Abstract
Purpose
Pro-inflammatory cytokines play a pivotal role in osteoarthritis, as well as in bone tunnel widening after ACL reconstructive surgery. A new treatment option is to administer autologous conditioned serum (ACS) containing endogenous anti-inflammatory cytokines including IL-1Ra and growth factors (IGF-1, PDGF, and TGF-β1, among others) in the liquid blood phase. The purpose of this trial was to establish whether the postoperative outcome could be affected by intraarticular application of ACS.
Methods
In a prospective, randomized, double-blinded, placebo-controlled trial with two parallel groups, 62 patients were treated. Bone tunnel width was measured by CT scans, while clinical efficacy was assessed by patient-administered outcome instruments (WOMAC, IKDC 2000) up to 1 year following the ACL reconstruction in patients receiving either ACS (Group A) or placebo (Group B). We compared the levels and dynamics of IL-1β concentrations in the synovial liquid and examined the correlation between the levels of IL-1β at three different postoperative points.
Results
Bone tunnel enlargement was significantly less (6 months: 8%, 12 months: 13%) in Group A than in Group B (6 months: 31%, 12 months: 38%). Clinical outcomes (WOMAC, IKDC 2000) were consistently better in patients treated with ACS at all data points and for all outcome parameters, and there were statistically significant differences in the WOMAC stiffness subscale after 1 year. The decrease in IL-1β synovial fluid concentration was more pronounced in the ACS group, and values were lower, to a statistically significant degree, in the ACS group at day 10.
Conclusion
The intraarticular administration/injection of ACS results in decreased bone tunnel widening after ACL reconstructive surgery.
Level of evidence
Therapeutic study, Randomized controlled trial (significant differences and narrow confidence intervals), Level I.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to available literature, anterior cruciate ligament (ACL) reconstruction can be unsuccessful in up to 25% of cases, due to a variety of causes [31, 62]. Postoperative widening of the tibial and the femoral tunnel, visible on radiological images, is one of the causes that has been identified. The impact of tunnel widening is still unclear, since tunnel widening results in greater knee laxity, but contrariwise is frequently observed in patients that are doing well, objectively and subjectively.
Tunnel widening can potentially complicate an ACL revision procedure [43, 70, 77]. Revision surgery involves staged reconstruction and additional surgical procedures and entails significant costs. Wide tunnels also alter the direction of the graft within a joint. Not only does this require a second surgery, but the potential for additional giving-way episodes further risks injury to the menisci and articular cartilage.
Bone tunnel widening occurs over time, not immediately after surgery [9]. Several authors [9, 24, 55, 66] found that tunnel widening occurred early and stabilized within a year. Others have observed tunnel widening to progress up to 2 years after surgery [24, 38, 56, 57, 73, 74].
The exact etiology of tunnel widening is unknown. The most prominent factor that contributes to enlargement is the choice of graft. It is more commonly found when pure tendon grafts are used, compared with bone patellar tendon autografts [8, 13, 14, 19, 32, 35, 42, 57, 65, 74, 76, 81]. However, a few surgeons have also reported contrary results [14].
A number of biological and mechanical etiologies have been suggested as the cause of tunnel widening. The postulated mechanical factors include local stress deprivation of the tunnel wall (cell necrosis caused by drilling and graft remodeling) [6], the windshield-wiper effect and the bungee cord effect [19, 38], graft-tunnel motion [6], aggressive rehabilitation [7, 20, 67, 81], graft fixation[4, 12, 19, 25, 39, 41, 48–50, 59, 60], and increased graft forces due to improper graft placement [8, 27, 31–33, 38, 63].
There are many potential biological explanations. Graft swelling, the use of allograft tissue (a cytokine-mediated nonspecific inflammatory response), synovial fluid propagation within bony tunnels (“synovial bathing effect”) [10, 14], and increased cytokine levels creating a hostile environment within the joint (as a result of toxic products including ethylene, oxide, and metal) are biological modes that could induce osteolysis and tunnel widening [15, 24, 33, 34, 36, 37, 52, 82].
After ligament injuries or ruptures, large increases in the pro-inflammatory cytokines occur acutely [15, 26, 82]. Elevated IL-1β and TNF-alpha were detected, supporting the hypothesis that theses cytokines were related to bone resorption induced by the biological activity of cytokines and depletion of growth factors, i.e., osteoclast activation and subsequent osteolysis. [16, 18, 29, 36, 40, 52, 68, 75]. Associated injuries such as bone bruises and meniscal tears may affect the cytokine profile. Of the cytokines identified as playing a interleukin-1 (IL-1) appears to be of pivotal importance [15, 33, 36]. Although much remains to be discovered, it is known that IL-1β inhibits the synthesis of the matrix and also promotes its degeneration, whereas certain cytokines and growth factors, especially insulin-like growth factor-1 (IGF-1), transforming growth factor-β (TGF-β), and several bone morphogenic proteins (BMPs), promote matrix and bone synthesis [16, 17, 29, 36, 40, 68, 69, 75]. Moreover, IL-1Ra apparently becomes depleted over time and tends to be undetectable in chronic ACL situations. Consequently, the cartilage-protective function of this cytokine is lost [82]. In animal models of arthritis and inflammation, as well as in clinical trials, blockage of IL-1β by IL-1Ra can slow the progression of disease [2, 3, 11, 21, 22, 30, 45, 54, 80] .Thus, the inhibition of IL-1β may be an important strategy for decreasing tissue degeneration and destruction [36, 45].
