Abstract
Plants synthetize and accumulate a various class of organic compounds known as secondary metabolites (SMs). Although SMs do not play a crucial role as primary metabolites in plant growth and development, they are of high ecological significance. Humans use these compounds in the pharmaceutical industry. One of the results of mutual symbiotic association between plants and arbuscular mycorrhizal fungi (AMF) is the reprogramming of metabolic pathways and modulating the range and content of plant SMs such as phenolics, terpenoids, and alkaloids. Many of SMs act as signals for multiple interactions between plants and AMF, from the pre-symbiotic stage to the creation/formation of a functional symbiosis. This chapter briefly reviews the current research status in the field of SM changes under the influence of AMF. The plant association with AMF increases the production and accumulation of SMs directly through improving water and nutrient uptake and enhancing the photosynthetic capacity or indirectly by provoking the biosynthetic pathways of SMs through generation of signaling molecules and changes in the concentration of phytohormones. The extent to which AMF affect plant SMs depends on the plant and fungus species and environmental factors.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
5.1 Introduction
In the long path of evolution, plants have developed a wide range of organic compounds known as secondary metabolites (SMs). SMs have no direct role in plant fundamental processes such as photosynthesis, respiration, and transport of nutrients or in the biosynthetic pathways of carbohydrates, proteins, or lipids. Unlike primary metabolites, SMs have a limited distribution in the plant kingdom; in other words, specific SMs are often found only in one species or plants related to that species (Ghamarnia et al. 2022; Yousefi and Safari 2022). Lack or reduced level of SMs does not cause an immediate death of plants, but it causes disruptions in survival, fertility, and ecological distribution and resistance of plants to natural enemies (Agostini-Costa et al. 2012).
SMs in plants are structurally and chemically diverse groups of products, and despite lack of an essential role for plant growth and development, they are economically important for human well-being in various fields, including pharmaceuticals, nutrients, food additives, and agrochemicals (Chandran et al. 2020). Meanwhile, medicinal plants, which are rich reservoirs of the main active ingredients of numerous medicinal compounds, have attracted special attention (Zhao et al. 2022; Walid et al. 2021).
The most significant and prevalent mutualistic symbiosis of microorganisms with plants is formed between roots and arbuscular mycorrhizal fungi (AMF). Mycorrhiza promotes plant growth; increases the uptake of micro- and macronutrients (Grümberg et al. 2015; Battini et al. 2017; Amani Machiani et al. 2021); improves water relations (Eulenstein et al. 2016; Li et al. 2019; Ostadi et al. 2022), soil fertility, and yield quality (Amani Machiani et al. 2022); enhances tolerance to environmental stresses, such as drought, salinity, and heavy metals; and boosts resistance to pests and pathogens (Moradtalab et al. 2019; Al-Arjani et al. 2020). As the most prevalent symbiotic relationship in nature, AMF symbiosis offers enormous potential for resource utilization in agriculture, especially in low-input and sustainable agriculture, and would cut agricultural expenses and further minimize environmental degradation (Begum et al. 2019; Igiehon and Babalola 2017).
A wide range of environmental (abiotic and biotic stresses) conditions and internal (morphological, developmental, and genetic) factors affect the production and accumulation of SMs (Simões et al. 2016). Moreover, the mutual relationships between plants and soil microorganisms, like AMF, also change the quality and quantity of plant SMs (Kapoor et al. 2017). AMF-mediated increase in the synthesis and accumulation of SMs may be one of the mechanisms for the positive effects of mycorrhization in host plants, particularly those associated with higher tolerance to suboptimal conditions (Kaur and Suseela 2020).
Although the colonization of plant by the fungus takes place morphologically in the root, it affects the metabolism and physiology of the plant systemically (Schweiger and Müller 2015). Due to its dependence on plant carbon (carbohydrates and lipids), AMF are a powerful carbon sink in the roots. They allocate up to 30% of plant’s photosynthetic carbon in return for water and nutrient supply (Drigo et al. 2010; Van Der Heijden et al. 2015). Such interaction regulates photosynthesis and other primary metabolic pathways to maintain the balance of carbon in plant. As an inevitable consequence of change in primary metabolism, the profile of plant SMs will change (Kaschuk et al. 2009; Kogel et al. 2010).
A group of SMs act as mediators of AMF-plant interactions, from the pre-symbiotic step to establishing and maintaining a successful symbiotic association. For example, in response to secretion of signaling compounds from roots (strigolactones, flavonoids), AMF release lipocyto-oligosaccharide compounds called “Myc factors” that lead to the induction of symbiotic responses in the host plant (Kaur and Suseela 2020). In addition to the release of molecules necessary for identification and formation of a functional symbiosis, the AMF association leads to reprogramming of the secondary metabolic pathways in the host plants (Pozo et al. 2009; Jung et al. 2012; Song et al. 2013). The host genome is affected by fungal metabolites, and thus, the microbe-host interaction causes changes in the transcriptome, proteome, and ultimately the metabolome of the plant (Mhlongo et al. 2018). This chapter briefly introduces the impacts of AMF on secondary metabolism in host plants and provides insights to achieve higher performance of commercial plants through AMF colonization in sustainable cultivation systems.
5.2 Secondary Metabolism
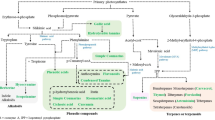
Unlike primary metabolism, secondary metabolism includes metabolic pathways and their related molecules that are not necessary for plant life (Yang et al. 2018). Products of SMs originate from primary metabolites or their intermediate compounds in the biosynthetic pathways (Piasecka et al. 2015). Methods such as mass spectrometry, gas chromatography, and liquid chromatography have made it possible to detect and identify a wide range of SMs in plants. By examining only 20–30% of all known plant species (Zhu et al. 2023), more than 100,000 different compounds have been isolated and identified (Wink 2008). Despite such a high diversity of SMs, there are limited numbers of corresponding basic biosynthetic pathways (Fig. 5.1). Substrates are usually derived from basic metabolic pathways such as the shikimic acid pathway, Krebs cycle, and glycolysis pathway (Wink 2008). According to the molecular structure and biological functions, three important classes of plant SMs are phenolics, terpenes or terpenoids, and alkaloids (Pandey et al. 2018). Terpenoids and phenolics are synthesized by almost all vascular plants, while alkaloids show a patchy occurrence pattern. So, specific alkaloid compounds are usually considered as indicators of specific taxonomic classes (Wink 2008). Another important group of SMs discussed in this chapter are saponins, a chemically heterogeneous group of triterpenoid and steroidal glycosides that are found in most medicinal plants and have a high molecular weight (Nasseri et al. 2020).
The main metabolic pathways leading to the synthesis of terpenes or terpenoids, phenolics, alkaloids, and saponins in plants (in gray, pink, green, and blue shaded sections, respectively). The brown color shows the category of regulatory compounds in medicinal plants colonized with AMF. Redrawn after Zhao et al. (2022)
Many SMs show positive effect on numerous aspects of plant growth and development including defense response (Isah 2019), innate immunity (Piasecka et al. 2015), and adaptations to environmental stress (Yang et al. 2018). Moreover, SMs act as signaling compounds for symbiosis between microorganisms and plants, including AMF (Guerrieri et al. 2019).
Apart from their functional significance in plants, the high commercial value of a large group of SMs has caused them to be used as special chemicals such as flavorings, perfumes, insecticides, dyes, and all kinds of anticancer, antidiabetic, antihypertensive, anti-AIDS, heart and blood vessel improvers, memory boosters, and antidepressants (Chandran et al. 2020). Since symbiosis of plants with AMF significantly modifies production and the pattern of accumulation of SMs (Fig. 5.2) (Tsiokanos et al. 2022), the extent to which the quality and quantity of SMs are affected by AMF association is one of the significant aspects in the production of medicinal and aromatic plants.
