Abstract
Purpose
Evaluate the effects of intra-operative leucocyte-poor-platelet-rich plasma (PRP) (type P3-Bβ with endogenous activation) injection in International Cartilage Repair Society (ICRS) grade III knee chondral injuries treated by chondroplasties, to increase and ameliorate the repair tissue.
Methods
Patients were divided into two groups. Group A (control) consisted of 31 patients and Group B (PRP) 33 patients, totaling 64 patients analyzed. Patients also could had associated injuries (meniscal and/or ACL) being equally divided between both groups to avoid bias. PRP was injected at the end of surgery in group B. The patient outcomes were assessed using subjective International Knee Documentation Committee (IKDC) form, Knee Injury and Osteoarthritis Outcome Score (KOOS), and Tegner activity forms, prior to the surgery and three, six, 12, and 24 months after surgery (medium-term follow up).
Results
IKDC and KOOS scores showed increase at each evaluation time points after surgery in both groups, but the treated Group (B) showed a higher increase with statistically significant difference. The Tegner activity scores were higher for the treated group only at six and 12 months.
Conclusion
Based on the subjective IKDC, KOOS, and Tegner scores, those patients affected by ICRS grade III chondral injuries undergoing arthroscopic chondroplasty who were also treated with PRP showed better and faster outcomes than the control group. Independently from the associated injury (meniscal or ACL). This difference could be measured for up to two years.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The treatment of chondral injuries remains a challenge. This tissue has limited healing potential because of its complex extracellular matrix and the absence of vessels. [1, 2] The final repair tissue is mostly fibrocartilage, which does not have the same load resistance when compared with hyaline cartilage. It also has a tendency for early degeneration [1, 2]. Results of surgical procedures vary and do not guarantee the prevention of degenerative joint disease progression [3].
Platelet-rich plasma (PRP) is a fraction of plasma with supraphysiological concentration of platelets that is rich in growth factors [4, 5]. There is evidence that PRP can improve the healing process and reduce pain when used for muscle and tendon injuries, as well as when it is used in the treatment of tendinopathy (especially for epicondylitis) and osteoarthritis [6,7,8,9].
Studies show that in cartilage PRP could increase the glycosaminoglycans and type II collagen synthesis and reduce the chondral degradation [2, 6]. It is also able to induce mesenchymal stem cell chondrogenesis and promote chondrocytes proliferation, differentiation, and adhesion [6, 10]. This product has also proven to have biological glue function, to regulate the homeostasis, and to restore the intra-articular hyaluronic acid quality [7, 8, 10]. PRP improves the quality of the synovial fluid and reduces inflammation and pain [4, 11,12,13,14]. Furthermore, it can improve the quality and increase the production of the superficial zone protein, which leads to better articular surface lubrication [15].
Some authors still have some concerns about the effectiveness of PRP [9, 16] but many recent articles show better clinical results when compared with placebo [17, 18] and hyaluronic acid (HA) in the treatment of knee osteoarthritis [17,18,19,20]. This variation may be due to the wide variety of products that can be named PRP. There are commercial and homemade products [10, 21, 22]; with different platelet concentration [2, 21,22,23]; activated by thrombin, CaCl2, or without activation [21, 24]; leukocyte-rich (LR) or leukocyte-poor (LP) [4, 10, 12, 21, 25]; made on the day of application or frozen for later use [16, 19, 20, 26]; number of applications required and time between these injections [19, 25, 27]; and with great discussion about the advantages or disadvantages of each characteristic presented.
Kon et al. [20] also suggests that even if PRP has no effect on the chondral structure or on the progression of degenerative joint disease, it may, however, influence articular homeostasis, reduce synovial tissue hypertrophy, and modulate cytokines. Thus, it could lead to clinical improvement in patients with cartilage injury, even if only temporarily.
