Abstract
Summary
Vitamin D insufficiency and sarcopenia are crucial risk factors for osteoporosis. In a study of noninstitutionalized elderly subjects, we investigated the simultaneous effect of vitamin D and sarcopenia on bone mineral density (BMD) and found that sarcopenia was associated with low BMD in the femur, especially in those with suboptimal vitamin D levels.
Introduction
Although vitamin D insufficiency and sarcopenia are prevalent in the elderly population worldwide, their possible influence on BMD has not been determined. We aimed to investigate the different effect of vitamin D insufficiency and sarcopenia on BMD in the elderly Korean population.
Methods
Individuals aged 60 or older were selected from those who participated in the Fourth and Fifth Korea National Health and Nutrition Examination Surveys conducted in 2009 and 2010; 1,596 males and 1,886 females were analyzed. Appendicular skeletal muscle mass (ASM) and BMD were assessed by dual-energy X-ray absorptiometry; serum 25-hydroxyvitamin D [25(OH)D] and a panel of clinical and laboratory parameters were also measured.
Results
The study population was divided into four groups according to their vitamin D and sarcopenic status. BMD in total femur and in the femoral neck but not the lumbar spine was markedly decreased in sarcopenic subjects with vitamin D insufficiency [25(OH)D < 20 ng/ml] comparing to other groups, regardless of gender. Multivariable linear regression models showed that BMD was significantly associated with ASM and high daily calcium intake as well as conventional risk factors such as age, body mass index (BMI), and history of fracture. Independent predictors for low femur BMD included sarcopenia, low daily calcium intake, low 25(OH)D levels, age, and BMI.
Conclusions
These data showed that an association between vitamin D insufficiency and low BMD was more prominent in elderly subjects with sarcopenia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As elderly population increases rapidly worldwide, age-related complications and morbidity place a considerable and increasing burden on the public health care system as well as individual patients. Osteoporosis, a common disease in the elderly, is characterized by low bone mass and deterioration of bone microarchitecture, which increases the risk of fragility fractures [1]. Vitamin D [25(OH)D] plays an important role in bone and mineral metabolism, and its deficiency is closely associated with metabolic bone disease [2]. Vitamin D insufficiency [25(OH)D < 20 ng/ml] is prevalent worldwide in both young and older populations and is particularly common in South Korea, where it was detected in 47.3 % of males and 64.5 % of females in a study population that included 3,047 males and 3,878 females aged 10 years or older [2, 3].
Sarcopenia is a syndrome characterized by progressive and generalized loss of skeletal muscle mass and strength [4]. Sarcopenia is prevalent among elderly people and is significantly associated with low BMD and possibly with increased risk of osteoporotic fracture [5–8]. This association may be attributed to the effects of muscle mass and muscle contraction on maintaining bone mass [9]. Furthermore, the inflammatory catabolic process underlying sarcopenia may induce bone loss [10]. A number of cardiometabolic disorders, including diabetes mellitus, metabolic syndrome, and coronary artery disease, have been also associated with sarcopenia [11–14]. Several recent studies have reported the effect of either vitamin D or sarcopenia on BMD in elderly populations [5–7, 15, 16]. However, no study has yet investigated the simultaneous effect of vitamin D and sarcopenia on BMD, particularly in an elderly Asian population.
Besides the effect of vitamin D on bone metabolism, vitamin D also plays an important role in muscle mass and strength [17]. This implies vitamin D insufficiency and sarcopenia, together, could induce low BMD through decreased mechanical load. Therefore, we evaluated the association of vitamin D insufficiency with BMD according to sarcopenia status among elderly South Koreans.
Methods
Study participants
This study is based on data obtained from the Korea National Health and Nutrition Examination Survey (KNHANES) conducted in 2009 and 2010. The KNHANES is a cross-sectional study regularly conducted by the Division of Chronic Disease Surveillance, Korea Centers for Disease Control and Prevention of the Ministry of Health and Welfare to examine the general health and nutritional status of the civilian noninstitutionalized population of the South Korea [18]. The KNHANES consists of four surveys: a health interview survey, a health behavior survey, a health examination survey, and a nutrition survey [19]. A total of 2,363 individuals aged ≥60 years participated in the 2009 KNHANES surveys (which included 7,798 subjects aged ≥20 years), and 2,028 individuals aged ≥60 years participated in the 2010 surveys (which included 6,665 subjects aged ≥20 years). Among these 4,391 participants aged ≥60 years, we excluded individuals whose BMD, appendicular skeletal muscle mass (ASM), or 25(OH)D level was not measured in the survey (n = 909). All participants provided written informed consent before participating in the survey.
