracy, and positive and negative predictive values of the PET/CT were also calculated on the basis of per-patient analysis. The κ statistics (Cohen κ) were used as measures of agreement between the PET/CT and the histological findings. The κ values up to 0.40 were considered to indicate poor agreement; values between 0.41 and 0.75, moderate to good agreement; and values greater than 0.75, excellent agreement [26]. The analysis was performed using SPSS software (Version 11.0; SPSS, Chicago, IL, USA). A p value of less than 0.05 was considered statistically significant.
Survival curves for progression-free survival (PFS) were plotted according to the method of Kaplan–Meier using SPSS software (Version 11.0; SPSS). Patients were grouped into two groups, negative PET/CT scan and positive PET/CT scan, and the time intervals between the PET/CT and the last follow-up or time of recurrence were calculated to assess the PFS.
Results
Patient characteristics
Thirty-one patients met the inclusion criteria and underwent PET/CT imaging for suspected recurrent endometrial cancer during the study period. Table 1 summarizes the clinical characteristics of the enrolled patients. The median patient age was 53 (range, 28–74) years, and the median time from treatment to PET/CT scanning was 24 (range, 1–87) months. At the initial diagnosis, most (21/31) of the tumors were endometrioid adenocarcinoma (n = 27). Overall, 12 of 31 patients (38.7%) had pathologically or clinically documented recurrent disease during the follow-up (Fig. 1). Of the 12 patients with recurrent disease, nine (75.0%) women were confirmed to have a recurrence by surgical biopsy, and three (25.0%) women were documented via clinical follow-up or imaging modalities (Table 2).
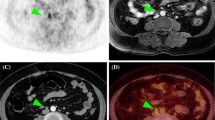
Detection and precise localization of disease recurrence in treated endometrial cancer. A 50-year-old woman (FIGO stage IIB, grade 1 endometrial cancer) 5 months after staging laparotomy was referred for a PET/CT for suspected recurrence. a An unenhanced CT transaxial image shows enlarged lymph node in the aortocaval area. b FDG-PET transaxial image shows an area of intense FDG uptake in the lesion. c Integrated PET/CT transaxial image shows that the abnormal FDG uptake corresponds to the enlarged lymph node on the CT image. Histopathological examination confirmed the presence of tumor involvement of the lymph node
Table 3 summarizes the indications for PET/CT scans in patients with suspected recurrent endometrial cancer, the number of positive PET/CT studies, and confirmed recurrences. More than half (54.8%) of the subjects had a PET/CT performed for evaluation of treatment response. In addition, 32.3% of patients had a PET/CT performed due to abnormal imaging studies during follow-up. There were 13 cases with positive PET/CT scans and no case where a recurrence was not detected by the PET/CT scan.
PET/CT diagnostic accuracy
The κ value in the current study was 0.933 (p < 0.001). This result suggests that although there were relatively few patients, the PET/CT results had excellent correlation with the histological and clinical findings. The overall patient-based sensitivity, specificity, positive and negative predictive values, and accuracy of the PET/CT for the detection of recurrent endometrial cancer among suspected patients were 100% (12 of 12 patients), 94.7% (18 of 19 patients), 92.3% (12 of 13 patients), 100% (18 of 18 patients), and 96.8% (30 of 31 patients), respectively (Table 4).
Clinical impact of PET/CT on patient management
The clinical impact of PET/CT scanning in the management of suspected endometrial cancer recurrence is summarized in Table 5. The PET/CT scan results modified the diagnosis or treatment plan in seven (22.6%) patients, mainly by introducing previously unplanned therapeutic procedures in five (16.1%) patients and eliminating previously planned diagnostic procedures in two (6.5%) patients (Fig. 2). Among five patients who had their treatment plans changed based on the PET/CT findings, three patients received chemotherapy, 2 twopatients received concurrent chemoradiation, and two patients are alive without evidence of disease. Of the 17 scans performed for surveillance of the treatment response, one scan identified a recurrence not detected by other modalities. The PET/CT results were also helpful in detecting recurrent disease in patients with unexplained elevated serum CA-125 levels during follow-up.