This study aims to evaluate the effects of PRP surgical application during arthroscopy in symptomatic patients with International Cartilage Repair Society (ICRS) grade III chondral knee injuries, who may or may not have another injury, if there is better function and quality of life when compared with patients with similar injuries who did not receive PRP (control group). The hypothesis is that the patients receiving PRP will have a faster recovery with better results at the end of follow up, when compared with the control group.
Methods
The study was approved by two Ethical Committees. Patients were selected from a private institution and accepted to participate in the study after orientation. All patients accepted the terms and signed a consent form prior to the study inclusion.
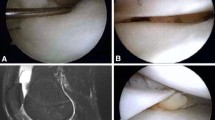
The study included a total of 80 patients selected between November 2011 and June 2014. The inclusion criteria were as follows: patients of both genders aged 18 to 50 (mean 35.65; SD 8.25). All patients had full-thickness chondral injuries without compromising the subchondral bone in the affected knee, therefore classified as ICRS grade III. All chondral injuries must be under 2 cm2 and treated by chondroplasty. Microfractures technique was avoided during the surgery to prevent bias. As some of the patients also had ACL and/or meniscus injuries, they were submitted to surgical treatment for the chondral injury as well as for the other injuries. The contralateral knee had to be asymptomatic. All tested patients took an MRI exam of the affected knee up to three months before surgery.
Patients were excluded if they had osteoarthrosis of any knee compartment (Kellgreen & Lawrence > 1), if they had any injuries to structures other than cartilage, meniscus or ACL, if they had indication for meniscus repair, if they did not follow the post-operative protocol, if they chose to leave the study, if their BMI (body mass index) was greater than 30, or if their pre-operative platelet dosage was lower than 150.000/mm3.
The subjects of the control group (no PRP application—Group A) and treated group (PRP application—Group B) were allocated by the author randomly the day before surgery using a random number table. The patients were not present at the allocation and were kept unaware of their group for the duration of the trial. Patients that needed ACL reconstruction were divided equally between the groups.
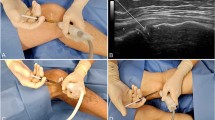
On the day of the procedure, the patients were sedated with 15 mg of Midazolam Maleate administered orally 30 minutes before entering the operating room. A peripheral vein was punctured, and 20 ml of blood was collected from the patients of the treated group, to obtain the PRP. The blood was stored in 5 ml tubes with 10% of sodium citrate. The blood in each tube was centrifuged at 1200 RPM for ten minutes in a 6.5-cm radius centrifuge (Fanem®, Guarulhos/SP). As a result of the centrifugation the blood split into three layers: red blood cells (bottom of the tube), white blood cells (thin layer over the red cells, also called buffy coat), and plasma (the upper layer). The plasma was then transferred to another 10 ml sterile tube, without the buffy coat and was centrifuged again for five minutes at the same rotation speed. The second centrifugation split the plasma into two further layers, the upper half, the platelet-poor plasma, and the lower half, the platelet-rich plasma, also called platelet concentrate. The PRP was then stored in a sterile syringe to be applied at the end of surgery. The PRP was always prepared by the same trained and experienced professional throughout the process.
Surgical technique
First the patient received a spinal anesthesia and then the limb was prepared. All surgeries were performed with proximal thigh tourniquet. The procedure began with standard arthroscopy portals. Chondroplasties was done in chondral injuries in order to regularize the injury. The technique consists of removing with a shaver the chondral tissue not attached to the subchondral bone, leaving stable walls. When present we remove the fibrous tissue covering the subchondral bone, always preserving these structure (Fig. 1).
Meniscal ruptures, when present, were treated with partial meniscectomy. Patients with indication of meniscal repair were excluded. ACL ruptures were treated with standard reconstruction with autograft hamstrings. At the end of the procedure, prior to portal closure, the PRP (5 ml) was applied on the chondral injuries.