Potential pathways of plant metabolism reprogramming by AMF. Mycorrhization is associated with the reprogramming of primary metabolites and SMs in plants. Reprogramming of SMs stimulates plant defense and increases plant tolerance to abiotic and biotic stresses. Changes in the secondary metabolic pathways contribute to the self-regulation of AMF colonization by modulating the synthesis of signaling compounds. SA salicylic acid, JA jasmonic acid, SPs secreted proteins, LCOs lipochitooligosaccharides. Redrawn after Kaur and Suseela (2020)
5.2.1 Terpenoids
Terpenoids (or isoprenoids) are the most diverse class of plant metabolites, with more than 27,000 compounds (Sacchettini and Poulter 1997; Pandey et al. 2018). Terpenoids contain a ring or chain structure that is obtained from the fusion of isoprene units (C5). Based on the number of isoprene units used in their structure, terpenoids can be classified as hemiterpenes (C5), monoterpenes (C10, geraniol, linalool, limonene, etc.), sesquiterpenes (C15, abscisic acid, humulene, germacrene, bisabolene, etc.), diterpenes (C20, gibberellic acid), triterpenes (C30, ursolic acid, oleanolic acid, betulinic acid, etc.), tetraterpenes (C40, lupeol, diosgenin, stigmasterols, lanosterol, etc.), and polyterpenes (C > 40) (Pusztahelyi et al. 2015; Pandey et al. 2018). Terpenoids are synthesized and stored in trichomes and epidermal structures of leaf in plants (Covello et al. 2007). These compounds play an essential role in plant adaptation to adverse environmental conditions, and some of them (e.g., abscisic acid and gibberellic acid) regulate plant growth and stress responses (Pusztahelyi et al. 2015). Some sesquiterpenes (such as caryophyllene) and monoterpenes (such as linalool) are toxic to pathogens and herbivorous insects and can also act as a signaling molecule to attract natural enemies of insects (Kappers et al. 2005; Bakkali et al. 2008; Sharma et al. 2017). In addition, when plants are attacked by pathogens or fungus-eating animals, AMF stimulate the production of some terpenoids (such as catalpol) by the host plant. Catalpol is transferred to soil or external hyphae and acts as a signal or defense substances to protect neighboring plants or symbiotic fungi (Babikova et al. 2013; Duhamel et al. 2013). Despite some reports regarding neutral (Morone-Fortunato and Avato 2008; Nell et al. 2009) and even negative effects (Khaosaad et al. 2006), many studies have shown that the symbiotic relationship between AMF and plants can stimulate the synthesis of terpenoids and affect the composition and content of terpenoids (Karagiannidis et al. 2011; Lermen et al. 2015; Weisany et al. 2015). The studies conducted so far show that the increase in the production of precursors through the methylerythritol 4-phosphate (MEP) pathway (Fig. 5.2) is the reason for the increased concentration of terpenoids after inoculation with AMF (Kapoor et al. 2017). On the other hand, terpenoids, especially volatile mono- and sesquiterpenes with small molecular weight, play an important role in the symbiosis between plants and AMF (Duhamel et al. 2013; Babikova et al. 2013; Sharma et al. 2017). In addition, some terpenoids are bioactive and are the main active component of medicinal plants, such as the sesquiterpene artemisinin found in Artemisia annua (Mandal et al. 2015b).
5.2.1.1 Carotenoid Pathway
Carotenoids are members of the 40-carbon (tetraterpenoids) subfamily of terpenoids, which are made by connecting 8 isoprene units (Li et al. 2020). The important function of the carotenoid pathway is known in various plant processes including signaling, synthesis, and release of hormones, light protection, and photosynthesis (Shumskaya and Wurtzel 2013). Upregulation of the primary enzymes of this pathway, 1-deoxy-D-xylulose 5-phosphate reductoisomerase (DXR) and 1-deoxy-D-xylulose 5-phosphate synthase (DXS) (Fig. 5.3), after inoculation with AMF was associated with the accumulation of the diterpene stevioside and sesquiterpene artemisinin in the leaves of Stevia rebadiana and Artemisia annua, respectively (Mandal et al. 2015a, b).
Biosynthesis pathway of DMAPP/IPP promoted or induced by AMF. GGPP geranylgeranyl diphosphate, FPP farnesyl diphosphate, SQC sesquiterpene cyclase, GPP geranyl diphosphate, DMAPP dimethylallyl diphosphate, IPP isopentenyl diphosphate, DXR 1-deoxy-D-xylulose 5-phosphate reductoisomerase, MEP 2-C-methyl-D-erythritol 4-phosphate, DXS 1-deoxy-D-xylulose 5-phosphate synthase, DOXP 1-deoxy-D-xylulose 5-phosphate, HMG-CoA β-hydroxy β-methylglutaryl-CoA. Redrawn after Zhi-lin et al. (2007)
A number of molecules derived from the carotenoid pathway (e.g., strigolactones) are released by mycorrhizal roots during the pre-symbiotic step and after colonization (Al-Babili and Bouwmeester 2015). Strigolactones act as signaling compounds and regulate spore germination, hyphal elongation, and hyphopodium formation leading to the start of AMF-plant symbiosis (Al-Babili and Bouwmeester 2015). Significant induction of PDR1 transporter (an ABC transporter) which is involved in the transport of strigolactones is known in mycorrhizal plants (Kretzschmar et al. 2012). AMF also induce the synthesis and accumulation of some apocarotenoids, such as C14 acyclic polyene mycorradicin and C13 cyclohexenone derivatives (e.g., blumenin) (Fig. 5.3), which are important regulators of the establishment and maintaining of AM symbiosis in colonized roots (Schliemann et al. 2008; Fiorilli et al. 2019). The development of mycorrhization in Hordeum vulgare plants was associated with an increased accumulation of cyclohexenone derivatives (Peipp et al. 1997). Blumenols (glycosidic derivatives) are among the most common compounds derived from cyclohexenone that accumulate in the mycorrhizal plants and act as biomarkers of AM symbiosis (Schliemann et al. 2006; Wang et al. 2018). In addition, increased levels of apocarotenoids in AM roots may be important in protecting plants against ROS and pathogens, self-regulation of fungal colonization, and synthesis of signaling molecules (Strack and Fester 2006). There are some reports that the activation of carotenoid biosynthesis is specific to the plant-AMF relationship (Fester et al. 2005; Maier et al. 1997). Furthermore, mycorrhization reduced the emission of volatile sesquiterpenes by herbivory in Plantago lanceolata, indicating that terpenoids are also involved in indirectly priming of the host defense system (Fontana et al. 2009).
5.2.1.2 Essential Oils
Plant essential oils (Eos) are aromatic and volatile oil compounds. Complex mixtures of monoterpenes, sesquiterpenes, diterpenes, and compounds derived from phenylpropene are the main bioactive components of Eos (Nurzynska-Wierdak 2013). These compounds are important in the plant growth and development, resistance of plants against adverse environmental conditions, and attraction of pollinating insects. They are used as antiseptic, medicinal, and aromatic compounds in various industries (Chiej 1984; Chukwuka et al. 2011). In addition, studies show that different species of AMF cause different responses in the same plant; specific components of Eos are also affected by AMF in different aromatic plants (Kapoor et al. 2017). Rezaei-Chiyaneh et al. (2021) indicated that colonization by a combination of Rhizophagus irregularis and Funneliformis mosseae increased the main essential oil compounds such as trans-caryophyllene, borneol, p-cymene, thymol, and geranyl-acetate in Nigella sativa. The content of g-terpinene, p-cymene, and thymol in Eos of thyme (Thymus vulgaris) also increased after inoculation with Funneliformis mosseae (Amani Machiani et al. 2021). Similarly, Eo content was improved in Mentha requienii and Origanum onites plants inoculated with Glomus lamellosum and G. etunicatum, respectively (Karagiannidis et al. 2011). Moreover, after inoculation with Gigaspora rosea, the accumulation of linalool in Ocimum basilicum L. leaves declined, while the accumulation of eugenol and alpha-terpineol enhanced, suggesting that AMF association changes the essential oil composition of plant leaves (Copetta et al. 2006).