Inflammatory uptake of FDG in treated endometrial cancer. A 52-year-old woman (FIGO stage IIIA, grade 1 endometrial cancer) 59 months after staging laparotomy referred for PET/CT for suspected recurrence. a An unenhanced CT transaxial image shows soft tissue mass right to the rectum. b FDG-PET transaxial image shows an area of FDG uptake; however, due to the proximity to the bladder, it is hard to discriminate the exact lesion. c Integrated PET/CT transaxial image shows a faint FDG uptake within the soft tissue mass with an SUV of 1.7, which was rather suggestive of inflammatory changes. Clinical follow-up revealed that the FDG uptake was due to the inflammatory reaction after concurrent chemoradiation
The median follow-up was 6 (range, 2–31) months from the time of the PET/CT until the last follow-up or the time of recurrence if present. As shown in Fig. 3, the 2-year PFS rate of patients with a negative PET/CT scan for recurrence was significantly better than for patients with a positive PET/CT (100 vs 33.7%, p = 0.015). All 18 patients with negative PET/CT scans are still alive without evidence of disease; however, 10 out of 13 patients with positive PET/CT scans are alive with disease.
Discussion
The purpose of this study was to evaluate the accuracy of integrated PET/CT for the identification of suspected recurrent endometrial carcinoma after treatment. Metabolic imaging using FDG-PET is now widely applied to the evaluation of cervical and ovarian carcinoma [6, 11, 12, 14, 15, 19, 27–33]. However, limited information is available regarding the clinical utility of FDG-PET for endometrial cancer, especially the indications for post-therapy surveillance [13, 34–36]. The diagnostic efficacy and benefit of FDG-PET in other clinical settings for the evaluation of endometrial cancer remain uncertain [37, 38]. Substantial variations in endometrial uptake have been reported in patients with benign endometrial pathology, in patients with normal menstrual cycles, and in postmenopausal patients [39]. These variations may explain the limited use of FDG-PET for endometrial cancer.
The current study was undertaken to evaluate the clinical feasibility and performance of integrated PET/CT for the detection of recurrent endometrial cancer in suspicious patients with otherwise clinically occult disease based on biochemical parameters or the results of other imaging modalities. Belhocine et al. [13] reported that FDG-PET had a sensitivity of 96% and a specificity of 78% for post-therapy surveillance and led to the alteration of treatment in 35% of cases. Saga et al. [35] reported that FDG-PET had a sensitivity of 100% and a specificity of 88.2% with the treatment plan affected in 33.3% of cases and demonstrated that negative PET results tended to be associated with a better disease-free course. Recently, Rebollo-Aguirre et al. [36] reported that PET modified the information obtained by conventional imaging techniques in more than half of the cases studied. In the current study, the PET/CT showed similar or better diagnostic performance than previous studies and a similar clinical impact on patient management. As most of the recurrent cases were confirmed by surgical biopsy, this provided additional support to the accuracy of PET/CT for detecting recurrent lesions. The positive predictive value of PET/CT in detecting disease recurrence was 92.3%; this value may be due to the small number of cases in the study. The PET/CT identified 13 abnormal scans, and recurrences were documented in 12 cases. A PET/CT scan was performed in a patient (age 46, endometrioid adenocarcinoma stage IB, grade 2) due to an unexplained elevated serum CA-125 level 28 months after surgery and concurrent chemoradiation. The PET/CT scan showed a hypermetabolic lesion in a left axillary lymph node with a SUVmax of 3.1; however, the lesion proved to be negative for malignancy after surgical biopsy (lymphoid follicular hyperplasia with no tumor involvement), and serum CA-125 level was normalized.
The results of this study demonstrate the potential value of PET/CT in patients with suspected recurrent endometrial cancer. Furthermore, the results of this study suggest a potential role for integrated PET/CT as a useful screening approach in the post-therapy surveillance of endometrial cancer. As shown in the Fig. 3, the 2-year PFS rate, of patients with a negative PET/CT scan for recurrence, was significantly better than for patients with a positive PET/CT (100 vs 33.7%, p = 0.015). Although we could not determine the overall survival rate because of the small number of patients and the short period of follow-up, this can be evaluated in future studies. We are planning a prospective study to determine the feasibility of using PET/CT for the detection of recurrence in patients with endometrial cancer and the potential role of using this technology for cancer surveillance.