During the immediate post-operative period, the patient was treated with dipyrone (1 g every 6 h), tramadol (50 mg every 6 h), and morphine (4 mg in case of pain). During hospitalization ice packs were applied for 20 minutes every hour. At discharge, 12 hours after surgery, 37.5 mg of Tramadol Hydrochloride was prescribed, along with 325 mg of Paracetamol as well as 500 mg of dipyrone (in case of pain) to be taken every eight hours for five days. The patients were also instructed to apply ice packs for 20 minutes every two hours.
Patients were instructed to begin the rehabilitation protocol during the immediate post-operative period with continuous passive mobilization and stimulation for isometrics quadriceps contraction. The patients were advised to start physical therapy the day following surgery, or as soon as possible. Crutches with partial weight bearing were to be used for 14 days, then with full weight bearing thereafter.
Patients were assessed using forms preoperatively as well as three, six, 12, and 24 months after surgery. These included the subjective International Knee Documentation Committee (IKDC) form, the Knee Injury and Osteoarthritis Outcome Score (KOOS), and the Tegner activity form. All scores of the forms were documented.
Patients who found performing the exercises of the physical therapy protocol difficult and painful after three months of surgery were treated with viscosupplementation as long as no new injuries were found. The treatment was 1 dose of 20 mg/2.0 ml of intraarticular sodium hyaluronate (Fermathron®) per week for three weeks.
Patients who developed intense joint effusion (more than 3+) had their knee drained.
PRP samples from six randomized patients were tested for platelet count, in order to confirm the quality of the technique. Further three PRP samples were tested for leukocyte count.
Statistical analysis
The sample size of 40 patients per group was calculated based on 5% margin of error and 80% of power. A chi-square test was used to ensure group homogeneity in terms of gender, side, age, need of viscosupplementation, joint aspiration, and associated injuries. An analysis of variance (ANOVA) for repeated measures was used to compare the groups at different time points in conjunction with the Turkey–Kramer test controlled by the presence of ACL injury. The tests were carried out at 5% (p < 0.05) significance level. SPSS® software for Windows®, version 19.0—IBM Corp. (Armonk, NY) was used for all analyses.
The comparative analysis of groups was controlled by the effect of ACL reconstruction and its interaction effect, both in the univariate variance analysis and for repeated measurements. All variables were evaluated for variance homogeneity between groups by the Levene test, in which the p values were greater than 0.05, except for the Tegner score assessed after intervention, which is why the F test was chosen in the variance analysis.
Results
Of the 80 selected patients, nine were excluded from Group A and seven from Group B. Two patients completed the forms incorrectly and 14 missed the follow up, representing a total loss of 20%. Thus, 33 patients completed the study in Group B (treated) and 31 in Group A (control).
Table 1 shows the demographic data of the subjects and associated injuries of both groups. The size of injuries ranged from 0.4 to 1.8 cm2 in group A, with a mean of 1.2 cm2. In Group B ranged from 0.3 to 1.9 cm2, with a mean of 1.0 cm2. This injuries are defining as small-medium cartilage defects. Tables 2 show the local distribution of chondral injuries broken down per group. Injuries classified as grade I or II were left untreated.
The groups were statistically similar in gender, age, BMI, operated side, injure size, and associated injuries.
Seven patients (22.58%) in Group A underwent supplementary treatment with viscosupplementation. However, in group B, only 2 patients (6.06%) required such treatment. Despite the wide range of values, the groups were not statistically different for this variable. One patient in Group A and 2 in Group B required joint aspiration post-operatively, which did not indicate statistical difference between the groups either.
The mean platelet count in the PRP samples of the six selected patients was 964,833/mm3, a 3.13 times of increase compared with the serum dosage. The leukocyte count of the three selected PRP samples was as follows: 1490/mm3, 4210/mm3, and 2680/mm3, with an average of 2793 leukocytes/mm3.
The IKDC and KOOS form results showed significant difference at three, six, 12, and 24 months. In each case, the treated group (B) showed statistically better results compared with those of the untreated group (A) (Tables 3 and 4; Figs. 2 and 3).
