Abstract
Data-driven models in a combination of optimization algorithms could be beneficial methods for predicting and optimizing in vitro culture processes. This study was aimed at modeling and optimizing a new embryogenesis medium for chrysanthemum. Three individual data-driven models, including multi-layer perceptron (MLP), adaptive neuro-fuzzy inference system (ANFIS), and support vector regression (SVR), were developed for callogenesis rate (CR), embryogenesis rate (ER), and somatic embryo number (SEN). Consequently, the best obtained results were used in the fusion process by a bagging method. For medium reformulation, effects of eight ionic macronutrients on CR, ER, and SEN and effects of four vitamins on SEN were evaluated using data fusion (DF)–non-dominated sorting genetic algorithm-II (NSGA-II) and DF-genetic algorithm (GA), respectively. Results showed that DF models with the highest R2 had superb performance in comparison with all other individual models. According to DF-NSGAII, the highest ER and SEN can be obtained from the medium containing 14.27 mM NH4+, 38.92 mM NO3−, 22.79 mM K+, 5.08 mM Cl−, 3.34 mM Ca2+, 1.67 mM Mg2+, 2.17 mM SO42−, and 1.44 mM H2PO4−. Based on the DF-GA model, the maximum SEN can be obtained from a medium containing 0.61 μM thiamine, 5.93 μM nicotinic acid, 0.25 μM biotin, and 0.26 μM riboflavin. The efficiency of the established-optimized medium was experimentally compared to Murashige and Skoog medium (MS) for embryogenesis of five chrysanthemum cultivars, and results indicated the efficiency of optimized medium over MS medium.
Key points
• MLP, SVR, and ANFIS were fused by a bagging method to develop a data fusion model.
• NSGA-II and GA were linked to the data fusion model for establishing and optimizing a new embryogenesis medium.
• The new culture medium (HNT) had better efficiency than MS medium.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chrysanthemum may be the world’s most widely grown ornamental species with over 20,000 cultivars in markets (da Silva and Kulus 2014). Also, chrysanthemum is broadly used in different parts of the world especially in China, Japan, and India either as an anti-inflammatory herb or a fragrant floral tea in conventional medicine due to the wide spectrum of its biomolecules and secondary metabolites (Yang et al. 2017). Antioxidant, antifungal, antibacterial, anti-inflammatory, and anti-spirochetal activities of chrysanthemum have been previously reported by several studies (Kim and Lee 2005; Lin et al. 2010; Yang et al. 2017). Moreover, chrysanthemum has been used as a model plant for color modification scientific researches because of its wide range of biomolecules involved in pigments (Noda et al. 2017; Hesami et al. 2020a). As a result, global demands for this valuable plant have been significantly increased. However, the current breeding efforts based on traditional reproduction are not able to cope with the pace of increasing market demand (Noda et al. 2017). Therefore, different new methods should be proposed based on novel biotechnological techniques such as in vitro culture tools to improve pharmacological, ornamental, and industrial traits of this plant.
In vitro propagation through somatic embryogenesis has become an efficient way of improving most economically important chrysanthemum cultivars (Naing et al. 2013). Culture stimuli, environment, and the cultural stimuli × environment interaction exert significant influence on cell differentiation during somatic embryogenesis (Guillou et al. 2018). Several studies (May and Trigiano 1991; Tanaka et al. 2000; da Silva 2003; Xu et al. 2012; Naing et al. 2013) have developed somatic embryogenesis protocol for chrysanthemum. However, those studies focused on the effects of different hormonal combinations as well as growth factors on embryogenesis. On the other hand, there is a lack of a comprehensive study on the establishment of a culture medium that can lead to high-frequency somatic embryogenesis. Such studies would be useful for further in vitro gene expression, genetic transformation, and functional gene studies involved in biomolecules synthesis pathways (Greenway et al. 2012).
Nutrient components are known as a major factor affected in vitro morphogenesis in different species or plant organs (Fisichella et al. 2000; Greenway et al. 2012). Essential nutrients can be provided by the basal salts that can be involved in the regulation of the morphology and growth of plant tissues (Samson et al. 2006). However, the formulation of the basal salts in a medium commonly is not considered in different tissue culture applications. Many studies have reported the major role of macroelements in somatic embryogenesis (Fisichella et al. 2000; Samson et al. 2006; Koleva-Gudeva et al. 2007).
Furthermore, the effects of vitamins and amino acids on embryogenesis have been reported by several studies (Al-Khayri 2001; Kintzios et al. 2001; Guillou et al. 2018). Although somatic embryogenesis studies of chrysanthemum have used MS medium as a basal medium (May and Trigiano 1991; Tanaka et al. 2000; da Silva 2003; Xu et al. 2012; Naing et al. 2013), the composition of MS medium is based on analysis of tissue ashes of tobacco (Gago et al. 2011). Since the nutrient requirements for different tissue culture systems and plant species would be varied and useful for specific applications, it is necessary to develop species-based medium formulations. However, design and modification of a medium for specific purpose need high expertise and would be time-consuming. Hildebrandt et al. (1946) indicated the need of Hildebrandt’s medium design on more than 16,000 cultures of tobacco. Murashige and Skoog (1962) spent 5 years for designing MS medium by using a broad spectrum of 43 factorial design along with 81 combinations for tobacco. To ease this problem, computer technologies such as artificial intelligence (AI) would be helpful to reduce this long and cumbersome process (Gago et al. 2011).
Somatic embryogenesis is a multi-variable procedure impacted by different factors such as plant genotype, culture medium composition, type and age of explants, and different types and concentrations of plant growth regulators (PGRs) (Hesami et al. 2017b, 2020c). Also, somatic embryogenesis consists of non-linear and non-deterministic developmental processes (Niazian et al. 2018; Hesami et al. 2020b). Therefore, conventional analytical techniques are inefficient to model nonlinearity in complex systems that exist in plant tissue culture, in particular, somatic embryogenesis. AI models have provided an appropriate approach for modeling the non-linearity and ill-defined systems in in vitro culture. Recently, different individual AI models such as neural networks (Barone 2019; Hesami et al. 2019c; Jamshidi et al. 2019; García-Pérez et al. 2020a; Salehi et al. 2020), fuzzy logic (Nezami-Alanagh et al. 2017; Hesami et al. 2019d; Farhadi et al. 2020; García-Pérez et al. 2020b), and decision trees (Khvatkov et al. 2019; Akin et al. 2020) have been successfully employed for predicting and optimizing various plant tissue culture processes. On the other hand, the necessity of increased precision and accuracy of data-driven models has encouraged researchers to develop applicable methods such as multi-model fusion-based approaches. The key idea of data fusion is fusing or combining data derived from fused information in order to provide more precise estimations in comparing with using individual model (Alizadeh and Nikoo 2018). Many researchers in several fields of study have used data fusion (Aiello et al. 2018; Alizadeh and Nikoo 2018; Hararuk et al. 2018; Wu et al. 2018). At more complex features such as medium establishment, in particular, multi-model fusion-based methods could be used to integrate the advantages and strengths of individual models. Several studies have demonstrated that data fusion models can be more reliable and accurate to model complex systems (Aiello et al. 2018; Alizadeh and Nikoo 2018; Hararuk et al. 2018; Wu et al. 2018; Hesami and Jones 2020).
This study shows high stability and accuracy of using the data fusion model in predicting and optimizing the somatic embryogenesis of chrysanthemum. However, the weakness of using AI models is that it is hard to obtain an optimized solution (Hosseini-Moghari and Araghinejad 2015; Hosseini-Moghari et al. 2015, 2017; Haddad Omid et al. 2016; Moravej and Hosseini-Moghari 2016; Bozorg-Haddad et al. 2017; Araghinejad et al. 2017, 2018; Dezfooli et al. 2018; Ebrahimian et al. 2020; Moravej et al. 2020; Sheikhi et al. 2020). Most studies selected the optimized solution by a lot of experiments (Gago et al. 2010, 2011, 2014; Alanagh et al. 2014; Hesami and Daneshvar 2018b; Hesami et al. 2018b; Niazian et al. 2018). Evaluating objective function can be useful for figuring out the performance of in vitro culture systems in optimization problems (Arab et al. 2017). However, different trials and errors are needed to optimize the plant tissue culture process. To tackle this time-consuming running (trials and errors), optimization algorithms can be considered a reliable solution. For instance, Jamshidi et al. (2019) used genetic algorithm (GA) to optimize nutrition for pear rootstock tissue culture medium formulation. On the one hand, GA as one of the best well-known optimization algorithms can only handle a single objective function (Bozorg-Haddad et al. 2016; Hesami et al. 2019b). On the other hand, sometimes, plant tissue culture processes are faced with multi-objective functions. Therefore, there is a dire need to apply multi-objective algorithms such as non-dominated sorting genetic algorithm-II (NSGA-II) for optimizing multi-objective functions (Wu et al. 2018). Recently, NSGA-II has been successfully employed to optimize different in vitro culture systems such as sterilization (Hesami et al. 2019c), somatic embryogenesis (Hesami et al. 2019d, 2020c), and shoot proliferation (Hesami et al. 2019c).
