Abstract
Purpose
The purpose of this study was to evaluate the efficacy of platelet-rich plasma (PRP) in reducing femoral and tibial tunnel enlargement in patients operated on for anterior cruciate ligament reconstruction with hamstrings.
Methods
Forty male patients, in which both femoral and tibial 9-mm tunnels were performed because of the graft size, were enrolled in this prospective study. They were randomly assigned to group A (20 patients, PRP group) and group B (20 patients, control group). All patients were followed up at a median of 14.7 months (range 10–16 months), with a physical examination, the Tegner, Lysholm and objective IKDC scoring scales, and with the KT-1000 arthrometer. Moreover, they underwent a CT evaluation in order to assess the amount of tunnel enlargement.
Results
Femoral tunnel diameter increased from 9.0 ± 0.1 mm to 9.8 ± 0.3 mm in group A (p = 0.032) and from 9.0 ± 0.1 mm to 9.4 ± 0.5 mm in group B (p = 0.043). Tibial tunnel diameter increased from 9.0 ± 0.2 mm to 10.9 ± 0.2 mm in group A (p = 0.029) and from 9.1 ± 0.1 mm to 10.1 ± 0.4 mm in group B (p = 0.028). Physical examination as well as the evaluation scales used showed no differences between the two groups.
Conclusions
The use of PRP does not seem to be effective in preventing tunnel enlargement.
Level of evidence
Therapeutic study, Level II.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anterior cruciate ligament (ACL) reconstruction is a widespread surgical procedure for functional instability of the knee. Among the possible complications related to this type of surgery, tibial and femoral bone tunnel widening is one of the most frequently detected and does not seem to be strictly related to the surgical technique performed or the fixation devices used [5, 13, 18, 20, 24, 25, 36, 47].
Despite the fact that this phenomenon tends to occur in the vast majority of patients, it has been shown how it does not seem to affect the long-term clinical results of the operation [2, 4, 8, 11, 17, 19, 44, 47, 53, 54].
Lately, many growth factors [6, 22, 52], such as bone morphogenetic protein-2 (BMP-2) [39], mesenchymal stem cells [45] and platelet-rich plasma (PRP) [3, 12, 14, 15, 23, 41], have been studied with the aim of promoting and accelerating graft-to-bone integration. It is reasonable to assume that a better and faster integration of the graft should reduce bone tunnel phenomenon by exposing the graft for a shorter period of time to mechanical and biological factors.
The aim of this study was to assess the effect of PRP in accelerating the graft-to-bone process and thus in reducing bone tunnel enlargement.
The hypothesis of the study is that the use of PRP would reduce the amount of bone tunnel enlargement.
Materials and methods
Forty patients affected by ACL instability were randomly assigned to group A (20 patients, ACL reconstruction with hamstrings + PRP, study group) and group B (20 patients, ACL reconstruction, control group). All patients were men, with a median age of 34.5 years (range: 18–48 years).
The presence of a chronic instability (more than 30 days from the trauma) was set as inclusion criteria to enter the study. Moreover, in order to gather an homogeneous group of patients, we only included patients who had received 9 mm intra-operative tunnel diameters, which was determined by the size of the hamstrings; furthermore, we decided to include only male patients in order to avoid bias potentially arising from lower bone density that could have been presented in female patients [16, 35, 38]. Exclusion criteria were as follows: an age higher than 50 years old, patients with associated concomitant medial or lateral collateral ligament injuries, and patients with degenerative joint disease or chondral damage detected with pre-operative standard radiographs or magnetic resonance imaging (MRI) examinations.
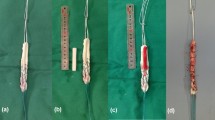
The PRP we used was obtained from the Immunohaematology and Transfusion Department of our hospital with the PRP Fast Biotech kit (MyCells® PPT-Platelet Preparation Tube). It was obtained by isolation and centrifugation of 10 ml of whole blood (taken about 60 min prior to surgery) in order to isolate platelets, which were infiltrated in their liquid form; furthermore, by the addition of thrombin and 10 % Ca-gluconate a few minutes before its use, a thick, adhesive gel was achieved and used as scaffold.