As for the Tegner score, the ANOVA shows significant difference between the groups with better outcomes for the treated group at 6 and 12 months (Tables 5; Fig. 4).
The interaction effect of the ACL reconstruction was not significant at either evaluation time point for all evaluated scores (Tables 3, 4, and 5).
Discussion
The most important finding of this study was that the intraarticular injection of leukocyte-poor-PRP after chondroplasty for the treatment of small to medium ICRS grade III chondral knee injuries may improve the functional outcomes within three months of surgery and this improvement remains for up to 24 months after surgery. This promising result is even more encouraging because it is independent of any other associated procedures that have been eventually performed with the cartilage defect treatment as partial meniscectomy or ACL reconstruction.
Intra-articular PRP injections have been used in the treatment of knee osteoarthritis with encouraging results [20]. However some articles have suggested that its effects are limited [14, 16]. Filardo et al. [16] conducted a study on 192 patients with chondropathy. The 96 patients of the study’s treated group received one application of PRP per week for three weeks, while another 96 patients in the control group received hyaluronate. After 12 months, they concluded that there was no difference between the groups. However, the patients’ average age was much higher than the one presented here (53.32 for men and 57.55 for women). Patients with focal chondral injuries were excluded, and the PRP were frozen for the last two applications, which could have caused bias. Hart et al. [25] examined the effects of nine PRP applications in the course of one year in a randomized clinical trial of grade II or III chondral injury patients with tendency to osteoarthritis. Although the treated group had better Lysholm, Tegner, Cincinnati, and subjective IKDC scores, the follow-up MRI scans did not confirm any improvement. This suggests that PRP affects the articular environment rather than the healing process. However, Sheth et al. [9] concluded in a meta-analysis, using pain VAS (Visual Analog Scale), that PRP does not provide better pain relief than other treatments.
There is no evidence of another control group study, similar to the one presented, which evaluates the effects of PRP on patients undergoing surgery for focal cartilage injuries. Siclari et al. [28] used microfractures to treat chondral injuries and applied a hyaluronate scaffold immersed in PRP in a study with 52 patients. Although the KOOS scores demonstrated early recovery (within 3 months) and the histological analysis showed a hyaline like chondral repair tissue in five patients, there was no control group to provide a baseline. A scaffold was used in the study, which increases the final costs and limits procedure access. Using microfractures can lead to a bias in the evaluation of applying PRP. When performing microfractures the results can be attributed to the surgical technique rather than the PRP application. That is why only chondroplasties were performed in our study and no microfractures.
PRP preparation type should be adapted to the tissue it will be applied to in order to optimize the effects [12]. PRPs with higher leukocyte concentration may favour a greater presence of catabolic and pro-inflammatory cytokines like MMP-9 and IL-1β which may not have a positive effect on the articular environment [4, 12, 23, 27]. Leukocytes can secrete inflammatory mediators, proteases, and reactive oxygen species in the joint which can lead to a greater inflammatory reaction and may cause limited pain and joint effusion [16, 27]. However, from another point of view, some white cells like peripheral blood mononuclear cells (lymphocytes and monocytes) could have a positive effect by releasing anabolic cytokines such as IL-6 as well as proteins and enzymes involved in preventing infection [27, 29]. In vitro studies suggest that LR-PRP may have this deleterious effect on chondral tissue [10], but in vivo studies do not support these results [26]. Belk et al. [30] in a literature review showed that the leukocytes concentration in PRP do not influence WOMAC or VAS scores, but LP-PRP had better results in subjective IKDC scores. However Filardo et al. [18] in a meta-analysis of RCTs showed that the leukocytes concentration does not influence the PRP functional results. Thus, the role of leukocytes in PRP remains under discussion.