Measurement of bone mineral density and appendicular skeletal muscle mass
BMD measurements of the femoral neck, total femur (which includes the femoral neck, trochanter, and intertrochanter area), and lumbar spine (L1–L4) were conducted by dual-energy X-ray absorptiometry (DXA) using Discovery QDR 4500 W fan-beam densitometers (Hologic Inc., Denver, Co, USA). Densitometers were calibrated daily with phantom supplied from the manufacturer to maintain the stability of DXA measurements. The coefficients of variation (in percent) for duplicated measurements in 30 adults satisfied the following precision criteria: lumbar spine, <1.9 %; femur neck, <2.5 %; and total femur, <1.8 %. In the analysis, we used BMD (in grams per square centimeter) or a T-score based on the gender-specific mean for healthy young Asian adults, which was provided by the manufacturer. A T-score ≥ −1.0 was considered normal, osteopenia was defined as a T-score between −1.0 and −2.5, and osteoporosis was defined as a T-score ≤ −2.5, in accordance with World Health Organization criteria [20]. Low BMD was defined as a T-score < −1.0. ASM was determined by using Discovery QDR 4500 W fan-beam densitometers (Hologic Inc.) To define sarcopenia, we evaluated ASM as a percentage of body weight (ASM/Wt), which was modified from the study of Janssen et al. [21]. We found that body weight in the denominator was the best method to minimize the effect of the strong correlation between ASM and body weight (Supplementary Table 1). Sarcopenia was defined as <1 SD below the sex-specific mean for a young reference group from the dataset of KNHANES 2009 and 2010 (780 men and 967 women, 20–30 years old). The cutoff point for sarcopenia was 32.2 % for men and 25.4 % for women.
Measurement of risk factors of low BMD
Possible risk factors were selected from a literature review of population-based cross-sectional and longitudinal studies assessing risk factors for low BMD. Waist circumference was measured during the health examination, and body mass index (BMI) was determined as the ratio of weight to height (in kilograms per square centimeter). Dietary calcium intake was estimated from a 24-h dietary recall. Standardized health questionnaires were used to determine behavior and lifestyle characteristics including alcohol consumption (drinks/week), cigarette smoking status (never/past/current), regular exercise (yes/no), and current use of vitamin and mineral supplements (yes/no). Alcohol consumption was recorded as the average number of drinks consumed per week regardless of the type of alcoholic beverage, as previously described [22]. Regular exercise was defined as engaging in moderate or intense exercise on a regular basis (>three times per week). Questionnaires also obtained information about reproductive health (e.g., early menopause and use of exogenous hormones), history of fracture, and family history of osteoporosis.
Blood samples collected from each participant during the survey were processed, refrigerated immediately, and transported in cold storage to the central laboratory (Neodin Medical Institute, Seoul, South Korea). All blood samples were analyzed within 24 h after transportation. Serum 25-hydroxy vitamin D [25(OH)D] concentration was determined by radioimmunoassay (DiaSorin Inc., Stillwater, MN) using a gamma counter (1470 Wizard; PerkinElmer, Turku, Finland). Vitamin D insufficiency was defined as serum 25(OH)D < 20 ng/ml [2, 23]. Total cholesterol, high-density lipoprotein cholesterol, triglyceride, fasting glucose, and alanine aminotransferase levels were determined by using a Hitachi 7600 automated chemistry analyzer (Hitachi, Tokyo, Japan) with reagents produced by Sekisui Medical Corporation (Tokyo, Japan). Insulin concentration was determined by immunoradiometry (INS-IRMA; Bio-Source, Nivelles, Belgium). Insulin resistance was assessed by using the homeostasis model assessment estimate of insulin resistance (HOMA-IR; fasting insulin [in micro units per millimeter] × fasting glucose [in millimoles per liter]/22.5) [24]. We used the definition of obesity in the Asian-Pacific region (BMI ≥ 25 kg/m2) to categorize participants as obese [25].