Surgical technique
All operations were performed by the same expert surgeon, with hamstrings, through an Out-In technique [1, 10]: in particular, the graft was fixed with the Swing-Bridge device (Citieffe, Bologna, Italy) on the femoral side and with the Evolgate (Citieffe, Bologna, Italy) on the tibial side; diameter of the Evolgate screw was 9.5 mm. In group B, the surgery was performed in a “standard” way. In group A, before passing the graft through the femoral tunnel, we added 5 ml of PRP between the peripheral part of the graft and the tunnel wall; plus, we put another 5 mL of PRP in its semisolid pattern above the graft before it was pulled down into the femoral tunnel. Similarly, on the tibial side, we added 5 ml of liquid and semisolid PRP before fixing the graft with the metallic screw.
Post-operatively, all patients started weight-bearing with the use of crutches the day after the operation. Moreover, they immediately started performing isometric exercises for muscular strength with the knee locked in a full extension brace. Two weeks after the operation, the brace was removed in all cases, and patients started active range of motion (ROM). Within the first 6 weeks, patients started progressive isotonic and isokinetic exercises. Patients involved in sports activities were allowed to return to practise their sport 6 months after surgery, and patients involved in noncontact sports after 4 months.
Follow-up
Patients were followed up at a median of 14.7 months (range 10–16 months). They were blindly evaluated by an independent orthopaedic surgeon (A.V.): range of motion and Lachman and pivot-shift tests were assessed. Moreover, they were objectively evaluated with Lysholm, Tegner and objective IKDC scoring scales, and with the KT-1000 arthrometer (MEDmetric corp., San Diego, CA, USA).
Radiological evaluation
Tunnel diameters of the operated knee were assessed with a computed tomography (CT) [27] that was performed the day after surgery and at final follow-up: a 16-slice MSCT scanner Philips MX 8000 with post-process multislab reconstruction on sagittal and coronal planes was used for the evaluation. Scanning was performed from a level just above the femoral foramen to a level right below the outer hole of the tibial tunnel. Slice thickness was 1 mm with retrorecons of 0.75 mm.
Measurements were taken at eight different levels, four each for femoral and tibial tunnels, according to what was previously described by Iorio et al. and Vadalà et al. [20, 47]. In particular, image acquisitions were obtained through a volumetric mode: a volume was scanned, and the raw data sets were manipulated afterwards, thus allowing post-process reformation along all the axes (perpendicular, horizontal and oblique).
All diameters were calculated in millimetres. All the measurements were performed by the same expert radiologist in a blinded fashion.
Statistical analysis
The data obtained from the study were analysed using the chi-square test and the Fisher exact test. The power analysis evaluation showed a 90 % power capability to detect differences between the two groups.
Results
No intra- or post-operative complications were detected in any patient of either group.
Clinical Evaluation
In Group A (study group), median Tegner value decreased from 7.7 (range 5–10) before trauma to 6.5 (range 4–10) at follow-up; mean Lysholm score was 55.4 ± 9.4 pre-operatively and 95.6 ± 5.8 after surgical procedure; the mean value of the objective IKDC form at follow-up was 92.4 ± 8.1: specifically, 16 patients (80 %) entered level A and four patients (20 %) level B; Lachman test was negative in all patients (100 %); pivot-shift test was found to be negative (grade 0) in 16 patients (80 %) and positive (grade +1) in 4 patients (20 %); and the mean anterior laxity difference between the involved knee and the contralateral healthy knee dropped from 9.5 ± 2.4 mm to 2.9 ± 1.2 mm at maximum manual handling (Table 1).
In the control group (Group B), the average score of the Tegner scale decreased from a pre-operative value of 7.1 (range 5–9) to 6.9 (range 3–9) at follow-up; the mean value of the Lysholm scale raised from 57.9 ± 5.6 to 94.1 ± 3.3. The mean value of the objective IKDC form at follow-up was 93.9 ± 6.7: sixteen patients (80 %) entered level A and 4 patients (20 %) level B; Lachman test was negative in all patients (100 %); pivot-shift test was judged as negative (grade 0) in 18 patients (90 %) and as positive (grade +1) in two patients (10 %); and the mean anterior laxity difference dropped from 10.1 ± 2.6 mm to 2.8 ± 3.1 mm at maximum manual handling.