Activating PRP in advance of the application can also influence the results. Using thrombin stimulates quick clot formation, which may cause inflammatory response in joints, probably due to the very rapid release of growth factors (GF) [13, 21, 24, 27]. This clot may reduce the availability of the GFs. Furthermore, thrombin can initiate an immune response [13, 21, 24]. Activation by CaCl2 is very similar to thrombin, but with less clot retraction and no antibody production [21]. Injection of PRP in its liquid form has the advantage of allow the product to reach areas of difficult access within the joint as it spreads [27]. PRP contact with collagen of the extra-cellular matrix causes a physiological activation inducing the formation of a bioscaffold with slow release of GFs, prolonging its effect [27]. However, there is still no consensus on whether the form of PRP activation influences the results. The PRP obtained in the present study would be classified as P3–Bβ [21]. It was decided that endogenous activation would be used with leukocyte poor PRP, which may have been beneficial for the treatment.
The platelet count varies depending on the obtaining method [21, 22]. Some studies suggest that it may influence the PRP action because of the difference in GFs and inflammatory mediator concentrations [14, 22, 23]. The method of the present study is classified as homemade, and it has been standardized and validated.
Including patients with ACL reconstruction could cause bias as bone tunnels are drilled (which increases the intra-articular stem cell concentration) and rehabilitation is longer. At present, there are no conclusive studies on the effects of using PRP in ACL reconstruction surgery [13, 31]. Del Torto et al. [7] in a comparative study with 28 patients undergoing ACL reconstruction applied PRP in the treatment of 14 patients and compared the results with the other 14 as control. They concluded that although the PRP application led to better subjective IKDC scores at six, 12, and 24 months after surgery, the MRI tests have revealed no changes in the healing process. The author suggests that PRP should restore a better articular homeostasis or stimulate intra-articular anabolism.
Therefore, in order to reduce the bias resulting from ACL reconstruction, the affected patients were equally distributed between the two groups. These patients were included in the study because of the high prevalence of this surgery in the routine of the knee surgeon and the high association of these injuries.
The IKDC and KOOS scores showed that the PRP group had better results at each stage of the post-operative evaluation. The difference was already statistically significant at the first evaluation time point at three months. The Tegner score only showed difference in favour of the treated group at six and 12 months. The different results of the Tegner form are due to some factors: the form evaluates the patients’ activity levels which may only change slightly during rehabilitation. Most of the selected patients were not athletes; their physical activity levels were classified as recreational; therefore, they did not depend on returning to their preinjury activity level within a short time.
Although there was no statistical difference in the need for viscosupplementation, there were more patients in the control group who needed this treatment (22.58%). This also suggests that PRP can have a better effect in the articular environment possibly due to its ability to improve synovial fluid quality and production [11, 14], balance intra-articular homeostasis [11,12,13,14] and its direct lubrication [15], and anti-inflammatory effect [14]. Patients who required viscosupplementation were not excluded from the trial because it was considered a symptomatic treatment which formed part of the recovery protocol and could be used when necessary.
PRP is also cited as a possible cause of higher joint effusion and self-limited pain, when applied intra-articularly [12]. However, the treated group in this study did not have higher joint effusion rates than the control group.
Post-operative MRI scans were not taken because although they are important tests, they display moderate sensitivity for detecting and classifying chondral knee injuries [32]. Alternatives to improve sensitivity of MRI analysis of chondral injuries would be the use of T2 maping or Delayed Gadolinioum-Enhanced MRI of Cartilage (dGEMRIC) techniques. The first one, although not so high cost, takes a little more time and requires a specific protocol, absent in our imaging service. The second technique has the disadvantage of requiring more time for the exam and a high cost, in addition to being used more for research [33]. The use of some of these techniques could maybe detect differences between the repair tissues of the groups of our study.
Regarding the histological analysis, the limitation was because no patient required further surgery until the end of follow-up.
Despite our results, better healing due to PRP application cannot be proved. However, this study as well as other articles suggests that PRP can improve joint homeostasis and affect the synovial tissue. The classic study of Dye et al. [34] in 1998 proved that the most sensitive structure in the knee is the synovial tissue. If PRP is able to decrease synovial tissue inflammation, improve synovial fluid quality, and stimulate its production, it could explain the better functional outcomes of the patients in the treated group. Even if these patients do not have better repair tissue the suggested PRP effect can lead to an extended tissue response slowing down joint tissue degeneration.