Statistical analysis
Participant characteristics were compared according to sarcopenia status using independent-sample Student’s t tests for continuous variables and chi-square tests for categorical variables. Participants were classified into four groups according to vitamin D level and sarcopenia status. Mean BMD values of the four groups were compared by analysis of covariance with adjustment for age and BMI, followed by Bonferroni post hoc analysis. Multivariable logistic regression analysis was used to determine independent predictors for low BMD. Relationships between BMD and clinical and laboratory values were also analyzed. Multivariable linear regression was used to evaluate the relationships between BMD and selected covariates that are considered clinically important risk factors for osteoporosis or osteopenia based on epidemiologic evidence. Continuous variables were expressed as mean ± SD. A P value < 0.05 was considered significant. Statistical analyses were carried out using PASW Statistics Release 18.0.0 (SPSS Inc., Chicago, IL).
Results
Participant characteristics according to sarcopenia status
General characteristics of study participants are presented in Table 1 according to sarcopenia status. We found that mean 25(OH)D concentration was significantly lower in participants with sarcopenia than in those without sarcopenia among both men (19.6 ± 6.7 vs. 22.4 ± 7.6 ng/ml, P < 0.001) and women (17.3 ± 6.5 vs. 19.5 ± 7.5 ng/ml, P < 0.001). In addition, subjects with sarcopenia had significantly higher waist circumference, BMI, and level of insulin resistance (high fasting glucose and HOMA-IR), regardless of gender, and were less likely to exercise regularly than with those without sarcopenia. However, the proportion of women receiving estrogen replacement therapy or with a history of early menopause did not differ according to sarcopenia status. When we use a different definition of sarcopenia (ASM divided by height squared, ASM/Ht2), BMI and waist circumference in sarcopenic subjects were significantly lower than those in non-sarcopenic subjects, which are contrary to the above findings (Supplementary Table 2). This implies that discrepancy in characteristics of sarcopenic subjects is shown depending on the different definition of sarcopenia.
Femoral BMD is markedly decreased in sarcopenic subjects with vitamin D insufficiency
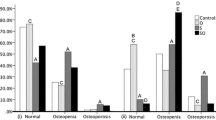
To determine whether vitamin D and sarcopenia exert a combined effect on BMD, subjects were divided into four groups according to 25(OH)D level and sarcopenia status. After adjusting for age and BMI, sarcopenic subjects with vitamin D insufficiency showed significantly lower femoral neck BMD compared with those with sufficient vitamin D levels [25(OH)D ≥ 20 ng/ml] and compared with participants without sarcopenia regardless of vitamin D status among both genders (Fig. 1a). Similarly, total femur BMD in sarcopenic subjects with vitamin D insufficiency was significantly lower than that of other groups, other than sarcopenic female subjects with normal vitamin D levels (Fig. 1b). In contrast, lumbar spine BMD did not differ significantly among the groups (Fig. 1c). Similarly, sarcopenic subjects with vitamin D insufficiency had significantly decreased T-scores at the femoral neck and total femur, but not at the lumbar spine (Supplementary Fig. 1). Femur BMD was also decreased in subjects with sarcopenia and vitamin D insufficiency when ASM/Ht2 as an index of relative skeletal muscle mass was used to define sarcopenia (Supplementary Fig. 2).
Relationships between BMD and clinical and laboratory values
Correlation and multivariable linear regression analyses were performed to evaluate relationships between BMD and clinical and laboratory values, including 25(OH)D and ASM (Tables 2 and 3). As expected, age had the strongest negative correlation with BMD, whereas BMI and ASM values showed strong positive correlations with BMD at each site. However, weak or no correlation was observed between BMD and ASM/Wt according to sarcopenia status and gender. With regard to vitamin D level, only sarcopenic subjects showed positive correlations between 25(OH)D and total femur BMD (men, r = 0.139; women, r = 0.070) and femoral neck BMD (men, r = 0.123; women, r = 0.092). No correlation between 25(OH)D values and BMD was observed at any site in the non-sarcopenic group. In addition, waist circumference, daily calcium intake, and alanine aminotransferase levels correlated positively with BMD at all sites regardless of gender and sarcopenia status. To adjust the effect of age and BMI on BMDs, partial correlation analyses were performed (Supplementary Table 3). Contrary to the findings from simple correlation analyses, correlation between ASM and vitamin D was significant in both men and women (P < 0.001 both) and BMDs at total femur and femur neck area were significantly correlated with ASM and vitamin D levels regardless gender (all P < 0.001). However, only spinal BMD was not significantly related to the levels of vitamin D (P = 0.214 in men, 0.228 in women).