5.2.2 Phenolics
Phenolics or phenylpropanoids are a large class of SMs produced in plant tissues to protect against predators, parasites, pathogens, and UV radiation (Singla et al. 2019). They are synthesized by the pentose phosphate and shikimate pathways through the phenylpropanoid pathway (Fig. 5.1) (Michalak 2006; Zaynab et al. 2018). The five main groups of phenolics are phenolic acids, flavonoids, coumarins, stilbenes, and monolignols (Deng and Lu 2017). Most of the compounds have antioxidant activity, which, apart from their beneficial effects under stress conditions, give the plant valuable medicinal properties (Krishnaiah et al. 2011). In addition, they are important signaling compounds in microbe-plant relationships and plant development. AMF create changes in the quantity and quality of different SMs by reprogramming the phenylpropanoid pathway, to establish a functional symbiosis (Schweiger and Müller 2015). Two important groups of compounds in the phenylpropanoid pathway are phenolic acids and flavonoids.
5.2.2.1 Flavonoids
Flavonoids form the largest group of phenolics and include flavonols, flavones, isoflavonoids, and anthocyanins (Reyes-Carmona et al. 2005). Flavonoids have high antioxidant activity and contribute to the flavor and color of plants (Mierziak et al. 2014). There are many reports of beneficial effects of AMF on flavonoid concentrations. Inoculation with Rhizoglomus intraradices and Funneliformis mosseae increased the flavonoid levels in the Solanum nigrum L. roots (Muniz et al. 2021). In inoculated corn plants, the appropriate combination of fungus-plant species is essential in determining the result of symbiosis and achieving higher yield. Moreover, the plants inoculated with Dentiscutata heterogama and Claroideoglomus etunicatum had higher flavonoid levels compared to the plants inoculated with Acaulospora longula (Silva et al. 2019; Avio et al. 2020). Furthermore, the levels of phenols and flavonoids in the weed Aphis gossypii elevated after inoculation with Septoglomus constrictum and Claroideoglomus etunicatum, and this was accompanied by a decrease in its herbivory (Wu et al. 2023). These findings indicate the ecological implications of AMF in weed management.
In the AMF-plant relationship, changes in the flavonoid pattern are a function of the developmental stage of the symbiosis. These compounds are essential in initiating and limiting mycorrhization (Kaur and Suseela 2020). Different types of flavonoids affect the inoculation of different AMF species differently. Some flavonoids, including ononin and formononetin, are responsible for autoregulation of AM symbiosis, by limiting the fungal colonization after reaching to a threshold level (Catford et al. 2006). In the same way, some isoflavonoids, such as malonylononin, ononin, and daidzein, are involved in the later stages of mycorrhizal development (Schliemann et al. 2008). AMF changed flavonoid accumulation in growing roots of Manihot esculenta Crantz 6 weeks after inoculation with Rhizophagus irregularis (Bag et al. 2022). Comparison of well-colonized roots of Medicago sativa and Medicago truncatula, and incompletely colonized roots of Medicago sativa, showed that the level of flavonoid 4′,7-dihydroxyflavone increased only in the roots with well-developed colonization (Harrison and Dixon 1993). In the same way, the levels of medicarpin, which were increased during initial colonization, were greatly reduced during the advanced stages of colonization. However, the abundance of medicarpin in incompletely colonized roots did not decrease, which means that growth-dependent changes in the flavonoid pattern are necessary for the formation and regulation of symbiotic association (Harrison and Dixon 1993). Moreover, the increase in the concentration of some types of flavonoids, such as coumestrol, daidzein, medicarpin malonyl glucoside, and formononetin malonyl glucoside, is related to the improved resistance of mycorrhizal plants to stressful conditions (Harrison and Dixon 1993).
5.2.2.2 Phenolic Acids
Phenolic acids are considered as important mediators of plant-microorganism interactions (Mandal et al. 2010). Higher level of these compounds in herbal products is an indication of their higher quality (Ghasemzadeh and Ghasemzadeh 2011). There are several reports of improved levels of phenolic acids by inoculation of AMF. AMF colonization enhanced the abundance of phenolic acids in Arachis hypogaea roots and leaves (Devi and Reddy 2002). The abundance of phenolic acid derivatives also changed in Cichorium intybus (Rozpądek et al. 2014) and Hordeum vulgare (Devi and Reddy 2002) plants inoculated with AMF. Hydroxycinnamate amides and caffeic acid derivatives were reported in mycorrhizal roots of chicory, and accumulation of hydroxycinnamate amides in barley mycorrhizal roots. Total phenol content in Eclipta prostrata L. plants increased 8 weeks after inoculation with a combination of AMF species (Acaulospora lacunosa, Funneliformis mosseae, and Gigaspora albida) (Duc et al. 2021). Colonization in Passiflora alata Curtis plants with Gigaspora albida and Acaulospora longula increased the total phenol by 94% and 111%, respectively (Muniz et al. 2021). In addition, resistance of Phoenix dactylifera against bayoud disease is improved through its colonization with various AMF species. This improvement was related to the increased enzymatic activities of polyphenol oxidases and peroxidases (Jaiti et al. 2007). Therefore, higher concentration of phenolic acids in AMF-inoculated plants implies the improvement of plant defense system against pathogens. In addition, it has been found that artichoke plants inoculated with Funneliformis mosseae IMA1 and Claroideoglomus claroideum 22W3 had higher phenolic compounds and antioxidant activities than control plants, while Glomus sp., Rhizophagus irregularis, and other varieties of Funneliformis mosseae had no effect on the plant antioxidant activity (Mandal et al. 2010). It has been reported that while ferulic acid level in Solanum lycopersicum roots decreased with AMF, the accumulation of caffeic acids increased. Such different response to mycorrhizal association depending on fungus and plant species suggests certain degrees of specificity in the AMF-plant interaction (López-Ráez et al. 2010). Similarly, Aliferis et al. (2015) and Maier et al. (2000) reported increased concentrations of other phenylpropanoid pathway compounds including coumarins and their hydroxyl derivatives, in mycorrhizal Salix purpurea L. leaves. These compounds have antioxidant and antimicrobial properties (Maier et al. 2000; Aliferis et al. 2015). Inoculation with Acaulospora longula also increased the tannin level in Libidibia ferrea fruits (Santos et al. 2020).
5.2.3 Alkaloids
Alkaloids are alkaline-like substances that contain one or more nitrogen atoms in the heterocyclic ring (Zhu et al. 2023). In plants, these metabolites are derived from amino acids, aromatic compounds, as well as terpenes (Herbert 2001). Based on their amino acid precursors, they are classified into pyridine, quinolizidine, indole-quinoline, benzyl-isoquinoline, pyrrolizidine, pyrrolidine, and tropane groups (Song et al. 2014; Dey et al. 2020). So far, 600 bioactive alkaloid compounds have been identified out of a total 20,000 alkaloid compounds discovered (mainly from plants). Duo to toxic properties, alkaloids play an important role in strengthening the plant’s defense system against biotic stresses (Yeshi et al. 2022). Some types of alkaloids have medicinal properties with a wide application in the pharmaceutical industry. In this case, we can mention morphine, cocaine, nicotine, colchicine, quinine, strychnine, and caffeine (Herbert 2001). Several studies reported the elevated concentrations of alkaloids after the inoculation of plants with AMF. For example, colonization with different species of AMF increased the concentration of alkaloid colchicine in different organs (aerial organ, tuber, and seed) of Gloriosa superba L. plant (Pandey et al. 2014). In addition, inoculation with Gigaspora rosea was associated with an increase in the content of alkaloid with therapeutic properties, trigonelline in Prosopis laevigata. Accumulation of alkaloids trigonelline (Rojas-Andrade et al. 2003), castanospermine (Abu-Zeyad et al. 1999), and camptothecin (Wei and Wang 1989) was also enhanced in plants colonized with different AMF species.
5.2.4 Saponins
An important class of SMs produced in plants are saponins. Their structure is characterized by a carbohydrate part (an oligo or monosaccharide chain) attached to a steroid or triterpene (Hussein and El-Anssary 2019). The various biological functions of saponins explain their wide application in pharmaceutical, cosmetic, and food industries (Isah 2019). An increase in saponin concentration has been reported in mycorrhizal plants. For example, inoculation of Glycyrrhiza glabra plants with Claroideoglomus etunicatum (Johny et al. 2021) and Funneliformis mosseae and/or Diversispora epigaea (Liu et al. 2007) was associated with the accumulation of triterpenoid saponin glycyrrhizic acid. Similarly, an accumulation of saponins was observed in the aerial parts of Anchusa officinalis L. (Cartabia et al. 2021) and Passiflora alata (Muniz et al. 2021) plants inoculated with Rhizophagus irregularis and Acaulospora longula, respectively.