As limitations of our study we can mention, in addition to the aforementioned (absence of post-operative images and histological evaluation), the small number of patients. The use of viscosupplementation can be a bias because it may change the final scores of the patients, but, as already mentioned, it is also a parameter of treatment success. The application of a single dose of PRP is debatable as there are studies that mention that the use of multiple doses may lead to better results [19, 25, 27]. We treated small to medium injuries, which usually have better prognosis, in a medium term follow-up. Longer term follow-up could show different results [26].
As future perspectives, PRP could be used in the same way in patient without associated injuries (like meniscal or ACL) to increase the strength of the results. The treatment could be used on larger injuries, also evaluated with more sensitive imaging exams and possible with histologic analysis. But the most promising future in our opinion would be the use of specific GFs according to the desired result.
Conclusion
Based on the subjective IKDC, KOOS, and Tegner forms scores, patients affected by small-to-medium symptomatic ICRS grades III knee chondral injuries may have a clinical benefit from an intra-articular injection of leukocyte-poor-PRP at the end of chondroplasty. This benefit has been observed up to a medium-term follow-up (2 years), and the results are independent of any other concomitant surgical procedures performed in association with the chondroplasty (i.e., partial meniscectomy or ACL reconstruction). This study, thus, further suggests the positive anabolic effect of the LP-PRP when associated to cartilage repair procedures.
References
Alford JW, Cole BJ (2005) Cartilage restoration. Part 1. Basic science historical perspective patient evaluation and treatment options. Am J Sports Med 33(2):295–306. https://doi.org/10.1177/0363546504273510
Bhosale AM, Richardson JB (2008) Articular cartilage: structure, injuries and review of management. Br Med Bull 87(1):77–95. https://doi.org/10.1093/bmb/ldn025
Filardo G, Andriolo L, Balboni F et al (2015) Cartilage failures. Systematic literature review critical survey analysis and definition. Knee Surg Sports Traumatol Arthrosc 23(12):3660–3669. https://doi.org/10.1007/s00167-014-3272-2
Mascarenhas R, Saltzman BM, Fortier LA, Cole BJ (2015) Role of platelet-rich plasma in articular cartilage injury and disease. J Knee Surg 28(1):3–10. https://doi.org/10.1055/s-0034-1384672
Mehta S, Watson JT (2008) Platelet rich concentrate: basic science and current clinical applications. J Orthop Trauma 22(6):433–438. https://doi.org/10.1097/BOT.0b013e31817e793f
Foster TE, Puskas BL, Mandelbaum BR et al (2009) Platelet-rich plasma: from basic science to clinical applications. Am J Sports Med 37(11):2259–2272. https://doi.org/10.1177/0363546509349921
Del Torto M, Enea D, Panfoli N et al (2015) Hamstrings anterior cruciate ligament reconstruction with and without platelet rich fibrin matrix. Knee Surg Sports Traumatol Arthrosc 23(12):3614–3622. https://doi.org/10.1007/s00167-014-3260-6
Lopez-Vidriero E, Goulding KA, Simon DA et al (2010) The use of platelet-rich plasma in arthroscopy and sports medicine: optimizing the healing environment. Arthroscopy 26(2):269–278. https://doi.org/10.1016/j.arthro.2009.11.015
Sheth U, Simunovic N, Klein G et al (2012) Efficacy of autologous platelet-rich plasma use for Orthopaedic indications: a meta-analysis. J Bone Joint Surg Am 94(4):298–307. https://doi.org/10.2106/JBJS.K.00154