In the current study, modeling the somatic embryogenesis was conducted through data fusion approach. Afterwards, NSGAII and GA were linked to data fusion for achieving the optimum concentrations of macronutrients and vitamins, respectively, which are necessary for successful somatic embryogenesis. Thus, the main objective of this study was to determine the best model and optimize the proper levels of vitamins and macronutrients in somatic embryogenesis of chrysanthemum. According to the best of our knowledge, this study is the first report of the application of the data fusion model for predicting and optimizing processes in the field of plant tissue culture.
Material and methods
Plant materials, media, and culture conditions
The leaf segments of chrysanthemum (Dendranthema × grandiflorum) “Hornbill Dark” were selected as explants from the newly developed shoots of 9-month-old greenhouse-grown mother plants. The surface sterilization of the explants, medium preparation, and culture conditions were performed based on Hesami et al.’s (2019c) protocol.
Selecting the best combination of plant growth regulators
In the first experiment, different concentrations of 2,4-dichlorophenoxyacetic acid (2,4-D) and 6-benzylaminopurine (BAP) (0, 1, 2, and 3 mg·l−1) in MS medium were tested for choosing the best PGR combination for further experiments. All experiments were carried out based on a completely randomized design (CRD) with the factorial arrangement, and there were 15 replicates per treatment, and each treatment was repeated in three sets.
Selecting the best common medium for the development of chrysanthemum embryogenesis media
For the second experiment, five conventional media including SH (Schenk and Hildebrandt 1972), MS (Murashige and Skoog 1962), B5 (Gamborg et al. 1968), White (White 1942), and NN (Nitsch and Nitsch 1969) were used for selecting the best medium for predicting and optimizing of chrysanthemum embryogenesis medium. The concentrations of micronutrients and vitamins for all formulated media were the same as MS medium.
Developing a new culture medium
In the third experiment, the effects of the primary macronutrients on embryogenesis of chrysanthemum were evaluated by designing 32 media for optimizing a new culture medium.
Five MS macronutrients with various concentrations were studied in multifactorial combinations including 0.5× and 1× MS for NH4NO3 (825 and 1650 mg·l−1); 0.5× and 1× MS for KNO3 (950 and 1900 mg·l−1); 0.5× and 1× MS for CaCl2 (220 and 440 mg·l−1); 0.5× and 1× MS for KH2PO4 (85 and 170 mg·l−1); and 0.5× and 1× MS for MgSO4·7H2O (185 and 370 mg·l−1) (Supplemental Table S1).
According to the fourth experiment, the effects of the macroelements on chrysanthemum embryogenesis were tested by proposing 37 media to optimize a new culture medium.
Five MS macronutrients with various concentrations were studied in multifactorial combinations including 0.75× and 1.25× MS for NH4NO3 (1237.5 and 2062.5 mg·l−1); 0.75× and 1.25× MS for KNO3 (1425 and 2.375 mg·l−1); 0.75× and 1.25× MS for CaCl2 (330 and 550 mg·l−1); 0.75× and 1.25× MS for KH2PO4 (127.5 and 212.5 mg·l−1); and 0.75× and 1.25× MS for MgSO4·7H2O (277.5 and 462.5 mg·l−1). Also, five MS-modified media containing 0.5, 0.75, 1, 1.25, or 1.5× MS for NH4NO3 along with 1.5× MS for other macronutrients were tested (Supplemental Table S2).
The concentrations of micronutrients and vitamins for all formulated media were the same as MS medium.
In the fifth experiment, the effects of thiamine, nicotinic acid, riboflavin, and biotin on embryogenesis of chrysanthemum were evaluated in multifactorial combinations by designing 36 media to optimize a new culture medium (Supplemental Table S3).
The concentrations of micronutrients, myo-inositol, glycine, and pyridoxine HCl for all formulated media in the fifth experiment were the same as MS medium. Also, the levels of macronutrients for all formulated media were the same as optimized macroelements obtained in previous experiments by a data fusion–NSGAII model.
Individual data-driven models
Three individual data-driven models, namely, multi-layer perceptron (MLP), adaptive neuro-fuzzy inference system (ANFIS), and support vector regression (SVR) were proposed as estimator models for embryogenesis estimation using formulated medium data. For constructing individual models, different concentrations of macroelements (NH4+, NO3−, K+, Ca2+, Cl−, Mg2+, SO42−, and H2PO4−) were considered inputs and callogenesis rate (CR) (Eq. 1), embryogenesis rate (ER) (Eq. 2), and somatic embryo number (SEN) per explant were considered output data for the modeling of macronutrients experiments; different concentrations of vitamins (thiamine, nicotinic acid, biotin, and riboflavin) were considered inputs, and SEN was selected as output data for the modeling of vitamins.
The training and testing data of the models were set to 75% and 25% of the datasets, respectively, and checked to confirm if the range of train set consists of the test data. In this study, the MLP model was run based on feed forward back-propagation (3-layer back-propagation network) as one of the common bases of network structure. The linear (purelin) and transfer functions of the hyperbolic tangent sigmoid (tansig) were applied for output and hidden layers, respectively. Moreover, a Levenberg-Marquardt algorithm was applied in the training step of the network to determine the optimal bias and weights. Afterward, the optimal number of neurons in the hidden layer was identified by using a trial and error method. The Gaussian membership function was used (between 3 and 5 membership functions for various variables) to run the ANFIS model based on a trial and error approach. Meanwhile, the number of epochs to training the models was set to 10. In this study, the SVR–radial basis function (RBF) kernels were considered and the tube size (ε), regularized constant (c), and γ were selected based on trial and error via nested loops.
The different values for the significant model’s parameters were examined based on a trial and error analysis for determining and improving the overall performance of the best-constructed model. To assess and compare the accuracy of individual and data-fusion models, three following performance measures including R2 (coefficient of determination), root mean square error (RMSE), and mean bias error (MBE) were used:
where yt, \( \overline{y} \), \( \hat{y_t} \), and T are the tth observed data, the mean of observed values, the mean of predicted values, and total number of predicted values, respectively. Greater R2 and smaller RMSE and MBE indicated better performance of the constructed models.
Data fusion model
Data fusion is known as the process of combining and mixing data from various sources such as single outputs of several data-driven models that the overall equation can be as follows:
where \( {\overset{\wedge }{y}}_i \) stands for target variable, x is a vector of independent estimators, ε stands for corresponding estimation error, and n is a number of observation data.
In order to develop data fusion models, Eq. (6) can be introduced to the following form where several individual models are used as follows:
where m stands for the number of individual model and [\( {\overset{\wedge }{y}}_i \)] stands as matrix of estimations provided by each model.
Subsequently, the matrix of [\( {\overset{\wedge }{y}}_i \)] will be considered input data infusion models.
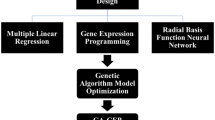
Many methods have been recommended for fusing individual models (Aiello et al. 2018; Alizadeh and Nikoo 2018; Hararuk et al. 2018; Wu et al. 2018; Hesami and Jones 2020), which reported that the most powerful and uncomplicated method among different approaches is the bagging method for data fusing (Hesami and Jones 2020). Therefore, the best-resulted outputs achieved by three individual models were fused through the bagging method (Fig. 1).
Optimization algorithms
NSGA-II
NSGA-II should mostly rely on a step-by-step procedure: crowding distance, elitist non-dominated sorting, and binary tournament selection, for choosing the best non-dominated solutions. The initialization of the chromosome/population is the start point of the computational process. The three major components for simulation process are crossover, selection, and mutation operation that would be useful for evaluating decision variables and objective functions. The next step would be categorize the solutions which are not dominated by the others as different non-dominated fronts of the population, derived based on the concept of non-dominated sorting. Also, each of the non-dominated front can be considered a level or rank data, and the population is leveled except the first Pareto front. Thus, the first rank (non-dominated front) is the last generation of the optimal Pareto. Removing the high-rank (lower priority) member and selecting others for generating parent population for the next generation are known as the NSGA-II algorithm procedure (Supplemental Fig. S1).
In optimizing macronutrient experiments, the ideal point of Pareto was selected such that ER and SEN became the maximum. Indeed, a point in the Pareto front was detected as the best optimal answer such that:
was minimal, where o and p were the highest ER and the maximum SEN, respectively.
GA
In order to achieve the highest SEN in the vitamin experiments, the trained data fusion model was processed as the fitness function by GA to identify the optimum levels of thiamine, nicotinic acid, biotin, and riboflavin.