A new treatment alternative that draws on knowledge of the role played by cytokines and growth factors is autologous conditioned serum (ACS) that is produced from the patient’s own blood [47, 75]. This novel, biologically based therapeutic agent was suggested from the observations of Meijer et al. [47], who found that exposure of blood to pyrogen-free surfaces selectively elicited a vigorous, rapid increase in the synthesis of anti-inflammatory cytokines, specifically IL-1Ra, fibroblastic growth factor-1 (FGF-1), hepatocyte growth factor (HGF), and transforming growth factor-β1 (TGF-β1), resulting in higher concentrations of those cytokines and factors in human blood serum. For the ACS production, venous blood is drawn into syringes containing pre-treated glass beads (Orthokine EOT Syringe, Orthogen, Duesseldorf, Germany). When the blood is incubated for 6–9 h, the increase in IL-1Ra is comparable to that observed in cultures of purified monocytes exposed to immobilized IgG. There is no noticeable induction of IL-1β or TNF-α. Extensive cell death does not occur, hemolysis is mild and not clinically remarkable, and the major serum protein composition is not affected. After incubation, the blood is centrifuged and the serum is extracted. The resulting product, which is known as autologous conditioned cell-free serum (ACS), is then portioned and either stored until needed or injected into the affected region of the patient.
The specific aim of the present randomized controlled trial (RCT) was to use intraarticular ACS injections as a way to reduce the IL-1β influence on the process of ACL healing. The study objectives were (1) to measure and compare the width of the tibial bone tunnel, at day 1, and 6 and 12 months after ACL reconstruction in patients treated with ACS or placebo injections (Bone tunnel widening); (2) to measure and compare the levels and dynamics of IL-1β concentrations in the synovial fluid at three different postoperative time points and to determine whether the IL-1β-dependent catabolic effects will decrease after postoperative intraarticular injection of ACS with the accompanying increased levels of IL-1Ra (IL-1β effects); (3) to demonstrate that patients’ state of health has improved from baseline assessment and to evaluate differences in the two treatment groups concerning clinical outcome (Effectiveness and patients’ state of health); (4) to assess the nature and duration of any serious and/or minor adverse events (AE) subsequent to ACS injections (Treatment safety).
Materials and methods
In a prospective, randomized, double-blinded, placebo-controlled trial with two parallel groups, 62 patients were treated. This trial was conducted over a period of 27 months; the first patient was enrolled in 2006, and the last patient completed follow-up in 2008. Each patient was fully informed about the purpose of the trial, expected benefits, possible risks, and all the other details pertaining to the study. All participants signed an informed consent. Approval for this study was obtained from the Ethical Board, School of Medicine, University of Zagreb, prior to performing the study.
Eligible patients were older than 18 years. Prior to each surgery, traumatic ACL rupture was determined on the basis of a clinical examination and confirmed by exploratory and therapeutic arthroscopic surgery in accordance with good surgical practice to confirm the ACL tear and address any meniscal or chondral injury. The present study included patients with the same preoperative diagnosis of isolated ACL rupture, a knee axis deviation up to 5° and knee chondral lesions up to grade 2 according to the Outerbridge classification.
Exclusion criteria were poor general health as judged by the orthopedic surgeon; concomitant painful or disabling disease of the spine, hips, or lower limbs that would interfere with the evaluation of the afflicted knee; any inflammatory and infectious arthropathies; alcohol/drug abuse and morbid obesity.
Surgical intervention
The data used in this study are from the study diaries, according to the patient’s operative and postoperative protocols. The same surgeon operated (ND) on all patients during a period of 6 months after the injury. The subjects underwent surgical reconstruction of a traumatic ACL rupture using autograft of the semitendinosus and gracilis tendons (HS), as well as of the patellar ligament (BTB). An endoscopic technique was used in all cases. Numerous anatomical, radiographic, cadaveric as well as clinical studies on the performed surgical procedures are available in literature. Thirty-two patients were randomized to receive BTB and 30 patients to receive HS reconstruction.
The drill diameter used for the tibial tunnel averaged 8.6 ± 0.7 mm (range 7–10 mm). At femoral site, 27 patients were fixed with the use of Bio Trans Fix (Arthrex, US) and 35 with Rigid Fix (Mytek, US). In all cases, at the tibial site, an interferent screw was used. In the present trial, procedures were equal in all patients.
Postoperative treatment protocol
Participants who met all inclusion and exclusion criteria were randomized to either the ACS or the saline group. A computer-generated randomization list was produced by a trial coordinator at the external blood-processing site, who allotted the participants either to the ACS or the saline group in a sequential manner according to the randomization code but was not otherwise involved in the trial. A 60 ml sample of whole blood was taken from each participant using the special syringe containing pre-treated glass beads. Processing including centrifugation, microfiltration (0.2 μm, Millipore), and aliquoting into four 2-ml portions was performed in accordance with the manufacturers’ recommendations. The ACS production as well as the reproducible elevation of biologic factors within the serum is described in detail in references [47, 75].
The aliquots were frozen at −20°C. Samples from patients who had been randomized to the saline group were discarded. Throughout the study, the participants and their observing (interviewing) doctors were blinded to the treatment to which the patients were assigned. All procedures were identical for both the ACS and the saline injections. Because the ACS and the saline solutions are different colors, blinding of the administering doctor was not possible. Therefore, the blind observer technique was chosen to obtain double-blind conditions.