5.3 Mechanisms of AMF Effects on SM Production in Plants
AM colonization alters the plant SM synthesis pattern in two different ways, nutritional (direct) and non-nutritional (indirect) effects (Fig. 5.4). In the nutritional mode of action, AMF improve water and nutrient uptake (phosphorus, nitrogen, copper, manganese, zinc) (Kapoor et al. 2017) and photosynthetic capacity and growth, which subsequently increase the production of precursor compounds required for the synthesis of SMs (Zhao et al. 2022). Some reports attribute the benefits of AMF on SMs to improved nutrient uptake, especially phosphorus. For example, inoculated and non-inoculated Foeniculum vulgare plants, with similar phosphorus levels, showed the similar essential oil content (Kapoor et al. 2017). Improved accumulation of glycyrrhizic acid in Glycyrrhiza glabra was also attributed to improved phosphorus uptake in these plants by AMF (Xie et al. 2018a, b). It seems that AMF cause C:N:P (carbon:nitrogen:phosphorus) stoichiometric changes in tissues by increasing phosphorus and nitrogen availability and regulating carbon distribution (Saia et al. 2014; Zhao et al. 2015). Based on the carbon-nutrient balance hypothesis, the synthesis of SMs is regulated by the ratio between carbon and nutrients. Low C:N ratios in plants lead to the synthesis of carbon-based SMs, while high C:N ratios increase the synthesis of nitrogen-based SMs (Gershenzon 1994; Hamilton et al. 2001). A recent study supported this hypothesis. Xie et al. (2018a, b) indicated that AMF-mediated increases in the C:N ratio and decreases in N:P ratio in Glycyrrhiza glabra plants were associated with an accumulation of glycyrrhizic acid in the roots. On the other hand, the improved water and nutrient uptake and higher photosynthetic capacity lead to increased division and development in the secretory ducts, glandular trichomes, and Eo channels (Amani Machiani et al. 2021). Moreover, some micronutrients act as cofactors for different enzymes. For example, manganese is an essential cofactor for the function of phytoene synthase, a key enzyme involved in the biosynthesis of carotenoids (tetraterpenoid) (Fraser et al. 2000). Two independent studies have reported increased manganese uptake in Stevia rebaudiana plants colonized by R. fasciculatus (Mandal et al. 2015b) and R. intraradices (Mandal et al. 2013). In addition, improved phosphorus nutrition in AMF plants increases metabolic precursors such as ATP, NADPH, phosphoenol-pyruvate, erythrose-4-phosphate, pyruvate, glyceraldehyde phosphate, and acetyl coenzyme A, which are necessary for the synthesis of SMs (Kapoor et al. 2017).
Proposed mechanisms affecting the production of SMs in AMF-colonized plants. Redrawn after Amani Machiani et al. (2022)
Although the AM symbiotic relationship increases the availability of nutrients, especially phosphorus, leading to higher levels of primary metabolites, the levels of phenols, flavonoids, and tannins were not affected by the phosphorus nutritional status in cebil (Anadenanthera colubrina) plants (Pedone-Bonfim et al. 2013). Similarly, the accumulation of anthocyanins and flavonoids in Medicago truncatula leaves was observed in mycorrhizal plants in the absence of phosphorus uptake benefits (Adolfsson et al. 2017). These findings emphasize the reprogramming of SMs in the phenylpropanoid pathway by AMF colonization, regardless of its nutritional function (Kaur and Suseela 2020). Furthermore, it has been reported that inoculation with F. mosseae significantly increased Eo accumulation in two genotypes of Origanum sp., while the same effect was not observed when the non-mycorrhizal plants are supplied with phosphorus (Khaosaad et al. 2006). The authors concluded that AMF-mediated increase in Eo concentration in Origanum sp. plants was not due to nutritional benefits (Khaosaad et al. 2006).
In non-nutritional (indirect) effects, AMF affect the biosynthetic pathways of SMs through modifications in the phytohormone levels such as cytokinins, gibberellic acid, and jasmonic acid (Erb and Kliebenstein 2020). For example, Liao et al. (2018) reported that gibberellic acid and jasmonic acid increased the levels of terpenoids by stimulating sesquiterpenoid biosynthetic gene expression and glandular trichome formation. Also, jasmonic acid stimulated two monoterpenes synthesis in Lycopersicon esculentum (Van Schie et al. 2007), and sesquiterpenoid biosynthesis gene in Foeniculum vulgare (Maes et al. 2011). In addition, the signaling molecules generated upon host plant-AMF interaction influence the levels of SMs in plant tissues. For example, the symbiosis between Trifolium repens and F. mosseae increased the levels of hydrogen peroxide, nitric oxide, and salicylic acid, which lead to enhanced enzyme activities in the biosynthetic pathway of phenolics (Zhang et al. 2013).
The two modes of action described above are not independent of each other. The expression of key genes involved in the biosynthetic pathways of SMs can also be affected by plant nutritional status (Lillo et al. 2008; Lazzara et al. 2017). For example, the gene expression pattern of various enzymes such as phenylalanine ammonia-lyase, flavonol synthase, chalcone isomerase, chalcone synthase, and cinnamate-4-hydroxylase, from the biosynthetic pathway of flavonoids, has been shown to be affected by the level of phosphorus and nitrogen supply (Lillo et al. 2008).
5.4 The Importance of AMF in Medicinal Plants
Medicinal plants are valuable and perpetual reservoirs of SMs, and in fact, they are the main effective sources of numerous medicinal compounds (Chandran et al. 2020). Due to the lipophilic properties of active oils in several medicinal plants, these substances are capable to entirely disrupt the membrane structure of bacteria, particularly Gram-negative type (Nazzaro et al. 2013). Some other SMs have immune response properties and antibacterial, antioxidant, antiviral, anticancer, and many other capabilities (Chandran et al. 2020).
The widespread approach of the world to the use of herbal medicines has increased the attention of most countries in the world to the identification and therapeutic use of medicinal plants and a return to nature. Chemical drugs as pure compounds, and despite their effectiveness, have several adverse effects, while the bioactive compounds found in medicinal plants are combined with other substances and thus have a state of biological balance. According to the statistics of WHO,Footnote 1 more than 80% of the people around the world, especially in developing and poor societies, meet their main medical needs from medicinal plants (Chandran et al. 2020; Zhao et al. 2022).
The growth of human and animal populations particularly in the developing countries, along with the constant increase in food, fodder, and fuel needs, put a huge pressure on the stability of the elements of the agricultural ecosystem. Since the fertility of the land is decreasing, farmers are turning towards the destruction of natural resource lands to obtain more crops. Therefore, if the current trend of population growth continues, in addition to the reduction of forests and rangelands, the production capacity of agricultural lands will also decrease (Shah et al. 2021). Thus, the utilization of phytomicrobiome components is suggested as one of the efficient and necessary solutions for the sustainable growth of crop production (Shah et al. 2021).
5.5 Production of Medicinal Plants
Despite the growing market demand, medicinal plants naturally have a very low performance in producing SMs (Thakur et al. 2019). So, several techniques have been developed aimed at improving the performance of these valuable metabolites in medicinal plants, such as Agrobacterium-mediated transformation, metabolic engineering, and mass propagation (Oksman-Caldentey and Inzé 2004; Liu et al. 2005). However, each of these methods is associated with limitations such as low practicality, high cost, and low effectiveness (Oksman-Caldentey and Inzé 2004; El-Sayed and Verpoorte 2007; Kapoor et al. 2017). On the other hand, plant productivity in intensive farming methods has a very close connection with the heavy use of chemicals including inorganic herbicides, pesticides, and fertilizers. Such widespread use of chemicals, in addition to the risks for human health and environmental issues, reduce the quality and quantity of bioactive molecules in medicinal herbs (Amani Machiani et al. 2022). So, there is a necessity for using environmentally friendly strategies to improve the production of SMs (Li et al. 2023). Symbiosis of plants with AMF can change the pattern of production and accumulation of SMs (Fig. 5.2), depending on the fungus and plant species and the growth environment (Tsiokanos et al. 2022). Therefore, as many studies have confirmed (Chandran et al. 2020; Zhao et al. 2022), inoculation of plants with AMF offers a promising tool and a simpler alternative to overcome these limitations (Kapoor et al. 2017).