Table 3 shows the results of multivariable linear regression analysis to identify covariates associated with BMD. The conventional risk factors age and BMI were significantly associated with BMD at nearly all sites in both men and women. Moreover, daily calcium intake (high/low tertile) and ASM were also associated with BMD at all sties regardless of gender, and 25(OH)D level was associated with BMD at the femoral neck and total femur, but not lumbar spine. Among postmenopausal women, estrogen replacement and history of early menopause were more closely associated with lumbar spine BMD than femoral bone BMD. Waist circumference was closely associated with BMD at the femoral neck and total femur in men, but not in women.
Independent predictors of low bone mass in elderly subjects
To assess the association between status of BMD and vitamin D insufficiency or sarcopenia, we performed chi-square tests in the elderly population after stratifying by BMI (Supplementary Table 4). Vitamin D insufficiency was significantly associated with osteopenia or osteoporosis in nonobese men and obese women. Sarcopenia was significantly associated with osteopenia or osteoporosis in obese women and all men, regardless of BMI. Then, multivariable logistic regression analysis was used to identify independent predictors of low bone mass of the total femur in elderly individuals (Fig. 2). We included clinically important conventional variables and factors that were significantly associated with BMD in the regression model. After adjusting for regular exercise, smoking status, alcohol consumption, and vitamin supplementation, we found that sarcopenia and low 25(OH)D levels were independent predictors for low bone mass at the total femur in both men and women. Age was closely associated with an increased risk of low bone mass, whereas high BMI and the upper tertile of daily calcium intake were associated with a decreased risk of low bone mass, regardless of gender. Waist circumference and history of fracture were also independent predictors of low bone mass among men, whereas no history of estrogen replacement therapy was associated with low bone mass at the total femur among women.
Multivariable logistic regression analysis to identify variables independently associated with low bone mass of total femur among (a) men and (b) women. Models were adjusted for regular exercise, smoking status, alcohol consumption, and vitamin supplementation (0 = T-score > −1.0; 1 = T-score ≤ −1.0)
Discussion
This study of elderly noninstitutionalized Korean subjects demonstrated that elderly sarcopenic subjects with vitamin D insufficiency had significantly decreased BMD at the total femur and femoral neck regions, but not at the lumbar spine, regardless of gender. Vitamin D insufficiency decreases BMD due to increased bone resorption due to a compensatory increase in parathyroid hormone. Vitamin D plays a role in bone formation through stimulation of osteoblasts, suppression of osteoblast apoptosis, and trans-differentiation of osteoblasts from bone marrow adipocytes [17]. In muscle cells, vitamin D induces de novo synthesis of proteins that regulate cell proliferation and differentiation and also increases the calcium pool essential for muscle contraction. Therefore, vitamin D is intimately involved in maintaining muscle mass and function [17] and bone integrity. Aging induces human skin to lose its ability to generate vitamin D3 from UV radiation and low dietary intake of vitamin D in the elderly can result in vitamin D insufficiency [26]. It could increase fracture risk due to BMD loss and muscle weakness in the aged people [27, 28]. Furthermore, sarcopenia is also known to cause low BMD, due to less mechanical stimulation and the pro-inflammatory cytokines underlying sarcopenia [9, 10].
Several recent epidemiological studies have examined the interaction between sarcopenia, osteoporosis, and vitamin D insufficiency. Our study has confirmed the higher prevalence of osteopenia and osteoporosis in subjects with vitamin D insufficiency, consistent with the findings of the Rancho Bernardo and other studies [29, 30]. We also identified a significant association between sarcopenia and BMD in accord with previous report of a correlation between sarcopenia, assessed by relative skeletal muscle mass index, and BMD in postmenopausal women [31]. Kim et al. found significantly lower 25(OH)D levels in older Korean subjects with sarcopenia, regardless of BMI, suggesting that vitamin D and sarcopenia may be associated [32]. We also found that vitamin D levels were significantly correlated with ASM, which reflects the amount of skeletal muscle mass.
The relationship between regional BMD and body composition (e.g., muscle or fat) in elderly subjects was demonstrated in several studies, but the physiological basis of the relationship is not well understood. The Michigan Bone Health Study reported that decreased BMD at the proximal femur could be attributed to premenopausal muscle loss [33]. Similarly, a 12-year longitudinal study [34] showed that changes in lean body mass contribute to proximal femur BMD, whereas changes in lean or fat body mass are not related to lumbar spine BMD in women. Schoffl et al. reported that lean mass was the strongest predictor of cortical bone mineral content of the femur in postmenopausal women [35]. Consistent with these findings, we observed that ASM was more strongly correlated with femoral BMD than lumbar spine BMD.