Subsequently, an injection regime of 4 injections was started in a rigid schedule comprising 11 days: injections were given on day 0 (day of surgery), day 1, day 6, and day 10. The synovial fluids present were aspirated to minimize drug dilution and for ELISA testing. The needles were left in place and either 2 ml of ACS or 2 ml of physiological saline was injected through a 0.20-mm-pore-size anti-bacterial sterile filter.
Demographic and baseline disease characteristics
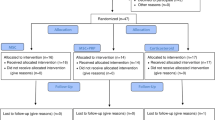
A total of 112 patients were screened for possible inclusion in the study. Fifty were excluded because they did not meet the inclusion/exclusion criteria or withdrew voluntarily after receiving written and oral information (Fig. 1).
Sixty-two patients met the inclusion criteria and did not have exclusion criteria and were randomized to receive ACS (n = 31) or placebo (n = 31). No patient was completely lost to follow-up. During the course of the study, we were not able to collect complete data sets from 6 patients from the ACS group and 9 patients from placebo group at different time points (Fig. 1). The data sets for the participants were completed using the “last observation carried forward” (LOCF) method.
Baseline values of both treatment groups were comparable. No significant differences between the treatment groups were found with respect to age (P = 0.12), gender, weight, or body mass index (BMI) before and during the study period (Table 1).
CT scans: bone tunnel widening
Tunnel widening was determined using standardized CT scans [46]. The CT was chosen for evaluating the tunnel diameter because it gives a true measurement, especially in the tunnel of patients who undergo BTB autografts [23, 32, 73]. The measurements of tibial tunnel width at a level of 1 cm below articular surface were taken on postoperative day 1, and 6 and 12 months after operative treatment by means of CT scans and assessment of the scans by two independent experts (intra- and inter-observer blinded).
Intraarticular IL-1 concentration
The intraarticular concentration of IL-1β was measured on postoperative day 1, day 6, and day 10. Measurements of IL-1β in serum and synovial fluid were performed by an independent expert using a Human IL-1β/IL-1F2 Quantikine ELISA Kit (Minneapolis, MN, USA).
Effectiveness, safety, and patients’ state of health
To evaluate the clinical efficacy and the possible correlation between the tunnel widening at follow-up and the clinical outcome, well-known patient-administered outcome instruments were used (Western Ontario and McMaster Universities Osteoarthritis Index [WOMAC]) and International Knee Documentation Committee Score [IKDC 2000]). At 6 and 12 months after surgery, the patients completed the same questionnaires as at baseline. At these follow-up visits, the treating orthopedic surgeon performed a physical examination of the knee, completed the surgeon part of the IKDC 2000, and checked for adverse events and changes in analgesic use.
Statistical analysis
Statistical analysis was done using SPSS for Windows on a personal computer. At baseline, homogeneity of demographic and disease characteristics was tested with an ANOVA one-way method for continued parameter and a Chi-square test for categorical parameter. A general linear model for repeated measurement was used to compare the longitudinal profile of the groups and a multiple comparison between the groups was performed. The changes from baseline and comparisons of groups for all scores were calculated using an ANOVA one-way test. Although randomization was stratified for graft types, sub-analysis was performed for these groups. Patients who were considered treatment failures were excluded from further follow-up. The data sets for the participants considered treatment failures were completed using the “last observation carried forward” method.
Results are presented graphically, using the mean profile per group and analytically reporting the P values for each contrast. P values less than 0.05 were considered statistically significant.
Results
CT scans: bone tunnel widening
Sixty-one of sixty-two tunnels had enlarged compared with the diameter of the drill in the 12 month post-op, regardless of trial medication, type of fixation, and irrespective of graft type. Statistically significant differences were found in the enlargement between surgery and 6 and 12-month post-op (Fig. 2a–c, Table 2).
Irrespective of treatment modality (ACS or placebo), tunnel widening was marked in the HS group. In the HS group, the mean increase in the tibial tunnel area was 33.2% (range 6–105%) after 6 months and 41.7% (range 8–108%) after 12 months. In the BTB group, it was only 11.1% (range −2–48%) after 6 months and 17.1% (range 3–69%) after 12 months.
The effect of ACS and placebo treatment over time on the bone tunnel widening is presented in Fig. 3. With respect to the treatment groups (ACS vs. placebo), the magnitude of tunnel enlargement was statistically significant higher in patient knees treated with placebo/saline at 12 months in the BTB group and at 6 and 12 months in the HS group (each P < 0.05) (Table 2). Note that the tunnel widening in the ACS group is only half that in the placebo group after 6 and 12 months.
Intraarticular IL-1 concentration
In the overall study population, synovial fluid (SF) concentrations postoperatively at day 1 were significantly higher than normal values (Fig. 4). Subsequently, the IL-1β SF concentration dropped off significantly in both groups and reached approximately normal values by day 6 (in both groups each P at day 6 was below 0.05 for the comparison with day 1 values). Beyond day 6, the ACS and placebo groups reacted differently. In the ACS group, the values continued to decline until day 10 (ACS day 10: mean 3.4 pg/l), whereas in the placebo group the IL-1β concentrations again tended to increase till day 10 (placebo day 10: mean 16.0 pg/l). The difference at day 10 was statistically significant (P = 0.038 for the comparison ACS vs. placebo). The IL-1β concentrations found in the ACS group at day 10 were a little lower than normal values given in the literature.