In addition, the selection method of roulette wheel was used for selecting elite populations for crossover for obtaining the appropriate fitness. Therefore, 200 initial population, 1000 generation, 0.7 crossover rate, 0.04 mutation rate, the uniform of mutation function, 2-point crossover function, and roulette wheel as a selection function were considered. The generational practice was performed repeatedly for obtaining the generation number. The determination of the best optimal solutions was limited between the ranges of input variable during the optimization process.
Sensitivity analyses
To determine the importance of each input variable on the studied parameters, the sensitivity analysis was performed and subsequently, important variables were recognized via sensitivity error (VSE) and variable sensitivity ratio (VSR) values.
Matlab (version 9.5) software was used to write mathematical codes for assessing and constructing models and optimization and sensitivity analysis.
Validation experiment
For validation experiment, the new medium formulated by data fusion–optimization algorithms (HNT) was compared to MS medium for evaluating the efficiency of the data fusion–NSGAII and data fusion–GA for modeling and optimizing the composition of the culture medium that would be influenced by embryogenesis parameters. The media were supplemented with 2 mg·l−1 2,4-D and 2 mg·l−1 BAP. Also, five cultivars of chrysanthemum including “Malibu,” “Ferry,” “Feeling White,” “Lollipop Yellow,” and “Hornbill Dark” were used to evaluate the potential of HNT medium for embryogenesis in different cultivars of chrysanthemum.
Results
Effect of PGR combinations on somatic embryogenesis
The callus formation from the cut end of leaf disc in chrysanthemum was achieved in MS medium supplemented with different 2,4-D/BAP combinations, and the whole explant’s surface was covered with callus after two weeks. Although different 2,4-D/BAP combinations had a significant effect on embryogenesis, there was no callus initiation in the absence of PGRs. Based on Table 1, the maximum CR, ER, and SEN were obtained in MS medium supplemented with 2 mg·l−1 2,4-D along with 2 mg·l−1 BAP. The results of the present study (Table 1) indicated that the lower or higher concentrations of 2,4-D/BAP (more or less than 2 mg·l−1) resulted in decreasing the ER and SEN. Therefore, 2 mg·l−1 2,4-D along with 2 mg·l−1 BAP as the best PGR combination was chosen for later experiments.
Effect of basal media on somatic embryogenesis
According to Table 2, the highest CR, ER, and SEN were obtained from MS medium. On the contrary, White medium caused the minimum CR, ER, and SEN. As a result, MS medium was selected for developing a new culture medium.
Effect of medium composition on somatic embryogenesis
The results of macronutrient modification showed that the highest CR (100%), ER (100%), and SEN (12.11) were obtained from a modified MS medium containing 15.45 mM NH4+, 38.95 mM NO3−, 25.12 mM K+, 7.5 mM Cl−, 3.75 mM Ca2+, 1.87 mM Mg2+, 1.87 mM SO42, and 1.62 mM H2PO4− (Supplemental Table S2). However, a modified MS medium containing 20.6 mM NH4+, 30 mM NO3−, 10 mM K+, 3 mM Cl−, 1.5 mM Ca2+, 0.78 mM Mg2+, 0.75 mM SO42−, and 0.65 mM H2PO4− caused the minimum CR (15.56%), ER (8.89%), and SEN (1.35) (Supplemental Table S1).
Since CR and ER were 100% for all treatments in the vitamin modification experiment, only SEN is presented in Supplemental Table S3. The results of vitamin modification (Supplemental Table S3) indicated that the highest SEN (22.94) was obtained from the medium containing 0.6 μM thiamine, 6 μM nicotinic acid, 0.2 μM biotin, and 0.26 μM riboflavin. However, the medium containing 0.3 μM thiamine, 4.06 μM nicotinic acid, 0.2 μM biotin, and 0.13 μM riboflavin caused the minimum SEN (14.2).
Data fusion modeling and evaluation
In the third and fourth stages of the experiments, three individual models including MLP, ANFIS, and SVR were applied for modeling and predicting the three studied parameters (CR, ER, and SEN) based on eight input variables including NH4+, NO3−, K+, Ca2+, Cl−, Mg2+, SO42−, and H2PO4−. In order to improve forecasting results, the best estimations obtained by three individual models were fused through the bagging method.
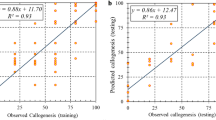
The efficiency of the individual and data fusion models was determined based on the assessment of forecasted and observed data. All of the R2 of testing and training datasets were over 78%, 80%, and 74% for MLP, ANFIS, and SVR models, respectively (Table 3). According to Table 3, the data fusion model had the better accuracy on modeling and predicting ER (R2 > 0.92), CR (R2 > 0.91), and SEN (R2 > 0.98) compared with individual models. The good fit of the data fusion model can be traced by the correlation between observed and predicted data for CR, ER, and SEN (Supplemental Fig. S2). Also, RMSE and MBE, same as R2, in data fusion models were better than individual models (Table 3). Based on the performance criteria that were mentioned in Table 3, data fusion models were able to efficiently explain the performances of somatic embryogenesis to different concentrations of macronutrients.
In the fifth set of the experiments, three individual models including MLP, ANFIS, and SVR were used for modeling the number of the somatic embryos based on four variables including thiamine, nicotinic acid, riboflavin, and biotin. The best estimation derived from three individual models was fused by the bagging method in order to improve forecasting results.
The efficiency of the individual models was determined based on the assessment of forecasted and observed data. Regarding Table 4, all R2 of testing and training datasets were over 85%, 82%, and 81% for MLP, ANFIS, and SVR models, respectively. Also, the data fusion model had a better accuracy on modeling and predicting SEN (R2 > 0.89) in comparison with individual models (Table 4). The good fit of the data fusion model can be traced by the correlation between observed and predicted data for SEN (Supplemental Fig. S3). Moreover, RMSE and MBE, same as R2, in data fusion models, were better than individual models (Table 4). SEN could be precisely predicted by using the data fusion model in the testing dataset. Moreover, balanced performance criteria were successfully obtained in both training and testing phases of data fusion models.
Sensitivity analysis of the models
In macronutrient formulation experiments, in order to identify the general VSR, the comparative rank of input data was evaluated via the entire 621 observations (testing and training). The VSR was obtained for the CR, ER, and SEN, with respect to modified macronutrients of MS medium (Table 5). Sensitivity analysis indicated that CR of chrysanthemum was more sensitive to SO42− concentration, followed by Mg2+, NH4+, NO3−, K+, H2PO4−, Ca2+, and Cl− (Table 5). In SEN and ER models, the feed efficiency possessed more sensitivity in SO42− concentration, followed by Mg2+, H2PO4−, K+, NO3−, NH4+, Ca2+, and Cl− (Table 5).
In vitamin formulation experiments, in order to identify the general VSR, the comparative rank of input data was evaluated via the entire 180 observations (testing and training). The VSR obtained for the model of SEN, with respect to modified vitamins are presented in Table 6. Sensitivity analysis showed that the NSE of chrysanthemum was more sensitive to thiamine concentration, followed by nicotinic acid, riboflavin, and biotin (Table 6).
Model optimization
The result of the optimization process for formulation of macronutrients via NSGA-II are provided in detailed in Supplemental Fig. S4 and Table 7. During the optimization process, the upper bound and the lower bound of input variables (Supplemental Table S1 and S2) were treated as constraints, and the ideal point was determined based on the point with the highest ER and SEN (Fig. S4). The optimal SEN (13.36) and ER (99.9%) can be achieved from a medium including 14.27 mM NH4+, 38.92 mM NO3−, 22.79 mM K+, 5.08 mM Cl−, 3.34 mM Ca2+, 1.67 mM Mg2+, 2.17 mM SO42−, and 1.44 mM H2PO4− (Table 7).
The aim of the vitamin formulation experiment was not only to model and predict the SEN but also to optimize vitamin levels for the maximum SEN of chrysanthemum. GA was linked to data fusion for achieving the optimum concentrations of vitamins which are necessary for successful embryogenesis. The result of the optimization process was provided in detail in Table 8. During the optimization process, the upper bound and the lower bound of input variables were determined as constraints (Supplemental Table S3). The optimal SEN (22.41) can be achieved from a medium including 0.61 μM thiamine, 5.93 μM nicotinic acid, 0.25 μM biotin, and 0.26 μM riboflavin (Table 8).
Assessment of new formulated medium (validation experiment)
High EF and NSE were achieved by using HNT medium including 2 mg/l 2,4-D and 2 mg/l BAP, which are significantly higher than those produced by using the same concentration of 2,4-D and BAP in MS medium for all cultivars (Table 9). These results showed that HNT medium could be used for somatic embryogenesis in different cultivars of chrysanthemum.