No significant differences were detected for any of the parameters assessed between the two groups (Table 1).
Radiological results
Femoral tunnel diameter increased from 9.0 ± 0.1 mm to 9.8 ± 0.3 mm in group A (p = 0.032) and from 9.0 ± 0.1 mm to 9.4 ± 0.5 mm in group B (p = 0.043) (Fig. 1). Tibial tunnel diameter increased from 9.0 ± 0.2 mm to 10.9 ± 0.2 mm in group A (p = 0.029) and from 9.0 ± 0.1 mm to 10.1 ± 0.4 mm in group B (n.s.) (Fig. 2). Even though a statistical difference was always found comparing pre- and post-operative bone tunnel diameters for femoral and tibial tunnels in all patients of both groups, comparison of final values between the two groups never showed statistically significant differences (Table 2).
Discussion
The most important finding of the present study was the inefficacy of the PRP in accelerating the graft-to-bone integration process and consequently in decreasing the tunnel enlargement phenomenon.
It has been widely shown how the ligament autograft maturation process goes through various stages [29, 50, 51] and how growth factors are present in each of these stages [32, 50, 51]. Orrego et al. [36] have shown good results from the use of PRP in accelerating and improving bone-tendon integration. However, the vast majority of such types of studies so far published were conducted in vitro or in animals [28, 30, 31, 33, 37, 49, 52]. The use of growth factors in humans has still not been adequately investigated, and results shown by authors in the last years are discordant [23, 34, 42, 46]. De Vos et al. [7] obtained the same results in pain and activity improvement compared with a saline injection in the treatment of Achilles tendinopathy, while Kon et al. [19], in treating patellar disorders, showed marked improvement in knee function and quality of life, stating also that PRP has to be associated with physiotherapy in order to obtain better outcomes.
ACL reconstruction is obviously one of the orthopaedic surgical fields in which the use of any type of growth factors would be useful, since the integration process of the reconstructed ACL is one of the keys for a successful long-term follow-up outcome. In this paper, we aimed to assess whether the use of PRP would have accelerated the bone-tendon integration process by evaluating the bone tunnel enlargement that is strictly related to the integration process itself. Indeed, both mechanical and biological factors [2, 8, 17, 19, 43, 44, 47, 53, 54], which are thought to work in sync in determining the widening phenomenon, play a role in the mismatch between the peripheral part of the tendon and the tunnel wall. Indeed, both the bungee [21] and the windshield wiper phenomena [9] (among mechanical factors) and the synovial bathing effect [48] (among biological factors) take place until this area fills up with Sharpey’s fibres and the ACL is fully incorporated within the bone tunnels.
As a consequence, a faster and better integration process should lead to a lower amount of femoral and tibial enlargement, because of the minor time in which these factors might have their effects. We hypothesized that a faster integration process due to the use of PRP would have significantly reduced the bungee and windshield wiper effects, thus avoiding a significant tunnel enlargement phenomenon.
Previous studies [5, 20, 24, 40] have already reported how different fixation devices might influence the amount of tunnel enlargement, probably because of the different capabilities of such constructs to minimize mechanical and biological factors. Similarly, Vadalà et al. [48] showed how different rehabilitation protocols significantly influence the tunnel widening process.
As a matter of fact, results of our study do not support the use of PRP in reducing bone tunnel enlargement: indeed, comparison of radiological data of the two groups shows similar values of tunnel widening both on the femoral (9.8 mm in group A vs. 9.4 mm in group B; n.s.) and on the tibial side (10.9 mm in group A vs. 10.1 mm in group B; n.s.). Similarly, the clinical examination and the scoring scales used showed no differences either.
Limitations of this study are certainly the small size of the groups and the lack of a histological analysis. Regarding the small size of the groups, we tried to gather the most homogeneous types of patients possible, in order to avoid bias due to associated pathologies. In the same way, a histological analysis would have provided more scientific data of how PRP really would act in the mismatch between the graft and bone tunnel [26]. Moreover, another important limitation of this study is the limited power of the study due to the small size of the groups considered.