However, nearly all patients had higher postoperative IL-1β synovial fluid concentrations (except ACS day 10), and there was no significant correlation between bone tunnel widening and postoperative IL-1β concentration (P > 0.05). Subsequent stratification of the results revealed no correlation between IL-1β SF concentrations and graft type used (HS vs. BTB).
Patients’ assessment of treatment efficacy
Both ACS-treated and placebo-treated patients showed a significant improvement on all outcome measures (P < 0.001) according to WOMAC and IKDC 2000, compared with baseline values. ACS-treated patients scored consistently better with the lowest pain scores and the largest reduction (Table 3) than did the placebo-treated patients.
As far as improvement over time is concerned, ACS resulted in significantly more improvement in WOMAC stiffness scores (P = 0.047, 12 months) compared with placebo treatment. Comparable improvements were found for ACS and placebo treatments in terms of the WOMAC global index (71% vs. 61% at 6 months, and 89% vs 79% after 12 months; n.s.).
Clinical examination
Baseline disease characteristics were comparable in the two groups according to the IKDC 2000 clinical examination and surgeon’s part. When the participants were stratified by groups after 6 months, there were significantly lower effusion values (assessed by ballotting the knee) in the ACS group. In the ACS group, 70% had no effusion and 30% had mild effusion, whereas in the placebo group only 35% had no effusion and 64% had mild effusion. Significantly more patients in the ACS group had better results on functional tests, achieving a ratio of >90% of one-leg hops with their index knee, compared with their normal knee. Other measurements were very similar (e.g., no difference in laxity, about 60% in ACS group vs. 65% in the placebo group were rated as tight).
Overall, after 12 months, there was a further improvement in both groups, but with diminished differences between groups. There was no longer any significant difference in effusion (e.g., 80% of patients with no effusion in ACS group vs. 76% in the placebo group) or in functional one-leg hops, and still no difference in laxity. About 62% of ACS patients and 69% of placebo patients were rated as tight.
The range of motion (ROM) was significantly better in the ACS group. About 97% of patients in this group had full range of motion. In the placebo group, 2 patients had a lack of extension (3–5°) and 4 had a lack of flexion (6–15°), whereas only 1 patient from the ACS group had a lack of flexion (6–15°). Additionally, we found less harvest site pathologies (tenderness, irritation, or numbness at the autograft harvest site) in the ACS group. Seventy-six percentage had no such problems, 21% had mild problems, and only 3.4 had moderate problems. In contrast, in the placebo group, only 58% had no problems at the harvest site, 23% had mild, and 19% had moderate problems.
Subsequent stratification of the patient-administered outcome parameters as well as the clinical examination result revealed no correlation between tunnel widening and clinical outcome. Interestingly, sub-analysis for graft type did not show statistically significant differences in clinical outcomes between the subgroups, as measured by WOMAC or IKDC. In addition, correlation analysis did not show significant relations between baseline patient characteristics (age, gender, and symptom severity) and treatment outcome.
Safety: adverse events
In general, all intraarticular injections were well tolerated: adverse events were only mild to moderate and most consisted of a subjective pain or pressure sensation directly after the IA injections and revealed within a few hours without secondary damage. There was no statistically significant difference in the prevalence of adverse events between the two groups as a very good risk/benefit ratio and excellent safety record has already been demonstrated for ACS in other clinical trials. There were no infections or severe adverse events related to the study procedure or medication in this trial.
Discussion
The most important finding of the present randomized controlled trial was that a standardized postoperative treatment regimen with intraarticular administration of ACS was able to influence outcome parameters different from placebo treatment. The statistically significant reduction in bone tunnel enlargement and the statistically significant improvement of WOMAC stiffness together with the consistently higher, though nonstatistically significant, improvement of clinical parameters demonstrate that ACS induces a biological response and warrant future investigations into the possible effects of ACS.
The cause of bone tunnel widening is unclear, but it is thought to be multifactorial, with mechanical and biological factors playing a role [61, 62]. Although this present study cannot prove it alone, there are findings that favor biological factors play a more important role in tunnel enlargement than mechanical ones. In the present trial, it was shown that there is an association between tibial bone tunnel enlargement and elevated synovial fluid concentrations of IL-1β concentrations postoperatively after ACL reconstruction, indicating a potential involvement of these biological mediators in the pathogenesis of bone tunnel widening [15]. However, there was no difference in the contents of synovial fluid between HS and BTB ACL, suggesting that the biological response in both of these types of autografts is similar. The present study as well as previously published trials show that the type of the fixation appears not to have a determinant effect on tunnel enlargement, since enlargement occurs irrespective of fixation type, suggesting that the enlargement may be more of a biological than a mechanical phenomenon. There was also variation in bone tunnel widening between patients, even among patients treated with similar grafts, fixation devices and methods, same surgeon, and rehabilitation protocol. These findings cannot be explained by biomechanical factors only. As in previously published studies, no significant correlations were found between the aforementioned outcome measures and the tunnel widening at 12-month follow-up in our patients, but bone tunnel enlargement after ACL reconstruction has been suggested to play an important role for revision ACL surgery, which is necessary in about 8% of patients after ACL reconstruction [77].