Discussion
High-frequency embryogenesis can be considered one of the most important prerequisites to obtain success in genetic engineering and genome editing (Naing et al. 2013). Medium compositions especially macronutrients and vitamins play pivotal roles in somatic embryogenesis (Guillou et al. 2018). The effects of various macroelements, as well as vitamins of culture medium on the somatic embryogenesis, were previously studied in different plants like Brassica napus L. ssp. Oleifera (Metzg.) Sink (Loh and Lim 1992), Capsicum annuum (Kintzios et al. 2001), Triticum aestivum (Greer et al. 2009), Cydonia oblonga (Fisichella et al. 2000), Manihot esculenta (Groll et al. 2002), Plantago ovata (Saha et al. 2011), and Eleusine coracana (Kothari et al. 2004). In the current study, a new culture medium, for the first time, has been established for somatic embryogenesis of chrysanthemum.
The results of this study showed the necessity of balancing auxin/cytokinin (2,4-D/BAP) for obtaining acceptable somatic embryogenesis. The highest ER was observed in 2 mg·l−1 2,4-D along with 2 mg·l−1 BAP. In accordance with our results, Naing et al. (2013) indicated that 100% embryogenesis in chrysanthemum was achieved under the combination of 2.0 mg·l−1 BAP and 2.0 mg·l−1 2,4-D. The ratio of cytokinins and auxins plays a significant role in developing events in in vitro culture (Zhao 2008; Hesami and Daneshvar 2018a; Hesami et al. 2017a, 2018a, 2018c, 2019a). Based on previous studies, several genes such as ARABIDOPSIS HISTIDINE KINASE4 (AHK4) and YUCCA (YUC) could be significantly expressed by applying external auxins during embryogenesis. Furthermore, external cytokinins can regulate auxin efflux carriers PINFORMED (PIN) (Jones et al. 2010).
The macronutrients of MS medium aroused maximized ER and SEN. This finding may be because of the higher level of nitrogen, which was at the minimum double in MS medium compared with other media. Moreover, MS medium has more balanced ratio between ammonium and nitrate. Similar results have been mentioned for three genotypes of quince, which indicated higher embryogenesis on MS macronutrients compared to media WPM, SH, NN, QL, White, B5, or DKW (Fisichella et al. 2000). By this finding, Taylor et al. (1996) reported that MS medium compared with B5, WPM, or SH had a better impact on somatic embryogenesis of cassava. Additionally, removal of NH4NO3 from MS caused the suppression of embryogenesis. Somatic embryogenesis in orchard grass was much higher on MS medium than on SH (Trigiano et al. 1992). However, the addition of ammonium into SH medium promoted embryogenesis.
In this study, a comprehensive understanding of the effect of various levels of macronutrients on chrysanthemum embryogenesis was achieved by using the data fusion–NSGAII model, which provides new insights into enhancing somatic embryogenesis of chrysanthemum. According to the best of our knowledge, this study is the first report of using the data fusion–NSGAII model for optimization and modeling macronutrient levels for chrysanthemum embryogenesis.
Data fusion that has been used by many researchers in several fields of the study was reported to be a reliable and accurate model for studying complex systems (Aiello et al. 2018; Alizadeh and Nikoo 2018; Hararuk et al. 2018; Wu et al. 2018).
The results of the sensitivity analysis showed that both EF and NSE are more sensitive to SO42− concentration, followed by Mg2+, H2PO4−, K+, NO3−, NH4+, Ca2+, and Cl−.
In plants, sulfate is subjected to a reduction-assimilation pathway that leads principally to cysteine, a vital component in sulfur assimilation (Saito 2004). Xu and Møller (2004) reported that cysteine is an important amino acid, consumed as a substrate for the synthesis of biotin and thiamine, to eschew Arabidopsis zygotic embryo lethality and deficiency in cysteine suppresses embryogenesis. Furthermore, cysteine directly provides sulfur for Fe–S groups, and the latter synthesis depends on the availability of sulfate (Smith et al. 2001; Ding et al. 2005). Al-Khayri (2001) showed that biotin and thiamine could be able to improve somatic embryogenesis in date palm (Phoenix dactylifera L.). Therefore, exogenous sulfate might be essential for the cysteine synthesis. Our results showed that increases in sulfate content caused higher embryogenesis. This finding is in parallel with Saha et al. (2011) and Minyaka et al. (2008) observations. Minyaka et al. (2008) reported that the sulfur supply of two- to threefold as compared to MS medium has increased somatic embryogenesis of cacao.
Our results showed that somatic embryogenesis of chrysanthemum was improved by increasing the magnesium (Mg) concentration of the medium. In accordance with our results, Dussert et al. (1995) reported that the somatic embryogenesis of Cocos nucifera was enhanced by increasing in the concentration of Mg in the medium. In sympathy with this finding, Walker and Sato (1981) demonstrated that somatic embryogenesis of Medicago sativa was significantly decreased by omitting Mg from the formulated medium. Also, Kintzios et al. (2004) and Saha et al. (2011) reported that the high level of Mg caused the increasing of embryogenesis of melon and P. ovata, respectively. Mg is known as an essential component of the chlorophyll molecule and essential for the activity of many enzymes in a nonspecific way, particularly those involved in the phosphate transfer (Yan et al. 2018). Mg is an absolute prerequisite for ATP synthesis and accumulation of ribosome subunits (Yan et al. 2018). Although the common culture media usually contain a moderately low concentration of Mg, somatic embryogenesis needs a higher concentration of Mg (Minyaka et al. 2008).
According to our results, somatic embryogenesis was enhanced by increasing the phosphorus concentration in the formulated medium. A similar result was reported by Kintzios et al. (2004) in melon. Phosphorus as a vital element involved in several macromolecules such as phospholipids, nucleic acids, ATP, and co-enzymes (Pang et al. 2018). An enhancement phosphorus accumulation under in vitro condition might have been associated with higher rates of synthesis and metabolism of the protein (Elkonin and Pakhomova 2000). Milazzo et al. (1999) demonstrated that proline-linked activation of synthesis of aromatic compounds and purine through the stimulation of the pentose phosphate pathway might be fundamental for embryogenesis.
According to the results of this study, increases in the potassium (K) concentration lead to a significant enhancement during the somatic embryogenesis of chrysanthemum. Similar with our results, Saha et al. (2011) reported that somatic embryogenesis of P. ovata was improved by increasing the K concentration in the medium. Also, Ghosh and Sen (1991) and Shetty and McKersie (1993) indicated that potassium supports the somatic embryogenesis of Asparagus cooperi and M. sativa, respectively. Also, K plays an important role in embryogenesis of sweet orange (Niedz and Evens 2008). K is a fundamental cation in plant metabolism, and no other elements can replace it. This macronutrient is vital for plant growth and development. K can considerably exert an impact on the tissue morphology and cause the change of metabolism (Knutsson et al. 2018). Moreover, many studies have indicated that K deficiency in the culture media resulted in the restriction of embryogenesis, hyperhydricity, reduction of the phosphate absorption rate, and finally decrease in the cell elongation (Ghosh and Sen 1991; Shetty and McKersie 1993; Niedz and Evens 2008; Saha et al. 2011).
Our results showed that improvements in somatic embryogenesis could be achieved by decreasing in the NH4+ concentrations. To some extent, the embryogenic response in different species such as sweet potato (Norton and LaBonte 1996) and melon (Kintzios et al. 2004) can be enhanced by decreasing in the levels of ammonium. Also, the negative impact of ammonium has been reported previously (Barcelo et al. 1992). According to Selby and Harvey (1990), the sensitiveness of the Picea sitchensis organogenesis to the different ratios of NH4+:NO3− is inevitable. Based on Selby and Harvey’s (1990) study, the 1:5 to 1:2 ratio could be optimal for adventitious bud development; also, such similar ratios were identified in Chlorophytum borivilianum callus cultures for stimulating optimal somatic embryogenesis (Arora et al. 1999).
The calcium (Ca) element is necessitated in in vitro morphogenesis and is essential for several responses regulated by plant growth substances, especially cytokinins and auxins (Demidchik and Shabala 2018). Our results showed that somatic embryogenesis of chrysanthemum was enhanced by increasing the Ca concentration in the medium. In sympathy with our finding, several studies showed that somatic embryogenesis of Astragalus adsurgens (Luo et al. 2003), Eucalyptus urophylla (Arruda et al. 2000), and Hevea brasiliensis (Etienne et al. 1997) was significantly increased by increasing Ca level in the formulated medium. Also, somatic embryogenesis of carrot emerged with a rise of Ca in the medium (Jansen et al. 1990).
Our results showed that decreases or increases in chlorine (Cl) lead to a significant reduction of the somatic embryogenesis of chrysanthemum. Cl is an essential component for osmoregulation (Flowers and Colmer 2015). Similar to our result, Luo et al. (2003) reported that too high or low concentration of Cl in media could be a sense in low regeneration frequency.
According to the data fusion–GA model, an optimal NSE can be obtained from a medium containing 0.61 μM thiamine, 5.93 μM nicotinic acid, 0.25 μM biotin, and 0.26 μM riboflavin.