With respect to the site-specific effect of vitamin D on BMD, vitamin D level has been more consistently associated with hip BMD than lumbar spinal BMD [36]. Several studies show the influence of extravertebral calcifications on lumbar spinal BMD [37–41]. In general, these calcifications are classified into two categories: vascular calcification, usually arising at the aorta and its major branches, and the osteophytic calcification found in degenerative spinal diseases [41]. These studies demonstrated that degenerative conditions, such as osteophyte formation and bone sclerosis, might erroneously increase measures of spinal BMD. However, aortic calcification had only a minimal effect on spinal BMD, probably due to the relatively low mineral density of vascular deposits [38–41]. Severe degenerative changes in spinal vertebrae are more common in elderly adults; therefore, spinal BMD is likely to be overestimated by DXA in these individuals [37], which may in turn underestimate the relationship between vitamin D level and spinal BMD. Therefore, we should be cautious in interpreting our results. In our view, there are two possible explanations for this correlation: either vitamin D has the true site-specific effect on BMD, or degenerative conditions in spine may introduce errors into estimation of the association between vitamin D and lumbar spinal BMD.
The present study shows that vitamin D insufficiency and sarcopenia are additively associated with low BMD, especially in the femur region. Osteoporotic hip fractures are a critical public health and economic burden and are serious life-threatening injuries for elderly individuals. Furthermore, low femoral neck BMD is generally considered a reliable predictor of future hip fracture [42, 43]. Our findings indicate that femoral BMD is significantly decreased in elderly sarcopenic subjects with vitamin D insufficiency, thus increasing the risk of osteoporotic hip fracture. A meta-analysis of randomized controlled trials reported that both monotherapy with alfacalcidol (active vitamin D) and combination therapy with vitamin D and calcium significantly reduced the incidence of hip fracture but not vertebral fractures in older adults, compared with a placebo [44]. This supports the idea that vitamin D replacement may be more effective in preventing hip fracture than vertebral fracture.
There are several explanations for our results that sarcopenia and vitamin D insufficiency are associated with femoral BMD, but not lumbar spine BMD. These results may be attributed to heterogeneity in the structural and metabolic characteristics of bones. Lumbar vertebrae, which consist primarily of cancellous and trabecular bone, are more affected by estrogen levels than the femur. In contrast, the femur contains more cortical bone, which has a lower metabolic rate than trabecular bone. Lean body mass may protect against bone loss in the femur, which must bear greater mechanical loads and stresses than the axial spine through muscular contraction. Muscle contraction stimulates periosteal apposition, which can also be directly induced by mechanical strain through mechanoreceptors in osteocytes [45, 46]. This may explain why sarcopenic patients with vitamin D insufficiency had lower BMD only in the femur, which is surrounded by strong thigh muscles. Consistent with our findings, a cross-sectional study from Brazil also showed that postmenopausal women with sarcopenia had lower BMD in the femoral neck and total femur, but not in the lumbar spine [47].
The present study has several important strengths. To the best of our knowledge, this is the first study to evaluate the relationship between BMD and sarcopenia status combined with vitamin D insufficiency among elderly adults. In addition, this is a large population-based study, which can increase the statistical reliability of the results. Finally, ethnicity influences not only the effects of anthropometric indices on BMD but also body composition; therefore, it is noteworthy that our study population consists of ethnically homogenous South Korean individuals.
However, our study has some limitations, which should be addressed by further investigation. First, the cross-sectional design did not permit conclusions regarding a causal relationship between BMD and sarcopenia combined with vitamin D insufficiency in elderly adults. Second, the precise mechanism underlying the effects of lean body mass and vitamin D level on bone density of the femur, but not vertebrae, has not yet been elucidated. Third, because standardized diagnostic criteria for sarcopenia have not been established, our results are affected by the definition of sarcopenia used (ASM/Wt or ASM/Ht2).
An important clinical implication of our study is that vitamin D supplementation should be emphasized for the elderly with sarcopenia, to prevent osteoporotic hip fractures. Indeed, recent randomized controlled trials and meta-analysis show that vitamin D supplementation does reduce the risk of fractures and falls in the elderly [48]. We showed that the elderly with sarcopenia have a greater need of vitamin D supplementation. Our study also underscores the importance of prevention of sarcopenia through physical exercise prior to aging because it is difficult to reverse loss of muscle mass in the elderly.