Advances in molecular and cellular biology are beginning to influence the clinical practice of orthopaedics. Novel, biologically based therapeutic agents like ACS promise to improve the management of a variety of orthopedic conditions through the inhibition of catabolic cytokines and the local administration of autologous anabolic growth factors [44, 51, 53, 72]. Autologous conditioned serum (ACS) is produced from the patient’s own blood and can be manufactured with ease at the point of care. When results like from the present trial need to be explained, we have to turn to the previous works on this new treatment strategy. ACS has been used in more than 30,000 humans and 10,000 horses. It has recently been shown to be effective in two randomized controlled osteoarthritis trials [3, 80] and in an RCT on the treatment of sciatic back pain [5]. It has been proved to improve clinical lameness in horses significantly [28], and to enhance healing of muscle and tendon injuries in animals [44, 79] as well as in human athletes [78].
There are many products and devices on the market derived from blood; however, this product is different from platelet-rich plasma (PRP) or autologous conditioned plasma (ACP) preparations, because besides the simple concentration of blood own factors, it leads through incubation in a special created surrounding to the production of IL-1Ra (partly de novo) in order to decrease the inflammation cytokine. Because of the autologous nature of the treatment (meaning that only substances originating from the patient’s own body are used) and the physiological levels of cytokines and growth factors that are achieved, the risk/benefit ratio is excellent.
There is an urgent need for consensus about postoperative treatment strategies to enhance the speed and safety in which a patient regains the pre-injury level of function or an athlete returns to sport. Preclinical patient education, an optimal timing of surgery, exact starting times of postoperative regimens, and control of the rehabilitation aims with reliable tests could enable a less-complicated rehabilitation course [1, 58, 64, 71]. In the present trial, absolute values of the WOMAC and IKDC outcome measures showed that ACS-treated patients scored consistently better, compared with placebo-treated patients. In addition, ACS treatment was found to result in statistically significant more improvement on the WOMAC stiffness score after 6 months. However, the differences disappeared over time, but when looking at the different results you can see that treating with ACS could reach earlier better functional outcomes and may be the subtle distinction that could cause the difference of success or failure after surgery. However, additional studies are necessary to determine which strategies could optimize outcome most efficiently and to better estimate the expected benefits of the treated patients.
One possible limitation of this study is that the CT scans were performed 12 months after surgery and the enlargement could not be finished yet. Revision surgery causes significant extra costs, but since the trial has a relatively short follow-up period, it is currently not possible to proof if this postoperative ACS treatment protocol has an influence on that or if there might be a decreased concern in revision surgeries about tunnel enlargement in ACS-treated patients. The main concern surgeons have about TW is the possibility of subsequent ligament laxity. Yet in the present study, as in other studies, ligament laxity did not occur. No significant differences in stability (elongation or rupture of the graft) comparing HS and BTB were found. However, besides standardized clinical examination by a specially trained surgeon, no objective measurement of knee stability was done in this trial.
One major point in this trial was that the surgery itself helped most in patients suffering from ACL deficiency independent of postsurgical treatment. Ultimately, the trial objective was mainly to determine effects of ACS as a new treatment, compared with placebo, and not focus on the differences between the surgical options. Different fixation methods could also lead to other clinical and functional outcomes, but there remains considerable debate regarding the actual optimal choice, making clinical application challenging. Yet, in the present trial, all surgical procedures of tibial fixation were performed by the same surgeon, and interferent screws lengths, materials, and application techniques were the same in all cases.
Conclusion
Novel technologies and standardized postoperative treatment regiments such as those described in the present RCT may provide useful therapeutic procedures to enhance biological healing after reconstructive surgery.
Autologous conditioned serum (ACS) appears to have a beneficial biological effect on patient-documented symptoms and structure modifying effects arising after ACL reconstructive surgery, although this hypothesis remains to be studied in future clinical trials.