The effects of nicotinic acid, riboflavin, biotin, and thiamine on somatic embryogenesis have been shown by several studies (Bonner 1937; Barwale et al. 1986; Asano et al. 1996; Guillou et al. 2018). Barwale et al. (1986) reported that 1.0 μM thiamine caused an increase in the embryogenesis of soybean compared with 0.2 μM thiamine (level of thiamine in MS medium). Also, these authors mentioned that embryogenesis was increased by using 32.4 μM nicotinic acid. In another study, Asano et al. (1996) indicated that increasing embryogenesis of Zoysia japonica Steud. was achieved by adding riboflavin and thiamine to the formulated media. Moreover, riboflavin alone did not affect embryogenesis frequency, but in the presence of thiamine at high concentration (4 mg/l or higher level) leads to an increase in embryogenesis. Riboflavin is a core component of two cofactors, namely, flavin mononucleotide (FMN) and flavin adenine dinucleotide (FAD), which participated in several reduction reactions which are essential to the core energy metabolism (Asensi-Fabado and Munné-Bosch 2010). FMN and FAD also play important roles in many physiological processes, such as photorepair of DNA, nitrogen fixation, phototropism, and circadian clocks (Sa et al. 2016). Al-Khayri (2001) showed the effect of biotin and thiamine on embryogenesis of date palm and reported the necessity of these compounds for somatic embryogenesis. Bonner (1937) also reported that obtaining the maximum number of somatic embryos is dependent on the presentence of both thiamine and biotin in the culture media. Thiamine is an essential cofactor in the metabolism of carbohydrate, which participated in the biosynthesis of some amino acids (Minhas et al. 2018). Biotin is a water-soluble component acting as a cofactor for several enzymes involved in carbohydrate metabolism and also has an important role in ATP production (Galili et al. 2016).
Nicotinic acid is the main compound of the electron transporter coenzyme nicotinamide adenine dinucleotide (NAD+) and nicotinamide adenine dinucleotide phosphate (NADP+) which are involved in a wide range of oxidation-reduction reactions, especially during oxidative phosphorylation and glycolysis (Ashihara et al. 2015). In line with our results, Asano et al. (1996) reported that increasing the concentration of nicotinic acid enhanced the somatic embryogenesis of Z. japonica.
Many studies demonstrated that the embryogenic process needs a high level of energy (Bonner 1937; Barwale et al. 1986; Asano et al. 1996; Guillou et al. 2018). So, the drastic effects of thiamine, nicotinic acid, riboflavin, and biotin on somatic embryogenesis of chrysanthemum might be due to the fact that these components play fundamental roles in carbohydrate and energy metabolisms as well as amino acid synthesis.
This study was aimed at providing a new procedure for prediction-optimization of the species-unique medium formulation and evaluating the effectiveness of this procedure by making a comparison between new formulated medium (HNT) and common MS medium in somatic embryogenesis of chrysanthemum. In this study, data fusion–NSGAII and GA was imposed to the somatic embryogenesis of chrysanthemum experiment dataset. Based on the results, identifying interaction effects can be done quickly and precisely in data fusion instead of common statistical analysis for a large number of experiments. Finally, this model could be able to specify the optimal combination of nutrients for obtaining the most proper achievements for the studied purposes. The HNT medium formulated by data fusion–NSGAII and GA is assumed to be more advantageous for somatic embryogenesis of chrysanthemum in comparison with MS medium. Also, our results revealed the appropriateness of HNT medium for somatic embryogenesis in various cultivars of chrysanthemum. Therefore, data fusion–NSGAII and GA can be recognized as a powerful tool for utilization in different areas of plant tissue culture such as medium formulations so proposing a new nutrient medium can be done quickly and effectively with an increased in the productivity in plant tissue culture area.
Data availability
All processed data are available without restriction upon inquiry.
References
Aiello G, Giovino I, Vallone M, Catania P, Argento A (2018) A decision support system based on multisensor data fusion for sustainable greenhouse management. J Clean Prod 172:4057–4065. https://doi.org/10.1016/j.jclepro.2017.02.197
Akin M, Eyduran SP, Eyduran E, Reed BM (2020) Analysis of macro nutrient related growth responses using multivariate adaptive regression splines. Plant Cell Tissue Organ Cult 140:661–670. https://doi.org/10.1007/s11240-019-01763-8
Al-Khayri JM (2001) Optimization of biotin and thiamine requirements for somatic embryogenesis of date palm (Phoenix dactylifera L.). In Vitro Cell Dev Biol Plant 37(4):453–456. https://doi.org/10.1007/s11627-001-0079-x
Alanagh EN, G-a G, Haddad R, Maleki S, Landín M, Gallego PP (2014) Design of tissue culture media for efficient Prunus rootstock micropropagation using artificial intelligence models. Plant Cell Tissue Organ Cult 117(3):349–359. https://doi.org/10.1007/s11240-014-0444-1
Alizadeh MR, Nikoo MR (2018) A fusion-based methodology for meteorological drought estimation using remote sensing data. Remote Sens Environ 211:229–247. https://doi.org/10.1016/j.rse.2018.04.001
Arab MM, Yadollahi A, Ahmadi H, Eftekhari M, Maleki M (2017) Mathematical modeling and optimizing of in vitro hormonal combination for G× N15 vegetative rootstock proliferation using artificial neural network-genetic algorithm (ANN-GA). Front Plant Sci 8:1853. https://doi.org/10.3389/fpls.2017.01853
Araghinejad S, Fayaz N, Hosseini-Moghari S-M (2018) Development of a hybrid data driven model for hydrological estimation. Water Resour Manag 32(11):3737–3750. https://doi.org/10.1007/s11269-018-2016-3
Araghinejad S, Hosseini-Moghari S-M, Eslamian S (2017) Application of data-driven models in drought forecasting. In: Handbook of Drought and Water Scarcity. CRC Press, pp. 423–440
Arora DK, Sun SS, Ramawat KG, Merillon J-M (1999) Factors affecting somation embryogenesis in long term callus cultures of ‘safed musli’ (Chlorophytum borivilianum), an endangered wonder herb. Indian J Exp Biol 31:75–82
Arruda S, Souza G, Almeida M, Gonçalves A (2000) Anatomical and biochemical characterization of the calcium effect on Eucalyptus urophylla callus morphogenesis in vitro. Plant Cell Tissue Organ Cult 63(2):142–154. https://doi.org/10.1023/A:1006482702094
Asano Y, Katsumoto H, Inokuma C, Kaneko S, Ito Y, Fujiie A (1996) Cytokinin and thiamine requirements and stimulative effects of riboflavin and α-ketoglutaric acid on embryogenic callus induction from the seeds of Zoysia japonica Steud. J Plant Physiol 149(3–4):413–417. https://doi.org/10.1016/S0176-1617(96)80142-8
Asensi-Fabado MA, Munné-Bosch S (2010) Vitamins in plants: occurrence, biosynthesis and antioxidant function. Trends Plant Sci 15(10):582–592. https://doi.org/10.1016/j.tplants.2010.07.003
Ashihara H, Ludwig IA, Katahira R, Yokota T, Fujimura T, Crozier A (2015) Trigonelline and related nicotinic acid metabolites: occurrence, biosynthesis, taxonomic considerations, and their roles in planta and in human health. Phytochem Rev 14(5):765–798. https://doi.org/10.1007/s11101-014-9375-z
Barcelo P, Lazzeri PA, Martin A, Lörz H (1992) Competence of cereal leaf cells. II. Influence of auxin, ammonium and explant age on regeneration. J Plant Physiol 139(4):448–454. https://doi.org/10.1016/S0176-1617(11)80493-1
Barone JO (2019) Use of multiple regression analysis and artificial neural networks to model the effect of nitrogen in the organogenesis of Pinus taeda L. Plant Cell Tissue Organ Cult 137(3):455–464. https://doi.org/10.1007/s11240-019-01581-y