From the clinician’s perspective, the PET/CT had a promising impact on patient care. The diagnosis or treatment plan was modified in 22.6% of patients mainly by the introduction of unplanned therapeutic procedures such as chemotherapy or concurrent chemoradiation in five (16.1%) patients and elimination of previously planned diagnostic procedures in some cases (6.5%). The value of an additional diagnostic tool should be judged based on whether the information obtained improves clinical decisions, although the definition of clinical benefit varies in different clinical situations. In the current study, the treatment plan was positively influenced in five (16.1%) patients. The PET/CT results decreased equivocal PET reports in two cases, which might have led to further imaging and biopsy procedures and associated additional cost and emotional stress.
The study had the following limitations. First, this was a retrospective study. The PET/CT was not performed in every case of endometrial cancer after primary treatment during the study period. The application of PET/CT to only selected cases might introduce bias and influence the study results. Second, the number of patients with suspected endometrial cancer recurrence in our series was small. Third, the median follow-up duration after PET/CT was relatively short. More long-term data are needed to confirm the importance of PET/CT in post-therapy surveillance of endometrial cancer.
In conclusion, this retrospective study demonstrated that integrated [18F]FDG PET/CT was a highly sensitive, specific, and accurate post-therapy surveillance modality for the detection of recurrent endometrial cancer in well-selected cases. The integrated PET/CT scan was a useful tool for the detection of subtle recurrences. In addition, the PET/CT allowed individualization of the optimal treatment plan by accurately identifying patients with disease recurrence and avoiding unnecessary diagnostic procedures in patients without recurrence. Well-designed prospective studies are necessary to confirm whether the use of integrated PET/CT scanning might improve overall survival of patients with recurrent endometrial cancer by earlier detection of recurrent disease.
References
Rose PG. Endometrial carcinoma. N Engl J Med 1996;335:640–9.
Kurihara T, Mizunuma H, Obara M, Andoh K, Ibuki Y, Nishimura T. Determination of a normal level of serum CA125 in postmenopausal women as a tool for preoperative evaluation and postoperative surveillance of endometrial carcinoma. Gynecol Oncol 1998;69:192–6.
Hida J, Yasutomi M, Shindoh K, Kitaoka M, Fujimoto K, Ieda S, et al. Second-look operation for recurrent colorectal cancer based on carcinoembryonic antigen and imaging techniques. Dis Colon Rectum 1996;39:74–9.
Torizuka T, Nobezawa S, Kanno T, Futatsubashi M, Yoshikawa E, Okada H, et al. Ovarian cancer recurrence: role of whole-body positron emission tomography using 2-[fluorine-18]-fluoro-2-deoxy-d-glucose. Eur J Nucl Med Mol Imaging 2002;29:797–803.
Ebner F, Kressel HY, Mintz MC, Carlson JA, Cohen EK, Schiebler M, et al. Tumor recurrence versus fibrosis in the female pelvis: differentiation with MR imaging at 1.5 T. Radiology 1988;166:333–40.
Kinkel K, Ariche M, Tardivon AA, Spatz A, Castaigne D, Lhomme C, et al. Differentiation between recurrent tumor and benign conditions after treatment of gynecologic pelvic carcinoma: value of dynamic contrast-enhanced subtraction MR imaging. Radiology 1997;204:55–63.
Connor JP, Andrews JI, Anderson B, Buller RE. Computed tomography in endometrial carcinoma. Obstet Gynecol 2000;95:692–6.
Rohren EM, Turkington TG, Coleman RE. Clinical applications of PET in oncology. Radiology 2004;231:305–32.
Anzai Y, Carroll WR, Quint DJ, Bradford CR, Minoshima S, Wolf GT, et al. Recurrence of head and neck cancer after surgery or irradiation: prospective comparison of 2-deoxy-2-[F-18]fluoro-d-glucose PET and MR imaging diagnoses. Radiology 1996;200:135–41.