5.6 Conclusion
It is well established that AMF symbiosis reprograms plant secondary metabolism. AMF affect the production and accumulation of SMs directly by improving the uptake of water and nutrients, enhancing the photosynthetic capacity, and increasing the biomass, or indirectly by stimulating the biosynthetic pathways of SMs through the production of signaling compounds and changes in the phytohormone levels. Some of these compounds show a characteristic pattern depending on the developmental stage of colonization and act as signals between two partners over the pre-symbiosis stage to the formation of a functional symbiosis and in the maintenance of AM symbiosis. In addition, modifications in secondary metabolism following plant mycorrhization increase the concentration of plant antioxidant compounds. As a result, plant’s ability to tolerate stressful conditions and the quality of plant products are improved.
A wide group of SMs have nutritional and medicinal value for human. Wide application and increasing demand for these valuable biological reservoirs make it necessary to invest in research and development to optimize their production. The use of AMF technique is considered as a potential tool and a promising opportunity to improve the quality and yield of plant active ingredients. AMF technique is a potential tool in crop management and plays a prominent role in promoting new environmentally friendly products in low-input and sustainable agricultural systems. In general, three research priorities are suggested to advance future studies: (1) more in-depth studies for understanding the mechanisms through which AMF association changes the SMs quantities and qualities, especially in terms of the species-specific nature of AMF-plant symbiosis; (2) the effects of environmental factors and developmental stages of the symbiosis, which have been greatly underestimated in the current studies, and (3) development of strategies for the implementation of mycorrhiza in the production of aromatic and medicinal compounds for industrial and commercial purposes.
Notes
- 1.
The World Health Organization.
References
Abu-Zeyad R, Khan AG, Khoo C (1999) Occurrence of arbuscular mycorrhiza in Castanospermum australe A. Cunn. & C. Fraser and effects on growth and production of castanospermine. Mycorrhiza 9(2):111–117
Adolfsson L, Nziengui H, Abreu IN, Šimura J, Beebo A, Herdean A, Spetea C (2017) Enhanced secondary-and hormone metabolism in leaves of arbuscular mycorrhizal Medicago truncatula. Plant Physiol 175(1):392–411
Agostini-Costa TDS, Vieira RF, Bizzo HR, Silveira D, Gimenes MA (2012) Secondary metabolites. In: Chromatography and its applications, vol 1. IntechOpen, London, pp 131–164
Al-Arjani ABF, Hashem A, Abd Allah EF (2020) Arbuscular mycorrhizal fungi modulates dynamics tolerance expression to mitigate drought stress in Ephedra foliata Boiss. Saudi J Biol Sci 27(1):380–394
Al-Babili S, Bouwmeester HJ (2015) Strigolactones, a novel carotenoid-derived plant hormone. Annu Rev Plant Biol 66:161–186
Aliferis KA, Chamoun R, Jabaji S (2015) Metabolic responses of willow (Salix purpurea L.) leaves to mycorrhization as revealed by mass spectrometry and 1H NMR spectroscopy metabolite profiling. Front Plant Sci 6:344
Amani Machiani M, Javanmard A, Morshedloo MR, Aghaee A, Maggi F (2021) Funneliformis mosseae inoculation under water deficit stress improves the yield and phytochemical characteristics of thyme in intercropping with soybean. Sci Rep 11(1):15279
Amani Machiani M, Javanmard A, Habibi Machiani R, Sadeghpour A (2022) Arbuscular mycorrhizal fungi and changes in primary and secondary metabolites. Plants 11(17):2183
Avio L, Maggini R, Ujvári G, Incrocci L, Giovannetti M, Turrini A (2020) Phenolics content and antioxidant activity in the leaves of two artichoke cultivars are differentially affected by six mycorrhizal symbionts. Sci Hortic 264:109153
Babikova Z, Gilbert L, Bruce TJ, Birkett M, Caulfield JC, Woodcock C, Johnson D (2013) Underground signals carried through common mycelial networks warn neighbouring plants of aphid attack. Ecol Lett 16(7):835–843
Bag S, Mondal A, Majumder A, Mondal SK, Banik A (2022) Flavonoid mediated selective cross-talk between plants and beneficial soil microbiome. Phytochem Rev 21(5):1739–1760
Bakkali F, Averbeck S, Averbeck D, Idaomar M (2008) Biological effects of essential oils–a review. Food Chem Toxicol 46(2):446–475
Battini F, Grønlund M, Agnolucci M, Giovannetti M, Jakobsen I (2017) Facilitation of phosphorus uptake in maize plants by mycorrhizosphere bacteria. Sci Rep 7(1):4686
Begum N, Qin C, Ahanger MA, Raza S, Khan MI, Ashraf M, Zhang L (2019) Role of arbuscular mycorrhizal fungi in plant growth regulation: implications in abiotic stress tolerance. Front Plant Sci 10:1068
Cartabia A, Tsiokanos E, Tsafantakis N, Lalaymia I, Termentzi A, Miguel M, Declerck S (2021) The arbuscular mycorrhizal fungus Rhizophagus irregularis MUCL 41833 modulates metabolites production of Anchusa officinalis L. under semi-hydroponic cultivation. Front Plant Sci 12:724352
Catford JG, Staehelin C, Larose G, Piché Y, Vierheilig H (2006) Systemically suppressed isoflavonoids and their stimulating effects on nodulation and mycorrhization in alfalfa split-root systems. Plant Soil 285:257–266
Chandran H, Meena M, Barupal T, Sharma K (2020) Plant tissue culture as a perpetual source for production of industrially important bioactive compounds. Biotechnol Rep 26:e00450
Chiej R (1984) The Macdonald encyclopedia of medicinal plants. Macdonald & Co (Publishers) Ltd., London
Chukwuka KS, Ikheloa JO, Okonko IO, Moody JO, Mankinde TA (2011) The antimicrobial activities of some medicinal plants on Escherichia coli as an agent of diarrhea in livestock. Adv Appl Sci Res 2(4):37–48
Copetta A, Lingua G, Berta G (2006) Effects of three AM fungi on growth, distribution of glandular hairs, and essential oil production in Ocimum basilicum L. var. Genovese. Mycorrhiza 16:485–494
Covello PS, Teoh KH, Polichuk DR, Reed DW, Nowak G (2007) Functional genomics and the biosynthesis of artemisinin. Phytochemistry 68(14):1864–1871
Deng Y, Lu S (2017) Biosynthesis and regulation of phenylpropanoids in plants. Crit Rev Plant Sci 36(4):257–290
Devi MC, Reddy MN (2002) Phenolic acid metabolism of groundnut (Arachis hypogaea L.) plants inoculated with VAM fungus and rhizobium. Plant Growth Regul 37:151–156
Dey P, Kundu A, Kumar A, Gupta M, Lee BM, Bhakta T, Kim HS (2020) Analysis of alkaloids (indole alkaloids, isoquinoline alkaloids, tropane alkaloids). In: Recent advances in natural products analysis. Elsevier, London, pp 505–567
Drigo B, Pijl AS, Duyts H, Kielak AM, Gamper HA, Houtekamer MJ, Kowalchuk GA (2010) Shifting carbon flow from roots into associated microbial communities in response to elevated atmospheric CO2. Proc Natl Acad Sci 107(24):10938–10942