References
Andersson D, Samuelsson K, Karlsson J (2009) Treatment of anterior cruciate ligament injuries with special reference to surgical technique and rehabilitation: an assessment of randomized controlled trials. Arthroscopy 25:653–685
Arend WP (1993) Interleukin-1 receptor antagonist. Adv Immunol 54:167–227
Baltzer AW, Moser C, Jansen SA et al (2009) Autologous conditioned serum (Orthokine) is an effective treatment for knee osteoarthritis. Osteoarthr Cartil 17:152–160
Baumfeld JA, Diduch DR, Rubino LJ et al (2008) Tunnel widening following anterior cruciate ligament reconstruction using hamstring autograft: a comparison between double cross-pin and suspensory graft fixation. Knee Surg Sports Traumatol Arthrosc 16:1108–1113
Becker C, Heidersdorf S, Drewlo S et al (2007) Efficacy of epidural perineural injections with autologous conditioned serum for lumbar radicular compression: an investigator-initiated, prospective, double-blind, reference-controlled study. Spine 32:1803–1808
Berg EE, Pollard ME, Kang Q (2001) Interarticular bone tunnel healing. Arthroscopy 17:189–195
Bohnsack M, Hehl S, Moller H et al (2006) Influence of the postoperative activity level on tibial bone tunnel enlargement and functional treatment results following anterior cruciate ligament reconstruction using a patellar tendon autograft. Z Orthop Ihre Grenzgeb 144:373–379
Brown CH Jr, Wilson DR, Hecker AT et al (2004) Graft-bone motion and tensile properties of hamstring and patellar tendon anterior cruciate ligament femoral graft fixation under cyclic loading. Arthroscopy 20:922–935
Buck DC, Simonian PT, Larson RV et al (2004) Timeline of tibial tunnel expansion after single-incision hamstring anterior cruciate ligament reconstruction. Arthroscopy 20:34–36
Buelow JU, Siebold R, Ellermann A (2002) A prospective evaluation of tunnel enlargement in anterior cruciate ligament reconstruction with hamstrings: extracortical versus anatomical fixation. Knee Surg Sports Traumatol Arthrosc 10:80–85
Chevalier X, Giraudeau B, Conrozier T et al (2005) Safety study of intraarticular injection of interleukin 1 receptor antagonist in patients with painful knee osteoarthritis: a multicenter study. J Rheumatol 32:1317–1323
Choi NH, Lee JH, Son KM et al (2010) Tibial tunnel widening after anterior cruciate ligament reconstructions with hamstring tendons using Rigidfix femoral fixation and Intrafix tibial fixation. Knee Surg Sports Traumatol Arthrosc 18:92–97
Clark JC, Rueff DE, Indelicato PA et al (2009) Primary ACL reconstruction using allograft tissue. Clin Sports Med 28:223–244 (viii)
Clatworthy MG, Annear P, Bulow JU et al (1999) Tunnel widening in anterior cruciate ligament reconstruction: a prospective evaluation of hamstring and patella tendon grafts. Knee Surg Sports Traumatol Arthrosc 7:138–145
Darabos N, Hundric-Haspl Z, Haspl M et al (2009) Correlation between synovial fluid and serum IL-1beta levels after ACL surgery-preliminary report. Int Orthop 33:413–418
Dinarello CM (1999) Proinflammatory and anti-inflammatory cytokines. Rheumatoid Arthritis
Evans CH, Ghivizzani SC, Robbins PD (2009) Orthopedic gene therapy in 2008. Mol Ther 17:231–244
Evans CH, Gouze JN, Gouze E et al (2004) Osteoarthritis gene therapy. Gene Ther 11:379–389
Fauno P, Kaalund S (2005) Tunnel widening after hamstring anterior cruciate ligament reconstruction is influenced by the type of graft fixation used: a prospective randomized study. Arthroscopy 21:1337–1341
Feller JA, Webster KE, Taylor NF et al (2004) Effect of physiotherapy attendance on outcome after anterior cruciate ligament reconstruction: a pilot study. Br J Sports Med 38:74–77
Fernandes J, Tardif G, Martel-Pelletier J et al (1999) In vivo transfer of interleukin-1 receptor antagonist gene in osteoarthritic rabbit knee joints: prevention of osteoarthritis progression. Am J Pathol 154:1159–1169
Fernandes JC, Martel-Pelletier J, Pelletier JP (2002) The role of cytokines in osteoarthritis pathophysiology. Biorheology 39:237–246
Figueroa D, Melean P, Calvo R et al (2010) Magnetic resonance imaging evaluation of the integration and maturation of semitendinosus-gracilis graft in anterior cruciate ligament reconstruction using autologous platelet concentrate. Arthroscopy 26:1318–1325
Fink C, Zapp M, Benedetto KP et al (2001) Tibial tunnel enlargement following anterior cruciate ligament reconstruction with patellar tendon autograft. Arthroscopy 17:138–143
Foldager C, Jakobsen BW, Lund B et al (2010) Tibial tunnel widening after bioresorbable poly-lactide calcium carbonate interference screw usage in ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 18:79–84
Frank C, Amiel D, Woo SL et al (1985) Normal ligament properties and ligament healing. Clin Orthop Relat Res 196:15–25
Frank RM, Seroyer ST, Lewis PB et al (2010) MRI analysis of tibial position of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc 18:1607–1611
Frisbie DD, Kawcak CE, Werpy NM et al (2007) Clinical, biochemical, and histologic effects of intra-articular administration of autologous conditioned serum in horses with experimentally induced osteoarthritis. Am J Vet Res 68:290–296
Goldring SR, Goldring MB (2004) The role of cytokines in cartilage matrix degeneration in osteoarthritis. Clin Orthop Relat Res 427:S27–S36
Granowitz EV, Clark BD, Mancilla J et al (1991) Interleukin-1 receptor antagonist competitively inhibits the binding of interleukin-1 to the type II interleukin-1 receptor. J Biol Chem 266:14147–14150