Barwale U, Kerns H, Widholm J (1986) Plant regeneration from callus cultures of several soybean genotypes via embryogenesis and organogenesis. Planta 167(4):473–481. https://doi.org/10.1007/BF00391223
Bonner J (1937) The role of vitamins in plant development. Bot Rev 3(12):616–640. https://doi.org/10.1007/BF02872294
Bozorg-Haddad O, Azarnivand A, Hosseini-Moghari S-M, Loáiciga HA (2016) Development of a comparative multiple criteria framework for ranking pareto optimal solutions of a multiobjective reservoir operation problem. J Irrig Drain Eng 142(7):e04016019. https://doi.org/10.1061/(ASCE)IR.1943-4774.0001028
Bozorg-Haddad O, Azarnivand A, Hosseini-Moghari S-M, Loáiciga Hugo A (2017) WASPAS application and evolutionary algorithm benchmarking in optimal reservoir optimization problems. J Water Resour Plan Manag 143(1):04016070. https://doi.org/10.1061/(ASCE)WR.1943-5452.0000716
da Silva JAT (2003) Chrysanthemum: advances in tissue culture, cryopreservation, postharvest technology, genetics and transgenic biotechnology. Biotechnol Adv 21(8):715–766. https://doi.org/10.1016/S0734-9750(03)00117-4
da Silva JAT, Kulus D (2014) Chrysanthemum biotechnology: discoveries from the recent literature. Folia Hortic 26(2):67–77. https://doi.org/10.2478/fhort-2014-0007
Demidchik V, Shabala S (2018) Mechanisms of cytosolic calcium elevation in plants: the role of ion channels, calcium extrusion systems and NADPH oxidase-mediated ‘ROS-Ca2+ hub’. Funct Plant Biol 45(2):9–27. https://doi.org/10.1071/FP16420
Dezfooli D, Hosseini-Moghari S-M, Ebrahimi K, Araghinejad S (2018) Classification of water quality status based on minimum quality parameters: application of machine learning techniques. Model Earth Syst Environ 4(1):311–324. https://doi.org/10.1007/s40808-017-0406-9
Ding B, Smith ES, Ding H (2005) Mobilization of the iron centre in IscA for the iron–sulphur cluster assembly in IscU. Biochem J 389(3):797–802. https://doi.org/10.1042/BJ20050405
Dussert S, Verdeil J-L, Buffard-Morel J (1995) Specific nutrient uptake during initiation of somatic embryogenesis in coconut calluses. Plant Sci 111(2):229–236. https://doi.org/10.1016/0168-9452(95)04248-S
Ebrahimian H, Dialameh B, Hosseini-Moghari S-M, Ebrahimian A (2020) Optimal conjunctive use of aqua-agriculture reservoir and irrigation canal for paddy fields (case study: Tajan irrigation network, Iran). Paddy Water Environ 18(3):499–514. https://doi.org/10.1007/s10333-020-00797-5
Elkonin LA, Pakhomova NV (2000) Influence of nitrogen and phosphorus on induction embryogenic callus of sorghum. Plant Cell Tissue Organ Cult 61(2):115–123. https://doi.org/10.1023/A:1006472418218
Etienne H, Lartaud M, Carron M-P, Michaux-Ferrière N (1997) Use of calcium to optimize long-term proliferation of friable embryogenic calluses and plant regeneration in Hevea brasiliensis (Müll. Arg.). J Exp Bot 48(1):129–137. https://doi.org/10.1093/jxb/48.1.129
Farhadi S, Salehi M, Moieni A, Safaie N, Sabet MS (2020) Modeling of paclitaxel biosynthesis elicitation in Corylus avellana cell culture using adaptive neuro-fuzzy inference system-genetic algorithm (ANFIS-GA) and multiple regression methods. PLoS One 15(8):e0237478. https://doi.org/10.1371/journal.pone.0237478
Fisichella M, Silvi E, Morini S (2000) Regeneration of somatic embryos and roots from quince leaves cultured on media with different macroelement composition. Plant Cell Tissue Organ Cult 63(2):101–107. https://doi.org/10.1023/A:1006407803660
Flowers TJ, Colmer TD (2015) Plant salt tolerance: adaptations in halophytes. Ann Bot 115(3):327–331. https://doi.org/10.1093/aob/mcu267
Gago J, Landín M, Gallego PP (2010) Artificial neural networks modeling the in vitro rhizogenesis and acclimatization of Vitis vinifera L. J Plant Physiol 167(15):1226–1231. https://doi.org/10.1016/j.jplph.2010.04.008
Gago J, Martinez-Nunez L, Landin M, Flexas J, Gallego PP (2014) Modeling the effects of light and sucrose on in vitro propagated plants: a multiscale system analysis using artificial intelligence technology. PLoS One 9(1):e85989. https://doi.org/10.1371/journal.pone.0085989
Gago J, Pérez-Tornero O, Landín M, Burgos L, Gallego PP (2011) Improving knowledge of plant tissue culture and media formulation by neurofuzzy logic: a practical case of data mining using apricot databases. J Plant Physiol 168(15):1858–1865. https://doi.org/10.1016/j.jplph.2011.04.008
Galili G, Amir R, Fernie AR (2016) The regulation of essential amino acid synthesis and accumulation in plants. Annu Rev Plant Biol 67:153–178. https://doi.org/10.1146/annurev-arplant-043015-112213
Gamborg OLC, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50(1):151–158. https://doi.org/10.1016/0014-4827(68)90403-5
García-Pérez P, Lozano-Milo E, Landín M, Gallego PP (2020a) Combining medicinal plant in vitro culture with machine learning technologies for maximizing the production of phenolic compounds. Antioxidants 9(3):210. https://doi.org/10.3390/antiox9030210
García-Pérez P, Lozano-Milo E, Landín M, Gallego PP (2020b) Machine learning technology reveals the concealed interactions of phytohormones on medicinal plant in vitro organogenesis. Biomolecules 10(5):746. https://doi.org/10.3390/biom10050746
Ghosh B, Sen S (1991) Plant regeneration through somatic embryogenesis from spear callus culture of Asparagus cooperi Baker. Plant Cell Rep 9(12):667–670. https://doi.org/10.1007/BF00235353
Greenway MB, Phillips IC, Lloyd MN, Hubstenberger JF, Phillips GC (2012) A nutrient medium for diverse applications and tissue growth of plant species in vitro. Vitro Cell Dev Biol Plant 48(4):403–410. https://doi.org/10.1007/s11627-012-9452-1
Greer MS, Kovalchuk I, Eudes F (2009) Ammonium nitrate improves direct somatic embryogenesis and biolistic transformation of Triticum aestivum. New Biotechnol 26(1–2):44–52. https://doi.org/10.1016/j.nbt.2009.02.003
Groll J, Mycock D, Gray V (2002) Effect of medium salt concentration on differentiation and maturation of somatic embryos of cassava (Manihot esculenta Crantz). Ann Bot 89(5):645–648. https://doi.org/10.1093/aob/mcf095
Guillou C, Fillodeau A, Brulard E, Breton D, Maraschin SDF, Verdier D, Simon M, Ducos J-P (2018) Indirect somatic embryogenesis of Theobroma cacao L. in liquid medium and improvement of embryo-to-plantlet conversion rate. In Vitro Cell Dev Biol Plant 54(4):377–391. https://doi.org/10.1007/s11627-018-9909-y
Haddad Omid B, Hosseini-Moghari S-M, Loáiciga Hugo A (2016) Biogeography-based optimization algorithm for optimal operation of reservoir systems. J Water Resour Plan Manag 142(1):04015034. https://doi.org/10.1061/(ASCE)WR.1943-5452.0000558
Hararuk O, Zwart JA, Jones SE, Prairie Y, Solomon CT (2018) Model-data fusion to test hypothesized drivers of lake carbon cycling reveals importance of physical controls. J Geophys Res-Biogeo 123(3):1130–1142. https://doi.org/10.1002/2017JG004084
Hesami M, Alizadeh M, Naderi R, Tohidfar M (2020a) Forecasting and optimizing Agrobacterium-mediated genetic transformation via ensemble model- fruit fly optimization algorithm: a data mining approach using chrysanthemum databases. PLoS One 15:e0239901. https://doi.org/10.1371/journal.pone.0239901
Hesami M, Condori-Apfata JA, Valderrama Valencia M, Mohammadi M (2020b) Application of artificial neural network for modeling and studying in vitro genotype-independent shoot regeneration in wheat. Appl Sci 10(15):5370. https://doi.org/10.3390/app10155370
Hesami M, Daneshvar MH (2018a) In vitro adventitious shoot regeneration through direct and indirect organogenesis from seedling-derived hypocotyl segments of Ficus religiosa L.: an important medicinal plant. HortScience 53(1):55–61. https://doi.org/10.21273/HORTSCI12637-17
Hesami M, Daneshvar MH (2018b) Indirect organogenesis through seedling-derived leaf segments of Ficus religiosa-a multipurpose woody medicinal plant. J Crop Sci Biotechnol 21(2):129–136. https://doi.org/10.1007/s12892-018-0024-0