Kreuz PC, Krüger JP, Metzlaff S et al (2015) Platelet-rich plasma preparation types show impact on chondrogenic differentiation. Migration and proliferation of human subchondral mesenchymal progenitor cells. Arthroscopy 31(10):1951–1961. https://doi.org/10.1016/j.arthro.2015.03.033
Cugat R, Cuscó X, Seijas R et al (2015) Biologic enhancement of cartilage repair: the role of platelet-rich plasma and other commercially available growth factors. Arthroscopy 31(4):777–783. https://doi.org/10.1016/j.arthro.2014.11.031
Filardo G, Kon E, Roffi A et al (2015) Platelet-rich plasma: why intra-articular? A systematic review of preclinical studies and clinical evidence on PRP for joint degeneration. Knee Surg Sports Traumatol Arthrosc 23(9):2459–2474. https://doi.org/10.1007/s00167-013-2743-1
Mishra A, Harmon K, Woodall J, Vieira A (2012) Sports medicine applications of platelet rich plasma. Curr Pharm Biotechnol 13(7):1185–1195. https://doi.org/10.2174/138920112800624283
Sundman EA, Cole BJ, Karas V et al (2014) The anti-inflammatory and matrix restorative mechanisms of platelet-rich plasma in osteoarthritis. Am J Sports Med 42(1):35–41. https://doi.org/10.1177/0363546513507766
Sakata R, McNary SM, Miyatake K et al (2015) Stimulation of the superficial zone protein and lubrication in the articular cartilage by human platelet-rich plasma. Am J Sports Med 43(6):1467–1473. https://doi.org/10.1177/0363546515575023
Filardo G, Di Matteo B, Di Martino A et al (2015) Platelet-rich plasma intra-articular knee injections show no superiority versus viscosupplementation: a randomized controlled trial. Am J Sports Med 43(7):1575–1582. https://doi.org/10.1177/0363546515582027
Kanchanatawan W, Arirachakaran A, Chaijenkij K et al (2016) Short-term outcomes of platelet-rich plasma injection for treatment of osteoarthritis of the knee. Knee Surg Sports Traumatol Arthrosc 24(5):1665–1677. https://doi.org/10.1007/s00167-015-3784-4
Filardo G, Previtali D, Napoli F et al (2020) PRP injections for the treatment of knee osteoarthritis: a meta-analysis of randomized controlled trials. Cartilage 1947603520931170. Advance online publication. https://doi.org/10.1177/1947603520931170
Tavassoli M, Janmohammadi N, Hosseini A et al (2019) Single- and double-dose of platelet-rich plasma versus hyaluronic acid for treatment of knee osteoarthritis: a randomized controlled trial. World J Orthop 10(9):310–326. https://doi.org/10.5312/wjo.v10.i9.310
Kon E, Mandelbaum B, Buda R et al (2011) Platelet-rich plasma intra-articular injection versus hyaluronic acid viscosupplementation as treatments for cartilage pathology: from early degeneration to osteoarthritis. Arthroscopy 27(11):1490–1501. https://doi.org/10.1016/j.arthro.2011.05.011
Delong JM, Russel RP, Mazzoca AD (2012) Platelet-rich plasma: the PAW classification system. Arthroscopy 28(7):998–1009. https://doi.org/10.1016/j.arthro.2012.04.148
Magalon J, Bausset O, Serratrice N et al (2014) Characterization and comparison of 5 platelet-rich plasma preparations in a single-donor model. Arthroscopy 30(5):629–638. https://doi.org/10.1016/j.arthro.2014.02.020
Oh JH, Kim W, Park KU, Roh YH (2015) Comparison of the cellular composition and cytokine-release kinetics of various platelet-rich plasma preparations. Am J Sports Med 43(12):3062–3070. https://doi.org/10.1177/0363546515608481
Harrison S, Vavken P, Kevy S et al (2011) Platelet activation by collagen provides sustained release of anabolic cytokines. Am J Sports Med 39(4):729–734. https://doi.org/10.1177/0363546511401576
Hart R, Safi A, Komzák M et al (2013) Platelet-rich plasma in patients with tibiofemoral cartilage degeneration. Arch Orthop Trauma Surg 133(9):1295–1301. https://doi.org/10.1007/s00402-013-1782-x