In conclusion, elderly sarcopenic subjects with vitamin D insufficiency exhibited significantly lower femoral BMD than individuals who were sarcopenic or vitamin D insufficient (but not both) in noninstitutionalized South Korean population. These findings suggest a site-specific effect of lean muscle mass and vitamin D level on BMD. The combination of sarcopenia and vitamin D insufficiency may thus accelerate femoral BMD loss in geriatric populations. In light of the worldwide epidemic of vitamin D insufficiency, the importance of vitamin D supplementation and maintaining muscle mass should be emphasized for elderly adults with a high risk of osteoporotic fracture. Prospective studies are needed to confirm that 25(OH)D can prevent femoral bone loss and hip fractures in sarcopenic elderly adults.
References
Rachner TD, Khosla S, Hofbauer LC (2011) Osteoporosis: now and the future. Lancet 377:1276–1287
Choi HS, Oh HJ, Choi H, Choi WH, Kim JG, Kim KM, Kim KJ, Rhee Y, Lim SK (2011) Vitamin D insufficiency in Korea—a greater threat to younger generation: the Korea National Health and Nutrition Examination Survey (KNHANES) 2008. J Clin Endocrinol Metab 96:643–651
McGreevy C, Williams D (2011) New insights about vitamin D and cardiovascular disease: a narrative review. Ann Intern Med 155:820–826
Lim JS, Hwang JS, Lee JA, Kim DH, Park KD, Cheon GJ, Shin CH, Yang SW (2010) Bone mineral density according to age, bone age, and pubertal stages in Korean children and adolescents. J Clin Densitom 13:68–76
Frisoli A, Chaves PH, Ingham SJ, Fried LP (2011) Severe osteopenia and osteoporosis, sarcopenia, and frailty status in community-dwelling older women: results from the Women's Health and Aging Study (WHAS) II. Bone 48:952–957
Pongchaiyakul C, Nguyen ND, Nguyen TV (2004) Development and validation of a new clinical risk index for prediction of osteoporosis in Thai women. Chot Mai Het Thang Phaet 87:910–916
Duan Y, Beck TJ, Wang X, Seeman E (2003) Structural and biomechanical basis of sexual dimorphism in femoral neck fragility has its origins in growth and aging. J Bone Miner Res 18:1766–1774
Pluijm SM, Visser M, Smit JH, Popp-Snijders C, Roos JC, Lips P (2001) Determinants of bone mineral density in older men and women: body composition as mediator. J Bone Miner Res 16:2142–2151
Frost HM (1987) The mechanostat: a proposed pathogenic mechanism of osteoporoses and the bone mass effects of mechanical and nonmechanical agents. Bone Miner 2:73–85
Montagnani A, Gonnelli S, Alessandri M, Nuti R (2011) Osteoporosis and risk of fracture in patients with diabetes: an update. Aging Clin and Exp Res 23:84–90
Kim TN, Park MS, Lim KI, Yang SJ, Yoo HJ, Kang HJ, Song W, Seo JA, Kim SG, Kim NH, Baik SH, Choi DS, Choi KM (2011) Skeletal muscle mass to visceral fat area ratio is associated with metabolic syndrome and arterial stiffness: The Korean Sarcopenic Obesity Study (KSOS). Diabetes Res Clin Pract 93:285–291
Lim S, Kim JH, Yoon JW, Kang SM, Choi SH, Park YJ, Kim KW, Lim JY, Park KS, Jang HC (2010) Sarcopenic obesity: prevalence and association with metabolic syndrome in the Korean Longitudinal Study on Health and Aging (KLoSHA). Diabetes Care 33:1652–1654
Cruz-Jentoft AJ, Landi F, Topinkova E, Michel JP (2010) Understanding sarcopenia as a geriatric syndrome. Curr Opin Clin Nutr Metab Care 13:1–7
Stephen WC, Janssen I (2009) Sarcopenic-obesity and cardiovascular disease risk in the elderly. J Nutr Health Aging 13:460–466
Waugh EJ, Lam MA, Hawker GA, McGowan J, Papaioannou A, Cheung AM, Hodsman AB, Leslie WD, Siminoski K, Jamal SA (2009) Risk factors for low bone mass in healthy 40–60 year old women: a systematic review of the literature. Osteoporos Int 20:1–21