Duc NH, Vo AT, Haddidi I, Daood H, Posta K (2021) Arbuscular mycorrhizal fungi improve tolerance of the medicinal plant Eclipta prostrata (L.) and induce major changes in polyphenol profiles under salt stresses. Front Plant Sci 11:612299
Duhamel M, Pel R, Ooms A, Bücking H, Jansa J, Ellers J, Kiers ET (2013) Do fungivores trigger the transfer of protective metabolites from host plants to arbuscular mycorrhizal hyphae? Ecology 94(9):2019–2029
El-Sayed M, Verpoorte R (2007) Catharanthus terpenoid indole alkaloids: biosynthesis and regulation. Phytochem Rev 6:277–305
Erb M, Kliebenstein DJ (2020) Plant secondary metabolites as defenses, regulators, and primary metabolites: the blurred functional trichotomy. Plant Physiol 184(1):39–52
Eulenstein F, Tauschke M, Behrendt A, Monk J, Schindler U, Lana MA, Monk S (2016) The application of mycorrhizal fungi and organic fertilisers in horticultural potting soils to improve water use efficiency of crops. Horticulturae 3(1):8
Fester T, Wray V, Nimtz M, Strack D (2005) Is stimulation of carotenoid biosynthesis in arbuscular mycorrhizal roots a general phenomenon? Phytochemistry 66(15):1781–1786
Fiorilli V, Wang JY, Bonfante P, Lanfranco L, Al-Babili S (2019) Apocarotenoids: old and new mediators of the arbuscular mycorrhizal symbiosis. Front Plant Sci 10:1186
Fontana A, Reichelt M, Hempel S, Gershenzon J, Unsicker SB (2009) The effects of arbuscular mycorrhizal fungi on direct and indirect defense metabolites of Plantago lanceolata L. J Chem Ecol 35:833–843
Fraser PD, Schuch W, Bramley PM (2000) Phytoene synthase from tomato (Lycopersicon esculentum) chloroplasts–partial purification and biochemical properties. Planta 211:361–369
Gershenzon J (1994) Metabolic costs of terpenoid accumulation in higher plants. J Chem Ecol 20:1281–1328
Ghamarnia H, Palash M, Dousti B (2022) Camelina zoning for different climate conditions in Kurdistan Province. Agrotechniques Ind Crops 2(1):49–56
Ghasemzadeh A, Ghasemzadeh N (2011) Flavonoids and phenolic acids: role and biochemical activity in plants and human. J Med Plant Res 5(31):6697–6703
Grümberg BC, Urcelay C, Shroeder MA, Vargas-Gil S, Luna CM (2015) The role of inoculum identity in drought stress mitigation by arbuscular mycorrhizal fungi in soybean. Biol Fertil Soils 51:1–10
Guerrieri A, Dong L, Bouwmeester HJ (2019) Role and exploitation of underground chemical signaling in plants. Pest Manag Sci 75(9):2455–2463
Hamilton JG, Zangerl AR, DeLucia EH, Berenbaum MR (2001) The carbon–nutrient balance hypothesis: its rise and fall. Ecol Lett 4(1):86–95
Harrison MJ, Dixon RA (1993) Isoflavonoid accumulation and expression of defense gene transcripts during the establishment of vesicular-arbuscular mycorrhizal associations in roots of Medicago truncatula. Mol Plant-Microbe Interact 6(5):643–654
Herbert RB (2001) The biosynthesis of plant alkaloids and nitrogenous microbial metabolites. Nat Prod Rep 18(1):50–65
Hussein RA, El-Anssary AA (2019) Plants secondary metabolites: the key drivers of the pharmacological actions of medicinal plants. Herb Med 1(3):76139
Igiehon NO, Babalola OO (2017) Biofertilizers and sustainable agriculture: exploring arbuscular mycorrhizal fungi. Appl Microbiol Biotechnol 101:4871–4881
Isah T (2019) Stress and defense responses in plant secondary metabolites production. Biol Res 52:39
Jaiti F, Meddich A, El Hadrami I (2007) Effectiveness of arbuscular mycorrhizal fungi in the protection of date palm (Phoenix dactylifera L.) against Bayoud disease. Physiol Mol Plant Pathol 71(4-6):166–173
Johny L, Cahill DM, Adholeya A (2021) AMF enhance secondary metabolite production in ashwagandha, licorice, and marigold in a fungi-host specific manner. Rhizosphere 17:100314
Jung SC, Martinez-Medina A, Lopez-Raez JA, Pozo MJ (2012) Mycorrhiza-induced resistance and priming of plant defenses. J Chem Ecol 38:651–664
Kapoor R, Anand G, Gupta P, Mandal S (2017) Insight into the mechanisms of enhanced production of valuable terpenoids by arbuscular mycorrhiza. Phytochem Rev 16:677–692
Kappers IF, Aharoni A, Van Herpen TW, Luckerhoff LL, Dicke M, Bouwmeester HJ (2005) Genetic engineering of terpenoid metabolism attracts bodyguards to Arabidopsis. Science 309(5743):2070–2072
Karagiannidis N, Thomidis T, Lazari D, Panou-Filotheou E, Karagiannidou C (2011) Effect of three Greek arbuscular mycorrhizal fungi in improving the growth, nutrient concentration, and production of essential oils of oregano and mint plants. Sci Hortic 129(2):329–334
Kaschuk G, Kuyper TW, Leffelaar PA, Hungria M, Giller KE (2009) Are the rates of photosynthesis stimulated by the carbon sink strength of rhizobial and arbuscular mycorrhizal symbioses? Soil Biol Biochem 41(6):1233–1244
Kaur S, Suseela V (2020) Unraveling arbuscular mycorrhiza-induced changes in plant primary and secondary metabolome. Metabolites 10(8):335
Khaosaad T, Vierheilig H, Nell M, Zitterl-Eglseer K, Novak J (2006) Arbuscular mycorrhiza alter the concentration of essential oils in oregano (Origanum sp., Lamiaceae). Mycorrhiza 16:443–446
Kogel KH, Voll LM, Schäfer P, Jansen C, Wu Y, Langen G, Sonnewald U (2010) Transcriptome and metabolome profiling of field-grown transgenic barley lack induced differences but show cultivar-specific variances. Proc Natl Acad Sci 107(14):6198–6203
Kretzschmar T, Kohlen W, Sasse J, Borghi L, Schlegel M, Bachelier JB, Martinoia E (2012) A petunia ABC protein controls strigolactone-dependent symbiotic signalling and branching. Nature 483(7389):341–344
Krishnaiah D, Sarbatly R, Nithyanandam R (2011) A review of the antioxidant potential of medicinal plant species. Food Bioprod Process 89(3):217–233
Lazzara S, Militello M, Carrubba A, Napoli E, Saia S (2017) Arbuscular mycorrhizal fungi altered the hypericin, pseudohypericin, and hyperforin content in flowers of Hypericum perforatum grown under contrasting P availability in a highly organic substrate. Mycorrhiza 27(4):345–354
Lermen C, Morelli F, Gazim ZC, da Silva AP, Goncalves JE, Dragunski DC, Alberton O (2015) Essential oil content and chemical composition of Cymbopogon citratus inoculated with arbuscular mycorrhizal fungi under different levels of lead. Ind Crop Prod 76:734–738
Li J, Meng B, Chai H, Yang X, Song W, Li S, Sun W (2019) Arbuscular mycorrhizal fungi alleviate drought stress in C3 (Leymus chinensis) and C4 (Hemarthria altissima) grasses via altering antioxidant enzyme activities and photosynthesis. Front Plant Sci 10:499
Li C, Swofford CA, Sinskey AJ (2020) Modular engineering for microbial production of carotenoids. Metabol Eng Commun 10:e00118
Li Z, Xiong K, Wen W, Li L, Xu D (2023) Functional endophytes regulating plant secondary metabolism: current status, prospects and applications. Int J Mol Sci 24(2):1153
Liao D, Wang S, Cui M, Liu J, Chen A, Xu G (2018) Phytohormones regulate the development of arbuscular mycorrhizal symbiosis. Int J Mol Sci 19(10):3146
Lillo C, Lea US, Ruoff P (2008) Nutrient depletion as a key factor for manipulating gene expression and product formation in different branches of the flavonoid pathway. Plant Cell Environ 31(5):587–601