Schelling M, Avril N, Nahrig J, Kuhn W, Romer W, Sattler D, et al. Positron emission tomography using [(18)F]Fluorodeoxyglucose for monitoring primary chemotherapy in breast cancer. J Clin Oncol 2000;18:1689–95.
Chung HH, Kim SK, Kim TH, Lee S, Kang KW, Kim JY, et al. Clinical impact of FDG-PET imaging in post-therapy surveillance of uterine cervical cancer: from diagnosis to prognosis. Gynecol Oncol 2006;103:165–70.
Chung HH, Jo H, Kang WJ, Kim JW, Park NH, Song YS, et al. Clinical impact of integrated PET/CT on the management of suspected cervical cancer recurrence. Gynecol Oncol 2007;104:529–34.
Belhocine T, De Barsy C, Hustinx R, Willems-Foidart J. Usefulness of (18)F-FDG PET in the post-therapy surveillance of endometrial carcinoma. Eur J Nucl Med Mol Imaging 2002;29:1132–9.
Ryu SY, Kim MH, Choi SC, Choi CW, Lee KH. Detection of early recurrence with 18F-FDG PET in patients with cervical cancer. J Nucl Med 2003;44:347–52.
Grigsby PW, Siegel BA, Dehdashti F, Rader J, Zoberi I. Posttherapy [18F] fluorodeoxyglucose positron emission tomography in carcinoma of the cervix: response and outcome. J Clin Oncol 2004;22:2167–71.
Charron M, Beyer T, Bohnen NN, Kinahan PE, Dachille M, Jerin J, et al. Image analysis in patients with cancer studied with a combined PET and CT scanner. Clin Nucl Med 2000;25:905–10.
Kluetz PG, Meltzer CC, Villemagne VL, Kinahan PE, Chander S, Martinelli MA, et al. Combined PET/CT Imaging in Oncology. Impact on Patient Management. Clin Positron Imaging 2000;3:223–30.
Makhija S, Howden N, Edwards R, Kelley J, Townsend DW, Meltzer CC. Positron emission tomography/computed tomography imaging for the detection of recurrent ovarian and fallopian tube carcinoma: a retrospective review. Gynecol Oncol 2002;85:53–8.
Chung HH, Kang WJ, Kim JW, Park NH, Song YS, Chung JK, et al. Role of [18F]FDG PET/CT in the assessment of suspected recurrent ovarian cancer: correlation with clinical or histological findings. Eur J Nucl Med Mol Imaging 2007;34:480–6.
Antoch G, Saoudi N, Kuehl H, Dahmen G, Mueller SP, Beyer T, et al. Accuracy of whole-body dual-modality fluorine-18-2-fluoro-2-deoxy-d-glucose positron emission tomography and computed tomography (FDG-PET/CT) for tumor staging in solid tumors: comparison with CT and PET. J Clin Oncol 2004;22:4357–68.
Metser U, Golan O, Levine CD, Even-Sapir E. Tumor lesion detection: when is integrated positron emission tomography/computed tomography more accurate than side-by-side interpretation of positron emission tomography and computed tomography. J Comput Assist Tomogr 2005;29:554–9.
Reinartz P, Wieres FJ, Schneider W, Schur A, Buell U. Side-by-side reading of PET and CT scans in oncology: which patients might profit from integrated PET/CT. Eur J Nucl Med Mol Imaging 2004;31:1456–61.
De Leyn P, Stroobants S, De Wever W, Lerut T, Coosemans W, Decker G, et al. Prospective comparative study of integrated positron emission tomography-computed tomography scan compared with remediastinoscopy in the assessment of residual mediastinal lymph node disease after induction chemotherapy for mediastinoscopy-proven stage IIIA-N2 Non-small-cell lung cancer: a Leuven Lung Cancer Group Study. J Clin Oncol 2006;24:3333–9.
Pelosi E, Messa C, Sironi S, Picchio M, Landoni C, Bettinardi V, et al. Value of integrated PET/CT for lesion localisation in cancer patients: a comparative study. Eur J Nucl Med Mol Imaging 2004;31:932–9.