Papaioannou A, Kennedy CC, Cranney A, Hawker G, Brown JP, Kaiser SM, Leslie WD, O'Brien CJ, Sawka AM, Khan A, Siminoski K, Tarulli G, Webster D, McGowan J, Adachi JD (2009) Risk factors for low BMD in healthy men age 50 years or older: a systematic review. Osteoporos Int 20:507–518
Montero-Odasso M, Duque G (2005) Vitamin D in the aging musculoskeletal system: an authentic strength preserving hormone. Mol Aspects Med 26:203–219
Korea Centers for Disease Control and Prevention, Ministry of Health and Welfare, South Korea. Korea National Health and Nutrition Examination Survey (KNHANES). http://knhanes.cdc.go.kr/. Accessed 20 Dec 2012
Lim S, Shin H, Song JH, Kwak SH, Kang SM, Won Yoon J, Choi SH, Cho SI, Park KS, Lee HK, Jang HC, Koh KK (2011) Increasing prevalence of metabolic syndrome in Korea: the Korean National Health and Nutrition Examination Survey for 1998–2007. Diabetes Care 34:1323–1328
Organization WH (1994) WHO Technical Report Series No. 843: assessment of fracture risk and its application to screening for postmenopausal osteoporosis. WHO, Geneva
Janssen I, Heymsfield SB, Ross R (2002) Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc 50:889–896
Lee YH, Bang H, Kim HC, Kim HM, Park SW, Kim DJ (2012) A simple screening score for diabetes for the Korean population: development, validation, and comparison with other scores. Diabetes Care 35:1723–1730
Hwang YC, Ahn HY, Jeong IK, Ahn KJ, Chung HY (2013) Optimal serum concentration of 25-hydroxyvitamin D for bone health in older Korean adults. Calcif Tissue Int 92:68–74
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419
World Health Organization Western Pacific Region IAftSoO (2000) The Asia-Pacific perspective: redefining obesity and its treatment. Health Communications Australia, Sydney, pp 15–21
MacLaughlin J, Holick MF (1985) Aging decreases the capacity of human skin to produce vitamin D3. J Clin Invest 76:1536–1538
Morley JE, Argiles JM, Evans WJ, Bhasin S, Cella D, Deutz NE, Doehner W, Fearon KC, Ferrucci L, Hellerstein MK, Kalantar-Zadeh K, Lochs H, MacDonald N, Mulligan K, Muscaritoli M, Ponikowski P, Posthauer ME, Rossi Fanelli F, Schambelan M, Schols AM, Schuster MW, Anker SD (2010) Nutritional recommendations for the management of sarcopenia. J Am Med Dir Assoc 11:391–396
Bischoff-Ferrari HA, Dawson-Hughes B, Willett WC, Staehelin HB, Bazemore MG, Zee RY, Wong JB (2004) Effect of vitamin D on falls: a meta-analysis. JAMA 291:1999–2006
Saquib N, von Muhlen D, Garland CF, Barrett-Connor E (2006) Serum 25-hydroxyvitamin D, parathyroid hormone, and bone mineral density in men: the Rancho Bernardo study. Osteoporos Int 17:1734–1741
von Muhlen DG, Greendale GA, Garland CF, Wan L, Barrett-Connor E (2005) Vitamin D, parathyroid hormone levels and bone mineral density in community-dwelling older women: the Rancho Bernardo Study. Osteoporos Int 16:1721–1726
Walsh MC, Hunter GR, Livingstone MB (2006) Sarcopenia in premenopausal and postmenopausal women with osteopenia, osteoporosis and normal bone mineral density. Osteoporos Int 17:61–67
Kim MK, Baek KH, Song KH, Il Kang M, Park CY, Lee WY, Oh KW (2011) Vitamin D deficiency is associated with sarcopenia in older Koreans, regardless of obesity: the Fourth Korea National Health and Nutrition Examination Surveys (KNHANES IV) 2009. J Clin Endocrinol Metab 96:3250–3256
Sowers M, Crutchfield M, Bandekar R, Randolph JF, Shapiro B, Schork MA, Jannausch M (1998) Bone mineral density and its change in pre-and perimenopausal white women: the Michigan Bone Health Study. J of Bone and Mineral Res 13:1134–1140
Liu-Ambrose T, Kravetsky L, Bailey D, Sherar L, Mundt C, Baxter-Jones A, Khan KM, McKay HA (2006) Change in lean body mass is a major determinant of change in areal bone mineral density of the proximal femur: a 12-year observational study. Calcif Tissue Int 79:145–151