Liu Y, Wang H, Ye HC, Li GF (2005) Advances in the plant isoprenoid biosynthesis pathway and its metabolic engineering. J Integr Plant Biol 47(7):769–782
Liu J, Wu L, Wei S, Xiao X, Su C, Jiang P, Yu Z (2007) Effects of arbuscular mycorrhizal fungi on the growth, nutrient uptake and glycyrrhizin production of licorice (Glycyrrhiza uralensis Fisch). Plant Growth Regul 52:29–39
López-Ráez JA, Flors V, García JM, Pozo MJ (2010) AM symbiosis alters phenolic acid content in tomato roots. Plant Signal Behav 5(9):1138–1140
Maes L, Van Nieuwerburgh FC, Zhang Y, Reed DW, Pollier J, Vande Casteele SR, Goossens A (2011) Dissection of the phytohormonal regulation of trichome formation and biosynthesis of the antimalarial compound artemisinin in Artemisia annua plants. New Phytol 189(1):176–189
Maier W, Hammer K, Dammann U, Schulz B, Strack D (1997) Accumulation of sesquiterpenoid cyclohexenone derivatives induced by an arbuscular mycorrhizal fungus in members of the Poaceae. Planta 202:36–42
Maier W, Schmidt J, Nimtz M, Wray V, Strack D (2000) Secondary products in mycorrhizal roots of tobacco and tomato. Phytochemistry 54(5):473–479
Mandal SM, Chakraborty D, Dey S (2010) Phenolic acids act as signaling molecules in plant-microbe symbioses. Plant Signal Behav 5(4):359–368
Mandal S, Evelin H, Giri B, Singh VP, Kapoor R (2013) Arbuscular mycorrhiza enhances the production of stevioside and rebaudioside-A in Stevia rebaudiana via nutritional and non-nutritional mechanisms. Appl Soil Ecol 72:187–194
Mandal S, Upadhyay S, Singh VP, Kapoor R (2015a) Enhanced production of steviol glycosides in mycorrhizal plants: a concerted effect of arbuscular mycorrhizal symbiosis on transcription of biosynthetic genes. Plant Physiol Biochem 89:100–106
Mandal S, Upadhyay S, Wajid S, Ram M, Jain DC, Singh VP, Kapoor R (2015b) Arbuscular mycorrhiza increase artemisinin accumulation in Artemisia annua by higher expression of key biosynthesis genes via enhanced jasmonic acid levels. Mycorrhiza 25:345–357
Mhlongo MI, Piater LA, Madala NE, Labuschagne N, Dubery IA (2018) The chemistry of plant–microbe interactions in the rhizosphere and the potential for metabolomics to reveal signaling related to defense priming and induced systemic resistance. Front Plant Sci 9:112
Michalak A (2006) Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress. Pol J Environ Stud 15(4):523–530
Mierziak J, Kostyn K, Kulma A (2014) Flavonoids as important molecules of plant interactions with the environment. Molecules 19(10):16240–16265
Moradtalab N, Hajiboland R, Aliasgharzad N, Hartmann TE, Neumann G (2019) Silicon and the association with an arbuscular-mycorrhizal fungus (Rhizophagus clarus) mitigate the adverse effects of drought stress on strawberry. Agronomy 9(1):41
Morone-Fortunato I, Avato P (2008) Plant development and synthesis of essential oils in micropropagated and mycorrhiza inoculated plants of Origanum vulgare L. ssp. hirtum (Link) Ietswaart. Plant Cell Tissue Organ Cult 93:139–149
Muniz BC, Falcão EL, de Paula Monteiro R, dos Santos EL, Bastos Filho CJA, da Silva FSB (2021) Acaulospora longula Spain & NC Schenck: a low-cost bioinsumption to optimize phenolics and saponins production in Passiflora alata Curtis. Ind Crop Prod 167:113498
Nasseri MA, Keshtkar H, Kazemnejadi M, Allahresani A (2020) Phytochemical properties and antioxidant activity of Echinops persicus plant extract: green synthesis of carbon quantum dots from the plant extract. SN Appl Sci 2:1–12
Nazzaro F, Fratianni F, De Martino L, Coppola R, De Feo V (2013) Effect of essential oils on pathogenic bacteria. Pharmaceuticals 6(12):1451–1474
Nell M, Voetsch M, Vierheilig H, Steinkellner S, Zitterl-Eglseer K, Franz C, Novak J (2009) Effect of phosphorus uptake on growth and secondary metabolites of garden sage (Salvia officinalis L.). J Sci Food Agric 89(6):1090–1096
Nurzynska-Wierdak R (2013) Does mineral fertilization modify essential oil content and chemical composition in medicinal plants? Acta Sci Pol Hortorum Cultus 12:5
Oksman-Caldentey KM, Inzé D (2004) Plant cell factories in the post-genomic era: new ways to produce designer secondary metabolites. Trends Plant Sci 9(9):433–440
Ostadi A, Javanmard A, Amani Machiani M, Sadeghpour A, Maggi F, Nouraein M, Lorenzo JM (2022) Co-application of TiO2 nanoparticles and arbuscular mycorrhizal fungi improves essential oil quantity and quality of sage (Salvia officinalis L.) in drought stress conditions. Plants 11(13):1659
Pandey DK, Malik T, Dey A, Singh J, Banik RM (2014) Improved growth and colchicine concentration in Gloriosa superba on mycorrhizal inoculation supplemented with phosphorus-fertilizer. Afr J Tradit Complement Altern Med 11(2):439–446
Pandey DK, Kaur P, Dey A (2018) Arbuscular mycorrhizal fungi: effects on secondary metabolite production in medicinal plants. In: Fungi and their role in sustainable development: current perspectives. Springer, Cham, pp 507–538
Pedone-Bonfim MV, Lins MA, Coelho IR, Santana AS, Silva FS, Maia LC (2013) Mycorrhizal technology and phosphorus in the production of primary and secondary metabolites in cebil (Anadenanthera colubrina (Vell.) Brenan) seedlings. J Sci Food Agric 93(6):1479–1484
Peipp H, Maier W, Schmidt J, Wray V, Strack D (1997) Arbuscular mycorrhizal fungus-induced changes in the accumulation of secondary compounds in barley roots. Phytochemistry 44(4):581–587
Piasecka A, Jedrzejczak-Rey N, Bednarek P (2015) Secondary metabolites in plant innate immunity: conserved function of divergent chemicals. New Phytol 206(3):948–964
Pozo MJ, Verhage A, García-Andrade J, García JM, Azcón-Aguilar C (2009) Priming plant defence against pathogens by arbuscular mycorrhizal fungi. In: Mycorrhizas-functional processes and ecological impact. Springer, Cham, pp 123–135
Pusztahelyi T, Holb IJ, Pócsi I (2015) Secondary metabolites in fungus-plant interactions. Front Plant Sci 6:573
Reyes-Carmona J, Yousef GG, Martínez-Peniche RA, Lila MA (2005) Antioxidant capacity of fruit extracts of blackberry (Rubus sp.) produced in different climatic regions. J Food Sci 70(7):s497–s503
Rezaei-Chiyaneh E, Battaglia ML, Sadeghpour A, Shokrani F, Nasab ADM, Raza MA, von Cossel M (2021) Optimizing intercropping systems of black cumin (Nigella sativa L.) and fenugreek (Trigonella foenum-graecum L.) through inoculation with bacteria and mycorrhizal fungi. Adv Sustainable Syst 5(9):2000269
Rojas-Andrade R, Cerda-García-Rojas C, Frías-Hernández J, Dendooven L, Olalde-Portugal V, Ramos-Valdivia A (2003) Changes in the concentration of trigonelline in a semi-arid leguminous plant (Prosopis laevigata) induced by an arbuscular mycorrhizal fungus during the presymbiotic phase. Mycorrhiza 13(1):49–52
Rozpądek P, Wężowicz K, Stojakowska A, Malarz J, Surówka E, Anielska T, Turnau K (2014) Mycorrhizal fungi modulate phytochemical production and antioxidant activity of Cichorium intybus L.(Asteraceae) under metal toxicity. Chemosphere 112:217–224