Herbort M, Lenschow S, Fu FH et al (2010) ACL mismatch reconstructions: influence of different tunnel placement strategies in single-bundle ACL reconstructions on the knee kinematics. Knee Surg Sports Traumatol Arthrosc 18:1551–1558
Hersekli MA, Akpinar S, Ozalay M et al (2004) Tunnel enlargement after arthroscopic anterior cruciate ligament reconstruction: comparison of bone-patellar tendon-bone and hamstring autografts. Adv Ther 21:123–131
Hoher J, Moller HD, Fu FH (1998) Bone tunnel enlargement after anterior cruciate ligament reconstruction: fact or fiction? Knee Surg Sports Traumatol Arthrosc 6:231–240
Hollis R, West H, Greis P et al (2009) Autologous bone effects on femoral tunnel widening in hamstring anterior cruciate ligament reconstruction. J Knee Surg 22:114–119
Iorio R, Vadala A, Argento G et al (2007) Bone tunnel enlargement after ACL reconstruction using autologous hamstring tendons: a CT study. Int Orthop 31:49–55
Irie K, Uchiyama E, Iwaso H (2003) Intraarticular inflammatory cytokines in acute anterior cruciate ligament injured knee. Knee 10:93–96
Jacobs JJ, Roebuck KA, Archibeck M et al (2001) Osteolysis: basic science. Clin Orthop Relat Res 393:71–77
Jagodzinski M, Foerstemann T, Mall G et al (2005) Analysis of forces of ACL reconstructions at the tunnel entrance: is tunnel enlargement a biomechanical problem? J Biomech 38:23–31
Jagodzinski M, Geiges B, von Falck C et al (2010) Biodegradable screw versus a press-fit bone plug fixation for hamstring anterior cruciate ligament reconstruction: a prospective randomized study. Am J Sports Med 38:501–508
Kohno T, Ishibashi Y, Tsuda E et al (2007) Immunohistochemical demonstration of growth factors at the tendon-bone interface in anterior cruciate ligament reconstruction using a rabbit model. J Orthop Sci 12:67–73
Kuskucu SM (2008) Comparison of short-term results of bone tunnel enlargement between EndoButton CL and cross-pin fixation systems after chronic anterior cruciate ligament reconstruction with autologous quadrupled hamstring tendons. J Int Med Res 36:23–30
L’Insalata JC, Klatt B, Fu FH et al (1997) Tunnel expansion following anterior cruciate ligament reconstruction: a comparison of hamstring and patellar tendon autografts. Knee Surg Sports Traumatol Arthrosc 5:234–238
Maak TG, Voos JE, Wickiewicz TL et al (2010) Tunnel widening in revision anterior cruciate ligament reconstruction. J Am Acad Orthop Surg 18:695–706
Majewski M (2009) Accelerated healing of the rat Achilles tendon in response to autologous conditioned serum. Am J Sports Med 37:2117–2125
Manolagas SC, Jilka RL (1995) Bone marrow, cytokines, and bone remodeling. Emerging insights into the pathophysiology of osteoporosis. N Engl J Med 332:305–311
Marchant MH Jr, Willimon SC, Vinson E et al (2010) Comparison of plain radiography, computed tomography, and magnetic resonance imaging in the evaluation of bone tunnel widening after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 18:1059–1064
Meijer H, Reinecke J, Becker C et al (2003) The production of anti-inflammatory cytokines in whole blood by physico-chemical induction. Inflamm Res 52:404–407
Moisala AS, Jarvela T, Paakkala A et al (2008) Comparison of the bioabsorbable and metal screw fixation after ACL reconstruction with a hamstring autograft in MRI and clinical outcome: a prospective randomized study. Knee Surg Sports Traumatol Arthrosc 16:1080–1086
Myers P, Logan M, Stokes A et al (2008) Bioabsorbable versus titanium interference screws with hamstring autograft in anterior cruciate ligament reconstruction: a prospective randomized trial with 2-year follow-up. Arthroscopy 24:817–823
Nebelung W, Becker R, Merkel M et al (1998) Bone tunnel enlargement after anterior cruciate ligament reconstruction with semitendinosus tendon using Endobutton fixation on the femoral side. Arthroscopy 14:810–815
Nin JR, Gasque GM, Azcarate AV et al (2009) Has platelet-rich plasma any role in anterior cruciate ligament allograft healing? Arthroscopy 25:1206–1213
Nordstrom DC, Santavirta S, Aho A et al (1999) Immune responses to osteoarticular allografts of the knee–cytokine studies. Arch Orthop Trauma Surg 119:195–198
Orrego M, Larrain C, Rosales J et al (2008) Effects of platelet concentrate and a bone plug on the healing of hamstring tendons in a bone tunnel. Arthroscopy 24:1373–1380
Pelletier JP, Caron JP, Evans C et al (1997) In vivo suppression of early experimental osteoarthritis by interleukin-1 receptor antagonist using gene therapy. Arthritis Rheum 40:1012–1019
Peyrache MD, Djian P, Christel P et al (1996) Tibial tunnel enlargement after anterior cruciate ligament reconstruction by autogenous bone-patellar tendon-bone graft. Knee Surg Sports Traumatol Arthrosc 4:2–8
Pinczewski LA, Deehan DJ, Salmon LJ et al (2002) A five-year comparison of patellar tendon versus four-strand hamstring tendon autograft for arthroscopic reconstruction of the anterior cruciate ligament. Am J Sports Med 30:523–536
Pinczewski LA, Lyman J, Salmon LJ et al (2007) A 10-year comparison of anterior cruciate ligament reconstructions with hamstring tendon and patellar tendon autograft: a controlled, prospective trial. Am J Sports Med 35:564–574
Samuelsson K, Andersson D, Karlsson J (2009) Treatment of anterior cruciate ligament injuries with special reference to graft type and surgical technique: an assessment of randomized controlled trials. Arthroscopy 25:1139–1174
Sastre S, Popescu D, Nunez M et al (2010) Double-bundle versus single-bundle ACL reconstruction using the horizontal femoral position: a prospective, randomized study. Knee Surg Sports Traumatol Arthrosc 18:32–36