Schoffl I, Kemmler W, Kladny B, Vonstengel S, Kalender WA, Engelke K (2008) In healthy elderly postmenopausal women variations in BMD and BMC at various skeletal sites are associated with differences in weight and lean body mass rather than by variations in habitual physical activity, strength or VO2max. J Musculoskelet Neuronal Interact 8:363–374
Binkley N (2012) Vitamin D and osteoporosis-related fracture. Arch Biochem Biophys 523:115–122
Muraki S, Yamamoto S, Ishibashi H, Horiuchi T, Hosoi T, Orimo H, Nakamura K (2004) Impact of degenerative spinal diseases on bone mineral density of the lumbar spine in elderly women. Osteoporos Int 15:724–728
von der Recke P, Hansen MA, Overgaard K, Christiansen C (1996) The impact of degenerative conditions in the spine on bone mineral density and fracture risk prediction. Osteoporos Int 6:43–49
Drinka PJ, DeSmet AA, Bauwens SF, Rogot A (1992) The effect of overlying calcification on lumbar bone densitometry. Calcif Tissue Int 50:507–510
Reid IR, Evans MC, Ames R, Wattie DJ (1991) The influence of osteophytes and aortic calcification on spinal mineral density in postmenopausal women. J Clin Endocrinol Metab 72:1372–1374
Orwoll ES, Oviatt SK, Mann T (1990) The impact of osteophytic and vascular calcifications on vertebral mineral density measurements in men. J Clin Endocrinol Metab 70:1202–1207
Cummings SR, Bates D, Black DM (2002) Clinical use of bone densitometry: scientific review. JAMA 288:1889–1897
Cummings SR, Black DM, Nevitt MC, Browner W, Cauley J, Ensrud K, Genant HK, Palermo L, Scott J, Vogt TM (1993) Bone density at various sites for prediction of hip fractures. The Study of Osteoporotic Fractures Research Group. Lancet 341:72–75
Avenell A, Gillespie WJ, Gillespie LD, O'Connell D (2009) Vitamin D and vitamin D analogues for preventing fractures associated with involutional and post-menopausal osteoporosis. Cochrane Database Syst Rev:CD000227
Cui LH, Shin MH, Kweon SS, Park KS, Lee YH, Chung EK, Nam HS, Choi JS (2007) Relative contribution of body composition to bone mineral density at different sites in men and women of South Korea. J Bone Miner Metab 25:165–171
Petit MA, Beck TJ, Lin HM, Bentley C, Legro RS, Lloyd T (2004) Femoral bone structural geometry adapts to mechanical loading and is influenced by sex steroids: the Penn State Young Women’s Health Study. Bone 35:750–759
Genaro PS, Pereira GA, Pinheiro MM, Szejnfeld VL, Martini LA (2010) Influence of body composition on bone mass in postmenopausal osteoporotic women. Arch Gerontol Geriatr 51:295–298
Lips P, Bouillon R, van Schoor NM, Vanderschueren D, Verschueren S, Kuchuk N, Milisen K, Boonen S (2010) Reducing fracture risk with calcium and vitamin D. Clin Endocrinol (Oxf) 73:277–285
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
S.-G. Lee and Y.-h. Lee contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 28.7 KB)
Supplementary Fig. 1
T-score of the (A) femoral neck, (B) total femur, (C) total lumbar spine according to vitamin D level and sarcopenia status (JPEG 43 kb)
High resolution image
(TIFF 636 KB)
Supplementary Fig. 2
Differences in bone mineral density of (A) femoral neck, (B) total femur, and (C) lumbar spine according to vitamin D level and sarcopenia status defined by ASM/Ht2 (JPEG 44 kb)
High resolution image
(TIFF 689 kb)
Rights and permissions
About this article
Cite this article
Lee, SG., Lee, Yh., Kim, K.J. et al. Additive association of vitamin D insufficiency and sarcopenia with low femoral bone mineral density in noninstitutionalized elderly population: the Korea National Health and Nutrition Examination Surveys 2009–2010. Osteoporos Int 24, 2789–2799 (2013). https://doi.org/10.1007/s00198-013-2378-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-013-2378-6