Hubner KF, McDonald TW, Niethammer JG, Smith GT, Gould HR, Buonocore E. Assessment of primary and metastatic ovarian cancer by positron emission tomography (PET) using 2-[18F]deoxyglucose (2-[18F]FDG). Gynecol Oncol 1993;51:197–204.
Fleiss J. Statistical methods for rates and proportions. 2nd ed. New York: Willey; 1981.
Sugawara Y, Eisbruch A, Kosuda S, Recker BE, Kison PV, Wahl RL. Evaluation of FDG PET in patients with cervical cancer. J Nucl Med 1999;40:1125–31.
Grigsby PW, Siegel BA, Dehdashti F, Mutch DG. Posttherapy surveillance monitoring of cervical cancer by FDG-PET. Int J Radiat Oncol Biol Phys 2003;55:907–13.
Belhocine TZ. 18F-FDG PET imaging in posttherapy monitoring of cervical cancers: from diagnosis to prognosis. J Nucl Med 2004;45:1602–4.
Sironi S, Messa C, Mangili G, Zangheri B, Aletti G, Garavaglia E, et al. Integrated FDG PET/CT in patients with persistent ovarian cancer: correlation with histologic findings. Radiology 2004;233:433–40.
Bristow RE, del Carmen MG, Pannu HK, Cohade C, Zahurak ML, Fishman EK, et al. Clinically occult recurrent ovarian cancer: patient selection for secondary cytoreductive surgery using combined PET/CT. Gynecol Oncol 2003;90:519–28.
Yen TC, Ng KK, Ma SY, Chou HH, Tsai CS, Hsueh S, et al. Value of dual-phase 2-fluoro-2-deoxy-d-glucose positron emission tomography in cervical cancer. J Clin Oncol 2003;21:3651–8.
Lai CH, Huang KG, See LC, Yen TC, Tsai CS, Chang TC, et al. Restaging of recurrent cervical carcinoma with dual-phase [18F]fluoro-2-deoxy-d-glucose positron emission tomography. Cancer 2004;100:544–52.
Chao A, Chang TC, Ng KK, Hsueh S, Huang HJ, Chou HH, et al. 18F-FDG PET in the management of endometrial cancer. Eur J Nucl Med Mol Imaging 2006;33:36–44.
Saga T, Higashi T, Ishimori T, Mamede M, Nakamoto Y, Mukai T, et al. Clinical value of FDG-PET in the follow up of post-operative patients with endometrial cancer. Ann Nucl Med 2003;17:197–203.
Rebollo-Aguirre AC, Ramos-Font C, Gallego Peinado M, Bellon-Guardia ME, Cabello Garcia D, Rodriguez-Fernandez A, et al. [Positron emission tomography with fluordesoxyglucose-F18 in follow-up of endometrial cancer]. Rev Esp Med Nucl 2006;25:359–66.
Horowitz NS, Dehdashti F, Herzog TJ, Rader JS, Powell MA, Gibb RK, et al. Prospective evaluation of FDG-PET for detecting pelvic and para-aortic lymph node metastasis in uterine corpus cancer. Gynecol Oncol 2004;95:546–51.
Suzuki R, Miyagi E, Takahashi N, Sukegawa A, Suzuki A, Koike I, et al. Validity of positron emission tomography using fluoro-2-deoxyglucose for the preoperative evaluation of endometrial cancer. Int J Gynecol Cancer 2007;17:890–6.
Lerman H, Metser U, Grisaru D, Fishman A, Lievshitz G, Even-Sapir E. Normal and abnormal 18F-FDG endometrial and ovarian uptake in pre- and postmenopausal patients: assessment by PET/CT. J Nucl Med 2004;45:266–71.
Acknowledgments
This study was supported by a grant of the Korea Health 21 R&D Project, Ministry of Health &Welfare, Republic of Korea (0412-CR01-0704-0001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chung, H.H., Kang, W.J., Kim, J.W. et al. The clinical impact of [18F]FDG PET/CT for the management of recurrent endometrial cancer: correlation with clinical and histological findings. Eur J Nucl Med Mol Imaging 35, 1081–1088 (2008). https://doi.org/10.1007/s00259-007-0687-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-007-0687-8