Di Martino A, Di Matteo B, Papio T et al (2019) Platelet-rich plasma versus hyaluronic acid injections for the treatment of knee osteoarthritis. Results at 5 years of a double-blind, Randomized Controlled Trial. Am J Sports Med 47(2):347–354. https://doi.org/10.1177/0363546518814532
Marmotti A, Rossi R, Castoldi F et al (2015) PRP and articular cartilage: a clinical update. Biomed Res Int 2015:542502. https://doi.org/10.1155/2015/542502
Siclari A, Mascaro G, Gentili C et al (2012) A cell-free scaffold-based cartilage repair provides improved function hyaline-like repair at one year. Clin Orthop Relat Res 470(3):910–919. https://doi.org/10.1007/s11999-011-2107-4
Yoshida R, Murray MM (2013) Peripheral blood mononuclear cells enhance the anabolic effects of platelet-rich plasma on anterior cruciate ligament fibroblasts. J Orthop Res 31(1):29–34. https://doi.org/10.1002/jor.22183
Belk JW, Kraeutler MJ, Houck DA et al (2020) Platelet-rich plasma versus hyaluronic acid for knee osteoarthritis. Am J Sports Med 363546520909397. Advance online publication. https://doi.org/10.1177/0363546520909397
Figueroa D, Figueroa F, Calvo R et al (2015) Platelet-rich plasma use in anterior cruciate ligament surgery: systematic review of the literature. Arthroscopy 31(5):981–988. https://doi.org/10.1016/j.arthro.2014.11.022
Danieli MV, Guerreiro JPF, Queiroz AO et al (2016) Diagnosis and classification of chondral knee injuries: comparison between magnetic resonance imaging and arthroscopy. Knee Surg Sports Traumatol Arthrosc 24(5):1627–1633. https://doi.org/10.1007/s00167-015-3622-8
Link TM, Neumann J, Li X (2017) Prestructural cartilage assessment using MRI. J Magn Reson Imaging 45(4):949–965. https://doi.org/10.1002/jmri.25554
Dye SF, Vaupel GL, Dye CC (1998) Conscious neurosensory mapping of the internal structures of the human knee without Intraarticular anesthesia. Am J Sports Med 26(6):773–777. https://doi.org/10.1177/03635465980260060601
Acknowledgements
We appreciate the work of the Research Support Department of Botucatu Medical School—UNESP, which helped with the statistical analyses. We also like to thank Ms. Tiemi Matsu for helping with statistical analyses. We are very grateful to Ms. Dora Horvath and FD English School with the work of improving the English of the text. Finally, we would like to show our gratitude to Ms. Madalena Sbizera who prepared the PRP for us.
Author information
Authors and Affiliations
Contributions
MVD carried out the conception and design, performed the surgeries and collected the data, has been involved in drafting the manuscript, and approved the final version.
JPFG contributed with the design, data collection, and revision of final version.
AOQ contributed with design and data collection.
HRP contributed with the conception and design and review the final version.
DCC contributed with the conception and design, has been involved in drafting the manuscript, and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was previously approved by the Ethics Research Committee of the Botucatu Medical School—UNESP (protocol number: CAAE: 0026.0.083.357-11) and by the Ethics Research Committee of the Irmandade Santa Casa de Londrina—BIO ISCAL (protocol number: 3995-2011).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Level of evidence: II, randomized controlled trial
Rights and permissions
About this article
Cite this article
Danieli, M.V., Guerreiro, J.P.F., Queiroz, A.O. et al. Leucocyte-poor-platelet-rich plasma intra-operative injection in chondral knee injuries improve patients outcomes. A prospective randomized trial. International Orthopaedics (SICOT) 45, 463–471 (2021). https://doi.org/10.1007/s00264-020-04830-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-020-04830-4