Sacchettini JC, Poulter CD (1997) Creating isoprenoid diversity. Science 277(5333):1788–1789
Saia S, Benítez E, García-Garrido JM, Settanni L, Amato G, Giambalvo D (2014) The effect of arbuscular mycorrhizal fungi on total plant nitrogen uptake and nitrogen recovery from soil organic material. J Agric Sci 152(3):370–378
Santos EL, Silva WAV, Ferreira MRA, Soares LA, Sampaio EVSB, Silva FA, Silva FSB (2020) Acaulospora longula increases the content of phenolic compounds and antioxidant activity in fruits of Libidibia ferrea. Open Microbiol J 14(1):132–139
Schliemann W, Schmidt J, Nimtz M, Wray V, Fester T, Strack D (2006) Accumulation of apocarotenoids in mycorrhizal roots of Ornithogalum umbellatum. Phytochemistry 67(12):1196–1205
Schliemann W, Ammer C, Strack D (2008) Metabolite profiling of mycorrhizal roots of Medicago truncatula. Phytochemistry 69(1):112–146
Schweiger R, Müller C (2015) Leaf metabolome in arbuscular mycorrhizal symbiosis. Curr Opin Plant Biol 26:120–126
Shah A, Nazari M, Antar M, Msimbira LA, Naamala J, Lyu D, Smith DL (2021) PGPR in agriculture: a sustainable approach to increasing climate change resilience. Front Sustainable Food Syst 5:667546
Sharma E, Anand G, Kapoor R (2017) Terpenoids in plant and arbuscular mycorrhiza-reinforced defence against herbivorous insects. Ann Bot 119(5):791–801
Shumskaya M, Wurtzel ET (2013) The carotenoid biosynthetic pathway: thinking in all dimensions. Plant Sci 208:58–63
Silva FA, Maia LC, Silva FS (2019) Arbuscular mycorrhizal fungi as biotechnology alternative to increase concentrate of secondary metabolites in Zea mays L. Rev Bras Bot 42:189–193
Simões CMO, Schenkel EP, de Mello JCP, Mentz LA, Petrovick PR (2016) Farmacognosia: do produto natural ao medicamento. Artmed Editora, Porto Alegre
Singla RK, Dubey AK, Garg A, Sharma RK, Fiorino M, Ameen SM, Al-Hiary M (2019) Natural polyphenols: chemical classification, definition of classes, subcategories, and structures. J AOAC Int 102(5):1397–1400
Song YY, Ye M, Li CY, Wang RL, Wei XC, Luo SM, Zeng RS (2013) Priming of anti-herbivore defense in tomato by arbuscular mycorrhizal fungus and involvement of the jasmonate pathway. J Chem Ecol 39:1036–1044
Song MC, Kim EJ, Kim E, Rathwell K, Nam SJ, Yoon YJ (2014) Microbial biosynthesis of medicinally important plant secondary metabolites. Nat Prod Rep 31(11):1497–1509
Strack D, Fester T (2006) Isoprenoid metabolism and plastid reorganization in arbuscular mycorrhizal roots. New Phytol 172(1):22–34
Thakur M, Bhattacharya S, Khosla PK, Puri S (2019) Improving production of plant secondary metabolites through biotic and abiotic elicitation. J Appl Res Med Aromat Plants 12:1–12
Tsiokanos E, Cartabia A, Tsafantakis N, Lalaymia I, Termentzi A, Miguel M, Fokialakis N (2022) The metabolic profile of Anchusa officinalis L. differs according to its associated arbuscular mycorrhizal fungi. Metabolites 12(7):573
Van Der Heijden MG, Martin FM, Selosse MA, Sanders IR (2015) Mycorrhizal ecology and evolution: the past, the present, and the future. New Phytol 205(4):1406–1423
Van Schie CC, Haring MA, Schuurink RC (2007) Tomato linalool synthase is induced in trichomes by jasmonic acid. Plant Mol Biol 64:251–263
Walid, N., Kassam, R., & Ashfaq, M. A. (2021). Interaction between mycorrhizal and medicinal plants towards enhancement of secondary metabolites. Int. J. Chem. Stud, 9(2), 140–152.
Wang M, Schäfer M, Li D, Halitschke R, Dong C, McGale E, Baldwin IT (2018) Blumenols as shoot markers of root symbiosis with arbuscular mycorrhizal fungi. elife 7:e37093
Wei GT, Wang HG (1989) Effects of VA mycorrhizal fungi on growth, nutrient uptake and effective compounds in Chinese medicinal herb Datura stramonium L. Sci Agric Sin 25(5):56–61
Weisany W, Raei Y, Pertot I (2015) Changes in the essential oil yield and composition of dill (Anethum graveolens L.) as response to arbuscular mycorrhiza colonization and cropping system. Ind Crop Prod 77:295–306
Wink M (2008) Plant secondary metabolism: diversity, function and its evolution. Nat Prod Commun 3(8):1934578X0800300801
Wu QS, Silva FS, Hijri M, Kapoor R (2023) Arbuscular mycorrhiza-mediated augmentation of plant secondary metabolite production. Front Plant Sci 14:1150900
Xie W, Hao Z, Yu M, Wu Z, Zhao A, Li J, Chen B (2018a) Improved phosphorus nutrition by arbuscular mycorrhizal symbiosis as a key factor facilitating glycyrrhizin and liquiritin accumulation in Glycyrrhiza uralensis. Plant Soil 439:243–257
Xie W, Hao Z, Zhou X, Jiang X, Xu L, Wu S, Chen B (2018b) Arbuscular mycorrhiza facilitates the accumulation of glycyrrhizin and liquiritin in Glycyrrhiza uralensis under drought stress. Mycorrhiza 28:285–300
Yang L, Wen KS, Ruan X, Zhao YX, Wei F, Wang Q (2018) Response of plant secondary metabolites to environmental factors. Molecules 23(4):762
Yeshi K, Crayn D, Ritmejerytė E, Wangchuk P (2022) Plant secondary metabolites produced in response to abiotic stresses has potential application in pharmaceutical product development. Molecules 27(1):313
Yousefi B, Safari H (2022) Evaluation of essential oil, morphological traits and crop yield in Satureja mutica Fisch. & CA Mey. in rainfed cultivation under different row spacing. Agrotechniques Ind Crops 2(4):187–197
Zaynab M, Fatima M, Abbas S, Sharif Y, Umair M, Zafar MH, Bahadar K (2018) Role of secondary metabolites in plant defense against pathogens. Microb Pathog 124:198–202
Zhang RQ, Zhu HH, Zhao HQ, Yao Q (2013) Arbuscular mycorrhizal fungal inoculation increases phenolic synthesis in clover roots via hydrogen peroxide, salicylic acid and nitric oxide signaling pathways. J Plant Physiol 170(1):74–79
Zhao R, Guo W, Bi N, Guo J, Wang L, Zhao J, Zhang J (2015) Arbuscular mycorrhizal fungi affect the growth, nutrient uptake and water status of maize (Zea mays L.) grown in two types of coal mine spoils under drought stress. Appl Soil Ecol 88:41–49
Zhao Y, Cartabia A, Lalaymia I, Declerck S (2022) Arbuscular mycorrhizal fungi and production of secondary metabolites in medicinal plants. Mycorrhiza 32(3-4):221–256
Zhi-lin Y, Chuan-chao D, Lian-qing C (2007) Regulation and accumulation of secondary metabolites in plant-fungus symbiotic system. Afr J Biotechnol 6(11):57436
Zhu F, Wen W, Cheng Y, Alseekh S, Fernie AR (2023) Integrating multiomics data accelerates elucidation of plant primary and secondary metabolic pathways. Abiotech 2023:1–10
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Rahmat, S., Soheilikhah, Z. (2024). Arbuscular Mycorrhizal Fungi and Plant Secondary Metabolism. In: Ahammed, G.J., Hajiboland, R. (eds) Arbuscular Mycorrhizal Fungi and Higher Plants. Springer, Singapore. https://doi.org/10.1007/978-981-99-8220-2_5
Download citation
DOI: https://doi.org/10.1007/978-981-99-8220-2_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-99-8219-6
Online ISBN: 978-981-99-8220-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)