Schultz WR, McKissick RC, DeLee JC (2007) Tibial tunnel widening after hamstring tendon anterior cruciate ligament reconstruction: the effect of supplemental aperture fixation with autogenous bone cores. Am J Sports Med 35:1725–1730
Siebold R, Cafaltzis K (2010) Differentiation between intraoperative and postoperative bone tunnel widening and communication in double-bundle anterior cruciate ligament reconstruction: a prospective study. Arthroscopy 26:1066–1073
Silva A, Sampaio R, Pinto E (2010) Femoral tunnel enlargement after anatomic ACL reconstruction: a biological problem? Knee Surg Sports Traumatol Arthrosc 18:1189–1194
Silva A, Sampaio R, Pinto E (2010) Placement of femoral tunnel between the AM and PL bundles using a transtibial technique in single-bundle ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 18:1245–1251
Strehl A, Eggli S (2007) The value of conservative treatment in ruptures of the anterior cruciate ligament (ACL). J Trauma 62:1159–1162
Streich NA, Friedrich K, Gotterbarm T et al (2008) Reconstruction of the ACL with a semitendinosus tendon graft: a prospective randomized single blinded comparison of double-bundle versus single-bundle technique in male athletes. Knee Surg Sports Traumatol Arthrosc 16:232–238
Ugutmen E, Ozkan K, Guven M et al (2007) Early tunnel enlargement after arthroscopic ACL reconstructions. Acta Orthop Belg 73:625–629
Vadala A, Iorio R, De Carli A et al (2007) The effect of accelerated, brace free, rehabilitation on bone tunnel enlargement after ACL reconstruction using hamstring tendons: a CT study. Knee Surg Sports Traumatol Arthrosc 15:365–371
van’t Hof RJ, Ralston SH (2001) Nitric oxide and bone. Immunology 103:255–261
van den Berg WB (1999) The role of cytokines and growth factors in cartilage destruction in osteoarthritis and rheumatoid arthritis. Z Rheumatol 58:136–141
van Eck CF, Schreiber VM, Liu TT et al (2010) The anatomic approach to primary, revision and augmentation anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 18:1154–1163
van Grinsven S, van Cingel RE, Holla CJ et al (2010) Evidence-based rehabilitation following anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 18:1128–1144
Vogrin M, Rupreht M, Dinevski D et al (2010) Effects of a platelet gel on early graft revascularization after anterior cruciate ligament reconstruction: a prospective, randomized, double-blind, clinical trial. Eur Surg Res 45:77–85
Webster KE, Feller JA, Elliott J et al (2004) A comparison of bone tunnel measurements made using computed tomography and digital plain radiography after anterior cruciate ligament reconstruction. Arthroscopy 20:946–950
Webster KE, Feller JA, Hameister KA (2001) Bone tunnel enlargement following anterior cruciate ligament reconstruction: a randomised comparison of hamstring and patellar tendon grafts with 2-year follow-up. Knee Surg Sports Traumatol Arthrosc 9:86–91
Wehling P, Moser C, Frisbie DD et al (2007) Autologous Conditioned Serum in the treatment of Orthopaedic diseases—The Orthokine Therapy. Biodrugs 21:223–232
Wilson TC, Kantaras A, Atay A et al (2004) Tunnel enlargement after anterior cruciate ligament surgery. Am J Sports Med 32:543–549
Wolf RS, Lemak LJ (2002) Revision anterior cruciate ligament reconstruction surgery. J South Orthop Assoc 11:25–32
Wright-Carpenter T, Klein P, Schaferhoff P et al (2004) Treatment of muscle injuries by local administration of autologous conditioned serum: a pilot study on sportsmen with muscle strains. Int J Sports Med 25:588–593
Wright-Carpenter T, Opolon P, Appell HJ et al (2004) Treatment of muscle injuries by local administration of autologous conditioned serum: animal experiments using a muscle contusion model. Int J Sports Med 25:582–587
Yang KG, Raijmakers NJ, van Arkel ER et al (2008) Autologous interleukin-1 receptor antagonist improves function and symptoms in osteoarthritis when compared to placebo in a prospective randomized controlled trial. Osteoarthr Cartil 16:498–505
Yu JK, Paessler HH (2005) Relationship between tunnel widening and different rehabilitation procedures after anterior cruciate ligament reconstruction with quadrupled hamstring tendons. Chin Med J (Engl) 118:320–326
Zysk SP, Fraunberger P, Veihelmann A et al (2004) Tunnel enlargement and changes in synovial fluid cytokine profile following anterior cruciate ligament reconstruction with patellar tendon and hamstring tendon autografts. Knee Surg Sports Traumatol Arthrosc 12:98–103
Acknowledgments
The trial was an investigator-initiated, prospective, double-blind, reference-controlled study. The source of all funding for the study has been obtained by the National Health Insurance. We thank Vera Srsan Zivanovic (Department of General Surgery and Traumatology, General Hospital Varazdin), Renata Jug (Institute of Imunology, Zagreb), Damir Poljak (Department of Transfusiology, General Hospital Varazdin), and Darko Kranjcec (Department of General Surgery and Traumatology, General Hospital Varazdin).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Darabos, N., Haspl, M., Moser, C. et al. Intraarticular application of autologous conditioned serum (ACS) reduces bone tunnel widening after ACL reconstructive surgery in a randomized controlled trial. Knee Surg Sports Traumatol Arthrosc 19 (Suppl 1), 36–46 (2011). https://doi.org/10.1007/s00167-011-1458-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-011-1458-4