As a matter of fact, the use of PRP did not provide any effect in reducing the amount of tunnel enlargement. The initial hypothesis that PRP would have avoided this phenomenon was not confirmed.
Conclusion
The use of PRP does not seem to be effective in preventing, or even reducing, femoral and tibial tunnel enlargement in patients operated on for ACL reconstruction with hamstrings.
References
Brown CH, Ferretti A, Conteduca F et al (2001) Biomechanics of the Swing-Bridge technique for anterior cruciate ligament reconstruction. Eur J Sports Traum Rel Res 23(2):69–73
Cameron M, Buchgraber A, Passler H (1997) The natural history of the anterior cruciate ligament-deficient knee: changes in synovial fluid cytokine and keratan sulfate concentrations. Am J Sports Med 25:751–754
Cervellin M, de Girolamo L, Bait C et al (2012) Autologous platelet-rich plasma gel to reduce donor-site morbidity after patellar tendon graft harvesting for anterior cruciate ligament reconstruction: a randomized, controlled clinical study. Knee Surg Sports Traumatol Arthrosc 20(1):114–120
Choi NH, Lee JH, Son KM et al (2010) Tibial tunnel widening after anterior cruciate ligament reconstructions with hamstring tendons using Rigidfix femoral fixation and Intrafix tibial fixation. Knee Surg Sports Traumatol Arthrosc 18(1):92–97
Clatworthy MG, Bartelett J, Howell S et al (1999) The effect of graft fixation techniques on tunnel widening in hamstring ACL reconstruction. Arthroscopy 15(Suppl):5
Darabos N, Haspl M, Moser C et al (2011) Intraarticular application of autologous conditioned serum (ACS) reduces bone tunnel widening after ACL reconstructive surgery in a randomized controller trial. Knee Surg Sports Traumatol Arthrosc 19(Suppl 1):S36–S46
De Vos RJ, Weir A, van Schie HT et al (2010) Platelet-rich plasma injection for chronic Achilles tendinopathy: a randomized controller trial. JAMA 13(303):144–149
Fahey M, Indelicato PA (1994) Bone tunnel enlargement after anterior cruciate ligament replacement. Am J Sports Med 22(3):410–414
Fauno P, Kaalund S (2005) Tunnel widening after hamstring anterior cruciate ligament reconstruction is influenced by the type of graft fixation used: a prospective randomized study. Arthroscopy 21(11):1337–1341
Ferretti A, Conteduca F, Morelli F et al (2003) The evolgate, a method to improve the pull-out strength of interference screw in Tibial Fixation of ACL reconstruction with DGST. Arthroscopy 19(9):936–940
Fink C, Zapp M, Benedetto KP et al (2001) Tibial tunnel enlargement following anterior cruciate ligament reconstruction with patellar tendon autograft. Arthroscopy 17(2):138–143
Floryan K, Berghoff WJ (2004) Intraoperative use of autologous platelet rich and platelet poor plasma for orthopaedic surgery patients. AORN J 80:668–674
Foldager C, Jakobsen BW, Lund B et al (2010) Tibial tunnel widening after bioresorbable poly-lactide calcium carbonate interference screw usage in ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 18(1):79–84
Foster TE, Puskas BL, Mandelbaum BR, Gerhart MB et al (2009) Platelet-rich plasma: from basic science to clinical applications. Am J Sports Med 37:2259–2272
Frechette JP, Martineau I, Gagnon G (2005) Platelet-rich plasmas: growth factor content and roles in wound healing. J Dent Res 84:434–439
Gobbi A, Domzalski M, Pascual J (2004) Comparison of anterior cruciate ligament reconstruction in male and female athletes using the patellar tendon and hamstring autografts. Knee Surg Sports Traumatol Arthrosc 12:534–539
Hantes ME, Mastrokalos DS, Yu J et al (2004) The effect of early motion on tibial tunnel widening after anterior cruciate ligament replacement using hamstring tendon grafts. Arthroscopy 20(6):572–580
Hersekli MA, Akpinar S, Ozalay M et al (2004) Tunnel enlargement after arthroscopic anterior cruciate ligament reconstruction: comparison of bone-patellar tendon-bone and hamstring autografts. Adv Ther 21(2):123–131