Hesami M, Daneshvar MH, Lotfi A (2017a) In vitro shoot proliferation through cotyledonary node and shoot tip explants of Ficus religiosa L. Plant Tissue Cult Biotechnol 27(1):85–88. https://doi.org/10.3329/ptcb.v27i1.35017
Hesami M, Daneshvar MH, Yoosefzadeh-Najafabadi M (2018a) Establishment of a protocol for in vitro seed germination and callus formation of Ficus religiosa L., an important medicinal plant. Jundishapur J Nat Pharm Prod 13(4):e62682. https://doi.org/10.5812/jjnpp.62682
Hesami M, Daneshvar MH, Yoosefzadeh-Najafabadi M (2019a) An efficient in vitro shoot regeneration through direct organogenesis from seedling-derived petiole and leaf segments and acclimatization of Ficus religiosa. J For Res 30(3):807–815
Hesami M, Jones AMP (2020) Application of artificial intelligence models and optimization algorithms in plant cell and tissue culture. Appl Microbiol Biotechnol 104:1–37. https://doi.org/10.1007/s00253-020-10888-2
Hesami M, Naderi R, Tohidfar M (2019b) Modeling and optimizing in vitro sterilization of chrysanthemum via multilayer perceptron-non-dominated sorting genetic algorithm-II (MLP-NSGAII). Front Plant Sci 10:282. https://doi.org/10.3389/fpls.2019.00282
Hesami M, Naderi R, Tohidfar M (2019c) Modeling and optimizing medium composition for shoot regeneration of chrysanthemum via radial basis function-non-dominated sorting genetic algorithm-II (RBF-NSGAII). Sci Rep 9(1):1–11. https://doi.org/10.1038/s41598-019-54257-0
Hesami M, Naderi R, Tohidfar M, Yoosefzadeh-Najafabadi M (2019d) Application of adaptive neuro-fuzzy inference system-non-dominated sorting genetic algorithm-II (ANFIS-NSGAII) for modeling and optimizing somatic embryogenesis of chrysanthemum. Front Plant Sci 10:869. https://doi.org/10.3389/fpls.2019.00869
Hesami M, Naderi R, Tohidfar M, Yoosefzadeh-Najafabadi M (2020c) Development of support vector machine-based model and comparative analysis with artificial neural network for modeling the plant tissue culture procedures: effect of plant growth regulators on somatic embryogenesis of chrysanthemum, as a case study. Plant Methods 16(1):112. https://doi.org/10.1186/s13007-020-00655-9
Hesami M, Naderi R, Yoosefzadeh-Najafabadi M (2018b) Optimizing sterilization conditions and growth regulator effects on in vitro shoot regeneration through direct organogenesis in Chenopodium quinoa. BioTechnologia 99(1):49–57. https://doi.org/10.5114/bta.2018.73561
Hesami M, Naderi R, Yoosefzadeh-Najafabadi M, Maleki M (2018c) In vitro culture as a powerful method for conserving Iranian ornamental geophytes. BioTechnologia 99(1):73–81. https://doi.org/10.5114/bta.2018.73563
Hesami M, Naderi R, Yoosefzadeh-Najafabadi M, Rahmati M (2017b) Data-driven modeling in plant tissue culture. J Appl Environ Biol Sci 7(8):37–44
Hildebrandt AC, Riker A, Duggar B (1946) The influence of the composition of the medium on growth in vitro of excised tobacco and sunflower tissue cultures. Am J Bot 33:591–597. https://doi.org/10.2307/2437399
Hosseini-Moghari S-M, Araghinejad S, Azarnivand A (2017) Drought forecasting using data-driven methods and an evolutionary algorithm. Model Earth Syst Environ 3(4):1675–1689. https://doi.org/10.1007/s40808-017-0385-x
Hosseini-Moghari S-M, Morovati R, Moghadas M, Araghinejad S (2015) Optimum operation of reservoir using two evolutionary algorithms: imperialist competitive algorithm (ICA) and cuckoo optimization algorithm (COA). Water Resour Manag 29(10):3749–3769. https://doi.org/10.1007/s11269-015-1027-6
Hosseini-Moghari SM, Araghinejad S (2015) Monthly and seasonal drought forecasting using statistical neural networks. Environ Earth Sci 74(1):397–412. https://doi.org/10.1007/s12665-015-4047-x
Jamshidi S, Yadollahi A, Arab MM, Soltani M, Eftekhari M, Sabzalipoor H, Sheikhi A, Shiri J (2019) Combining gene expression programming and genetic algorithm as a powerful hybrid modeling approach for pear rootstocks tissue culture media formulation. Plant Methods 15(1):136. https://doi.org/10.1186/s13007-019-0520-y
Jansen MA, Booij H, Schel JH, de Vries SC (1990) Calcium increases the yield of somatic embryos in carrot embryogenic suspension cultures. Plant Cell Rep 9(4):221–223. https://doi.org/10.1007/BF00232184
Jones B, Gunnerås SA, Petersson SV, Tarkowski P, Graham N, May S, Dolezal K, Sandberg G, Ljung K (2010) Cytokinin regulation of auxin synthesis in Arabidopsis involves a homeostatic feedback loop regulated via auxin and cytokinin signal transduction. Plant Cell 22:2956–2969. https://doi.org/10.1105/tpc.110.074856
Khvatkov P, Chernobrovkina M, Okuneva A, Dolgov S (2019) Creation of culture media for efficient duckweeds micropropagation (Wolffia arrhiza and Lemna minor) using artificial mathematical optimization models. Plant Cell Tissue Organ Cult 136(1):85–100. https://doi.org/10.1007/s11240-018-1494-
Kim HJ, Lee YS (2005) Identification of new dicaffeoylquinic acids from Chrysanthemum morifolium and their antioxidant activities. Planta Med 71(09):871–876. https://doi.org/10.1055/s-2005-873115
Kintzios S, Drossopoulos J, Lymperopoulos C (2001) Effect of vitamins and inorganic micronutrients on callus growth and somatic embryogenesis from leaves of chilli pepper. Plant Cell Tissue Organ Cult 67(1):55–62. https://doi.org/10.1023/A:1011610413177
Kintzios S, Stavropoulou E, Skamneli S (2004) Accumulation of selected macronutrients and carbohydrates in melon tissue cultures: association with pathways of in vitro dedifferentiation and differentiation (organogenesis, somatic embryogenesis). Plant Sci 167(3):655–664. https://doi.org/10.1016/j.plantsci.2004.05.021
Knutsson P, Cantatore V, Seemann M, Tam PL, Panas I (2018) Role of potassium in the enhancement of the catalytic activity of calcium oxide towards tar reduction. Appl Catal B-Environ 229:88–95. https://doi.org/10.1016/j.apcatb.2018.02.002
Koleva-Gudeva LR, Spasenoski M, Trajkova F (2007) Somatic embryogenesis in pepper anther culture: the effect of incubation treatments and different media. Sci Hortic 111(2):114–119. https://doi.org/10.1016/j.scienta.2006.10.013
Kothari S, Agarwal K, Kumar S (2004) Inorganic nutrient manipulation for highly improved in vitro plant regeneration in finger millet—Eleusine coracana (L.) Gaertn. In Vitro Cell Dev Biol Plant 40(5):515–519. https://doi.org/10.1079/IVP2004564
Lin G-H, Lin L, Liang H-W, Ma X, Wang J-Y, Wu L-P, Jiang H-D, Bruce IC, Xia Q (2010) Antioxidant action of a Chrysanthemum morifolium extract protects rat brain against ischemia and reperfusion injury. J Med Food 13(2):306–311. https://doi.org/10.1089/jmf.2009.1184
Loh C, Lim G (1992) The influence of medium components on secondary embryogenesis of winter oilseed rape, Brassica napus L. ssp. oleifera (Metzg.) Sink. New Phytol 121(3):425–430. https://doi.org/10.1111/j.1469-8137.1992.tb02942.x
Luo J-P, Jiang S-T, Pan L-J (2003) Cold-enhanced somatic embryogenesis in cell suspension cultures of Astragalus adsurgens Pall.: relationship with exogenous calcium during cold pretreatment. Plant Growth Regul 40(2):171–177. https://doi.org/10.1023/A:1024295901808
May R, Trigiano R (1991) Somatic embryogenesis and plant regeneration from leaves of Dendranthema grandiflora. J Am Soc Hortic Sci 116(2):366–371
Milazzo MC, Zheng Z, Kellett G, Haynesworth K, Shetty K (1999) Stimulation of benzyladenine-induced in vitro shoot organogenesis and endogenous proline in melon (Cucumis melo L.) by fish protein hydrolysates in combination with proline analogues. J Agric Food Chem 47(4):1771–1775. https://doi.org/10.1021/jf9812883
Minhas AP, Tuli R, Puri S (2018) Pathway editing targets for thiamine biofortification in rice grains. Front Plant Sci 9:e975. https://doi.org/10.3389/fpls.2018.00975
Minyaka E, Niemenak N, Sangare A, Omokolo DN (2008) Effect of MgSO4 and K2SO4 on somatic embryo differentiation in Theobroma cacao L. Plant Cell Tissue Organ Cult 94(2):149–160. https://doi.org/10.1007/s11240-008-9398-5