Hogervorst T, Van der Hart CP, Pels Rijcken TH et al (2000) Abnormal bone scans of the tibial tunnel 2 years after patella ligament anterior cruciate ligament reconstruction: correlation with tunnel enlargement and tibial graft length. Knee Surg Sports Traumatol Arthrosc 8(6):322–328
Iorio R, Vadalà A, Argento G et al (2007) Bone tunnel enlargement after ACL reconstruction using autologous hamstring tendons: a CT study. Int Orthop 31:49–55
Jagodzinski M, Foerstemann T, Mall G et al (2005) Analysis of forces of ACL reconstructions at the tunnel entrance: is tunnel enlargement a biomechanical problem? J Biomech 38:23–31
Ju YJ, Muneta T, Yoshimura H et al (2008) Synovial mesenchymal stem cells accelerate early remodelling of tendon-bone healing. Cell Tissue Res 332(3):469–478
Kon E, Filardo G, Di Martino A et al (2011) Platelet-rich plasma (PRP) to treat sports injuries: evidence to support its use. Knee Surg Sports Traumatol Arthrosc 19(4):516–527
L’Insalata JC, Klatt B, Fu FH et al (1997) Tunnel expansion following anterior cruciate ligament reconstruction: a comparison of hamstring and patellar tendon autografts. Knee Surg Sports Traumatol Arthrosc 5(4):234–238
Lind M, Feller J, Webster KE (2009) Bone tunnel widening after anterior cruciate ligament reconstruction using EndoButton or EndoButton continuous loop. Arthroscopy 25(11):1275–1280
Lyras DN, Kazakos K, Georgiadis G et al (2011) Does a single application of PRP alter the expression of IGF-I in the early phase of tendon healing? J Foot Ankle Surg 50:276–282
Marchant MH Jr, Willimon SC, Vinson E et al (2010) Comparison of plain radiography, computed tomography, and magnetic resonance imaging in the evaluation of bone tunnel widening after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 18(8):1059–1064
Martinek V, Latterman C, Usas A et al (2002) Enhancement of tendon-bone integration of anterior cruciate ligament grafts with bone morphogenetic protein-2 gene transfer: a histological and biomechanical study. J Bone Joint Surg Am 84:1123–1131
Marumo K, Saito M, Yamagishi T et al (2005) The “ligamentization” process in human anterior cruciate ligament reconstruction with autogenous patellar and hamstring tendons: a biochemical study. Am J Sports Med 33:1166–1173
Meller R, Neddermann A, Willbold E et al (2010) The relation between tunnel widening and bone mineral density after anterior cruciate ligament reconstruction: an experimental study in sheep. Arthroscopy 26(4):481–487
Mihelic R, Pecina M, Jelic M et al (2004) Bone morphogenetic protein-7 (osteogenic protein-1) promotes tendon graft integration in anterior cruciate ligament reconstruction in sheep. Am J Sports Med 32:1619–1625
Molloy T, Wang Y, Murrell AC (2003) The roles of growth factors in tendon and ligament healing. Sports Med 33:381–394
Murray MM, Spindler KP, Devin C et al (2006) Use of a collagen platelet rich plasma scaffold to stimulate healing of a central defect in the canine ACL. J Orthop Res 24:820–830
Nin JR, Gasque GM, Azcárate AV et al (2009) Has platelet-rich plasma any role in anterior cruciate ligament allograft healing? Arthroscopy 25(11):1206–1213
Noojin FK, Barrett GR, Hartzog CW et al (2000) Clinical comparison of intraarticular anterior cruciate ligament reconstruction using autogenous semitendinosus and gracilis tendons in men versus women. Am J Sports Med 28(6):783–789
Orrego M, Larrain C, Rosales J et al (2008) Effects of platelet concentrate and a bone plug on the healing of hamstring tendons in a bone tunnel. Arthroscopy 24(12):1373–1380
Petrigliano FA, McAllister DR, Wu BM (2006) Tissue engineering for anterior cruciate ligament reconstruction: a review of current strategies. Arthroscopy 22:441–451
Pinczewski LA, Deehan DJ, Salmon LJ et al (2002) A five-year comparison of patellar tendon versus four-strand hamstring tendon autograft for arthroscopic reconstruction of the anterior cruciate ligament. Am J Sports Med 30(4):523–536
Rodeo SA, Suzuki K, Deng XH et al (1999) Use of recombinant human bone morphogenetic protein-2 to enhance tendon healing in a bone tunnel. Am J Sports Med 27(4):476–488
Sabat D, Kundu K, Arora S et al (2011) Tunnel widening after anterior cruciate ligament reconstruction: a prospective randomized computed tomography-based study comparing 2 different femoral fixation methods for hamstring graft. Arthroscopy 27(6):776–783
Sanchez AR, Sheridan PJ, Kupp LI (2003) Is platelet-rich plasma the perfect enhancement factor? A current review. Int J Oral Maxillofac Implants 18:93–103
Silva A, Sampaio R (2009) Anatomic ACL reconstruction: does the platelet-rich plasma accelerate tendon healing? Knee Surg Sports Traumatol Arthrosc 17(6):676–682
Silva A, Sampaio R, Pinto E (2010) Femoral tunnel enlargement after anatomic ACL reconstruction: a biological problem? Knee Surg Sports Traumatol Arthrosc 18(9):1189–1194
Simonian PT, Erickson MS, Larson RV et al (2000) Tunnel expansion after hamstring anterior cruciate ligament reconstruction with 1-incision endobutton femoral fixation. Arthroscopy 16(7):707–714
Soon MY, Hassan A, Hui JH et al (2007) An analysis of soft tissue allograft anterior cruciate ligament reconstruction in a rabbit model: a short-term study of the use of mesemchymas stem cells to enhance tendon osteointegration. Am J Sports Med 35(6):962–971
Taylor DW, Petrera M, Hendry M et al (2011) A systematic review of the use of platelet-rich plasma in sports medicine as a new treatment for tendon and ligament injuries. Clin J Sport Med 21:344–352
Vadalà A, Iorio R, De Carli A et al (2006) The effect of accelerated, brace free, rehabilitation on bone tunnel enlargement after ACL reconstruction using hamstring tendons: a CT study. Knee Surg Sports Traumatol Arthrosc 15(4):365–371
Webster KE, Feller JA, Hameister KA (2001) Bone tunnel enlargement following anterior cruciate ligament reconstruction: a randomised comparison of hamstring and patellar tendon grafts with 2-year follow-up. Knee Surg Sports Traumatol Arthrosc 9(2):86–91
Weiler A, Forster C, Hunt P et al (2004) The influence of locally applied platelet-derived growth factor-BB on free tendon graft remodelling after anterior cruciate ligament reconstruction. Am J Sports Med 32:881–891
Weiler A, Hoffmann RF, Bail HJ et al (2002) Tendon healing in a bone tunnel. Part II: Histologic analysis after biodegradable interference fit fixation in a model of anterior cruciate ligament reconstruction in sheep. Arthroscopy 18:124–135
Weiler A, Peine R, Pashmineh-Azar A et al (2002) Tendon healing in a bone tunnel. Part I: Biomechanical results after biodegradable interference fit fixation in a model of anterior cruciate ligament reconstruction in sheep. Arthroscopy 18:113–123
Yamazaki S, Yasuda K, Tomita F et al (2005) The effect of transforming growth factor-beta1 on intraosseous healing of flexor tendon autograft replacement of anterior cruciate ligament in dogs. Arthroscopy 21(9):1034–1041
Yu JK, Paessler HH (2005) Relationship between tunnel widening and different rehabilitation procedure after cruciate ligament reconstruction with quadrupled hamstring tendons. Chin Med J (Engl) 118(4):320–326
Zysk SP, Fraunberger P, Veihelmann A (2004) Tunnel enlargement and changes in synovial fluid cytokine profile following anterior cruciate ligament reconstruction with patellar tendon and hamstring tendon autografts. Knee Surg Sports Traumatol Arthrosc 12(2):98–103
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vadalà, A., Iorio, R., De Carli, A. et al. Platelet-rich plasma: does it help reduce tunnel widening after ACL reconstruction?. Knee Surg Sports Traumatol Arthrosc 21, 824–829 (2013). https://doi.org/10.1007/s00167-012-1980-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-012-1980-z