Moravej M, Amani P, Hosseini-Moghari S-M (2020) Groundwater level simulation and forecasting using interior search algorithm-least square support vector regression (ISA-LSSVR). Groundw Sustain Dev 11:1–18
Moravej M, Hosseini-Moghari S-M (2016) Large scale reservoirs system operation optimization: the interior search algorithm (ISA) approach. Water Resour Manag 30(10):3389–3407. https://doi.org/10.1007/s11269-016-1358-y
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Naing AH, Kim CK, Yun BJ, Jin JY, Lim KB (2013) Primary and secondary somatic embryogenesis in Chrysanthemum cv. Euro Plant Cell Tiss Org Cult 112(3):361–368. https://doi.org/10.1007/s11240-012-0243-5
Nezami-Alanagh E, Garoosi G-A, Maleki S, Landín M, Gallego PP (2017) Predicting optimal in vitro culture medium for Pistacia vera micropropagation using neural networks models. Plant Cell Tissue Organ Cult 129(1):19–33. https://doi.org/10.1007/s11240-016-1152-9
Niazian M, Sadat-Noori SA, Abdipour M, Tohidfar M, Mortazavian SMM (2018) Image processing and artificial neural network-based models to measure and predict physical properties of embryogenic callus and number of somatic embryos in ajowan (Trachyspermum ammi (L.) Sprague). In Vitro Cell Dev-Pl 54(1):54–68. https://doi.org/10.1007/s11627-017-9877-7
Niedz RP, Evens TJ (2008) The effects of nitrogen and potassium nutrition on the growth of nonembryogenic and embryogenic tissue of sweet orange (Citrus sinensis (L.) Osbeck). BMC Plant Biol 8(1):126. https://doi.org/10.1186/1471-2229-8-126
Nitsch JP, Nitsch C (1969) Haploid plants from pollen grains. Science 163(3862):85–87. https://doi.org/10.1126/science.163.3862.85
Noda N, Yoshioka S, Kishimoto S, Nakayama M, Douzono M, Tanaka Y, Aida R (2017) Generation of blue chrysanthemums by anthocyanin B-ring hydroxylation and glucosylation and its coloration mechanism. Sci Adv 3(7):e1602785. https://doi.org/10.1126/sciadv.1602785
Norton M, LaBonte D (1996) Cultural conditions affecting the frequency of embryoid formation in sweetpotato (Ipomoea batatas). HortScience 31(4):630–630
Pang J, Ryan MH, Lambers H, Siddique KH (2018) Phosphorus acquisition and utilisation in crop legumes under global change. Curr Opin Plant Biol 45:248–254. https://doi.org/10.1016/j.pbi.2018.05.012
Sa N, Rawat R, Thornburg C, Walker KD, Roje S (2016) Identification and characterization of the missing phosphatase on the riboflavin biosynthesis pathway in Arabidopsis thaliana. Plant J 88(5):705–716. https://doi.org/10.1111/tpj.13291
Saha P, Bandyopadhyay S, Raychaudhuri SS (2011) Formulation of nutrient medium for in vitro somatic embryo induction in Plantago ovata Forsk. Biol Trace Elem Res 140(2):225–243. https://doi.org/10.1007/s12011-010-8684-3
Saito K (2004) Sulfur assimilatory metabolism. The long and smelling road. Plant Physiol 136(1):2443–2450. https://doi.org/10.1104/pp.104.046755
Salehi M, Farhadi S, Moieni A, Safaie N, Ahmadi H (2020) Mathematical modeling of growth and paclitaxel biosynthesis in Corylus avellana cell culture responding to fungal elicitors using multilayer perceptron-genetic algorithm. Front Plant Sci 11:1148. https://doi.org/10.3389/fpls.2020.01148
Samson NP, Campa C, Le Gal L, Noirot M, Thomas G, Lokeswari T, De Kochko A (2006) Effect of primary culture medium composition on high frequency somatic embryogenesis in different Coffea species. Plant Cell Tissue Organ Cult 86(1):37–45. https://doi.org/10.1007/s11240-006-9094-2
Schenk RU, Hildebrandt A (1972) Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can J Bot 50(1):199–204. https://doi.org/10.1139/b72-026
Selby C, Harvey B (1990) The influence of composition of the basal medium on the growth and morphogenesis of cultured Sitka spruce (Picea sitchensis) tissues. Ann Bot 65(4):395–407. https://doi.org/10.1093/oxfordjournals.aob.a087950
Sheikhi A, Mirdehghan SH, Arab MM, Eftekhari M, Ahmadi H, Jamshidi S, Gheysarbigi S (2020) Novel organic-based postharvest sanitizer formulation using Box Behnken design and mathematical modeling approach: a case study of fresh pistachio storage under modified atmosphere packaging. Postharvest Biol Technol 160:111047. https://doi.org/10.1016/j.postharvbio.2019.111047
Shetty K, McKersie BD (1993) Proline, thioproline and potassium mediated stimulation of somatic embryogenesis in alfalfa (Medicago sativa L.). Plant Sci 88(2):185–193. https://doi.org/10.1016/0168-9452(93)90090-M
Smith AD, Agar JN, Johnson KA, Frazzon J, Amster IJ, Dean DR, Johnson MK (2001) Sulfur transfer from IscS to IscU: the first step in iron− sulfur cluster biosynthesis. J Am Chem Soc 123(44):11103–11104. https://doi.org/10.1021/ja016757n
Tanaka K, Kanno Y, Kudo S, Suzuki M (2000) Somatic embryogenesis and plant regeneration in chrysanthemum (Dendranthema grandiflorum (Ramat.) Kitamura). Plant Cell Rep 19(10):946–953. https://doi.org/10.1007/s002990000225
Taylor NJ, Edwards M, Kiernan RJ, Davey CD, Blakesley D, Henshaw GG (1996) Development of friable embryogenic callus and embryogenic suspension culture systems in cassava (Manihot esculenta Crantz). Nat Biotechnol 14(6):726–730. https://doi.org/10.1038/nbt0696-726
Trigiano R, May R, Conger B (1992) Reduced nitrogen influences somatic embryo quality and plant regeneration from suspension cultures of orchardgrass. Vitro Cell Dev Biol Plant 28(4):187–191. https://doi.org/10.1007/BF02823315
Walker K, Sato S (1981) Morphogenesis in callus tissue of Medicago sativa: the role of ammonium ion in somatic embryogenesis. Plant Cell Tissue Organ Cult 1(1):109–121. https://doi.org/10.1007/BF02318910
White PR (1942) Plant tissue cultures. Annu Rev Biochem 11(1):615–628. https://doi.org/10.1146/annurev.bi.11.070142.003151
Wu X-M, Zhang Q-Z, Wang Y-Z (2018) Traceability of wild Paris polyphylla Smith var. yunnanensis based on data fusion strategy of FT-MIR and UV–Vis combined with SVM and random forest. Spectrochim Acta A Mol Biomol Spectrosc 205:479–488. https://doi.org/10.1016/j.saa.2018.07.067
Xu P, Zhang Z, Wang B, Xia X, Jia J (2012) Somatic embryogenesis and plant regeneration in chrysanthemum (Yuukou). Plant Cell Tissue Organ Cult 111(3):393–397. https://doi.org/10.1007/s11240-012-0201-2
Xu XM, Møller SG (2004) AtNAP7 is a plastidic SufC-like ATP-binding cassette/ATPase essential for Arabidopsis embryogenesis. Proc Natl Acad Sci 101(24):9143–9148. https://doi.org/10.1073/pnas.0400799101
Yan Y-W, Mao D-D, Yang L, Qi J-L, Zhang X-X, Tang Q-L, Li Y-P, Tang R-J, Luan S (2018) Magnesium transporter MGT6 plays an essential role in maintaining magnesium homeostasis and regulating high magnesium tolerance in Arabidopsis. Front Plant Sci 9:e274. https://doi.org/10.3389/fpls.2018.00274
Yang L, Aobulikasimu N, Cheng P, Wang J-H, Li H (2017) Analysis of floral volatile components and antioxidant activity of different varieties of Chrysanthemum morifolium. Molecules 22(10):1790. https://doi.org/10.3390/molecules22101790
Zhao Y (2008) The role of local biosynthesis of auxin and cytokinin in plant development. Curr Opin Plant Biol 11(1):16–22. https://doi.org/10.1016/j.pbi.2007.10.008
Author information
Authors and Affiliations
Contributions
M.H. was responsible for performing the experiments, data modeling, summing up, and writing the manuscript. R.N. and M.T. were responsible for designing and leading the experiments and revising the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
This work does not involve any human participation nor live animals performed by any of the listed authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 567 kb)
Rights and permissions
About this article
Cite this article
Hesami, M., Naderi, R. & Tohidfar, M. Introducing a hybrid artificial intelligence method for high-throughput modeling and optimizing plant tissue culture processes: the establishment of a new embryogenesis medium for chrysanthemum, as a case study. Appl Microbiol Biotechnol 104, 10249–10263 (2020). https://doi.org/10.1007/s00253-020-10978-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10978-1